Clinically Relevant Immune Responses against Cytomegalovirus: Implications for Precision Medicine
Abstract
1. Introduction
2. CMV–Host Interactions in Cancer
T-Cell Reactivity Against CMV Targets in T Cells from Patients with Cancer—Experience at the ImmunoSurgery Unit, Champalimaud Centre for the Unknown (CCU)
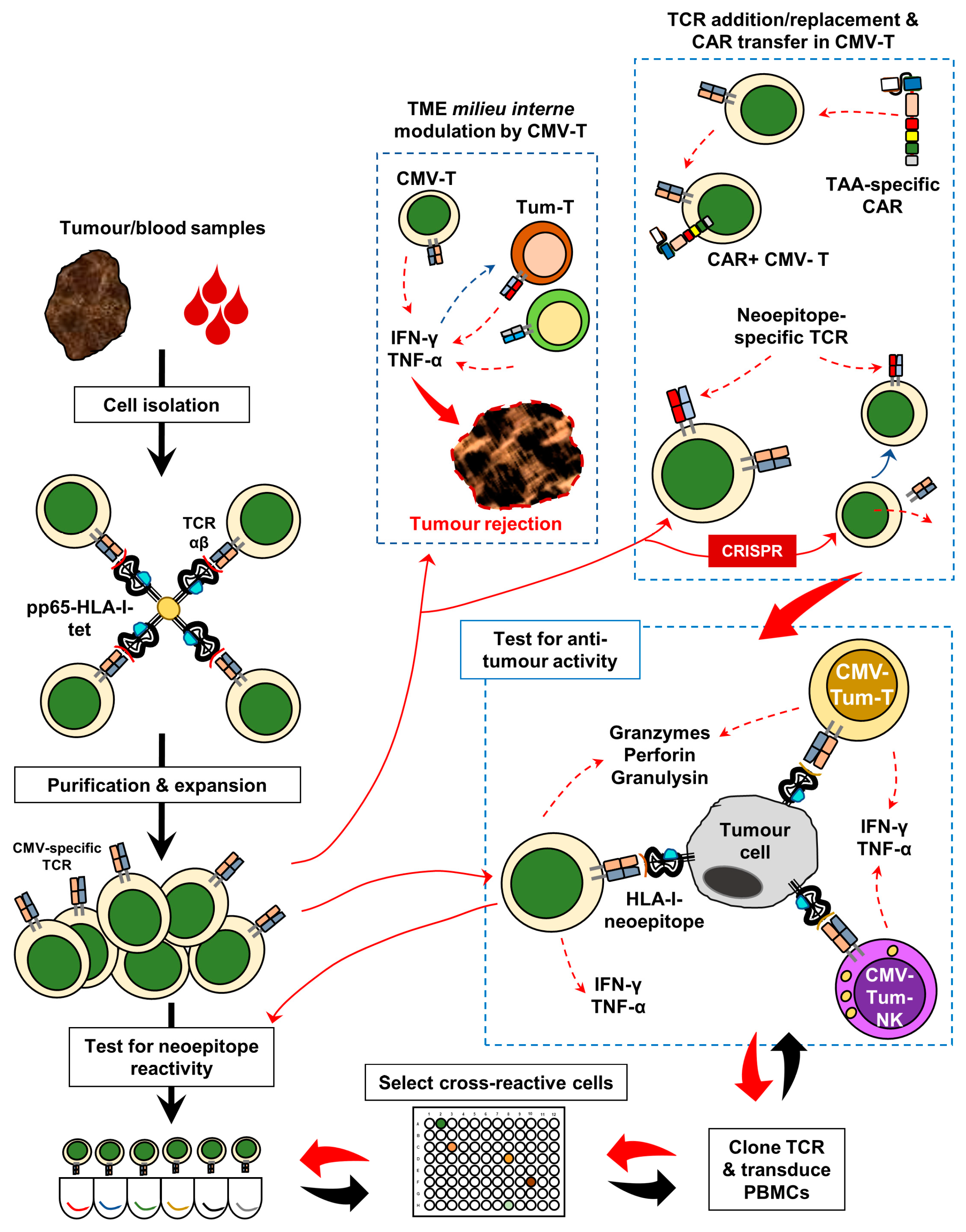
3. Harnessing Anti-CMV Immunomodulation for Precision Oncology
4. Material and Methods (Original Data from the CCU)
T-Cell Exposure to CMV pp65 and EBNA-1 Peptides
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| CCU | Champalimaud Centre for the Unknown |
| CMV | Cytomegalovirus |
| EBV | Epstein–Barr virus |
| MCMV | Murine cytomegalovirus |
| TIL | Tumour-infiltrating lymphocytes |
| TIB | Tumour-infiltrating B cells |
| PBMCs | Peripheral blood mononuclear cells |
| TCR | T-cell receptor |
| BCR | B-cell receptor |
| HLA | Human leukocyte antigen |
| MHC | Major histocompatibility complex |
| TME | Tumour microenvironment |
| PD-1 | Programmed cell death 1 |
| TIM-3 | T-cell immunoglobulin mucin-domain-containing 3 |
| IgG | Immunoglobulin gamma |
| HSCT | Haematopoietic stem cell transplantation |
| ADCC | Antibody-dependent cellular cytotoxicity |
| WBA | Whole-blood assay |
| ELISA | Enzyme-linked immunosorbent assay |
| IFN-γ | Interferon gamma |
| pg | picogrammes |
| IU | International units |
| GBM | Glioblastoma multiforme |
| PC | Pancreatic cancer |
| CRC | Colorectal cancer |
| EsoCA | Oesophageal cancer |
| BCC | Basal cell carcinoma |
| ICI | Immune checkpoint inhibitor |
| CMV-IVIG | Cytomegalovirus-intravenous immunoglibulin |
| PE | Pleural effusion |
| EBNA-1 | Epstein–Barr nuclear antigen 1 |
| PHA | Phytohaemagglutinin |
| CAR | Chimeric antigen receptor |
| Th1 | T helper 1 |
References
- Souquette, A.; Frere, J.; Smithey, M.; Sauce, D.; Thomas, P.G. A constant companion: Immune recognition and response to cytomegalovirus with aging and implications for immune fitness. GeroScience 2017, 39, 293–303. [Google Scholar] [CrossRef]
- Kotton, C.N.; Kumar, D.; Caliendo, A.M.; Huprikar, S.; Chou, S.; Danziger-Isakov, L.; Humar, A. The Third International Consensus Guidelines on the Management of Cytomegalovirus in Solid-organ Transplantation. Transplantation 2018, 102, 900–931. [Google Scholar] [CrossRef] [PubMed]
- Chen, K.; Cheng, M.P.; Hammond, S.P.; Einsele, H.; Marty, F.M. Antiviral prophylaxis for cytomegalovirus infection in allogeneic hematopoietic cell transplantation. Blood Adv. 2018, 2, 2159–2175. [Google Scholar] [CrossRef] [PubMed]
- Fowler, K.B.; Boppana, S.B. Congenital cytomegalovirus infection. Semin. Perinatol. 2018, 42, 149–154. [Google Scholar] [CrossRef]
- Adland, E.; Klenerman, P.; Goulder, P.; Matthews, P.C. Ongoing burden of disease and mortality from HIV/CMV coinfection in Africa in the antiretroviral therapy era. Front. Microbiol. 2015, 6, 1016. [Google Scholar] [CrossRef] [PubMed]
- Sehrawat, S.; Kumar, D.; Rouse, B.T. Herpesviruses: Harmonious Pathogens but Relevant Cofactors in Other Diseases? Front. Cell. Infect. Microbiol. 2018, 8. [Google Scholar] [CrossRef] [PubMed]
- La Rosa, C.; Diamond, D.J. The immune response to human CMV. Future Virol. 2012, 7, 279–293. [Google Scholar] [CrossRef]
- Muccio, L.; Bertaina, A.; Falco, M.; Pende, D.; Meazza, R.; Lopez-Botet, M.; Moretta, L.; Locatelli, F.; Moretta, A.; Della Chiesa, M. Analysis of memory-like natural killer cells in human cytomegalovirus-infected children undergoing αβ+T and B cell-depleted hematopoietic stem cell transplantation for hematological malignancies. Haematologica 2016, 101, 371–381. [Google Scholar] [CrossRef]
- Iancu, E.M.; Corthesy, P.; Baumgaertner, P.; Devevre, E.; Voelter, V.; Romero, P.; Speiser, D.E.; Rufer, N. Clonotype selection and composition of human CD8 T cells specific for persistent herpes viruses varies with differentiation but is stable over time. J. Immunol. 2009, 183, 319–331. [Google Scholar] [CrossRef] [PubMed]
- Okhrimenko, A.; Grun, J.R.; Westendorf, K.; Fang, Z.; Reinke, S.; von Roth, P.; Wassilew, G.; Kuhl, A.A.; Kudernatsch, R.; Demski, S.; et al. Human memory T cells from the bone marrow are resting and maintain long-lasting systemic memory. Proc. Natl. Acad. Sci. USA 2014, 111, 9229–9234. [Google Scholar] [CrossRef] [PubMed]
- Turula, H.; Smith, C.J.; Grey, F.; Zurbach, K.A.; Snyder, C.M. Competition between T cells maintains clonal dominance during memory inflation induced by MCMV. Eur. J. Immunol. 2013, 43, 1252–1263. [Google Scholar] [CrossRef]
- Wang, G.C.; Dash, P.; McCullers, J.A.; Doherty, P.C.; Thomas, P.G. T cell receptor alphabeta diversity inversely correlates with pathogen-specific antibody levels in human cytomegalovirus infection. Sci. Transl. Med. 2012, 4, 128ra142. [Google Scholar] [CrossRef]
- Huth, A.; Liang, X.; Krebs, S.; Blum, H.; Moosmann, A. Antigen-Specific TCR Signatures of Cytomegalovirus Infection. J. Immunol. 2019, 202, 979–990. [Google Scholar] [CrossRef]
- van de Berg, P.J.; Heutinck, K.M.; Raabe, R.; Minnee, R.C.; Young, S.L.; van Donselaar-van der Pant, K.A.; Bemelman, F.J.; van Lier, R.A.; ten Berge, I.J. Human cytomegalovirus induces systemic immune activation characterized by a type 1 cytokine signature. J. Infect. Dis. 2010, 202, 690–699. [Google Scholar] [CrossRef]
- Turner, J.E.; Campbell, J.P.; Edwards, K.M.; Howarth, L.J.; Pawelec, G.; Aldred, S.; Moss, P.; Drayson, M.T.; Burns, V.E.; Bosch, J.A. Rudimentary signs of immunosenescence in Cytomegalovirus-seropositive healthy young adults. Age 2014, 36, 287–297. [Google Scholar] [CrossRef]
- Di Benedetto, S.; Gaetjen, M.; Müller, L. The Modulatory Effect of Gender and Cytomegalovirus-Seropositivity on Circulating Inflammatory Factors and Cognitive Performance in Elderly Individuals. Int. J. Mol. Sci. 2019, 20, 990. [Google Scholar] [CrossRef]
- Liu, Z.; Poiret, T.; Meng, Q.; Rao, M.; von Landenberg, A.; Schoutrop, E.; Valentini, D.; Dodoo, E.; Peredo-Harvey, I.; Maeurer, M. Epstein-Barr virus- and cytomegalovirus-specific immune response in patients with brain cancer. J. Transl. Med. 2018, 16, 182. [Google Scholar] [CrossRef]
- Nagu, T.; Aboud, S.; Rao, M.; Matee, M.; Axelsson, R.; Valentini, D.; Mugusi, F.; Zumla, A.; Maeurer, M. Strong anti-Epstein Barr virus (EBV) or cytomegalovirus (CMV) cellular immune responses predict survival and a favourable response to anti-tuberculosis therapy. Int. J. Infect. Dis. IJID Off. Publ. Int. Soc. Infect. Dis. 2017, 56, 136–139. [Google Scholar] [CrossRef]
- Rahbar, A.; Peredo, I.; Solberg, N.W.; Taher, C.; Dzabic, M.; Xu, X.; Skarman, P.; Fornara, O.; Tammik, C.; Yaiw, K.; et al. Discordant humoral and cellular immune responses to Cytomegalovirus (CMV) in glioblastoma patients whose tumors are positive for CMV. Oncoimmunology 2015, 4, e982391. [Google Scholar] [CrossRef]
- Mitchell, D.A.; Xie, W.; Schmittling, R.; Learn, C.; Friedman, A.; McLendon, R.E.; Sampson, J.H. Sensitive detection of human cytomegalovirus in tumors and peripheral blood of patients diagnosed with glioblastoma. Neuro-Oncology 2008, 10, 10–18. [Google Scholar] [CrossRef]
- Priel, E.; Wohl, A.; Teperberg, M.; Nass, D.; Cohen, Z.R. Human cytomegalovirus viral load in tumor and peripheral blood samples of patients with malignant gliomas. J. Clin. Neurosci. Off. J. Neurosurg. Soc. Australas 2015, 22, 326–330. [Google Scholar] [CrossRef]
- Stragliotto, G.; Rahbar, A.; Solberg, N.W.; Lilja, A.; Taher, C.; Orrego, A.; Bjurman, B.; Tammik, C.; Skarman, P.; Peredo, I.; et al. Effects of valganciclovir as an add-on therapy in patients with cytomegalovirus-positive glioblastoma: A randomized, double-blind, hypothesis-generating study. Int. J. Cancer 2013, 133, 1204–1213. [Google Scholar] [CrossRef]
- Söderberg-Nauclér, C.; Rahbar, A.; Stragliotto, G. Survival in patients with glioblastoma receiving valganciclovir. N. Engl. J. Med. 2013, 369, 985–986. [Google Scholar] [CrossRef]
- Solomon, I.H.; Ramkissoon, S.H.; Milner, D.A., Jr.; Folkerth, R.D. Cytomegalovirus and glioblastoma: A review of evidence for their association and indications for testing and treatment. J. Neuropathol. Exp. Neurol. 2014, 73, 994–998. [Google Scholar] [CrossRef]
- Nair, S.K.; De Leon, G.; Boczkowski, D.; Schmittling, R.; Xie, W.; Staats, J.; Liu, R.; Johnson, L.A.; Weinhold, K.; Archer, G.E.; et al. Recognition and killing of autologous, primary glioblastoma tumor cells by human cytomegalovirus pp65-specific cytotoxic T cells. Clin. Cancer Res. Off. J. Am. Assoc. Cancer Res. 2014, 20, 2684–2694. [Google Scholar] [CrossRef]
- Crough, T.; Beagley, L.; Smith, C.; Jones, L.; Walker, D.G.; Khanna, R. Ex vivo functional analysis, expansion and adoptive transfer of cytomegalovirus-specific T-cells in patients with glioblastoma multiforme. Immunol. Cell Biol. 2012, 90, 872–880. [Google Scholar] [CrossRef]
- Xing, Y.; Wang, Y.; Wang, S.; Wang, X.; Fan, D.; Zhou, D.; An, J. Human cytomegalovirus infection contributes to glioma disease progression via upregulating endocan expression. Transl. Res. J. Lab. Clin. Med. 2016, 177, 113–126. [Google Scholar] [CrossRef]
- Cobbs, C.S.; Soroceanu, L.; Denham, S.; Zhang, W.; Britt, W.J.; Pieper, R.; Kraus, M.H. Human cytomegalovirus induces cellular tyrosine kinase signaling and promotes glioma cell invasiveness. J. Neuro-Oncol. 2007, 85, 271–280. [Google Scholar] [CrossRef]
- Bai, B.; Wang, X.; Chen, E.; Zhu, H. Human cytomegalovirus infection and colorectal cancer risk: A meta-analysis. Oncotarget 2016, 7, 76735–76742. [Google Scholar] [CrossRef]
- Herbein, G.; Kumar, A. The oncogenic potential of human cytomegalovirus and breast cancer. Front. Oncol. 2014, 4, 230. [Google Scholar] [CrossRef]
- Franklin, C.; Rooms, I.; Fiedler, M.; Reis, H.; Milsch, L.; Herz, S.; Livingstone, E.; Zimmer, L.; Schmid, K.W.; Dittmer, U.; et al. Cytomegalovirus reactivation in patients with refractory checkpoint inhibitor-induced colitis. Eur. J. Cancer 2017, 86, 248–256. [Google Scholar] [CrossRef]
- Lankes, K.; Hundorfean, G.; Harrer, T.; Pommer, A.J.; Agaimy, A.; Angelovska, I.; Tajmir-Riahi, A.; Gohl, J.; Schuler, G.; Neurath, M.F.; et al. Anti-TNF-refractory colitis after checkpoint inhibitor therapy: Possible role of CMV-mediated immunopathogenesis. Oncoimmunology 2016, 5, e1128611. [Google Scholar] [CrossRef]
- Schulz, U.; Solidoro, P.; Müller, V.; Szabo, A.; Gottlieb, J.; Wilkens, H.; Enseleit, F. CMV Immunoglobulins for the Treatment of CMV Infections in Thoracic Transplant Recipients. Transplantation 2016, 100, S5–S10. [Google Scholar] [CrossRef]
- Schampera, M.S.; Schweinzer, K.; Abele, H.; Kagan, K.O.; Klein, R.; Rettig, I.; Jahn, G.; Hamprecht, K. Comparison of cytomegalovirus (CMV)-specific neutralization capacity of hyperimmunoglobulin (HIG) versus standard intravenous immunoglobulin (IVIG) preparations: Impact of CMV IgG normalization. J. Clin. Virol. Off. Publ. Pan Am. Soc. Clin. Virol. 2017, 90, 40–45. [Google Scholar] [CrossRef]
- Alsuliman, T.; Kitel, C.; Dulery, R.; Guillaume, T.; Larosa, F.; Cornillon, J.; Labussiere-Wallet, H.; Mediavilla, C.; Belaiche, S.; Delage, J.; et al. Cytotect(R)CP as salvage therapy in patients with CMV infection following allogeneic hematopoietic cell transplantation: A multicenter retrospective study. Bone Marrow Transplant. 2018, 53, 1328–1335. [Google Scholar] [CrossRef]
- Tan, B.H. Cytomegalovirus Treatment. Curr. Treat. Options Infect. Dis. 2014, 6, 256–270. [Google Scholar] [CrossRef]
- Meng, Q.; Valentini, D.; Rao, M.; Dodoo, E.; Maeurer, M. CMV and EBV targets recognized by tumor-infiltrating B lymphocytes in pancreatic cancer and brain tumors. Sci. Rep. 2018, 8, 17079–17089. [Google Scholar] [CrossRef]
- Scheper, W.; van Dorp, S.; Kersting, S.; Pietersma, F.; Lindemans, C.; Hol, S.; Heijhuurs, S.; Sebestyen, Z.; Gründer, C.; Marcu-Malina, V. γδT cells elicited by CMV reactivation after allo-SCT cross-recognize CMV and leukemia. Leukemia 2013, 27, 1328–1338. [Google Scholar] [CrossRef]
- Pfirrmann, V.; Oelsner, S.; Rettinger, E.; Huenecke, S.; Bonig, H.; Merker, M.; Wels, W.S.; Cinatl, J.; Schubert, R.; Klingebiel, T.; et al. Cytomegalovirus-specific cytokine-induced killer cells: Concurrent targeting of leukemia and cytomegalovirus. Cytotherapy 2015, 17, 1139–1151. [Google Scholar] [CrossRef]
- Lee, M.; Park, C.; Woo, J.; Kim, J.; Kho, I.; Nam, D.-H.; Park, W.-Y.; Kim, Y.-S.; Kong, D.-S.; Lee, H.W.; et al. Preferential Infiltration of Unique Vγ9Jγ2-Vδ2 T Cells Into Glioblastoma Multiforme. Front. Immunol. 2019, 10. [Google Scholar] [CrossRef]
- Chen, G.; Yang, X.; Ko, A.; Sun, X.; Gao, M.; Zhang, Y.; Shi, A.; Mariuzza, R.A.; Weng, N.P. Sequence and Structural Analyses Reveal Distinct and Highly Diverse Human CD8(+) TCR Repertoires to Immunodominant Viral Antigens. Cell Rep. 2017, 19, 569–583. [Google Scholar] [CrossRef]
- Weile, J.; Streeck, B.; Muck, J.; Krebs, G.; Jakobus, K.-H.; Knabbe, C.; Weber, F. Severe cytomegalovirus-associated esophagitis in an immunocompetent patient after short-term steroid therapy. J. Clin. Microbiol. 2009, 47, 3031–3033. [Google Scholar] [CrossRef]
- Chan, A.; Bazerbachi, F.; Hanson, B.; Alraies, M.C.; Duran-Nelson, A. Cytomegalovirus hepatitis and pancreatitis in the immunocompetent. Ochsner J. 2014, 14, 295–299. [Google Scholar]
- Bedri, S.; Sultan, A.A.; Alkhalaf, M.; Al Moustafa, A.E.; Vranic, S. Epstein-Barr virus (EBV) status in colorectal cancer: A mini review. Hum. Vaccines Immunother. 2018. [Google Scholar] [CrossRef]
- Erkes, D.A.; Smith, C.J.; Wilski, N.A.; Caldeira-Dantas, S.; Mohgbeli, T.; Snyder, C.M. Virus-Specific CD8+ T Cells Infiltrate Melanoma Lesions and Retain Function Independently of PD-1 Expression. J. Immunol. 2017. [Google Scholar] [CrossRef]
- Bentz, G.L.; Yurochko, A.D. Human CMV infection of endothelial cells induces an angiogenic response through viral binding to EGF receptor and beta1 and beta3 integrins. Proc. Natl. Acad. Sci. USA 2008, 105, 5531–5536. [Google Scholar] [CrossRef]
- Heiske, A.; Roettger, Y.; Bacher, M. Cytomegalovirus upregulates vascular endothelial growth factor and its second cellular kinase domain receptor in human fibroblasts. Viral Immunol. 2012, 25, 360–367. [Google Scholar] [CrossRef]
- Muz, B.; de la Puente, P.; Azab, F.; Azab, A.K. The role of hypoxia in cancer progression, angiogenesis, metastasis, and resistance to therapy. Hypoxia 2015, 3, 83–92. [Google Scholar] [CrossRef]
- Datta, M.; Via, L.E.; Kamoun, W.S.; Liu, C.; Chen, W.; Seano, G.; Weiner, D.M.; Schimel, D.; England, K.; Martin, J.D.; et al. Anti-vascular endothelial growth factor treatment normalizes tuberculosis granuloma vasculature and improves small molecule delivery. Proc. Natl. Acad. Sci. USA 2015, 112, 1827–1832. [Google Scholar] [CrossRef]
- Dong, H.; Qian, D.; Wang, Y.; Meng, L.; Chen, D.; Ji, X.; Feng, W. Survivin expression and serum levels in pancreatic cancer. World J. Surg. Oncol. 2015, 13, 189. [Google Scholar] [CrossRef]
- Meng, Q.; Liu, Z.; Rangelova, E.; Poiret, T.; Ambati, A.; Rane, L.; Xie, S.; Verbeke, C.; Dodoo, E.; Del Chiaro, M.; et al. Expansion of Tumor-reactive T Cells From Patients With Pancreatic Cancer. J. Immunother. 2016, 39, 81–89. [Google Scholar] [CrossRef]
- Liu, Z.; Poiret, T.; Persson, O.; Meng, Q.; Rane, L.; Bartek, J., Jr.; Karbach, J.; Altmannsberger, H.M.; Illies, C.; Luo, X.; et al. NY-ESO-1- and survivin-specific T-cell responses in the peripheral blood from patients with glioma. Cancer Immunol. Immunother. CII 2018, 67, 237–246. [Google Scholar] [CrossRef]
- Kim, J.; Kim, A.R.; Shin, E.C. Cytomegalovirus Infection and Memory T Cell Inflation. Immune Netw. 2015, 15, 186–190. [Google Scholar] [CrossRef] [PubMed]
- Makler, O.; Oved, K.; Netzer, N.; Wolf, D.; Reiter, Y. Direct visualization of the dynamics of antigen presentation in human cells infected with cytomegalovirus revealed by antibodies mimicking TCR specificity. Eur. J. Immunol. 2010, 40, 1552–1565. [Google Scholar] [CrossRef]
- Luo, X.H.; Meng, Q.; Rao, M.; Liu, Z.; Paraschoudi, G.; Dodoo, E.; Maeurer, M. The impact of inflationary cytomegalovirus-specific memory T cells on anti-tumour immune responses in patients with cancer. Immunology 2018, 155, 294–308. [Google Scholar] [CrossRef]
- Antonioli, L.; Pacher, P.; Vizi, E.S.; Haskó, G. CD39 and CD73 in immunity and inflammation. Trends Mol. Med. 2013, 19, 355–367. [Google Scholar] [CrossRef] [PubMed]
- Simoni, Y.; Becht, E.; Fehlings, M.; Loh, C.Y.; Koo, S.L.; Teng, K.W.W.; Yeong, J.P.S.; Nahar, R.; Zhang, T.; Kared, H.; et al. Bystander CD8(+) T cells are abundant and phenotypically distinct in human tumour infiltrates. Nature 2018, 557, 575–579. [Google Scholar] [CrossRef]
- Baumann, N.S.; Torti, N.; Welten, S.P.M.; Barnstorf, I.; Borsa, M.; Pallmer, K.; Oduro, J.D.; Cicin-Sain, L.; Ikuta, K.; Ludewig, B.; et al. Tissue maintenance of CMV-specific inflationary memory T cells by IL-15. PLoS Pathog. 2018, 14, e1006993. [Google Scholar] [CrossRef] [PubMed]
- Waldmann, T.A. Interleukin-15 in the treatment of cancer. Expert Rev. Clin. Immunol. 2014, 10, 1689–1701. [Google Scholar] [CrossRef] [PubMed]
- Hu, Q.; Ye, X.; Qu, X.; Cui, D.; Zhang, L.; Xu, Z.; Wan, H.; Zhang, L.; Tao, W. Discovery of a novel IL-15 based protein with improved developability and efficacy for cancer immunotherapy. Sci. Rep. 2018, 8, 7675. [Google Scholar] [CrossRef] [PubMed]
- Conlon, K.C.; Lugli, E.; Welles, H.C.; Rosenberg, S.A.; Fojo, A.T.; Morris, J.C.; Fleisher, T.A.; Dubois, S.P.; Perera, L.P.; Stewart, D.M.; et al. Redistribution, hyperproliferation, activation of natural killer cells and CD8 T cells, and cytokine production during first-in-human clinical trial of recombinant human interleukin-15 in patients with cancer. J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 2015, 33, 74–82. [Google Scholar] [CrossRef] [PubMed]
- Bahador, M.; Gras Navarro, A.; Rahman, M.A.; Dominguez-Valentin, M.; Sarowar, S.; Ulvestad, E.; Njolstad, G.; Lie, S.A.; Kristoffersen, E.K.; Bratland, E.; et al. Increased infiltration and tolerised antigen-specific CD8(+) TEM cells in tumor but not peripheral blood have no impact on survival of HCMV(+) glioblastoma patients. Oncoimmunology 2017, 6, e1336272. [Google Scholar] [CrossRef] [PubMed]
- Prado-Garcia, H.; Romero-Garcia, S.; Puerto-Aquino, A.; Rumbo-Nava, U. The PD-L1/PD-1 pathway promotes dysfunction, but not “exhaustion”, in tumor-responding T cells from pleural effusions in lung cancer patients. Cancer Immunol. Immunother. CII 2017, 66, 765–776. [Google Scholar] [CrossRef] [PubMed]
- Scheper, W.; Kelderman, S.; Fanchi, L.F.; Linnemann, C.; Bendle, G.; de Rooij, M.A.J.; Hirt, C.; Mezzadra, R.; Slagter, M.; Dijkstra, K.; et al. Low and variable tumor reactivity of the intratumoral TCR repertoire in human cancers. Nat. Med. 2019, 25, 89–94. [Google Scholar] [CrossRef] [PubMed]
- Legut, M.; Dolton, G.; Mian, A.A.; Ottmann, O.G.; Sewell, A.K. CRISPR-mediated TCR replacement generates superior anticancer transgenic T cells. Blood 2018, 131, 311–322. [Google Scholar] [CrossRef]
- Abken, H.; Wels, W.S.; Kühlcke, K. The Express Drivers: Chimeric Antigen Receptor-redirected T cells Make it to the Clinic. In Cancer Immunotherapy Meets Oncology: In Honor of Christoph Huber; Britten, C., Kreiter, S., Diken, M., Rammensee, H.-G., Eds.; Springers: Berlin/Heidelberg, Germany, 2014; p. 248. [Google Scholar]
- Liu, Z.; Meng, Q.; Bartek, J.; Poiret, T.; Persson, O.; Rane, L.; Rangelova, E.; Illies, C.; Peredo, I.; Luo, X.; et al. Tumor-infiltrating T-cells (TIL) from patients with glioma. Oncoimmunology 2016, 6, e1252894. [Google Scholar] [CrossRef] [PubMed]
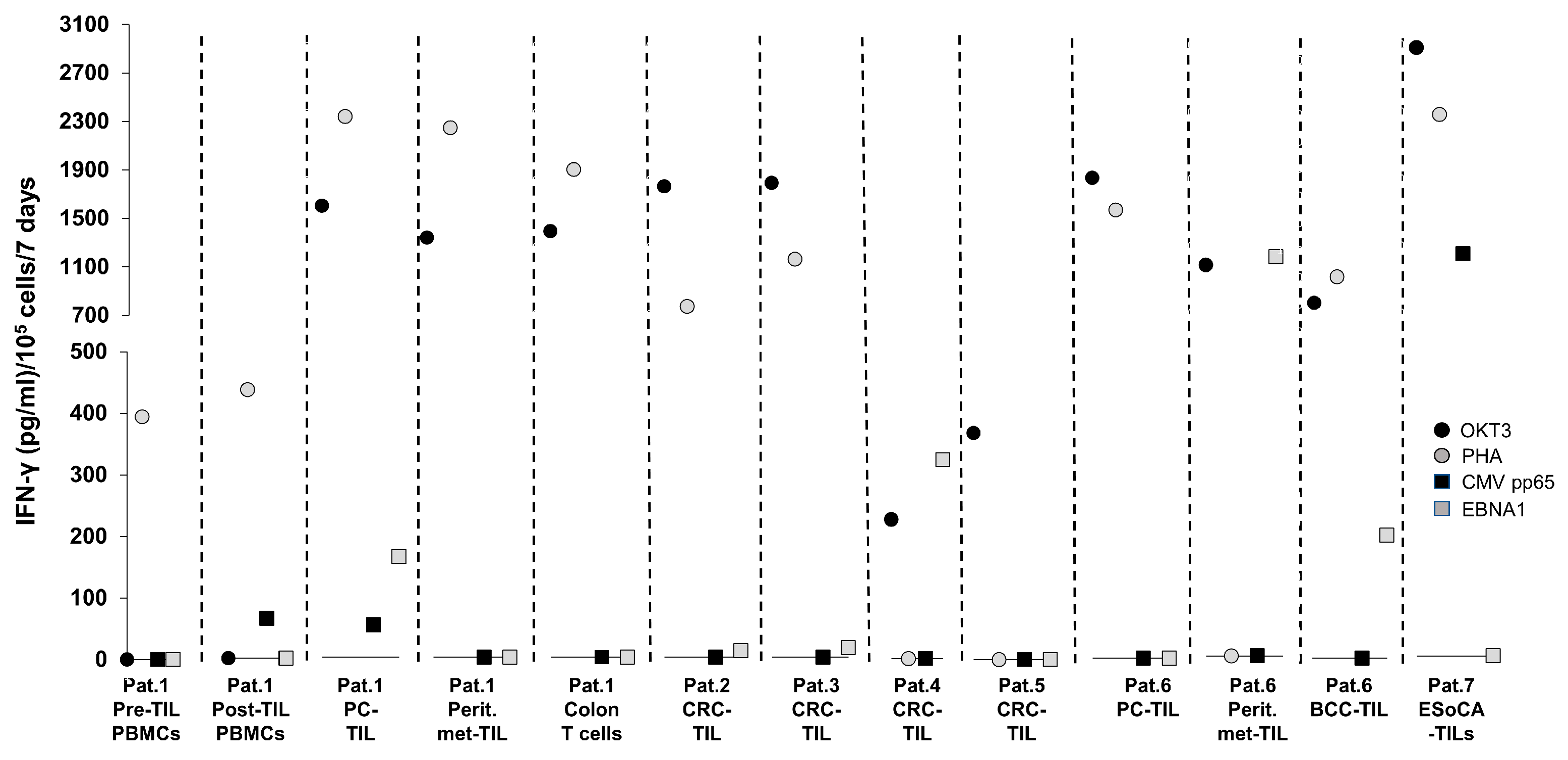
| Patient | Description | Samples Tested for CMV/EBV Reactivity |
|---|---|---|
| 1 | Metastatic pancreatic cancer (PC), peritoneal spread. Patient received TIL therapy. A colon biopsy was taken due to a clostridium difficile infection one month post-TIL therapy. PBMCs were sampled before and after TIL infusion. |
|
| 2 | Colorectal cancer (CRC) | TIL from CRC tissue |
| 3 | CRC | TIL from CRC tissue |
| 4 | CRC | TIL from CRC tissue |
| 5 | CRC | TIL from CRC tissue |
| 6 | Metastatic pancreatic cancer which spread to the peritoneum. Patient also had basal-cell carcinoma (BCC/basalioma). |
|
| 7 | Oesophageal cancer (EsoCA) | TIL from EsoCA tissue |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lérias, J.R.; Paraschoudi, G.; Silva, I.; Martins, J.; de Sousa, E.; Condeço, C.; Figueiredo, N.; Carvalho, C.; Dodoo, E.; Jäger, E.; et al. Clinically Relevant Immune Responses against Cytomegalovirus: Implications for Precision Medicine. Int. J. Mol. Sci. 2019, 20, 1986. https://doi.org/10.3390/ijms20081986
Lérias JR, Paraschoudi G, Silva I, Martins J, de Sousa E, Condeço C, Figueiredo N, Carvalho C, Dodoo E, Jäger E, et al. Clinically Relevant Immune Responses against Cytomegalovirus: Implications for Precision Medicine. International Journal of Molecular Sciences. 2019; 20(8):1986. https://doi.org/10.3390/ijms20081986
Chicago/Turabian StyleLérias, Joana R., Georgia Paraschoudi, Inês Silva, João Martins, Eric de Sousa, Carolina Condeço, Nuno Figueiredo, Carlos Carvalho, Ernest Dodoo, Elke Jäger, and et al. 2019. "Clinically Relevant Immune Responses against Cytomegalovirus: Implications for Precision Medicine" International Journal of Molecular Sciences 20, no. 8: 1986. https://doi.org/10.3390/ijms20081986
APA StyleLérias, J. R., Paraschoudi, G., Silva, I., Martins, J., de Sousa, E., Condeço, C., Figueiredo, N., Carvalho, C., Dodoo, E., Jäger, E., Rao, M., & Maeurer, M. (2019). Clinically Relevant Immune Responses against Cytomegalovirus: Implications for Precision Medicine. International Journal of Molecular Sciences, 20(8), 1986. https://doi.org/10.3390/ijms20081986






