Involvement of ACE2/Ang-(1-7)/MAS1 Axis in the Regulation of Ovarian Function in Mammals
Abstract
1. Introduction
2. Angiotensin-(1-7) Synthesis and Metabolism
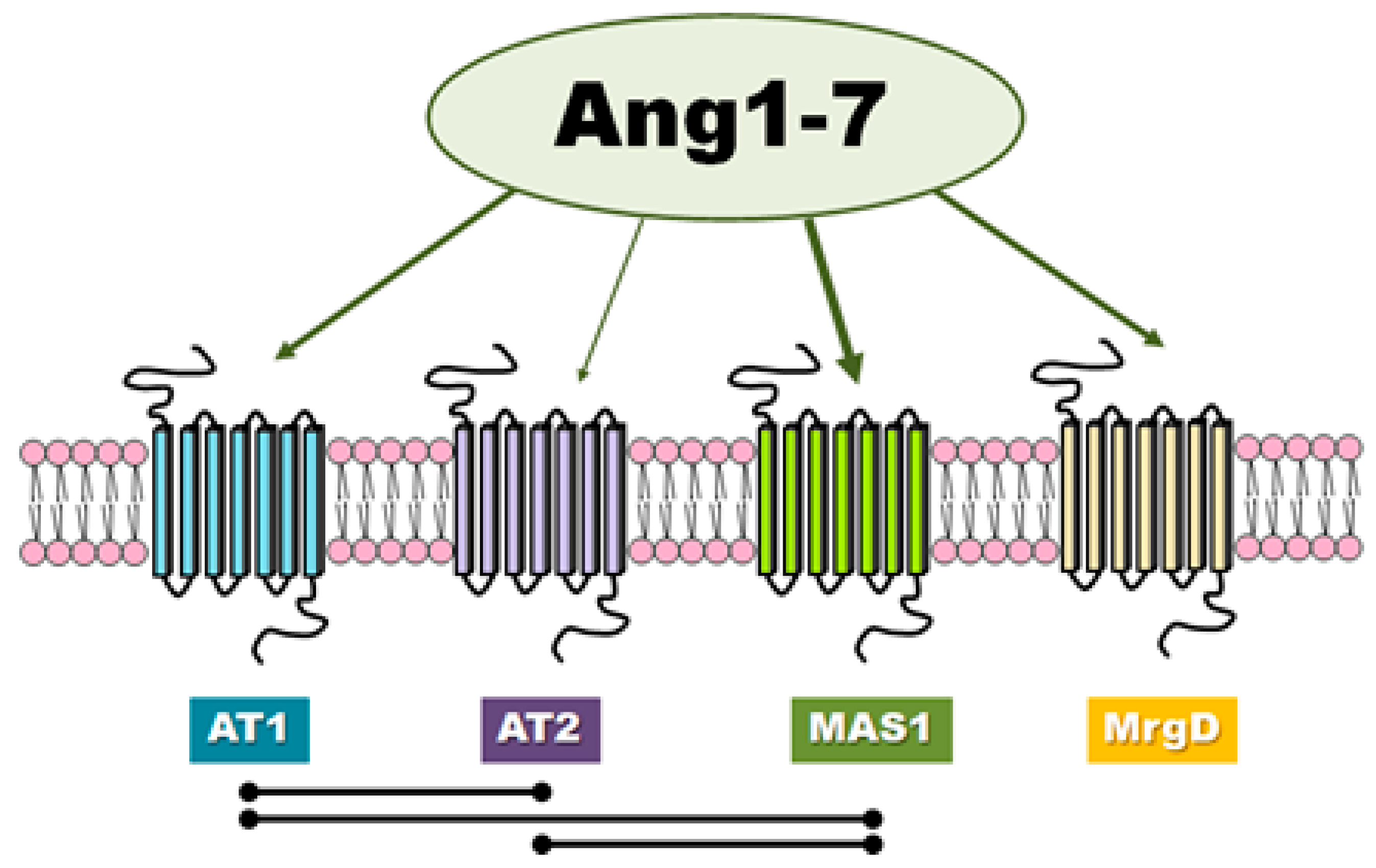
3. The Receptors Involved in Ang-(1-7) Activity
4. Localization and Level of Ang-(1-7) Axis Components in the Ovary
4.1. In the Rat Ovary
4.2. In the Rabbit Ovary
4.3. In the Ewe Ovary
4.4. In the Bovine Ovary
4.5. In the Human Ovary
5. The Relationship between Gonadotropins, Sex Hormones, and ACE 2/Ang-(1-7)/MAS1 Axis in the Ovary
5.1. In the Rat and Mouse Ovary
5.2. In the Rabbit Ovary
5.3. In the Ewe Ovary
5.4. In the Bovine Ovary
5.5. In the Human Ovary
6. Ovarian Diseases Related to the ACE2/Ang-(1-7)/MAS1 Axis
6.1. Polycystic Ovary Syndrome (PCOS)
6.2. Ovarian Hyperstimulation Syndrome (OHSS)
6.3. Ovarian Cancer (OC)
7. Conclusions
Funding
Conflicts of Interest
Abbreviations
| Ang-(1-7) | Angiotensin 1-7 |
| OC | Ovarian cancer |
| VEGF | Vascular endothelial growth factor |
| ACE | Angiotensin converting enzyme |
| LH | Luteinizing hormone |
| FSH | Follicle-stimulating hormone |
| RAS | Renin-angiotensin system |
| AP | Aminopeptidase |
| DC | Decarboxylase |
| DPP3 | Dipeptidyl peptidase 3 |
| CP | Carboxypeptidase |
| PRCP | Prolyl-carboxypeptidase |
| GPCRs | G protein-coupled receptors |
| hGC | Human urinary gonadotropin |
| eCG | Equine chorionic gonadotropins |
| EFOs | Follicle-enclosed oocytes |
| GVB | Germinal vesicle breakdown |
| IGF-1 | Insulin-like growth factor 1 |
| P4 | Progesterone |
| E2 | Estradiol |
| T | Testosterone |
| GnRH | Gonadotropin-releasing hormone |
| rFSH | Recombinant FSH |
| FF | Human ovarian follicular fluid |
| COS | Ovarian stimulation |
| IVF | In vitro fertilization |
| PCOS | Polycystic ovary syndrome |
| AR | Androgen receptor |
| DHEAS | Dehydroepiandrosterone sulfate |
| SHBG | Sex hormone-binding globulin |
| OHSS | Ovarian hyperstimulation syndrome |
| MCS | Promote multicellular spheroid |
References
- Akajewski, A. Ovary. In Anatomy of Domestic Animals; PWRIL: Poznan, Poland, 1994; pp. 150–153. [Google Scholar]
- Skinner, M. Encyclopedia of Reproduction; Elsevier Academic Press: Cambridge, MA, USA, 2018. [Google Scholar]
- Krzymowski, T.; Przały, J. Reproductive Processes in Sexually Mature Females. In Animal Physiology; PWRiL: Poznan, Poland, 2015; pp. 629–648. [Google Scholar]
- Costa, A.P.; Fagundes-Moura, C.R.; Pereira, V.M.; Silva, L.F.; Vieira, M.A.; Santos, R.A.; Dos Reis, A.M. Angiotensin-(1-7): A novel peptide in the ovary. Endocrinology 2003, 144, 1942–1948. [Google Scholar] [CrossRef] [PubMed]
- Iwai, M.; Horiuchi, M. Devil and angel in the renin-angiotensin system: ACE-angiotensin II-AT1 receptor axis vs. ACE2-angiotensin-(1-7)-Mas receptor axis. Hypertens. Res. 2009, 32, 533–536. [Google Scholar] [CrossRef] [PubMed]
- Schwentner, L.; Wöckel, A.; Herr, D.; Wulff, C. Is there a role of the local tissue RAS in the regulation of physiologic and pathophysiologic conditions in the reproductive tract? J. Renin Angiotensin Aldosterone Syst. 2011, 12, 385–393. [Google Scholar] [CrossRef] [PubMed]
- Herr, D.; Bekes, I.; Wulff, C. Local Renin-Angiotensin system in the reproductive system. Front. Endocrinol. (Lausanne) 2013, 4, 150. [Google Scholar] [CrossRef]
- Rice, G.I.; Thomas, D.A.; Grant, P.J.; Turner, A.J.; Hooper, N.M. Evaluation of angiotensin-converting enzyme (ACE), its homologue ACE2 and neprilysin in angiotensin peptide metabolism. Biochem. J. 2004, 383, 45–51. [Google Scholar] [CrossRef]
- Santos, R.A.S.; Sampaio, W.O.; Alzamora, A.C.; Motta-Santos, D.; Alenina, N.; Bader, M.; Campagnole-Santos, M.J. The ACE2/Angiotensin-(1-7)/MAS Axis of the Renin-Angiotensin System: Focus on Angiotensin-(1-7). Physiol. Rev. 2018, 98, 505–553. [Google Scholar] [CrossRef]
- Vickers, C.; Hales, P.; Kaushik, V.; Dick, L.; Gavin, J.; Tang, J.; Godbout, K.; Parsons, T.; Baronas, E.; Hsieh, F.; et al. Hydrolysis of biological peptides by human angiotensin-converting enzyme-related carboxypeptidase. J. Biol. Chem. 2002, 277, 14838–14843. [Google Scholar] [CrossRef]
- Brosnihan, K.B.; Neves, L.A.A.; Anton, L.; Joyner, J.; Valdes, G.; Merrill, D.C. Enhanced expression of Ang-(1-7) during pregnancy. Braz. J. Med. Biol. Res. 2004, 37, 1255–1262. [Google Scholar] [CrossRef]
- Burrell, L.M.; Johnston, C.I.; Tikellis, C.; Cooper, M.E. ACE2, a new regulator of the renin–angiotensin system. Trends Endocrinol. Metab. 2004, 15, 166–169. [Google Scholar] [CrossRef]
- Cruz-Diaz, N.; Wilson, B.A.; Pirro, N.T.K.; Brosnihan, B.; Marshall, A.C.; Chappell, M.C. Identification of dipeptidyl peptidase 3 as the Angiotensin-(1–7) degrading peptidase in human HK-2 renal epithelial cells. Peptides 2016, 83, 29–37. [Google Scholar] [CrossRef]
- Villela, D.C.; Passos-Silva, D.G.; Santos, R.A. Alamandine: A new member of the angiotensin family. Curr. Opin. Nephrol. Hypertens. 2014, 23, 130–134. [Google Scholar] [CrossRef] [PubMed]
- Qaradakhi, T.; Apostolopoulos, V.; Zulli, A. Angiotensin (1-7) and Alamandine: Similarities and differences. Pharmacol. Res. 2016, 111, 820–826. [Google Scholar] [CrossRef] [PubMed]
- Teixeira, L.B.; Parreiras-E.-Silva, L.T.; Bruder-Nascimento, T.; Duarte, D.A.; Simões, S.C.; Costa, R.M.; Rodríguez, D.Y.; Ferreira, P.A.B.; Silva, C.A.A.; Abrao, E.P.; et al. Ang-(1-7) is an endogenous β-arrestin-biased agonist of the AT1 receptor with protective action in cardiac hypertrophy. Sci. Rep. 2017, 7, 11903. [Google Scholar] [CrossRef] [PubMed]
- Gironacci, M.M.; Coba, M.P.; Peña, C. Angiotensin-(1-7) binds at the type 1 angiotensin II receptors in rat renal cortex. Regul. Pept. 1999, 84, 51–54. [Google Scholar] [CrossRef]
- Rowe, B.P.; Saylor, D.L.; Speth, R.C.; Absher, D.R. Angiotensin-(1-7) binding at angiotensin II receptors in the rat brain. Regul. Pept. 1995, 56, 139–146. [Google Scholar] [CrossRef]
- Clark, M.A.; Tallant, E.A.; Diz, D.I. Downregulation of the AT1A receptor by pharmacologic concentrations of Angiotensin-(1-7). J. Cardiovasc. Pharmacol. 2001, 37, 437–448. [Google Scholar] [CrossRef] [PubMed]
- Bosnyak, S.; Jones, E.S.; Christopoulos, A.; Aguilar, M.I.; Thomas, W.G.; Widdop, R.E. Relative affinity of angiotensin peptides and novel ligands at AT1 and AT2 receptors. Clin. Sci. (London) 2011, 121, 297–303. [Google Scholar] [CrossRef]
- Noor, N.; Patel, C.B.; Rockman, H.A. Β-arrestin: A signaling molecule and potential therapeutic target for heart failure. J. Mol. Cell. Cardiol. 2011, 51, 534–541. [Google Scholar] [CrossRef]
- van Gastel, J.; Hendrickx, J.O.; Leysen, H.; Santos-Otte, P.; Luttrell, L.M.; Martin, B. Maudsley, S. β-Arrestin Based Receptor Signaling Paradigms: Potential Therapeutic Targets for Complex Age-Related Disorders. Front. Pharmacol. 2018, 9, 1369. [Google Scholar] [CrossRef]
- Jean-Charles, P.; Kaur, S.; Shenoy, S.K. GPCR signaling via β-arrestin-dependent mechanisms. J. Cardiovasc. Pharmacol. 2017, 70, 142–158. [Google Scholar] [CrossRef]
- Walters, P.E.; Gaspari, T.A.; Widdop, R.E. Angiotensin-(1-7) acts as a vasodepressor agent via angiotensin II type 2 receptors in conscious rats. Hypertension 2005, 45, 960–966. [Google Scholar] [CrossRef] [PubMed]
- Jiang, F.; Yang, J.; Zhang, Y.; Dong, M.; Wang, S.; Zhang, Q.; Liu, F.F.; Zhang, K.; Zhang, C. Nat Angiotensin-converting enzyme 2 and angiotensin 1–7: Novel therapeutic targets. Rev. Cardiol. 2014, 11, 413–426. [Google Scholar] [CrossRef]
- Tetzner, A.; Gebolys, K.; Meinert, C.; Klein, S.; Uhlich, A.; Trebicka, J.; Villacañas, Ó.; Walther, T. G-Protein-Coupled Receptor MrgD Is a Receptor for Angiotensin-(1-7) Involving Adenylyl Cyclase, cAMP, and Phosphokinase, A. Hypertension 2016, 68, 185–194. [Google Scholar] [CrossRef] [PubMed]
- Gironacci, M.M.; Adamo, H.P.; Corradi, G.; Santos, R.A.; Ortiz, P.; Carretero, O.A. Angiotensin (1-7) induces MAS receptor internalization. Hypertension 2011, 58, 176–181. [Google Scholar] [CrossRef] [PubMed]
- Santos, R.A.; Simoes e Silva, A.C.; Maric, C.; Silva, D.M.; Machado, R.P.; de Buhr, I.; Heringer-Walther, S.; Pinheiro, S.V.; Lopes, M.T.; Bader, M.; et al. Angiotensin-(1-7) is an endogenous ligand for the G protein-coupled receptor Mas. Proc. Natl. Acad. Sci. USA 2003, 100, 8258–8263. [Google Scholar] [CrossRef]
- Tallant, E.A.; Ferrario, C.M.; Gallagher, P.E. Angiotensin-(1-7) inhibits growth of cardiac myocytes through activation of the mas receptor. Am. J. Physiol. Heart Circ. Physiol. 2005, 289, H1560–H1566. [Google Scholar] [CrossRef]
- Leal, M.C.; Pinheiro, S.V.; Ferreira, A.J.; Santos, R.A.; Bordoni, L.; Alenina, N.; Bader, M.; França, L.R. The role of angiotensin-(1-7) receptor Mas in spermatogenesis in mice and rats. J. Anat. 2009, 214, 736–743. [Google Scholar] [CrossRef]
- Gaidarov, I.; Adams, J.; Frazer, J.; Anthony, T.; Chen, X.; Gatlin, J.; Semple, G.; Unett, D.J. Angiotensin (1-7) does not interact directly with MAS1, but can potently antagonize signaling from the AT1 receptor. Cell. Signal. 2018, 50, 9–24. [Google Scholar] [CrossRef]
- Dominici, F.P.; Burghi, V.; Muñoz, M.C.; Giani, J.F. Modulation of the action of insulin by angiotensin-(1-7). Clin. Sci. (London) 2014, 126, 613–630. [Google Scholar] [CrossRef]
- Honorato-Sampaio, K.; Pereira, V.M.; Santos, R.A.; Reis, A.M. Evidence that angiotensin-(1-7) is an intermediate of gonadotrophin-induced oocyte maturation in the rat preovulatory follicle. Exp. Physiol. 2012, 97, 642–650. [Google Scholar] [CrossRef]
- Thomas, W.G.; Sernia, C. The immunocytochemical localization of angiotensinogen in the rat ovary. Cell. Tissue Res. 1990, 261, 367–373. [Google Scholar] [CrossRef] [PubMed]
- Pereira, V.M.; Reis, F.M.; Santos, R.A.; Cassali, G.D.; Santos, S.H.; Honorato-Sampaio, K.; dos Reis, A.M. Gonadotropin stimulation increases the expression of angiotensin-(1-7) and MAS receptor in the rat ovary. Reprod. Sci. 2009, 16, 1165–1174. [Google Scholar] [CrossRef] [PubMed]
- Paul, M.; Poyan Mehr, A.; Kreutz, R. Physiology of local renin-angiotensin systems. Physiol. Rev. 2006, 86, 747–803. [Google Scholar] [CrossRef]
- Viana, G.E.; Pereira, V.M.; Honorato-Sampaio, K.; Oliveira, C.A.; Santos, R.A.; Reis, A.M. Angiotensin-(1-7) induces ovulation and steroidogenesis in perfused rabbit ovaries. Exp. Physiol. 2011, 96, 957–965. [Google Scholar] [CrossRef] [PubMed]
- Zappulla, J.P.; DesGroseillers, L. Neutral endopeptidase is expressed on the follicular granulosa cells of rabbit ovaries. Comp. Biochem. Physiol. B Biochem. Mol. Biol. 2001, 129, 863–870. [Google Scholar] [CrossRef]
- Yoshimura, Y.; Karube, M.; Aoki, H.; Oda, T.; Koyama, N.; Nagai, A.; Akimoto, Y.; Hirano, H.; Nakamura, Y. Angiotensin II induces ovulation and oocyte maturation in rabbit ovaries via the AT2receptor subtype. Endocrinology 1996, 137, 1204–1211. [Google Scholar] [CrossRef] [PubMed]
- Féral, C.; Benhaïm, A.; Leymarie, P. Angiotensin II receptor type 1 on granulosa and thecal cells of rabbit preovulatory follicles. Biochim. Biophys. Acta 1996, 1284, 221–226. [Google Scholar] [CrossRef][Green Version]
- Pereira, A.M.; de Souza, A., Jr.; Machado, F.B.; Gonçalves, G.K.; Feitosa, L.C.; Reis, A.M.; Santos, R.A.; Honorato-Sampaio, K.; Costa, A.R. The effect of angiotensin-converting enzyme inhibition throughout a superovulation protocol in ewes. Res. Vet. Sci. 2015, 103, 205–210. [Google Scholar] [CrossRef]
- Barreta, M.H.; Gasperin, B.G.; Ferreira, R.; Rovani, M.; Pereira, G.R.; Bohrer, R.C.; de Oliveira, J.F.; Gonçalves, P.B. The components of the angiotensin-(1-7) system are differentially expressed during follicular wave in cattle. J. Renin Angiotensin Aldosterone Syst. 2015, 16, 275–283. [Google Scholar] [CrossRef]
- Schultze, D.; Brunswig, B.; Mukhopadhyay, A.K. Renin and prorenin-like activities in bovine ovarian follicles. Endocrinology 1989, 124, 1389–1398. [Google Scholar] [CrossRef]
- Tonellotto dos Santos, J.; Ferreira, R.; Gasperin, B.G.; Siqueira, L.C.; de Oliveira, J.F.; Santos, R.A.; Reis, A.M.; Gonçalves, P.B. Molecular characterization and regulation of the angiotensin-converting enzyme type 2/angiotensin-(1-7)/MAS receptor axis during the ovulation process in cattle. J. Renin Angiotensin Aldosterone Syst. 2012, 13, 91–98. [Google Scholar] [CrossRef] [PubMed]
- Schauser, K.H.; Nielsen, A.H.; Winther, H.; Dantzer, V.; Poulsen, K. Localization of the Renin-Angiotensin System in the Bovine Ovary: Cyclic Variation of the Angiotensin II Receptor Expression. Biol. Reprod. 2001, 65, 1672–1680. [Google Scholar] [CrossRef] [PubMed]
- Palumbo, A.; Pourmotabbed, G.; Carcangiu, M.L.; Andrade-Gordon, P.; Roa, L.; DeCherney, A.; Naftolin, F. Immunohistochemical localization of renin and angiotensin in the ovary: Comparison between normal women and patients with histologically proven polycystic ovarian disease. Fertil. Steril. 1993, 60, 280–284. [Google Scholar] [CrossRef]
- Reis, F.M.; Bouissou, D.R.; Pereira, V.M.; Camargos, A.F.; dos Reis, A.M.; Santos, R.A. Angiotensin-(1-7), its receptor Mas, and the angiotensin-converting enzyme type 2 are expressed in the human ovary. Fertil. Steril. 2011, 95, 176–181. [Google Scholar] [CrossRef] [PubMed]
- Cavallo, I.K.; Dela Cruz, C.; Oliveira, M.L.; Del Puerto, H.L.; Dias, J.A.; Lobach, V.N.; Casalechi, M.; Camargos, M.G.; Reis, A.M.; Santos, R.A.; et al. Angiotensin-(1-7) in human follicular fluid correlates with oocyte maturation. Hum. Reprod. 2017, 32, 1318–1324. [Google Scholar] [CrossRef]
- Erickson, G.F.; Ryan, K.J. Stimulation of testosterone production in isolated rabbit thecal tissue by LH/FSH, dibutyryl cyclic AMP, PGE2 alpha, and PGE2. Endocrinology 1976, 99, 452–458. [Google Scholar] [CrossRef]
- Fortune, J.E.; Armstrong, D.T. Androgen production by theca and granulosa isolated from proestrous rat follicles. Endocrinology 1977, 100, 1341–1347. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.X.; Hsueh, A.J. Synergism between granulosa and theca-interstitial cells in estrogen biosynthesis by gonadotropin-treated rat ovaries: Studies on the two-cell, two-gonadotropin hypothesis using steroid antisera. Biol. Reprod. 1986, 35, 27–36. [Google Scholar] [CrossRef]
- Torres-Ramírez, B.N.; Ortiz-Hernández, R.; Escobar-Sánchez, M.L.; Echeverría-Martínez, O.M.; Vázquez-Nin, H.G. Endoplasmic Reticulum Stress during Mammalian Follicular. In Endoplasmic Reticulum; IntechOpen: London, UK, 2019. [Google Scholar] [CrossRef]
- Ginther, O.J.; Bergfelt, D.R.; Kulick, L.J.; Kot, K. Selection of the dominant follicle in cattle: Establishment of follicle deviation in less than 8 hours through depression of FSH concentrations. Theriogenology 1999, 52, 1079–1093. [Google Scholar] [CrossRef]
- Mihm, M.; Crowe, M.A.; Knight, P.G.; Austin, E.J. Follicle wave growth in cattle. Reprod. Domest. Anim. 2002, 37, 191–200. [Google Scholar] [CrossRef]
- Erman, A.; Chen-Gal, B.; van Dijk, D.J.; Sulkes, J.; Kaplan, B.; Boner, G.; Neri, A. Ovarian angiotensin-converting enzyme activity in humans: Relationship to estradiol, age, and uterine pathology. J. Clin. Endocrinol. Metab. 1996, 81, 1104–1107. [Google Scholar] [CrossRef][Green Version]
- Billig, H.; Furuta, I.; Hsueh, A.J. Estrogens inhibit and androgens enhance ovarian granulosa cell apoptosis. Endocrinology 1993, 133, 2204–2212. [Google Scholar] [CrossRef] [PubMed]
- Peluso, J.J.; Pappalardo, A. Progesterone and cell-cell adhesion interact to regulate rat granulosa cell apoptosis. Biochem. Cell. Biol. 1994, 72, 547–551. [Google Scholar] [CrossRef] [PubMed]
- Honorato-Sampaio, K.; Andrade, R.F.; Bader, M.; de Souza Martins, A.; Santos, R.A.S.; Adelina Martha Reis, A.M. Genetic Deletion of the Angiotensin-(1-7) Receptor Mas Leads to a Reduced Ovulatory Rate. Peptides 2018, 107, 83–88. [Google Scholar] [CrossRef] [PubMed]
- Maggio, M.; Ceda, G.P.; Lauretani, F.; Pahor, M.; Bandinelli, S.; Najjar, S.S.; Ling, S.M.; Basaria, S.; Ruggiero, C.; Valenti, G.; et al. Relation of angiotensin-converting enzyme inhibitor treatment to insulin-like growth factor-1 serum levels in subjects >65 years of age (the InCHIANTI study). Am. J. Cardiol. 2006, 97, 1525–1529. [Google Scholar] [CrossRef][Green Version]
- Szczepanska-Sadowska, E.; Czarzasta, K.; Cudnoch-Jedrzejewska, A. Dysregulation of the Renin-Angiotensin System and the Vasopressinergic System Interactions in Cardiovascular Disorders. Curr. Hypertens. Rep. 2018, 20, 19. [Google Scholar] [CrossRef]
- Giovannini, S.; Cesari, M.; Marzetti, E.; Leeuwenburgh, C.; Maggio, M.; Pahor, M. Effects of ACE-inhibition on IGF-1 and IGFBP-3 concentrations in older adults with high cardiovascular risk profile. J. Nutr. Health Aging 2010, 14, 457–460. [Google Scholar] [CrossRef]
- Sahin, Y.; Kontaş, O.; Müderris, I.I.; Cankurtaran, M. Effects of angiotensin converting enzyme inhibitor cilazapril and angiotensin II antagonist saralasin in ovarian hyperstimulation syndrome in the rabbit. Gynecol. Endocrinol. 1997, 11, 231–236. [Google Scholar] [CrossRef]
- Peña, O.; Palumbo, A.; González-Fernández, R.; Hernández, J.; Naftolin, F.; Avila, J. Expression of angiotensin II type 1 (AT1) and angiotensin II type 2 (AT2) receptors in human granulosa-lutein (GL) cells: Correlation with infertility diagnoses. Fertil. Steril. 2010, 93, 1601–1608. [Google Scholar] [CrossRef]
- Palumbo, A.; Ávila, J.; Naftolin, F. The Ovarian Renin-Angiotensin System (OVRAS): A Major Factor in Ovarian Function and Disease. Reprod. Sci. 2016, 23, 1644–1655. [Google Scholar] [CrossRef]
- Ndefo, U.A.; Eaton, A.; Green, M.R. Polycystic ovary syndrome: A review of treatment options with a focus on pharmacological approaches. Pharm. Ther. 2013, 38, 336–355. [Google Scholar]
- Qin, S.; Zhou, Y.J.; Liu, Y.; Shen, H.M.; Li, X.D.; Yan, X.; Tang, H.J. Expression and significance of ACE2-Ang-(1-7)-Mas axis in the endometrium of patients with polycystic ovary syndrome. Zhonghua Yi Xue Za Zhi 2013, 93, 1989–1992. [Google Scholar] [PubMed]
- Jaatinen, T.A.; Matinlauri, I.; Anttila, L.; Koskinen, P.; Erkkola, R.; Irjala, K. Serum total renin is elevated in women with polycystic ovarian syndrome. Fertil. Steril. 1995, 63, 1000–1004. [Google Scholar] [CrossRef]
- Arefi, S.; Mottaghi, S.; Sharifi, A.M. Studying the correlation of renin-angiotensin-system (RAS) components and insulin resistance in polycystic ovary syndrome (PCOs). Gynecol. Endocrinol. 2013, 29, 470–473. [Google Scholar] [CrossRef]
- Alphan, Z.; Berberoglu, Z.; Gorar, S.; Candan, Z.; Aktas, A.; Aral, Y.; Ademoglu, E. Increased total Renin levels but not Angiotensin-converting enzyme activity in obese patients with polycystic ovary syndrome. Med. Princ. Pract. 2013, 22, 475–479. [Google Scholar] [CrossRef]
- Jia, H.; Wang, B.; Yu, L.; Jiang, Z. Association of angiotensin-converting enzyme gene insertion/deletion polymorphism with polycystic ovary syndrome: A meta-analysis. J. Renin Angiotensin Aldosterone Syst. 2013, 14, 255–262. [Google Scholar] [CrossRef]
- Falkner, B.; Canessa, M.; Anzalone, D. Effect of angiotensin converting enzyme inhibitor (lisinopril) on insulin sensitivity and sodium transport in mild hypertension. Am. J. Hypertens. 1995, 8, 454–460. [Google Scholar] [CrossRef]
- Underwood, P.C.; Adler, G.K. The Renin Angiotensin Aldosterone System and Insulin Resistance in Humans. Curr. Hypertens. Rep. 2013, 15, 59–70. [Google Scholar] [CrossRef]
- Paris, R.V.; Bertoldo, M.J. The Mechanism of Androgen Actions in PCOS. Etiol. Med. Sci. (Basel) 2019, 7, 89. [Google Scholar] [CrossRef]
- Hacihanefioglu, B.; Somunkiran, A.; Mahmutoglu, I.; Sercelik, A.; Toptani, S.; Kervancioglu, E. Effect of hypertension therapy with the angiotensin-converting enzyme inhibitor lisinopril on hyperandrogenism in women with polycystic ovary syndrome. Fertil. Steril. 2002, 77, 526–538. [Google Scholar] [CrossRef]
- Yoshimura, Y. The ovarian renin-angiotensin system in reproductive physiology. Front. Neuroendocrinol. 1997, 18, 247–291. [Google Scholar] [CrossRef] [PubMed]
- Deepika, M.L.N.; Ranjith Reddy, K.; Usha Rani, V.; Balakrishna, N.; Prasanna Latha, K.; Parveen, J. Do ACE I/D gene polymorphism serve as a predictive marker for age at onset in PCOS? J. Assist. Reprod. Genet. 2013, 30, 125–130. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.; Fan, H.; Che, Y.; Cao, Y.; Wu, X.; Sun, H.; Liang, F.; Yi, L.; Wang, Y. Association between ACE gene I/D polymorphisms and hyperandrogenism in women with Polycystic Ovary Syndrome (PCOS) and controls. BMC Med. Genet. 2009, 10, 64. [Google Scholar] [CrossRef] [PubMed]
- Kumar, P.; Sait, S.F.; Sharma, A.; Kumar, M. Ovarian hyperstimulation syndrome. J. Hum. Reprod. Sci. 2011, 4, 70–75. [Google Scholar] [CrossRef] [PubMed]
- Navot, D.; Margalioth, E.J.; Laufer, N.; Birkenfeld, A.; Relou, A.; Rosler, A.; Schenker, J.G. Direct correlation between plasma renin activity and severity of the ovarian hyperstimulation syndrome. Fertil. Steril. 1987, 48, 57–61. [Google Scholar] [CrossRef]
- Delbaere, A.; Bergmann, P.J.; Englert, Y. Features of the Renin-angiotensin system in ascites and pleural effusion during severe ovarian hyperstimulation syndrome. J. Assist. Reprod. Genet. 1997, 14, 241–244. [Google Scholar] [CrossRef]
- Ong, A.C.M.; Eisen, V.; Rennie, D.P.; Homburg, R.; Lachelin, G.C.L.; Jacobs, H.S.; Slater, J.D. The pathogenesis of the ovarian hyperstimulation syndrome (OHS): A possible role for ovarian renin. Clin. Endocrinol. (Oxford) 1991, 34, 43–49. [Google Scholar] [CrossRef]
- Morris, R.S.; Wong, I.L.; Kirkman, E.; Gentschein, E.; Paulson, R.J. Inhibition of ovarian-derived pro-renin to angiotensin cascade in the treatment of ovarian hyperstimulation syndrome. Hum. Reprod. 1995, 10, 1355–1358. [Google Scholar] [CrossRef]
- Gul, T.G.; Posaci, C.; Caliskan, S. The role of enalapril in the prevention of ovarian hyperstimulation syndrome: A rabbit model. Hum. Reprod. 2001, 16, 2253–2257. [Google Scholar] [CrossRef][Green Version]
- Ando, H.; Furugori, K.; Shibata, D.; Harata, T.; Murata, Y.; Mizutani, S. Dual renin-angiotensin blockade therapy in patients at high risk of early ovarian hyperstimulation syndrome receiving IVF and elective embryo cryopreservation: A case series. Hum. Reprod. 2003, 18, 1219–1222. [Google Scholar] [CrossRef][Green Version]
- Baris Ata, M.D.; Kayhan Yakin, M.D.; Cengiz Alatas, M.D.; Bulent Urman, M.D. Dual renin-angiotensin blockage and total embryo cryopreservation is not a risk-free strategy in patients at high risk for ovarian hyperstimulation syndrome. Fertil. Steril. 2008, 90, 531–536. [Google Scholar] [CrossRef]
- Manno, M.; Tomei, F. Renin-angiotensin system activation during severe OHSS: Cause or effect? Fertil. Steril. 2008, 89, 488. [Google Scholar] [CrossRef] [PubMed]
- Nowak-Markwitz, E.; Spaczyński, M. Ovarian cancer—Modern approach to its origin and histogenesis. Ginekol. Pol. 2012, 83, 454–457. [Google Scholar] [PubMed]
- Kujawa, K.A.; Lisowska, K.M. Ovarian cancer—From biology to clinic. Postepy Hig. Med. Dosw. (online) 2015, 69, 1275–1290. [Google Scholar] [CrossRef]
- Wolgien, M.D.C.G.M.; Alves Correa, S.A.; Breuel, P.A.F.; Nazário, A.P.P.; Facina, G. Renin Angiotensin System Components and Cancer. Rep. Associ. J. Biosci. Med. 2016, 4, 65–75. [Google Scholar] [CrossRef][Green Version]
- Domińska, K.; Lachowicz-Ochedalska, A. The involvement of the renin-angiotensin system (RAS) in cancerogenesis. Postepy Biochem. 2008, 54, 294–300. [Google Scholar]
- Ishikane, S.; Takahashi-Yanaga, F. The role of angiotensin II in cancer metastasis: Potential of renin-angiotensin system blockade as a treatment for cancer metastasis. Biochem. Pharmacol. 2018, 151, 96–103. [Google Scholar] [CrossRef]
- Gallagher, P.E.; Arter, A.L.; Deng, G.; Tallant, E.A. Angiotensin-(1-7): A peptide hormone with anti-cancer activity. Curr. Med. Chem. 2014, 21, 242417–242423. [Google Scholar] [CrossRef]
- Xu, J.; Fann, J.; Wu, F.; Huang, Q.; Guo, M.; Lv, Z.; Han, J.; Duan, L.; Hu, G.; Chen, L.; et al. The ACE2/Angiotensin-(1–7)/Mas Receptor Axis: Pleiotropic Roles in Cancer. Front. Physiol. 2017, 8, 276. [Google Scholar] [CrossRef]
- Beyazit, F.; Ayhan, S.; Celik, H.T.; Gungor, T. Assessment of serum angiotensin-converting enzyme in patients with epithelial ovarian cancer. Arch. Gynecol. Obstet. 2015, 292, 415–420. [Google Scholar] [CrossRef]
- Cotter, T.P.; Kealy, N.P.; Duggan, P.F.; Curtain, A.; Bredin, C.P. Elevated serum angiotensin converting enzyme levels in metastatic ovarian dysgerminoma. Respir. Med. 1997, 91, 237–239. [Google Scholar] [CrossRef][Green Version]
- Correa-Noronha, S.A.; Noronha, S.M.; Alecrim, C.; de Carvalho Mesquita, A.; Brito, G.S.; Junqueira, M.G.; Leite, D.B.; Carvalho, C.V.; Silva, I.D. Association of angiotensin-converting enzyme I gene I/D polymorphism with endometrial but not with ovarian cancer. Gynecol. Endocrinol. 2012, 28, 889–891. [Google Scholar] [CrossRef] [PubMed]
- Ino, K.; Shibata, K.; Kajiyama, H.; Yamamoto, E.; Nagasaka, T.; Nawa, A.; Nomura, S.; Kikkawa, F. Angiotensin II type 1 receptor expression in ovarian cancer and its correlation with tumour angiogenesis and patient survival. Br. J. Cancer 2006, 94, 552–560. [Google Scholar] [CrossRef] [PubMed]
- Suganuma, T.; Ino, K.; Shibata, K.; Kajiyama, H.; Nagasaka, T.; Mizutani, S.; Kikkawa, F. Functional expression of the angiotensin II type 1 receptor in human ovarian carcinoma cells and its blockade therapy resulting in suppression of tumor invasion, angiogenesis, and peritoneal dissemination. Clin. Cancer Res. 2005, 11, 2686–2694. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Yu, S.; Ting Lam, M.M.; Poon, T.C.W.; Sun, L.; Jiao, Y.; Wong, A.S.T.; Lee, L.T.O. Angiotensin II promotes ovarian cancer spheroid formation and metastasis by upregulation of lipid desaturation and suppression of endoplasmic reticulum stress. J. Exp. Clin. Cancer Res. 2019, 38, 116. [Google Scholar] [CrossRef]
- Song, L.; Zhang, S.L.; Bai, K.H.; Yang, J.; Xiong, H.Y.; Li, X.; Liu, T.; Liu, H.R. Serum agonistic autoantibodies against type-1 angiotensin II receptor titer in patients with epithelial ovarian cancer: A potential role in tumor cell migration and angiogenesis. J. Ovarian Res. 2013, 6, 22–28. [Google Scholar] [CrossRef]


| Elements OF ACE2/ANG1-7 Axis | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| PRORENIN, RENIN | ANGIOTENSINOGEN | Ang-(1-7) | ACE2 | NEP | PEP | MAS1 | AT1 | AT2 | |
| Rat Ovary |
|
| no data | no data | no data |
|
|
| |
| Rabbit Ovary | no data | no data | no data | no data |
|
|
| ||
| Ewe Ovary | no data | no data |
| no data | no data | no data | no data | no data | |
| Bovine Ovary |
| no data |
|
|
|
|
|
| |
| Human Ovary |
| no data |
| no data | no data |
| no data | no data | |
| Species | Gonadotropins | FSH | LH | Ang-(1-7) | ACE1 Inhibitor |
|---|---|---|---|---|---|
| Rat | stimulates Ang-(1-7) and MAS1 expression in the ovarian theca- interstitial cells [35] increases ACE2 mRNA but not their activity in ovaries [33,35] increases PEP activity but inhibites NEP activity in ovaries [35] | stimulates renin mRNA expression in the ovary [36] | inreases Ang-(1-7) and ACE2 immunoreactivity in the preovulatory follicles [33] | non impact on sex hormone production [33] | reduces levels of P4 and increased T after LH-stimulation [33] |
| Rabbit | no data | no data | no data | stimulates release E2 but not P4 in ovaries [37] | not influence on P4 production but E2 level decreased after gonadotropins-stymulation [37] |
| Ewe | no data | no data | no data | no data | decreases E2 plasma concentration but not influence on T and P4 production [41] showes increased P4:E2 ratio [41] |
| Bovine | increases Ang-(1-7) concentration in the follicular fluid of preovulatory follicles [44] downregulates ACE2 and PEP expression in granulosa cells but not near ovulation [44] | no data | no data | no data | no data |
| Human | elevates level of plasma Ang-(1-7) [47,48] | increases plasma concentrations of Ang-(1–7) [48] | no data | no data | no data |
© 2020 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Domińska, K. Involvement of ACE2/Ang-(1-7)/MAS1 Axis in the Regulation of Ovarian Function in Mammals. Int. J. Mol. Sci. 2020, 21, 4572. https://doi.org/10.3390/ijms21134572
Domińska K. Involvement of ACE2/Ang-(1-7)/MAS1 Axis in the Regulation of Ovarian Function in Mammals. International Journal of Molecular Sciences. 2020; 21(13):4572. https://doi.org/10.3390/ijms21134572
Chicago/Turabian StyleDomińska, Kamila. 2020. "Involvement of ACE2/Ang-(1-7)/MAS1 Axis in the Regulation of Ovarian Function in Mammals" International Journal of Molecular Sciences 21, no. 13: 4572. https://doi.org/10.3390/ijms21134572
APA StyleDomińska, K. (2020). Involvement of ACE2/Ang-(1-7)/MAS1 Axis in the Regulation of Ovarian Function in Mammals. International Journal of Molecular Sciences, 21(13), 4572. https://doi.org/10.3390/ijms21134572






