TRP Channels in the Focus of Trigeminal Nociceptor Sensitization Contributing to Primary Headaches
Abstract
1. Anatomical Basis of Headache Generation
2. Morphology and Role of Meningeal Nociceptors in Headache Generation
3. The Trigeminal Ganglion Neuron as a Model for Its Peripheral and Central Terminals
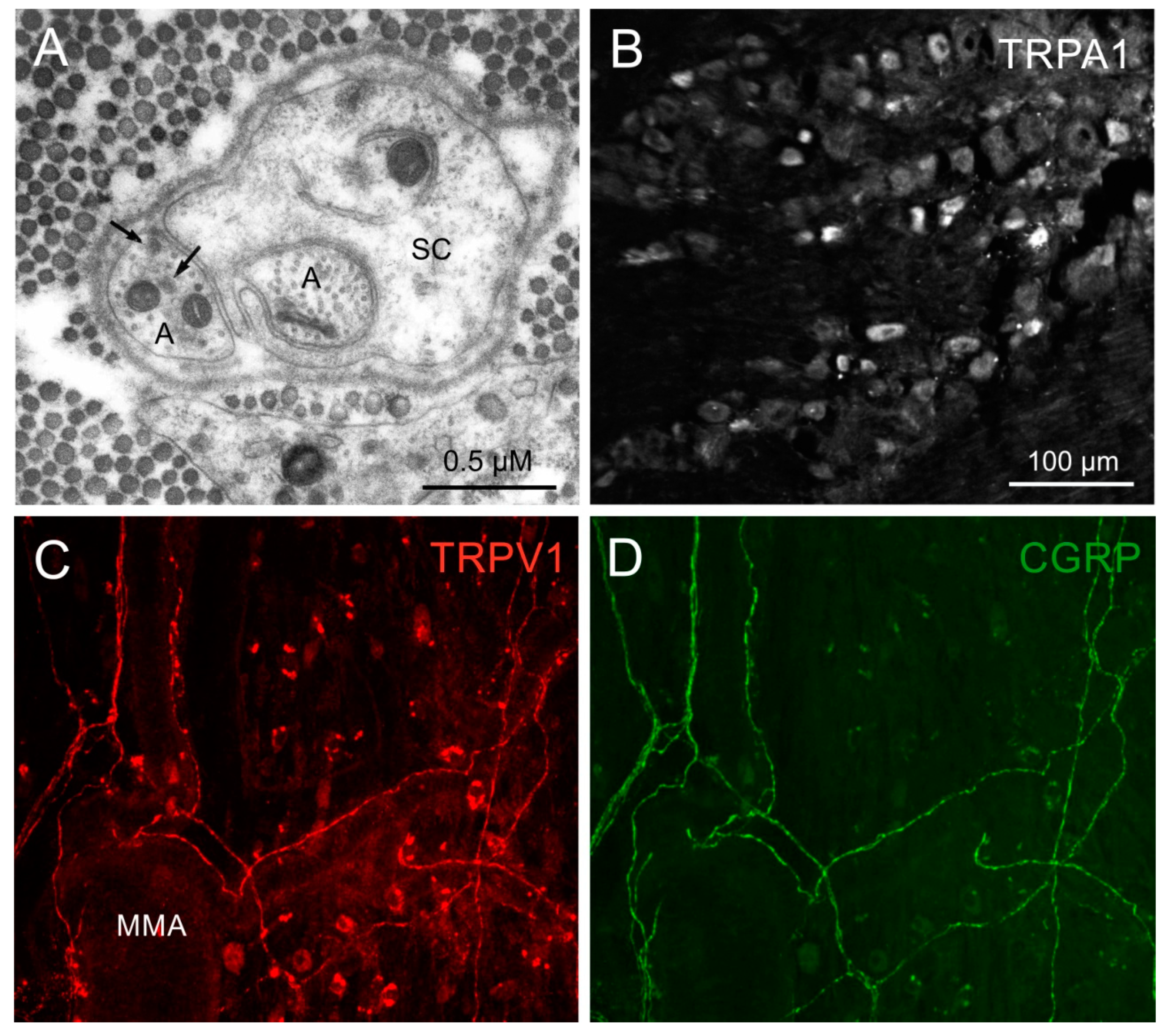
3.1. Immunohistochemistry
3.2. Neuropeptide Release
3.3. Electrophysiological Recordings
3.4. Calcium Imaging
4. Expression of TRPV1 and TRPA1 and Co-Expression with Neuropeptides
5. Functional Significance of Mediator Trafficking to Afferent Fiber Terminals
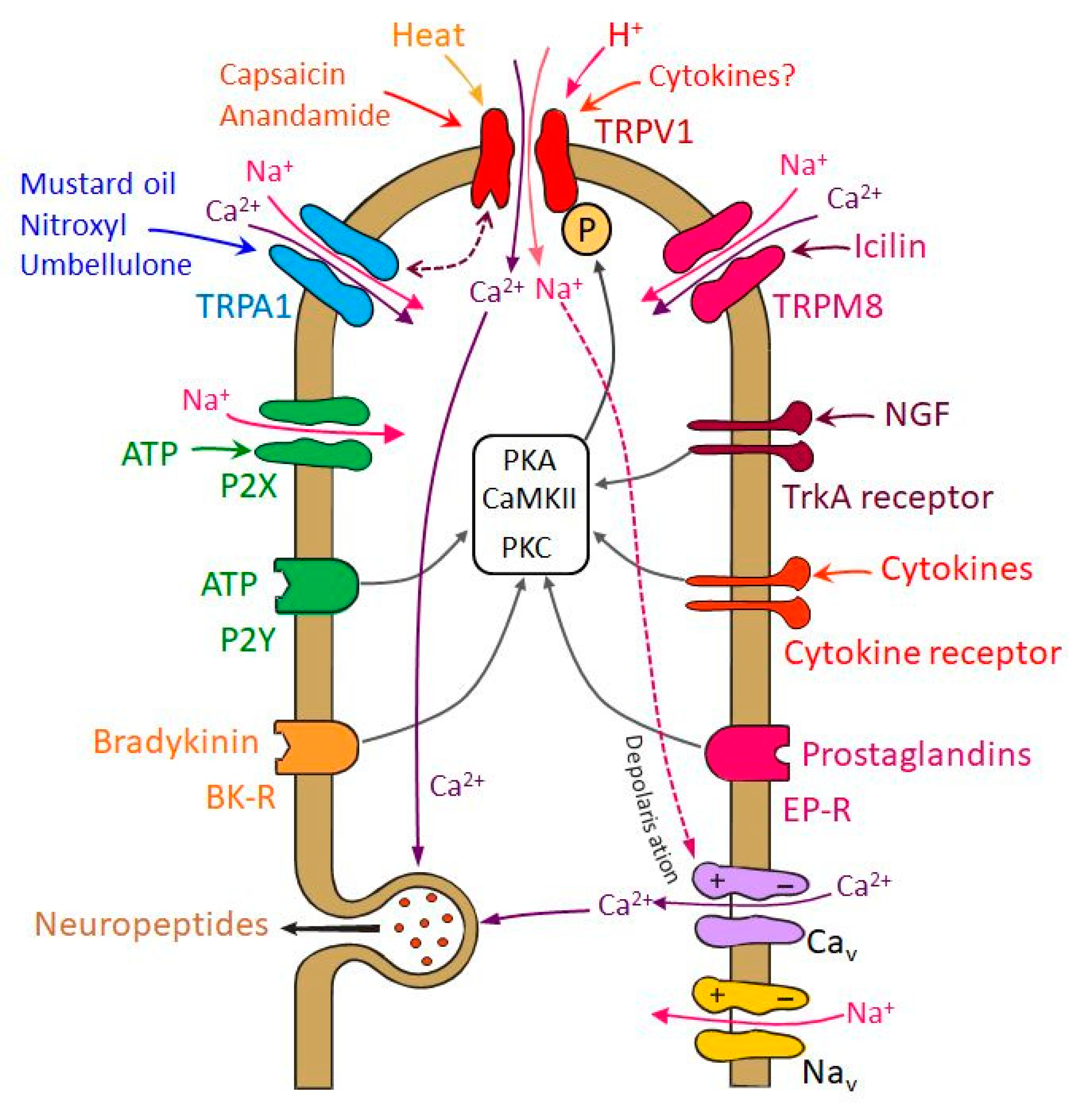
6. Signals Sensitizing Trigeminal Nociceptors—Involvement and Interaction of TRP Receptors
7. Sensitization of Trigeminal Nociceptors Following Cortical Spreading Depression
8. Role of Metabolic States in Trigeminal Sensitization
9. Intraganglionic Mechanisms Involved in Nociceptor Sensitization
10. Sensitization Processes within the Trigeminovascular System
11. Medication-Induced Trigeminal Nociception and Pain
12. Synopsis: Relevance of TRPV1 and TRPA1 in Primary Headaches
Funding
Conflicts of Interest
References
- Bernstein, C.; Burstein, R. Sensitization of the Trigeminovascular Pathway: Perspective and Implications to Migraine Pathophysiology. J. Clin. Neurol. 2012, 8, 89–99. [Google Scholar] [CrossRef] [PubMed]
- Mickle, A.D.; Shepherd, A.J.; Mohapatra, D.P. Sensory TRP Channels: The Key Transducers of Nociception and Pain. Prog. Mol. Biol. Transl. Sci. 2015, 131, 73–118. [Google Scholar] [PubMed]
- Bolay, H.; Messlinger, K.; Dux, M.; Akcali, D. Anatomy of headache. In Pathophysiology of Headaches, 1st ed.; Ashina, M., Geppetti, P., Eds.; Springer International Publishing: Cham, Switzerland, 2015; pp. 1–29. ISBN 978-3-319-15621-7. [Google Scholar]
- Messlinger, K.; Dux, M. Functional anatomy of trigeminovascular pain. In Neurobiological Basis of Migraine; Dalkara, T., Moskowitz, M., Eds.; John Wiley & Sons Inc.: Hoboken, USA, 2017; pp. 3–29. ISBN 978-1-118-96719-5. [Google Scholar]
- Goadsby, P.J.; Holland, P.R.; Martins-Oliveira, M.; Hoffmann, J.; Schankin, C.; Akerman, S. Pathophysiology of Migraine: A Disorder of Sensory Processing. Physiol. Rev. 2017, 97, 553–622. [Google Scholar] [CrossRef] [PubMed]
- Penfield, W.; McNaughton, M. Dural headache and innervation of the dura mater. Arch. Neurol. Psychiatr. 1940, 44, 43–75. [Google Scholar] [CrossRef]
- Ray, B.S.; Wolff, H.G. Experimental studies on headache: Pain sensitive structures of the head and their significance in headache. Arch. Surg. 1940, 1, 813–856. [Google Scholar] [CrossRef]
- Messlinger, K.; Hanesch, U.; Baumgärtel, M.; Trost, B.; Schmidt, R.F. Innervation of the dura mater encephali of cat and rat: Ultrastructure and calcitonin gene-related peptide-like and substance P-like immunoreactivity. Anat. Embryol. 1993, 188, 219–237. [Google Scholar] [CrossRef]
- Fricke, B.; von Düring, M.; Andres, K.H. Topography and immunocytochemical characterization of nerve fibers in the leptomeningeal compartments of the rat. A light- and electron-microscopical study. Cell Tissue Res. 1997, 287, 11–22. [Google Scholar] [CrossRef]
- Fontaine, D.; Almairac, F.; Santucci, S.; Fernandez, C.; Dallel, R.; Pallud, J.; Lanteri-Minet, M. Dural and pial pain-sensitive structures in humans: New inputs from awake craniotomies. Brain 2018, 141, 1040–1048. [Google Scholar] [CrossRef]
- Graham, J.R.; Wolff, H.G. Mechanism of migraine headache and action of ergotamine tartrate. Arch. Neurol Psychiatr. 1938, 39, 737–763. [Google Scholar] [CrossRef]
- Schueler, M.; Messlinger, K.; Dux, M.; Neuhuber, W.L.; De Col, R. Extracranial projections of meningeal afferents and their impact on meningeal nociception and headache. Pain 2013, 154, 1622–1631. [Google Scholar] [CrossRef]
- Zhao, J.; Levy, D. The sensory innervation of the calvarial periosteum is nociceptive and contributes to headache-like behavior. Pain 2014, 155, 1392–1400. [Google Scholar] [CrossRef] [PubMed]
- Andres, K.H.; von Düring, M.; Muszynski, K.; Schmidt, R.F. Nerve fibres and their terminals of the dura mater encephali of the rat. Anat. Embryol. 1987, 175, 289–301. [Google Scholar] [CrossRef] [PubMed]
- Abdo, H.; Calvo-Enrique, L.; Lopez, J.M.; Song, J.; Zhang, M.-D.; Usoskin, D.; El Manira, A.; Adameyko, I.; Hjerling-Leffler, J.; Ernfors, P. Specialized cutaneous Schwann cells initiate pain sensation. Science 2019, 365, 695–699. [Google Scholar] [CrossRef] [PubMed]
- Heppelmann, B.; Messlinger, K.; Neiss, W.F.; Schmidt, R.F. Ultrastructural three-dimensional reconstruction of group III and group IV sensory nerve endings (free nerve endings) in the knee joint capsule of the cat: Evidence for multiple receptive sites. J. Comp. Neurol. 1990, 292, 103–116. [Google Scholar] [CrossRef]
- Messlinger, K. Functional Morphology of Nociceptive and Other Fine Sensory Endings (Free Nerve Endings) in Different Tissues. In The Polymodal Receptor—a Gateway to Pathological Pain; Elsevier: Amsterdam, Netherlands, 1996; pp. 273–298. [Google Scholar]
- Munger, B.L.; Ide, C. The structure and function of cutaneous sensory receptors. Arch. Histol. Cytol. 1988, 51, 1–34. [Google Scholar] [CrossRef]
- Byers, M.R. Sensory innervation of periodontal ligament of rat molars consists of unencapsulated Ruffini-like mechanoreceptors and free nerve endings. J. Comp. Neurol. 1985, 231, 500–518. [Google Scholar] [CrossRef]
- Von Düring, M.; Andres, K.H. Sensory nerve fiber terminals in the arachnoid granulations of non-human primates. Neurosci. Lett. 1991, 127, 121–124. [Google Scholar] [CrossRef]
- Strassman, A.M.; Raymond, S.A.; Burstein, R. Sensitization of meningeal sensory neurons and the origin of headaches. Nature 1996, 384, 560–564. [Google Scholar] [CrossRef]
- Schepelmann, K.; Ebersberger, A.; Pawlak, M.; Oppmann, M.; Messlinger, K. Response properties of trigeminal brain stem neurons with input from dura mater encephali in the rat. Neuroscience 1999, 90, 543–554. [Google Scholar] [CrossRef]
- Zhang, X.; Levy, D.; Noseda, R.; Kainz, V.; Jakubowski, M.; Burstein, R. Activation of meningeal nociceptors by cortical spreading depression: Implications for migraine with aura. J. Neurosci. 2010, 30, 8807–8814. [Google Scholar] [CrossRef]
- Olesen, J.; Burstein, R.; Ashina, M.; Tfelt-Hansen, P. Origin of pain in migraine: Evidence for peripheral sensitisation. Lancet Neurol. 2009, 8, 679–690. [Google Scholar] [CrossRef]
- Roch, M.; Messlinger, K.; Kulchitsky, V.; Tichonovich, O.; Azev, O.; Koulchitsky, S. Ongoing activity in trigeminal wide-dynamic range neurons is driven from the periphery. Neuroscience 2007, 150, 681–691. [Google Scholar] [CrossRef] [PubMed]
- Cabanes, C.; Viana, F.; Belmonte, C. Differential thermosensitivity of sensory neurons in the guinea pig trigeminal ganglion. J. Neurophysiol. 2003, 90, 2219–2231. [Google Scholar] [CrossRef][Green Version]
- De Col, R.; Messlinger, K.; Carr, R.W. Repetitive activity slows axonal conduction velocity and concomitantly increases mechanical activation threshold in single axons of the rat cranial dura. J. Physiol. 2012, 590, 725–736. [Google Scholar] [CrossRef] [PubMed]
- Levy, D.; Burstein, R.; Strassman, A.M. Calcitonin gene-related peptide does not excite or sensitize meningeal nociceptors: Implications for the pathophysiology of migraine. Ann. Neurol. 2005, 58, 698–705. [Google Scholar] [CrossRef] [PubMed]
- Yan, J.; Wei, X.; Bischoff, C.; Edelmayer, R.M.; Dussor, G. pH-evoked dural afferent signaling is mediated by ASIC3 and is sensitized by mast cell mediators. Headache 2013, 53, 1250–1261. [Google Scholar] [CrossRef]
- Wiesenfeld-Hallin, Z.; Hökfelt, T.; Lundberg, J.M.; Forssmann, W.G.; Reinecke, M.; Tschopp, F.A.; Fischer, J.A. Immunoreactive calcitonin gene-related peptide and substance P coexist in sensory neurons to the spinal cord and interact in spinal behavioral responses of the rat. Neurosci. Lett. 1984, 52, 199–204. [Google Scholar] [CrossRef]
- Lee, Y.; Kawai, Y.; Shiosaka, S.; Takami, K.; Kiyama, H.; Hillyard, C.J.; Girgis, S.; MacIntyre, I.; Emson, P.C.; Tohyama, M. Coexistence of calcitonin gene-related peptide and substance P-like peptide in single cells of the trigeminal ganglion of the rat: Immunohistochemical analysis. Brain Res. 1985, 330, 194–196. [Google Scholar] [CrossRef]
- Eftekhari, S.; Salvatore, C.A.; Calamari, A.; Kane, S.A.; Tajti, J.; Edvinsson, L. Differential distribution of calcitonin gene-related peptide and its receptor components in the human trigeminal ganglion. Neuroscience 2010, 169, 683–696. [Google Scholar] [CrossRef]
- Quartu, M.; Serra, M.P.; Boi, M.; Poddighe, L.; Picci, C.; Demontis, R.; Del Fiacco, M. TRPV1 receptor in the human trigeminal ganglion and spinal nucleus: Immunohistochemical localization and comparison with the neuropeptides CGRP and SP. J. Anat. 2016, 229, 755–767. [Google Scholar] [CrossRef]
- Martins, D.O.; Santos, F.M.; Britto, L.R.G.; Lemos, J.B.D.; Chacur, M. Neurochemical effects of photobiostimulation in the trigeminal ganglion after inferior alveolar nerve injury. J. Biol. Regul. Homeost. Agents 2017, 31, 147–152. [Google Scholar]
- Kageneck, C.; Nixdorf-Bergweiler, B.E.; Messlinger, K.; Fischer, M.J. Release of CGRP from mouse brainstem slices indicates central inhibitory effect of triptans and kynurenate. J. Headache Pain 2014, 15, 7. [Google Scholar] [CrossRef] [PubMed]
- Dux, M.; Babes, A.; Manchen, J.; Sertel-Nakajima, J.; Vogler, B.; Schramm, J.; Messlinger, K. High-dose phenylephrine increases meningeal blood flow through TRPV1 receptor activation and release of calcitonin gene-related peptide. Eur. J. Pain 2019. [Google Scholar] [CrossRef] [PubMed]
- Thalakoti, S.; Patil, V.V.; Damodaram, S.; Vause, C.V.; Langford, L.E.; Freeman, S.E.; Durham, P.L. Neuron-glia signaling in trigeminal ganglion: Implications for migraine pathology. Headache 2007, 47, 1008–1023, discussion 24–25. [Google Scholar] [CrossRef] [PubMed]
- Meng, J.; Ovsepian, S.V.; Wang, J.; Pickering, M.; Sasse, A.; Aoki, K.R.; Lawrence, G.W.; Dolly, J.O. Activation of TRPV1 mediates calcitonin gene-related peptide release, which excites trigeminal sensory neurons and is attenuated by a retargeted botulinum toxin with anti-nociceptive potential. J. Neurosci. 2009, 29, 4981–4992. [Google Scholar] [CrossRef]
- Vellani, V.; Moschetti, G.; Franchi, S.; Giacomoni, C.; Sacerdote, P.; Amodeo, G. Effects of NSAIDs on the Release of Calcitonin Gene-Related Peptide and Prostaglandin E2 from Rat Trigeminal Ganglia. Mediat. Inflamm. 2017, 2017, 9547056. [Google Scholar] [CrossRef]
- Lehmann, R.; Schöbel, N.; Hatt, H.; van Thriel, C. The involvement of TRP channels in sensory irritation: A mechanistic approach toward a better understanding of the biological effects of local irritants. Arch. Toxicol. 2016, 90, 1399–1413. [Google Scholar] [CrossRef]
- Klein, A.H.; Joe, C.L.; Davoodi, A.; Takechi, K.; Carstens, M.I.; Carstens, E. Eugenol and carvacrol excite first- and second-order trigeminal neurons and enhance their heat-evoked responses. Neuroscience 2014, 271, 45–55. [Google Scholar] [CrossRef]
- Lee, P.R.; Lee, J.Y.; Kim, H.B.; Lee, J.H.; Oh, S.B. TRPM8 Mediates Hyperosmotic Stimuli-Induced Nociception in Dental Afferents. J. Dent. Res. 2019, 99, 107–114. [Google Scholar] [CrossRef]
- Huang, D.; Li, S.; Dhaka, A.; Story, G.M.; Cao, Y.-Q. Expression of the transient receptor potential channels TRPV1, TRPA1 and TRPM8 in mouse trigeminal primary afferent neurons innervating the dura. Mol. Pain 2012, 8, 66. [Google Scholar] [CrossRef]
- Geppetti, P.; Benemei, S.; De Cesaris, F. CGRP receptors and TRP channels in migraine. J. Headache Pain 2015, 16, A21. [Google Scholar] [CrossRef] [PubMed]
- Raisinghani, M.; Zhong, L.; Jeffry, J.A.; Bishnoi, M.; Pabbidi, R.M.; Pimentel, F.; Cao, D.-S.; Evans, M.S.; Premkumar, L.S. Activation characteristics of transient receptor potential ankyrin 1 and its role in nociception. Am. J. Physiol. Cell Physiol. 2011, 301, C587–C600. [Google Scholar] [CrossRef] [PubMed]
- Vedder, H.; Affolter, H.U.; Otten, U. Nerve growth factor (NGF) regulates tachykinin gene expression and biosynthesis in rat sensory neurons during early postnatal development. Neuropeptides 1993, 24, 351–357. [Google Scholar] [CrossRef]
- Price, T.J.; Louria, M.D.; Candelario-Soto, D.; Dussor, G.O.; Jeske, N.A.; Patwardhan, A.M.; Diogenes, A.; Trott, A.A.; Hargreaves, K.M.; Flores, C.M. Treatment of trigeminal ganglion neurons in vitro with NGF, GDNF or BDNF: Effects on neuronal survival, neurochemical properties and TRPV1-mediated neuropeptide secretion. BMC Neurosci. 2005, 6, 4. [Google Scholar] [CrossRef]
- Matsuka, Y.; Edmonds, B.; Mitrirattanakul, S.; Schweizer, F.E.; Spigelman, I. Two types of neurotransmitter release patterns in isolectin B4-positive and negative trigeminal ganglion neurons. Neuroscience 2007, 144, 665–674. [Google Scholar] [CrossRef]
- Price, T.J.; Flores, C.M. Critical evaluation of the colocalization between calcitonin gene-related peptide, substance P, transient receptor potential vanilloid subfamily type 1 immunoreactivities, and isolectin B4 binding in primary afferent neurons of the rat and mouse. J. Pain 2007, 8, 263–272. [Google Scholar] [CrossRef]
- Price, T.J.; Patwardhan, A.; Akopian, A.N.; Hargreaves, K.M.; Flores, C.M. Modulation of trigeminal sensory neuron activity by the dual cannabinoid-vanilloid agonists anandamide, N-arachidonoyl-dopamine and arachidonyl-2-chloroethylamide. Br. J. Pharmacol. 2004, 141, 1118–1130. [Google Scholar] [CrossRef]
- Jordt, S.-E.; Bautista, D.M.; Chuang, H.-H.; McKemy, D.D.; Zygmunt, P.M.; Högestätt, E.D.; Meng, I.D.; Julius, D. Mustard oils and cannabinoids excite sensory nerve fibres through the TRP channel ANKTM1. Nature 2004, 427, 260–265. [Google Scholar] [CrossRef]
- Salas, M.M.; Hargreaves, K.M.; Akopian, A.N. TRPA1-mediated responses in trigeminal sensory neurons: Interaction between TRPA1 and TRPV1. Eur. J. Neurosci. 2009, 29, 1568–1578. [Google Scholar] [CrossRef]
- Benemei, S.; De Cesaris, F.; Fusi, C.; Rossi, E.; Lupi, C.; Geppetti, P. TRPA1 and other TRP channels in migraine. J. Headache Pain 2013, 14, 71. [Google Scholar] [CrossRef]
- Nassini, R.; Materazzi, S.; Vriens, J.; Prenen, J.; Benemei, S.; De Siena, G.; la Marca, G.; Andrè, E.; Preti, D.; Avonto, C.; et al. The “headache tree” via umbellulone and TRPA1 activates the trigeminovascular system. Brain 2012, 135, 376–390. [Google Scholar] [CrossRef]
- Denner, A.C.; Vogler, B.; Messlinger, K.; De Col, R. Role of transient receptor potential ankyrin 1 receptors in rodent models of meningeal nociception-Experiments in vitro. Eur. J. Pain 2017, 21, 843–854. [Google Scholar] [CrossRef] [PubMed]
- Teicher, C.; De Col, R.; Messlinger, K. Hydrogen Sulfide Mediating both Excitatory and Inhibitory Effects in a Rat Model of Meningeal Nociception and Headache Generation. Front. Neurol. 2017, 8, 336. [Google Scholar] [CrossRef] [PubMed]
- Bautista, D.M.; Siemens, J.; Glazer, J.M.; Tsuruda, P.R.; Basbaum, A.I.; Stucky, C.L.; Jordt, S.-E.; Julius, D. The menthol receptor TRPM8 is the principal detector of environmental cold. Nature 2007, 448, 204–208. [Google Scholar] [CrossRef]
- Dussor, G.; Cao, Y.-Q. TRPM8 and Migraine. Headache 2016, 56, 1406–1417. [Google Scholar] [CrossRef] [PubMed]
- Moskowitz, M.A. Neurogenic inflammation in the pathophysiology and treatment of migraine. Neurology 1993, 43, S16–S20. [Google Scholar] [PubMed]
- Williamson, D.J.; Hargreaves, R.J.; Hill, R.G.; Shepheard, S.L. Sumatriptan inhibits neurogenic vasodilation of dural blood vessels in the anaesthetized rat—intravital microscope studies. Cephalalgia 1997, 17, 525–531. [Google Scholar] [CrossRef] [PubMed]
- Storer, R.J.; Akerman, S.; Goadsby, P.J. Calcitonin gene-related peptide (CGRP) modulates nociceptive trigeminovascular transmission in the cat. Br. J. Pharmacol. 2004, 142, 1171–1181. [Google Scholar] [CrossRef]
- Coste, J.; Voisin, D.L.; Miraucourt, L.S.; Dallel, R.; Luccarini, P. Dorsal horn NK1-expressing neurons control windup of downstream trigeminal nociceptive neurons. Pain 2008, 137, 340–351. [Google Scholar] [CrossRef]
- Hou, M.; Kanje, M.; Longmore, J.; Tajti, J.; Uddman, R.; Edvinsson, L. 5-HT(1B) and 5-HT(1D) receptors in the human trigeminal ganglion: Co-localization with calcitonin gene-related peptide, substance P and nitric oxide synthase. Brain Res. 2001, 909, 112–120. [Google Scholar] [CrossRef]
- Xiao, Y.; Richter, J.A.; Hurley, J.H. Release of glutamate and CGRP from trigeminal ganglion neurons: Role of calcium channels and 5-HT1 receptor signaling. Mol. Pain 2008, 4, 12. [Google Scholar] [CrossRef]
- Lennerz, J.K.; Rühle, V.; Ceppa, E.P.; Neuhuber, W.L.; Bunnett, N.W.; Grady, E.F.; Messlinger, K. Calcitonin receptor-like receptor (CLR), receptor activity-modifying protein 1 (RAMP1), and calcitonin gene-related peptide (CGRP) immunoreactivity in the rat trigeminovascular system: Differences between peripheral and central CGRP receptor distribution. J. Comp. Neurol. 2008, 507, 1277–1299. [Google Scholar] [CrossRef]
- Eftekhari, S.; Warfvinge, K.; Blixt, F.W.; Edvinsson, L. Differentiation of nerve fibers storing CGRP and CGRP receptors in the peripheral trigeminovascular system. J. Pain 2013, 14, 1289–1303. [Google Scholar] [CrossRef]
- Takhshid, M.A.; Owji, A.A.; Panjehshahin, M.R. In vitro effects of adrenomedullin and calcitonin gene related peptide on the release of serotonin and amino acids from rat dorsal spinal cord. Neurosci. Lett. 2007, 420, 193–197. [Google Scholar] [CrossRef]
- Edvinsson, J.C.A.; Warfvinge, K.; Krause, D.N.; Blixt, F.W.; Sheykhzade, M.; Edvinsson, L.; Haanes, K.A. C-fibers may modulate adjacent Aδ-fibers through axon-axon CGRP signaling at nodes of Ranvier in the trigeminal system. J. Headache Pain 2019, 20, 105. [Google Scholar] [CrossRef]
- Buldyrev, I.; Tanner, N.M.; Hsieh, H.; Dodd, E.G.; Nguyen, L.T.; Balkowiec, A. Calcitonin gene-related peptide enhances release of native brain-derived neurotrophic factor from trigeminal ganglion neurons. J. Neurochem. 2006, 99, 1338–1350. [Google Scholar] [CrossRef]
- Ciobanu, C.; Reid, G.; Babes, A. Acute and chronic effects of neurotrophic factors BDNF and GDNF on responses mediated by thermo-sensitive TRP channels in cultured rat dorsal root ganglion neurons. Brain Res. 2009, 1284, 54–67. [Google Scholar] [CrossRef]
- Kermani, P.; Hempstead, B. BDNF Actions in the Cardiovascular System: Roles in Development, Adulthood and Response to Injury. Front. Physiol. 2019, 10, 455. [Google Scholar] [CrossRef]
- Garraway, S.M.; Huie, J.R. Spinal Plasticity and Behavior: BDNF-Induced Neuromodulation in Uninjured and Injured Spinal Cord. Neural Plast. 2016, 2016, 9857201. [Google Scholar] [CrossRef]
- Johnson, K.W.; Morin, S.M.; Wroblewski, V.J.; Johnson, M.P. Peripheral and central nervous system distribution of the CGRP neutralizing antibody [125I] galcanezumab in male rats. Cephalalgia 2019, 39, 1241–1248. [Google Scholar] [CrossRef]
- Russo, A.F. Calcitonin Gene-Related Peptide (CGRP). Annu. Rev. Pharm. Toxicol. 2015, 55, 533–552. [Google Scholar] [CrossRef]
- DosSantos, M.F.; Holanda-Afonso, R.C.; Lima, R.L.; DaSilva, A.F.; Moura-Neto, V. The role of the blood–brain barrier in the development and treatment of migraine and other pain disorders. Front. Cell Neurosci. 2014, 8, 302. [Google Scholar] [CrossRef]
- Edvinsson, L. CGRP receptor antagonists and antibodies against CGRP and its receptor in migraine treatment. Br. J. Clin. Pharmacol. 2015, 80, 193–199. [Google Scholar] [CrossRef]
- Sand, T.; Zhitniy, N.; Nilsen, K.B.; Helde, G.; Hagen, K.; Stovner, L.J. Thermal pain thresholds are decreased in the migraine preattack phase. Eur. J. Neurol. 2008, 15, 1199–1205. [Google Scholar] [CrossRef]
- Burstein, R.; Yarnitsky, D.; Goor-Aryeh, I.; Ransil, B.J.; Bajwa, Z.H. An association between migraine and cutaneous allodynia. Ann. Neurol. 2000, 47, 614–624. [Google Scholar] [CrossRef]
- Lipton, R.B.; Bigal, M.E.; Ashina, S.; Burstein, R.; Silberstein, S.; Reed, M.L.; Serrano, D.; Stewart, W.F. American Migraine Prevalence Prevention Advisory Group Cutaneous allodynia in the migraine population. Ann. Neurol. 2008, 63, 148–158. [Google Scholar] [CrossRef]
- Lopshire, J.C.; Nicol, G.D. The cAMP transduction cascade mediates the prostaglandin E2 enhancement of the capsaicin-elicited current in rat sensory neurons: Whole-cell and single-channel studies. J. Neurosci. 1998, 18, 6081–6092. [Google Scholar] [CrossRef]
- Moriyama, T.; Higashi, T.; Togashi, K.; Iida, T.; Segi, E.; Sugimoto, Y.; Tominaga, T.; Narumiya, S.; Tominaga, M. Sensitization of TRPV1 by EP1 and IP reveals peripheral nociceptive mechanism of prostaglandins. Mol. Pain 2005, 1, 3. [Google Scholar] [CrossRef]
- Morenilla-Palao, C.; Planells-Cases, R.; García-Sanz, N.; Ferrer-Montiel, A. Regulated exocytosis contributes to protein kinase C potentiation of vanilloid receptor activity. J. Biol. Chem. 2004, 279, 25665–25672. [Google Scholar] [CrossRef]
- Tohda, C.; Sasaki, M.; Konemura, T.; Sasamura, T.; Itoh, M.; Kuraishi, Y. Axonal transport of VR1 capsaicin receptor mRNA in primary afferents and its participation in inflammation-induced increase in capsaicin sensitivity. J. Neurochem. 2001, 76, 1628–1635. [Google Scholar] [CrossRef]
- Amaya, F.; Oh-hashi, K.; Naruse, Y.; Iijima, N.; Ueda, M.; Shimosato, G.; Tominaga, M.; Tanaka, Y.; Tanaka, M. Local inflammation increases vanilloid receptor 1 expression within distinct subgroups of DRG neurons. Brain Res. 2003, 963, 190–196. [Google Scholar] [CrossRef]
- Kunkler, P.E.; Ballard, C.J.; Oxford, G.S.; Hurley, J.H. TRPA1 receptors mediate environmental irritant-induced meningeal vasodilatation. Pain 2011, 152, 38–44. [Google Scholar] [CrossRef]
- Dux, M.; Sántha, P.; Jancsó, G. Capsaicin-sensitive neurogenic sensory vasodilatation in the dura mater of the rat. J. Physiol. 2003, 552, 859–867. [Google Scholar] [CrossRef]
- Gupta, S.; Akerman, S.; van den Maagdenberg, A.M.J.M.; Saxena, P.R.; Goadsby, P.J.; van den Brink, A.M. Intravital microscopy on a closed cranial window in mice: A model to study trigeminovascular mechanisms involved in migraine. Cephalalgia 2006, 26, 1294–1303. [Google Scholar] [CrossRef]
- Dux, M.; Will, C.; Eberhardt, M.; Fischer, M.J.M.; Messlinger, K. Stimulation of rat cranial dura mater with potassium chloride causes CGRP release into the cerebrospinal fluid and increases medullary blood flow. Neuropeptides 2017, 64, 61–68. [Google Scholar] [CrossRef]
- Capuano, A.; Greco, M.C.; Navarra, P.; Tringali, G. Correlation between algogenic effects of calcitonin-gene-related peptide (CGRP) and activation of trigeminal vascular system, in an in vivo experimental model of nitroglycerin-induced sensitization. Eur. J. Pharmacol. 2014, 740, 97–102. [Google Scholar] [CrossRef]
- Holzer, P. Local effector functions of capsaicin-sensitive sensory nerve endings: Involvement of tachykinins, calcitonin gene-related peptide and other neuropeptides. Neuroscience 1988, 24, 739–768. [Google Scholar] [CrossRef]
- Levy, D.; Burstein, R.; Kainz, V.; Jakubowski, M.; Strassman, A.M. Mast cell degranulation activates a pain pathway underlying migraine headache. Pain 2007, 130, 166–176. [Google Scholar] [CrossRef]
- Schwenger, N.; Dux, M.; de Col, R.; Carr, R.; Messlinger, K. Interaction of calcitonin gene-related peptide, nitric oxide and histamine release in neurogenic blood flow and afferent activation in the rat cranial dura mater. Cephalalgia 2007, 27, 481–491. [Google Scholar] [CrossRef]
- Smith, J.A.; Davis, C.L.; Burgess, G.M. Prostaglandin E2-induced sensitization of bradykinin-evoked responses in rat dorsal root ganglion neurons is mediated by cAMP-dependent protein kinase A. Eur. J. Neurosci. 2000, 12, 3250–3258. [Google Scholar] [CrossRef]
- Premkumar, L.S.; Ahern, G.P. Induction of vanilloid receptor channel activity by protein kinase C. Nature 2000, 408, 985–990. [Google Scholar] [CrossRef]
- Tominaga, M.; Wada, M.; Masu, M. Potentiation of capsaicin receptor activity by metabotropic ATP receptors as a possible mechanism for ATP-evoked pain and hyperalgesia. Proc. Natl. Acad. Sci. USA 2001, 98, 6951–6956. [Google Scholar] [CrossRef]
- Sarchielli, P.; Mancini, M.L.; Floridi, A.; Coppola, F.; Rossi, C.; Nardi, K.; Acciarresi, M.; Pini, L.A.; Calabresi, P. Increased levels of neurotrophins are not specific for chronic migraine: Evidence from primary fibromyalgia syndrome. J. Pain 2007, 8, 737–745. [Google Scholar] [CrossRef] [PubMed]
- Dallos, A.; Kiss, M.; Polyánka, H.; Dobozy, A.; Kemény, L.; Husz, S. Effects of the neuropeptides substance P, calcitonin gene-related peptide, vasoactive intestinal polypeptide and galanin on the production of nerve growth factor and inflammatory cytokines in cultured human keratinocytes. Neuropeptides 2006, 40, 251–263. [Google Scholar] [CrossRef]
- Bonnington, J.K.; McNaughton, P.A. Signalling pathways involved in the sensitisation of mouse nociceptive neurones by nerve growth factor. J. Physiol. 2003, 551, 433–446. [Google Scholar] [CrossRef]
- Van Buren, J.J.; Bhat, S.; Rotello, R.; Pauza, M.E.; Premkumar, L.S. Sensitization and translocation of TRPV1 by insulin and IGF-I. Mol. Pain 2005, 1, 17. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Huang, J.; McNaughton, P.A. NGF rapidly increases membrane expression of TRPV1 heat-gated ion channels. EMBO J. 2005, 24, 4211–4223. [Google Scholar] [CrossRef]
- Zhang, L.; Kunkler, P.E.; Knopp, K.L.; Oxford, G.S.; Hurley, J.H. Role of intraganglionic transmission in the trigeminovascular pathway. Mol. Pain 2019, 15. [Google Scholar] [CrossRef]
- Ji, R.-R.; Samad, T.A.; Jin, S.-X.; Schmoll, R.; Woolf, C.J. p38 MAPK activation by NGF in primary sensory neurons after inflammation increases TRPV1 levels and maintains heat hyperalgesia. Neuron 2002, 36, 57–68. [Google Scholar] [CrossRef]
- Por, E.D.; Samelson, B.K.; Belugin, S.; Akopian, A.N.; Scott, J.D.; Jeske, N.A. PP2B/calcineurin-mediated desensitization of TRPV1 does not require AKAP150. Biochem. J. 2010, 432, 549–556. [Google Scholar] [CrossRef][Green Version]
- Por, E.D.; Bierbower, S.M.; Berg, K.A.; Gomez, R.; Akopian, A.N.; Wetsel, W.C.; Jeske, N.A. β-Arrestin-2 desensitizes the transient receptor potential vanilloid 1 (TRPV1) channel. J. Biol. Chem. 2012, 287, 37552–37563. [Google Scholar] [CrossRef]
- Masuoka, T.; Kudo, M.; Yamashita, Y.; Yoshida, J.; Imaizumi, N.; Muramatsu, I.; Nishio, M.; Ishibashi, T. TRPA1 Channels Modify TRPV1-Mediated Current Responses in Dorsal Root Ganglion Neurons. Front. Physiol. 2017, 8, 272. [Google Scholar] [CrossRef]
- Carreño, O.; Corominas, R.; Fernández-Morales, J.; Camiña, M.; Sobrido, M.-J.; Fernández-Fernández, J.M.; Pozo-Rosich, P.; Cormand, B.; Macaya, A. SNP variants within the vanilloid TRPV1 and TRPV3 receptor genes are associated with migraine in the Spanish population. Am. J. Med. Genet. B Neuropsychiatr. Genet. 2012, 159B, 94–103. [Google Scholar] [CrossRef]
- Chasman, D.I.; Schürks, M.; Anttila, V.; de Vries, B.; Schminke, U.; Launer, L.J.; Terwindt, G.M.; van den Maagdenberg, A.M.J.M.; Fendrich, K.; Völzke, H.; et al. Genome-wide association study reveals three susceptibility loci for common migraine in the general population. Nat. Genet. 2011, 43, 695–698. [Google Scholar] [CrossRef]
- Freilinger, T.; Anttila, V.; de Vries, B.; Malik, R.; Kallela, M.; Terwindt, G.M.; Pozo-Rosich, P.; Winsvold, B.; Nyholt, D.R.; van Oosterhout, W.P.J.; et al. Genome-wide association analysis identifies susceptibility loci for migraine without aura. Nat. Genet. 2012, 44, 777–782. [Google Scholar] [CrossRef]
- Gavva, N.R.; Sandrock, R.; Arnold, G.E.; Davis, M.; Lamas, E.; Lindvay, C.; Li, C.-M.; Smith, B.; Backonja, M.; Gabriel, K.; et al. Reduced TRPM8 expression underpins reduced migraine risk and attenuated cold pain sensation in humans. Sci. Rep. 2019, 9, 19655. [Google Scholar] [CrossRef]
- Almeida, M.C.; Hew-Butler, T.; Soriano, R.N.; Rao, S.; Wang, W.; Wang, J.; Tamayo, N.; Oliveira, D.L.; Nucci, T.B.; Aryal, P.; et al. Pharmacological blockade of the cold receptor TRPM8 attenuates autonomic and behavioral cold defenses and decreases deep body temperature. J. Neurosci. 2012, 32, 2086–2099. [Google Scholar] [CrossRef]
- Burgos-Vega, C.C.; Ahn, D.D.-U.; Bischoff, C.; Wang, W.; Horne, D.; Wang, J.; Gavva, N.; Dussor, G. Meningeal transient receptor potential channel M8 activation causes cutaneous facial and hindpaw allodynia in a preclinical rodent model of headache. Cephalalgia 2016, 36, 185–193. [Google Scholar] [CrossRef]
- Kayama, Y.; Shibata, M.; Takizawa, T.; Ibata, K.; Shimizu, T.; Ebine, T.; Toriumi, H.; Yuzaki, M.; Suzuki, N. Functional interactions between transient receptor potential M8 and transient receptor potential V1 in the trigeminal system: Relevance to migraine pathophysiology. Cephalalgia 2017. [Google Scholar] [CrossRef]
- Key, F.M.; Abdul-Aziz, M.A.; Mundry, R.; Peter, B.M.; Sekar, A.; D’Amato, M.; Dennis, M.Y.; Schmidt, J.M.; Andrés, A.M. Human local adaptation of the TRPM8 cold receptor along a latitudinal cline. PLoS Genet. 2018, 14, e1007298. [Google Scholar] [CrossRef]
- Kym, P.R.; Kort, M.E.; Hutchins, C.W. Analgesic potential of TRPV1 antagonists. Biochem. Pharmacol. 2009, 78, 211–216. [Google Scholar] [CrossRef]
- Garami, A.; Pakai, E.; McDonald, H.A.; Reilly, R.M.; Gomtsyan, A.; Corrigan, J.J.; Pinter, E.; Zhu, D.X.D.; Lehto, S.G.; Gavva, N.R.; et al. TRPV1 antagonists that cause hypothermia, instead of hyperthermia, in rodents: Compounds’ pharmacological profiles, in vivo targets, thermoeffectors recruited and implications for drug development. Acta Physiol. 2018, 223, e13038. [Google Scholar] [CrossRef]
- Arngrim, N.; Hougaard, A.; Ahmadi, K.; Vestergaard, M.B.; Schytz, H.W.; Amin, F.M.; Larsson, H.B.W.; Olesen, J.; Hoffmann, M.B.; Ashina, M. Heterogenous migraine aura symptoms correlate with visual cortex functional magnetic resonance imaging responses. Ann. Neurol. 2017, 82, 925–939. [Google Scholar] [CrossRef]
- Hadjikhani, N.; Vincent, M. Neuroimaging clues of migraine aura. J. Headache Pain 2019, 20, 32. [Google Scholar] [CrossRef]
- Zhang, X.; Levy, D.; Kainz, V.; Noseda, R.; Jakubowski, M.; Burstein, R. Activation of central trigeminovascular neurons by cortical spreading depression. Ann. Neurol. 2011, 69, 855–865. [Google Scholar] [CrossRef]
- Bolay, H.; Reuter, U.; Dunn, A.K.; Huang, Z.; Boas, D.A.; Moskowitz, M.A. Intrinsic brain activity triggers trigeminal meningeal afferents in a migraine model. Nat. Med. 2002, 8, 136–142. [Google Scholar] [CrossRef]
- Brinley, F.J.; Kandel, E.R.; Marshall, W.H. Potassium outflux from rabbit cortex during spreading depression. J. Neurophysiol. 1960, 23, 246–256. [Google Scholar] [CrossRef]
- Gasparini, C.F.; Smith, R.A.; Griffiths, L.R. Genetic insights into migraine and glutamate: A protagonist driving the headache. J. Neurol. Sci. 2016, 367, 258–268. [Google Scholar] [CrossRef]
- Shatillo, A.; Koroleva, K.; Giniatullina, R.; Naumenko, N.; Slastnikova, A.A.; Aliev, R.R.; Bart, G.; Atalay, M.; Gu, C.; Khazipov, R.; et al. Cortical spreading depression induces oxidative stress in the trigeminal nociceptive system. Neuroscience 2013, 253, 341–349. [Google Scholar] [CrossRef]
- Sakaguchi, R.; Mori, Y. Transient receptor potential (TRP) channels: Biosensors for redox environmental stimuli and cellular status. Free Radic. Biol. Med. 2019. [Google Scholar] [CrossRef]
- Melo-Carrillo, A.; Noseda, R.; Nir, R.-R.; Schain, A.J.; Stratton, J.; Strassman, A.M.; Burstein, R. Selective Inhibition of Trigeminovascular Neurons by Fremanezumab: A Humanized Monoclonal Anti-CGRP Antibody. J. Neurosci. 2017, 37, 7149–7163. [Google Scholar] [CrossRef]
- Schain, A.J.; Melo-Carrillo, A.; Borsook, D.; Grutzendler, J.; Strassman, A.M.; Burstein, R. Activation of pial and dural macrophages and dendritic cells by cortical spreading depression. Ann. Neurol. 2018, 83, 508–521. [Google Scholar] [CrossRef]
- Levy, D. Endogenous mechanisms underlying the activation and sensitization of meningeal nociceptors: The role of immuno-vascular interactions and cortical spreading depression. Curr. Pain Headache Rep. 2012, 16, 270–277. [Google Scholar] [CrossRef]
- Split, W.; Szydlowska, M. Headaches in non insulin-dependent diabetes mellitus. Funct. Neurol. 1997, 12, 327–332. [Google Scholar]
- Chai, N.C.; Scher, A.I.; Moghekar, A.; Bond, D.S.; Peterlin, B.L. Obesity and headache: Part I--a systematic review of the epidemiology of obesity and headache. Headache 2014, 54, 219–234. [Google Scholar] [CrossRef]
- Marics, B.; Peitl, B.; Pázmándi, K.; Bácsi, A.; Németh, J.; Oszlács, O.; Jancsó, G.; Dux, M. Diet-Induced Obesity Enhances TRPV1-Mediated Neurovascular Reactions in the Dura Mater. Headache 2017, 57, 441–454. [Google Scholar] [CrossRef]
- Marics, B.; Peitl, B.; Varga, A.; Pázmándi, K.; Bácsi, A.; Németh, J.; Szilvássy, Z.; Jancsó, G.; Dux, M. Diet-induced obesity alters dural CGRP release and potentiates TRPA1-mediated trigeminovascular responses. Cephalalgia 2017, 37, 581–591. [Google Scholar] [CrossRef]
- Wentworth, J.M.; Naselli, G.; Brown, W.A.; Doyle, L.; Phipson, B.; Smyth, G.K.; Wabitsch, M.; O’Brien, P.E.; Harrison, L.C. Pro-inflammatory CD11c+ CD206+ adipose tissue macrophages are associated with insulin resistance in human obesity. Diabetes 2010, 59, 1648–1656. [Google Scholar] [CrossRef]
- Calder, P.C.; Ahluwalia, N.; Brouns, F.; Buetler, T.; Clement, K.; Cunningham, K.; Esposito, K.; Jönsson, L.S.; Kolb, H.; Lansink, M.; et al. Dietary factors and low-grade inflammation in relation to overweight and obesity. Br. J. Nutr. 2011, 106, S5–S78. [Google Scholar] [CrossRef]
- Harriott, A.M.; Gold, M.S. Electrophysiological properties of dural afferents in the absence and presence of inflammatory mediators. J. Neurophysiol. 2009, 101, 3126–3134. [Google Scholar] [CrossRef]
- Dux, M.; Rosta, J.; Pintér, S.; Sántha, P.; Jancsó, G. Loss of capsaicin-induced meningeal neurogenic sensory vasodilatation in diabetic rats. Neuroscience 2007, 150, 194–201. [Google Scholar] [CrossRef]
- Troger, J.; Humpel, C.; Kremser, B.; Kralinger, M.; Teuchner, B.; Kunze, C.; Philipp, W.; Kieselbach, G. The effect of streptozotocin-induced diabetes mellitus on substance P and calcitonin gene-related peptide expression in the rat trigeminal ganglion. Brain Res. 1999, 842, 84–91. [Google Scholar] [CrossRef]
- Spanou, I.; Bougea, A.; Liakakis, G.; Rizonaki, K.; Anagnostou, E.; Duntas, L.; Kararizou, E. Relationship of Migraine and Tension-Type Headache With Hypothyroidism: A Literature Review. Headache 2019, 59, 1174–1186. [Google Scholar] [CrossRef]
- Bevilaqua Grossi, D.; Lipton, R.B.; Bigal, M.E. Temporomandibular disorders and migraine chronification. Curr. Pain Headache Rep. 2009, 13, 314–318. [Google Scholar] [CrossRef]
- Eberhardt, M.; Neeb, L.; Vogel, E.-M.; Tiegs, G.; Reuter, U.; Messlinger, K.; Fischer, M.J.M. Glyceroltrinitrate facilitates stimulated CGRP release but not gene expression of CGRP or its receptor components in rat trigeminal ganglia. Neuropeptides 2009, 43, 483–489. [Google Scholar] [CrossRef]
- Cheng, J.-K.; Ji, R.-R. Intracellular signaling in primary sensory neurons and persistent pain. Neurochem. Res. 2008, 33, 1970–1978. [Google Scholar] [CrossRef]
- Kung, L.-H.; Gong, K.; Adedoyin, M.; Ng, J.; Bhargava, A.; Ohara, P.T.; Jasmin, L. Evidence for glutamate as a neuroglial transmitter within sensory ganglia. PLoS ONE 2013, 8, e68312. [Google Scholar] [CrossRef]
- Goto, T.; Iwai, H.; Kuramoto, E.; Yamanaka, A. Neuropeptides and ATP signaling in the trigeminal ganglion. Jpn. Dent. Sci. Rev. 2017, 53, 117–124. [Google Scholar] [CrossRef]
- Zhang, Y.; Song, N.; Liu, F.; Lin, J.; Liu, M.; Huang, C.; Liao, D.; Zhou, C.; Wang, H.; Shen, J. Activation of mitogen-activated protein kinases in satellite glial cells of the trigeminal ganglion contributes to substance P-mediated inflammatory pain. Int. J. Oral. Sci. 2019, 11, 24. [Google Scholar] [CrossRef]
- Boye Larsen, D.; Ingemann Kristensen, G.; Panchalingam, V.; Laursen, J.C.; Nørgaard Poulsen, J.; Skallerup Andersen, M.; Kandiah, A.; Gazerani, P. Investigating the expression of metabotropic glutamate receptors in trigeminal ganglion neurons and satellite glial cells: Implications for craniofacial pain. J. Recept. Signal Transduct. Res. 2014, 34, 261–269. [Google Scholar] [CrossRef]
- Dux, M.; Deák, É.; Tassi, N.; Sántha, P.; Jancsó, G. Endovanilloids are potential activators of the trigeminovascular nocisensor complex. J. Headache Pain 2016, 17, 53. [Google Scholar] [CrossRef]
- Dinh, Q.T.; Cryer, A.; Dinh, S.; Trevisani, M.; Georgiewa, P.; Chung, F.; Geppetti, P.; Heppt, W.; Klapp, B.F.; Fischer, A. Protease-activated receptor 2 expression in trigeminal neurons innervating the rat nasal mucosa. Neuropeptides 2005, 39, 461–466. [Google Scholar] [CrossRef]
- Dux, M.; Rosta, J.; Sántha, P.; Jancsó, G. Involvement of capsaicin-sensitive afferent nerves in the proteinase-activated receptor 2-mediated vasodilatation in the rat dura mater. Neuroscience 2009, 161, 887–894. [Google Scholar] [CrossRef]
- Dai, Y.; Wang, S.; Tominaga, M.; Yamamoto, S.; Fukuoka, T.; Higashi, T.; Kobayashi, K.; Obata, K.; Yamanaka, H.; Noguchi, K. Sensitization of TRPA1 by PAR2 contributes to the sensation of inflammatory pain. J. Clin. Investig. 2007, 117, 1979–1987. [Google Scholar] [CrossRef]
- Dux, M.; Sántha, P.; Jancsó, G. The role of chemosensitive afferent nerves and TRP ion channels in the pathomechanism of headaches. Pflug. Arch. 2012, 464, 239–248. [Google Scholar] [CrossRef]
- Wang, R. Physiological implications of hydrogen sulfide: A whiff exploration that blossomed. Physiol. Rev. 2012, 92, 791–896. [Google Scholar] [CrossRef]
- Kimura, H. The physiological role of hydrogen sulfide and beyond. Nitric Oxide 2014, 41, 4–10. [Google Scholar] [CrossRef]
- Koroleva, K.; Mustafina, A.; Yakovlev, A.; Hermann, A.; Giniatullin, R.; Sitdikova, G. Receptor Mechanisms Mediating the Pro-Nociceptive Action of Hydrogen Sulfide in Rat Trigeminal Neurons and Meningeal Afferents. Front. Cell Neurosci. 2017, 11, 226. [Google Scholar] [CrossRef]
- Eberhardt, M.; Dux, M.; Namer, B.; Miljkovic, J.; Cordasic, N.; Will, C.; Kichko, T.I.; de la Roche, J.; Fischer, M.; Suárez, S.A.; et al. H2S and NO cooperatively regulate vascular tone by activating a neuroendocrine HNO-TRPA1-CGRP signalling pathway. Nat. Commun. 2014, 5, 4381. [Google Scholar] [CrossRef]
- Miao, X.; Meng, X.; Wu, G.; Ju, Z.; Zhang, H.-H.; Hu, S.; Xu, G.-Y. Upregulation of cystathionine-β-synthetase expression contributes to inflammatory pain in rat temporomandibular joint. Mol. Pain 2014, 10, 9. [Google Scholar] [CrossRef]
- Hatakeyama, Y.; Takahashi, K.; Tominaga, M.; Kimura, H.; Ohta, T. Polysulfide evokes acute pain through the activation of nociceptive TRPA1 in mouse sensory neurons. Mol. Pain 2015, 11, 24. [Google Scholar] [CrossRef]
- Kimura, H. Hydrogen polysulfide (H2S n) signaling along with hydrogen sulfide (H2S) and nitric oxide (NO). J. Neural Transm. 2016, 123, 1235–1245. [Google Scholar] [CrossRef]
- Fukuto, J.M.; Switzer, C.H.; Miranda, K.M.; Wink, D.A. Nitroxyl (HNO): Chemistry, biochemistry, and pharmacology. Annu. Rev. Pharmacol. Toxicol. 2005, 45, 335–355. [Google Scholar] [CrossRef]
- Dux, M.; Will, C.; Vogler, B.; Filipovic, M.R.; Messlinger, K. Meningeal blood flow is controlled by H2 S-NO crosstalk activating a HNO-TRPA1-CGRP signalling pathway. Br. J. Pharmacol. 2016, 173, 431–445. [Google Scholar] [CrossRef]
- Diener, H.-C.; Holle, D.; Solbach, K.; Gaul, C. Medication-overuse headache: Risk factors, pathophysiology and management. Nat. Rev. Neurol. 2016, 12, 575–583. [Google Scholar] [CrossRef]
- Masaaki, I.; Hideyo, K.; Hirotaka, K.; Masakazu, I. Lack of Association between TRP Gene Polymorphisms and Complication of Medication Overuse Headache in Migraine Patients. J. Med. Clin. Res. Rev. 2018, 2, 1–5. [Google Scholar]
- Van Hoogstraten, W.S.; MaassenVanDenBrink, A. The need for new acutely acting antimigraine drugs: Moving safely outside acute medication overuse. J. Headache Pain 2019, 20, 54. [Google Scholar] [CrossRef]
- Srikiatkhachorn, A.; le Grand, S.M.; Supornsilpchai, W.; Storer, R.J. Pathophysiology of medication overuse headache—an update. Headache 2014, 54, 204–210. [Google Scholar] [CrossRef]
- Carvalho, C.; Santos, R.X.; Cardoso, S.; Correia, S.; Oliveira, P.J.; Santos, M.S.; Moreira, P.I. Doxorubicin: The good, the bad and the ugly effect. Curr. Med. Chem. 2009, 16, 3267–3285. [Google Scholar] [CrossRef]
- Kondo, A.; Ohnishi, A.; Nagara, H.; Tateishi, J. Neurotoxicity in primary sensory neurons of adriamycin administered through retrograde axoplasmic transport in rats. Neuropathol. Appl. Neurobiol. 1987, 13, 177–192. [Google Scholar] [CrossRef]
- Katona, M.; Boros, K.; Sántha, P.; Ferdinandy, P.; Dux, M.; Jancsó, G. Selective sensory denervation by capsaicin aggravates adriamycin-induced cardiomyopathy in rats. Naunyn Schmiedebergs Arch. Pharmacol. 2004, 370, 436–443. [Google Scholar] [CrossRef]
- Deák, É.; Rosta, J.; Boros, K.; Kis, G.; Sántha, P.; Messlinger, K.; Jancsó, G.; Dux, M. Chronic adriamycin treatment impairs CGRP-mediated functions of meningeal sensory nerves. Neuropeptides 2018, 69, 46–52. [Google Scholar] [CrossRef]
- Edvinsson, L.; Warfvinge, K. Recognizing the role of CGRP and CGRP receptors in migraine and its treatment. Cephalalgia 2017, 366–373. [Google Scholar] [CrossRef]
- Eftekhari, S.; Salvatore, C.A.; Johansson, S.; Chen, T.-B.; Zeng, Z.; Edvinsson, L. Localization of CGRP, CGRP receptor, PACAP and glutamate in trigeminal ganglion. Relation to the blood-brain barrier. Brain Res. 2015, 1600, 93–109. [Google Scholar] [CrossRef]
- Edvinsson, L.; Tajti, J.; Szalárdy, L.; Vécsei, L. PACAP and its role in primary headaches. J. Headache Pain 2018, 19, 21. [Google Scholar] [CrossRef]
- Benemei, S.; Cortese, F.; Labastida-Ramírez, A.; Marchese, F.; Pellesi, L.; Romoli, M.; Vollesen, A.L.; Lampl, C.; Ashina, M. School of Advanced Studies of the European Headache Federation (EHF-SAS) Triptans and CGRP blockade—impact on the cranial vasculature. J. Headache Pain 2017, 18, 103. [Google Scholar] [CrossRef]
- Lambert, G.A.; Davis, J.B.; Appleby, J.M.; Chizh, B.A.; Hoskin, K.L.; Zagami, A.S. The effects of the TRPV1 receptor antagonist SB-705498 on trigeminovascular sensitisation and neurotransmission. Naunyn Schmiedebergs Arch. Pharmacol. 2009, 380, 311–325. [Google Scholar] [CrossRef]
- Meents, J.E.; Hoffmann, J.; Chaplan, S.R.; Neeb, L.; Schuh-Hofer, S.; Wickenden, A.; Reuter, U. Two TRPV1 receptor antagonists are effective in two different experimental models of migraine. J. Headache Pain 2015, 16, 57. [Google Scholar] [CrossRef]
- Summ, O.; Holland, P.R.; Akerman, S.; Goadsby, P.J. TRPV1 receptor blockade is ineffective in different in vivo models of migraine. Cephalalgia 2011, 31, 172–180. [Google Scholar] [CrossRef]
- Edelmayer, R.M.; Le, L.N.; Yan, J.; Wei, X.; Nassini, R.; Materazzi, S.; Preti, D.; Appendino, G.; Geppetti, P.; Dodick, D.W.; et al. Activation of TRPA1 on dural afferents: A potential mechanism of headache pain. Pain 2012, 153, 1949–1958. [Google Scholar] [CrossRef]
- Materazzi, S.; Benemei, S.; Fusi, C.; Gualdani, R.; De Siena, G.; Vastani, N.; Andersson, D.A.; Trevisan, G.; Moncelli, M.R.; Wei, X.; et al. Parthenolide inhibits nociception and neurogenic vasodilatation in the trigeminovascular system by targeting the TRPA1 channel. Pain 2013, 154, 2750–2758. [Google Scholar] [CrossRef]
- Leamy, A.W.; Shukla, P.; McAlexander, M.A.; Carr, M.J.; Ghatta, S. Curcumin ((E,E)-1,7-bis(4-hydroxy-3-methoxyphenyl)-1,6-heptadiene-3,5-dione) activates and desensitizes the nociceptor ion channel TRPA1. Neurosci. Lett. 2011, 503, 157–162. [Google Scholar] [CrossRef]
- Wang, S.; Yamamoto, S.; Kogure, Y.; Zhang, W.; Noguchi, K.; Dai, Y. Partial Activation and Inhibition of TRPV1 Channels by Evodiamine and Rutaecarpine, Two Major Components of the Fruits of Evodia rutaecarpa. J. Nat. Prod. 2016, 79, 1225–1230. [Google Scholar] [CrossRef]
- Lipton, R.B.; Göbel, H.; Einhäupl, K.M.; Wilks, K.; Mauskop, A. Petasites hybridus root (butterbur) is an effective preventive treatment for migraine. Neurology 2004, 63, 2240–2244. [Google Scholar] [CrossRef]
- Benemei, S.; De Logu, F.; Li Puma, S.; Marone, I.M.; Coppi, E.; Ugolini, F.; Liedtke, W.; Pollastro, F.; Appendino, G.; Geppetti, P.; et al. The anti-migraine component of butterbur extracts, isopetasin, desensitizes peptidergic nociceptors by acting on TRPA1 cation channel. Br. J. Pharmacol. 2017, 174, 2897–2911. [Google Scholar] [CrossRef]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dux, M.; Rosta, J.; Messlinger, K. TRP Channels in the Focus of Trigeminal Nociceptor Sensitization Contributing to Primary Headaches. Int. J. Mol. Sci. 2020, 21, 342. https://doi.org/10.3390/ijms21010342
Dux M, Rosta J, Messlinger K. TRP Channels in the Focus of Trigeminal Nociceptor Sensitization Contributing to Primary Headaches. International Journal of Molecular Sciences. 2020; 21(1):342. https://doi.org/10.3390/ijms21010342
Chicago/Turabian StyleDux, Mária, Judit Rosta, and Karl Messlinger. 2020. "TRP Channels in the Focus of Trigeminal Nociceptor Sensitization Contributing to Primary Headaches" International Journal of Molecular Sciences 21, no. 1: 342. https://doi.org/10.3390/ijms21010342
APA StyleDux, M., Rosta, J., & Messlinger, K. (2020). TRP Channels in the Focus of Trigeminal Nociceptor Sensitization Contributing to Primary Headaches. International Journal of Molecular Sciences, 21(1), 342. https://doi.org/10.3390/ijms21010342




