Antioxidant Activity and Toxicity of Fullerenols via Bioluminescence Signaling: Role of Oxygen Substituents
Abstract
1. Introduction
2. Results and Discussion
2.1. Toxicity and Antioxidant Activity of Fullerenols via Bioluminescent Assays
2.1.1. Fullerenol Toxicity
2.1.2. Fullerenol Antioxidant Activity
Change in General Toxicity (GT) of Oxidizer Solutions Under Exposure to Fullerenols
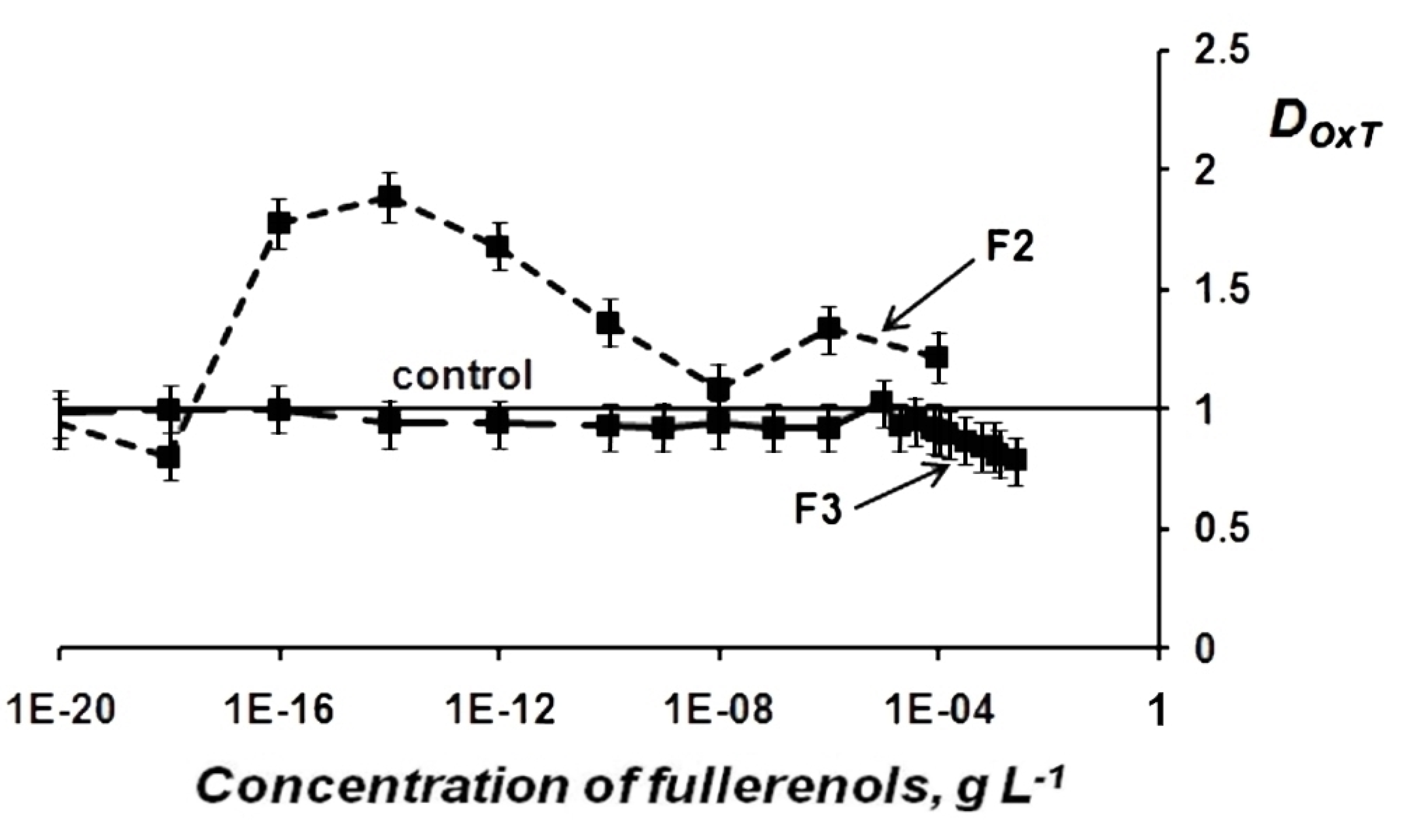
Change in Oxidative Toxicity (OxT) of Oxidizer Solutions Under Exposure to Fullerenols
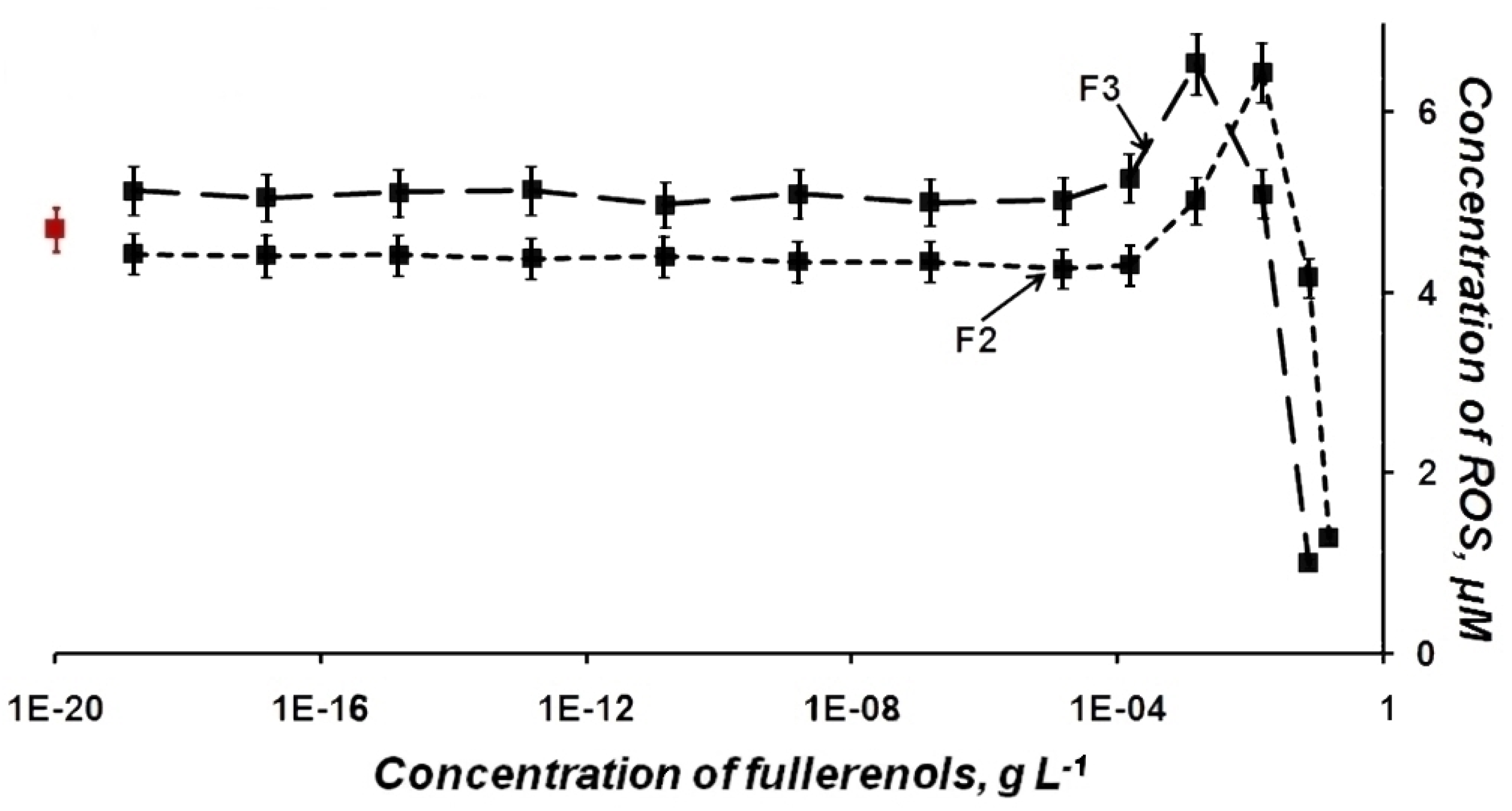
2.2. ROS Content in Fullerenol Solutions—Luminol Chemiluminescence Assay
3. Materials and Methods
3.1. Preparations of Fullerenols
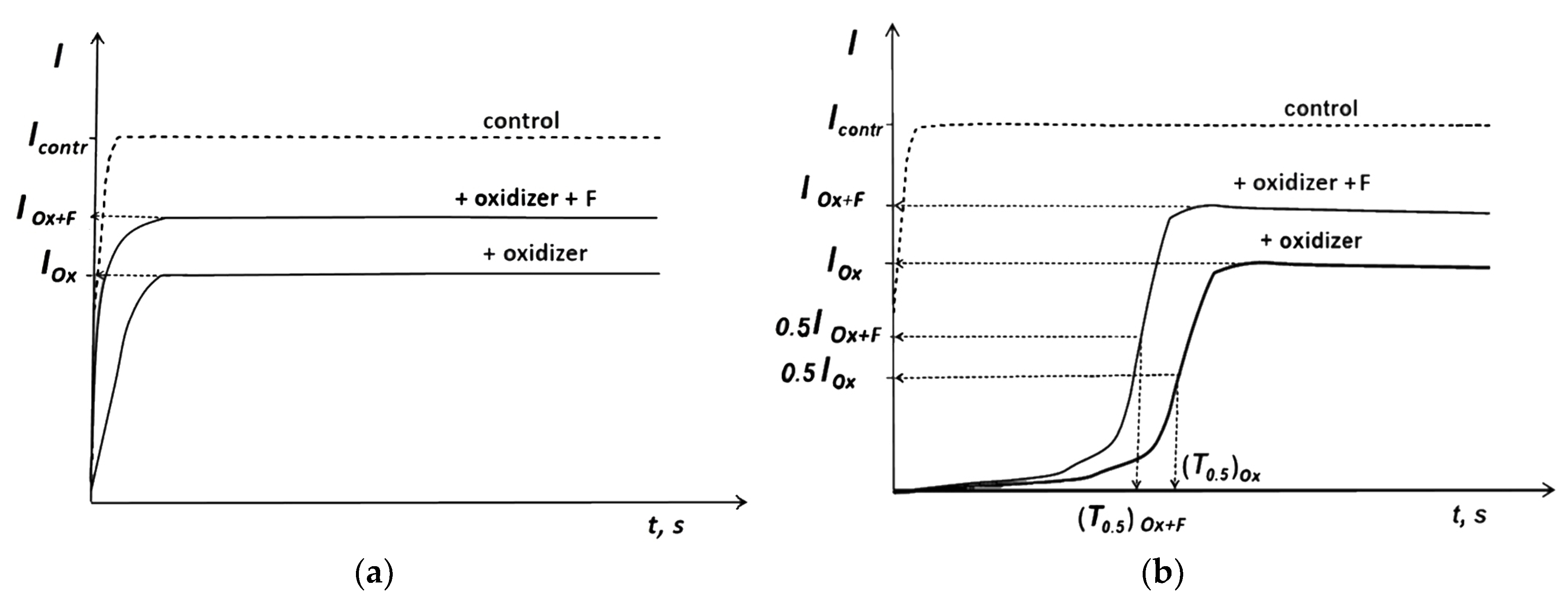
3.2. Bioluminescence Assay Systems and Experimental Data Processing
3.3. Luminol Chemiluminescence Assay
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| CR | concentration range |
| F | fullerenol |
| F1 | fullerenol C60Oy(OH)x, where x+y = 24–28 |
| F2 | fullerenol C60,70Oy(OH)x, where x+y = 24–28 |
| F3 | fullerenol C60,70Oy(OH)x, where x+y = 40–42 |
| F4 | fullerenol Fe0,5C60Oy(OH)x, where x+y = 40–42 |
| F5 | fullerenol Gd@C82Oy(OH)x, where x+y = 40–42 |
| FMN | flavin mononucleotide |
| GT | general toxicity |
| NADH | nicotinamide adenine dinucleotide disodium salt-reduced |
| OxT | oxidative toxicity |
| ROS | reactive oxygen species |
References
- Foley, S.; Crowley, C.; Smaihi, M.; Bonfils, C.; Erlanger, B.F.; Seta, P.; Larroque, C. Cellular localization of a water-soluble fullerene derivative. Biochem. Biophys. Res. Commun. 2002, 294, 116–119. [Google Scholar] [CrossRef]
- Grebowski, J.; Krokosz, A.; Puchala, M. Fullerenol C60(OH)36 could associate to band 3 protein of human erythrocyte membranes. Biochim. Biophys. Acta (BBA) Biomembr. 2013, 1828, 2007–2014. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Y.; Hou, L.; Liu, M.; Newell, S.E.; Yin, G.; Yu, C.; Zhang, H.; Li, X.; Gao, D.; Gao, J.; et al. Effects of silver nanoparticles on nitrification and associated nitrous oxide production in aquatic environments. Sci. Adv. 2017, 3, e1603229. [Google Scholar] [CrossRef] [PubMed]
- Iavicoli, I.; Leso, V.; Fontana, L.; Calabrese, E.J. Nanoparticle exposure and hormetic dose–responses: An update. Int. J. Mol. Sci. 2018, 19, 805. [Google Scholar] [CrossRef] [PubMed]
- Grebowski, J.; Kazmierska, P.; Krokosz, A. Fullerenols as a new therapeutic approach in nanomedicine. Biomed. Res. Int. 2013, 2013, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Cai, X.; Hao, J.; Zhang, X.; Yu, B.; Ren, J.; Luo, C.; Li, Q.; Huang, Q.; Shi, X.; Li, W.; et al. The polyhydroxylated fullerene derivative C60(OH)24 protects mice from ionizing-radiation-induced immune and mitochondrial dysfunction. Toxicol. Appl. Pharmacol. 2010, 243, 27–34. [Google Scholar] [CrossRef] [PubMed]
- Slavic, M.; Djordjevic, A.; Radojicic, R.; Milovanovic, S.; Orescanin-Dusic, Z.; Rakocevic, Z.; Spasic, M.B.; Blagojevic, D. FullerenolC60(OH)24 nanoparticles decrease relaxing effects of dimethyl sulfoxide on rat uterus spontaneous contraction. J. Nanopart. Res. 2013, 15, 1–10. [Google Scholar] [CrossRef]
- Mirkov, S.M.; Djordjevic, A.N.; Andric, N.L.; Andric, S.A.; Kostic, T.S.; Bogdanovic, G.M.; Vojinovic-Miloradov, M.B.; Kovacevic, R.Z. Nitric oxide-scavenging activity of polyhydroxylated fullerenol, C60(OH)24. Nitric Oxide 2004, 11, 201–207. [Google Scholar] [CrossRef] [PubMed]
- Injac, R.; Prijatelj, M.; Strukelj, B. Fullerenol nanoparticles: Toxicity and antioxidant activity. Methods Mol. Biol. 2013, 1028, 75–100. [Google Scholar] [CrossRef] [PubMed]
- Djordjevic, A.; Srdjenovic, B.; Seke, M.; Petrovic, D.; Injac, R.; Mrdjanovic, J. Review of synthesis and antioxidant potential of fullerenol nanoparticles. J. Nanomater. 2015, 2015, 1–15. [Google Scholar] [CrossRef]
- Wang, Z.; Wang, S.; Lu, Z.; Gao, X. Syntheses, structures and antioxidant activities of fullerenols: Knowledge learned at the atomistic level. J. Clust. Sci. 2015, 26, 375–388. [Google Scholar] [CrossRef]
- Djordjevic, A.; Canadanovic-Brunet, J.M.; Vojinovic-Miloradov, M.; Bogdanovic, G. Antioxidant properties and hypothetic radical mechanism of fullerenol C60(OH)24. Oxid. Commun. 2005, 27, 806–812. [Google Scholar]
- Jiao, F.; Liu, Y.; Qu, Y.; Li, W.; Zhou, G.; Ge, C.; Li, Y.; Sun, B.; Chen, C. Studies on anti-tumor and antimetastatic activities of fullerenol in a mouse breast cancer model. Carbon 2010, 48, 2231–2243. [Google Scholar] [CrossRef]
- Eropkin, M.Y.; Melenevskaya, E.Y.; Nasonova, K.V.; Bryazzhikova, T.S.; Eropkina, E.M.; Danilenko, D.M.; Kiselev, O.I. Synthesis and biological activity of fullerenols with various contents of hydroxyl groups. Pharm. Chem. J. 2013, 47, 87–91. [Google Scholar] [CrossRef]
- Chaban, V.V.; Fileti, E.E. Which fullerenols are water soluble? Systematic atomistic investigation. New J. Chem. 2017, 41, 184–189. [Google Scholar] [CrossRef]
- Hayyan, M.; Hashim, M.A.; AlNashef, I.M. Superoxide ion: Generation and chemical implications. Chem. Rev. 2016, 116, 3029–3085. [Google Scholar] [CrossRef]
- Hancock, J.T.; Desikan, R.; Neill, S.J. Role of reactive oxygen species in cell signalling pathways. Biochem. Soc. Trans. 2001, 29 Pt 2, 345–350. [Google Scholar] [CrossRef]
- Devasagayam, T.P.; Tilak, J.C.; Boloor, K.K.; Sane, K.S.; Ghaskadbi, S.S.; Lele, R.D. Free radicals and antioxidants in human health: Current status and future prospects. J. Assoc. Phys. India 2004, 52, 794–804. [Google Scholar]
- Klaunig, J.E.; Kamendulis, L.M. The role of oxidative stress in carcinogenesis. Annu. Rev. Pharmacol. Toxicol. 2004, 44, 239–267. [Google Scholar] [CrossRef]
- Sinha, K.; Das, J.; Pal, P.B.; Sil, P.C. Oxidative stress: The mitochondria-dependent and mitochondria-independent pathways of apoptosis. Arch. Toxicol. 2013, 87, 1157–1180. [Google Scholar] [CrossRef]
- Anderson, G.; Maes, M. Neurodegeneration in Parkinson’s disease: Interactions of oxidative stress, tryptophan catabolites and depression with mitochondria and sirtuins. Mol. Neurobiol. 2014, 49, 771–783. [Google Scholar] [CrossRef] [PubMed]
- Giacco, F.; Brownlee, M. Oxidative stress and diabetic complications. Circ. Res. 2010, 107, 1058–1070. [Google Scholar] [CrossRef] [PubMed]
- Zenker, M.; Aigner, T.; Wendler, O.; Tralau, T.; Müntefering, H.; Fenski, R.; Pitz, S.; Schumacher, V.; Royer-Pokora, B.; Wühl, E.; et al. Human laminin β2 deficiency causes congenital nephrosis with mesangial sclerosis and distinct eye abnormalities. Hum. Mol. Genet. 2004, 13, 2625–2632. [Google Scholar] [CrossRef] [PubMed]
- Caldeira, G.L.; Ferreira, I.L.; Rego, A.C. Impaired transcription in Alzheimer’s disease: Key role in mitochondrial dysfunction and oxidative stress. J. Alzheimer’s Dis. 2013, 34, 115–131. [Google Scholar] [CrossRef]
- Dharmaraja, A.T. Role of reactive oxygen species (ROS) in therapeutics and drug resistance in cancer and bacteria. J. Med. Chem. 2017, 60, 3221–3240. [Google Scholar] [CrossRef] [PubMed]
- Forrester, S.J.; Kikuchi, D.S.; Hernandes, M.S.; Xu, Q.; Griendling, K.K. Reactive oxygen species in metabolic and inflammatory signaling. Circ. Res. 2018, 122, 877–902. [Google Scholar] [CrossRef] [PubMed]
- Nugud, A.; Sandeep, D.; El-Serafi, A.T. Two faces of the coin: Minireview for dissecting the role of reactive oxygen species in stem cell potency and lineage commitment. J. Adv. Res. 2018, 14, 73–79. [Google Scholar] [CrossRef]
- Bulich, A.A.; Isenberg, D.L. Use of the luminescent bacterial system for rapid assessment of aquatic toxicity. ISA Trans. 1981, 20, 29–33. [Google Scholar] [PubMed]
- Fedorova, E.; Kudryasheva, N.; Kuznetsov, A.; Mogil’naya, O.; Stom, D. Bioluminescent monitoring of detoxification processes: Activity of humic substances in quinone solutions. J. Photochem. Photobiol. B 2007, 88, 131–136. [Google Scholar] [CrossRef]
- Girotti, S.; Ferri, E.N.; Fumo, M.G.; Maiolini, E. Monitoring of environmental pollutants by bioluminescent bacteria. Anal. Chim. Acta 2008, 608, 2–29. [Google Scholar] [CrossRef]
- Kudryasheva, N.; Kratasyuk, V.; Esimbekova, E.; Vetrova, E.; Nemtseva, E.; Kudinova, I. Development of bioluminescent bioindicators for analyses of environmental pollution. Field Anal. Chem. Technol. 1998, 2, 277–280. [Google Scholar] [CrossRef]
- Roda, A.; Pasini, P.; Mirasoni, M.; Michchelini, E.; Guardigli, M. Biotechnological application of bioluminescence and chemiluminescence. Trends Biotechnol. 2004, 22, 295–303. [Google Scholar] [CrossRef] [PubMed]
- Abbas, M.; Adil, M.; Ehtisham-Ul-Haque, S.; Munir, B.; Yameen, M.; Ghaffar, A.; Shar, G.A.; Asif Tahir, M.; Iqbal, M. Vibrio fischeri bioluminescence inhibition assay for ecotoxicity assessment: A review. Sci. Total Environ. 2018, 626, 1295–1309. [Google Scholar] [CrossRef] [PubMed]
- Kratasyuk, V.A.; Esimbekova, E.N. Applications of luminous bacteria enzymes in toxicology. Comb. Chem. High Throughput Screen. 2015, 18, 952–959. [Google Scholar] [CrossRef] [PubMed]
- Rozhko, T.V.; Kudryasheva, N.S.; Kuznetsov, A.M.; Vydryakova, G.A.; Bondareva, L.G.; Bolsunovsky, A.Y. Effect of low-level α-radiation on bioluminescent assay systems of various complexity. Photochem. Photobiol. Sci. 2007, 6, 67–70. [Google Scholar] [CrossRef]
- Selivanova, M.A.; Mogilnaya, O.A.; Badun, G.A.; Vydryakova, G.A.; Kuznetsov, A.M.; Kudryasheva, N.S. Effect of tritium on luminous marine bacteria and enzyme reactions. J. Environ. Radioact. 2013, 120, 19–25. [Google Scholar] [CrossRef]
- Esimbekova, E.N.; Kondik, A.M.; Kratasyuk, V.A. Bioluminescent enzymatic rapid assay of water integral toxicity. Environ. Monit. Assess. 2013, 185, 5909–5916. [Google Scholar] [CrossRef]
- Efremenko, E.N.; Maslova, O.V.; Kholstov, A.V.; Senko, O.V.; Ismailov, A.D. Biosensitive element in the form of immobilized luminescent photobacteria for detecting ecotoxicants in aqueous flow-through systems. Luminescence 2016, 31, 1283–1289. [Google Scholar] [CrossRef]
- Ismailov, A.D.; Aleskerova, L.E. Photobiosensors containing luminescent bacteria. Biochemistry 2015, 80, 733–744. [Google Scholar] [CrossRef]
- Ranjan, R.; Rastogi, N.K.; Thakur, M.S. Development of immobilized biophotonic beads consisting of Photobacterium leiognathi for the detection of heavy metals and pesticide. J. Hazard. Mater. 2012, 225–226, 114–123. [Google Scholar] [CrossRef]
- Kudryasheva, N.S. Bioluminescence and exogenous compounds: Physicochemical basis for bioluminescence assay. J. Photochem. Photobiol. B 2006, 83, 77–86. [Google Scholar] [CrossRef]
- Kirillova, T.N.; Kudryasheva, N.S. Effect of heavy atom in bioluminescent reactions. Anal. Bioanal. Chem. 2007, 387, 2009–2016. [Google Scholar] [CrossRef]
- Kirillova, T.N.; Gerasimova, M.A.; Nemtseva, E.V.; Kudryasheva, N.S. Effect of halogenated fluorescent compounds on bioluminescent reactions. Anal. Bioanal. Chem. 2011, 400, 343–351. [Google Scholar] [CrossRef]
- Nemtseva, E.V.; Kudryasheva, N.S. The mechanism of electronic excitation in bacterial bioluminescent reaction. Russ. Chem. Rev. 2007, 76, 91–100. [Google Scholar] [CrossRef]
- Vetrova, E.V.; Kudryasheva, N.S.; Kratasyuk, V.A. Redox compounds influence on the NAD(P)H:FMN-oxidoreductase-luciferase bioluminescent system. Photochem. Photobiol. Sci. 2007, 6, 35–40. [Google Scholar] [CrossRef]
- Fedorova, G.F.; Kancheva, V.D.; Menshov, V.A.; Naumov, V.V.; Vasil’ev, R.F.; Veprintsev, T.L.; Trofimov, A.V.; Tsaplev, Y.B.; Yablonskaya, O.I. Exogenous and endogenous mediators of oxygen metabolism: Alternatives for chemical and biological activity. Stud. Nat. Prod. Chem. 2016, 47, 357–385. [Google Scholar] [CrossRef]
- Slavova-Kazakova, A.K.; Angelova, S.E.; Veprintsev, T.L.; Denev, P.; Fabbri, D.; Dettori, M.A.; Kratchanova, M.; Naumov, V.V.; Trofimov, A.V.; Vasil’ev, R.F.; et al. Antioxidant potential of curcumin-related compounds studied by chemiluminescence kinetics, chain-breaking efficiencies, scavenging activity (ORAC) and DFT calculations. Beilstein J. Org. Chem. 2015, 11, 1398–1411. [Google Scholar] [CrossRef]
- Kudryasheva, N.; Vetrova, E.; Kuznetsov, A.; Kratasyuk, V.; Stom, D. Bioluminescent assays: Effects of quinones and phenols. Ecotoxicol. Environ. Saf. 2002, 53, 221–225. [Google Scholar] [CrossRef]
- Tarasova, A.S.; Stom, D.I.; Kudryasheva, N.S. Effect of humic substances on toxicity of inorganic oxidizer bioluminescent monitoring. Environ. Toxicol. Chem. 2011, 30, 1013–1017. [Google Scholar] [CrossRef]
- Kudryasheva, N.S.; Tarasova, A.S. Pollutant toxicity and detoxification by humic substances: Mechanisms and quantitative assessment via luminescent Biomonitoring. Environ. Sci. Pollut. Res. Int. 2015, 22, 155–167. [Google Scholar] [CrossRef]
- Tarasova, A.S.; Stom, D.I.; Kudryasheva, N.S. Antioxidant activity of humic substances via bioluminescent monitoring in vitro. Environ. Monit. Assess. 2015, 187, 89. [Google Scholar] [CrossRef]
- Tarasova, A.S.; Kislan, S.L.; Fedorova, E.S.; Kuznetsov, A.M.; Mogilnaya, O.A.; Stom, D.I.; Kudryasheva, N.S. Bioluminescence as a tool for studying detoxification processes in metal salt solutions involving humic substances. J. Photochem. Photobiol. B 2012, 117, 164–170. [Google Scholar] [CrossRef]
- Kudryasheva, N.S.; Kovel, E.S.; Sachkova, A.S.; Vorobeva, A.A.; Isakova, V.G.; Churilov, G.N. Bioluminescent enzymatic assay as a tool for studying antioxidant activity and toxicity of bioactive compounds. J. Photochem. Photobiol. 2017, 93, 536–540. [Google Scholar] [CrossRef]
- Sachkova, A.S.; Kovel, E.S.; Churilov, G.N.; Guseynov, O.A.; Bondar, A.A.; Dubinina, I.A.; Kudryasheva, N.S. On mechanism of antioxidant effect of fullerenols. Biochem. Biophys. Rep. 2017, 9, 1–8. [Google Scholar] [CrossRef]
- Sachkova, A.S.; Kovel, E.S.; Churilov, G.N.; Stom, D.I.; Kudryasheva, N.S. Biological activity of carbonic nano-structures—Comparison via enzymatic bioassay. J. Soils Sediments 2018. [Google Scholar] [CrossRef]
- Meng, J.; Liang, X.; Chen, X.; Zhao, Y. Biological characterizations of [Gd@C82(OH)22]n nanoparticles as fullerene derivatives for cancer therapy. Integr. Biol. 2013, 5, 43–47. [Google Scholar] [CrossRef]
- Sosnovik, D.E.; Caravan, P. Molecular MRI of the Cardiovascular System in the Post-NSF Era. Curr. Cardiovasc. Imaging Rep. 2013, 6, 61–68. [Google Scholar] [CrossRef]
- Kanda, T.; Osawa, M.; Oba, H.; Toyoda, K.; Kotoku, J.; Haruyama, T.; Takeshita, K.; Furui, S. High signal intensity in dentate nucleus on unenhanced T1-weighted MR Images: Association with linear versus macrocyclic gadolinium chelate administration. Radiology 2015, 275, 803–809. [Google Scholar] [CrossRef]
- Remmel’, N.N.; Titova, N.M.; Kratasyuk, V.A. Oxidative stress monitoring in biological samples by bioluminescent method. Bull. Exp. Biol. Med. 2003, 136, 209–211. [Google Scholar] [CrossRef]
- Alexandrova, M.; Rozhko, T.; Vydryakova, G.; Kudryasheva, N. Effect of americium-241 on luminous bacteria. Role of peroxides. J. Environ. Radioact. 2011, 102, 407–411. [Google Scholar] [CrossRef]
- Andrievsky, G.V.; Bruskov, V.I.; Tykhomyrov, A.A.; Gudkov, S.V. Peculiarities of the antioxidant and radioprotective effects of hydrated C60 fullerene nanostuctures in vitro and in vivo. Free Radic. Biol. Med. 2009, 47, 786–793. [Google Scholar] [CrossRef]
- Andrievsky, G.; Klochkov, V.; Derevyanchenko, L. Is the C60 fullerene molecule toxic? Fuller. Nanotub. Carbon Nanostruct. 2005, 13, 363–376. [Google Scholar] [CrossRef]
- Voeikov, V.L.; Yablonskaya, O.I. Stabilizing effects of hydrated fullerenes C60 in a wide range of concentrations on luciferase, alkaline phosphatase, and peroxidase in vitro. Electromagn. Biol. Med. 2015, 34, 160–166. [Google Scholar] [CrossRef]
- Novikov, K.N.; Berdnikova, N.G.; Novikov, A.K.; Lyusina, O.Y.; Muhitova, O.G.; Yablonskaya, O.I.; Minh, H.D.; Voeikov, V.L. Changes in chemiluminescence of whole blood of COPD patients treated with Hypoxen® and effects of C60 fullerenes on blood chemiluminescence. Med. Sci. Monit. 2012, 18, BR76–BR83. [Google Scholar] [CrossRef]
- Goncharova, E.A.; Isakova, V.G.; Tomashevich, E.V.; Churilov, G.N. Obtaining of water-soluble polyhydroxylated fullerenols with iron nanoparticles as catalyzers. Vestn. SibGAU 2009, 22, 90–93. [Google Scholar]
- Sun, D.; Huang, H.; Yang, S. Synthesis and characterization of a water-soluble endohedral metallofullerol. Chem. Mater. 1999, 11, 1003–1006. [Google Scholar] [CrossRef]
- Churilov, G.N.; Kratschmer, W.; Osipova, I.V.; Glushenko, G.A.; Vnukova, N.G.; Kolonenko, A.L.; Dudnik, A.I. Synthesis of fullerenes in a high-frequency arc plasma under elevated helium pressure. Carbon 2013, 62, 389–392. [Google Scholar] [CrossRef]
- Isakova, V.G.; Goncharova, E.A.; Bayukov, O.A.; Churilov, G.N. Hydroxylation of fullerenes modified with iron nanoparticles. Russ. J. Appl. Chem. 2011, 84, 1165–1169. [Google Scholar] [CrossRef]
- Churilov, G.N.; Popov, A.A.; Vnukova, N.G.; Dudnik, A.I.; Glushchenko, G.A.; Samoylova, N.A.; Dubinina, I.A.; Gulyaeva, U.E. A method and apparatus for high-throughput controlled synthesis of fullerenes and endohedral metal fullerenes. Tech. Phys. Lett. 2016, 42, 475–477. [Google Scholar] [CrossRef]
- Churilov, G.; Popov, A.; Vnukova, N.; Dudnik, A.; Samoylova, N.; Glushenko, G. Controlled synthesis of fullerenes and endohedral metallofullerenes in high frequency arc discharge. Fuller. Nanotub. Carbon Nanostruct. 2016, 24, 675–678. [Google Scholar] [CrossRef]
- Akiyama, K.; Hamano, T.; Nakanishi, Y.; Takeuchi, E.; Noda, S.; Wang, Z.; Kubuki, S.; Shinohara, H. Non-HPLC rapid separation of metallofullerenes and empty cages with TiCl4 Lewis acid. J. Am. Chem. Soc. 2012, 134, 9762–9767. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Zhang, M.; Sun, B.; Xing, G.; Song, Y.; Guo, H.L.; Chang, Y.; Ge, Y.; Zhao, Y. Separation and purification of fullerenols for improved biocompatibility. Carbon 2012, 50, 460–469. [Google Scholar] [CrossRef]
- Li, J.; Wang, T.; Feng, Y.; Zhang, Y.; Zhen, M.; Shu, C.; Jiang, L.; Wang, Y.; Wang, C. A water-soluble gadolinium metallofullerenol: Facile preparation, magnetic properties and magnetic resonance imaging application. Dalton Trans. 2016, 45, 8696–8699. [Google Scholar] [CrossRef] [PubMed]
- Kuznetsov, A.M.; Rodicheva, E.K.; Shilova, E.V. Bioassay based on lyophilized bacteria. Biotekhnologiya 1996, 9, 57–61. [Google Scholar]
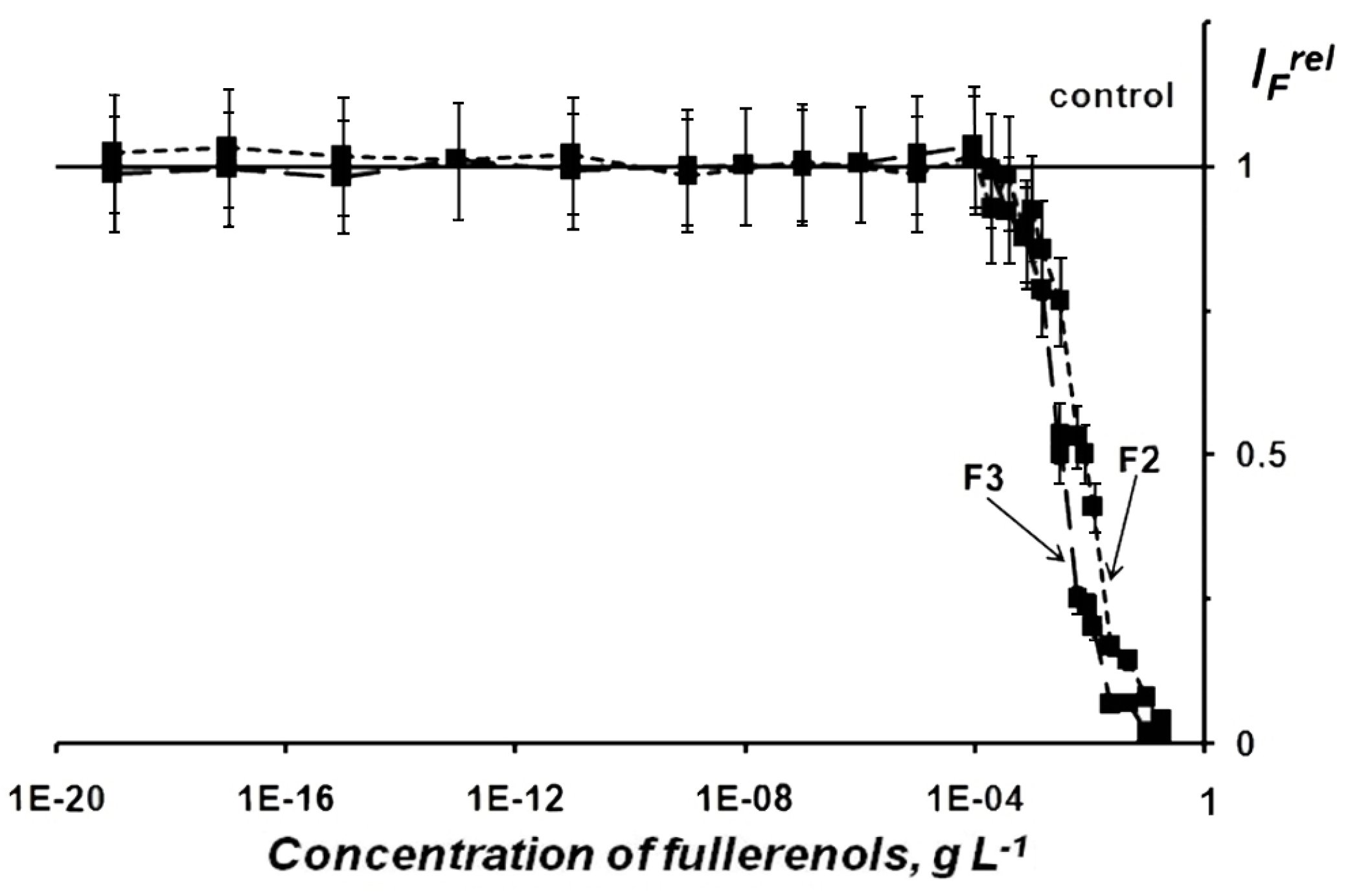
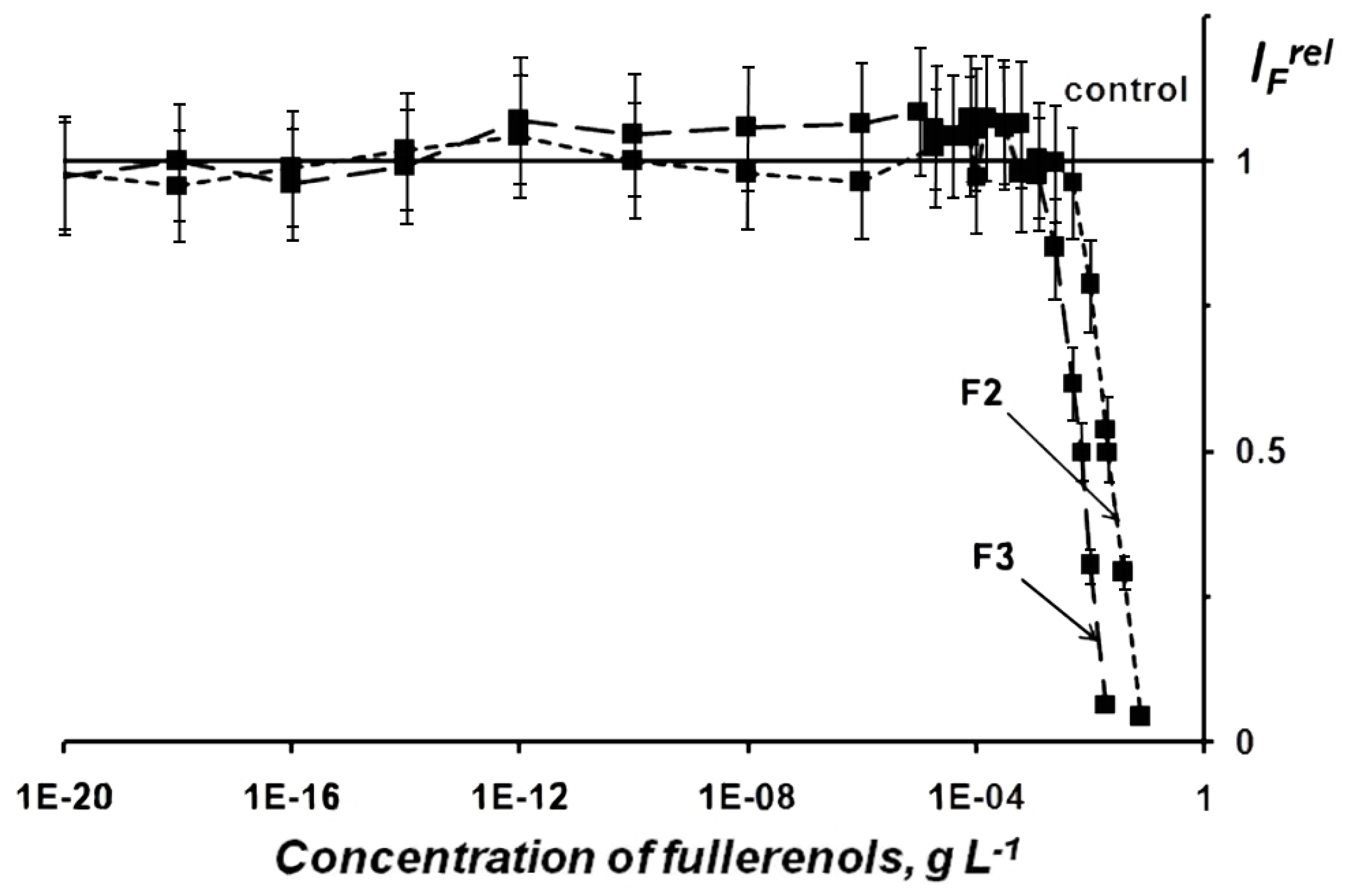
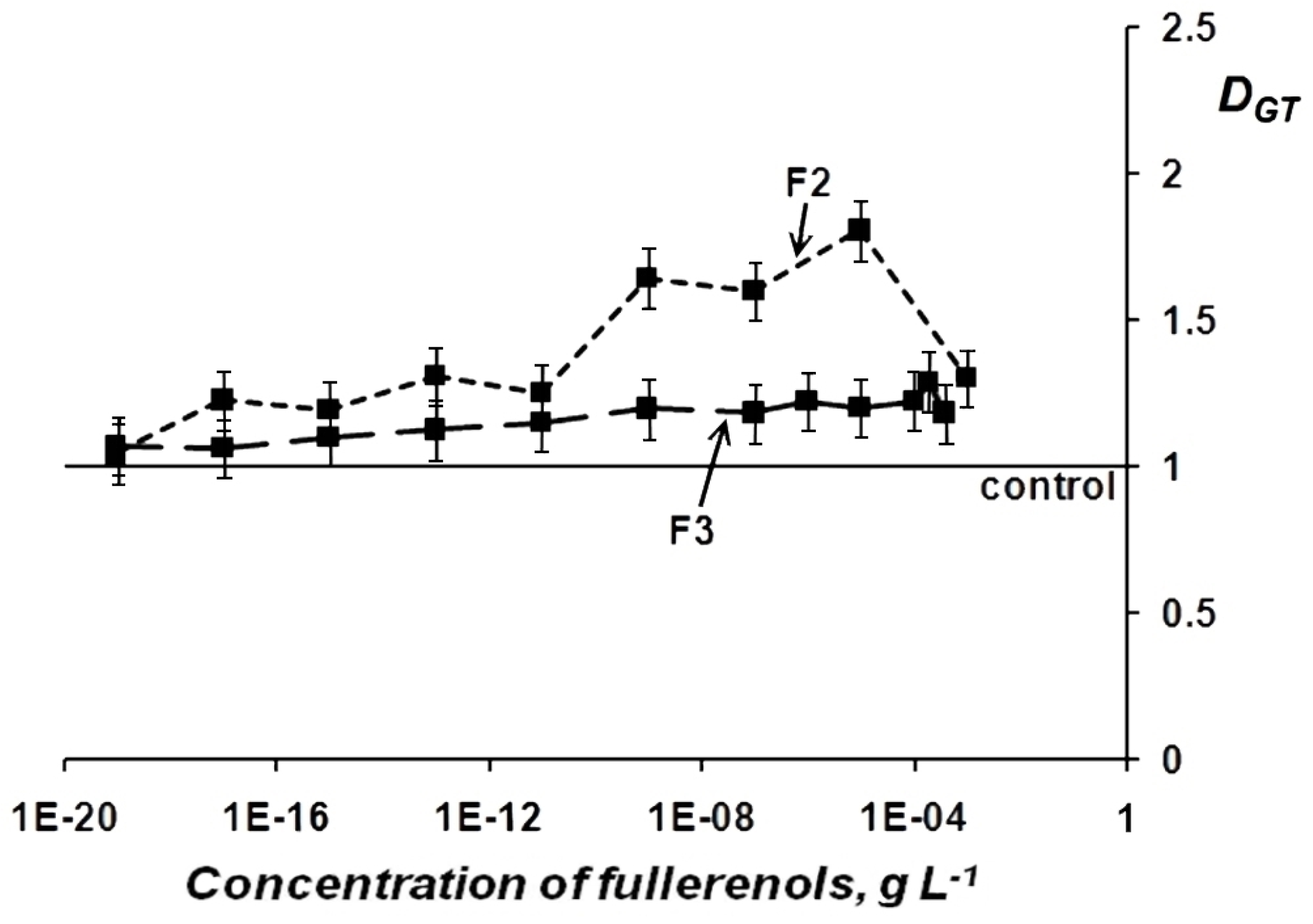

| Chemical Formula | x+y | Abbreviation |
|---|---|---|
| C60Oy(OH)x | 24–28 | F1 |
| C60,70Oy(OH)x | 24–28 | F2 |
| C60,70Oy(OH)x | 40–42 | F3 |
| Fe0,5C60Oy(OH)x | 40–42 | F4 |
| Gd@C82Oy(OH)x | 40–42 | F5 |
| Fullerenols | Cellular Assay | Enzymatic Assay | ||
|---|---|---|---|---|
| Fullerenol Concentration (g L−1) | ||||
| EC50 | CR | EC50 | CR | |
| F1 | 0.031 | <0.010 | 0.092 | <0.010 |
| F2 | 0.008 | <0.002 | 0.021 | <0.010 |
| F3 | 0.003 | <0.001 | 0.007 | <0.003 |
| F4 | 0.021 | <0.012 | 0.007 | <0.001 |
| F5 | - | - | 0.018 | <0.005 |
| Fullerenols | Maximal Value of DGT | |
|---|---|---|
| Enzymatic Assay | Cellular Assay | |
| F1 | 2.2 | 1.4 |
| F2 | 1.5 | 1.8 |
| F3 | 1.3 | 1.3 |
| F4 | 1.1 | 1.3 |
| Fullerenols | Maximal Value of DOxT |
|---|---|
| F1 | 2.0 |
| F2 | 1.9 |
| F3 | 1.0 |
| F4 | 1.3 |
| Fullerenols | EC50, g L−1 |
|---|---|
| F1 | 0.179 |
| F2 | 0.124 |
| F3 | 0.056 |
| F4 | 0.105 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kovel, E.S.; Sachkova, A.S.; Vnukova, N.G.; Churilov, G.N.; Knyazeva, E.M.; Kudryasheva, N.S. Antioxidant Activity and Toxicity of Fullerenols via Bioluminescence Signaling: Role of Oxygen Substituents. Int. J. Mol. Sci. 2019, 20, 2324. https://doi.org/10.3390/ijms20092324
Kovel ES, Sachkova AS, Vnukova NG, Churilov GN, Knyazeva EM, Kudryasheva NS. Antioxidant Activity and Toxicity of Fullerenols via Bioluminescence Signaling: Role of Oxygen Substituents. International Journal of Molecular Sciences. 2019; 20(9):2324. https://doi.org/10.3390/ijms20092324
Chicago/Turabian StyleKovel, Ekaterina S., Anna S. Sachkova, Natalia G. Vnukova, Grigoriy N. Churilov, Elena M. Knyazeva, and Nadezhda S. Kudryasheva. 2019. "Antioxidant Activity and Toxicity of Fullerenols via Bioluminescence Signaling: Role of Oxygen Substituents" International Journal of Molecular Sciences 20, no. 9: 2324. https://doi.org/10.3390/ijms20092324
APA StyleKovel, E. S., Sachkova, A. S., Vnukova, N. G., Churilov, G. N., Knyazeva, E. M., & Kudryasheva, N. S. (2019). Antioxidant Activity and Toxicity of Fullerenols via Bioluminescence Signaling: Role of Oxygen Substituents. International Journal of Molecular Sciences, 20(9), 2324. https://doi.org/10.3390/ijms20092324







