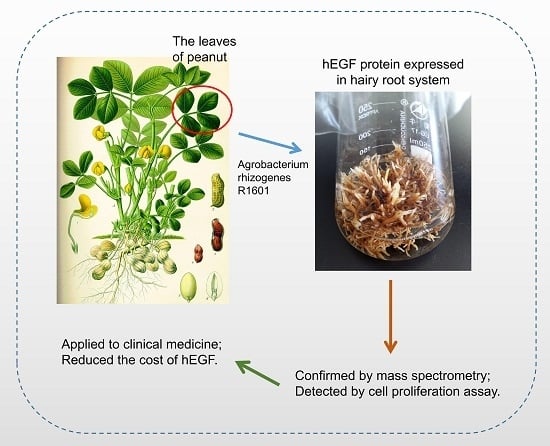
High Efficient Expression and Purification of Human Epidermal Growth Factor in Arachis Hypogaea L.
Abstract
:1. Introduction
2. Results
2.1. The Establishment of Protein Expression System in Peanut Hairy Root
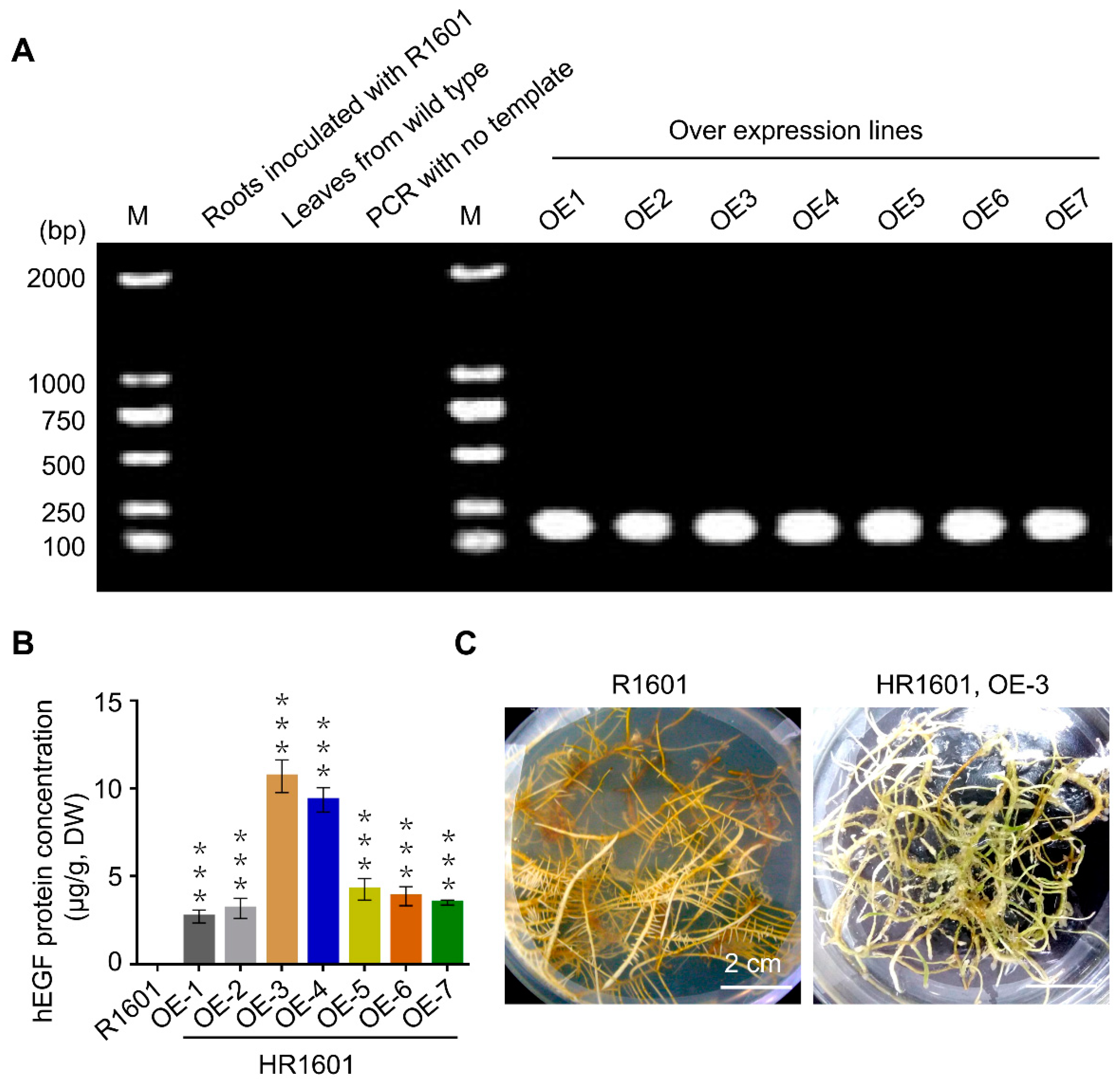
2.2. The Identification of the hEGF Transgenic Hairy Roots
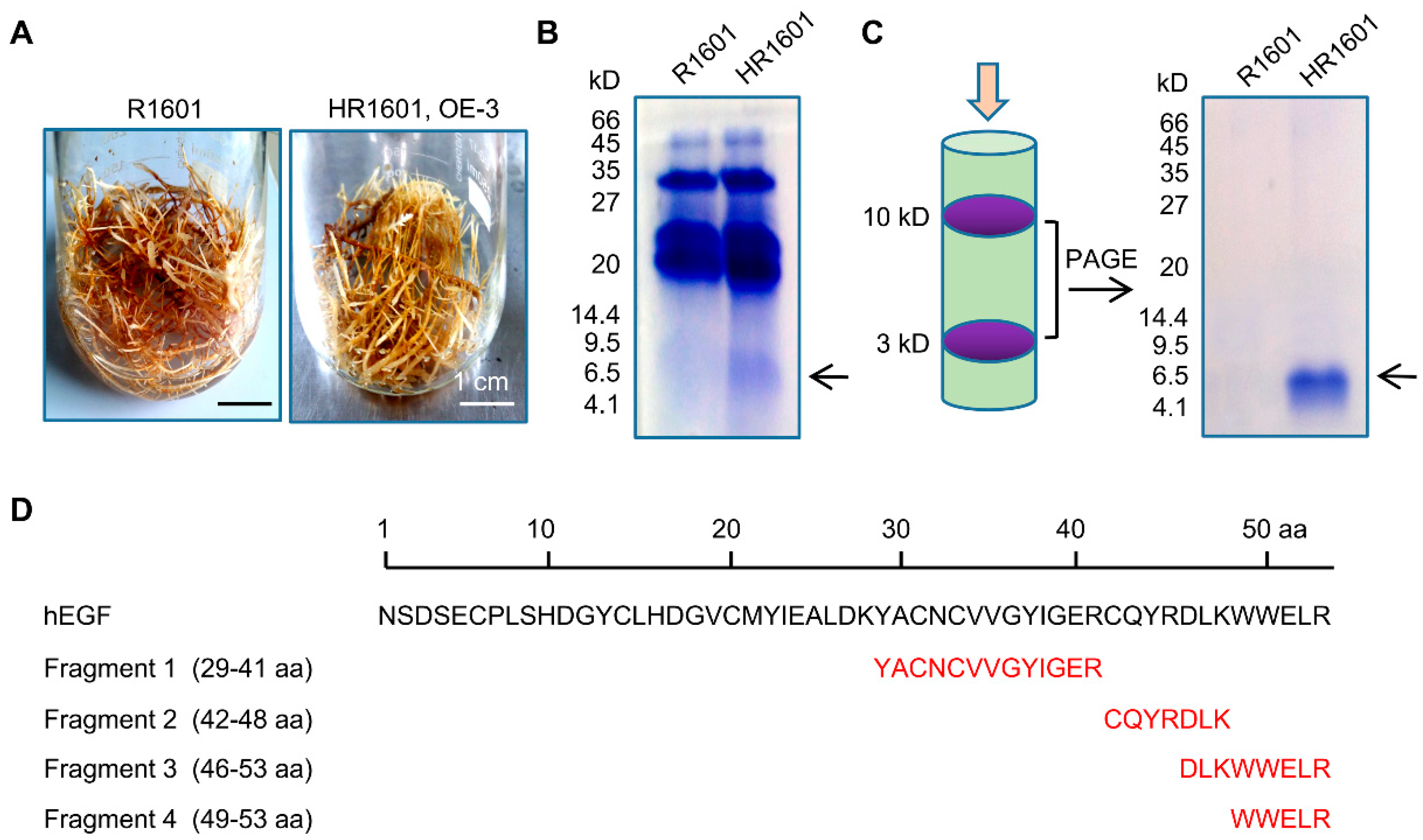
2.3. The Isolation and Purification of hEGF Peptide in Hairy Roots
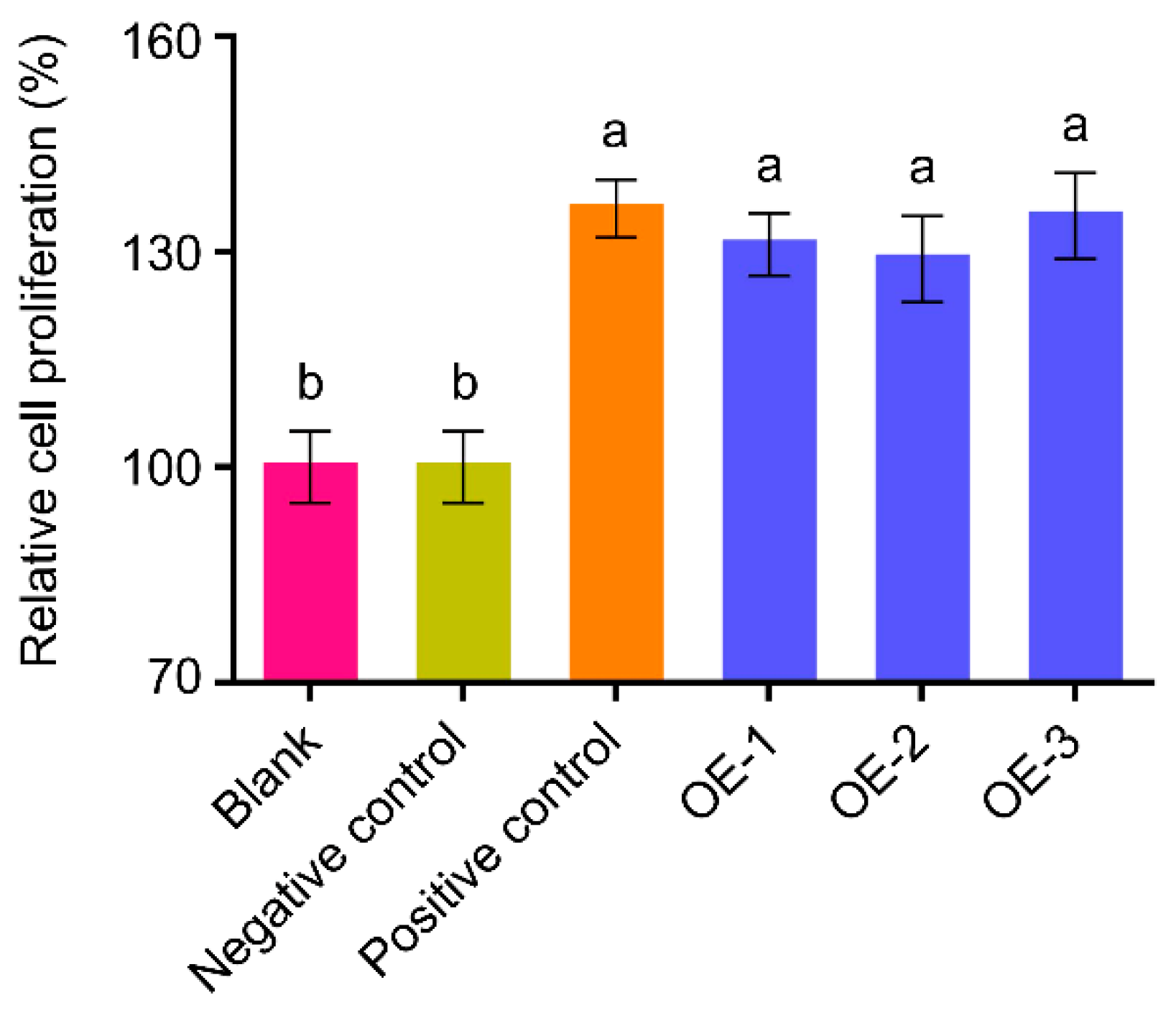
2.4. The hEGF Protein Purified from Peanut Hairy Roots Has Biological Activity
3. Discussion
4. Materials and Methods
4.1. Plant Material and Bacteria Strains
4.2. The Construction of HR1601 Strain
4.3. The Establishment of Peanut Hairy Root Culture System
4.4. Hairy Root Growth and hEGF Protein Content Analyses
4.5. The Detection of the hEGF Protein by Matrix-Assisted Laser Desorption/Ionization Time of Flight Mass Spectrometry
4.6. Digestion Before MS Analysis
4.7. MS Analysis
4.8. Cell Proliferation Assay
4.9. Statistical Tests
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| hEGF | Human epidermal growth factor |
| EGFR | hEGF receptor |
| MTT | Methylthiazolyldiphenyl-tetrazolium bromide |
| OE | overexpression lines |
| bp | base pair |
| MS | Murashige and Skoog medium |
| MALDI-TOF-MS | matrix-assisted laser desorption/ionization time of flight mass spectrometry |
References
- Cooke, R.M.; Wilkinson, A.J.; Baron, M.; Pastore, A.; Tappin, M.J.; Campbell, I.D.; Gregory, H.; Sheard, B. The solution structure of human epidermal growth factor. Nature 1987, 327, 339–341. [Google Scholar] [CrossRef] [PubMed]
- Mount, C.D.; Lukas, T.J.; Orth, D.N. Purification and characterization of epidermal growth factor (beta-urogastrone) and epidermal growth factor fragments from large volumes of human urine. Arch. Biochem. Biophys. 1985, 240, 33–42. [Google Scholar] [CrossRef]
- Stanley, M.A.; Dahlenburg, K. The effect of choleragen and epidermal growth factor on proliferation and maturation in vitro of human ectocervical cells. In Vitro 1984, 20, 144–151. [Google Scholar] [CrossRef] [PubMed]
- Cohen, S.; Carpenter, G. Human epidermal growth factor: Isolation and chemical and biological properties. Proc. Natl. Acad. Sci. USA 1975, 72, 1317–1321. [Google Scholar] [CrossRef] [PubMed]
- Jorissen, R.N.; Walker, F.; Pouliot, N.; Garrett, T.P.; Ward, C.W.; Burgess, A.W. Epidermal growth factor receptor: Mechanisms of activation and signalling. Exp. Cell Res. 2003, 284, 31–53. [Google Scholar] [CrossRef] [PubMed]
- Parries, G.; Chen, K.; Misono, K.S.; Cohen, S. The human urinary epidermal growth factor (EGF) precursor. Isolation of a biologically active 160-kilodalton heparin-binding pro-EGF with a truncated carboxyl terminus. J. Biol. Chem. 1995, 270, 27954–27960. [Google Scholar] [CrossRef]
- Lee, Y.S.; Suh, C.W.; Park, S.K.; Lee, E.K. Purification of soluble human epidermal growth factor (hEGF) from recombinant Escherichia coli culture broth by using expanded-bed adsorption chromatography. Biotechnol. Appl. Biochem. 2003, 38, 9–13. [Google Scholar] [CrossRef]
- Wu, C.S.; Kuo, W.T.; Chang, C.Y.; Kuo, J.Y.; Tsai, Y.T.; Yu, S.M.; Wu, H.T.; Chen, P.W. The modified rice alphaAmy8 promoter confers high-level foreign gene expression in a novel hypoxia-inducible expression system in transgenic rice seedlings. Plant. Mol. Biol. 2014, 85, 147–161. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.; Yu, J.; Lin, J.; Wu, S.; Li, S.; Wang, J. High Efficient Expression, Purification, and Functional Characterization of Native Human Epidermal Growth Factor in Escherichia coli. Biomed. Res. Int. 2016, 2016, 3758941. [Google Scholar] [CrossRef] [PubMed]
- Bai, J.Y.; Zeng, L.; Hu, Y.L.; Li, Y.F.; Lin, Z.P.; Shang, S.C.; Shi, Y.S. Expression and characteristic of synthetic human epidermal growth factor (hEGF) in transgenic tobacco plants. Biotechnol. Lett. 2007, 29, 2007–2012. [Google Scholar] [CrossRef] [PubMed]
- Eissazadeh, S.; Moeini, H.; Dezfouli, M.G.; Heidary, S.; Nelofer, R.; Abdullah, M.P. Production of recombinant human epidermal growth factor in Pichia pastoris. Braz. J. Microbiol. 2017, 48, 286–293. [Google Scholar] [CrossRef]
- Parsons, J.; Wirth, S.; Dominguez, M.; Bravo-Almonacid, F.; Giulietti, A.M.; Talou, J.R. Production of Human Epidermal Growth Factor (hEGF) by in Vitro Cultures of Nicotiana tabacum: Effect of Tissue Differentiation and Sodium Nitroprusside Addition. Int. J. Biotechnol. Biochem. 2010, 6, 133–140. [Google Scholar]
- Banerjee, S.; Singh, S.; Ur Rahman, L. Biotransformation studies using hairy root cultures—A review. Biotechnol. Adv. 2012, 30, 461–468. [Google Scholar] [CrossRef] [PubMed]
- Woods, R.R.; Geyer, B.C.; Mor, T.S. Hairy-root organ cultures for the production of human acetylcholinesterase. BMC Biotechnol. 2008, 8, 95. [Google Scholar] [CrossRef]
- Liu, J.; Dolan, M.C.; Reidy, M.; Cramer, C.L. Expression of bioactive single-chain murine IL-12 in transgenic plants. J. Interferon Cytokine Res. 2008, 28, 381–392. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.R.; Sim, J.S.; Ajjappala, H.; Kim, Y.H.; Hahn, B.S. Expression and large-scale production of the biochemically active human tissue-plasminogen activator in hairy roots of Oriental melon (Cucumis melo). J. Biosci. Bioeng. 2012, 113, 106–111. [Google Scholar] [CrossRef]
- Ono, N.N.; Tian, L. The multiplicity of hairy root cultures: Prolific possibilities. Plant. Sci. 2011, 180, 439–446. [Google Scholar] [CrossRef]
- Takeo, M.; Lee, W.; Ito, M. Wound healing and skin regeneration. Cold Spring Harb. Perspect. Med. 2015, 5, a023267. [Google Scholar] [CrossRef]
- Guimaraes, L.A.; Pereira, B.M.; Araujo, A.C.G.; Guimaraes, P.M.; Brasileiro, A.C.M. Ex vitro hairy root induction in detached peanut leaves for plant-nematode interaction studies. Plant. Methods 2017, 13, 25. [Google Scholar] [CrossRef]
- Yang, T.; Fang, L.; Nopo-Olazabal, C.; Condori, J.; Nopo-Olazabal, L.; Balmaceda, C.; Medina-Bolivar, F. Enhanced Production of Resveratrol, Piceatannol, Arachidin-1, and Arachidin-3 in Hairy Root Cultures of Peanut Co-treated with Methyl Jasmonate and Cyclodextrin. J. Agric. Food Chem. 2015, 63, 3942–3950. [Google Scholar] [CrossRef]
- Zhang, N.; Wright, T.; Wang, X.; Karki, U.; Savary, B.J.; Xu, J. Engineering ‘designer’ glycomodules for boosting recombinant protein secretion in tobacco hairy root culture and studying hydroxyproline-O-glycosylation process in plants. Plant. Biotechnol. J. 2018. [Google Scholar] [CrossRef] [PubMed]
- Zhou, M.L.; Zhu, X.M.; Shao, J.R.; Tang, Y.X.; Wu, Y.M. Production and metabolic engineering of bioactive substances in plant hairy root culture. Appl. Microbiol. Biotechnol. 2011, 90, 1229–1239. [Google Scholar] [CrossRef] [PubMed]
- Cardon, F.; Pallisse, R.; Bardor, M.; Caron, A.; Vanier, J.; Ele Ekouna, J.P.; Lerouge, P.; Boitel-Conti, M.; Guillet, M. Brassica rapa hairy root based expression system leads to the production of highly homogenous and reproducible profiles of recombinant human alpha-L-iduronidase. Plant. Biotechnol. J. 2019, 17, 505–516. [Google Scholar] [CrossRef] [PubMed]
- Abbott, J.A.; Medina-Bolivar, F.; Martin, E.M.; Engelberth, A.S.; Villagarcia, H.; Clausen, E.C.; Carrier, D.J. Purification of resveratrol, arachidin-1, and arachidin-3 from hairy root cultures of peanut (Arachis hypogaea) and determination of their antioxidant activity and cytotoxicity. Biotechnol. Prog. 2010, 26, 1344–1351. [Google Scholar] [CrossRef] [PubMed]
- Yamagata, H.; Nakahama, K.; Suzuki, Y.; Kakinuma, A.; Tsukagoshi, N.; Udaka, S. Use of Bacillus brevis for efficient synthesis and secretion of human epidermal growth factor. Proc. Natl. Acad. Sci. USA 1989, 86, 3589–3593. [Google Scholar] [CrossRef] [PubMed]
- Katayama, H.; Nagasu, T.; Oda, Y. Improvement of in-gel digestion protocol for peptide mass fingerprinting by matrix-assisted laser desorption/ionization time-of-flight mass spectrometry. Rapid Commun. Mass Spectrom. 2001, 15, 1416–1421. [Google Scholar] [CrossRef] [PubMed]
- Pan, X.; Zhang, X.; Sun, H.; Zhang, J.; Yan, M.; Zhang, H. Autophagy inhibition promotes 5-fluorouraci-induced apoptosis by stimulating ROS formation in human non-small cell lung cancer A549 cells. PLoS One 2013, 8, e56679. [Google Scholar] [CrossRef] [PubMed]

© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yao, Q.; Yu, Z.; Liu, P.; Zheng, H.; Xu, Y.; Sai, S.; Wu, Y.; Zheng, C. High Efficient Expression and Purification of Human Epidermal Growth Factor in Arachis Hypogaea L. Int. J. Mol. Sci. 2019, 20, 2045. https://doi.org/10.3390/ijms20082045
Yao Q, Yu Z, Liu P, Zheng H, Xu Y, Sai S, Wu Y, Zheng C. High Efficient Expression and Purification of Human Epidermal Growth Factor in Arachis Hypogaea L. International Journal of Molecular Sciences. 2019; 20(8):2045. https://doi.org/10.3390/ijms20082045
Chicago/Turabian StyleYao, Qingshou, Zipeng Yu, Pu Liu, Hao Zheng, Yang Xu, Sixiang Sai, Yuyong Wu, and Chengchao Zheng. 2019. "High Efficient Expression and Purification of Human Epidermal Growth Factor in Arachis Hypogaea L." International Journal of Molecular Sciences 20, no. 8: 2045. https://doi.org/10.3390/ijms20082045
APA StyleYao, Q., Yu, Z., Liu, P., Zheng, H., Xu, Y., Sai, S., Wu, Y., & Zheng, C. (2019). High Efficient Expression and Purification of Human Epidermal Growth Factor in Arachis Hypogaea L. International Journal of Molecular Sciences, 20(8), 2045. https://doi.org/10.3390/ijms20082045