Chaperone-Like Activity of HSPB5: The Effects of Quaternary Structure Dynamics and Crowding
Abstract
1. Introduction
2. Results
2.1. Protein Aggregation. Theoretical Analysis of the Initial Parts of the Kinetic Curves
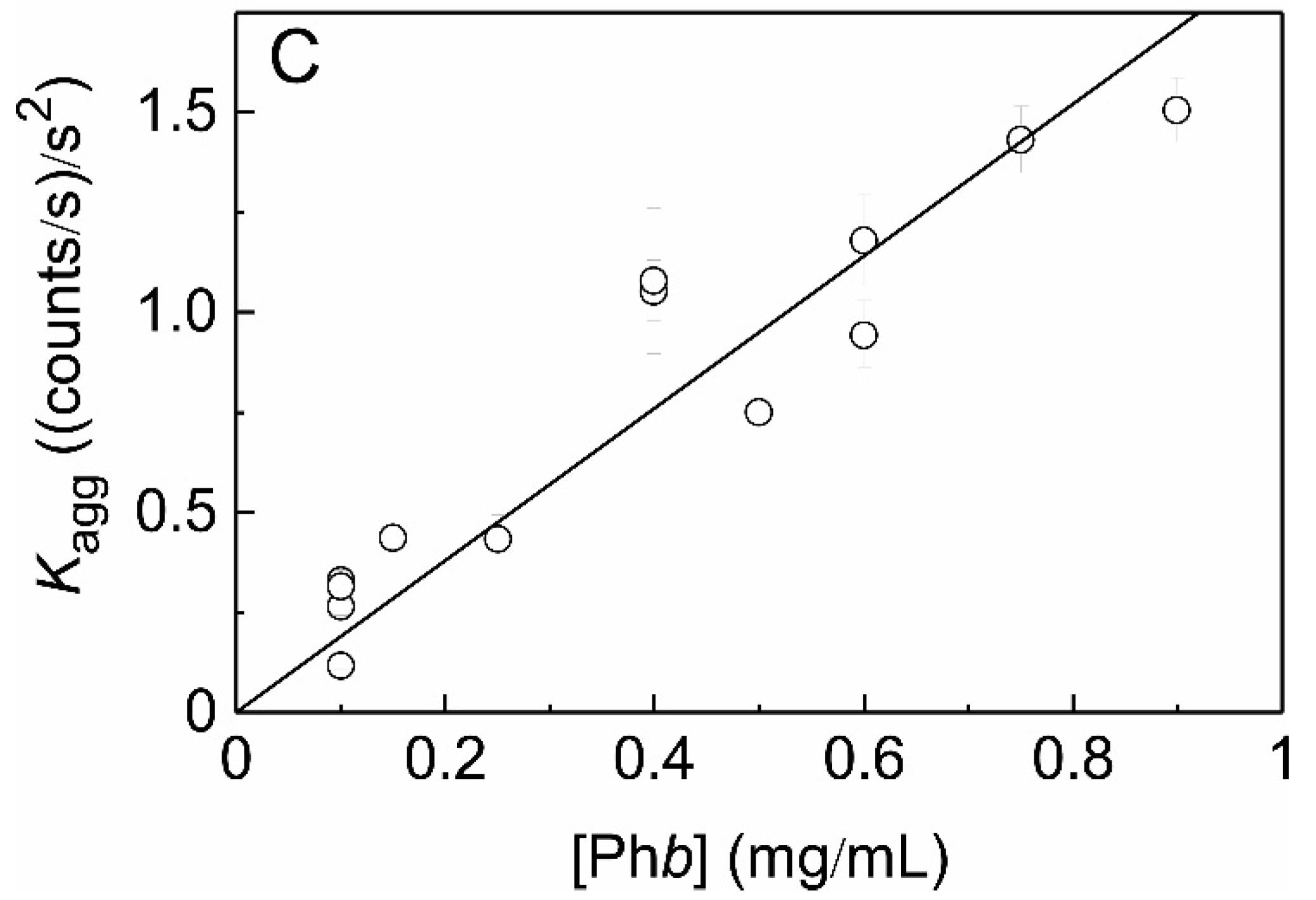
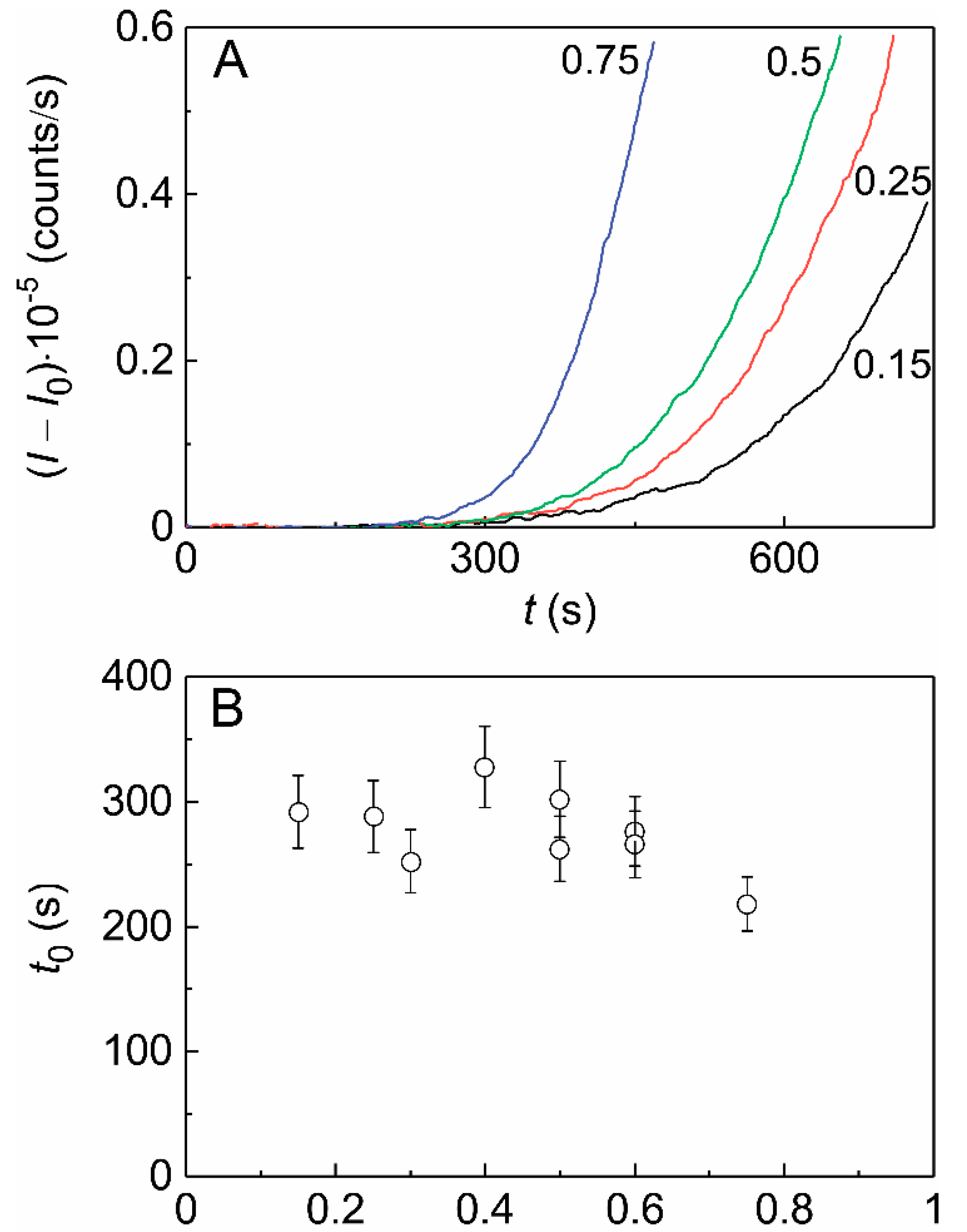
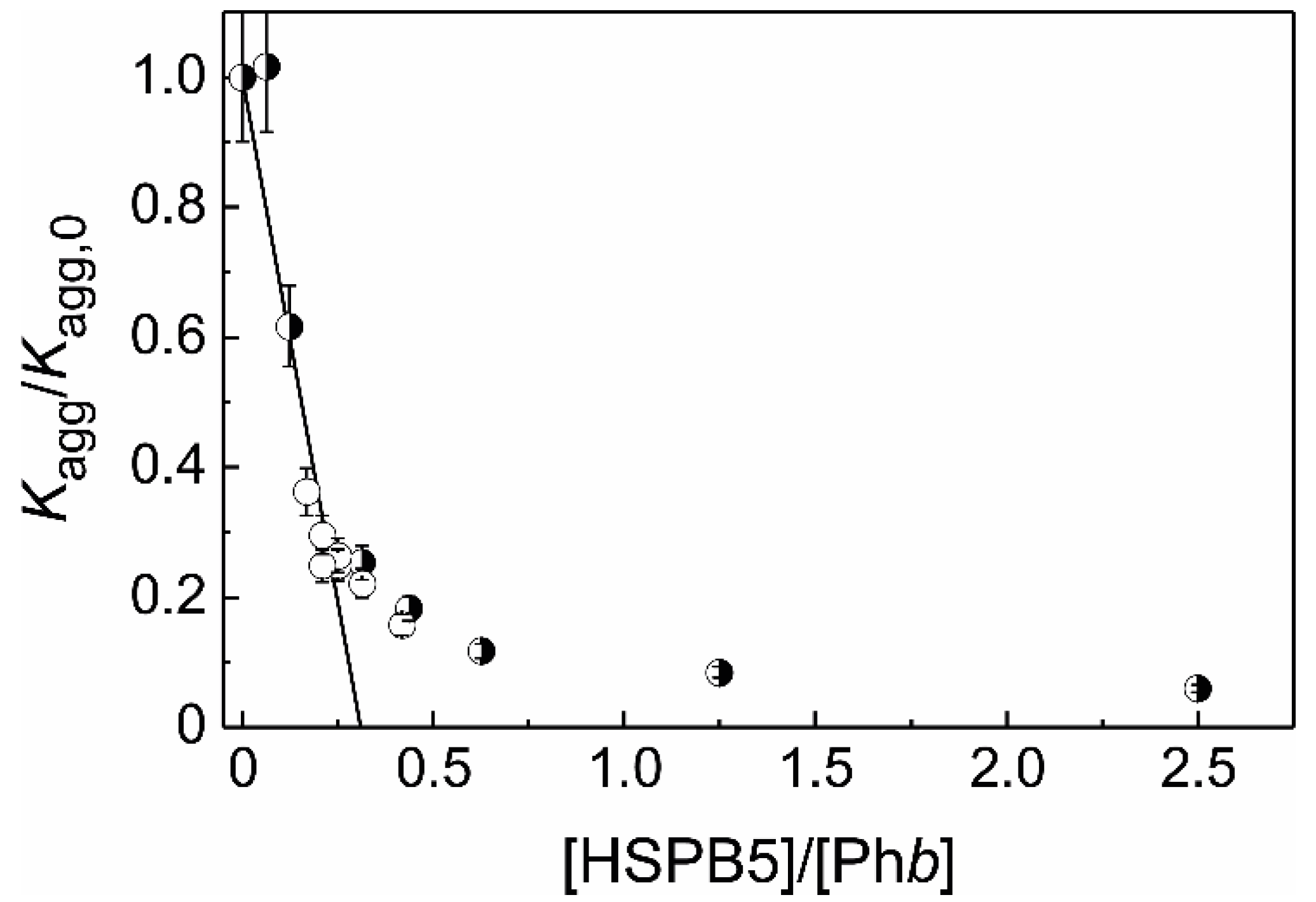
2.2. The Effect of HSPB5 on Thermal Aggregation of Phb
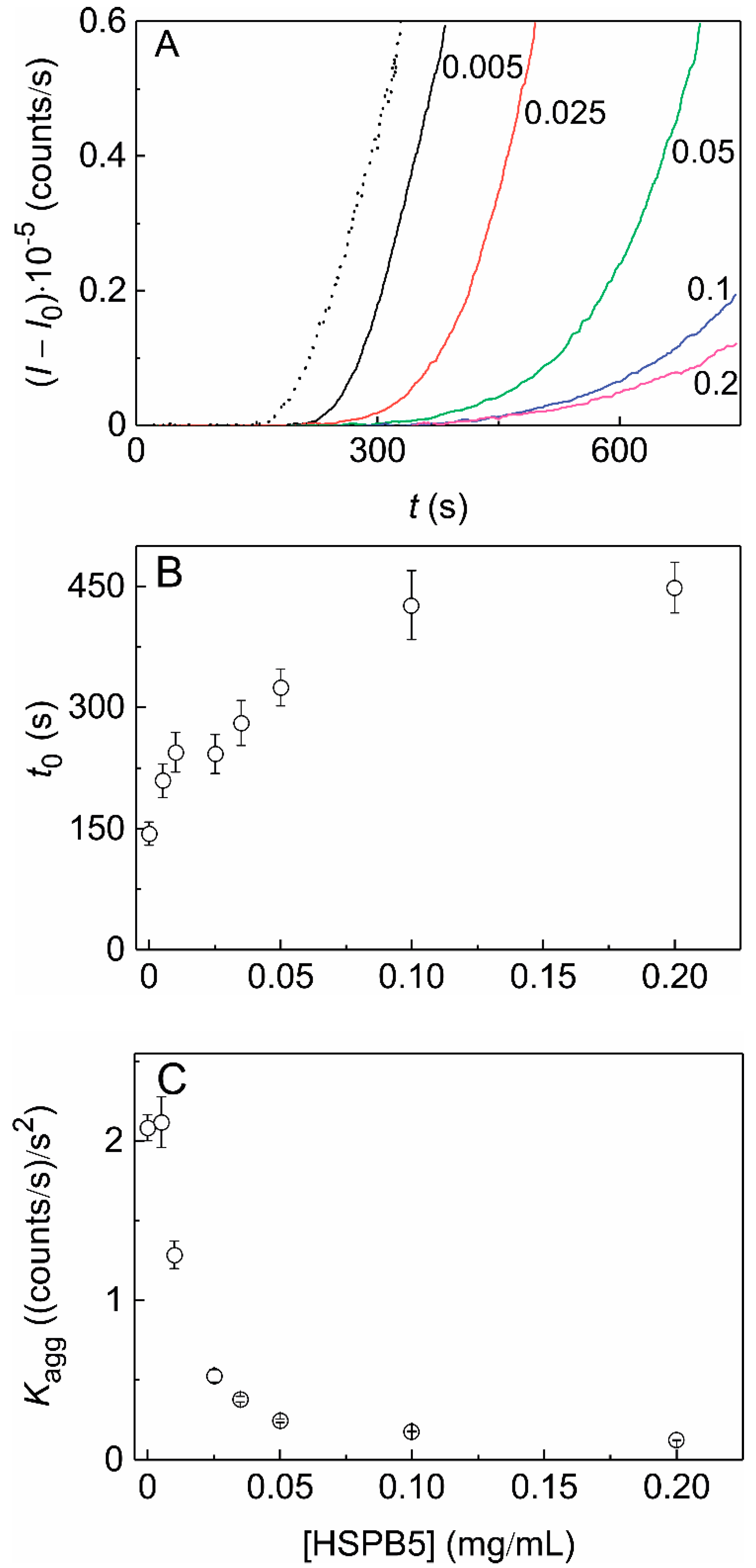
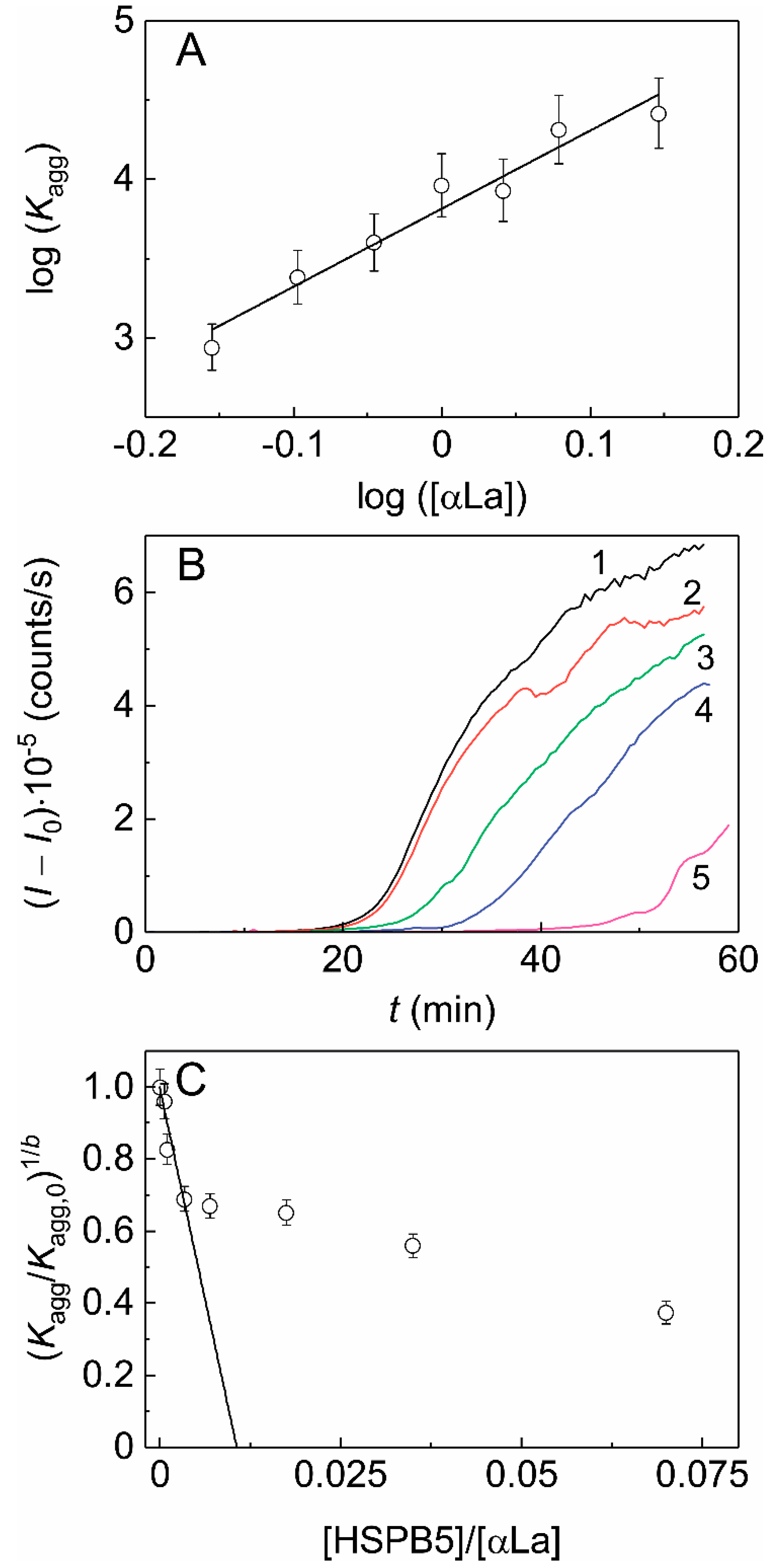
2.3. The Effect of HSPB5 on DTT-Induced Aggregation of α-Lactalbumin (αLa)
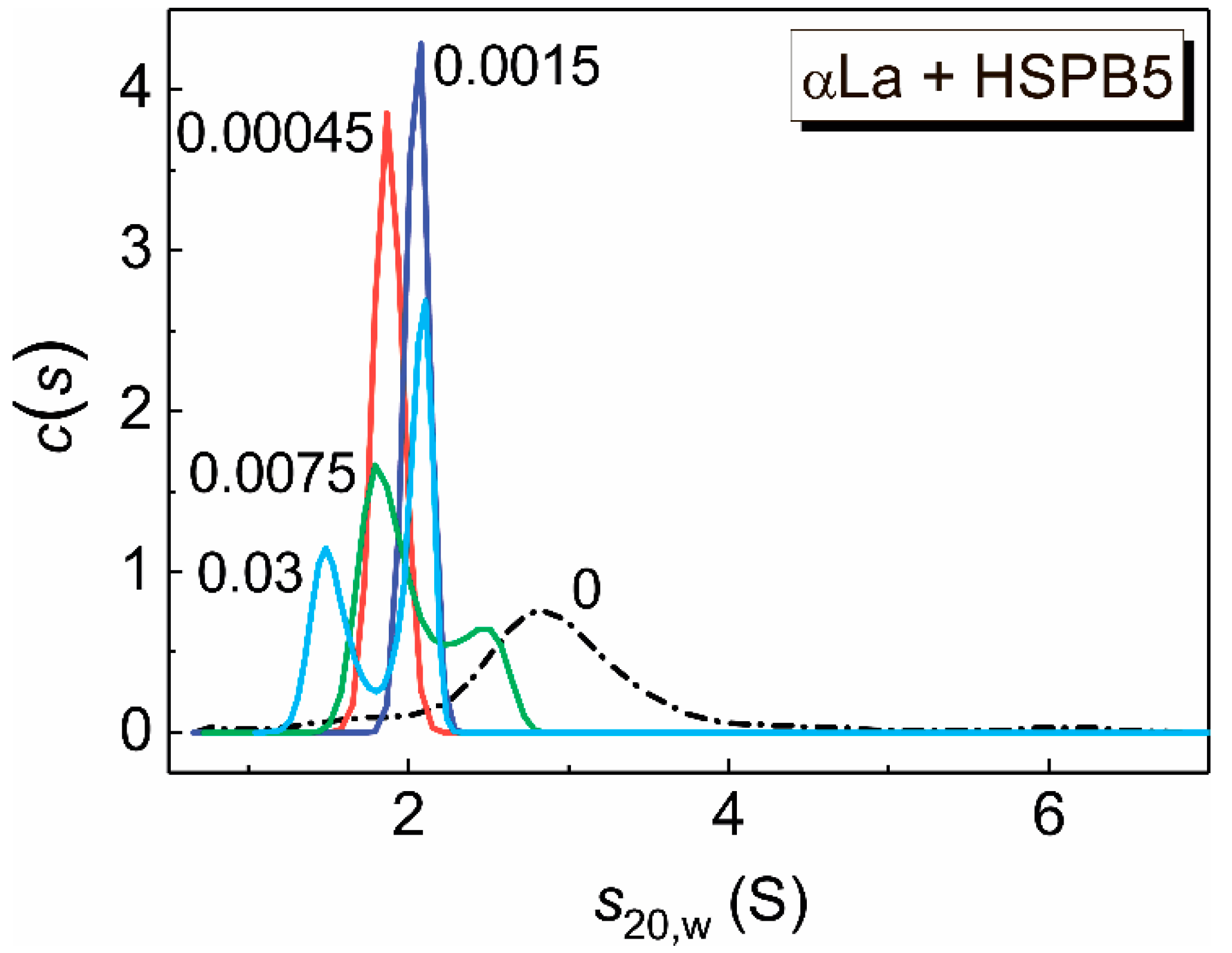
2.4. Sedimentation Velocity (SV) Analysis of HSPB5 and α-Lactalbumin at 37 °C
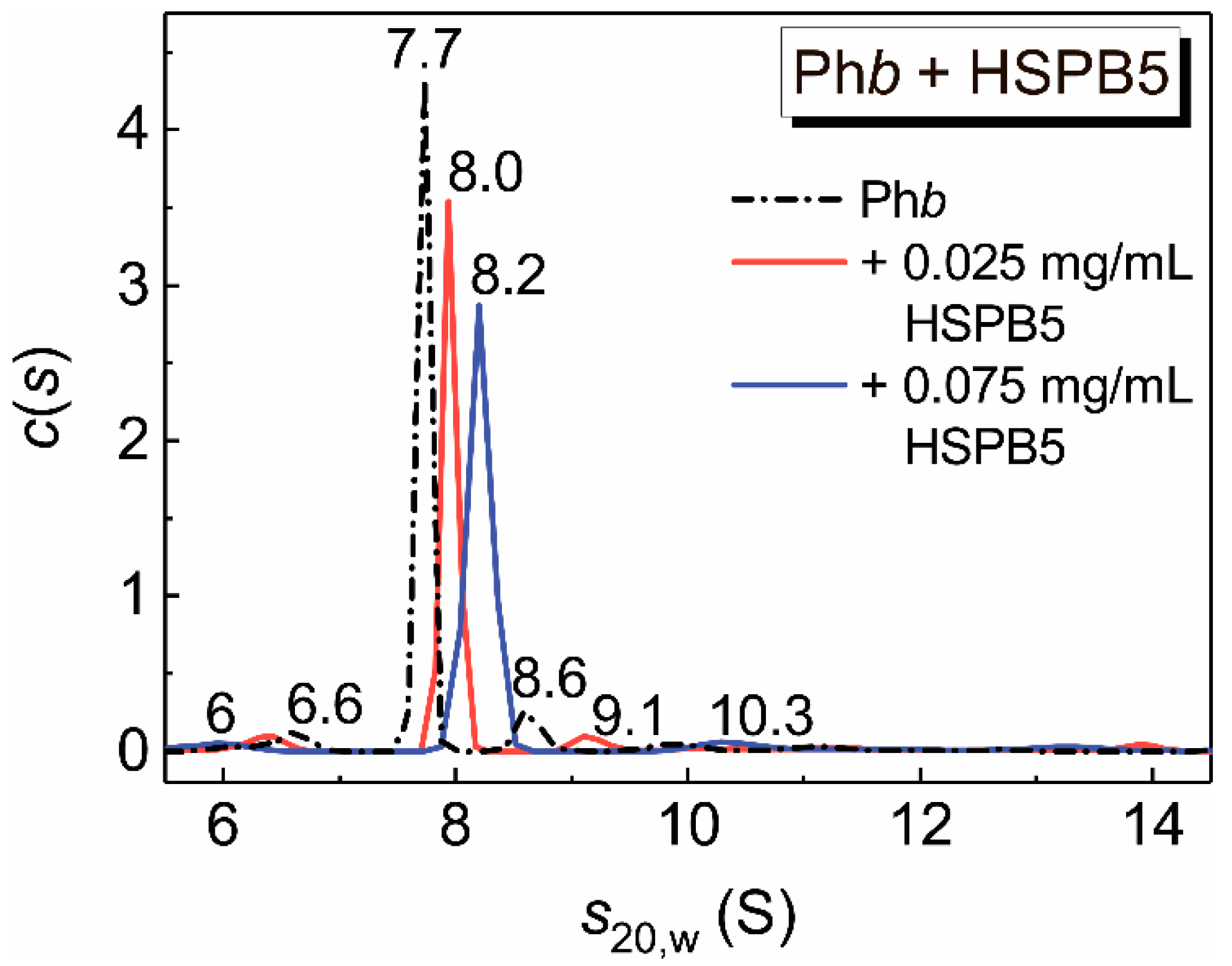
2.5. Sedimentation Velocity Analysis of HSPB5 and Phb at 48 °C
2.6. Sedimentation Velocity Analysis of HSPB5 and Phb at 48 °C Under Crowded Conditions
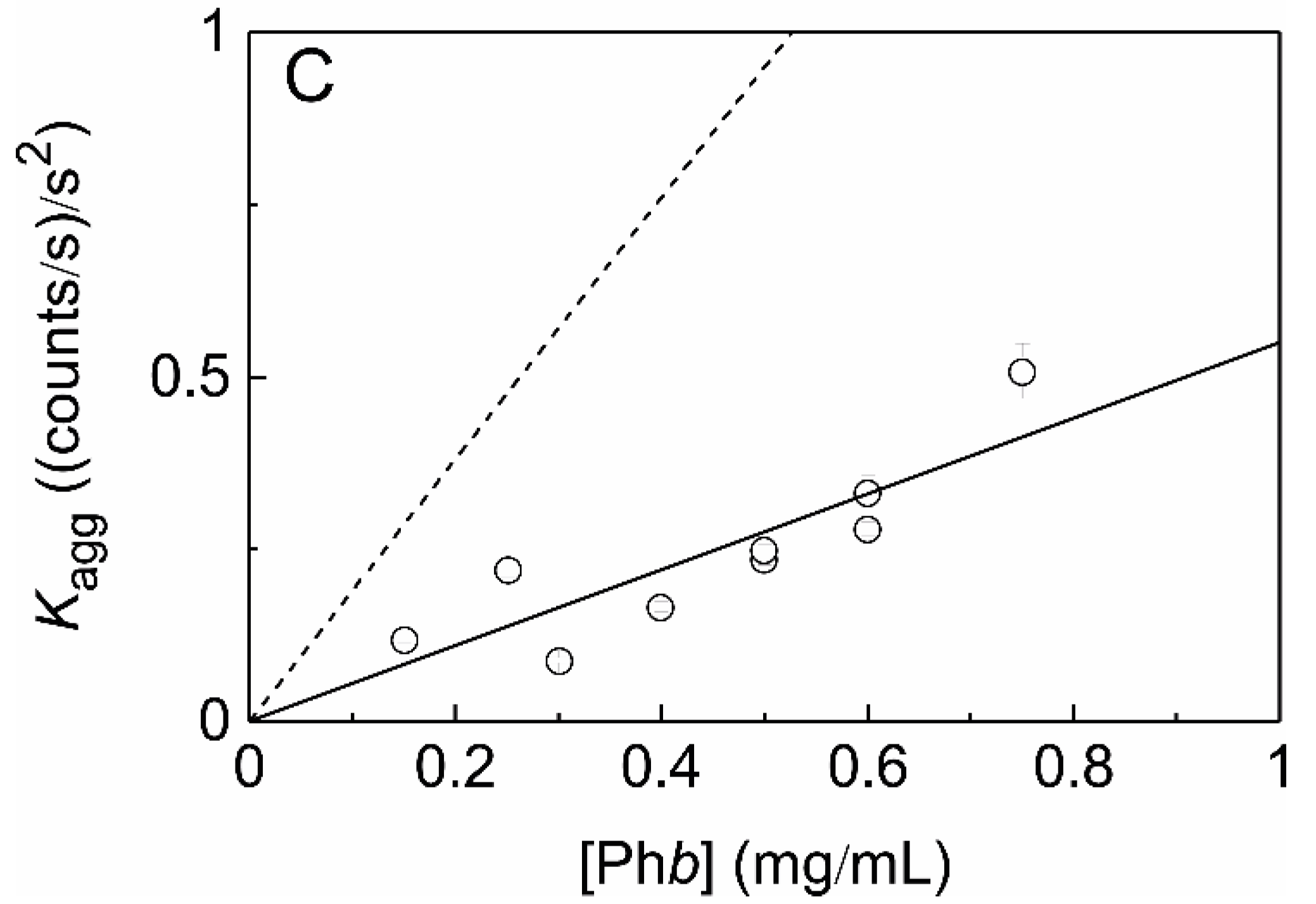
2.7. Effect of Crowding on the Chaperone-Like Activity of HSPB5
3. Discussion
4. Materials and Methods
4.1. Materials
4.2. Dynamic Light Scattering (DLS)
4.3. Analytical Ultracentrifugation (AUC)
4.4. Calculations
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| ACD | α-Crystalline domain |
| ATP | Adenosine triphosphate |
| AUC | Analytical ultracentrifugation |
| DLS | Dynamic light scattering |
| DTT | Dithiothreitol |
| EDTA | Ethylenediaminetetraacetic acid |
| EVE | Excluded volume effect |
| IDR | Intrinsically disordered region |
| PEG | Polyethylene glycol |
| PVP | Polyvinylpyrrolidone |
| sHSPs | Small heat shock proteins |
| SV | Sedimentation velocity |
References
- Haslbeck, M.; Vierling, E.A. First line of stress defense: Small heat shock proteins and their function in protein homeostasis. J. Mol. Biol. 2015, 427, 1537–1548. [Google Scholar] [CrossRef] [PubMed]
- Tyedmers, J.; Mogk, A.; Bukau, B. Cellular strategies for controlling protein aggregation. Nat. Rev. Mol. Cell Biol. 2010, 11, 777–788. [Google Scholar] [CrossRef] [PubMed]
- Chen, B.; Retzlaff, M.; Roos, T.; Frydman, J. Cellular strategies of protein quality control. Cold Spring Harb. Perspect. Biol. 2011, 3, a004374. [Google Scholar] [CrossRef]
- Mymrikov, E.V.; Seit-Nebi, A.S.; Gusev, N.B. Large potentials of small heat shock proteins. Physiol. Rev. 2011, 91, 1123–1159. [Google Scholar] [CrossRef] [PubMed]
- Webster, J.M.; Darling, A.L.; Uversky, V.N.; Blair, L.J. Small heat shock proteins, big impact on protein aggregation in neurodegenerative disease. Front. Pharmacol. 2019, 10, 1047. [Google Scholar] [CrossRef]
- Landsbury, A.; Perng, M.D.; Pohl, E.; Quinlan, R.A. Functional symbiosis between the intermediate filament cytoskeleton and small heat shock proteins. In Small Stress Proteins and Human Diseases; Arrigo, A.P., Simon, S., Eds.; Nova Science: New York, NY, USA, 2010; pp. 55–87. [Google Scholar]
- Arrigo, A.P. HSP27: Novel regulator of intracellular redox state. IUBMB Life 2001, 52, 303–307. [Google Scholar] [CrossRef]
- Basha, E.; O’Neill, H.; Vierling, E. Small heat shock proteins and α-crystallins: Dynamic proteins with flexible functions. Trends Biochem. Sci. 2012, 37, 106–117. [Google Scholar] [CrossRef]
- Hilton, G.R.; Lioe, H.; Stengel, F.; Baldwin, A.J.; Benesch, J.L.P. Small heat-shock proteins: Paramedics of the cell. In Molecular Chaperones. Topics in Current Chemistry; Jackson, S., Ed.; Springer: Berlin/Heidelberg, Germany, 2013; Volume 328, pp. 69–98. [Google Scholar] [CrossRef]
- Carra, S.; Alberti, S.; Benesch, J.L.P.; Boelens, W.; Buchner, J.; Carver, J.A.; Cecconi, C.; Ecroyd, H.; Gusev, N.B.; Hightower, L.E.; et al. Small heat shock proteins: Multifaceted proteins with important implications for life. Cell Stress Chaperones 2019, 24, 295–308. [Google Scholar] [CrossRef]
- Ciocca, D.R.; Calderwood, S.K. Heat shock proteins in cancer: Diagnostic, prognostic, predictive, and treatment implications. Cell Stress Chaperones 2005, 10, 86–103. [Google Scholar] [CrossRef]
- Clark, A.R.; Lubsen, N.H.; Slingsby, C. sHSP in the eye lens: Crystallin mutations, cataract and proteostasis. Int. J. Biochem. Cell Biol. 2012, 44, 1687–1697. [Google Scholar] [CrossRef] [PubMed]
- Makley, L.N.; McMenimen, K.A.; DeVree, B.T.; Goldman, J.W.; McGlasson, B.N.; Rajagopal, P.; Dunyak, B.M.; McQuade, T.J.; Thompson, A.D.; Sunahara, R.; et al. Pharmacological chaperone for α-crystallin partially restores transparency in cataract models. Science 2015, 350, 674–677. [Google Scholar] [CrossRef] [PubMed]
- Gerasimovich, E.S.; Strelkov, S.V.; Gusev, N.B. Some properties of three αB-crystallin mutants carrying point substitutions in the C-terminal domain and associated with congenital diseases. Biochimie 2017, 142, 168–178. [Google Scholar] [CrossRef] [PubMed]
- Nefedova, V.V.; Muranova, L.K.; Sudnitsyna, M.V.; Ryzhavskaya, A.S.; Gusev, N.B. Small heat shock proteins and distal hereditary neuropathies. Biochemistry 2015, 80, 1734–1747. [Google Scholar] [CrossRef] [PubMed]
- Ryazantsev, S.N.; Poliansky, N.B.; Chebotareva, N.A.; Muranov, O.K. 3D structure of the native α-crystallin from bovine eye lens. Int. J. Biol. Macromol. 2018, 117, 1289–1298. [Google Scholar] [CrossRef] [PubMed]
- Delaye, M.; Tardieu, A. Short-range order of crystallin proteins accounts for eye lens transparency. Nature 1983, 302, 415–417. [Google Scholar] [CrossRef] [PubMed]
- Sudnitsyna, M.V.; Mymrikov, E.V.; Seit-Nebi, A.S.; Gusev, N.B. The role of intrinsically disordered regions in the structure and functioning of small heat shock proteins. Curr. Protein Pept. Sci. 2012, 13, 76–85. [Google Scholar] [CrossRef]
- Delbecq, C.P.; Klevit, R.E. One size does not fit all: The oligomeric states of αB crystallin. FEBS Lett. 2013, 587, 1073–1080. [Google Scholar] [CrossRef]
- Carver, J.A.; Grosas, A.B.; Ecroyd, H.; Quinlan, R.A. The functional roles of the unstructured N- and C-terminal regions in αB-crystallin and other mammalian small heat-shock proteins. Cell Stress Chaperones 2017, 22, 627–638. [Google Scholar] [CrossRef]
- Peschek, J.; Braun, N.; Rohrberg, J.; Back, K.C.; Kriehuber, T.; Kastenmuller, A.; Weinkauf, S.; Buchner, J. Regulated structural transitions unleash the chaperone activity of αB-crystallin. Proc. Natl. Acad. Sci. USA 2013, 110, E3780–E3789. [Google Scholar] [CrossRef]
- Hochberg, G.K.A.; Shepherd, D.A.; Marklund, E.G.; Santhanagoplan, I.; Degiacomi, M.T.; Laganowsky, A.; Allison, T.M.; Basha, E.; Marty, M.T.; Galpin, M.R.; et al. Structural principles that enable oligomeric small heat shock protein paralogs to evolve distinct functions. Science 2018, 359, 930–935. [Google Scholar] [CrossRef]
- Hochberg, G.K.A.; Benesch, J.L.P. Dynamical structure of αB-crystallin. Prog. Biophys. Mol. Biol. 2014, 115, 11–20. [Google Scholar] [CrossRef] [PubMed]
- Braun, N.; Zacharias, M.; Peschek, J.; Kastenmuller, A.; Zou, J.; Hanzlik, M.; Haslbeck, M.; Rappsilber, J.; Buchner, J.; Weinkauf, S. Multiple molecular architectures of the eye lens chaperone αB-crystallin elucidated by a triple hybrid approach. Proc. Natl. Acad. Sci. USA 2011, 108, 20491–20496. [Google Scholar] [CrossRef] [PubMed]
- Chebotareva, N.A.; Eronina, T.B.; Roman, S.G.; Mikhaylova, V.V.; Sluchanko, N.N.; Gusev, N.B.; Kurganov, B.I. Oligomeric state of αB-crystallin under crowded conditions. Biochem. Biophys. Res. Commun. 2019, 508, 1101–1105. [Google Scholar] [CrossRef]
- Mchaourab, H.S.; Godar, J.A.; Stewart, P.L. Structure and mechanism of protein stability sensors: The chaperone activity of small heat-shock proteins. Biochemistry 2009, 48, 3828–3837. [Google Scholar] [CrossRef] [PubMed]
- Inoue, R.; Takata, T.; Fujii, N.; Ishii, K.; Uchiyama, S.; Sato, N.; Oba, Y.; Wood, K.; Kato, K.; Fujii, N.; et al. New insight into the dynamical system of αB-crystallin oligomers. Sci. Rep. 2016, 6, 29208. [Google Scholar] [CrossRef]
- Stengel, F.; Baldwin, A.J.; Painter, A.J.; Jaya, N.; Basha, E.; Kay, L.E.; Vierling, E.; Robinson, C.V.; Benesch, J.L.P. Quaternary dynamics and plasticity underlie small heat shock protein chaperone function. Proc. Natl. Acad. Sci. USA 2010, 107, 2007–2012. [Google Scholar] [CrossRef]
- Hayashi, J.; Carver, J.A. The multifaceted nature of αB-crystallin. Cell Stress Chaperones 2020, 25, 639–654. [Google Scholar] [CrossRef]
- Alderson, T.R.; Roche, J.; Gastall, H.Y.; Dias, D.M.; Pritišanac, I.; Ying, J.; Bax, A.; Benesch, J.L.P.; Baldwin, A.J. Local unfolding of the HSP27 monomer regulates chaperone activity. Nat. Commun. 2019, 10, 1068. [Google Scholar] [CrossRef]
- Liu, L.; Ghosh, J.G.; Clark, J.I.; Jiang, S. Studies of αB-crystallin subunit dynamics by surface plasmon resonance. Anal. Biochem. 2006, 350, 186–195. [Google Scholar] [CrossRef]
- Chebotareva, N.A.; Eronina, T.B.; Sluchanko, N.N.; Kurganov, B.I. Effect of Ca2+ and Mg2+ ions on oligomeric state and chaperone-like activity of αB-crystallin in crowded media. Int. J. Biol. Macromol. 2015, 76, 86–93. [Google Scholar] [CrossRef]
- Ecroyd, H.; Meehan, S.; Horwitz, J.; Aquilina, J.A.; Benesch, J.L.; Robinson, C.V.; Macphee, C.E.; Carver, J.A. Mimicking phosphorylation of αB-crystallin affects its chaperone activity. Biochem. J. 2007, 401, 129–141. [Google Scholar] [CrossRef]
- Jovcevski, B.; Kelly, M.A.; Rote, A.P.; Berg, T.; Gastall, H.Y.; Benesch, J.L.P.; Aquilina, J.A.; Ecroyd, H. Phosphomimics destabilize Hsp27 oligomeric assemblies and enhance chaperone activity. Chem. Biol. 2015, 22, 186–195. [Google Scholar] [CrossRef]
- Muranova, L.K.; Sudnitsyna, M.V.; Gusev, N.B. αB-Crystallin phosphorylation: Advances and problems. Biochemistry 2018, 83, 1196–1206. [Google Scholar] [CrossRef] [PubMed]
- Roman, S.G.; Chebotareva, N.A.; Kurganov, B.I. Anti-aggregation activity of small heat shock proteins under crowded conditions. Int. J. Biol. Macromol. 2017, 100, 97–103. [Google Scholar] [CrossRef]
- Fonin, A.V.; Darling, A.L.; Kuznetsova, I.M.; Turoverov, K.K.; Uversky, V.N. Intrinsically disordered proteins in crowded milieu: When chaos prevails within the cellular gumbo. Cell. Mol. Life Sci. 2018, 75, 3907–3929. [Google Scholar] [CrossRef]
- Kuznetsova, I.M.; Turoverov, K.K.; Uversky, V.N. What macromolecular crowding can do to a protein. Int. J. Mol. Sci. 2014, 15, 23090–23140. [Google Scholar] [CrossRef] [PubMed]
- Fu, X. Insights into how small heat shock proteins bind a great diversity of substrate proteins: A super-transformer model. In The Big Book on Small Heat Shock Proteins; Tanguay, R.M., Hightower, L.E., Eds.; Springer: Cham, Switzerland, 2015; pp. 101–117. [Google Scholar] [CrossRef]
- Zimmerman, S.B.; Trach, S.O. Estimation of macromolecule concentrations and excluded volume effects for the cytoplasm of Escherichia coli. J. Mol. Biol. 1991, 222, 599–620. [Google Scholar] [CrossRef]
- Zimmerman, S.B.; Minton, A.P. Macromolecular crowding: Biochemical, biophysical, and physiological consequences. Annu. Rev. Biophys. Biomol. Struct. 1993, 22, 23–65. [Google Scholar] [CrossRef]
- Ellis, R.J. Macromolecular crowding: Obvious but underappreciated. Trends Biochem. Sci. 2001, 26, 597–604. [Google Scholar] [CrossRef]
- Minton, A.P. The influence of macromolecular crowding and macromolecular confinement on biochemical reactions in physiological media. J. Biol. Chem. 2001, 276, 10577–10580. [Google Scholar] [CrossRef]
- Minton, A.P.; Wilf, J. Effect of macromolecular crowding upon the structure and function of an enzyme: Glyceraldehyde-3-phosphate dehydrogenase. Biochemistry 1981, 20, 4821–4826. [Google Scholar] [CrossRef] [PubMed]
- Chebotareva, N.A.; Kurganov, B.I.; Livanova, N.B. Biochemical effects of molecular crowding. Biochemistry 2004, 69, 1239–1251. [Google Scholar] [CrossRef]
- Ellis, R.J.; Minton, A.P. Protein aggregation in crowded environments. Biol. Chem. 2006, 387, 485–497. [Google Scholar] [CrossRef] [PubMed]
- Zhou, H.X.; Rivas, G.; Minton, A.P. Macromolecular crowding and confinement: Biochemical, biophysical, and potential physiological consequences. Annu. Rev. Biophys. 2008, 37, 375–397. [Google Scholar] [CrossRef] [PubMed]
- Ellis, R.J. Protein aggregation: Opposing effects of chaperones and crowding. In Folding for the Synapse; Wyttenbach, A., O’Connor, V., Eds.; Springer: Boston, MA, USA, 2011; pp. 9–34. [Google Scholar]
- Dewavrin, I.-Y.; Abdurrahiem, M.; Blocki, A.; Musib, M.; Piazza, F.; Raghunath, M. Synergistic rate of collagen fibrillogenesis in heterogeneous mixture of crowding agents. J. Phys. Chem. B 2015, 119, 4350–4358. [Google Scholar] [CrossRef]
- Shah, D.; Tan, A.L.; Ramakrishnan, V.; Jiang, J.; Rajagopalan, R. Effects of polydisperse crowders on aggregation reactions: A molecular thermodynamic analysis. J. Chem. Phys. 2011, 134, 064704. [Google Scholar] [CrossRef]
- Du, F.; Zhou, Z.; Mo, Z.-Y.; Shi, J.-Z.; Chen, J.; Liang, Y. Mixed macromolecular crowding accelerates the refolding of rabbit muscle creatine kinase: Implications for protein folding in physiological environments. J. Mol. Biol. 2006, 364, 469–482. [Google Scholar] [CrossRef]
- Batra, J.; Xu, K.; Zhou, H.-X. Nonadditive effects of mixed crowding on protein stability. Proteins 2009, 77, 133–138. [Google Scholar] [CrossRef]
- Phillip, Y.; Schreiber, G. Formation of protein complexes in crowded environments—From in vitro to in vivo. FEBS Lett. 2013, 587, 1046–1052. [Google Scholar] [CrossRef]
- Nakano, S.; Miyoshi, D.; Sugimoto, N. Effects of molecular crowding on the structures, interactions, and functions of nucleic acids. Chem. Rev. 2014, 114, 2733–2758. [Google Scholar] [CrossRef]
- Sarkar, M.; Smith, A.E.; Pielak, G.J. Impact of reconstituted cytosol on protein stability. Proc. Natl. Acad. Sci. USA 2013, 110, 19342–19347. [Google Scholar] [CrossRef] [PubMed]
- Mittal, S.; Chowhan, R.K.; Singh, L.R. Macromolecular crowding: Macromolecules friend or foe. Biochim. Biophys. Acta 2015, 1850, 1822–1831. [Google Scholar] [CrossRef]
- Stepanenko, O.V.; Povarova, O.I.; Sulatskaya, A.I.; Ferreira, L.A.; Zaslavsky, B.Y.; Kuznetsova, I.M.; Turoverov, K.K.; Uversky, V.N. Protein unfolding in crowded milieu: What crowding can do to a protein undergoing unfolding? J. Biomol. Struct. Dyn. 2016, 34, 2155–2170. [Google Scholar] [CrossRef]
- Shahid, S.; Hassan, M.I.; Islam, A.; Ahmad, F. Size-dependent studies of macromolecular crowding on the thermodynamic stability, structure and functional activity of proteins: In vitro and in silico approaches. Biochim. Biophys. Acta, Gen. Subj. 2017, 1861, 178–197. [Google Scholar] [CrossRef] [PubMed]
- Shahid, S.; Ahmad, F.; Hassan, M.I.; Islam, A. Relationship between protein stability and functional activity in the presence of macromolecular crowding agents alone and in mixture: An insight into stability-activity trade-off. Arch. Biochem. Biophys. 2015, 584, 42–50. [Google Scholar] [CrossRef] [PubMed]
- Sharp, K.A. Analysis of the size dependence of macromolecular crowding shows that smaller is better. Proc. Natl. Acad. Sci. USA 2015, 112, 7990–7995. [Google Scholar] [CrossRef] [PubMed]
- Chebotareva, N.A.; Makeeva, V.F.; Bazhina, S.G.; Eronina, T.B.; Gusev, N.B.; Kurganov, B.I. Interaction of Hsp27 with native phosphorylase kinase under crowding conditions. Macromol. Biosci. 2010, 10, 783–789. [Google Scholar] [CrossRef]
- Roman, S.G.; Chebotareva, N.A.; Eronina, T.B.; Kleymenov, S.Y.; Makeeva, V.F.; Poliansky, N.B.; Muranov, K.O.; Kurganov, B.I. Does the crowded cell-like environment reduce the chaperone-like activity of α-crystallin? Biochemistry 2011, 50, 10607–10623. [Google Scholar] [CrossRef]
- Chebotareva, N.A.; Eronina, T.B.; Roman, S.G.; Poliansky, N.B.; Muranov, K.O.; Kurganov, B.I. Effect of crowding and chaperones on self-association, aggregation and reconstitution of apophosphorylase b. Int. J. Biol. Macromol. 2013, 60, 69–76. [Google Scholar] [CrossRef]
- Sluchanko, N.N.; Chebotareva, N.A.; Gusev, N.B. Quaternary structure of human small heat shock protein HSPB6 (Hsp20) in crowded media modeled by trimethylamine N-oxide (TMAO): Effect of protein phosphorylation. Biochimie 2015, 108, 68–75. [Google Scholar] [CrossRef]
- Eronina, T.B.; Mikhaylova, V.V.; Chebotareva, N.A.; Kurganov, B.I. Kinetic regime of thermal aggregation of holo- and apoglycogen phosphorylases b. Int. J. Biol. Macromol. 2016, 92, 1252–1257. [Google Scholar] [CrossRef] [PubMed]
- Bumagina, Z.M.; Gurvits, B.Y.; Artemova, N.V.; Muranov, K.O.; Yudin, I.K.; Kurganov, B.I. Mechanism of suppression of dithiothreitol-induced aggregation of bovine α-lactalbumin by α-crystallin. Biophys. Chem. 2010, 146, 108–117. [Google Scholar] [CrossRef] [PubMed]
- Borzova, V.A.; Markossian, K.A.; Kurganov, B.I. Relationship between the initial rate of protein aggregation and the lag period for amorphous aggregation. Int. J. Biol. Macromol. 2014, 68, 144–150. [Google Scholar] [CrossRef] [PubMed]
- Chebotareva, N.A.; Filippov, D.O.; Kurganov, B.I. Effect of crowding on several stages of protein aggregation in test systems in the presence of α-crystallin. Int. J. Biol. Macromol. 2015, 80, 358–365. [Google Scholar] [CrossRef] [PubMed]
- Barford, D.; Johnson, L.N. The allosteric transition of glycogen phosphorylase. Nature 1989, 340, 609–616. [Google Scholar] [CrossRef]
- Permyakov, E.A.; Berliner, L.J. α-Lactalbumin: Structure and function. FEBS Lett. 2000, 473, 269–274. [Google Scholar] [CrossRef]
- Kuwajima, K. The molten globule state of α-lactalbumin. FASEB J. 1996, 10, 102–109. [Google Scholar] [CrossRef]
- Kulig, M.; Ecroyd, H. The small heat-shock protein αB-crystallin uses different mechanisms of chaperone action to prevent the amorphous versus fibrillar aggregation of α-lactalbumin. Biochem. J. 2012, 448, 343–352. [Google Scholar] [CrossRef]
- Carver, J.A.; Lindner, R.A.; Lyon, C.; Canet, D.; Hernandez, H.; Dobson, C.M.; Redfield, C. The interaction of the molecular chaperone α-crystallin with unfolding α-lactalbumin: A structural and kinetic spectroscopic study. J. Mol. Biol. 2002, 318, 815–827. [Google Scholar] [CrossRef]
- Lindner, R.A.; Treweek, T.M.; Carver, J.A. The molecular chaperone α-crystallin is in kinetic competition with aggregation to stabilize a monomeric molten-globule form of α-lactalbumin. Biochem. J. 2001, 354, 79–87. [Google Scholar] [CrossRef]
- Ecroyd, H.; Carver, J.A. The effect of small molecules in modulating the chaperone activity of αB-crystallin against ordered and disordered protein aggregation. FEBS J. 2008, 275, 935–947. [Google Scholar] [CrossRef] [PubMed]
- Sanders, H.M.; Jovcevski, B.; Carver, J.A.; Pukala, T.L. The molecular chaperone β-casein prevents amorphous and fibrillar aggregation of α-lactalbumin by stabilization of dynamic disorder. Biochem. J. 2020, 477, 629–643. [Google Scholar] [CrossRef] [PubMed]
- Patro, S.Y.; Przybycien, T.M. Simulations of reversible protein aggregate and crystal structure. Biophys. J. 1996, 70, 2888–2902. [Google Scholar] [CrossRef]
- Ferrone, F. Analysis of protein aggregation kinetics. Methods Enzymol. 1999, 309, 256–274. [Google Scholar] [CrossRef]
- Powers, E.T.; Ferrone, F.A. Kinetic models for protein misfolding and association. In Protein Misfolding Diseases: Current and Emerging Principles and Therapies; Ramirez-Alvarado, M., Kelly, J.W., Dobson, C.M., Eds.; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2010; pp. 73–92. [Google Scholar]
- Hirota, N.; Edskes, H.; Hall, D. Unified theoretical description of the kinetics of protein aggregation. Biophys. Rev. 2019, 11, 191–208. [Google Scholar] [CrossRef]
- Chen, S.; Ferrone, F.A.; Wetzel, R. Huntington’s disease age-of-onset linked to polyglutamine aggregation nucleation. Proc. Natl. Acad. Sci. USA 2002, 99, 11884–11889. [Google Scholar] [CrossRef]
- Kurganov, B.I. Kinetics of heat aggregation of proteins. Biochemistry 1998, 63, 364–366. [Google Scholar] [PubMed]
- Kurganov, B.I. Antiaggregation activity of chaperones and its quantification. Biochemistry 2013, 78, 1554–1566. [Google Scholar] [CrossRef]
- Kurganov, B.I. Quantification of anti-aggregation activity of chaperones. Int. J. Biol. Macromol. 2017, 100, 104–117. [Google Scholar] [CrossRef]
- Golub, N.; Meremyanin, A.; Markossian, K.; Eronina, T.; Chebotareva, N.; Asryants, R.; Muronets, V.; Kurganov, B. Evidence for the formation of start aggregates as an initial stage of protein aggregation. FEBS Lett. 2007, 581, 4223–4227. [Google Scholar] [CrossRef]
- Khanova, H.A.; Markossian, K.A.; Kurganov, B.I.; Samoilov, A.M.; Kleimenov, S.Y.; Levitsky, D.I.; Yudin, I.K.; Timofeeva, A.C.; Muranov, K.O.; Ostrovsky, M.A. Mechanism of chaperone-like activity. Suppression of thermal aggregation of βL-crystallin by α-crystallin. Biochemistry 2005, 44, 15480–15487. [Google Scholar] [CrossRef]
- Golub, N.V.; Markossian, K.A.; Kasilovich, N.V.; Sholukh, M.V.; Orlov, V.N.; Kurganov, B.I. Thermal inactivation, denaturation and aggregation of mitochondrial aspartate aminotransferase. Biophys. Chem. 2008, 135, 125–131. [Google Scholar] [CrossRef]
- Eronina, T.B.; Chebotareva, N.A.; Roman, S.G.; Kleymenov, S.Y.; Makeeva, V.F.; Polansky, N.B.; Muranov, K.O.; Kurganov, B.I. Thermal denaturation and aggregation of apoform of glycogen phosphorylase b. Effect of crowding agents and chaperones. Biopolymers 2014, 101, 504–516. [Google Scholar] [CrossRef] [PubMed]
- Borzova, V.A.; Markossian, K.A.; Kara, D.A.; Chebotareva, N.A.; Makeeva, V.F.; Poliansky, N.B.; Muranov, K.O.; Kurganov, B.I. Quantification of anti-aggregation activity of chaperones: A test-system based on dithiothreitol-induced aggregation of bovine serum albumin. PLoS ONE 2013, 8, e74367. [Google Scholar] [CrossRef] [PubMed]
- Benesch, J.L.P.; Ayoub, M.; Robinson, C.V.; Aquilina, J.A. Small heat shock protein activity is regulated by variable oligomeric substructure. J. Biol. Chem. 2008, 283, 28513–28517. [Google Scholar] [CrossRef] [PubMed]
- Haslbeck, M.; Peschek, J.; Buchner, J.; Weinkauf, S. Structure and function of α-crystallins: Traversing from in vitro to in vivo. Biochim. Biophys. Acta 2016, 1860, 159–166. [Google Scholar] [CrossRef] [PubMed]
- Bova, M.; Ding, L.-L.; Horwitz, J.; Fung, B.K.-K. Subunit exchange of αA-crystallin. J. Biol. Chem. 1997, 272, 29511–29517. [Google Scholar] [CrossRef]
- Abgar, S.; Backmann, J.; Aerts, T.; Vanhoudt, J.; Clauwaert, J. The structural differences between bovine lens αA- and αB-crystallin. Eur. J. Biochem. 2000, 267, 5916–5925. [Google Scholar] [CrossRef]
- Vanhoudt, J.; Abgar, S.; Aerts, T.; Clauwaert, J. Native quaternary structure of bovine α-crystallin. Biochemistry 2000, 39, 4483–4492. [Google Scholar] [CrossRef]
- Bakthisaran, R.; Akula, K.K.; Tangirala, R.; Rao, C.M. Phosphorylation of αB-crystallin: Role in stress, aging and pathophysiological conditions. Biochim. Biophys. Acta Gen. Subj. 2016, 1860, 167–182. [Google Scholar] [CrossRef]
- Aquilina, J.A.; Benesch, J.L.P.; Bateman, O.A.; Slingsby, C.; Robinson, C.V. Polydispersity of a mammalian chaperone: Mass spectrometry reveals the population of oligomers in αB-crystallin. Proc. Natl. Acad. Sci. USA 2003, 100, 10611–10616. [Google Scholar] [CrossRef] [PubMed]
- Hu, Z.; Jiang, J.; Rajagopalan, R. Effects of macromolecular crowding on biochemical reaction equilibria: A molecular thermodynamic perspective. Biophys. J. 2007, 93, 1464–1473. [Google Scholar] [CrossRef] [PubMed]
- Schlesinger, A.P.; Wang, Y.; Tadeo, X.; Millet, O.; Pielak, G.J. Macromolecular crowding fails to fold a globular protein in cell. J. Am. Chem. Soc. 2011, 133, 8082–8085. [Google Scholar] [CrossRef] [PubMed]
- Sukenik, S.; Harries, D. Balance of enthalpy and entropy in depletion forces. Curr. Opin. Colloid Interface Sci. 2013, 18, 495–501. [Google Scholar] [CrossRef]
- Mukherjee, S.K.; Gautam, S.; Biswas, S.; Kundu, J.; Chowdhury, P.K. Do macromolecular crowding agents exert only an excluded volume effect? A protein solvation study. J. Phys. Chem. B 2015, 119, 14145–14156. [Google Scholar] [CrossRef] [PubMed]
- Hall, D. Protein self-association in the cell: A mechanism for fine tuning the level of macromolecular crowding? Eur. Biophys. J. 2006, 35, 276–280. [Google Scholar] [CrossRef]
- Hall, D.; Dobson, C.M. Expanding to fill the gap: A possible role for inert biopolymers in regulating the extent of the “macromolecular crowding” effect. FEBS Lett. 2006, 580, 2584–2590. [Google Scholar] [CrossRef]
- Eronina, T.B.; Chebotareva, N.A.; Roman, S.G.; Makeeva, V.F.; Kleymenov, S.Y.; Kurganov, B.I. Effect of proline on thermal inactivation, denaturation and aggregation of glycogen phosphorylase b from rabbit skeletal muscle. Biophys. Chem. 2009, 141, 66–74. [Google Scholar] [CrossRef]
- Mymrikov, E.V.; Bukach, O.V.; Seit-Nebi, A.S.; Gusev, N.B. The pivotal role of the β7 strand in the intersubunit contacts of different human small heat shock proteins. Cell Stress Chaperones 2010, 15, 365–377. [Google Scholar] [CrossRef]
- Kastenschmidt, L.L.; Kastenschmidt, J.; Helmreich, E. Subunit interactions and their relationship to the allosteric properties of rabbit skeletal muscle phosphorylase b. Biochemistry 1968, 7, 3590–3608. [Google Scholar] [CrossRef]
- Kronman, M.J.; Andreotti, R.E. Inter- and intramolecular interactions of α-lactalbumin. I. The apparent heterogeneity at acid pH. Biochemistry 1964, 3, 1145–1151. [Google Scholar] [CrossRef] [PubMed]
- Brown, P.H.; Schuck, P. Macromolecular size-and-shape distributions by sedimentation ultracentrifugation. Biophys. J. 2006, 90, 4651–4661. [Google Scholar] [CrossRef] [PubMed]
- Scientist for Experimental Data Fitting. Microsoft Windows Version 2.0; MicroMath, Inc.: Salt Lake City, UT, USA, 1995. [Google Scholar]





| [HSPB5] (mg/mL) | s20,w (S) | Friction Ratio, f/f0 | Molecular Mass (kDa) |
|---|---|---|---|
| 0 | 2.9 ± 0.8 6.1 ± 0.4 | 2.7 | |
| 0.00045 | 1.85 ± 0.13 | 1.614 | 25.4 |
| 0.0015 | 2.0 ± 0.1 | 1.545 | 26 |
| 0.0075 | 1.9 ± 0.2 2.4 ± 0.3 | 1.925 | 33 47 |
| 0.03 | 1.6 ± 0.2 2.0 ± 0.1 | 1.836 | 23.9 37.4 |
| Additions | Kagg ((counts/s)/s2) | t0 (s) | 1 | j |
|---|---|---|---|---|
| Without addition of crowding agents | ||||
| – | 0.123 ± 0.007 | 323 ± 9 | 1.0 | - |
| Action of individual crowding agents | ||||
| PEG20kDa 25 mg/mL | 1.85 ± 0.04 | 287 ± 2 | 15.0 ± 0.9 | - |
| PVP10kDa 25 mg/mL | 0.28 ± 0.01 | 266 ± 5 | 2.3 ± 0.2 | - |
| PVP25kDa 25 mg/mL | 0.52 ± 0.02 | 232 ± 5 | 4.2 ± 0.3 | - |
| Ficoll70kDa 75 mg/mL | 0.144 ± 0.015 | 252 ± 12 | 1.17 ± 0.14 | - |
| Combined action of crowding agents | ||||
| PEG20kDa 25 mg/mL + Ficoll70kDa 75 mg/mL | 1.37 ± 0.05 | 196 ± 4 | 11.1 ± 0.8 | 0.71 ± 0.06 |
| PVP10kDa 25 mg/mL + Ficoll70kDa 75 mg/mL | 1.69 ± 0.05 | 263 ± 3 | 13.7 ± 0.9 | 8.8 ± 0.6 |
| PVP25kDa 25 mg/mL + Ficoll70kDa 75 mg/mL | 3.76 ± 0.21 | 240 ± 3 | 30.6 ± 2.4 | 8.7 ± 0.7 |
| PVP10kDa 25 mg/mL + PEG20kDa 25 mg/mL | 4.00 ± 0.22 | 227 ± 3 | 32.5 ± 2.6 | 2.06 ± 0.17 |
| PVP25kDa 25 mg/mL + PEG20kDa 25 mg/mL | 6.76 ± 0.29 | 203 ± 2 | 55 ± 4 | 3.12 ± 0.23 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chebotareva, N.A.; Roman, S.G.; Borzova, V.A.; Eronina, T.B.; Mikhaylova, V.V.; Kurganov, B.I. Chaperone-Like Activity of HSPB5: The Effects of Quaternary Structure Dynamics and Crowding. Int. J. Mol. Sci. 2020, 21, 4940. https://doi.org/10.3390/ijms21144940
Chebotareva NA, Roman SG, Borzova VA, Eronina TB, Mikhaylova VV, Kurganov BI. Chaperone-Like Activity of HSPB5: The Effects of Quaternary Structure Dynamics and Crowding. International Journal of Molecular Sciences. 2020; 21(14):4940. https://doi.org/10.3390/ijms21144940
Chicago/Turabian StyleChebotareva, Natalia A., Svetlana G. Roman, Vera A. Borzova, Tatiana B. Eronina, Valeriya V. Mikhaylova, and Boris I. Kurganov. 2020. "Chaperone-Like Activity of HSPB5: The Effects of Quaternary Structure Dynamics and Crowding" International Journal of Molecular Sciences 21, no. 14: 4940. https://doi.org/10.3390/ijms21144940
APA StyleChebotareva, N. A., Roman, S. G., Borzova, V. A., Eronina, T. B., Mikhaylova, V. V., & Kurganov, B. I. (2020). Chaperone-Like Activity of HSPB5: The Effects of Quaternary Structure Dynamics and Crowding. International Journal of Molecular Sciences, 21(14), 4940. https://doi.org/10.3390/ijms21144940





