Molecular Mapping of Hydrogen Sulfide Targets in Normal Human Keratinocytes
Abstract
1. Introduction
2. Results
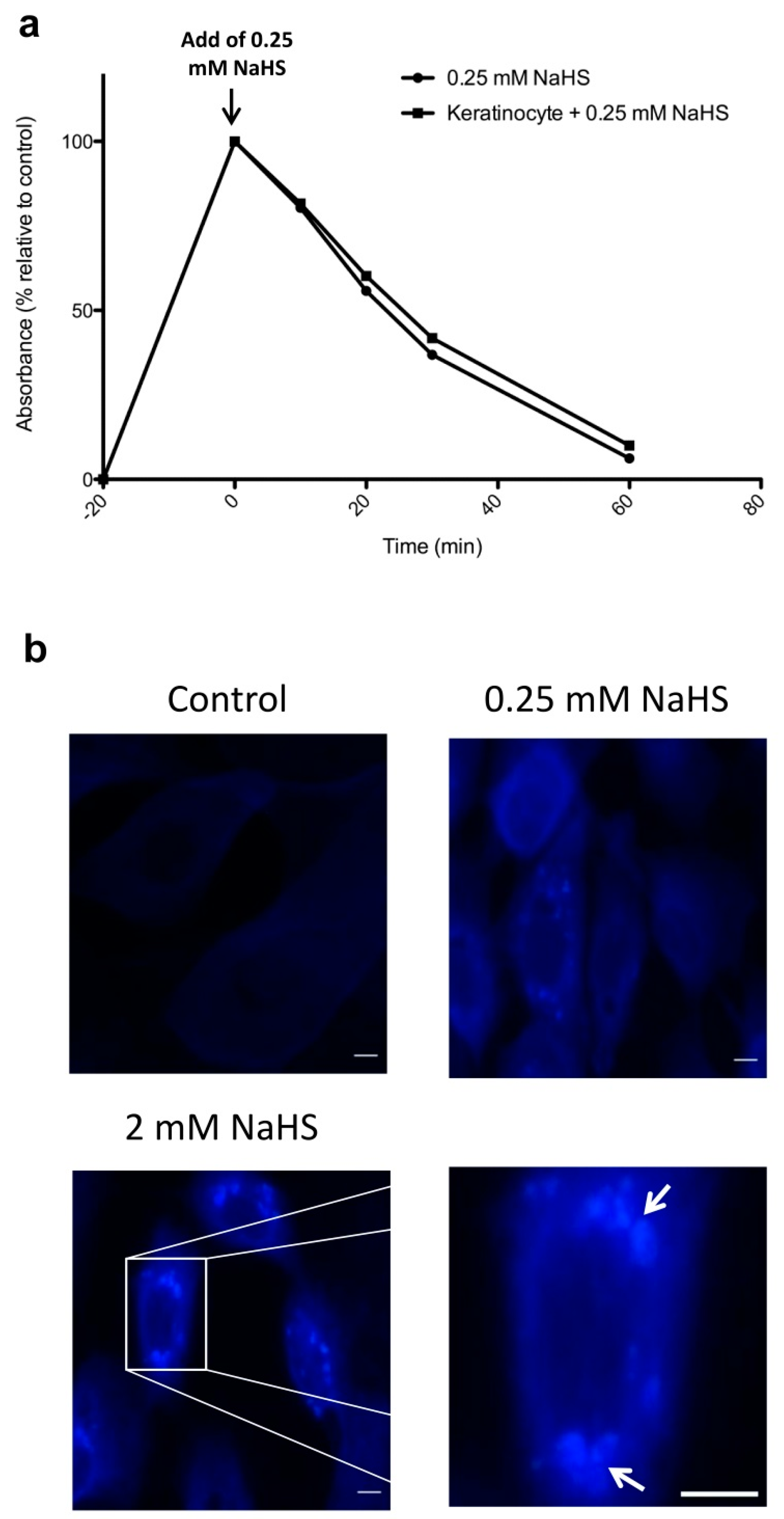
2.1. NaHS Induces a Rapid and Transient Increase in H2S in Human Keratinocyte Cultures
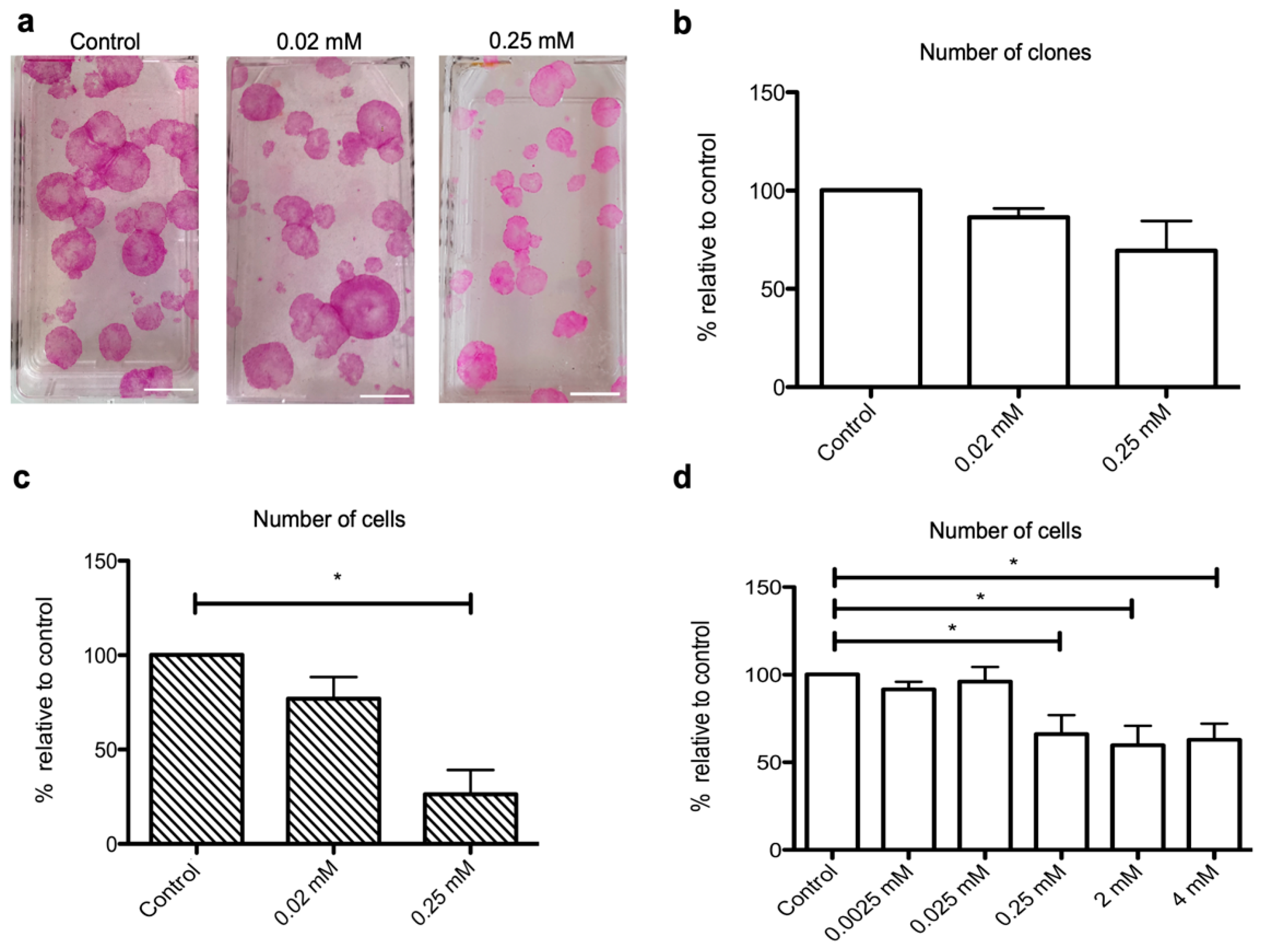
2.2. NaHS Impairs the Growth of Human Keratinocyte Progenitors and Stem Cells
2.3. NaHS Stimulates the Synthesis of Superoxide Dismutase 2 (SOD2) by Cultured Human Keratinocytes
2.4. NaHS Exerts Specific Effects on the RNA Profile of Cultured Human Keratinocytes
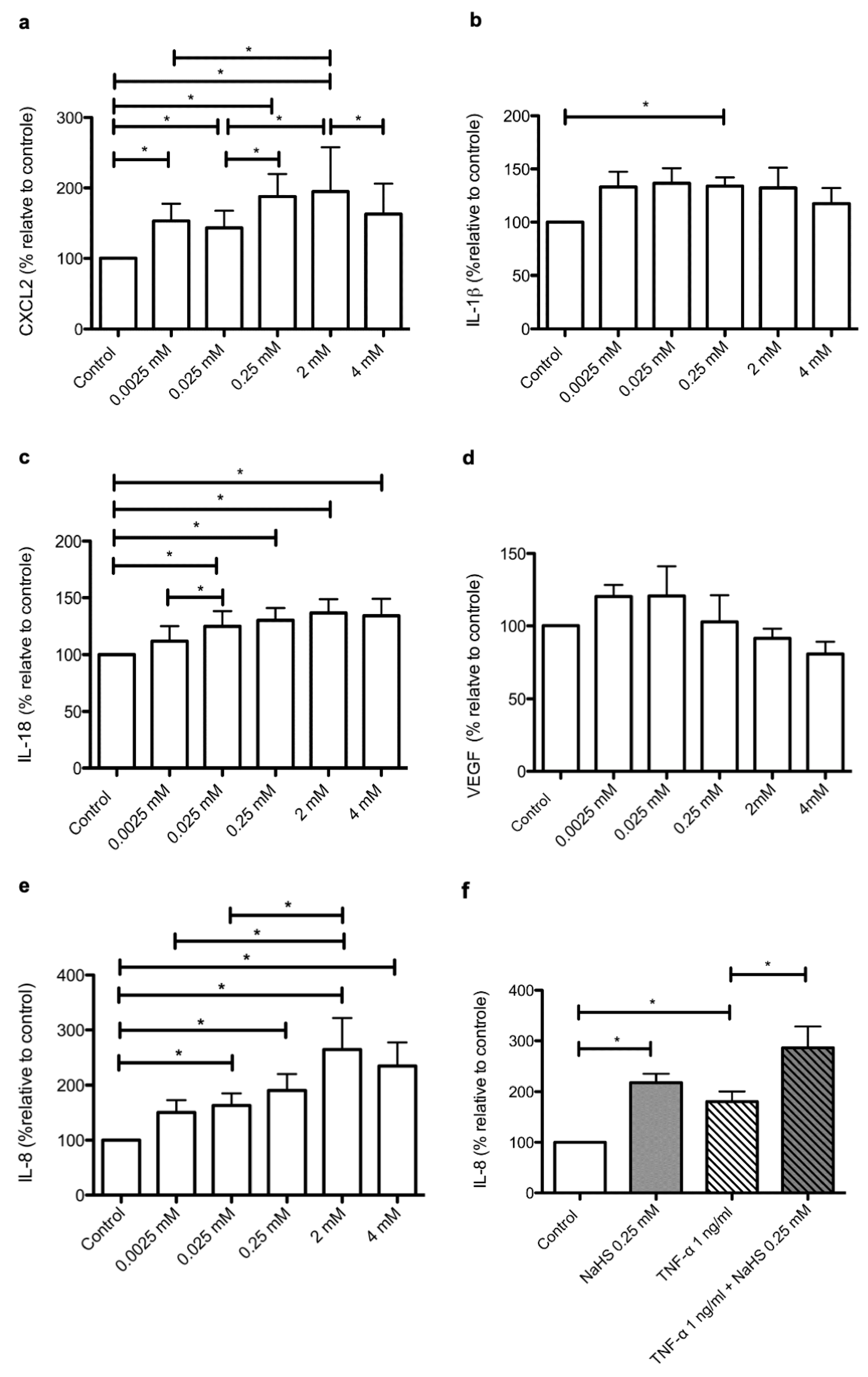
2.5. NaHS Modifies the Secretary Profile of Cultured Human Keratinocytes
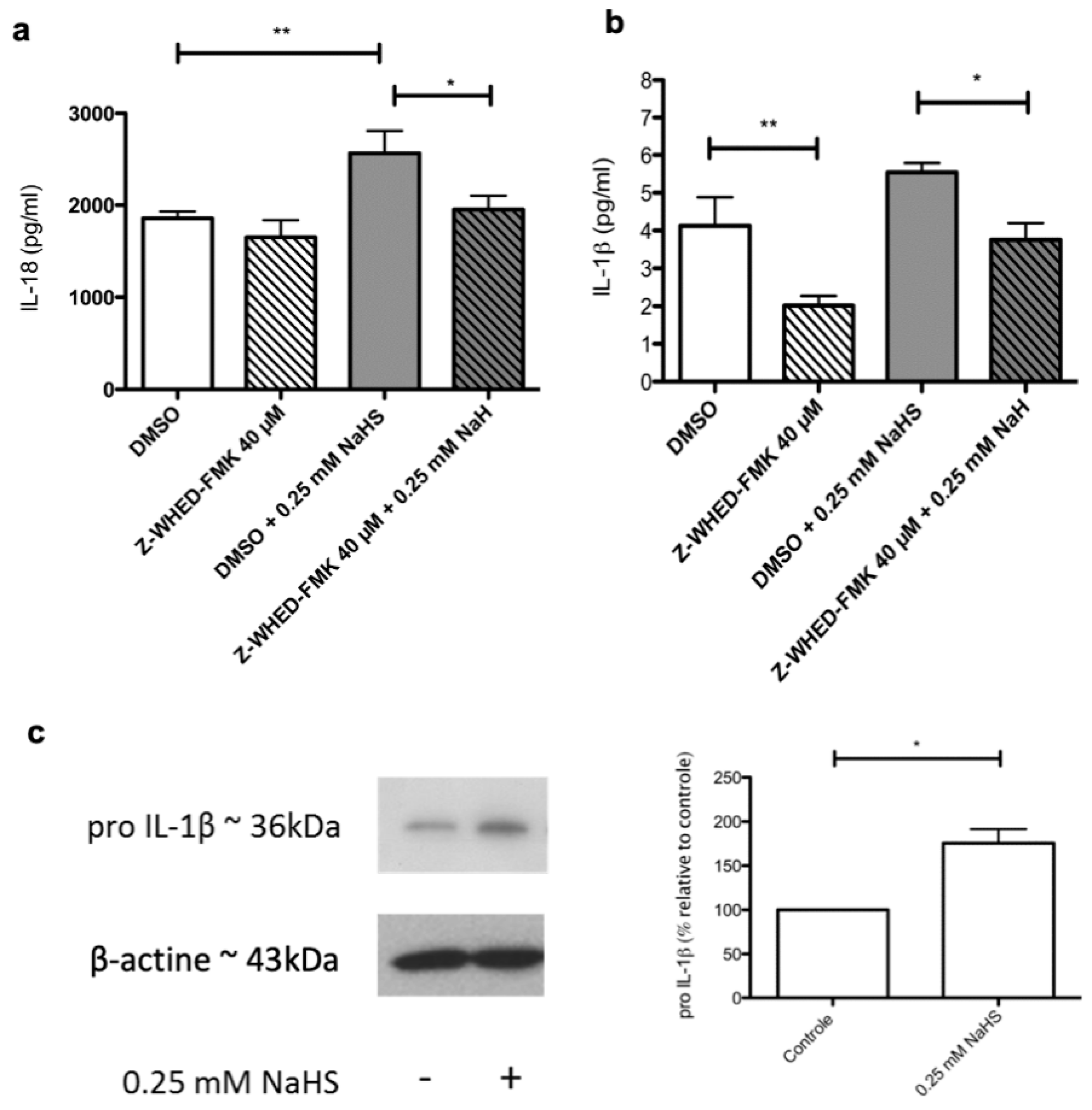
2.6. Distinct Molecular Pathways Mediate the Stimulating Effects of NaHS on the Synthesis of IL-1β and IL-18 by Cultured Human Keratinocytes
3. Discussion
4. Materials and Methods
4.1. Ethical Statement
4.2. Generation of Cultured Human Epidermal Sheets
4.3. NaHS
4.4. Measurements of H2S in Culture Supernatants
4.5. Fluorescence-Based Detection of Intracellular H2S
4.6. Clonogenic Assay
4.7. RNA–seq and Proteomics Analyses
4.8. Enzyme-Linked Immunosorbent Assay (ELISA)
4.9. Western Blot Analysis
4.10. Statistical Analysis
4.11. Data Availability
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| NAHS | Sodium hydrosulfide |
| H2S | Hydrogen sulfide |
| IL | Interleukin |
References
- Carbajo, J.M.; Maraver, F. Sulphurous Mineral Waters: New Applications for Health. Evid.-Based Complement. Altern. Med. 2017, 2017, 1–11. [Google Scholar] [CrossRef]
- Gianfaldoni, S.; Tchernev, G.; Wollina, U.; Roccia, M.G.; Fioranelli, M.; Gianfaldoni, R.; Lotti, T. History of the Baths and Thermal Medicine. Open Access Maced. J. Med. Sci. 2017, 5, 566–568. [Google Scholar] [CrossRef]
- Huang, A.; Seité, S.; Adar, T. The use of balneotherapy in dermatology. Clin. Dermatol. 2018, 36, 363–368. [Google Scholar] [CrossRef]
- Matz, H.; Orion, E.; Wolf, R. Balneotherapy in dermatology. Dermatol. Ther. 2003, 16, 132–140. [Google Scholar] [CrossRef] [PubMed]
- Davinelli, S.; Bassetto, F.; Vitale, M.; Scapagnini, G. Chapter 10—Thermal Waters and the Hormetic Effects of Hydrogen Sulfide on Inflammatory Arthritis and Wound Healing. In The Science of Hormesis in Health and Longevity; Academic Press: Cambridge, MA, USA, 2019; pp. 121–126. [Google Scholar]
- Xie, X.; Dai, H.; Zhuang, B.; Chai, L.; Xie, Y.; Li, Y. Exogenous hydrogen sulfide promotes cell proliferation and differentiation by modulating autophagy in human keratinocytes. Biochem. Biophys. Res. Commun. 2016, 472, 437–443. [Google Scholar] [CrossRef]
- Mirandola, P.; Gobbi, G.; Micheloni, C.; Vaccarezza, M.; Di Marcantonio, D.; Ruscitti, F.; de Panfilis, G.; Vitale, M. Hydrogen sulfide inhibits IL-8 expression in human keratinocytes via MAP kinase signaling. Lab. Investig. 2011, 91, 1188–1194. [Google Scholar] [CrossRef]
- Gobbi, G.; Ricci, F.; Malinverno, C.; Carubbi, C.; Pambianco, M.; de Panfilis, G.; Vitale, M.; Mirandola, P. Hydrogen sulfide impairs keratinocyte cell growth and adhesion inhibiting mitogen-activated protein kinase signaling. Lab. Investig. 2009, 89, 994–1006. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.; Yang, Z.; Zhang, M.; Dong, Q.; Wang, X.; Lan, A.; Zeng, F.; Chen, P.; Wang, C.; Feng, J. Hydrogen sulfide protects against chemical hypoxia-induced cytotoxicity and inflammation in HaCaT cells through inhibition of ROS/NF-κB/COX-2 pathway. PLoS ONE 2011, 6, e21971. [Google Scholar] [CrossRef]
- Merighi, S.; Gessi, S.; Varani, K.; Fazzi, D.; Borea, P.A. Hydrogen sulfide modulates the release of nitric oxide and VEGF in human keratinocytes. Pharmacol. Res. 2012, 66, 428–436. [Google Scholar] [CrossRef] [PubMed]
- Li, A.; Simmons, P.J.; Kaur, P. Identification and isolation of candidate human keratinocyte stem cells based on cell surface phenotype. Proc. Natl. Acad. Sci. USA 1998, 95, 3902–3907. [Google Scholar] [CrossRef] [PubMed]
- Krebsbach, P.H.; Villa-Diaz, L.G. The Role of Integrin α6 (CD49f) in Stem Cells: More than a Conserved Biomarker. Stem Cells Dev. 2017, 26, 1090–1099. [Google Scholar] [CrossRef] [PubMed]
- Davis, A.P.; Grondin, C.J.; Johnson, R.J.; Sciaky, D.; McMorran, R.; Wiegers, J.; Wiegers, T.C.; Mattingly, C.J. The Comparative Toxicogenomics Database: Update 2019. Nucleic Acids Res. 2019, 47, D948–D954. [Google Scholar] [CrossRef] [PubMed]
- Kuleshov, M.V.; Jones, M.R.; Rouillard, A.D.; Fernandez, N.F.; Duan, Q.; Wang, Z.; Koplev, S.; Jenkins, S.L.; Jagodnik, K.M.; Lachmann, A.; et al. Enrichr: A comprehensive gene set enrichment analysis web server 2016 update. Nucleic Acids Res. 2016, 44, W90–W97. [Google Scholar] [CrossRef] [PubMed]
- Ross, D.; Siegel, D. NQO1 in protection against oxidative stress. Curr. Opin. Toxicol. 2018, 7, 67–72. [Google Scholar] [CrossRef]
- Kobayashi, A.; Kang, M.-I.; Okawa, H.; Ohtsuji, M.; Zenke, Y.; Chiba, T.; Igarashi, K.; Yamamoto, M. Oxidative Stress Sensor Keap1 Functions as an Adaptor for Cul3-Based E3 Ligase to Regulate Proteasomal Degradation of Nrf2. Mol. Cell. Biol. 2004, 24, 7130–7139. [Google Scholar] [CrossRef]
- Boro, M.; Balaji, K.N. CXCL1 and CXCL2 Regulate NLRP3 Inflammasome Activation via G-Protein–Coupled Receptor CXCR2. J. Immunol. 2017, 199, 1660–1671. [Google Scholar] [CrossRef]
- De Filippo, K.; Dudeck, A.; Hasenberg, M.; Nye, E.; van Rooijen, N.; Hartmann, K.; Gunzer, M.; Roers, A.; Hogg, N. Mast cell and macrophage chemokines CXCL1/CXCL2 control the early stage of neutrophil recruitment during tissue inflammation. Blood 2013, 121, 4930–4937. [Google Scholar] [CrossRef]
- Rajarathnam, K.; Schnoor, M.; Richardson, R.M.; Rajagopal, S. How do chemokines navigate neutrophils to the target site: Dissecting the structural mechanisms and signaling pathways. Cell. Signal. 2019, 54, 69–80. [Google Scholar] [CrossRef]
- Rouault, C.; Pellegrinelli, V.; Schilch, R.; Cotillard, A.; Poitou, C.; Tordjman, J.; Sell, H.; Clément, K.; Lacasa, D. Roles of chemokine ligand-2 (CXCL2) and neutrophils in influencing endothelial cell function and inflammation of human adipose tissue. Endocrinology 2013, 154, 1069–1079. [Google Scholar] [CrossRef]
- Guo, H.; Callaway, J.B.; Ting, J.P.-Y. Inflammasomes: Mechanism of action, role in disease, and therapeutics. Nat. Med. 2015, 21, 677–687. [Google Scholar] [CrossRef]
- Lee, D.-J.; Du, F.; Chen, S.-W.; Nakasaki, M.; Rana, I.; Shih, V.F.S.; Hoffmann, A.; Jamora, C. Regulation and function of the caspase-1 in an inflammatory microenvironment. J. Investig. Dermatol. 2015, 135, 2012–2020. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.; Kang, J.S.; Lee, W.J. The Production IL-21 and VEGF in UVB-irradiated Human Keratinocyte Cell Line, HaCaT. Immune Netw. 2010, 10, 75–80. [Google Scholar] [CrossRef] [PubMed]
- Viac, J.; Palacio, S.; Schmitt, D.; Claudy, A. Expression of vascular endothelial growth factor in normal epidermis, epithelial tumors and cultured keratinocytes. Arch. Dermatol. Res. 1997, 289, 158–163. [Google Scholar] [CrossRef] [PubMed]
- Rennekampff, H.-O.; Hansbrough, J.F.; Kiessig, V.; Doré, C.; Sticherling, M.; Schröder, J.-M. Bioactive Interleukin-8 Is Expressed in Wounds and Enhances Wound Healing. J. Surg. Res. 2000, 93, 41–54. [Google Scholar] [CrossRef] [PubMed]
- Sticherling, M.; Bornscheuer, E.; Schröder, J.M.; Christophers, E. Localization of neutrophil-activating peptide-1/interleukin-8-immunoreactivity in normal and psoriatic skin. J. Investig. Dermatol. 1991, 96, 26–30. [Google Scholar] [CrossRef]
- Bhattacharyya, S.; Dudeja, P.K.; Tobacman, J.K. Tumor Necrosis Factor α-induced Inflammation Is Increased but Apoptosis Is Inhibited by Common Food Additive Carrageenan. J. Biol. Chem. 2010, 285, 39511–39522. [Google Scholar] [CrossRef]
- Zhou, P.; Lu, S.; Luo, Y.; Wang, S.; Yang, K.; Zhai, Y.; Sun, G.; Sun, X. Attenuation of TNF-α-Induced Inflammatory Injury in Endothelial Cells by Ginsenoside Rb1 via Inhibiting NF-κB, JNK and p38 Signaling Pathways. Front. Pharmacol. 2017, 8, 464. [Google Scholar] [CrossRef]
- Carubbi, C.; Gobbi, G.; Bucci, G.; Gesi, M.; Vitale, M.; Mirandola, P. Skin, Inflammation and Sulfurous Waters: What is Known, What is Believed, Skin, Inflammation and Sulfurous Waters: What is Known, What is Believed. Eur. J. Inflamm. 2013, 11, 591–599. [Google Scholar] [CrossRef]
- Zhao, F.; Lei, F.; Yan, X.; Zhang, S.; Wang, W.; Zheng, Y. Protective Effects of Hydrogen Sulfide Against Cigarette Smoke Exposure-Induced Placental Oxidative Damage by Alleviating Redox Imbalance via Nrf2 Pathway in Rats. Cell. Physiol. Biochem. 2018, 48, 1815–1828. [Google Scholar] [CrossRef]
- Liang, Y.; Zheng, P.; Li, S.; Li, K.; Xu, H. Nitrate reductase-dependent NO production is involved in H2S-induced nitrate stress tolerance in tomato via activation of antioxidant enzymes. Sci. Hortic. 2018, 229, 207–214. [Google Scholar] [CrossRef]
- Wen, Y.-D.; Wang, H.; Kho, S.; Rinkiko, S.; Sheng, X.; Shen, H.-M.; Zhu, Y.-Z. Hydrogen Sulfide Protects HUVECs against Hydrogen Peroxide Induced Mitochondrial Dysfunction and Oxidative Stress. PLoS ONE 2013, 8, e53147. [Google Scholar] [CrossRef] [PubMed]
- Glory, A.; Averill-Bates, D.A. The antioxidant transcription factor Nrf2 contributes to the protective effect of mild thermotolerance (40 °C) against heat shock-induced apoptosis. Free Radic. Biol. Med. 2016, 99, 485–497. [Google Scholar] [CrossRef] [PubMed]
- Shen, J.; Rasmussen, M.; Dong, Q.-R.; Tepel, M.; Scholze, A. Expression of the NRF2 Target Gene NQO1 Is Enhanced in Mononuclear Cells in Human Chronic Kidney Disease. Oxid. Med. Cell. Longev. 2017, 2017, 9091879. [Google Scholar] [CrossRef] [PubMed]
- Xie, Z.-Z.; Liu, Y.; Bian, J.-S. Hydrogen Sulfide and Cellular Redox Homeostasis. Oxid. Med. Cell. Longev. 2016, 2016, 6043038. [Google Scholar] [CrossRef]
- Meng, W.; Pei, Z.; Feng, Y.; Zhao, J.; Chen, Y.; Shi, W.; Xu, Q.; Lin, F.; Sun, M.; Xiao, K. Neglected role of hydrogen sulfide in sulfur mustard poisoning: Keap1 S-sulfhydration and subsequent Nrf2 pathway activation. Sci. Rep. 2017, 7, 1–17. [Google Scholar] [CrossRef]
- Corsello, T.; Komaravelli, N.; Casola, A. Role of Hydrogen Sulfide in NRF2- and Sirtuin-Dependent Maintenance of Cellular Redox Balance. Antioxidants (Basel) 2018, 7, 129. [Google Scholar] [CrossRef]
- Kaspar, J.W.; Jaiswal, A.K. An Autoregulatory Loop between Nrf2 and Cul3-Rbx1 Controls Their Cellular Abundance. J. Biol. Chem. 2010, 285, 21349–21358. [Google Scholar] [CrossRef]
- Li, R.; Jia, Z.; Zhu, H. Regulation of Nrf2 Signaling. React. Oxyg. Species (Apex) 2019, 8, 312–322. [Google Scholar] [CrossRef]
- Olson, K.R.; Gao, Y.; Arif, F.; Arora, K.; Patel, S.; DeLeon, E.R.; Sutton, T.R.; Feelisch, M.; Cortese-Krott, M.M.; Straub, K.D. Metabolism of hydrogen sulfide (H2S) and Production of Reactive Sulfur Species (RSS) by superoxide dismutase. Redox Biol. 2017, 15, 74–85. [Google Scholar] [CrossRef]
- Olson, K.R.; Gao, Y. Effects of inhibiting antioxidant pathways on cellular hydrogen sulfide and polysulfide metabolism. Free Radic. Biol. Med. 2019, 135, 1–14. [Google Scholar] [CrossRef]
- Olaru, F.; Jensen, L.E. Staphylococcus aureus stimulates neutrophil targeting chemokine expression in keratinocytes through an autocrine IL-1alpha signaling loop. J. Investig. Dermatol. 2010, 130, 1866–1876. [Google Scholar] [CrossRef] [PubMed]
- Ridiandries, A.; Tan, J.T.M.; Bursill, C.A. The Role of Chemokines in Wound Healing. Int. J. Mol. Sci. 2018, 19, 3217. [Google Scholar] [CrossRef] [PubMed]
- Gillitzer, R.; Goebeler, M. Chemokines in cutaneous wound healing. J. Leukoc. Biol. 2001, 69, 513–521. [Google Scholar] [PubMed]
- Barrandon, Y.; Green, H. Three clonal types of keratinocyte with different capacities for multiplication. Proc. Natl. Acad. Sci. USA 1987, 84, 2302–2306. [Google Scholar] [CrossRef] [PubMed]
- Black, A.F.; Bouez, C.; Perrier, E.; Schlotmann, K.; Chapuis, F.; Damour, O. Optimization and characterization of an engineered human skin equivalent. Tissue Eng. 2005, 11, 723–733. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.Z.; Wang, Z.J.; Ho, P.; Loke, Y.Y.; Zhu, Y.C.; Huang, S.H.; Tan, C.S.; Whiteman, M.; Lu, J.; Moore, P.K. Hydrogen sulfide and its possible roles in myocardial ischemia in experimental rats. J. Appl. Physiol. 2007, 102, 261–268. [Google Scholar] [CrossRef]
- Li, L.; Bhatia, M.; Zhu, Y.Z.; Zhu, Y.C.; Ramnath, R.D.; Wang, Z.J.; Anuar, F.B.M.; Whiteman, M.; Salto-Tellez, M.; Moore, P.K. Hydrogen sulfide is a novel mediator of lipopolysaccharide-induced inflammation in the mouse. FASEB J. 2005, 19, 1196–1198. [Google Scholar] [CrossRef]
- Jobeili, L.; Rousselle, P.; Béal, D.; Blouin, E.; Roussel, A.-M.; Damour, O.; Rachidi, W. Selenium preserves keratinocyte stemness and delays senescence by maintaining epidermal adhesion. Aging (Albany NY) 2017, 9, 2302–2315. [Google Scholar] [CrossRef]
- Arab, T.; Raffo-Romero, A.; Van Camp, C.; Lemaire, Q.; Le Marrec-Croq, F.; Drago, F.; Aboulouard, S.; Slomianny, C.; Lacoste, A.-S.; Guigon, I.; et al. Proteomic characterisation of leech microglia extracellular vesicles (EVs): Comparison between differential ultracentrifugation and OptiprepTM density gradient isolation. J. Extracell. Vesicles 2019, 8, 1603048. [Google Scholar] [CrossRef]
- Sinel, C.; Cacaci, M.; Meignen, P.; Guérin, F.; Davies, B.W.; Sanguinetti, M.; Giard, J.-C.; Cattoir, V. Subinhibitory Concentrations of Ciprofloxacin Enhance Antimicrobial Resistance and Pathogenicity of Enterococcus faecium. Antimicrob. Agents Chemother. 2017, 61, e02763-16. [Google Scholar] [CrossRef]
- Cardon, T.; Hervé, F.; Delcourt, V.; Roucou, X.; Salzet, M.; Franck, J.; Fournier, I. Optimized Sample Preparation Workflow for Improved Identification of Ghost Proteins. Anal. Chem. 2020, 92, 1122–1129. [Google Scholar] [CrossRef] [PubMed]
- Delloye-Bourgeois, C.; Bertin, L.; Thoinet, K.; Jarrosson, L.; Kindbeiter, K.; Buffet, T.; Tauszig-Delamasure, S.; Bozon, M.; Marabelle, A.; Combaret, V.; et al. Microenvironment-Driven Shift of Cohesion/Detachment Balance within Tumors Induces a Switch toward Metastasis in Neuroblastoma. Cancer Cell 2017, 32, 427–443.e8. [Google Scholar] [CrossRef] [PubMed]
- Bringuier, P.-P.; Schalken, J.A.; Hervieu, V.; Giroldi, L.A. Involvement of orphan nuclear receptor COUP-TFII in cadherin-6 and cadherin-11 regulation: Implications in development and cancer. Mech. Dev. 2015, 136, 64–72. [Google Scholar] [CrossRef] [PubMed]
| GO Term | Adjusted p-Value |
|---|---|
| response to reactive oxygen species | 5.79 × 10−19 |
| extrinsic apoptotic signaling pathway | 9.14 × 10−18 |
| activation of cysteine-type endopeptidase activity involved in apoptotic process | 3.08 × 10−17 |
| cellular response to reactive oxygen species | 6.10 × 10−17 |
| glutathione metabolic process | 7.22 × 10−16 |
| negative regulation of extrinsic apoptotic signaling pathway | 2.05 × 10−15 |
| cellular response to mechanical stimulus | 1.00 × 10−14 |
| I-kappaB kinase/NF-kappaB signaling | 2.73 × 10−13 |
| interleukin-1-mediated signaling pathway | 2.73 × 10−13 |
| toll-like receptor signaling pathway | 3.46 × 10−13 |
| Gene Symbol | Gene Name |
|---|---|
| Up-regulated genes | |
| CASP1 | Caspase-1 |
| CAST | Calpastatin |
| CUL3 | Cullin 3 |
| CXCL2 | C-X-C motif chemokine ligand 2 |
| GDF15 | Growth differentiation factor 15 |
| NQO1 | NAD(P)H quinone dehydrogenase 1 |
| SLC3A2 | Solute carrier family 3 member 2 |
| UGT1A6 | UDP glucuronosyltransferase family 1 member A6 |
| Down-regulated genes | |
| CAMKK2 | Calcium/calmodulin dependent protein kinase kinase 2 |
| HOMER3 | Homer scaffold protein 3 |
| PTK2 | Protein tyrosine kinase 2 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gross-Amat, O.; Guillen, M.; Gimeno, J.-P.; Salzet, M.; Lebonvallet, N.; Misery, L.; Auxenfans, C.; Nataf, S. Molecular Mapping of Hydrogen Sulfide Targets in Normal Human Keratinocytes. Int. J. Mol. Sci. 2020, 21, 4648. https://doi.org/10.3390/ijms21134648
Gross-Amat O, Guillen M, Gimeno J-P, Salzet M, Lebonvallet N, Misery L, Auxenfans C, Nataf S. Molecular Mapping of Hydrogen Sulfide Targets in Normal Human Keratinocytes. International Journal of Molecular Sciences. 2020; 21(13):4648. https://doi.org/10.3390/ijms21134648
Chicago/Turabian StyleGross-Amat, Olivia, Marine Guillen, Jean-Pascal Gimeno, Michel Salzet, Nicolas Lebonvallet, Laurent Misery, Céline Auxenfans, and Serge Nataf. 2020. "Molecular Mapping of Hydrogen Sulfide Targets in Normal Human Keratinocytes" International Journal of Molecular Sciences 21, no. 13: 4648. https://doi.org/10.3390/ijms21134648
APA StyleGross-Amat, O., Guillen, M., Gimeno, J.-P., Salzet, M., Lebonvallet, N., Misery, L., Auxenfans, C., & Nataf, S. (2020). Molecular Mapping of Hydrogen Sulfide Targets in Normal Human Keratinocytes. International Journal of Molecular Sciences, 21(13), 4648. https://doi.org/10.3390/ijms21134648






