Calcium-Binding Proteins in the Nervous System during Hibernation: Neuroprotective Strategies in Hypometabolic Conditions?
Abstract
:1. Introduction
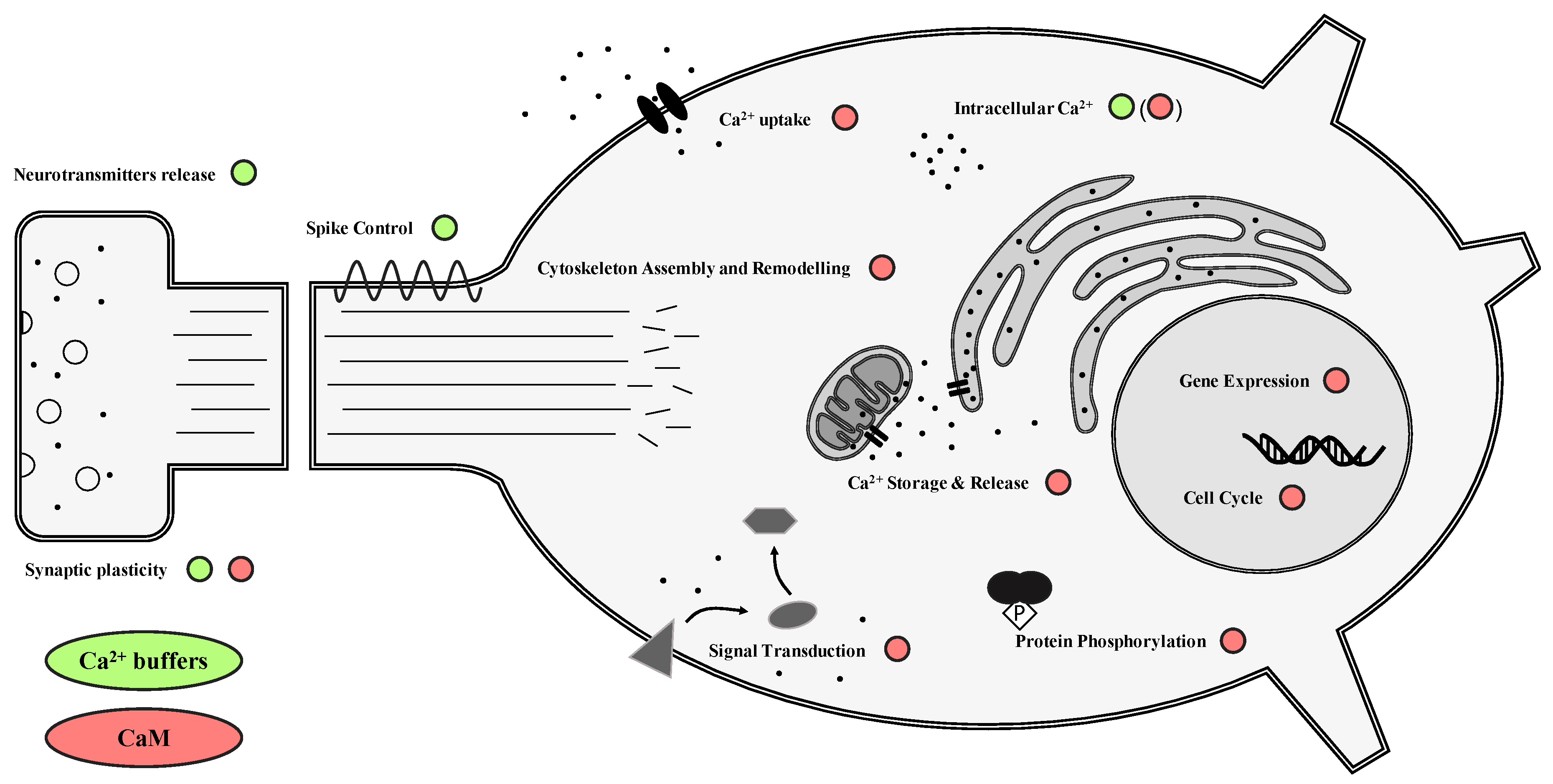
2. CBPs in the Central Nervous System
2.1. Calcium Buffers
2.2. Calcium Sensors
3. Hibernation: Who, How, and Why is it Interesting?
- It is a convenient and efficient model to study reaction and adaptation to extreme conditions at different levels of organization, from behavior down to the genetic control and the changes in molecular interactions.
- Hibernating animals show unmatched tolerance to these conditions thanks to protective mechanisms; this is especially true for the nervous system, which requires large amounts of energy to be maintained.
4. CBPs in the Hibernating Nervous System: A Role in Neuroprotection and Plasticity?
4.1. Ca2+ and CBPs
4.2. CBPs and Cytoskeleton Components
5. Hibernation and Translational Medicine: the Case of Neurodegeneration
6. Conclusions and Future Directions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| AD | Alzheimer’s disease |
| ARG | Autoradiography |
| CA1, 2 | Cornu Ammonis 1, 2 |
| CaM | Calmodulin |
| CAMK | Ca2+/calmodulin-dependent kinase |
| CAMKII | Ca2+/calmodulin-dependent kinase 2 |
| CB | Calbindin |
| CBPs | Calcium-binding proteins |
| CC | Cell count |
| CisPt | Cisplatin |
| CNS | Central nervous system |
| CR | Calretinin |
| DADLE | [D-Ala2, D-Leu5]-enkephalin |
| DG | Dentate gyrus |
| ERK1 | Extracellular regulated protein kinase 1 |
| ERK2 | Extracellular regulated protein kinase 2 |
| GABA | γ-aminobutyric acid |
| HIT | Hibernation induction trigger |
| HP | Hibernation-specific protein |
| ID | Immunodot |
| IHC | Immunohistochemistry |
| ISH | In situ hybridization |
| MAP | Microtubule-associated protein |
| MAP-2 | Microtubule-associated protein 2 |
| NF-H | Heavy subunit of neurofilaments |
| NFT | Neurofibrillary tangles |
| P | Phosphorylated |
| P-NP | Phosphorylated and non-phosphorylated |
| P-Tau | Phosphorylated tau |
| PKA | Protein kinase A |
| pNF-H | Phosphorylated heavy subunit of neurofilaments |
| PP1 | Protein phosphatase 1 |
| PP2A | Protein phosphatase 2A |
| PP2C | Protein phosphatase 2C |
| PtAcacDMS | [Pt(O,O′-acac)(γ-acac)(DMS)] |
| PV | Parvalbumin |
| RIA | Radioimmunoassay |
| SAPK/JNK | Stress-activated protein kinase/Jun-amino-terminal kinase |
| SI | Signal intensity |
| WB | Western blot |
References
- Choi, D.W. Excitotoxic cell death. J. Neurobiol. 1992, 23, 1261–1276. [Google Scholar] [CrossRef]
- Hutchins, J.B.; Barger, S.W. Why neurons die: Cell death in the nervous system. Anat. Rec. 1998, 253, 79–90. [Google Scholar] [CrossRef] [Green Version]
- Deshpande, J.K.; Siesjo, B.K.; Wieloch, T. Calcium accumulation and neuronal damage in the rat hippocampus following cerebral ischemia. J. Cereb. Blood Flow Metab. 1987, 7, 89–95. [Google Scholar] [CrossRef] [PubMed]
- Ming, G.; Song, H. Adult neurogenesis in the mammalian central nervous system. Annu. Rev. Neurosci. 2005, 28, 223–250. [Google Scholar] [CrossRef] [PubMed]
- Giorgi, C.; Danese, A.; Missiroli, S.; Patergnani, S.; Pinton, P. Calcium dynamics as a machine for decoding signals. Trends Cell Biol. 2018, 28, 258–273. [Google Scholar] [CrossRef]
- Humeau, J.; Bravo-San Pedro, J.M.; Vitale, I.; Nunez, L.; Villalobo, C.; Kroemer, G.; Senovilla, L. Calcium signaling and cell cycle: Progression or death. Cell Calcium 2018, 70, 3–15. [Google Scholar] [CrossRef] [PubMed]
- Gasperini, R.J.; Pavez, M.; Thompson, A.C.; Mitchell, C.B.; Hardy, H.; Young, K.M.; Chilton, J.K.; Foa, L. How does calcium interact with the cytoskeleton to regulate growth cone motility during axon path finding? Mol. Cell. Neurosci. 2017, 84, 29–35. [Google Scholar] [CrossRef]
- Berridge, M.J.; Bootman, M.D.; Roderick, H.L. Calcium signalling: Dynamics, homeostasis and remodelling. Nat. Rev. Mol. Cell Biol. 2003, 4, 517–529. [Google Scholar] [CrossRef]
- Elliott, E.; Malouf, A.; Catterall, W. Role of calcium channel subtypes in calcium transients in hippocampal CA3 neurons. J. Neurosci. 2018, 15, 6433–6444. [Google Scholar] [CrossRef]
- Carafoli, E.; Santella, L.; Branca, D.; Brini, M. Generation, control, and processing of cellular calcium signals. Crit. Rev. Biochem. Mol. Biol. 2001, 36, 107–260. [Google Scholar] [CrossRef]
- Bagur, R.; Hajnoczky, G. Intracellular Ca2+ sensing: Its role in calcium homeostasis and signaling. Mol. Cell 2017, 66, 780–788. [Google Scholar] [CrossRef]
- Kreutz, M.R.; Naranjo, K.R.; Koch, K.-W.; Schwaller, B. The neuronal functions of EF-hand Ca2+-binding proteins. Front. Mol. Neurosci. 2012, 5, 92. [Google Scholar] [CrossRef]
- Pchitskaya, E.; Popugaeva, E.; Bezprozvanny, I. Calcium signaling and molecular mechanisms underlying neurodegenerative diseases. Cell Calcium 2018, 70, 87–94. [Google Scholar] [CrossRef]
- Bezprozvanny, I. Calcium signaling and neurodegenerative diseases. Trends Mol. Med. 2009, 15, 89–100. [Google Scholar] [CrossRef] [Green Version]
- Bezprozvanny, I.; Mattson, M.P. Neuronal calcium mishandling and the pathogenesis of Alzheimer’s disease. Trends Neurosci. 2008, 31, 454–463. [Google Scholar] [CrossRef]
- Berridge, M.J. Calcium signalling and Alzheimer’s disease. Neurochem. Res. 2011, 36, 1149–1156. [Google Scholar] [CrossRef]
- Vignola, C.; Necchi, D.; Scherini, E.; Bernocchi, G. MPTP-induced changes in the monkey cerebellum - immunohistochemistry of calcium-binding and cytoskeletal proteins. Neurodeg. 1994, 3, 25–31. [Google Scholar]
- Scherini, E.; Bernocchi, G. CisDDP treatment and development of the rat cerebellum. Prog. Neurobiol. 1994, 42, 161–196. [Google Scholar] [CrossRef]
- Cerri, S.; Piccolini, V.M.; Santin, G.; Bottone, M.G.; de Pascali, S.A.; Migoni, D.; Iadarola, P.; Fanizzi, F.P.; Bernocchi, G. The developmental neurotoxicity study of platinum compounds. Effects of cisplatin versus a novel Pt(II) complex on rat cerebellum. Neurotoxicol. Teratol. 2011, 33, 273–281. [Google Scholar] [CrossRef]
- Permyakov, E.A.; Kretsinger, R.H. Calcium binding proteins; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2010; ISBN 9780470525845. [Google Scholar]
- Kretsinger, R.H. Calcium-binding proteins. Annu. Rev. Biochem. 1976, 45, 239–266. [Google Scholar] [CrossRef]
- Schwaller, B. Emerging functions of the “Ca2+ buffers” parvalbumin, calbindin D-28k and calretinin in the brain. In Handbook of Neurochemistry and Molecular Neurobiology: Neural Protein Metabolism and Function; Lajtha, A., Banik, N., Eds.; Springer: Boston, MA, USA, 2007; pp. 117–221. ISBN 9780387303468. [Google Scholar]
- Schwaller, B. Cytosolic Ca2+ buffers. Cold Spring Harb. Perspect. Biol. 2010, 2, a004051. [Google Scholar] [CrossRef]
- Schwaller, B.; Meyer, M.; Schiffmann, S. ‘New’ functions for ‘old’ proteins: The role of the calcium-binding proteins calbindin D-28k, calretinin and parvalbumin, in cerebellar physiology. Studies with knockout mice. The Cerebellum 2002, 1, 241–258. [Google Scholar] [CrossRef]
- Andressen, C.; Blumcke, I.; Celio, M.R. Calcium-binding proteins: Selective markers of nerve cells. Cell Tissue Res. 1993, 271, 181–208. [Google Scholar] [CrossRef]
- Bastianelli, E. Distribution of calcium-binding proteins in the cerebellum. Cerebellum 2003, 2, 242–262. [Google Scholar] [CrossRef]
- Gonchar, Y.; Wang, Q.; Burkhalter, A. Multiple distinct subtypes of GABAergic neurons in mouse visual cortex identified by triple immunostaining. Front. Neuroanat. 2008, 1, 1–11. [Google Scholar] [CrossRef]
- Litwinowicz, B.; Labuda, C.; Kowiański, P.; Spodnik, J.H.; Ludkiewicz, B.; Wojcik, S.; Morys, J. Developmental pattern of calbindin D28k protein expression in the rat striatum and cerebral cortex. Folia Morphol. (Warsz). 2003, 62, 327–329. [Google Scholar]
- Piccolini, V.M.; Bottone, M.G.; Bottiroli, G.; Pascali, S.A.; De Fanizzi, F.P.; Bernocchi, G. Platinum drugs and neurotoxicity: Effects on intracellular calcium homeostasis. Cell. Biol. Toxicol. 2013, 29, 339–353. [Google Scholar] [CrossRef]
- Ahn, J.H.; Hong, S.; Park, J.H.; Kim, I.H.; Cho, J.H.; Lee, T.-K.; Lee, J.-C.; Chen, B.H.; Shin, B.-N.; Bae, E.J.; et al. Immunoreactivities of calbindin-D28k, calretinin and parvalbumin in the somatosensory cortex of rodents during normal aging. Mol. Med. Rep. 2017, 16, 7191–7198. [Google Scholar] [CrossRef] [Green Version]
- Bae, J.E.; Chen, H.B.; Shin, B.-N.; Cho, J.H.; Kim, I.H.; Park, J.H.; Lee, J.-C.; Tae, H.J.; Choi, S.Y.; Kim, J.-D.; et al. Comparison of immunoreactivities of calbindin-D28k, calretinin and parvalbumin in the striatum between young, adult and aged mice, rats and gerbils. Neurochem. Res. 2015, 40, 864–872. [Google Scholar] [CrossRef]
- Schwaller, B. The use of transgenic mouse models to reveal the functions of Ca2+ buffer proteins in excitable cells. Biochim. Biophys. Acta 2012, 1820, 1294–1303. [Google Scholar] [CrossRef]
- Sayer, R.J.; Turnbull, C.I.; Hubbard, M.J. Calbindin(28kDa) is specifically associated with extranuclear constituents of the dense particulate fraction. Cell Tissue Res. 2000, 302, 171–180. [Google Scholar] [CrossRef]
- Necchi, D.; Soldani, C.; Pisu, M.B.; Bernocchi, G.; Scherini, E. Distribution of calretinin-like immunoreactivity in the brain of Rana esculenta. J. Chem. Neuroanat. 1999, 16, 233–243. [Google Scholar] [CrossRef]
- Schwaller, B. Calretinin: From a “simple” Ca2+ buffer to a multifunctional protein implicated in many biological processes. Front. Neuroanat. 2014, 8, 1–7. [Google Scholar] [CrossRef]
- Caballero, A.; Flores-Barrera, E.; Cass, D.K.; Tseng, K.Y. Differential regulation of parvalbumin and calretinin interneurons in the prefrontal cortex during adolescence. Brain Struct. Funct. 2014, 219, 395–406. [Google Scholar] [CrossRef] [PubMed]
- Ueno, H.; Suemitsu, S.; Okamoto, M.; Matsumoto, Y.; Ishihara, T. Parvalbumin neurons and perineuronal nets in the mouse prefrontal cortex. Neuroscience 2017, 343, 115–127. [Google Scholar] [CrossRef]
- Yamada, J.; Jinno, S. Spatio-temporal differences in perineuronal net expression in the mouse hippocampus, with reference to parvalbumin. Neuroscience 2013, 253, 368–379. [Google Scholar] [CrossRef]
- Pisu, M.B.; Roda, E.; Avella, D.; Bernocchi, G. Developmental plasticity of rat cerebellar cortex after cisplatin injury: Inhibitory synapses and differentiating Purkinje neurons. Neuroscience 2004, 129, 655–664. [Google Scholar] [CrossRef] [PubMed]
- McMahon, S.M.; Jackson, M.B. An inconvenient truth: Calcium sensors are calcium buffers. Trends Neurosci. 2018, 41, 880–884. [Google Scholar] [CrossRef]
- Burgoyne, R.D. Neuronal calcium sensor proteins: Generating diversity in neuronal Ca2+ signalling. Nat. Rev. Neurosci. 2007, 8, 182–193. [Google Scholar] [CrossRef]
- Zhang, M.; Abrams, C.; Wang, L.; Gizzi, A.; He, L.; Lin, R.; Chen, Y.; Loll, P.J.; Pascal, J.M.; Zhang, J. Structural basis for calmodulin as a dynamic calcium sensor. Structure 2012, 20, 911–923. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Villarroel, A.; Taglialatela, M.; Bernardo-Seisdedos, G.; Alaimo, A.; Agirre, J.; Alberdi, A.; Gomis-Perez, C.; Soldovieri, M.V.; Ambrosino, P.; Malo, C.; et al. The ever changing moods of calmodulin: How structural plasticity entails transductional adaptability. J. Mol. Biol. 2014, 426, 2717–2735. [Google Scholar] [CrossRef]
- Klee, C.B.; Vanaman, T.C. Calmodulin. Adv. Protein Chem. 1982, 35, 213–321. [Google Scholar]
- Means, A.R.; Tash, J.S.; Chafouleas, J.G. Physiological implications in the presence, distribution and regulation of calmodulin in eukaryotic cells. Physiol. Rev. 1982, 62, 1–39. [Google Scholar] [CrossRef]
- Cheung, W.Y. Calmodulin plays a pivotal role in cellular regulation. Sciencee 1978, 207, 19–27. [Google Scholar] [CrossRef]
- Sharma, R.K.; Parameswaran, S. Calmodulin-binding proteins: A journey of 40 years. Cell Calcium 2018, 75, 89–100. [Google Scholar] [CrossRef]
- Chin, D.; Means, A.R. Calmodulin: A prototypical calcium sensor. Trends Cell Biol. 2000, 10, 322–328. [Google Scholar] [CrossRef]
- Xia, Z.; Storm, D.R. The role of calmodulin as a signal integrator for synaptic plasticity. Nat. Rev. Neurosci. 2005, 6, 267–276. [Google Scholar] [CrossRef] [PubMed]
- Berchtold, M.W.; Villalobo, A. The many faces of calmodulin in cell proliferation, programmed cell death, autophagy, and cancer. Biochim. Biophys. Acta 2014, 1843, 398–435. [Google Scholar] [CrossRef] [Green Version]
- Takemoto-Kimura, S.; Suzuki, K.; Horigane, S.; Kamijo, S.; Inoue, M.; Sakamoto, M.; Fujii, H.; Bito, H. Calmodulin kinases: Essential regulators in health and disease. J. Neurochem. 2017, 141, 808–818. [Google Scholar] [CrossRef]
- Simon, B.; Huart, A.; Wilmanns, M. Molecular mechanisms of protein kinase regulation by calcium/calmodulin. Bioorg. Med. Chem. 2015, 23, 2749–2760. [Google Scholar] [CrossRef] [PubMed]
- Colbran, R.J.; Schworer, C.M.; Hashimoto, Y.; Fong, Y.; Rich, D.P.; Smith, M.K.; Soderling, T.R. Calcium/calmodulin-dependent protein kinase II. Biochem. J. 1989, 258, 313–325. [Google Scholar] [CrossRef] [Green Version]
- Liu, X.; Murray, K.D. Neuronal excitability and calcium/calmodulin-dependent protein kinase type II: Location, location, location. Epilepsia 2012, 53, 45–52. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zadrozna, M.; Nowak, B.; Lason-Tyburkiewicz, M.; Wolak, M.; Sowa-Kucma, M.; Papp, M.; Ossowska, G.; Pilc, A.; Nowak, G. Different pattern of changes in calcium binding proteins immunoreactivity in the medial prefrontal cortex of rats exposed to stress models of depression. Pharmacol. Reports 2011, 63, 1539–1546. [Google Scholar] [CrossRef]
- Eyles, D.W.; Mcgrath, J.J.; Reynolds, G.P. Neuronal calcium-binding proteins and schizophrenia. Schizophr. Res. 2002, 57, 27–34. [Google Scholar] [CrossRef]
- Freund, T.F.; Buzsaki, G.; Leon, A.; Baimbridge, K.G.; Somogyi, P. Relationship of neural vulnerability and calcium binding protein immunoreactivity in ischemia. Exp. Brain Res. 1990, 83, 55–66. [Google Scholar] [CrossRef]
- O’Day, D.H.; Eshak, K.; Myre, M.A. Calmodulin binding proteins and Alzheimer’s disease. J. Alzheimer’s Dis. 2015, 46, 553–569. [Google Scholar] [CrossRef]
- Calixto, A. Life without food and the implications for neurodegeneration. Adv. Genet. 2015, 92, 53–74. [Google Scholar]
- Geiser, F. Hibernation. Curr. Biol. 2013, 23, 188–193. [Google Scholar] [CrossRef]
- Staples, J.F. Metabolic flexibility: Hibernation, torpor, and estivation. Compr. Physiol. 2016, 6, 737–771. [Google Scholar]
- Rubtsov, A.M. Hibernation: Protein adaptations. In Protein Adaptations and Signal Transductions; Storey, K.B., Storey, J.M., Eds.; Elsevier Science B.V.: Amsterdam, The Netherlands, 2001; pp. 57–71. [Google Scholar]
- Carey, H.V.; Andrews, M.T.; Martin, S.L. Mammalian hibernation: Cellular and molecular responses to depressed metabolism and low temperature. Am. Physiol. Soc. 2003, 83, 1153–1181. [Google Scholar] [CrossRef] [PubMed]
- Boutilier, R.G. Mechanisms of cell survival in hypoxia and hypothermia. J. Exp. Biol. 2001, 204, 3171–3181. [Google Scholar] [PubMed]
- Schwartz, C.; Andrews, M.T. Circannual transitions in gene expression: Lessons from seasonal adaptations. Curr. Top. Dev. Biol. 2013, 105, 247–273. [Google Scholar] [PubMed] [Green Version]
- Andrews, M.T. Molecular interactions underpinning the phenotype of hibernation in mammals. J. Exp. Zool. 2019, 222, jeb160606. [Google Scholar] [CrossRef] [PubMed]
- Lust, W.D.; Wheaton, A.B.; Feussner, G.; Passonneau, J. Metabolism in the hamster brain during hibernation and arousal. Brain Res. 1989, 489, 12–20. [Google Scholar] [CrossRef]
- Morin, P.J.; Storey, K.B. Mammalian hibernation: Differential gene expression and novel application of epigenetic controls. Int. J. Dev. Biol. 2009, 53, 433–442. [Google Scholar] [CrossRef]
- Williams, C.T.; Barnes, B.M.; Buck, C.L. Integrating physiology, behavior, and energetics: Biologging in a free-living arctic hibernator. Comp. Biochem. Physiol. Part A Mol. Integr. Physiol. 2016, 202, 53–62. [Google Scholar] [CrossRef]
- Yan, J.; Barnes, B.M.; Kohl, F.; Marr, T.G. Modulation of gene expression in hibernating arctic ground squirrels. Physiol. Genomicsl 2008, 32, 170–181. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Antón-Fernández, A.; León-Espinosa, G.; DeFelipe, J.; Munoz, A. Changes in the Golgi apparatus of neocortical and hippocampal neurons in the hibernating hamster. Front. Neuroanat. 2015, 9, 1–15. [Google Scholar] [CrossRef]
- Storey, K.B.; Heldmaier, G.; Rider, M.H. Mammalian hibernation: Physiology, cell signaling, and gene controls on metabolic rate depression. In Dormancy and Resistance in Harsh Environments; Lubzens, E., Cerda, J., Clark, M.S., Eds.; Springer: Berlin/Heidelberg, Germany, 2010; pp. 227–252. ISBN 9783642124228. [Google Scholar]
- Horowitz, J.M.; Horrigan, D.J. Hibernation in mammals: Central nervous system function. Compr. Physiol. 2011, 1, 533–539. [Google Scholar]
- Storey, K.B. Out cold: Biochemical regulation of mammalian hibernation—A mini-review. Gerontology 2010, 56, 220–230. [Google Scholar] [CrossRef]
- Tsukamoto, D.; Hasegawa, T.; Hirose, S.; Sakurai, Y.; Ito, M.; Takamatsu, N. Circadian transcription factor HSF1 regulates differential HSP70 gene transcription during the arousal-torpor cycle in mammalian hibernation. Sci. Rep. 2019, 9, 1–11. [Google Scholar] [CrossRef]
- Ma, Y.L.; Zhu, X.; Rivera, P.M.; Tøien, Ø.; Barnes, B.M.; Lamanna, J.C.; Smith, M.A.; Drew, K.L. Absence of cellular stress in brain after hypoxia induced by arousal from hibernation in Arctic ground squirrels. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2005, 289, R1297–R1306. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bogren, L.K.; Drew, K.L. Ischemia/reperfusion injury resistance in hibernators is more than an effect of reduced body temperature or winter season. Temperature 2014, 1, 87–88. [Google Scholar] [CrossRef] [Green Version]
- Drew, K.L.; Harris, M.B.; LaManna, J.C.; Smith, M.A.; Zhu, X.W.; Ma, Y.L. Hypoxia tolerance in mammalian heterotherms. J. Exp. Biol. 2004, 207, 3155–3162. [Google Scholar] [CrossRef] [Green Version]
- Dave, K.R.; Prado, R.; Raval, A.P.; Drew, K.L.; Perez-Pinzon, M.A. The Arctic ground squirrel brain is resistant to injury from cardiac arrest during euthermia. Stroke 2006, 37, 1261–1265. [Google Scholar] [CrossRef]
- Bhowmick, S.; Moore, J.T.; Kirschner, D.L.; Drew, K.L. Arctic ground squirrel hippocampus tolerates oxygen glucose deprivation independent of hibernation season even when not hibernating and after ATP depletion, acidosis, and glutamate efflux. J. Neurochem. 2017, 142, 160–170. [Google Scholar] [CrossRef]
- Larson, J.; Drew, K.L.; Folkow, L.P.; Milton, S.L.; Park, T.J. No oxygen? No problem! Intrinsic brain tolerance to hypoxia in vertebrates. J. Exp. Biol. 2014, 217, 1024–1039. [Google Scholar] [CrossRef] [PubMed]
- Fu, W.; Hu, H.; Dang, K.; Chang, H.; Du, B.; Wu, X.; Gao, Y. Remarkable preservation of Ca2+ homeostasis and inhibition of apoptosis contribute to anti-muscle atrophy effect in hibernating Daurian ground squirrels. Sci. Rep. 2016, 6, 27020. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.Q.; Lakatta, E.G.; Cheng, H.; Zhou, Z.Q. Adaptive mechanisms of intracellular calcium homeostasis in mammalian hibernators. J. Exp. Biol. 2002, 205, 2957–2962. [Google Scholar]
- Andrews, M.T. Advances in molecular biology of hibernation in mammals. BioEssays 2007, 29, 431–440. [Google Scholar] [CrossRef] [PubMed]
- Milner, R.E.; Michalak, M.; Wang, L.C.H. Altered properties of calsequestrin and the ryanodine receptor in the cardiac sarcoplasmic reticulum of hibernating mammals. Biochim. Biophys. Acta Biomembr. 1991, 1063, 120–128. [Google Scholar] [CrossRef]
- Càceres, C.E. Dormancy in invertebrates. Invertebr. Biol. 1997, 116, 371–383. [Google Scholar] [CrossRef]
- Storey, K.B.; Storey, J.M. Hibernation: Poikilotherms. In eLS; John Wiley & Sons: Hoboken, NJ, USA, 2011; ISBN 9780470015902. [Google Scholar] [Green Version]
- Costanzo, J.P. Overwintering adaptations and extreme freeze tolerance in a subarctic population of the wood frog, Rana sylvatica. J. Comp. Physiol. B 2019, 189, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Ansart, A.; Vernon, P.; Daguzan, J. Photoperiod is the main cue that triggers supercooling ability in the land snail, Helix aspersa (Gastropoda, Helicidae). Cryobiology 2001, 42, 266–273. [Google Scholar] [CrossRef]
- Ansart, A.; Vernon, P. Cold hardiness in molluscs. Acta Oecologica 2003, 24, 95–102. [Google Scholar] [CrossRef]
- Ansart, A.; Nicolai, A.; Vernon, P.; Madec, L. Do ice nucleating agents limit the supercooling ability of the land snail Cornu aspersum? Cryo-Letters 2010, 31, 329–340. [Google Scholar]
- Nowakowska, A.; Caputa, M.; Rogalska, J. Seasonal changes in cryoprotectants concentrations in Helix pomatia snails. J. Physiol. Pharmacol. 2006, 8, 93–105. [Google Scholar]
- Nicolai, A.; Filser, J.; Lenz, R.; Bertrand, C.; Charrier, M. Quantitative assessment of hemolymph metabolites in two physiological states and two populations of the land snail Helix pomatia. Physiol. Biochem. Zool. 2012, 85, 274–284. [Google Scholar] [CrossRef] [PubMed]
- León-Olea, M.; Miller-Pérez, C.; Cruz, R.; Antón, B.; Vega, R.; Soto, E. Immunohistochemical localization and electrophysiological action of nociceptin/orphanin-FQ in the snail (Helix aspersa) neurons. Neurosci. Lett. 2001, 316, 141–144. [Google Scholar] [CrossRef]
- Vignola, C.; Fenoglio, C.; Scherini, E.; Bernocchi, G. The cerebral neurons of Helix aspersa during hibernation. Changes in the cytochemical detection of calmodulin, cytoskeletal components and phosphatases. Tissue Cell 1995, 27, 185–196. [Google Scholar] [CrossRef]
- Moreton, R.B. Electrophysiology and ionic movements in the central nervous system of the snail, Helix aspersa. J. Exp. Biol. 1972, 57, 513–541. [Google Scholar] [PubMed]
- Korobtsov, G.N.; Sakharov, D.A. Effect of serotonin and acetylcholine on neurons in the central nervous system of snails. Neurophysiology 1974, 6, 512–517. [Google Scholar]
- Pirger, Z.; Lubics, A.; Reglodi, D.; Laszlo, Z.; Mark, L.; Kiss, T. Mass spectrometric analysis of activity-dependent changes of neuropeptide profile in the snail, Helix pomatia. Neuropeptides 2010, 44, 475–483. [Google Scholar] [CrossRef]
- Kiss, T.; Battonyai, I.; Pirger, Z. Down regulation of sodium channels in the central nervous system of hibernating snails. Physiol. Behav. 2014, 131, 93–98. [Google Scholar] [CrossRef]
- Hiripi, L.; Salánki, J. Seasonal and activity-dependent changes of the serotonin level in the C.N.S. and heart of the snail (Helix Pomatia L.). Comp. Gen. Pharmacol. 1973, 4, 285–292. [Google Scholar] [CrossRef]
- Pisu, M.B.; Conforti, E.; Fenoglio, C.; Necchi, D.; Scherini, E.; Bernocchi, G. Nitric oxide-containing neurons in the nervous ganglia of Helix aspersa during rest and activity: Immunocytochemical and enzyme histochemical detection. J. Comp. Neurol. 1999, 409, 274–284. [Google Scholar] [CrossRef]
- Pisu, M.B.; Conforti, E.; Scherini, E.; Bernocchi, G. Gastrin-Cholecystokinin immunoreactivity in the central nervous system of Helix aspersa during rest and activity. J. Exp. Zool. 2000, 287, 29–37. [Google Scholar] [CrossRef]
- Bernocchi, G.; Vignola, C.; Scherini, E.; Necchi, D.; Pisu, M.B. Bioactive peptides and serotonin immunocytochemistry in the cerebral ganglia of hibernating Helix aspersa. J. Exp. Zool. 1998, 280, 354–367. [Google Scholar] [CrossRef]
- Michaelidis, B.; Loumbourdis, N.S.; Kapaki, E. Analysis of monoamines, adenosine and GABA in tissues of the land snail Helix lucorum and lizard Agama stellio stellio during hibernation. J. Exp. Biol. 2002, 205, 1135–1143. [Google Scholar]
- Breukelen, F.; Van Krumschnabel, G.; Podrabsky, J.E. Vertebrate cell death in energy-limited conditions and how to avoid it: What we might learn from mammalian hibernators and other stress-tolerant vertebrates. Apoptosis 2010, 15, 386–399. [Google Scholar] [CrossRef]
- Jinka, T.R.; Combs, V.M.; Drew, K.L. Translating drug-induced hibernation to therapeutic hypothermia. ACS Chem. Neurosci. 2015, 6, 899–904. [Google Scholar] [CrossRef]
- Drew, K.L.; Zuckerman, J.A.; Shenk, P.E.; Bogren, L.K.; Jinka, T.R.; Moore, J.T. Hibernation: A natural model of tolerance to cerebral ischemia/reperfusion. In Innate tolerance in the CNS; Gidday, J.M., Perez-Pinzon, M.A., Zhang, J.H., Eds.; Springer: New York, NY, USA, 2012; pp. 37–50. [Google Scholar]
- Malatesta, M.; Biggiogera, M.; Zancanaro, C. Hypometabolic induced state: A potential tool in biomedicine and space exploration. In Life in Extreme Environments; Amils, R., Ellis-Evans, C., Hinghofer-Szalkay, H., Eds.; Springer: Dordrecht, The Netherlands, 2006; pp. 47–60. [Google Scholar]
- Dave, K.R.; Christian, S.L.; Perez-Pinzon, M.A.; Drew, K.L. Neuroprotection: Lessons from hibernators. Comp. Biochem. Physiol. Part B Biochem. Mol. Biol. 2012, 162, 1–9. [Google Scholar] [CrossRef] [Green Version]
- Kondo, N. Endogenous circannual clock and HP complex in a hibernation control system. Cold Spring Harb. Symp. Quant. Biol. 2007, 72, 607–613. [Google Scholar] [CrossRef]
- Drew, K.L.; Buck, C.L.; Barnes, B.M.; Christian, S.L.; Rasley, B.T.; Harris, M.B. Central nervous system regulation of mammalian hibernation: Implications for metabolic suppression and ischemia tolerance. J. Neurochem. 2007, 102, 1713–1726. [Google Scholar] [CrossRef] [PubMed]
- Miller, L.P.; Hsu, C. Therapeutic potential for adenosine receptor activation in ischemic brain injury. J. Neurotrauma 1992, 9, S563–S567. [Google Scholar] [PubMed]
- Hanegan, J.L.; Williams, B.A. Ca2+ induced hypothermia in a hibernator (Citellus beechyi). Comp. Biochem. Physiol. 1975, 50, 247–252. [Google Scholar] [CrossRef]
- Myers, R.D.; Buckman, J.E. Deep hypothermia induced in the golden hamster by altering cerebral calcium levels. Am. J. Physiol. 1972, 223, 1313–1318. [Google Scholar] [CrossRef] [PubMed]
- Zhou, F.; Zhu, X.; Castellani, R.J.; Stimmelmayr, R.; Perry, G.; Smith, M.A.; Drew, K.L. Hibernation, a model of neuroprotection. Am. J. Pathol. 2001, 158, 2145–2151. [Google Scholar] [CrossRef]
- Cogut, V.; Bruintjes, J.J.; Eggen, B.J.L.; van der Zee, E.A.; Henning, R.H. Brain inflammatory cytokines and microglia morphology changes throughout hibernation phases in Syrian hamster. Brain Behav. Immun. 2018, 68, 17–22. [Google Scholar] [CrossRef]
- Malatesta, M.; Biggiogera, M.; Baldelli, B.; Barabino, S.M.L.; Martin, T.E.; Zancanaro, C. Hibernation as a far-reaching program for the modulation of RNA transcription. Microsc. Res. Tech. 2008, 71, 564–572. [Google Scholar] [CrossRef]
- Bernocchi, G.; Barni, S.; Scherini, E. The annual cycle of Erinaceus europaeus L. as a model for a further study of cytochemical heterogeneity in Purkinje neuron nuclei. Neuroscience 1986, 17, 427–437. [Google Scholar] [CrossRef]
- Giacometti, S.; Scherini, E.; Bernocchi, G. Seasonal changes in the nucleoli of Purkinje cells of the hedgehog cerebellum. Brain Res. 1989, 488, 365–368. [Google Scholar] [CrossRef]
- Bai, L.; Liu, E.; Bai, L.; Liu, B.; Ji, C.; Zhao, S.; Liu, S.; Wang, R.; Wang, W.; Yao, P.; et al. Hypoxic and cold adaptation insights from the himalayan marmot genome. iScience 2018, 11, 519–530. [Google Scholar] [CrossRef] [PubMed]
- Gonzales-Riano, C.; Leon-Espinosa, G.; Regalado-Reyes, M.; Garcia, A.; DeFelipe, J.; Barbas, C. Metabolomic study of hibernating syrian hamster brains: In search of neuroprotective agents. J. Proteome Res. 2019, 18, 1175–1190. [Google Scholar] [CrossRef]
- Hindle, A.G.; Martin, S.L. Cytoskeletal regulation dominates temperature-sensitive proteomic changes of hibernation in forebrain of 13-lined ground squirrels. PLoS One 2013, 8, e71627. [Google Scholar] [CrossRef]
- Gentile, N.T.; Brenner, M.; Spatz, M.; McCarron, R.M.; Hallenbeck, J.M. Decreased calcium accumulation in isolated nerve endings during hibernation in ground squirrels. Neurochem. Res. 2006, 21, 947–954. [Google Scholar] [CrossRef]
- Brauch, K.M.; Dhruv, N.D.; Hanse, E.A.; Andrews, M.T. Digital transcriptome analysis indicates adaptive mechanisms in the heart of a hibernating mammal. Physiol. Genomics 2005, 23, 227–234. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, J.; Yuan, L.; Sun, M.; Zhang, L.; Zhang, S. Screening of hibernation-related genes in the brain of Rhinolophus ferrumequinum during hibernation. Comp. Biochem. Physiol. Part B 2008, 149, 388–393. [Google Scholar] [CrossRef] [PubMed]
- Pisu, M.B.; Scherini, E.; Bernocchi, G. Immunocytochemical changes of cytoskeleton components and calmodulin in the frog cerebellum and optic tectum during hibernation. J. Chem. Neuroanat. 1998, 72, 63–73. [Google Scholar] [CrossRef]
- Roda, E.; Bottone, M.G.; Insolia, V.; Barni, S.; Bernocchi, G. Changes in the cerebellar cytoarchitecture of hibernating hedgehog Erinaceus europaeus L. (Mammalia): An immunocytochemical approach. Eur. Zool. J. 2017, 84, 496–511. [Google Scholar] [CrossRef]
- Schwartz, C.; Hampton, M.; Andrews, M.T. Seasonal and regional differences in gene expression in the brain of a hibernating mammal. PLoS One 2013, 8, e58427. [Google Scholar] [CrossRef] [PubMed]
- Frerichs, K.U.; Smith, C.B.; Brenner, M.; DeGracia, D.J.; Krause, G.S.; Marrone, L.; Dever, T.E.; Hallenbeck, J.M. Suppression of protein synthesis in brain during hibernation involves inhibition of protein initiation and elongation. Proc. Natl. Acad. Sci. USA 1998, 95, 14511–14516. [Google Scholar] [CrossRef] [Green Version]
- Chase, R. Structure and function in the cerebral ganglion. Microsc. Res. Tech. 2000, 49, 511–520. [Google Scholar] [CrossRef]
- Kerkut, G.A.; Lambert, J.D.C.; Gayton, R.J.; Loker, J.E.; Walker, R.J. Mapping of nerve cells in the suboesophageal ganglia of Helix aspersa. Comp. Biochem. Physiol. Part A Mol. Integr. Physiol. 1975, 50, 1–25. [Google Scholar] [CrossRef]
- Alvarez-Leefmans, F.J.; Rink, T.J.; Tsien, R.Y. Free calcium ions in neurones of Helix aspersa measured with ion-selective microelectrodes. J. Physiol. 1981, 315, 531–548. [Google Scholar] [CrossRef]
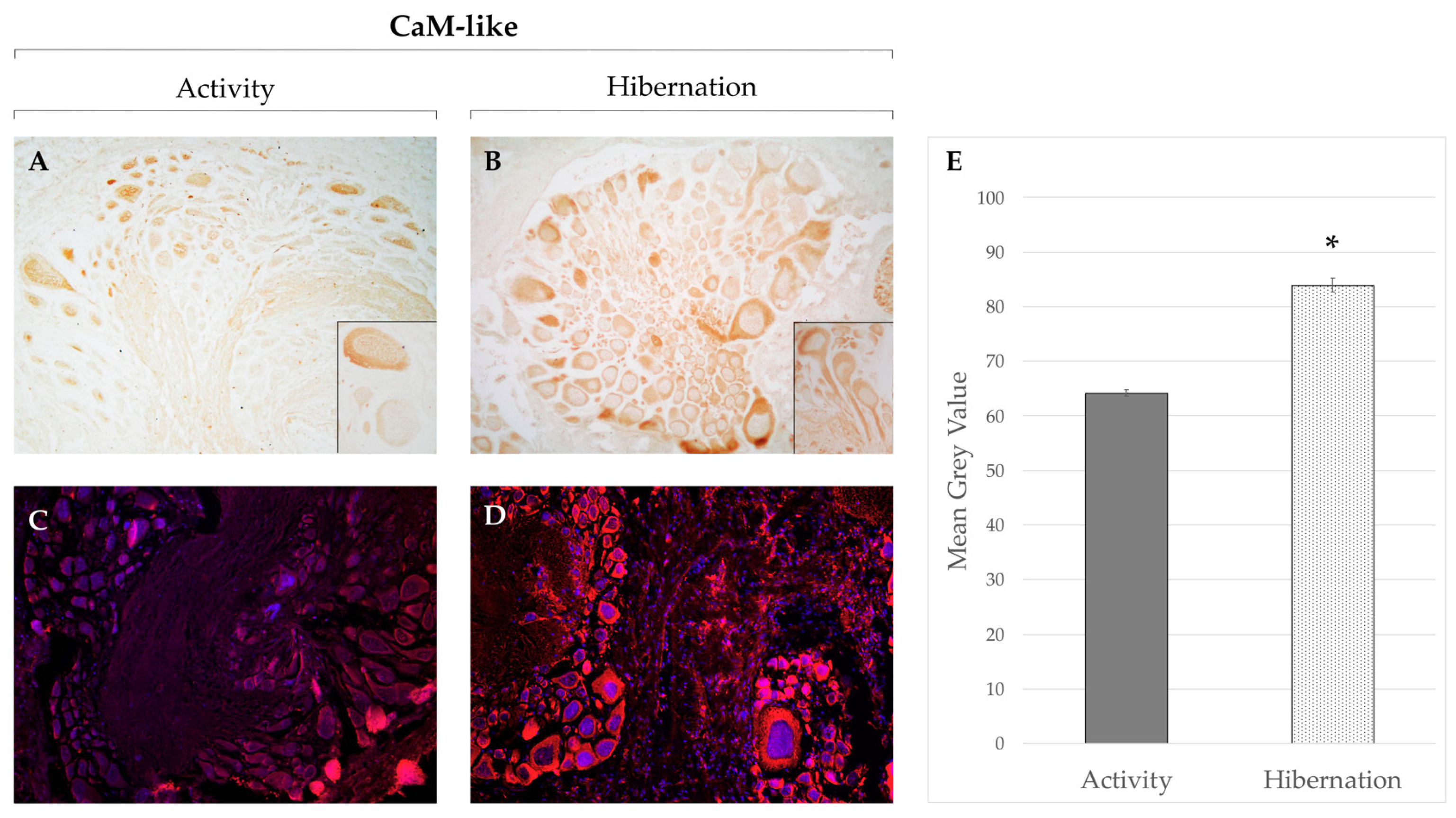
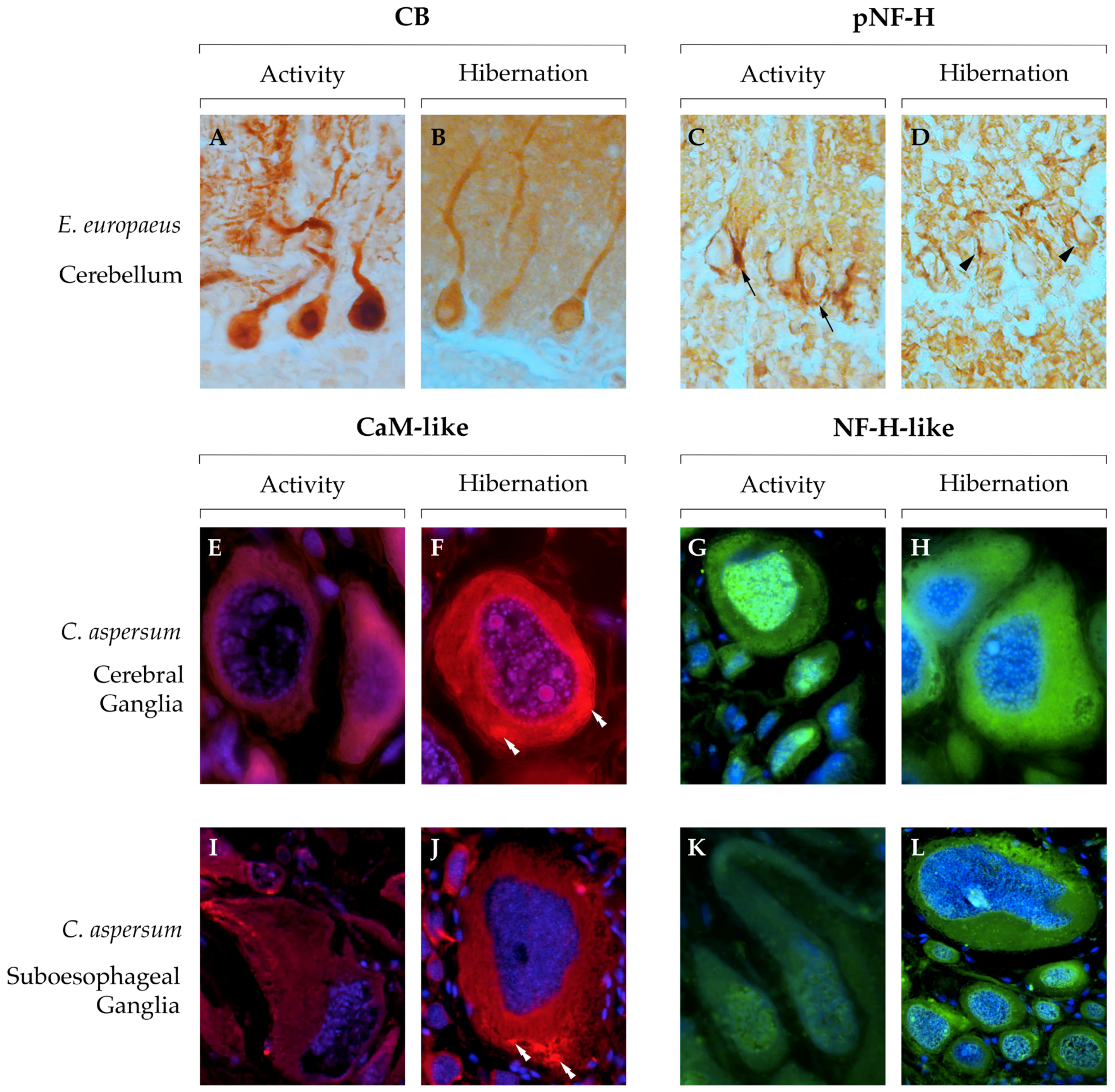
- Gattoni, G.; Insolia, V.; Bernocchi, G. Hibernation induces changes in the metacerebral neurons of Cornu aspersum: Distribution and co-localization of cytoskeletal and calcium-binding proteins. Invertebr. Neurosci. 2018, 18, 13. [Google Scholar] [CrossRef]
- Arendt, T.; Stieler, J.; Strijkstra, A.M.; Hut, R.A.; Rudiger, J.; Can der Zee, E.A.; Harkany, T.; Holzer, M.; Hartig, W. Reversible paired helical filament-like phosphorylation of tau is an adaptive process associated with neuronal plasticity in hibernating animals. Development/Plasticity/Repair 2003, 23, 6972–6981. [Google Scholar] [CrossRef]
- Su, B.; Wang, X.; Srew, K.L.; Perry, G.; Smith, M.A.; Zhu, X. Physiological regulation of tau phosphorylation during hibernation. J. Neurochem. 2008, 105, 2098–2108. [Google Scholar] [CrossRef] [Green Version]
- von der Ohe, C.G.; Garner, C.C.; Darian-smith, C.; Heller, H.C. Synaptic protein dynamics in hibernation. J. Neurosci. 2007, 27, 84–92. [Google Scholar] [CrossRef]
- Stieler, J.T.; Bullmann, T.; Kohl, F.; Tøien, Ø.; Bruckner, M.K.; Hartig, W.; Barnes, B.M.; Arendt, T. The physiological link between metabolic rate depression and tau phosphorylation in mammalian hibernation. PLoS ONE 2011, 6, e14530. [Google Scholar] [CrossRef] [PubMed]
- Härtig, W.; Stieler, J.; Boerema, A.S.; Wolf, J.; Schmidt, U.; Weißfuß, J.; Bullmann, T.; Strijkstra, A.M.; Arendt, T. Hibernation model of tau phosphorylation in hamsters: Selective vulnerability of cholinergic basal forebrain neurons – implications for Alzheimer’s disease. Eur. J. Neurosci. 2007, 25, 69–80. [Google Scholar] [CrossRef] [PubMed]
- Bullmann, T.; Seeger, G.; Stieler, J.; Hanics, J.; Reimann, K.; Kretzschmann, T.P.; Hilbrich, I.; Holzer, M.; Alpar, A.; Arendt, T. Tau phosphorylation-associated spine regression does not impair hippocampal-dependent memory in hibernating golden hamsters. Hippocampus 2016, 26, 301–318. [Google Scholar] [CrossRef]
- Stieler, J.T.; Borema, A.S.; Bullmann, T.; Kohl, F.; Strijkstra, A.M.; Barnes, B.M.; Arendt, T. Activity-state profile of tau kinases in hibernating animals. In Hypometabolism in Animals, Hibernation, Torpor and Cryobiology; Lovegrove, B.G., Mckechnie, A.E., Eds.; University of KwaZulu-Natal: Pietermaritzburg, South Africa, 2008; pp. 133–142. [Google Scholar]
- Bao, B.; Xu, W. Identification of gene expression changes associated with the initiation of diapause in the brain of the cotton bollworm, Helicoverpa armigera. BMC Genomics 2011, 12, 224. [Google Scholar] [CrossRef] [PubMed]
- Ahn, J.J.; Choi, K.; Huang, S.; Al Baki, A.; Ahmed, S.; Kim, Y. Calcium/calmodulin-dependent protein kinase II of the oriental fruit fly, Bactrocera dorsalis, and its association with rapid cold hardiness. J. Asia. Pac. Entomol. 2018, 21, 1275–1282. [Google Scholar] [CrossRef]
- MacDonald, J.A.; Storey, K.B. The effect of hibernation on protein phosphatases from ground squirrel organs. Arch. Biochem. Biophys. 2007, 468, 234–243. [Google Scholar] [CrossRef]
- Wu, C.; Reardon, A.J.; Storey, K.B. Effects of hibernation on regulation of mammalian protein phosphatase type-2-A. Cryobiology 2013, 66, 267–274. [Google Scholar] [CrossRef]
- Chen, Y.; Matsushita, M.; Nairn, A.C.; Damuni, Z.; Cai, D.; Frerichs, K.U.; Hallenbeck, J.M. Mechanisms for increased levels of phosphorylation of Elongation Factor-2 during hibernation in ground squirrels. Biochemistry 2001, 40, 11565–11570. [Google Scholar] [CrossRef]
- Popov, V.I.; Bocharova, L.S.; Bragin, A.G. Repeated changes of dendritic morphology in the hippocampus of ground squirrels in the course of hibernation. Neuroscience 1992, 48, 45–51. [Google Scholar] [CrossRef]
- Popov, V.I.; Bocharova, L.S. Hibernation-induced structural changes in synaptic contacts between mossy fibers and hippocampal pyramidal neurons. Neuroscience 1992, 48, 53–62. [Google Scholar] [CrossRef]
- von der Ohe, C.G.; Darian-smith, C.; Garner, C.C.; Heller, H.C. Ubiquitous and temperature-dependent neural plasticity in hibernators. J. Neurosci. 2006, 26, 10590–10598. [Google Scholar] [CrossRef]
- Magarinos, A.M.; Mcewen, B.S.; Saboureau, M.; Pevet, P. Rapid and reversible changes in intrahippocampal connectivity during the course of hibernation in European hamsters. Proc. Natl. Acad. Sci. USA 2006, 103, 18775–18780. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Horowitz, J.M.; Horwitz, B.A. Extreme neuroplasticity of hippocampal CA1 pyramidal neurons in hibernating mammalian species. Front. Neuroanat. 2019, 13, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Ruczynski, I.; Siemers, B.M. Hibernation does not affect memory retention in bats. Biol. Lett. 2011, 7, 153–155. [Google Scholar] [CrossRef] [PubMed]
- Clemens, L.E.; Heldmaier, G.; Exner, C. Keep cool: Memory is retained during hibernation in Alpine marmots. Physiol. Behav. 2009, 98, 78–84. [Google Scholar] [CrossRef] [PubMed]
- McNamara, M.C.; Riedesel, M.L. Memory and hibernation in Citellus lateralis. Science 1973, 179, 92–94. [Google Scholar] [CrossRef]
- Emirbekov, E.Z.; Pashaeva, M.E. Expression of cytoskeleton proteins in hypothalamic cells in winter sleeping ground squirrels Citellus pygmaeus Pallas during hibernation. Neurochem. J. 2014, 8, 178–183. [Google Scholar] [CrossRef]
- Dehmelt, L.; Halpain, S. The MAP2/Tau family of microtubule-associated proteins. Genome Biol. 2004, 6, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Penazzi, L.; Bakota, L.; Brandt, R. Microtubule dynamics in neuronal development, plasticity, and neurodegeneration. In International Review of Cell and Molecular Biology; Vanpouille-Box, C., Galluzzi, L., Eds.; Elsevier Inc.: Amsterdam, The Netherlands, 2016; Volume 321, pp. 89–169. [Google Scholar]
- Johnson, G.V.W.; Jope, R.S. The role of microtubule-associated protein 2 (MAP-2) in neuronal growth, plasticity and degeneration. J. Neurosci. Res. 1992, 33, 505–512. [Google Scholar] [CrossRef]
- Arendt, T.; Bullmann, T. Neuronal plasticity in hibernation and the proposed role of the microtubule-associated protein tau as a “master switch” regulating synaptic gain in neuronal networks. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2013, 305, 478–489. [Google Scholar] [CrossRef]
- Gómez-Ramos, A.; Smith, M.A.; Perry, G.; Avila, J. Tau phosphorylation and assembly. Acta Neurobiol. Exp. (Wars). 2004, 64, 33–39. [Google Scholar]
- Baudier, J.; Cole, R.D. Phosphorylation of tau proteins to a state like that in Alzheimer’s brain is catalyzed by a calcium/calmodulin-dependent kinase and modulated by phospholipids. J. Biol. Chem. 1987, 262, 17577–17583. [Google Scholar]
- Leon-Espinosa, G.; Garcia, E.; Garcia-Escudero, V.; Felix, H.; DeFelipe, F.; Avila, J. Changes in tau phosphorylation in hibernating rodents. J. Neurosci. Res. 2013, 91, 954–962. [Google Scholar] [CrossRef] [Green Version]
- Stieler, J.T.; Bullmann, T.; Kohl, F.; Barnes, B.M.; Arendt, T. PHF-like tau phosphorylation in mammalian hibernation is not associated with p25-formation. J. Neural Transm. 2009, 116, 345–350. [Google Scholar] [CrossRef] [PubMed]
- Oka, M.; Fukisaki, N.; Maruko-Otake, A.; Ohtake, Y.; Shimizu, S.; Saito, T.; Hisanaga, S.-I.; Iijima, K.M.; Ando, K. Calcium/Calmodulin-dependent protein kinase II promotes neurodegeneration caused by tau phosphorylated at Ser262/356 in a transgenic Drosophila model of tauopathy. J. Biochem. 2017, 162, 1–8. [Google Scholar] [CrossRef]
- Sánchez, C.; Díaz-Nido, J.; Avila, J. Phosphorylation of microtubule-associated protein 2 (MAP2) and its relevance for the regulation of the neuronal cytoskeleton function. Prog. Neurobiol. 2000, 61, 133–168. [Google Scholar] [CrossRef]
- Wei, Y.; Ye, J.; Wang, X.; Li-ping, Z.; Hu, Q.; Wang, Q.; Ke, D.; Tian, Q.; Wang, J.-Z. Tau-induced calcium/calmodulin-dependent protein kinase-IV activation aggravates nuclear tau hyperphosphorylation. Neurosci. Bull. 2017, 34, 261–269. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.C.; Wolff, J. Calmodulin binds to both microtubule-associated protein 2 and tau proteins. J. Biol. Chem. 1984, 259, 1226–1230. [Google Scholar] [PubMed]
- Padilla, R.; Maccioni, R.B.; Avila, J. Calmodulin binds to a tubulin binding site of the microtubule-associated protein tau. Mol. Cell. Biochem. 1990, 97, 35–41. [Google Scholar] [CrossRef] [PubMed]
- Kakiuchi, S.; Sobue, K. Calcium and calmodulin-dependent flip-flop mechanism in microtubule assembly-disassembly. FEBS Lett. 1981, 132, 141–143. [Google Scholar] [CrossRef]
- Liu, Q.; Xie, F.; Siedlak, S.L.; Nunomura, A.; Honda, K.; Moreira, P.I.; Zhua, X.; Smith, M.A.; Perry, G. Neurofilament proteins in neurodegenerative diseases. Cell. Mol. Life Sci. 2004, 61, 3057–3075. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ackerley, S.; Thornhill, P.; Grierson, A.J.; Brownlees, J.; Anderton, B.H.; Leigh, P.N.; Shaw, C.E.; Miller, C.C.J. Neurofilament heavy chain side arm phosphorylation regulates axonal transport of neurofilaments. J. Cell Biol. 2003, 161, 489–495. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Goldstein, M.E.; Sternberger, N.H.; Sternberger, L.A. Phosphorylation protects neurofilaments against proteolysis. J. Neuroimmunol. 1987, 14, 149–160. [Google Scholar] [CrossRef]
- Drew, K.L.; Rice, M.E.; Kuhn, T.B.; Smith, M.A. Neuroprotective adaptations in hibernation: Therapeutic implications for ischemia-reperfusion, traumatic brain injury ad neurodegenerative diseases. Free Radic. Biol. Med. 2001, 31, 563–573. [Google Scholar] [CrossRef]
- Stenzel-Poore, M.P.; Stevens, S.L.; Xiong, Z.; Lessov, N.S.; Harrington, C.A.; Mori, M.; Meller, R.; Rosenzweig, H.L.; Tobar, E.; Shaw, T.E.; et al. Effect of ischaemic preconditioning on genomic response to cerebral ischaemia: Similarity to neuroprotective strategies in hibernation and hypoxia-tolerant states. Lancet 2003, 362, 1028–1037. [Google Scholar] [CrossRef]
- Zancanaro, C.; Biggiogera, M.; Malatesta, M. Mammalian Hibernation:Relevance to a Possible Human Hypometabolic State; European Space Agency: Paris, France, 2004. [Google Scholar]
- Liska, M.G.; Crowley, M.G.; Tuazon, J.P.; Borlongan, C.V. Neuroprotective and neuroregenerative potential of pharmacologically-induced hypothermia with D-alanine D-leucine enkephalin in brain injury. Neural Regen. Res. 2018, 13, 2029–2037. [Google Scholar] [PubMed]
- Oeltgen, P.R.; Nilekani, S.P.; Nuchols, P.A.; Spurrier, W.A.; Su, T.P. Further studies on opioids and hibernation: Delta opioid receptor ligand selectively induced hibernation in summer-active ground squirrels. Life Sci. 1988, 43, 1565–1574. [Google Scholar] [CrossRef]
- Vecchio, L.; Soldani, C.; Bottone, M.G.; Malatesta, M.; Martin, T.E.; Rothblum, L.I.; Pellicciari, C.; Biggiogera, M. DADLE induces a reversible hibernation-like state in HeLa cells. Histochem. Cell Biol. 2006, 125, 193–201. [Google Scholar] [CrossRef] [PubMed]
- Su, T. Delta opioid peptide [D-Ala2,D-Leu5]Enkephalin promotes cell survival. J. Biomed. Sci. 2000, 7, 195–199. [Google Scholar] [CrossRef]
- Borlongan, C.V.; Wang, Y.; Su, T. Delta opioid peptide ((D-ALA2,D-LEU 5)Enkephalin: Linking hibernation and neuroprotection. Front. Biosci. 2004, 9, 3392–3398. [Google Scholar] [CrossRef]
- Logan, S.M.; Storey, K.B. Avoiding apoptosis during mammalian hibernation. Temperature 2017, 4, 15–17. [Google Scholar] [CrossRef]
- Cerri, S.; Bottiroli, G.; Bottone, M.G.; Barni, S.; Bernocchi, G. Cell proliferation and death in the brain of active and hibernating frogs. J. Anat. 2009, 215, 124–131. [Google Scholar] [CrossRef] [Green Version]
- McWhinney, S.R.; Goldberg, R.M.; McLeod, H.L. Platinum neurotoxicity pharmacogenetics. Mol. Cancer Ther. 2009, 8, 10–16. [Google Scholar] [CrossRef] [PubMed]
- Amptoulach, S.; Tsavaris, N. Neurotoxicity caused by the treatment with platinum analogues. Chemother. Res. Pract. 2011, 2011, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Bernocchi, G.; Bottone, M.G.; Piccolini, V.M.; Dal Bo, V.; Santin, G.; De Pascali, S.A.; Migoni, D.; Fanizzi, F.P. Developing central nervous system and vulnerability to platinum compounds. Chemother. Res. Pract. 2011, 2011, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Unel, C.C.; Erol, K. The role of ionic homeostasis in cisplatin-induced neurotoxicity: A preliminary study. Eurasian J. Med. 2018, 50, 81–85. [Google Scholar] [PubMed]
- Bernocchi, G.; Fanizzi, F.P.; Pascali, S.A.; de Piccolini, V.M.; Gasperini, C.; Insolia, V.; Bottone, M.G. Neurotoxic effects of platinum compounds: Studies in vivo on intracellular calcium homeostasis in the immature central nervous system. Toxics 2015, 3, 224–248. [Google Scholar] [CrossRef] [PubMed]
- Piccolini, V.M.; Esposito, A.; Dal Bo, V.; Insolia, V.; Bottone, M.G.; De Pascali, S.A.; Fanizzi, F.P.; Bernocchi, G. Cerebellum neurotransmission during postnatal development: [Pt(O,O′-acac)(γ-acac)(DMS)] vs cisplatin and neurotoxicity. Int. J. Dev. Neurosci. 2015, 40, 24–34. [Google Scholar] [CrossRef]
- Necchi, D.; Scherini, E.; Bernocchi, G. Glial cell reaction to cis-dichlorodiammine platinum treatment in the immature rat cerebellum. Exp. Neurol. 1997, 144, 219–226. [Google Scholar] [CrossRef]
- Scherini, E.; Biggiogera, M.; Bernocchi, G.; Mares, V. Damage and repair of the immature rat cerebellum after cis-dichlorodiammineplatinum II (cis-DDP) treatment. An ultrastructural study. Acta Neuropathol. 1987, 72, 218–228. [Google Scholar] [CrossRef]
- Bertram, L.; Tanzi, R.E. The genetic epidemiology of neurodegenerative disease. J. Clin. Invest. 2005, 115, 1449–1457. [Google Scholar] [CrossRef] [Green Version]
- LaFerla, F.M.; Oddo, S. Alzheimer’s disease: Aβ, tau and synaptic dysfunction. Trends Mol. Med. 2005, 11, 170–176. [Google Scholar] [CrossRef]
- Bakota, L.; Brandt, R. Tau biology and tau-directed therapies for Alzheimer’s disease. Drugs 2016, 76, 301–313. [Google Scholar] [CrossRef] [PubMed]
- Alzheimer’s Association Calcium Hypothesis Workgroup. Calcium hypothesis of Alzheimer’s disease and brain aging: A framework for integrating new evidence into a comprehensive theory of pathogenesis. Alzheimer’s Dement. 2017, 13, 178–182. [Google Scholar] [CrossRef] [PubMed]
- Llinas, R.; Moreno, H. Perspective on calcium and Alzheimer’s disease. Alzheimer’s Dement. 2017, 13, 196–197. [Google Scholar] [CrossRef]
- Iritani, S.; Niizato, K.; Emson, P.C. Relationship of calbindin D28K-immunoreactive cells and neuropathological changes in the hippocampal formation of Alzheimer’s disease. Neuropathology 2001, 21, 162–167. [Google Scholar] [CrossRef] [PubMed]
- Hoskins, B.; Ho, I.K. Effects of maturation and aging on calmodulin and calmodulin-regulated enzymes in various regions of mouse brain. Mech. Ageing Dev. 1986, 36, 173–186. [Google Scholar] [CrossRef]
- Lu, T.; Pan, Y.; Kao, S.; Li, C.; Kohane, I.; Chan, J.; Yankner, B.A. Gene regulation and DNA damage in the ageing human brain. Nature 2004, 429, 883–891. [Google Scholar] [CrossRef]
- Lesort, M.; Blanchard, C.; Yardin, C.; Esclaire, F.; Hugon, J. Cultured neurons expressing phosphorylated tau are more resistant to apoptosis induced by NMDA or serum deprivation. Mol. Brain Res. 1997, 45, 127–132. [Google Scholar] [CrossRef]
- Arendt, T.; Holzer, M.; Fruth, R.; Brückner, M.K.; Gärtner, U. Phosphorylation of tau, aβ-formation, and apoptosis after in vivo inhibition of PP-1 and PP-2A. Neurobiol. Aging 1998, 19, 3–13. [Google Scholar] [CrossRef]
- Bu, J.; Sathyendra, V.; Nagykery, N.; Geula, C. Age-related changes in calbindin-D28k, calretinin, and parvalbumin-immunoreactive neurons in the human cerebral cortex. Exp. Neurol. 2003, 182, 220–231. [Google Scholar] [CrossRef]
- Ueno, H.; Takao, K.; Suemitsu, S.; Murakami, S.; Kitamura, N.; Wani, K.; Okamoto Aoki, S.; Ishihara, T. Age-dependent and region-specific alteration of parvalbumin neurons and perineuronal nets in the mouse cerebral cortex. Neurochem. Int. 2018, 112, 59–70. [Google Scholar] [CrossRef] [PubMed]
- Villa, A.; Podini, P.; Panzeri, M.C.; Racchetti, G.; Meldolesi, J. Cytosolic Ca2+ binding proteins during rat brain ageind: Loss of calbindin and calretinin hippocampus, with no change in the cerebellum. Eur. J. Neurosci. 1994, 6, 1491–1499. [Google Scholar] [CrossRef]
- Ouda, L.; Burianova, J.; Syka, J. Age-related changes in calbindin and calretinin immunoreactivity in the central auditory system of the rat. Exp. Gerontol. 2012, 47, 497–506. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.H.; Hwang, I.K.; Yoo, K.-Y.; Choi, J.H.; Park, O.K.; Lee, J.-C.; Jeong, Y.-G.; Lee, I.S.; Won, M.-H. Calbindin D-28k immunoreactivity and its protein level in hippocampal subregions during normal aging in gerbils. Cell. Mol. Neurobiol. 2009, 29, 665–672. [Google Scholar] [CrossRef] [PubMed]
- Kishimoto, J.; Tsuchiya, T.; Cox, H.; Emson, P.C.; Nakayama, Y. Age-related changes of calbindin-D28k, calretinin and parvalbumin mRNAs in the hamster brain. Neurobiol. Aging 1998, 19, 77–82. [Google Scholar] [CrossRef]
- McLachlan, D.R.; Wong, L.; Bergeron, C.; Baimbridge, K.G. Calmodulin and calbindin D28K in Alzheimer disease. Alzheimer Dis. Assoc. Disord. 1987, 1, 171–179. [Google Scholar] [CrossRef]
- Ichimiya, Y.; Emson, P.C.; Mountjoy, C.Q.; Lawson, D.E.M.; Heizmann, C.W. Loss of calbindin-28K immunoreactive neurones from the cortex in Alzheimer-type dementia. Brain Res. 1988, 475, 156–159. [Google Scholar] [CrossRef]
- Ichimiya, Y.; Emson, P.C.; Mountjoy, C.Q.; Lawson, D.E.M.; Iizuka, R. Caibindin-immunoreactive cholinergic neurones in the nucleus basalis of Meynert in Alzheimer-type dementia. Brain Res. 1989, 499, 402–406. [Google Scholar] [CrossRef]
- Hof, P.R.; Morrison, J.H. Neocortical neuronal subpopulations labeled by a monoclonal antibody to calbindin exhibit differential vulnerability in Alzheimer’s disease. Exp. Neurol. 1991, 111, 293–301. [Google Scholar] [CrossRef]
- Nishiyama, E.; Ohwada, J.; Iwamoto, N.; Arai, H. Selective loss of calbindin D28K-immunoreative neurons in the cortical layer II in brains of Alzheimer’s disease: A morphometric study. Neurosci. Lett. 1993, 163, 223–226. [Google Scholar] [CrossRef]
- Satoh, J.; Tabira, T.; Sano, M.; Nakayama, H.; Tateishi, J. Parvalbumin-immunoreactive neurons in the human central nervous system are decreased in Alzheimer’s disease. Acta Neuropathol. 1991, 81, 388–395. [Google Scholar] [CrossRef] [PubMed]
- Brady, D.R.; Mufson, E.J. Parvalbumin-immunoreactive neurons in the hippocampal formation of Alzheimer’s diseased brain. Neuroscience 1997, 80, 1113–1125. [Google Scholar] [CrossRef]
- Solomon, B.; Koppel, R.; Jossiphov, J. Immunostaining of calmodulin and aluminium in Alzheimer’s disease-affected brains. Brain Res. Bull. 2001, 55, 253–256. [Google Scholar] [CrossRef]
- Palop, J.J.; Jones, B.; Kekonius, L.; Chin, J.; Yu, G.-Q.; Raber, J.; Masliah, E.; Mucke, L. Neuronal depletion of calcium-dependent proteins in the dentate gyrus is tightly linked to Alzheimer’s disease-related cognitive deficits. Proc. Natl. Acad. Sci. USA 2003, 100, 9572–9577. [Google Scholar] [CrossRef]
- Palop, J.J.; Chin, J.; Roberson, E.D.; Wang, J.; Thwin, M.T.; Bien-Ly, N.; Yoo, J.; Ho, K.O.; Yu, G.-Q.; Kreitzer, A.; et al. Aberrant excitatory neuronal activity and compensatory remodeling of inhibitory hippocampal circuits in mouse models of Alzheimer’s disease. Neuron 2007, 55, 697–711. [Google Scholar] [CrossRef]
- Zallo, F.; Gardenal, E.; Verkhratsky, A.; Julio, J. Loss of calretinin and parvalbumin positive interneurones in the hippocampal CA1 of aged Alzheimer’s disease mice. Neurosci. Lett. 2018, 681, 19–25. [Google Scholar] [CrossRef] [PubMed]
- Mosconi, L.; Pupi, A.; De Leon, M.J. Brain glucose hypometabolism and oxidative stress in preclinical Alzheimer’s disease. Ann. N. Y. Acad. Sci. 2008, 1147, 180–195. [Google Scholar] [CrossRef]
- Arendt, T.; Stieler, J.; Holzer, M. Brain hypometabolism triggers PHF-like phosphorylation of tau, a major hallmark of Alzheimer’s disease pathology. J. Neural Transm. 2015, 122, 531–539. [Google Scholar] [CrossRef]
- Nestor, P.J.; Fryer, T.D.; Smielewski, P.; Hodges, J.R. Limbic hypometabolism in Alzheimer’s disease and mild cognitive impairment. Ann. Neurol. 2003, 54, 343–351. [Google Scholar] [CrossRef]
- Bretteville, A.; Marcouiller, F.; Julien, C.; El Khoury, N.B.; Petry, F.R.; Poitras, I.; Mouginot, D.; Lévesque, G.; Hébert, S.S.; Planel, E. Hypothermia-induced hyperphosphorylation: A new model to study tau kinase inhibitors. Sci. Rep. 2012, 2, 1–8. [Google Scholar] [CrossRef]
- Mosconi, L.; Sorbi, S.; de Leon, M.J.; Li, Y.; Nacmias, B.; Myoung, P.S.; Tsui, W.; Ginestroni, A.; Bessi, V.; Fayyazz, M.; et al. Hypometabolism exceeds atrophy in presymptomatic early-onset familial Alzheimer’s disease. J. Nucl. Med. 2006, 47, 1778–1786. [Google Scholar] [PubMed]
- Hirono, N.; Mori, E.; Ishii, K.; Ikejiri, Y.; Imamura, T.; Shimomura, T.; Hashimoto, M.; Yamashita, H.; Sasaki, M. Frontal lobe hypometabolism and depression in Alzheimer’s disease. Neurology 1998, 50, 380–383. [Google Scholar] [CrossRef] [PubMed]
- Tascedda, F.; Malagoli, D.; Accorsi, A.; Rigillo, G.; Blom, J.M.C.; Ottaviani, E. Molluscs as models for translational medicine. Med. Sci. Monit. Basic Res. 2015, 21, 96–99. [Google Scholar] [PubMed]
- Pan, M. Hibernation induction in non-hibernating species. Biosci. Horizons 2018, 11, 1–10. [Google Scholar] [CrossRef]
- Frerichs, K.U.; Hallenbeck, J.M. Hibernation in ground squirrels induces state and species-specific tolerance to hypoxia and aglycemia: An in vitro study in hippocampal slices. J. Cereb. Blood Flow Metab. 1998, 18, 168–175. [Google Scholar] [CrossRef] [PubMed]
| Species | Calcium-Binding Proteins | Cytoskeletal Proteins | Area | Ref. | |||||
|---|---|---|---|---|---|---|---|---|---|
| CB | PV | CR | CaM | MAP-2 | P-TAU | NF-H | |||
| Ictidomys tridecemlineatus | No change | No change | Decrease No change | Decrease Decrease | No change | Increase No change | Cortex Hypothalamus | [128] | |
| Increase (P-NP) | Forebrain | [122] | |||||||
| Spermophilus citellus | Decrease | Increase | Brain | [134] | |||||
| Increase | Forebrain | [135] | |||||||
| Spermophilus lateralis | Decrease | Brain (synapses) | [136] | ||||||
| Spermophilus parryii | Increase | Brain | [137] | ||||||
| Mesocricetus auratus | Increase | Forebrain | [138] | ||||||
| Increase | Brain | [137] | |||||||
| Increase | Brain | [71] | |||||||
| Increase | Hippocampus | [139] | |||||||
| Increase | Brain | [121] | |||||||
| Erinaceus europaeus | Decrease | Decrease | Decrease | Decrease | Decrease (P) | Cerebellum | [127] | ||
| Ursus americanus | Increase | Brain | [137] | ||||||
| Rana esculenta | Decrease No change | Decrease (f) Decrease | Decrease (P) Decrease (P) | Cerebellum Optic tectum | [126] | ||||
| Cornu aspersum | Increase | Decrease (f) | Increase (P) | Cerebral ganglia | [95] | ||||
| No change | Increase | Increase | Increase | Increase (P-NP) | Cerebral ganglia | [133] | |||
| Increase | Increase (P-NP) | Suboesophageal Ganglion | This work | ||||||
| Calcium-Binding Proteins | pNF-H | Compound | Area | Reference | |
|---|---|---|---|---|---|
| CB | PV | ||||
| Decrease | Decrease | Increase | CisPt | Cerebellum | [18] |
| Decrease | Decrease | Increase | CisPt | Cerebellum | [188] |
| Decrease Decrease + no change | CisPt PtAcacDMS | Hippocampus | [186] | ||
| Decrease No change | CisPt PtAcacDMS | Cerebellum | [29] | ||
| Decrease | CisPt | Cerebellum | [39] | ||
| Age | ||||||
| Species | CB | PV | CaM | Method | Area | Reference |
| Homo sapiens | Decrease | No effect | CC | Cortex, Hippocampus | [200] | |
| Decrease | No effect | Decrease | tot mRNA | Frontal cortex | [197] | |
| Mus musculus | Decrease | No effect | CC, WB | Striatum | [31] | |
| Decrease | No effect | CC, WB | Somatosensory cortex | [30] | ||
| Decrease No effect | SI | Hippocampus (CA1) Hippocampus (CA3, DG) | [38] | |||
| Decrease | CC, SI | Brain | [201] | |||
| No effect Increase Decrease | RIA | Striatum Cortex, Cerebellum Diencephalon, Medulla | [196] | |||
| Rattus norvegicus | Decrease No effect | WB, ID | Hippocampus Cerebellum | [202] | ||
| Decrease | WB | Striatum | [28] | |||
| Decrease | CC, WB | Auditory system | [203] | |||
| Decrease | No effect | CC, WB | Striatum | [31] | ||
| Decrease | Increase | CC | Somatosensory cortex | [30] | ||
| Meriones unguiculatus | Decrease | SI, WB | Hippocampus | [204] | ||
| Decrease | No effect | CC, WB | Striatum | [31] | ||
| Decrease | Increase | CC, WB | Somatosensory cortex | [30] | ||
| Mesocricetus auratus | Decrease | No effect | ISH, ARG | Hippocampus, Striatum, Cerebellum | [205] | |
| Alzheimer’s Disease | ||||||
| Species | CB | PV | CaM | Method | Area | Reference |
| Homo sapiens | Decrease | Decrease | RIA | Frontal, Temporal, Parietal Cortex | [206] | |
| Decrease | CC, SI | Cortex | [207,208] | |||
| Decrease | CC | Prefrontal cortex | [209] | |||
| Decrease | CC | Temporal cortex | [210] | |||
| Decrease | CC | Hippocampus | [195] | |||
| Decrease | CC | Temporal, Parahippocampal, Parietal cortex, Cerebellum | [211] | |||
| Decrease No effect | CC | Hippocampus | [212] | |||
| Decrease | SI | Cortex | [213] | |||
| Mus musculus | Decrease | SI, WB, RT-PCR | Dentate gyrus | [214] | ||
| Decrease | CC, SI | Hippocampus | [215] | |||
| Decrease | SI, WB | Hippocampus | [216] | |||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gattoni, G.; Bernocchi, G. Calcium-Binding Proteins in the Nervous System during Hibernation: Neuroprotective Strategies in Hypometabolic Conditions? Int. J. Mol. Sci. 2019, 20, 2364. https://doi.org/10.3390/ijms20092364
Gattoni G, Bernocchi G. Calcium-Binding Proteins in the Nervous System during Hibernation: Neuroprotective Strategies in Hypometabolic Conditions? International Journal of Molecular Sciences. 2019; 20(9):2364. https://doi.org/10.3390/ijms20092364
Chicago/Turabian StyleGattoni, Giacomo, and Graziella Bernocchi. 2019. "Calcium-Binding Proteins in the Nervous System during Hibernation: Neuroprotective Strategies in Hypometabolic Conditions?" International Journal of Molecular Sciences 20, no. 9: 2364. https://doi.org/10.3390/ijms20092364
APA StyleGattoni, G., & Bernocchi, G. (2019). Calcium-Binding Proteins in the Nervous System during Hibernation: Neuroprotective Strategies in Hypometabolic Conditions? International Journal of Molecular Sciences, 20(9), 2364. https://doi.org/10.3390/ijms20092364





