PHLDA1 Does Not Contribute Directly to Heat Shock-Induced Apoptosis of Spermatocytes
Abstract
1. Introduction
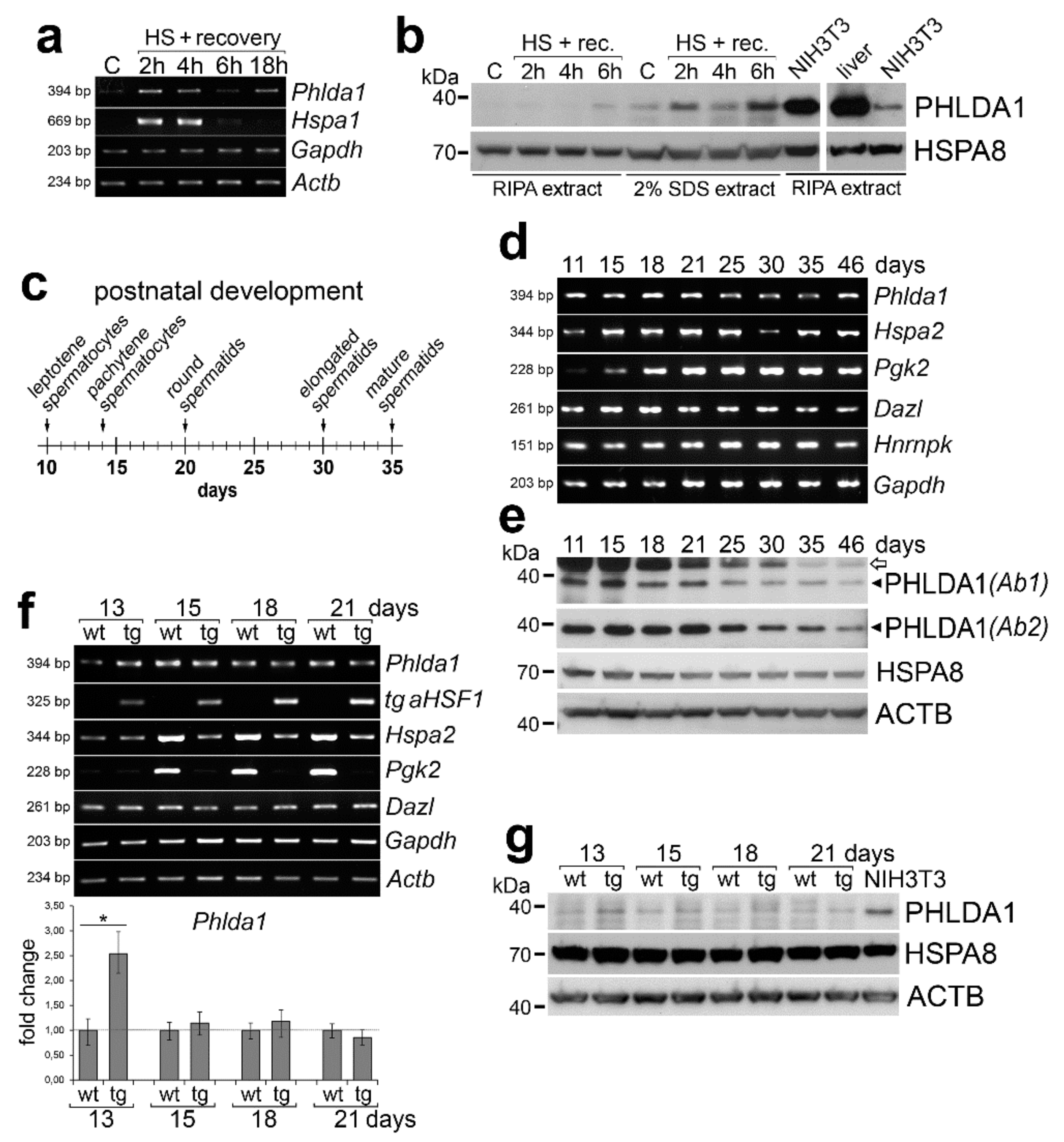
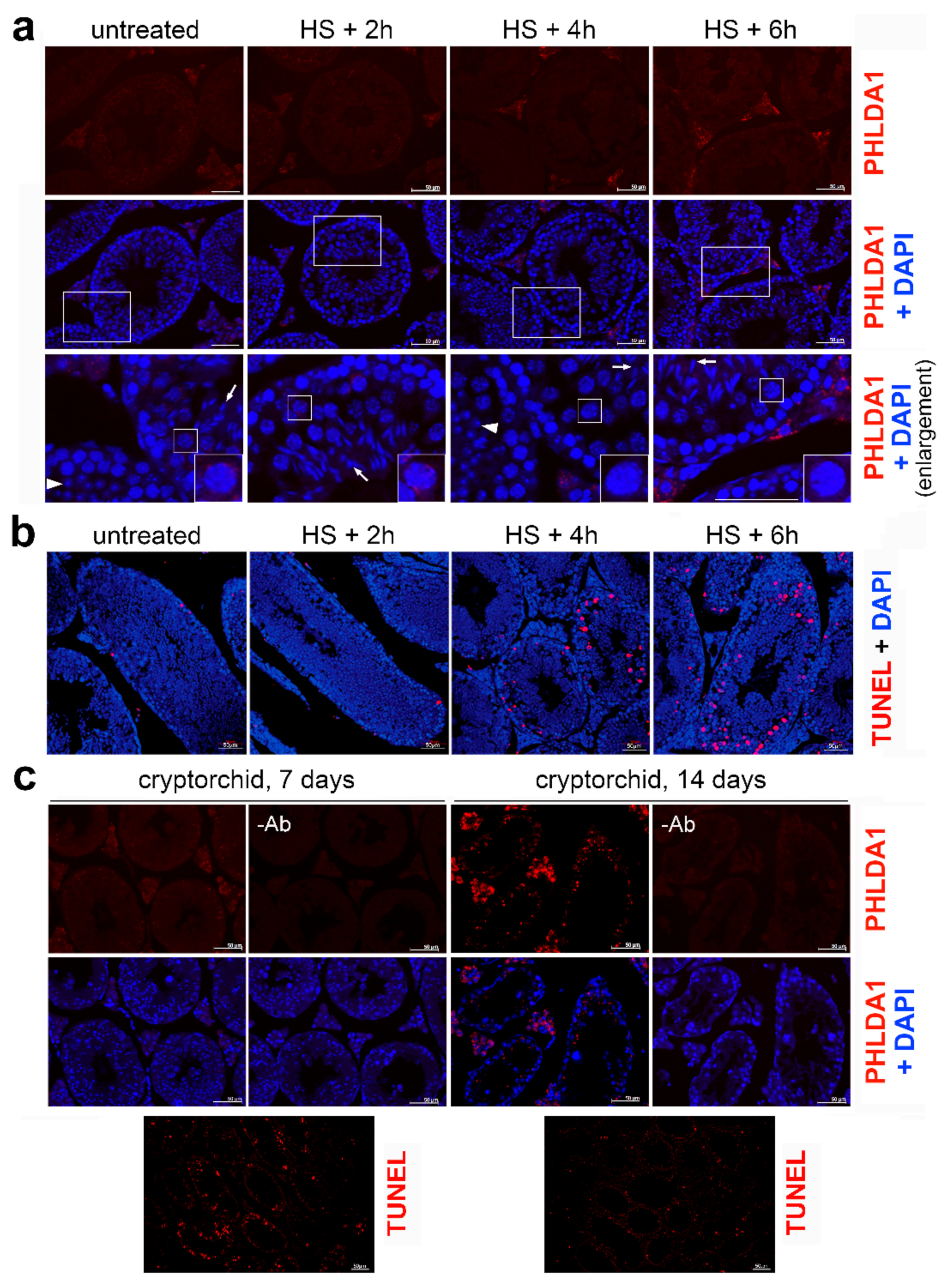
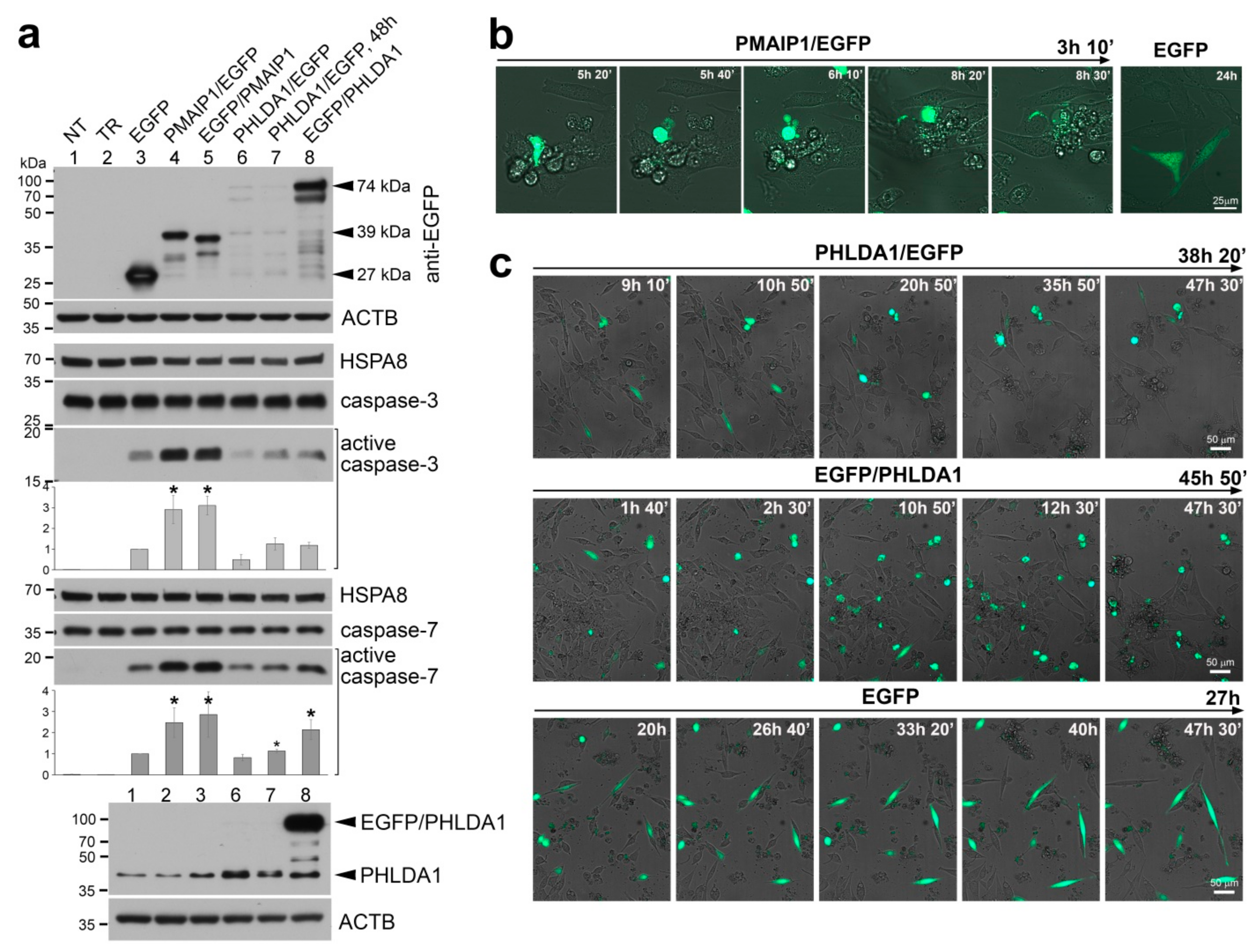
2. Results
3. Discussion
4. Materials and Methods
4.1. Animals and Heat Shock Treatment
4.2. RNA Isolation, cDNA Synthesis, and (q)PCR
4.3. Transient Transfections, Protein Extraction, and Western Blotting
4.4. Immunofluorescence (IF) and TUNEL Assay
4.5. Phlda1 and Pmaip1 Cloning
4.6. Life Cell Imaging
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Rupik, W.; Jasik, K.; Bembenek, J.; Widłak, W. The expression patterns of heat shock genes and proteins and their role during vertebrate’s development. Comp. Biochem. Physiol. Part. A Mol. Integr. Physiol. 2011, 159, 349–366. [Google Scholar] [CrossRef]
- Izu, H.; Inouye, S.; Fujimoto, M.; Shiraishi, K.; Naito, K.; Nakai, A. Heat shock transcription factor 1 is involved in quality-control mechanisms in male germ cells. Biol. Reprod. 2004, 70, 18–24. [Google Scholar] [CrossRef]
- Vydra, N.; Malusecka, E.; Jarzab, M.; Lisowska, K.; Glowala-Kosinska, M.; Benedyk, K.; Widlak, P.; Krawczyk, Z.; Widlak, W. Spermatocyte-specific expression of constitutively active heat shock factor 1 induces HSP70i-resistant apoptosis in male germ cells. Cell Death Differ. 2006, 13, 212–222. [Google Scholar] [CrossRef] [PubMed]
- Widlak, W.; Vydra, N.; Malusecka, E.; Dudaladava, V.; Winiarski, B.; Scieglińska, D.; Widlak, P. Heat shock transcription factor 1 down-regulates spermatocyte-specific 70 kDa heat shock protein expression prior to the induction of apoptosis in mouse testes. Genes Cells 2007, 12, 487–499. [Google Scholar] [CrossRef] [PubMed]
- Nakai, A.; Suzuki, M.; Tanabe, M. Arrest of spermatogenesis in mice expressing an active heat shock transcription factor 1. EMBO J. 2000, 19, 1545–1554. [Google Scholar] [CrossRef] [PubMed]
- Widłak, W.; Benedyk, K.; Vydra, N.; Głowala, M.; Scieglińska, D.; Małusecka, E.; Nakai, A.; Krawczyk, Z. Expression of a constitutively active mutant of heat shock factor 1 under the control of testis-specific hst70 gene promoter in transgenic mice induces degeneration of seminiferous epithelium. Acta Biochim. Pol. 2003, 50, 535–541. [Google Scholar] [CrossRef] [PubMed]
- Yin, Y.; Hawkins, K.L.; DeWolf, W.C.; Morgentaler, A. Heat stress causes testicular germ cell apoptosis in adult mice. J. Androl. 1997, 18, 159–165. [Google Scholar]
- Kim, B.; Park, K.; Rhee, K. Heat stress response of male germ cells. Cell. Mol. Life Sci. 2013, 70, 2623–2636. [Google Scholar] [CrossRef]
- Hayashida, N.; Inouye, S.; Fujimoto, M.; Tanaka, Y.; Izu, H.; Takaki, E.; Ichikawa, H.; Rho, J.; Nakai, A. A novel HSF1-mediated death pathway that is suppressed by heat shock proteins. EMBO J. 2006, 25, 4773–4783. [Google Scholar] [CrossRef]
- Liu, F.; Xu, Z.-L.; Qian, X.-J.; Qiu, W.-Y.; Huang, H. Expression of Hsf1, Hsf2, and Phlda1 in cells undergoing cryptorchid-induced apoptosis in rat testes. Mol. Reprod. Dev. 2011, 78, 283–291. [Google Scholar] [CrossRef]
- Frank, D.; Mendelsohn, C.L.; Ciccone, E.; Svensson, K.; Ohlsson, R.; Tycko, B. A novel pleckstrin homology-related gene family defined by Ipl/Tssc3, TDAG51, and Tih1: Tissue-specific expression, chromosomal location, and parental imprinting. Mamm. Genome 1999, 10, 1150–1159. [Google Scholar] [CrossRef] [PubMed]
- Gomes, I.; Xiong, W.; Miki, T.; Rosner, M.R. A proline- and glutamine-rich protein promotes apoptosis in neuronal cells. J. Neurochem. 1999, 73, 612–622. [Google Scholar] [CrossRef] [PubMed]
- Sakthianandeswaren, A.; Christie, M.; D’Andreti, C.; Tsui, C.; Jorissen, R.N.; Li, S.; Fleming, N.I.; Gibbs, P.; Lipton, L.; Malaterre, J.; et al. PHLDA1 expression marks the putative epithelial stem cells and contributes to intestinal tumorigenesis. Cancer Res. 2011, 71, 3709–3719. [Google Scholar] [CrossRef] [PubMed]
- Nagai, M.A. Pleckstrin homology-like domain, family A, member 1 (PHLDA1) and cancer. Biomed. Rep. 2016, 4, 275–281. [Google Scholar] [CrossRef]
- Coleman, S.K.; Cao, A.W.; Rebalka, I.A.; Gyulay, G.; Chambers, P.J.; Tupling, A.R.; Austin, R.C.; Hawke, T.J. The Pleckstrin homology like domain family member, TDAG51, is temporally regulated during skeletal muscle regeneration. Biochem. Biophys. Res. Commun. 2018, 495, 499–505. [Google Scholar] [CrossRef]
- Wang, S.; Zhang, H.; Wang, A.; Huang, D.; Fan, J.; Lu, L.; Chen, X. PHLDA1 Promotes Lung Contusion by Regulating the Toll-Like Receptor 2 Signaling Pathway. Cell. Physiol. Biochem. 2016, 40, 1198–1206. [Google Scholar] [CrossRef]
- Basseri, S.; Lhoták, S.; Fullerton, M.D.; Palanivel, R.; Jiang, H.; Lynn, E.G.; Ford, R.J.; Maclean, K.N.; Steinberg, G.R.; Austin, R.C. Loss of TDAG51 results in mature-onset obesity, hepatic steatosis, and insulin resistance by regulating lipogenesis. Diabetes 2013, 62, 158–169. [Google Scholar] [CrossRef]
- Oberg, H.-H.; Sipos, B.; Kalthoff, H.; Janssen, O.; Kabelitz, D. Regulation of T-cell death-associated gene 51 (TDAG51) expression in human T-cells. Cell Death Differ. 2004, 11, 674–684. [Google Scholar] [CrossRef][Green Version]
- Park, E.-S.; Kim, J.; Ha, T.; Choi, J.-S.; Soo Hong, K.; Rho, J. TDAG51 deficiency promotes oxidative stress-induced apoptosis through the generation of reactive oxygen species in mouse embryonic fibroblasts. Exp. Mol. Med. 2013, 45, e35. [Google Scholar] [CrossRef]
- Park, C.G.; Lee, S.Y.; Kandala, G.; Lee, S.Y.; Choi, Y. A novel gene product that couples TCR signaling to Fas(CD95) expression in activation-induced cell death. Immunity 1996, 4, 583–591. [Google Scholar] [CrossRef]
- Rho, J.; Gong, S.; Kim, N.; Choi, Y. TDAG51 is not essential for Fas/CD95 regulation and apoptosis in vivo. Mol. Cell. Biol. 2001, 21, 8365–8370. [Google Scholar] [CrossRef] [PubMed]
- Hossain, G.S.; Van Thienen, J.V.; Werstuck, G.H.; Zhou, J.; Sood, S.K.; Dickhout, J.G.; De Koning, A.B.L.; Tang, D.; Wu, D.; Falk, E.; et al. TDAG51 is induced by homocysteine, promotes detachment-mediated programmed cell death, and contributes to the cevelopment of atherosclerosis in hyperhomocysteinemia. J. Biol. Chem. 2003, 278, 30317–30327. [Google Scholar] [CrossRef] [PubMed]
- Bellve, A.; Cavicchia, J.; Millette, C.; O’Brien, D.; Bhatnagar, Y.; Dym, M. Spermatogenic cells of the prepuberal mouse: Isolation and morphological characterization. J. Cell Biol. 1977, 74, 68–85. [Google Scholar] [CrossRef] [PubMed]
- Scieglinska, D.; Krawczyk, Z. Expression, function, and regulation of the testis-enriched heat shock HSPA2 gene in rodents and humans. Cell Stress Chaperones 2015, 20, 221–235. [Google Scholar] [CrossRef]
- Goto, M.; Koji, T.; Mizuno, K.; Tamaru, M.; Koikeda, S.; Nakane, P.K.; Mori, N.; Masamune, Y.; Nakanishi, Y. Transcription switch of two phosphoglycerate kinase genes during spermatogenesis as determined with mouse testis sections in situ. Exp. Cell Res. 1990, 186, 273–278. [Google Scholar] [CrossRef]
- Niederberger, C.; Agulnik, A.I.; Cho, Y.; Lamb, D.; Bishop, C.E. In situ hybridization shows that Dazla expression in mouse testis is restricted to premeiotic stages IV-VI of spermatogenesis. Mamm. Genome 1997, 8, 277–278. [Google Scholar] [CrossRef]
- Oda, E.; Ohki, R.; Murasawa, H.; Nemoto, J.; Shibue, T.; Yamashita, T.; Tokino, T.; Taniguchi, T.; Tanaka, N. Noxa, a BH3-only member of the Bcl-2 family and candidate mediator of p53-induced apoptosis. Science 2000, 288, 1053–1058. [Google Scholar] [CrossRef]
- Carlisle, R.E.; Heffernan, A.; Brimble, E.; Liu, L.; Jerome, D.; Collins, C.A.; Mohammed-Ali, Z.; Margetts, P.J.; Austin, R.C.; Dickhout, J.G. TDAG51 mediates epithelial-to-mesenchymal transition in human proximal tubular epithelium. Am. J. Physiol. Renal Physiol. 2012, 303, F467–F481. [Google Scholar] [CrossRef]
- Brentnall, M.; Rodriguez-Menocal, L.; De Guevara, R.L.; Cepero, E.; Boise, L.H. Caspase-9, caspase-3 and caspase-7 have distinct roles during intrinsic apoptosis. BMC Cell Biol. 2013, 14, 32. [Google Scholar] [CrossRef]
- Luaces, J.P.; Rossi, L.F.; Sciurano, R.B.; Rebuzzini, P.; Merico, V.; Zuccotti, M.; Merani, M.S.; Garagna, S. Loss of Sertoli-germ cell adhesion determines the rapid germ cell elimination during the seasonal regression of the seminiferous epithelium of the large hairy armadillo Chaetophractus villosus. Biol. Reprod. 2014, 90, 48. [Google Scholar] [CrossRef]
- Widlak, W.; Winiarski, B.; Krawczyk, A.; Vydra, N.; Malusecka, E.; Krawczyk, Z. Inducible 70 kDa heat shock protein does not protect spermatogenic cells from damage induced by cryptorchidism. Int. J. Androl. 2007, 30, 80–87. [Google Scholar] [CrossRef] [PubMed]
- Kus-Liśkiewicz, M.; Polańska, J.; Korfanty, J.; Olbryt, M.; Vydra, N.; Toma, A.; Widłak, W. Impact of heat shock transcription factor 1 on global gene expression profiles in cells which induce either cytoprotective or pro-apoptotic response following hyperthermia. BMC Genomics 2013, 14, 456. [Google Scholar] [CrossRef] [PubMed]
- Vydra, N.; Janus, P.; Toma-Jonik, A.; Stokowy, T.; Mrowiec, K.; Korfanty, J.; Długajczyk, A.; Wojtaś, B.; Gielniewski, B.; Widłak, W. 17β-Estradiol Activates HSF1 via MAPK Signaling in ERα-Positive Breast Cancer Cells. Cancers 2019, 11, 1533. [Google Scholar] [CrossRef] [PubMed]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Janus, P.; Mrowiec, K.; Vydra, N.; Widłak, P.; Toma-Jonik, A.; Korfanty, J.; Smolarczyk, R.; Widłak, W. PHLDA1 Does Not Contribute Directly to Heat Shock-Induced Apoptosis of Spermatocytes. Int. J. Mol. Sci. 2020, 21, 267. https://doi.org/10.3390/ijms21010267
Janus P, Mrowiec K, Vydra N, Widłak P, Toma-Jonik A, Korfanty J, Smolarczyk R, Widłak W. PHLDA1 Does Not Contribute Directly to Heat Shock-Induced Apoptosis of Spermatocytes. International Journal of Molecular Sciences. 2020; 21(1):267. https://doi.org/10.3390/ijms21010267
Chicago/Turabian StyleJanus, Patryk, Katarzyna Mrowiec, Natalia Vydra, Piotr Widłak, Agnieszka Toma-Jonik, Joanna Korfanty, Ryszard Smolarczyk, and Wiesława Widłak. 2020. "PHLDA1 Does Not Contribute Directly to Heat Shock-Induced Apoptosis of Spermatocytes" International Journal of Molecular Sciences 21, no. 1: 267. https://doi.org/10.3390/ijms21010267
APA StyleJanus, P., Mrowiec, K., Vydra, N., Widłak, P., Toma-Jonik, A., Korfanty, J., Smolarczyk, R., & Widłak, W. (2020). PHLDA1 Does Not Contribute Directly to Heat Shock-Induced Apoptosis of Spermatocytes. International Journal of Molecular Sciences, 21(1), 267. https://doi.org/10.3390/ijms21010267





