Protocatechuic Aldehyde Attenuates UVA-induced Photoaging in Human Dermal Fibroblast Cells by Suppressing MAPKs/AP-1 and NF-κB Signaling Pathways
Abstract
1. Introduction
2. Results
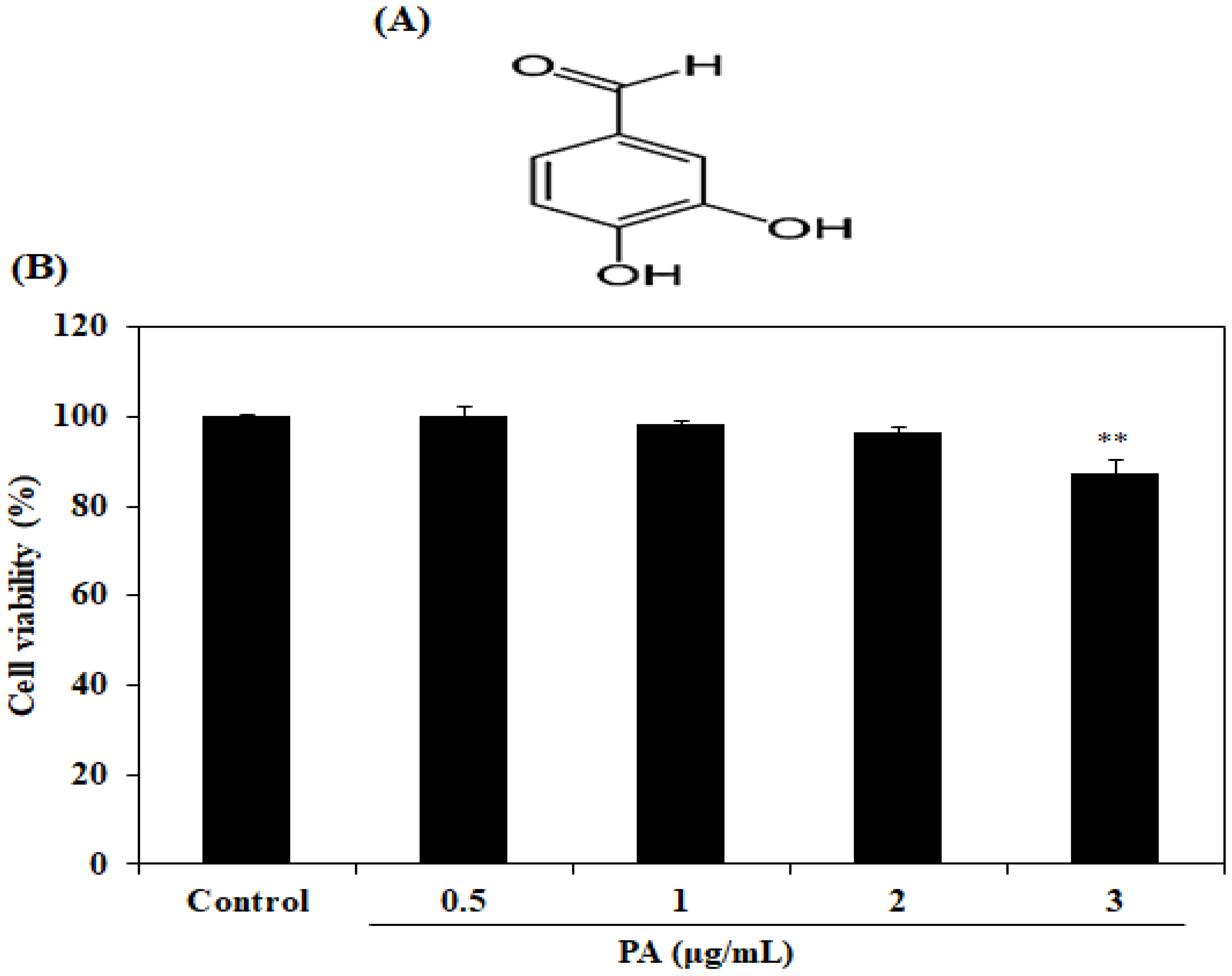
2.1. Cytotoxicity Effect of PA in HDF Cells
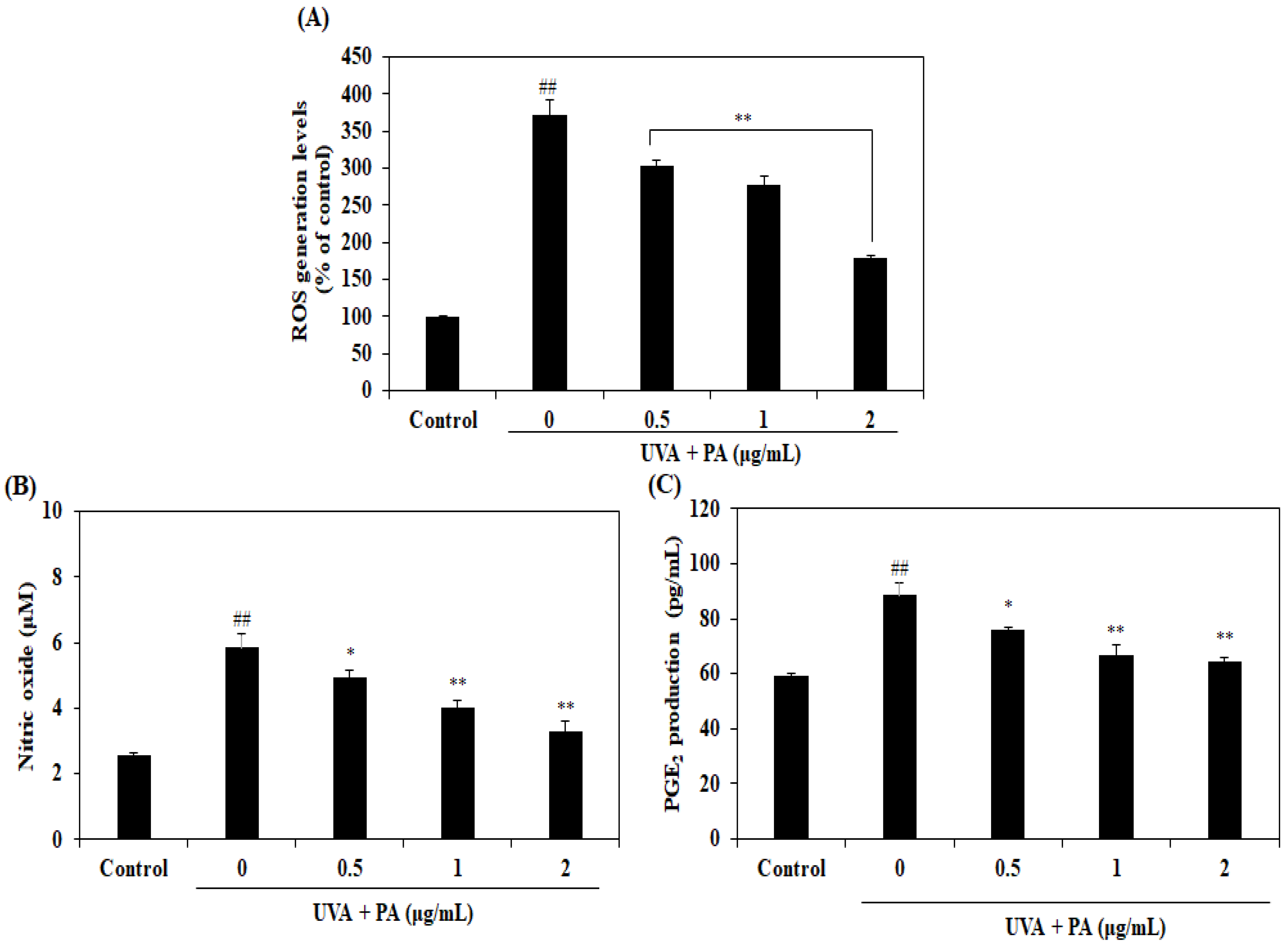
2.2. Effect of PA on Intracellular ROS and Pro-Inflammatory Mediators NO and PGE2 Production in UVA-Induced HDF Cells
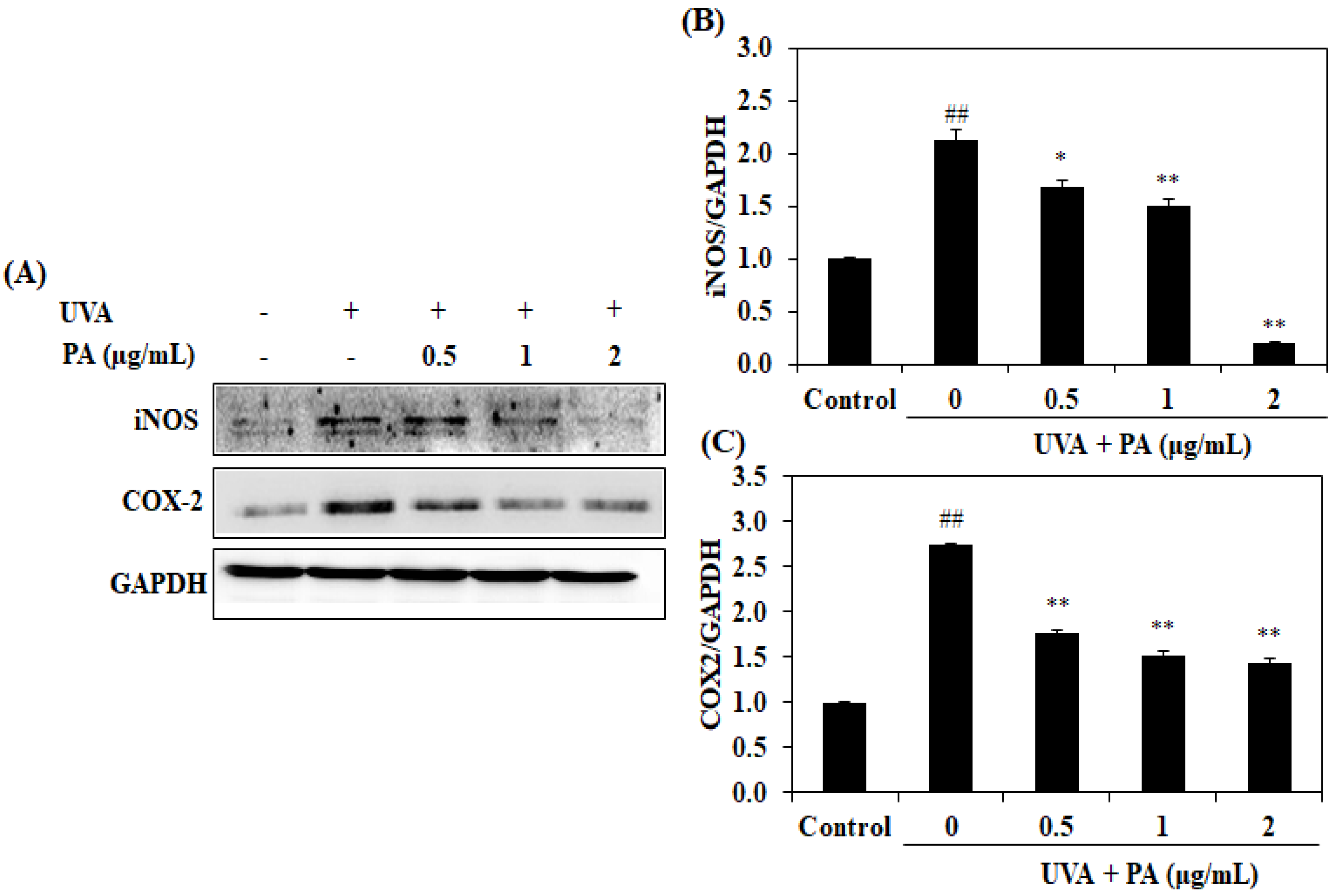
2.3. Effect of PA on COX-2 and iNOS Expression in UVA-Induced HDF Cells
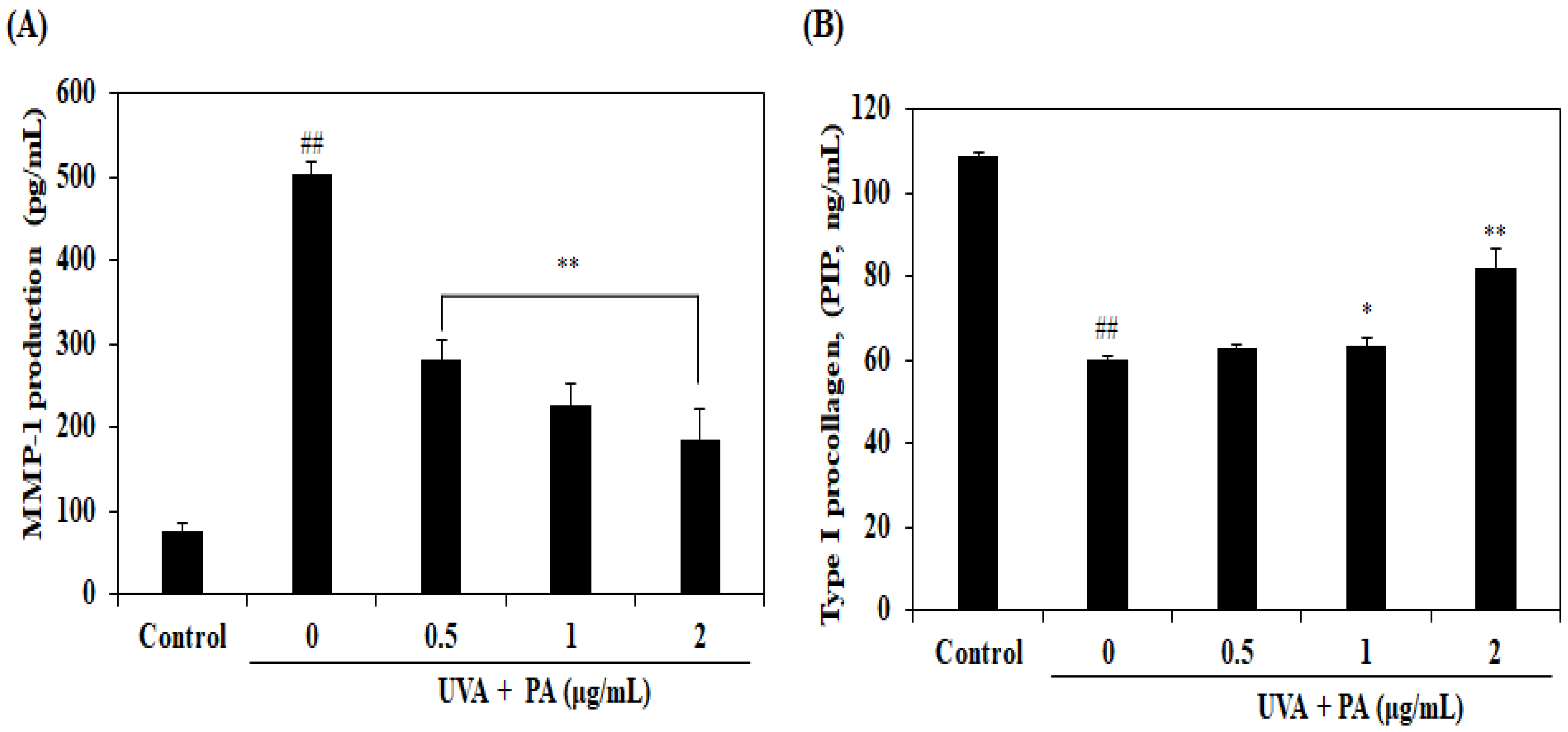
2.4. Effects of PA on MMP-1 Expression Level and Collagen Synthesis in UVA-Induced HDF Cells
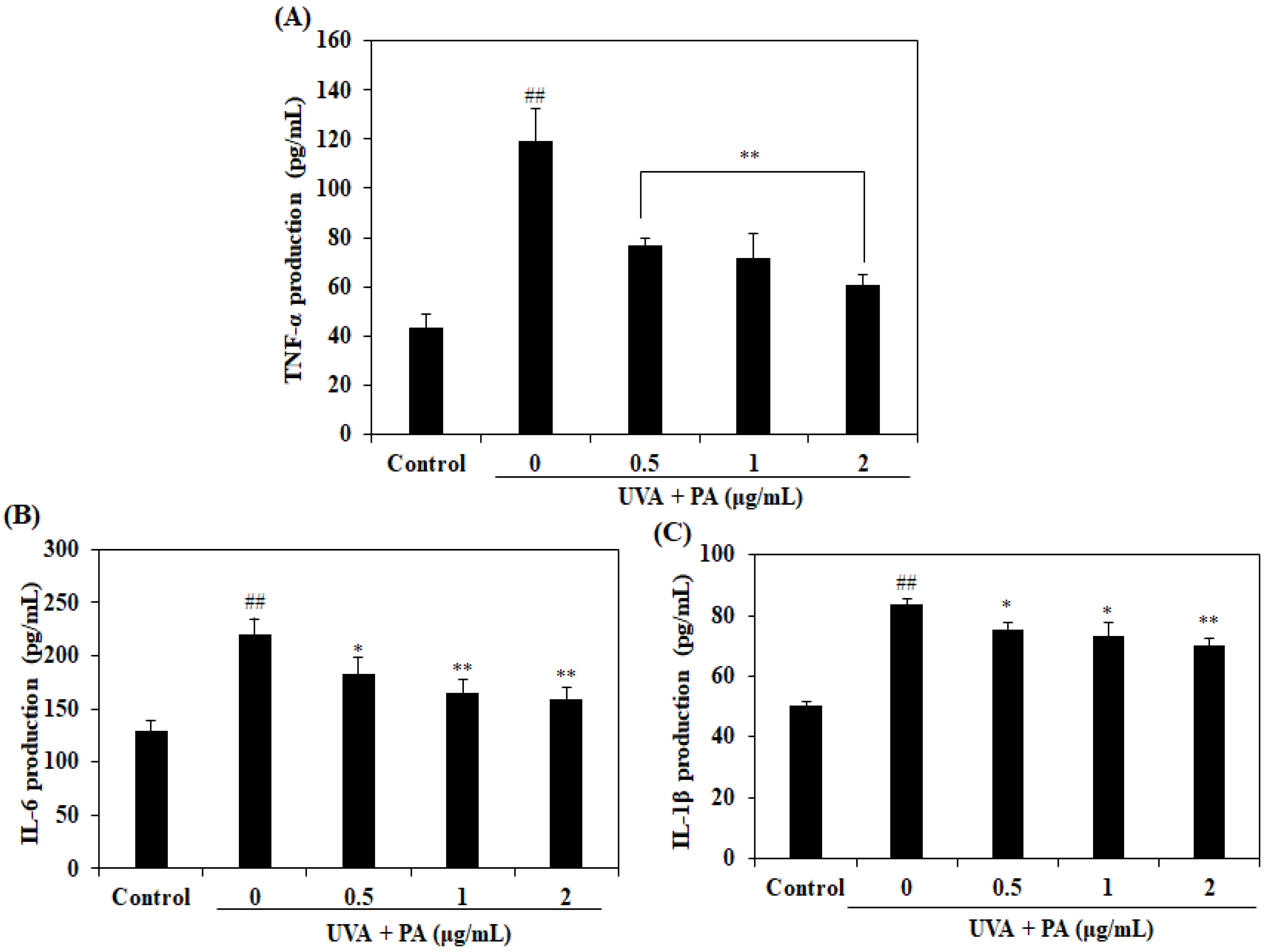
2.5. Effect of PA on Pro-Inflammatory Cytokines Levels in UVA-Induced HDF Cells
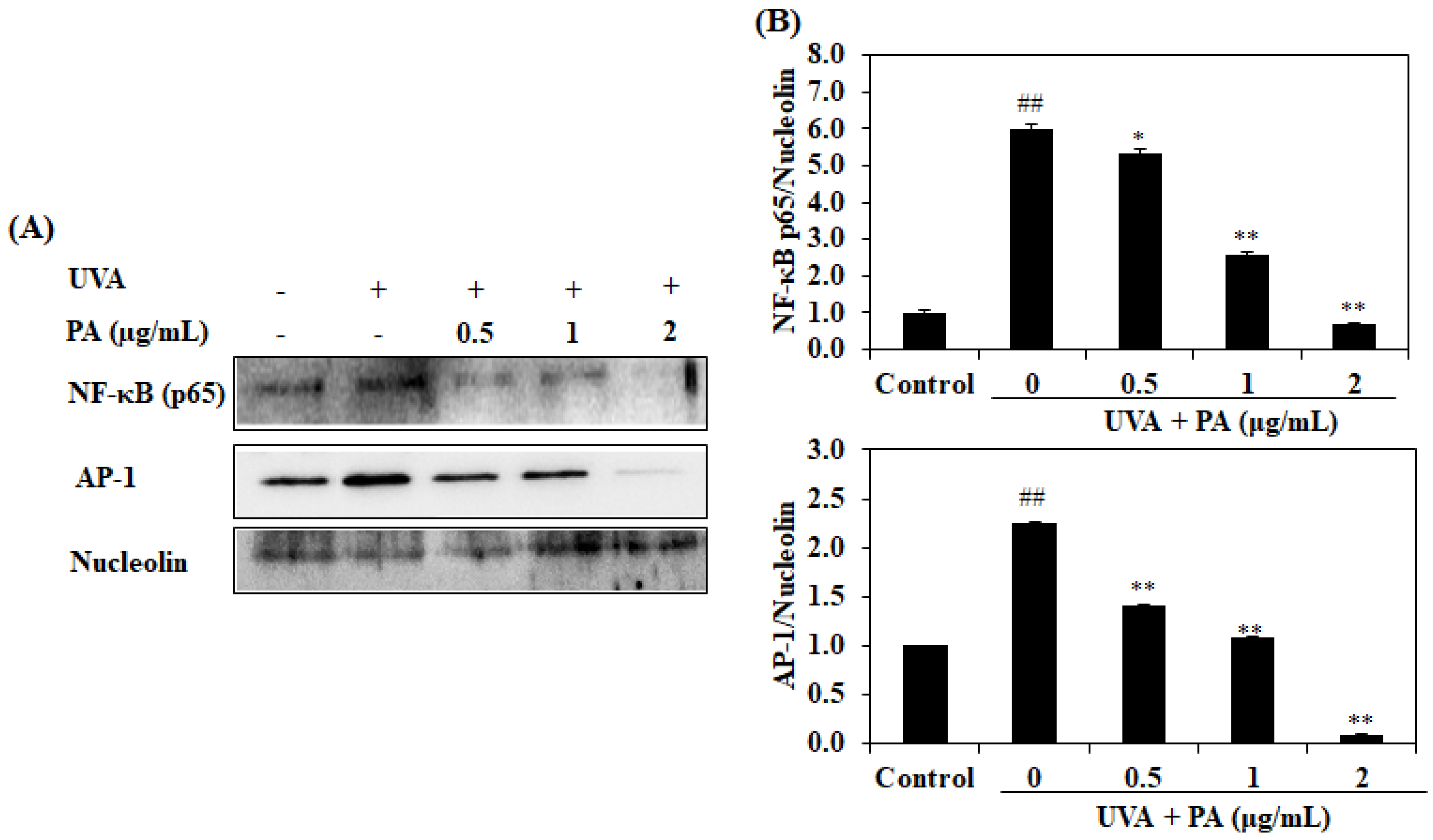
2.6. Effect of PA on AP-1 and NF-κB Expression in UVA-Induced HDF Cells
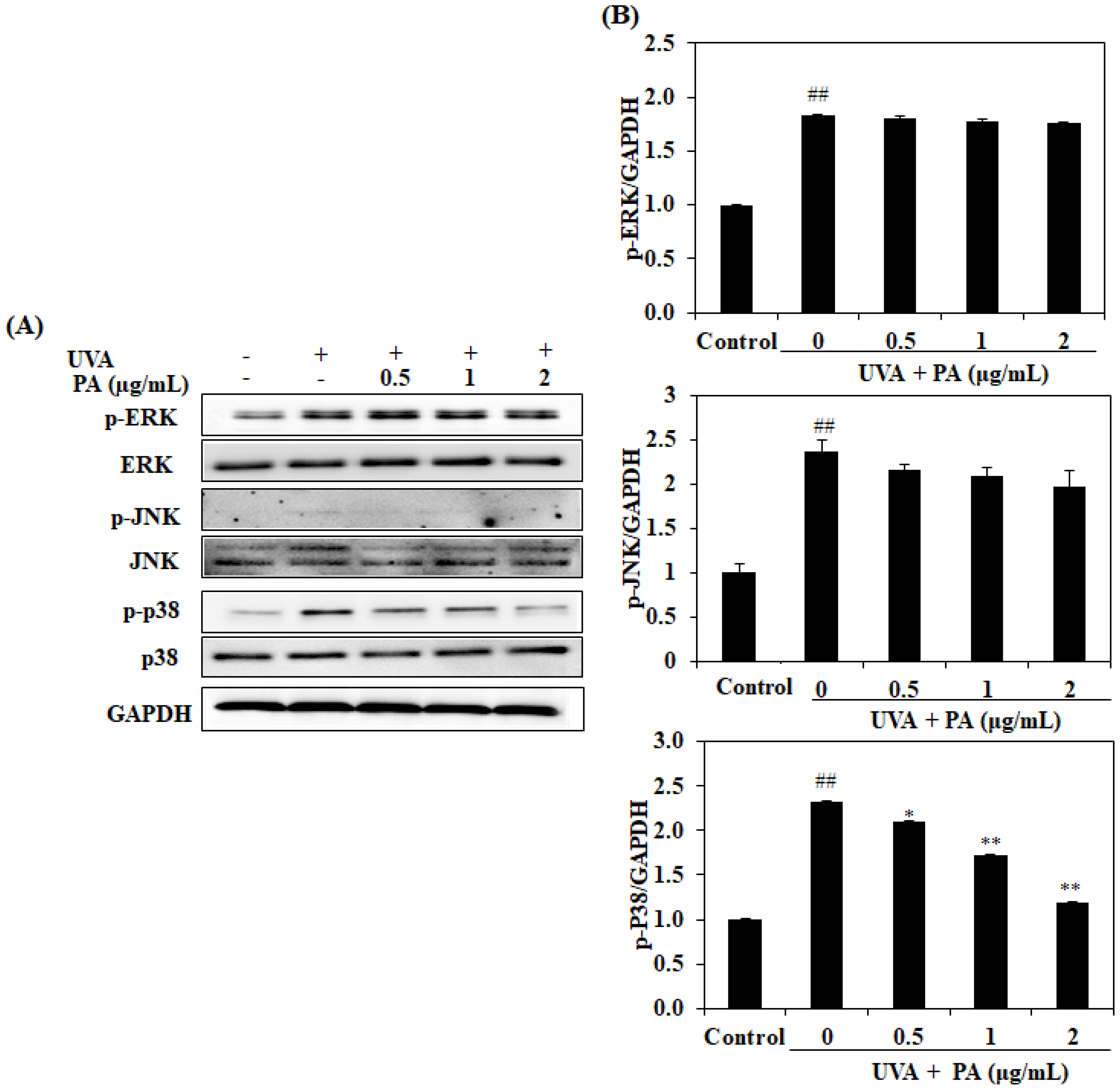
2.7. Effect of PA on Mitogen-Activated Protein Kinase (MAPK) Expression in UVA-Induced HDF Cells
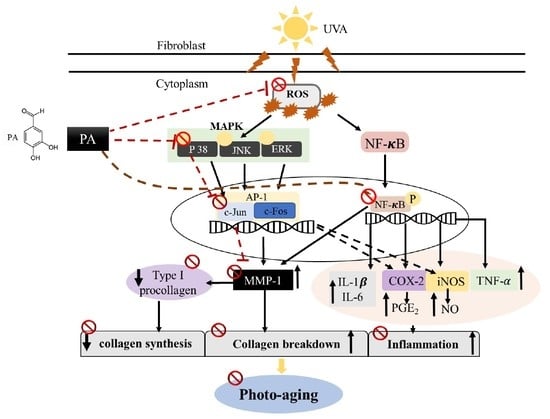
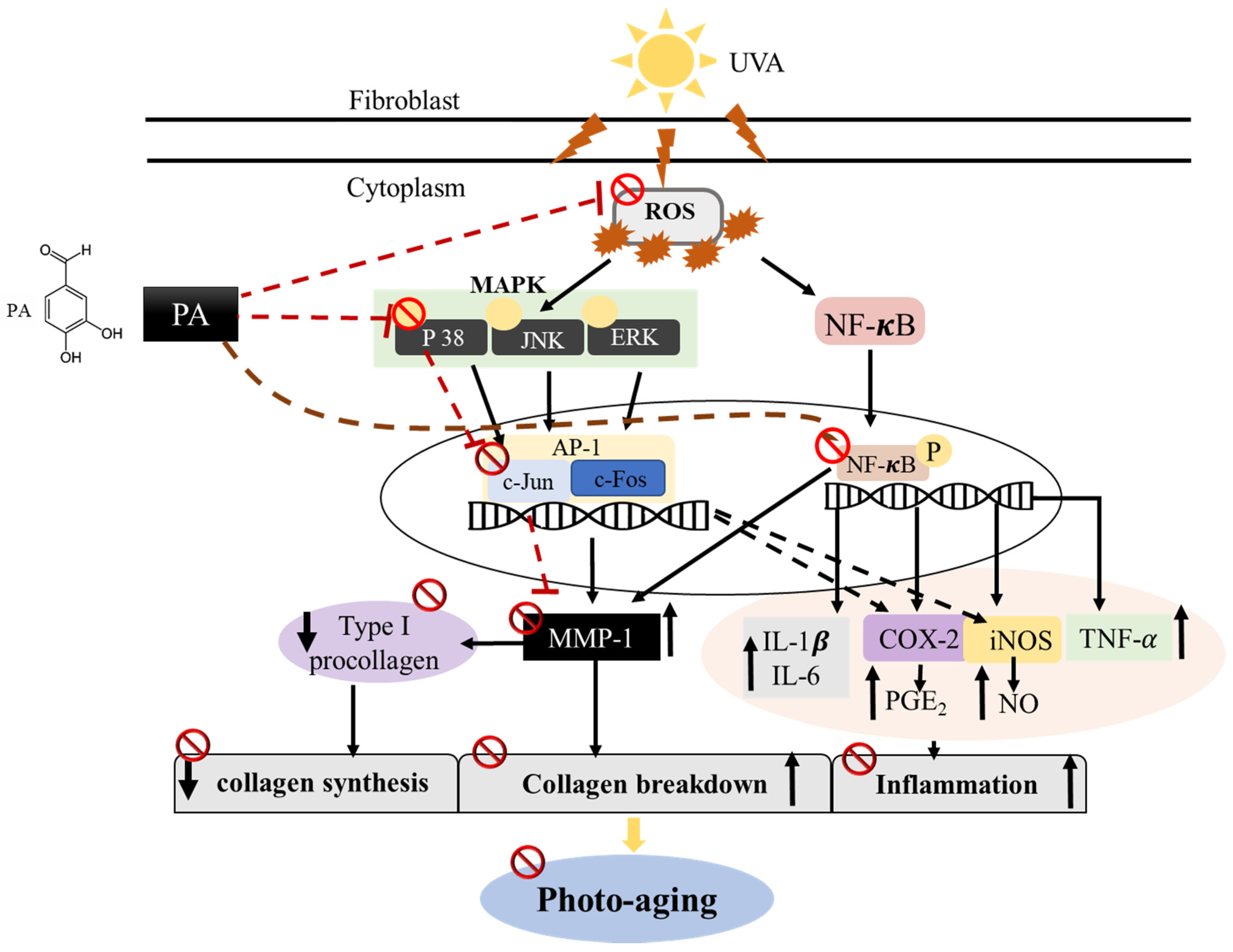
3. Discussion
4. Materials and Methods
4.1. Materials
4.2. Cell Culture and UVA Irradiation
4.3. Measurement of Cytotoxicity
4.4. Determination of Intracellular ROS Levels
4.5. Determination of Nitric Oxide (NO) Production
4.6. Enzyme-Linked Immunosorbent Assay (ELISA) Determination of MMP-1 and Type I Procollagen Production
4.7. Western Blot Analysis
4.8. Statistical Analysis
Author Contributions
Funding
Conflicts of Interest
References
- Adil, M.D.; Kaiser, P.; Satti, N.K.; Zargar, M.A.; Vishwakarma, R.A.; Tasduq, S.A. Effect of Emblica officinalis (fruit) against UVB-induced photo-aging in human skin fibroblasts. J. Ethnopharmacol. 2010, 132, 109–114. [Google Scholar] [CrossRef] [PubMed]
- Papakonstantinou, E.; Roth, M.; Karakiulakis, G. Hyaluronic acid: A key molecule in skin aging. Dermato-Endocrinology 2012, 4, 253–258. [Google Scholar] [CrossRef]
- Wang, L.; Lee, W.; Oh, J.Y.; Cui, Y.R.; Ryu, B.; Jeon, Y.-J. Protective Effect of Sulfated Polysaccharides from Celluclast-Assisted Extract of Hizikia fusiforme Against Ultraviolet B-Induced Skin Damage by Regulating NF-κB, AP-1, and MAPKs Signaling Pathways In Vitro in Human Dermal Fibroblasts. Mar. Drugs 2018, 16, 239. [Google Scholar] [CrossRef]
- Wang, X.; Hong, H.; Wu, J. Hen collagen hydrolysate alleviates UVA-induced damage in human dermal fibroblasts. J. Funct. Foods 2019, 63, 103574. [Google Scholar] [CrossRef]
- Jeon, S.; Choi, M. Anti-inflammatory and anti-aging effects of hydroxytyrosol on human dermal fibroblasts (HDFs). Biomed. Dermatol. 2018, 2, 21. [Google Scholar] [CrossRef]
- Anderson, A.; Bowman, A.; Boulton, S.J.; Manning, P.; Birch-Machin, M.A. A role for human mitochondrial complex II in the production of reactive oxygen species in human skin. Redox Biol. 2014, 2, 1016–1022. [Google Scholar] [CrossRef]
- Philips, N.; Ding, X.; Kandalai, P.; Marte, I.; Krawczyk, H.; Richardson, R. The Beneficial Regulation of Extracellular Matrix and Heat Shock Proteins, and the Inhibition of Cellular Oxidative Stress Effects and Inflammatory Cytokines by 1α, 25 dihydroxyvitaminD3 in Non-Irradiated and Ultraviolet Radiated Dermal Fibroblasts. Cosmetics 2019, 6, 46. [Google Scholar] [CrossRef]
- Lee, B.-C.; Lee, S.Y.; Lee, H.J.; Sim, G.-S.; Kim, J.-H.; Kim, J.-H.; Cho, Y.-H.; Lee, D.-H.; Pyo, H.-B.; Choe, T.-B.; et al. Anti-oxidative and photo-protective effects of coumarins isolated from Fraxinus chinensis. Arch. Pharmacal Res. 2007, 30, 1293–1301. [Google Scholar] [CrossRef]
- Bickers, D.R.; Athar, M. Oxidative Stress in the Pathogenesis of Skin Disease. J. Investig. Dermatol. 2006, 126, 2565–2575. [Google Scholar] [CrossRef] [PubMed]
- Franceschi, C.; Campisi, J. Chronic Inflammation (Inflammaging) and Its Potential Contribution to Age-Associated Diseases. J. Gerontol. Ser. A Biomed. Sci. Med. Sci. 2014, 69, S4–S9. [Google Scholar] [CrossRef] [PubMed]
- Fuller, B. Role of PGE-2 and Other Inflammatory Mediators in Skin Aging and Their Inhibition by Topical Natural Anti-Inflammatories. Cosmetics 2019, 6, 6. [Google Scholar] [CrossRef]
- Liu, Y.; Xie, P.; Wang, B. Evaluation of radix Salviae Miltiorrhizae and its preparation. China J. Chin. Mater. Medica 1990, 15, 159–162. [Google Scholar]
- Weber, B.; Hoesch, L.; Rast, D.M. Protocatechualdehyde and other phenols as cell wall components of grapevine leaves. Phytochemistry 1995, 40, 433–437. [Google Scholar] [CrossRef]
- Kim, T.W.; Kim, T.H. Pancreatic lipase inhibitors in the roots of Taraxacum ohwianum, a herb used in Korean traditional medicine. Korean J. Food Preserv. 2011, 18, 53–58. [Google Scholar] [CrossRef]
- Wei, G.; Guan, Y.; Yin, Y.; Duan, J.; Zhou, D.; Zhu, Y.; Quan, W.; Xi, M.; Wen, A. Anti-inflammatory Effect of Protocatechuic Aldehyde on Myocardial Ischemia/Reperfusion Injury In Vivo and In Vitro. Inflammation 2012, 36, 592–602. [Google Scholar] [CrossRef]
- Kim, K.-J.; Kim, M.-A.; Jung, J.-H. Antitumor and antioxidant activity of protocatechualdehyde produced from Streptomyces lincolnensis M-20. Arch. Pharmacal Res. 2008, 31, 1572–1577. [Google Scholar] [CrossRef]
- Zhou, Z.; Zhang, Y.; Ding, X.-R.; Chen, S.-H.; Yang, J.; Wang, X.; Jia, G.-L.; Chen, H.-S.; Bo, X.-C.; Wang, S. Protocatechuic aldehyde inhibits hepatitis B virus replication both in vitro and in vivo. Antivir. Res. 2007, 74, 59–64. [Google Scholar] [CrossRef]
- Li, S.; Yu, Y.; Chen, J.; Guo, B.; Yang, L.; Ding, W. Evaluation of the Antibacterial Effects and Mechanism of Action of Protocatechualdehyde against Ralstonia solanacearum. Molecules 2016, 21, 754. [Google Scholar] [CrossRef]
- Chou, T.H.; Ding, H.Y.; Lin, R.J.; Liang, J.Y.; Liang, C.H. Inhibition of melanogenesis and oxidation by proticatechuic acid from Origanun vulgare (Oregano). J. Nat. Prod. 2010, 73, 1767–1774. [Google Scholar] [CrossRef]
- Poljsak, B.; Dahmane, R. Free Radicals and Extrinsic Skin Aging. Dermatol. Res. Pr. 2012, 2012, 135206. [Google Scholar] [CrossRef]
- Kashif, M.; Akhtar, N.; Mustafa, R. An overview of dermatological and cosmeceutical benefits of Diospyros kaki and its phytoconstituents. Rev. Bras. de Farm. 2017, 27, 650–662. [Google Scholar] [CrossRef]
- Kerdudo, A.; Burger, P.; Merck, F.; Dingas, A.; Rolland, Y.; Michel, T.; Fernandez, X. Development of a natural ingredient – Natural preservative: A case study. Comptes Rendus Chim. 2016, 19, 1077–1089. [Google Scholar] [CrossRef]
- Bruge, F.; Tiano, L.; Astolfi, P.; Emanuelli, M.; Damiani, E. Prevention of UVA-Induced Oxidative Damage in Human Dermal Fibroblasts by New UV Filters, Assessed Using a Novel In Vitro Experimental System. PLoS ONE 2014, 9, e83401. [Google Scholar] [CrossRef]
- Kuo, Y.-H.; Chen, C.-W.; Chu, Y.; Lin, P.; Chiang, H.-M. In Vitro and In Vivo Studies on Protective Action of N-Phenethyl Caffeamide against Photodamage of Skin. PLoS ONE 2015, 10, e0136777. [Google Scholar] [CrossRef]
- Kageyama, H.; Waditee-Sirisattha, R. Antioxidative, Anti-Inflammatory, and Anti-Aging Properties of Mycosporine-Like Amino Acids: Molecular and Cellular Mechanisms in the Protection of Skin-Aging. Mar. Drugs 2019, 17, 222. [Google Scholar] [CrossRef] [PubMed]
- Xia, W.; Hammerberg, C.; Li, Y.; He, T.; Quan, T.; Voorhees, J.J.; Fisher, G.J. Expression of catalytically active matrix metalloproteinase-1 in dermal fibroblasts induces collagen fragmentation and functional alterations that resemble aged human skin. Aging Cell 2013, 12, 661–671. [Google Scholar] [CrossRef] [PubMed]
- Freitas-Rodríguez, S.; Folgueras, A.; Lopez-Otin, C. The role of matrix metalloproteinases in aging: Tissue remodeling and beyond. Biochim. et Biophys. Acta (BBA)-Bioenerg. 2017, 1864, 2015–2025. [Google Scholar] [CrossRef]
- Jung, Y.R.; Kim, D.H.; Kim, S.R.; An, H.J.; Lee, E.K.; Tanaka, T.; Kim, N.D.; Yokozawa, T.; Park, J.N.; Chung, H.Y. Anti-Wrinkle Effect of Magnesium Lithospermate B from Salvia miltiorrhiza BUNGE: Inhibition of MMPs via NF-kB Signaling. PLoS ONE 2014, 9, e102689. [Google Scholar] [CrossRef]
- Quan, T.; Qin, Z.; Xia, W.; Shao, Y.; Voorhees, J.J.; Fisher, G.J. Matrix-Degrading Metalloproteinases in Photoaging. J. Investig. Dermatol. Symp. Proc. 2009, 14, 20–24. [Google Scholar] [CrossRef]
- Wlaschek, M.; Heinen, G.; Poswig, A.; Schwarz, A.; Krieg, T.; Scharffetter-Kochanek, K. Uva-Induced Autocrine Stimulation of Fibroblast-Derived Collagenase/Mmp-1 by Interrelated Loops Ofinterleukin–1 Andinterleukin–6. Photochem. Photobiol. 1994, 59, 550–556. [Google Scholar] [CrossRef]
- Maes, M. The cytokine hypothesis of depression: Inflammation, oxidative & nitrosative stress (IO&NS) and leaky gut as new targets for adjunctive treatments in depression. Neuro Endocrinol. Lett. 2008, 29, 287–291. [Google Scholar] [PubMed]
- Lee, Y.R.; Noh, E.M.; Han, J.H.; Kim, J.M.; Hwang, J.K.; Hwang, B.M.; Chung, E.Y.; Kim, B.S.; Lee, S.H.; Lee, S.J.; et al. Brazilin inhibits UVB-induced MMP-1/3 expressions and secretions by suppressing the NF-κB pathway in human dermal fibroblasts. Eur. J. Pharmacol. 2012, 674, 80–86. [Google Scholar] [CrossRef] [PubMed]
- Li, X.-N.; Su, J.; Zhao, L.; Xiang, J.-B.; Wang, W.; Liu, F.; Li, H.-Y.; Zhong, J.-T.; Bai, X.; Sun, L.-K. The p38 MAPK inhibitor JLU1124 inhibits the inflammatory response induced by lipopolysaccharide through the MAPK-NF-κB pathway in RAW264.7 macrophages. Int. Immunopharmacol. 2013, 17, 785–792. [Google Scholar] [CrossRef] [PubMed]
- Pittayapruek, P.; Meephansan, J.; Prapapan, O.; Komine, M.; Ohtsuki, M. Role of Matrix Metalloproteinases in Photoaging and Photocarcinogenesis. Int. J. Mol. Sci. 2016, 17, 868. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Huang, S.; Li, Y.; Li, Y.; Li, D.; Wu, P.; Wang, Q.; Zheng, X.; Zhang, K. Glycyrrhizic acid from licorice down-regulates inflammatory responses via blocking MAPK and PI3K/Akt-dependent NF-κB signalling pathways in TPA-induced skin inflammation. MedChemComm 2018, 9, 1502–1510. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.-H.; Li, H.-J.; Wu, N.-L.; Hsiao, C.-Y.; Lin, C.-N.; Chang, H.-H.; Hung, C.-F. Photoprotective Effects of Cycloheterophyllin against UVA-Induced Damage and Oxidative Stress in Human Dermal Fibroblasts. PLoS ONE 2016, 11, e0161767. [Google Scholar] [CrossRef]
- Bell, S.; Degitz, K.; Quirling, M.; Jilg, N.; Page, S.; Brand, K. Involvement of NF-κB signalling in skin physiology and disease. Cell Signal. 2003, 15, 1–7. [Google Scholar] [CrossRef]
- Hwang, B.-M.; Noh, E.-M.; Kim, J.-S.; Kim, J.-M.; Hwang, J.-K.; Kim, H.-K.; Kang, J.; Kim, D.-S.; Chae, H.-J.; You, Y.-O.; et al. Decursin inhibits UVB-induced MMP expression in human dermal fibroblasts via regulation of nuclear factor-κB. Int. J. Mol. Med. 2012, 31, 477–483. [Google Scholar] [CrossRef]
- Ding, Y.; Jiratchayamaethasakul, C.; Kim, J.; Kim, E.A.; Heo, S.J.; Lee, S.H. Antioxidant and anti-melanogenic activities of ultrasonic extract from Stichopus japonicas. Asian Pac. J. Trop. Biomed. 2020, 10, 33–41. [Google Scholar]
- Heo, S.-J.; Jeon, Y.-J. Protective effect of fucoxanthin isolated from Sargassum siliquastrum on UV-B induced cell damage. J. Photochem. Photobiol. B Biol. 2009, 95, 101–107. [Google Scholar] [CrossRef]
- Chang, Y.-C.; Li, P.-C.; Chen, B.-C.; Chang, M.-S.; Wang, J.-L.; Chiu, W.-T.; Lin, C.-H. Lipoteichoic acid-induced nitric oxide synthase expression in RAW 264.7 macrophages is mediated by cyclooxygenase-2, prostaglandin E2, protein kinase A, p38 MAPK, and nuclear factor-κB pathways. Cell Signal. 2006, 18, 1235–1243. [Google Scholar] [CrossRef] [PubMed]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ding, Y.; Jiratchayamaethasakul, C.; Lee, S.-H. Protocatechuic Aldehyde Attenuates UVA-induced Photoaging in Human Dermal Fibroblast Cells by Suppressing MAPKs/AP-1 and NF-κB Signaling Pathways. Int. J. Mol. Sci. 2020, 21, 4619. https://doi.org/10.3390/ijms21134619
Ding Y, Jiratchayamaethasakul C, Lee S-H. Protocatechuic Aldehyde Attenuates UVA-induced Photoaging in Human Dermal Fibroblast Cells by Suppressing MAPKs/AP-1 and NF-κB Signaling Pathways. International Journal of Molecular Sciences. 2020; 21(13):4619. https://doi.org/10.3390/ijms21134619
Chicago/Turabian StyleDing, Yuling, Chanipa Jiratchayamaethasakul, and Seung-Hong Lee. 2020. "Protocatechuic Aldehyde Attenuates UVA-induced Photoaging in Human Dermal Fibroblast Cells by Suppressing MAPKs/AP-1 and NF-κB Signaling Pathways" International Journal of Molecular Sciences 21, no. 13: 4619. https://doi.org/10.3390/ijms21134619
APA StyleDing, Y., Jiratchayamaethasakul, C., & Lee, S.-H. (2020). Protocatechuic Aldehyde Attenuates UVA-induced Photoaging in Human Dermal Fibroblast Cells by Suppressing MAPKs/AP-1 and NF-κB Signaling Pathways. International Journal of Molecular Sciences, 21(13), 4619. https://doi.org/10.3390/ijms21134619