An Overview of Coumarin as a Versatile and Readily Accessible Scaffold with Broad-Ranging Biological Activities
Abstract
1. Introduction
Metabolism
2. Biological Activities
2.1. Antioxidant Activity
2.2. Anticancer Activity
2.3. Carbonic Anhydrase Inhibition
2.4. Antibacterial Activity
2.5. Antifungal Activity
2.6. Antiviral Activity
2.7. Anti-Inflammatory Activity
2.8. Neuroprotection: Effect on Alzheimer’s Disease
2.9. Anticonvulsant Activity
2.10. Anticoagulant Activity
2.11. Antidiabetic Activity
3. Other Applications of Coumarin Scaffold
3.1. Coumarins Photoproperties
3.1.1. Coumarins as Photocleavable Protecting Groups (PPGs)
3.1.2. Coumarins as Fluorescent Probes
3.2. Food Systems
3.3. Coumarin-Metal Complexes
4. Synthesis of Coumarin Scaffold
4.1. New Approaches in Coumarins Synthesis
4.1.1. Flow Chemistry and Immobilized Reagents
4.1.2. Photocatalysis
4.1.3. Solvent-Free Reactions
4.1.4. Microwave Assisted Reactions
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Borges, F.; Roleira, F.; Milhanzes, N.; Santana, L.; Uriarte, E. Simple coumarins and analogues in medicinal chemistry: Occurrence, synthesis and biological activity. Curr. Med. Chem. 2005, 12, 887–916. [Google Scholar] [CrossRef]
- Hoult, J.R.S.; Payá, M. Pharmacological and biochemical actions of simple coumarins: Natural products with therapeutic potential. Gen. Pharmacol. 1996, 27, 713–722. [Google Scholar] [CrossRef]
- Kummerle, A.E.; Vitorio, F.; Franco, D.P.; Pereira, T.M. Coumarin Compounds in Medicinal Chemistry: Some Important Examples from the Last Year. Curr. Top. Med. Chem. 2018, 18, 124–128. [Google Scholar] [CrossRef]
- Barot, K.P.; Jain, S.V.; Kremer, L.; Singh, S.; Ghate, M.D. Recent advances and therapeutic journey of coumarins: Current status and perspectives. Med. Chem. Res. 2015, 24, 2771–2798. [Google Scholar] [CrossRef]
- IUPAC. Nomenclature of Organic Chemistry; Pergamon Press: Oxford, UK, 1979. [Google Scholar]
- Zhu, J.J.; Jiang, J.G. Pharmacological and Nutritional Effects of Natural Coumarins and Their Structure–Activity Relationships. Mol. Nutr. Food Res. 2018, 62. [Google Scholar] [CrossRef]
- Stefanachi, A.; Leonetti, F.; Pisani, L.; Catto, M.; Carotti, A. Coumarin: A natural, privileged and versatile scaffold for bioactive compounds. Molecules 2018, 23, 250. [Google Scholar] [CrossRef] [PubMed]
- Srikrishna, D.; Godugu, C.; Dubey, P.K. A Review on Pharmacological Properties of Coumarins. Mini Rev. Med. Chem. 2016, 18. [Google Scholar] [CrossRef] [PubMed]
- Santra, H.K.; Banerjee, D. Natural Products as Fungicide and Their Role in Crop Protection. In Natural Bioactive Products in Sustainable Agriculture; Singh, J., Yadav, A., Eds.; Springer: Singapore, 2020; pp. 131–219. [Google Scholar]
- Mark, R.; Lyu, X.; Lee, J.J.L.; Parra-Saldívar, R.; Chen, W.N. Sustainable production of natural phenolics for functional food applications. J. Funct. Foods 2019, 57, 233–254. [Google Scholar] [CrossRef]
- DellaGreca, M.; Fiorentino, A.; Isidori, M.; Previtera, L.; Temussi, F.; Zarrelli, A. Benzocoumarins from the rhizomes of Juncus acutus. Tetrahedron 2003, 59, 4821–4825. [Google Scholar] [CrossRef]
- Tasior, M.; Kim, D.; Singha, S.; Krzeszewski, M.; Ahn, K.H.; Gryko, D.T. π-Expanded Coumarins: Synthesis, Optical Properties and Applications. R. Soc. Chem. 2014, 3, 1421–1446. [Google Scholar] [CrossRef]
- Zhang, Z.R.; Leung, W.N.; Cheung, H.Y.; Chan, C.W. Osthole: A Review on Its Bioactivities, Pharmacological Properties and Potential as Alternative Medicine. Evid. Based Complement. Altern. Med. 2015, 2015. [Google Scholar] [CrossRef] [PubMed]
- Bourgaud, F.; Hehn, A.; Larbat, R.; Doerper, S.; Gontier, E.; Kellner, S.; Matern, U. Biosynthesis of coumarins in plants: A major pathway still to be unravelled for cytochrome P450 enzymes. Phytochem. Rev. 2006, 5, 293–308. [Google Scholar] [CrossRef]
- Chakthong, S.; Weaaryee, P.; Puangphet, P.; Mahabusarakam, W.; Plodpai, P.; Voravuthikunchai, S.P.; Kanjana-Opas, A. Alkaloid and coumarins from the green fruits of Aegle marmelos. Phytochemistry 2012, 75, 108–113. [Google Scholar] [CrossRef] [PubMed]
- Basile, A.; Sorbo, S.; Spadaro, V.; Bruno, M.; Maggio, A.; Faraone, N.; Rosselli, S. Antimicrobial and antioxidant activities of coumarins from the roots of Ferulago campestris (apiaceae). Molecules 2009, 14, 939–952. [Google Scholar] [CrossRef]
- Patil, A.D.; Freyer, A.J.; Eggleston, D.S.; Haltiwanger, R.C.; Bean, M.F.; Taylor, P.B.; Caranfa, M.J.; Breen, A.L.; Bartus, H.R. The inophyllums, novel inhibitors of HIV-1 reverse transcriptase isolated from the Malaysian tree, Calophyllum inophyllum Linn. J. Med. Chem. 1993, 36, 4131–4138. [Google Scholar] [CrossRef]
- Wardrop, D.; Keeling, D. The story of the discovery of heparin and warfarin. Br. J. Haematol. 2008, 141, 757–763. [Google Scholar] [CrossRef]
- Pal, S.; Chatare, V.; Pal, M. Isocoumarin and Its Derivatives: An Overview on their Synthesis and Applications. Curr. Org. Chem. 2011, 15, 782–800. [Google Scholar] [CrossRef]
- Crombie, L.; Games, D.E.; McCormick, A. Isolation and structure ofmammea A/BA, A/AB and A/BB: A group of 4-arylcoumarin extractives of Mammea americana L. Tetrahedron Lett. 1966, 7, 145–149. [Google Scholar] [CrossRef]
- Egan, D.; O’kennedy, R.; Moran, E.; Cox, D.; Prosser, E.; Thornes, R.D. The pharmacology, metabolism, analysis and applications of coumarin and coumarin-related compounds. Drug Metab. Rev. 1990, 22, 503–529. [Google Scholar] [CrossRef]
- Leonart, L.P.; Gasparetto, J.C.; Pontes, F.L.D.; Cerqueira, L.B.; De Francisco, T.M.G.; Pontarolo, R. New metabolites of coumarin detected in human urine using ultra performance liquid chromatography/quadrupole-time-of-flight tandem mass spectrometry. Molecules 2017, 22, 2031. [Google Scholar] [CrossRef]
- Gasparetto, J.C.; Peccinini, R.G.; de Francisco, T.M.; Cerqueira, L.B.; Campos, F.R.; Pontarolo, R. A kinetic study of the main guaco metabolites using syrup formulation and the identification of an alternative route of coumarin metabolism in humans. PLoS ONE 2015, 10, e0118922. [Google Scholar] [CrossRef] [PubMed]
- Traykova, M.; Kostova, I. Coumarin derivatives and oxidative stress. Int. J. Pharm. 2005, 1, 29–32. [Google Scholar] [CrossRef]
- Liu, R.H.; Hotchkiss, J.H. Potential genotoxicity of chronically elevated nitric oxide. Mutat. Res. 1995, 339, 73–89. [Google Scholar] [CrossRef]
- Ames, B.N.; Shigenaga, M.K.; Gold, L.S. DNA lesions, inducible DNA repair and cell division: Three key factors in mutagenesis and carcinogenesis. Environ. Health Perspect. 1993, 101, 35–44. [Google Scholar] [CrossRef] [PubMed]
- Andreescu, S.; Hepel, M. Oxidative stress: Diagnosis, prevention and therapy. In ACS Symposium Series; American Chemical Society: Washington, DC, USA, 2011. [Google Scholar]
- Galano, A.; Mazzone, G.; Alvarez-Diduk, R.; Marino, T.; Alvarez-Idaboy, J.R.; Russo, N. Food Antioxidants: Chemical Insights at the Molecular Level. Annu. Rev. Food Sci. Technol. 2016, 7, 335–352. [Google Scholar] [CrossRef] [PubMed]
- Wahy, A.H.M.E.; Ismail, A.R.; Kana, M.T.H.A.; Negm, N.A. Synthesis and characterization of novel bis-(4-methylcoumarin) derivatives as photosensitizers in antimicrobial photodynamic therapy. J. Taiwan Inst. Chem. Eng. 2017, 77, 83–91. [Google Scholar] [CrossRef]
- Couttolenc, A.; Díaz-Porras, Á.; Espinoza, C.; Medina, M.E.; Trigos, Á. On the primary and secondary antioxidant activity from hydroxy-methylcoumarins: Experimental and theoretical studies. J. Phys. Org. Chem. 2020, 33, 1–10. [Google Scholar] [CrossRef]
- Thavasi, V.; Leong, L.P.; Bettens, R.P.A. Investigation of the influence of hydroxy groups on the radical scavenging ability of polyphenols. J. Phys. Chem. A 2006, 110, 4918–4923. [Google Scholar] [CrossRef]
- Medina, M.E.; Iuga, C.; Álvarez-Idaboy, J.R. Antioxidant activity of fraxetin and its regeneration in aqueous media. A density functional theory study. RSC Adv. 2014, 4, 52920–52932. [Google Scholar] [CrossRef]
- Medina, M.E.; Galano, A.; Alvarez-Idaboy, J.R. Theoretical study on the peroxyl radicals scavenging activity of esculetin and its regeneration in aqueous solution. Phys. Chem. Chem. Phys. 2014, 16, 1197–1207. [Google Scholar] [CrossRef]
- Wang, G.; Liu, Y.; Zhang, L.; An, L.; Chen, R.; Liu, Y.; Luo, Q.; Li, Y.; Wang, H.; Xue, Y. Computational study on the antioxidant property of coumarin-fused coumarins. Food Chem. 2020, 304, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Xi, G.L.; Liu, Z.Q. Coumarin-Fused Coumarin: Antioxidant Story from N,N -Dimethylamino and Hydroxyl Groups. J. Agric. Food Chem. 2015, 63, 3516–3523. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Wei, L.; Zhang, J.; Gu, G.; Guo, Z. Significantly enhanced antioxidant activity of chitosan through chemical modification with coumarins. Polym. Chem. 2019, 10, 1480–1488. [Google Scholar] [CrossRef]
- Luan, F.; Li, Q.; Tan, W.; Wei, L.; Zhang, J.; Dong, F.; Gu, G.; Guo, Z. The evaluation of antioxidant and antifungal properties of 6-amino-6-deoxychitosan in vitro. Int. J. Biol. Macromol. 2018, 107, 595–603. [Google Scholar] [CrossRef] [PubMed]
- Popova, S.A.; Shevchenko, O.G.; Chukicheva, I.Y.; Kutchin, A.V. Synthesis and Biological Evaluation of Novel Coumarins with tert-Butyl and Terpene Substituents. Chem. Biodivers. 2019, 16. [Google Scholar] [CrossRef]
- Pierce, B.A. Genetics. A Conceptual Approach, 3rd ed.; W. H. Freeman and Company: New York, NY, USA, 2010. [Google Scholar]
- Hanahan, D.; Weinberg, R.A. The hallmarks of cancer. Cell 2000, 100, 57–70. [Google Scholar] [CrossRef]
- Maleki, E.H.; Bahrami, A.R.; Sadeghian, H.; Matin, M.M. Discovering the structure–activity relationships of different O-prenylated coumarin derivatives as effective anticancer agents in human cervical cancer cells. Toxicol. In Vitro 2020, 63, 104745. [Google Scholar] [CrossRef]
- Comba, A.; Pasqualini, M.E. Primers on molecular pathways lipoxygenases: Their role as an oncogenic pathway in pancreatic cancer. Pancreatology 2009, 9, 724–728. [Google Scholar] [CrossRef]
- Kelavkar, U.P.; Glasgow, W.; Olson, S.J.; Foster, B.A.; Shappell, S.B. Overexpression of 12/15-lipoxygenase, an ortholog of human 15-lipoxygenase-1, in the prostate tumors of TRAMP mice. Neoplasia 2004, 6, 821–830. [Google Scholar] [CrossRef]
- Iranshahi, M.; Jabbari, A.; Orafaie, A.; Mehri, R.; Zeraatkar, S.; Ahmadi, T.; Alimardani, M.; Sadeghian, H. Synthesis and SAR studies of mono O-prenylated coumarins as potent 15-lipoxygenase inhibitors. Eur. J. Med. Chem. 2012, 57, 134–142. [Google Scholar] [CrossRef]
- Orafaie, A.; Sadeghian, H.; Bahrami, A.R.; Saboormaleki, S.; Matin, M.M. 5-farnesyloxycoumarin: A potent 15-LOX-1 inhibitor, prevents prostate cancer cell growth. Med. Chem. Res. 2017, 26, 227–234. [Google Scholar] [CrossRef]
- Hosseinymehr, M.; Matin, M.M.; Sadeghian, H.; Bahrami, A.R.; Kaseb-Mojaver, N. 8-Farnesyloxycoumarin induces apoptosis in PC-3 prostate cancer cells by inhibition of 15-lipoxygenase-1 enzymatic activity. Anticancer Drugs 2016, 27, 854–862. [Google Scholar] [CrossRef] [PubMed]
- Halawa, A.H.; Eliwa, E.M.; Hassan, A.A.; Nassar, H.S.; El-Eisawy, R.A.; Ismail, M.; Frese, M.; Shaaban, M.; El-Agrody, A.M.; Bedair, A.H.; et al. Synthesis, in vitro cytotoxicity activity against the human cervix carcinoma cell line and in silico computational predictions of new 4-arylamino-3-nitrocoumarin analogues. J. Mol. Struct. 2020, 1200, 127047. [Google Scholar] [CrossRef]
- Herrera-R, A.; Castrillón, W.; Otero, E.; Ruiz, E.; Carda, M.; Agut, R.; Naranjo, T.; Moreno, G.; Maldonado, M.E.; Cardona-G, W. Synthesis and antiproliferative activity of 3- and 7-styrylcoumarins. Med. Chem. Res. 2018, 27, 1893–1905. [Google Scholar] [CrossRef]
- Herrera-R, A.; Naranjo, T.W.; Maldonado, M.E.; Moreno-Q, G.; Yepes, A.; Cardona-G, W. Styrylcoumarin 7-SC2 induces apoptosis in SW480 human colon adenocarcinoma cells and inhibits azoxymethane-induced aberrant crypt foci formation in BALB/c mice. Med. Chem. Res. 2020, 29, 377–395. [Google Scholar] [CrossRef]
- Diao, Q.P.; Guo, H.; Wang, G.Q. Design, Synthesis and In Vitro Anticancer Activities of Diethylene Glycol Tethered Isatin-1,2,3-triazole-coumarin Hybrids. J. Heterocycl. Chem. 2019, 56, 1667–1671. [Google Scholar] [CrossRef]
- Li, Z.H.; Liu, X.Q.; Geng, P.F.; Suo, F.Z.; Ma, J.L.; Yu, B.; Zhao, T.Q.; Zhou, Z.Q.; Huang, C.X.; Zheng, Y.C.; et al. Discovery of [1,2,3]Triazolo[4,5-d]pyrimidine Derivatives as Novel LSD1 Inhibitors. ACS Med. Chem. Lett. 2017, 8, 384–389. [Google Scholar] [CrossRef]
- Yu, H.; Hou, Z.; Tian, Y.; Mou, Y.; Guo, C. Design, synthesis, cytotoxicity and mechanism of novel dihydroartemisinin-coumarin hybrids as potential anti-cancer agents. Eur. J. Med. Chem. 2018, 151, 434–449. [Google Scholar] [CrossRef]
- Singh, H.; Singh, J.V.; Gupta, M.K.; Saxena, A.K.; Sharma, S.; Nepali, K.; Bedi, P.M.S. Triazole tethered isatin-coumarin based molecular hybrids as novel antitubulin agents: Design, synthesis, biological investigation and docking studies. Bioorg. Med. Chem. Lett. 2017, 27, 3974–3979. [Google Scholar] [CrossRef]
- Xu, Z.; Song, X.F.; Hu, Y.Q.; Qiang, M.; Lv, Z.S. Azide-alkyne cycloaddition towards 1H-1,2,3-triazole-tethered gatifloxacin and isatin conjugates: Design, synthesis and in vitro anti-mycobacterial evaluation. Eur. J. Med. Chem. 2017, 138, 66–71. [Google Scholar] [CrossRef]
- Cai, G.; Yu, W.; Song, D.; Zhang, W.; Guo, J.; Zhu, J.; Ren, Y.; Kong, L. Discovery of fluorescent coumarin-benzo[b]thiophene 1, 1-dioxide conjugates as mitochondria-targeting antitumor STAT3 inhibitors. Eur. J. Med. Chem. 2019, 174, 236–251. [Google Scholar] [CrossRef] [PubMed]
- Aggarwal, B.B.; Kunnumakkara, A.B.; Harikumar, K.B.; Gupta, S.R.; Tharakan, S.T.; Koca, C.; Dey, S.; Sung, B. Signal transducer and activator of transcription-3, inflammation and cancer: How intimate is the relationship? Ann. N. Y. Acad. Sci. 2009, 1171, 59–76. [Google Scholar] [CrossRef] [PubMed]
- Supuran, C.T. Carbonic anhydrases: Novel therapeutic applications for inhibitors and activators. Nat. Rev. Drug Discov. 2008, 7, 168–181. [Google Scholar] [CrossRef] [PubMed]
- Alterio, V.; di Fiore, A.; D’Ambrosio, K.; Supuran, C.T.; De Simone, G. Multiple Binding Modes of Inhibitors to Carbonic Anhydrases: How to Design Specific Drugs Targeting 15 Different Isoforms? Chem. Rev. 2012, 112, 4421–4468. [Google Scholar] [CrossRef]
- Supuran, C.T.; Altamimi, A.S.A.; Carta, F. Carbonic Anhydrase inhibition and the management of glaucoma: A literature inhibition and the management of glaucoma: A literature. Expert Opin. Ther. Patents 2019, 29, 781–792. [Google Scholar] [CrossRef]
- Masini, E.; Carta, F.; Scozzafava, A. Antiglaucoma Carbonic anhydrase inhibitors: A patent review. Expert Opin. Ther. Patents 2013, 23, 705–716. [Google Scholar] [CrossRef]
- Aggarwal, M.; Kondeti, B.; McKenna, R. Anticonvulsant/antiepileptic carbonic anhydrase inhibitors: A patent review. Expert Opin. Ther. Patents 2013, 23, 717–724. [Google Scholar] [CrossRef]
- Lou, Y.; McDonald, P.C.; Oloumi, A.; Dedhar, S. Targeting tumour hypoxia: Suppression of breast tumor growth and metastasis by novel carbonic anhydrase IX inhibitors. Cancer Res. 2011, 71, 3364–3376. [Google Scholar] [CrossRef]
- Neri, D.; Supuran, C.T. Interfering with pH regulation in tumours as a therapeutic strategy. Nat. Rev. Drug Discov. 2011, 10, 767–777. [Google Scholar] [CrossRef]
- Maresca, A.; Temperini, C.; Vu, H.; Pham, N.B.; Poulsen, S.A.; Scozzafava, A.; Quinn, R.J.; Supuran, C.T. Non-zinc mediated inhibition of carbonic anhydrases: Coumarins are a new class of suicide inhibitors. J. Am. Chem. Soc. 2009, 131, 3057–3062. [Google Scholar] [CrossRef]
- Maresca, A.; Temperini, C.; Pochet, L.; Masereel, B.; Scozzafava, A.; Supuran, C.T. Deciphering the Mechanism of Carbonic Anhydrase Inhibition with Coumarins and Thiocoumarins. J. Med. Chem. 2010, 53, 335–344. [Google Scholar] [CrossRef] [PubMed]
- Maresca, A.; Scozzafava, A.; Supuran, C.T. 7,8-Disubstituted- but not 6,7-disubstituted coumarins selectively inhibit the transmembrane, tumor-associated carbonic anhydrase isoforms IX and XII over the cytosolic ones I and II in the low nanomolar/subnanomolar range. Bioorg. Med. Chem. Lett. 2010, 20, 7255–7258. [Google Scholar] [CrossRef] [PubMed]
- Touisni, N.; Maresca, A.; McDonald, P.C.; Lou, Y.; Scozzafava, A.; Dedhar, S.; Winum, J.Y.; Supuran, C.T. Glycosyl coumarin carbonic anhydrase IX and XII inhibitors strongly attenuate the growth of primary breast tumors. J. Med. Chem. 2011, 54, 8271–8277. [Google Scholar] [CrossRef] [PubMed]
- Ferraroni, M.; Carta, F.; Scozzafava, A.; Supuran, C.T. Thioxocoumarins Show an Alternative Carbonic Anhydrase Inhibition Mechanism Compared to Coumarins. J. Med. Chem. 2016, 59, 462–473. [Google Scholar] [CrossRef] [PubMed]
- De Luca, L.; Mancuso, F.; Ferro, S.; Buemi, M.R.; Angeli, A.; Del Prete, S.; Capasso, C.; Supuran, C.T.; Gitto, R. Inhibitory effects and structural insights for a novel series of coumarin-based compounds that selectively target human CA IX and CA XII carbonic anhydrases. Eur. J. Med. Chem. 2018, 143, 276–282. [Google Scholar] [CrossRef]
- Fois, B.; Distinto, S.; Meleddu, R.; Deplano, S.; Maccioni, E.; Floris, C.; Rosa, A.; Nieddu, M.; Caboni, P.; Sissi, C.; et al. Coumarins from Magydaris pastinacea as inhibitors of the tumour-associated carbonic anhydrases IX and XII: Isolation, biological studies and in silico evaluation. J. Enzyme Inhib. Med. Chem. 2020, 35, 539–548. [Google Scholar] [CrossRef]
- Buran, K.; Bua, S.; Poli, G.; Bayram, F.E.Ö.; Tuccinardi, T.; Supuran, C.T. Novel 8-substituted coumarins that selectively inhibit human carbonic anhydrase IX and XII. Int. J. Mol. Sci. 2019, 20, 1208. [Google Scholar] [CrossRef]
- Krasavin, M.; Žalubovskis, R.; Grandāne, A.; Domračeva, I.; Zhmurov, P.; Supuran, C.T. Sulfocoumarins as dual inhibitors of human carbonic anhydrase isoforms IX/XII and of human thioredoxin reductase. J. Enzyme Inhib. Med. Chem. 2020, 35, 506–510. [Google Scholar] [CrossRef]
- Kurt, B.Z.; Dag, A.; Doğan, B.; Durdagi, S.; Angeli, A.; Nocentini, A.; Supuran, C.T.; Sonmez, F. Synthesis, biological activity and multiscale molecular modeling studies of bis-coumarins as selective carbonic anhydrase IX and XII inhibitors with effective cytotoxicity against hepatocellular carcinoma. Bioorg. Chem. 2019, 87, 838–850. [Google Scholar] [CrossRef] [PubMed]
- Narella, S.G.; Shaik, M.G.; Mohammed, A.; Alvala, M.; Angeli, A.; Supuran, C.T. Synthesis and biological evaluation of coumarin-1,3,4-oxadiazole hybrids as selective carbonic anhydrase IX and XII inhibitors. Bioorg. Chem. 2019, 87, 765–772. [Google Scholar] [CrossRef] [PubMed]
- Bassetti, M.; Merelli, M.; Temperoni, C.; Astilean, A. New antibiotics for bad bugs: Where are we? Ann. Clin. Microbiol. Antimicrob. 2013, 12, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Brown, E.D.; Wright, G.D. Antibacterial drug discovery in the resistance era. Nature 2016, 529, 336–343. [Google Scholar] [CrossRef] [PubMed]
- Cowan, M.M. Plant products as antimicrobial agents. Clin. Microbiol. Rev. 1999, 12, 564–582. [Google Scholar] [CrossRef] [PubMed]
- De Souza, S.M.; Delle Monache, F.; Smânia, A. Antibacterial activity of coumarins. Z. Naturforsch. Sect. C J. Biosci. 2005, 60, 693–700. [Google Scholar] [CrossRef]
- Nagamallu, R.; Srinivasan, B.; Ningappa, M.B.; Kariyappa, A.K. Synthesis of novel coumarin appended bis(formylpyrazole) derivatives: Studies on their antimicrobial and antioxidant activities. Bioorg. Med. Chem. Lett. 2016, 26, 690–694. [Google Scholar] [CrossRef]
- Chougala, B.M.; Samundeeswari, S.; Holiyachi, M.; Naik, N.S.; Shastri, L.A.; Dodamani, S.; Jalalpure, S.; Dixit, S.R.; Joshi, S.D.; Sunagar, V.A. Green, unexpected synthesis of bis-coumarin derivatives as potent anti-bacterial and anti-inflammatory agents. Eur. J. Med. Chem. 2018, 143, 1744–1756. [Google Scholar] [CrossRef]
- Rice, L.B. Federal Funding for the Study of Antimicrobial Resistance in Nosocomial Pathogens: No ESKAPE. J. Infect. Dis. 2008, 197, 1079–1081. [Google Scholar] [CrossRef]
- Naik, N.S.; Shastri, L.A.; Joshi, S.D.; Sunagar, V.; Dixit, S.R.; Chougala, B.M.; Samundeeswari, S.; Holiyachi, M.; Shaikh, F.; Madar, J.; et al. 3,4-Dihydropyrimidinone-coumarin analogues as a new class of selective agent against S. aureus: Synthesis, biological evaluation and molecular modelling study. Bioorg. Med. Chem. 2017, 25, 1413–1422. [Google Scholar] [CrossRef]
- Chavan, R.R.; Hosamani, K.M. Microwave-assisted synthesis, computational studies and antibacterial/ anti-inflammatory activities of compounds based on coumarin-pyrazole hybrid. R. Soc. Open Sci. 2018, 5. [Google Scholar] [CrossRef]
- Rahman, A.; Choudhary, M.I.; Thomsen, W.J. Bioassay Techniques for Drug Develompent, 1st ed.; Harwood Academic Publishers: Amsterdam, The Netherlands, 2001. [Google Scholar]
- Madeiro, S.A.L.; Borges, N.H.P.B.; Souto, A.L.; de Figueiredo, P.T.R.; Siqueira-Junior, J.P.; Tavares, J.F. Modulation of the antibiotic activity against multidrug resistant strains of coumarins isolated from Rutaceae species. Microb. Pathog. 2017, 104, 151–154. [Google Scholar] [CrossRef]
- Widelski, J.; Luca, S.V.; Skiba, A.; Chinou, I.; Marcourt, L.; Wolfender, J.L.; Skalicka-Wozniak, K. Isolation and antimicrobial activity of coumarin derivatives from fruits of peucedanum luxurians tamamsch. Molecules 2018, 23, 1222. [Google Scholar] [CrossRef]
- Liu, H.; Ren, Z.L.; Wang, W.; Gong, J.X.; Chu, M.J.; Ma, Q.W.; Wang, J.C.; Lv, X.H. Novel coumarin-pyrazole carboxamide derivatives as potential topoisomerase II inhibitors: Design, synthesis and antibacterial activity. Eur. J. Med. Chem. 2018, 157, 81–87. [Google Scholar] [CrossRef] [PubMed]
- Brown, G.D.; Denning, D.W.; Gow, N.A.R.; Levitz, S.M.; Netea, M.G.; White, T.C. Hidden killers: Human fungal infections. Sci. Transl. Med. 2012, 4, 1–10. [Google Scholar] [CrossRef] [PubMed]
- GAFFI Fungal Disease Frequency|Gaffi—Global Action Fund for Fungal Infections. Available online: https://www.gaffi.org/why/fungal-disease-frequency/ (accessed on 21 March 2020).
- How Common are Fungal Diseases?—Fungal Infection Trust. Available online: https://www.fungalinfectiontrust.org/how-common-are-fungal-diseases (accessed on 21 March 2020).
- Bartlett, J.G. Nosocomial bloodstream infections in US hospitals: Analysis of 24,179 cases from a prospective nationwide surveillance study. Infect. Dis. Clin. Pract. 2004, 12, 376. [Google Scholar] [CrossRef]
- Pfaller, M.A.; Diekema, D.J. Epidemiology of invasive candidiasis: A persistent public health problem. Clin. Microbiol. Rev. 2007, 20, 133–163. [Google Scholar] [CrossRef]
- Shi, Y.; Zhou, C.H. Synthesis and evaluation of a class of new coumarin triazole derivatives as potential antimicrobial agents. Bioorg. Med. Chem. Lett. 2011, 21, 956–960. [Google Scholar] [CrossRef] [PubMed]
- Shaikh, M.H.; Subhedar, D.D.; Kalam Khan, F.A.; Sangshetti, J.N.; Shingate, B.B. 1,2,3-Triazole incorporated coumarin derivatives as potential antifungal and antioxidant agents. Chin. Chem. Lett. 2016, 27, 295–301. [Google Scholar] [CrossRef]
- Tiwari, S.; Seijas, J.; Vazquez-Tato, M.; Sarkate, A.; Karnik, K.; Nikalje, A. Facile Synthesis of Novel Coumarin Derivatives, Antimicrobial Analysis, Enzyme Assay, Docking Study, ADMET Prediction and Toxicity Study. Molecules 2017, 22, 1172. [Google Scholar] [CrossRef]
- Elias, R.; Benhamou, R.I.; Jaber, Q.Z.; Dorot, O.; Zada, S.L.; Oved, K.; Pichinuk, E.; Fridman, M. Antifungal activity, mode of action variability and subcellular distribution of coumarin-based antifungal azoles. Eur. J. Med. Chem. 2019, 179, 779–790. [Google Scholar] [CrossRef]
- Dikpınar, T.; Süzgeç-Selçuk, S.; Çelik, B.Ö.; Uruşak, E.A. Antimicrobial activity of rhizomes of Ferulago trachycarpa Boiss. and bioguided isolation of active coumarin constituents. Ind. Crops Prod. 2018, 123, 762–767. [Google Scholar] [CrossRef]
- Lee, J.H.; Kim, Y.G.; Cho, H.S.; Ryu, S.Y.; Cho, M.H.; Lee, J. Coumarins reduce biofilm formation and the virulence of Escherichia coli O157:H7. Phytomedicine 2014, 21, 1037–1042. [Google Scholar] [CrossRef] [PubMed]
- Reen, F.J.; Gutiérrez-Barranquero, J.A.; Parages, M.L.; O’Gara, F. Coumarin: A novel player in microbial quorum sensing and biofilm formation inhibition. Appl. Microbiol. Biotechnol. 2018, 102, 2063–2073. [Google Scholar] [CrossRef] [PubMed]
- Xu, K.; Wang, J.L.; Chu, M.P.; Jia, C. Activity of coumarin against Candida albicans biofilms. J. Mycol. Med. 2019, 29, 28–34. [Google Scholar] [CrossRef] [PubMed]
- Barry, A.L.; Thornsberry, J.C. Susceptibility tests: Diffusion test procedures. In Manual of Clinical Microbiology, 4th ed.; Balows, A., Hausler, W.H., Jr., Shadomy, J., Lenette, E.H., Eds.; American Society of Microbiology: Washington, DC, USA, 1985; pp. 978–987. [Google Scholar]
- Dharavath, R.; Nagaraju, N.; Reddy, M.R.; Ashok, D.; Sarasija, M.; Vijjulatha, M.; T, V.; Jyothi, K.; Prashanthi, G. Microwave-assisted synthesis, biological evaluation and molecular docking studies of new coumarin-based 1,2,3-triazoles. RSC Adv. 2020, 10, 11615–11623. [Google Scholar] [CrossRef]
- WHO Coronavirus (COVID-19) Events as They Happen. Available online: https://www.who.int/emergencies/diseases/novel-coronavirus-2019/events-as-they-happen (accessed on 21 March 2020).
- Mishra, S.; Pandey, A.; Manvati, S. Coumarin: An emerging antiviral agent. Heliyon 2020, 6, e03217. [Google Scholar] [CrossRef]
- Lilly Ganju, S.S. Plant Derived Antivirals: A Potential Source of Drug Development. J. Virol. Antivir. Res. 2013, 2. [Google Scholar] [CrossRef]
- Kwon, J.E.; Lee, Y.G.; Kang, J.H.; Bai, Y.F.; Jeong, Y.J.; Baek, N.I.; Seo, Y.J.; Kang, S.C. Anti-viral activity of compounds from Agrimonia pilosa and Galla rhois extract mixture. Bioorg. Chem. 2019, 93, 103320. [Google Scholar] [CrossRef]
- Guo, H.; Wan, X.; Niu, F.; Sun, J.; Shi, C.; Ye, J.M.; Zhou, C. Evaluation of antiviral effect and toxicity of total flavonoids extracted from Robinia pseudoacacia cv. idaho. Biomed. Pharmacother. 2019, 118, 109335. [Google Scholar] [CrossRef]
- Dewi, B.E.; Ratningpoeti, E.; Desti, H.; Angelina, M. In vitro and in silico study to evaluate the effectiveness of quercitrin as antiviral drug to dengue virus. AIP Conf. Proc. 2019, 2193. [Google Scholar] [CrossRef]
- Sahuc, M.; Sahli, R.; Rivière, C.; Pène, V.; Lavie, M. Vandeputte, Dehydrojuncusol, a Natural Phenanthrene Compound Extracted from Juncus maritimus, Is a New Inhibitor of Hepatitis C Virus RNA Replication. J. Virol. 2019, 93, 1–15. [Google Scholar] [CrossRef]
- Teplova, V.V.; Isakova, E.P.; Klein, O.I.; Dergachova, D.I.; Gessler, N.N.; Deryabina, Y.I. Natural Polyphenols: Biological Activity, Pharmacological Potential, Means of Metabolic Engineering (Review). Appl. Biochem. Microbiol. 2018, 54, 221–237. [Google Scholar] [CrossRef]
- Campagna, M.; Rivas, C. Antiviral activity of resveratrol. Biochem. Soc. Trans. 2010, 38, 50–53. [Google Scholar] [CrossRef] [PubMed]
- Hassan, M.Z.; Osman, H.; Ali, M.A.; Ahsan, M.J. Therapeutic potential of coumarins as antiviral agents. Eur. J. Med. Chem. 2016, 123, 236–255. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.P.; Yan, G.; Xie, Y.T.; Lin, T.C.; Zhang, W.; Li, J.; Wu, Y.J.; Zhou, J.Y.; Fu, Y.H. Bioactive prenylated coumarins as potential anti-inflammatory and anti-HIV agents from Clausena lenis. Bioorg. Chem. 2020, 97, 103699. [Google Scholar] [CrossRef]
- Liu, Y.P.; Liu, Q.L.; Zhang, X.L.; Niu, H.Y.; Guan, C.Y.; Sun, F.K.; Xu, W.; Fu, Y.H. Bioactive monoterpene indole alkaloids from Nauclea officinalis. Bioorg. Chem. 2019, 83, 1–5. [Google Scholar] [CrossRef]
- Liu, Y.P.; Zhao, W.H.; Feng, X.Y.; Zhang, Z.J.; Zong, K.; Sun, Z.G.; Zheng, Y.T.; Fu, Y.H. Novel tetrahydrofuran derivatives from Trigonostemon howii with their potential anti-HIV-1 activities. Bioorg. Chem. 2018, 79, 111–114. [Google Scholar] [CrossRef]
- Liu, Y.P.; Yan, G.; Guo, J.M.; Liu, Y.Y.; Li, Y.J.; Zhao, Y.Y.; Qiang, L.; Fu, Y.H. Prenylated Coumarins from the Fruits of Manilkara zapota with Potential Anti-inflammatory Effects and Anti-HIV Activities. J. Agric. Food Chem. 2019, 67, 11942–11947. [Google Scholar] [CrossRef]
- Jesumoroti, O.J.; Faridoon; Mnkandhla, D.; Isaacs, M.; Hoppe, H.C.; Klein, R. Evaluation of novel N′-(3-hydroxybenzoyl)-2-oxo-2H-chromene-3-carbohydrazide derivatives as potential HIV-1 integrase inhibitors. Medchemcomm 2019, 10, 80–88. [Google Scholar] [CrossRef]
- Goldgur, Y.; Dyda, F.; Hickman, A.B.; Jenkins, T.M.; Craigie, R.; Davies, D.R. Three new structures of the core domain of HIV-1 integrase: An active site that binds magnesium. Proc. Natl. Acad. Sci. USA 1998, 95, 9150–9154. [Google Scholar] [CrossRef]
- Neamati, N.; Sunder, S.; Pommier, Y. Design and discovery of HIV-1 integrase inhibitors. Drug Discov. Today 1997, 2, 487–498. [Google Scholar] [CrossRef]
- Zhao, H.; Neamati, N.; Sunder, S.; Hong, H.; Wang, S.; Milne, G.W.A.; Pommier, Y.; Burke, T.R. Hydrazide-Containing Inhibitors of HIV-1 Integrase. J. Med. Chem. 1997, 2623, 937–941. [Google Scholar] [CrossRef] [PubMed]
- Park, J.H.; Lee, S.U.; Kim, S.H.; Shin, S.Y.; Lee, J.Y.; Shin, C.G.; Yoo, K.H.; Lee, Y.S. Chromone and chromanone derivatives as strand transfer inhibitors of HIV-1 integrase. Arch. Pharm. Res. 2008, 31, 1–5. [Google Scholar] [CrossRef] [PubMed]
- McColl, D.J.; Chen, X. Strand transfer inhibitors of HIV-1 integrase: Bringing IN a new era of antiretroviral therapy. Antiviral Res. 2010, 85, 101–118. [Google Scholar] [CrossRef] [PubMed]
- WHO -Up to 650 000 People Die of Respiratory Diseases Linked to Seasonal Flu Each Year. Available online: https://www.who.int/news-room/detail/14-12-2017-up-to-650-000-people-die-of-respiratory-diseases-linked-to-seasonal-flu-each-year (accessed on 21 March 2020).
- Kilbourne, E.D. Influenza pandemics of the 20th century. Emerg. Infect. Dis. 2006, 12, 9–14. [Google Scholar] [CrossRef] [PubMed]
- Osman, H.; Yusufzai, S.K.; Khan, M.S.; Abd Razik, B.M.; Sulaiman, O.; Mohamad, S.; Gansau, J.A.; Ezzat, M.O.; Parumasivam, T.; Hassan, M.Z. New thiazolyl-coumarin hybrids: Design, synthesis, characterization, X-ray crystal structure, antibacterial and antiviral evaluation. J. Mol. Struct. 2018, 1166, 147–154. [Google Scholar] [CrossRef]
- Arshad, A.; Osman, H.; Bagley, M.C.; Lam, C.K.; Mohamad, S.; Zahariluddin, A.S.M. Synthesis and antimicrobial properties of some new thiazolyl coumarin derivatives. Eur. J. Med. Chem. 2011, 46, 3788–3794. [Google Scholar] [CrossRef]
- Reddy, G.M.; Garcia, J.R.; Reddy, V.H.; de Andrade, A.M.; Camilo, A.; Pontes Ribeiro, R.A.; de Lazaro, S.R. Synthesis, antimicrobial activity and advances in structure-activity relationships (SARs) of novel tri-substituted thiazole derivatives. Eur. J. Med. Chem. 2016, 123, 508–513. [Google Scholar] [CrossRef]
- Pavurala, S.; Vaarla, K.; Kesharwani, R.; Naesens, L.; Liekens, S.; Vedula, R.R. Bis coumarinyl bis triazolothiadiazinyl ethane derivatives: Synthesis, antiviral activity evaluation and molecular docking studies. Synth. Commun. 2018, 48, 1494–1503. [Google Scholar] [CrossRef]
- Bizzarri, B.M.; Botta, L.; Capecchi, E.; Celestino, I.; Checconi, P.; Palamara, A.T.; Nencioni, L.; Saladino, R. Regioselective IBX-Mediated Synthesis of Coumarin Derivatives with Antioxidant and Anti-influenza Activities. J. Nat. Prod. 2017, 80, 3247–3254. [Google Scholar] [CrossRef]
- Tsay, S.C.; Lin, S.Y.; Huang, W.C.; Hsu, M.H.; Hwang, K.C.; Lin, C.C.; Horng, J.C.; Chen, I.C.; Hwu, J.R.; Shieh, F.K.; et al. Synthesis and structure-activity relationships of imidazole-coumarin conjugates against hepatitis C virus. Molecules 2016, 21, 228. [Google Scholar] [CrossRef]
- Huang, S.X.; Mou, J.F.; Luo, Q.; Mo, Q.H.; Zhou, X.L.; Huang, X.; Xu, Q.; Tan, X.D.; Chen, X.; Liang, C.Q. Anti-Hepatitis B Virus Activity of Esculetin from Microsorium fortunei in Vitro and in Vivo. Molecules 2019, 24, 3475. [Google Scholar] [CrossRef] [PubMed]
- Taramelli, D.; Miglietta, A.; D’Alessandro, S.; Corbett, Y. Risposta del tessuto al danno. In Patologia Generale e Fisiopatologia, 2nd ed.; Celotti, F., Ed.; EdiSES: Napoli, Italy, 2013; pp. 86–127. [Google Scholar]
- Medzhitov, R. Origin and physiological roles of inflammation. Nature 2008, 454, 428–435. [Google Scholar] [CrossRef] [PubMed]
- Vane, J.; Botting, R. Inflammation and the mechanism of action of anti-inflammatory drugs. FASEB J. 1987, 89–96. [Google Scholar] [CrossRef]
- Gaddi, A.; Cicero, A.F.G.; Pedro, E.J. Clinical perspectives of anti-inflammatory therapy in the elderly: The lipoxigenase (LOX)/cycloxigenase (COX) inhibition concept. Arch. Gerontol. Geriatr. 2004, 38, 201–212. [Google Scholar] [CrossRef]
- Sanghi, S.; MacLaughlin, E.; Jewell, C.; Chaffer, S.; Naus, P.; Watson, L.; Dostal, D. Cyclooxygenase-2 Inhibitors: A Painful Lesson. Cardiovasc. Hematol. Disord. Targets 2012, 6, 83–98. [Google Scholar] [CrossRef]
- Schjerning Olsen, A.M.; Fosbøl, E.L.; Lindhardsen, J.; Folke, F.; Charlot, M.; Selmer, C.; Lamberts, M.; Bjerring Olesen, J.; Køber, L.; Hansen, P.R.; et al. Duration of treatment with nonsteroidal anti-inflammatory drugs and impact on risk of death and recurrent myocardial infarction in patients with prior myocardial infarction: A nationwide cohort study. Circulation 2011, 123, 2226–2235. [Google Scholar] [CrossRef]
- Woods, K.W.; McCroskey, R.W.; Michaelides, M.R.; Wada, C.K.; Hulkower, K.I.; Bell, R.L. Thiazole analogues of the NSAID indomethacin as selective COX-2 inhibitors. Bioorg. Med. Chem. Lett. 2001, 11, 1325–1328. [Google Scholar] [CrossRef]
- El-Gamal, M.I.; Oh, C.H. Synthesis, in vitro antiproliferative activity and in silico studies of fused tricyclic coumarin sulfonate derivatives. Eur. J. Med. Chem. 2014, 84, 68–76. [Google Scholar] [CrossRef]
- Unsal-Tan, O.; Ozadali, K.; Piskin, K.; Balkan, A. Molecular modeling, synthesis and screening of some new 4-thiazolidinone derivatives with promising selective COX-2 inhibitory activity. Eur. J. Med. Chem. 2012, 57, 59–64. [Google Scholar] [CrossRef]
- Rambabu, D.; Mulakayala, N.; Ismail; Ravi Kumar, K.; Pavan Kumar, G.; Mulakayala, C.; Kumar, C.S.; Kalle, A.M.; Basaveswara Rao, M.V.; Oruganti, S.; et al. Synthesis and pharmacological evaluation of N-substituted 2-(2-oxo-2H-chromen-4-yloxy)propanamide as cyclooxygenase inhibitors. Bioorg. Med. Chem. Lett. 2012, 22, 6745–6749. [Google Scholar] [CrossRef]
- Kontogiorgis, C.A.; Hadjipavlou-Litina, D.J. Synthesis and antiinflammatory activity of coumarin derivatives. J. Med. Chem. 2005, 48, 6400–6408. [Google Scholar] [CrossRef] [PubMed]
- Dawood, D.H.; Batran, R.Z.; Farghaly, T.A.; Khedr, M.A.; Abdulla, M.M. New Coumarin Derivatives as Potent Selective COX-2 Inhibitors: Synthesis, Anti-Inflammatory, QSAR and Molecular Modeling Studies. Arch. Pharm. 2015, 348, 875–888. [Google Scholar] [CrossRef] [PubMed]
- Ford-Hutchinson, A.W.; Gresser, M.; Young, R.N. 5-Lipoxygenase. Annu. Rev. Biochem. 1994, 63, 383–417. [Google Scholar] [CrossRef] [PubMed]
- Werz, O.; Steinhilber, D. Development of 5-lipoxygenase inhibitors—Lessons from cellular enzyme regulation. Biochem. Pharmacol. 2005, 70, 327–333. [Google Scholar] [CrossRef]
- Srivastava, P.; Vyas, V.K.; Variya, B.; Patel, P.; Qureshi, G.; Ghate, M. Synthesis, anti-inflammatory, analgesic, 5-lipoxygenase (5-LOX) inhibition activities and molecular docking study of 7-substituted coumarin derivatives. Bioorg. Chem. 2016, 67, 130–138. [Google Scholar] [CrossRef]
- Liu, B.Y.; Zhang, C.; Zeng, K.W.; Li, J.; Guo, X.Y.; Zhao, M.B.; Tu, P.F.; Jiang, Y. Anti-Inflammatory Prenylated Phenylpropenols and Coumarin Derivatives from Murraya exotica. J. Nat. Prod. 2018, 81, 22–33. [Google Scholar] [CrossRef]
- Courtois, G. The NF-κB signaling pathway in human genetic diseases. Cell. Mol. Life Sci. 2005, 62, 1682–1691. [Google Scholar] [CrossRef]
- Karin, M.; Ben-Neriah, Y. Phosphorilation meets ubiquitination: The control of NF-kB activity. Annu. Rev. Immunol. 2000, 621–663. [Google Scholar] [CrossRef]
- Fan, H.; Gao, Z.; Ji, K.; Li, X.; Wu, J.; Liu, Y.; Wang, X.; Liang, H.; Liu, Y.; Li, X.; et al. The in vitro and in vivo anti-inflammatory effect of osthole, the major natural coumarin from Cnidium monnieri (L.) Cuss, via the blocking of the activation of the NF-κB and MAPK/p38 pathways. Phytomedicine 2019, 58, 152864. [Google Scholar] [CrossRef]
- Mu, C.; Wu, M.; Li, Z. Anti-Inflammatory Effect of Novel 7-Substituted Coumarin Derivatives through Inhibition of NF-κB Signaling Pathway. Chem. Biodivers. 2019, 16, e1800559. [Google Scholar] [CrossRef]
- Alzheimer-Riese. s.d. Available online: https://www.alzheimer-riese.it/ (accessed on 20 June 2018).
- Vitthalrao, B.K.; Sunanda, V.K.; Sharad, G.J. Alzheimer’s disease: Overview. Int. Acad. Inst. Sci. Technol. 2016, 3, 23–38. [Google Scholar]
- Bartus, R.T.; Dean, R.L.; Beer, B.; Lippa, A.S. The cholinergic hypothesis of geriatric memory dysfunction. Science 1982, 217, 408–417. [Google Scholar] [CrossRef] [PubMed]
- Furukawa-Hibi, Y.; Alkam, T.; Nitta, A.; Matsuyama, A.; Mizoguchi, H.; Suzuki, K.; Moussaoui, S.; Yu, Q.S.; Greig, N.H.; Nagai, T.; et al. Butyrylcholinesterase inhibitors ameliorate cognitive dysfunction induced by amyloid-β peptide in mice. Behav. Brain Res. 2011, 225, 222–229. [Google Scholar] [CrossRef] [PubMed]
- Querfurth, H.W.; Laferla, F.M. Mechanisms of disease: Alzheimer’s disease. N. Engl. J. Med. 2010, 362, 329–344. [Google Scholar] [CrossRef] [PubMed]
- Vassar, R.; Kandalepas, P. The b-secretase enzyme BACE1 as a therapeutic target for Alzheimer’s disease. Alzheimers Res. Ther. 2011, 3, 20–26. [Google Scholar] [CrossRef]
- Karakaya, S.; Koca, M.; Sytar, O.; Dursunoglu, B.; Ozbek, H.; Duman, H.; Guvenalp, Z.; Kılıc, C.S. Antioxidant and anticholinesterase potential of Ferulago cassia with farther bio-guided isolation of active coumarin constituents. S. Afr. J. Bot. 2019, 121, 536–542. [Google Scholar] [CrossRef]
- Ellman, G.L.; Courtney, K.D.; Andresjr, V.; Featherstone, R.M. A new and rapid colorimetric determination of acetylcholinesterase activity. Biochem. Pharmacol. 1961, 7, 88–95. [Google Scholar] [CrossRef]
- Najafi, Z.; Mahdavi, M.; Saeedi, M.; Karimpour-Razkenari, E.; Edraki, N.; Sharifzadeh, M.; Khanavi, M.; Akbarzadeh, T. Novel tacrine-coumarin hybrids linked to 1,2,3-triazole as anti-Alzheimer’s compounds: In vitro and in vivo biological evaluation and docking study. Bioorg. Chem. 2019, 83, 303–316. [Google Scholar] [CrossRef]
- De Souza, L.G.; Rennã, M.N.; Figueroa-Villar, J.D. Coumarins as cholinesterase inhibitors: A review. Chem. Biol. Interact. 2016, 254, 11–23. [Google Scholar] [CrossRef]
- Najafi, Z.; Mahdavi, M.; Saeedi, M.; Karimpour-Razkenari, E.; Asatouri, R.; Vafadarnejad, F.; Moghadam, F.H.; Khanavi, M.; Sharifzadeh, M.; Akbarzadeh, T. Novel tacrine-1,2,3-triazole hybrids: In vitro, in vivo biological evaluation and docking study of cholinesterase inhibitors. Eur. J. Med. Chem. 2017, 125, 1200–1212. [Google Scholar] [CrossRef]
- Rastegari, A.; Nadri, H.; Mahdavi, M.; Moradi, A.; Mirfazli, S.S.; Edraki, N.; Moghadam, F.H.; Larijani, B.; Akbarzadeh, T.; Saeedi, M. Design, synthesis and anti-Alzheimer’s activity of novel 1,2,3-triazole-chromenone carboxamide derivatives. Bioorg. Chem. 2019, 83, 391–401. [Google Scholar] [CrossRef] [PubMed]
- Asadipour, A.; Alipour, M.; Jafari, M.; Khoobi, M.; Emami, S.; Nadri, H.; Sakhteman, A.; Moradi, A.; Sheibani, V.; Homayouni Moghadam, F.; et al. Novel coumarin-3-carboxamides bearing N-benzylpiperidine moiety as potent acetylcholinesterase inhibitors. Eur. J. Med. Chem. 2013, 70, 623–630. [Google Scholar] [CrossRef] [PubMed]
- Iraji, A.; Firuzi, O.; Khoshneviszadeh, M.; Tavakkoli, M.; Mahdavi, M.; Nadri, H.; Edraki, N.; Miri, R. Multifunctional iminochromene-2H-carboxamide derivatives containing different aminomethylene triazole with BACE1 inhibitory, neuroprotective and metal chelating properties targeting Alzheimer’s disease. Eur. J. Med. Chem. 2017, 141, 690–702. [Google Scholar] [CrossRef] [PubMed]
- De Souza, G.A.; da Silva, S.J.; de Nigris Del Cistia, C.; Pitasse-Santos, P.; de Oliveira Pires, L.; Passos, Y.M.; Cordeiro, Y.; Cardoso, C.M.; Castro, R.N.; Sant’Anna, C.M.R.; et al. Discovery of novel dual-active 3-(4-(dimethylamino)phenyl)-7-aminoalcoxy-coumarin as potent and selective acetylcholinesterase inhibitor and antioxidant. J. Enzyme Inhib. Med. Chem. 2019, 34, 631–637. [Google Scholar] [CrossRef]
- Yang, J.; Zhang, P.; Hu, Y.; Liu, T.; Sun, J.; Wang, X. Synthesis and biological evaluation of 3-arylcoumarins as potential anti-Alzheimer’s disease agents. J. Enzyme Inhib. Med. Chem. 2019, 34, 651–656. [Google Scholar] [CrossRef]
- Knez, D.; Sova, M.; Košak, U.; Gobec, S. Dual inhibitors of cholinesterases and monoamine oxidases for Alzheimer’s disease. Future Med. Chem. 2017, 9, 811–832. [Google Scholar] [CrossRef]
- Joubert, J.; Foka, G.B.; Repsold, B.P.; Oliver, D.W.; Kapp, E.; Malan, S.F. Synthesis and evaluation of 7-substituted coumarin derivatives as multimodal monoamine oxidase-B and cholinesterase inhibitors for the treatment of Alzheimer’s disease. Eur. J. Med. Chem. 2017, 125, 853–864. [Google Scholar] [CrossRef]
- Shi, D.H.; Min, W.; Song, M.Q.; Si, X.X.; Li, M.C.; Zhang, Z.Y.; Liu, Y.W.; Liu, W.W. Synthesis, characterization, crystal structure and evaluation of four carbazole-coumarin hybrids as multifunctional agents for the treatment of Alzheimer’s disease. J. Mol. Struct. 2020, 1209, 127897. [Google Scholar] [CrossRef]
- Tang, Y.-Z.; Liu, Z.-Q. Free-radical-scavenging effect of carbazole derivatives on AAPH-induced hemolysis of human erythrocytes. Biorgan. Maed. Chem. 2007, 15, 1903–1913. [Google Scholar] [CrossRef]
- Yang, W.; Wong, Y.; Ng, O.T.W.; Bai, L.P.; Kwong, D.W.J.; Ke, Y.; Jiang, Z.H.; Li, H.W.; Yung, K.K.L.; Wong, M.S. Inhibition of beta-amyloid peptide aggregation by multifunctional carbazole-based fluorophores. Angew. Chem. Int. Ed. 2012, 51, 1804–1810. [Google Scholar] [CrossRef]
- Fang, L.; Fang, X.; Gou, S.; Lupp, A.; Lenhardt, I.; Sun, Y.; Huang, Z.; Chen, Y.; Zhang, Y.; Fleck, C. Design, synthesis and biological evaluation of D-ring opened galantamine analogs as multifunctional anti-Alzheimer agents. Eur. J. Med. Chem. 2014, 76, 376–386. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Qiang, X.; Luo, L.; Yang, X.; Xiao, G.; Liu, Q.; Ai, J.; Tan, Z.; Deng, Y. Aurone Mannich base derivatives as promising multifunctional agents with acetylcholinesterase inhibition, anti-β-amyloid aggragation and neuroprotective properties for the treatment of Alzheimer’s disease. Eur. J. Med. Chem. 2017, 126, 762–775. [Google Scholar] [CrossRef] [PubMed]
- McNamara, J.O. Cellular and molecular basis of epilepsy. J. Neurosci. 1994, 14, 3413–3425. [Google Scholar] [CrossRef] [PubMed]
- Aboul-Enein, M.N.; El-Azzouny, A.A.; Attia, M.I.; Maklad, Y.A.; Aboutabl, M.E.; Ragab, F.; El-Hamid, W.H.A.A. Anticonvulsant profiles of certain new 6-Aryl-9-substituted- 6,9-diazaspiro-[4.5]decane-8,10-diones and 1-Aryl-4-substituted- 1,4-diazaspiro[5.5]undecane-3,5-diones. Int. J. Mol. Sci. 2014, 15, 16911–16935. [Google Scholar] [CrossRef] [PubMed]
- Kamiński, K.; Rapacz, A.; Łuszczki, J.J.; Latacz, G.; Obniska, J.; Kieć-Kononowicz, K.; Filipek, B. Design, synthesis and biological evaluation of new hybrid anticonvulsants derived from N-benzyl-2-(2,5-dioxopyrrolidin-1-yl)propanamide and 2-(2,5-dioxopyrrolidin-1-yl)butanamide derivatives. Bioorg. Med. Chem. 2015, 23, 2548–2561. [Google Scholar] [CrossRef]
- Abd-Allah, W.H.; Aboutabl, M.E.; Aboul-Enein, M.N.; El-Azzouny, A.A.S. Synthesis, molecular modeling studies and anticonvulsant activity of certain (1-(benzyl (aryl) amino) cyclohexyl) methyl esters. Bioorg. Chem. 2017, 71, 135–145. [Google Scholar] [CrossRef]
- Sahu, M.; Siddiqui, N.; Naim, M.J.; Alam, O.; Yar, M.S.; Sharma, V.; Wakode, S. Design, Synthesis and Docking Study of Pyrimidine–Triazine Hybrids for GABA Estimation in Animal Epilepsy Models. Arch. Pharm. 2017, 350, 1–18. [Google Scholar] [CrossRef]
- Abd-Allah, W.H.; Osman, E.E.A.; Anwar, M.A.E.M.; Attia, H.N.; El Moghazy, S.M. Design, synthesis and docking studies of novel benzopyrone derivatives as anticonvulsants. Bioorg. Chem. 2020, 98, 103738. [Google Scholar] [CrossRef]
- Aboutabl, M.E.; Hassan, R.M.; El-Azzouny, A.A.S.; Aboul-Enein, M.N.; Abd-Allah, W.H. Design and synthesis of novel parabanic acid derivatives as anticonvulsants. Bioorg. Chem. 2020, 94, 103473. [Google Scholar] [CrossRef]
- Partap, S.; Yar, M.S.; Hassan, M.Z.; Akhtar, M.J.; Siddiqui, A.A. Design, Synthesis and Pharmacological Screening of Pyridazinone Hybrids as Anticonvulsant Agents. Arch. Pharm. 2017, 350, 1–14. [Google Scholar] [CrossRef]
- Mohammadi-Khanaposhtani, M.; Ahangar, N.; Sobhani, S.; Masihi, P.H.; Shakiba, A.; Saeedi, M.; Akbarzadeh, T. Design, synthesis, in vivo and in silico evaluation of new coumarin-1,2,4-oxadiazole hybrids as anticonvulsant agents. Bioorg. Chem. 2019, 89, 102989. [Google Scholar] [CrossRef] [PubMed]
- Ahangar, N.; Ayati, A.; Alipour, E.; Pashapour, A.; Foroumadi, A.; Emami, S. 1-[(2-arylthiazol-4-yl)methyl]azoles as a new class of anticonvulsants: Design, synthesis, in vivo screening and in silico drug-like properties. Chem. Biol. Drug Des. 2011, 78, 844–852. [Google Scholar] [CrossRef] [PubMed]
- Lankau, H.J.; Unverferth, K.; Grunwald, C.; Hartenhauer, H.; Heinecke, K.; Bernöster, K.; Dost, R.; Egerland, U.; Rundfeldt, C. New GABA-modulating 1,2,4-oxadiazole derivatives and their anticonvulsant activity. Eur. J. Med. Chem. 2007, 42, 873–879. [Google Scholar] [CrossRef] [PubMed]
- Faizi, M.; Sheikhha, M.; Ahangar, N.; Ghomi, H.T.; Shafaghi, B.; Shafiee, A.; Tabatabai, S.A. Design, synthesis and pharmacological evaluation of novel 2-[2-(2-Chlorophenoxy) phenyl]-1,3,4-oxadiazole derivatives as benzodiazepine receptor agonists. Iran. J. Pharm. Res. 2012, 11, 83–90. [Google Scholar] [CrossRef] [PubMed]
- Mohammadi-Khanaposhtani, M.; Shabani, M.; Faizi, M.; Aghaei, I.; Jahani, R.; Sharafi, Z.; Shamsaei Zafarghandi, N.; Mahdavi, M.; Akbarzadeh, T.; Emami, S.; et al. Design, synthesis, pharmacological evaluation and docking study of new acridone-based 1,2,4-oxadiazoles as potential anticonvulsant agents. Eur. J. Med. Chem. 2016, 112, 91–98. [Google Scholar] [CrossRef] [PubMed]
- Zolfagharian, F.; Razavi, B.M.; Hosseinzadeh, H. Anticonvulsant effect of Satureja hortensis aerial parts extracts in mice. Avicenna J. Phytomed. 2016, 6, 305–312. [Google Scholar] [CrossRef]
- Mokrov, G.V.; Savel’ev, V.L.; Voronina, T.A.; Litvinova, S.A.; Kovalev, I.G.; Nerobkova, L.N.; Dmitrienko, A.O.; Gudasheva, T.A.; Seredenin, S.B. Synthesis and Anticonvulsant Activity of N-Substituted 4-Amino-3-Nitrocoumarins. Pharm. Chem. J. 2019, 53, 118–124. [Google Scholar] [CrossRef]
- Wadelius, M.; Pirmohamed, M. Pharmacogenetics of warfarin: Current status and future challenges. Pharmacogen. J. 2007, 7, 99–111. [Google Scholar] [CrossRef]
- Kamali, F.; Pirmohamed, M. The future prospects of pharmacogenetics in oral anticoagulation therapy. Br. J. Clin. Pharmacol. 2006, 61, 746–751. [Google Scholar] [CrossRef]
- Pirmohamed, M. Warfarin: Almost 60 years old and still causing problems. Br. J. Clin. Pharmacol. 2006, 62, 509–511. [Google Scholar] [CrossRef]
- Trailokya, A. Acenocoumarol in Thromoembolic Disorders. Cardiovasc. Pharmacol. Open Access 2015, 4. [Google Scholar] [CrossRef]
- Lippi, G.; Gosselin, R.; Favaloro, E.J. Current and Emerging Direct Oral Anticoagulants: State-of-the-Art. Semin. Thromb. Hemost. 2019, 45, 490–501. [Google Scholar] [CrossRef] [PubMed]
- Székely, O.; Miyazawa, K.; Lip, G.Y.H. Current and emerging pharmacotherapy for ischemic stroke prevention in patients with atrial fibrillation. Expert Opin. Pharmacother. 2018, 19, 1999–2009. [Google Scholar] [CrossRef] [PubMed]
- Bavisotto, L.M.; Ellis, D.J.; Milner, P.G.; Combs, D.L.; Irwin, I.; Canafax, D.M. Tecarfarin, a novel vitamin K reductase antagonist, is not affected by CYP2C9 and CYP3A4 inhibition following concomitant administration of fluconazole in healthy participants. J. Clin. Pharmacol. 2011, 51, 561–574. [Google Scholar] [CrossRef] [PubMed]
- Albrecht, D.; Ellis, D.; Canafax, D.M.; Combs, D.; Druzgala, P.; Milner, P.G.; Midei, M.G. Pharmacokinetics and pharmacodynamics of tecarfarin, a novel vitamin K antagonist oral anticoagulant. Thromb. Haemost. 2017, 117, 706–717. [Google Scholar] [CrossRef] [PubMed]
- Albrecht, D.; Turakhia, M.P.; Ries, D.; Marbury, T.; Smith, W.; Dillon, D.; Milner, P.G.; Midei, M.G. Pharmacokinetics of tecarfarin and warfarin in patients with severe chronic kidney disease. Thromb. Haemost. 2017, 117, 2026–2033. [Google Scholar] [CrossRef] [PubMed]
- Bang, N.C.; Abyshev, A.Z.; Ivkin, D.Y. Synthesis and In Vivo Evaluation of New Coumarin Conjugates as Potential Indirect-Action Anticoagulants. Pharm. Chem. J. 2019, 53, 419–422. [Google Scholar] [CrossRef]
- Montagut-Romans, A.; Boulven, M.; Jacolot, M.; Moebs-Sanchez, S.; Hascoët, C.; Hammed, A.; Besse, S.; Lemaire, M.; Benoit, E.; Lattard, V.; et al. Synthesis and biological evaluation of C-3 aliphatic coumarins as vitamin K antagonists. Bioorg. Med. Chem. Lett. 2017, 27, 1598–1601. [Google Scholar] [CrossRef]
- Gebauer, M. Synthesis and structure-activity relationships of novel warfarin derivatives. Bioorg. Med. Chem. 2007, 15, 2414–2420. [Google Scholar] [CrossRef]
- Au, N.; Rettie, A.E. Pharmacogenomics of 4-hydroxycoumarin anticoagulants. Drug Metab. Rev. 2008, 40, 355–375. [Google Scholar] [CrossRef]
- Lei, L.; Xue, Y.B.; Liu, Z.; Peng, S.S.; He, Y.; Zhang, Y.; Fang, R.; Wang, J.P.; Luo, Z.W.; Yao, G.M. Coumarin derivatives from Ainsliaea fragrans and their anticoagulant activity. Sci. Rep. 2015, 5, 1–9. [Google Scholar] [CrossRef]
- World Health Organization (WHO). Diabetic. Available online: http://www.who.int/diabetic/en/ (accessed on 18 March 2020).
- Lebovitz, H.A. A new oral therapy for diabetes management: Alpha-glucosidase inhibition with acarbose. Clin. Diabetes 1995, 13, 99–103. [Google Scholar] [CrossRef]
- Playford, R.J.; Pither, C.; Gao, R.; Middleton, S.J. Use of the alpha-glucosidase inhibitor acarbose in patients with ‘Middleton syndrome’: Normal gastric anatomy but with accelerated gastric emptying causing postprandial reactive hypoglycemia and diarrhea. Can. J. Gastroenterol. 2013, 27, 403–404. [Google Scholar] [CrossRef] [PubMed]
- Menteşe, E.; Baltaş, N.; Bekircan, O. Synthesis and kinetics studies of N′-(2-(3,5-disubstituted-4H-1,2,4-triazol-4-yl)acetyl)-6/7/8-substituted-2-oxo-2H-chromen-3-carbohydrazide derivatives as potent antidiabetic agents. Arch. Pharm. 2019, 352, 2–8. [Google Scholar] [CrossRef] [PubMed]
- Kharb, R.; Sharma, P.C.; Yar, M.S. Pharmacological significance of triazole scaffold. J. Enzyme Inhib. Med. Chem. 2011, 26, 1–21. [Google Scholar] [CrossRef]
- Kaur, P.; Chawla, A. 1,2,4-Triazole: A Review of Pharmacological Activities. Int. Res. J. Pharm. 2017, 8, 10–29. [Google Scholar] [CrossRef]
- Pillai, R.R.; Karrouchi, K.; Fettach, S.; Armaković, S.; Armaković, S.J.; Brik, Y.; Taoufik, J.; Radi, S.; El Abbes Faouzi, M.; Ansar, M. Synthesis, spectroscopic characterization, reactive properties by DFT calculations, molecular dynamics simulations and biological evaluation of Schiff bases tethered 1,2,4-triazole and pyrazole rings. J. Mol. Struct. 2019, 1177, 47–54. [Google Scholar] [CrossRef]
- Dhameja, M.; Gupta, P. Synthetic heterocyclic candidates as promising α-glucosidase inhibitors: An overview. Eur. J. Med. Chem. 2019, 176, 343–377. [Google Scholar] [CrossRef]
- Taha, M.; Shah, S.A.A.; Afifi, M.; Imran, S.; Sultan, S.; Rahim, F.; Khan, K.M. Synthesis, α-glucosidase inhibition and molecular docking study of coumarin based derivatives. Bioorg. Chem. 2018, 77, 586–592. [Google Scholar] [CrossRef]
- Salar, U.; Taha, M.; Khan, K.M.; Ismail, N.H.; Imran, S.; Perveen, S.; Gul, S.; Wadood, A. Syntheses of new 3-thiazolyl coumarin derivatives, in vitro α-glucosidase inhibitory activity and molecular modeling studies. Eur. J. Med. Chem. 2016, 122, 196–204. [Google Scholar] [CrossRef]
- Hu, Y.; Wang, B.; Yang, J.; Liu, T.; Sun, J.; Wang, X. Synthesis and biological evaluation of 3-arylcoumarin derivatives as potential anti-diabetic agents. J. Enzyme Inhib. Med. Chem. 2019, 34, 15–30. [Google Scholar] [CrossRef] [PubMed]
- Asgari, M.S.; Mohammadi-Khanaposhtani, M.; Kiani, M.; Ranjbar, P.R.; Zabihi, E.; Pourbagher, R.; Rahimi, R.; Faramarzi, M.A.; Biglar, M.; Larijani, B.; et al. Biscoumarin-1,2,3-triazole hybrids as novel anti-diabetic agents: Design, synthesis, in vitro α-glucosidase inhibition, kinetic and docking studies. Bioorg. Chem. 2019, 92, 103206. [Google Scholar] [CrossRef] [PubMed]
- Wang, G.; Peng, Z.; Li, X.; Li, J. Synthesis, in vitro evaluation and molecular docking studies of novel triazine-triazole derivatives as potential α-glucosidase inhibitors. Eur. J. Med. Chem. 2017, 125, 423–429. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, S.; Nur-e-Alam, M.; Parveen, I.; Coles, S.J.; Hafizur, R.M.; Hameed, A.; Orton, J.B.; Threadgill, M.D.; Yousaf, M.; Alqahtani, A.M.; et al. Stimulation of insulin secretion by 5-methylcoumarins and its sulfur analogues isolated from Clutia lanceolata Forssk. Phytochemistry 2020, 170, 112213. [Google Scholar] [CrossRef] [PubMed]
- Ford, R.A.; Hawkins, D.R.; Mayo, B.C.; Api, A.M. The in vivo dermal absorption and metabolism of [4-14C]coumarin by rats and by human volunteers under simulated conditions of use in fragrances. Food Chem. Toxicol. 2001, 39, 153–162. [Google Scholar] [CrossRef]
- Azim, S.A.; Al-Hazmy, S.M.; Ebeid, E.M.; El-Daly, S.A. A new coumarin laser dye 3-(benzothiazol-2-yl)-7-hydroxycoumarin. Opt. Laser Technol. 2005, 37, 245–249. [Google Scholar] [CrossRef]
- Vocanson, M.; Valeyrie, M.; Rozières, A.; Hennino, A.; Floc’h, F.; Gard, A.; Nicolas, J.F. Lack of evidence for allergenic properties of coumarin in a fragrance allergy mouse model. Contact Dermatitis 2007, 57, 361–364. [Google Scholar] [CrossRef] [PubMed]
- Xiao, H.; Chen, K.; Cui, D.; Jiang, N.; Yin, G.; Wang, J.; Wang, R. Two novel aggregation-induced emission active coumarin-based Schiff bases and their applications in cell imaging. New J. Chem. 2014, 38, 2386–2393. [Google Scholar] [CrossRef]
- Srinivasan, G.; Chen, J.; Parisi, J.; Brückner, C.; Yao, X.; Lei, Y. An Injectable PEG-BSA-Coumarin-GOx Hydrogel for Fluorescence Turn-on Glucose Detection. Appl. Biochem. Biotechnol. 2015, 177, 1115–1126. [Google Scholar] [CrossRef]
- Song, P.P.; Zhao, J.; Liu, Z.L.; Duan, Y.B.; Hou, Y.P.; Zhao, C.Q.; Wu, M.; Wei, M.; Wang, N.H.; Lv, Y.; et al. Evaluation of antifungal activities and structure–activity relationships of coumarin derivatives. Pest Manag. Sci. 2017, 73, 94–101. [Google Scholar] [CrossRef] [PubMed]
- Gandioso, A.; Contreras, S.; Melnyk, I.; Oliva, J.; Nonell, S.; Velasco, D.; García-Amorós, J.; Marchán, V. Development of Green/Red-Absorbing Chromophores Based on a Coumarin Scaffold That Are Useful as Caging Groups. J. Org. Chem. 2017, 82, 5398–5408. [Google Scholar] [CrossRef] [PubMed]
- Gualandi, A.; Rodeghiero, G.; Della Rocca, E.; Bertoni, F.; Marchini, M.; Perciaccante, R.; Jansen, T.P.; Ceroni, P.; Cozzi, P.G. Application of coumarin dyes for organic photoredox catalysis. Chem. Commun. 2018, 54, 10044–10047. [Google Scholar] [CrossRef] [PubMed]
- Liu, D.; Yang, F.; Xiong, F.; Gu, N. The smart drug delivery system and its clinical potential. Theranostics 2016, 6, 1306–1323. [Google Scholar] [CrossRef] [PubMed]
- Silva, J.M.; Silva, E.; Reis, R.L. Light-triggered release of photocaged therapeutics—Where are we now? J. Control. Release 2019, 298, 154–176. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Mu, J.; Xing, B. Photoactivated drug delivery and bioimaging. Wiley Interdiscip. Rev. Nanomed. Nanobiotechnol. 2017, 9, e1408. [Google Scholar] [CrossRef]
- Velema, W.A.; Szymanski, W.; Feringa, B.L. Photopharmacology: Beyond proof of principle. J. Am. Chem. Soc. 2014, 136, 2178–2191. [Google Scholar] [CrossRef]
- Brieke, C.; Rohrbach, F.; Gottschalk, A.; Mayer, G.; Heckel, A. Light-controlled tools. Angew. Chem. Int. Ed. 2012, 51, 8446–8476. [Google Scholar] [CrossRef]
- Fournier, L.; Aujard, I.; Le Saux, T.; Maurin, S.; Beaupierre, S.; Baudin, J.B.; Jullien, L. Coumarinylmethyl caging groups with redshifted absorption. Chem. A Eur. J. 2013, 19, 17494–17507. [Google Scholar] [CrossRef]
- Fournier, L.; Gauron, C.; Xu, L.; Aujard, I.; Le Saux, T.; Gagey-Eilstein, N.; Maurin, S.; Dubruille, S.; Baudin, J.B.; Bensimon, D.; et al. A blue-absorbing photolabile protecting group for in vivo chromatically orthogonal photoactivation. ACS Chem. Biol. 2013, 8, 1528–1536. [Google Scholar] [CrossRef]
- Bojtár, M.; Kormos, A.; Kis-Petik, K.; Kellermayer, M.; Kele, P. Green-Light Activatable, Water-Soluble Red-Shifted Coumarin Photocages. Org. Lett. 2019, 21, 9410–9414. [Google Scholar] [CrossRef]
- Iturmendi, A.; Theis, S.; Maderegger, D.; Monkowius, U.; Teasdale, I. Coumarin-Caged Polyphosphazenes with a Visible-Light Driven On-Demand Degradation. Macromol. Rapid Commun. 2018, 39, 1–6. [Google Scholar] [CrossRef]
- Sarkar, A.R.; Heo, C.H.; Xu, L.; Lee, H.W.; Si, H.Y.; Byun, J.W.; Kim, H.M. A ratiometric two-photon probe for quantitative imaging of mitochondrial pH values. Chem. Sci. 2016, 7, 766–773. [Google Scholar] [CrossRef]
- Gong, H.L.; Jiang, Y.; Hou, R.C.; Ding, X.Q. A Sensitive and Selective Fluorescent Coumarin-Based Probe for Detection of Hypochlorite ion and its Application to Cellular Imaging. J. Fluoresc. 2016, 26, 403–406. [Google Scholar] [CrossRef] [PubMed]
- Hu, Z.Q.; Sun, L.L.; Gu, Y.Y.; Jiang, Y. A sensitive and selective fluorescent probe for detection of glutathione in the presence of Cu2+ and its application to biological imaging. Sens. Actuators B Chem. 2015, 212, 220–224. [Google Scholar] [CrossRef]
- Shangguan, M.; Jiang, X.; Lu, Z.; Zou, W.; Chen, Y.; Xu, P.; Pan, Y.; Hou, L. A coumarin-based fluorescent probe for hypochlorite ion detection in environmental water samples and living cells. Talanta 2019, 202, 303–307. [Google Scholar] [CrossRef] [PubMed]
- Tang, Y.; Li, Y.; Han, J.; Mao, Y.; Ni, L.; Wang, Y. A coumarin based fluorescent probe for rapidly distinguishing of hypochlorite and copper (II) ion in organisms. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2019, 208, 299–308. [Google Scholar] [CrossRef] [PubMed]
- Starzak, K.; Matwijczuk, A.; Creaven, B.; Matwijczuk, A.; Wybraniec, S.; Karcz, D. Fluorescence quenching-based mechanism for determination of hypochlorite by coumarin-derived sensors. Int. J. Mol. Sci. 2019, 20, 281. [Google Scholar] [CrossRef] [PubMed]
- Bekhradnia, A.; Domehri, E.; Khosravi, M. Novel coumarin-based fluorescent probe for selective detection of Cu(II). Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2016, 152, 18–22. [Google Scholar] [CrossRef]
- He, G.; Liu, X.; Xu, J.; Ji, L.; Yang, L.; Fan, A.; Wang, S.; Wang, Q. Synthesis and application of a highly selective copper ions fluorescent probe based on the coumarin group. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2018, 190, 116–120. [Google Scholar] [CrossRef]
- Mani, K.S.; Rajamanikandan, R.; Murugesapandian, B.; Shankar, R.; Sivaraman, G.; Ilanchelian, M.; Rajendran, S.P. Coumarin based hydrazone as an ICT-based fluorescence chemosensor for the detection of Cu2+ ions and the application in HeLa cells. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2019, 214, 170–176. [Google Scholar] [CrossRef]
- Li, H.; Sun, X.; Zheng, T.; Xu, Z.; Song, Y.; Gu, X. Coumarin-based multifunctional chemosensor for arginine/lysine and Cu2+/Al3+ ions and its Cu2+ complex as colorimetric and fluorescent sensor for biothiols. Sens. Actuators B Chem. 2019, 279, 400–409. [Google Scholar] [CrossRef]
- Chen, X.Z.; Ma, X.D.; Wang, H.M.; Wang, M.; Zhang, Y.Y.; Gao, G.; Liu, J.J.; Hou, S.C. A coumarin-based colorimetric and fluorescent dual probe for palladium(II) ions that can be used in live cells. New J. Chem. 2017, 41, 8026–8030. [Google Scholar] [CrossRef]
- Cheng, X.; Qu, S.; Xiao, L.; Li, W.; He, P. Thioacetalized coumarin-based fluorescent probe for mercury(II): Ratiometric response, high selectivity and successful bioimaging application. J. Photochem. Photobiol. A Chem. 2018, 364, 503–509. [Google Scholar] [CrossRef]
- Warrier, S.; Kharkar, P.S. Highly selective on-off fluorescence recognition of Fe3+ based on a coumarin derivative and its application in live-cell imaging. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2018, 188, 659–665. [Google Scholar] [CrossRef] [PubMed]
- Jiao, S.; Wang, X.; Sun, Y.; Zhang, L.; Sun, W.; Sun, Y.; Wang, X.; Ma, P.; Song, D. A novel fluorescein-coumarin-based fluorescent probe for fluoride ions and its applications in imaging of living cells and zebrafish in vivo. Sens. Actuators B Chem. 2018, 262, 188–194. [Google Scholar] [CrossRef]
- Yao, K.; Chang, Y.; Li, B.; Yang, H.; Xu, K. A novel coumarin-based fluorescent sensor for Ca2+ and sequential detection of F− and its live cell imaging. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2019, 216, 385–394. [Google Scholar] [CrossRef] [PubMed]
- Reddy, T.S.; Choi, M.S. Dicyanovinylcoumarin as a turn-on fluorescent sensor for cyanide ion. J. Photochem. Photobiol. A Chem. 2018, 351, 108–114. [Google Scholar] [CrossRef]
- Zhang, Q.; Zhang, Y.; Ding, S.; Zhang, H.; Feng, G. A near-infrared fluorescent probe for rapid, colorimetric and ratiometric detection of bisulfite in food, serum and living cells. Sens. Actuators B Chem. 2015, 211, 377–384. [Google Scholar] [CrossRef]
- Fazio, T.; Warner, C.R. A review of sulphites in foods: Analytical methodologyand reported findings. Food Addit. Contam. 1990, 7, 433–454. [Google Scholar] [CrossRef]
- Taylor, S.L.; Highley, N.A.; Bush, R.K. Bush, Sulfites in foods: Uses, analytical methods, residues, fate, exposure assessment, metabolism, toxicity and hypersensitivity. Adv. Food Res. 1986, 30, 1–76. [Google Scholar] [CrossRef]
- Vally, H.; Misso, N.L.; Madan, V. Clinical effects of sulphite additives. Clin. Exp. Allergy 2009, 39, 1643–1651. [Google Scholar] [CrossRef] [PubMed]
- Hansell, A.; Oppenheimer, C. Health hazards from volcanic gases: A systematicliterature review. Arch. Environ. Health 2004, 59, 628–639. [Google Scholar] [CrossRef]
- Nair, R.R.; Raju, M.; Bhai, S.; Raval, I.H.; Haldar, S.; Ganguly, B.; Chatterjee, P.B. Estimation of bisulfate in edible plant foods, dog urine and drugs: Picomolar level detection and bio-imaging in living organisms. Analyst 2019, 144, 5724–5737. [Google Scholar] [CrossRef] [PubMed]
- Fujioka, N.; Fritz, V.; Upadhyaya, P.; Kassie, F.; Hetch, S.S. Research on cruciferous vegetables, indole-3-carbinol and cancer prevention: A tribute to Lee W. Wattenberg. Mol. Nutr. Food Res. 2016, 60, 1228–1238. [Google Scholar] [CrossRef] [PubMed]
- Keck, A.-S.; Finley, J.W. Crouciferous Vegetables: Cancer Protective Mechanism of Glcisunolate Hydrolisi Products and Selenium. Crucif. Veg. Cancer Prot. 2004, 3, 5–12. [Google Scholar] [CrossRef]
- Botti, M.G.; Taylor, M.G.; Botting, N.P. Studies on the mechanism of Myrosinase: Investigation of the effect of glycosil acceptors on enzyme activity. J. Biol. Chem. 1995, 35, 20530–20535. [Google Scholar] [CrossRef]
- Marx, J.A.; Hockberger, R.S.; Walls, R.M. Rosen’s Emergency Medicines, 8th ed.; Elsevier: Amsterdam, The Netherlands, 2014. [Google Scholar]
- Yang, L.; Ding, W.; Xu, Y.; Wu, D.; Li, S.; Chen, J.; Guo, B. New insights into the antibacterial activity of hydroxycoumarins against ralstonia solanacearum. Molecules 2016, 21, 468. [Google Scholar] [CrossRef]
- Genin, S. Molecular traits controlling host range and adaptation to plants in Ralstonia solanacearum. New Phytol. 2010, 187, 920–928. [Google Scholar] [CrossRef]
- De Oliveira Monteiro, F. Environmental Cues Controlling the Pathogenicity of “Ralstonia Solanacearum” on Plants. Master’s Thesis, Universitat de Barcelona, Barcelona, Spain, 2013. [Google Scholar]
- Yai, J.; Allen, C. The plant pathogen Ralstonia solanacearum needs aerotaxis for normal biofilm formation and interactions with its tomato host. J. Bacteriol. 2007, 189, 6415–6524. [Google Scholar] [CrossRef]
- Ramírez-Pelayo, C.; Martínez-Quiñones, J.; Gil, J.; Durango, D. Coumarins from the peel of citrus grown in Colombia: Composition, elicitation and antifungal activity. Heliyon 2019, 5. [Google Scholar] [CrossRef]
- Piasecka, A.; Jedrzejczak-Rey, N.; Bednarek, P. Secondary metabolites in plant innate immunity: Conserved function of divergent chemicals. New Phytol. 2015, 206, 948–964. [Google Scholar] [CrossRef]
- VanEtten, H.; Mansfield, J.; Bailey, J.; Farmer, E. Two classes of plant antibiotics: Phytoalexins versus phytoanticipins. Plant Cell 1994, 6, 1191–1192. [Google Scholar] [CrossRef] [PubMed]
- Yu, X.; Teng, P.; Zhang, Y.L.; Xu, Z.J.; Zhang, M.Z.; Zhang, W.H. Design, synthesis and antifungal activity evaluation of coumarin-3-carboxamide derivatives. Fitoterapia 2018, 127, 387–395. [Google Scholar] [CrossRef] [PubMed]
- Reddy, N.S.; Mallireddigari, M.R.; Cosenza, S.; Gumireddy, K.; Bell, S.C.; Premkumar Reddy, E.; Ramana Reddy, M. Synthesis of new coumarin 3-(N-aryl) sulfonamides and their anticancer activity. Bioorg. Med. Chem. Lett. 2004, 14, 4093–4097. [Google Scholar] [CrossRef] [PubMed]
- Wehrstedt, K.D.; Wandrey, P.A.; Heitkamp, D. Explosive properties of 1-hydroxybenzotriazoles. J. Hazard. Mater. 2005, 126, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Grande, F.; Aiello, F.; Grazie, O.D.; Brizzi, A.; Garofaloa, A.; Neamati, N. Synthesis and antitumor activities of a series of novel quinoxalinhydrazides. Bioorg. Med. Chem. 2007, 15, 288–294. [Google Scholar] [CrossRef] [PubMed]
- Zou, M.; Lu, J.R.; Chen, H.P. Synthesis, characterization and antibacterial activities of N-[1-(substituted phenyl) ethyl]-2-hydroxybenzohydrazide. Chem. J. Chin. Univ. 2012, 33, 2235–2238. [Google Scholar] [CrossRef]
- Yang, G.; Jin, Q.; Xu, C.; Fan, S.; Wang, C.; Xie, P. Synthesis, characterization and antifungal activity of coumarin-functionalized chitosan derivatives. Int. J. Biol. Macromol. 2018, 106, 179–184. [Google Scholar] [CrossRef]
- Lippard, S.J.; Berg, J.M. Principles of Bioinorganic Chemistry; University Science Books: Mill Valley, CA, USA, 1999; ISBN 0-935702-73-3. [Google Scholar]
- Karaliota, A.; Kretsi, O.; Tzougraki, C. Synthesis and characterization of a binuclear coumarin-3-carboxylate copper(II) complex. J. Inorg. Biochem. 2001, 84, 33–37. [Google Scholar] [CrossRef]
- MacLean, L.; Karcz, D.; Jenkins, H.; McClean, S.; Devereux, M.; Howe, O.; Pereira, M.D.; May, N.V.; Enyedy, É.A.; Creaven, B.S. Copper(II) complexes of coumarin-derived Schiff base ligands: Pro- or antioxidant activity in MCF-7 cells? J. Inorg. Biochem. 2019, 197, 110702. [Google Scholar] [CrossRef]
- Dharmaraja, A.T. Role of Reactive Oxygen Species (ROS) in Therapeutics and Drug Resistance in Cancer and Bacteria. J. Med. Chem. 2017, 60, 3221–3240. [Google Scholar] [CrossRef] [PubMed]
- Reczek, C.R.; Chandel, N.S. The Two Faces of Reactive Oxygen Species in Cancer. Annu. Rev. Cancer Biol. 2017, 1, 79–98. [Google Scholar] [CrossRef]
- Barrera, G.; Peroxidation, L. Oxidative Stress and Lipid Peroxidation Products in cancer progression and therapy. ISRN Oncol. 2012, 2012, 137289. [Google Scholar] [CrossRef]
- Dwyer, F.P.; Mayhew, E.; Roe, E.M.; Shulman, A. Inhibition of Landschutz Ascites Tumour Growth By metal chelates derived from 3,4,7,8-tetramethyl- 1,1 0-phenanthroline. Br. J. Cancer 1965, 19, 195–199. [Google Scholar] [CrossRef] [PubMed]
- Qin, Q.P.; Wang, Z.F.; Huang, X.L.; Tan, M.X.; Zou, B.Q.; Liang, H. Strong in vitro and vivo cytotoxicity of novel organoplatinum(II) complexes with quinoline-coumarin derivatives. Eur. J. Med. Chem. 2019, 184, 111751. [Google Scholar] [CrossRef]
- Aslkhademi, S.; Noshiranzadeh, N.; Sadjadi, M.S.; Mehrani, K.; Farhadyar, N. Synthesis, crystal structure and investigation of the catalytic and spectroscopic properties of a Zn(II) complex with coumarin-hydrazone ligand. Polyhedron 2019, 160, 115–122. [Google Scholar] [CrossRef]
- Jaiswal, S.; Bhattacharya, K.; Sullivan, M.; Walsh, M.; Creaven, B.S.; Laffir, F.; Duffy, B.; McHale, P. Non-cytotoxic antibacterial silver-coumarin complex doped sol-gel coatings. Colloids Surf. B Biointerfaces 2013, 102, 412–419. [Google Scholar] [CrossRef]
- Vekariya, R.H.; Patel, H.D. Recent Advances in the Synthesis of Coumarin Derivatives via Knoevenagel Condensation: A Review. Synth. Commun. 2014, 44, 2756–2788. [Google Scholar] [CrossRef]
- Salem, M.A.; Helal, M.H.; Gouda, M.A.; Ammar, Y.A.; El-Gaby, M.S.A.; Abbas, S.Y. An overview on synthetic strategies to coumarins. Synth. Commun. 2018, 48, 1534–1550. [Google Scholar] [CrossRef]
- Hartman, R.L.; McMullen, J.P.; Jensen, K.F. Deciding Wether To Go with the Flow: Evaluating the Merits of Flow Reactors for Synthesis. Angew. Chem. Int. Ed. 2011, 50, 7502–7519. [Google Scholar] [CrossRef]
- Taghavi-Moghadam, S.; Kleemann, A.; Golbig,, K.G. Microreaction Technology as a Novel Approach to Drug Design, Process Development and Reliability. Org. Process Res. Dev. 2001, 5, 652–658. [Google Scholar] [CrossRef]
- Gutmann, B.; Cantillo, D.; Kapper, C.O. Continuous-Flow Technology—A Tool fo the Safe Manifacturing of Active Pharmaceutical Ingredients. Angew. Chem. Int. Ed. 2015, 54, 6688–6728. [Google Scholar] [CrossRef]
- Li, X.; Chen, A.; Zhou, Y.; Huang, L.; Fang, Z.; Gan, H.; Guo, K. Two-stage flow synthesis of coumarin via O-acetylation of salicylaldehyde. J. Flow Chem. 2015, 5, 82–86. [Google Scholar] [CrossRef]
- Mhiri, C.; Ternane, R.; Hamdi, N.; Baklouti, L. Synthesis of coumarin derivative using polymer supported reagents. Eur. J. Chem. 2018, 9, 89–91. [Google Scholar] [CrossRef]
- Metternich, J.B.; Gilmour, R. One Photocatalyst, n Activation Modes Strategy for Cascade Catalysis: Emulating Coumarin Biosynthesis with (-)-Riboflavin. J. Am. Chem. Soc. 2016, 138, 1040–1045. [Google Scholar] [CrossRef]
- Song, D.; Wang, C.M.; Ye, Z.P.; Xia, P.J.; Deng, Z.X.; Xiao, J.A.; Xiang, H.Y.; Yang, H. Visible-light-driven, photoredox-catalyzed cascade of ortho-hydroxycinnamic esters to access 3-fluoroalkylated coumarins. J. Org. Chem. 2019, 84, 7480–7487. [Google Scholar] [CrossRef]
- Eivgi, O.; Sutar, R.L.; Reany, O.; Lemcoff, N.G. Bichromatic Photosynthesis of Coumarins by UV Filter-Enabled Olefin Metathesis. Adv. Synth. Catal. 2017, 359, 2352–2357. [Google Scholar] [CrossRef]
- Reichardt, C.; Welton, T. Solvents and Solvent Effects in Organic Chemistry, 4th ed.; Wiley-VCH: Weinheim, Germany, 2011. [Google Scholar]
- Horning, E.C. Organic Syntheses, Coll. Vol. III; John Wiley & Sons, Inc.: New York, NY, USA, 1955. [Google Scholar]
- Kalita, P.; Kumar, R. Solvent-free coumarin synthesis via Pechmann reaction using solid catalysts. Microporous Mesoporous Mater. 2012, 149, 1–9. [Google Scholar] [CrossRef]
- Loupy, A. Solvent-Free Reactions. In Modern Solvents in Organic Synthesis; Knochel, P., Ed.; Springer: Berlin, Germany, 1999. [Google Scholar]
- Sugino, T.; Tanaka, K. Solvent-free coumarin synthesis. Chem. Lett. 2001, 110–111. [Google Scholar] [CrossRef]
- Ebrahimi, S.E.S.; Ghadirian, P.; Emtiazi, H.; Yahya-Meymandi, A.; Saeedi, M.; Mahdavi, M.; Nadri, H.; Moradi, A.; Sameem, B.; Vosooghi, M.; et al. Hetero-annulated coumarins as new AChE/BuChE inhibitors: Synthesis and biological evaluation. Med. Chem. Res. 2016, 25, 1831–1841. [Google Scholar] [CrossRef]
- Emtiazi, H.; Amrollahi, M.A.; Mirjalili, B.B.F. Nano-silica sulfuric acid as an efficient catalyst for the synthesis of substituted pyrazoles. Arab. J. Chem. 2015, 8, 793–797. [Google Scholar] [CrossRef]
- Kour, M.; Paul, S.; Clark, J.H.; Gupta, V.K.; Kant, R. Preparation and characterization of Lewis acid grafted sulfonated carbon@titania composites for the multicomponent synthesis of 4H-pyrimido[2,1-b]benzothiazoles and benzoxanthenones under solvent-free conditions. J. Mol. Catal. A Chem. 2016, 411, 299–310. [Google Scholar] [CrossRef]
- Kour, M.; Paul, S. A green and convenient approach for the one-pot solvent-free synthesis of coumarins and β-amino carbonyl compounds using Lewis acid grafted sulfonated carbon@titania composite. Monatshefte fur Chem. 2017, 148, 327–337. [Google Scholar] [CrossRef]
- Ghomi, J.S.; Akbarzadeh, Z. Ultrasonic accelerated Knoevenagel condensation by magnetically recoverable MgFe2O4 nanocatalyst: A rapid and green synthesis of coumarins under solvent-free conditions. Ultrason. Sonochem. 2018, 40, 78–83. [Google Scholar] [CrossRef]
- Pakdel, S.; Akhlaghinia, B.; Mohammadinezhad, A. Fe3O4@Boehmite-NH2-CoII NPs: An Environment Friendly Nanocatalyst for Solvent Free Synthesis of Coumarin Derivatives Through Pechmann Condensation Reaction. Chem. Afr. 2019, 2, 367–376. [Google Scholar] [CrossRef]
- Jenkins, H.D.B. Ionic liquids—An overview. Sci. Prog. 2011, 94, 265–297. [Google Scholar] [CrossRef]
- Earle, M.J.; Seddon, K.R. Ionic liquids. Green solvents for the future. Pure Appl. Chem. 2000, 72, 1391–1398. [Google Scholar] [CrossRef]
- Deng, Y.; Beadham, I.; Ghacre, M.; Costa Gomes, M.F.; Gathergood, N.; Husson, P.; Lègeret, B.; Quilty, B.; Sancelme, M.; Besse-Hoggan, P. When can ionic liquids be considered readily biodegradable? Biodegradation pathways of pyridinium, pyrrolidinium and ammonium-based ionic liquids. Green Chem. 2015, 17, 1479–1491. [Google Scholar] [CrossRef]
- Coleman, D.; Gathergood, N. Biodegradation studies of ionic liquids. Chem. Soc. Rev. 2010, 39, 600–637. [Google Scholar] [CrossRef]
- Egorova, K.S.; Ananikov, V.P. Toxicity of ionic liquids: Eco(cyto)activity as complicated but unavoidable parameter for task-specific optimization. ChemSusChem 2014, 7, 336–360. [Google Scholar] [CrossRef]
- Mahato, S.; Santra, S.; Chatterjee, R.; Zyryanov, G.V.; Hajra, A.; Majee, A. Brønsted acidic ionic liquid-catalyzed tandem reaction: An efficient approach towards regioselective synthesis of pyrano[3,2-: C] coumarins under solvent-free conditions bearing lower E-factors. Green Chem. 2017, 19, 3282–3295. [Google Scholar] [CrossRef]
- Cole, A.C.; Jensen, J.L.; Ntai, I.; Tran, K.L.T.; Weaver, K.J.; Forbes, D.C.; Davis, J.H. Novel brønsted acidic ionic liquids and their use as dual solvent-catalysts. J. Am. Chem. Soc. 2002, 124, 5962–5963. [Google Scholar] [CrossRef]
- Moosavi-Zare, A.R.; Zolfigol, M.A.; Khaledian, O.; Khakyzadeh, V. Condensation of 2-naphtol with arylaldehydes using acetic acid functionalized ionic liquids as highly efficient and reusable catalysts. Chin. J. Catal. 2014, 35, 573–578. [Google Scholar] [CrossRef]
- Moosavi-Zare, A.R.; Zolfigol, M.A.; Khaledian, O.; Khakyzadeh, V.; Farahani, M.D.; Kruger, H.G. Tandem Knoevenagel-Michael-cyclocondensation reactions of malononitrile, various aldehydes and dimedone using acetic acid functionalized ionic liquid. New J. Chem. 2014, 38, 2342–2347. [Google Scholar] [CrossRef]
- Noroozizadeh, E.; Moosavi-Zare, A.R.; Zolfigol, M.A.; Zarei, M.; Karamian, R.; Asadbegy, M.; Yari, S.; Farida, S.H.M. Synthesis of bis-coumarins over acetic acid functionalized poly(4-vinylpyridinum) bromide (APVPB) as a green and efficient catalyst under solvent-free conditions and their biological activity. J. Iran. Chem. Soc. 2018, 15, 471–481. [Google Scholar] [CrossRef]
- Lidstrom, P.; Tierney, J.; Wathey, B.; Westman, J. Microwave-assisted organic synthesis—A review. Tetra 2001, 57, 9225–9283. [Google Scholar] [CrossRef]
- Gabriel, C.; Gabriel, S.; Grant, E.H.; Halstead, B.S.J.; Michael P Mingos, D. Dielectric parameters relevant to microwave dielectric heating. Chem. Soc. Rev. 1998, 27, 213–223. [Google Scholar] [CrossRef]
- Singh, V.; Singh, J.; Kaur, K.P.; Kad, G.L. Acceleration of the Pechmann Reaction by Microwave Irradiation: Application to the Preparation of Coumarins. J. Chem. Res. Part S 1997, 58–59. [Google Scholar] [CrossRef]
- Saidi, M.R.; Bigdeli, K. Microwave Promoted and Improved Thermal Synthesis of Pyranocoumarins and Furocoumarins. J. Chem. Res. Part S 1998, 800–801. [Google Scholar] [CrossRef]
- Valizadeh, H.; Shockravi, A. Task-specific ionic liquid as reagent and reaction medium for the one-pot Horner-Wadsworth-Emmons-type reaction under microwave irradiation. Synth. Commun. 2009, 39, 4341–4349. [Google Scholar] [CrossRef]
- Valizadeh, H.; Vaghefi, S. One-Pot wittig and knoevenagel reactions in ionic liquid as convenient methods for the synthesis of coumarin derivatives. Synth. Commun. 2009, 39, 1666–1678. [Google Scholar] [CrossRef]
- Fiorito, S.; Epifano, F.; Taddeo, V.A.; Genovese, S. Ytterbium triflate promoted coupling of phenols and propiolic acids: Synthesis of coumarins. Tetrahedron Lett. 2016, 57, 2939–2942. [Google Scholar] [CrossRef]
- Genovese, S.; Fiorito, S.; Specchiulli, M.C.; Taddeo, V.A.; Epifano, F. Microwave-assisted synthesis of xanthones promoted by ytterbium triflate. Tetrahedron Lett. 2015, 56, 847–850. [Google Scholar] [CrossRef]
- Mollica, A.; Genovese, S.; Pinnen, F.; Stefanucci, A.; Curini, M.; Epifano, F. Ytterbium triflate catalysed Meerwein-Ponndorf-Verley (MPV) reduction. Tetrahedron Lett. 2012, 53, 890–892. [Google Scholar] [CrossRef]
- Wang, L.; Xia, J.; Tian, H.; Qian, C.; Ma, Y. Synthesis of coumarin by Yb(OTf)3 catalyzed Pechmann reaction under the solvent-free conditions. Indian J. Chem. Sect. B Org. Med. Chem. 2003, 42, 2097–2099. [Google Scholar] [CrossRef]
- Sabou, R.; Hoelderich, W.F.; Ramprasad, D.; Weinand, R. Synthesis of 7-hydroxy-4-methylcoumarin via the Pechmann reaction with Amberlyst ion-exchange resins as catalysts. J. Catal. 2005, 232, 34–37. [Google Scholar] [CrossRef]
- Bouasla, S.; Amaro-Gahete, J.; Esquivel, D.; López, M.I.; Jiménez-Sanchidrián, C.; Teguiche, M.; Romero-Salguero, F.J. Coumarin derivatives solvent-free synthesis under microwave irradiation over heterogeneous solid catalysts. Molecules 2017, 22, 2072. [Google Scholar] [CrossRef]
- Konrádová, D.; Kozubíková, H.; Doležal, K.; Pospíšil, J. Microwave-Assisted Synthesis of Phenylpropanoids and Coumarins: Total Synthesis of Osthol. Eur. J. Org. Chem. 2017, 2017, 5204–5213. [Google Scholar] [CrossRef]
- Gu, C.X.; Chen, W.W.; Xu, B.; Xu, M.H. Synthesis of indolo[2,3-c]coumarins and indolo[2,3-c]quinolinones via microwave-assisted base-free intramolecular cross dehydrogenative coupling. Tetrahedron 2019, 75, 1605–1611. [Google Scholar] [CrossRef]
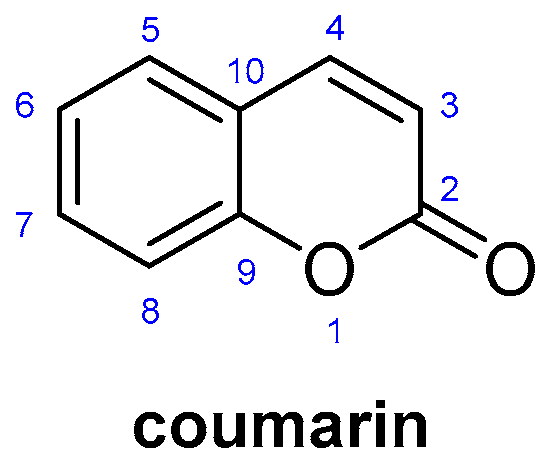
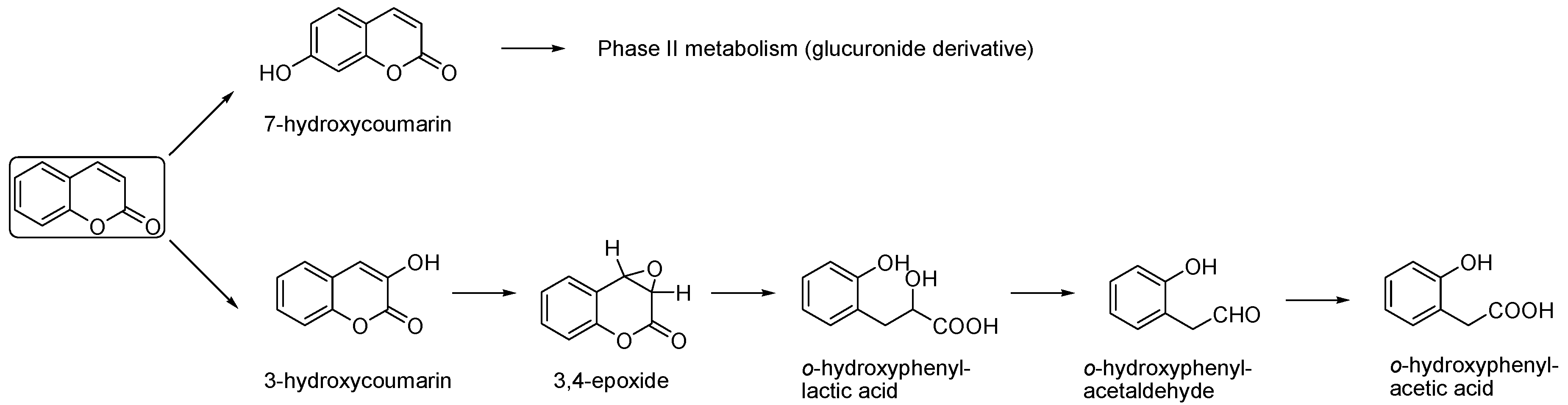

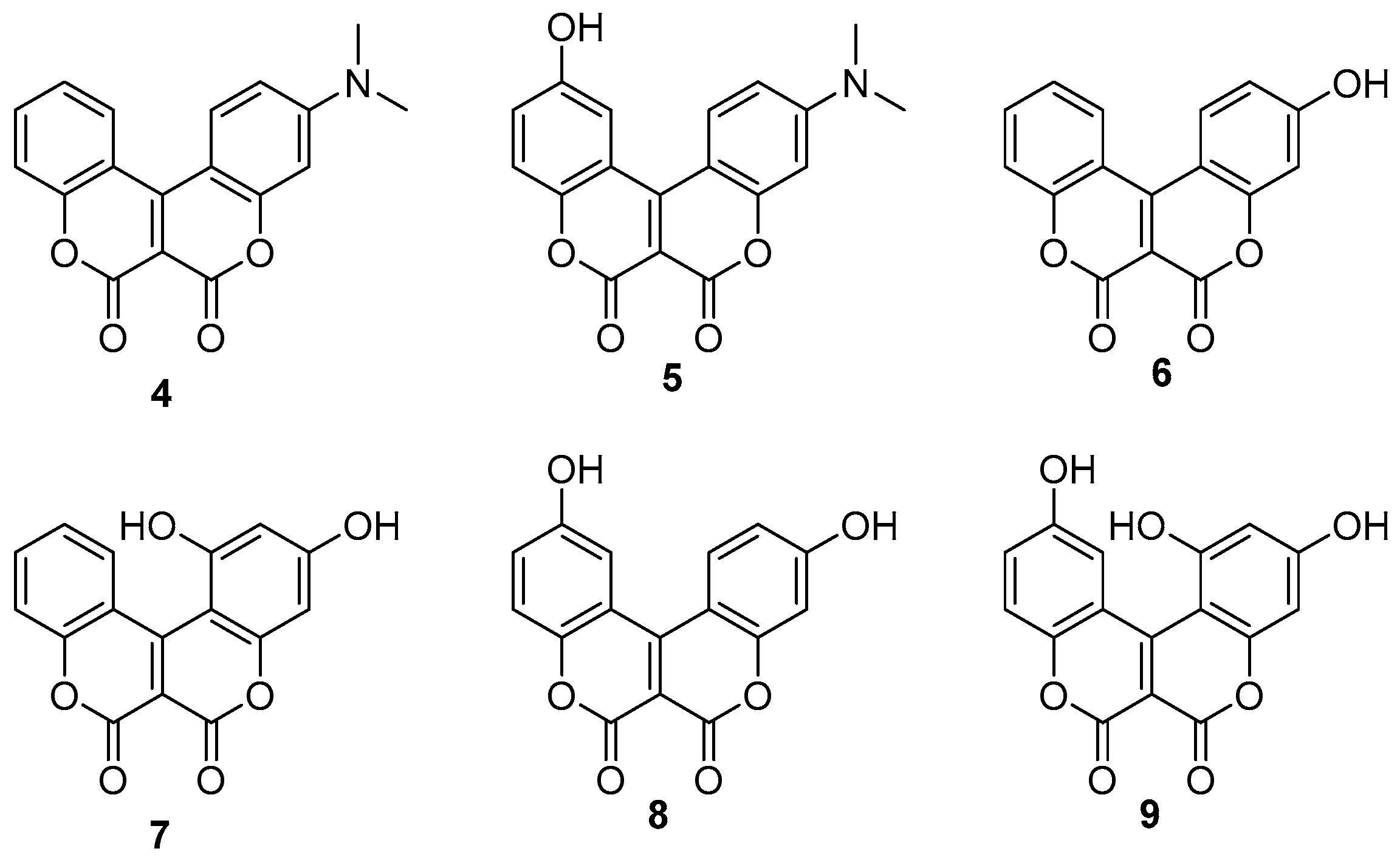
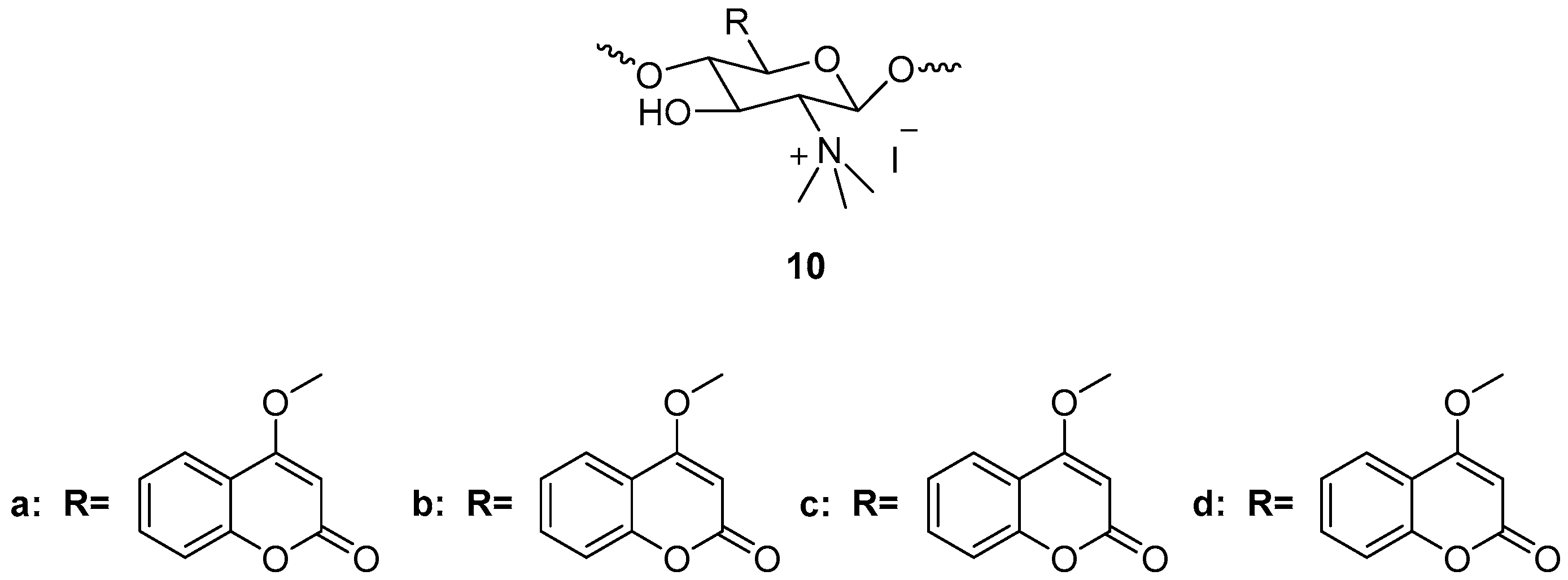
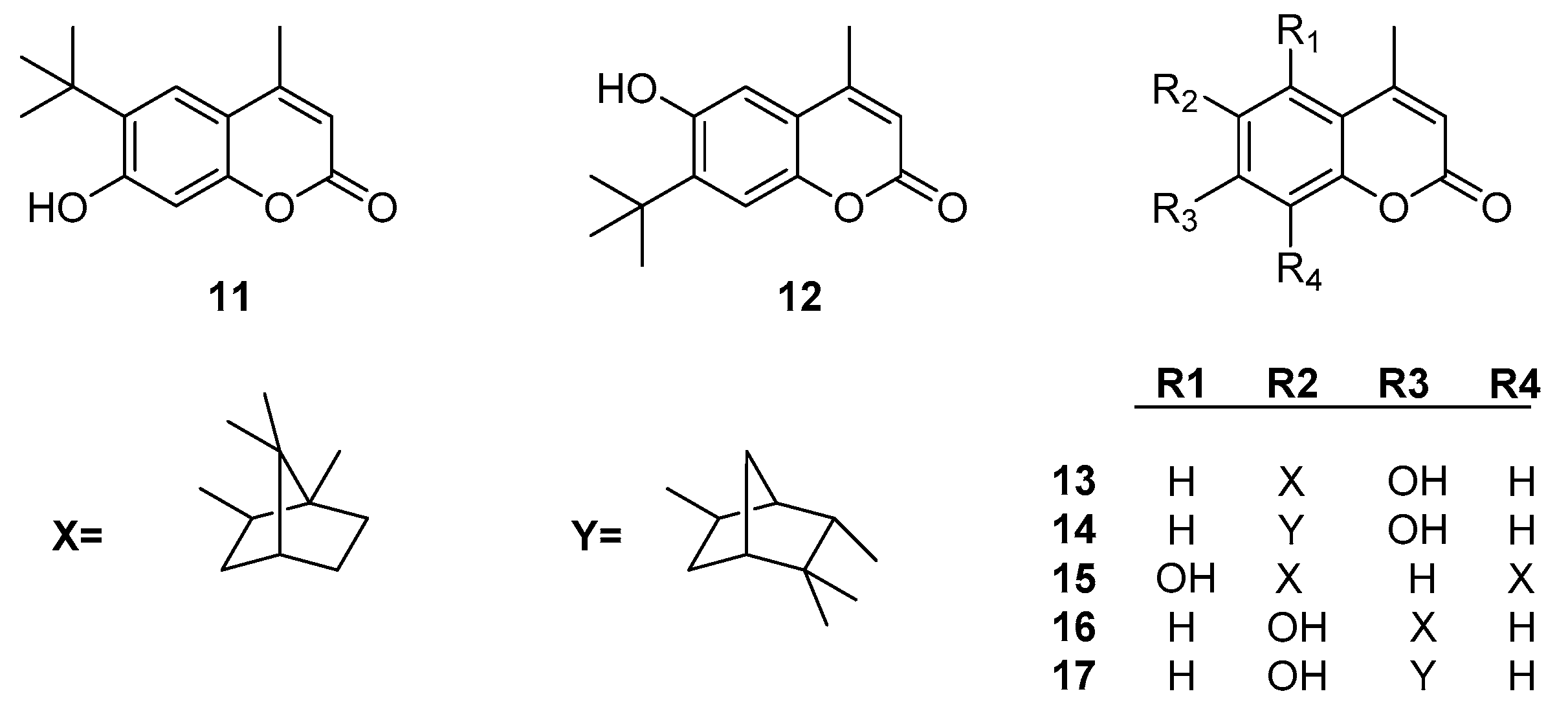
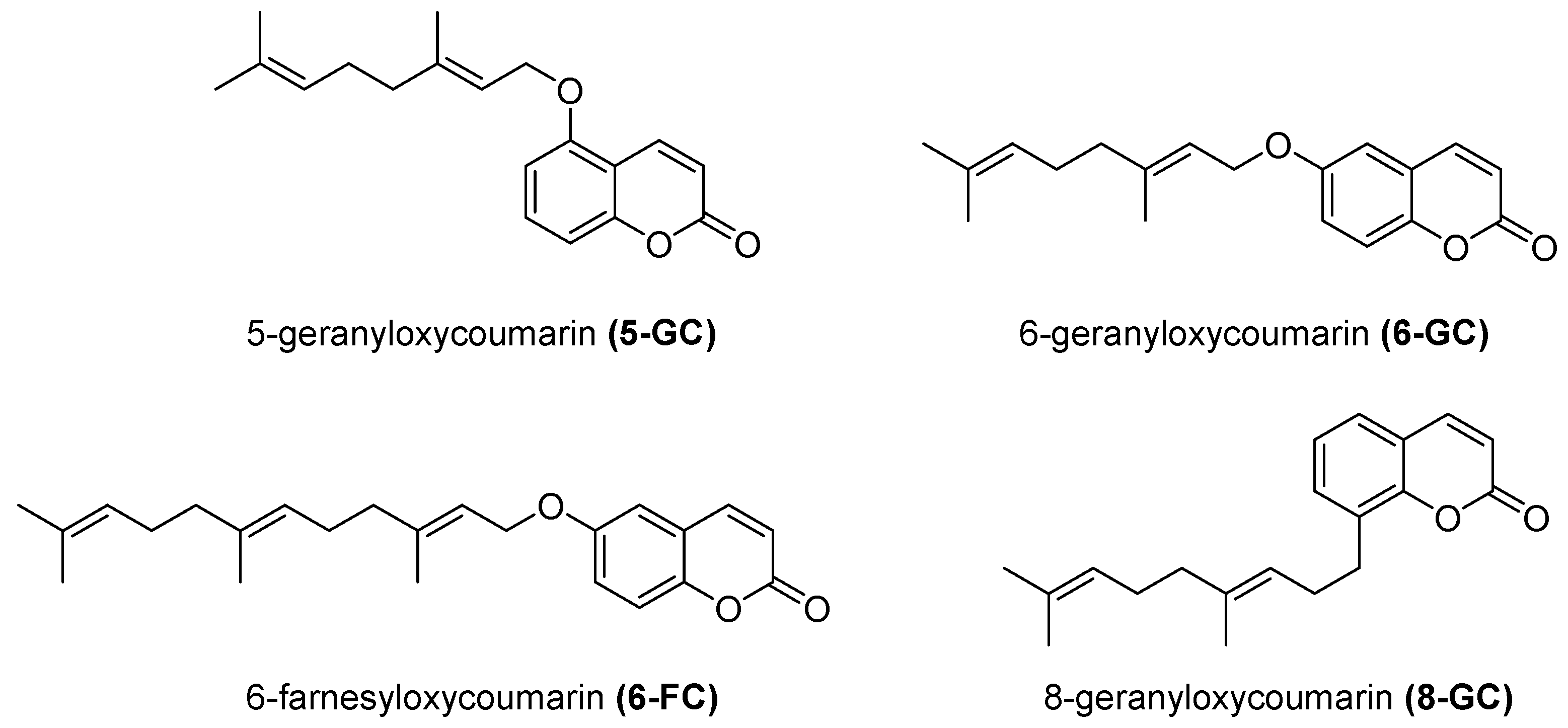
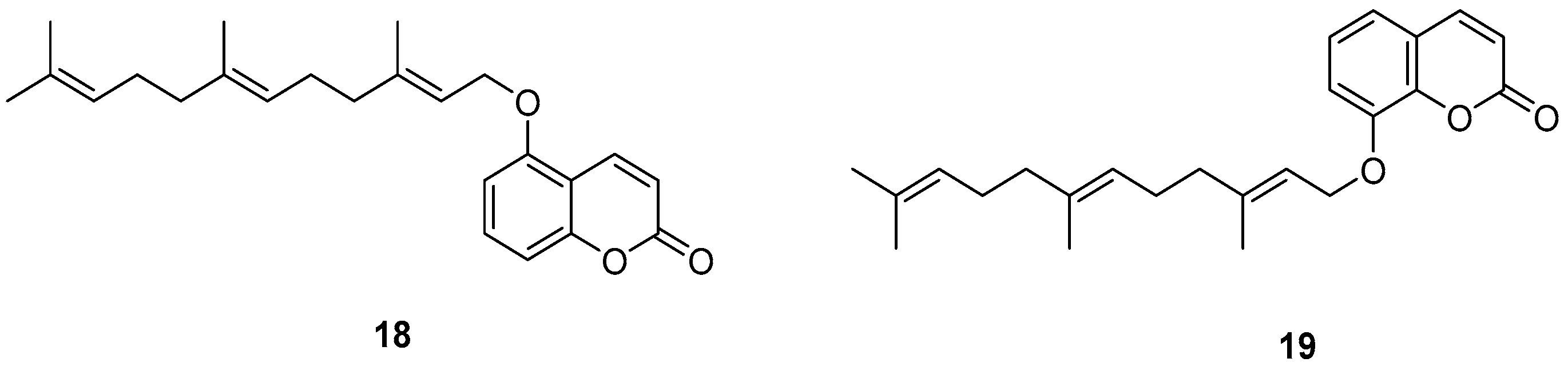
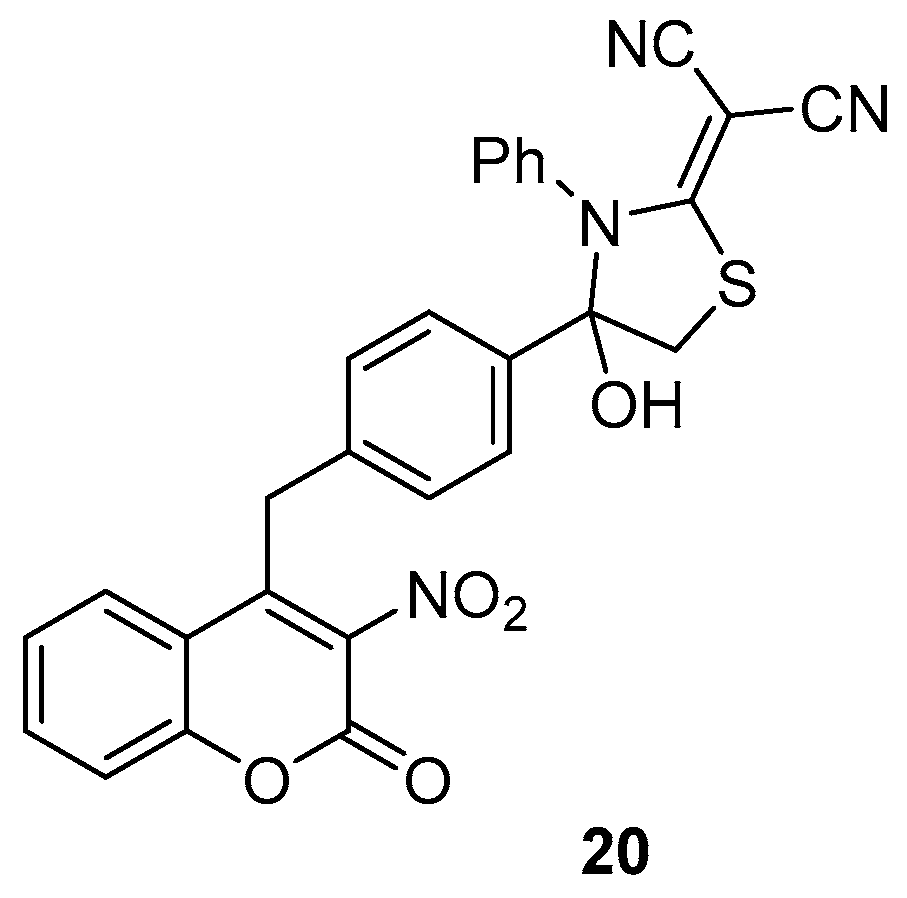
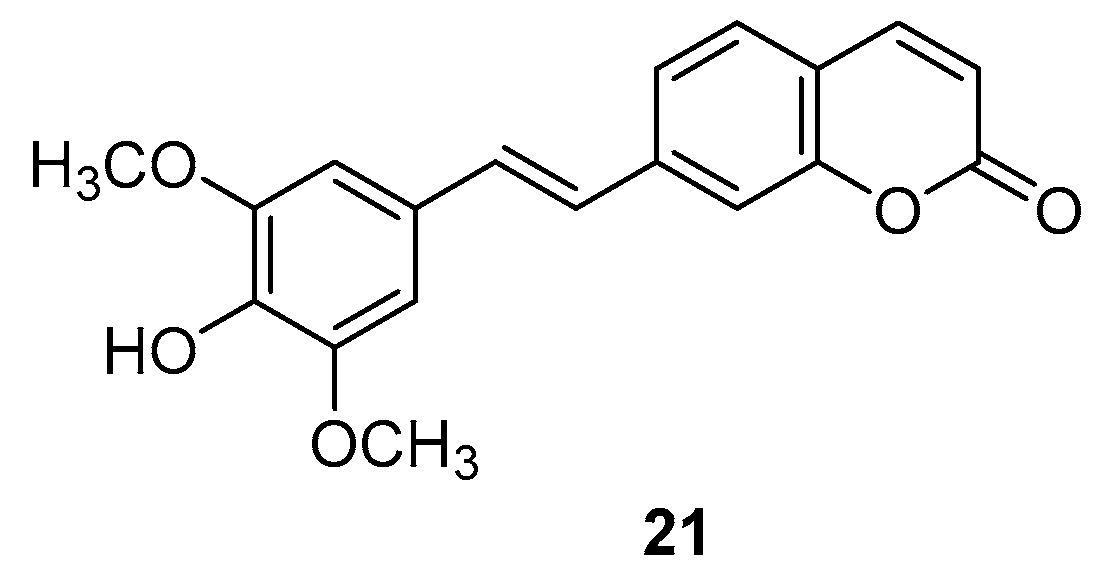
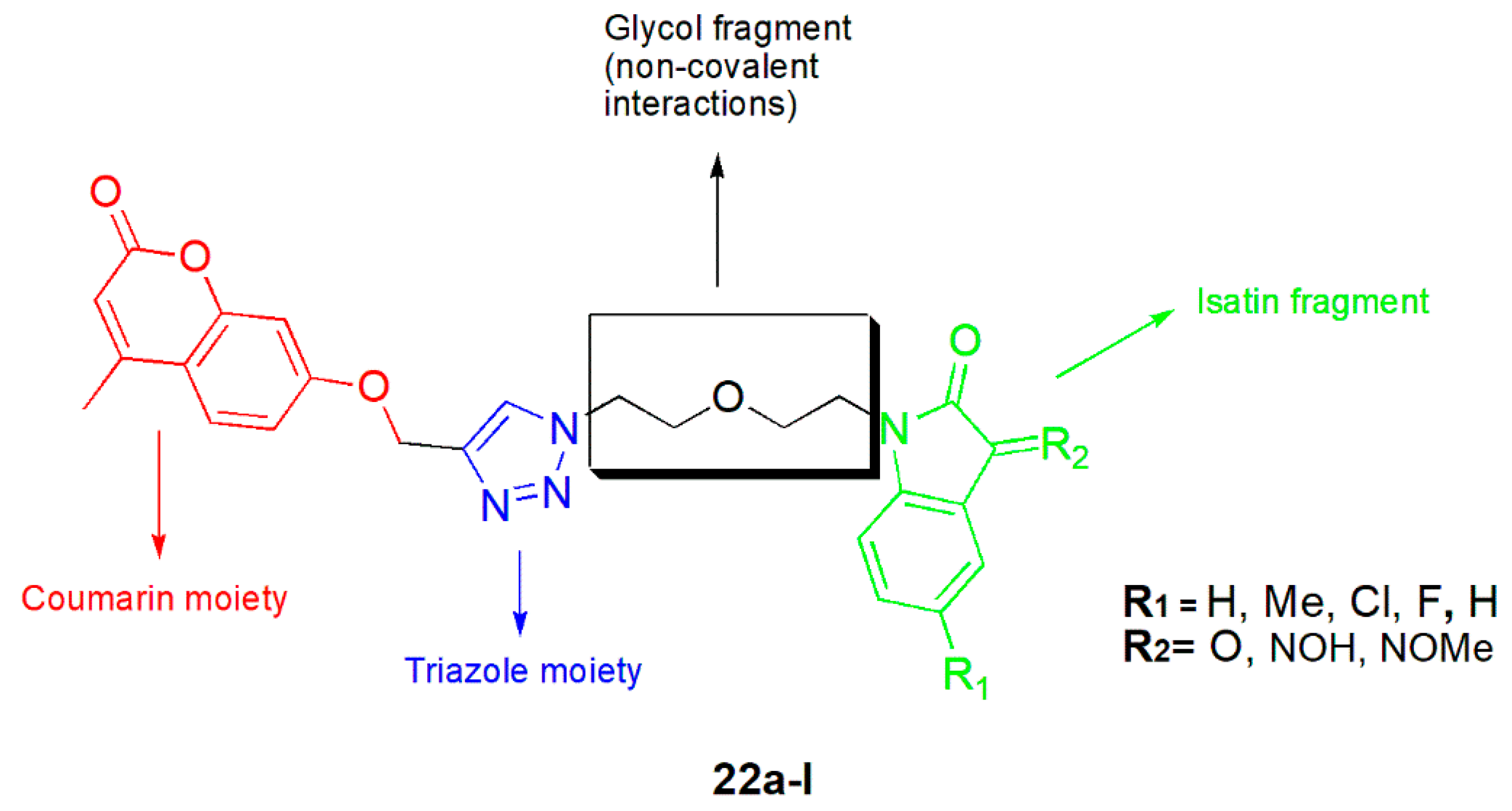
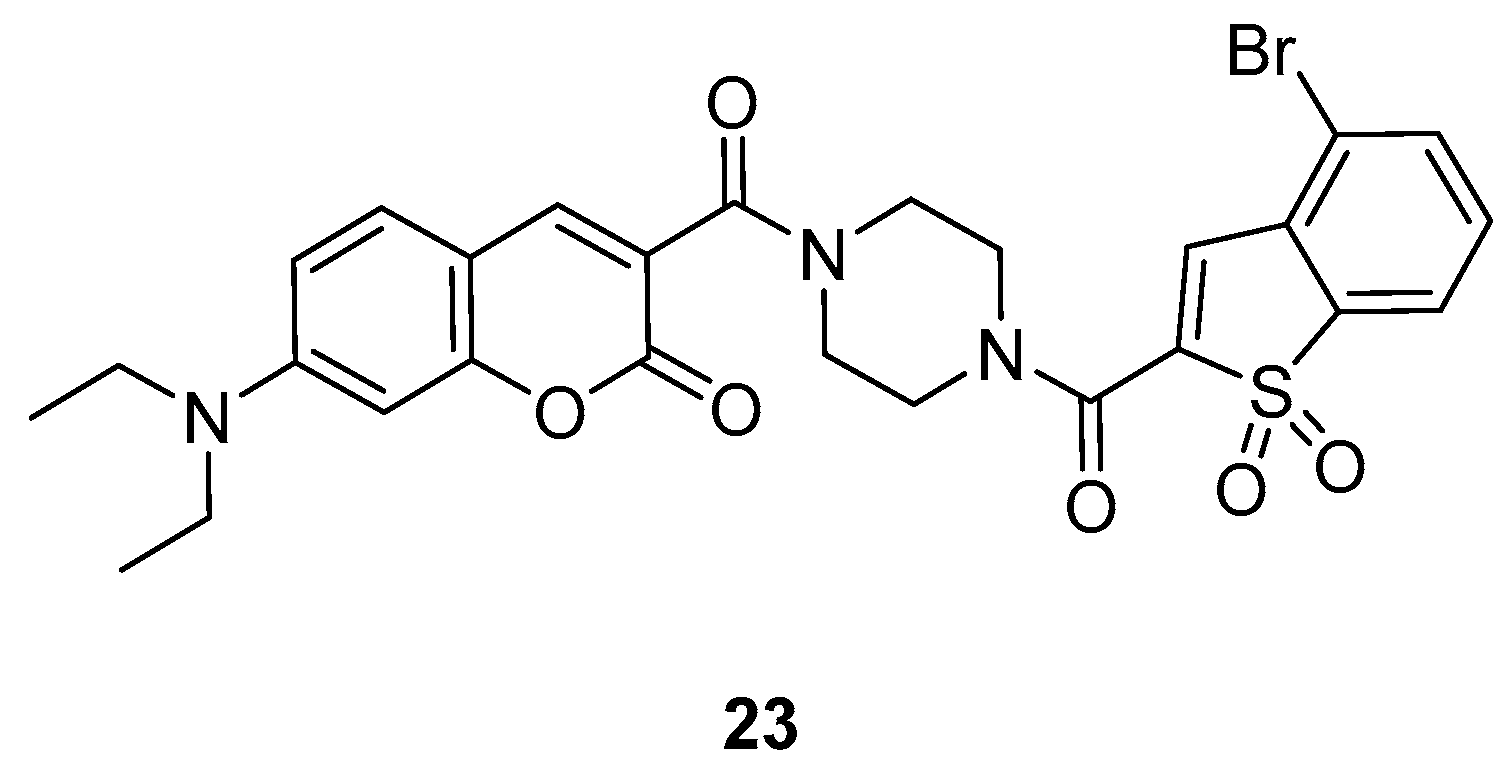
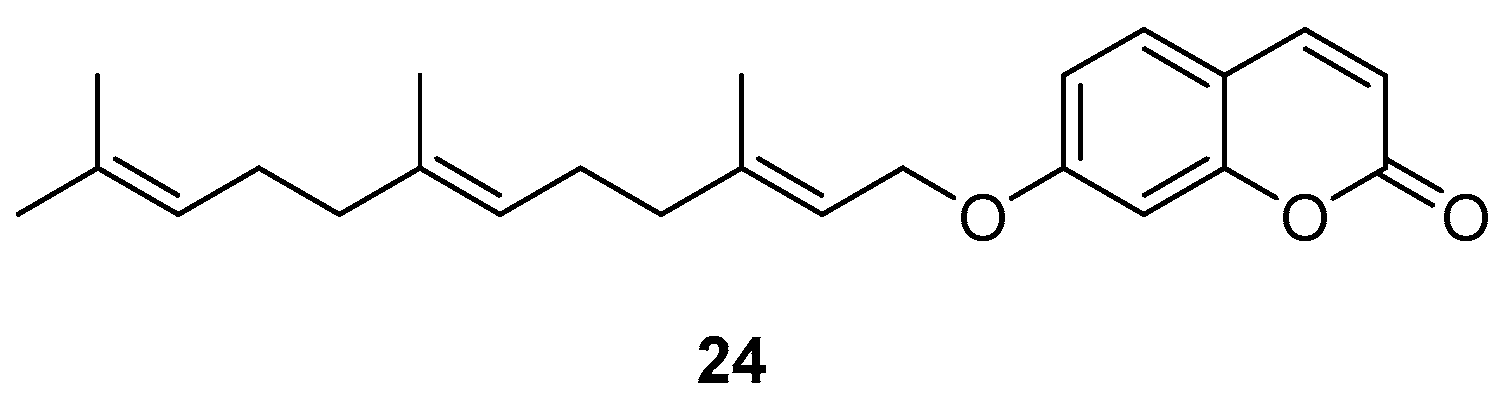
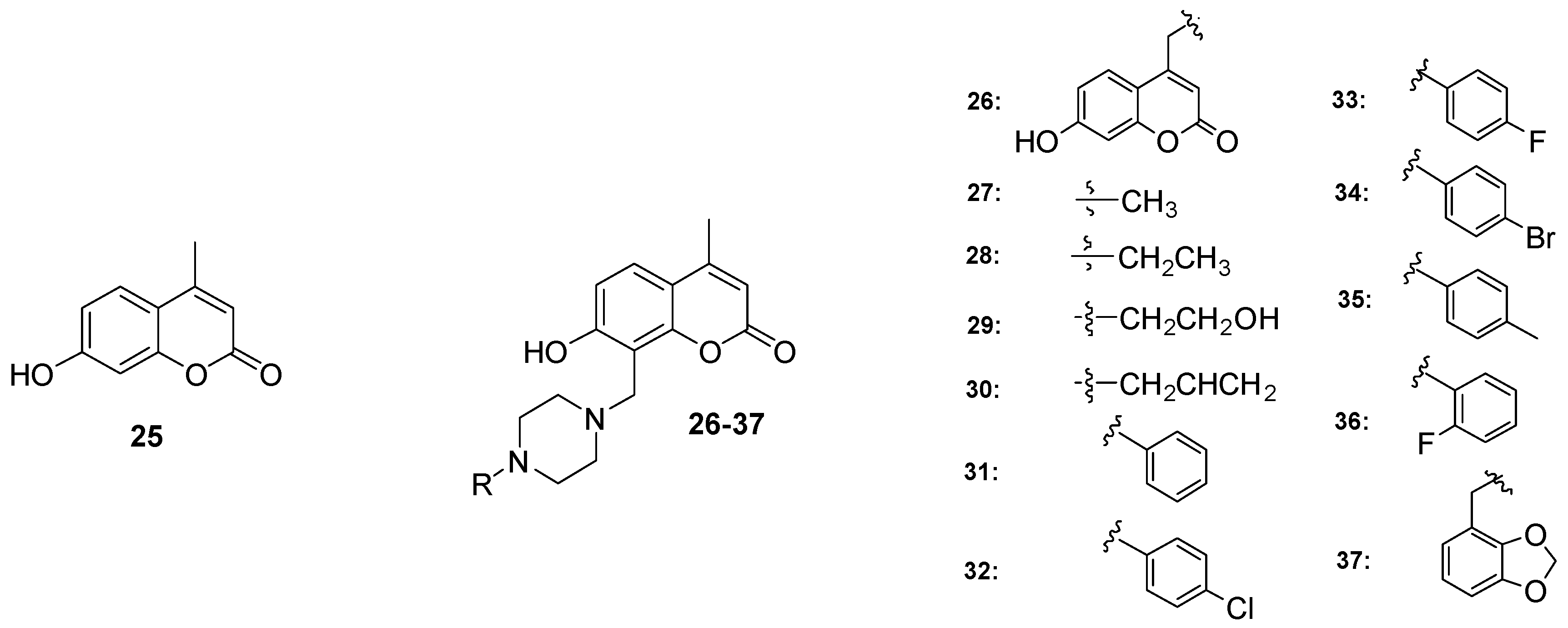
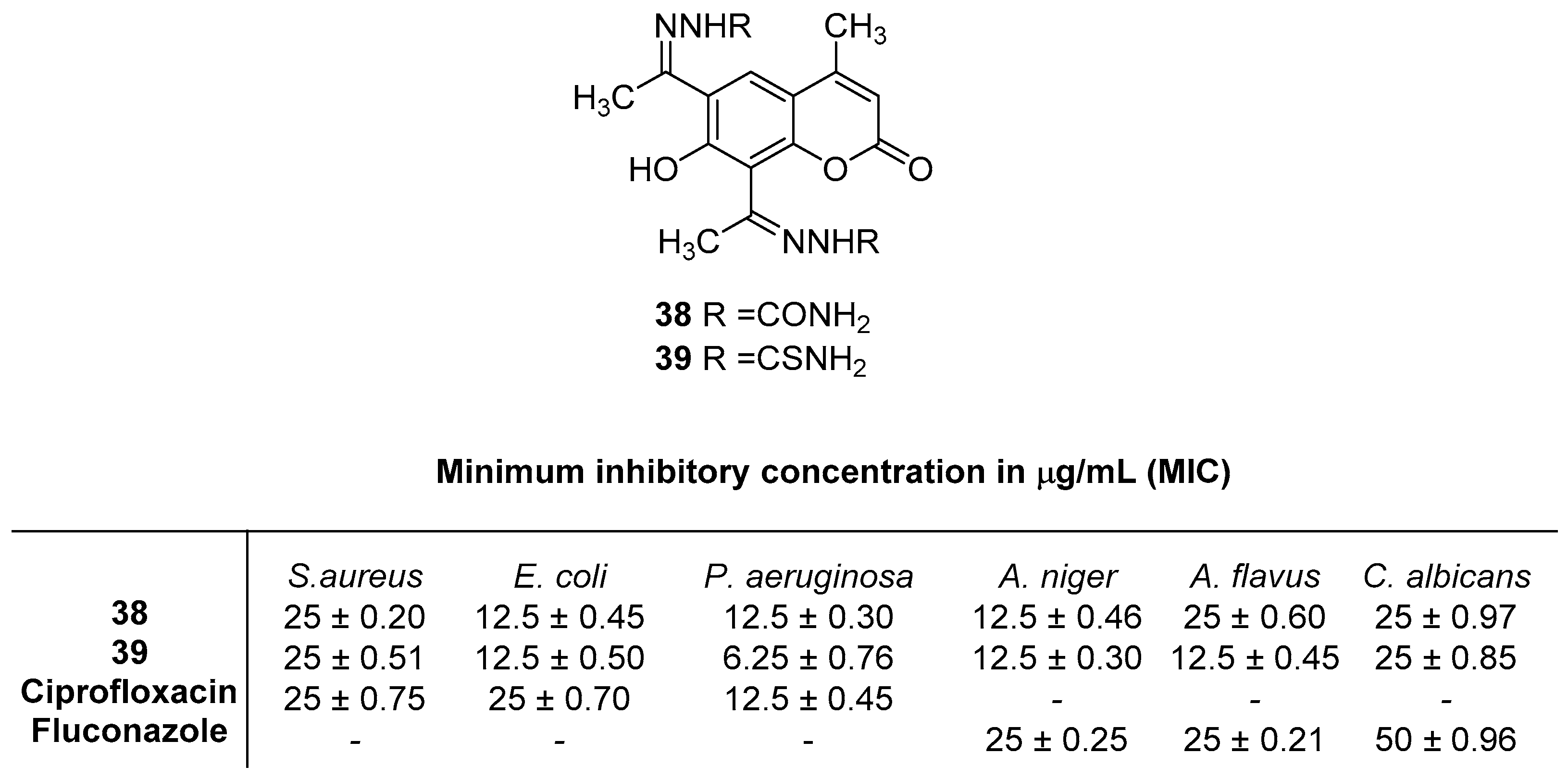
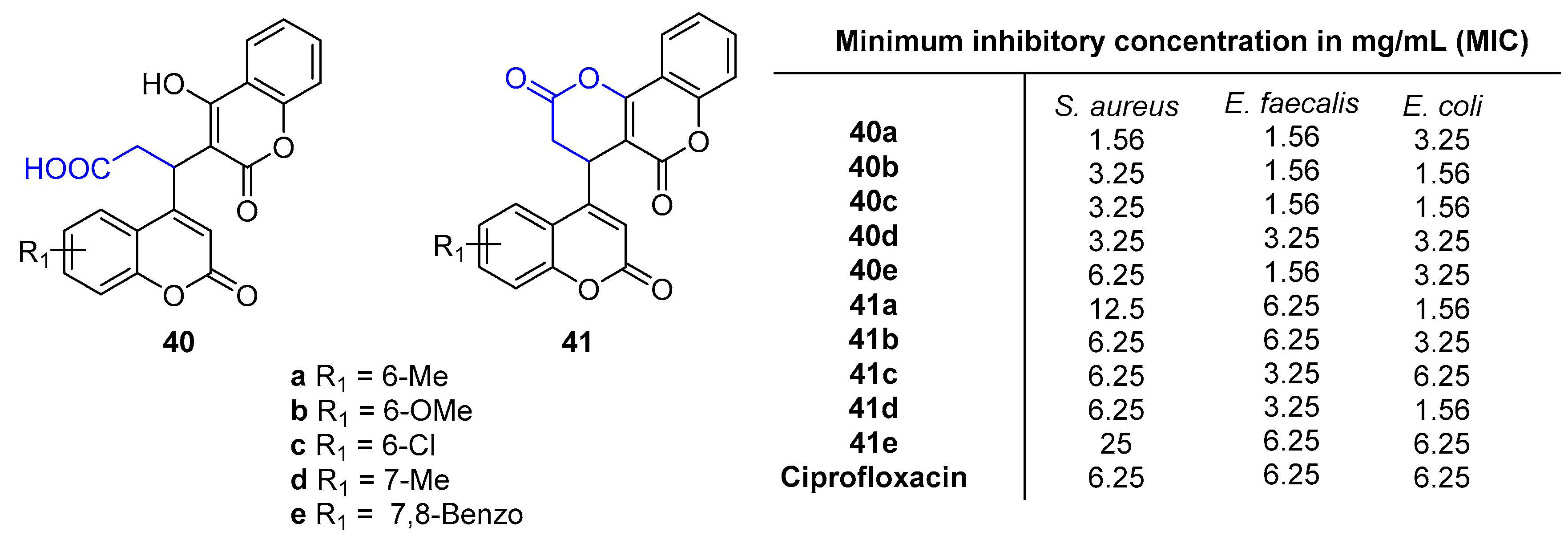
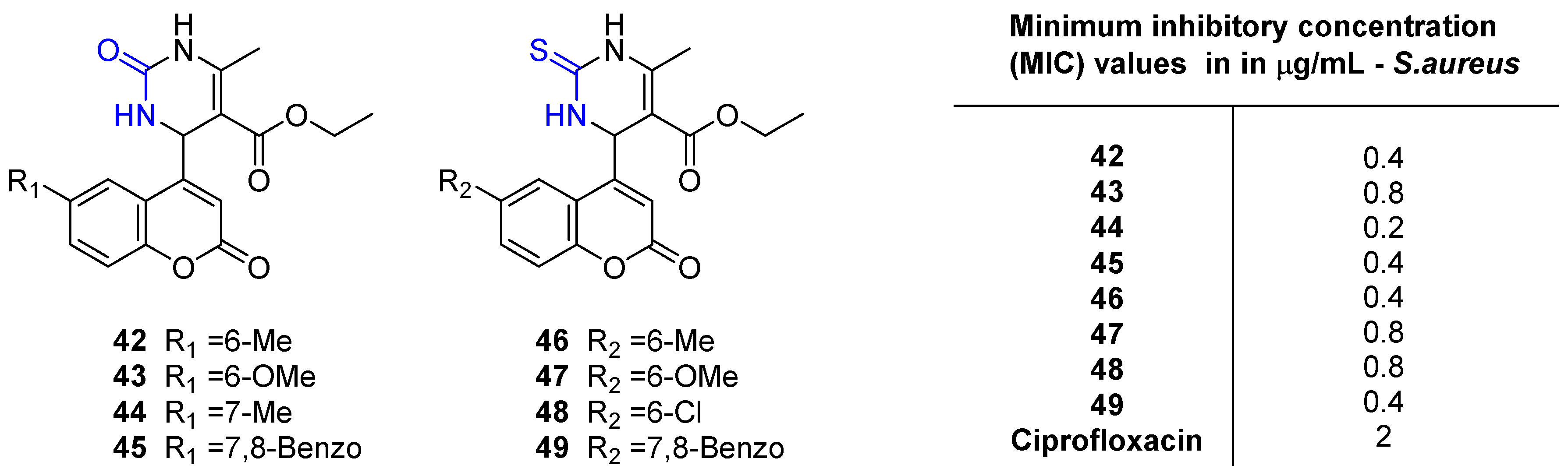
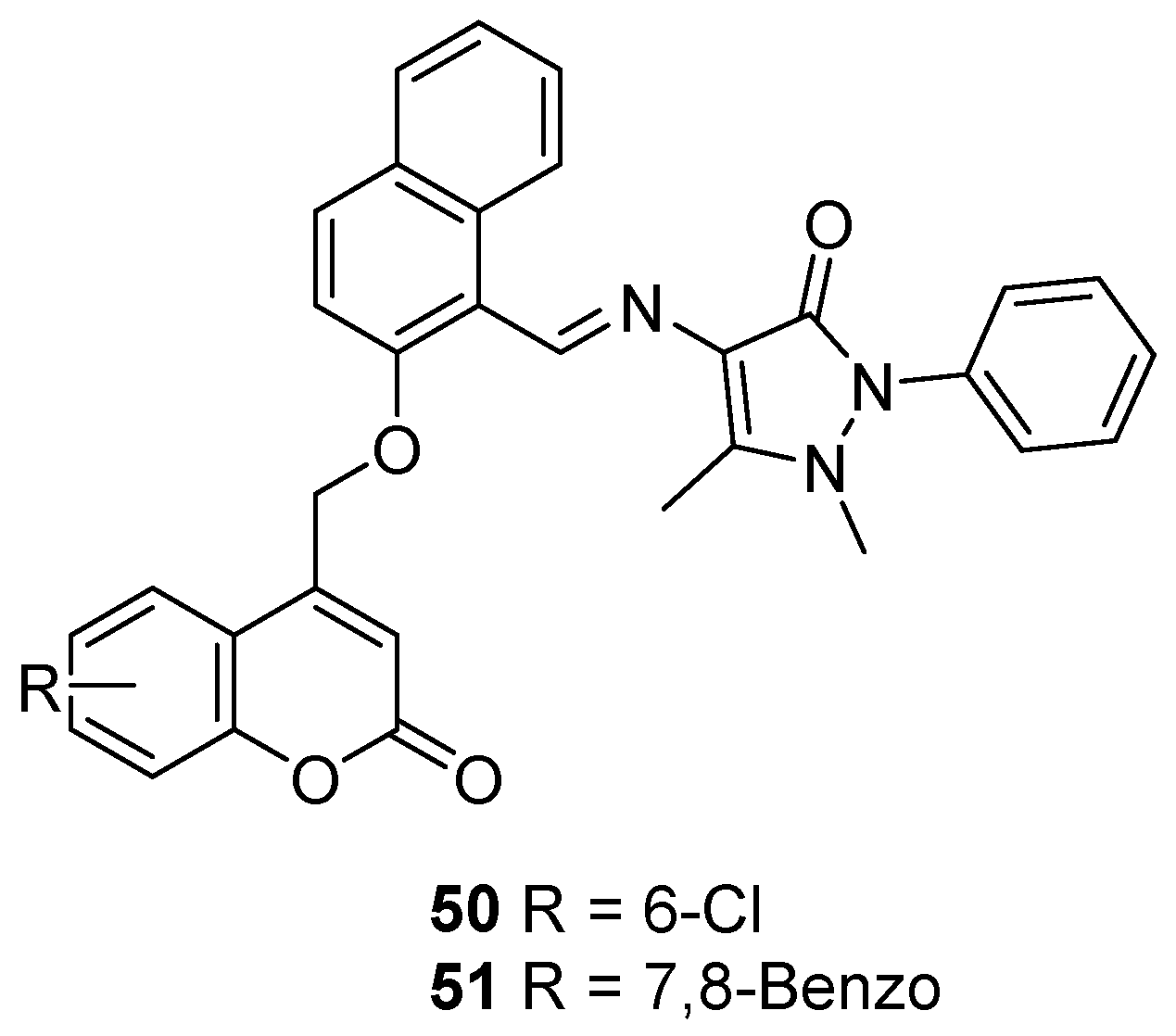
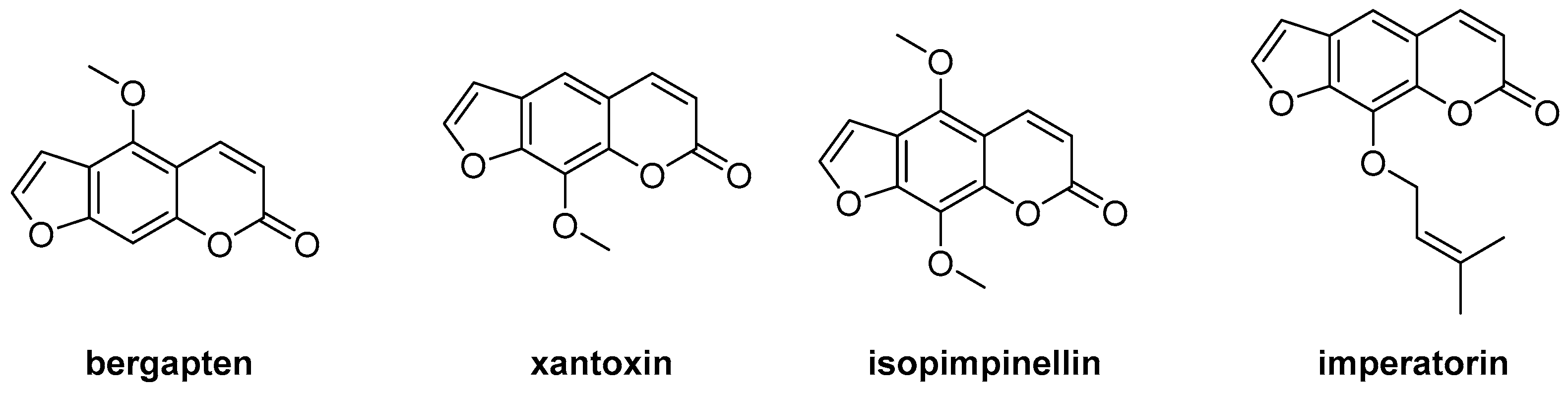
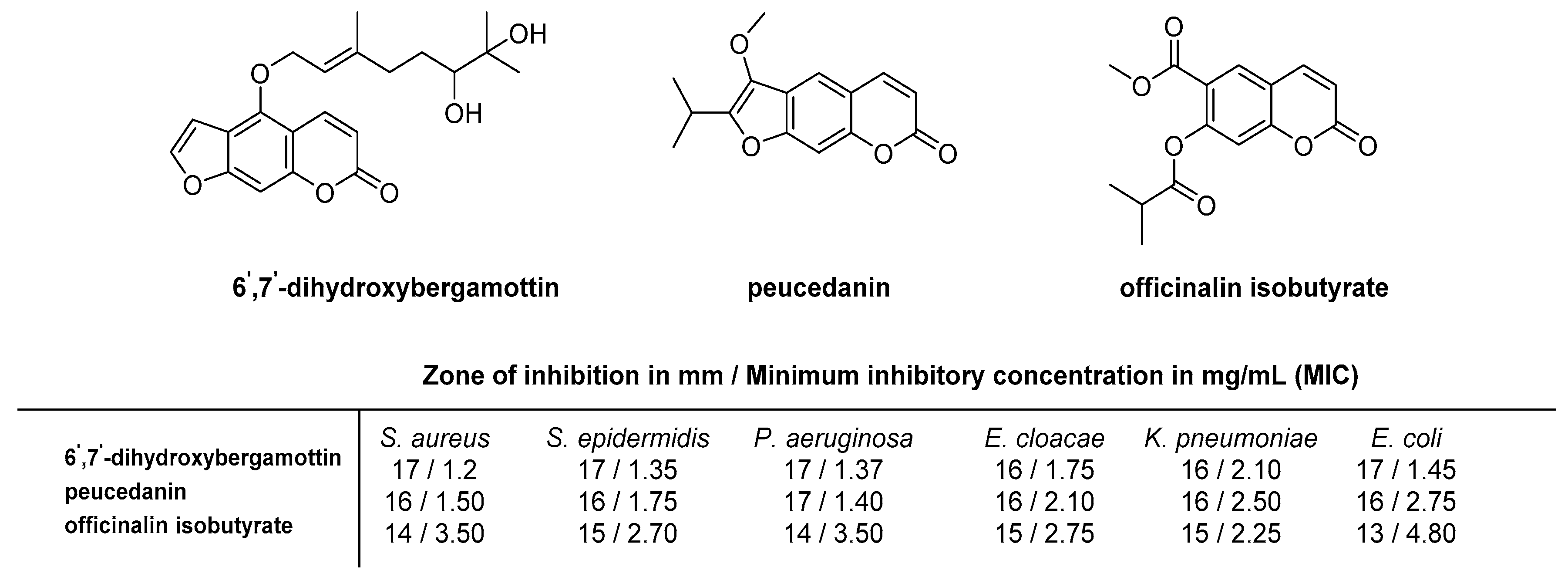
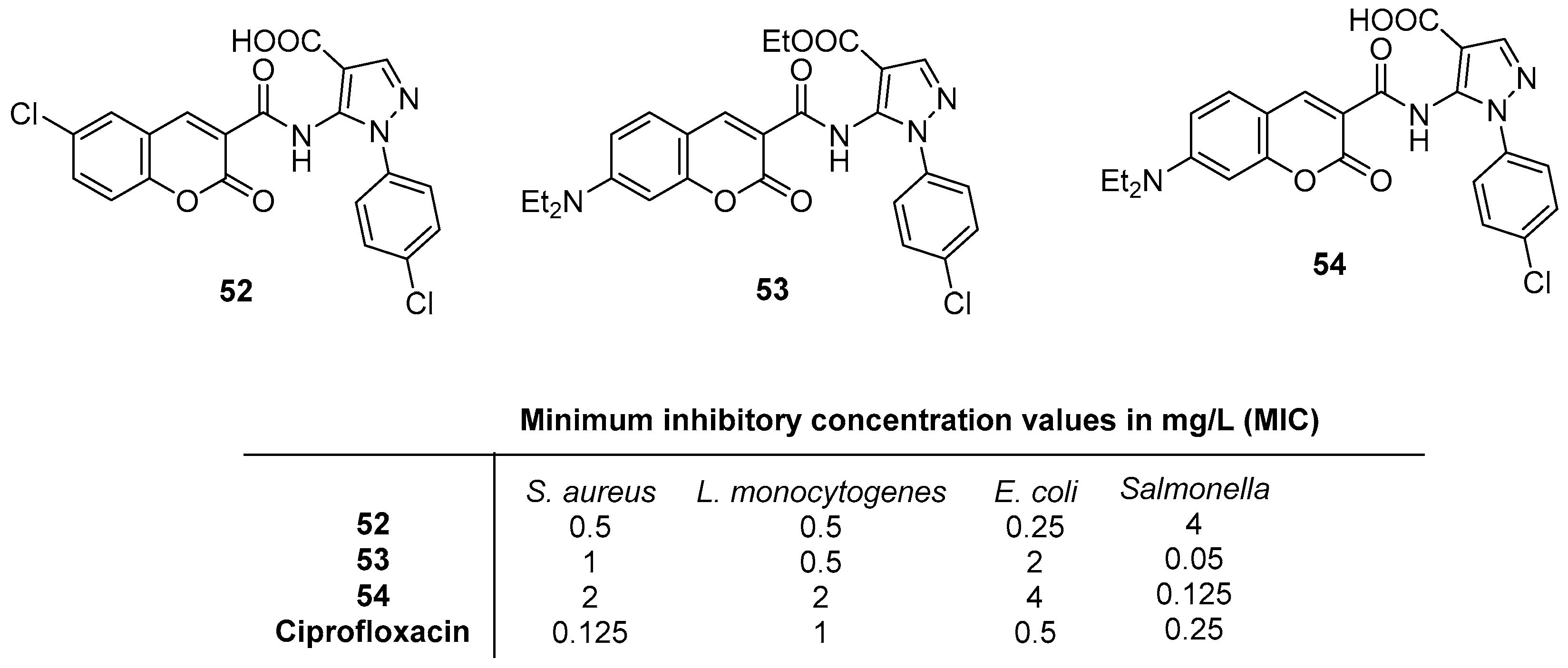

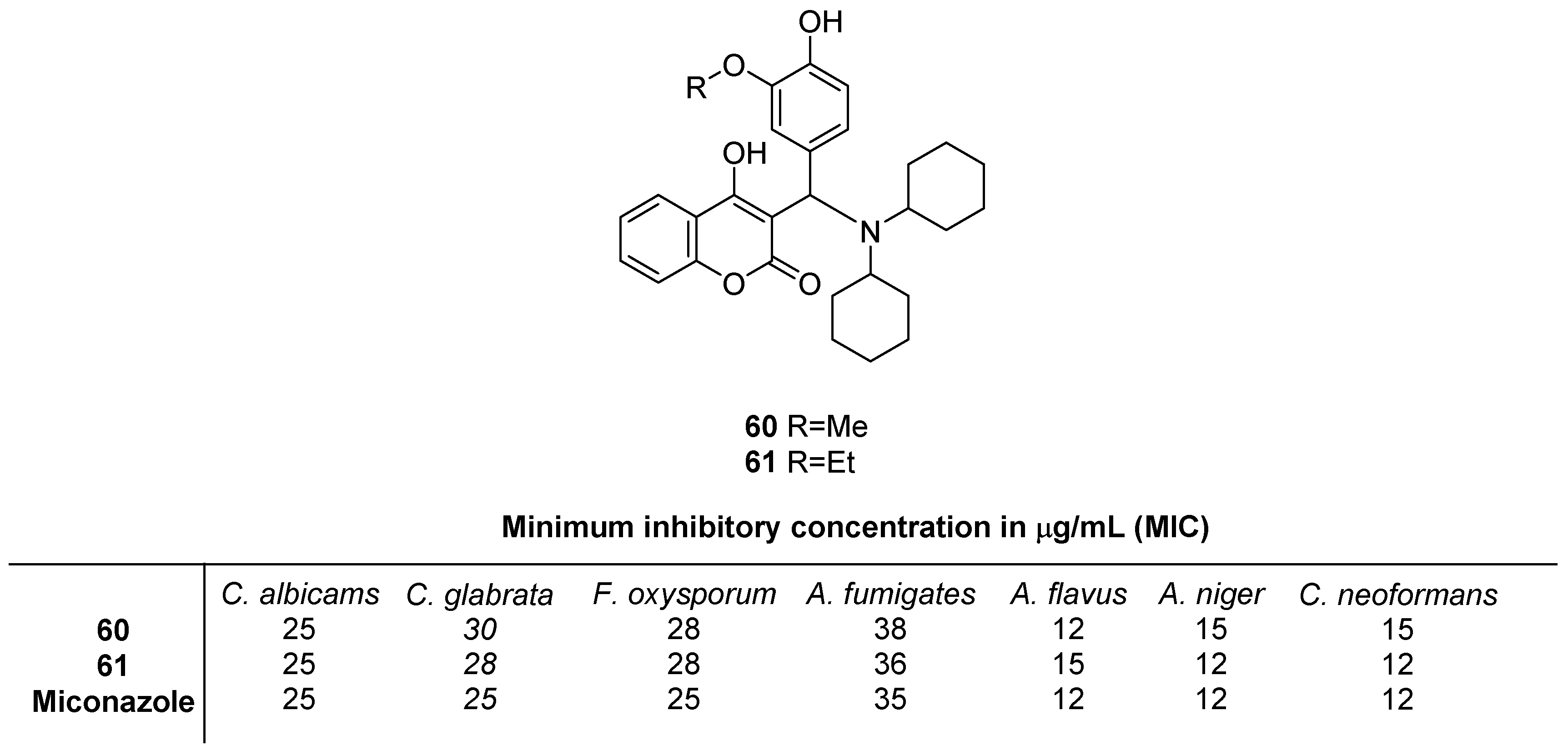
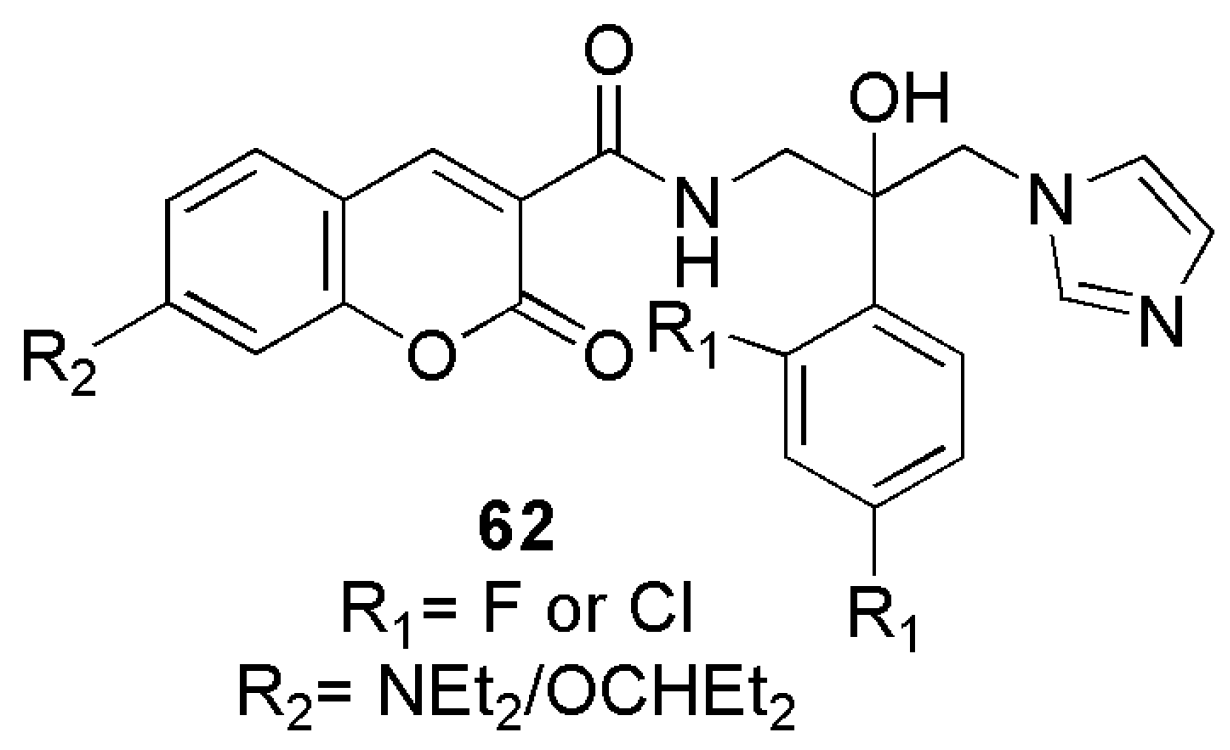
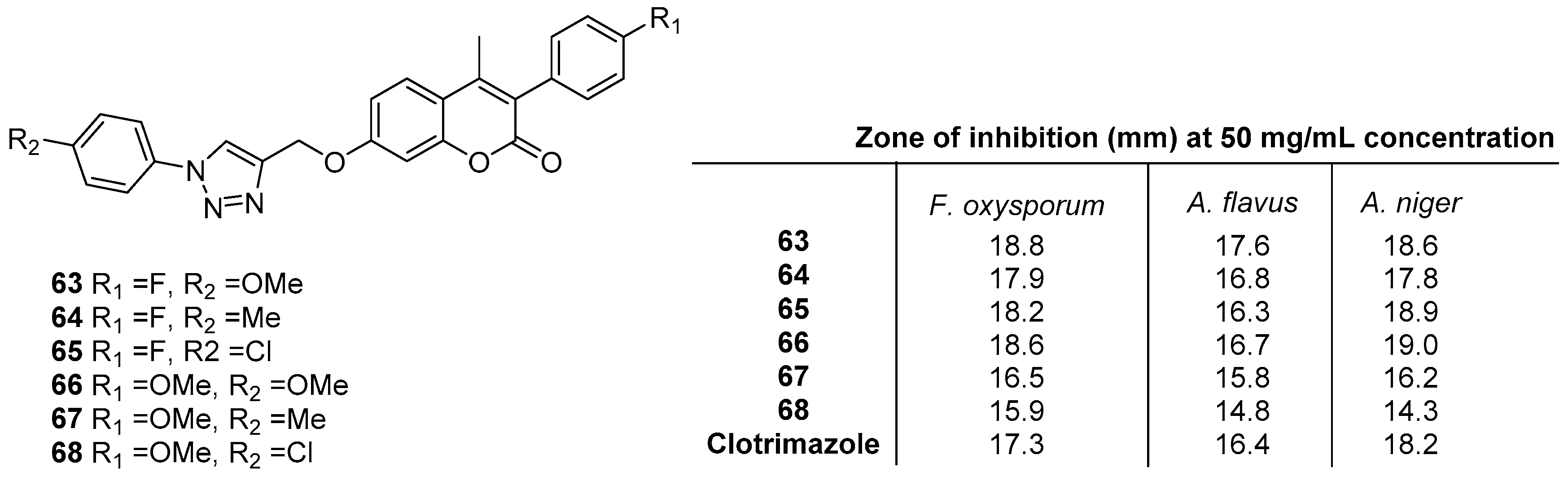
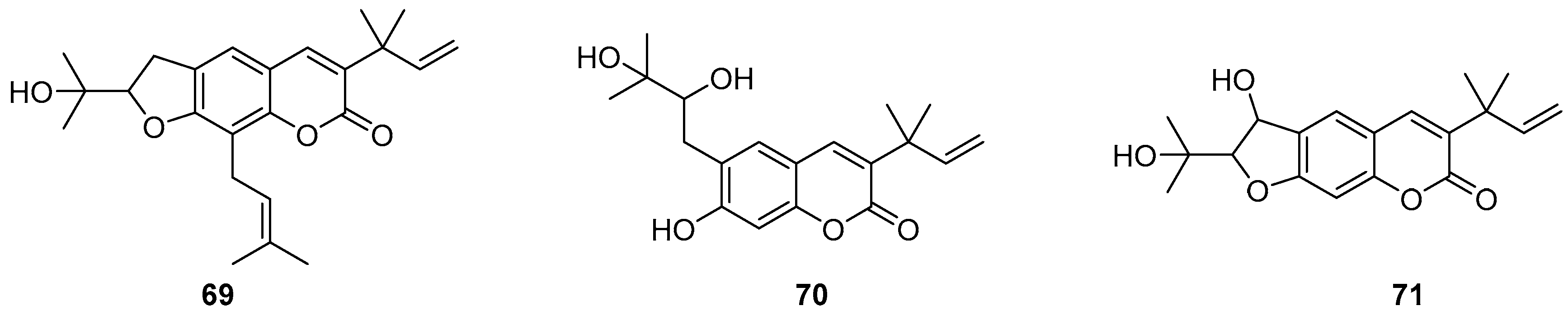
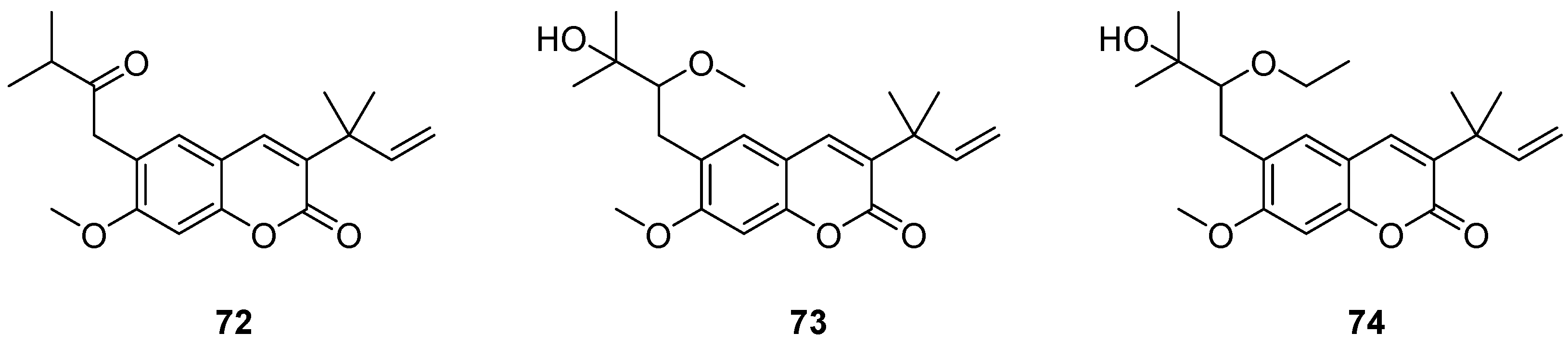
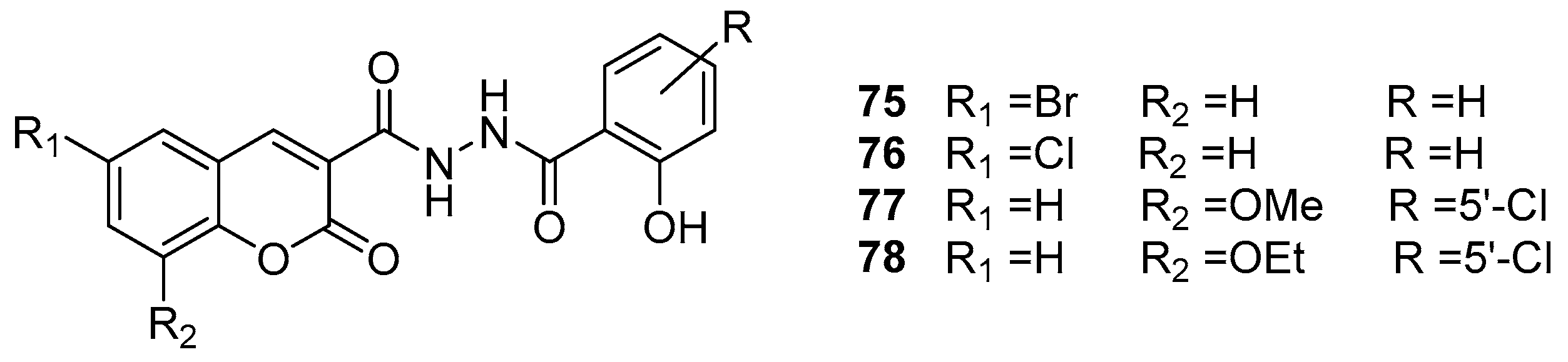
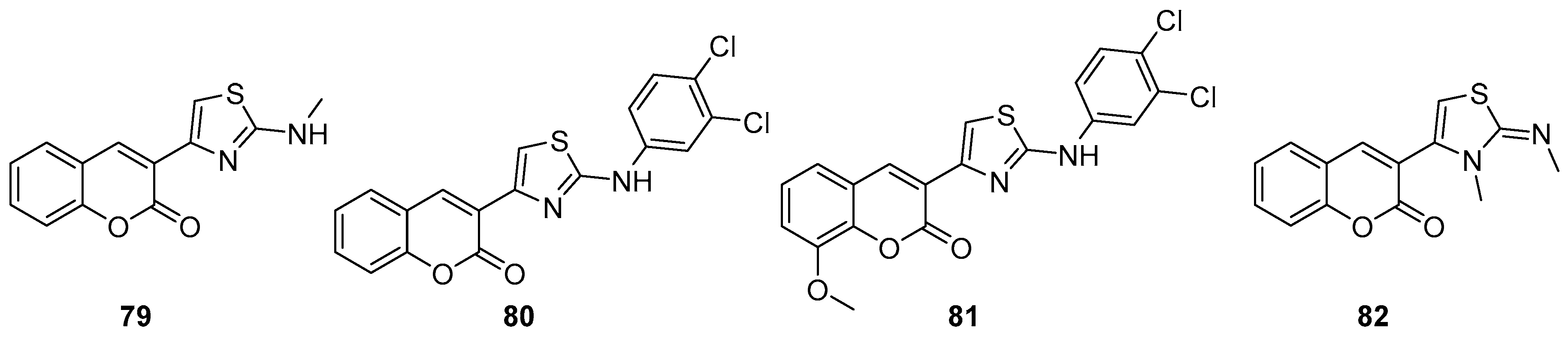
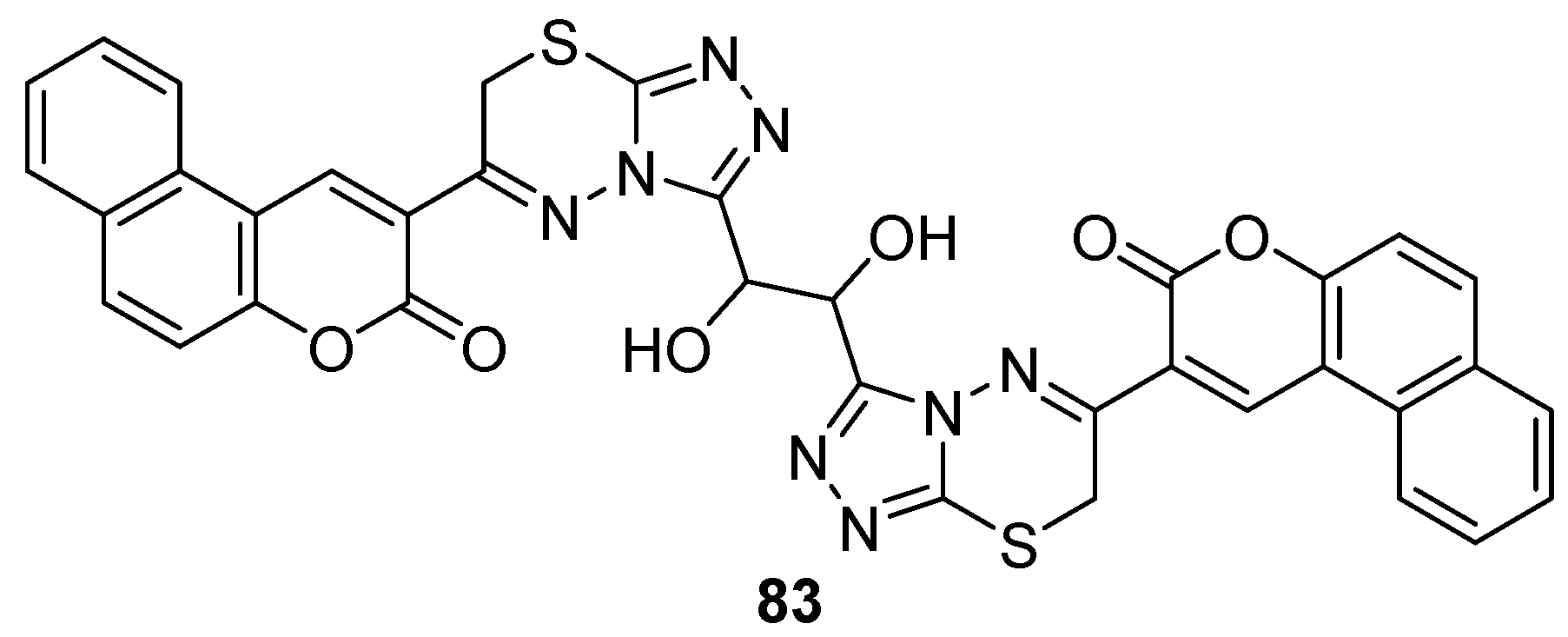

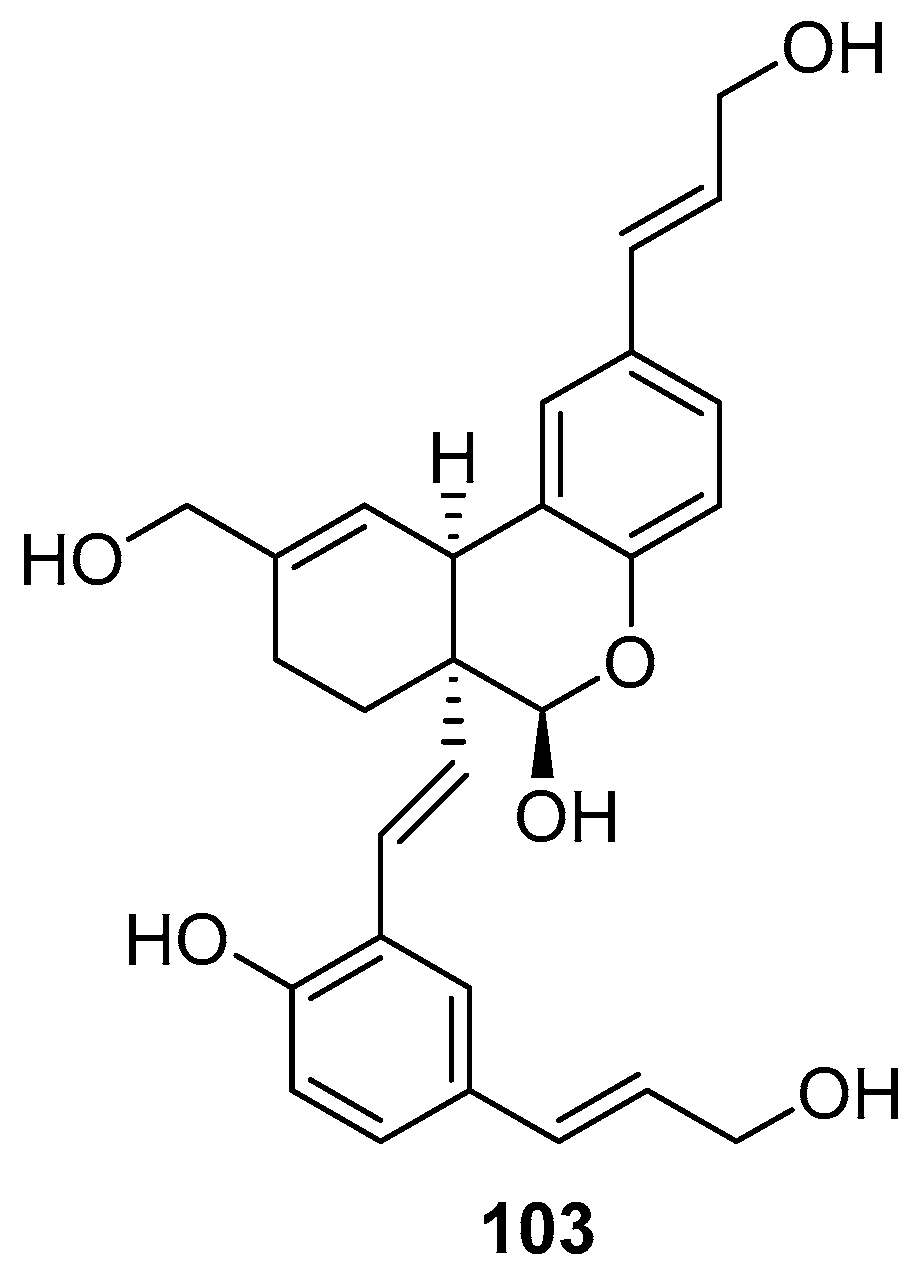

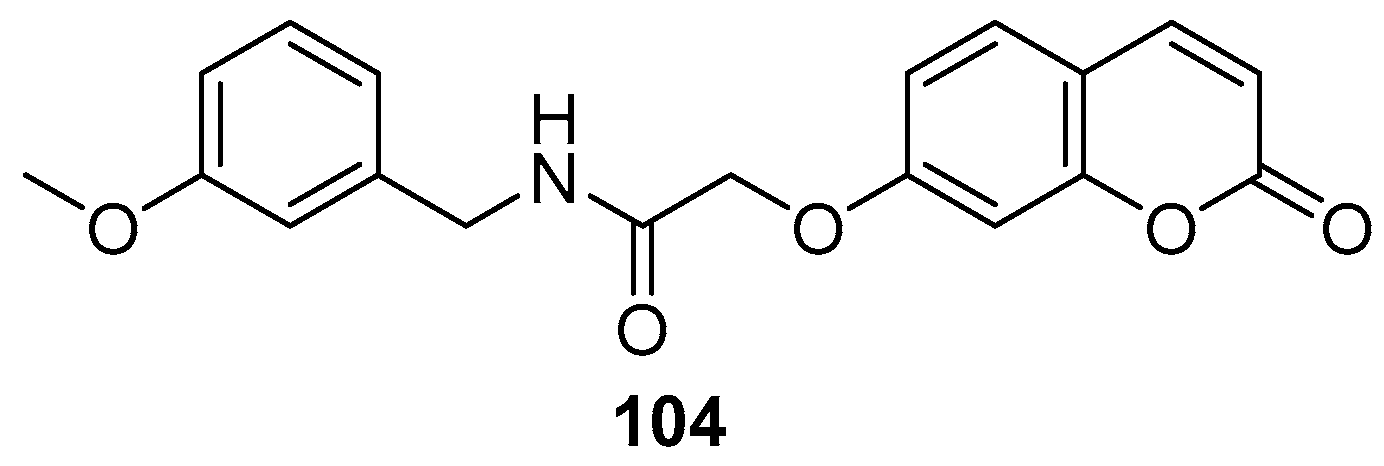
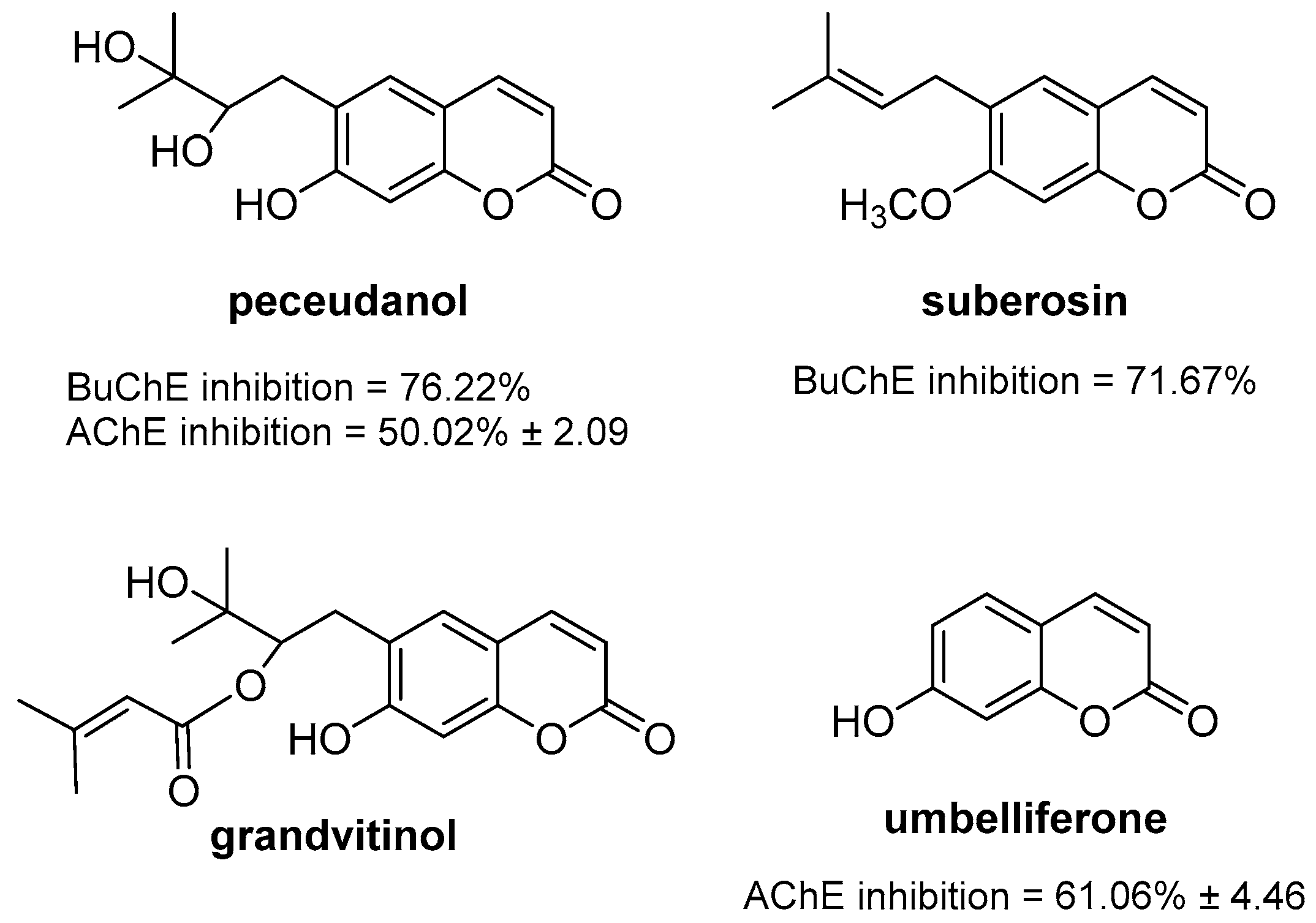
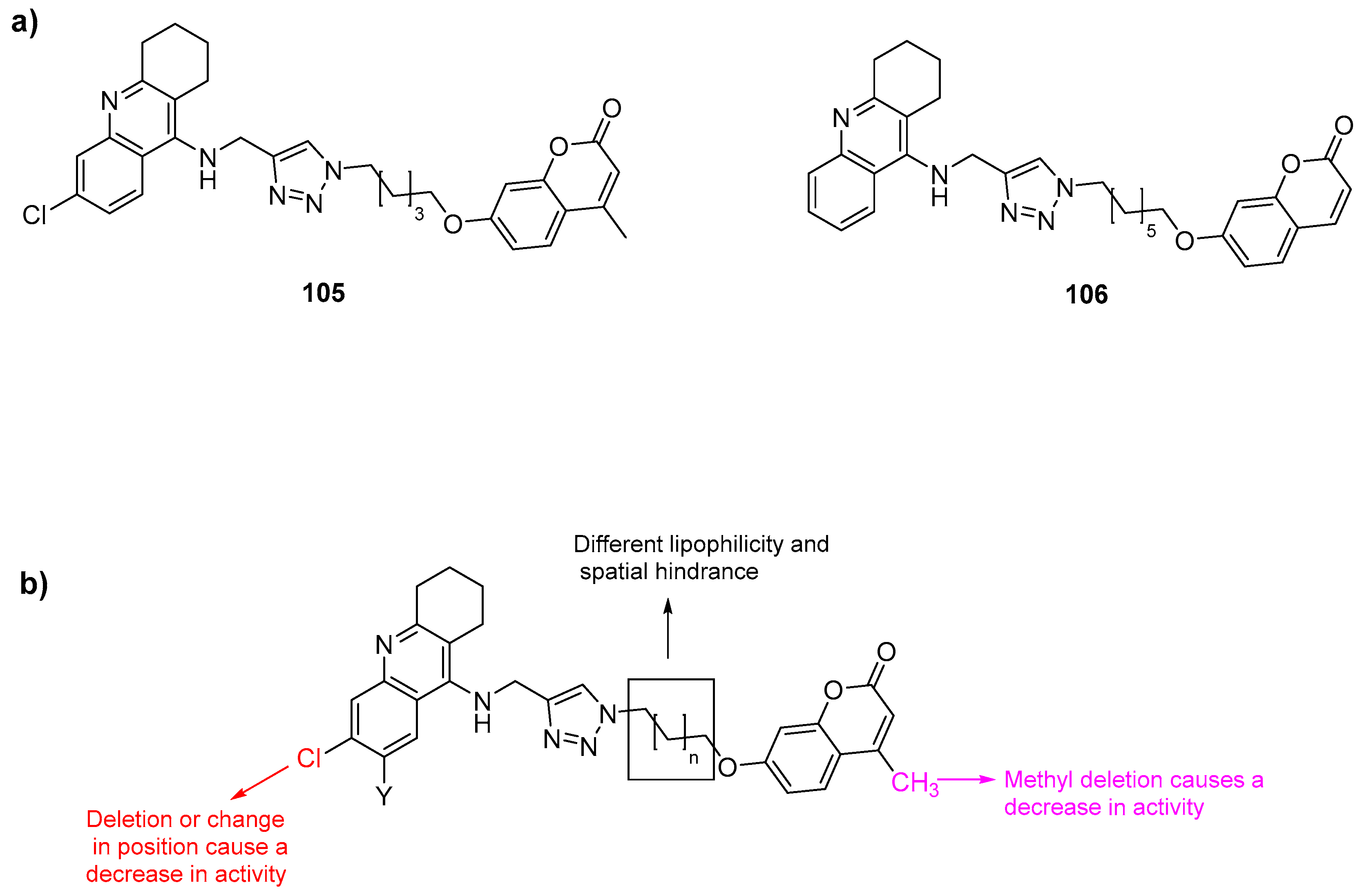
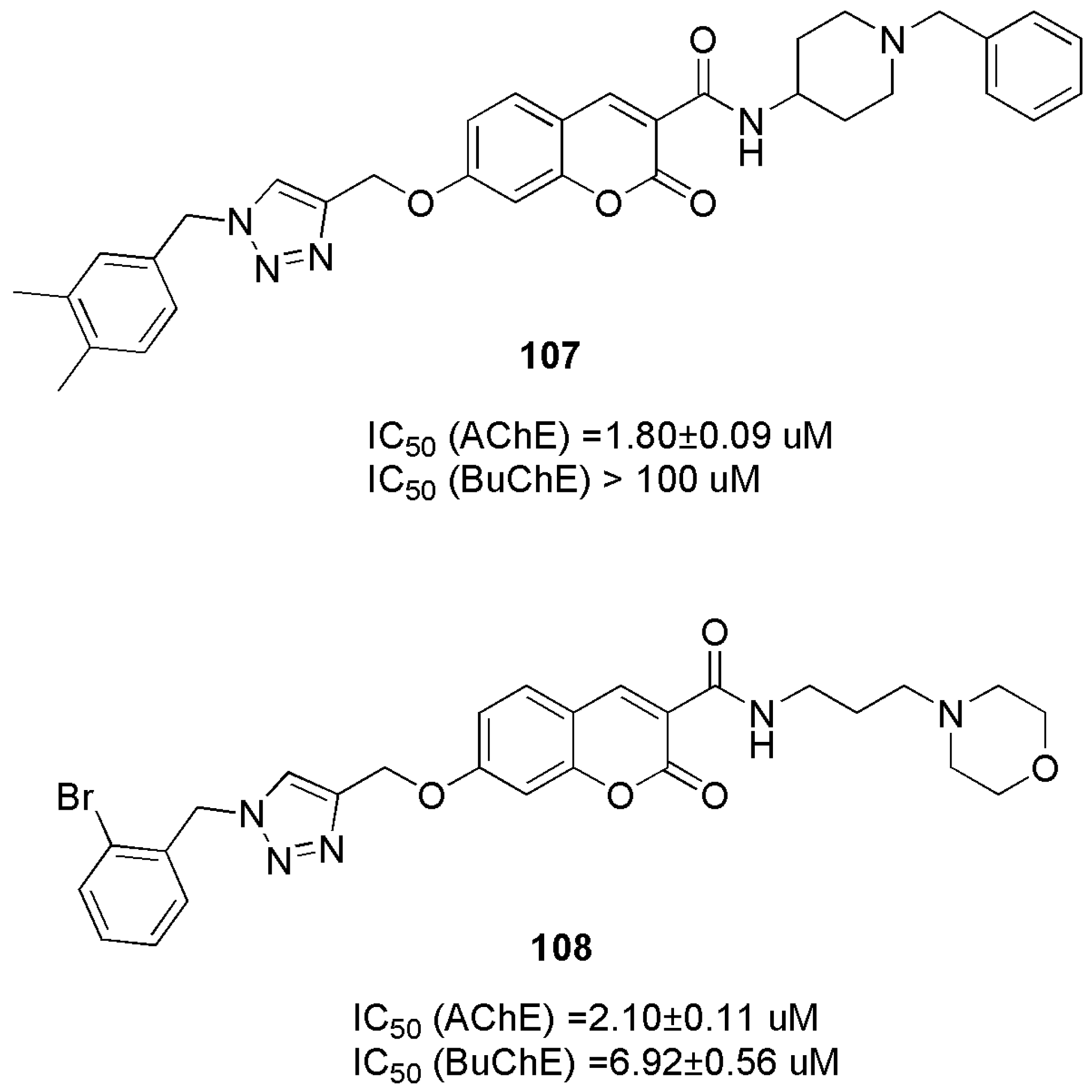
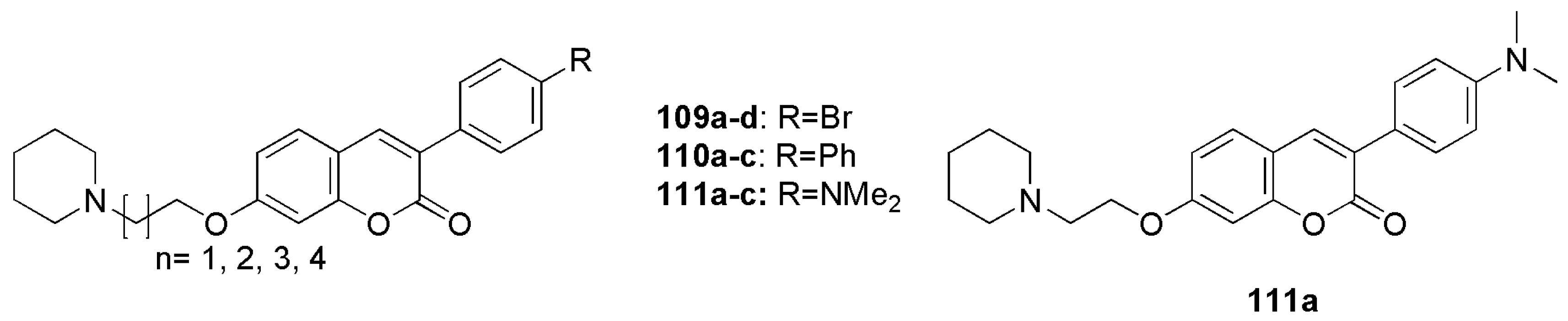
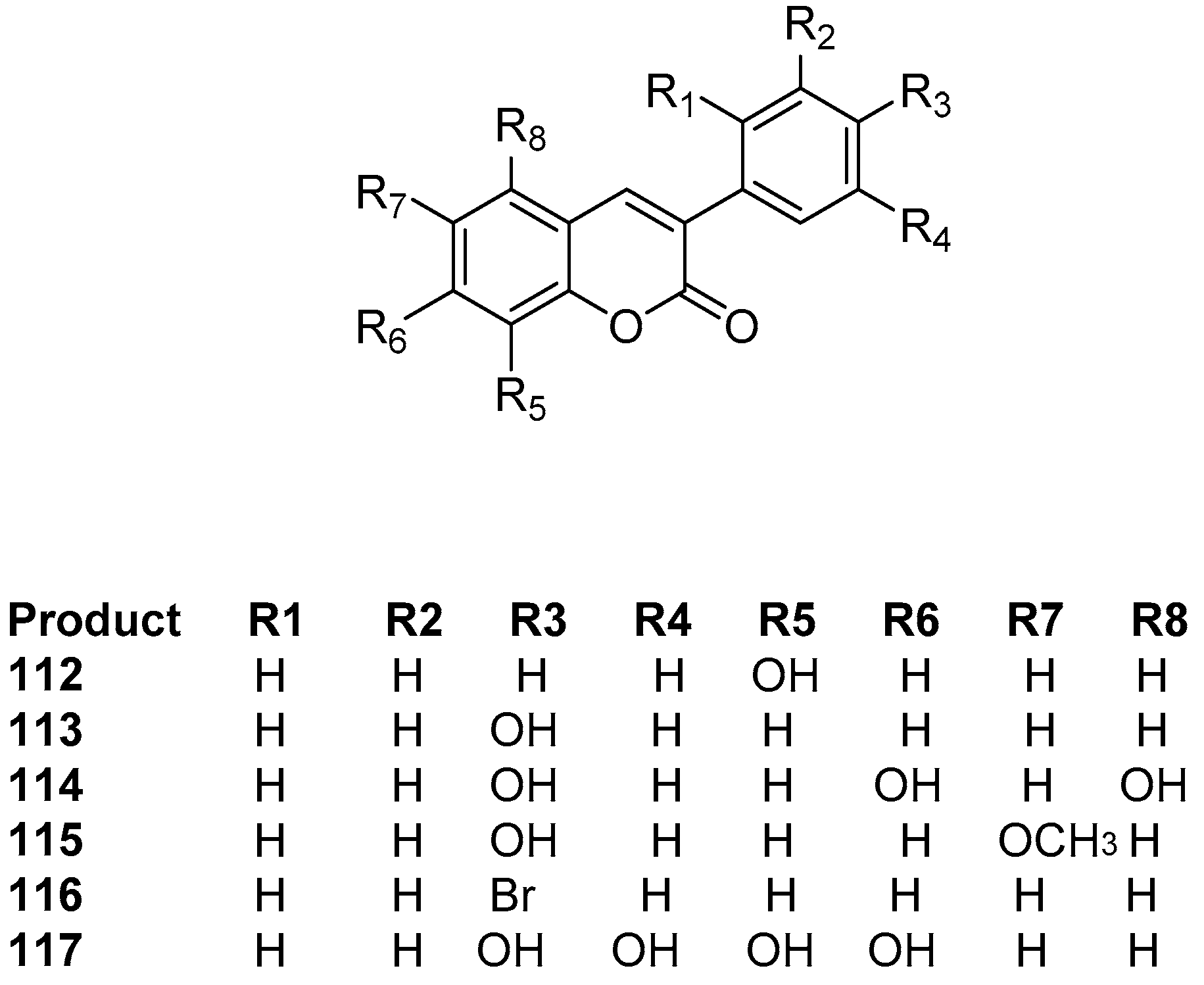
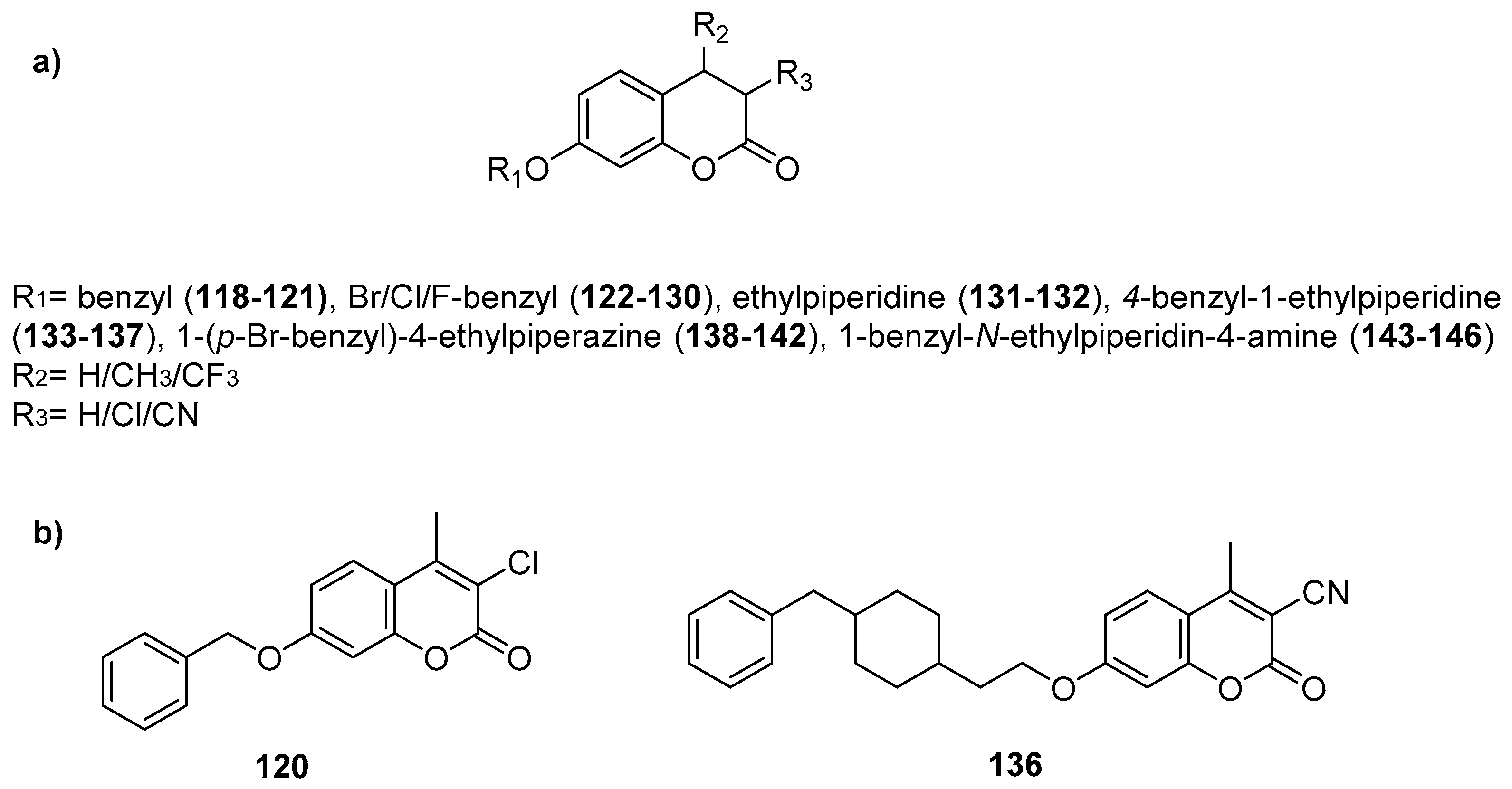
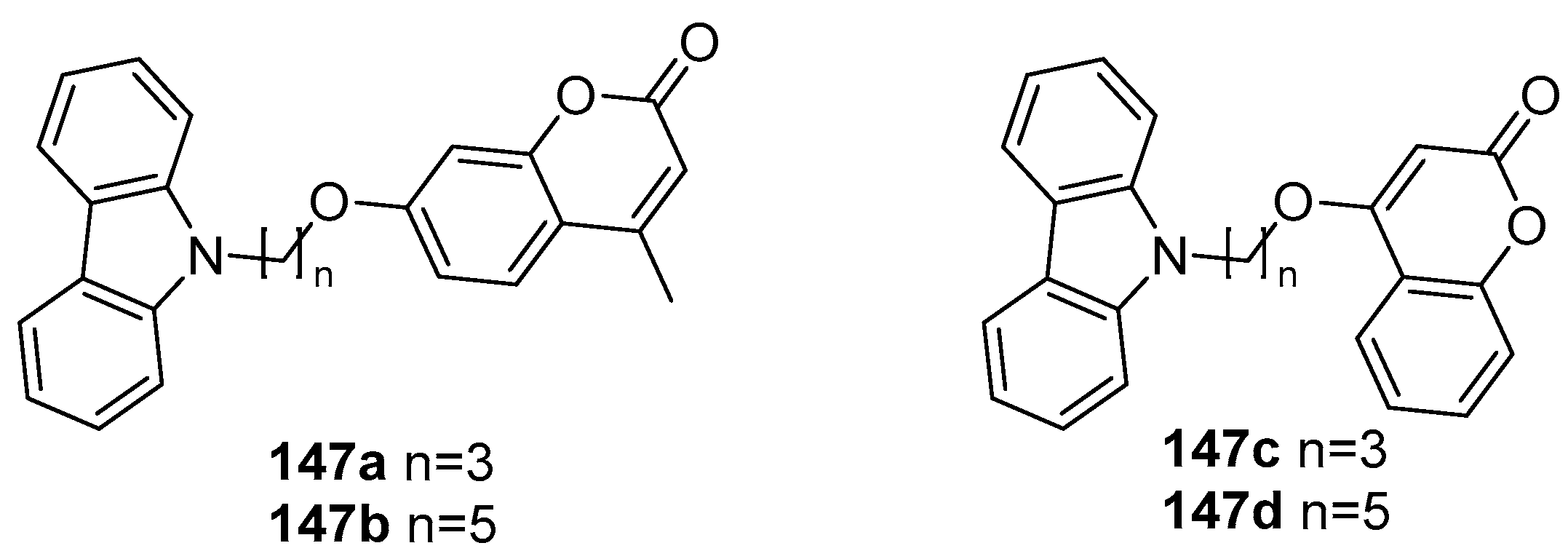
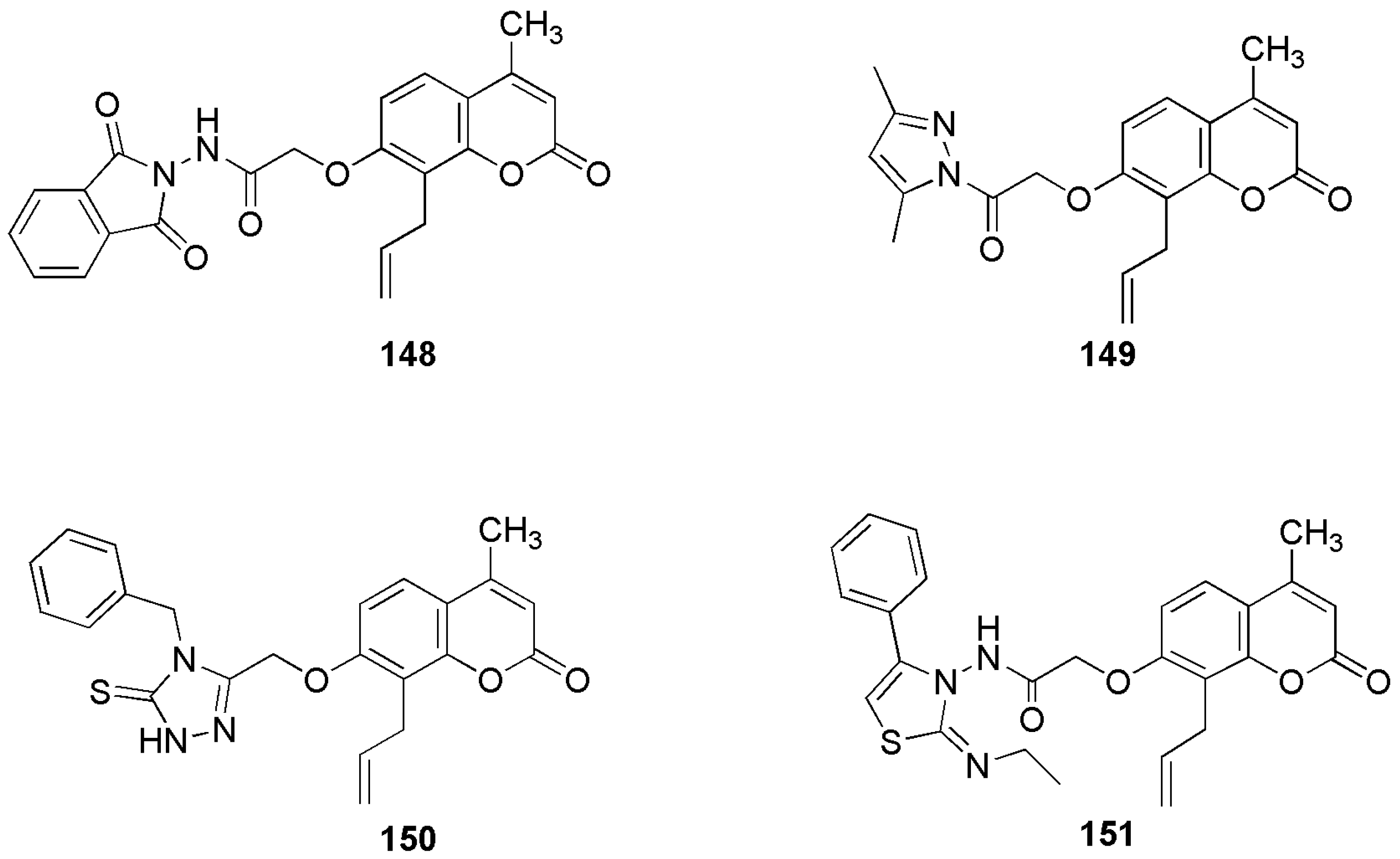
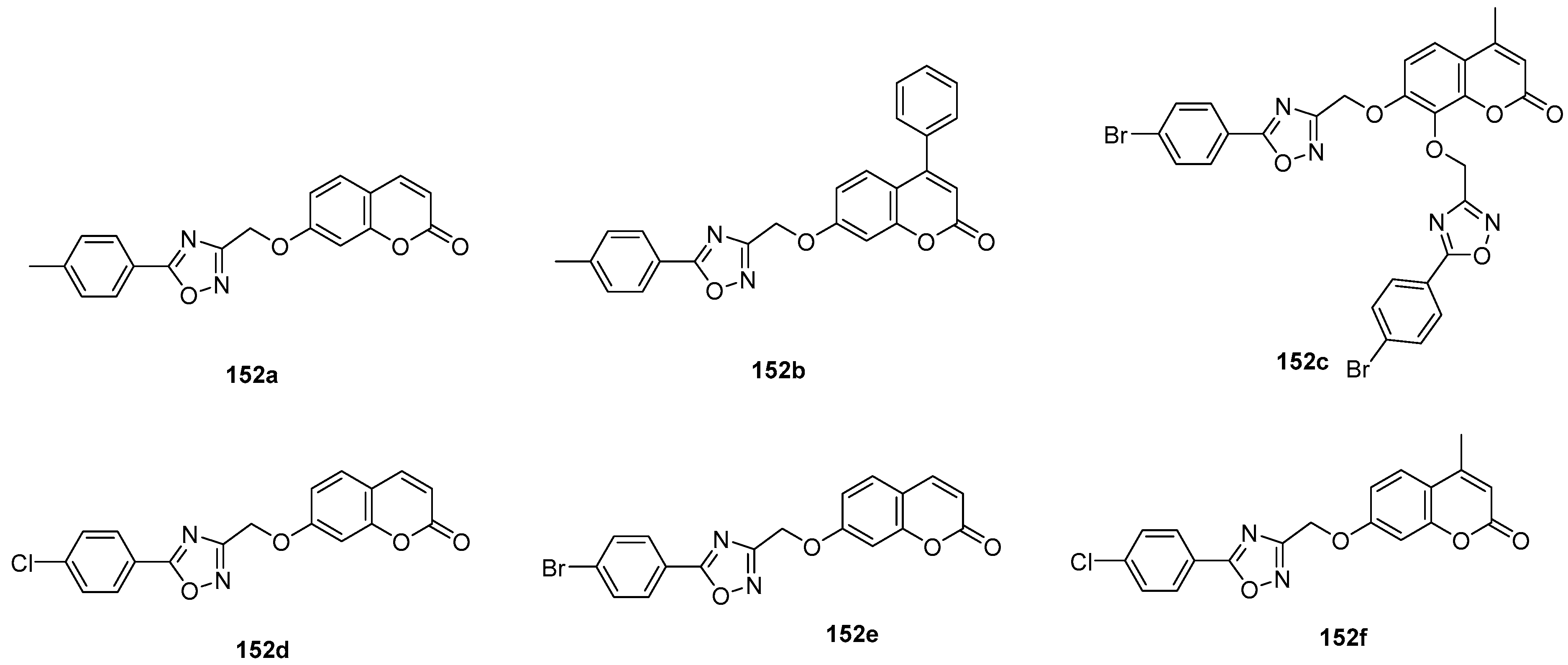
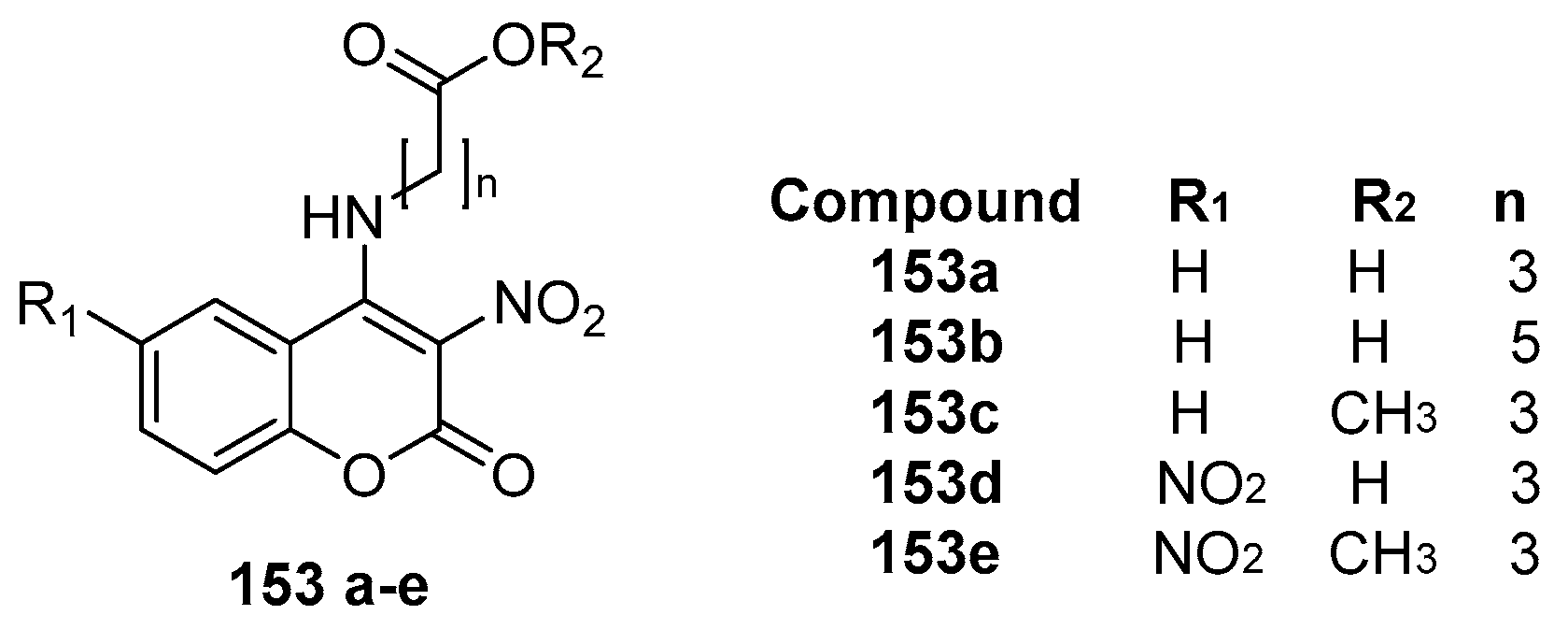
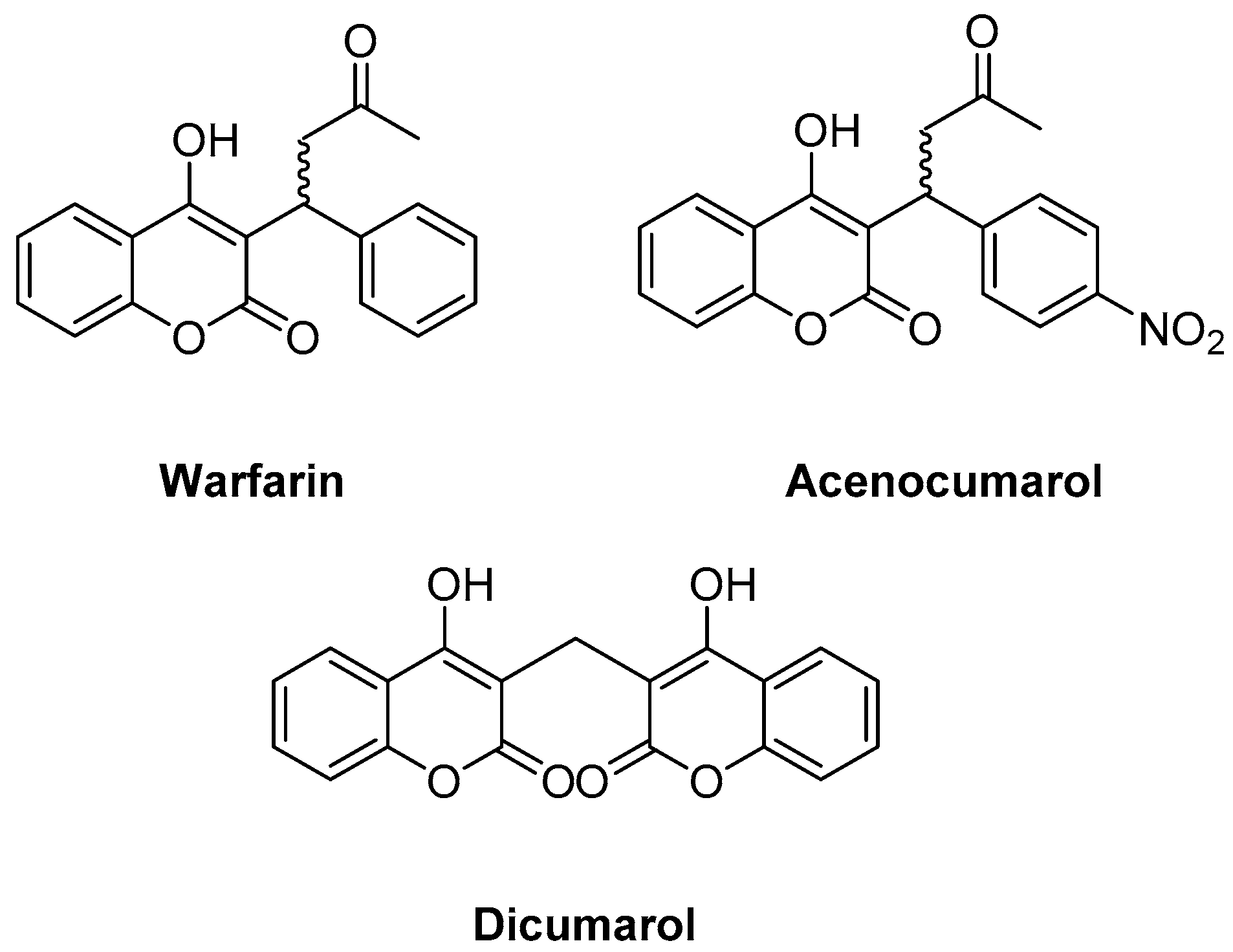
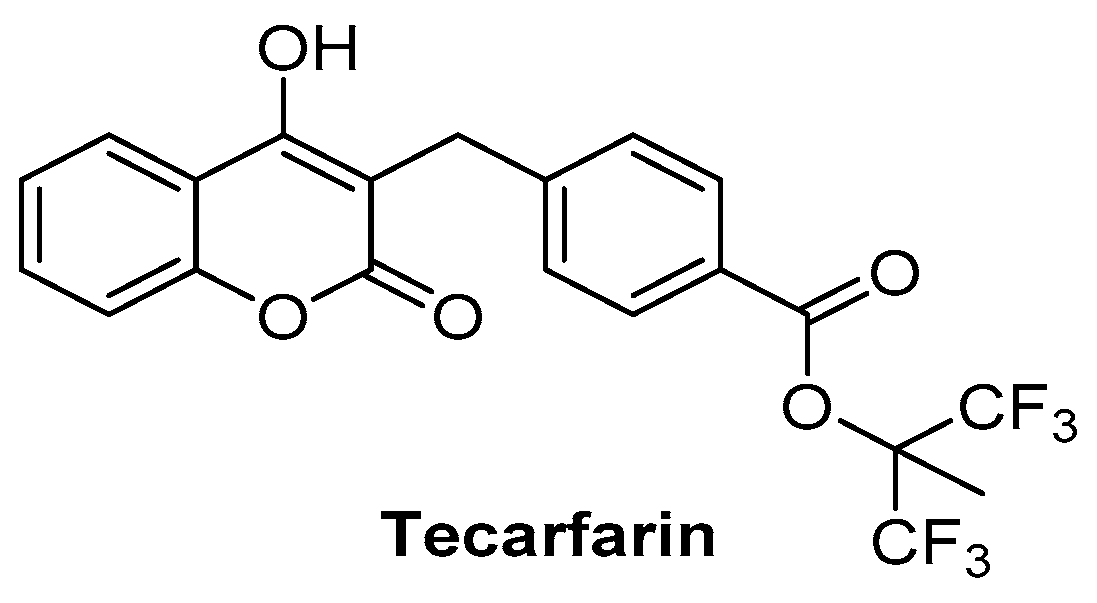
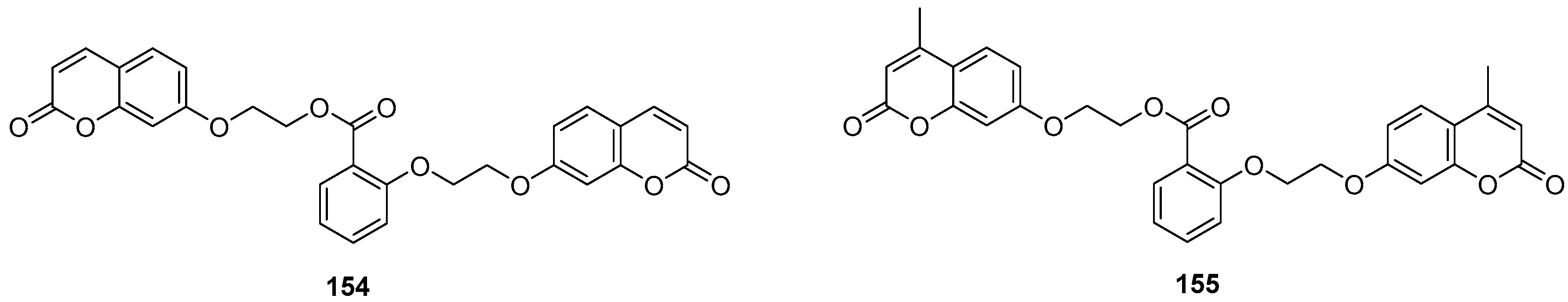
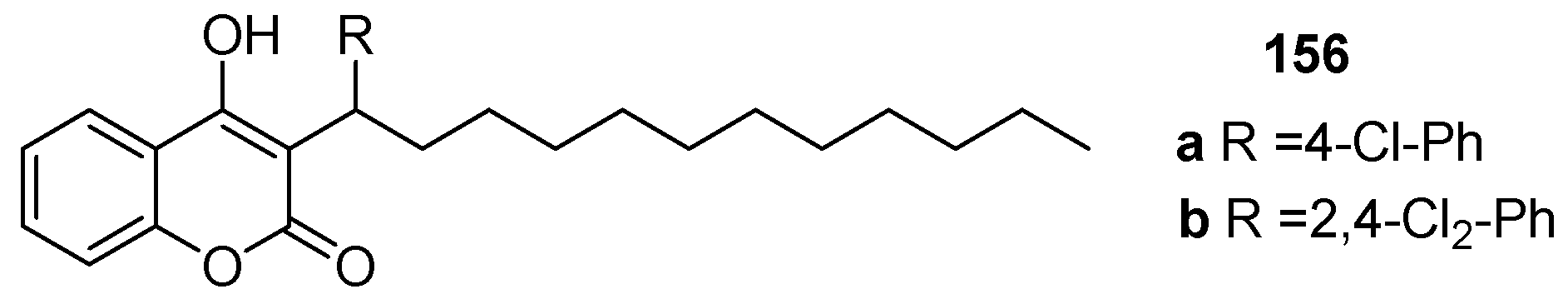
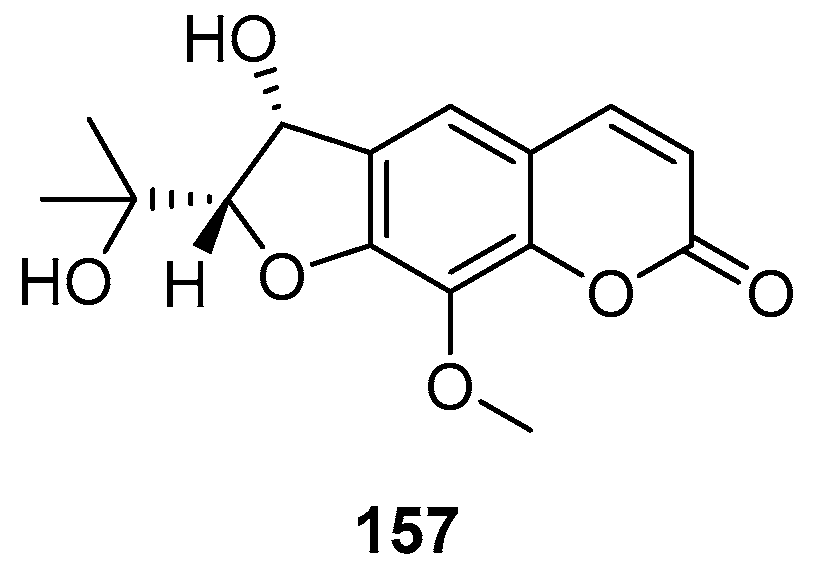
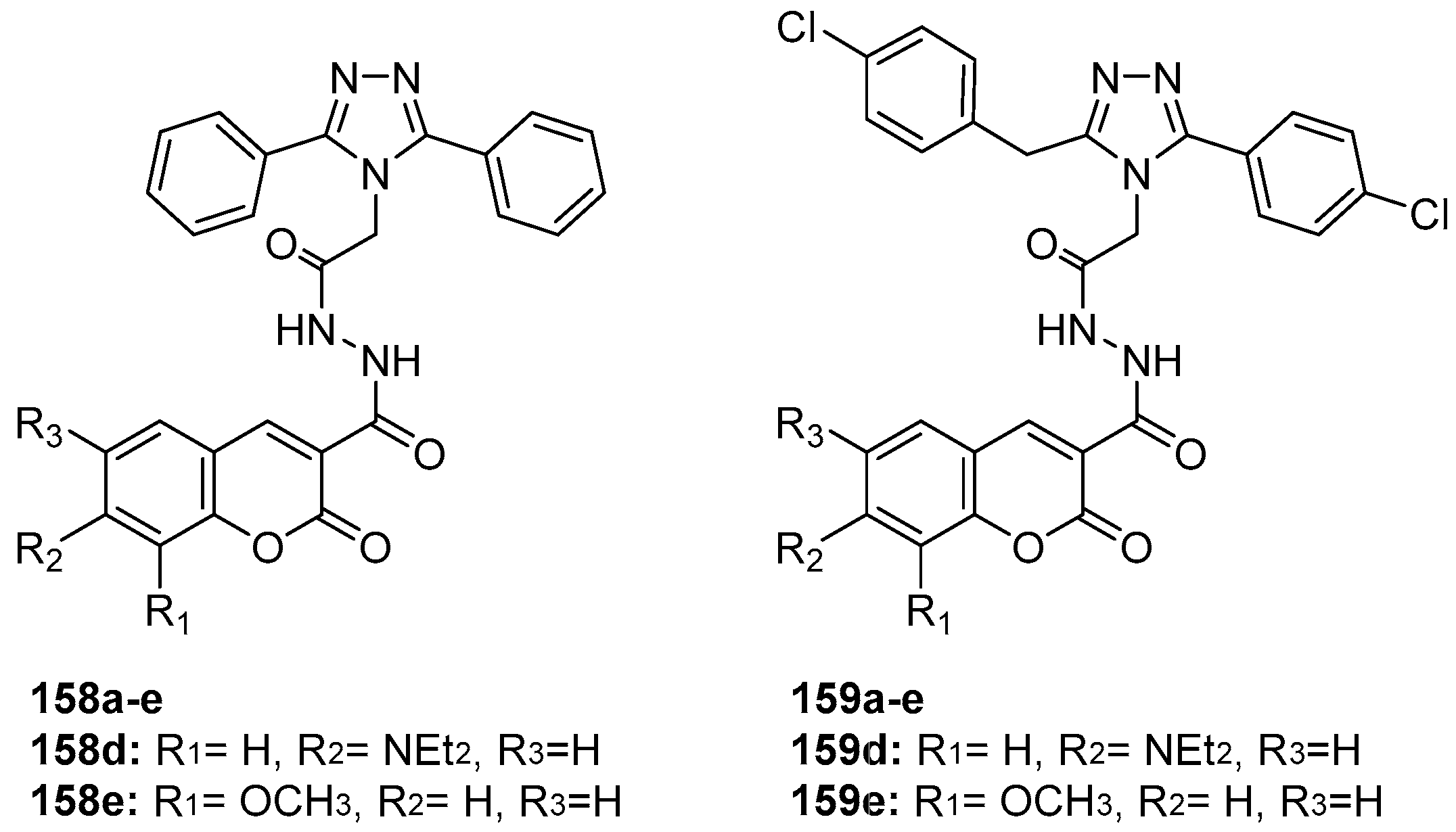
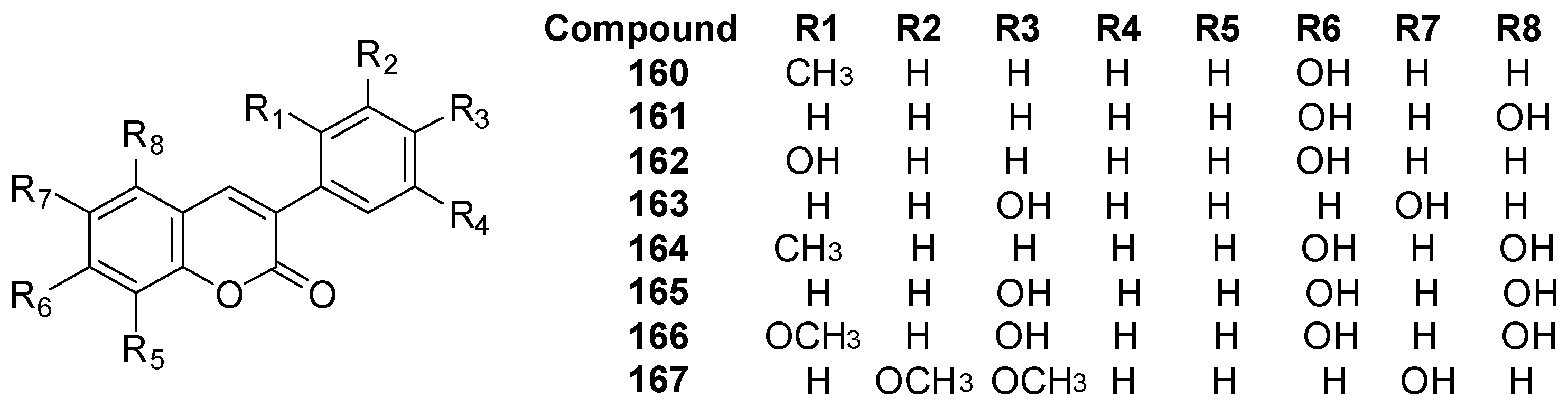
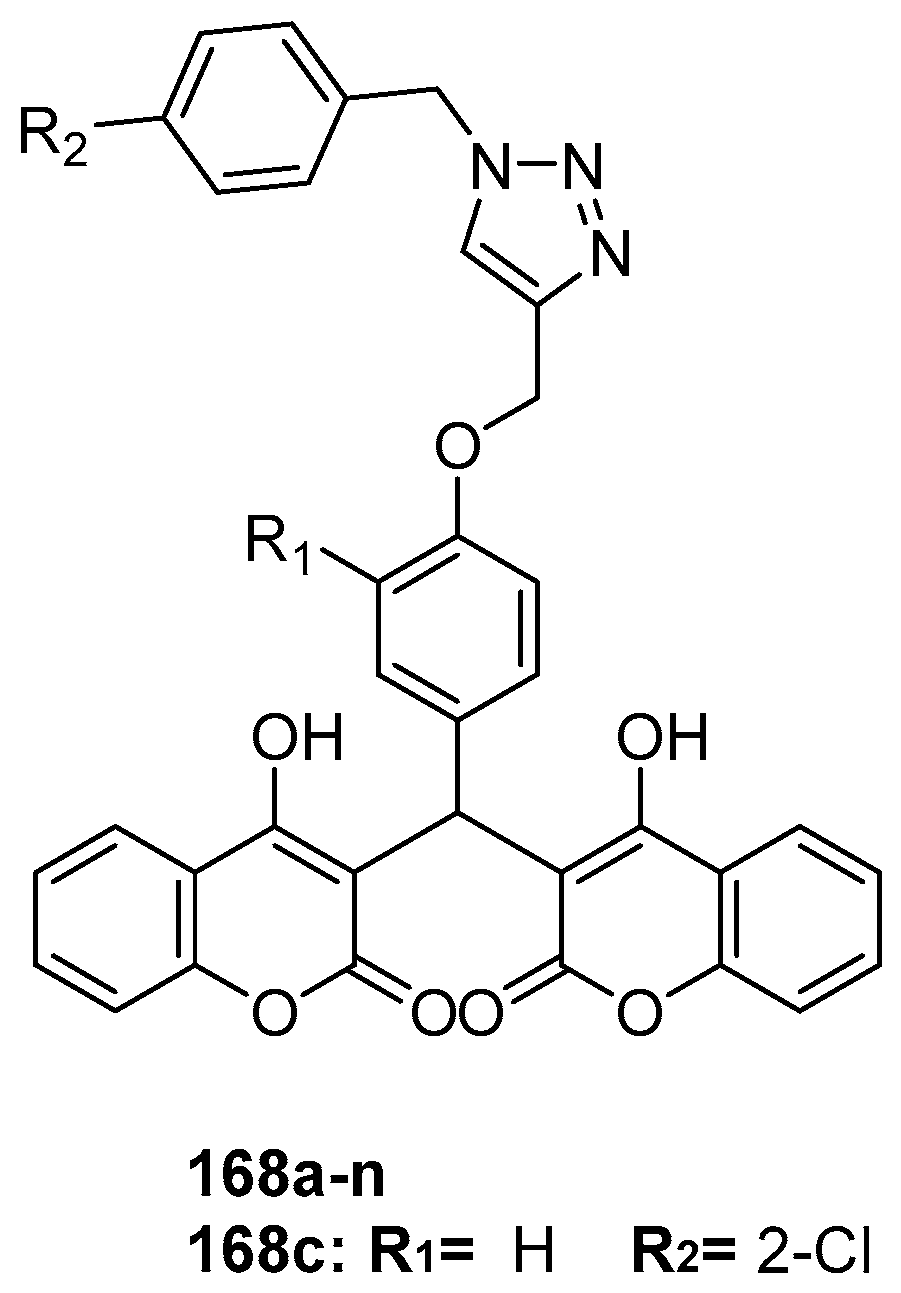

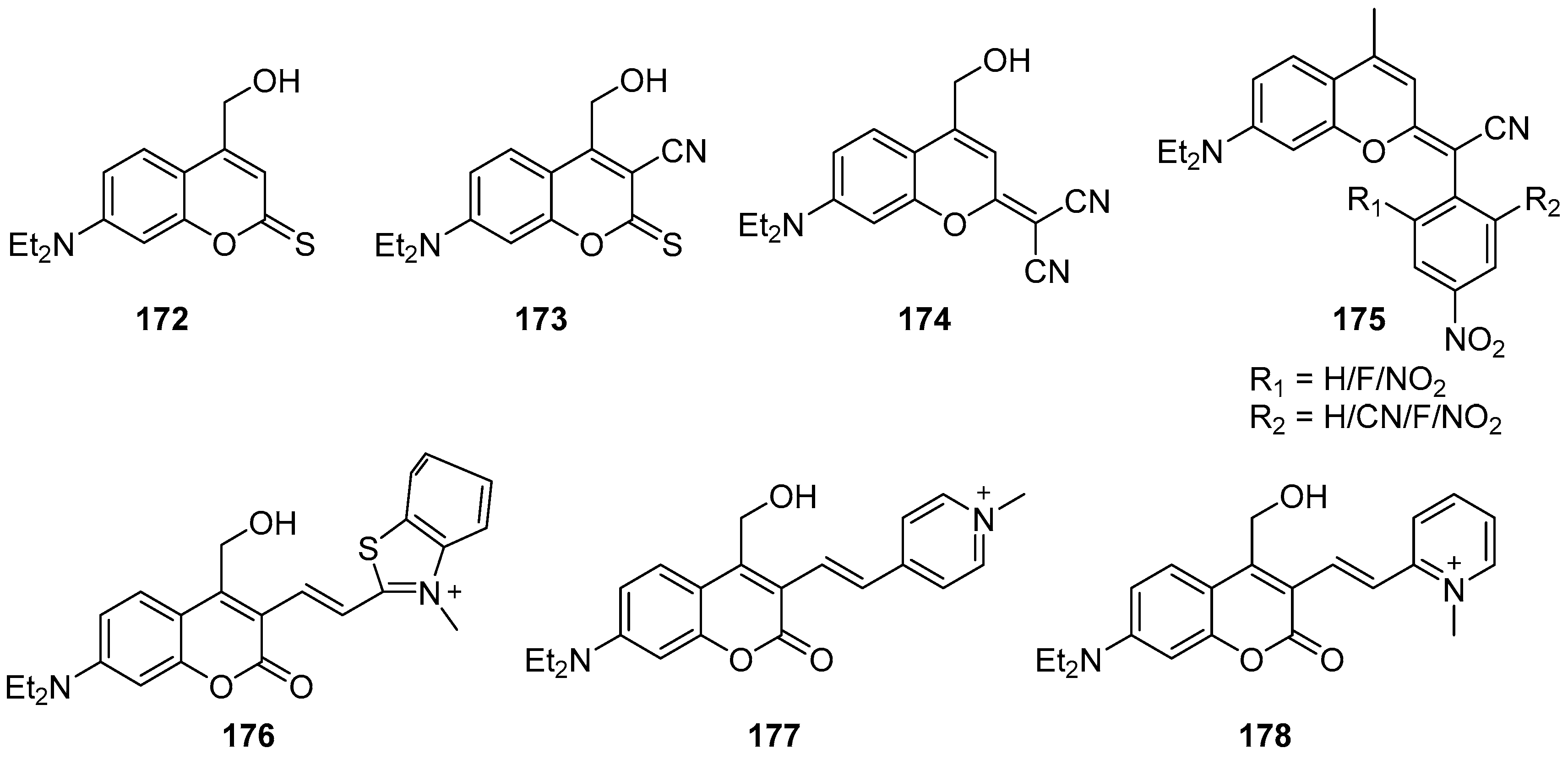
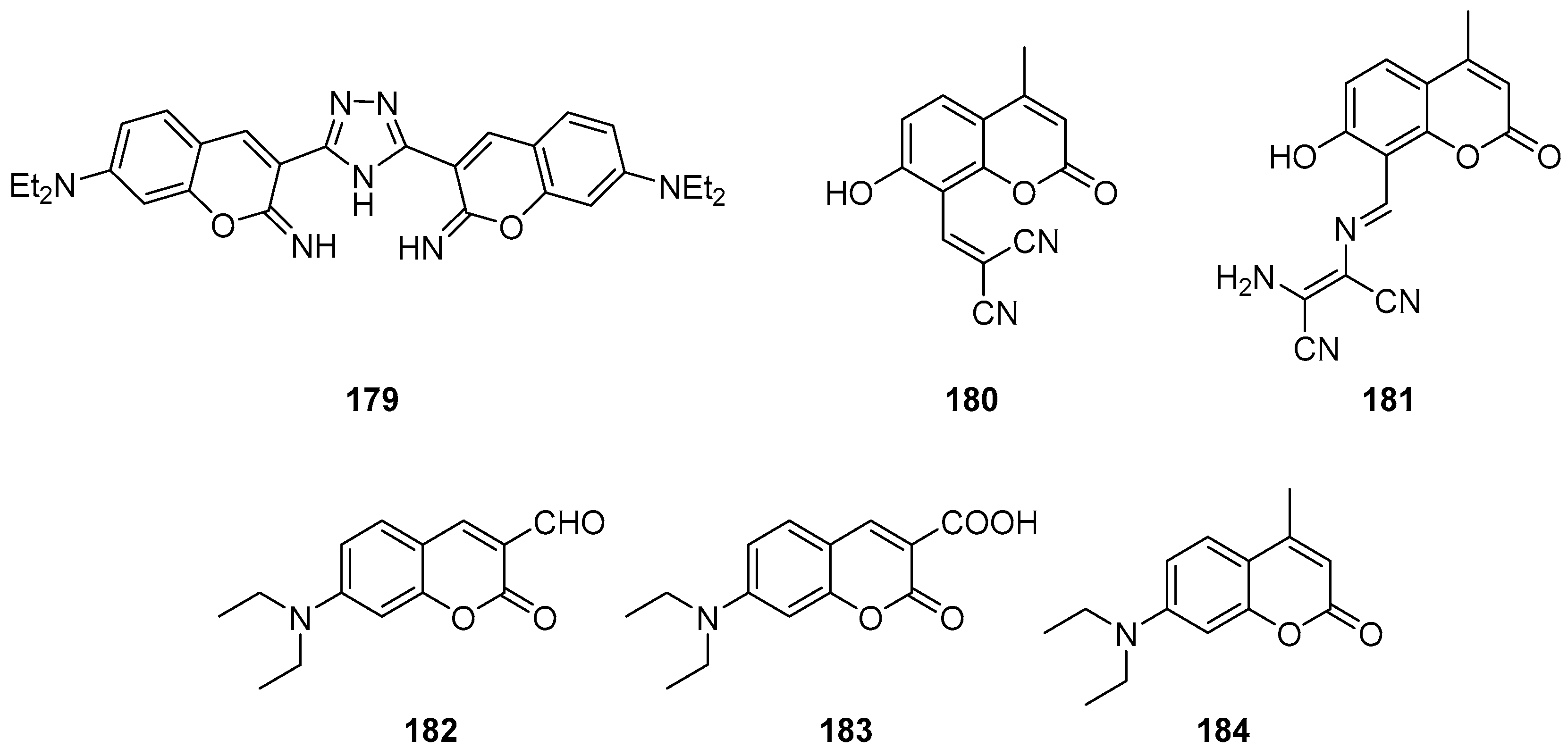
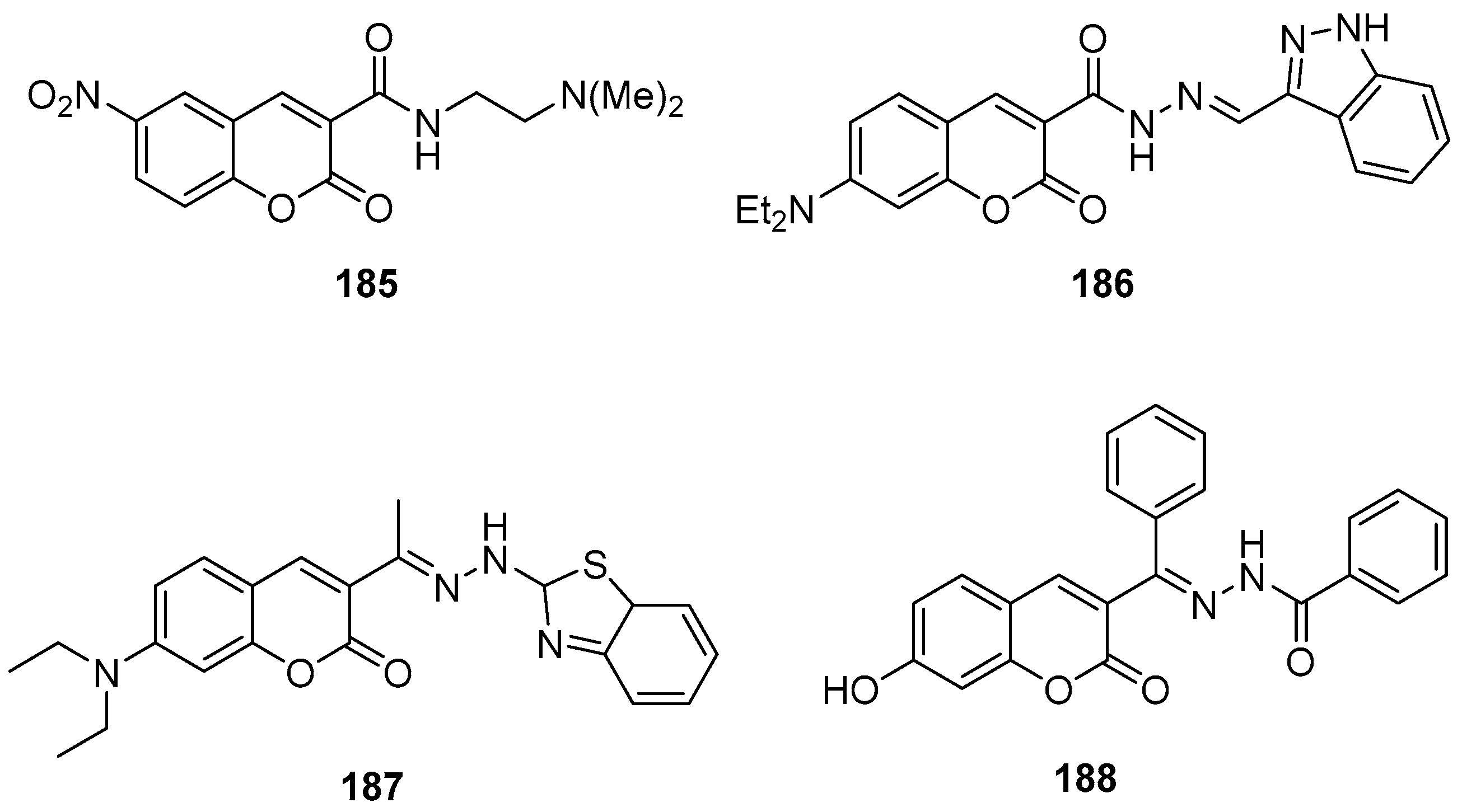

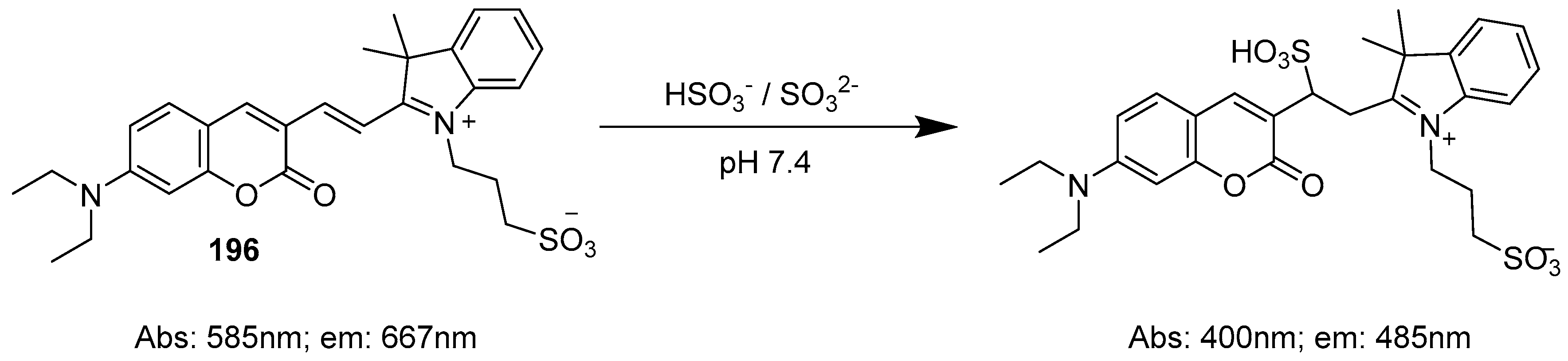
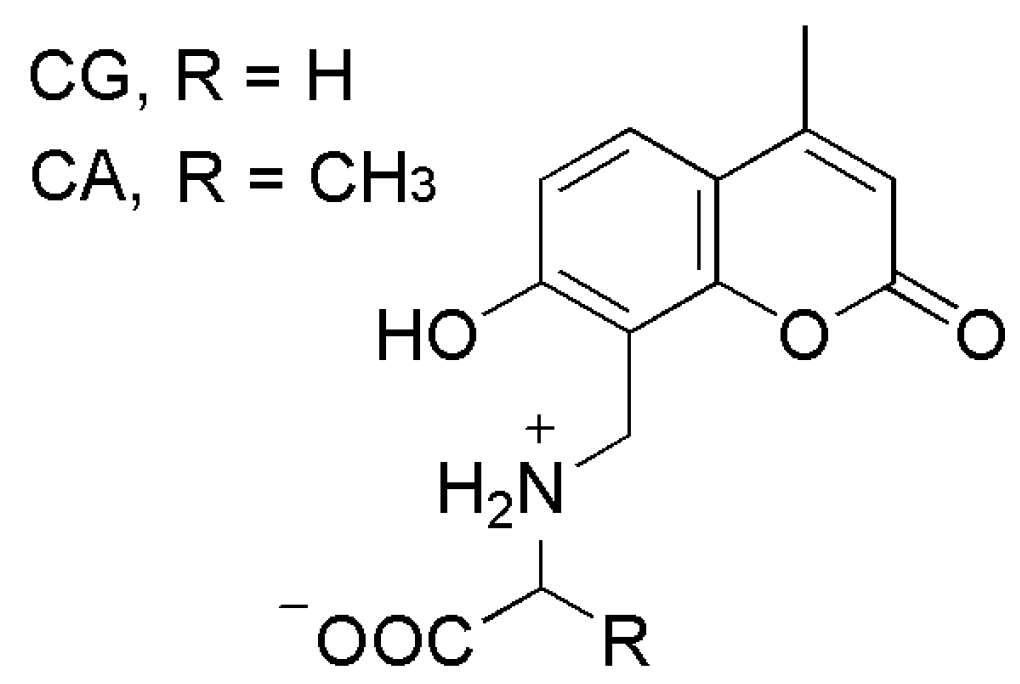
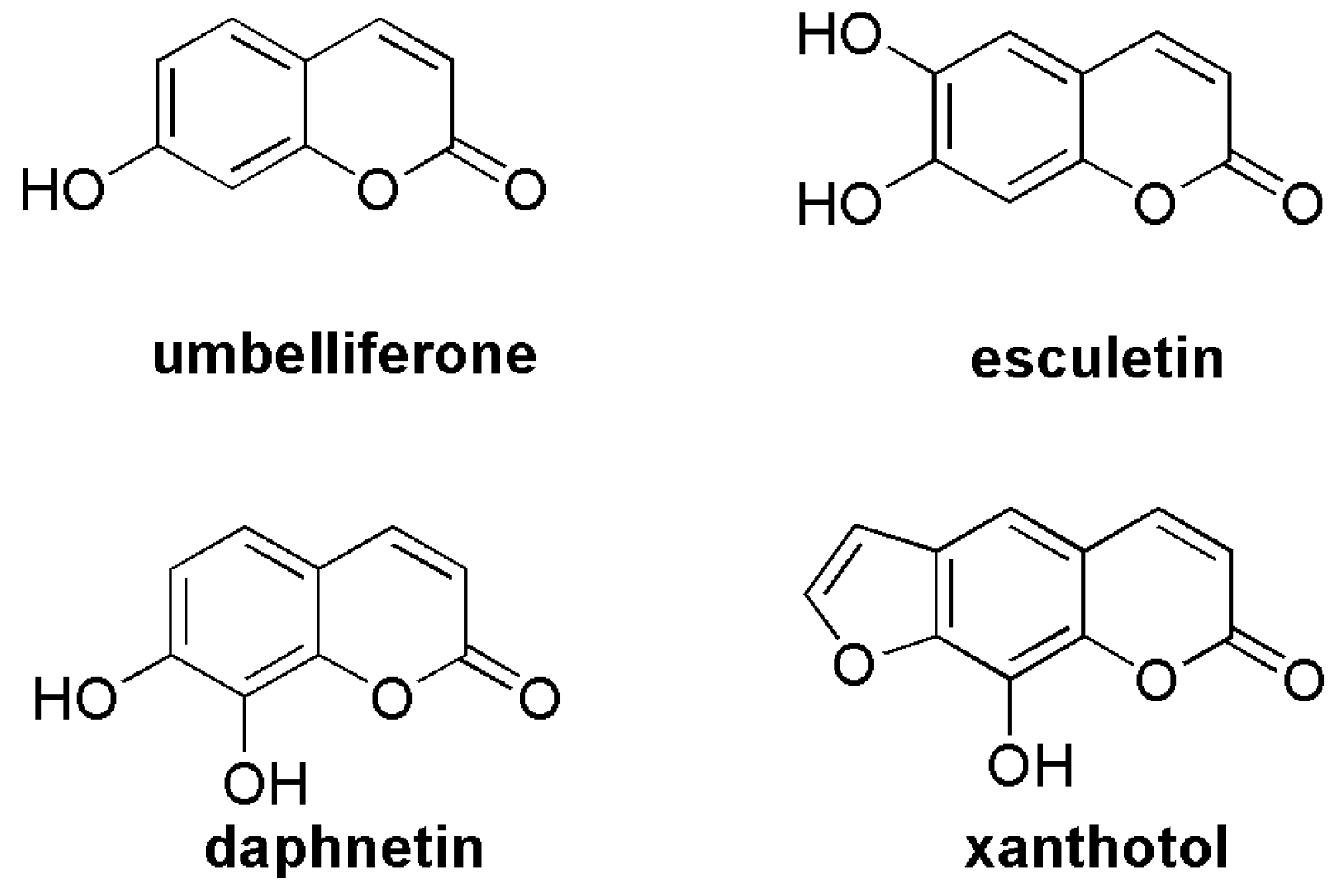
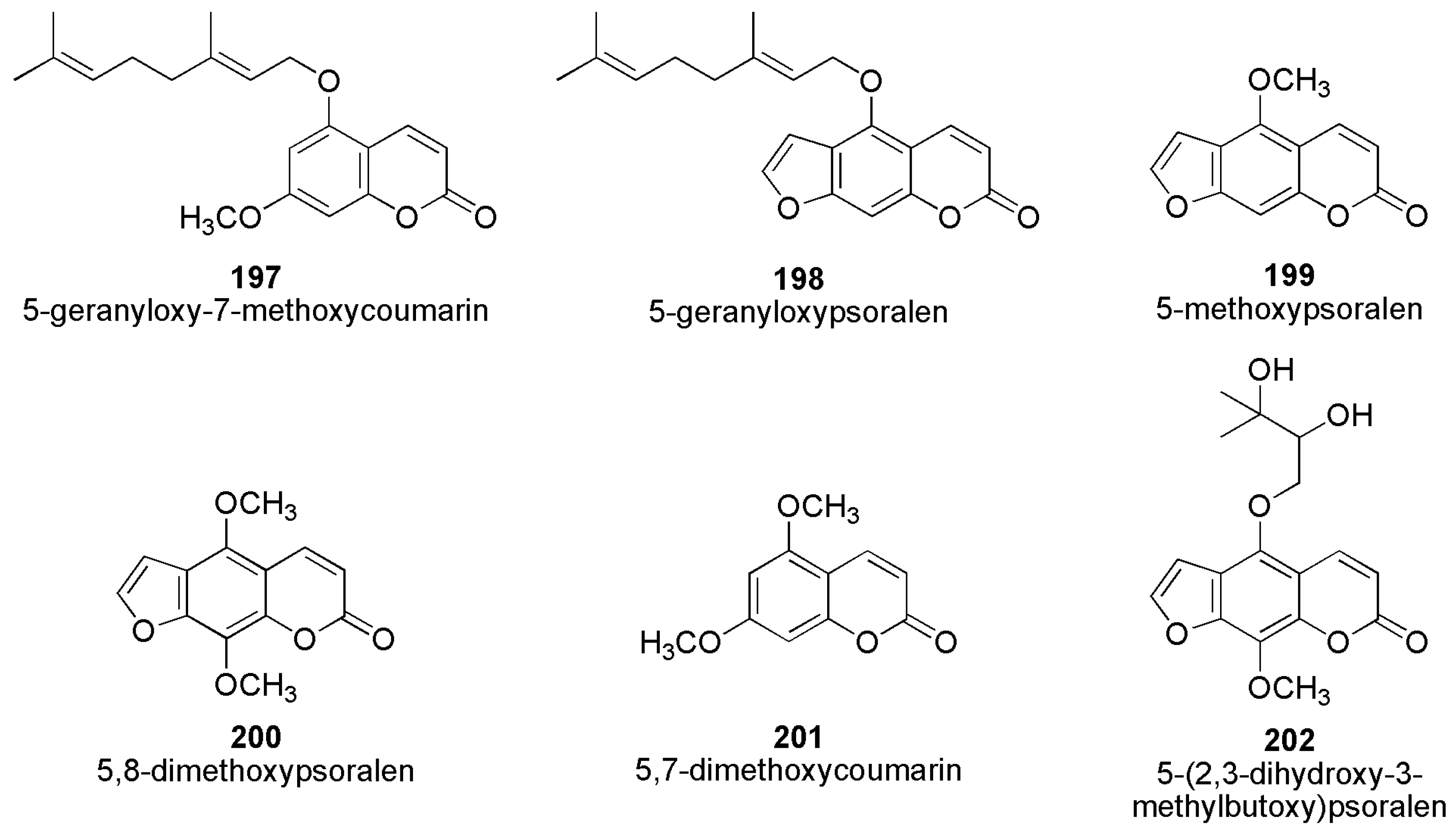

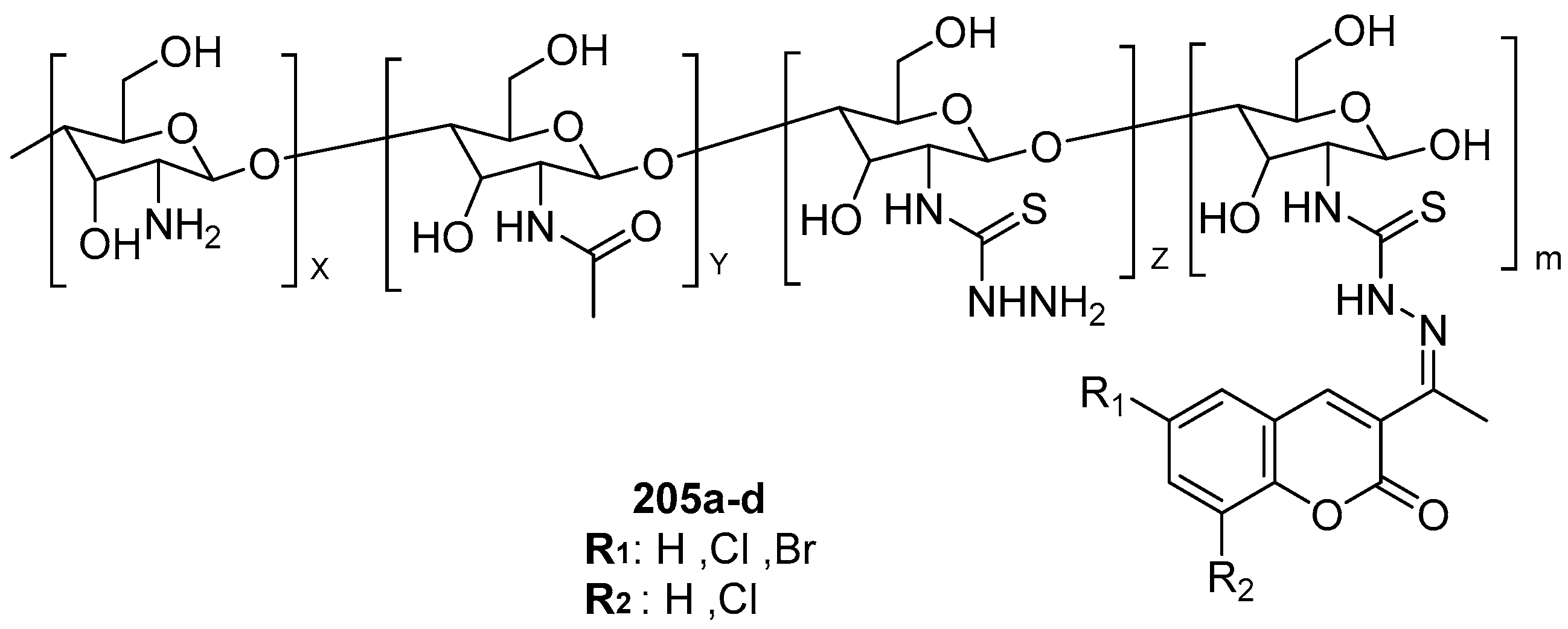
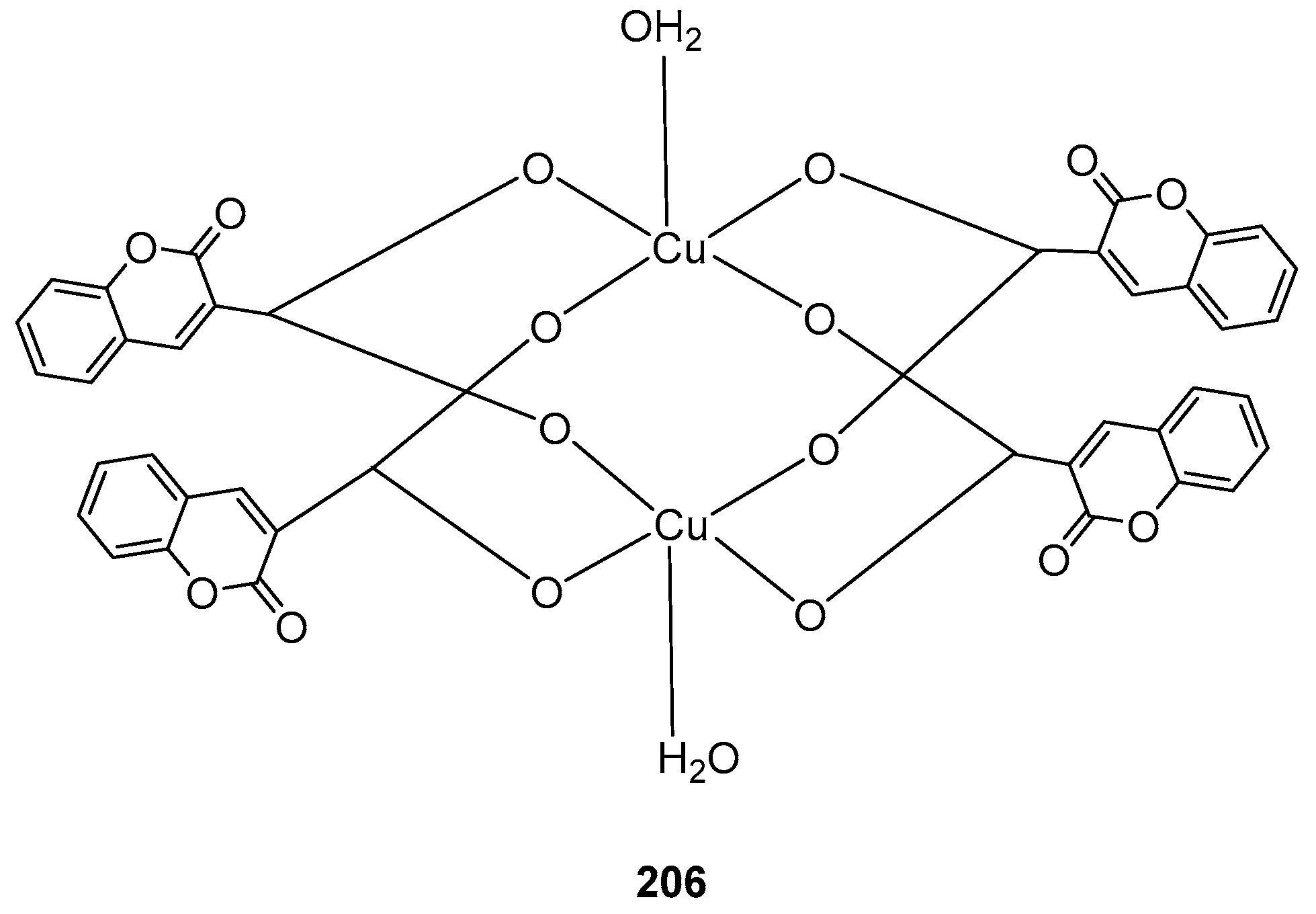
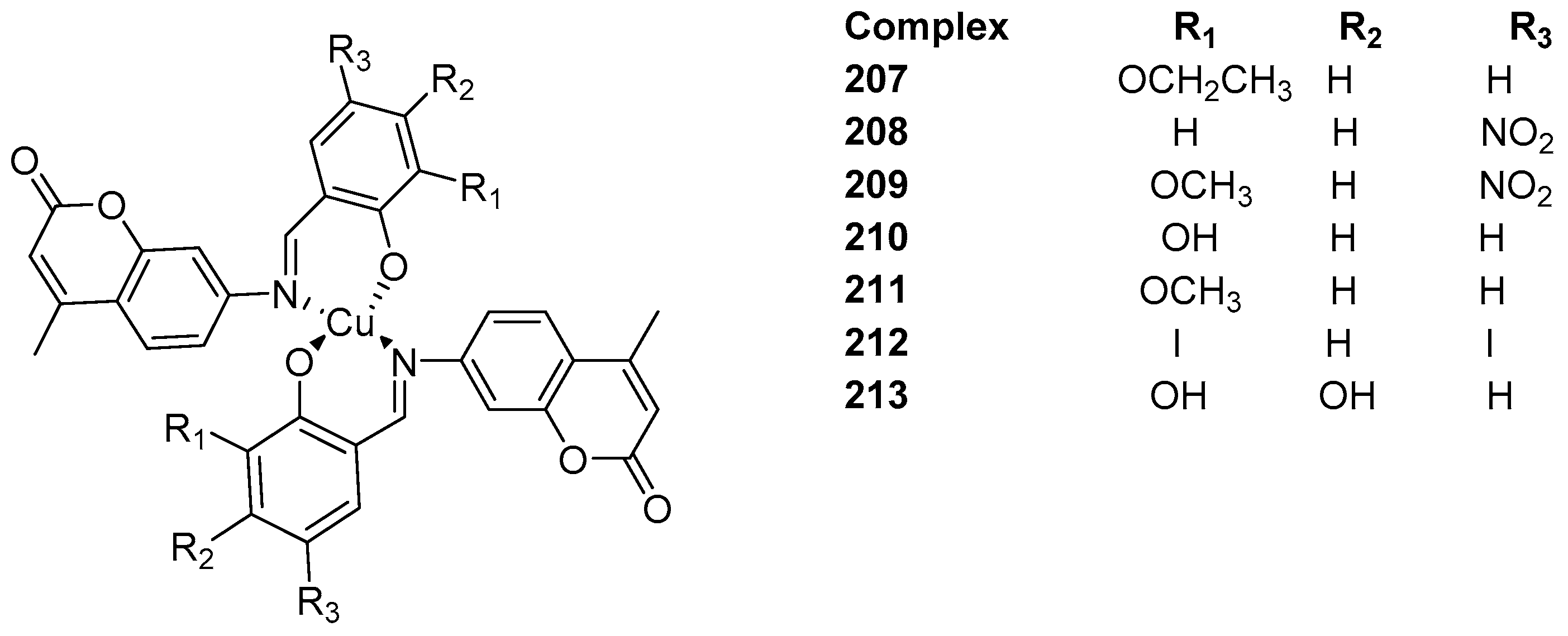
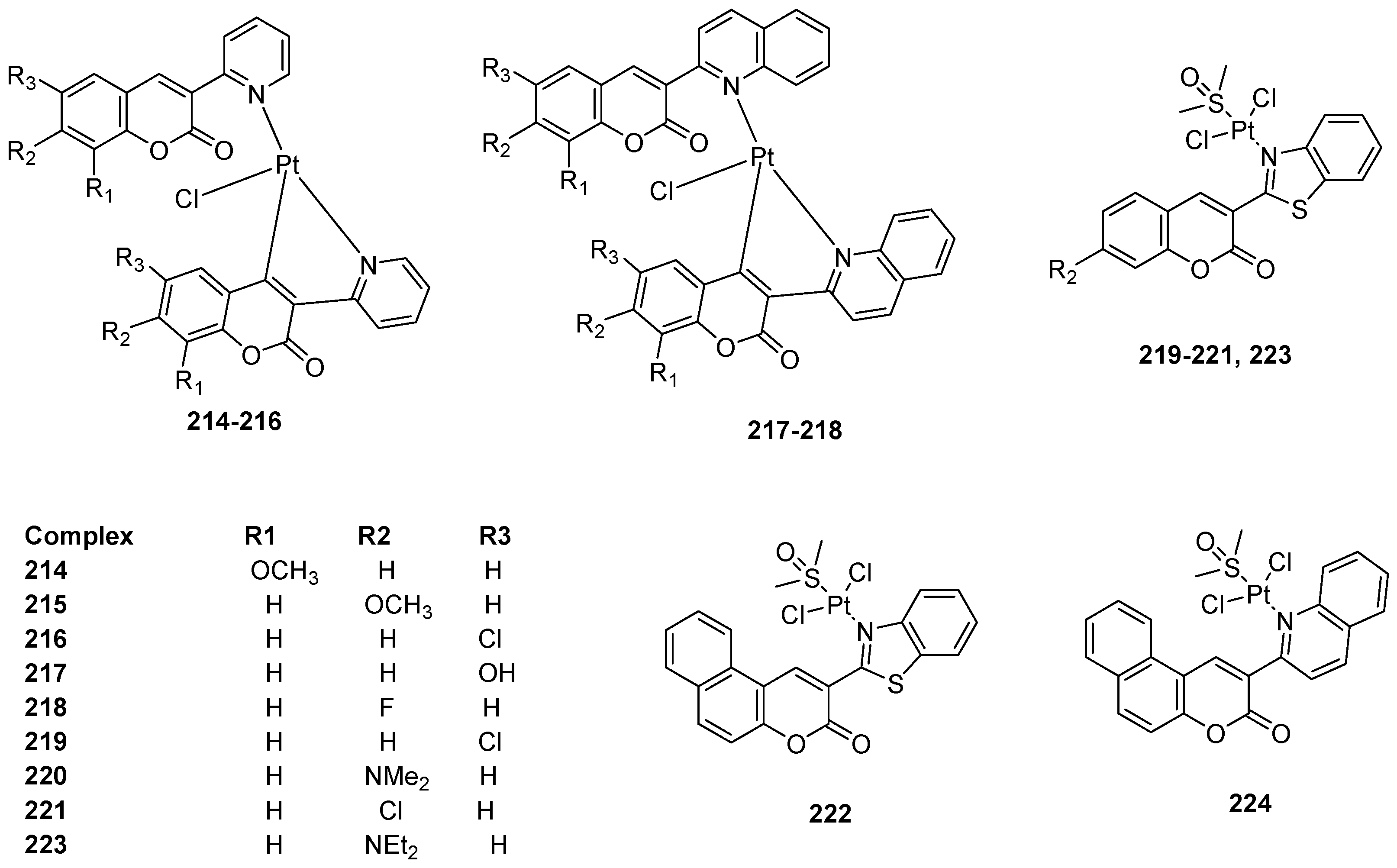
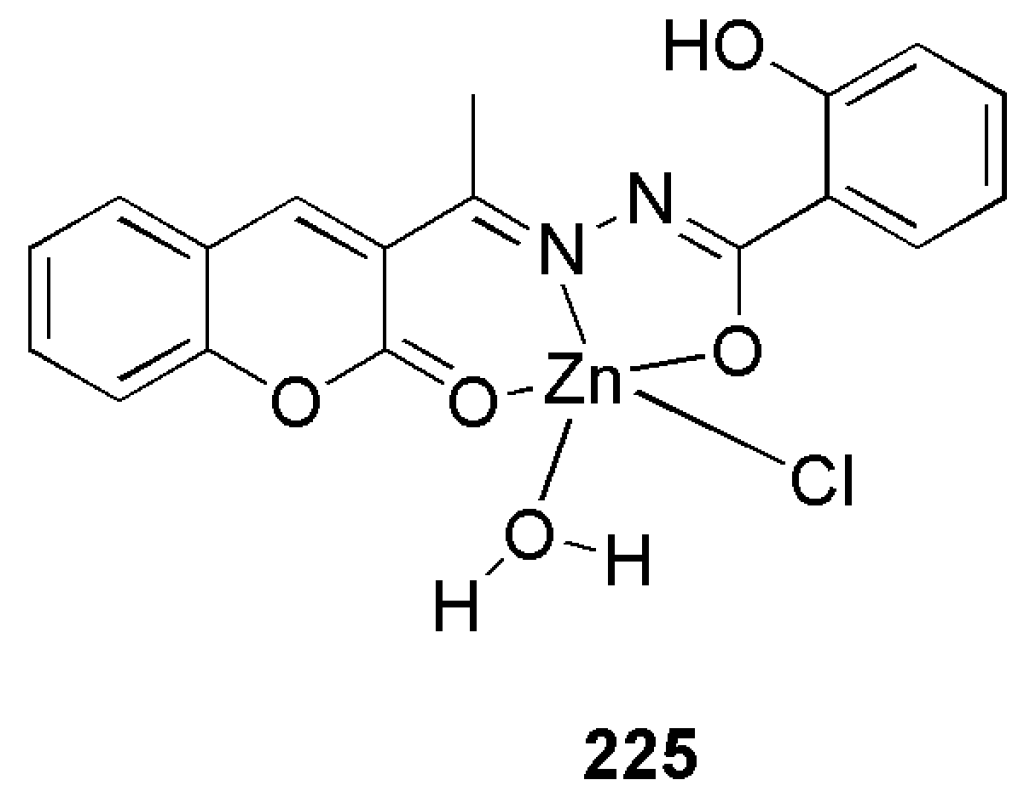
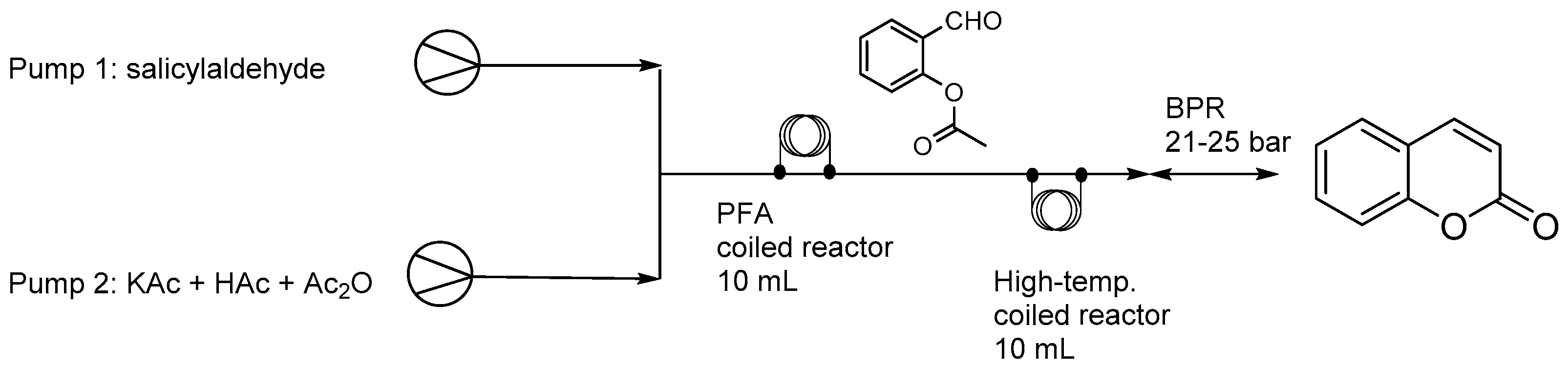
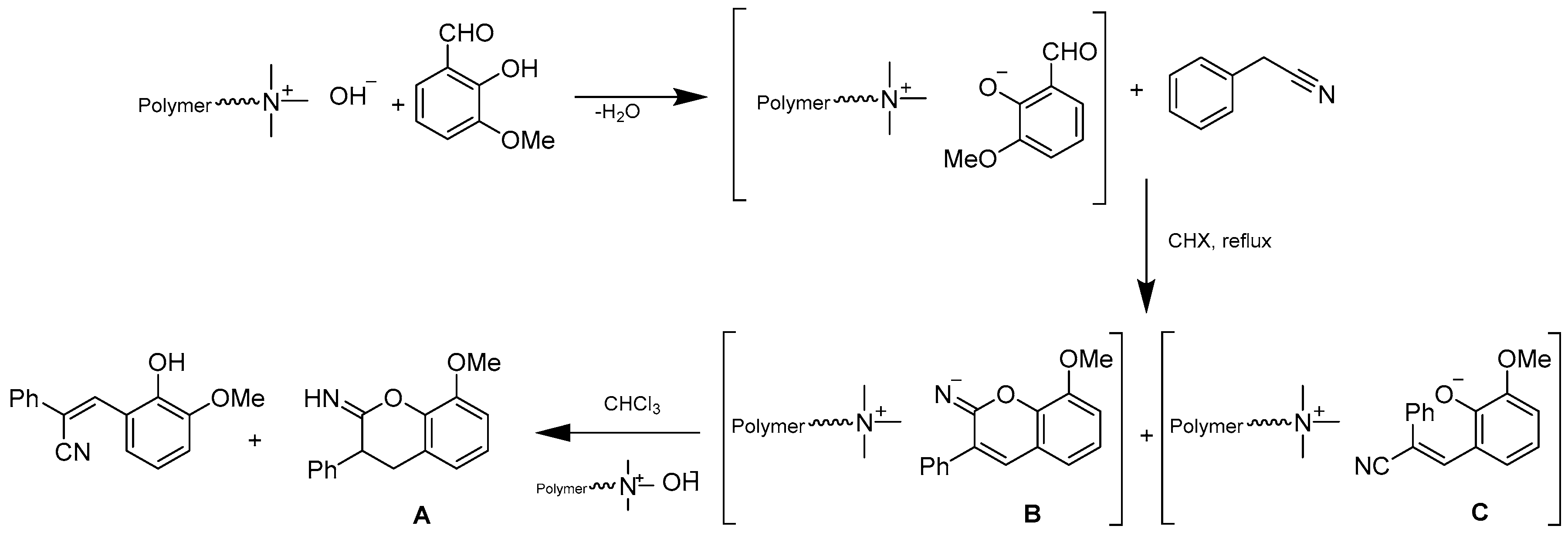
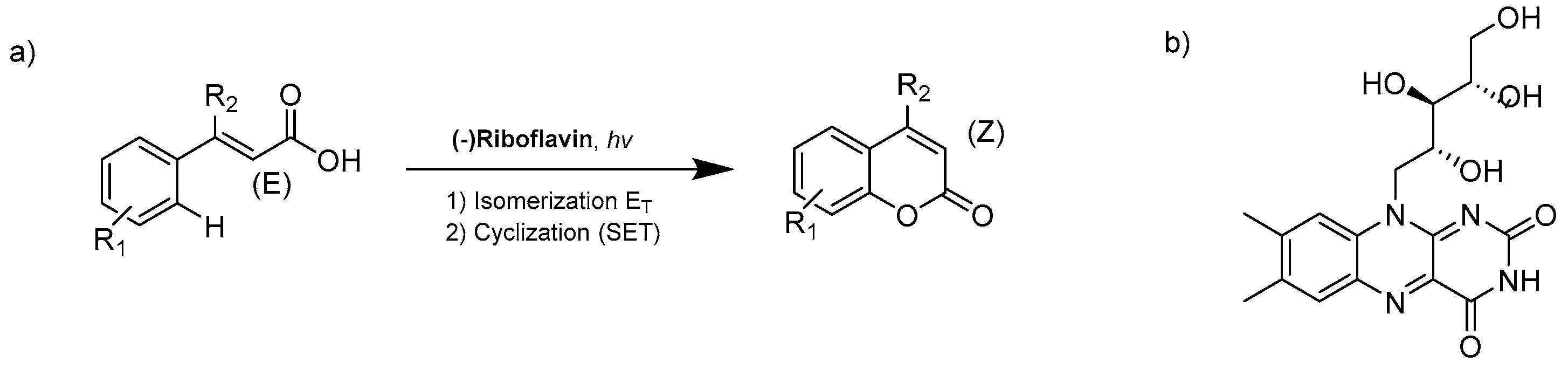

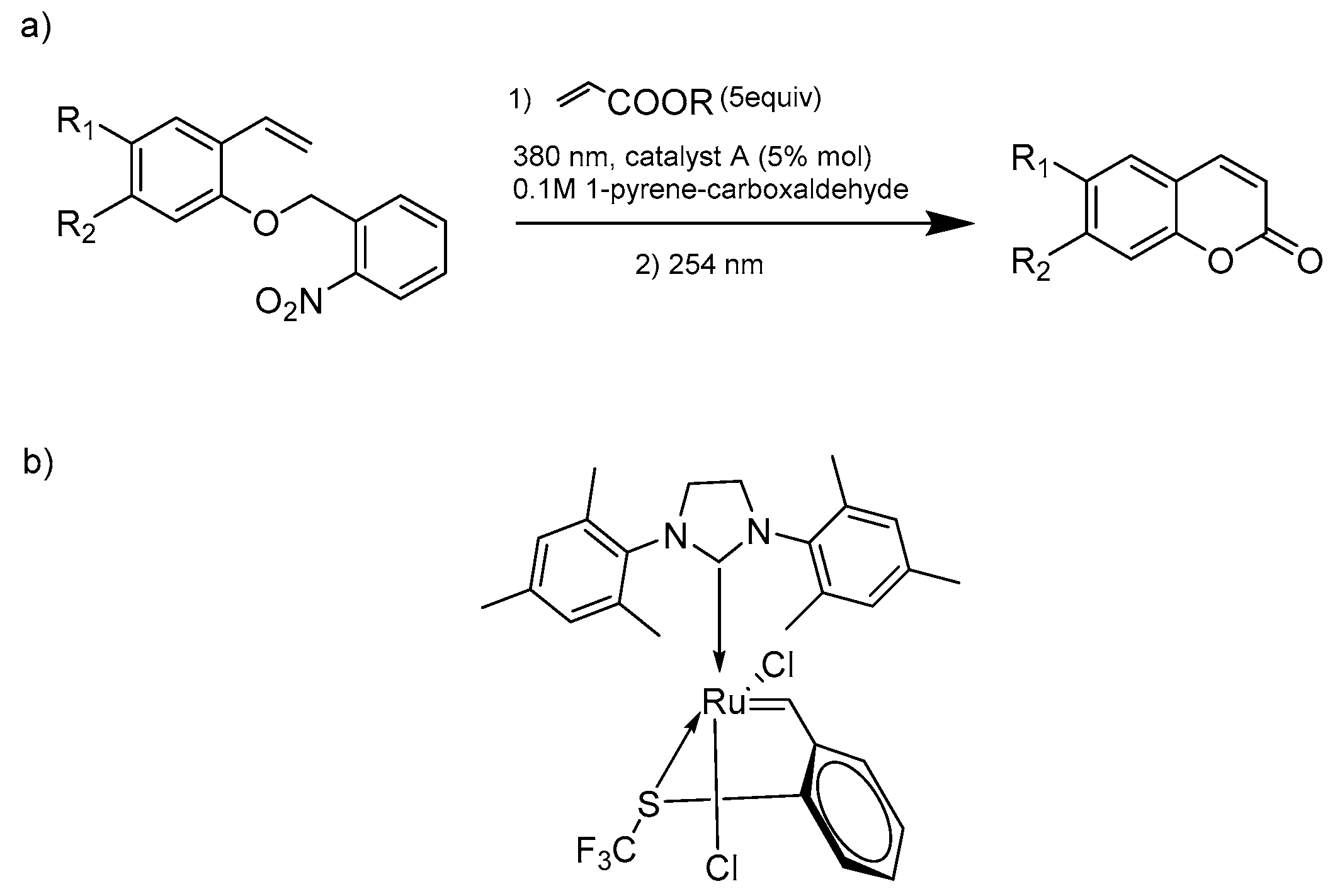
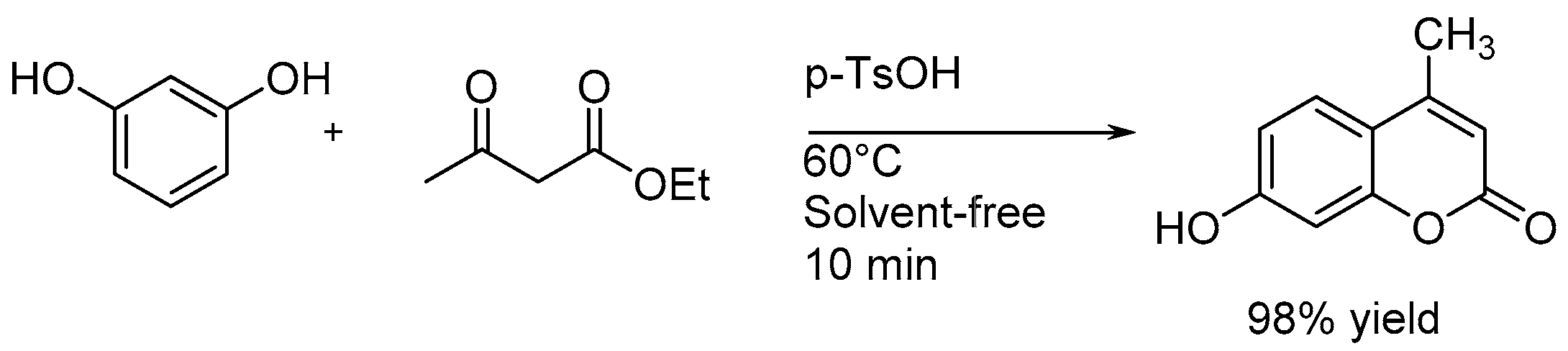
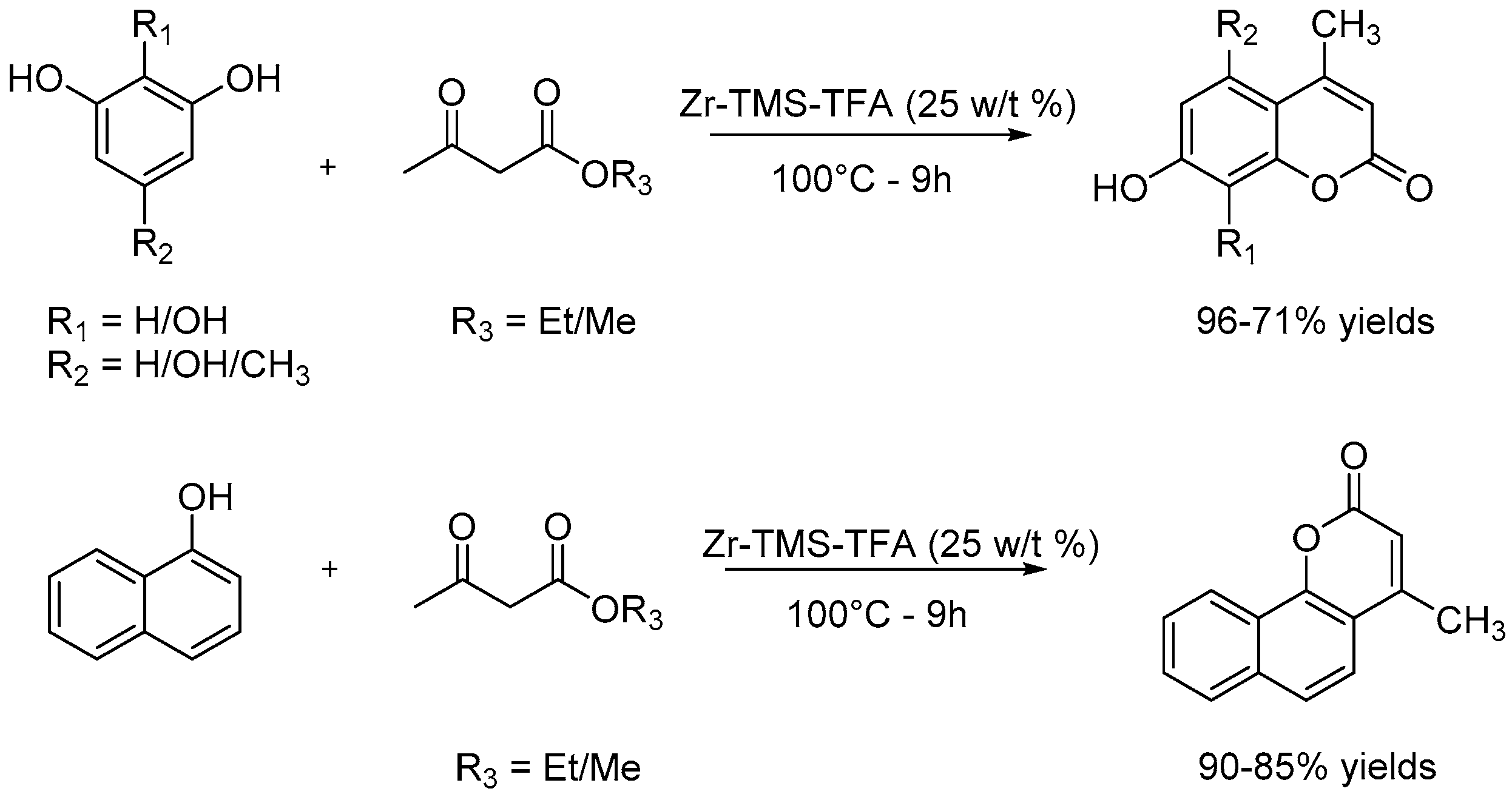
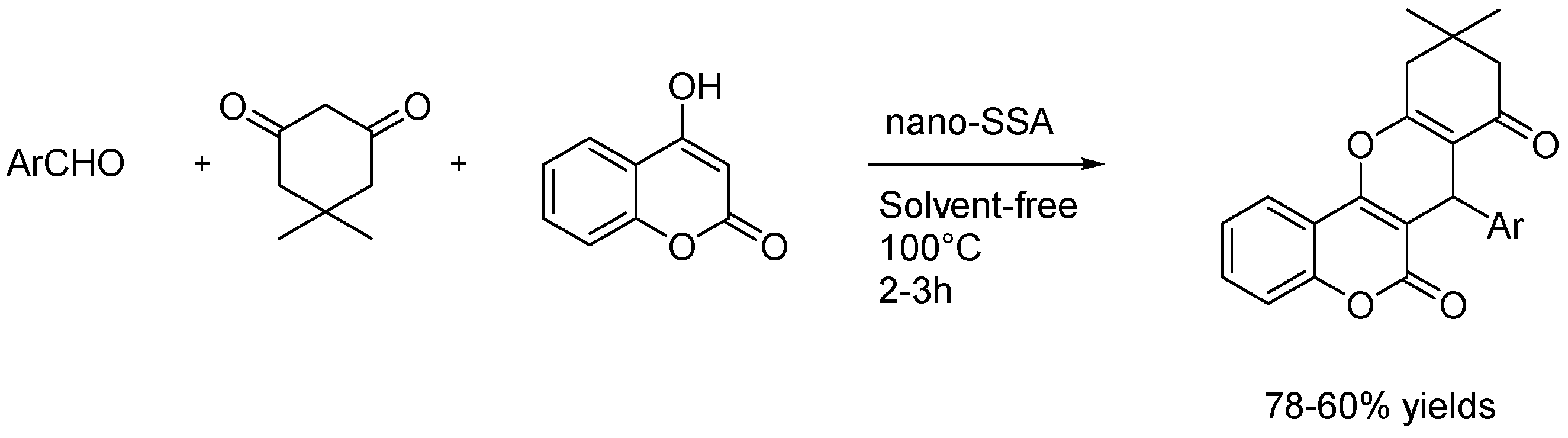
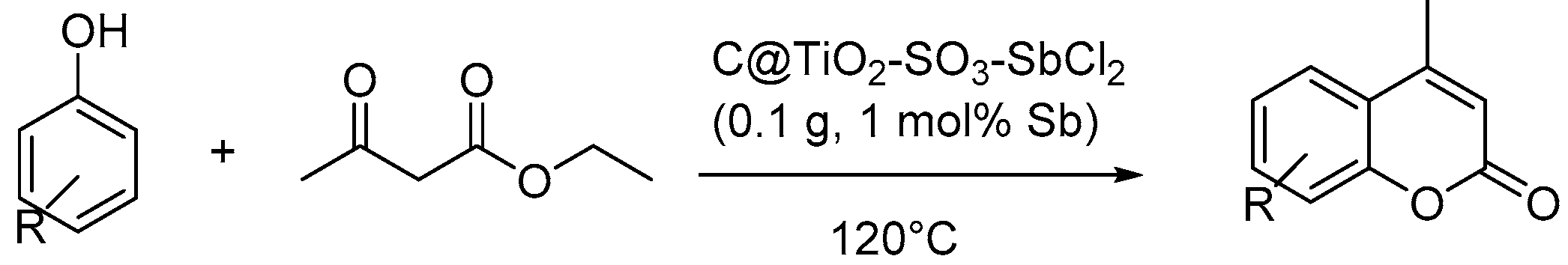
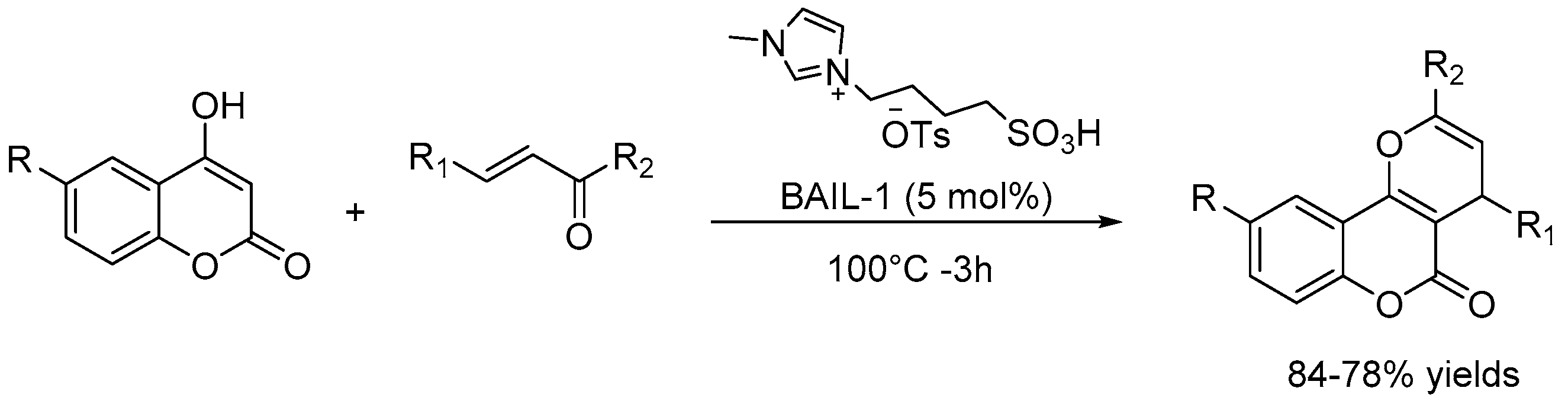
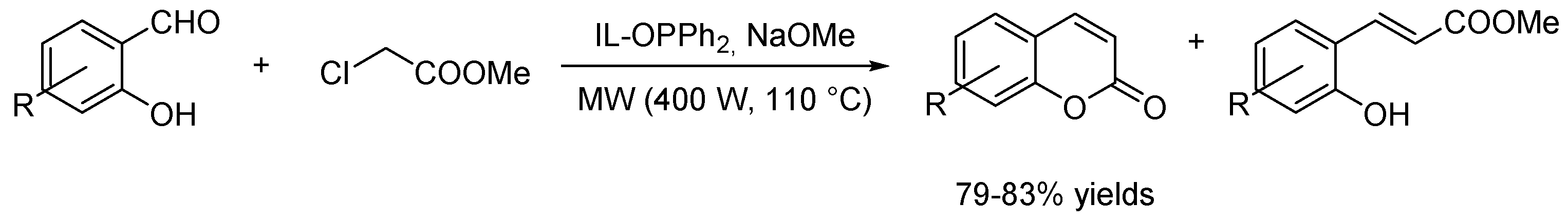
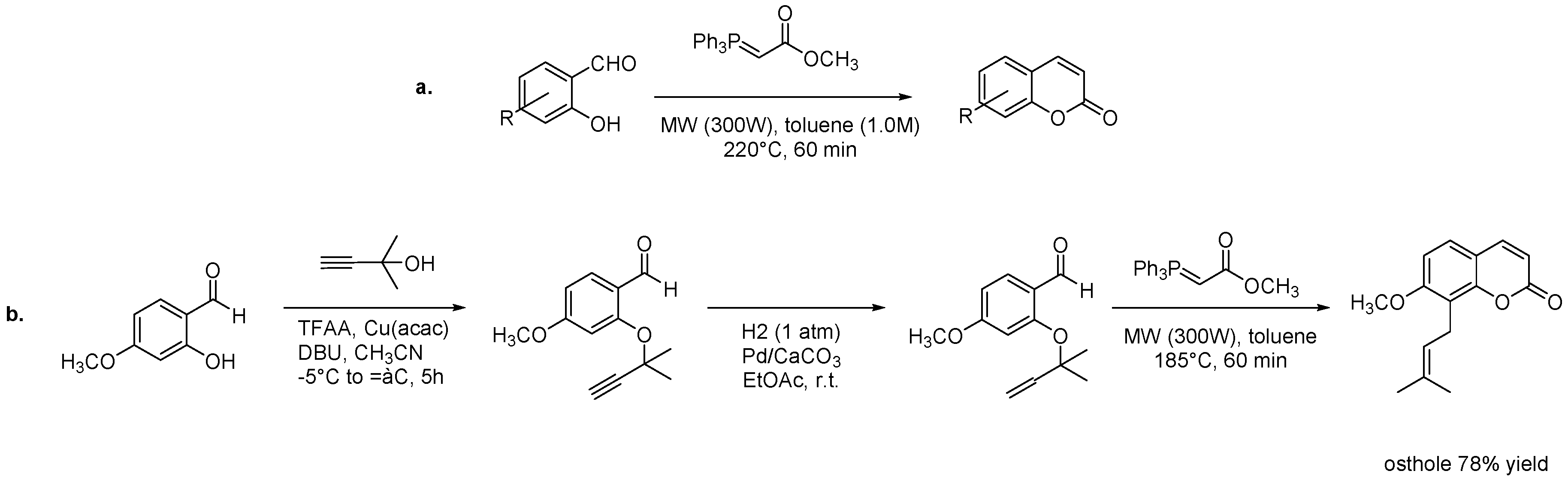
| Class | General Structure | Examples |
|---|---|---|
| Simple coumarins |  | Osthole (neuroprotective, osteogenic, immunomodulatory, anticancer, hepatoprotective, cardiovascular protective and antimicrobial) [13] |
| Furanocoumarins | 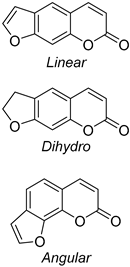 | Psoralen (antifungal) [14] Antoghenol (antibacterial) [15] Columbianedin (anti-inflammatory) [15] |
| Pyranocoumarins | 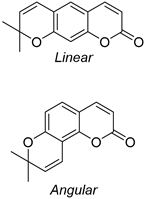 | Grandivittin (antibacterial) [16] Inophyllum A, B, C, E, P, G1, andG2 (antiviral) [17] |
| Biscoumarins |  | Dicoumarol (anticoagulant) [18] |
| Isocoumarins |  | Thunberginols (antidiabetic) [19] |
| Phenylcoumarins |  | Isodispar B (anti-inflammatory) [20] |
| Structure | Biological Activity | Molecular Target | Name/Number | Ref. | Origin |
|---|---|---|---|---|---|
 | Antioxidant | Free radicals | 3 | [30] | Synthetic |
 | Antioxidant | Free radicals | 9 | [35] | Synthetic |
 | Antioxidant | Free radicals | 10d | [36] | Synthetic |
 | Antioxidant | Free radicals | 15 | [38] | Synthetic |
 | Antiprolifera-tive | DNA, chromatin | 18 | [45] | Natural |
 | Antiprolifera-tive | Topoisomerase 1 | 20 | [47] | Synthetic |
 | Antiprolifera-tive | Tumor-suppressor protein p53 | 21 | [49] | Synthetic |
 | Anticancer | 22a-l | [50] | Synthetic | |
 | Anticancer | STAT3 | 23 | [55] | Synthetic |
 | Anticancer | Carbonic Anhydrase IX and XII | 24 | [70] | Natural |
 | Anticancer | Carbonic Anhydrase IX and XII | 26–37 | [71] | Synthetic |
 | Antibacterial/antifungal | S. aureus, E. coli, P. aeruginosa, A. niger, A. flavus, C. albicans | 38,39 | [79] | Synthetic |
 | Antibacterial | S. aureusus, E.faecalis, E. coli | 40,41a–e | [80] | Synthetic |
 | Antibacterial | S. aureus DNA gyrase | 42–49 | [82] | Synthetic |
 | Antibacterial | S. aureus | 50,51 | [83] | Synthetic |
 | Modulator of antibiotics | n.g. | isopimpinellin imperatorin | [85] | Natural |
 | Antibacterial | S. aureus, S. epidermidis, P. aeruginosa, E. cloacae, K. Pneumoniae, E. coli | 6′,7′-dihydroxybergamottin, peucedanin, officinali isobutyrate | [86] | Natural |
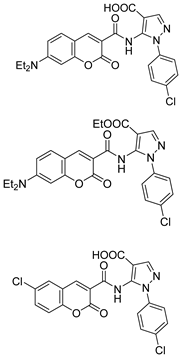 | Antibacterial | S. aureus, L. monocytogenes, E. coli, Salmonella—inhibitors of Topo II and Topo IV | 52–54 | [87] | Synthetic |
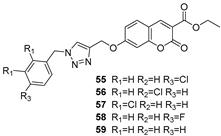 | Antifungal | C. albicans -P450 cytochrome lanosterol 14α-demethylase | 55–59 | [94] | Synthetic |
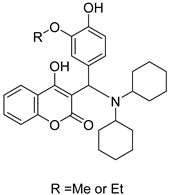 | Antifungal | C. albicans—inhibition ergosterol biosynthesis by binding lanosterol 14a-demethylase | 60,61 | [95] | Synthetic |
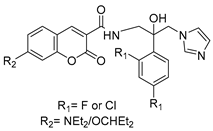 | Antifungal | Several Candida strains—mode of action involving CYP51 and additional unidentified mechanism | 62 | [96] | Synthetic |
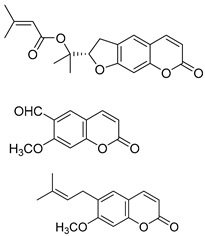 | Antifungal | C. albicans | marmesin senesioate, suberosin, crenulatin | [97] | Natural |
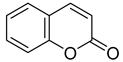 | Antifungal | C. albicans—antibiofilm action | coumarin | [100] | Natural |
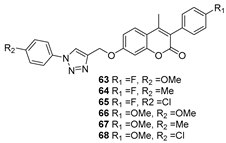 | Antifungal | F. oxysporum, A. flavus, A. niger | 63–68 | [102] | Synthetic |
 | Antiviral | Anti-HIV reverse transcriptase | 69–71 | [113] | Natural |
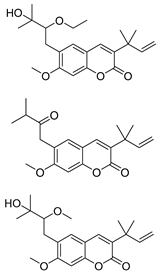 | Antiviral | HIV reverse transcriptase | 72–74 | [116] | Natural |
 | Antiviral | HIV1-IN | 75–78 | [117] | Synthetic |
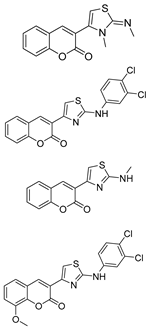 | Antiviral | H1N1 virus | 79–82 | [125] | Synthetic |
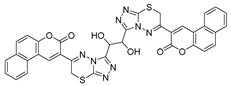 | Antiviral | Influenza A virus | 83 | [128] | Synthetic |
 | Antiviral | Influenza A/PR8/H1N1 viruses—probably could affect intracellular redox-sensitive pathways | 84–89 | [129] | Synthetic |
 | Antiviral | Hepatitis C virus | 90–92 | [130] | Synthetic |
 | Antiviral | Hepatitis B virus | esculetin | [131] | Natural |
 | Antiinflamma-tory | COX2 | 93–100 | [143] | Synthetic |
 | Antiinflamma-tory | 5-LOX | 101,102 | [146] | Synthetic |
 | Antiinflamma-tory | NO production | 103 | [147] | Natural |
 | Antiinflamma-tory | NF-kB and p38/MAPK signaling pathways | osthole | [150] | Natural |
 | Antiinflamma-tory | NF-kB signaling pathways | 104 | [151] | Synthetic |
 | Anti-Alzhemer’s disease | AChE, BuChE, BACE1 | 105 | [162] | Synthetic |
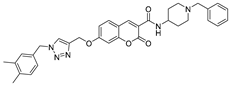 | Anti-Alzhemer’s disease | AChE, BuChE, BACE1, metal chelation | 107 | [163] | Synthetic |
 | Anti-Alzhemer’s disease | AChE, BuChE, BACE1 | 108 | [163] | Synthetic |
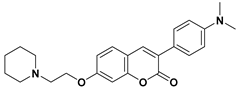 | Anti-Alzhemer’s disease | AChE, BuChE | 111a | [166] | Synthetic |
 | Anti-Alzhemer’s disease | AChE, BuChE, MAO-B | 117 | [167] | Synthetic |
 | Anti-Alzhemer’s disease | AChE, BuChE, MAO-B, β-amyloid aggregation | 136 | [169] | Synthetic |
 | Anticonvulsant | GABAergic system | 148 | [180] | Synthetic |
 | Anticonvulsant | BDZ receptor | 152d | [183] | Synthetic |
 | Anticoagulant | VKOR inhibitor | Tecarfarin(ATI-5923) | [195,196,197,198] | Synthetic |
 | Anticoagulant | n.g. | 154,155 | [199] | Synthetic |
 | Anticoagulant | murine VKORC1 inhibitor | 156 | [200] | Synthetic |
 | Anticoagulant | n.g. | 157 | [203] | Natural |
 | Antidiabetic | α-glucosidases | 158d,e | [207] | Synthetic |
 | Antidiabetic | α-glucosidases | 159d,e | [207] | Synthetic |
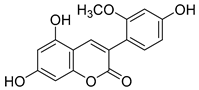 | Antidiabetic | α-glucosidases | 165 | [214] | Synthetic |
 | Antidiabetic | α-glucosidases | 168c | [215] | Synthetic |
 | Antidiabetic | Insulin release | 169–171 | [217] | Synthetic |
 | Photocleavable protecting groups | - | 172–174 | [231,232] | Synthetic |
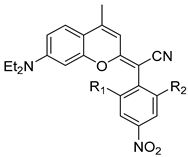 | Photocleavable protecting group | - | 175 | [224] | Synthetic |
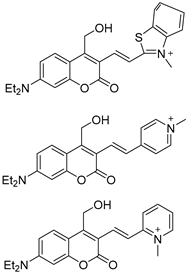 | Photocleavable protecting groups | - | 176–178 | [233] | Synthetic |
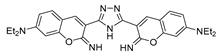 | Ionic probe | GHS (in the presence of Cu2+) and ClO− | 179 | [236,237] | Syntehtic |
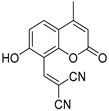 | Ionic probe | ClO− | 180 | [238] | Synthetic |
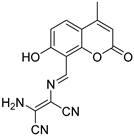 | Ionic probe | ClO−, Cu2+ | 181 | [239] | Synthetic |
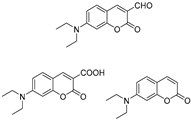 | Ionic probe | ClO− | 182–184 | [240] | Synthetic |
 | Ionic probe | Cu2+ | 185 | [241] | Synthetic |
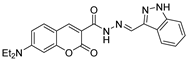 | Ionic probe | Cu2+ | 186 | [242] | Synthetic |
 | Ionic probe | Cu2+ | 187 | [243] | Synthetic |
 | Ionic probe | Cu2+, Al3+, Arg, Lys, Cys, Hcy, GSH | 188 | [244] | Synthetic |
 | Ionic probe | Pd2+ | 189 | [245] | Synthetic |
 | Ionic probe | Hg2+ | 190,191 | [246] | Synthetic |
 | Ionic probe | Fe3+ | 192 | [247] | Synthetic |
 | Ionic probe | F− | 193 | [248] | Synthetic |
 | Ionic probe | F−, Ca2+ | 194 | [249] | Synthetic |
 | Ionic probe | F−, CN− | 195 | [250] | Synthetic |
 | Probe | HSO3−, SO32− ion | 196 | [251] | Synthetic |
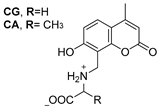 | Probe | HSO4− and SO42− | CG, CA | [256] | Synthetic |
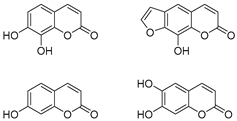 | Antibacterial | R. Solanacearum, genes fliA, flhC, biofilm formation | umbelliferone, esculetin, daphnetin, xanthotol | [261] | Natural |
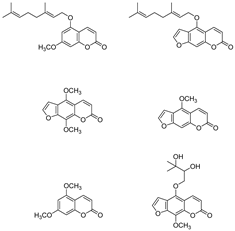 | Fungicide | Colletotrichum sp. | 197–202 | [265] | Natural |
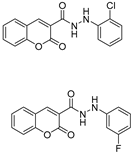 | Fungicide | Botrytis cinerea, Alternaria solani, Gibberella zeae, Rhizoctorzia solani, Cucumber anthrax and Alternaria | 203,204 | [268] | Synthetic |
 | Fungicide: | Alternaria solani sorauer, Fusarium oxysporum f.sp. vasinfectum and Fusarium moniliforme | 205a–d | [273] | Synthetic |
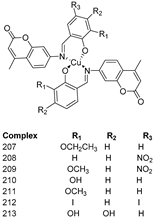 | Anticancer | MCF-7 human breast cancer cells | 207–213 | [276] | Synthetic |
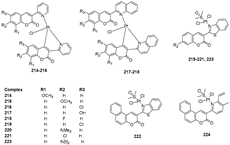 | Anticancer | Human lung adenocarcinoma (A549/DDP) and HeLa cells | 214–224 | [281] | Synthetic |
 | Catalyst | 225 | [282] | Synthetic |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Annunziata, F.; Pinna, C.; Dallavalle, S.; Tamborini, L.; Pinto, A. An Overview of Coumarin as a Versatile and Readily Accessible Scaffold with Broad-Ranging Biological Activities. Int. J. Mol. Sci. 2020, 21, 4618. https://doi.org/10.3390/ijms21134618
Annunziata F, Pinna C, Dallavalle S, Tamborini L, Pinto A. An Overview of Coumarin as a Versatile and Readily Accessible Scaffold with Broad-Ranging Biological Activities. International Journal of Molecular Sciences. 2020; 21(13):4618. https://doi.org/10.3390/ijms21134618
Chicago/Turabian StyleAnnunziata, Francesca, Cecilia Pinna, Sabrina Dallavalle, Lucia Tamborini, and Andrea Pinto. 2020. "An Overview of Coumarin as a Versatile and Readily Accessible Scaffold with Broad-Ranging Biological Activities" International Journal of Molecular Sciences 21, no. 13: 4618. https://doi.org/10.3390/ijms21134618
APA StyleAnnunziata, F., Pinna, C., Dallavalle, S., Tamborini, L., & Pinto, A. (2020). An Overview of Coumarin as a Versatile and Readily Accessible Scaffold with Broad-Ranging Biological Activities. International Journal of Molecular Sciences, 21(13), 4618. https://doi.org/10.3390/ijms21134618








