Plasma Extracellular Vesicle-Derived TIMP-1 mRNA as a Prognostic Biomarker in Clear Cell Renal Cell Carcinoma: A Pilot Study
Abstract
1. Introduction
2. Results
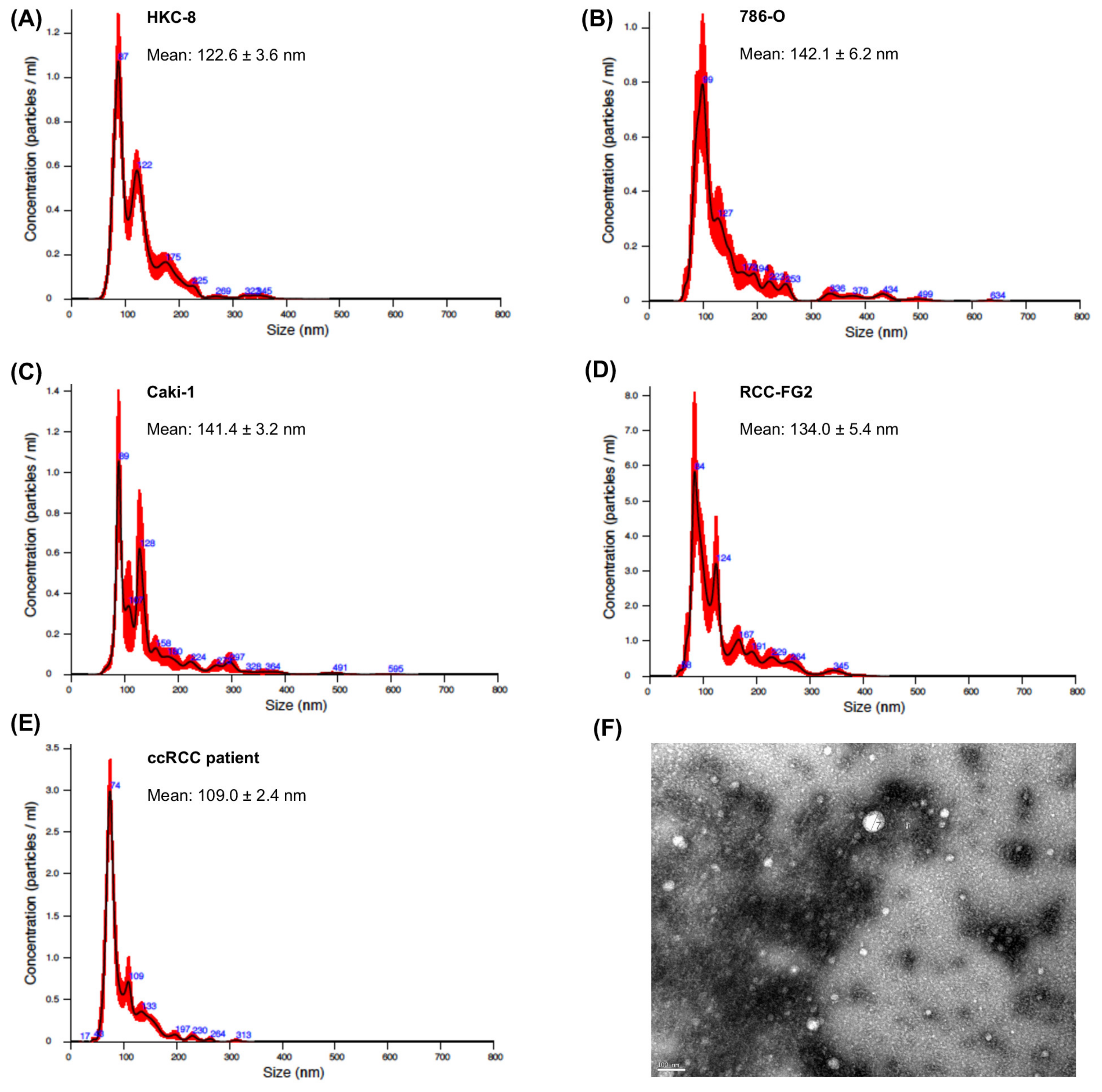
2.1. EVs Characterization
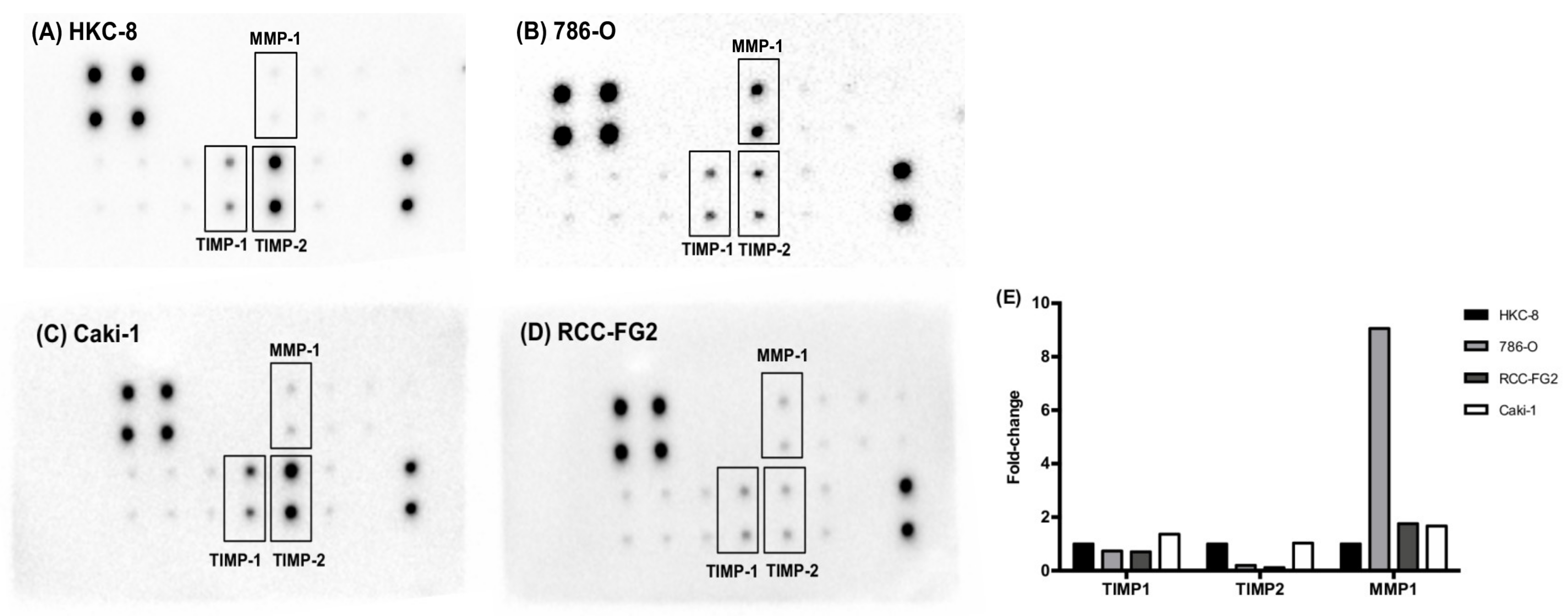
2.2. MMP-1, TIMP-1 and TIMP-2 Present Different Protein Levels among Renal Cell-Derived EVs
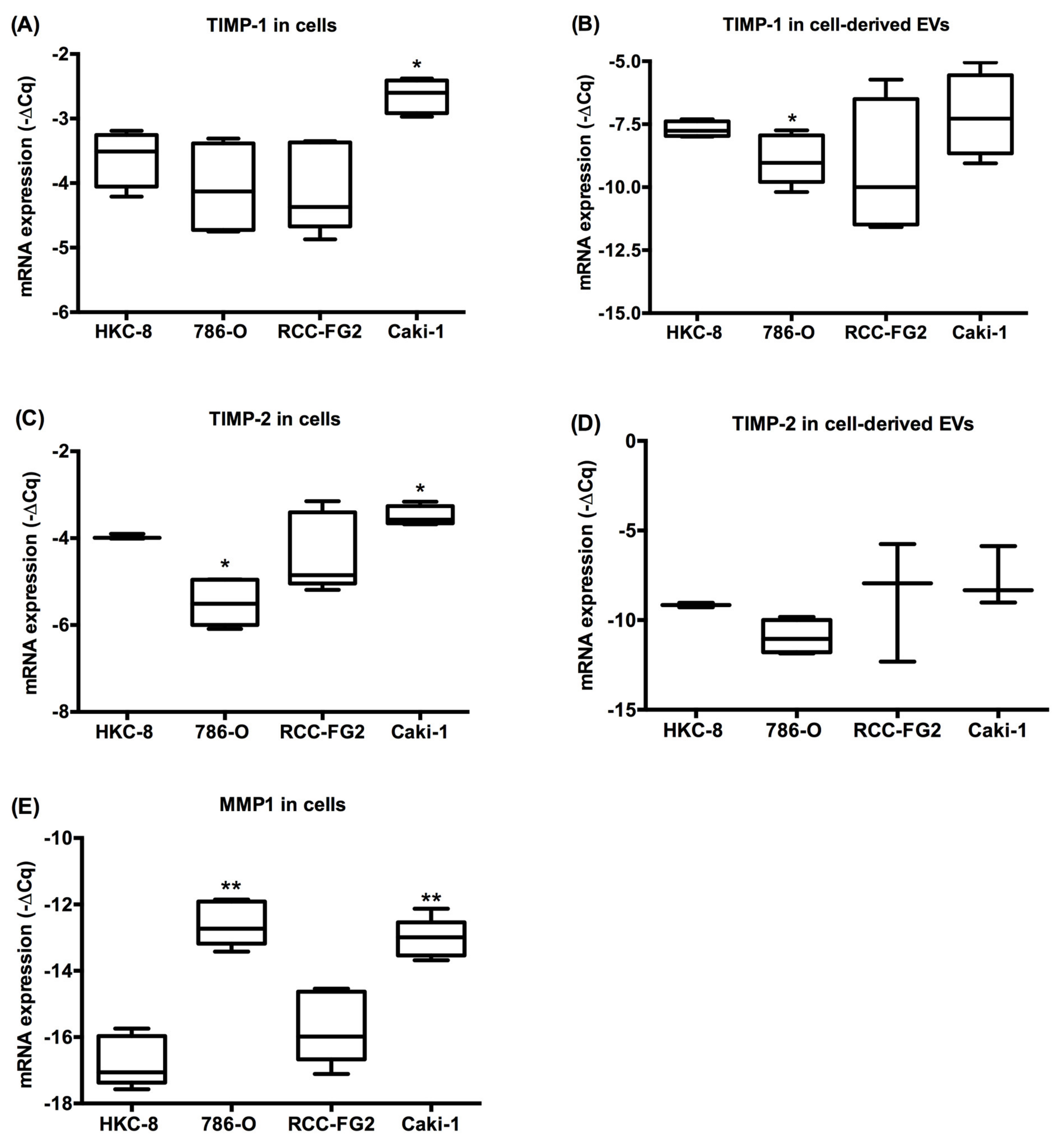
2.3. MMP-1, TIMP-1 and TIMP-2 mRNA Levels in Renal Cell Lines and Cell Line-Derived EVs
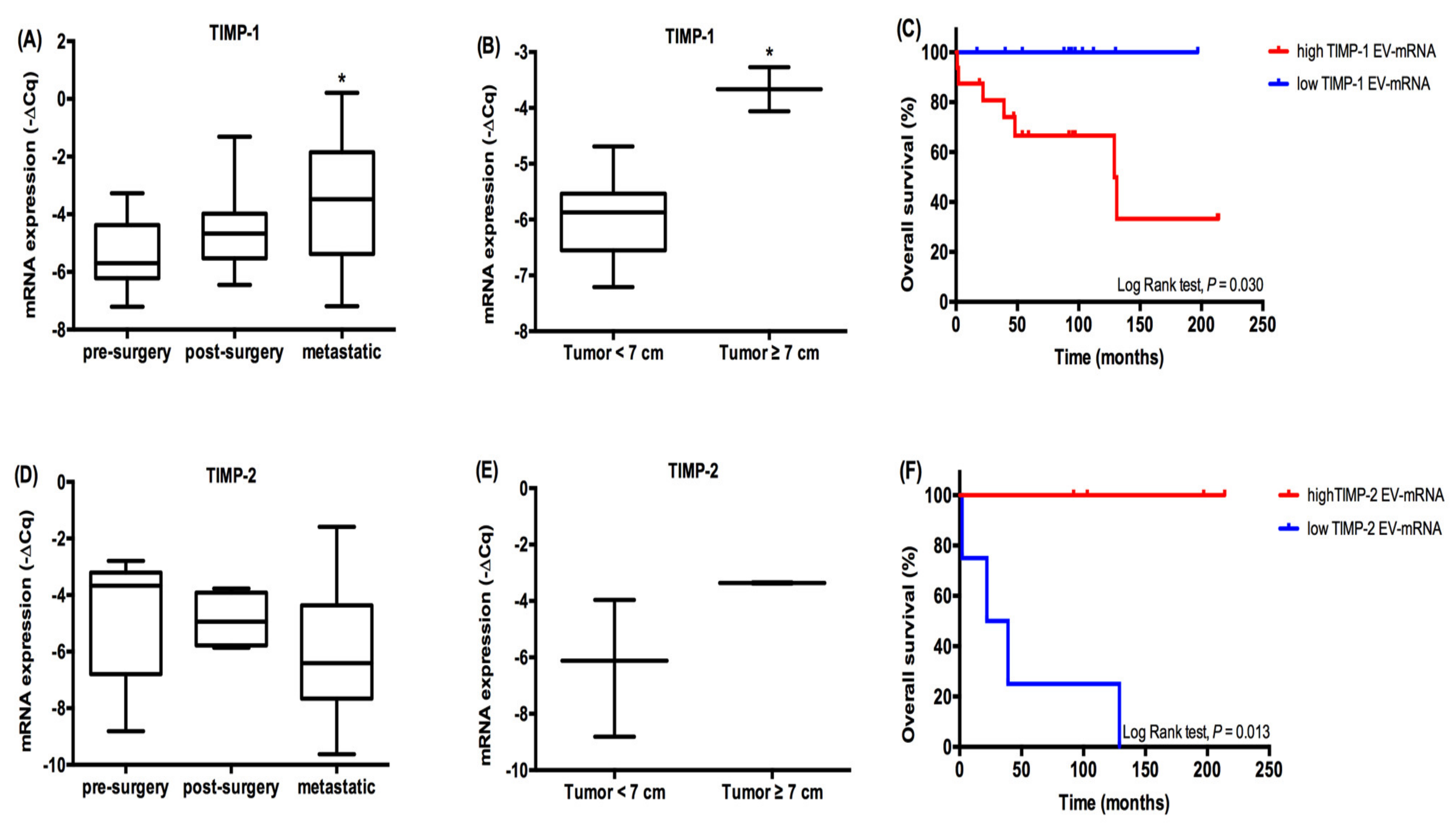
2.4. TIMP-1, TIMP-2 and MMP-1 mRNA Expression in ccRCC Patients’ EVs
2.5. EV-Derived TIMP1 mRNA as a Prognostic Marker in ccRCC Patients: The Example of Two Patients
3. Discussion
4. Materials and Methods
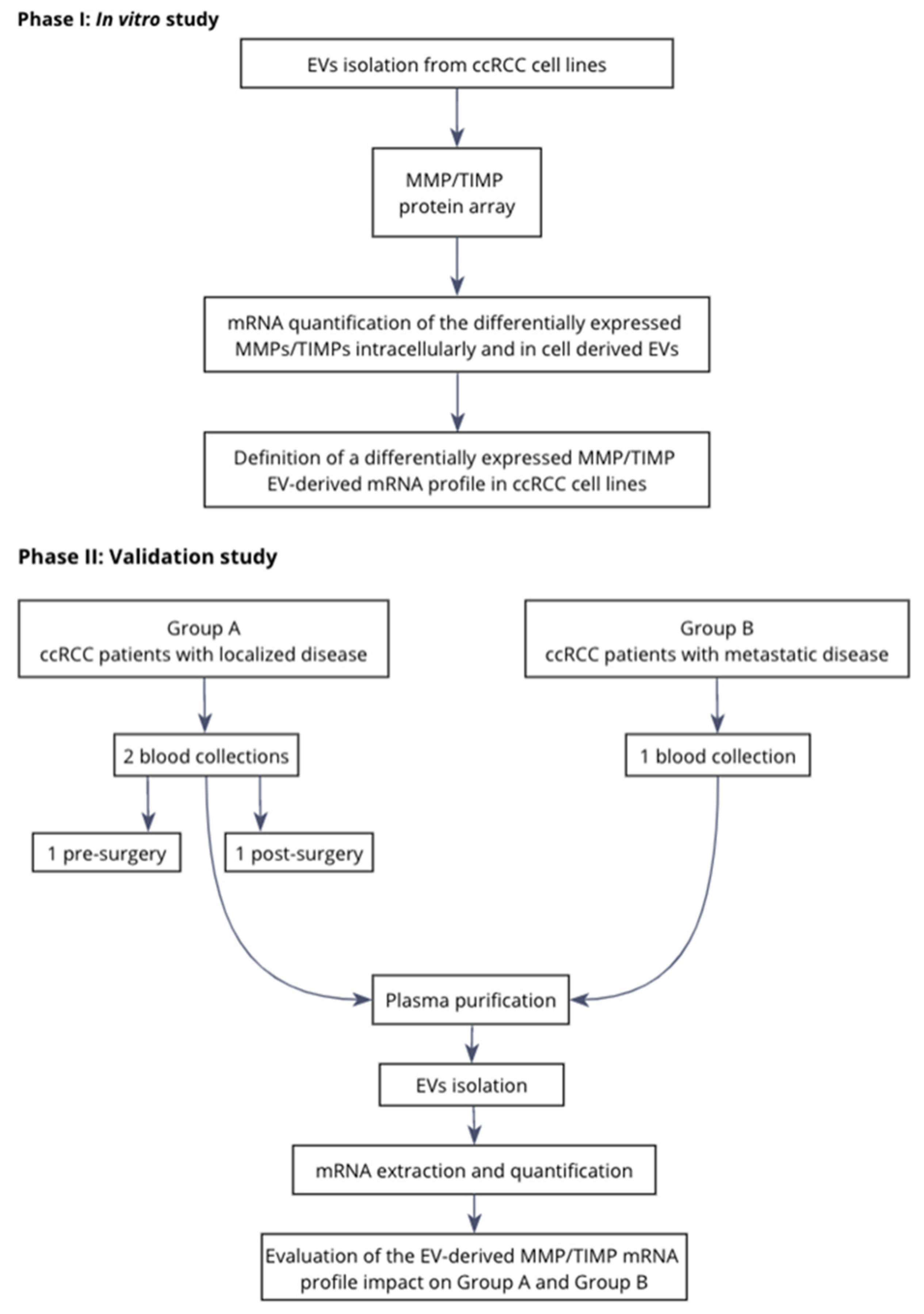
4.1. Study Design
4.2. Ethics Statement
4.3. Study Population
4.4. EVs Isolation
4.5. EVs NTA Analysis
4.6. Quantification of Vesicular Structures by EVs Flow Cytometry
4.7. Protein Extraction and Multiplexed MMP Array
4.8. RNA Extraction and cDNA Synthesis
4.9. Quantitative Real Time PCR
4.10. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| ACTB | Actin Beta |
| AJCC | American Joint Committee on Cancer |
| AKT | AKT Serine/Threonine Kinase |
| ccRCC | Clear cell Renal Cell Carcinoma |
| CFSE | Carboxyfluorescein Diacetate Succinimidyl Ester |
| Cq | Quantification cycle |
| EMT | Epithelial Mesenchymal Transition |
| EVs | Extracellular Vesicles |
| GAPDH | Glyceraldehyde 3-phosphate dehydrogenase |
| HIF1 | Hypoxia Inducible Factor 1 |
| ISUP | International Society of Urological Pathology |
| MMP-1 | Matrix metalloproteinase 1 |
| NTA | Nanoparticle Tracking Analysis |
| PFP | Platelet-free-plasma |
| PI3K | Phosphatidylinositol 3-kinase |
| TIMP-1 | Tissue inhibitor of metalloproteinase 1 |
| TIMP-2 | Tissue inhibitor of metalloproteinase 1 |
| TNM | Tumor-node-metastasis |
References
- Hanahan, D.; Weinberg, R.A. Hallmarks of cancer: The next generation. Cell 2011, 144, 646–674. [Google Scholar] [CrossRef] [PubMed]
- Hu, M.; Polyak, K. Microenvironmental regulation of cancer development. Curr. Opin. Genet. Dev. 2008, 18, 27–34. [Google Scholar] [CrossRef]
- Latifkar, A.; Hur, Y.H.; Sanchez, J.C.; Cerione, R.A.; Antonyak, M.A. New insights into extracellular vesicle biogenesis and function. J. Cell Sci. 2019, 132. [Google Scholar] [CrossRef] [PubMed]
- Wortzel, I.; Dror, S.; Kenific, C.M.; Lyden, D. Exosome-Mediated Metastasis: Communication from a Distance. Dev. Cell 2019, 49, 347–360. [Google Scholar] [CrossRef] [PubMed]
- Nogues, L.; Benito-Martin, A.; Hergueta-Redondo, M.; Peinado, H. The influence of tumour-derived extracellular vesicles on local and distal metastatic dissemination. Mol. Aspects Med. 2018, 60, 15–26. [Google Scholar] [CrossRef] [PubMed]
- Nawaz, M.; Shah, N.; Zanetti, B.R.; Maugeri, M.; Silvestre, R.N.; Fatima, F.; Neder, L.; Valadi, H. Extracellular Vesicles and Matrix Remodeling Enzymes: The Emerging Roles in Extracellular Matrix Remodeling, Progression of Diseases and Tissue Repair. Cells 2018, 7, 167. [Google Scholar] [CrossRef] [PubMed]
- You, Y.; Shan, Y.; Chen, J.; Yue, H.; You, B.; Shi, S.; Li, X.; Cao, X. Matrix metalloproteinase 13-containing exosomes promote nasopharyngeal carcinoma metastasis. Cancer Sci. 2015, 106, 1669–1677. [Google Scholar] [CrossRef]
- Han, K.-Y. Evidence for the Involvement of MMP14 in MMP2 Processing and Recruitment in Exosomes of Corneal Fibroblasts. Invest. Ophthalmol. Vis. Sci. 2015, 56, 5323–5329. [Google Scholar] [CrossRef]
- Yokoi, A.; Yoshioka, Y.; Yamamoto, Y.; Ishikawa, M.; Ikeda, S.-I.; Kato, T.; Kiyono, T.; Takeshita, F.; Kajiyama, H.; Kikkawa, F.; et al. Malignant extracellular vesicles carrying MMP1 mRNA facilitate peritoneal dissemination in ovarian cancer. Nat. Commun. 2017, 8, 14470. [Google Scholar] [CrossRef]
- Peinado, H.; Zhang, H.; Matei, I.R.; Costa-Silva, B.; Hoshino, A.; Rodrigues, G.; Psaila, B.; Kaplan, R.N.; Bromberg, J.F.; Kang, Y.; et al. Pre-metastatic niches: Organ-specific homes for metastases. Nat. Rev. Cancer 2017, 17, 302–317. [Google Scholar] [CrossRef]
- Shimoda, M.; Khokha, R. Metalloproteinases in extracellular vesicles. Biochim. Biophys. Acta Mol. Cell Res. 2017, 1864, 1989–2000. [Google Scholar] [CrossRef] [PubMed]
- Jackson, H.W.; Defamie, V.; Waterhouse, P.; Khokha, R. TIMPs: Versatile extracellular regulators in cancer. Nat. Rev. Cancer 2017, 17, 38–53. [Google Scholar] [CrossRef] [PubMed]
- Salimi Sartakhti, J.; Manshaei, M.H.; Sadeghi, M. MMP-TIMP interactions in cancer invasion: An evolutionary game-theoretical framework. J. Theor. Biol. 2017, 412, 17–26. [Google Scholar] [CrossRef] [PubMed]
- Shimoda, M. Extracellular vesicle-associated MMPs: A modulator of the tissue microenvironment. Adv. Clin. Chem. 2019, 88, 35–66. [Google Scholar] [PubMed]
- Capitanio, U.; Bensalah, K.; Bex, A.; Boorjian, S.A.; Bray, F.; Coleman, J.; Gore, J.L.; Sun, M.; Wood, C.; Russo, P. Epidemiology of Renal Cell Carcinoma. Eur. Urol. 2019, 75, 74–84. [Google Scholar] [CrossRef]
- Escudier, B.; Porta, C.; Schmidinger, M.; Rioux-Leclercq, N.; Bex, A.; Khoo, V.; Grunwald, V.; Gillessen, S.; Horwich, A. Renal cell carcinoma: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-updagger. Ann. Oncol. 2019, 30, 706–720. [Google Scholar] [CrossRef]
- Dabestani, S.; Thorstenson, A.; Lindblad, P.; Harmenberg, U.; Ljungberg, B.; Lundstam, S. Renal cell carcinoma recurrences and metastases in primary non-metastatic patients: A population-based study. World J. Urol. 2016, 34, 1081–1086. [Google Scholar] [CrossRef]
- Gill, D.M.; Agarwal, N.; Vaishampayan, U. Evolving Treatment Paradigm in Metastatic Renal Cell Carcinoma. Am. Soc. Clin. Oncol. Educ. Book 2017, 37, 319–329. [Google Scholar] [CrossRef]
- Morales-Kastresana, A.; Telford, B.; Musich, T.A.; McKinnon, K.; Clayborne, C.; Braig, Z.; Rosner, A.; Demberg, T.; Watson, D.C.; Karpova, T.S.; et al. Labeling Extracellular Vesicles for Nanoscale Flow Cytometry. Sci. Rep. 2017, 7, 1878. [Google Scholar] [CrossRef]
- Pospichalova, V.; Svoboda, J.; Dave, Z.; Kotrbova, A.; Kaiser, K.; Klemova, D.; Ilkovics, L.; Hampl, A.; Crha, I.; Jandakova, E.; et al. Simplified protocol for flow cytometry analysis of fluorescently labeled exosomes and microvesicles using dedicated flow cytometer. J. Extracell. Vesicles 2015, 4, 25530. [Google Scholar] [CrossRef]
- Mastoridis, S.; Bertolino, G.M.; Whitehouse, G.; Dazzi, F.; Sanchez-Fueyo, A.; Martinez-Llordella, M. Multiparametric Analysis of Circulating Exosomes and Other Small Extracellular Vesicles by Advanced Imaging Flow Cytometry. Front. Immunol. 2018, 9, 1583. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Zhang, Y.; Liu, M.; Wang, Y.; Yang, T.; Li, D.; Ding, F.; Bai, G.; Li, Q. TIMP2 is a Poor Prognostic Factor and Predicts Metastatic Biological Behavior in Gastric Cancer. Sci. Rep. 2018, 8, 9629. [Google Scholar] [CrossRef] [PubMed]
- Reis, S.T.; Antunes, A.A.; Pontes-Junior, J.; de Sousa-Canavez, J.M.; Dall’Oglio, M.F.; Piantino, C.B.; Cruz, J.A.S.D.; Morais, D.R.; Srougi, M.; Leite, K.R.M. Underexpression of MMP-2 and its regulators, TIMP2, MT1-MMP and IL-8, is associated with prostate cancer. Int. Braz. J. Urol. 2012, 38, 167–174. [Google Scholar] [CrossRef] [PubMed]
- Kai, A.K.-L.; Chan, L.K.; Lo, R.C.-L.; Lee, J.M.-F.; Wong, C.C.-L.; Wong, J.C.-M.; Ng, I.O.-L. Down-regulation of TIMP2 by HIF-1alpha/miR-210/HIF-3alpha regulatory feedback circuit enhances cancer metastasis in hepatocellular carcinoma. Hepatology 2016, 64, 473–487. [Google Scholar] [CrossRef] [PubMed]
- D’Costa, Z.; Jones, K.; Azad, A.; van Stiphout, R.; Lim, S.Y.; Gomes, A.L.; Kinchesh, P.; Smart, S.C.; Gillies McKenna, W.; Buffa, F.M.; et al. Gemcitabine-Induced TIMP1 Attenuates Therapy Response and Promotes Tumor Growth and Liver Metastasis in Pancreatic Cancer. Cancer Res. 2017, 77, 5952–5962. [Google Scholar]
- Song, G.; Xu, S.; Zhang, H.; Wang, Y.; Xiao, C.; Jiang, T.; Wu, L.; Zhang, T.; Sun, X.; Zhong, L.; et al. TIMP1 is a prognostic marker for the progression and metastasis of colon cancer through FAK-PI3K/AKT and MAPK pathway. J. Exp. Clin. Cancer Res. 2016, 35, 148. [Google Scholar] [CrossRef]
- Vallabhaneni, K.C.; Penfornis, P.; Dhule, S.; Guillonneau, F.; Adams, K.V.; Mo, Y.Y.; Xu, R.; Liu, Y.; Watabe, K.; Vemuri, M.C.; et al. Extracellular vesicles from bone marrow mesenchymal stem/stromal cells transport tumor regulatory microRNA, proteins, and metabolites. Oncotarget 2015, 6, 4953–4967. [Google Scholar] [CrossRef]
- Skog, J.; Würdinger, T.; van Rijn, S.; Meijer, D.H.; Gainche, L.; Sena-Esteves, M.; Curry, W.T.J.; Carter, B.S.; Krichevsky, A.M.; Breakefield, X.O. Glioblastoma microvesicles transport RNA and proteins that promote tumour growth and provide diagnostic biomarkers. Nat. Cell Biol. 2008, 10, 1470–1476. [Google Scholar] [CrossRef]
- Tan, K.H.; Tan, S.S.; Ng, M.J.; Tey, W.S.; Sim, W.K.; Allen, J.C.; Lim, S.K. Extracellular vesicles yield predictive pre-eclampsia biomarkers. J. Extracell. Vesicles 2017, 6, 1408390. [Google Scholar] [CrossRef]
- Skerenova, M.; Mikulova, V.; Capoun, O.; Zima, T.; Tesarova, P. Circulating tumor cells and serum levels of MMP-2, MMP-9 and VEGF as markers of the metastatic process in patients with high risk of metastatic progression. Biomed. Pap. Med. Fac. Univ. Palacky Olomouc Czech Repub. 2017, 161, 272–280. [Google Scholar] [CrossRef]
- Cai, X.; Zhu, H.; Li, Y. PKCzeta, MMP2 and MMP9 expression in lung adenocarcinoma and association with a metastatic phenotype. Mol. Med. Rep. 2017, 16, 8301–8306. [Google Scholar] [CrossRef] [PubMed]
- Etheridge, A.; Gomes, C.P.C.; Pereira, R.W.; Galas, D.; Wang, K. The complexity, function and applications of RNA in circulation. Front. Genet. 2013, 4, 115. [Google Scholar] [CrossRef] [PubMed]
- Bhome, R.; Goh, R.W.; Bullock, M.D.; Pillar, N.; Thirdborough, S.M.; Mellone, M.; Mirnezami, R.; Galea, D.; Veselkov, K.; Gu, Q.; et al. Exosomal microRNAs derived from colorectal cancer-associated fibroblasts: Role in driving cancer progression. Aging (Albany NY) 2017, 9, 2666–2694. [Google Scholar] [CrossRef] [PubMed]
- Luo, F.; Sun, Z.; Han, Q.; Xue, C.; Bai, C. Effect of Human Hepatocellular Carcinoma HepG2 Cell-derived Exosome on the Differentiation of Mesenchymal Stem Cells and Their Interaction. Zhongguo Yi Xue Ke Xue Yuan Xue Bao 2017, 39, 312–317. [Google Scholar]
- Hu, Y.; Rao, S.-S.; Wang, Z.-X.; Cao, J.; Tan, Y.-J.; Luo, J.; Li, H.-M.; Zhang, W.-S.; Chen, C.-Y.; Xie, H. Exosomes from human umbilical cord blood accelerate cutaneous wound healing through miR-21-3p-mediated promotion of angiogenesis and fibroblast function. Theranostics 2018, 8, 169–184. [Google Scholar] [CrossRef]
- Kahlert, C.; Kalluri, R. Exosomes in tumor microenvironment influence cancer progression and metastasis. J. Mol. Med. (Berl.) 2013, 91, 431–437. [Google Scholar] [CrossRef]
- Lobb, R.J.; Lima, L.G.; Möller, A. Exosomes: Key mediators of metastasis and pre-metastatic niche formation. Semin. Cell Dev. Biol. 2017, 67, 3–10. [Google Scholar] [CrossRef]
- Abdella, A.M.; Attia, G.A.; Eed, M.A.; Eldib, A.S.; Haleem, S.S. Evaluation of matrix metalloproteinase-9 and tissue inhibitor metalloproteinase-1 levels in bronchoalveolar lavage of apparently healthy smokers. Egypt. J. Chest Dis. Tuberc. 2015, 64, 371–378. [Google Scholar] [CrossRef]
- Watson, A.M.; Benton, A.S.; Rose, M.C.; Freishtat, R.J. Cigarette smoke alters tissue inhibitor of metalloproteinase 1 and matrix metalloproteinase 9 levels in the basolateral secretions of human asthmatic bronchial epithelium in vitro. J. Investig. Med. 2010, 58, 725–729. [Google Scholar] [CrossRef]
- Cui, H.; Seubert, B.; Stahl, E.; Dietz, H.; Reuning, U.; Moreno-Leon, L.; Ilie, M.; Hofman, P.; Nagase, H.; Mari, B.; et al. Tissue inhibitor of metalloproteinases-1 induces a pro-tumourigenic increase of miR-210 in lung adenocarcinoma cells and their exosomes. Oncogene 2015, 34, 3640–3650. [Google Scholar] [CrossRef]
- Dias, F.; Teixeira, A.L.; Ferreira, M.; Adem, B.; Bastos, N.; Vieira, J.; Fernandes, M.; Sequeira, M.I.; Maurício, J.; Lobo, F.; et al. Plasmatic miR-210, miR-221 and miR-1233 profile: Potential liquid biopsies candidates for renal cell carcinoma. Oncotarget 2017, 8, 103315–103326. [Google Scholar] [CrossRef] [PubMed]
- Dias, F.; Teixeira, A.L.; Nogueira, I.; Morais, M.; Maia, J.; Bodo, C.; Ferreira, M.; Silva, A.; Vilhena, M.; Lobo, J.; et al. Extracellular Vesicles Enriched in hsa-miR-301a-3p and hsa-miR-1293 Dynamics in Clear Cell Renal Cell Carcinoma Patients: Potential Biomarkers of Metastatic Disease. Cancers 2020, 12, 1450. [Google Scholar] [CrossRef] [PubMed]
- Maiyo, F.; Singh, M. Folate-Targeted mRNA Delivery Using Chitosan-Functionalized Selenium Nanoparticles: Potential in Cancer Immunotherapy. Pharmaceuticals 2019, 12, 164. [Google Scholar] [CrossRef] [PubMed]
- Barry, M.; Pearce, H.; Cross, L.; Tatullo, M.; Gaharwar, A.K. Advances in Nanotechnology for the Treatment of Osteoporosis. Curr. Osteoporos. Rep. 2016, 14, 87–94. [Google Scholar] [CrossRef] [PubMed]
- Ohno, S.-I.; Drummen, G.P.C.; Kuroda, M. Focus on Extracellular Vesicles: Development of Extracellular Vesicle-Based Therapeutic Systems. Int. J. Mol. Sci. 2016, 17, 172. [Google Scholar] [CrossRef] [PubMed]
- Srigley, J.R.; Delahunt, B.; Eble, J.N.; Egevad, L.; Epstein, J.I.; Grignon, D.; Hes, O.; Moch, H.; Montironi, R.; Tickoo, S.K.; et al. The International Society of Urological Pathology (ISUP) Vancouver Classification of Renal Neoplasia. Am. J. Surg. Pathol. 2013, 37, 1469–1489. [Google Scholar] [CrossRef]
- Maia, J.; Batista, S.; Couto, N.; Gregorio, A.; Bodo, C.; Elzanowska, J.; Moraes, M.; Costa-Silva, B. Population Analysis of Extracellular Vesicles in Microvolumes of Biofluids. Unpublished work. 2020. [Google Scholar]
- Zeringer, E.; Li, M.; Barta, T.; Schageman, J.; Pedersen, K.W.; Neurauter, A.; Magdaleno, S.; Setterquist, R.; Vlassov, A.V. Methods for the extraction and RNA profiling of exosomes. World J. Methodol. 2013, 3, 11–18. [Google Scholar] [CrossRef]
- Pfaffl, M.W.; Tichopad, A.; Prgomet, C.; Neuvians, T.P. Determination of stable housekeeping genes, differentially regulated target genes and sample integrity: BestKeeper--Excel-based tool using pair-wise correlations. Biotechnol. Lett. 2004, 26, 509–515. [Google Scholar] [CrossRef]
| A | B | C | D | E | F | G | H | |
|---|---|---|---|---|---|---|---|---|
| 1 | Pos | Pos | Neg | Neg | MMP-1 | MMP-2 | MMP-3 | MMP-8 |
| 2 | Pos | Pos | Neg | Neg | MMP-1 | MMP-2 | MMP-3 | MMP-8 |
| 3 | MMP-9 | MMP-10 | MMP-13 | TIMP-1 | TIMP-2 | TIMP-4 | Neg | Pos |
| 4 | MMP-9 | MMP-10 | MMP-13 | TIMP-1 | TIMP-2 | TIMP-4 | Neg | Pos |
| Gender | Age | Tumor Size | TNM Stage | ISUP | Time to Recurrence | Metastatic Site | |
|---|---|---|---|---|---|---|---|
| Patient A | male | 83 | 6 cm | pT3aNxM0R0 | 4 | 9 months | lung |
| Patient B | male | 74 | 8 cm | pT3cN0M0R0 | 3 | 21 months | bones |
| Group A (n = 32) | Group B (n = 29) | ||||
|---|---|---|---|---|---|
| n | % | n | % | ||
| Gender | |||||
| Male | 24 | 75.0 | 21 | 72.4 | |
| Female | 8 | 25.0 | 8 | 27.6 | |
| Age | Average ± SD | 61.9 ± 12.4 | 62.4 ± 9.9 | ||
| Type of surgery | |||||
| Partial nephrectomy | 18 | 56.3 | 0 | 0 | |
| Radical nephrectomy | 14 | 43.8 | 27 | 93.1 | |
| No surgery | 0 | 0 | 2 | 6.9 | |
| Tumor size | |||||
| <7 cm | 24 | 75.0 | 7 | 24.1 | |
| ≥7 cm | 8 | 25.0 | 18 | 62.1 | |
| Undetermined | 0 | 0 | 4 | 13.8 | |
| T | |||||
| T1 | 17 | 53.1 | 8 | 27.6 | |
| T2 | 1 | 3.1 | 7 | 24.1 | |
| T3 | 13 | 40.6 | 10 | 34.5 | |
| T4 | 0 | 0 | 1 | 3.4 | |
| Tx | 1 | 3.1 | 3 | 10.6 | |
| n | |||||
| N0 | 0 | 0 | 15 | 51.7 | |
| N1-N2 | 0 | 0 | 3 | 10.3 | |
| Nx | 0 | 0 | 9 | 31.0 | |
| M | |||||
| M0 | 32 | 100 | 20 | 68.9 | |
| M1 | 0 | 0 | 7 | 24.1 | |
| Mx | 0 | 0 | 2 | 7.0 | |
| Clinical stage | |||||
| I | 17 | 53.1 | 7 | 24.1 | |
| II | 1 | 3.1 | 6 | 20.7 | |
| III | 13 | 40.6 | 8 | 27.6 | |
| IV | 0 | 0 | 5 | 17.2 | |
| Not available | 1 | 3.1 | 3 | 10.4 | |
| ISUP classification | |||||
| 1 | 2 | 6.3 | 1 | 3.45 | |
| 2 | 18 | 56.3 | 12 | 41.4 | |
| 3 | 11 | 34.3 | 7 | 24,1 | |
| 4 | 1 | 3.1 | 5 | 17.2 | |
| Not available | 0 | 0 | 4 | 13.8 | |
| Smoking status | |||||
| Smoker | 6 | 18.8 | 2 | 6.9 | |
| Ex-smoker | 11 | 34.4 | 4 | 13.8 | |
| Non-smoker | 14 | 43.8 | 23 | 79.3 | |
| Not available | 1 | 3.1 | 0 | 0 | |
| Hypertension | |||||
| Yes | 20 | 62.5 | 14 | 48.3 | |
| No | 11 | 34.4 | 15 | 51.7 | |
| Not available | 1 | 3.1 | 0 | 0 | |
| Diabetes mellitus | |||||
| DM I | 0 | 0 | 0 | 0 | |
| DM II | 9 | 28.1 | 12 | 41.4 | |
| No | 22 | 68.8 | 17 | 58.6 | |
| Not available | 1 | 3.1 | 0 | 0 | |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dias, F.; Teixeira, A.L.; Nogueira, I.; Morais, M.; Maia, J.; Bodo, C.; Ferreira, M.; Vieira, I.; Silva, J.; Lobo, J.; et al. Plasma Extracellular Vesicle-Derived TIMP-1 mRNA as a Prognostic Biomarker in Clear Cell Renal Cell Carcinoma: A Pilot Study. Int. J. Mol. Sci. 2020, 21, 4624. https://doi.org/10.3390/ijms21134624
Dias F, Teixeira AL, Nogueira I, Morais M, Maia J, Bodo C, Ferreira M, Vieira I, Silva J, Lobo J, et al. Plasma Extracellular Vesicle-Derived TIMP-1 mRNA as a Prognostic Biomarker in Clear Cell Renal Cell Carcinoma: A Pilot Study. International Journal of Molecular Sciences. 2020; 21(13):4624. https://doi.org/10.3390/ijms21134624
Chicago/Turabian StyleDias, Francisca, Ana Luísa Teixeira, Inês Nogueira, Mariana Morais, Joana Maia, Cristian Bodo, Marta Ferreira, Isabel Vieira, José Silva, João Lobo, and et al. 2020. "Plasma Extracellular Vesicle-Derived TIMP-1 mRNA as a Prognostic Biomarker in Clear Cell Renal Cell Carcinoma: A Pilot Study" International Journal of Molecular Sciences 21, no. 13: 4624. https://doi.org/10.3390/ijms21134624
APA StyleDias, F., Teixeira, A. L., Nogueira, I., Morais, M., Maia, J., Bodo, C., Ferreira, M., Vieira, I., Silva, J., Lobo, J., Sequeira, J. P., Maurício, J., Oliveira, J., Palmeira, C., Martins, G., Kok, K., Costa-Silva, B., & Medeiros, R. (2020). Plasma Extracellular Vesicle-Derived TIMP-1 mRNA as a Prognostic Biomarker in Clear Cell Renal Cell Carcinoma: A Pilot Study. International Journal of Molecular Sciences, 21(13), 4624. https://doi.org/10.3390/ijms21134624











