Oncogenic Effect of the Novel Fusion Gene VAPA-Rab31 in Lung Adenocarcinoma
Abstract
1. Introduction
2. Results
2.1. Identification of the VAPA-Rab31 Fusion Gene in a Patient with Lung Adenocarcinoma
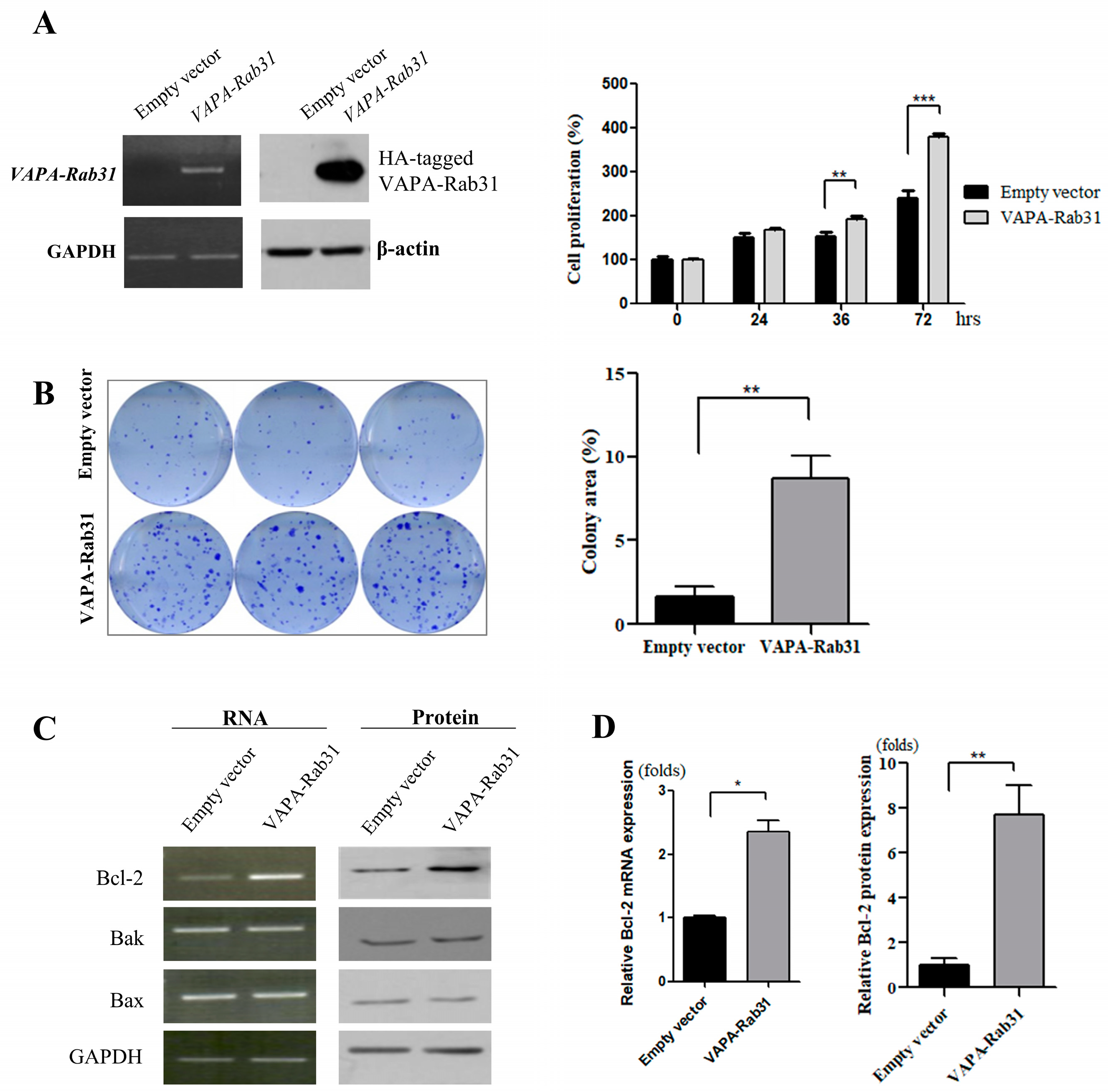
2.2. Overexpression of VAPA-Rab31 Increases Colony Formation and Upregulates Bcl-2 Expression
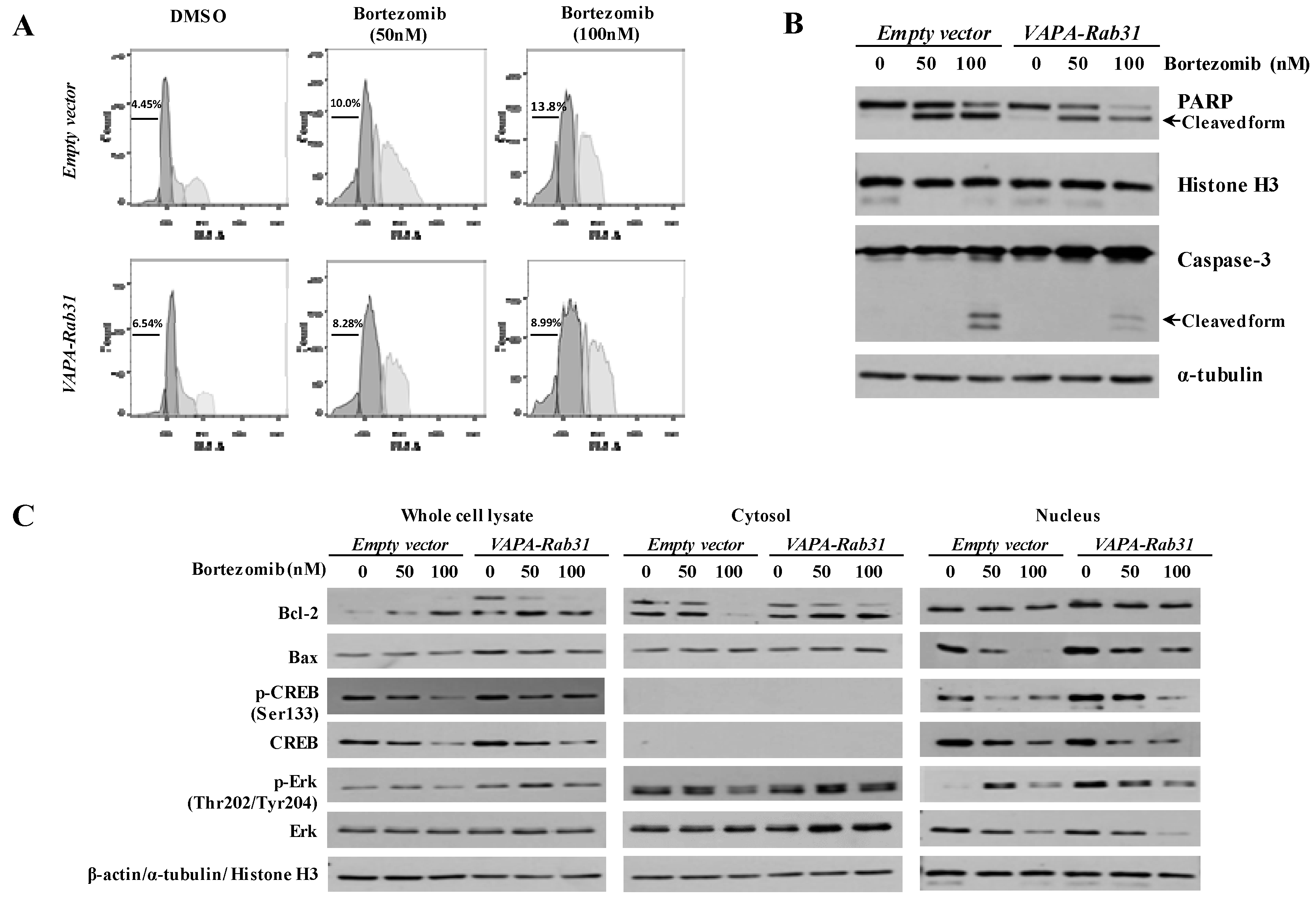
2.3. VAPA-Rab31 Fusion Induces Resistance to Apoptosis
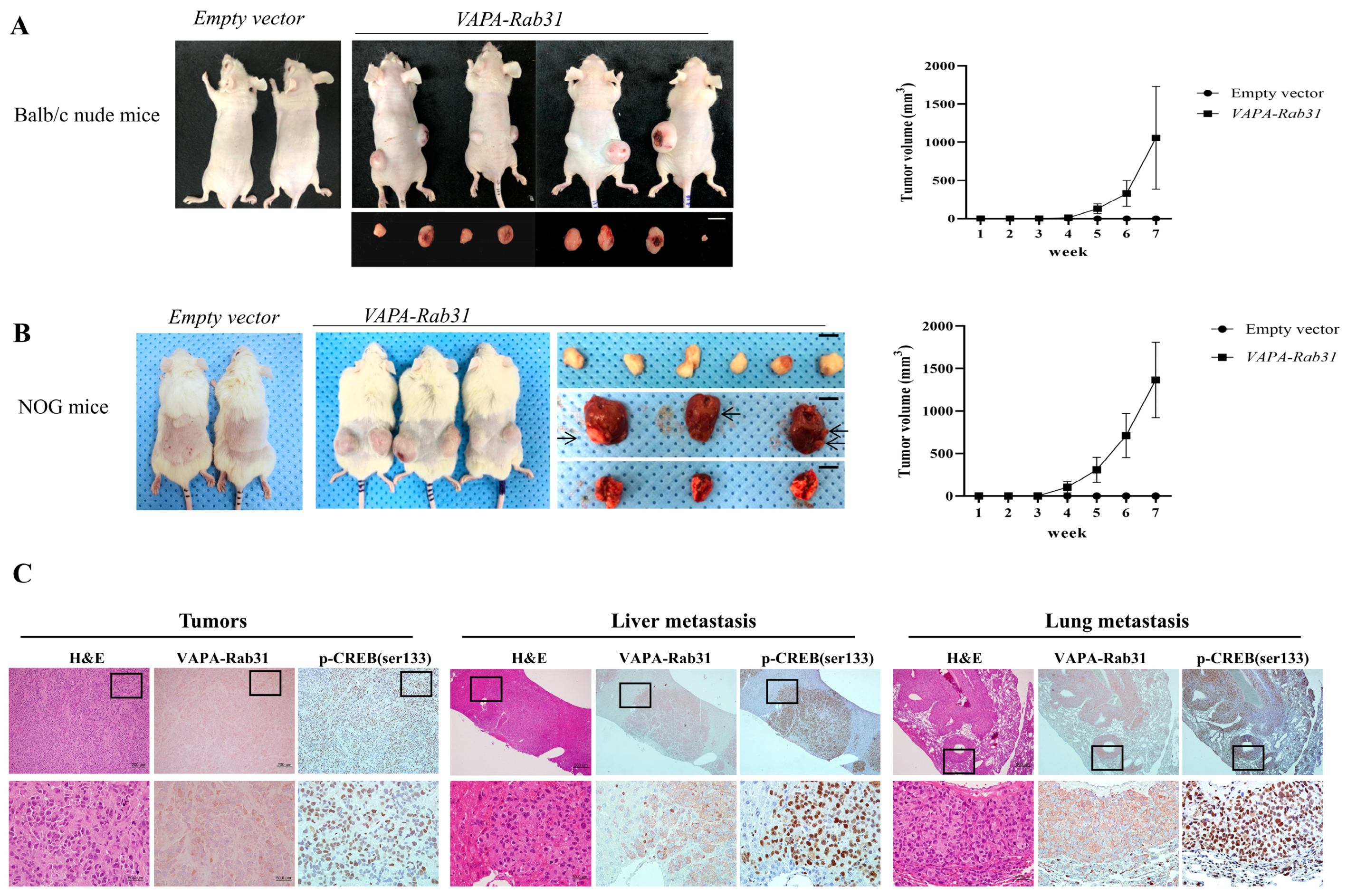
2.4. Tumorigenic Effect of VAPA-Rab31 Fusion is Confirmed Using Xenografts
3. Discussion
4. Materials and Methods
4.1. Patient’s Specimen and Cell Lines
4.2. RNA Extraction and RT-PCR
4.3. Overexpression of VAPA-Rab31
4.4. Proliferation and Colony Formation Assay
4.5. Apoptosis Analysis
4.6. Western Blot Analysis
4.7. Induction of In Vivo Tumorigenicity
4.8. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| VAPA | Vesicle-associated membrane protein (VAMP)-associated protein A |
| Rab31 | Ras-related protein 31 |
| CREB | CAMP responsive element binding protein |
| PARP | Poly (ADP-ribose) polymerase |
| Bax | Bcl-2 associated X |
| Bak | Bcl-2-antagonist/Killer |
| H&E | Hematoxylin and eosin |
| FFPE | Formalin-fixed paraffin-embedded |
References
- Bray, F.; Ferlay, J.; Soerjomataram, I.; Siegel, R.L.; Torre, L.A.; Jemal, A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2018, 68, 394–424. [Google Scholar] [CrossRef] [PubMed]
- Ferlay, J.; Soerjomataram, I.; Dikshit, R.; Eser, S.; Mathers, C.; Rebelo, M.; Parkin, D.M.; Forman, D.; Bray, F. Cancer incidence and mortality worldwide: Sources, methods and major patterns in GLOBOCAN 2012. Int. J. Cancer 2015, 136, E359–E386. [Google Scholar] [CrossRef] [PubMed]
- Travis, W.D.; Brambilla, E.; Nicholson, A.G.; Yatabe, Y.; Austin, J.H.M.; Beasley, M.B.; Chirieac, L.R.; Dacic, S.; Duhig, E.; Flieder, D.B.; et al. The 2015 World Health Organization Classification of Lung Tumors: Impact of Genetic, Clinical and Radiologic Advances Since the 2004 Classification. J. Thorac. Oncol. 2015, 10, 1243–1260. [Google Scholar] [CrossRef] [PubMed]
- Cheng, L.; Alexander, R.E.; Maclennan, G.T.; Cummings, O.W.; Montironi, R.; Lopez-Beltran, A.; Cramer, H.M.; Davidson, D.D.; Zhang, S. Molecular pathology of lung cancer: Key to personalized medicine. Mod. Pathol. 2012, 25, 347–369. [Google Scholar] [CrossRef] [PubMed]
- The Cancer Genome Atlas Research Network. Comprehensive molecular profiling of lung adenocarcinoma. Nature 2014, 511, 543–550. [Google Scholar] [CrossRef]
- Jordan, E.J.; Kim, H.R.; Arcila, M.E.; Barron, D.; Chakravarty, D.; Gao, J.; Chang, M.T.; Ni, A.; Kundra, R.; Jonsson, P.; et al. Prospective Comprehensive Molecular Characterization of Lung Adenocarcinomas for Efficient Patient Matching to Approved and Emerging Therapies. Cancer Discov. 2017, 7, 596–609. [Google Scholar] [CrossRef]
- Soda, M.; Choi, Y.L.; Enomoto, M.; Takada, S.; Yamashita, Y.; Ishikawa, S.; Fujiwara, S.; Watanabe, H.; Kurashina, K.; Hatanaka, H.; et al. Identification of the transforming EML4-ALK fusion gene in non-small-cell lung cancer. Nature 2007, 448, 561–566. [Google Scholar] [CrossRef]
- Pan, Y.; Zhang, Y.; Li, Y.; Hu, H.; Wang, L.; Li, H.; Wang, R.; Ye, T.; Luo, X.; Zhang, Y.; et al. ALK, ROS1 and RET fusions in 1139 lung adenocarcinomas: A comprehensive study of common and fusion pattern-specific clinicopathologic, histologic and cytologic features. Lung Cancer 2014, 84, 121–126. [Google Scholar] [CrossRef] [PubMed]
- Stransky, N.; Cerami, E.; Schalm, S.; Kim, J.L.; Lengauer, C. The landscape of kinase fusions in cancer. Nat. Commun. 2014, 5, 4846. [Google Scholar] [CrossRef] [PubMed]
- Campbell, J.D.; Alexandrov, A.; Kim, J.; Wala, J.; Berger, A.H.; Pedamallu, C.S.; Shukla, S.A.; Guo, G.; Brooks, A.N.; Murray, B.A.; et al. Distinct patterns of somatic genome alterations in lung adenocarcinomas and squamous cell carcinomas. Nat. Genet. 2016, 48, 607–616. [Google Scholar] [CrossRef] [PubMed]
- Shaw, A.T.; Ou, S.H.; Bang, Y.J.; Camidge, D.R.; Solomon, B.J.; Salgia, R.; Riely, G.J.; Varella-Garcia, M.; Shapiro, G.I.; Costa, D.B.; et al. Crizotinib in ROS1-rearranged non-small-cell lung cancer. N. Engl. J. Med. 2014, 371, 1963–1971. [Google Scholar] [CrossRef]
- Solomon, B.J.; Mok, T.; Kim, D.W.; Wu, Y.L.; Nakagawa, K.; Mekhail, T.; Felip, E.; Cappuzzo, F.; Paolini, J.; Usari, T.; et al. First-line crizotinib versus chemotherapy in ALK-positive lung cancer. N. Engl. J. Med. 2014, 371, 2167–2177. [Google Scholar] [CrossRef]
- Kelly, E.E.; Horgan, C.P.; Goud, B.; McCaffrey, M.W. The Rab family of proteins: 25 years on. Biochem. Soc. Trans. 2012, 40, 1337–1347. [Google Scholar] [CrossRef]
- Chua, C.E.; Tang, B.L. Engagement of the small GTPase Rab31 protein and its effector, early endosome antigen 1, is important for trafficking of the ligand-bound epidermal growth factor receptor from the early to the late endosome. J. Biol. Chem. 2014, 289, 12375–12389. [Google Scholar] [CrossRef]
- Chua, C.E.; Tang, B.L. The role of the small GTPase Rab31 in cancer. J. Cell. Mol. Med. 2015, 19, 1–10. [Google Scholar] [CrossRef]
- Pan, Y.; Zhang, Y.; Chen, L.; Liu, Y.; Feng, Y.; Yan, J. The Critical Role of Rab31 in Cell Proliferation and Apoptosis in Cancer Progression. Mol. Neurobiol. 2016, 53, 4431–4437. [Google Scholar] [CrossRef]
- Grismayer, B.; Solch, S.; Seubert, B.; Kirchner, T.; Schafer, S.; Baretton, G.; Schmitt, M.; Luther, T.; Kruger, A.; Kotzsch, M.; et al. Rab31 expression levels modulate tumor-relevant characteristics of breast cancer cells. Mol. Cancer 2012, 11, 62. [Google Scholar] [CrossRef]
- Sui, Y.; Zheng, X.; Zhao, D. Rab31 promoted hepatocellular carcinoma (HCC) progression via inhibition of cell apoptosis induced by PI3K/AKT/Bcl-2/BAX pathway. Tumour Biol. 2015, 36, 8661–8670. [Google Scholar] [CrossRef]
- Nishimura, Y.; Hayashi, M.; Inada, H.; Tanaka, T. Molecular cloning and characterization of mammalian homologues of vesicle-associated membrane protein-associated (VAMP-associated) proteins. Biochem. Biophys. Res. Commun. 1999, 254, 21–26. [Google Scholar] [CrossRef]
- Shaw, A.T.; Hsu, P.P.; Awad, M.M.; Engelman, J.A. Tyrosine kinase gene rearrangements in epithelial malignancies. Nat. Rev. Cancer 2013, 13, 772–787. [Google Scholar] [CrossRef] [PubMed]
- Mertens, F.; Johansson, B.; Fioretos, T.; Mitelman, F. The emerging complexity of gene fusions in cancer. Nat. Rev. Cancer 2015, 15, 371–381. [Google Scholar] [CrossRef]
- Yoshihara, K.; Wang, Q.; Torres-Garcia, W.; Zheng, S.; Vegesna, R.; Kim, H.; Verhaak, R.G. The landscape and therapeutic relevance of cancer-associated transcript fusions. Oncogene 2015, 34, 4845–4854. [Google Scholar] [CrossRef]
- Mani, R.S.; Chinnaiyan, A.M. Triggers for genomic rearrangements: Insights into genomic, cellular and environmental influences. Nat. Rev. Genet. 2010, 11, 819–829. [Google Scholar] [CrossRef]
- Mani, R.S.; Tomlins, S.A.; Callahan, K.; Ghosh, A.; Nyati, M.K.; Varambally, S.; Palanisamy, N.; Chinnaiyan, A.M. Induced chromosomal proximity and gene fusions in prostate cancer. Science 2009, 326, 1230. [Google Scholar] [CrossRef] [PubMed]
- Maher, C.A.; Kumar-Sinha, C.; Cao, X.; Kalyana-Sundaram, S.; Han, B.; Jing, X.; Sam, L.; Barrette, T.; Palanisamy, N.; Chinnaiyan, A.M. Transcriptome sequencing to detect gene fusions in cancer. Nature 2009, 458, 97–101. [Google Scholar] [CrossRef] [PubMed]
- Maher, C.A.; Palanisamy, N.; Brenner, J.C.; Cao, X.; Kalyana-Sundaram, S.; Luo, S.; Khrebtukova, I.; Barrette, T.R.; Grasso, C.; Yu, J.; et al. Chimeric transcript discovery by paired-end transcriptome sequencing. Proc. Natl. Acad. Sci. USA 2009, 106, 12353–12358. [Google Scholar] [CrossRef]
- Albright, C.D.; Jones, R.T.; Hudson, E.A.; Fontana, J.A.; Trump, B.F.; Resau, J.H. Transformed human bronchial epithelial cells (BEAS-2B) alter the growth and morphology of normal human bronchial epithelial cells in vitro. Cell Biol. Toxicol. 1990, 6, 379–398. [Google Scholar] [CrossRef]
- Hanahan, D.; Weinberg, R.A. Hallmarks of cancer: The next generation. Cell 2011, 144, 646–674. [Google Scholar] [CrossRef]
- Wilson, B.E.; Mochon, E.; Boxer, L.M. Induction of bcl-2 expression by phosphorylated CREB proteins during B-cell activation and rescue from apoptosis. Mol. Cell. Biol. 1996, 16, 5546–5556. [Google Scholar] [CrossRef]
- Naqvi, S.; Martin, K.J.; Arthur, J.S. CREB phosphorylation at Ser133 regulates transcription via distinct mechanisms downstream of cAMP and MAPK signalling. Biochem. J. 2014, 458, 469–479. [Google Scholar] [CrossRef]
- Sakamoto, K.M.; Frank, D.A. CREB in the pathophysiology of cancer: Implications for targeting transcription factors for cancer therapy. Clin. Cancer Res. 2009, 15, 2583–2587. [Google Scholar] [CrossRef] [PubMed]
- Tiscornia, G.; Singer, O.; Verma, I.M. Production and purification of lentiviral vectors. Nat. Protoc. 2006, 1, 241–245. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.Y.; Kim, M.S.; Kim, E.H.; Chung, N.; Jeong, Y.K. Retrospective growth kinetics and radiosensitivity analysis of various human xenograft models. Lab. Anim. Res. 2016, 32, 187–193. [Google Scholar] [CrossRef] [PubMed]

© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yoon, D.; Bae, K.; Kim, J.-H.; Choi, Y.-K.; Yoon, K.-A. Oncogenic Effect of the Novel Fusion Gene VAPA-Rab31 in Lung Adenocarcinoma. Int. J. Mol. Sci. 2019, 20, 2309. https://doi.org/10.3390/ijms20092309
Yoon D, Bae K, Kim J-H, Choi Y-K, Yoon K-A. Oncogenic Effect of the Novel Fusion Gene VAPA-Rab31 in Lung Adenocarcinoma. International Journal of Molecular Sciences. 2019; 20(9):2309. https://doi.org/10.3390/ijms20092309
Chicago/Turabian StyleYoon, Daseul, Kieun Bae, Jin-Hee Kim, Yang-Kyu Choi, and Kyong-Ah Yoon. 2019. "Oncogenic Effect of the Novel Fusion Gene VAPA-Rab31 in Lung Adenocarcinoma" International Journal of Molecular Sciences 20, no. 9: 2309. https://doi.org/10.3390/ijms20092309
APA StyleYoon, D., Bae, K., Kim, J.-H., Choi, Y.-K., & Yoon, K.-A. (2019). Oncogenic Effect of the Novel Fusion Gene VAPA-Rab31 in Lung Adenocarcinoma. International Journal of Molecular Sciences, 20(9), 2309. https://doi.org/10.3390/ijms20092309




