A Novel Standardized Cannabis sativa L. Extract and Its Constituent Cannabidiol Inhibit Human Polymorphonuclear Leukocyte Functions
Abstract
1. Introduction
2. Results
2.1. Effect of CM5 and Cannabidiol (CBD) on Cell Viability
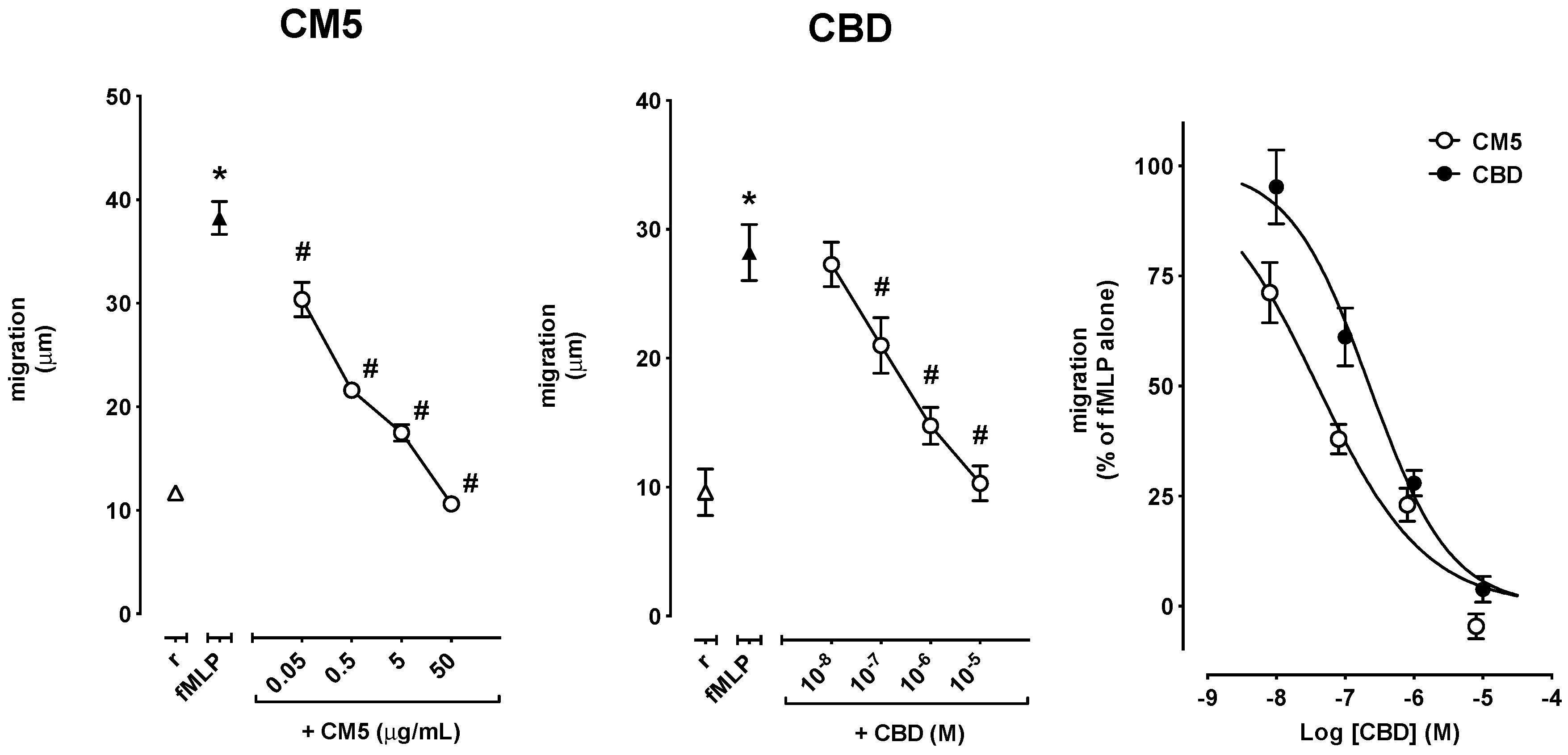
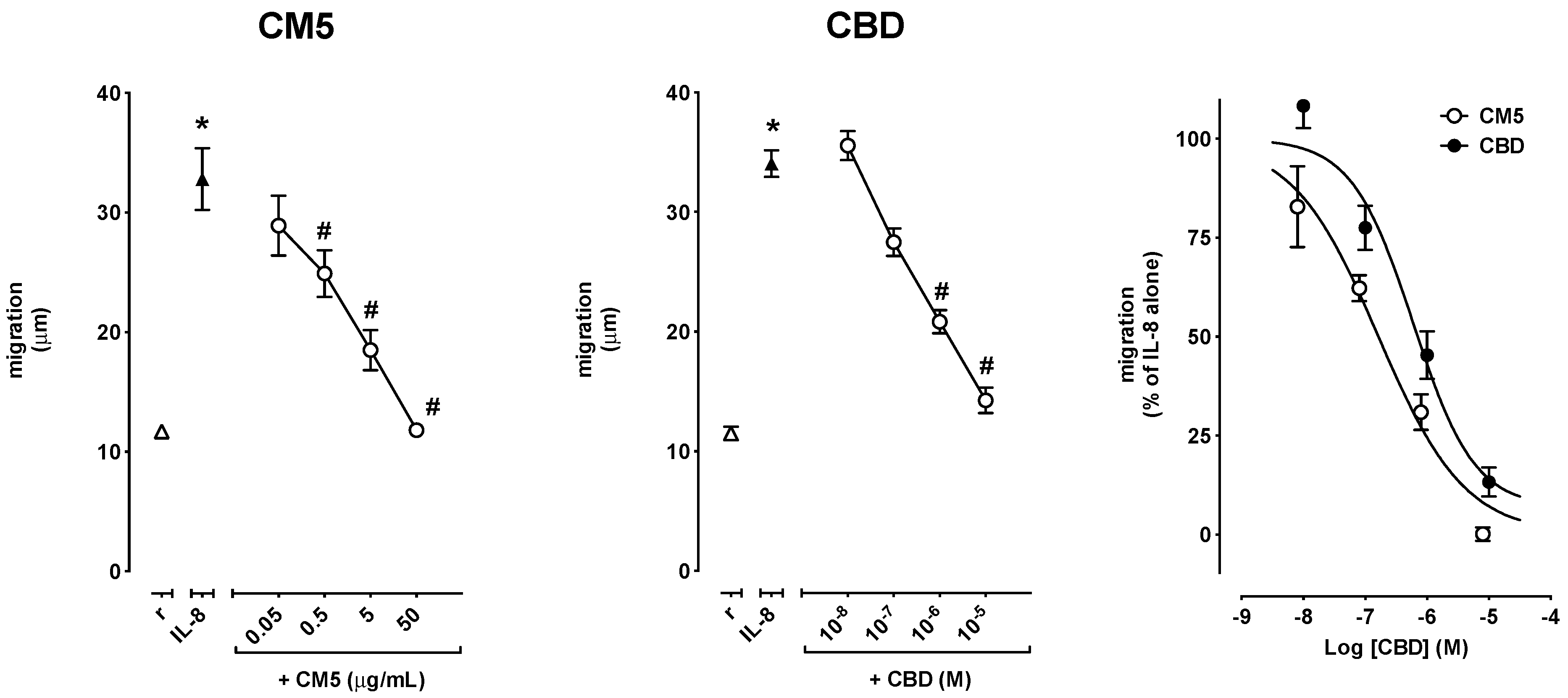
2.2. Effect of CM5 and CBD on Cell Migration
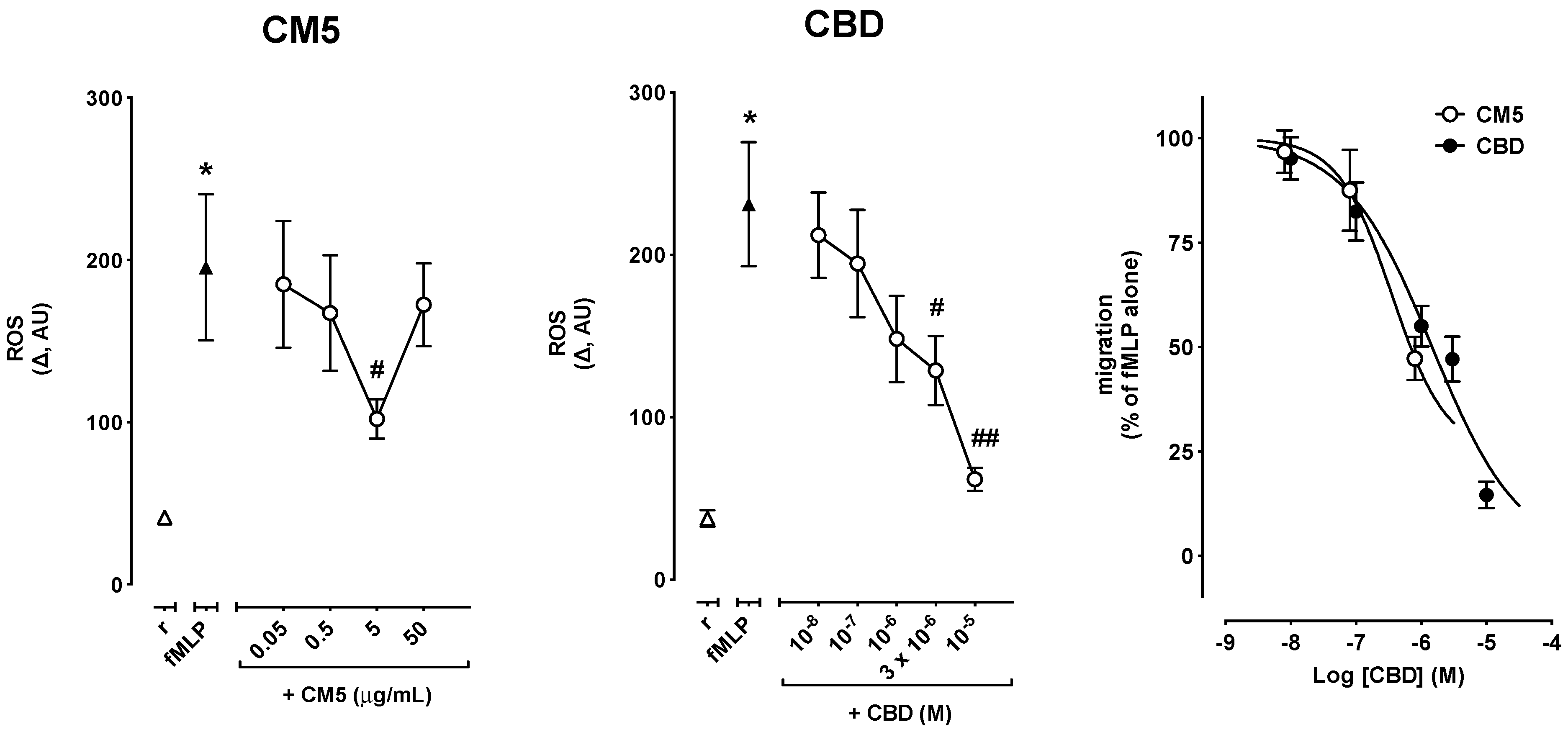
2.3. Effects of CM5 and CBD on Reactive Oxygen Species (ROS) Production
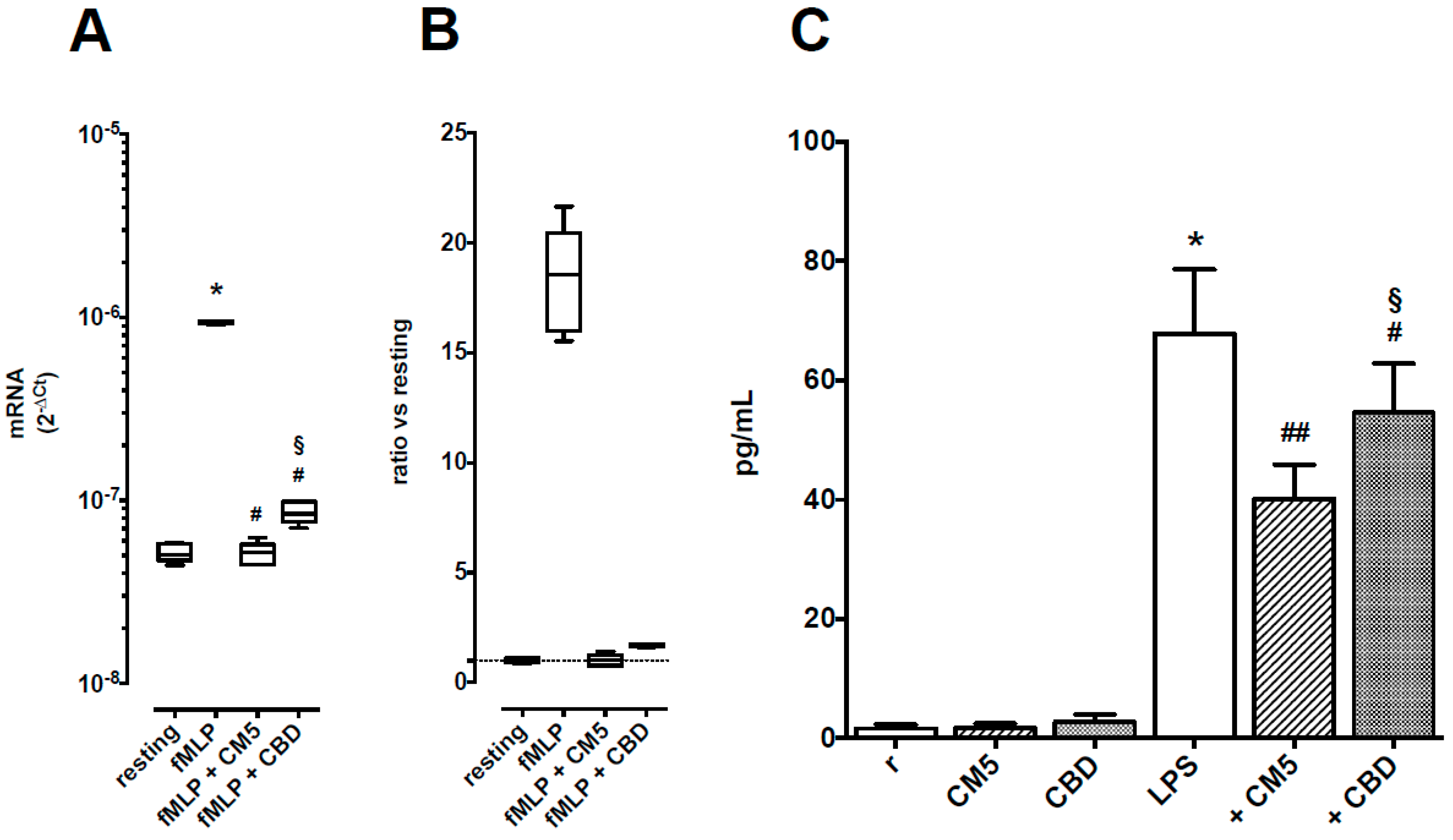
2.4. Effects of CM5 and CBD on TNF-α Production
3. Discussion
4. Materials and Methods
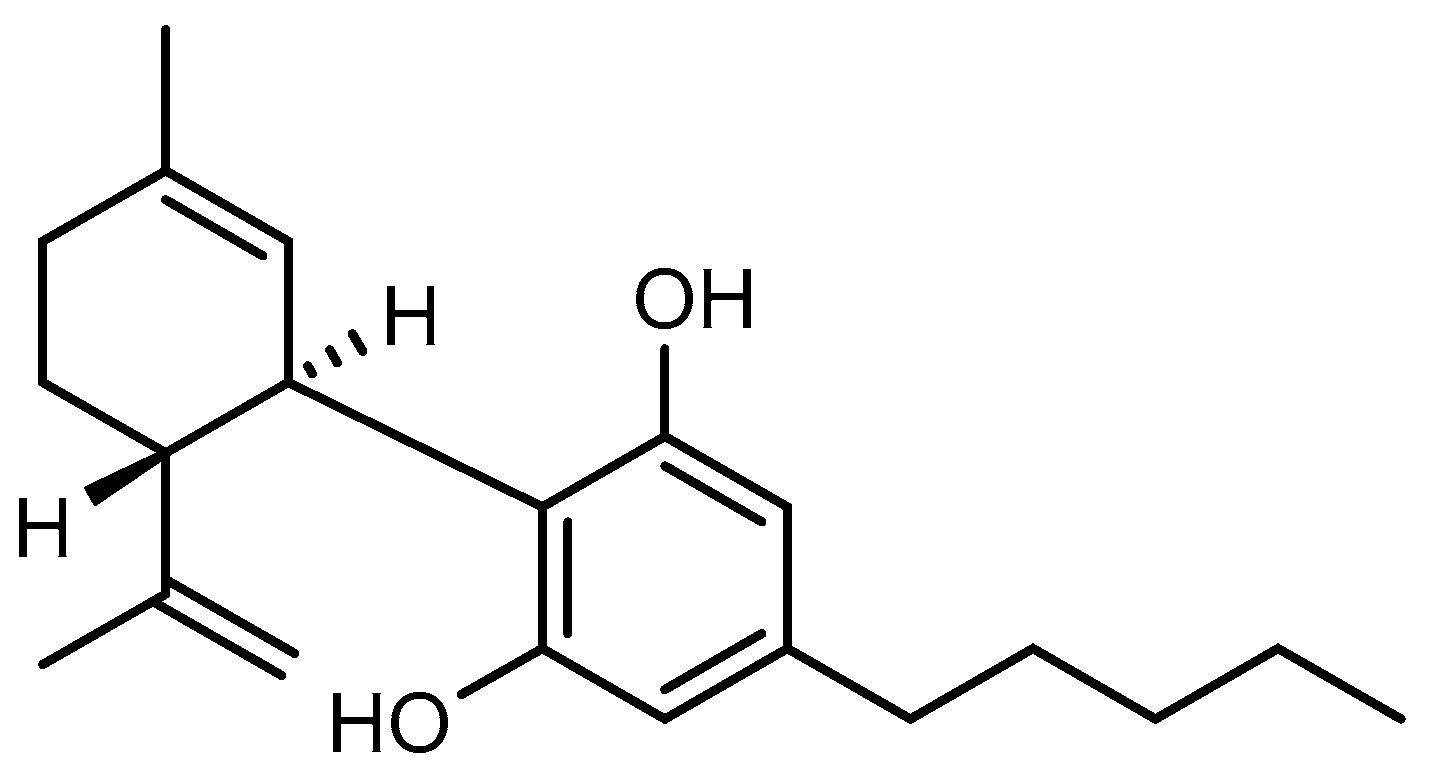
4.1. Test Substances
4.2. Isolation of Human Polymorphonuclear Leukocytes (PMN)
4.3. Cytotoxicity Assays
4.4. Cell Migration Assay
4.5. Reactive Oxygen Species (ROS) Production Assay
4.6. Measurement of TNF-α Production
4.7. Statistical Analysis
Supplementary Materials
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Zlas, J.; Stark, H.; Seligman, J.; Levy, R.; Werker, E.; Breuer, A.; Mechoulam, R. Early medical use of cannabis. Nature 1993, 363, 215. [Google Scholar] [CrossRef] [PubMed]
- Abrams, D.I. The therapeutic effects of Cannabis and cannabinoids: An update from the National Academies of Sciences, Engineering and Medicine report. Eur. J. Intern. Med. 2018, 49, 7–11. [Google Scholar] [CrossRef] [PubMed]
- Mechoulam, R.; Gaoni, Y. The absolute configuration of delta-1-tetrahydrocannabinol, the major active constituent of hashish. Tetrahedron. Lett. 1967, 12, 1109–1111. [Google Scholar] [CrossRef]
- European Monitoring Centre for Drugs and Drug Addiction. Cannabis Legislation in Europe: An Overview. 2018. Available online: www.emcdda.europa.eu (accessed on 7 November 2018).
- Andre, C.M.; Hausman, J.-F.; Guerriero, G. Cannabis sativa: The Plant of the Thousand and One Molecules. Front. Plant Sci. 2016, 7, 19. [Google Scholar]
- Babalonis, S.; Haney, M.; Malcolm, R.J.; Lofwall, M.R.; Votaw, V.R.; Sparenborg, S.; Walsh, S.L. Oral cannabidiol does not produce a signal for abuse liability in frequent marijuana smokers. Drug Alcohol. Depend 2017, 172, 9–13. [Google Scholar] [CrossRef]
- Bergamaschi, M.M.; Queiroz, R.H.C.; Zuardi, A.W.; Crippa, J.A.S. Safety and side effects of cannabidiol, a Cannabis sativa constituent. Curr. Drug Saf. 2011, 6, 237–249. [Google Scholar] [CrossRef]
- Iffland, K.; Grotenhermen, F. An Update on Safety and Side Effects of Cannabidiol: A Review of Clinical Data and Relevant Animal Studies. Cannabis Cannabinoid Res. 2017, 2, 139–154. [Google Scholar] [CrossRef]
- Taylor, L.; Gidal, B.; Blakey, G.; Tayo, B.; Morrison, G. A Phase, I, Randomized, Double-Blind, Placebo-Controlled, Single Ascending Dose, Multiple Dose, and Food Effect Trial of the Safety, Tolerability and Pharmacokinetics of Highly Purified Cannabidiol in Healthy Subjects. CNS Drugs 2018, 32, 1053–1067. [Google Scholar] [CrossRef]
- Chen, J.W.; Borgelt, L.M.; Blackmer, A.B. Epidiolex (Cannabidiol): A New Hope for Patients With Dravet or Lennox-Gastaut Syndromes. Ann. Pharmacother. 2019. [Google Scholar] [CrossRef] [PubMed]
- Pisanti, S.; Malfitano, A.M.; Ciaglia, E.; Lamberti, A.; Ranieri, R.; Cuomo, G.; Abate, M.; Faggiana, G.; Proto, M.C.; Fiore, D.; et al. Cannabidiol: State of the art and new challenges for therapeutic applications. Pharmacol. Ther. 2017, 175, 133–150. [Google Scholar] [CrossRef]
- Burstein, S. Cannabidiol (CBD) and its analogs: A review of their effects on inflammation. Bioorg. Med. Chem. 2015, 23, 1377–1385. [Google Scholar] [CrossRef]
- Kolaczkowska, E.; Kubes, P. Neutrophil recruitment and function in health and inflammation. Nat. Rev. Immunol. 2013, 13, 159–175. [Google Scholar] [CrossRef]
- Hallett, M.B.; Lloyds, D. Neutrophil priming: The cellular signals that say ‘amber’ but not ‘green’. Immunol. Today 1995, 16, 264–268. [Google Scholar] [CrossRef]
- Mortaz, E.; Alipoor, S.D.; Adcock, I.M.; Mumby, S.; Koenderman, L. Update on Neutrophil Function in Severe Inflammation. Front. Immunol. 2018, 9, 2171. [Google Scholar] [CrossRef]
- Perretti, M.; Di Filippo, C.; D’Amico, M.; Dalli, J. Characterizing the anti-inflammatory and tissue protective actions of a novel Annexin A1 peptide. PLoS ONE 2017, 12, e0175786. [Google Scholar] [CrossRef]
- Khajah, M.A. Role of Neutrophils in Disease Pathogenesis [Internet]; Khajah, M.A., Ed.; InTech: Rijeka, Croatia, 2017. [Google Scholar]
- Baban, B.; Hoda, N.; Malik, A.; Khodadadi, H.; Simmerman, E.; Vaibhav, K.; Mozaffari, M.S. Impact of cannabidiol treatment on regulatory T-17 cells and neutrophil polarization in acute kidney injury. Am. J. Physiol. 2018, 315, F1149–F1158. [Google Scholar] [CrossRef]
- Hayakawa, K.; Mishima, K.; Nozako, M.; Hazekawa, M.; Irie, K.; Fujioka, M.; Orito, K.; Abe, K.; Hasebe, N.; Egashira, N.; et al. Delayed treatment with cannabidiol has a cerebroprotective action via a cannabinoid receptor-independent myeloperoxidase-inhibiting mechanism. J. Neurochem. 2007, 102, 1488–1496. [Google Scholar] [CrossRef]
- Wang, Y.; Mukhopadhyay, P.; Cao, Z.; Wang, H.; Feng, D.; Haskó, G.; Mechoulam, R.; Gao, B.; Pacher, P. Cannabidiol attenuates alcohol-induced liver steatosis, metabolic dysregulation, inflammation and neutrophil-mediated injury. Sci Rep. 2017, 7, 12064. [Google Scholar] [CrossRef]
- Mukhopadhyay, P.; Rajesh, M.; Horváth, B.; Bátkai, S.; Park, O.; Tanchian, G.; Gao, R.Y.; Patel, V.; Wink, D.A.; Liaudet, L.; et al. Cannabidiol protects against hepatic ischemia/reperfusion injury by attenuating inflammatory signaling and response, oxidative/nitrative stress, and cell death. Free Radic. Biol. Med. 2011, 50, 1368–1381. [Google Scholar] [CrossRef]
- McHugh, D.; Tanner, C.; Mechoulam, R.; Pertwee, R.G.; Ross, R.A. Inhibition of Human Neutrophil Chemotaxis by Endogenous Cannabinoids and Phytocannabinoids: Evidence for a Site Distinct from CB1 and CB2. Mol. Pharmacol. 2007, 73, 441–450. [Google Scholar] [CrossRef]
- Devinsky, O.; Cross, J.H.; Laux, L.; Marsh, E.; Miller, I.; Nabbout, R.; Scheffer, I.E.; Thiele, E.A.; Wright, S. Trial of Cannabidiol for Drug-Resistant Seizures in the Dravet Syndrome. N. Engl. J. Med. 2017, 376, 2011–2020. [Google Scholar] [CrossRef]
- Kapellos, T.S.; Taylor, L.; Feuerborn, A.; Valaris, S.; Hussain, M.T.; Rainger, G.E.; Greaves, D.R.; Iqbal, A.J. Cannabinoid receptor 2 deficiency exacerbates inflammation and neutrophil recruitment. FASEB J. 2019. [Google Scholar] [CrossRef] [PubMed]
- Kurihara, R.; Tohyama, Y.; Matsusaka, S.; Naruse, H.; Kinoshita, E.; Tsujioka, T.; Katsumata, Y.; Yamamura, H. Effects of peripheral cannabinoid receptor ligands on motility and polarization in neutrophil-like HL60 cells and human neutrophils. J. Biol. Chem. 2006, 281, 12908–12918. [Google Scholar] [CrossRef]
- Balenga, N.A.B.; Aflaki, E.; Kargl, J.; Platzer, W.; Schröder, R.; Blättermann, S.; Kostenis, E.; Brown, A.J.; Heinemann, A.; Waldhoer, M. GPR55 regulates cannabinoid 2 receptor-mediated responses in human neutrophils. Nat. Publ. Gr. 2011, 21. [Google Scholar] [CrossRef]
- Reddy, R.C.; Narala, V.R.; Keshamouni, V.G.; Milam, J.E.; Newstead, M.W.; Standiford, T.J. Sepsis-induced inhibition of neutrophil chemotaxis is mediated by activation of peroxisome proliferator-activated receptor-{gamma}. Blood 2008, 112, 4250–4258. [Google Scholar] [CrossRef] [PubMed]
- Heiner, I.; Eisfeld, J.; Halaszovich, C.R.; Wehage, E.; Jüngling, E.; Zitt, C.; Lückhoff, A. Expression profile of the transient receptor potential (TRP) family in neutrophil granulocytes: Evidence for currents through long TRP channel 2 induced by ADP-ribose and NAD. Biochem. J. 2003, 371 Pt 3, 1045–1053. [Google Scholar] [CrossRef]
- Lehrer, R.I.; Ganz, T. Antimicrobial peptides in mammalian and insect host defence. Curr. Opin. Immunol. 1999, 11, 23–27. [Google Scholar] [CrossRef]
- Segal, A.W. How neutrophils kill microbes. Annu. Rev. Immunol. 2005, 23, 197–223. [Google Scholar] [CrossRef]
- Brinkmann, V.; Reichard, U.; Goosmann, C.; Fauler, B.; Uhlemann, Y.; Weiss, D.S.; Weinrauch, Y.; Zychlinsky, A. Neutrophil extracellular traps kill bacteria. Science 2004, 303, 1532–1535. [Google Scholar] [CrossRef]
- Angulo, J.; Martínez-Valdebenito, C.; Marco, C.; Galeno, H.; Villagra, E.; Vera, L.; Lagos, N.; Becerra, N.; Mora, J.; Bermúdez, A.; et al. Serum levels of interleukin-6 are linked to the severity of the disease caused by Andes Virus. PLoS Negl. Trop. Dis. 2017, 11, e0005757. [Google Scholar] [CrossRef]
- Cantin, A.M.; Hartl, D.; Konstan, M.W.; Chmiel, J.F. Inflammation in cystic fibrosis lung disease: Pathogenesis and therapy. J. Cyst. Fibros. 2015, 14, 419–430. [Google Scholar] [CrossRef]
- Tsukamoto, T.; Savanh Chanthaphavong, R.; Pape, H.-C. Current theories on the pathophysiology of multiple organ failure after trauma. Inj. Int. J. Care Inj. 2010, 41, 21–26. [Google Scholar] [CrossRef]
- Wright, H.L.; Moots, R.J.; Edwards, S.W. The multifactorial role of neutrophils in rheumatoid arthritis. Nat. Rev. Rheumatol. 2014, 10, 593–601. [Google Scholar] [CrossRef]
- Yoshida, S.; Hagiwara, Y.; Tsuchiya, M.; Shinoda, M.; Koide, M.; Hatakeyama, H.; Chaweewannakorn, C.; Yano, T.; Sogi, Y.; Itaya, N.; et al. Involvement of neutrophils and interleukin-18 in nociception in a mouse model of muscle pain. Mol. Pain 2018, 14. [Google Scholar] [CrossRef]
- Woodberry, T.; Bouffler, S.E.; Wilson, A.S.; Buckland, R.L.; Brüstle, A. The Emerging Role of Neutrophil Granulocytes in Multiple Sclerosis. J. Clin. Med. 2018, 7, 511. [Google Scholar] [CrossRef]
- Huang, J.; Xiao, Y.; Xu, A.; Zhou, Z. Neutrophils in type 1 diabetes. J. Diabetes Investig. 2016, 7, 652–663. [Google Scholar] [CrossRef]
- Cao, M.; Yu, M.; Zhang, Y.; Tong, D.; Guo, L.; Wang, C.; Li, T.; Yang, X.; Zhang, C.; Kou, J.; et al. Neutrophil Extracellular Traps Exacerbate Inflammatory Responses and Thrombotic Tendency in Both a Murine Colitis Model and Patients with Inflammatory Bowel Disease. Blood 2017, 130 (Suppl. 1), 994. [Google Scholar]
- Gaul, D.S.; Stein, S.; Matter, C.M. Neutrophils in cardiovascular disease. Eur. Heart J. 2017, 38, 1702–1704. [Google Scholar] [CrossRef]
- Hudome, S.; Palmer, C.; Roberts, R.L.; Mauger, D.; Housman, C.; Towfighi, J. The Role of Neutrophils in the Production of Hypoxic-Ischemic Brain Injury in the Neonatal Rat. Pediatr. Res. 1997, 41, 607–616. [Google Scholar] [CrossRef]
- Powell, D.R.; Huttenlocher, A. Neutrophils in the Tumor Microenvironment. Trends Immunol. 2016, 37, 41–52. [Google Scholar] [CrossRef]
- Segal, A.W. The role of neutrophils in the pathogenesis of Crohn’s disease. Eur. J. Clin. Investig. 2018, 48, e12983. [Google Scholar] [CrossRef]
- Scanzano, A.; Schembri, L.; Rasini, E.; Luini, A.; Dallatorre, J.; Legnaro, M.; Bombelli, R.; Congiu, T.; Cosentino, M.; Marino, F. Adrenergic modulation of migration, CD11b and CD18 expression, ROS and interleukin-8 production by human polymorphonuclear leukocytes. Inflamm. Res. 2015, 64, 127–135. [Google Scholar] [CrossRef]
- Mosmann, T. Rapid colorimetric assay for cellular growth and survival: Application to proliferation and cytotoxicity assays. J. Immunol. Methods 1983, 65, 55–63. [Google Scholar] [CrossRef]
- Marino, F.; Scanzano, A.; Pulze, L.; Pinoli, M.; Rasini, E.; Luini, A.; Bombelli, R.; Legnaro, M.; de Eguileor, M.; Cosentino, M. β2-Adrenoceptors inhibit neutrophil extracellular traps in human polymorphonuclear leukocytes. J. Leukoc. Biol. 2018, 104, 603–614. [Google Scholar] [CrossRef]
- Cosentino, M.; Bombelli, R.; Carcano, E.; Luini, A.; Marino, F.; Crema, F.; Dajas, F.; Lecchini, S. Immunomodulatory properties of Achyrocline satureioides (Lam.) D.C. infusion: A study on human leukocytes. J. Ethnopharmacol. 2008, 116, 501–507. [Google Scholar] [CrossRef]

| PMN Functions | CM5 (Mean ± SEM) | CBD (Mean ± SEM) | p-Value |
|---|---|---|---|
| fMLP-induced migration | −7.401 ± 0.111 | −6.645 ± 0.106 | 0.0008 |
| IL-8-induced migration | −6.786 ± 0.124 | −6.225 ± 0.219 | 0.0648 |
| fMLP-induced ROS production | −6.449 ± 0.148 | −5.832 ± 0.089 | 0.0023 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mabou Tagne, A.; Marino, F.; Legnaro, M.; Luini, A.; Pacchetti, B.; Cosentino, M. A Novel Standardized Cannabis sativa L. Extract and Its Constituent Cannabidiol Inhibit Human Polymorphonuclear Leukocyte Functions. Int. J. Mol. Sci. 2019, 20, 1833. https://doi.org/10.3390/ijms20081833
Mabou Tagne A, Marino F, Legnaro M, Luini A, Pacchetti B, Cosentino M. A Novel Standardized Cannabis sativa L. Extract and Its Constituent Cannabidiol Inhibit Human Polymorphonuclear Leukocyte Functions. International Journal of Molecular Sciences. 2019; 20(8):1833. https://doi.org/10.3390/ijms20081833
Chicago/Turabian StyleMabou Tagne, Alex, Franca Marino, Massimiliano Legnaro, Alessandra Luini, Barbara Pacchetti, and Marco Cosentino. 2019. "A Novel Standardized Cannabis sativa L. Extract and Its Constituent Cannabidiol Inhibit Human Polymorphonuclear Leukocyte Functions" International Journal of Molecular Sciences 20, no. 8: 1833. https://doi.org/10.3390/ijms20081833
APA StyleMabou Tagne, A., Marino, F., Legnaro, M., Luini, A., Pacchetti, B., & Cosentino, M. (2019). A Novel Standardized Cannabis sativa L. Extract and Its Constituent Cannabidiol Inhibit Human Polymorphonuclear Leukocyte Functions. International Journal of Molecular Sciences, 20(8), 1833. https://doi.org/10.3390/ijms20081833






