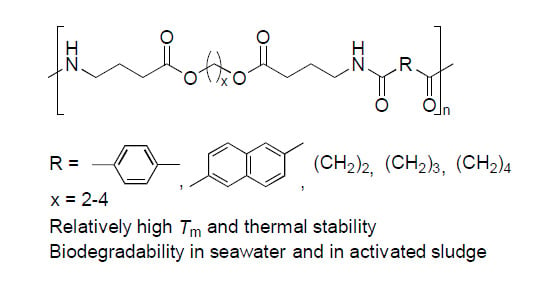
Synthesis, Properties, and Biodegradation of Sequential Poly(Ester Amide)s Containing γ-Aminobutyric Acid
Abstract
1. Introduction
2. Results
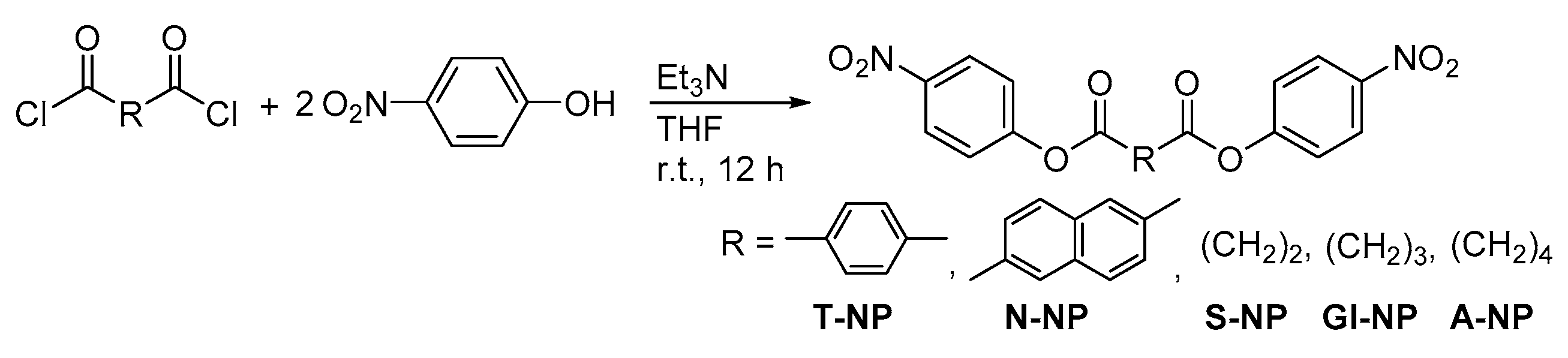
2.1. Synthesis of the Sequential Poly(Ester Amide)s
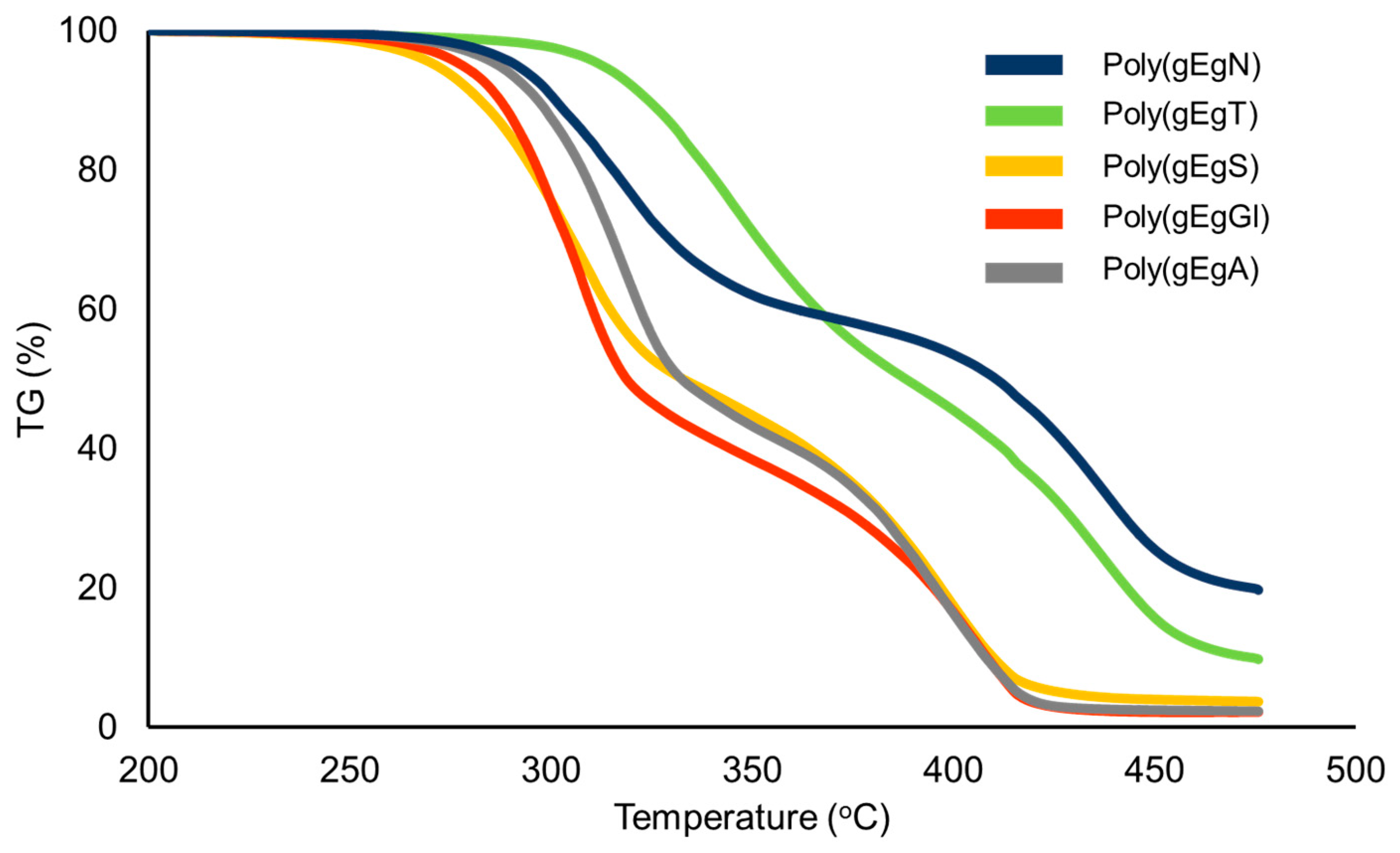
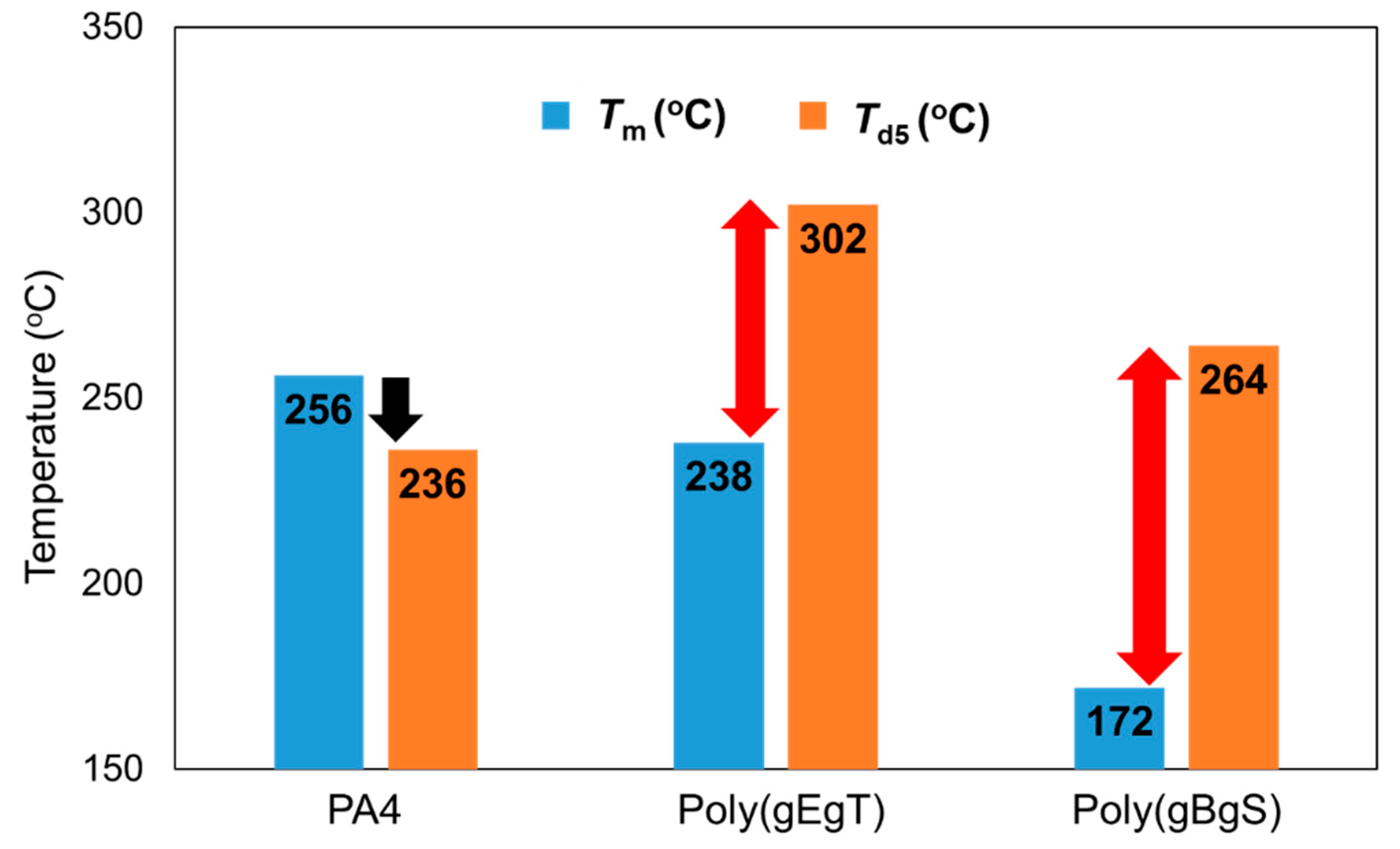
2.2. Thermal Properties of the Poly(Ester Amide)s Containing GABA Unit
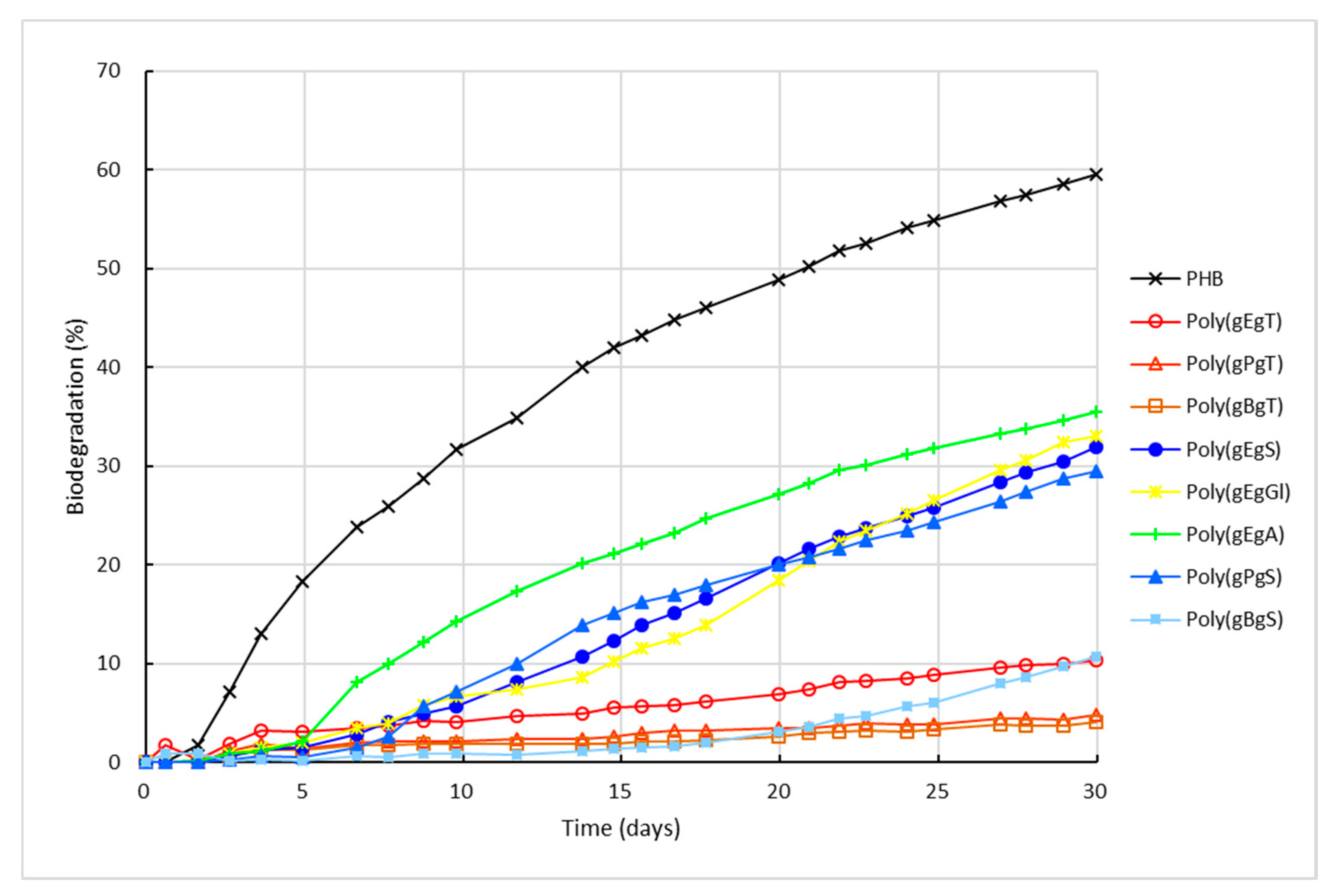
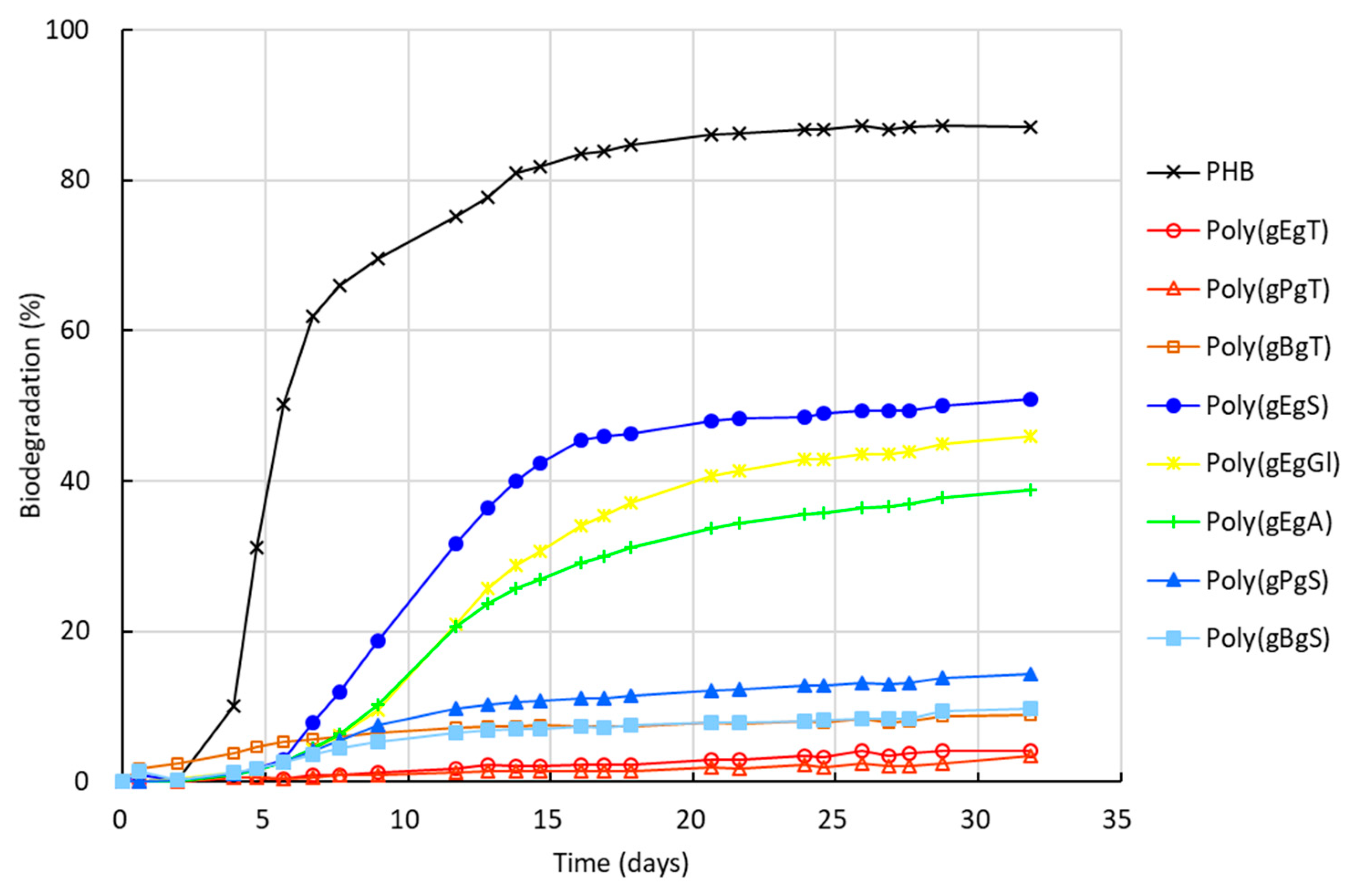
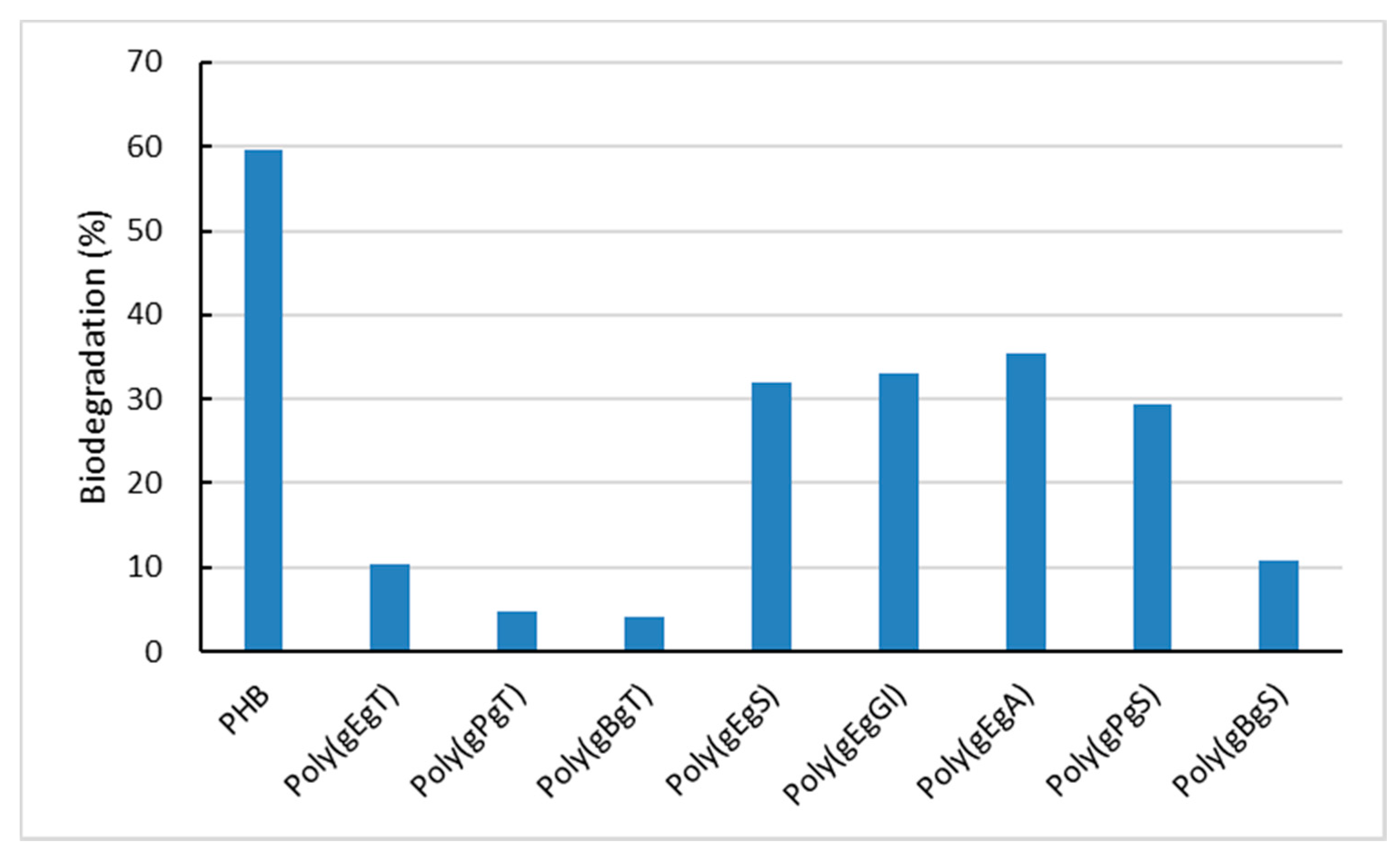
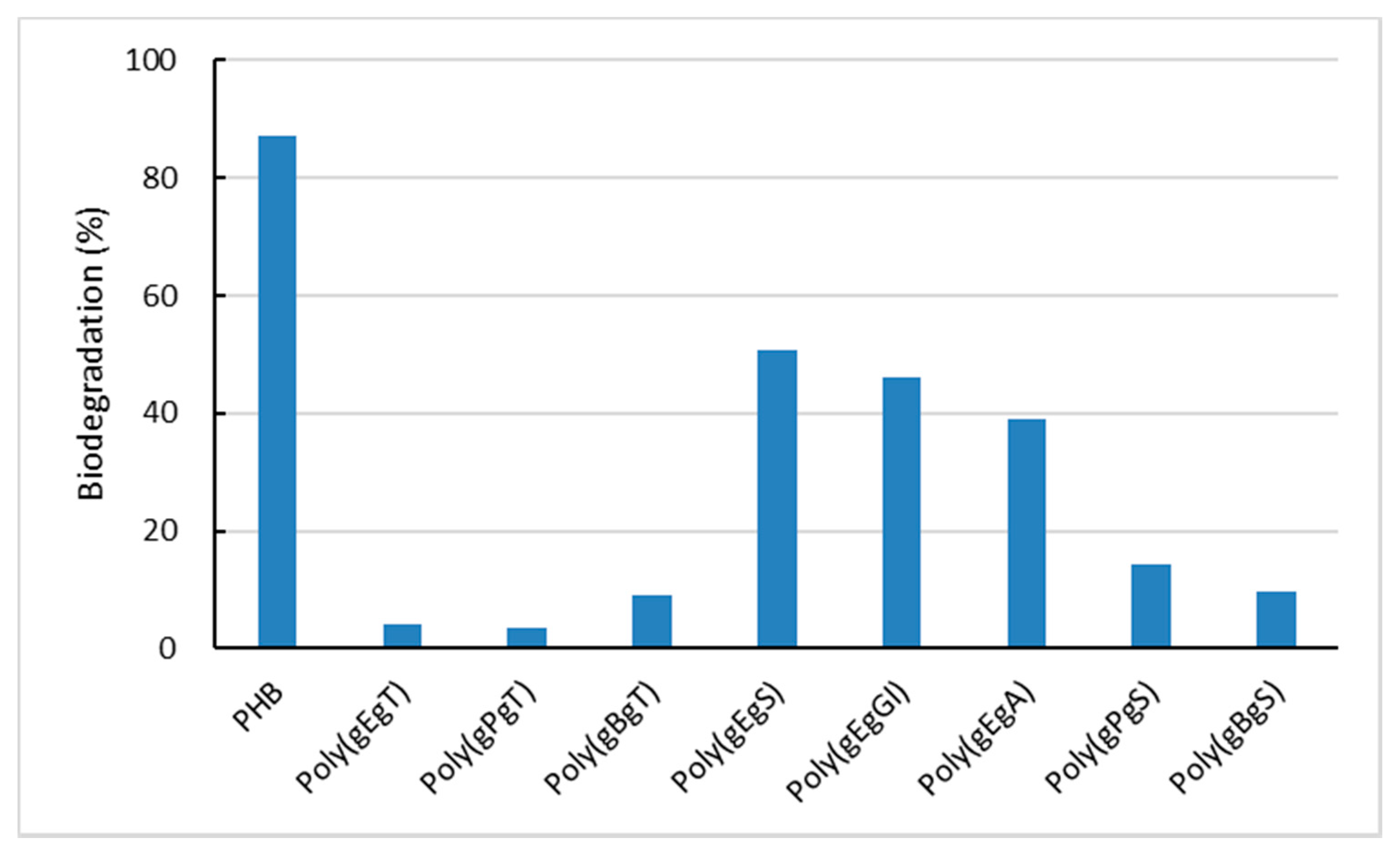
2.3. Biodegradation of the Poly(Ester Amide)s Containing GABA Unit
3. Discussion
4. Materials and Methods
4.1. Chemicals
4.2. Measurements
4.3. Synthesis of Ditosylates of Diesterdiamines
4.4. Synthesis of Sequential Poly(Ester Amide)s with GABA Unit
4.5. Biodegradation Tests of the Poly(Ester Amide)s in Seawater and in Activated Sludge
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| GABA | γ-Aminobutyric acid |
| PA4 | Polyamide 4 |
| PHA | Poly(3-hydroxyalkanoate) |
| PLLA | Poly(L-lactic acid) |
| PGA | Poly(glycolic acid) |
| PCL | Poly(ε-caprolactone) |
| PBS | Poly(butylene succinate) |
| PET | Poly(butylene succinate) |
| Tm | Melting temperature |
| Tg | Glass-transition temperature |
| TsOH·H2O | p-Toluenesulfonic acid monohydrate |
| gEg-OTs | Ditosylate of ethylene bis(γ-aminobutyrate) |
| gPg-OTs | Ditosylate of trimethylene bis(γ-aminobutyrate) |
| gBg-OTs | Ditosylate of butylene bis(γ-aminobutyrate) |
| T-NP | Bis(p-nitrophenyl) terephthalate |
| N-NP | Bis(p-nitrophenyl) 2.6-naphthalenedicarboxylate |
| S-NP | Bis(p-nitrophenyl) succinate |
| Gl-NP | Bis(p-nitrophenyl) glutarate |
| A-NP | Bis(p-nitrophenyl) adipate |
| DMSO | Dimethyl sulfoxide |
| THF | Tetrahydrofuran |
| HFIP | 1,1,1,3,3,3-Hexafluoro-2-propanol |
| GPC | Gel permeation chromatography |
| PMMA | Poly(methyl methacrylate) |
| Mn | Number-averaged molecular weight |
| Mw | Weight-averaged molecular weight |
| Poly(gEgT) | Poly((ethylene bis(γ-aminobutyrate)) terephthalate) |
| Poly(gPgT) | Poly((trimethylene bis(γ-aminobutyrate)) terephthalate) |
| Poly(gBgT) | Poly((butylene bis(γ-aminobutyrate)) terephthalate) |
| Poly(gEgN) | Poly((ethylene bis(γ-aminobutyrate)) 2.6-naphthalenedicarboxylate) |
| Poly(gPgN) | Poly((trimethylene bis(γ-aminobutyrate)) 2.6-naphthalenedicarboxylate) |
| Poly(gBgN) | Poly((butylene bis(γ-aminobutyrate)) 2.6-naphthalenedicarboxylate) |
| Poly(gEgS) | Poly((ethylene bis(γ-aminobutyrate)) succinate) |
| Poly(gPgS) | Poly((trimethylene bis(γ-aminobutyrate)) succinate) |
| Poly(gBgS) | Poly((butylene bis(γ-aminobutyrate)) succinate) |
| Poly(gEgGl) | Poly((ethylene bis(γ-aminobutyrate)) glutarate) |
| Poly(gPgGl) | Poly((trimethylene bis(γ-aminobutyrate)) glutarate) |
| Poly(gBgGl) | Poly((butylene bis(γ-aminobutyrate)) glutarate) |
| Poly(gEgA) | Poly((ethylene bis(γ-aminobutyrate)) adipate) |
| Poly(gPgA) | Poly((trimethylene bis(γ-aminobutyrate)) adipate) |
| Poly(gBgA) | Poly((butylene bis(γ-aminobutyrate)) adipate) |
| DSC | Differential scanning calorimetry |
| TG | Thermogravimetric analysis |
| Td5 | Degradation temperatures at 5% weight loss |
| Td10 | Degradation temperatures at 10% weight loss |
| PHB | Poly(3-hydroxybutyrate) |
| PES | Poly(ethylene succinate) |
| AIST | National Institute of Advanced Industrial Science and Technology |
References
- Mallapragada, S.K.; Narasimhan, B. Handbook of Biodegradable Polymeric Materials and Their Applications; American Scientific Publishers: Stevenson Ranch, CA, USA, 2005. [Google Scholar]
- Narancic, T.; Verstichel, S.; Chaganti, S.R.; Morales-Gamez, L.; Kenny, S.T.; Wilde, B.D.; Padamati, R.B.; O’Connor, K.E. Biodegradable Plastic Blends Create New Possibilities for End-of-Life Management of Plastics but They Are Not a Panacea for Plastic Pollution. Environ. Sci. Technol. 2018, 52, 10441–10452. [Google Scholar] [CrossRef]
- Haider, T.P.; Völker, C.; Kramm, J.; Landfester, K.; Wurm, F.R. Plastics of the Future? The Impact of Biodegradable Polymers on the Environment and on Society. Angew. Chem. Int. Ed. 2019, 58, 50–62. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, S. Biodegradable Polymers: Present Opportunities and Challenges in Providing a Microplastic-Free Environment. Macromol. Chem. Phys. 2020, 221, 2000017. [Google Scholar] [CrossRef]
- Jambeck, J.R.; Geyer, R.; Wilcox, C.; Siegler, T.R.; Perryman, M.; Andrady, A.; Narayan, R.; Law, K.L. Plastic waste inputs from land into the ocean. Science 2015, 347, 768–771. [Google Scholar] [CrossRef] [PubMed]
- Lönnstedt, O.M.; Eklöv, P. Environmentally relevant concentrations of microplastic particles influence larval fish ecology. Science 2016, 352, 1213–1216. [Google Scholar] [CrossRef]
- Isobe, A.; Iwasaki, S.; Uchida, K.; Tokai, T. Abundance of non-conservative microplastics in the upper ocean from 1957 to 2066. Nat. Commun. 2019, 10, 417. [Google Scholar] [CrossRef]
- Volova, T.G.; Boyandin, A.N.; Vasiliev, A.D.; Karpov, V.A.; Prudnikova, S.V.; Mishukova, O.V.; Boyarskikh, U.A.; Filipenko, M.L.; Rudnev, V.P.; Xuân, B.B.; et al. Biodegradation of polyhydroxyalkanoates (PHAs) in tropical coastal waters and identification of PHA-degrading bacteria. Polym. Degrad. Stab. 2010, 95, 2350–2359. [Google Scholar] [CrossRef]
- Bagheri, A.R.; Laforsch, C.; Greiner, A.; Agarwal, S. Fate of So-Called Biodegradable Polymers in Seawater and Freshwater. Glob. Chall. 2017, 1, 1700048. [Google Scholar] [CrossRef]
- Nakayama, A.; Yamano, N.; Kawasaki, N. Biodegradation in seawater of aliphatic polyesters. Polym. Degrad. Stab. 2019, 166, 290–299. [Google Scholar] [CrossRef]
- Yoshida, S.; Hiraga, K.; Takehana, T.; Taniguchi, I.; Yamaji, H.; Maeda, Y.; Toyohara, K.; Miyamoto, K.; Kimura, Y.; Oda, K. A bacterium that degrades and assimilates poly(ethylene terephthalate). Science 2016, 351, 1196–1199. [Google Scholar] [CrossRef]
- Nakayama, Y.; Yagumo, W.; Tanaka, R.; Shiono, T.; Inumaru, K.; Tsutsumi, C.; Kawasaki, N.; Yamano, N.; Nakayama, A. Synthesis, properties and biodegradation of periodic copolyesters composed of hydroxy acids, ethylene glycol, and terephthalic acid. Polym. Degrad. Stab. 2020, 174, 109095. [Google Scholar] [CrossRef]
- Hashimoto, S.; Yamane, K.; Wakabayashi, J.; Matsui, H. Filament of Polyglycolic Acid Resin and Process for Producing the Same. U.S. Patent 2010/0148391 A1, 17 June 2010. [Google Scholar]
- Tsuji, H.; Suzuyoshi, K. Environmental degradation of biodegradable polyesters 1. Poly(ε-caprolactone), poly[(R)-3-hydroxybutyrate], and poly(L-lactide) films in controlled static seawater. Polym. Degrad. Stab. 2002, 75, 347–355. [Google Scholar] [CrossRef]
- Krasowska, K.; Brzeska, J.; Rutkowska, M.; Dacko, P.; Sobota, M.; Kowalczuk, M. The effect of poly(D,L-lactide) modification with poli[(R,S)-3-hydroxybutyrate] on the course of its degradation in natural environments. Polimery 2008, 53, 730–736. [Google Scholar] [CrossRef]
- Chen, X.; Wang, L.; Shi, J.; Shi, H.; Liu, Y. Environmental degradation of starch/poly(lactic acid) composite in seawater. Polym. Polym. Compos. 2011, 19, 559–565. [Google Scholar] [CrossRef]
- Yamano, N.; Kawasaki, N.; Takeda, S.; Nakayama, A. Production of 2-Pyrrolidone from Biobased Glutamate by Using Escherichia coli. J. Polym. Environ. 2013, 21, 528–533. [Google Scholar] [CrossRef]
- Hashimoto, K.; Hamano, T.; Okada, M. Degradation of several polyamides in soils. J. Appl. Polym. Sci. 1994, 54, 1579–1583. [Google Scholar] [CrossRef]
- Kawasaki, N.; Nakayama, A.; Yamano, N.; Takeda, S.; Kawata, Y.; Yamamoto, N.; Aiba, S.-I. Synthesis, thermal and mechanical properties and biodegradation of branched polyamide 4. Polymer 2005, 46, 9987–9993. [Google Scholar] [CrossRef]
- Yamano, N.; Nakayama, A.; Kawasaki, N.; Yamamoto, N.; Aiba, S. Mechanism and Characterization of Polyamide 4 Degradation by Pseudomonas sp. J. Polym. Environ. 2008, 16, 141–146. [Google Scholar] [CrossRef]
- Tachibana, K.; Hashimoto, K.; Yoshikawa, M.; Okawa, H. Isolation and characterization of microorganisms degrading nylon 4 in the composted soil. Polym. Degrad. Stab. 2010, 95, 912–917. [Google Scholar] [CrossRef]
- Tachibana, K.; Urano, Y.; Numata, K. Biodegradability of nylon 4 film in a marine environment. Polym. Degrad. Stab. 2013, 98, 1847–1851. [Google Scholar] [CrossRef]
- Yamano, N.; Kawasaki, N.; Ida, S.; Nakayama, Y.; Nakayama, A. Biodegradation of polyamide 4 in vivo. Polym. Degrad. Stab. 2017, 137, 281–288. [Google Scholar] [CrossRef]
- Tachibana, K.; Hashimoto, K.; Tansho, N.; Okawa, H. Chemical modification of chain end in nylon 4 and improvement of its thermal stability. J. Polym. Sci. Part A Polym. Chem. 2011, 49, 2495–2503. [Google Scholar] [CrossRef]
- Goodman, I.; Sheahan, R.J. Copolyesteramides—V. Hexamethylene adipamide/hexamethylene adipate random and ordered copolymers: Preparation and general properties. Eur. Polym. J. 1990, 26, 1081–1088. [Google Scholar] [CrossRef]
- Murthy, N.S.; Aharoni, S.M. Structural studies of liquid-crystalline poly(ester amides). Macromolecules 1992, 25, 1177–1183. [Google Scholar] [CrossRef]
- Pivsa-Art, S.; Nakayama, A.; Kawasaki, N.; Yamamoto, N.; Aiba, S. Biodegradability study of copolyesteramides based on diacid chlorides, diamines, and diols. J. Appl. Polym. Sci. 2002, 85, 774–784. [Google Scholar] [CrossRef]
- Puiggalí, J.; Subirana, J.A. Synthetic polymers containing α-amino acids: From polyamides to poly(ester amide)s. J. Pept. Sci. 2005, 11, 247–249. [Google Scholar] [CrossRef]
- Gomurashvili, Z.; Zhang, H.; Da, J.; Jenkins, T.D.; Hughes, J.; Wu, M.; Lambert, L.; Grako, K.A.; DeFife, K.M.; MacPherson, K.; et al. From Drug-Eluting Stents to Biopharmaceuticals: Poly(ester amide) a Versatile New Bioabsorbable Biopolymer. In Polymers for Biomedical Applications; ACS Publications: Washington, DC, USA, 2008; pp. 10–26. [Google Scholar]
- Fonseca, A.C.; Gil, M.H.; Simões, P.N. Biodegradable poly(ester amide)s—A remarkable opportunity for the biomedical area: Review on the synthesis, characterization and applications. Prog. Polym. Sci. 2014, 39, 1291–1311. [Google Scholar] [CrossRef]
- Díaz, A.; Katsarava, R.; Puiggalí, J. Synthesis, Properties and Applications of Biodegradable Polymers Derived from Diols and Dicarboxylic Acids: From Polyesters to Poly(ester amide)s. Int. J. Mol. Sci. 2014, 15, 7064–7123. [Google Scholar] [CrossRef]
- Ghosal, K.; Latha, M.S.; Thomas, S. Poly(ester amides) (PEAs)—Scaffold for tissue engineering applications. Eur. Polym. J. 2014, 60, 58–68. [Google Scholar] [CrossRef]
- Winnacker, M.; Rieger, B. Poly(ester amide)s: Recent insights into synthesis, stability and biomedical applications. Polym. Chem. 2016, 7, 7039–7046. [Google Scholar] [CrossRef]
- NgaNguyen, T.H.; Balligand, F.; Bormann, A.; Bennevault, V.; Guégan, P. Synthesis of new biobased linear poly(ester amide)s. Eur. Polym. J. 2019, 121, 109314. [Google Scholar] [CrossRef]
- Grigat, E.; Koch, R.; Timmermann, R. BAR 1095 and BAK 2195: Completely biodegradable synthetic thermoplastics. Polym. Degrad. Stab. 1998, 59, 223–226. [Google Scholar] [CrossRef]
- Asín, L.; Armelin, E.; Montané, J.; Rodríguez-Galán, A.; Puiggalí, J. Sequential poly(ester amide)s based on glycine, diols, and dicarboxylic acids: Thermal polyesterification versus interfacial polyamidation. Characterization of polymers containing stiff units. J. Polym. Sci. Part A Polym. Chem. 2001, 39, 4283–4293. [Google Scholar] [CrossRef]
- Yamano, N.; Kawasaki, N.; Ida, S.; Nakayama, A. Biodegradation of polyamide 4 in seawater. Polym. Degrad. Stab. 2019, 166, 230–236. [Google Scholar] [CrossRef]
- Lutz, J.-F.; Ouchi, M.; Liu, D.R.; Sawamoto, M. Sequence-Controlled Polymers. Science 2013, 341, 1238149. [Google Scholar] [CrossRef] [PubMed]
- Shaffer, T.D.; Canich, J.A.M.; Squire, K.R. Metallocene-Catalyzed Copolymerization of Ethylene and Isobutylene to Substantially Alternating Copolymers. Macromolecules 1998, 31, 5145–5147. [Google Scholar] [CrossRef]
- Katsarava, R.; Beridze, V.; Arabuli, N.; Kharadze, D.; Chu, C.C.; Won, C.Y. Amino acid-based bioanalogous polymers. Synthesis, and study of regular poly(ester amide)s based on bis(α-amino acid) α,ω-alkylene diesters, and aliphatic dicarboxylic acids. J. Polym. Sci. Part A Polym. Chem. 1999, 37, 391–407. [Google Scholar] [CrossRef]
- Atkins, K.M.; Lopez, D.; Knight, D.K.; Mequanint, K.; Gillies, E.R. A versatile approach for the syntheses of poly(ester amide)s with pendant functional groups. J. Polym. Sci. Part A Polym. Chem. 2009, 47, 3757–3772. [Google Scholar] [CrossRef]
- Yamada, Y.; Yoshida, M. Preparation of Diamine Derivatives Having Naphthalene Skeleton. JP Patent 1991-279350, 10 December 1991. [Google Scholar]
- Ashley, J.N.; Collins, R.F.; Davis, M.; Sirett, N.E. 673. The chemotherapy of schistosomiasis. Part I. Derivatives and analogues of αω-di-(p-aminophenoxy)alkanes. J. Chem. Soc. 1958, 3298–3313. [Google Scholar] [CrossRef]
- Arabuli, N.; Tsitlanadze, G.; Edilashvili, L.; Kharadze, D.; Goguadze, T.; Beridze, V.; Gomurashvili, Z.; Katsarava, R. Heterochain polymers based on natural amino acids. Synthesis and enzymatic hydrolysis of regular poly(ester amide)s based on bis(L-phenylalanine) α,ω-alkylene diesters and adipic acid. Macromol. Chem. Phys. 1994, 195, 2279–2289. [Google Scholar] [CrossRef]
- Hill, R.; Walker, E.E. Polymer constitution and fiber properties. J. Polym. Sci. Part A Gen. Pap. 1948, 3, 609–630. [Google Scholar] [CrossRef]
- Bunn, C.W. The melting points of chain polymers. J. Polym. Sci. 1955, 16, 323–343. [Google Scholar] [CrossRef]
- Brandrup, J.; Immergut, E.H.; Grulke, E.A. Polymer Handbook, 4th ed.; John Wiley & Sons: New York, NY, USA, 2003. [Google Scholar]
- Jin, X.; Ellis, T.S.; Karasz, F.E. Studies on the water induced plasticization behavior of nylon 4. The influence of crystallinity and radiation crosslinking. Makromol. Chem. 1985, 186, 191–201. [Google Scholar] [CrossRef]
- Zuo, J.; Li, S.; Bouzidi, L.; Narine, S.S. Thermoplastic polyester amides derived from oleic acid. Polymer 2011, 52, 4503–4516. [Google Scholar] [CrossRef]
- Ge, Y.P.; Yuan, D.; Luo, Z.L.; Wang, B.B. Synthesis and characterization of poly(ester amide) from remewable resources through melt polycondensation. Express Polym. Lett. 2014, 8, 50–54. [Google Scholar] [CrossRef]
- Kasuyaa, K.-I.; Takagi, K.-I.; Ishiwatari, S.-I.; Yoshida, Y.; Doi, Y. Biodegradabilities of various aliphatic polyesters in natural waters. Polym. Degrad. Stab. 1998, 59, 327–332. [Google Scholar] [CrossRef]
- Maeda, Y.; Nakayama, A.; Kawasaki, N.; Hayashi, K.; Aiba, S.; Yamamoto, N. Properties and biodegradability of chain-extended copoly(succinic anhydride/ethylene oxide). J. Environ. Polym. Degrad. 1996, 4, 225–233. [Google Scholar] [CrossRef]
- Yoo, E.S.; Im, S.S.; Yoo, Y. Morphology and degradation behavior of aliphatic/aromatic copolyesters. Macromol. Symp. 1997, 118, 739–745. [Google Scholar] [CrossRef]
- Sashiwa, H.; Kawasaki, N.; Nakayama, A.; Muraki, E.; Yamamoto, N.; Zhu, H.; Nagano, H.; Omura, Y.; Saimoto, H.; Shigemasa, Y.; et al. Chemical Modification of Chitosan. 13. Synthesis of Organosoluble, Palladium Adsorbable, and Biodegradable Chitosan Derivatives toward the Chemical Plating on Plastics. Biomacromolecules 2002, 3, 1120–1125. [Google Scholar] [CrossRef]
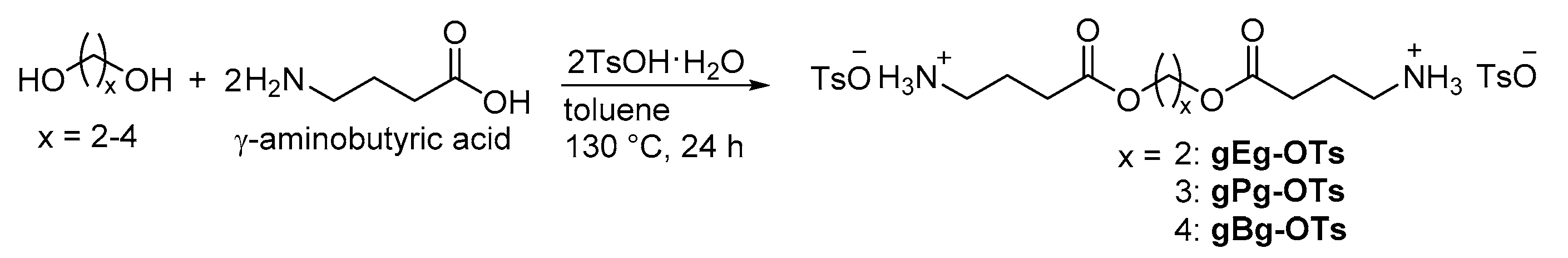
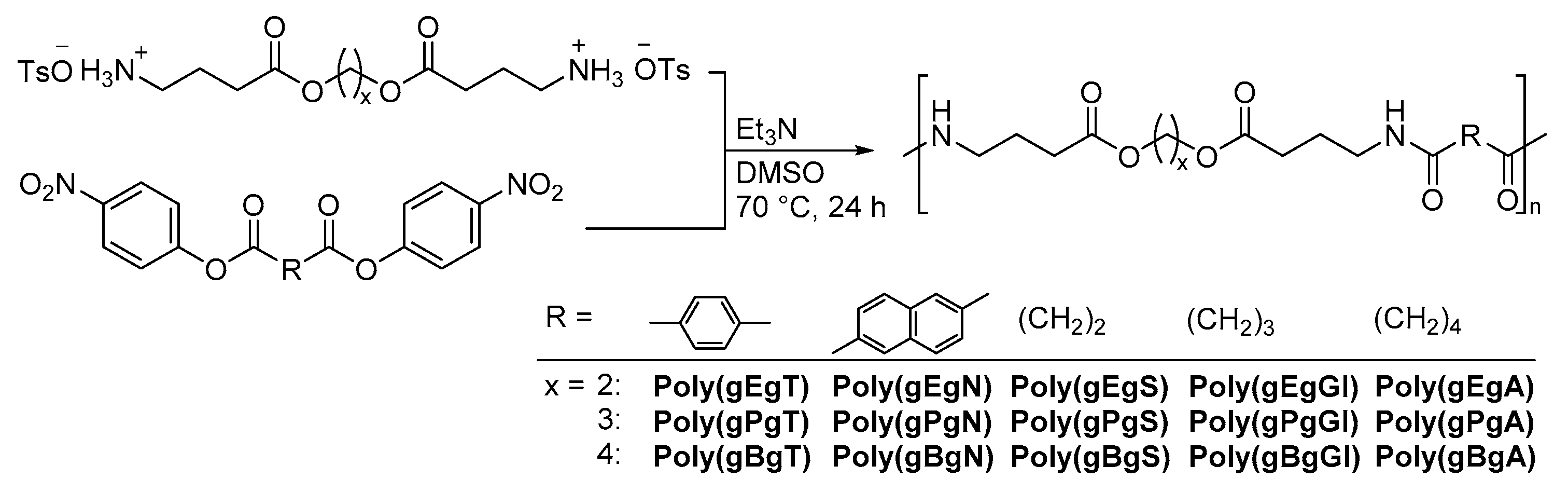
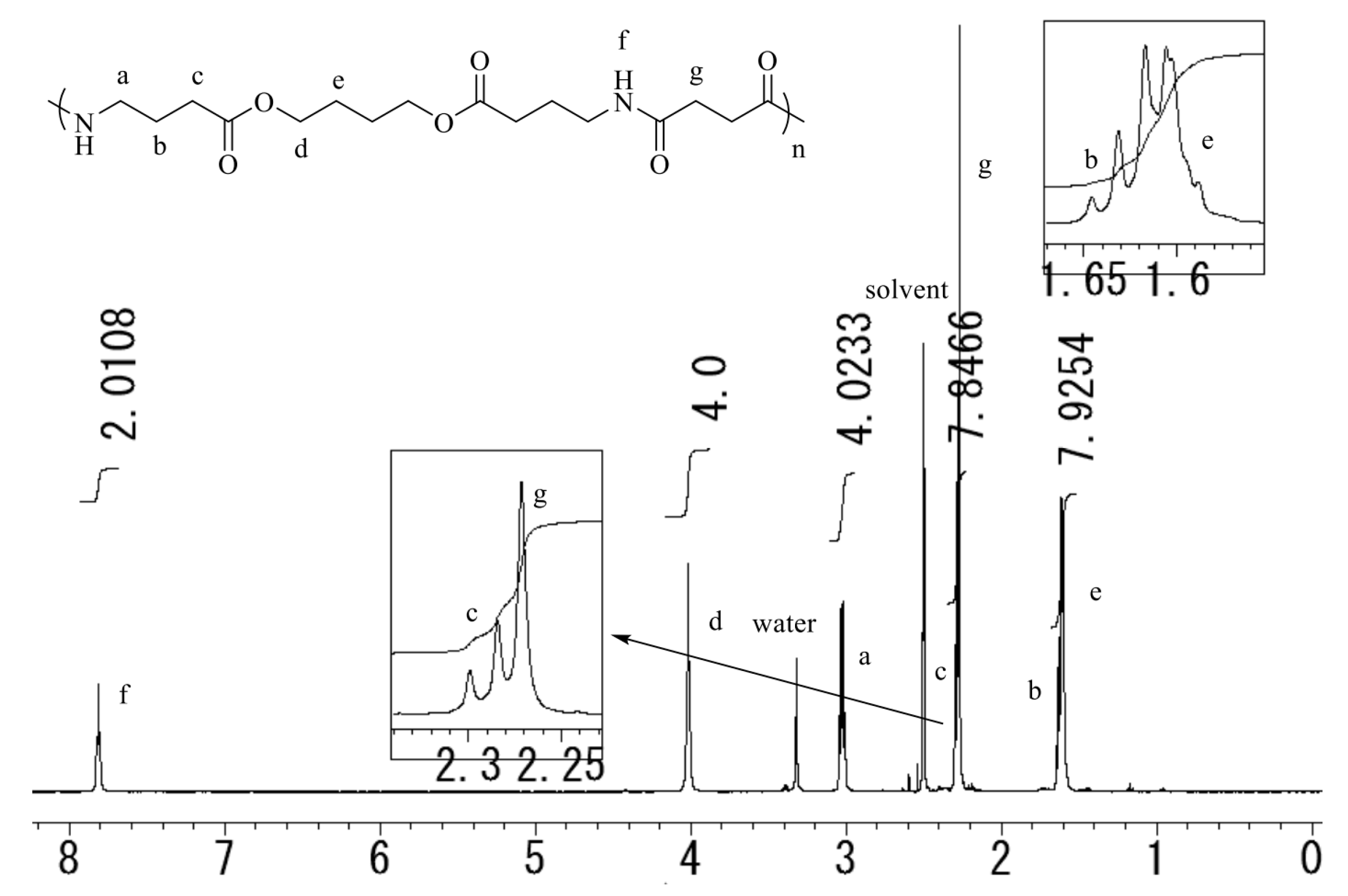
| Entry | Polymer | x | R | Yield | Mna | Mwa | Mw/Mna |
|---|---|---|---|---|---|---|---|
| (%) | (g/mol) | (g/mol) | |||||
| 1 | Poly(gEgT) | 2 | p-phenylene | 72 | 2600 | 4100 | 1.6 |
| 2 | Poly(gPgT) | 3 | p-phenylene | 92 | 6500 | 13,800 | 2.1 |
| 3 | Poly(gBgT) | 4 | p-phenylene | 80 | 4900 | 8300 | 1.7 |
| 4 | Poly(gEgN) | 2 | 2,5-naphthalenediyl | 92 | 5000 | 9900 | 2 |
| 5 | Poly(gPgN) | 3 | 2,5-naphthalenediyl | 97 | 5500 | 12,400 | 2.2 |
| 6 | Poly(gBgN) | 4 | 2,5-naphthalenediyl | 96 | 4900 | 8800 | 1.8 |
| 7 | Poly(gEgS) | 2 | (CH2)2 | 86 | 6100 | 12,400 | 2 |
| 8 | Poly(gPgS) | 3 | (CH2)2 | 83 | 5700 | 10,700 | 1.9 |
| 9 | Poly(gBgS) | 4 | (CH2)2 | 79 | 7600 | 15,300 | 2 |
| 10 | Poly(gEgGl) | 2 | (CH2)3 | 77 | 4100 | 10,500 | 2.6 |
| 11 | Poly(gPgGl) | 3 | (CH2)3 | 88 | 6700 | 17,800 | 2.7 |
| 12 | Poly(gBgGl) | 4 | (CH2)3 | 83 | 5700 | 14,900 | 2.6 |
| 13 | Poly(gEgA) | 2 | (CH2)4 | 83 | 4400 | 11,000 | 2.5 |
| 14 | Poly(gPgA) | 3 | (CH2)4 | 83 | 7300 | 18,700 | 2.5 |
| 15 | Poly(gBgA) | 4 | (CH2)4 | 78 | 6600 | 15,500 | 2.3 |
| Entry | Polymer | Tga | Tma | ΔHma | Td5b | Td10b |
|---|---|---|---|---|---|---|
| (°C) | (°C) | (J/g) | (°C) | (°C) | ||
| 1 | Poly(gEgT) | 103 | 212, 238 | 9.6, 11.0 | 302 | 320 |
| 2 | Poly(gPgT) | 75 | 200, 218 | 5.6, 8.6 | 276 | 294 |
| 3 | Poly(gBgT) | 72 | 188, 207 | 13.4, 5.2 | 268 | 280 |
| 4 | Poly(gEgN) | 84 | 229, 238 | 10.6 | 290 | 300 |
| 5 | Poly(gPgN) | 90 | 218, 232 | 5.6, 6.2 | 297 | 311 |
| 6 | Poly(gBgN) | 71 | 217, 236 | 14.2 | 300 | 315 |
| 7 | Poly(gEgS) | 71 | 165, 181 | 17 | 245 | 274 |
| 8 | Poly(gPgS) | 84 | 146, 166 | 11.0, 8.2 | 259 | 277 |
| 9 | Poly(gBgS) | 79 | 152, 172 | 11.1, 7.9 | 264 | 278 |
| 10 | Poly(gEgGl) | 78 | 112, 133 | 1.2, 11.0 | 269 | 284 |
| 11 | Poly(gPgGl) | 74 | 121, 140 | 13.1, 0.6 | 269 | 281 |
| 12 | Poly(gBgGl) | 77 | 111, 128 | 12.7, 1.1 | 271 | 289 |
| 13 | Poly(gEgA) | 75 | 162 | 19.2 | 279 | 292 |
| 14 | Poly(gPgA) | 82 | 140, 151, 162 | 18.4, 3.3 | 295 | 310 |
| 15 | Poly(gBgA) | 79 | 148, 162 | 15.0, 3.7 | 279 | 290 |
| PET [47] | 81 | 240–260 | 396 | |||
| PA4 [47,48] | 240–260 | 236 | ||||
| PBS [47] | 113 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nakayama, Y.; Watanabe, K.; Tanaka, R.; Shiono, T.; Kawasaki, N.; Yamano, N.; Nakayama, A. Synthesis, Properties, and Biodegradation of Sequential Poly(Ester Amide)s Containing γ-Aminobutyric Acid. Int. J. Mol. Sci. 2020, 21, 3674. https://doi.org/10.3390/ijms21103674
Nakayama Y, Watanabe K, Tanaka R, Shiono T, Kawasaki N, Yamano N, Nakayama A. Synthesis, Properties, and Biodegradation of Sequential Poly(Ester Amide)s Containing γ-Aminobutyric Acid. International Journal of Molecular Sciences. 2020; 21(10):3674. https://doi.org/10.3390/ijms21103674
Chicago/Turabian StyleNakayama, Yuushou, Kazumasa Watanabe, Ryo Tanaka, Takeshi Shiono, Norioki Kawasaki, Naoko Yamano, and Atsuyoshi Nakayama. 2020. "Synthesis, Properties, and Biodegradation of Sequential Poly(Ester Amide)s Containing γ-Aminobutyric Acid" International Journal of Molecular Sciences 21, no. 10: 3674. https://doi.org/10.3390/ijms21103674
APA StyleNakayama, Y., Watanabe, K., Tanaka, R., Shiono, T., Kawasaki, N., Yamano, N., & Nakayama, A. (2020). Synthesis, Properties, and Biodegradation of Sequential Poly(Ester Amide)s Containing γ-Aminobutyric Acid. International Journal of Molecular Sciences, 21(10), 3674. https://doi.org/10.3390/ijms21103674