Expression of Genes Involved in Axon Guidance: How Much Have We Learned?
Abstract
1. Introduction
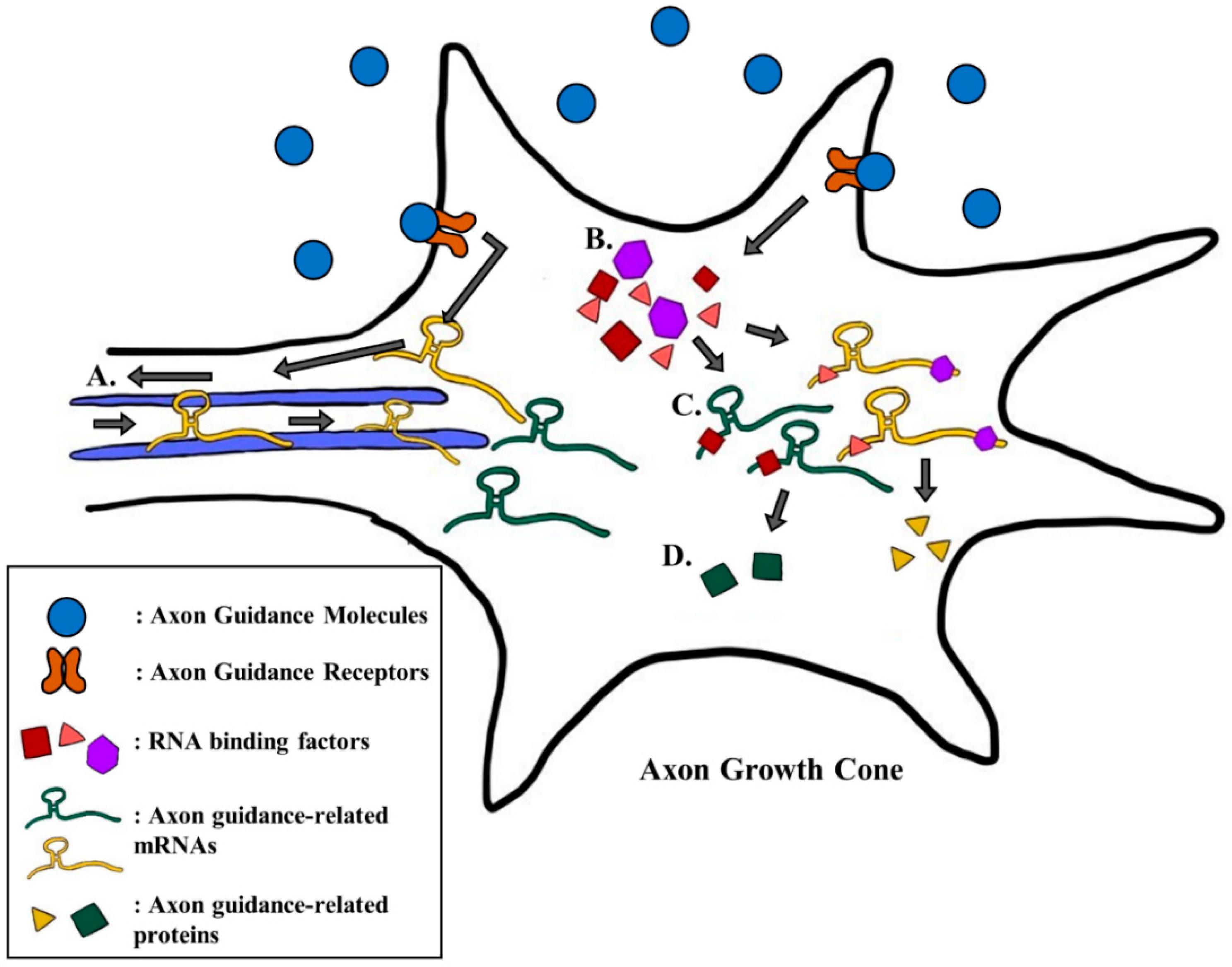
2. Regulation of Local Gene Expression by Guidance Cues and Receptors through RNA Binding Factors
2.1. BDNF Increases the Local Translation of DSCR1.4 mRNA
2.2. Netrin-1 Regulates the Local Translation of β-actin, Dscam, and tctp mRNAs
2.3. Sema3A Enhances Local Translation of fmr1 and the Processing of miRNAs
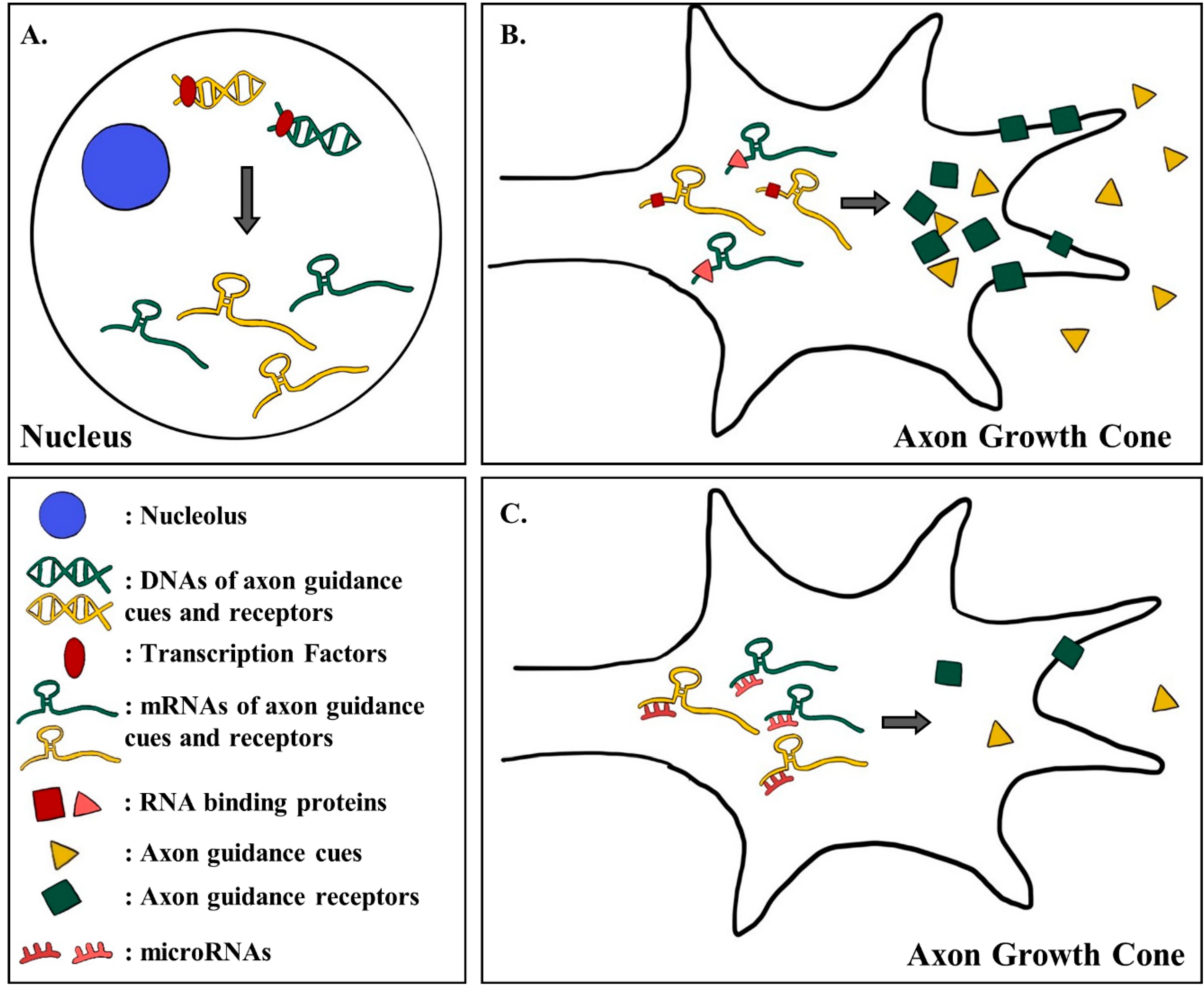
3. Transcriptional and Post-Transcriptional Regulation of the Expression of Guidance Proteins and Receptors
3.1. The Expression of Guidance Cues and Receptors are Regulated by Different Transcription Factors
3.2. RNA Binding Factors Regulate the Translation of Axon Guidance mRNAs
4. Pathological Relevance
4.1. The Association of Sema5A and Sema3F with Autism
4.2. The Protective Role of Netrin-1 against Alzheimer’s Disease
5. Concluding Remarks
Funding
Conflicts of Interest
References
- Kandel, E.R. The molecular biology of memory storage: A dialogue between genes and synapses. Science 2001, 294, 1030–1038. [Google Scholar] [CrossRef]
- Bakos, J.; Bacova, Z.; Grant, S.G.; Castejon, A.M.; Ostatnikova, D. Are Molecules Involved in Neuritogenesis and Axon Guidance Related to Autism Pathogenesis? Neuromolecular Med. 2015, 17, 297–304. [Google Scholar] [CrossRef] [PubMed]
- Gilman, S.R.; Chang, J.; Xu, B.; Bawa, T.S.; Gogos, J.A.; Karayiorgou, M.; Vitkup, D. Diverse types of genetic variation converge on functional gene networks involved in schizophrenia. Nat. Neurosci. 2012, 15, 1723–1728. [Google Scholar] [CrossRef]
- Iossifov, I.; O’Roak, B.J.; Sanders, S.J.; Ronemus, M.; Krumm, N.; Levy, D.; Stessman, H.A.; Witherspoon, K.T.; Vives, L.; Patterson, K.E.; et al. The contribution of de novo coding mutations to autism spectrum disorder. Nature 2014, 515, 216–221. [Google Scholar] [CrossRef] [PubMed]
- Ronemus, M.; Iossifov, I.; Levy, D.; Wigler, M. The role of de novo mutations in the genetics of autism spectrum disorders. Nat. Rev. Genet. 2014, 15, 133–141. [Google Scholar] [CrossRef]
- Bossers, K.; Meerhoff, G.; Balesar, R.; van Dongen, J.W.; Kruse, C.G.; Swaab, D.F.; Verhaagen, J. Analysis of gene expression in Parkinson’s disease: Possible involvement of neurotrophic support and axon guidance in dopaminergic cell death. Brain Pathol. 2009, 19, 91–107. [Google Scholar] [CrossRef]
- Antonell, A.; Llado, A.; Altirriba, J.; Botta-Orfila, T.; Balasa, M.; Fernandez, M.; Ferrer, I.; Sanchez-Valle, R.; Molinuevo, J.L. A preliminary study of the whole-genome expression profile of sporadic and monogenic early-onset Alzheimer’s disease. Neurobiol. Aging 2013, 34, 1772–1778. [Google Scholar] [CrossRef] [PubMed]
- Kashyap, G.; Bapat, D.; Das, D.; Gowaikar, R.; Amritkar, R.E.; Rangarajan, G.; Ravindranath, V.; Ambika, G. Synapse loss and progress of Alzheimer’s disease -A network model. Scientific Reports 2019, 9, 6555. [Google Scholar] [CrossRef]
- Bellon, A.; Mann, F. Keeping up with advances in axon guidance. Curr. Opin. Neurobiol. 2018, 53, 183–191. [Google Scholar] [CrossRef]
- Tessier-Lavigne, M.; Placzek, M.; Lumsden, A.G.; Dodd, J.; Jessell, T.M. Chemotropic guidance of developing axons in the mammalian central nervous system. Nature 1988, 336, 775–778. [Google Scholar] [CrossRef]
- Russell, S.A.; Bashaw, G.J. Axon guidance pathways and the control of gene expression. Dev. Dyn. 2018, 247, 571–580. [Google Scholar] [CrossRef] [PubMed]
- Squarzoni, P.; Thion, M.S.; Garel, S. Neuronal and microglial regulators of cortical wiring: Usual and novel guideposts. Front. Neurosci. 2015, 9, 248. [Google Scholar] [CrossRef] [PubMed]
- Coles, C.H.; Bradke, F. Coordinating neuronal actin-microtubule dynamics. Curr. Biol. 2015, 25, R677–R691. [Google Scholar] [CrossRef]
- Cammarata, G.M.; Bearce, E.A.; Lowery, L.A. Cytoskeletal social networking in the growth cone: How +TIPs mediate microtubule-actin cross-linking to drive axon outgrowth and guidance. Cytoskeleton 2016, 73, 461–476. [Google Scholar] [CrossRef] [PubMed]
- Omotade, O.F.; Pollitt, S.L.; Zheng, J.Q. Actin-based growth cone motility and guidance. Mol. Cell Neurosci. 2017, 84, 4–10. [Google Scholar] [CrossRef] [PubMed]
- O’Donnell, M.; Chance, R.K.; Bashaw, G.J. Axon growth and guidance: Receptor regulation and signal transduction. Annu. Rev. Neurosci. 2009, 32, 383–412. [Google Scholar] [CrossRef] [PubMed]
- Ng, J.; Nardine, T.; Harms, M.; Tzu, J.; Goldstein, A.; Sun, Y.; Dietzl, G.; Dickson, B.J.; Luo, L. Rac GTPases control axon growth, guidance and branching. Nature 2002, 416, 442–447. [Google Scholar] [CrossRef] [PubMed]
- Ye, X.; Qiu, Y.; Gao, Y.; Wan, D.; Zhu, H. A Subtle Network Mediating Axon Guidance: Intrinsic Dynamic Structure of Growth Cone, Attractive and Repulsive Molecular Cues, and the Intermediate Role of Signaling Pathways. Neural. Plast. 2019, 2019, 1719829. [Google Scholar] [CrossRef]
- Yam, P.T.; Charron, F. Signaling mechanisms of non-conventional axon guidance cues: The Shh, BMP and Wnt morphogens. Curr. Opin. Neurobiol. 2013, 23, 965–973. [Google Scholar] [CrossRef]
- O’Rourke, N.A.; Weiler, N.C.; Micheva, K.D.; Smith, S.J. Deep molecular diversity of mammalian synapses: Why it matters and how to measure it. Nat. Rev. Neurosci. 2012, 13, 365–379. [Google Scholar] [CrossRef]
- Shinmyo, Y.; Asrafuzzaman Riyadh, M.; Ahmed, G.; Bin Naser, I.; Hossain, M.; Takebayashi, H.; Kawasaki, H.; Ohta, K.; Tanaka, H. Draxin from neocortical neurons controls the guidance of thalamocortical projections into the neocortex. Nat. Commun. 2015, 6, 10232. [Google Scholar] [CrossRef] [PubMed]
- Blockus, H.; Chedotal, A. The multifaceted roles of Slits and Robos in cortical circuits: From proliferation to axon guidance and neurological diseases. Curr. Opin. Neurobiol. 2014, 27, 82–88. [Google Scholar] [CrossRef]
- Blockus, H.; Chedotal, A. Slit-Robo signaling. Development 2016, 143, 3037–3044. [Google Scholar] [CrossRef]
- Pignata, A.; Ducuing, H.; Castellani, V. Commissural axon navigation: Control of midline crossing in the vertebrate spinal cord by the semaphorin 3B signaling. Cell Adh. Migr. 2016, 10, 604–617. [Google Scholar] [CrossRef]
- Bruce, F.M.; Brown, S.; Smith, J.N.; Fuerst, P.G.; Erskine, L. DSCAM promotes axon fasciculation and growth in the developing optic pathway. Proc. Natl. Acad. Sci. USA 2017, 114, 1702–1707. [Google Scholar] [CrossRef] [PubMed]
- Hornberg, H.; Cioni, J.M.; Harris, W.A.; Holt, C.E. Hermes Regulates Axon Sorting in the Optic Tract by Post-Trancriptional Regulation of Neuropilin 1. J. Neurosci. 2016, 36, 12697–12706. [Google Scholar] [CrossRef] [PubMed]
- Choi, J.H.; Kim, S.H.; Jeong, Y.H.; Kim, S.W.; Min, K.T.; Kim, K.T. hnRNP Q Regulates Internal Ribosome Entry Site-Mediated fmr1 Translation in Neurons. Mol. Cell Biol. 2019, 39. [Google Scholar] [CrossRef]
- Choi, J.H.; Wang, W.; Park, D.; Kim, S.H.; Kim, K.T.; Min, K.T. IRES-mediated translation of cofilin regulates axonal growth cone extension and turning. EMBO J. 2018, 37. [Google Scholar] [CrossRef]
- Seo, J.Y.; Jung, Y.; Kim, D.Y.; Ryu, H.G.; Lee, J.; Kim, S.W.; Kim, K.T. DAP5 increases axonal outgrowth of hippocampal neurons by enhancing the cap-independent translation of DSCR1.4 mRNA. Cell Death Dis. 2019, 10, 49. [Google Scholar] [CrossRef]
- Cagnetta, R.; Wong, H.H.; Frese, C.K.; Mallucci, G.R.; Krijgsveld, J.; Holt, C.E. Noncanonical Modulation of the eIF2 Pathway Controls an Increase in Local Translation during Neural Wiring. Mol. Cell 2019, 73, 474–489. [Google Scholar] [CrossRef]
- Yam, P.T.; Langlois, S.D.; Morin, S.; Charron, F. Sonic hedgehog guides axons through a noncanonical, Src-family-kinase-dependent signaling pathway. Neuron 2009, 62, 349–362. [Google Scholar] [CrossRef] [PubMed]
- Landis, S.C. Neuronal Growth Cones. Annu. Rev. Physiol. 1983, 45, 567–580. [Google Scholar] [CrossRef] [PubMed]
- Chedotal, A. Roles of axon guidance molecules in neuronal wiring in the developing spinal cord. Nat. Rev. Neurosci. 2019, 20, 380–396. [Google Scholar] [CrossRef] [PubMed]
- Stoeckli, E. Where does axon guidance lead us? F1000.Res. 2017, 6, 78. [Google Scholar] [CrossRef] [PubMed]
- Stoeckli, E.T. Understanding axon guidance: Are we nearly there yet? Development 2018, 145. [Google Scholar] [CrossRef] [PubMed]
- Leung, K.M.; Lu, B.; Wong, H.H.; Lin, J.Q.; Turner-Bridger, B.; Holt, C.E. Cue-Polarized Transport of beta-actin mRNA Depends on 3’UTR and Microtubules in Live Growth Cones. Front. Cell Neurosci. 2018, 12, 300. [Google Scholar] [CrossRef] [PubMed]
- Jain, S.; Welshhans, K. Netrin-1 induces local translation of down syndrome cell adhesion molecule in axonal growth cones. Dev. Neurobiol. 2016, 76, 799–816. [Google Scholar] [CrossRef]
- Cagnetta, R.; Frese, C.K.; Shigeoka, T.; Krijgsveld, J.; Holt, C.E. Rapid Cue-Specific Remodeling of the Nascent Axonal Proteome. Neuron 2018, 99, 29–46 e24. [Google Scholar] [CrossRef]
- Ernfors, P.; Lee, K.F.; Jaenisch, R. Mice lacking brain-derived neurotrophic factor develop with sensory deficits. Nature 1994, 368, 147–150. [Google Scholar] [CrossRef]
- Cohen-Cory, S.; Kidane, A.H.; Shirkey, N.J.; Marshak, S. Brain-derived neurotrophic factor and the development of structural neuronal connectivity. Dev. Neurobiol. 2010, 70, 271–288. [Google Scholar] [CrossRef]
- Chen, T.J.; Gehler, S.; Shaw, A.E.; Bamburg, J.R.; Letourneau, P.C. Cdc42 participates in the regulation of ADF/cofilin and retinal growth cone filopodia by brain derived neurotrophic factor. J. Neurobiol. 2006, 66, 103–114. [Google Scholar] [CrossRef] [PubMed]
- Ohashi, K. Roles of cofilin in development and its mechanisms of regulation. Dev. Growth Differ. 2015, 57, 275–290. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Zhu, J.Z.; Chang, K.T.; Min, K.T. DSCR1 interacts with FMRP and is required for spine morphogenesis and local protein synthesis. EMBO J. 2012, 31, 3655–3666. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Rai, A.; Hur, E.M.; Smilansky, Z.; Chang, K.T.; Min, K.T. DSCR1 is required for both axonal growth cone extension and steering. J. Cell Biol. 2016, 213, 451–462. [Google Scholar] [CrossRef]
- Serafini, T.; Colamarino, S.A.; Leonardo, E.D.; Wang, H.; Beddington, R.; Skarnes, W.C.; Tessier-Lavigne, M. Netrin-1 is required for commissural axon guidance in the developing vertebrate nervous system. Cell 1996, 87, 1001–1014. [Google Scholar] [CrossRef]
- Colamarino, S.A.; Tessier-Lavigne, M. The axonal chemoattractant netrin-1 is also a chemorepellent for trochlear motor axons. Cell 1995, 81, 621–629. [Google Scholar] [CrossRef]
- Jarjour, A.A.; Manitt, C.; Moore, S.W.; Thompson, K.M.; Yuh, S.J.; Kennedy, T.E. Netrin-1 is a chemorepellent for oligodendrocyte precursor cells in the embryonic spinal cord. J. Neurosci. 2003, 23, 3735–3744. [Google Scholar] [CrossRef]
- Kennedy, T.E.; Wang, H.; Marshall, W.; Tessier-Lavigne, M. Axon guidance by diffusible chemoattractants: A gradient of netrin protein in the developing spinal cord. J. Neurosci. 2006, 26, 8866–8874. [Google Scholar] [CrossRef]
- Briancon-Marjollet, A.; Ghogha, A.; Nawabi, H.; Triki, I.; Auziol, C.; Fromont, S.; Piche, C.; Enslen, H.; Chebli, K.; Cloutier, J.F.; et al. Trio mediates netrin-1-induced Rac1 activation in axon outgrowth and guidance. Mol. Cell Biol. 2008, 28, 2314–2323. [Google Scholar] [CrossRef]
- Wang, G.X.; Poo, M.M. Requirement of TRPC channels in netrin-1-induced chemotropic turning of nerve growth cones. Nature 2005, 434, 898–904. [Google Scholar] [CrossRef]
- Strohl, F.; Lin, J.Q.; Laine, R.F.; Wong, H.H.; Urbancic, V.; Cagnetta, R.; Holt, C.E.; Kaminski, C.F. Single Molecule Translation Imaging Visualizes the Dynamics of Local beta-Actin Synthesis in Retinal Axons. Sci. Rep. 2017, 7, 709. [Google Scholar] [CrossRef] [PubMed]
- Leung, K.M.; van Horck, F.P.; Lin, A.C.; Allison, R.; Standart, N.; Holt, C.E. Asymmetrical beta-actin mRNA translation in growth cones mediates attractive turning to netrin-1. Nat. Neurosci. 2006, 9, 1247–1256. [Google Scholar] [CrossRef] [PubMed]
- Gouveia Roque, C.; Holt, C.E. Growth Cone Tctp Is Dynamically Regulated by Guidance Cues. Front. Mol. Neurosci. 2018, 11, 399. [Google Scholar] [CrossRef] [PubMed]
- Campbell, D.S.; Holt, C.E. Apoptotic pathway and MAPKs differentially regulate chemotropic responses of retinal growth cones. Neuron 2003, 37, 939–952. [Google Scholar] [CrossRef]
- Campbell, D.S.; Holt, C.E. Chemotropic responses of retinal growth cones mediated by rapid local protein synthesis and degradation. Neuron 2001, 32, 1013–1026. [Google Scholar] [CrossRef]
- Sahoo, P.K.; Smith, D.S.; Perrone-Bizzozero, N.; Twiss, J.L. Axonal mRNA transport and translation at a glance. J. Cell Sci. 2018, 131. [Google Scholar] [CrossRef]
- Mayr, C. Regulation by 3’-Untranslated Regions. Annu. Rev. Genet. 2017, 51, 171–194. [Google Scholar] [CrossRef]
- Meyer, K.D.; Saletore, Y.; Zumbo, P.; Elemento, O.; Mason, C.E.; Jaffrey, S.R. Comprehensive analysis of mRNA methylation reveals enrichment in 3’ UTRs and near stop codons. Cell 2012, 149, 1635–1646. [Google Scholar] [CrossRef]
- Lesbirel, S.; Viphakone, N.; Parker, M.; Parker, J.; Heath, C.; Sudbery, I.; Wilson, S.A. The m(6)A-methylase complex recruits TREX and regulates mRNA export. Sci. Rep. 2018, 8, 13827. [Google Scholar] [CrossRef]
- Yu, J.; Chen, M.; Huang, H.; Zhu, J.; Song, H.; Zhu, J.; Park, J.; Ji, S.-J. Dynamic m6A modification regulates local translation of mRNA in axons. Nucleic Acids Res. 2017, 46, 1412–1423. [Google Scholar] [CrossRef]
- Yamakawa, K.; Huot, Y.K.; Haendelt, M.A.; Hubert, R.; Chen, X.N.; Lyons, G.E.; Korenberg, J.R. DSCAM: A novel member of the immunoglobulin superfamily maps in a Down syndrome region and is involved in the development of the nervous system. Hum. Mol. Genet. 1998, 7, 227–237. [Google Scholar] [CrossRef] [PubMed]
- Santos, R.A.; Fuertes, A.J.C.; Short, G.; Donohue, K.C.; Shao, H.; Quintanilla, J.; Malakzadeh, P.; Cohen-Cory, S. DSCAM differentially modulates pre- and postsynaptic structural and functional central connectivity during visual system wiring. Neural Dev. 2018, 13, 22. [Google Scholar] [CrossRef] [PubMed]
- Hutchinson, K.M.; Vonhoff, F.; Duch, C. Dscam1 is required for normal dendrite growth and branching but not for dendritic spacing in Drosophila motoneurons. J. Neurosci. 2014, 34, 1924–1931. [Google Scholar] [CrossRef] [PubMed]
- Dascenco, D.; Erfurth, M.L.; Izadifar, A.; Song, M.; Sachse, S.; Bortnick, R.; Urwyler, O.; Petrovic, M.; Ayaz, D.; He, H.; et al. Slit and Receptor Tyrosine Phosphatase 69D Confer Spatial Specificity to Axon Branching via Dscam1. Cell 2015, 162, 1140–1154. [Google Scholar] [CrossRef] [PubMed]
- Sachse, S.M.; Lievens, S.; Ribeiro, L.F.; Dascenco, D.; Masschaele, D.; Horre, K.; Misbaer, A.; Vanderroost, N.; De Smet, A.S.; Salta, E.; et al. Nuclear import of the DSCAM-cytoplasmic domain drives signaling capable of inhibiting synapse formation. Embo J. 2019, 38. [Google Scholar] [CrossRef]
- Ly, A.; Nikolaev, A.; Suresh, G.; Zheng, Y.; Tessier-Lavigne, M.; Stein, E. DSCAM is a netrin receptor that collaborates with DCC in mediating turning responses to netrin-1. Cell 2008, 133, 1241–1254. [Google Scholar] [CrossRef]
- Liu, G.; Li, W.; Wang, L.; Kar, A.; Guan, K.L.; Rao, Y.; Wu, J.Y. DSCAM functions as a netrin receptor in commissural axon pathfinding. Proc. Natl. Acad. Sci. USA 2009, 106, 2951–2956. [Google Scholar] [CrossRef]
- Huang, H.; Shao, Q.; Qu, C.; Yang, T.; Dwyer, T.; Liu, G. Coordinated interaction of Down syndrome cell adhesion molecule and deleted in colorectal cancer with dynamic TUBB3 mediates Netrin-1-induced axon branching. Neuroscience 2015, 293, 109–122. [Google Scholar] [CrossRef]
- Jojic, B.; Amodeo, S.; Bregy, I.; Ochsenreiter, T. Distinct 3’ UTRs regulate the life-cycle-specific expression of two TCTP paralogs in Trypanosoma brucei. J. Cell Sci. 2018, 131. [Google Scholar] [CrossRef]
- Koziol, M.J.; Gurdon, J.B. TCTP in development and cancer. Biochem. Res. Int. 2012, 2012, 105203. [Google Scholar] [CrossRef]
- Bommer, U.A. The Translational Controlled Tumour Protein TCTP: Biological Functions and Regulation. Results Probl. Cell Differ. 2017, 64, 69–126. [Google Scholar] [CrossRef]
- Roque, C.G.; Wong, H.H.; Lin, J.Q.; Holt, C.E. Tumor protein Tctp regulates axon development in the embryonic visual system. Development 2016, 143, 1134–1148. [Google Scholar] [CrossRef] [PubMed]
- Kolodkin, A.L.; Matthes, D.J.; Goodman, C.S. The semaphorin genes encode a family of transmembrane and secreted growth cone guidance molecules. Cell 1993, 75, 1389–1399. [Google Scholar] [CrossRef]
- Nakamura, F.; Kalb, R.G.; Strittmatter, S.M. Molecular basis of semaphorin-mediated axon guidance. J. Neurobiol. 2000, 44, 219–229. [Google Scholar] [CrossRef]
- Rohm, B.; Ottemeyer, A.; Lohrum, M.; Puschel, A.W. Plexin/neuropilin complexes mediate repulsion by the axonal guidance signal semaphorin 3A. Mech. Dev. 2000, 93, 95–104. [Google Scholar] [CrossRef]
- Takamatsu, H.; Takegahara, N.; Nakagawa, Y.; Tomura, M.; Taniguchi, M.; Friedel, R.H.; Rayburn, H.; Tessier-Lavigne, M.; Yoshida, Y.; Okuno, T.; et al. Semaphorins guide the entry of dendritic cells into the lymphatics by activating myosin II. Nat. Immunol. 2010, 11, 594–600. [Google Scholar] [CrossRef] [PubMed]
- Barao, S.; Gartner, A.; Leyva-Diaz, E.; Demyanenko, G.; Munck, S.; Vanhoutvin, T.; Zhou, L.; Schachner, M.; Lopez-Bendito, G.; Maness, P.F.; et al. Antagonistic Effects of BACE1 and APH1B-gamma-Secretase Control Axonal Guidance by Regulating Growth Cone Collapse. Cell Rep. 2015, 12, 1367–1376. [Google Scholar] [CrossRef] [PubMed]
- Corradi, E.; Dalla Costa, I.; Gavoci, A.; Iyer, A.; Roccuzzo, M.; Otto, T.A.; Oliani, E.; Bridi, S.; Strohbuecker, S.; Santos-Rodriguez, G.; et al. Axonal precursor miRNAs hitchhike on endosomes and locally regulate the development of neural circuits. Embo. J. 2020, 39, e102513. [Google Scholar] [CrossRef]
- Price, T.J.; Flores, C.M.; Cervero, F.; Hargreaves, K.M. The RNA binding and transport proteins staufen and fragile X mental retardation protein are expressed by rat primary afferent neurons and localize to peripheral and central axons. Neuroscience 2006, 141, 2107–2116. [Google Scholar] [CrossRef]
- Sawicka, K.; Hale, C.R.; Park, C.Y.; Fak, J.J.; Gresack, J.E.; Van Driesche, S.J.; Kang, J.J.; Darnell, J.C.; Darnell, R.B. FMRP has a cell-type-specific role in CA1 pyramidal neurons to regulate autism-related transcripts and circadian memory. Elife 2019, 8. [Google Scholar] [CrossRef]
- Li, C.; Bassell, G.J.; Sasaki, Y. Fragile X Mental Retardation Protein is Involved in Protein Synthesis-Dependent Collapse of Growth Cones Induced by Semaphorin-3A. Front. Neural Circuits 2009, 3, 11. [Google Scholar] [CrossRef] [PubMed]
- Bartel, D.P. MicroRNAs: Target recognition and regulatory functions. Cell 2009, 136, 215–233. [Google Scholar] [CrossRef] [PubMed]
- Iyer, A.N.; Bellon, A.; Baudet, M.L. microRNAs in axon guidance. Front. Cell Neurosci. 2014, 8, 78. [Google Scholar] [CrossRef] [PubMed]
- Hutvagner, G.; McLachlan, J.; Pasquinelli, A.E.; Balint, E.; Tuschl, T.; Zamore, P.D. A cellular function for the RNA-interference enzyme Dicer in the maturation of the let-7 small temporal RNA. Science 2001, 293, 834–838. [Google Scholar] [CrossRef] [PubMed]
- Gershoni-Emek, N.; Altman, T.; Ionescu, A.; Costa, C.J.; Gradus-Pery, T.; Willis, D.E.; Perlson, E. Localization of RNAi Machinery to Axonal Branch Points and Growth Cones Is Facilitated by Mitochondria and Is Disrupted in ALS. Front. Mol. Neurosci. 2018, 11, 311. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.H.; Kim, P.; Phay, M.; Yoo, S. Identification of precursor microRNAs within distal axons of sensory neuron. J. Neurochem. 2015, 134, 193–199. [Google Scholar] [CrossRef]
- Vargas, J.N.; Kar, A.N.; Kowalak, J.A.; Gale, J.R.; Aschrafi, A.; Chen, C.Y.; Gioio, A.E.; Kaplan, B.B. Axonal localization and mitochondrial association of precursor microRNA 338. Cell Mol. Life Sci. 2016, 73, 4327–4340. [Google Scholar] [CrossRef]
- Schuz, A.; Palm, G. Density of neurons and synapses in the cerebral cortex of the mouse. J. Comp. Neurol. 1989, 286, 442–455. [Google Scholar] [CrossRef]
- Turner, A.M.; Greenough, W.T. Differential Rearing Effects on Rat Visual-Cortex Synapses.1. Synaptic and Neuronal Density and Synapses Per Neuron. Brain Res. 1985, 329, 195–203. [Google Scholar] [CrossRef]
- Shin, M.; Wang, Y.; Borgus, J.R.; Venton, B.J. Electrochemistry at the Synapse. Annu. Rev. Anal. Chem. 2019, 12, 297–321. [Google Scholar] [CrossRef]
- De Wit, J.; Ghosh, A. Specification of synaptic connectivity by cell surface interactions. Nat. Rev. Neurosci. 2016, 17, 22–35. [Google Scholar] [CrossRef]
- Miller, K.E.; Suter, D.M. An Integrated Cytoskeletal Model of Neurite Outgrowth. Front. Cell Neurosci. 2018, 12, 447. [Google Scholar] [CrossRef]
- Gallo, G. RhoA-kinase coordinates F-actin organization and myosin II activity during semaphorin-3A-induced axon retraction. J. Cell Sci. 2006, 119, 3413–3423. [Google Scholar] [CrossRef]
- Kodo, K.; Shibata, S.; Miyagawa-Tomita, S.; Ong, S.G.; Takahashi, H.; Kume, T.; Okano, H.; Matsuoka, R.; Yamagishi, H. Regulation of Sema3c and the Interaction between Cardiac Neural Crest and Second Heart Field during Outflow Tract Development. Sci. Rep. 2017, 7, 6771. [Google Scholar] [CrossRef]
- Ozmadenci, D.; Feraud, O.; Markossian, S.; Kress, E.; Ducarouge, B.; Gibert, B.; Ge, J.; Durand, I.; Gadot, N.; Plateroti, M.; et al. Netrin-1 regulates somatic cell reprogramming and pluripotency maintenance. Nat. Commun. 2015, 6, 7398. [Google Scholar] [CrossRef]
- Peng, J.; Santiago, I.J.; Ahn, C.; Gur, B.; Tsui, C.K.; Su, Z.; Xu, C.; Karakhanyan, A.; Silies, M.; Pecot, M.Y. Drosophila Fezf coordinates laminar-specific connectivity through cell-intrinsic and cell-extrinsic mechanisms. Elife 2018, 7. [Google Scholar] [CrossRef]
- Sun, Y.; Liu, C.H.; Wang, Z.; Meng, S.S.; Burnim, S.B.; SanGiovanni, J.P.; Kamenecka, T.M.; Solt, L.A.; Chen, J. RORalpha modulates semaphorin 3E transcription and neurovascular interaction in pathological retinal angiogenesis. Faseb J. 2017, 31, 4492–4502. [Google Scholar] [CrossRef]
- Wang, J.; Zhou, X.; Lu, H.; Song, M.; Zhao, J.; Wang, Q. Fluoxetine induces vascular endothelial growth factor/Netrin over-expression via the mediation of hypoxia-inducible factor 1-alpha in SH-SY5Y cells. J. Neurochem. 2016, 136, 1186–1195. [Google Scholar] [CrossRef][Green Version]
- Castillo-Paterna, M.; Moreno-Juan, V.; Filipchuk, A.; Rodriguez-Malmierca, L.; Susin, R.; Lopez-Bendito, G. DCC functions as an accelerator of thalamocortical axonal growth downstream of spontaneous thalamic activity. EMBO Rep. 2015, 16, 851–862. [Google Scholar] [CrossRef]
- Kandemir, B.; Dag, U.; Bakir Gungor, B.; Durasi, I.M.; Erdogan, B.; Sahin, E.; Sezerman, U.; Aksan Kurnaz, I. In silico analyses and global transcriptional profiling reveal novel putative targets for Pea3 transcription factor related to its function in neurons. PLoS ONE 2017, 12, e0170585. [Google Scholar] [CrossRef]
- Takahashi, K.; Okita, K.; Nakagawa, M.; Yamanaka, S. Induction of pluripotent stem cells from fibroblast cultures. Nat. Protoc. 2007, 2, 3081–3089. [Google Scholar] [CrossRef]
- Takahashi, K.; Yamanaka, S. Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell 2006, 126, 663–676. [Google Scholar] [CrossRef]
- Dos Santos, R.L.; Tosti, L.; Radzisheuskaya, A.; Caballero, I.M.; Kaji, K.; Hendrich, B.; Silva, J.C.R. MBD3/NuRD Facilitates Induction of Pluripotency in a Context-Dependent Manner. Cell Stem Cell 2014, 15, 392. [Google Scholar] [CrossRef]
- Tan, L.; Zhang, K.X.; Pecot, M.Y.; Nagarkar-Jaiswal, S.; Lee, P.T.; Takemura, S.Y.; McEwen, J.M.; Nern, A.; Xu, S.; Tadros, W.; et al. Ig Superfamily Ligand and Receptor Pairs Expressed in Synaptic Partners in Drosophila. Cell 2015, 163, 1756–1769. [Google Scholar] [CrossRef]
- Pecot, M.Y.; Tadros, W.; Nern, A.; Bader, M.; Chen, Y.; Zipursky, S.L. Multiple interactions control synaptic layer specificity in the Drosophila visual system. Neuron 2013, 77, 299–310. [Google Scholar] [CrossRef]
- Kolodziej, P.A.; Timpe, L.C.; Mitchell, K.J.; Fried, S.R.; Goodman, C.S.; Jan, L.Y.; Jan, Y.N. frazzled encodes a Drosophila member of the DCC immunoglobulin subfamily and is required for CNS and motor axon guidance. Cell 1996, 87, 197–204. [Google Scholar] [CrossRef]
- Santiago, C.; Bashaw, G.J. Islet Coordinately Regulates Motor Axon Guidance and Dendrite Targeting through the Frazzled/DCC Receptor. Cell Rep. 2017, 18, 1646–1659. [Google Scholar] [CrossRef]
- Thor, S.; Thomas, J.B. The Drosophila islet gene governs axon pathfinding and neurotransmitter identity. Neuron 1997, 18, 397–409. [Google Scholar] [CrossRef]
- Sugiyama, C.; Nakamichi, N.; Ogura, M.; Honda, E.; Maeda, S.; Taniura, H.; Yoneda, Y. Activator protein-1 responsive to the group II metabotropic glutamate receptor subtype in association with intracellular calcium in cultured rat cortical neurons. Neurochem. Int. 2007, 51, 467–475. [Google Scholar] [CrossRef]
- Gazon, H.; Barbeau, B.; Mesnard, J.M.; Peloponese, J.M., Jr. Hijacking of the AP-1 Signaling Pathway during Development of ATL. Front. Microbiol. 2017, 8, 2686. [Google Scholar] [CrossRef]
- Lee, H.; Kim, M.; Kim, N.; Macfarlan, T.; Pfaff, S.L.; Mastick, G.S.; Song, M.R. Slit and Semaphorin signaling governed by Islet transcription factors positions motor neuron somata within the neural tube. Exp. Neurol. 2015, 269, 17–27. [Google Scholar] [CrossRef]
- Haklai-Topper, L.; Mlechkovich, G.; Savariego, D.; Gokhman, I.; Yaron, A. Cis interaction between Semaphorin6A and Plexin-A4 modulates the repulsive response to Sema6A. EMBO J. 2010, 29, 2635–2645. [Google Scholar] [CrossRef]
- Binet, F.; Mawambo, G.; Sitaras, N.; Tetreault, N.; Lapalme, E.; Favret, S.; Cerani, A.; Leboeuf, D.; Tremblay, S.; Rezende, F.; et al. Neuronal ER stress impedes myeloid-cell-induced vascular regeneration through IRE1alpha degradation of netrin-1. Cell Metab. 2013, 17, 353–371. [Google Scholar] [CrossRef]
- Belle, M.; Parray, A.; Belle, M.; Chédotal, A.; Nguyen-Ba-Charvet, K.T. PlexinA2 and Sema6A are required for retinal progenitor cell migration. Dev. Growth Differ. 2016, 58, 492–502. [Google Scholar] [CrossRef]
- Kensler, T.W.; Wakabayashi, N.; Biswal, S. Cell survival responses to environmental stresses via the Keap1-Nrf2-ARE pathway. Annu. Rev. Pharmacol. Toxicol. 2007, 47, 89–116. [Google Scholar] [CrossRef]
- Wei, Y.; Gong, J.; Xu, Z.; Thimmulappa, R.K.; Mitchell, K.L.; Welsbie, D.S.; Biswal, S.; Duh, E.J. Nrf2 in ischemic neurons promotes retinal vascular regeneration through regulation of semaphorin 6A. Proc. Natl. Acad. Sci. USA 2015, 112, E6927–E6936. [Google Scholar] [CrossRef]
- Liu, X.F.; Zhou, D.D.; Xie, T.; Malik, T.H.; Lu, C.B.; Li, H.J.; Wang, F.; Shu, C.; Liu, C.; Lu, C.W.; et al. Nrf2, a Potential Therapeutic Target against Oxidative Stress in Corneal Diseases. Oxid. Med. Cell Longev. 2017, 2017, 2326178. [Google Scholar] [CrossRef]
- Nakagami, Y. Nrf2 Is an Attractive Therapeutic Target for Retinal Diseases. Oxid. Med. Cell. Longev. 2016, 2016, 7469326. [Google Scholar] [CrossRef]
- Villain, G.; Poissonnier, L.; Noueihed, B.; Bonfils, G.; Rivera, J.C.; Chemtob, S.; Soncin, F.; Mattot, V. miR-126-5p promotes retinal endothelial cell survival through SetD5 regulation in neurons. Development 2018, 145. [Google Scholar] [CrossRef]
- Jetten, A.M. Retinoid-related orphan receptors (RORs): Critical roles in development, immunity, circadian rhythm, and cellular metabolism. Nucl. Recept. Signal. 2009, 7, e003. [Google Scholar] [CrossRef]
- Kuechler, A.; Zink, A.M.; Wieland, T.; Ludecke, H.J.; Cremer, K.; Salviati, L.; Magini, P.; Najafi, K.; Zweier, C.; Czeschik, J.C.; et al. Loss-of-function variants of SETD5 cause intellectual disability and the core phenotype of microdeletion 3p25.3 syndrome. Eur. J. Hum. Genet. 2015, 23, 753–760. [Google Scholar] [CrossRef] [PubMed]
- Parenti, I.; Teresa-Rodrigo, M.E.; Pozojevic, J.; Ruiz Gil, S.; Bader, I.; Braunholz, D.; Bramswig, N.C.; Gervasini, C.; Larizza, L.; Pfeiffer, L.; et al. Mutations in chromatin regulators functionally link Cornelia de Lange syndrome and clinically overlapping phenotypes. Hum. Genet. 2017, 136, 307–320. [Google Scholar] [CrossRef] [PubMed]
- Szczaluba, K.; Brzezinska, M.; Kot, J.; Rydzanicz, M.; Walczak, A.; Stawinski, P.; Werner, B.; Ploski, R. SETD5 loss-of-function mutation as a likely cause of a familial syndromic intellectual disability with variable phenotypic expression. Am. J. Med. Genet. A 2016, 170, 2322–2327. [Google Scholar] [CrossRef] [PubMed]
- Belkina, A.C.; Denis, G.V. BET domain co-regulators in obesity, inflammation and cancer. Nat. Rev. Cancer 2012, 12, 465–477. [Google Scholar] [CrossRef]
- Batista, A.F.; Hengst, U. Intra-axonal protein synthesis in development and beyond. Int. J. Dev. Neurosci. 2016, 55, 140–149. [Google Scholar] [CrossRef]
- Hornberg, H.; Holt, C. RNA-binding proteins and translational regulation in axons and growth cones. Front. Neurosci. 2013, 7, 81. [Google Scholar] [CrossRef]
- Jain, S.; Welshhans, K. Local translation of cell adhesion molecules in axons. Neural. Regen. Res. 2016, 11, 543–544. [Google Scholar] [CrossRef]
- Zhang, L.; Chen, Y.; Li, C.; Liu, J.; Ren, H.; Li, L.; Zheng, X.; Wang, H.; Han, Z. RNA binding protein PUM2 promotes the stemness of breast cancer cells via competitively binding to neuropilin-1 (NRP-1) mRNA with miR-376a. Biomed. Pharm. 2019, 114, 108772. [Google Scholar] [CrossRef]
- Hornberg, H.; Wollerton-van Horck, F.; Maurus, D.; Zwart, M.; Svoboda, H.; Harris, W.A.; Holt, C.E. RNA-binding protein Hermes/RBPMS inversely affects synapse density and axon arbor formation in retinal ganglion cells in vivo. J. Neurosci. 2013, 33, 10384–10395. [Google Scholar] [CrossRef]
- Takahashi, T.; Fournier, A.; Nakamura, F.; Wang, L.H.; Murakami, Y.; Kalb, R.G.; Fujisawa, H.; Strittmatter, S.M. Plexin-neuropilin-1 complexes form functional semaphorin-3A receptors. Cell 1999, 99, 59–69. [Google Scholar] [CrossRef]
- Fiore, R.; Rajman, M.; Schwale, C.; Bicker, S.; Antoniou, A.; Bruehl, C.; Draguhn, A.; Schratt, G. MiR-134-dependent regulation of Pumilio-2 is necessary for homeostatic synaptic depression. EMBO J. 2014, 33, 2231–2246. [Google Scholar] [CrossRef] [PubMed]
- Hentze, M.W.; Castello, A.; Schwarzl, T.; Preiss, T. A brave new world of RNA-binding proteins. Nature Rev. Mol. Cell Biol. 2018, 19, 327–341. [Google Scholar] [CrossRef] [PubMed]
- O’Brien, J.; Hayder, H.; Zayed, Y.; Peng, C. Overview of MicroRNA Biogenesis, Mechanisms of Actions, and Circulation. Front. Endocrinol. 2018, 9, 402. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.; Chen, J.; Tang, C.X.; Zhou, X.Y.; Gao, D.S. Inverse Expression Levels of EphrinA3 and EphrinA5 Contribute to Dopaminergic Differentiation of Human SH-SY5Y Cells. J. Mol. Neurosci. 2016, 59, 483–492. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Bi, J.; Li, Z.; Li, Z.; Liu, X.; Kong, C. miR214 reduces cisplatin resistance by targeting netrin1 in bladder cancer cells. Int. J. Mol. Med. 2018, 41, 1765–1773. [Google Scholar] [CrossRef] [PubMed]
- Yang, T.; Huang, H.; Shao, Q.; Yee, S.; Majumder, T.; Liu, G. miR-92 Suppresses Robo1 Translation to Modulate Slit Sensitivity in Commissural Axon Guidance. Cell Rep. 2018, 24, 2694–2708.e2696. [Google Scholar] [CrossRef]
- Jiang, M.; Li, X.; Quan, X.; Li, X.; Zhou, B. MiR-92a Family: A Novel Diagnostic Biomarker and Potential Therapeutic Target in Human Cancers. Front. Mol. Biosci. 2019, 6, 98. [Google Scholar] [CrossRef]
- Wang, X.; Chen, Q.; Yi, S.; Liu, Q.; Zhang, R.; Wang, P.; Qian, T.; Li, S. Correction: The microRNAs let-7 and miR-9 down-regulate the axon-guidance genes Ntn1 and Dcc during peripheral nerve regeneration. J. Biol. Chem. 2019, 294, 6695. [Google Scholar] [CrossRef]
- Cuesta, S.; Restrepo-Lozano, J.M.; Silvestrin, S.; Nouel, D.; Torres-Berrio, A.; Reynolds, L.M.; Arvanitogiannis, A.; Flores, C. Non-Contingent Exposure to Amphetamine in Adolescence Recruits miR-218 to Regulate Dcc Expression in the VTA. Neuropsychopharmacology 2018, 43, 900–911. [Google Scholar] [CrossRef]
- Torres-Berrio, A.; Lopez, J.P.; Bagot, R.C.; Nouel, D.; Dal Bo, G.; Cuesta, S.; Zhu, L.; Manitt, C.; Eng, C.; Cooper, H.M.; et al. DCC Confers Susceptibility to Depression-like Behaviors in Humans and Mice and Is Regulated by miR-218. Biol. Psychiatry 2017, 81, 306–315. [Google Scholar] [CrossRef]
- Reynolds, L.M.; Pokinko, M.; Torres-Berrio, A.; Cuesta, S.; Lambert, L.C.; Del Cid Pellitero, E.; Wodzinski, M.; Manitt, C.; Krimpenfort, P.; Kolb, B.; et al. DCC Receptors Drive Prefrontal Cortex Maturation by Determining Dopamine Axon Targeting in Adolescence. Biol. Psychiatry 2018, 83, 181–192. [Google Scholar] [CrossRef] [PubMed]
- Vosberg, D.E.; Leyton, M.; Flores, C. The Netrin-1/DCC guidance system: Dopamine pathway maturation and psychiatric disorders emerging in adolescence. Mol. Psychiatry 2020, 25, 297–307. [Google Scholar] [CrossRef] [PubMed]
- Korsak, L.I.; Mitchell, M.E.; Shepard, K.A.; Akins, M.R. Regulation of neuronal gene expression by local axonal translation. Curr. Genet. Med. Rep. 2016, 4, 16–25. [Google Scholar] [CrossRef] [PubMed]
- Van Battum, E.Y.; Brignani, S.; Pasterkamp, R.J. Axon guidance proteins in neurological disorders. Lancet Neurol. 2015, 14, 532–546. [Google Scholar] [CrossRef]
- Li, Z.; Jagadapillai, R.; Gozal, E.; Barnes, G. Deletion of Semaphorin 3F in Interneurons Is Associated with Decreased GABAergic Neurons, Autism-like Behavior, and Increased Oxidative Stress Cascades. Mol. Neurobiol. 2019, 56, 5520–5538. [Google Scholar] [CrossRef]
- Ridge, P.G.; Hoyt, K.B.; Boehme, K.; Mukherjee, S.; Crane, P.K.; Haines, J.L.; Mayeux, R.; Farrer, L.A.; Pericak-Vance, M.A.; Schellenberg, G.D.; et al. Assessment of the genetic variance of late-onset Alzheimer’s disease. Neurobiol. Aging 2016, 41, 200.e213–200.e220. [Google Scholar] [CrossRef]
- Geschwind, D.H. Advances in autism. Annu. Rev. Med. 2009, 60, 367–380. [Google Scholar] [CrossRef]
- Zaslavsky, K.; Zhang, W.B.; McCready, F.P.; Rodrigues, D.C.; Deneault, E.; Loo, C.; Zhao, M.; Ross, P.J.; El Hajjar, J.; Romm, A.; et al. SHANK2 mutations associated with autism spectrum disorder cause hyperconnectivity of human neurons. Nat. Neurosci. 2019, 22, 556–564. [Google Scholar] [CrossRef]
- Grasselli, C.; Carbone, A.; Panelli, P.; Giambra, V.; Bossi, M.; Mazzoccoli, G.; De Filippis, L. Neural Stem Cells from Shank3-ko Mouse Model Autism Spectrum Disorders. Mol. Neurobiol. 2020, 57, 1502–1515. [Google Scholar] [CrossRef]
- Guo, B.; Chen, J.; Chen, Q.; Ren, K.; Feng, D.; Mao, H.; Yao, H.; Yang, J.; Liu, H.; Liu, Y.; et al. Anterior cingulate cortex dysfunction underlies social deficits in Shank3 mutant mice. Nat. Neurosci. 2019, 22, 1223–1234. [Google Scholar] [CrossRef]
- Zhou, Y.; Sharma, J.; Ke, Q.; Landman, R.; Yuan, J.; Chen, H.; Hayden, D.S.; Fisher, J.W., 3rd; Jiang, M.; Menegas, W.; et al. Atypical behaviour and connectivity in SHANK3-mutant macaques. Nature 2019, 570, 326–331. [Google Scholar] [CrossRef] [PubMed]
- Kang, M.-S.; Choi, T.-Y.; Ryu, H.G.; Lee, D.; Lee, S.-H.; Choi, S.-Y.; Kim, K.-T. Autism-like behavior caused by deletion of vaccinia-related kinase 3 is improved by TrkB stimulation. J. Exp. Med. 2017, 214, 2947–2966. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Lee, S.; Ryu, Y.J.; Lee, D.; Kim, S.; Seo, J.Y.; Oh, E.; Paek, S.H.; Kim, S.U.; Ha, C.M.; et al. Vaccinia-related kinase 2 plays a critical role in microglia-mediated synapse elimination during neurodevelopment. Glia 2019, 67, 1667–1679. [Google Scholar] [CrossRef]
- Weiss, L.A. Autism genetics: Emerging data from genome-wide copy-number and single nucleotide polymorphism scans. Expert Rev. Mol. Diagn. 2009, 9, 795–803. [Google Scholar] [CrossRef] [PubMed]
- Weiss, L.A.; Arking, D.E.; Gene Discovery Project of Johns, H.; the Autism, C.; Daly, M.J.; Chakravarti, A. A genome-wide linkage and association scan reveals novel loci for autism. Nature 2009, 461, 802–808. [Google Scholar] [CrossRef]
- Kim, S.A.; Kim, B.N.; Kim, J.W.; Shin, M.S.; Park, T.W.; Son, J.W.; Chung, U.S.; Park, M. Polymorphism in the Promoter Region of SEMA5A Is Associated with Sociality Traits in Korean Subjects with Autism Spectrum Disorders. Psychiatry Investig. 2017, 14, 876–878. [Google Scholar] [CrossRef]
- Mosca-Boidron, A.L.; Gueneau, L.; Huguet, G.; Goldenberg, A.; Henry, C.; Gigot, N.; Pallesi-Pocachard, E.; Falace, A.; Duplomb, L.; Thevenon, J.; et al. A de novo microdeletion of SEMA5A in a boy with autism spectrum disorder and intellectual disability. Eur. J. Hum. Genet. 2016, 24, 838–843. [Google Scholar] [CrossRef]
- Ziak, J.; Weissova, R.; Jerabkova, K.; Janikova, M.; Maimon, R.; Petrasek, T.; Pukajova, B.; Kleisnerova, M.; Wang, M.; Brill, M.S.; et al. CRMP2 mediates Sema3F-dependent axon pruning and dendritic spine remodeling. EMBO Rep. 2020, 21, e48512. [Google Scholar] [CrossRef]
- Duan, Y.; Wang, S.H.; Song, J.; Mironova, Y.; Ming, G.L.; Kolodkin, A.L.; Giger, R.J. Semaphorin 5A inhibits synaptogenesis in early postnatal- and adult-born hippocampal dentate granule cells. Elife 2014, 3. [Google Scholar] [CrossRef]
- Hilario, J.D.; Rodino-Klapac, L.R.; Wang, C.; Beattie, C.E. Semaphorin 5A is a bifunctional axon guidance cue for axial motoneurons in vivo. Dev. Biol. 2009, 326, 190–200. [Google Scholar] [CrossRef]
- Kantor, D.B.; Chivatakarn, O.; Peer, K.L.; Oster, S.F.; Inatani, M.; Hansen, M.J.; Flanagan, J.G.; Yamaguchi, Y.; Sretavan, D.W.; Giger, R.J.; et al. Semaphorin 5A is a bifunctional axon guidance cue regulated by heparan and chondroitin sulfate proteoglycans. Neuron 2004, 44, 961–975. [Google Scholar] [CrossRef]
- Bagri, A.; Cheng, H.J.; Yaron, A.; Pleasure, S.J.; Tessier-Lavigne, M. Stereotyped pruning of long hippocampal axon branches triggered by retraction inducers of the semaphorin family. Cell 2003, 113, 285–299. [Google Scholar] [CrossRef]
- Low, L.K.; Liu, X.B.; Faulkner, R.L.; Coble, J.; Cheng, H.J. Plexin signaling selectively regulates the stereotyped pruning of corticospinal axons from visual cortex. Proc. Natl. Acad. Sci. USA 2008, 105, 8136–8141. [Google Scholar] [CrossRef] [PubMed]
- Demyanenko, G.P.; Mohan, V.; Zhang, X.; Brennaman, L.H.; Dharbal, K.E.; Tran, T.S.; Manis, P.B.; Maness, P.F. Neural cell adhesion molecule NrCAM regulates Semaphorin 3F-induced dendritic spine remodeling. J. Neurosci. 2014, 34, 11274–11287. [Google Scholar] [CrossRef] [PubMed]
- Evans, T.L.; Blice-Baum, A.C.; Mihailescu, M.R. Analysis of the Fragile X mental retardation protein isoforms 1, 2 and 3 interactions with the G-quadruplex forming semaphorin 3F mRNA. Mol. Biosyst. 2012, 8, 642–649. [Google Scholar] [CrossRef] [PubMed]
- Matsuda, I.; Shoji, H.; Yamasaki, N.; Miyakawa, T.; Aiba, A. Comprehensive behavioral phenotyping of a new Semaphorin 3 F mutant mouse. Mol. Brain 2016, 9, 15. [Google Scholar] [CrossRef]
- Bondi, M.W.; Edmonds, E.C.; Salmon, D.P. Alzheimer’s Disease: Past, Present, and Future. J. Int. Neuropsychol. Soc. 2017, 23, 818–831. [Google Scholar] [CrossRef]
- Hardy, J.; Allsop, D. Amyloid deposition as the central event in the aetiology of Alzheimer’s disease. Trends. Pharmacol. Sci. 1991, 12, 383–388. [Google Scholar] [CrossRef]
- Chen, J.X.; Yan, S.D. Amyloid-beta-induced mitochondrial dysfunction. J. Alzheimers Dis. 2007, 12, 177–184. [Google Scholar] [CrossRef]
- Cheignon, C.; Tomas, M.; Bonnefont-Rousselot, D.; Faller, P.; Hureau, C.; Collin, F. Oxidative stress and the amyloid beta peptide in Alzheimer’s disease. Redox. Biol. 2018, 14, 450–464. [Google Scholar] [CrossRef]
- Shabani, M.; Haghani, M.; Tazangi, P.E.; Bayat, M.; Shid Moosavi, S.M.; Ranjbar, H. Netrin-1 improves the amyloid-beta-mediated suppression of memory and synaptic plasticity. Brain Res. Bull. 2017, 131, 107–116. [Google Scholar] [CrossRef] [PubMed]
- Spilman, P.R.; Corset, V.; Gorostiza, O.; Poksay, K.S.; Galvan, V.; Zhang, J.; Rao, R.; Peters-Libeu, C.; Vincelette, J.; McGeehan, A.; et al. Netrin-1 Interrupts Amyloid-beta Amplification, Increases sAbetaPPalpha in vitro and in vivo, and Improves Cognition in a Mouse Model of Alzheimer’s Disease. J. Alzheimers Dis. 2016, 52, 223–242. [Google Scholar] [CrossRef] [PubMed]
- Zamani, E.; Parviz, M.; Roghani, M.; Mohseni-Moghaddam, P. Key mechanisms underlying netrin-1 prevention of impaired spatial and object memory in Abeta1-42 CA1-injected rats. Clin. Exp. Pharmacol. Physiol. 2019, 46, 86–93. [Google Scholar] [CrossRef] [PubMed]
- Borel, F.; Marzocca, F.; Delcros, J.G.; Rama, N.; Mehlen, P.; Ferrer, J.L. Molecular characterization of Netrin-1 and APP receptor binding: New leads to block the progression of senile plaques in Alzheimer’s disease. Biochem. Biophys. Res. Commun. 2017, 488, 466–470. [Google Scholar] [CrossRef]
- Lourenco, F.C.; Galvan, V.; Fombonne, J.; Corset, V.; Llambi, F.; Muller, U.; Bredesen, D.E.; Mehlen, P. Netrin-1 interacts with amyloid precursor protein and regulates amyloid-beta production. Cell Death Differ. 2009, 16, 655–663. [Google Scholar] [CrossRef]
- Sun, L.; Ju, T.; Wang, T.; Zhang, L.; Ding, F.; Zhang, Y.; An, R.; Sun, Y.; Li, Y.; Lu, Y.; et al. Decreased Netrin-1 and Correlated Th17/Tregs Balance Disorder in Abeta1-42 Induced Alzheimer’s Disease Model Rats. Front. Aging Neurosci. 2019, 11, 124. [Google Scholar] [CrossRef]
| Regulated During: | Targeted Genes | Regulating Factors |
|---|---|---|
| Transcription | netrin-1 | Oct4 [95], Sox2 [95], NuRD [95], dFezf [96] |
| DCC | Islet [107], AP-1 [99] | |
| sema6a | Nrf2 [116] | |
| sema3e | RORα [97] | |
| sema3a | SetD5 [119] | |
| Post-transcription | neuropilin-1 | Hermes [26], PUM2 [128] |
| robo-1 | miR-92 [136] | |
| netrin-1 | let-7 [138] | |
| DCC | miR-9 [138], miR-218 [139,140] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, S.W.; Kim, K.-T. Expression of Genes Involved in Axon Guidance: How Much Have We Learned? Int. J. Mol. Sci. 2020, 21, 3566. https://doi.org/10.3390/ijms21103566
Kim SW, Kim K-T. Expression of Genes Involved in Axon Guidance: How Much Have We Learned? International Journal of Molecular Sciences. 2020; 21(10):3566. https://doi.org/10.3390/ijms21103566
Chicago/Turabian StyleKim, Sung Wook, and Kyong-Tai Kim. 2020. "Expression of Genes Involved in Axon Guidance: How Much Have We Learned?" International Journal of Molecular Sciences 21, no. 10: 3566. https://doi.org/10.3390/ijms21103566
APA StyleKim, S. W., & Kim, K.-T. (2020). Expression of Genes Involved in Axon Guidance: How Much Have We Learned? International Journal of Molecular Sciences, 21(10), 3566. https://doi.org/10.3390/ijms21103566






