The Cytotoxicity and Genotoxicity of Three Dental Universal Adhesives—An In Vitro Study
Abstract
1. Introduction
2. Results
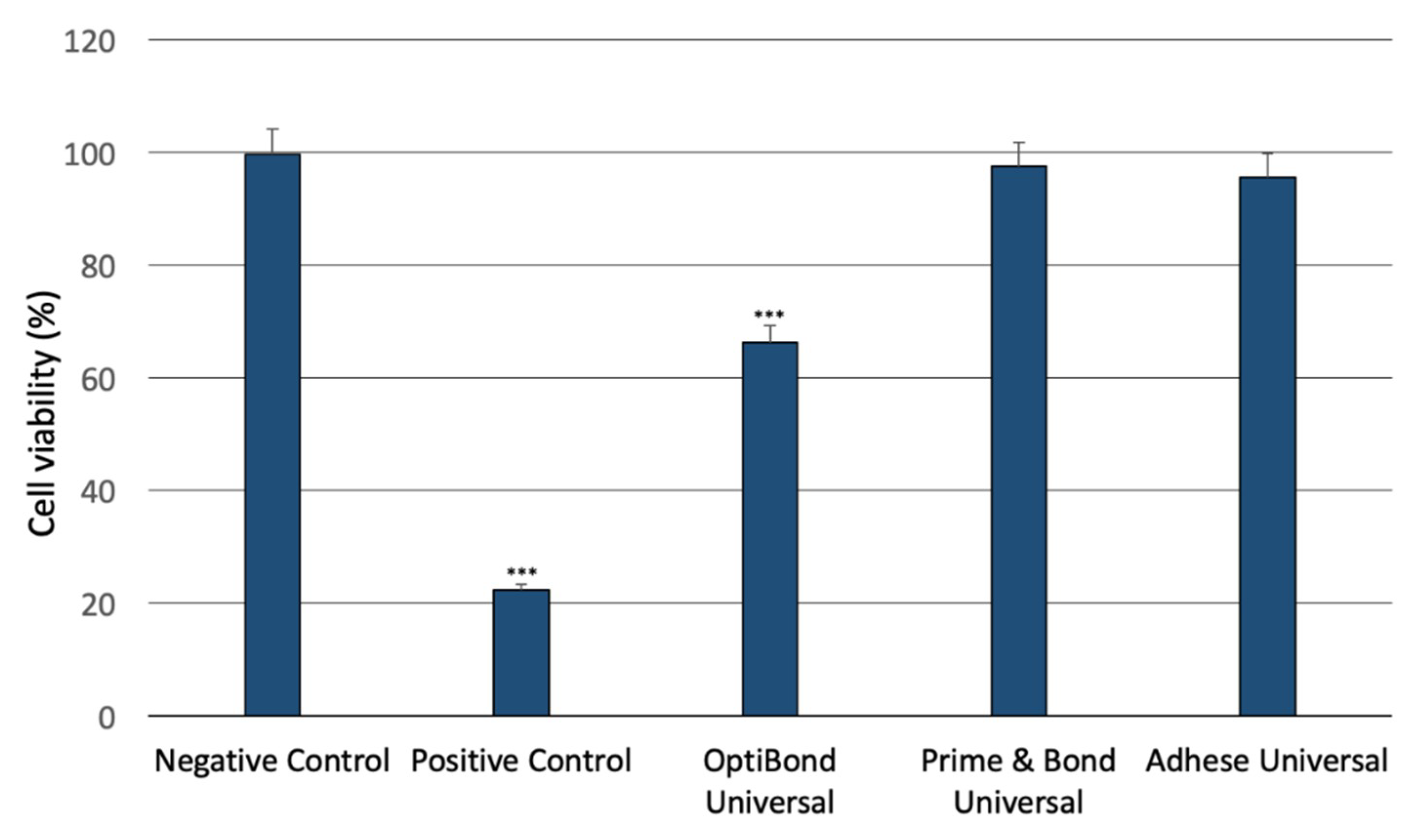
2.1. Analysis of the Cytotoxicity of the Dental Universal Adhesives
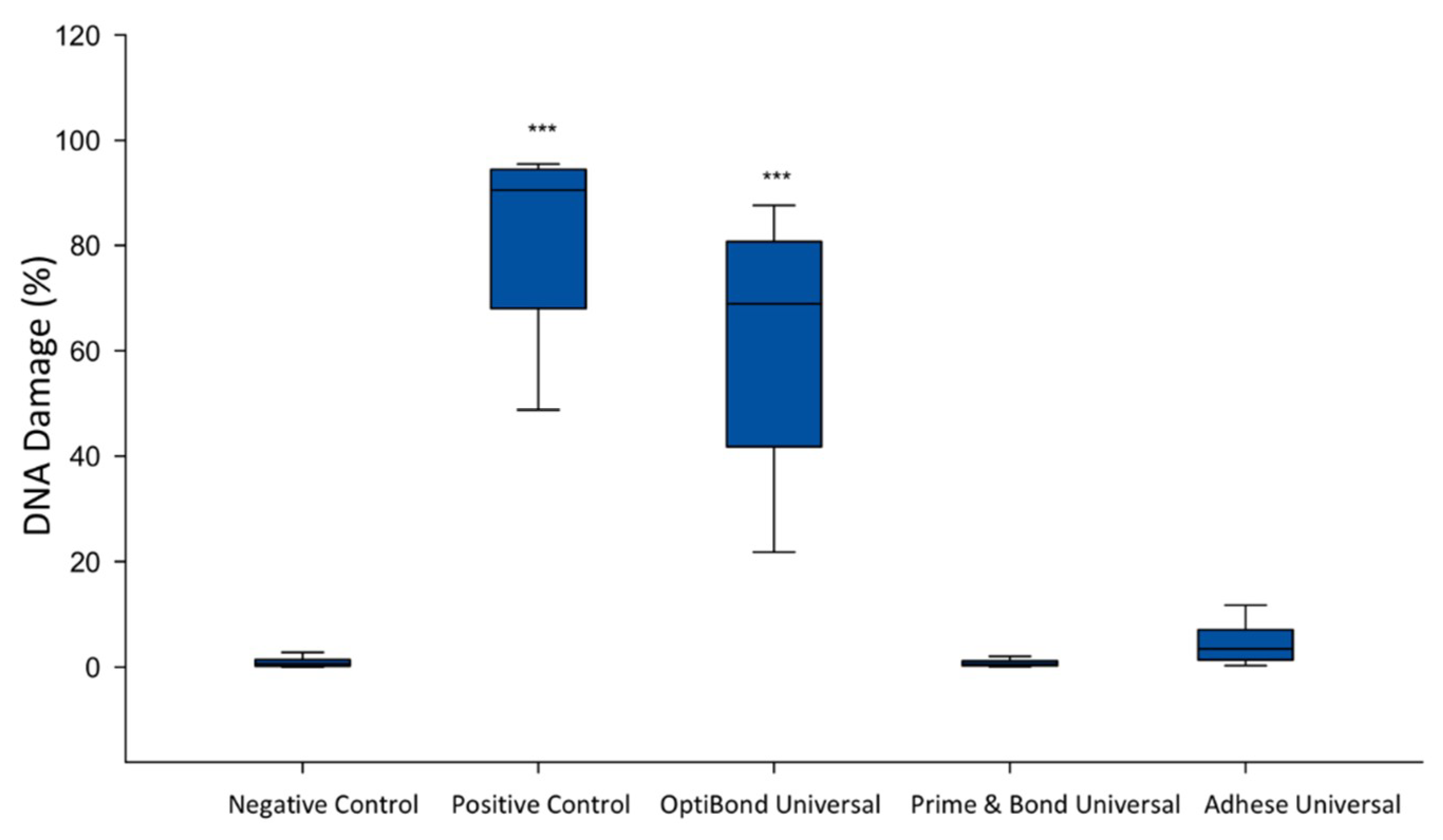
2.2. Analysis of the Genotoxicity of the Dental Universal Adhesives
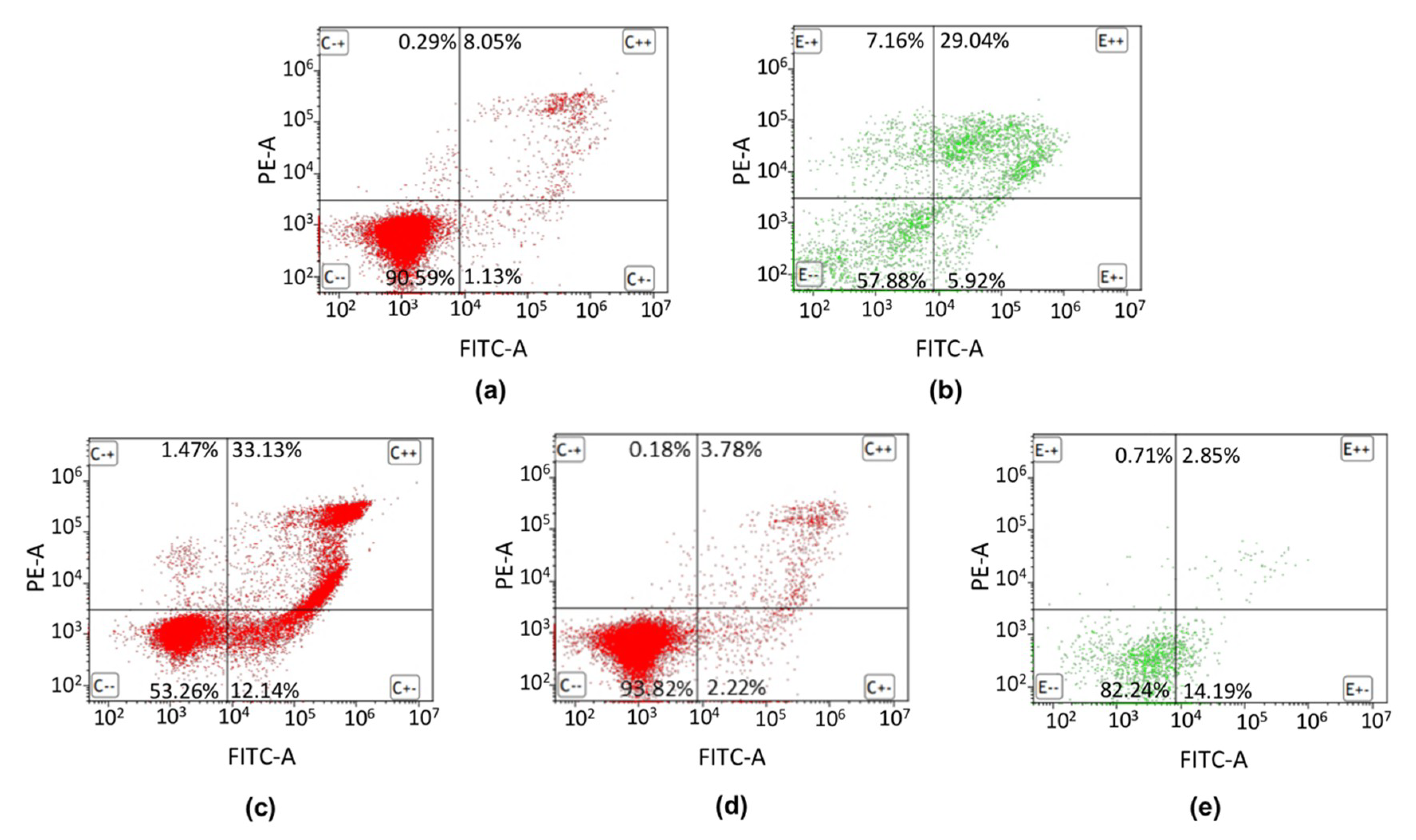
2.3. Apoptosis Detection by FITC Annexin V/PI Double Staining of the Dental Universal Adhesives
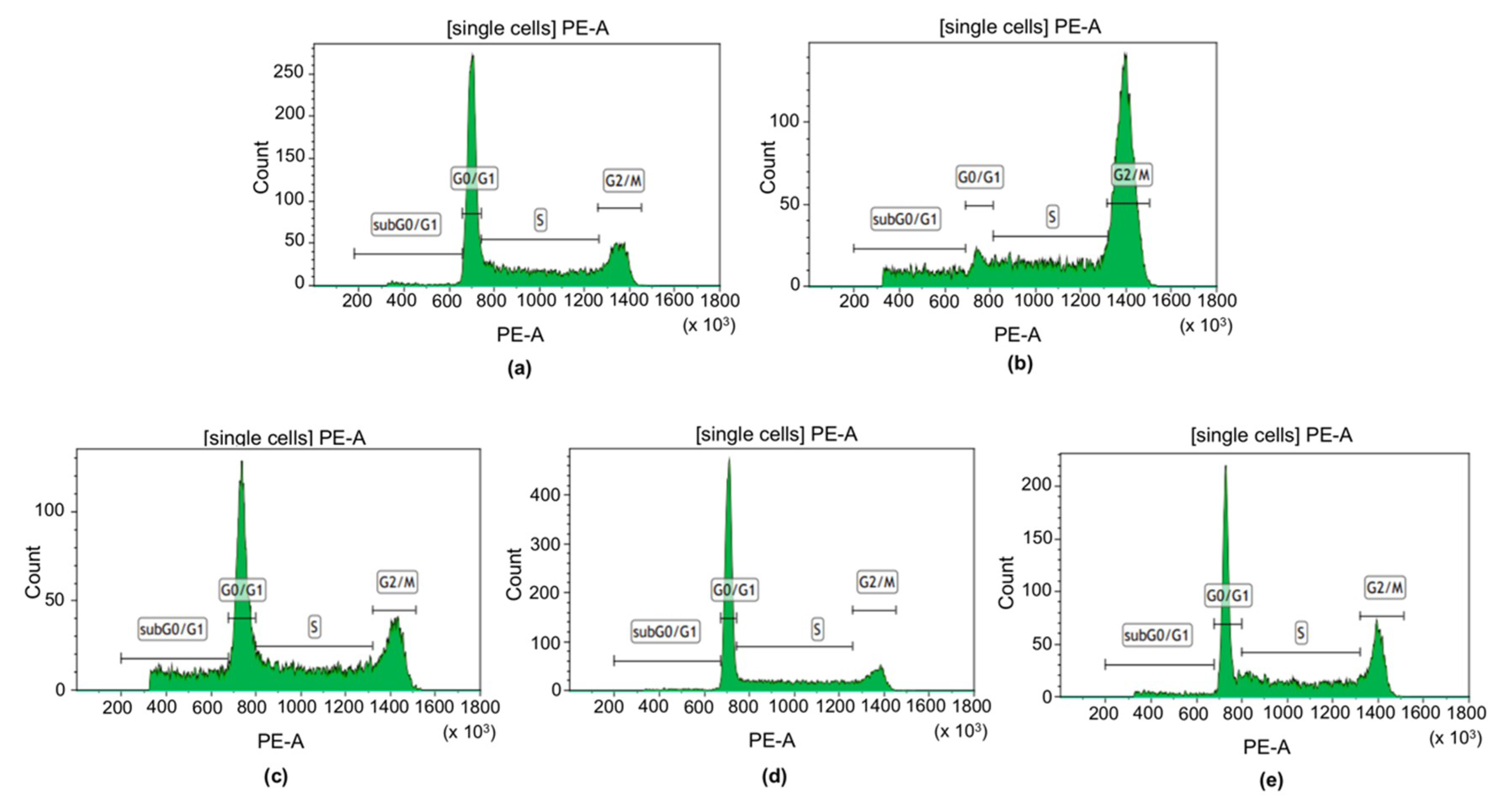
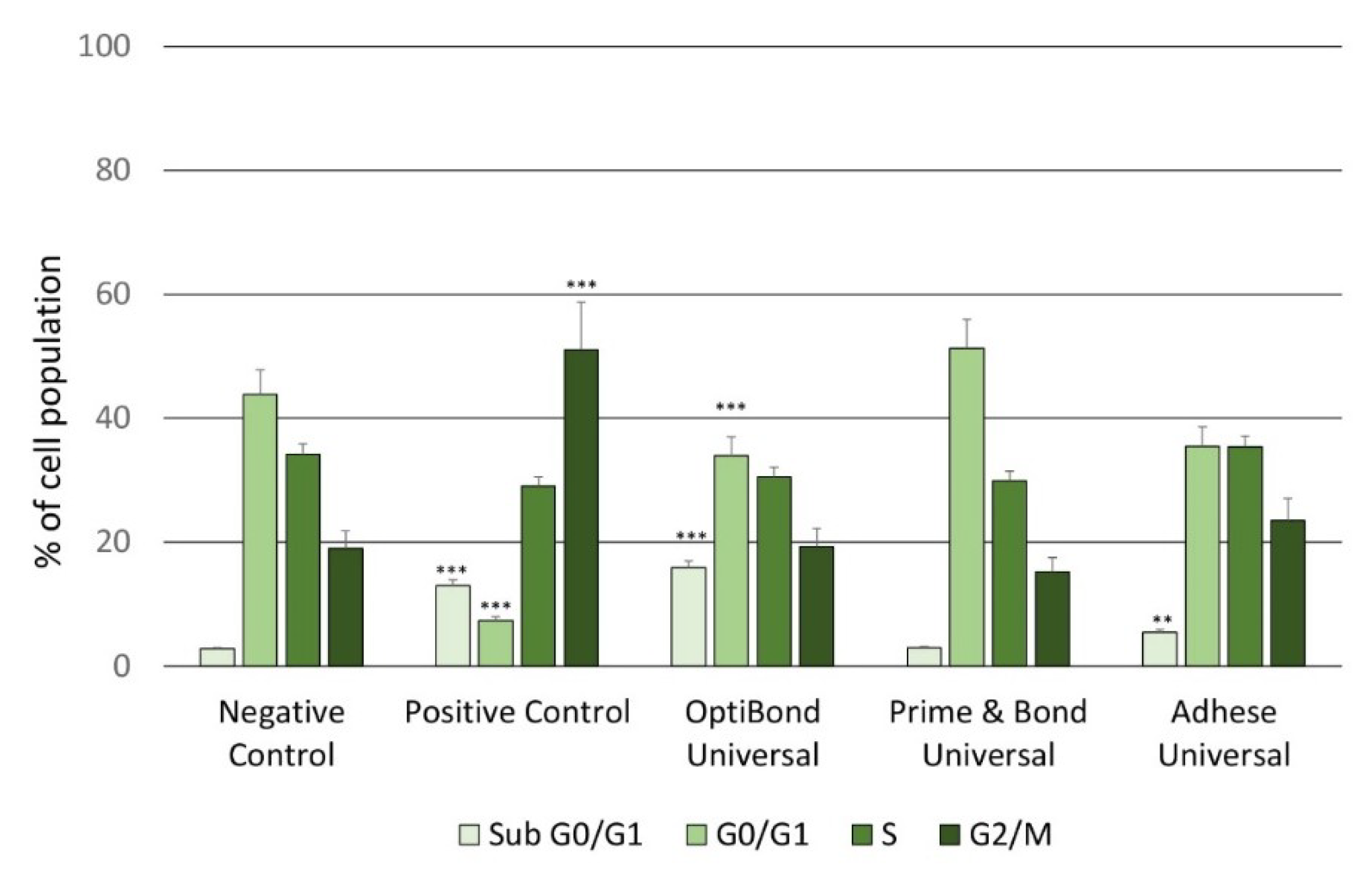
2.4. Analysis of the Cell Cycle Progression by PI Staining of the Dental Universal Adhesives
3. Discussion
4. Materials and Methods
4.1. Cell Line and Eluate Preparation
4.2. Cytotoxicity Analysis
4.3. Genotoxicity Assessment
4.4. Apoptosis Detection
4.5. Cell Cycle Analysis
4.6. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| BPDM | biphenyl dimethacrylate |
| PENTA | dipentaerythritol pentaacrylate phosphoric acid ester |
| D3MA | decanediol dimethacrylate |
| HEMA | 2-hydroxyethyl methacrylate |
| bis-GMA | bisphenol A-glycidyl methacrylate |
| MDP | methacryloyloxi-decyl-dihydrogen-phosphate |
| Phenyl-P | N-Phenyl-p-phenylenediamine |
| 4-MET | 4-methacryloxyethyl trimellitic acid |
| UDMA | urethane dimethacrylate |
| TEGDMA | triethylene glycol dimethacrylate |
| XTT | Cell Proliferation Kit II |
| MTT | Cell Proliferation Kit I |
| FC | flow cytometry |
| PI | propidium iodide |
| DAPI | 4′,6-Diamidino-2-Phenylindole |
| SC | monocyte/macrophage peripheral blood cell line |
| PBS | Phosphate-buffered saline |
References
- Milia, E.; Cumbo, E.; Jose, A.; Cardoso, R.; Gallina, G. Current Dental Adhesives Systems. A Narrative Review. Curr. Pharm. Des. 2012, 18, 5542–5552. [Google Scholar] [CrossRef] [PubMed]
- Sofan, E.; Sofan, A.; Palaia, G.; Tenore, G.; Romeo, U.; Migliau, G. Classification review of dental adhesive systems: From the IV generation to the universal type. Ann. Stomatol. 2017, 8, 1–17. [Google Scholar]
- Mante, F.K.; Ozer, F.; Walter, R.; Atlas, A.M.; Saleh, N.; Dietschi, D.; Blatz, M.B. The current state of adhesive dentistry: A guide for clinical practice. Compend. Contin. Educ. Dent. 2013, 34, 2–8. [Google Scholar] [PubMed]
- Van Landuyt, K.L.; Snauwaert, J.; De Munck, J.; Peumans, M.; Yoshida, Y.; Poitevin, A.; Coutinho, E.; Suzuki, K.; Lambrechts, P.; Van Meerbeek, B. Systematic review of the chemical composition of contemporary dental adhesives. Biomaterials 2007, 28, 3757–3785. [Google Scholar] [CrossRef] [PubMed]
- Perdigão, J.; Sezinando, A.; Monteiro, P.C. Laboratory bonding ability of a multi-purpose dentin adhesive. Am. J. Dent. 2012, 25, 153–158. [Google Scholar]
- Hanabusa, M.; Mine, A.; Kuboki, T.; Momoi, Y.; Van Ende, A.; Van Meerbeek, B.; De Munck, J. Bonding effectiveness of a new “multi-mode” adhesive to enamel and dentine. J. Dent. 2012, 40, 475–484. [Google Scholar] [CrossRef]
- Zecin-Deren, A.; Lukomska-Szymanska, M.; Szczesio-Wlodarczyk, A.; Piwonski, I.; Sokolowski, J.; Lapinska, B. The Influence of Application Protocol of Simplified and Universal Adhesives on the Dentin Bonding Performance. Appl. Sci. 2019, 10, 124. [Google Scholar] [CrossRef]
- Yoshida, Y.; Yoshihara, K.; Nagaoka, N.; Hayakawa, S.; Torii, Y.; Ogawa, T.; Osaka, A.; Meerbeek, B. Van Self-assembled nano-layering at the adhesive interface. J. Dent. Res. 2012, 91, 376–381. [Google Scholar] [CrossRef]
- Alex, G. Universal adhesives: The next evolution in adhesive dentistry? Compend. Contin. Educ. Dent. 2015, 36, 15–26. [Google Scholar]
- Kaczor-Wiankowska, K.; Lipa, S.; Krasowski, M.; Sokołowski, J.; Lewusz-Butkiewicz, K.; Nowicka, A. Evaluation of gap formation at the composite resin-tooth interface after using universal adhesives: In vitro SEM study using the replica technique. Microsc. Res. Tech. 2020, 83, 176–185. [Google Scholar] [CrossRef]
- Carvalho, A.A.; Leite, M.M.; Zago, J.K.M.; Nunes, C.A.B.C.M.; Barata, T.J.E.; de Freitas, G.C.; de Torres, É.M.; Lopes, L.G. Influence of different application protocols of universal adhesive system on the clinical behavior of Class I and II restorations of composite resin—A randomized and double-blind controlled clinical trial. BMC Oral Health 2019, 19, 252. [Google Scholar] [CrossRef] [PubMed]
- Cuevas-Suárez, C.E.; de Oliveira da Rosa, W.L.; Vitti, R.P.; da Silva, A.F.; Piva, E. Bonding Strength of Universal Adhesives to Indirect Substrates: A Meta-Analysis of in Vitro Studies. J. Prosthodont. 2020, 29, 298–308. [Google Scholar] [CrossRef] [PubMed]
- Nagarkar, S.; Theis-Mahon, N.; Perdigão, J. Universal dental adhesives: Current status, laboratory testing, and clinical performance. J. Biomed. Mater. Res. Part B Appl. Biomater. 2019, 107, 2121–2131. [Google Scholar] [CrossRef] [PubMed]
- Lümkemann, N.; Eichberger, M.; Stawarczyk, B. Different surface modifications combined with universal adhesives: The impact on the bonding properties of zirconia to composite resin cement. Clin. Oral Investig. 2019, 23, 3941–3950. [Google Scholar] [CrossRef]
- Papadogiannis, D.; Dimitriadi, M.; Zafiropoulou, M.; Gaintantzopoulou, M.-D.; Eliades, G. Universal Adhesives: Setting Characteristics and Reactivity with Dentin. Materials 2019, 12, 1720. [Google Scholar] [CrossRef]
- Fukegaw a, D.; Hayakawa, S.; Yoshida, Y.; Suzuki, K.; Osaka, A.; Van Meerbeek, B. Chemical interaction of phosphoric acid ester with hydroxyapatite. J. Dent. Res. 2006, 85, 941–944. [Google Scholar] [CrossRef]
- Van Landuyt, K.L.; Yoshida, Y.; Hirata, I.; Snauwaert, J.; De Munck, J.; Okazaki, M.; Suzuki, K.; Lambrechts, P.; Van Meerbeek, B. Influence of the chemical structure of functional monomers on their adhesive performance. J. Dent. Res. 2008, 87, 757–761. [Google Scholar] [CrossRef]
- Lukomska-Szymanska, M.; Sokolowski, J.; Lapinska, B. Degradation of a hybrid layer—Review of literature. J. Stomatol. 2017, 70, 88–94. [Google Scholar]
- Yoshihara, K.; Yoshida, Y.; Hayakawa, S.; Nagaoka, N.; Kamenoue, S.; Okihara, T.; Ogawa, T.; Nakamura, M.; Osaka, A.; Van Meerbeek, B. Novel fluoro-carbon functional monomer for dental bonding. J. Dent. Res. 2014, 93, 189–194. [Google Scholar] [CrossRef]
- Yoshida, Y.; Nagakane, K.; Fukuda, R.; Nakayama, Y.; Okazaki, M.; Shintani, H.; Inoue, S.; Tagawa, Y.; Suzuki, K.; De Munck, J.; et al. Comparative study on adhesive performance of functional monomers. J. Dent. Res. 2004, 83, 454–458. [Google Scholar] [CrossRef]
- Tsuchimoto, Y.; Yoshida, Y.; Mine, A.; Nakamura, M.; Nishiyama, N.; Van Meerbeek, B.; Suzuki, K.; Kuboki, T. Effect of 4-MET- and 10-MDP-based primers on resin bonding to titanium. Dent. Mater. J. 2006, 25, 120–124. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Ikemura, K.; Kojima, K.; Endo, T.; Kadoma, Y. Effect of the combination of dithiooctanoate monomers and acidic adhesive monomers on adhesion to precious metals, precious metal alloys and non-precious metal alloys. Dent. Mater. J. 2011, 30, 469–477. [Google Scholar] [CrossRef] [PubMed]
- Thompson, J.Y.; Stoner, B.R.; Piascik, J.R.; Smith, R. Adhesion/cementation to zirconia and other non-silicate ceramics: Where are we now? Dent. Mater. 2011, 27, 71–82. [Google Scholar] [CrossRef] [PubMed]
- Llerena-Icochea, A.E.; Costa, R.M.; Borges, A.F.S.; Bombonatti, J.F.S.; Furuse, A.Y. Bonding polycrystalline zirconia with 10-MDP-containing adhesives. Oper. Dent. 2017, 42, 335–341. [Google Scholar] [CrossRef] [PubMed]
- Van Meerbeek, B.; Yoshihara, K.; Yoshida, Y.; Mine, A.; De Munck, J.; Van Landuyt, K.L. State of the art of self-etch adhesives. Dent. Mater. 2011, 27, 17–28. [Google Scholar] [CrossRef] [PubMed]
- Pashley, D.H.; Tay, F.R.; Breschi, L.; Tjäderhane, L.; Carvalho, R.M.; Carrilho, M.; Tezvergil-Mutluay, A. State of the art etch-and-rinse adhesives. Dent. Mater. 2011, 27, 1–16. [Google Scholar] [CrossRef]
- Ergün, G.; Eǧilmez, F.; Üçtaşli, M.B.; Yilmaz, Ş. Effect of light curing type on cytotoxicity of dentine-bonding agents. Int. Endod. J. 2007, 40, 216–223. [Google Scholar] [CrossRef]
- Nowicka, A.; Łagocka, R.; Lipski, M.; Parafiniuk, M.; Grocholewicz, K.; Sobolewska, E.; Witek, A.; Buczkowska-Radlińska, J.; Nowicka, A.; Lagocka, R.; et al. Clinical and Histological Evaluation of Direct Pulp Capping on Human Pulp Tissue Using a Dentin Adhesive System. Biomed Res. Int. 2016, 2016, 2591273. [Google Scholar] [CrossRef]
- Fernandes, A.M.; Silva, G.A.B.; Lopes, N.; Napimoga, M.H.; Benatti, B.B.; Alves, J.B. Direct capping of human pulps with a dentin bonding system and calcium hydroxide: An immunohistochemical analysis. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endodontol. 2008, 105, 385–390. [Google Scholar] [CrossRef]
- Silva, G.A.B.; Gava, E.; Lanza, L.D.; Estrela, C.; Alves, J.B. Subclinical failures of direct pulp capping of human teeth by using a dentin bonding system. J. Endod. 2013, 39, 182–189. [Google Scholar] [CrossRef]
- Lu, Y.; Liu, T.; Li, H.; Pi, G. Histological evaluation of direct pulp capping with a self-etching adhesive and calcium hydroxide on human pulp tissue. Int. Endod. J. 2008, 41, 643–650. [Google Scholar] [CrossRef] [PubMed]
- Paula, A.B.; Laranjo, M.; Marto, C.M.; Paulo, S.; Abrantes, A.M.; Casalta-Lopes, J.; Marques-Ferreira, M.; Botelho, M.F.; Carrilho, E. Direct Pulp Capping: What is the Most Effective Therapy?—Systematic Review and Meta-Analysis. J. Evid. Based. Dent. Pract. 2018, 18, 298–314. [Google Scholar] [CrossRef] [PubMed]
- Pereira, J.C.; Segala, A.D.; Costa, C.A.S. Human pulpal response to direct pulp capping with an adhesive system. Am. J. Dent. 2000, 13, 139–147. [Google Scholar] [PubMed]
- Galler, K.M.; Schweikl, H.; Hiller, K.A.; Cavender, A.C.; Bolay, C.; D’Souza, R.N.; Schmalz, G. TEGDMA reduces mineralization in dental pulp cells. J. Dent. Res. 2011, 90, 257–262. [Google Scholar] [CrossRef]
- Dammaschke, T.; Stratmann, U.; Fischer, R.J.; Sagheri, D.; Schäfer, E. Proliferation of rat molar pulp cells after direct pulp capping with dentine adhesive and calcium hydroxide. Clin. Oral Investig. 2011, 15, 577–587. [Google Scholar] [CrossRef]
- Pagano, S.; Lombardo, G.; Balloni, S.; Bodo, M.; Cianetti, S.; Barbati, A.; Montaseri, A.; Marinucci, L. Cytotoxicity of universal dental adhesive systems: Assessment in vitro assays on human gingival fibroblasts. Toxicol. Vitr. 2019, 60, 252–260. [Google Scholar] [CrossRef]
- Hanks, C.T.; Strawn, S.E.; Watahai, J.C.; Craig, R.G. Cytotoxic Effects of Resin Components on Cultured Mammalian Fibroblasts. J. Dent. Res. 1991, 70, 1450–1455. [Google Scholar] [CrossRef]
- Oilo, G. Biodegradation of dental composites/glass-ionomer cements. Adv. Dent. Res. 1992, 6, 50–54. [Google Scholar] [CrossRef]
- Murray, P.E.; García Godoy, C.; García Godoy, F. How is the biocompatibilty of dental biomaterials evaluated? Med. Oral Patol. Oral Cir. Bucal 2007, 12, E258–E266. [Google Scholar]
- Schmalz, G.; Galler, K.M. Biocompatibility of biomaterials–Lessons learned and considerations for the design of novel materials. Dent. Mater. 2017, 33, 382–393. [Google Scholar] [CrossRef]
- Moharamzadeh, K.; Brooki, I.M.; Van Noortr, R. Biocompatibility of resin-based dental materials. Materials (Basel) 2009, 2, 514–548. [Google Scholar] [CrossRef]
- Ausiello, P.; Cassese, A.; Miele, C.; Beguinot, F.; Garcia-Godoy, F.; Jeso, B.D.; Ulianich, L. Cytotoxicity of dental resin composites: An in vitro evaluation. J. Appl. Toxicol. 2013, 33, 451–457. [Google Scholar] [CrossRef] [PubMed]
- Ergun, G.; Egilmez, F.; Cekic-Nagas, I. The effect of light curing units and modes on cytotoxicity of resin-core systems. Med. Oral Patol. Oral Cir. Bucal 2010, 15, e962-8. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Willershausen, I.; Callaway, A.; Briseño, B.; Willershausen, B. In vitro analysis of the cytotoxicity and the antimicrobial effect of four endodontic sealers. Head Face Med. 2011, 7, 15. [Google Scholar] [CrossRef] [PubMed]
- Baraba, A.; Želježić, D.; Kopjar, N.; Mladinić, M.; Anić, I.; Miletić, I. Evaluation of cytotoxic and genotoxic effects of two resin-based root-canal sealers and their components on human leucocytes in vitro. Int. Endod. J. 2011, 44, 652–661. [Google Scholar] [CrossRef]
- Pawlowska, E.; Poplawski, T.; Ksiazek, D.; Szczepanska, J.; Blasiak, J. Genotoxicity and cytotoxicity of 2-hydroxyethyl methacrylate. Mutat. Res. Genet. Toxicol. Environ. Mutagen. 2010, 696, 122–129. [Google Scholar] [CrossRef]
- Di Pietro, A.; Visalli, G.; La Maestra, S.; Micale, R.; Baluce, B.; Matarese, G.; Cingano, L.; Scoglio, M.E. Biomonitoring of DNA damage in peripheral blood lymphocytes of subjects with dental restorative fillings. Mutat. Res. Genet. Toxicol. Environ. Mutagen. 2008, 650, 115–122. [Google Scholar] [CrossRef]
- ISO/EN10993-5 ISO 10993-5 Biological Evaluation of Medical Devices—Part 5: Tests for Cytotoxicity: In Vitro Methods. Int. Stand. ISO 2009. Available online: https://www.iso.org/standard/36406.html (accessed on 30 May 2020).
- Bushnell, P.J.; Kavlock, R.J.; Crofton, K.M.; Weiss, B.; Rice, D.C. Behavioral toxicology in the 21st century: Challenges and opportunities for behavioral scientists. Summary of a symposium presented at the annual meeting of the Neurobehavioral Teratology Society, June, 2009. Neurotoxicol. Teratol. 2010, 32, 313–328. [Google Scholar] [CrossRef]
- Tuncer, S.; Demirci, M.; Schweikl, H.; Erguven, M.; Bilir, A.; Kara Tuncer, A. Inhibition of cell survival, viability and proliferation by dentin adhesives after direct and indirect exposure in vitro. Clin. Oral Investig. 2012, 16, 1635–1646. [Google Scholar] [CrossRef]
- Lee, Y.; An, S.-Y.Y.; Park, Y.-J.J.; Yu, F.H.; Park, J.-C.C.; Seo, D.-G.G. Cytotoxic effects of one-step self-etching adhesives on an odontoblast cell line. Scanning 2016, 38, 36–42. [Google Scholar] [CrossRef]
- Bianchi, L.; Ribeiro, A.P.D.; De Oliveira Carrilho, M.R.; Pashley, D.H.; De Souza Costa, C.A.; Hebling, J. Cytotoxicity of adhesive systems of different hydrophilicities on cultured odontoblast-like cells. J. Biomed. Mater. Res. Part B Appl. Biomater. 2013, 101, 1498–1507. [Google Scholar] [CrossRef] [PubMed]
- Bianchi, L.; Ribeiro, A.P.D.; De Oliveira Carrilho, M.R.; Pashley, D.H.; De Souza Costa, C.A.; Hebling, J. Transdentinal cytotoxicity of experimental adhesive systems of different hydrophilicity applied to ethanol-saturated dentin. Dent. Mater. 2013, 29, 980–990. [Google Scholar] [CrossRef] [PubMed]
- Lukomska-Szymanska, M.; Konieczka, M.; Zarzycka, B.; Lapinska, B.; Grzegorczyk, J.; Sokolowski, J. Antibacterial activity of commercial dentine bonding systems against E. faecalis-flow cytometry study. Materials 2017, 10, 481. [Google Scholar] [CrossRef] [PubMed]
- Lapinska, B.; Konieczka, M.; Zarzycka, B.; Sokolowski, K.; Grzegorczyk, J.; Lukomska-Szymanska, M. Flow cytometry analysis of antibacterial effects of universal dentin bonding agents on streptococcus mutans. Molecules 2019, 24, 532. [Google Scholar] [CrossRef]
- Collins, A.R. The comet assay for DNA damage and repair: Principles, applications, and limitations. Appl. Biochem. Biotechnol. Part B Mol. Biotechnol. 2004, 26, 249–261. [Google Scholar] [CrossRef]
- Tice, R.R.; Agurell, E.; Anderson, D.; Burlinson, B.; Hartmann, A.; Kobayashi, H.; Miyamae, Y.; Rojas, E.; Ryu, J.C.; Sasaki, Y.F. Single cell gel/comet assay: Guidelines for in vitro and in vivo genetic toxicology testing. Environ. Mol. Mutagen. 2000, 35, 206–221. [Google Scholar] [CrossRef]
- Kitasako, Y.; Arakawa, M.; Sonoda, H.; Tagami, J. Light and scanning electron microscopy of the inner surfaces of resins used in direct pulp capping. Am. J. Dent. 1999, 12, 217–221. [Google Scholar]
- Koulaouzidou, E.A.; Helvatjoglu-Antoniades, M.; Palaghias, G.; Karanika-Kouma, A.; Antoniades, D. Cytotoxicity of Dental Adhesives In Vitro. Eur. J. Dent. 2009, 3, 3–9. [Google Scholar] [CrossRef]
- Caldas, I.P.; Alves, G.G.; Barbosa, I.B.; Scelza, P.; de Noronha, F.; Scelza, M.Z. In vitro cytotoxicity of dental adhesives: A systematic review. Dent. Mater. 2019, 35, 195–205. [Google Scholar] [CrossRef]
- Williams, D.F. On the mechanisms of biocompatibility. Biomaterials 2008, 29, 2941–2953. [Google Scholar] [CrossRef]
- Loison-Robert, L.S.; Tassin, M.; Bonte, E.; Berbar, T.; Isaac, J.; Berdal, A.; Simon, S.; Fournier, B.P.J. In vitro effects of two silicate-based materials, Biodentine and BioRoot RCS, on dental pulp stem cells in models of reactionary and reparative dentinogenesis. PLoS ONE 2018, 13, e0190014. [Google Scholar] [CrossRef]
- Wegehaupt, F.J.; Lunghi, N.; Belibasakis, G.N.; Attin, T. Influence of light-curing distance on degree of conversion and cytotoxicity of etch-and-rinse and self-etch adhesives. BMC Oral Health 2016, 17, 12. [Google Scholar] [CrossRef]
- Almaroof, A.; Niazi, S.A.; Rojo, L.; Mannocci, F.; Deb, S. Evaluation of dental adhesive systems incorporating an antibacterial monomer eugenyl methacrylate (EgMA) for endodontic restorations. Dent. Mater. 2017, 33, e239–e254. [Google Scholar] [CrossRef]
- Jiang, R.D.; Lin, H.; Zheng, G.; Zhang, X.M.; Du, Q.; Yang, M. In vitro dentin barrier cytotoxicity testing of some dental restorative materials. J. Dent. 2017, 58, 28–33. [Google Scholar] [CrossRef]
- Lanza, C.R.M.; De Souza Costa, C.A.; Furlan, M.; Alécio, A.; Hebling, J. Transdentinal diffusion and cytotoxicity of self-etching adhesive systems. Cell Biol. Toxicol. 2009, 25, 533–543. [Google Scholar] [CrossRef]
- Cavalcanti, B.N.; Marques, M.M. Cytotoxicity of substances leached from a conventional and a self-etching adhesive system on human pulp fibroblasts. Brazilian Dent. Sci. 2011, 13, 10–14. [Google Scholar] [CrossRef]
- Rajić, V.B.; Želježić, D.; Ivanišević, A.M.; Verzak, Ž.; Baraba, A.; Miletić, I. Cytotoxicity and genotoxicity of resin based dental materials in human lymphocytes in vitro. Acta Clin. Croat. 2018, 57, 278–285. [Google Scholar]
- Huang, F.M.; Tai, K.W.; Chou, M.Y.; Chang, Y.C. Cytotoxicity of resin-, zinc oxide-eugenol-, and calcium hydroxide-based root canal sealers on human periodontal ligament cells and permanent V79 cells. Int. Endod. J. 2002, 35, 153–158. [Google Scholar] [CrossRef]
- Tadin, A.; Galic, N.; Zeljezic, D.; Mikelic Vitasovic, B.; Marovic, D.; Kovacic, I. Ex vivo evaluation of genotoxic effects of four dental adhesives on human leukocytes. J. Dent. Sci. 2013, 8, 37–43. [Google Scholar] [CrossRef][Green Version]
- Sideridou, I.D.; Achilias, D.S. Elution study of unreacted Bis-GMA, TEGDMA, UDMA, and Bis-EMA from light-cured dental resins and resin composites using HPLC. J. Biomed. Mater. Res. Part B Appl. Biomater. 2005, 74, 617–626. [Google Scholar] [CrossRef]
- Geurtsen, W.; Lehmann, F.; Spahl, W.; Leyhausen, G. Cytotoxicity of 35 dental resin composite monomers/additives in permanent 3T3 and three human primary fibroblast cultures. J. Biomed. Mater. Res. 1998, 41, 474–480. [Google Scholar] [CrossRef]
- Koulaouzidou, E.A.; Helvatjoglu-Antoniades, M.; Palaghias, G.; Karanika-Kouma, A.; Antoniades, D. Cytotoxicity evaluation of an antibacterial dentin adhesive system on established cell lines. J. Biomed. Mater. Res. Part B Appl. Biomater. 2008, 84, 271–276. [Google Scholar] [CrossRef] [PubMed]
- Huang, F.M.; Li, Y.C.; Lee, S.S.; Chang, Y.C. Cytotoxicity of dentine bonding agents on human pulp cells is related to intracellular glutathione levels. Int. Endod. J. 2010, 43, 1091–1097. [Google Scholar] [CrossRef] [PubMed]
- Kusdemir, M.; Gunal, S.; Ozer, F.; Imazato, S.; Izutani, N.; Ebisu, S.; Blatz, M.B. Evaluation of cytotoxic effects of six self-etching adhesives with direct and indirect contact tests. Dent. Mater. J. 2011, 30, 799–805. [Google Scholar] [CrossRef]
- Volk, J.; Ziemann, C.; Leyhausen, G.; Geurtsen, W. Non-irradiated campherquinone induces DNA damage in human gingival fibroblasts. Dent. Mater. 2009, 25, 1556–1563. [Google Scholar] [CrossRef]
- Yeh, H.W.; Chang, M.C.; Lin, C.C.P.; Tseng, W.Y.; Chang, H.H.; Wang, T.M.; Chen, Y.J.; Lin, C.C.P.; Yang, T.T.; Lin, L.D.; et al. Comparative cytotoxicity of five current dentin bonding agents: Role of cell cycle deregulation. Acta Biomater. 2009, 5, 3404–3410. [Google Scholar] [CrossRef]
- Huang, F.M.; Chang, Y.C. Cytotoxicity of dentine-bonding agents on human pulp cells in vitro. Int. Endod. J. 2002, 35, 905–909. [Google Scholar] [CrossRef]
- Kostoryz, E.L.; Eick, J.D.; Chappelow, C.C.; Glaros, A.G.; Wetmore, L.; Yourtee, D.M. In vitro effect of light-cure dental adhesive on IL-6 release from LPS-stimulated and unstimulated macrophages. J. Biomed. Mater. Res. 2003, 65, 89–94. [Google Scholar] [CrossRef]
- Gerzina, T.M.; Hume, W.R. Diffusion of monomers from bonding resin-resin composite combinations through dentine in vitro. J. Dent. 1996, 24, 125–128. [Google Scholar] [CrossRef]
- Spagnuolo, G.; Mauro, C.; Leonardi, A.; Santillo, M.; Paternò, R.; Schweikl, H.; Avvedimento, E.V.; Rengo, S. NF-κB protection against apoptosis induced by HEMA. J. Dent. Res. 2004, 83, 837–842. [Google Scholar] [CrossRef]
- Altintas, S.H.; Usumez, A. Evaluation of monomer leaching from a dual cured resin cement. J. Biomed. Mater. Res. Part B Appl. Biomater. 2008, 86, 523–529. [Google Scholar] [CrossRef]
- Bouillaguet, S.; Wataha, J.C.; Hanks, C.T.; Ciucchi, B.; Holz, J. In vitro cytotoxicity and dentin permeability of HEMA. J. Endod. 1996, 22, 244–248. [Google Scholar] [CrossRef]
- About, I.; Camps, J.; Mitsiadis, T.A.; Bottero, M.J.; Butler, W.; Franquin, J.C. Influence of resinous monomers on the differentiation in vitro of human pulp cells into odontoblasts. J. Biomed. Mater. Res. 2002, 63, 418–423. [Google Scholar] [CrossRef]
- da Silva, J.M.F.; Rodrigues, J.R.; Camargo, C.H.R.; Fernandes, V.V.B.; Hiller, K.A.; Schweikl, H.; Schmalz, G. Effectiveness and biological compatibility of different generations of dentin adhesives. Clin. Oral Investig. 2014, 18, 607–613. [Google Scholar] [CrossRef]
- Schweikl, H.; Spagnuolo, G.; Schmalz, G. Genetic and cellular toxicology of dental resin monomers. J. Dent. Res. 2006, 85, 870–877. [Google Scholar] [CrossRef]
- Mavrogonatou, E.; Eliades, T.; Eliades, G.; Kletsas, D. The effect of triethylene glycol dimethacrylate on p53-dependent G2 arrest in human gingival fibroblasts. Biomaterials 2010, 31, 8530–8538. [Google Scholar] [CrossRef]
- Elias, S.T.; dos Santos, A.F.; Garcia, F.C.P.; Pereira, P.N.R.; Hilgert, L.A.; Fonseca-Bazzo, Y.M.; Guerra, E.N.S.; Ribeiro, A.P.D. Cytotoxicity of universal, self-etching and etch-and-rinse adhesive systems according to the polymerization time. Braz. Dent. J. 2015, 26, 160–168. [Google Scholar] [CrossRef]
- Ruschel, V.C.; Stolf, S.C.; Shibata, S.; Chung, Y.; Boushell, L.W.; Baratieri, L.N.; Walter, R. Three-year clinical evaluation of universal adhesives in non-carious cervical lesions. Am. J. Dent. 2019, 32, 223–228. [Google Scholar]
- Wang, R.; Shi, Y.; Li, T.; Pan, Y.; Cui, Y.; Xia, W. Adhesive interfacial characteristics and the related bonding performance of four self-etching adhesives with different functional monomers applied to dentin. J. Dent. 2017, 62, 72–80. [Google Scholar] [CrossRef]
| Name | Manufacturer | Composition |
|---|---|---|
| OptiBond Universal | Kerr, Brea, CA, USA | Acetone (30–60%), HEMA (5–10%), glycerol dimethacrylate (1–5%), ethanol (5–10%) |
| Prime&Bond Universal | Dentsply Sirona, Charlotte, NC, USA | Phosphoric acid modified acrylate resin, multifunctional acrylate, bifunctional acrylate, acidic acrylate, isopropanol, water, initiator |
| Adhese Universal | Ivoclar Vivadent, Schaan, Liechtenstein | MDP, 3–10%, MCAP methacrylated carboxylic acid polymer, HEMA (10–25%), Bis-GMA (10–25%), D3MA (3–10%), 2-dimethylaminoethyl methacrylate (1–2.5%), camphorquinone (1–2.5%), ethanol (10–25%) |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wawrzynkiewicz, A.; Rozpedek-Kaminska, W.; Galita, G.; Lukomska-Szymanska, M.; Lapinska, B.; Sokolowski, J.; Majsterek, I. The Cytotoxicity and Genotoxicity of Three Dental Universal Adhesives—An In Vitro Study. Int. J. Mol. Sci. 2020, 21, 3950. https://doi.org/10.3390/ijms21113950
Wawrzynkiewicz A, Rozpedek-Kaminska W, Galita G, Lukomska-Szymanska M, Lapinska B, Sokolowski J, Majsterek I. The Cytotoxicity and Genotoxicity of Three Dental Universal Adhesives—An In Vitro Study. International Journal of Molecular Sciences. 2020; 21(11):3950. https://doi.org/10.3390/ijms21113950
Chicago/Turabian StyleWawrzynkiewicz, Adam, Wioletta Rozpedek-Kaminska, Grzegorz Galita, Monika Lukomska-Szymanska, Barbara Lapinska, Jerzy Sokolowski, and Ireneusz Majsterek. 2020. "The Cytotoxicity and Genotoxicity of Three Dental Universal Adhesives—An In Vitro Study" International Journal of Molecular Sciences 21, no. 11: 3950. https://doi.org/10.3390/ijms21113950
APA StyleWawrzynkiewicz, A., Rozpedek-Kaminska, W., Galita, G., Lukomska-Szymanska, M., Lapinska, B., Sokolowski, J., & Majsterek, I. (2020). The Cytotoxicity and Genotoxicity of Three Dental Universal Adhesives—An In Vitro Study. International Journal of Molecular Sciences, 21(11), 3950. https://doi.org/10.3390/ijms21113950






