Identification and Characterization of the DMRT11E Gene in the Oriental River Prawn Macrobrachium nipponense
Abstract
1. Introduction
2. Results
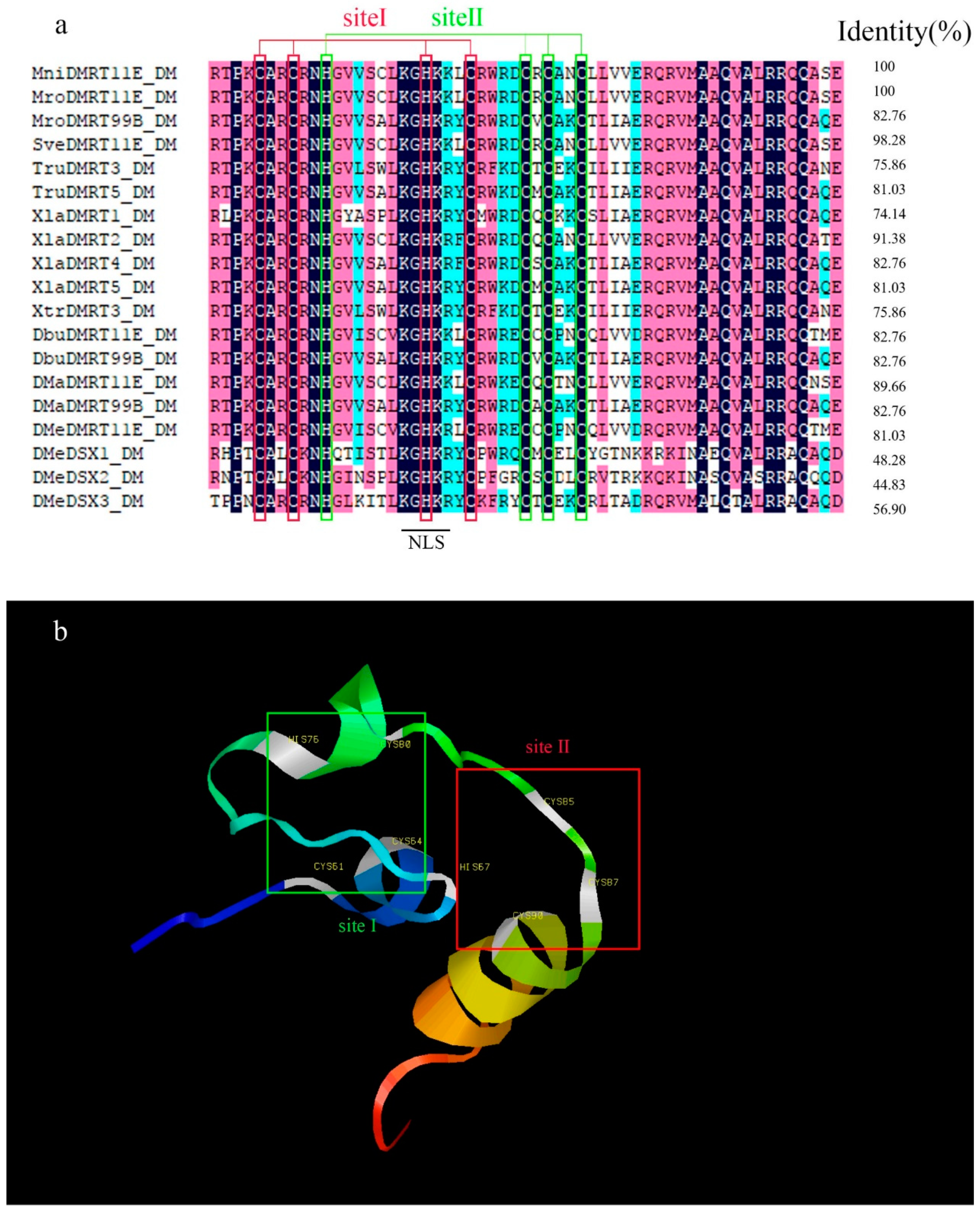
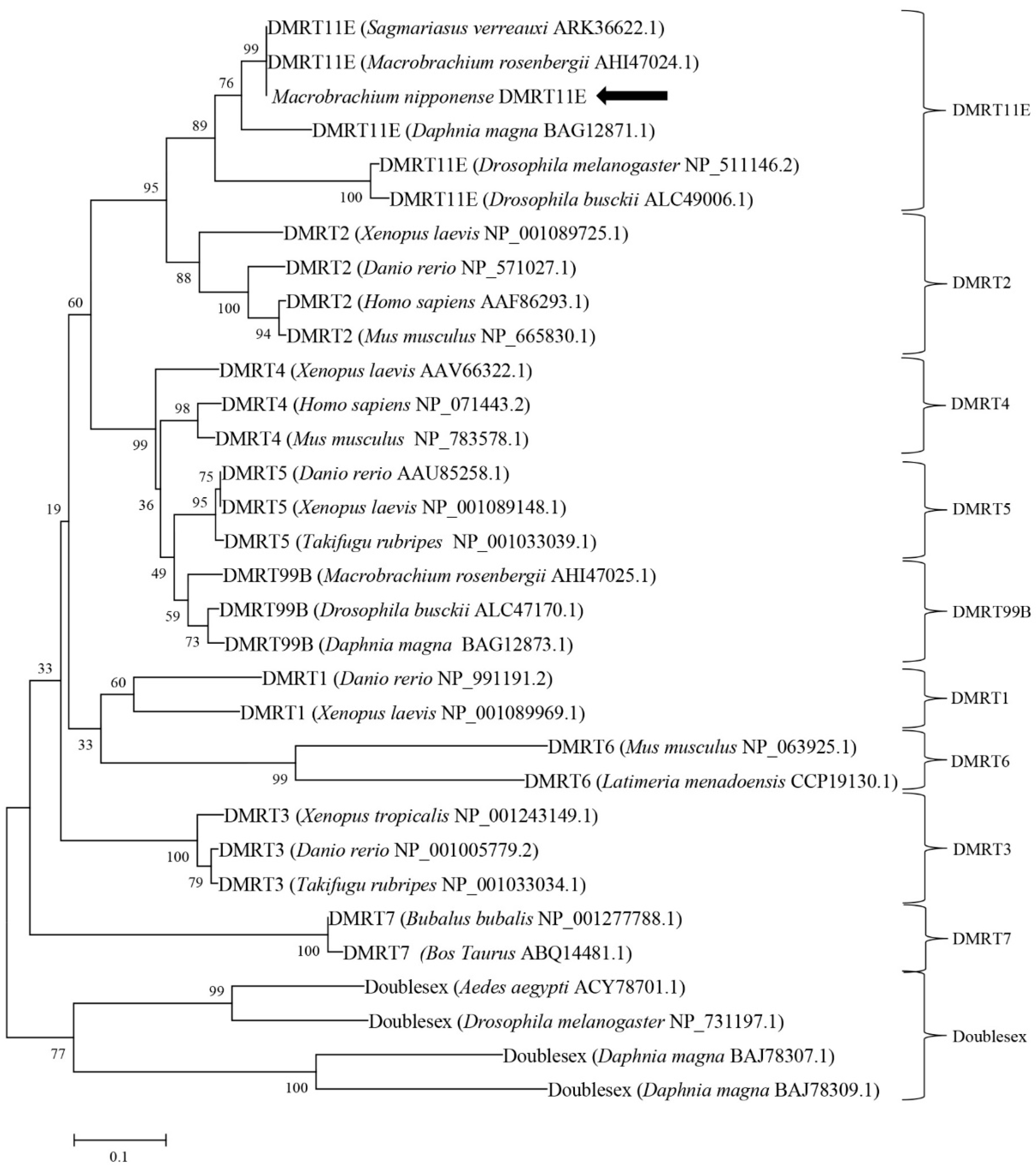
2.1. Molecular Cloning and Structural Analysis of DMRT11E Gene
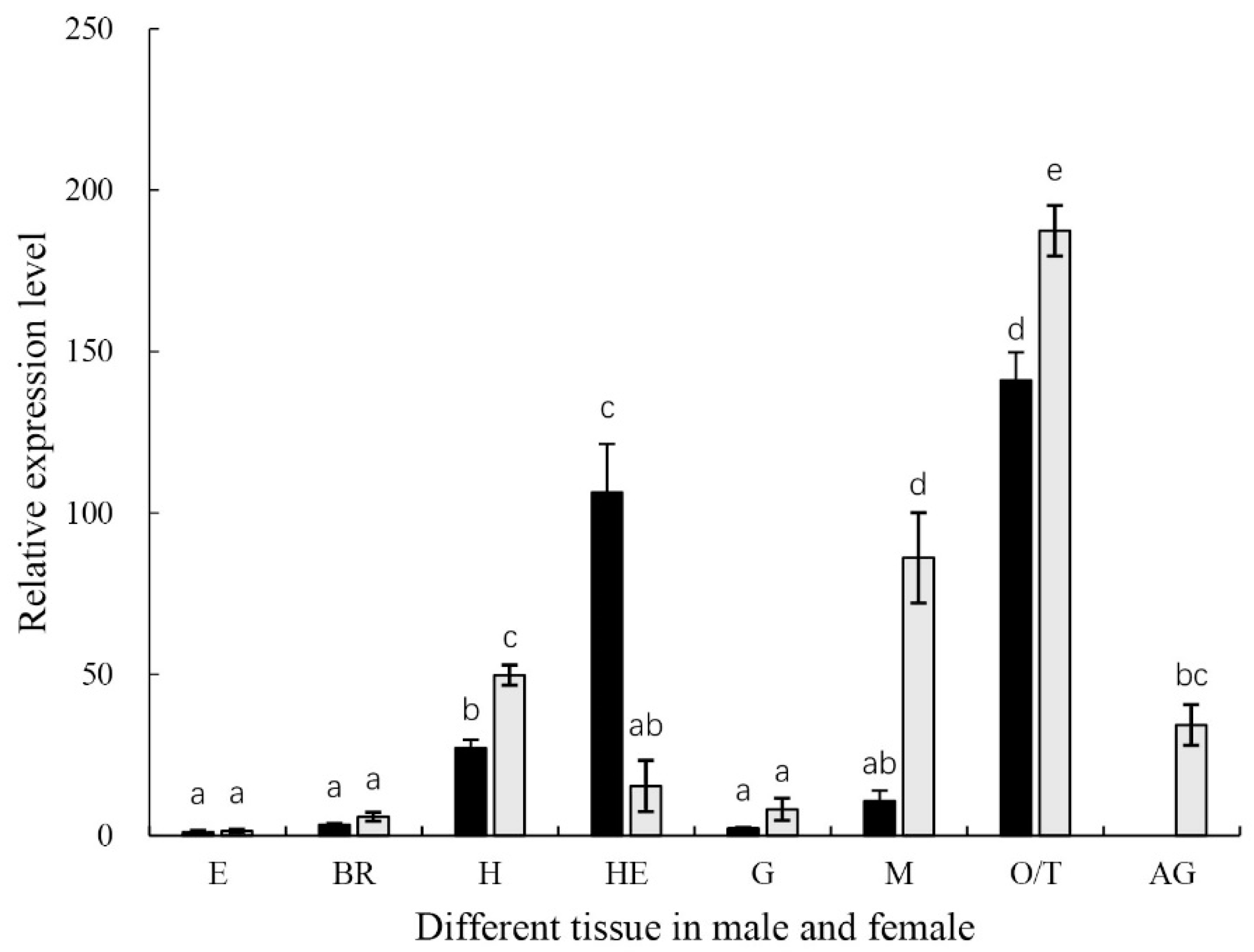
2.2. Tissue-Specific Expression Patterns of MniDMRT11E
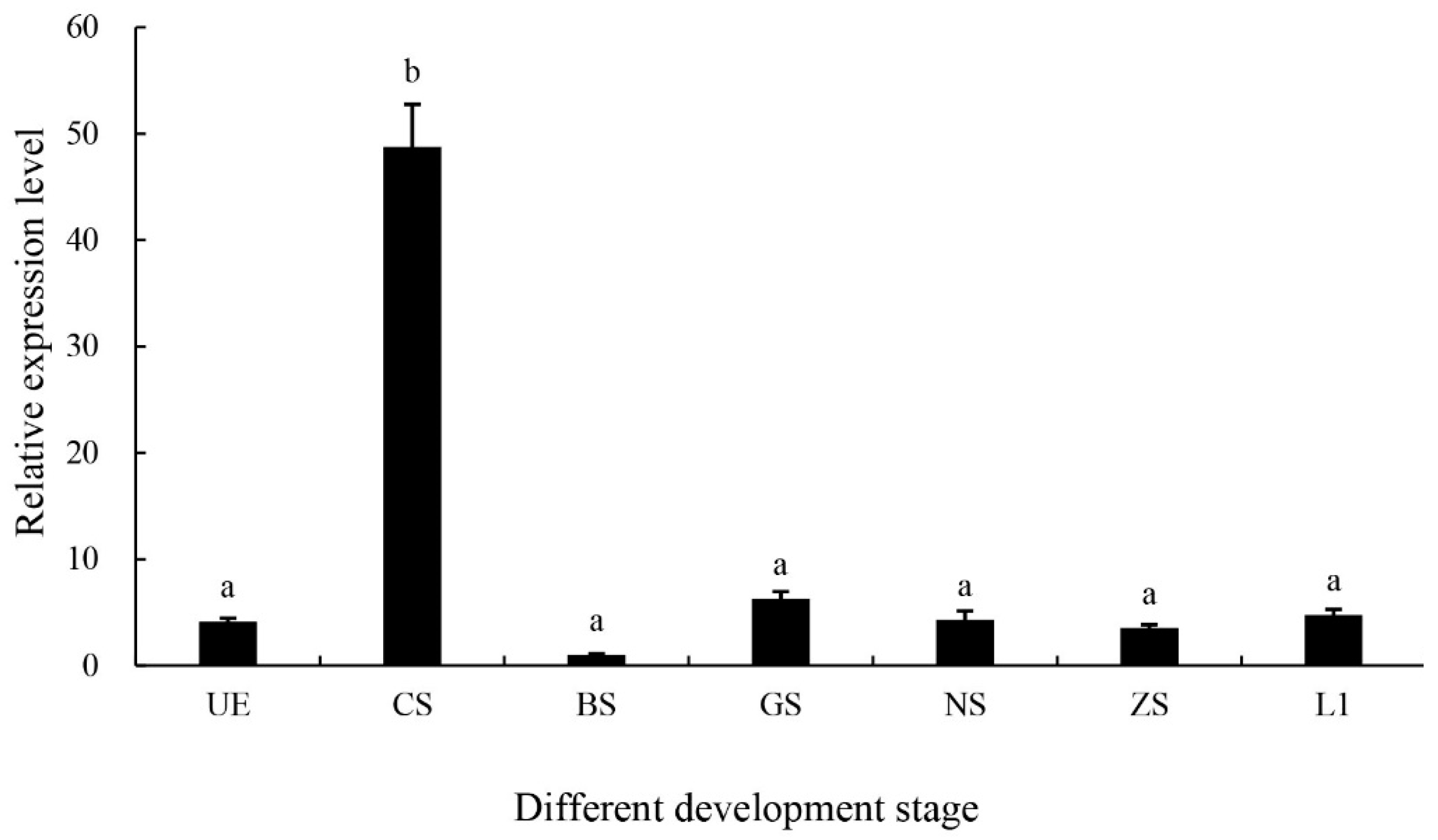
2.3. Expression of the MniDMRT11E Gene During Embryo Stages
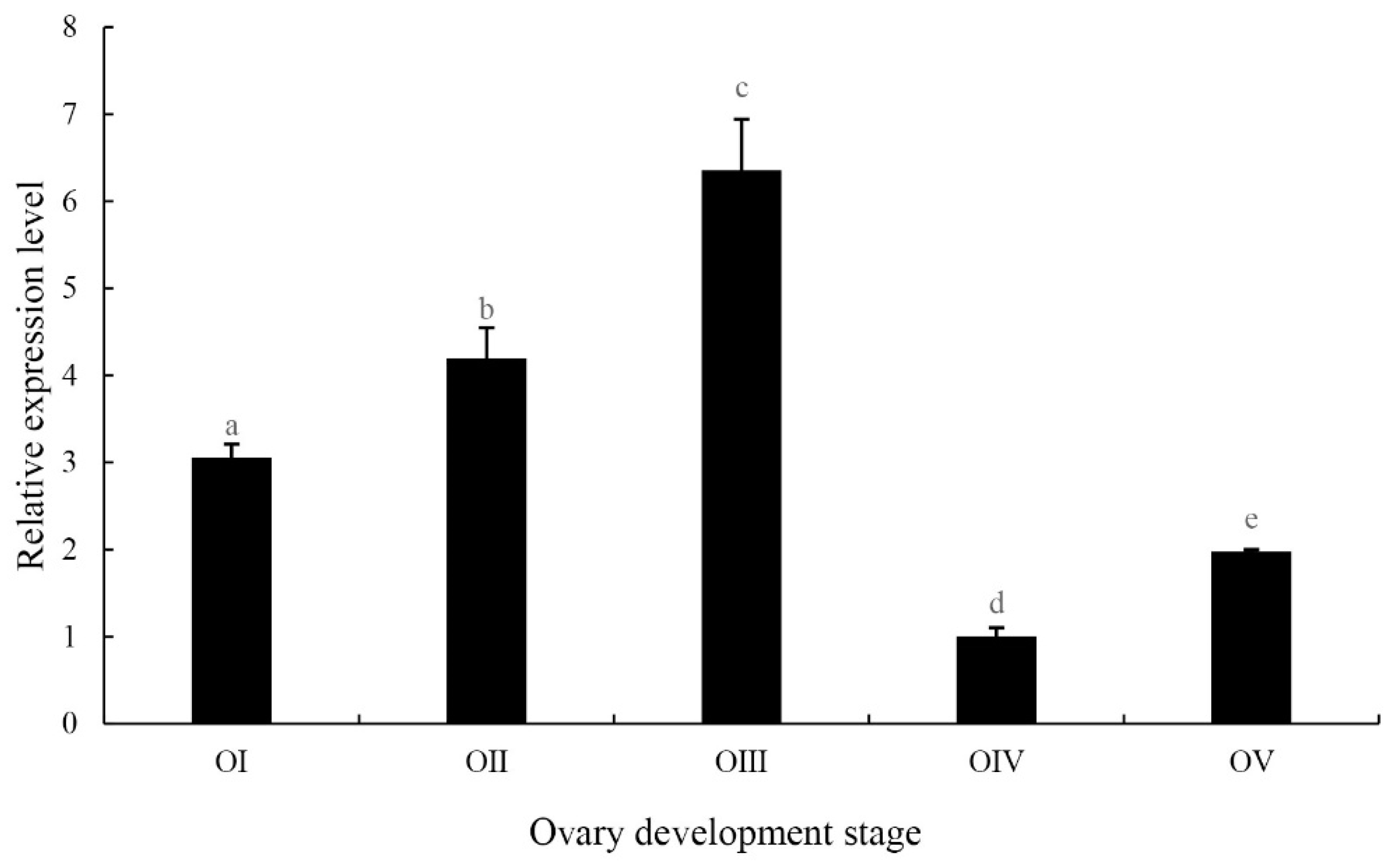
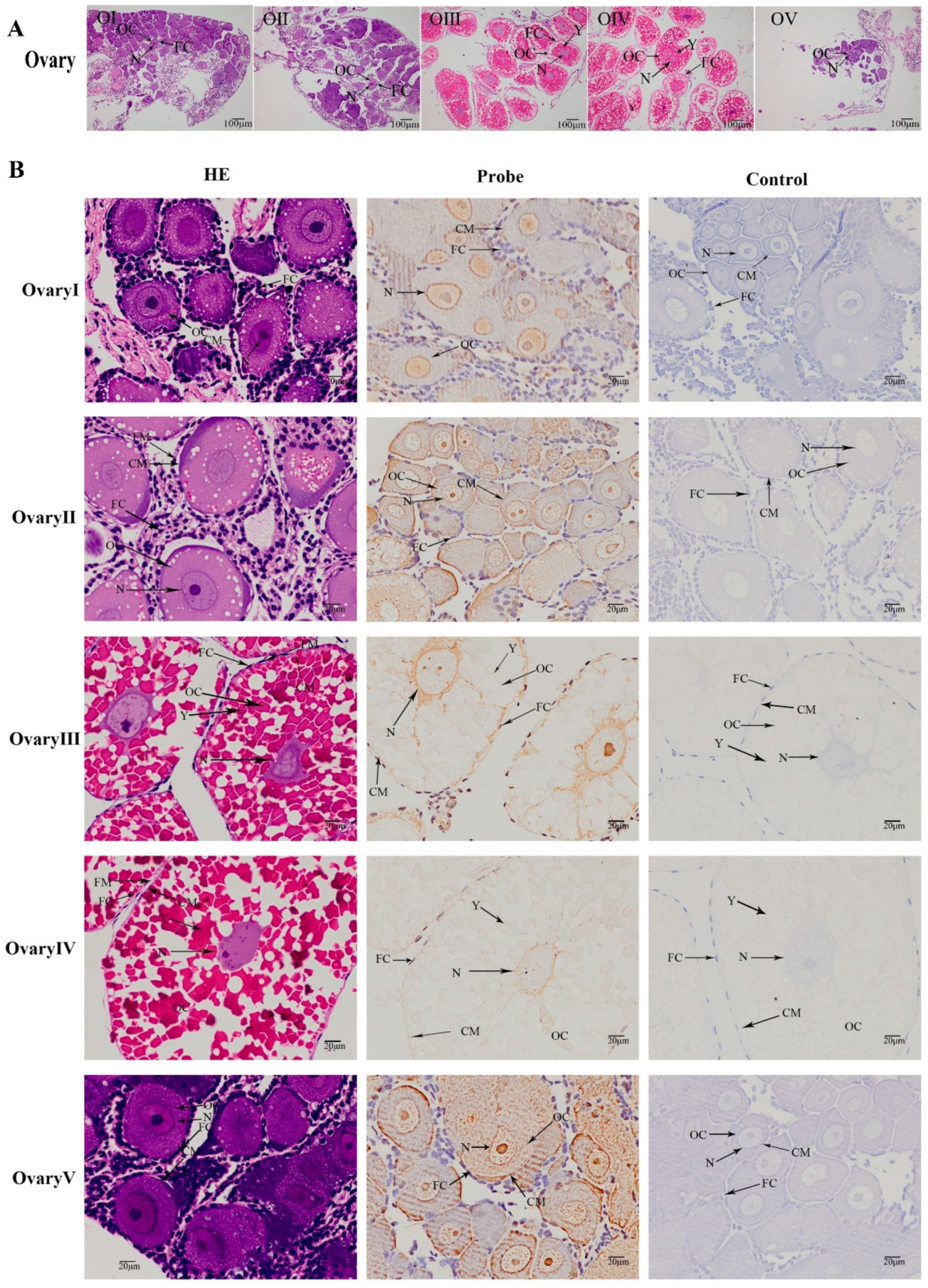
2.4. Expression of the MniDMRT11E Gene in Different Developmental Stages of the Ovaries
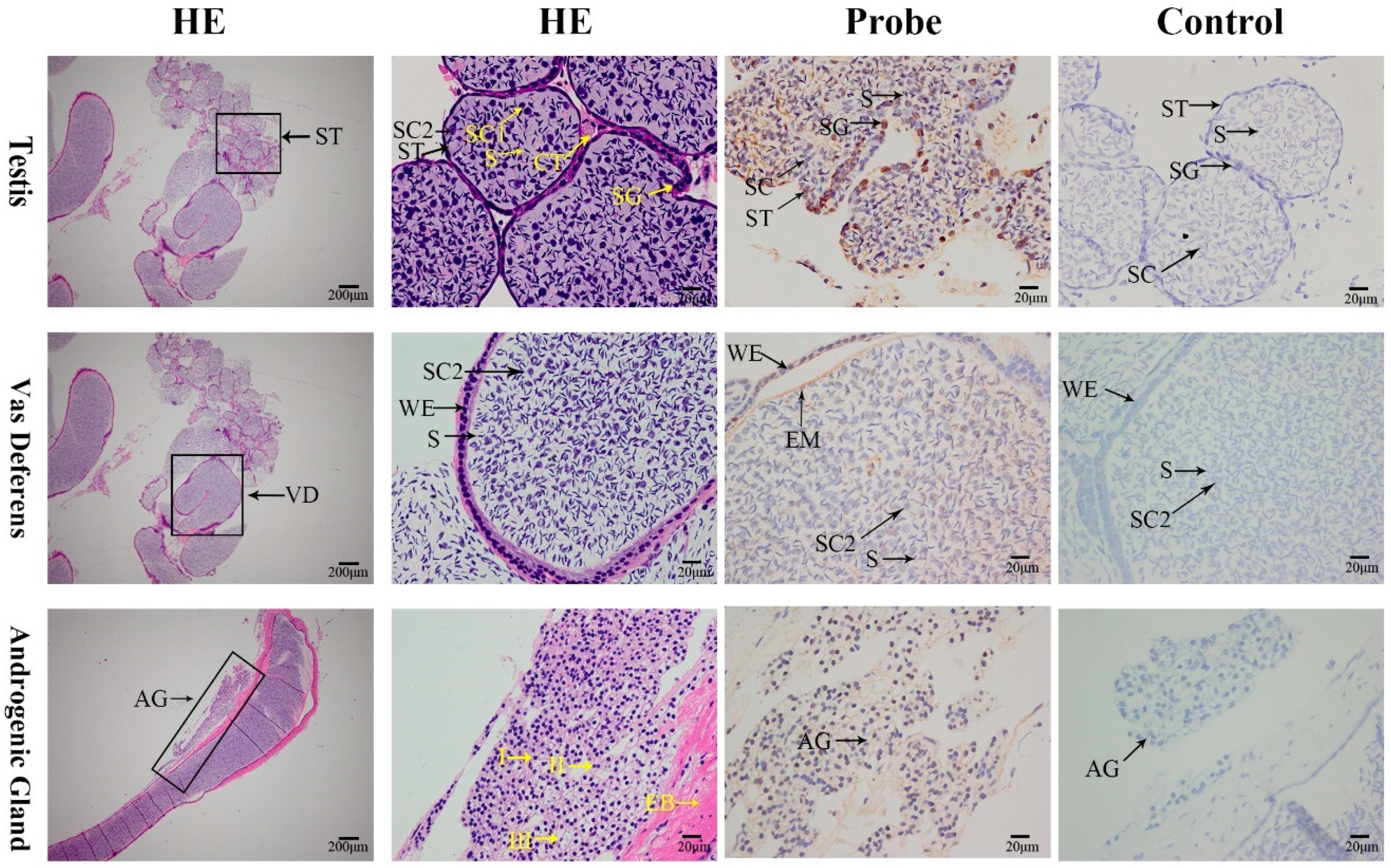
2.5. Localization of the MniDMRT11E Gene in the Gonad
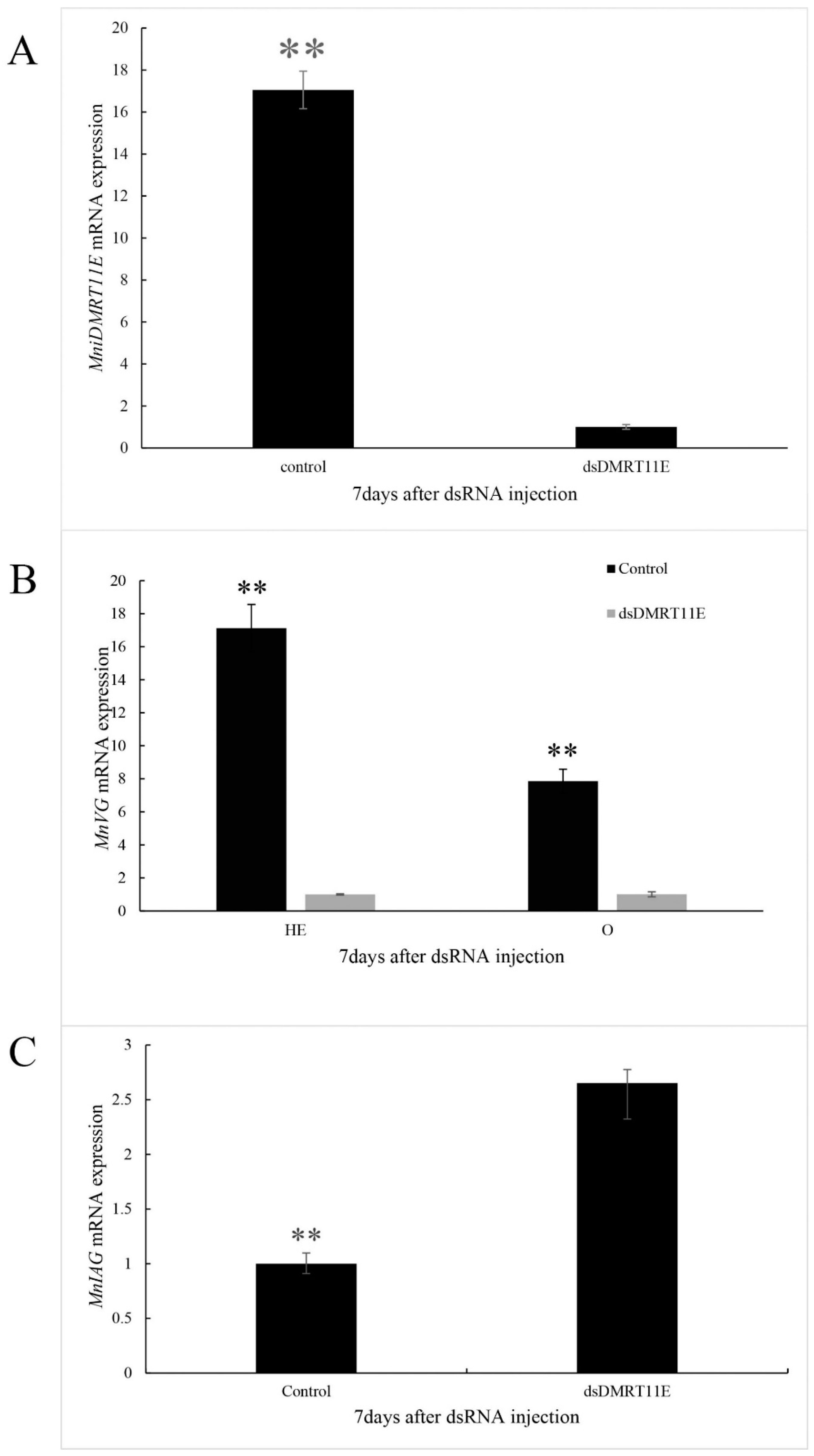
2.6. Effects of MniDMRT11E Knockdown on Gonad by RNAi
3. Discussion
4. Materials and Methods
4.1. Experimental Animals and Sampling
4.2. Nucleotide Sequence and Bioinformatics Analysis
4.3. Tissue Expression Analysis by Quantitative Real-Time PCR
4.4. Expression Profiles of DMRT11E in Ovarian Cycle
4.5. In Situ Hybridization (ISH)
4.6. RNA Interference of DMRT11E
4.7. Data Analysis
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| DMRT | doublesex and mab-3 related transcription factor |
| VG | vitellogenin |
| IAG | insulin-like androgenic gland factor |
| dsx | doublesex |
| qPCR | quantitative real-time reverse transcription PCR |
| EM | eosinophilic matrix |
| E | eyestalk |
| BR | brain |
| H | heart |
| HE | hepatopancreas |
| G | gill |
| M | muscle; |
| O | ovary |
| T | testis |
| AG | abdominal ganglia |
| UE | unfertilized egg |
| CS | cleavage stage |
| BS | blastula stage |
| GS | gastrula stage |
| NS | nauplius stage |
| ZS | zoea stage |
| L1 | the first day larvae after hatching |
| O I | undeveloped stage |
| O II | developing stage |
| O III | nearly-ripe stage |
| O IV | ripe stage |
| O V | spent stage |
| OG | oogonium |
| OC | oocyte |
| N | nucleus |
| CM | cytoplasmic membrane |
| Y | yolk granule |
| FC | follicle cell |
| FM | follicle membrane |
| CT | collecting tissue |
| SG | spermatogonium |
| SC | spermatocyte |
| SC1 | primary spermatocyte |
| SC2 | second spermatocyte |
| ST | spermatid |
| S | sperm |
| WE | wall epithelium |
| EM | eosinophilic matrix |
| VD | vas deferens |
| EB | the ejaculatory bulb |
| ORF | open reading frame |
| M. nipponense | Macrobrachium nipponense |
| M. rosenbergii | Macrobrachium rosenbergii |
| cDNA | complementary DNA |
| FFRC | freshwater Fisheries Research Centre |
| ISH | in situ hybridization |
| CISH | chromogenic in situ hybridization |
References
- Bureau of Fishery, Ministry of Agriculture, People’s Republic of China. Fisheries Economic Statistics. In China Fishery Yearbook; China Agricultural Press: Beijing, China, 2018; p. 34. [Google Scholar]
- Burtis, K.C.; Baker, B.S. Drosophila doublesex gene controls somatic sexual differentiation by producing alternatively spliced mRNAs encoding related sex-specific polypeptides. Cell 1989, 56, 997–1010. [Google Scholar] [CrossRef]
- Martín, I.; Ruiz, M.F.; Sánchez, L. The gene transformer-2 of Sciara (Diptera, Nematocera) and its effect on Drosophila sexual development. BMC Dev. Biol. 2011, 11, 19. [Google Scholar] [CrossRef]
- Ventura, T.; Manor, R.; Aflalo, E.D.; Weil, S.; Raviv, S.; Glazer, L.; Sagi, A. Temporal silencing of an androgenic gland-specific insulin-like gene affecting phenotypical gender differences and spermatogenesis. Endocrinology 2009, 150, 1278–1286. [Google Scholar] [CrossRef]
- Chung, J.S.; Manor, R.; Sagi, A. Cloning of an insulin-like androgenic gland factor (IAG) from the blue crab, Callinectes sapidus: Implications for eyestalk regulation of IAG expression. Gen. Comp. Endocrinol. 2011, 173, 4–10. [Google Scholar] [CrossRef] [PubMed]
- Ma, K.Y.; Lin, J.Y.; Guo, S.Z.; Chen, Y.; Li, J.L.; Qiu, G.F. Molecular characterization and expression analysis of an insulin-like gene from the androgenic gland of the oriental river prawn, Macrobrachium nipponense. Gen. Comp. Endocrinol. 2013, 185, 90–96. [Google Scholar] [CrossRef]
- Vega-Alpízar, J.L.; Alfaro-Montoya, J.; Hernández-Noguera, L.; Umaña-Castro, R.; Aflalo, E.D.; Sagic, A. Implant recognition and gender expression following ampoule-androgenic gland implantation in Litopenaeus vannamei females (Penaeidae). Aquaculture 2017, 468, 471–480. [Google Scholar] [CrossRef]
- Guo, Q.; Li, S.; Lv, X.; Xiang, J.; Sagi, A.; Manor, R.; Li, F. A Putative Insulin-like Androgenic Gland Hormone Receptor Gene Specifically Expressed in Male Chinese Shrimp. Endocrinology 2018, 159, 2173–2185. [Google Scholar] [CrossRef]
- Zhang, E.F.; Qiu, G.F. A novel Dmrt gene is specifically expressed in the testis of Chinese mitten crab, Eriocheir sinensis. Dev. Genes Evol. 2010, 220, 151–159. [Google Scholar] [CrossRef] [PubMed]
- Yu, Y.Q.; Ma, W.M.; Zeng, Q.G.; Qian, Y.Q.; Yang, J.S.; Yang, W.J. Molecular Cloning and Sexually Dimorphic Expression of Two Dmrt, Genes in the Giant Freshwater Prawn, Macrobrachium rosenbergii. Agric. Res. 2014, 3, 3181–3191. [Google Scholar] [CrossRef]
- Raymond, C.S.; Murphy, M.W.; O’Sullivan, M.G.; Bardwell, V.J.; Zarkower, D. Dmrt1, a gene related to worm and fly sexual regulators, is required for mammalian testis differentiation. Genes Dev. 2000, 14, 2587–2595. [Google Scholar] [CrossRef]
- Raymond, C.S.; Shamu, C.E.; Shen, M.M.; Seifert, K.J.; Hirsch, B.; Hodgkin, J.; Zarkower, D. Evidence for evolutionary conservation of sex-determining genes. Nature 1998, 391, 691–695. [Google Scholar] [CrossRef]
- Artyom, K. Dmrt, genes in the development and evolution of sexual dimorphism. Trends Genet. 2012, 28, 175–184. [Google Scholar]
- Erdman, S.E.; Burtis, K.C. The Drosophila doublesex proteins share a novel zinc finger related DNA binding domain. The EMBO journal 1993, 12, 527–535. [Google Scholar] [CrossRef]
- Murphy, M.W.; Lee, J.K.; Rojo, S.; Gearhart, M.D.; Kurahashi, K.; Banerjee, S.; Loeuille, G.A.; Bashamboo, A.; McElreavey, K.; Zarkower, D.; et al. An ancient protein-DNA interaction underlying metazoan sex determination. Nat. Struct. Mol. Biol. 2015, 22, 442–451. [Google Scholar] [CrossRef]
- Zhang, T.; Zarkower, D. DMRT proteins and coordination of mammalian spermatogenesis. Stem Cell Res. 2017, 24, 195–202. [Google Scholar] [CrossRef]
- Hildreth, P.E. doublesex, recessive gene that transforms both males and females of drosophila into intersexes. Genetics 1965, 51, 659–678. [Google Scholar]
- Shen, M.M.; Hodgkin, J. mab-3, a gene required for sex-specific yolk protein expression and a male-specific lineage in C. elegans. Cell 1988, 54, 1019–1031. [Google Scholar] [CrossRef]
- Picard, M.A.; Cosseau, C.; Mouahid, G.; Duval, D.; Grunau, C.; Toulza, E.; Allienne, J.F.; Boissier, J. The roles of Dmrt (Double sex/Male-abnormal-3 Related Transcription factor) genes in sex determination and differentiation mechanisms: Ubiquity and diversity across the animal kingdom. Comptes Rendus Biol. 2015, 338, 451–462. [Google Scholar] [CrossRef] [PubMed]
- Kato, Y.; Kobayashi, K.; Oda, S.; Colbourn, J.K.; Tatarazako, N.; Watanabe, H.; Iguchi, T. Molecular cloning and sexually dimorphic expression of DM-domain genes in Daphnia magna. Genomics 2008, 91, 94–101. [Google Scholar] [CrossRef]
- Li, F.; Qiao, H.; Fu, H.; Sun, S.; Zhang, W.; Jin, S.; Jiang, S.; Gong, Y.; Xiong, Y.; Wu, Y.; et al. Identification and characterization of opsin gene and its role in ovarian maturation in the oriental river prawn Macrobrachium nipponense. Comp. Biochem. Physiol. Part B Biochem. Mol. Biol. 2018, 218, 1–12. [Google Scholar] [CrossRef]
- Kim, B.M.; Jeong, C.B.; Kim, I.C.; Yim, J.H.; Lee, Y.S.; Rhee, J.; Lee, J. Identification of three doublesex genes in the monogonont rotifer Brachionus koreanus and their transcriptional responses to environmental stressor-triggered population growth retardation. Comp. Biochem. Physiol. Part B Biochem. Mol. Biol. 2014, 174, 36–44. [Google Scholar] [CrossRef]
- Qiu, G.; Du, N.; Lai, W. Studies on the male reproductive system of the freshwater praan, Macrobrachium nipponense: II. The morphology and ultrastructure of the sperm. Acta Zool. Sin. 1996, 42, 349–354. [Google Scholar]
- Qiao, H.; Xiong, Y.; Zhang, W.; Fu, H.; Jiang, S.; Sun, S.; Bai, H.; Jin, S.; Gong, Y. Characterization, expression, and function analysis of gonad-inhibiting hormone in Oriental River prawn, Macrobrachium nipponense and its induced expression by temperature. Comp. Biochem. Physiol. Part A Mol. Integr. Physiol. 2015, 185, 1–8. [Google Scholar] [CrossRef]
- Phoungpetchara, I.; Tinikul, Y.; Poljaroen, J.; Chotwiwatthanakun, C.; Vanichviriyakit, R.; Sroyraya, M.; Hanna, P.J.; Sobhon, P. Cells producing insulin-like androgenic gland hormone of the giant freshwater prawn, Macrobrachium rosenbergii, proliferate following bilateral eyestalk-ablation. Tissue Cell 2011, 43, 165–177. [Google Scholar] [CrossRef] [PubMed]
- Bai, H.; Qiao, H.; Li, F.; Fu, H.; Jiang, S.; Zhang, W.; Yan, Y.; Xiong, Y.; Sun, S.; Jin, S.; et al. Molecular and functional characterization of the vitellogenin receptor in oriental river prawn, Macrobrachium nipponense. Comp. Biochem. Physiol. Part A Mol. Integr. Physiol. 2016, 194, 45–55. [Google Scholar] [CrossRef] [PubMed]
- Bellefroid, E.J.; Leclere, L.; Saulnier, A.; Keruzore, M.; Sirakov, M.; Vervoort, M.; De Clercq, S. Expanding roles for the evolutionarily conserved Dmrt sex transcriptional regulators during embryogenesis. Cell. Mol. Llife Sci. 2013, 70, 3829–3845. [Google Scholar] [CrossRef] [PubMed]
- Wexler, J.R.; Plachetzki, D.C.; Kopp, A. Pan-metazoan phylogeny of the DMRT gene family: A framework for functional studies. Dev. Genes Evol. 2014, 224, 175–181. [Google Scholar] [CrossRef] [PubMed]
- Zhu, L.; Wilken, J.; Phillips, N.B.; Narendra, U.; Chan, G.; Stratton, S.M.; Kent, S.B.; Weiss, M.A. Sexual dimorphism in diverse metazoans is regulated by a novel class of intertwined zinc fingers. Genes Dev. 2000, 14, 1750–1764. [Google Scholar]
- Lourenço, R.; Lopes, S.S.; Saúde, L. Left-Right Function of dmrt2 Genes Is Not Conserved between Zebrafish and Mouse. PLoS ONE 2010, 5, e14438. [Google Scholar] [CrossRef] [PubMed]
- Seo, K.W.; Wang, Y.; Kokubo, H.; Kettlewell, J.R.; Zarkower, D.A.; Johnson, R.L. Targeted disruption of the DM domain containing transcription factor Dmrt2 reveals an essential role in somite patterning. Dev. Biol. 2006, 290, 200–210. [Google Scholar] [CrossRef]
- Wilder, M.N.; Okumura, T.; Tsutsui, N. Reproductive mechanisms in Crustacea focusing on selected prawn species: Vitellogenin structure, processing and synthetic control. Aqua Biosci. Monogr. 2010, 3, 73–110. [Google Scholar] [CrossRef]
- Yi, W.; Zarkower, D. Similarity of DNA binding and transcriptional regulation by Caenorhabditis elegans MAB-3 and Drosophila melanogaster DSX suggests conservation of sex determining mechanisms. Development 1999, 126, 873–881. [Google Scholar]
- Zhang, X.; Zha, J.; Wang, Z. Influences of 4-nonylphenol on doublesex-and mab-3–related transcription factor 1 gene expression and vitellogenin mRNA induction of adult rare minnow (Gobiocypris rarus). Environ. Toxicol. Chem. 2008, 27, 196–205. [Google Scholar] [CrossRef]
- Shukla, J.N.; Palli, S.R. Doublesex target genes in the red flour beetle, Tribolium castaneum. Sci. Rep. 2012, 2, 948. [Google Scholar] [CrossRef]
- Ventura, T.; Manor, R.; Aflalo, E.D.; Weil, S.; Rosen, O.; Sagi, A. Timing sexual differentiation: Full functional sex reversal achieved through silencing of a single insulin-like gene in the prawn, Macrobrachium rosenbergii. Biol. Rreprod. 2012, 86, 1–6. [Google Scholar] [CrossRef]
- Shechter, A.; Glazer, L.; Cheled, S.; Mor, E.; Weil, S.; Berman, A.; Bentov, S.; Aflalo, E.D.; Khalaila, I.; Sagi, A. A gastrolith protein serving a dual role in the formation of an amorphous mineral containing extracellular matrix. Proc. Natl. Acad. Sci. USA 2008, 105, 7129–7134. [Google Scholar] [CrossRef]
- Bai, H.; Qiao, H.; Li, F.; Fu, H.; Sun, S.; Zhang, W.; Jin, S.; Gong, Y.; Jiang, S.; Xiong, Y. Molecular characterization and developmental expression of vitellogenin in the oriental river prawn Macrobrachium nipponense and the effects of RNA interference and eyestalk ablation on ovarian maturation. Gene 2015, 562, 22–31. [Google Scholar] [CrossRef]
- French, A.S.; Meisner, S.; Liu, H.; Weckström, M.; Torkkeli, P.H. Transcriptome analysisand RNA interference of cockroach phototransduction indicate three opsins and suggest a major role for TRPL channels. Front. Physiol. 2015, 6, 207. [Google Scholar] [CrossRef]
- Subramoniam, T. Mechanisms and control of vitellogenesis in crustaceans. Fisheries. Sci. 2011, 77, 1–21. [Google Scholar] [CrossRef]
- Zhang, Y.; Jiang, S.; Xiong, Y.; Sun, S.; Qiao, H.; Jin, S.; Gong, Y.; Fu, H. Molecular cloning and expression analysis of extra sex combs gene during development in Macrobrachium nipponense. Turk. J. Fish. Aquat. Sci. 2013, 13, 331–340. [Google Scholar] [CrossRef]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCT method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef]
- Hu, Y.; Fu, H.; Qiao, H.; Sun, S.; Zhang, W.; Jin, S.; Jiang, S.; Gong, Y.; Xiong, Y.; Wu, Y. Validation and evaluation of reference genes for quantitative real-time PCR in Macrobrachium Nipponense. Int. J. Mol. Sci. 2018, 19, 2258. [Google Scholar] [CrossRef] [PubMed]

| Primer Name | Sequence (5′-3′) | Description |
|---|---|---|
| DMRTD-F1 | TGCGCCMGRTGYMGRAAYCAYGG | For DM-domain RACE |
| DMRTD-R1 | ARSGCSACYTGSGCSGCCATNAC | For DM-domain RACE |
| DMRTD-R2 | TGSGCSGCCATNACCCKYTGCC | For DM-domain RACE |
| 5′-RACE outer | ATTACCCGTTGCCTCT | For 5′-RACE |
| 5′-RACE inner | ACCACCAGCAGGCAGTTG | For 5′-RACE |
| DMRT-5′R | GCTTCTTGTGCCCCTTCA | For 5′-RACE |
| 3′-RACE outer | GGTGGTGTCCTGCCTGAA | For 3′-RACE |
| 3′-RACE inner | CCTGAAGGGGCACAAGAA | For 3′-RACE |
| DMRT-3′F | CCTGAAGGGGCACAAGAA | For 3′-RACE |
| DMRT-qF | ACGACCTTAGTAGGATGGACAGT | For RT-PCR |
| DMRT-qR | GAGTGGAGGCAATAGAATGGGTA | For RT-PCR |
| β-actinF | TATGCACTTCCTCATGCCATC | For RT-PCR |
| β-actinR | AGGAGGCGGCAGTGGTCAT | For RT-PCR |
| DMRT-P | CACTCCAGAATACTCGCCGAAGTCCTACGCGAAGAC | Probe foe ISH |
| DMRT-iF | TAATACGACTCACTATAGGGCTTAGTGTCAGACGCGACCA | For DMRT dsRNA |
| DMRT-iR | TAATACGACTCACTATAGGGCTTCGGCGAGTATTCTGGAG | For DMRT dsRNA |
| IAG-qF | CGCCTCCGTCTGCCTGAGATAC | For RT-PCR |
| IAG-qR | CCTCCTCCTCCACCTTCAATGC | For RT-PCR |
| VG-qF | GAAGTTAGCGGAGATCTGAGGT | For RT-PCR |
| VG-qR | CCTCGTTGACCAATCTTGAGAG | For RT-PCR |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, Y.; Jin, S.; Fu, H.; Qiao, H.; Sun, S.; Zhang, W.; Jiang, S.; Gong, Y.; Xiong, Y.; Wu, Y. Identification and Characterization of the DMRT11E Gene in the Oriental River Prawn Macrobrachium nipponense. Int. J. Mol. Sci. 2019, 20, 1734. https://doi.org/10.3390/ijms20071734
Wang Y, Jin S, Fu H, Qiao H, Sun S, Zhang W, Jiang S, Gong Y, Xiong Y, Wu Y. Identification and Characterization of the DMRT11E Gene in the Oriental River Prawn Macrobrachium nipponense. International Journal of Molecular Sciences. 2019; 20(7):1734. https://doi.org/10.3390/ijms20071734
Chicago/Turabian StyleWang, Yabing, Shubo Jin, Hongtuo Fu, Hui Qiao, Shengming Sun, Wenyi Zhang, Sufei Jiang, Yongsheng Gong, Yiwei Xiong, and Yan Wu. 2019. "Identification and Characterization of the DMRT11E Gene in the Oriental River Prawn Macrobrachium nipponense" International Journal of Molecular Sciences 20, no. 7: 1734. https://doi.org/10.3390/ijms20071734
APA StyleWang, Y., Jin, S., Fu, H., Qiao, H., Sun, S., Zhang, W., Jiang, S., Gong, Y., Xiong, Y., & Wu, Y. (2019). Identification and Characterization of the DMRT11E Gene in the Oriental River Prawn Macrobrachium nipponense. International Journal of Molecular Sciences, 20(7), 1734. https://doi.org/10.3390/ijms20071734







