Discovery of the Involvement in DNA Oxidative Damage of Human Sperm Nuclear Basic Proteins of Healthy Young Men Living in Polluted Areas
Abstract
1. Introduction
2. Results
2.1. Characteristics of Impacted Areas Used for the Recruitment
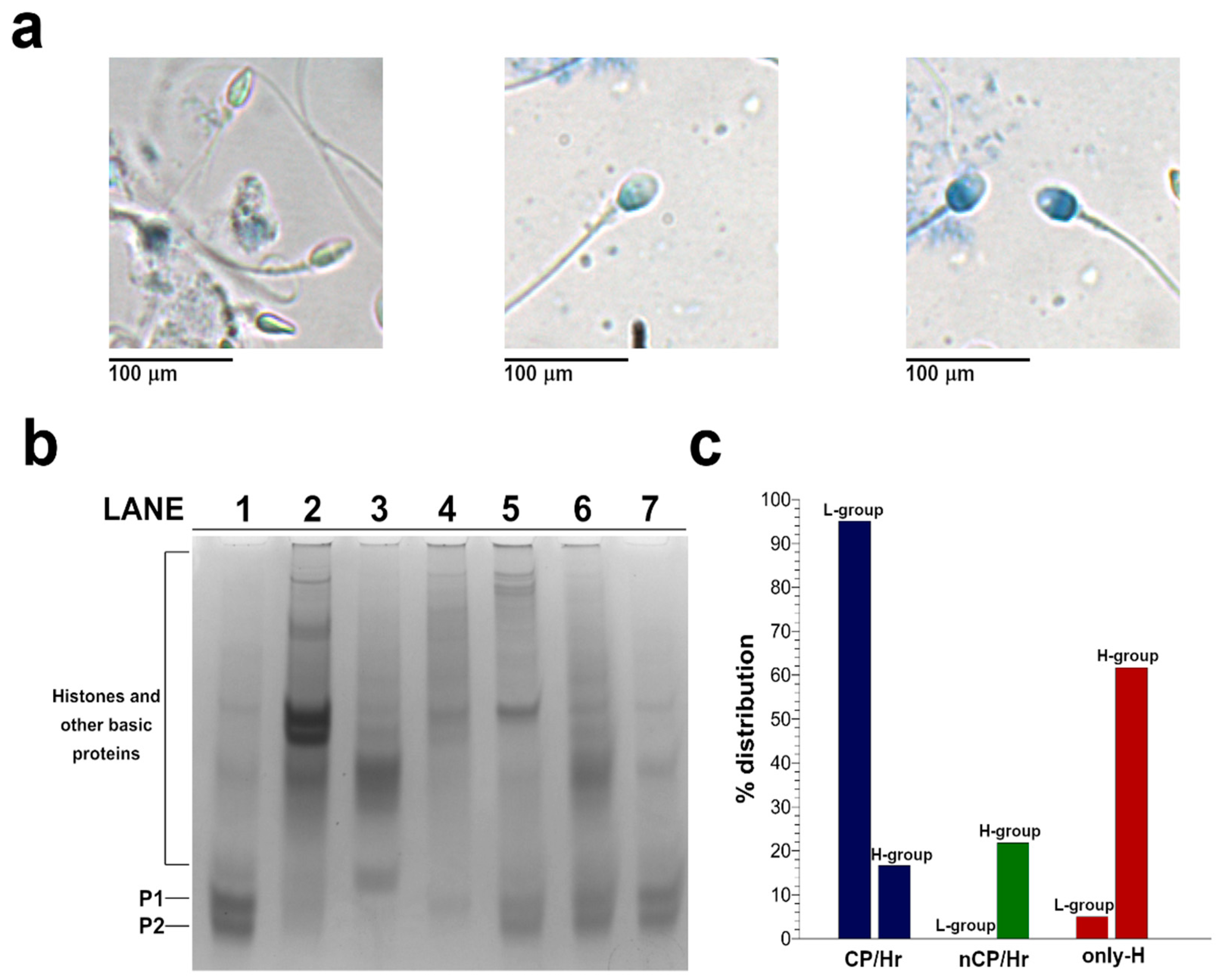
2.2. Analysis of SNBP
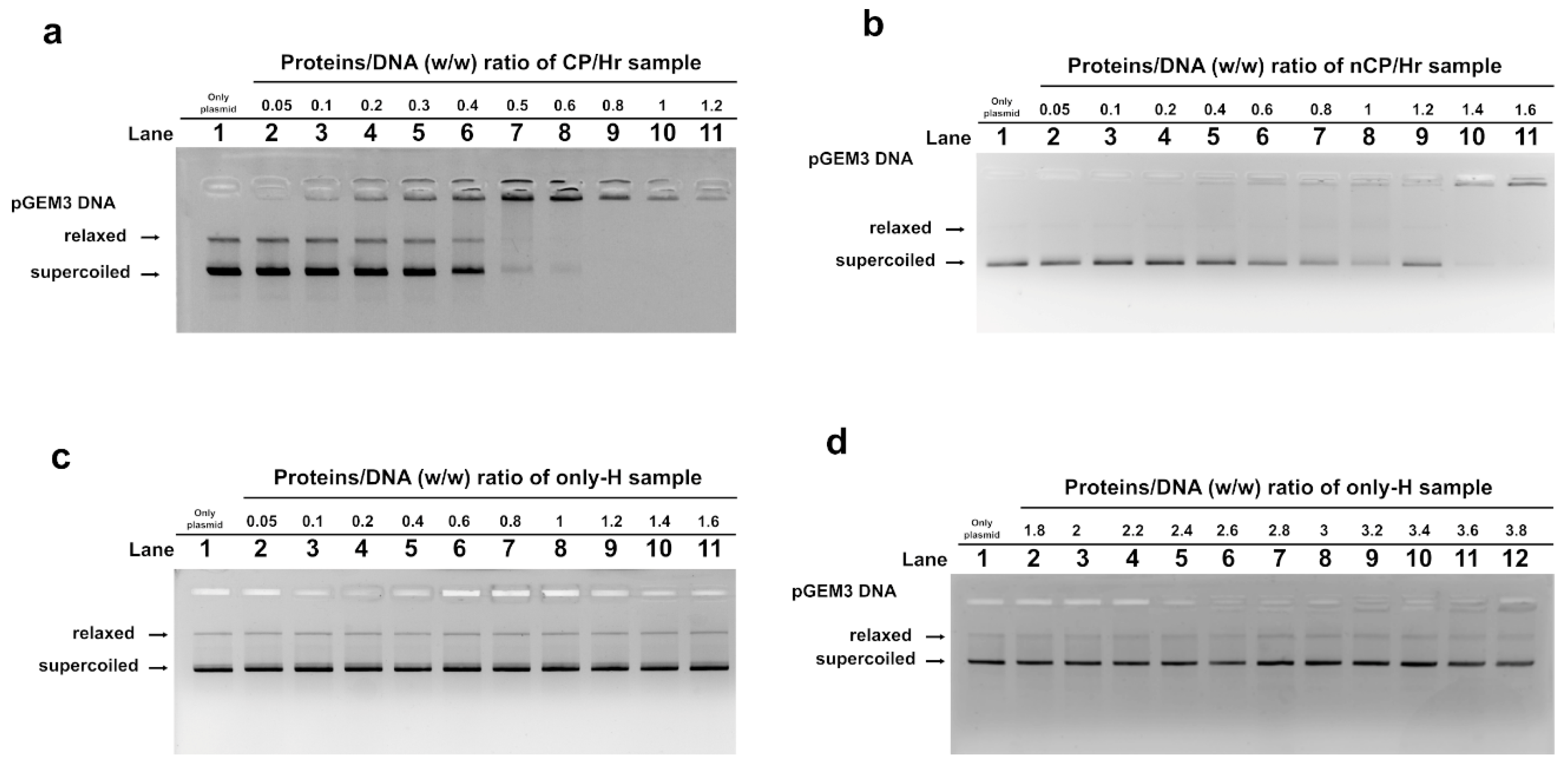
2.3. DNA Binding Ability of SNBP Analyzed by EMSA
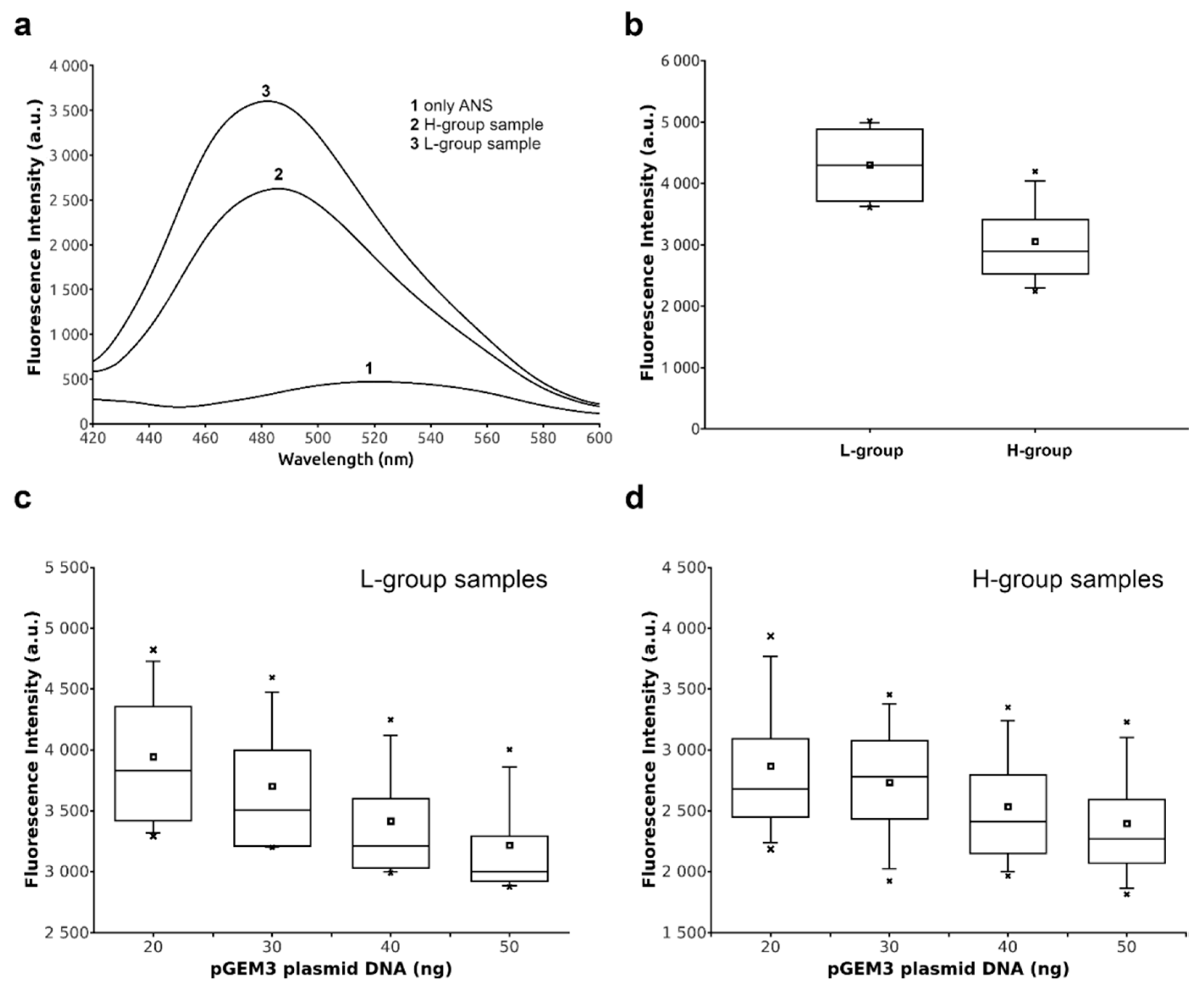
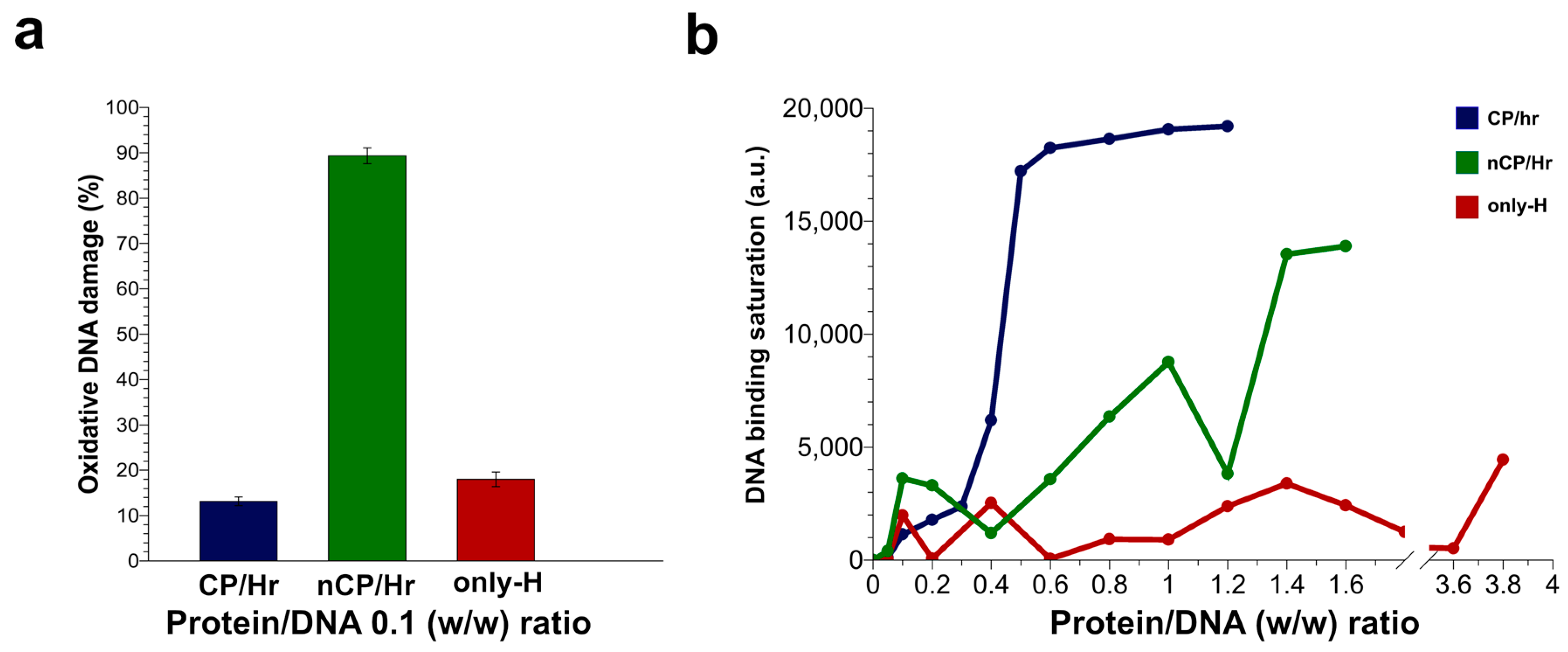
2.4. DNA Binding Ability of Sperm Proteins Determined by Fluorescence Analysis
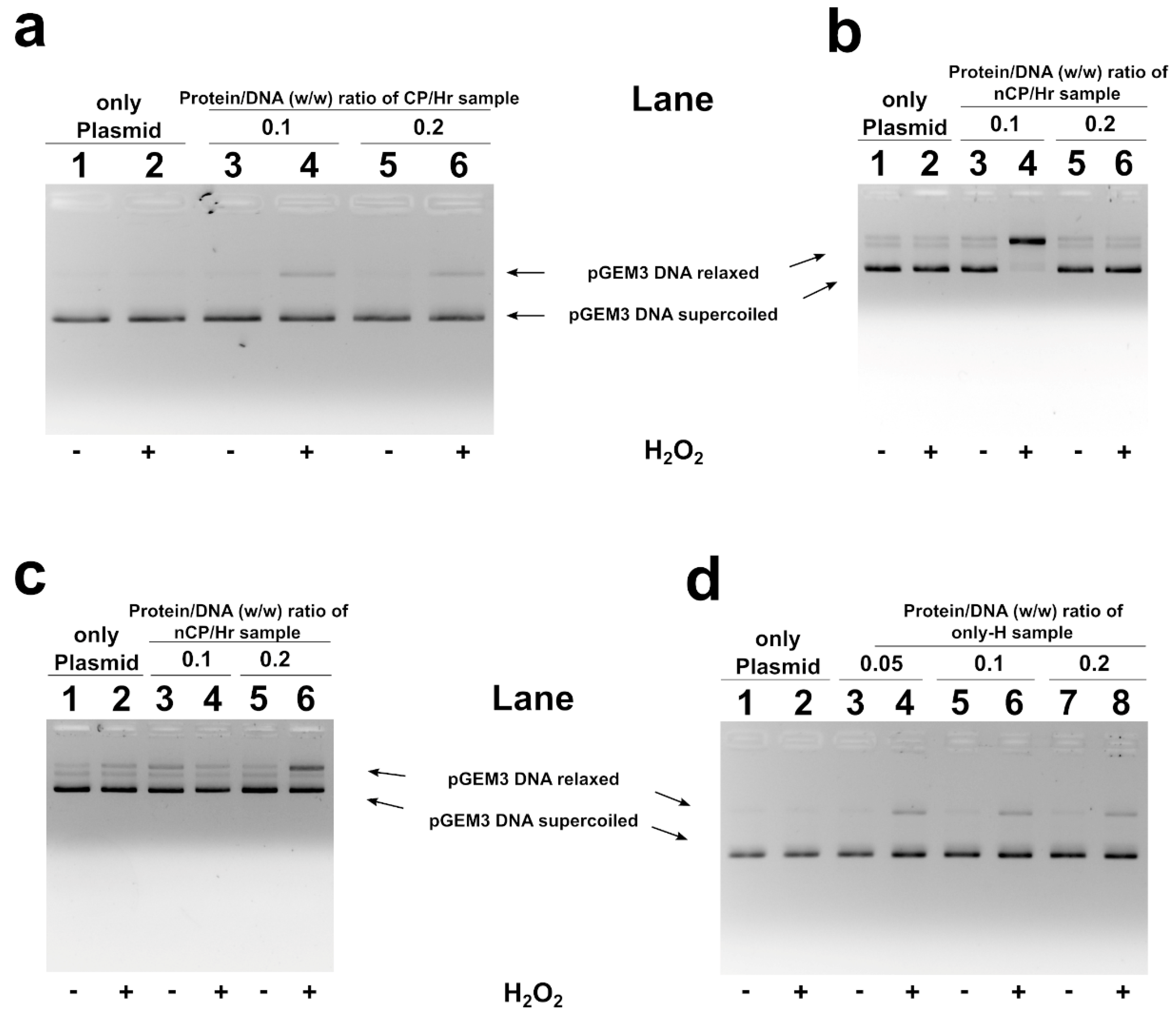
2.5. H2O2-Induced DNA Breakage in the Presence of Human SNBP
3. Discussion
4. Materials and Methods
4.1. Reagents
4.2. Ethical Statements
4.3. Recruitment
4.4. Spermatozoa Collection and SNBP Extraction
4.5. Acid-Urea Polyacrylamide Gel Electrophoresis
4.6. Plasmid DNA Preparation
4.7. DNA Binding Affinity of SNBP by EMSA
4.8. Fluorescence Spectroscopy
4.9. DNA Breakage Analyses
4.10. Aniline Blue Staining
4.11. Densitometric Gel Analysis
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| ROS | Reactive oxygen species |
| AU-PAGE | Acid-urea Polyacrylamide Gel Electrophoresis |
| EMSA | Electrophoretic Mobility Shift Assays |
| ANS | 8-Anilinonaphthalene-1-sulfonic acid |
| SNBP | Sperm Nuclear Basic Proteins |
| H-group | Man living in the high environmental impact areas |
| L-group | Man living in the low environmental impact areas |
| CP/Hr | Canonical protamines/histones ratio |
| nCP/Hr | Not canonical protamines/histones ratio |
| only-H | Only histones and other basic proteins |
| DFI | DNA fragmentation index |
References
- Abbotts, R.; Wilson, D.M. Coordination of DNA single strand break repair. Free Radic. Biol. Med. 2017, 107, 228–244. [Google Scholar] [CrossRef] [PubMed]
- Chatterjee, N.; Walker, G.C. Mechanisms of DNA damage, repair, and mutagenesis. Environ. Mol. Mutagen. 2017, 58, 235–263. [Google Scholar] [CrossRef]
- Nair, S.; Engelbrecht, M.; Miles, X.; Ndimba, R.; Fisher, R.; Du Plessis, P.; Bolcaen, J.; Nieto-Camero, J.; De Kock, E.; Vandevoorde, C. The Impact of Dose Rate on DNA Double-Strand Break Formation and Repair in Human Lymphocytes Exposed to Fast Neutron Irradiation. Int. J. Mol. Sci. 2019, 20, 5350. [Google Scholar] [CrossRef] [PubMed]
- Piscopo, M.; Trifuoggi, M.; Scarano, C.; Gori, C.; Giarra, A.; Febbraio, F. Relevance of arginine residues in Cu(II)-induced DNA breakage and Proteinase K resistance of H1 histones. Sci. Rep. 2018, 8, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Lettieri, G.; Mollo, V.; Ambrosino, A.; Caccavale, F.; Troisi, J.; Febbraio, F.; Piscopo, M. Molecular effects of copper on the reproductive system of Mytilus galloprovincialis. Mol. Reprod. Dev. 2019, 86, 1357–1368. [Google Scholar] [CrossRef] [PubMed]
- Dutta, A.; Yang, C.; Sengupta, S.; Mitra, S.; Hegde, M.L. New paradigms in the repair of oxidative damage in human genome: Mechanisms ensuring repair of mutagenic base lesions during replication and involvement of accessory proteins. Cell. Mol. Life Sci. 2015, 72, 1679–1698. [Google Scholar] [CrossRef]
- Alahmar, A.T. Role of Oxidative Stress in Male Infertility: An Updated Review. J. Hum. Reprod. Sci. 2019, 12, 4–18. [Google Scholar] [CrossRef]
- Barati, E.; Nikzad, H.; Karimian, M. Oxidative stress and male infertility: Current knowledge of pathophysiology and role of antioxidant therapy in disease management. Cell. Mol. Life Sci. 2020, 77, 93–113. [Google Scholar] [CrossRef]
- Collin, F. Chemical Basis of Reactive Oxygen Species Reactivity and Involvement in Neurodegenerative Diseases. Int. J. Mol. Sci. 2019, 20, 2407. [Google Scholar] [CrossRef]
- Dumont, E.; Monari, A. Understanding DNA under oxidative stress and sensitization: The role of molecular modeling. Front. Chem. 2015, 3, 43. [Google Scholar] [CrossRef]
- Bergamo, P.; Volpe, M.G.; Lorenzetti, S.; Mantovani, A.; Notari, T.; Cocca, E.; Cerullo, S.; Di Stasio, M.; Cerino, P.; Montano, L. Human semen as an early, sensitive biomarker of highly polluted living environment in healthy men: A pilot biomonitoring study on trace elements in blood and semen and their relationship with sperm quality and RedOx status. Reprod. Toxicol. 2016, 66, 1–9. [Google Scholar] [CrossRef]
- Bosco, L.; Notari, T.; Ruvolo, G.; Roccheri, M.C.; Martino, C.; Chiappetta, R.; Carone, D.; Lo Bosco, G.; Carrillo, L.; Raimondo, S.; et al. Sperm DNA fragmentation: An early and reliable marker of air pollution. Environ. Toxicol. Pharmacol. 2018, 58, 243–249. [Google Scholar] [CrossRef]
- Maresca, V.; Fusaro, L.; Sorbo, S.; Siciliano, A.; Loppi, S.; Paoli, L.; Monaci, F.; Karam, E.A.; Piscopo, M.; Guida, M.; et al. Functional and structural biomarkers to monitor heavy metal pollution of one of the most contaminated freshwater sites in Southern Europe. Ecotoxicol. Environ. Saf. 2018, 163, 665–673. [Google Scholar] [CrossRef]
- Basile, A.; Loppi, S.; Piscopo, M.; Paoli, L.; Vannini, A.; Monaci, F.; Sorbo, S.; Lentini, M.; Esposito, S. The biological response chain to pollution: A case study from the “Italian Triangle of Death” assessed with the liverwort Lunularia cruciata. Environ. Sci. Pollut. Res. Int. 2017, 24, 26185–26193. [Google Scholar] [CrossRef]
- Vecoli, C.; Montano, L.; Borghini, A.; Notari, T.; Guglielmino, A.; Mercuri, A.; Turchi, S.; Andreassi, M.G. Effects of Highly Polluted Environment on Sperm Telomere Length: A Pilot Study. Int. J. Mol. Sci. 2017, 18, 1703. [Google Scholar] [CrossRef]
- Ovári, L.; Sati, L.; Stronk, J.; Borsos, A.; Ward, D.C.; Huszar, G. Double probing individual human spermatozoa: Aniline blue staining for persistent histones and fluorescence in situ hybridization for aneuploidies. Fertil. Steril. 2010, 93, 2255–2261. [Google Scholar] [CrossRef]
- Soler-Ventura, A.; Castillo, J.; De la Iglesia, A.; Jodar, M.; Barrachina, F.; Ballesca, J.L.; Oliva, R. Mammalian Sperm Protamine Extraction and Analysis: A Step-By-Step Detailed Protocol and Brief Review of Protamine Alterations. Protein Pept. Lett. 2018, 25, 424–433. [Google Scholar] [CrossRef]
- Piscopo, M.; Notariale, R.; Rabbito, D.; Ausió, J.; Olanrewaju, O.S.; Guerriero, G. Mytilus galloprovincialis (Lamarck, 1819) spermatozoa: Hsp70 expression and protamine-like protein property studies. Environ. Sci. Pollut. Res. Int. 2018, 25, 12957–12966. [Google Scholar] [CrossRef]
- Piscopo, M.; Conte, M.; Di Paola, F.; Conforti, S.; Rana, G.; De Petrocellis, L.; Fucci, L.; Geraci, G. Relevance of arginines in the mode of binding of H1 histones to DNA. DNA Cell Biol. 2010, 29, 339–347. [Google Scholar] [CrossRef]
- Piscopo, M.; De Petrocellis, L.; Conte, M.; Pulcrano, G.; Geraci, G. On the possibility that H1 histone interaction with DNA occurs through phosphates connecting lysine and arginine side chain groups. Acta Biochim. Pol. 2006, 53, 507–513. [Google Scholar] [CrossRef]
- Slavík, J. Anilinonaphthalene sulfonate as a probe of membrane composition and function. Biochim. Biophys. Acta (BBA) Rev. Biomembr. 1982, 694, 1–25. [Google Scholar] [CrossRef]
- Klymchenko, A.S.; Mely, Y. Chapter Two—Fluorescent Environment-Sensitive Dyes as Reporters of Biomolecular Interactions. In Fluorescence-Based Biosensors; Progress in Molecular Biology and Translational Science; Morris, M.C., Ed.; Academic Press: Cambridge, MA, USA, 2013; Volume 113, pp. 35–58. [Google Scholar]
- Wong, E.W.P.; Cheng, C.Y. Impacts of environmental toxicants on male reproductive dysfunction. Trends Pharmacol. Sci. 2011, 32, 290–299. [Google Scholar] [CrossRef] [PubMed]
- Manfo, F.P.T.; Nantia, E.A.; Mathur, P.P. Effect of environmental contaminants on mammalian testis. Curr. Mol. Pharmacol. 2014, 7, 119–135. [Google Scholar] [CrossRef] [PubMed]
- Schulte, R.T.; Ohl, D.A.; Sigman, M.; Smith, G.D. Sperm DNA damage in male infertility: Etiologies, assays, and outcomes. J. Assist. Reprod. Genet. 2010, 27, 3–12. [Google Scholar] [CrossRef]
- González-Marín, C.; Gosálvez, J.; Roy, R. Types, causes, detection and repair of DNA fragmentation in animal and human sperm cells. Int. J. Mol. Sci. 2012, 13, 14026–14052. [Google Scholar] [CrossRef]
- Nelson, W.G.; Pienta, K.J.; Barrack, E.R.; Coffey, D.S. The role of the nuclear matrix in the organization and function of DNA. Annu. Rev. Biophys. Biophys. Chem. 1986, 15, 457–475. [Google Scholar] [CrossRef]
- Liang, Q.; Dedon, P.C. Cu(II)/H2O2-induced DNA damage is enhanced by packaging of DNA as a nucleosome. Chem. Res. Toxicol. 2001, 14, 416–422. [Google Scholar] [CrossRef]
- De Guglielmo, V.; Puoti, R.; Notariale, R.; Maresca, V.; Ausió, J.; Troisi, J.; Verrillo, M.; Basile, A.; Febbraio, F.; Piscopo, M. Alterations in the properties of sperm protamine-like II protein after exposure of Mytilus galloprovincialis (Lamarck 1819) to sub-toxic doses of cadmium. Ecotoxicol. Environ. Saf. 2019, 169, 600–606. [Google Scholar] [CrossRef] [PubMed]
- Piscopo, M.; Ricciardiello, M.; Palumbo, G.; Troisi, J. Selectivity of metal bioaccumulation and its relationship with glutathione S-transferase levels in gonadal and gill tissues of Mytilus galloprovincialis exposed to Ni (II), Cu (II) and Cd (II). Rend. Lincei Sci. Fis. Nat. 2016, 27, 737–748. [Google Scholar] [CrossRef]
- Gaetke, L.M.; Chow, C.K. Copper toxicity, oxidative stress, and antioxidant nutrients. Toxicology 2003, 189, 147–163. [Google Scholar] [CrossRef]
- Theophanides, T.; Anastassopoulou, J. Copper and carcinogenesis. Crit. Rev. Oncol. Hemat. 2002, 42, 57–64. [Google Scholar] [CrossRef]
- Govindaraju, M.; Shekar, H.S.; Sateesha, S.B.; Vasudeva Raju, P.; Sambasiva Rao, K.R.; Rao, K.S.J.; Rajamma, A.J. Copper interactions with DNA of chromatin and its role in neurodegenerative disorders. J. Pharm. Anal. 2013, 3, 354–359. [Google Scholar] [CrossRef] [PubMed]
- Arif, H.; Rehmani, N.; Farhan, M.; Ahmad, A.; Hadi, S.M. Mobilization of Copper ions by Flavonoids in Human Peripheral Lymphocytes Leads to Oxidative DNA Breakage: A Structure Activity Study. Int. J. Mol. Sci. 2015, 16, 26754–26769. [Google Scholar] [CrossRef] [PubMed]
- Cabelli, D.E.; Bielski, B.H.J.; Holcman, J. Interaction between copper(II)-arginine complexes and HO2/O2- radicals, a pulse radiolysis study. J. Am. Chem. Soc. 1987, 109, 3665–3669. [Google Scholar] [CrossRef]
- Patra, A.K.; Bhowmick, T.; Roy, S.; Ramakumar, S.; Chakravarty, A.R. Copper(II) Complexes of l-Arginine as Netropsin Mimics Showing DNA Cleavage Activity in Red Light. Inorg. Chem. 2009, 48, 2932–2943. [Google Scholar] [CrossRef]
- Wojciechowska, A.; Gągor, A.; Zierkiewicz, W.; Jarząb, A.; Dylong, A.; Duczmal, M. Metal–organic framework in an L-arginine copper(II) ion polymer: Structure, properties, theoretical studies and microbiological activity. RSC Adv. 2015, 5, 36295–36306. [Google Scholar] [CrossRef]
- Linder, M.C. The relationship of copper to DNA damage and damage prevention in humans. Mutat. Res. 2012, 733, 83–91. [Google Scholar] [CrossRef]
- Bal, W.; Jeżowska-Bojczuk, M.; Kasprzak, K.S. Binding of Nickel(II) and Copper(II) to the N-Terminal Sequence of Human Protamine HP2. Chem. Res. Toxicol. 1997, 10, 906–914. [Google Scholar] [CrossRef]
- Bal, W.; Lukszo, J.; Kasprzak, K.S. Mediation of oxidative DNA damage by nickel(II) and copper(II) complexes with the N-terminal sequence of human protamine HP2. Chem. Res. Toxicol. 1997, 10, 915–921. [Google Scholar] [CrossRef]
- Khorsandi, K.; Rabbani-Chadegani, A. Investigation on the chromium oxide interaction with soluble chromatin and histone H1: A spectroscopic study. Int. J. Biol. Macromol. 2014, 70, 57–63. [Google Scholar] [CrossRef]
- Luo, H.; Lu, Y.; Shi, X.; Mao, Y.; Dalal, N.S. Chromium (IV)-mediated fenton-like reaction causes DNA damage: Implication to genotoxicity of chromate. Ann. Clin. Lab. Sci. 1996, 26, 185–191. [Google Scholar] [PubMed]
- Zhitkovich, A. Importance of chromium-DNA adducts in mutagenicity and toxicity of chromium(VI). Chem. Res. Toxicol. 2005, 18, 3–11. [Google Scholar] [CrossRef]
- Holmes, A.L.; Wise, S.S.; Sandwick, S.J.; Lingle, W.L.; Negron, V.C.; Thompson, W.D.; Wise, J.P. Chronic exposure to lead chromate causes centrosome abnormalities and aneuploidy in human lung cells. Cancer Res. 2006, 66, 4041–4048. [Google Scholar] [CrossRef]
- Karthikeyan, K.S.; Polasa, H.; Sastry, K.S.; Reddy, G. Metabolism of lysine-chromium complex in Saccharomyces cerevisiae. Indian J. Microbiol. 2008, 48, 397–400. [Google Scholar] [CrossRef] [PubMed]
- Gasymov, O.K.; Glasgow, B.J. ANS fluorescence: Potential to augment the identification of the external binding sites of proteins. Biochim. Biophys. Acta 2007, 1774, 403–411. [Google Scholar] [CrossRef] [PubMed]
- Fioretti, F.M.; Febbraio, F.; Carbone, A.; Branno, M.; Carratore, V.; Fucci, L.; Ausió, J.; Piscopo, M. A sperm nuclear basic protein from the sperm of the marine worm Chaetopterus variopedatus with sequence similarity to the arginine-rich C-termini of chordate protamine-likes. DNA Cell Biol. 2012, 31, 1392–1402. [Google Scholar] [CrossRef] [PubMed]
- Yamaguchi, K.; Hada, M.; Fukuda, Y.; Inoue, E.; Makino, Y.; Katou, Y.; Shirahige, K.; Okada, Y. Re-evaluating the Localization of Sperm-Retained Histones Revealed the Modification-Dependent Accumulation in Specific Genome Regions. Cell Rep. 2018, 23, 3920–3932. [Google Scholar] [CrossRef] [PubMed]
- Brykczynska, U.; Hisano, M.; Erkek, S.; Ramos, L.; Oakeley, E.J.; Roloff, T.C.; Beisel, C.; Schübeler, D.; Stadler, M.B.; Peters, A.H.F.M. Repressive and active histone methylation mark distinct promoters in human and mouse spermatozoa. Nat. Struct. Mol. Biol. 2010, 17, 679–687. [Google Scholar] [CrossRef]
- Jung, Y.H.; Sauria, M.E.G.; Lyu, X.; Cheema, M.S.; Ausio, J.; Taylor, J.; Corces, V.G. Chromatin States in Mouse Sperm Correlate with Embryonic and Adult Regulatory Landscapes. Cell Rep. 2017, 18, 1366–1382. [Google Scholar] [CrossRef]
- Hammoud, S.S.; Nix, D.A.; Zhang, H.; Purwar, J.; Carrell, D.T.; Cairns, B.R. Distinctive chromatin in human sperm packages genes for embryo development. Nature 2009, 460, 473–478. [Google Scholar] [CrossRef]
- Sun, H.; Zhou, X.; Chen, H.; Li, Q.; Costa, M. Modulation of histone methylation and MLH1 gene silencing by hexavalent chromium. Toxicol. Appl. Pharmacol. 2009, 237, 258–266. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, L.M.R.; Cunha-Oliveira, T.; Sobral, M.C.; Abreu, P.L.; Alpoim, M.C.; Urbano, A.M. Impact of Carcinogenic Chromium on the Cellular Response to Proteotoxic Stress. Int. J. Mol. Sci. 2019, 20, 4901. [Google Scholar] [CrossRef] [PubMed]
- Schnekenburger, M.; Talaska, G.; Puga, A. Chromium cross-links histone deacetylase 1-DNA methyltransferase 1 complexes to chromatin, inhibiting histone-remodeling marks critical for transcriptional activation. Mol. Cell. Biol. 2007, 27, 7089–7101. [Google Scholar] [CrossRef] [PubMed]
- Chervona, Y.; Costa, M. The control of histone methylation and gene expression by oxidative stress, hypoxia and metals. Free Radic. Biol. Med. 2012, 53, 1041–1047. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, L.T.H.; Janssen, C.R. Comparative sensitivity of embryo-larval toxicity assays with African catfish (Clarias gariepinus) and zebra fish (Danio rerio). Environ. Toxicol. 2001. [Google Scholar] [CrossRef]
- Goldberg, S.J.; Lebowitz, M.D.; Graver, E.J.; Hicks, S. An association of human congenital cardiac malformations and drinking water contaminants. J. Am. Coll. Cardiol. 1990, 16, 155–164. [Google Scholar] [CrossRef]
- Vecoli, C.; Montano, L.; Andreassi, M.G. Environmental pollutants: Genetic damage and epigenetic changes in male germ cells. Environ. Sci. Pollut. Res. Int. 2016, 23, 23339–23348. [Google Scholar] [CrossRef]
- De Yebra, L.; Oliva, R. Rapid analysis of mammalian sperm nuclear proteins. Anal. Biochem. 1993, 209, 201–203. [Google Scholar] [CrossRef]
- Vassalli, Q.A.; Caccavale, F.; Avagnano, S.; Murolo, A.; Guerriero, G.; Fucci, L.; Ausió, J.; Piscopo, M. New insights into protamine-like component organization in Mytilus galloprovincialis’ sperm chromatin. DNA Cell Biol. 2015, 34, 162–169. [Google Scholar] [CrossRef] [PubMed]
- Carbone, A.; Fioretti, F.M.; Fucci, L.; Ausió, J.; Piscopo, M. High efficiency method to obtain supercoiled DNA with a commercial plasmid purification kit. Acta Biochim. Pol. 2012, 59, 275–278. [Google Scholar] [CrossRef] [PubMed]
- Piscopo, M.; Trifuoggi, M.; Notariale, R.; Labar, S.; Troisi, J.; Giarra, A.; Rabbito, D.; Puoti, R.; De Benedictis, D.; Brundo, M.V.; et al. Protamine-like proteins’ analysis as an emerging biotechnique for cadmium impact assessment on male mollusk Mytilus galloprovincialis (Lamarck 1819). Acta Biochim. Pol. 2018, 65, 259–267. [Google Scholar] [CrossRef] [PubMed]
- Rahiminia, T.; Yazd, E.F.; Fesahat, F.; Moein, M.R.; Mirjalili, A.M.; Talebi, A.R. Sperm chromatin and DNA integrity, methyltransferase mRNA levels, and global DNA methylation in oligoasthenoteratozoospermia. Clin. Exp. Reprod. Med. 2018, 45, 17–24. [Google Scholar] [CrossRef] [PubMed]
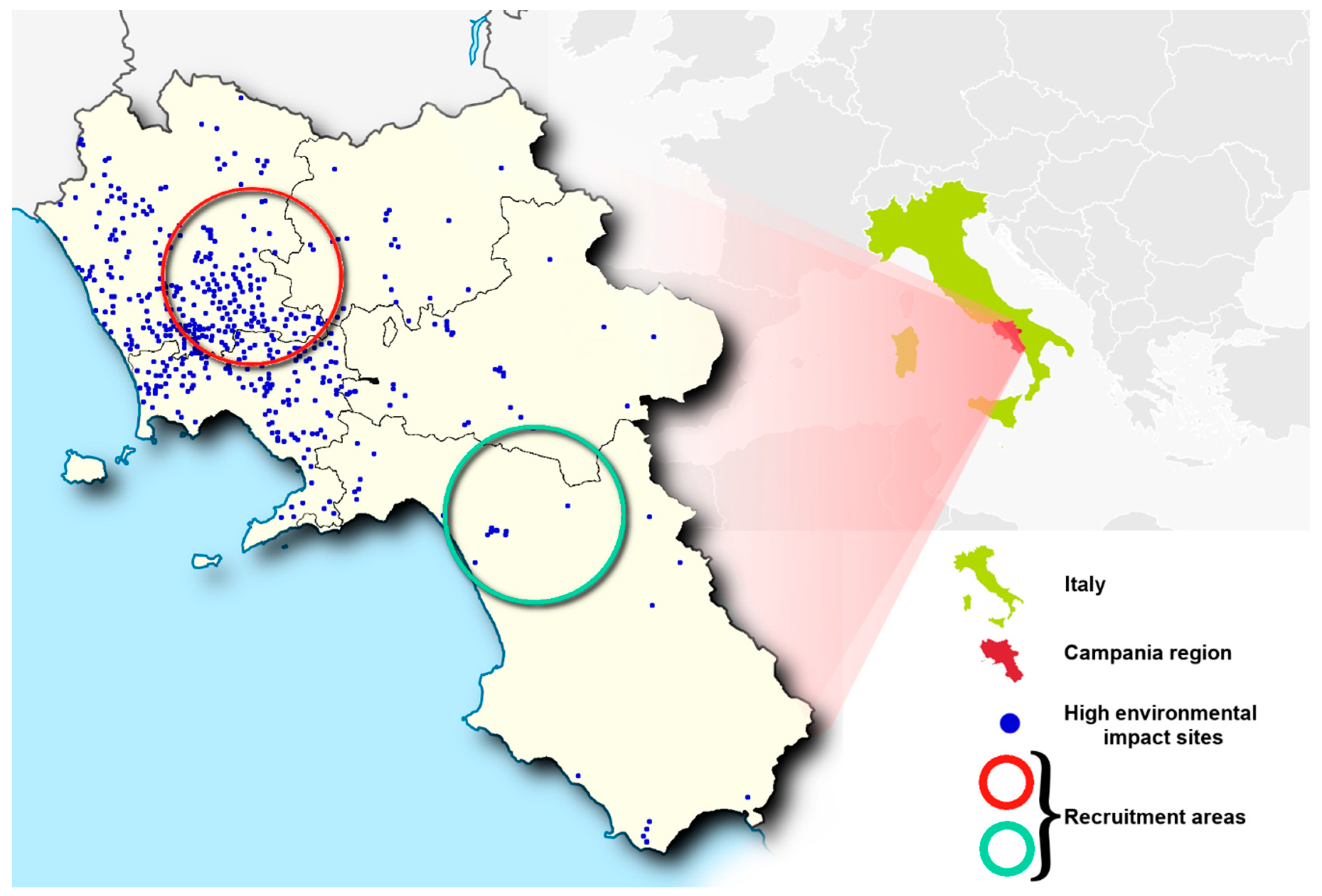
| Municipalities | |
|---|---|
| Low Impact Areas | High Impact Areas |
| Oliveto Citra–Contursi Terme–San Gregorio Magno–Buccino–Ricigliano–Valva–Colliano | Acerra–Caivano–Afragola–Casalnuovo-Pomigliano d’Arco–Brusciano–Giugliano–Cardito–Marigliano |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lettieri, G.; D’Agostino, G.; Mele, E.; Cardito, C.; Esposito, R.; Cimmino, A.; Giarra, A.; Trifuoggi, M.; Raimondo, S.; Notari, T.; et al. Discovery of the Involvement in DNA Oxidative Damage of Human Sperm Nuclear Basic Proteins of Healthy Young Men Living in Polluted Areas. Int. J. Mol. Sci. 2020, 21, 4198. https://doi.org/10.3390/ijms21124198
Lettieri G, D’Agostino G, Mele E, Cardito C, Esposito R, Cimmino A, Giarra A, Trifuoggi M, Raimondo S, Notari T, et al. Discovery of the Involvement in DNA Oxidative Damage of Human Sperm Nuclear Basic Proteins of Healthy Young Men Living in Polluted Areas. International Journal of Molecular Sciences. 2020; 21(12):4198. https://doi.org/10.3390/ijms21124198
Chicago/Turabian StyleLettieri, Gennaro, Giovanni D’Agostino, Elena Mele, Carolina Cardito, Rosa Esposito, Annalinda Cimmino, Antonella Giarra, Marco Trifuoggi, Salvatore Raimondo, Tiziana Notari, and et al. 2020. "Discovery of the Involvement in DNA Oxidative Damage of Human Sperm Nuclear Basic Proteins of Healthy Young Men Living in Polluted Areas" International Journal of Molecular Sciences 21, no. 12: 4198. https://doi.org/10.3390/ijms21124198
APA StyleLettieri, G., D’Agostino, G., Mele, E., Cardito, C., Esposito, R., Cimmino, A., Giarra, A., Trifuoggi, M., Raimondo, S., Notari, T., Febbraio, F., Montano, L., & Piscopo, M. (2020). Discovery of the Involvement in DNA Oxidative Damage of Human Sperm Nuclear Basic Proteins of Healthy Young Men Living in Polluted Areas. International Journal of Molecular Sciences, 21(12), 4198. https://doi.org/10.3390/ijms21124198










