Mangiferin Alleviates Ovalbumin-Induced Allergic Rhinitis via Nrf2/HO-1/NF-κB Signaling Pathways
Abstract
1. Introduction
2. Results
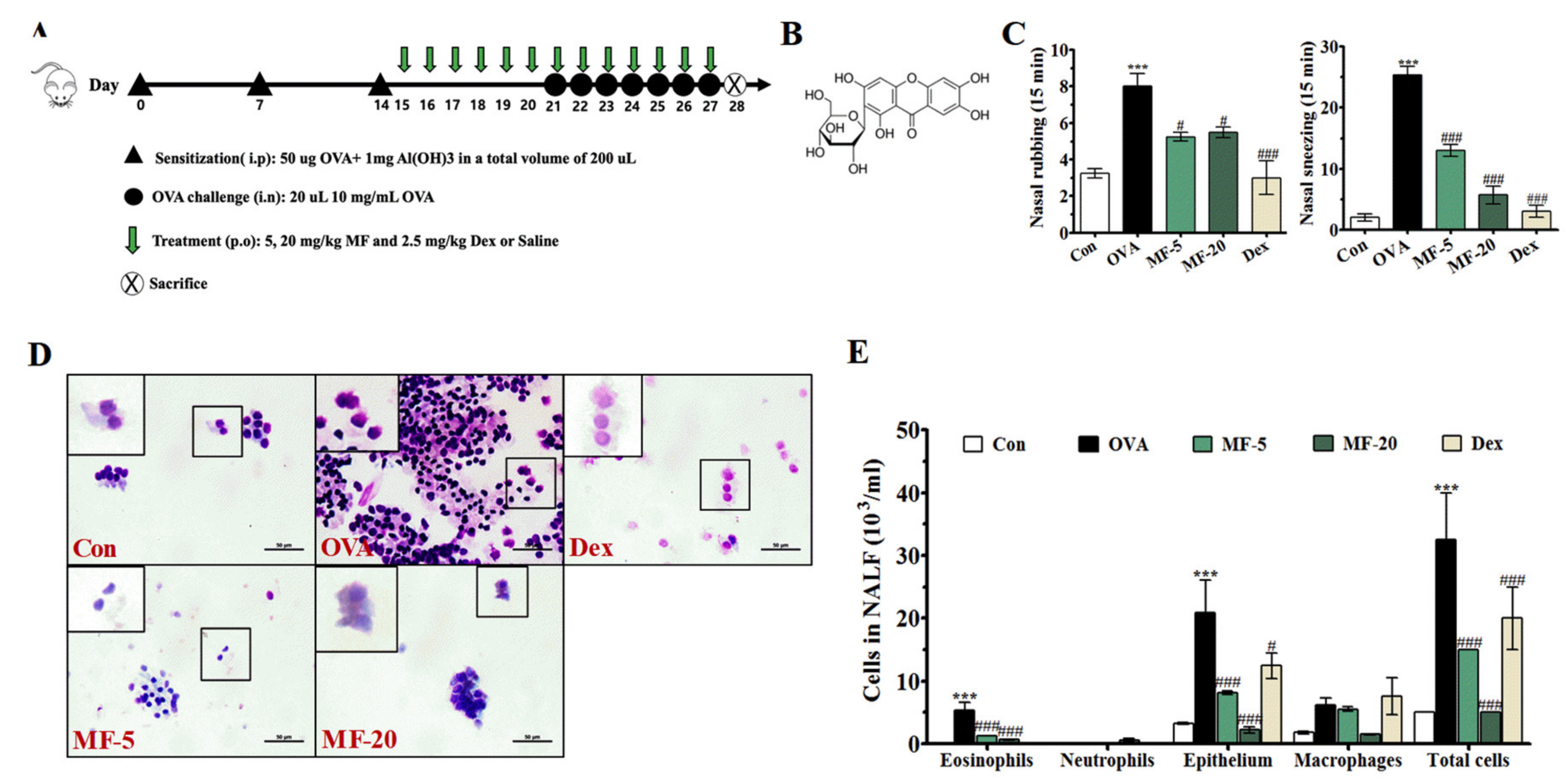
2.1. Effects of MF on Nasal Symptoms of OVA-Induced AR Mice
2.2. Effects of MF on the Infiltration of Inflammatory Cells in the NALF of OVA-induced AR Mice
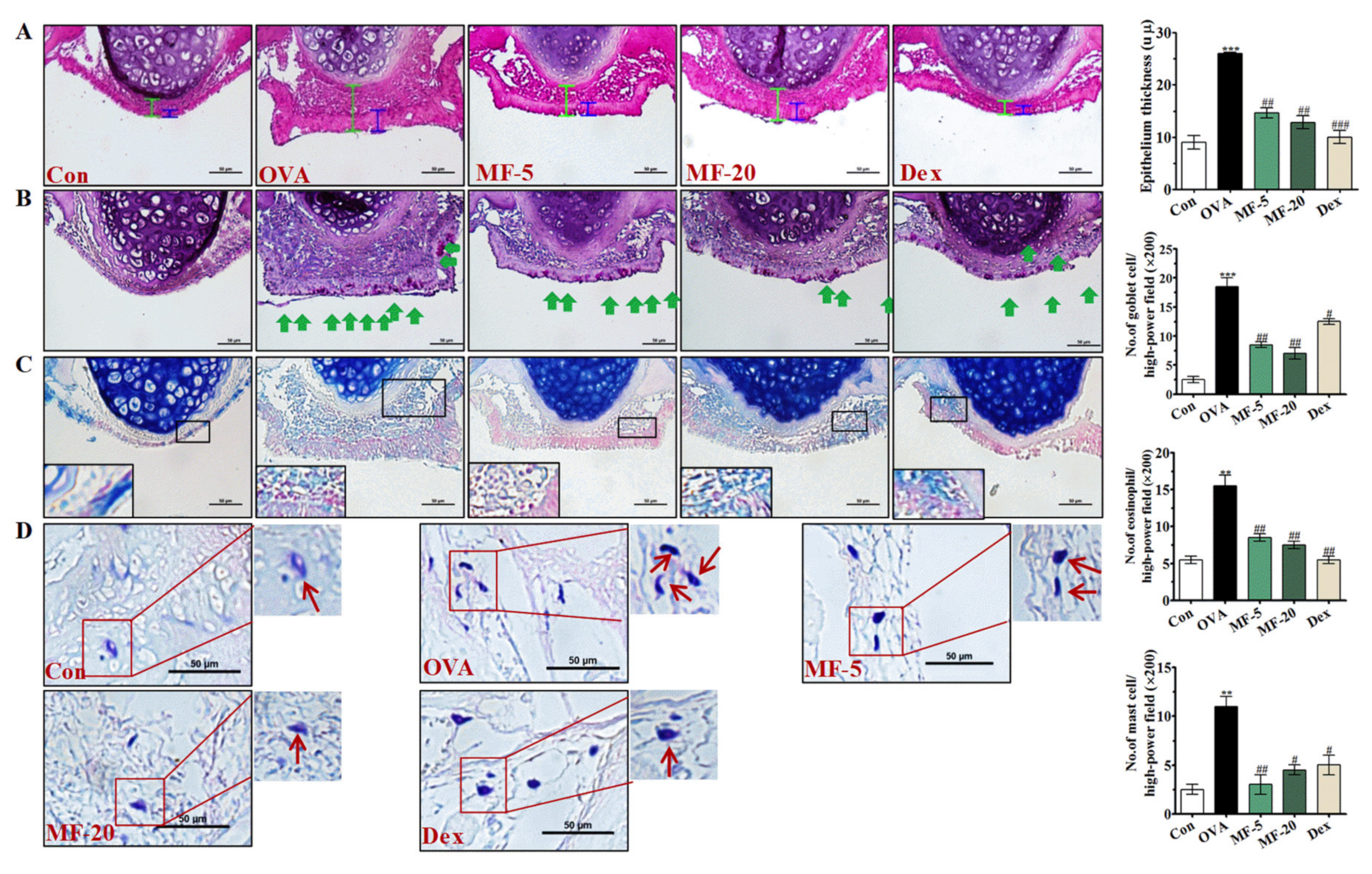
2.3. Effects of MF on the Histopathological Changes of the Nasal Mucosa of OVA-Induced AR Mice
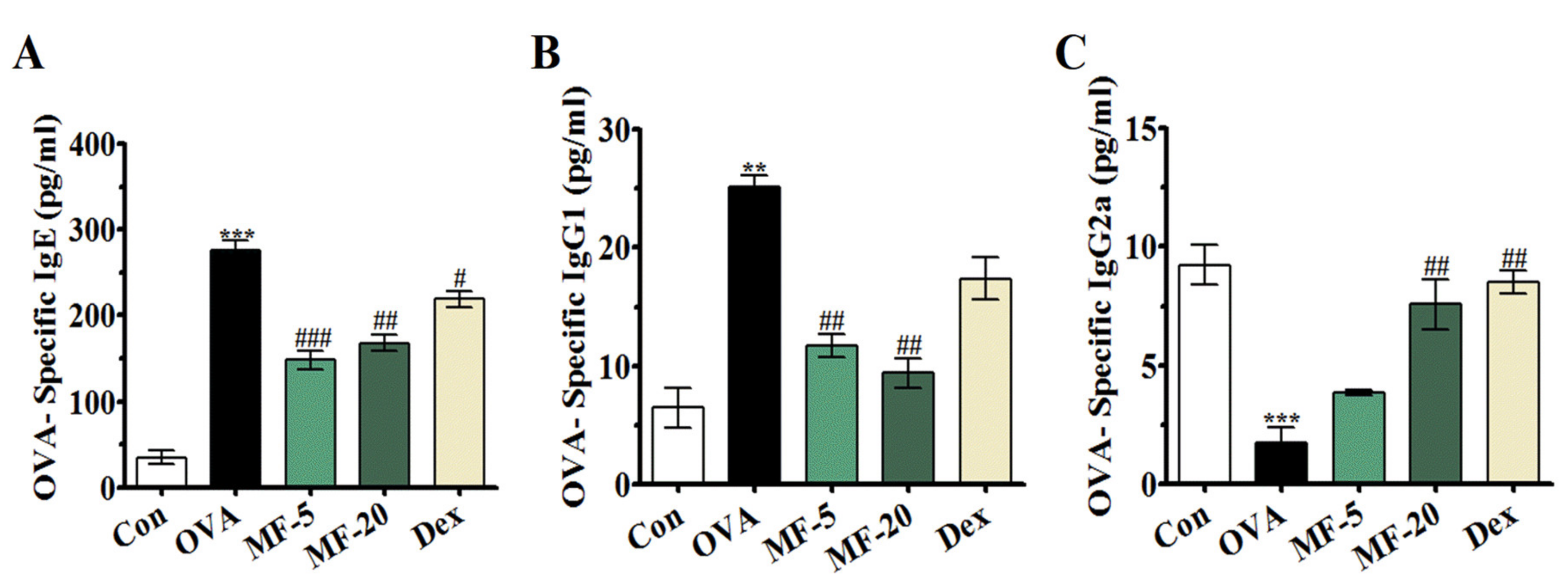
2.4. Effects of MF on the Serum Levels of OVA-Specific IgE, IgG1, and IgG2a of OVA-Induced AR Mice
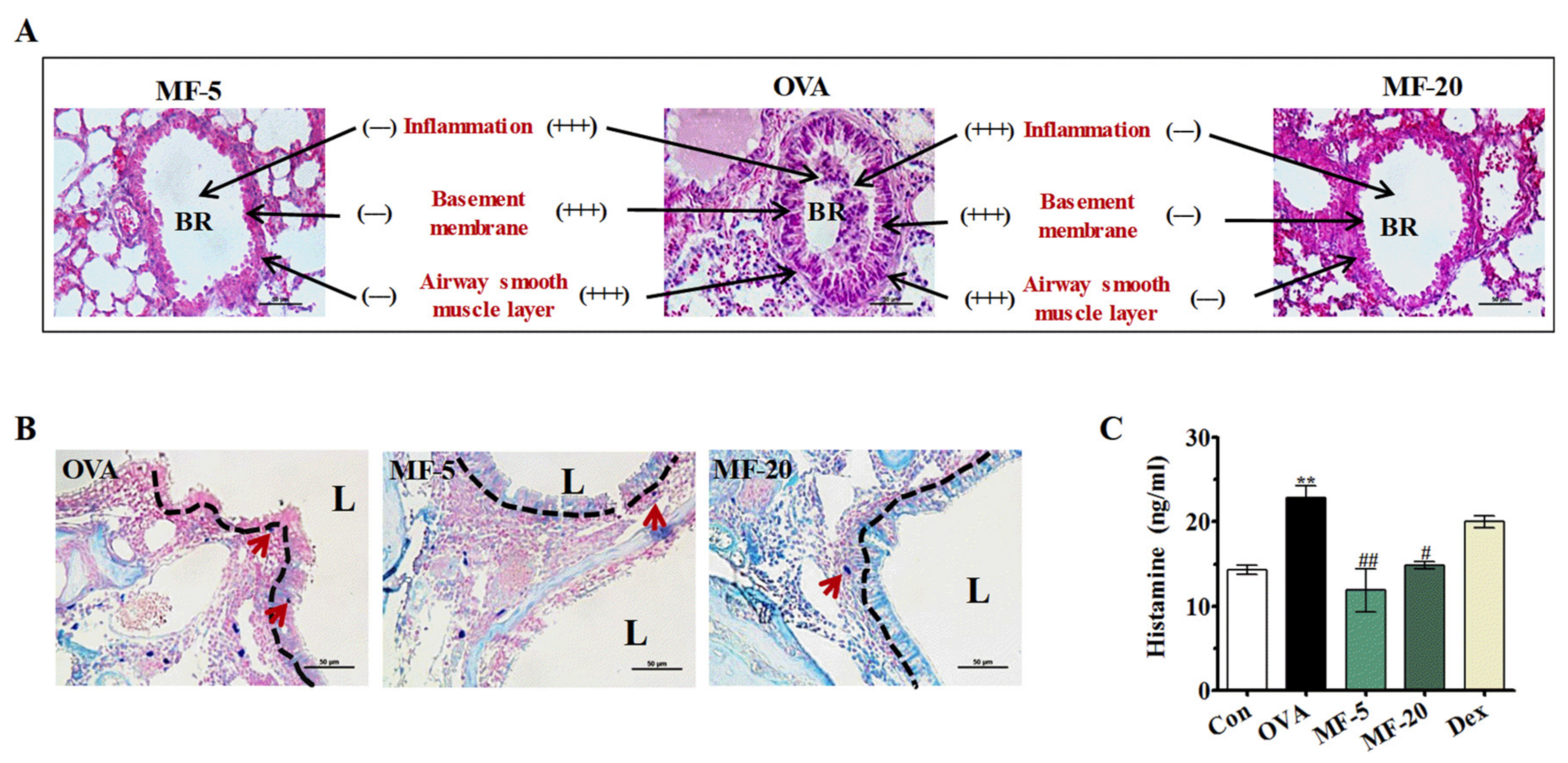
2.5. Effects of MF on Lung Histology and Mast Cell Migration in OVA-Induced AR Mice.
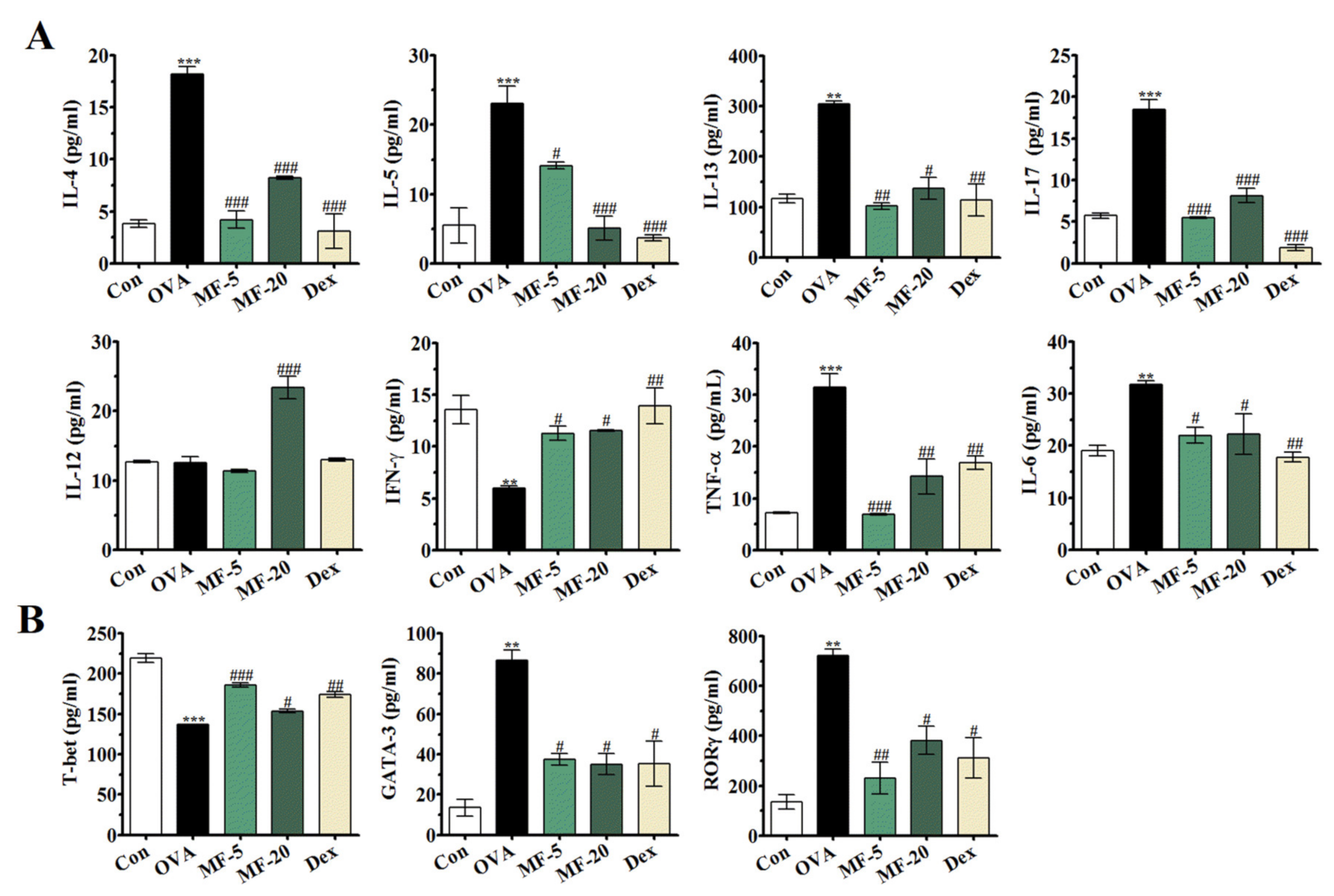
2.6. Effects of MF on the Levels of Th1, Th2, Th17 and Proinflammatory Cytokines, and Related Transcription Factors in OVA-induced AR Mice
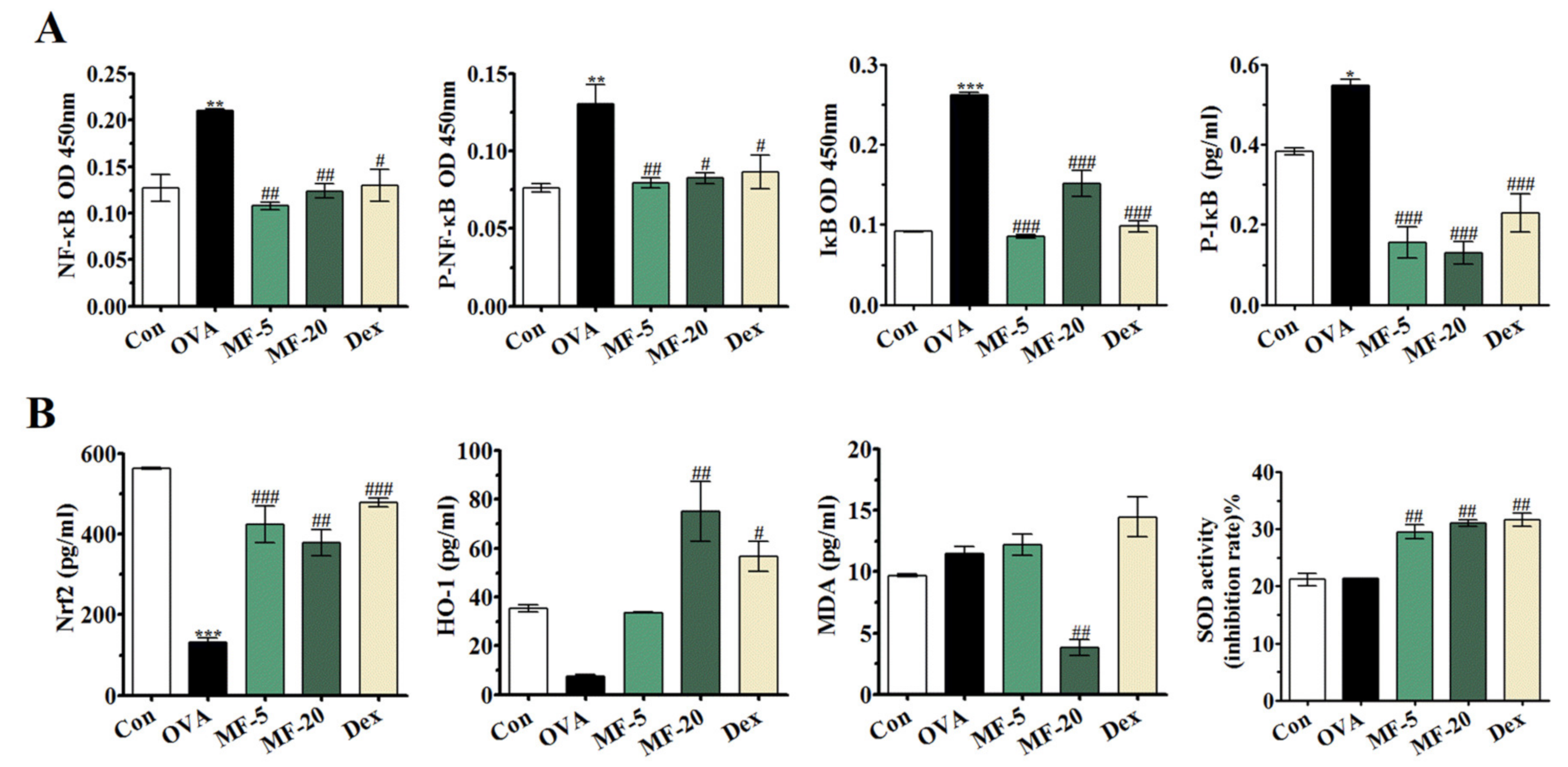
2.7. MF Inhibits OVA-Induced Activation of NF-κB Signaling in NALF
2.8. Effects of MF on Lipid Peroxidation and Antioxidant Enzyme Activity, and Nrf2/HO-1 Signaling Pathway
3. Discussion
4. Materials and Methods
4.1. Chemicals and Reagents
4.2. Animals
4.3. The OVA-induced AR Model
4.4. Lung Wet/Dry Weight Ratio
4.5. Nasal Symptom Scores
4.6. Collection and Measurement of Serum Levels of OVA-Specific Immunoglobulins
4.7. Collection and Analysis of NALF
4.8. Histopathological Evaluation of Nasal Mucosa and Lung Tissues
4.9. Assay of Lipid Peroxidation, Antioxidant Enzyme Activities, and Nrf2/HO-1 and NF-κB Signaling Pathways
4.10. Measurement of Th1/Th2/Th17 and Proinflammatory Cytokines and Related Transcription Factor Levels
4.11. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Pawankar, R.; Mori, S.; Ozu, C.; Kimura, S. Overview on the pathomechanisms of allergic rhinitis. Asia Pac. Allergy 2011, 1, 157–167. [Google Scholar] [CrossRef] [PubMed]
- Ercan, H.; Birben, E.; Dizdar, E.A.; Keskin, O.; Karaaslan, C.; Soyer, O.U.; Dut, R.; Sackesen, C.; Besler, T.; Kalayci, O. Oxidative stress and genetic and epidemiologic determinants of oxidant injury in childhood asthma. J. Allergy Clin. Immunol. 2006, 118, 1097–1104. [Google Scholar] [CrossRef] [PubMed]
- Marple, B.F. Allergic rhinitis and inflammatory airway disease: Interactions within the unified airspace. Am. J. Rhinol. Allergy 2010, 24, 249–254. [Google Scholar] [CrossRef] [PubMed]
- Harvey, A.L. Natural products in drug discovery. Drug Discov. Today 2008, 13, 894–901. [Google Scholar] [CrossRef] [PubMed]
- Pan, C.W.; Pan, Z.Z.; Hu, J.J.; Chen, W.L.; Zhou, G.Y.; Lin, W.; Jin, L.X.; Xu, C.L. Mangiferin alleviates lipopolysaccharide and D-galactosamine-induced acute liver injury by activating the Nrf2 pathway and inhibiting NLRP3 inflammasome activation. Eur. J. Pharmacol. 2016, 770, 85–91. [Google Scholar] [CrossRef]
- Pal, P.B.; Sinha, K.; Sil, P.C. Mangiferin, a natural xanthone, protects murine liver in Pb(II) induced hepatic damage and cell death via MAP kinase, NF-kappaB and mitochondria dependent pathways. PLoS ONE 2013, 8, e56894. [Google Scholar] [CrossRef]
- Saha, S.; Sadhukhan, P.; Sil, P.C. Mangiferin: A xanthonoid with multipotent anti-inflammatory potential. Biofactors 2016, 42, 459–474. [Google Scholar] [CrossRef]
- He, L.; Peng, X.; Zhu, J.; Chen, X.; Liu, H.; Tang, C.; Dong, Z.; Liu, F.; Peng, Y. Mangiferin attenuate sepsis-induced acute kidney injury via antioxidant and anti-inflammatory effects. Am. J. Nephrol. 2014, 40, 441–450. [Google Scholar] [CrossRef]
- Leiro, J.; Arranz, J.A.; Yanez, M.; Ubeira, F.M.; Sanmartin, M.L.; Orallo, F. Expression profiles of genes involved in the mouse nuclear factor-kappa B signal transduction pathway are modulated by mangiferin. Int. Immunopharmacol. 2004, 4, 763–778. [Google Scholar] [CrossRef]
- Wang, Z.; Guo, S.; Wang, J.; Shen, Y.; Zhang, J.; Wu, Q. Nrf2/HO-1 mediates the neuroprotective effect of mangiferin on early brain injury after subarachnoid hemorrhage by attenuating mitochondria-related apoptosis and neuroinflammation. Sci. Rep. 2017, 7, 11883. [Google Scholar] [CrossRef]
- Li, W.; Khor, T.O.; Xu, C.; Shen, G.; Jeong, W.S.; Yu, S.; Kong, A.N. Activation of Nrf2-antioxidant signaling attenuates NFkappaB-inflammatory response and elicits apoptosis. Biochem. Pharmacol. 2008, 76, 1485–1489. [Google Scholar] [CrossRef] [PubMed]
- Sanden, C.; Mori, M.; Jogdand, P.; Jonsson, J.; Krishnan, R.; Wang, X.; Erjefalt, J.S. Broad Th2 neutralization and anti-inflammatory action of pentosan polysulfate sodium in experimental allergic rhinitis. Immun. Inflamm. Dis. 2017, 5, 300–309. [Google Scholar] [CrossRef] [PubMed]
- Stone, K.D.; Prussin, C.; Metcalfe, D.D. IgE, mast cells, basophils, and eosinophils. J. Allergy Clin. Immunol. 2010, 125, S73–S80. [Google Scholar] [CrossRef] [PubMed]
- Melvin, T.A.; Ramanathan, M., Jr. Role of innate immunity in the pathogenesis of allergic rhinitis. Curr. Opin. Otolaryngol. Head Neck Surg. 2012, 20, 194–198. [Google Scholar] [CrossRef]
- Guo, L.Y.; Hung, T.M.; Bae, K.H.; Shin, E.M.; Zhou, H.Y.; Hong, Y.N.; Kang, S.S.; Kim, H.P.; Kim, Y.S. Anti-inflammatory effects of schisandrin isolated from the fruit of Schisandra chinensis Baill. Eur. J. Pharmacol. 2008, 591, 293–299. [Google Scholar] [CrossRef]
- Bui, T.T.; Piao, C.H.; Song, C.H.; Shin, H.S.; Chai, O.H. Bupleurum chinense extract ameliorates an OVA-induced murine allergic asthma through the reduction of the Th2 and Th17 cytokines production by inactivation of NFkappaB pathway. Biomed. Pharmacother. 2017, 91, 1085–1095. [Google Scholar] [CrossRef]
- Wei, D.Z.; Guo, X.Y.; Lin, L.N.; Lin, M.X.; Gong, Y.Q.; Ying, B.Y.; Huang, M.Y. Effects of Angelicin on Ovalbumin (OVA)-Induced Airway Inflammation in a Mouse Model of Asthma. Inflammation 2016, 39, 1876–1882. [Google Scholar] [CrossRef]
- Dar, A.; Faizi, S.; Naqvi, S.; Roome, T.; Zikr-ur-Rehman, S.; Ali, M.; Firdous, S.; Moin, S.T. Analgesic and antioxidant activity of mangiferin and its derivatives: The structure activity relationship. Biol. Pharm. Bull. 2005, 28, 596–600. [Google Scholar] [CrossRef]
- Rivera, D.G.; Hernandez, I.; Merino, N.; Luque, Y.; Alvarez, A.; Martin, Y.; Amador, A.; Nuevas, L.; Delgado, R. Mangifera indica L. extract (Vimang) and mangiferin reduce the airway inflammation and Th2 cytokines in murine model of allergic asthma. J. Pharm. Pharmacol. 2011, 63, 1336–1345. [Google Scholar] [CrossRef]
- Romagnani, S. Cytokines and chemoattractants in allergic inflammation. Mol. Immunol. 2002, 38, 881–885. [Google Scholar] [CrossRef]
- Minai-Fleminger, Y.; Levi-Schaffer, F. Mast cells and eosinophils: The two key effector cells in allergic inflammation. Inflamm. Res. 2009, 58, 631–638. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Zhang, W.; Shang, J.; Yang, J.; Zhang, L.; Bachert, C. Immunomodulatory effects of IL-23 and IL-17 in a mouse model of allergic rhinitis. Clin. Exp. Allergy 2013, 43, 956–966. [Google Scholar] [CrossRef] [PubMed]
- Bischoff, S.C. Role of mast cells in allergic and non-allergic immune responses: Comparison of human and murine data. Nat. Rev. Immunol. 2007, 7, 93–104. [Google Scholar] [CrossRef] [PubMed]
- Eifan, A.O.; Furukido, K.; Dumitru, A.; Jacobson, M.R.; Schmidt-Weber, C.; Banfield, G.; Durham, S.R.; Nouri-Aria, K.T. Reduced T-bet in addition to enhanced STAT6 and GATA3 expressing T cells contribute to human allergen-induced late responses. Clin. Exp. Allergy 2012, 42, 891–900. [Google Scholar] [CrossRef] [PubMed]
- Deo, S.S.; Mistry, K.J.; Kakade, A.M.; Niphadkar, P.V. Role played by Th2 type cytokines in IgE mediated allergy and asthma. Lung India 2010, 27, 66–71. [Google Scholar] [CrossRef]
- Coffman, R.L.; Seymour, B.W.; Hudak, S.; Jackson, J.; Rennick, D. Antibody to interleukin-5 inhibits helminth-induced eosinophilia in mice. Science 1989, 245, 308–310. [Google Scholar] [CrossRef]
- Grunig, G.; Warnock, M.; Wakil, A.E.; Venkayya, R.; Brombacher, F.; Rennick, D.M.; Sheppard, D.; Mohrs, M.; Donaldson, D.D.; Locksley, R.M.; et al. Requirement for IL-13 independently of IL-4 in experimental asthma. Science 1998, 282, 2261–2263. [Google Scholar] [CrossRef]
- Kang, O.H.; Jang, H.J.; Chae, H.S.; Oh, Y.C.; Choi, J.G.; Lee, Y.S.; Kim, J.H.; Kim, Y.C.; Sohn, D.H.; Park, H.; et al. Anti-inflammatory mechanisms of resveratrol in activated HMC-1 cells: Pivotal roles of NF-kappaB and MAPK. Pharmacol. Res. 2009, 59, 330–337. [Google Scholar] [CrossRef]
- Imran, M.; Arshad, M.S.; Butt, M.S.; Kwon, J.H.; Arshad, M.U.; Sultan, M.T. Mangiferin: A natural miracle bioactive compound against lifestyle related disorders. Lipids Health Dis. 2017, 16, 84. [Google Scholar] [CrossRef]
- Motohashi, H.; Yamamoto, M. Nrf2-Keap1 defines a physiologically important stress response mechanism. Trends Mol. Med. 2004, 10, 549–557. [Google Scholar] [CrossRef]
- Nguyen, T.; Nioi, P.; Pickett, C.B. The Nrf2-antioxidant response element signaling pathway and its activation by oxidative stress. J. Biol. Chem. 2009, 284, 13291–13295. [Google Scholar] [CrossRef] [PubMed]
- Das, J.; Ghosh, J.; Roy, A.; Sil, P.C. Mangiferin exerts hepatoprotective activity against D-galactosamine induced acute toxicity and oxidative/nitrosative stress via Nrf2-NFkappaB pathways. Toxicol. Appl. Pharmacol. 2012, 260, 35–47. [Google Scholar] [CrossRef] [PubMed]
- Mahmoud-Awny, M.; Attia, A.S.; Abd-Ellah, M.F.; El-Abhar, H.S. Mangiferin Mitigates Gastric Ulcer in Ischemia/ Reperfused Rats: Involvement of PPAR-gamma, NF-kappaB and Nrf2/HO-1 Signaling Pathways. PLoS ONE 2015, 10, e0132497. [Google Scholar] [CrossRef] [PubMed]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Piao, C.H.; Fan, Y.J.; Nguyen, T.V.; Song, C.H.; Chai, O.H. Mangiferin Alleviates Ovalbumin-Induced Allergic Rhinitis via Nrf2/HO-1/NF-κB Signaling Pathways. Int. J. Mol. Sci. 2020, 21, 3415. https://doi.org/10.3390/ijms21103415
Piao CH, Fan YJ, Nguyen TV, Song CH, Chai OH. Mangiferin Alleviates Ovalbumin-Induced Allergic Rhinitis via Nrf2/HO-1/NF-κB Signaling Pathways. International Journal of Molecular Sciences. 2020; 21(10):3415. https://doi.org/10.3390/ijms21103415
Chicago/Turabian StylePiao, Chun Hua, Yan Jing Fan, Thi Van Nguyen, Chang Ho Song, and Ok Hee Chai. 2020. "Mangiferin Alleviates Ovalbumin-Induced Allergic Rhinitis via Nrf2/HO-1/NF-κB Signaling Pathways" International Journal of Molecular Sciences 21, no. 10: 3415. https://doi.org/10.3390/ijms21103415
APA StylePiao, C. H., Fan, Y. J., Nguyen, T. V., Song, C. H., & Chai, O. H. (2020). Mangiferin Alleviates Ovalbumin-Induced Allergic Rhinitis via Nrf2/HO-1/NF-κB Signaling Pathways. International Journal of Molecular Sciences, 21(10), 3415. https://doi.org/10.3390/ijms21103415





