Fritillariae Thunbergii Bulbus: Traditional Uses, Phytochemistry, Pharmacodynamics, Pharmacokinetics and Toxicity
Abstract
1. Introduction
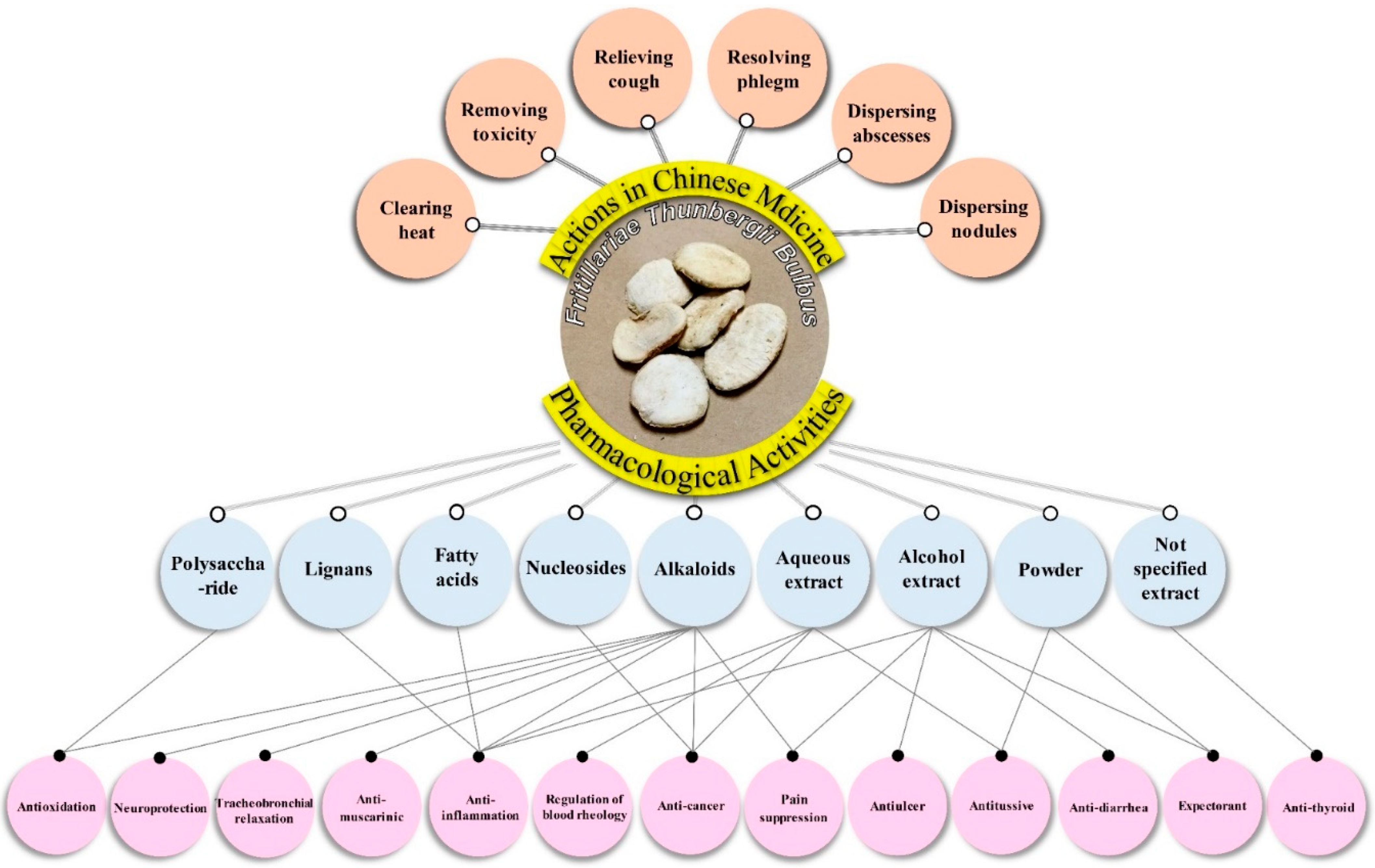
2. Traditional Uses of FTB
3. Modern Exploration of FTB
3.1. Phytochemistry of FTB
3.1.1. Alkaloids
3.1.2. Compounds from Essential Oils
3.1.3. Diterpenoids
3.1.4. Nucleosides
3.1.5. Elements
3.1.6. Other Constituents
3.2. Pharmacology of FTB
3.2.1. Anti-Cancer Effect
3.2.2. Tracheobronchial Relaxation
3.2.3. Antitussive Effect
3.2.4. Expectorant Effect
3.2.5. Anti-inflammation
3.2.6. Pain Suppression
3.2.7. Antioxidation
3.2.8. Other Pharmacological Effects
3.3. Pharmacokinetics of FTB
3.4. Toxicity of FTB
4. Conclusions
Supplementary Materials
Funding
Conflicts of Interest
References
- Bensky, D.; Clavey, S.; Stöger, E. Chinese Herbal Medicine. Materia Medica, 3rd ed.; Eastland Press: Seattle, WA, USA, 2015. [Google Scholar]
- Chinese Pharmacopoeia Commission (Ed.) Pharmacopoeia of the People’s Republic of China; China Medical Science and Technology Press: Beijing, China, 2015; Volume 1. [Google Scholar]
- Li, H.; Hung, A.; Yang, A.W.H. A Classic Herbal Formula Danggui Beimu Kushen Wan for Chronic Prostatitis: From Traditional Knowledge to Scientific Exploration. Evid. Based Complement. Altern. Med. 2018, 2018, 1612948. [Google Scholar] [CrossRef]
- Zhang, Z.; Wiseman, N.; Wilms, S. Jin Gui Yao Lue: Essential Prescriptions of the Golden Cabinet; Paradigm Publications: Taos, NM, USA, 2013. [Google Scholar]
- Wang, C.Y.; Bai, X.Y.; Wang, C.H. Traditional Chinese medicine: A treasured natural resource of anticancer drug research and development. Am. J. Chin. Med. 2014, 42, 543–559. [Google Scholar] [CrossRef] [PubMed]
- Jiangsu New Medical Academy (Ed.) Zhong Yao Da Ci Dian [Great Compendium of Chinese Medicines]; Shanghai Scientific and Technical Publishers: Shanghai, China, 1994. [Google Scholar]
- State Administration of Traditional Chinese Medicine ‘Chinese Materia Medica Committee’ (Ed.) Zhong Hua Ben Cao [Chinese Materia Medica]; Shanghai Scientific and Technical Publishers: Shanghai, China, 1999.
- Suh, W.S.; Lee, S.Y.; Park, J.E.; Kim, D.H.; Kim, S.; Lee, K.R. Two new steroidal alkaloids from the bulbs of fritillaria thunbergii. Heterocycles 2018, 96, 921–930. [Google Scholar] [CrossRef]
- Liu, W.; Zou, F.; Li, D. Studies on P-glycoprotein inhibitor of multidrug tumor in Bulbus Fritillariae Thunbergii. Chin. J. Surg. Integr. Tradit. West. Med. 2015, 21, 379–382. [Google Scholar]
- China Association of Chinese Medicine. Zhong Hua Yi Dian (Encyclopaedia of Traditional Chinese Medicine) [CD-ROM], 5th ed.; Hunan Electronic and Audio-Visual Publishing House: Changsha, China, 2015.
- Chinese Medicine Board of Australia. Nomenclature List of Commonly Used Chinese Herbal Medicines. Available online: http://www.chinesemedicineboard.gov.au/documents/default.aspx?record=WD15%2f18746&dbid=AP&chksum=Cs%2baCFhYVrzbzL2%2bYnhtRA%3d%3d (accessed on 22 January 2019).
- Culley, T.M. Why vouchers matter in botanical research. Appl. Plant Sci. 2013, 1, 1300076. [Google Scholar] [CrossRef] [PubMed]
- Kreiner, J.; Pang, E.; Lenon, G.B.; Yang, A.W.H. Saposhnikoviae divaricata: A phytochemical, pharmacological, and pharmacokinetic review. Chin. J. Nat. Med. 2017, 15, 255–264. [Google Scholar] [CrossRef]
- Cai, W.; Xiong, Y.; Sheng, Z.; Xia, M.; Cui, M. Determination of eighteen trace elements in Fritillaria from different places by ICP-OES. Chin. J. Mod. Appl. Pharm. 2013, 30, 277–280. [Google Scholar]
- Cai, X.; Zhang, Y.; Du, W.; Sheng, Z.; Yin, Z.; Fang, L.; Cai, B.; Ge, W. Determination of heavy metal and trace elements in fresh-cut Bulbus Fritillaria thunbergii slices by ICP-OES. Chin. Arch. Tradit. Chin. Med. 2014, 32, 2357–2359. [Google Scholar]
- Cao, Y.; Zhu, X.; Tan, L. Analysis of chemical composition by GC/MS and antimicrobial activities of essential oil from Fritillaria thunbergii Miq. J. Zhejiang Sci.-Tech. Univ. 2012, 29, 129–132. [Google Scholar]
- Chan, S.W. Pharmacological and Chemical Investigations into Bulbus Fritillariae. Ph.D. Thesis, Chinese University of Hong Kong, Hong Kong, China, 2000. [Google Scholar]
- Chen, X.; Li, X.; Mao, X.; Huang, H.; Miao, J.; Gao, W. Study on the effects of different drying methods on physicochemical properties, structure, and in vitro digestibility of Fritillaria thunbergii Miq. (Zhebeimu) flours. Food Bioprod. Process. 2016, 98, 266–274. [Google Scholar] [CrossRef]
- Chen, Y.; Wang, S. β-D-glu 4-1β-D-gal and sucrose determination of different Fritillarias by HPLC-ELSD. Lishizhen Med. Mater. Med. Res. 2012, 23, 1605–1606. [Google Scholar]
- Chen, Z.; Zhou, X.; Zhang, Z.; Chen, W. Disscussion on the necessity of preserving the bulb of Zhebeimu (Fritillariae thunbergii bulbus) based on modern chemical analysis. Guid. J. Tradit. Chin. Med. Pharm. 2016, 22, 44–46. [Google Scholar]
- Cheng, X.; Xiao, X.; Zhang, N.; Ma, S. Determination the contents of peimine and peiminine in Thumberg Fritillary Bulb by HPLC-MS. China Pharm. 2008, 11, 770–772. [Google Scholar]
- Cui, M.; Zhang, J.; Chen, S.; Jiang, H.; Zhou, H.; Ling, Q. Identification of alkaloids and flavonoids in all parts of Fritillaria thunbergii using LC-LTQ-Qrbitrap MSn. Zhongguo Zhong Yao Za Zhi 2016, 41, 2124–2130. [Google Scholar] [PubMed]
- Dai, G.; Liu, Z.; Chen, X.; Zhou, Q. Studies on the effect on the quantities of alkaloids of Bulbs Fritillariae thunbergii in different menstruum. Lishizhen Med. Mater. Med. Res. 2001, 12, 779–780. [Google Scholar]
- Ding, X.; Li, R.; Fang, K. Contents mensuration of total alkaloid in Bulbus Fritillariae by acid dye colorinetry. China Pharma. 1999, 8, 46–47. [Google Scholar]
- Du, W.; Zhang, H.; Yue, X.; Zhu, T.; Ge, W. The analysis on volatile components of Zhejiang Fritillary slices with different primary processing methods. Lishizhen Med. Mater. Med. Res. 2018, 29, 73–76. [Google Scholar]
- Duan, B.; Huang, L.; Chen, S. Study on the destructive effect to inherent quality of Fritillaria thunbergii Miq. (Zhebeimu) by sulfur-fumigated process using chromatographic fingerprinting analysis. Phytomedicine 2012, 19, 562–568. [Google Scholar] [CrossRef]
- Guo, J. Compariative Research on the Pharmaceutic and Pharmacodynamics among Different Breed Bulbus Fritillaria Thunbergii. Master’s Thesis, Heilongjiang University of Chinese Medicine, Haerbin, China, 2007. [Google Scholar]
- Huang, C. Determination of the contents of peimine A and peimine B in Fritillaria thunbergii by ELSD. China Health Stand. Manag. 2016, 7, 139–141. [Google Scholar]
- Jiang, Y.; Li, P. Determination of verticine and verticinone in Bulbus Fritillariae Thunbergii by HPLC-ELSD. Zhongguo Yao Xue Za Zhi 2005, 40, 1257–1259. [Google Scholar]
- Jin, X.; Xu, D.; Xu, Y.; Cui, D.; Xiao, X.; Tian, Z.; Lu, Y.; Zhen, Q. The structure identification of zhepiresionol. Acta Pharm. Sin. 1993, 28, 212–215. [Google Scholar]
- Jin, Y.; Shen, P.; Zhang, J.; Chao, Z.; Chao, X.; Yan, Y. Separation and purification of peimine and peiminine from Bulbus Fritillariae Thunbergi by high speed countercurrent chromatography. Res. Inf. Tradit. Chin. Med. 2005, 7, 13–15. [Google Scholar]
- Jin, Y.; Zeng, G.; Wang, L.; Wang, Z. Effects of different soaking time on extraction of active components of Fritillary Bulb. China Pharm. 2012, 21, 24–25. [Google Scholar]
- Kim, E.J.; Yoon, Y.P.; Woo, K.W.; Kim, J.H.; Min, S.Y.; Lee, H.J.; Lee, S.K.; Hong, J.H.; Lee, K.R.; Lee, C.J. Verticine, ebeiedine and suchengbeisine isolated from the bulbs of Fritillaria thunbergii Miq. inhibited the gene expression and production of MUC5AC mucin from human airway epithelial cells. Phytomedicine 2016, 23, 95–104. [Google Scholar] [CrossRef] [PubMed]
- Kitajima, J.; Komori, T.; Kawasaki, T. Studies on the constituents of the crude drug “Fritillariae bulbus.” III. On the diterpenoid constituents of fresh bulbs of Fritillaria thunbergii Miq. Chem. Pharm. Bull. 1982, 30, 3912–3921. [Google Scholar] [CrossRef]
- Kitajima, J.; Noda, N.; Ida, Y.; Komori, T.; Kawasaki, T. Studies on the constituents of the crude drug “Fritillariae bulbus.” IV. On the diterpenoid constituents of the crudr “Fritillariae bulbus”. Chem. Pharm. Bull. 1982, 30, 3922–3931. [Google Scholar] [CrossRef]
- Li, S.L.; Lin, G.; Chan, S.W.; Li, P. Determination of the major isosteroidal alkaloids in bulbs of Fritillaria by high-performance liquid chromatography coupled with evaporative light scattering detection. J. Chromatogr. A 2001, 909, 207–214. [Google Scholar] [CrossRef]
- Li, S.-L.; Li, P.; Lin, G.; Chan, S.-W.; Ho, Y.-P. Simultaneous determination of seven major isosteroidal alkaloids in bulbs of Fritillaria by gas chromatography. J. Chromatogr. A 2000, 873, 221–228. [Google Scholar] [CrossRef]
- Li, W.; Bi, K.; Qian, Y.; Ning, L.; Luo, X. A pre-column derivatization HPLC method for the determination of peimine and peiminine in Bulbus Fritillariae. J. Chin. Pharm. Sci. 1996, 5, 213–218. [Google Scholar]
- Liu, B.; Shi, R.; Lu, Y.; Zhou, Y. Study on the contents mensuration of total alkaloid in Bulbus Fritillariae by acid dye colorinetry. In Proceedings of the Fifth National Academic Conference on the Identification of Traditional Chinese Medicine, Weihai, China, 1 August 2001; pp. 152–155. [Google Scholar]
- Liu, J.; Wang, X.; Xiang, Z.; Li, Y. Study on the principal component analysis method to determination of trace elements of Fritillaria thunbergii Miq. Guangdong Trace Elem. Sci. 2008, 15, 31–35. [Google Scholar]
- Liu, L.; You, W.; Zheng, L.; Chen, F.; Jia, Z. Determination of peimine and peiminine in Bulbus Fritillariae Thunbergii by capillary electrophoresis by indirect UV detection using N-(1-naphthyl)ethylenediamine dihydrochloride as probe. Electrophoresis 2012, 33, 2152–2158. [Google Scholar] [CrossRef] [PubMed]
- Long, Z.; Guo, Z.; Acworth, I.N.; Liu, X.; Jin, Y.; Liu, X.; Liu, L.; Liang, L. A non-derivative method for the quantitative analysis of isosteroidal alkaloids from Fritillaria by high performance liquid chromatography combined with charged aerosol detection. Talanta 2016, 151, 239–244. [Google Scholar] [CrossRef] [PubMed]
- Lou, Y.; Cai, H.; Liu, X.; Tu, S.; Pei, K.; Zhao, Y.; Cao, G.; Li, S.; Qin, K.; Cai, B. Element analysis and characteristic identification of non-fumigated and sulfur-fumigated Fritillaria thunbergii Miq. using microwave digestion-inductively coupled plasma atomic emission spectrometry combined with Fourier transform infrared spectrometry. Pharmacogn. Mag. 2014, 10, S30–S36. [Google Scholar] [CrossRef]
- Ma, W.; Rong, J.; Sheng, Z.; Hu, J. Study on HPLC specific chromatogram of Fritillaric thunbergii. Chin. J. Hosp. Pharm. 2008, 28, 1458–1460. [Google Scholar]
- Ma, Y.; Wen, Q.; Tong, X.; Fan, B.; Sun, J. Study on serum pharmacochemistry of Fritillaria thunbergii Miq. by LC-MS in rats. Chin. J. Pharmacovigil. 2014, 11, 714–720. [Google Scholar]
- Morimoto, H.; Kimata, S. Studies on the components of Fritillaria Thunbergii Miq. I. isolation of peimine and its new glycoside. Chem. Pharm. Bull. 1960, 8, 302–307. [Google Scholar] [CrossRef]
- Qin, J.; Chen, Y.; Luo, M.; Zheng, X.; Deng, G.; Lin, H. Comparison of in vitro dissolution rates of three constituents in superfine powder, coarse powder and decoction pieces of Fritillaria thunbergii. Chin. Tradit. Patent Med. 2017, 39, 1232–1235. [Google Scholar]
- Que, L.; Lin, Z. Determination and comparision of alkaloids in Dongbeimu, Zhebeimu and Chuanbeimu by thin layer chromatography. Zhejiang Pharm. 1984, 1, 18–20. [Google Scholar]
- Tong, X. Reversing Multi-Drug Resistance on Tumor Cells and Pharmacokinetics Study on Ingredients in a Traditional Chinese Medicine Fritillaria thunbergii Miq. Master’s Thesis, Yunnan University of Traditional Chinese Medicine, Kunming, China, 2016. [Google Scholar]
- Wang, A.; Hu, M. Cotents mensuration of total alkaloid in Bulbus Fritillariae by acid dye two-phase titration. Chin. J. Mod. Appl. Pharm. 1998, 15, 50–51. [Google Scholar]
- Wang, L.; Xia, J. Determination of the proportion of peimine in Thunbery Fritillary Bulb. Heilongjiang Sci. Technol. Inf. 2011, 34. [Google Scholar] [CrossRef]
- Wang, X.; Yang, K.; Du, L. Determination of the proportion of Cu, Fe, Zn, and Ca elements in Thunbery Fritillary Bulb and Tendrilleaf Fritillary Bulb. Sichuang Food Ferment. 2007, 43, 65–67. [Google Scholar]
- Wang, Y.; Zheng, Y. The measurement and analysis of eight elements in several sorts of Fritillaria of traditional Chinese medicine. Stud. Trace Elem. Health 2004, 21, 30–33. [Google Scholar]
- Wu, D.; Wang, H.; Shen, P. Determination of selenium with different valences in Zhebeimu by spectrophotometry. Guangdong Trace Elem. Sci. 1998, 5, 66–68. [Google Scholar]
- Wu, J.; Zou, Y.; Zhan, X.; Chen, S.; Lu, G.; Lai, F. Survey of heavy metal pollution in four chinese crude drugs and their cultivated soils. Bull. Environ. Contam. Toxicol. 2008, 81, 571–573. [Google Scholar] [CrossRef] [PubMed]
- Wu, X.; Chan, S.W.; Ma, J.; Li, P.; Shaw, P.C.; Lin, G. Investigation of association of chemical profiles with the tracheobronchial relaxant activity of Chinese medicinal herb Beimu derived from various Fritillaria species. J. Ethnopharmacol. 2018, 210, 39–46. [Google Scholar] [CrossRef]
- Wu, Z.; Li, Q. Comparative analysis of the effective components (alkaloids) of Fritillaria anhuiensis S.C.Chen et S.P.Yin, Fritillariae Thunbergii Bulbus and Fritillariae Cirrhosae Bulbus. Chin. Tradit. Herbal Drugs 1993, 24, 351–353. [Google Scholar]
- Wu, Z.; Zheng, S. Grey relational analysis and determination of elements in Fritillaria Thunbergii Bulb and Fritillariae Cirrhosae Bulb. J. Chin. Med. Mater. 1997, 20, 291–293. [Google Scholar]
- Xu, Y.; Li, Y.; Zhang, P.; Yang, B.; Wu, H.; Guo, X.; Li, Y.; Zhang, Y. Sensitive UHPLC-MS/MS quantitation and pharmacokinetic comparisons of multiple alkaloids from Fuzi-Beimu and single herb aqueous extracts following oral delivery in rats. J. Chromatogr. B 2017, 1058, 24–31. [Google Scholar] [CrossRef] [PubMed]
- Xue, Y.; Gu, H. Determination of peimine and peiminine in Fritillaria thunbergii by HPLC-ELSD. Acta Pharm. Sin. 2005, 40, 550–552. [Google Scholar]
- Yu, R.; Cheng, Z.; Gong, X. Determination and comparison of trace elements in four sort of Bulbus Fritillariae by microwave digestion-atomic absorption spectrometry. Spectrosc. Spectr. Anal. 2007, 27, 2591–2594. [Google Scholar]
- Yuan, X.; Shi, J.; Yang, Y.; Luan, J.; Gao, J.; Wang, Y. Establishment of element fingerprint and multielement analysis of Fritillaria thunbergii by inductively coupled plasma optical emission spectrometry. Biol. Trace Elem. Res. 2010, 135, 304–313. [Google Scholar] [CrossRef] [PubMed]
- Zeng, R.; Liu, L.; Chen, L.; Zhao, Y.; Yi, W. Study on the determination of peimine in Fritillaria thunbergii Miq from different sources by using HPLC-ELSD. J. Jaingxi Univ. Tradit. Chin. Med. 2009, 21, 51–52. [Google Scholar]
- Zhang, C.; Sun, L.; Chen, R.; Su, J.H.; Zhang, H.F.; Gu, B.R.; Xing, Y.W.; Xue, M. Multiple analytical methods for identification and quality evaluation of Fritillariae Thunbergii Bulbus based on biological single molecules by high-performance liquid chromatography. J. Sep. Sci. 2016, 39, 3536–3543. [Google Scholar] [CrossRef]
- Zhang, J.; Lao, A.; Huang, H.; Ma, G.; Xu, R. Studies on chemical constituents of Fritillaria thunbergii Miq. III. isolation and identification of Zhebeinone. Acta Pharm. Sin. 1992, 27, 472–475. [Google Scholar]
- Zhang, J.; Lao, A.; Ma, G.; Xu, R. Studies on chemical constituents of Fritillaria thunbergii Miq. II. Acta Pharm. Sin. 1991, 33, 923–926. [Google Scholar]
- Zhang, J.; Lao, A.; Xu, R. Studies on chemical constituents of Fritillaria thunbergii Miq. IV. Chin. Bull. Bot. 1993, 35, 238–241. [Google Scholar]
- Zhang, J.; Lao, A.; Xu, R. Studies on the chemical constituents of fresh bulbs of Fritillaria thunbergii Miq. China J. Chin. Mater. Med. 1993, 18, 354–355. [Google Scholar]
- Zhang, J.; Ma, G.; Lao, A.; Xu, R. Studies on chemical constituents of Fritillaria thunbergii Miq. Acta Pharm. Sin. 1991, 26, 231–233. [Google Scholar]
- Zhang, W. Study on the Determination of Alkaloid and Nucleosides in Various Species of Fritillaria. Master’s Thesis, Hunan Normal University, Changsha, China, 2008. [Google Scholar]
- Zhang, X.; Li, F.; Wang, M. HPLC quantitative analysis of uracil and three nucleotides in Bulbus Fritillariae and the application for variety discrimination. In Proceedings of the Chinese Chemical Society: The 16th National Conference on Organic Analysis and Bioanalysis Proceeding, Neimenggu, China, 12 August 2011; p. 8. [Google Scholar]
- Zhou, J.L.; Xin, G.Z.; Shi, Z.Q.; Ren, M.T.; Qi, L.W.; Li, H.J.; Li, P. Characterization and identification of steroidal alkaloids in Fritillaria species using liquid chromatography coupled with electrospray ionization quadrupole time-of-flight tandem mass spectrometry. J. Chromatogr. A 2010, 1217, 7109–7122. [Google Scholar] [CrossRef]
- Zhou, M.; Ma, X.; Ding, G.; Wang, Z.; Liu, D.; Tong, Y.; Zhou, H.; Gao, J.; Hou, Y.; Jiang, M.; et al. Comparison and evaluation of antimuscarinic and anti-inflammatory effects of five Bulbus fritillariae species based on UPLC-Q/TOF integrated dual-luciferase reporter assay, PCA and ANN analysis. J. Chromatogr. B 2017, 1041–1042, 60–69. [Google Scholar] [CrossRef]
- Zhou, Y.; Gao, X.; Wu, C.; Wu, Y. Bioaccessibility and safety assessment of trace elements from decoction of “Zhebawei” herbal medicines by in vitro digestion method. J. Trace Elem. Med. Biol. 2014, 28, 173–178. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Ji, H.; Lin, B.Q.; Jiang, Y.; Li, P. The effects of five alkaloids from Bulbus Fritillariae on the concentration of cAMP in HEK cells transfected with muscarinic M2 receptor plasmid. Am. J. Chin. Med. 2006, 34, 901–910. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Liu, L.; Zhu, W.; Zhang, H.; Yan, Z.; Liu, H. Comparative pharmacokinetic studies of peimine and peiminine in rat plasma by LC-MS-MS after oral administration of Fritillaria thunbergii Miq. and Fritillaria thunbergii Miq.—Glycyrrhiza uralensis Fisch. couple extract. Pharmazie 2011, 66, 684–689. [Google Scholar] [CrossRef]
- Chen, L.-H.; Zhang, H.-M.; Guan, Z.-Y.; Zhu, W.-F.; Yi, W.-J.; Guan, Y.-M.; Wang, S.; Liu, H.-N. Sex dependent pharmacokinetics, tissue distribution and excretion of peimine and peiminine in rats assessed by liquid chromatography–tandem mass spectrometry. J. Ethnopharmacol. 2013, 145, 77–84. [Google Scholar] [CrossRef] [PubMed]
- Jiang, W.; Yang, Y.; Li, Y. Effects of some drugs for resolving phlegm on blood rehological property in rats. J. Tradit. Chin. Med. 2002, 3, 38. [Google Scholar]
- Li, Y.; Zhou, H.; Xie, J.; Ally, M.S.; Hou, Z.; Xu, Y.; Zhang, Y. A novel method for evaluating the cardiotoxicity of traditional Chinese medicine compatibility by using support vector machine model combined with metabonomics. Evid. Based Complement. Altern. Med. 2016, 2016, e6012761. [Google Scholar] [CrossRef]
- Li, Z.; An, C.; Hu, K.; Zhou, K.; Duan, H.; Tang, M. Multidrug resistance reversal activity of total alkaloid from Fritillaria thunbergii on cisplatin-resistant human lung adenocarcinoma A549/DDP cells. Chin. J. Pharmacol. Toxicol. 2013, 27, 315–320. [Google Scholar]
- Li, Z.; Qiao, Y.; Li, J.; An, C.; Hu, K.; Tang, M. Acute and sub-chronic toxicity studies of the extract of Thunberg Fritillary Bulb. Regul. Toxicol. Pharmacol. 2014, 68, 370–377. [Google Scholar] [CrossRef]
- Lin, M.; Zhou, Z.; Wang, L. Protection effect of Fritillaria thunbergii aginst hyperthyroidism in rats and mice. China Pharm. 2010, 21, 1362–1363. [Google Scholar]
- Lin, X.; Peng, X. Experimental study on percutaneous absorption of main alkaloids in Fritillaira Thunbergii in vitro. Asia-Pac. Tradit. Med. 2011, 7, 15–18. [Google Scholar]
- Ma, W. Antioxidant activity of polysaccharide from Fritillaria thunbergii Bulbus in vitro. Chin. Arch. Tradit. Chin. Med. 2014, 32, 1191–1193. [Google Scholar]
- Ruan, X.; Yang, L.; Cui, W.-X.; Zhang, M.-X.; Li, Z.-H.; Liu, B.; Wang, Q. Optimization of supercritical fluid extraction of total alkaloids, peimisine, peimine and peiminine from the Bulb of Fritillaria thunbergii Miq, and evaluation of antioxidant activities of the extracts. Materials 2016, 9, 524. [Google Scholar] [CrossRef]
- Wang, L.; Han, C.; Wang, P. Pharmacodynamic compariason on anti-tussive and expectorant effects between Fritillaria anhuiensis S.C.Chen et S.P.Yin, Fritillariae Cirrhosae Bulbus and Fritillariae Thunbergii Bulbus. Anhui Med. J. 1993, 14, 57–58. [Google Scholar]
- Xia, J.; Han, L.; Zhou, X.; Wei, B. The effect of Zhejiang Fritillaria Thunbergii against immunological CP/CPPS. Chin. Arch. Tradit. Chin. Med. 2011, 28, 1023–1025. [Google Scholar]
- Xu, J.; Zhao, W.; Pan, L.; Zhang, A.; Chen, Q.; Xu, K.; Lu, H.; Chen, Y. Peimine, a main active ingredient of Fritillaria, exhibits anti-inflammatory and pain suppression properties at the cellular level. Fitoterapia 2016, 111, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Yan, X.; Tong, Z.; Luo, Y.; Tang, L.; Wu, S.; Yang, J.; Peng, C. Pharmacodynamic compariason of Fritillaria unibracteata, Fritillaria unibracteata var. wabensis and Fritillaria thunbergii cultivated. Chin. J. Exp. Tradit. Med. Formulae 2012, 18, 244–248. [Google Scholar]
- Yan, Z.; Luo, Y.; Li, Z.; Tang, L.; Wu, S.; Yan, X.; Peng, C. Comparative studies on antiussive effect between Fritillaria unibracteata Hisao et K. C. Hisa and Fritillaria thunbergii Miq with chemical stimulation induced cough method. Lishizhen Med. Mater. Med. Res. 2012, 23, 2522–2525. [Google Scholar]
- Yang, Q.; Nie, S.; Weng, X.; Li, L.; Huang, L. Experiment studies on anti-tumor effect in vivo and in vitro of Aconitum carmichaelii Debx. and Fritillaria thunbergii Miq. used singly or matched. Chin. J. Exp. Tradit. Med. Formulae 2005, 11, 25–28. [Google Scholar]
- Zhang, M.; Shen, Y.; Zhu, Z.; Wang, H.; Li, F. Study on the anti-ulcer and analgesia effects of Fritillaria thunbergii Miq. Northwest Pharm. J. 1998, 13, 208–209. [Google Scholar]
- Zhang, M.; Shen, Y.; Zhu, Z.; Wang, H.; Ma, D. Study on the anti-inflammatory and anti-diarrhea effects of Fritillaria thunbergii Miq. Hunan Guiding J. Tradit. Chin. Med. Pharmacol. 1998, 4, 30–31. [Google Scholar]
- Zhang, W.; Xu, T.; Wang, G. Effect of extracts from Fritillaria thunbergii on oral ulcer model in rats. Mod. Pract. Med. 2018, 30, 242–243. [Google Scholar]
- Jian, B.; Zhang, H.; Han, C.; Liu, J. Anti-cancer activities of diterpenoids derived from euphorbia fischeriana steud. Molecules 2018, 23, 23020387. [Google Scholar] [CrossRef]
- Rohr, M.; Oleinikov, K.; Jung, M.; Sandjo, L.P.; Opatz, T.; Erkel, G. Anti-inflammatory tetraquinane diterpenoids from a Crinipellis species. Bioorg. Med. Chem. 2017, 25, 514–522. [Google Scholar] [CrossRef]
- Kolak, U.; Kabouche, A.; Ozturk, M.; Kabouche, Z.; Topcu, G.; Ulubelen, A. Antioxidant diterpenoids from the roots of Salvia barrelieri. Phytochem. Anal. 2009, 20, 320–327. [Google Scholar] [CrossRef]
- Hattori, H.; Tanaka, M.; Fukushima, M.; Sasaki, T.; Matsuda, A. Nucleosides and nucleotides. 158. 1-(3-C-ethynyl-beta-D-ribo-pentofuranosyl)-cytosine, 1-(3-C-ethynyl-beta-D-ribo-pentofuranosyl)uracil, and their nucleobase analogues as new potential multifunctional antitumor nucleosides with a broad spectrum of activity. J. Med. Chem. 1996, 39, 5005–5011. [Google Scholar] [CrossRef] [PubMed]
- Ichikawa, S.; Yamaguchi, M.; Matsuda, A. Antibacterial nucleoside natural products inhibiting phospho-murNAc-pentapeptide translocase: Chemistry and structure-activity relationship. Curr. Med. Chem. 2015, 22, 3951–3979. [Google Scholar] [CrossRef] [PubMed]
- Ichikawa, S.; Matsuda, A. Nucleoside natural products and related analogs with potential therapeutic properties as antibacterial and antiviral agents. Expert Opin. Ther. Pat. 2007, 17, 487–498. [Google Scholar] [CrossRef]
- Jan, A.T.; Azam, M.; Siddiqui, K.; Ali, A.; Choi, I.; Haq, Q.M. Heavy metals and human health: Mechanistic insight into toxicity and counter defense system of antioxidants. Int. J. Mol. Sci. 2015, 16, 29592–29630. [Google Scholar] [CrossRef]
- World Health Organization. Cancer. Available online: https://www.who.int/cancer/en/ (accessed on 22 January 2019).
- National Cancer Institute. Side Effects of Cancer Treatment. Available online: https://www.cancer.gov/about-cancer/treatment/side-effects (accessed on 22 January 2019).
- Cao, H.; Mu, Y.; Li, X.; Wang, Y.; Chen, S.; Liu, J.P. A Systematic Review of Randomized Controlled Trials on Oral Chinese Herbal Medicine for Prostate Cancer. PLoS ONE 2016, 11, e0160253. [Google Scholar] [CrossRef]
- Lin, X.; Peng, Z.; Su, C. Potential anti-cancer activities and mechanisms of costunolide and dehydrocostuslactone. Int. J. Mol. Sci. 2015, 16, 10888–10906. [Google Scholar] [CrossRef]
- Neubig, R.R.; Spedding, M.; Kenakin, T.; Christopoulos, A. International Union of Pharmacology Committee on Receptor Nomenclature and Drug Classification. XXXVIII. Update on terms and symbols in quantitative pharmacology. Pharmacol. Rev. 2003, 55, 597–606. [Google Scholar] [CrossRef]
- Koo, G.C.; Blake, J.T.; Talento, A.; Nguyen, M.; Lin, S.; Sirotina, A.; Shah, K.; Mulvany, K.; Hora, D., Jr.; Cunningham, P.; et al. Blockade of the voltage-gated potassium channel Kv1.3 inhibits immune responses in vivo. J. Immunol. 1997, 158, 5120–5128. [Google Scholar] [PubMed]
- Li, Z.; Liu, W.H.; Han, S.; Peng, B.W.; Yin, J.; Wu, Y.L.; He, X.H.; Li, W.X. Selective inhibition of CCR7(-) effector memory T cell activation by a novel peptide targeting Kv1.3 channel in a rat experimental autoimmune encephalomyelitis model. J. Biol. Chem. 2012, 287, 29479–29494. [Google Scholar] [CrossRef]
- Catterall, W.A.; Goldin, A.L.; Waxman, S.G. International Union of Pharmacology. XLVII. Nomenclature and structure-function relationships of voltage-gated sodium channels. Pharmacol. Rev. 2005, 57, 397–409. [Google Scholar] [CrossRef]
- Raymond, C.K.; Castle, J.; Garrett-Engele, P.; Armour, C.D.; Kan, Z.; Tsinoremas, N.; Johnson, J.M. Expression of alternatively spliced sodium channel alpha-subunit genes. Unique splicing patterns are observed in dorsal root ganglia. J. Biol. Chem. 2004, 279, 46234–46241. [Google Scholar] [CrossRef] [PubMed]
- Rush, A.M.; Dib-Hajj, S.D.; Liu, S.; Cummins, T.R.; Black, J.A.; Waxman, S.G. A single sodium channel mutation produces hyper- or hypoexcitability in different types of neurons. Proc. Natl. Acad. Sci. USA 2006, 103, 8245–8250. [Google Scholar] [CrossRef]
- Cox, J.J.; Reimann, F.; Nicholas, A.K.; Thornton, G.; Roberts, E.; Springell, K.; Karbani, G.; Jafri, H.; Mannan, J.; Raashid, Y.; et al. An SCN9A channelopathy causes congenital inability to experience pain. Nature 2006, 444, 894–898. [Google Scholar] [CrossRef] [PubMed]
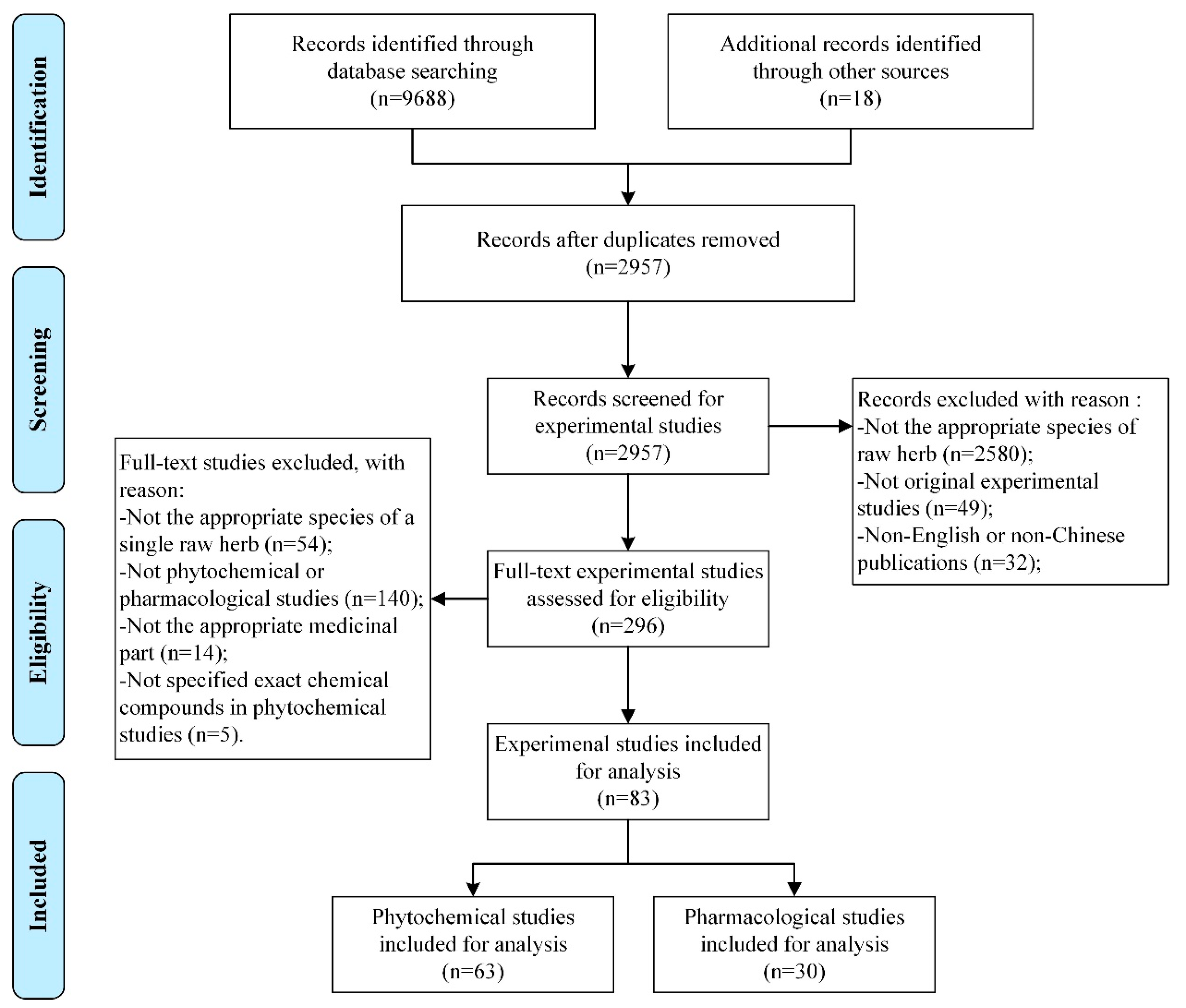
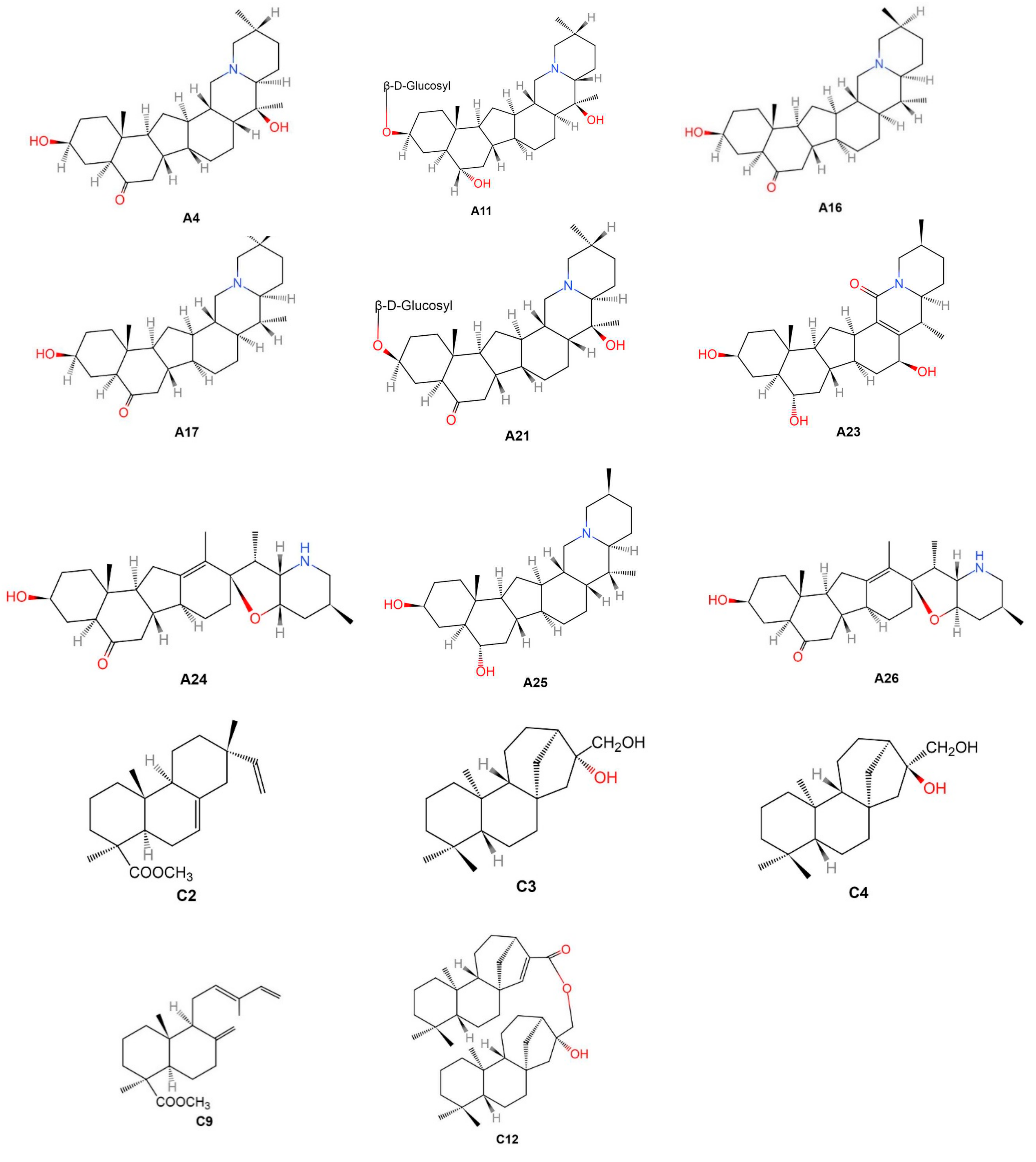
| No. | Derivatives and Constitutions | Molecular Formula | PubChem CID/SID | Molecule Weight (g/mol) | Method | |
|---|---|---|---|---|---|---|
| SRV Group | SNRV Group | |||||
| A | Alkaloids (n = 26) | |||||
| A1 | Peimine | C27H45NO3 | 131900 | 431.661 | HPLC [33]; HPLC-ELSD [44,60,63]; HPLC-ESI/MS [70]; HPLC-MS [21]; UPLC-ELSD [26]; LC/ESI-QTOF-MS/MS [72]; Pre-column derivatization HPLC [38]; GC [55]; 1HNMR/13CNMR/HR-FAB-MS [8] | HPLC [18,20]; HPLC-ELSD [17,27,28,29,32,36,47,51]; HSCCC-ELSD [31]; UPLC-CAD/HPLC-ELSD [42]; UHPLC-MS/MS [59]; LC-LTQ-Orbitrap MSn [22]; LC-TRAP-MS/LC-ESI-MS [45]; ESI-MS [73]; TLCS [23,48,57]; 13CNMR [69]; Determination of colorimetry [23]; RRLC-MS/MS [49]; MS/IR/1HNMR/13CNMR [75]; GC [37]; GC direct [17]; Indirect UV detection [41]; Paper partition chromatography [46]; Aid-dye ofnrimetry [24,39] |
| A2 | Peiminine | C27H43NO3 | 167691 | 429.645 | HPLC-MS [21]; HPLC-ELSD [60]; HPLC-ESI/MS [70]; Pre-column derivatization HPLC [38]; UPLC-ELSD [26]; LC/ESI-QTOF-MS/MS [72]; GC [56]; 1HNMR/13CNMR/HR-FAB-MS [8]; Acid dye two-phase titration [50] | HPLC [17,18,20]; HPLC-ELSD [27,28,29,32,36,47]; HPLC-CAD/HPLC-ELSD [42]; HSCCC-ELSD [31]; ESI-MS [73]; UHPLC–MS/MS [59]; TLCS [23,48,57]; LC-LTQ-Orbitrap MSn [22]; LC-TRAP-MS/LC-ESI-MS [45]; 13CNMR [69]; Determination of colorimetry [23]; RRLC-MS/MS [49]; MS/IR/1HNMR/13CNMR [75]; GC [37]; GC direct [17]; Indirect UV detection [41] |
| A3 | Zhebeinine | C27H45NO3 | 21121503 | 431.661 | N/A | TLC [69]; 13CNMR [69] |
| A4 | Zhebeinone | C27H43NO3 | NF (Figure 2) | 429.645 | N/A | GC-MS [65] |
| A5 | Ebeiedine | C27H45NO2 | 101324888 | 415.662 | HPLC [33]; GC [56]; LC/ESI-QTOF-MS/MS [72]; 1HNMR/13CNMR/HR-FAB-MS [8] | GC [37]; GC direct [17]; LC-TRAP-MS/LC-ESI-MS [45] |
| A6 | Ebeiedinone | C27H43NO2 | 102062796 | 413.646 | GC [56]; 1HNMR/13CNMR/HR-FAB-MS [8] | GC [37]; GC direct [17]; LC-TRAP-MS/LC-ESI-MS [44]; LC-LTQ-Orbitrap MSn [22]; HPLC-ELSD [17,36] |
| A7 | Isoverticine | C27H45NO3 | 21573744 | 431.661 | GC [56]; LC/ESI-QTOF-MS/MS [72]; 1HNMR/13CNMR/HR-FAB-MS [8] | GC [37]; GC direct [17]; LC-TRAP-MS/LC-ESI-MS [45]; HPLC-ELSD [17,36]; ESI-MS [73]; TLCS [57] |
| A8 | Suchengbeisine | C27H43NO3 | 102112537 | 429.645 | HPLC [33]; 1HNMR/13CNMR/HR-FAB-MS [8] | N/A |
| A9 | Peimisine | C27H41NO3 | 161294 | 427.629 | LC/ESI-QTOF-MS/MS [72]; UPLC-ELSD [26] | LC-TRAP-MS/LC-ESI-MS [45]; HPLC-ELSD [17,36,47]; TLC [68]; TLCS [57]; LC-LTQ-Orbitrap MSn [22]; RRLC-MS/MS [49] |
| A10 | Peimisine-N-oxide | C27H42NO4 | NF | 444.636 | N/A | LC-LTQ-Orbitrap MSn [22] |
| A11 | Zhebeininoside | C33H55NO8 | NF (Figure 2) | 593.802 | LC/ESI-QTOF-MS/MS [72] | LC-TRAP-MS/LC-ESI-MS [45]; LC-LTQ-Orbitrap MSn [22]; 13CNMR/Infrared spectra analysis [67]; ESI-MS [73] |
| A12 | Verticinone-3-β-d-glucoside | C33H53NO8 | 90479257 | 591.786 | LC/ESI-QTOF-MS/MS [72] | N/A |
| A13 | Puqietinone | C28H47NO2 | 10693900 | 429.689 | LC/ESI-QTOF-MS/MS [72] | N/A |
| A14 | Puqiedinone | C27H43NO2 | 126149 | 413.646 | LC/ESI-QTOF-MS/MS [72] | LC-TRAP-MS/LC-ESI-MS [45]; LC-LTQ-Orbitrap MSn [22] |
| A15 | Puqiedine | C27H45NO2 | 101400593 | 415.662 | LC/ESI-QTOF-MS/MS [72] | LC-TRAP-MS/LC-ESI-MS [45]; ESI-MS [73] |
| A16 | Eduardine | C27H43NO2 | NF (Figure 2) | 413.646 | N/A | GC-MS [66] |
| A17 | Zhebeirine | C27H43NO2 | NF (Figure 2) | 413.646 | N/A | GC-MS [66]; ESI-MS [73] |
| A18 | 2,3-deoxyladenosine | C10H11N5O2 | NF | 233.231 | N/A | ESI-MS [73] |
| A19 | N-demethylpuqietinone | C27H45NO2 | 11304576 | 415.662 | N/A | ESI-MS [73] |
| A20 | Puqienine B | C28H45NO2 | 11419389 | 443.672 | N/A | ESI-MS [73] |
| A21 | Zhebeinone-3-β-d-glucoside | C33H55NO8 | NF (Figure 2) | 593.802 | N/A | ESI-MS [73] |
| A22 | Peiminoside | C33H55NO7 | 90479565 | 577.803 | N/A | Paper partition chromatography [46] |
| A23 | Frithunbol A | C27H41NO4 | NF (Figure 2) | 443.628 | 1HNMR/13CNMR/HR-FAB-MS [8] | N/A |
| A24 | Frithunbol B | C27H42NO3 | NF (Figure 2) | 428.637 | 1HNMR/13CNMR/HR-FAB-MS [8] | N/A |
| A25 | Eduardinine | C27H45NO2 | NF (Figure 2) | 415.662 | 1HNMR/13CNMR/HR-FAB-MS [8] | N/A |
| A26 | 3β-hydroxy-5α-jervanin-12-en-6-one | C27H42NO3 | NF (Figure 2) | 428.637 | 1HNMR/13CNMR/HR-FAB-MS [8] | N/A |
| B | Compounds from essential oils (n = 29) | |||||
| B1 | δ-elemene | C15H24 | 12309449 | 204.357 | N/A | GC-MS [16] |
| B2 | δ-selinene | C15H24 | 520383 | 204.357 | N/A | GC-MS [16] |
| B3 | Tetradecanoic acid | C14H28O2 | 11005 | 228.376 | N/A | GC-MS [16] |
| B4 | Pentadecanoic acid | C15H30O2 | 13849 | 242.403 | N/A | GC-MS [16] |
| B5 | Hexadecanoic acid, methyl ester | C17H34O2 | 8181 | 270.457 | N/A | GC-MS [16,25] |
| B6 | 9-hexadecenoic acid | C16H30O2 | 5282745 | 254.414 | N/A | GC-MS [16] |
| B7 | N-hexadecanoic acid | C16H32O2 | 985 | 256.43 | N/A | GC-MS [16] |
| B8 | Kaur-15-ene | C20H32 | 521318 | 272.476 | N/A | GC-MS [16] |
| B9 | Heptadecanoic acid | C17H34O2 | 10465 | 270.457 | N/A | GC-MS [16] |
| B10 | Kaurene | C20H32 | 91746569 | 272.476 | N/A | GC-MS [16] |
| B11 | 9,12-Octadecadienoic acid (Z,Z)-, methyl ester | C19H34O2 | 5284421 | 294.479 | N/A | GC-MS [16] |
| B12 | 9-Tetradecenal, (Z)- | C14H26O | 5364471 | 210.361 | N/A | GC-MS [16] |
| B13 | 9,12-Octadecadienoic acid, methyl ester, (E, E)- | C19H34O2 | 135058711 | 294.479 | N/A | GC-MS [16] |
| B14 | Oleic acid | C18H34O2 | 445639 | 282.468 | N/A | GC-MS [16] |
| B15 | Linoleic acid, ethyl ester | C2036O2 | 5282184 | 308.506 | N/A | GC-MS [16] |
| B16 | Butylated hydroxytoluene | C15H24O | 31404 | 220.356 | N/A | GC-MS (Du et al., 2018) |
| B17 | L-(+)-Ascorbic acid 2,6- dihexadecanoate | C38H68O8 | 54722209 | 652.954 | N/A | GC-MS (Du et al., 2018) |
| B18 | Ethyl 9-hexadecenoate | C18H34O2 | 5364759 | 282.468 | N/A | GC-MS [25] |
| B19 | Hexadecanoic acid, ethyl ester | C18H36O2 | 12366 | 284.484 | N/A | GC-MS [25] |
| B20 | 1H-Naphtho [2,1-B] pyran, 3-ethenyldodecahydro-3,4a,7,7,10a-pentamethyl- | C20H34O | 273540178 | 290.491 | N/A | GC-MS [25] |
| B21 | Kaur-16-ene | C20H32O | 520687 | 272.476 | N/A | GC-MS [25] |
| B22 | 9,11-Octadecadienoic acid, methyl ester, (E, E)- | C19H34O2 | 319301067 | 294.479 | N/A | GC-MS [25] |
| B23 | 9,12-Octadecadienoic acid | C18H32O2 | 5282457 | 280.452 | N/A | GC-MS [25] |
| B24 | Linoleic acid, ethyl ester | C20H36O2 | 5282184 | 308.506 | N/A | GC-MS [25] |
| B25 | Octadecanoic acid, ethyl ester | C20H40O2 | 8122 | 312.538 | N/A | GC-MS [25] |
| B26 | 2(1H)-Phenanthrenone, 3,4,4a,4b,5,6,7,8,10,10a-decahydro- 1,1,4a,7,7-pentamethyl, [4aR-(4a.α., 4b.β.,10a.β.)]- | C19H30O | 621255 | 274.448 | N/A | GC-MS [25] |
| B27 | 3-Methyleneandrostan-17-ol | C20H32O | 625647 | 288.475 | N/A | GC-MS [25] |
| B28 | Androst-4-en-3-one, 17-hydroxy-, (17.β.) | C19H28O2 | 50049744 | 288.431 | N/A | GC-MS [25] |
| B29 | Podocarp-7-en-3.β.-ol, 13.β.-methyl-13-vinyl- | C20H32O | 620519 | 288.475 | N/A | GC-MS [25] |
| C | Diterpenoids (n = 13) | |||||
| C1 | Isopimaran-19-ol | C20H32O | 75399514 | 288.475 | N/A | 1HNMR/13CNMR [35] |
| C2 | Isopimaran-19-oic acid, methyl ester | C21H32O2 | NF (Figure 2) | 316.485 | N/A | 1HNMR/13CNMR [34,35] |
| C3 | Ent-kauran-16β, 17-diol | C20H34O2 | NF (Figure 2) | 306.49 | N/A | TLC [68]; 1HNMR/13CNMR [34,35] |
| C4 | Ent-kauran-16α, 17-diol | C20H34O2 | NF (Figure 2) | 306.49 | N/A | 1HNMR/13CNMR [34,35] |
| C5 | Ent-16β, 17-epoxy-kaurane | C20H32O | 79592848 | 288.475 | N/A | 1HNMR/13CNMR [35] |
| C6 | Ent-16α-methoxy-kauran-17-ol | C21H36O2 | 51842049 | 320.517 | N/A | 1HNMR/13CNMR [35] |
| C7 | Ent-kaur-15-en-17-ol | C20H32O | 3082069 | 288.475 | N/A | 1HNMR/13CNMR [35] |
| C8 | Trans-communol | C20H32O | 51909318 | 288.475 | N/A | 1HNMR/13CNMR [34] |
| C9 | Trans-comminic acid, methyl ester | C21H32O2 | NF (Figure 2) | 316.485 | N/A | 1HNMR/13CNMR [34] |
| C10 | Ent-17-norkauran-16-one | C19H30O | 12740861 | 274.448 | N/A | 1HNMR/13CNMR [34] |
| C11 | Ent-15β,16-epoxy-kauran-17-ol | C20H32O2 | 51511087 | 304.474 | N/A | 1HNMR/13CNMR [34] |
| C12 | Ent-16β-hydroxy-kauran-17-yl ent-kaur-15-en-17-oate | C40H63O3 | NF (Figure 2) | 591.941 | N/A | 1HNMR/13CNMR [34] |
| C13 | Ent-(16S)-atisan-13, 17-oxide | C20H32O | 50418337 | 288.475 | N/A | 1HNMR/13CNMR [34] |
| D | Carbohydrates (n = 2) | |||||
| D1 | β-d-glucose4-1β-d-galactose | C12H22O11 | NF | 342.297 | N/A | HPLC-ELSD [19] |
| D2 | Sucrose | C12H22O11 | 5988 | 342.297 | N/A | HPLC-ELSD [19] |
| E | Sterols (n = 2) | |||||
| E1 | β-sitosterol | C29H50O | 222284 | 414.718 | N/A | TLC [68] |
| E2 | Daucosterol | C35H60O6 | 5742590 | 576.859 | N/A | TLC [68] |
| F | Amino acids (n = 18) | |||||
| F1 | Glycine | C2H5NO2 | 750 | 75.067 | N/A | HPLC [64] |
| F2 | Leucine | C6H13NO2 | 6106 | 131.175 | N/A | HPLC [64] |
| F3 | Methionine | C5H11NO2S | 6137 | 149.208 | N/A | HPLC [64] |
| F4 | Tyrosine | C9H11NO3 | 6057 | 181.191 | N/A | HPLC [64] |
| F5 | Histidine | C6H9N3O2 | 6274 | 155.157 | N/A | HPLC [64] |
| F6 | Threonine | C4H9NO3 | 6288 | 119.12 | N/A | HPLC [64] |
| F7 | Alanine | C3H7NO2 | 5950 | 89.094 | N/A | HPLC [64] |
| F8 | Isoleucine | C6H13NO2 | 6306 | 131.175 | N/A | HPLC [64] |
| F9 | Tryptophan | C11H12N2O2 | 6305 | 204.229 | N/A | HPLC [64] |
| F10 | Cystine | C6H12N2O4S2 | 67678 | 240.292 | N/A | HPLC [64] |
| F11 | Lysine | C6H14N2O2 | 5962 | 146.19 | N/A | HPLC [64] |
| F12 | Aspartic acid | C4H7NO4 | 5960 | 133.103 | N/A | HPLC [64] |
| F13 | Valine | C5H11NO2 | 6287 | 117.148 | N/A | HPLC [64] |
| F14 | Phenylalanine | C8H8O2 | 6140 | 165.192 | N/A | HPLC [64] |
| F15 | Proline | C5H9NO2 | 145742 | 115.132 | N/A | HPLC [64] |
| F16 | Serine | C3H7NO3 | 5951 | 105.093 | N/A | HPLC [64] |
| F17 | Glutamic acid | C5H9NO4 | 33032 | 147.13 | N/A | HPLC [64] |
| F18 | Arginine | C6H14N4O2 | 6322 | 174.204 | N/A | HPLC [64] |
| G | Nucleosides (n = 6) | |||||
| G1 | Uridine | C9H12N2O6 | 6029 | 244.203 | LC-ESI-MSn [70]; HPLC-UV-ESI/MS [70]; HPLC [71] | HPLC [64] |
| G2 | Guanosine | C10H13N5O5 | 6802 | 283.244 | LC-ESI-MSn (Zhang, 2008); HPLC-UV-ESI/MS [70]; HPLC [71] | HPLC [64] |
| G3 | Adenosine | C10H13N5O4 | 60961 | 267.245 | LC-ESI-MSn (Zhang, 2008); HPLC-UV-ESI/MS [70]; HPLC [71] | HPLC [64] |
| G4 | Thymidine | C10H14N2O5 | 5789 | 242.231 | LC-ESI-MSn [70]; HPLC-UV-ESI/MS [70] | HPLC [64] |
| G5 | Cytidine | C9H13N3O5 | 6175 | 243.219 | N/A | HPLC [64] |
| G6 | Inosine | C10H12N4O5 | 6021 | 268.229 | N/A | HPLC [64] |
| H | Nucleobases (n = 4) | |||||
| H1 | Adenine | C5H5N5 | 190 | 135.13 | LC-ESI-MSn (Zhang, 2008); HPLC-UV-ESI/MS (Zhang, 2008) | HPLC [64] |
| H2 | Hypoxanthine | C5H4N4O | 790 | 136.114 | N/A | HPLC [64] |
| H3 | Uracil | C4H4N2O2 | 1174 | 112.088 | N/A | HPLC [64] |
| H4 | Thymine | C5H6N2O2 | 1135 | 126.115 | N/A | HPLC [64] |
| I | Fatty acids (n = 4) | |||||
| I1 | Vernolic acid | C18H32O3 | 6449780 | 296.451 | N/A | ESI-MS [73] |
| I2 | 2-monopalmitin | C19H38O4 | 123409 | 330.509 | N/A | ESI-MS [73] |
| I3 | 13(R)-hydroxy-octadeca-(9Z,11E ,15Z)-trien-oic acid | C18H30O3 | 643726 | 294.435 | N/A | ESI-MS [73] |
| I4 | Picropodophyllotoxin | C22H22O8 | 72435 | 414.41 | N/A | 13CNMR/Infrared spectra analysis [67] |
| J | Lignans (n = 3) | |||||
| J1 | Octahydrocurcumin | C21H28O6 | 11068834 | 376.449 | N/A | ESI-MS [73] |
| J2 | Zhebeiresinol | C14H16O6 | 192547 | 280.276 | N/A | ESI-MS [73]; 1HNMR/13CNMR [30] |
| J3 | Sauriol B | C21H28O6 | 15965508 | 376.449 | N/A | ESI-MS [73] |
| K | Elements (n = 27) | |||||
| K1 | Aluminum | Al | 5359268 | 26.982 | ICP-OES [62] | FAAS [61]; GFAAS [61] |
| K2 | Arsenic | As | 5359596 | 74.922 | ICP-AES [43]; ICP-OES [14] | ICP-AES [74]; ICP-OES [15]; ICAP [55]; FAAS [61]; GFAAS [61] |
| K3 | Boron | B | 5462311 | 10.81 | ICP-OES [14,62] | N/A |
| K4 | Barium | Ba | 5355457 | 137.327 | ICP-AES [58]; ICP-OES [14] | N/A |
| K5 | Bismuth | Bi | 5359367 | 208.98 | ICP-OES [14] | N/A |
| K6 | Calcium | Ca | 5460341 | 40.078 | ICP-OES [62] | FAAS [52,53,61]; GFAAS [61] |
| K7 | Cadmium | Cd | 23973 | 112.414 | ICP-AES [43]; ICP-OES [14,62] | ICP-AES [74]; ICP-OES [15]; ICAP [55]; FAAS [61]; GFAAS [61]; AAS [40] |
| K8 | Cobalt | Co | 104730 | 58.933 | ICP-AES [58]; ICP-OES [14] | FAAS [61]; GFAAS [61] |
| K9 | Chromium | Cr | 23976 | 51.996 | ICP-AES [58] | FAAS [61]; GFAAS [61]; AAS [40] |
| K10 | Copper | Cu | 23978 | 63.546 | ICP-AES [43,58]; ICP-OES [14] | ICP-AES [74]; ICP-OES [15]; ICAP [55]; FAAS [52,53,61]; GFAAS [61]; AAS [40] |
| K11 | Iron | Fe | 23925 | 55.845 | ICP-AES [43,58]; ICP-OES [14] | ICP-AES [74]; FAAS [52,53,61]; GFAAS [61]; AAS [40] |
| K12 | Mercury | Hg | 23931 | 200.592 | ICP-AES [43]; ICP-OES [14] | ICP-OES [15] |
| K13 | Indium | In | 5359967 | 114.818 | ICP-OES [14] | N/A |
| K14 | Potassium | K | 5462222 | 39.098 | ICP-OES [62] | FAAS [53,61]; GFAAS [61] |
| K15 | Lithium | Li | 3028194 | 6.94 | ICP-AES [43] | N/A |
| K16 | Magnesium | Mg | 5462224 | 24.305 | ICP-AES [43]; ICP-OES [62] | FAAS [53,61]; GFAAS [61]; AAS [40] |
| K17 | Manganese | Mn | 23930 | 54.938 | ICP-AES [43,58]; ICP-OES [14] | ICP-AES [74]; FAAS [53,61]; GFAAS [61]; ICAP [55] |
| K18 | Molybdenum | Mo | 23932 | 95.95 | ICP-OES [62] | N/A |
| K19 | Sodium | Na | 5360545 | 22.99 | ICP-OES [62] | FAAS [53,61]; GFAAS [61] |
| K20 | Nickel | Ni | 935 | 58.693 | ICP-AES [58]; ICP-OES [14,62] | ICAP [55] |
| K21 | Phosphorus | P | 5462309 | 30.974 | ICP-OES [62] | N/A |
| K22 | Lead | Pb | 5352425 | 207.2 | ICP-AES [43]; ICP-OES [14] | ICP-OES [15]; AAS [40]; ICAP [55] |
| K23 | Sulfur | S | 5362487 | 32.06 | ICP-AES [43] | N/A |
| K24 | Selenium | Se | 6326970 | 78.971 | N/A | Spectrophotometry [54] |
| K25 | Strontium | Sr | 5359327 | 87.62 | ICP-AES [43,58]; ICP-OES | FAAS [61]; GFAAS [61] |
| K26 | Vanadium | V | 23990 | 50.941 | ICP-AES [58] | N/A |
| K27 | Zinc | Zn | 23994 | 65.379 | ICP-AES [43,58]; ICP-OES [14] | ICP-AES [74]; ICP-OES; ICAP [55]; FAAS [52,53,61]; GFAAS [61]; AAS [40] |
| Pharmacological Effects/ Included Studies | Study Type | Extract | Characteristics of the Sample | Interventions | Duration | Primary Results |
|---|---|---|---|---|---|---|
| 1. Anti-cancer | ||||||
| Li et al., 2013 [80] | In vitro | Total alkaloids | Human lung adenocarcinoma parental cells A549; Resistant cells A549/DDP | Cytotoxicity: FTB 12.5, 25, 50, 100, 200 mg/L and vehicle 0.5% CMC-Na. Multidrug resistance reversal effect: FTB 9 mg/L, cyclosporine A 1 mg/L or tetrandrine 1 mg/L plus DDP (final concentration: 0.01, 0.1, 1, 10, 100 mg/L). MDR1 mRNA and P-gp expression: A549/DDP + vehicle; A549/DDP + FTB 9 mg/L; A549/DDP + DDP 14 mg/L; A549/DDP + DDP 14 mg/L + FTB 9 mg/L; A549 + vehicle | 72 h | IC50 of TAF to A549: 141 ± 5 mg/L; IC50 of TAF to A549/DDP: 298 ± 22 mg/L; FTB was superior to closporine A and tetrandrines in increasing the reversal fold; FTB alone was superior to vehicle in decreasing the MDR1 mRNA and P-gp expression. |
| In vivo | Total alkaloids | 60 BALB/c nude mice (A549/DDP model) | Vehicle 0.5% CMC-Na; DDP 5 mg/kg, ig, qd; TAF 2 mg/kg, ig, qd; DDP 5 mg/kg + FTB 0.5 mg/kg, ig, qd; DDP 5 mg/kg + FTB 1 mg/kg, ig, qd; DDP 5 mg/kg + FTB 2 mg/kg, ig, qd | 13 days | DDP + TAF was superior to DDP alone in increasing the tumor inhibitory rate. | |
| Liu et al., 2015 [9] | In vitro | Total alkaloids; Total nucleosides | Human hepatocellular carcinoma cell line HepG2, Resistant cell line HepG2/MDR | Blank control; P-gp positive inhibitor verapamil; Total alkaloid, nucleoside, or polysaccharide 5 μg/mL respectively; Total alkaloid, nucleoside, or polysaccharide 50 μg/mL respectively | 1 h | Total alkaloids or Total nucleosides in FTB was superior to the control in increasing the restriction of efflux activity of P-gp. |
| Yang et al., 2005 [91] | In vitro | Aqueous extract | Human lung adenocarcinoma parental cells LM2 | Blank control; FZ + FTB (75 + 75, 25 + 25, 5 + 5, 1 + 1 mg/mL respectively); FZ (75, 25, 5 mg/mL respectively); FTB (75, 25, 5 mg/mL respectively) | 48 h | FTB alone was superior to control and FZ + FTB in increasing the apoptosis rate. |
| In vivo | Aqueous extract | 142 SPF C57 mice (Human lung adenocarcinoma parental cells LM2 model) | Blank control; FZ + FTB (0.8 + 1.6 g/kg, 0.64 + 1.28 g/kg, 0.51 + 1.02 g/kg respectively, ig, qd); FZ (0.8 g/kg, 0.64 g/kg, 0.51 g/kg respectively, ig, qd); FTB (1.6 g/kg, 1.28 g/kg, 1.02 g/kg respectively, ig, qd) | 18 days | FTB alone was superior to control and FZ + FTB in increasing the tumor inhibitory rate and reducing the number of metastases. | |
| Tong, 2016 [49] | In vitro | Peimine; Peiminine | Human breast carcinoma cells MCF-7; Resistant cell line MCF-7/ADM | Peimine 12.5–400 μg/mL; Peiminine 12.5–400 μg/mL; ADM (0.78125–100 μg/mL) + Peimine or Peiminine; Paclitaxel (2.5–80 μg/mL) + Peimine or Peiminine | 48 h | Both peimine and peiminine could reverse the multi-drug resistant tumor resistance of ADM or paclitaxel |
| 2. Tracheobronchial relaxation | ||||||
| Chan, 2000 [17] | In vitro | Peimine; Peiminine; Ebeiedine | Rat tracheal and bronchial rings | Peimine, Peiminine, Ebeiedine, Imperialine, puqietinone, Salbutamol, Diphenhydramine, Codeine cumulative concentrations 1 nM-100 μM respectively | Immediate | Peimine, peiminine, ebeiedine and puqietinone in FTB could relax the tracheobronch of rats. |
| Wu et al., 2018 [56] | In vitro | Total alkaloids | Rat tracheal and bronchial rings | Total alkaloids cumulative concentrations 0-3 g/mL | Immediate | Total alkaloids in FTB were superior to control in increasing the pD2 value. |
| 3. Antitussive | ||||||
| Yan et al., 2012 [90] | In vivo | Micro powders | 44 guinea pigs (citric acid induced cough model) | Blank control: purified water, ig,qd; AZTB: 1.5 g/kg, ig, qd; FTB: 1.5 g/kg, ig, qd; Codeine phosphate: 0.02 g/kg, ig, qd | 5 min | FTB was superior to control in reduding the frequency of cough and prolonging the remission period. |
| Yan et al., 2012 [89] | In vivo | Micro powders | 53 guinea pigs (citric acid induced cough model) | Blank control: purified water, ig,qd; FTB: 1.5 g/kg, ig, qd; AZTB: 1.5 g/kg, ig, qd; WBBM: 1.5 g/kg, ig, qd; Codeine phosphate: 0.02 g/kg, ig, qd | 5 min | FTB was superior to control in reduding the frequency of cough and prolonging the remission period. |
| Guo, 2007 [27] | In vivo | Aqueous extract | 140 Kunming mice (ammonium hydroxide induced cough model) | Blank control: NS, ig,qd; FTB: 0.026, 0.052, 0.104 g/kg respectively, ig, qd; Codeine phosphate: 0.03 g/kg, ig, qd | 3 days | FTB harvested in all four places was superior to control in reduding the frequency of cough and prolonging the remission period. |
| 4. Expectorant | ||||||
| Yan et al., 2012 [89] | In vivo | Fine powders | 40 mice | Blank control: purified water, ig,qd; FTB: 2 g/kg, ig, qd; AZTB: 2 g/kg, ig, qd; WBBM: 2 g/kg, ig, qd; Ammonium chloride: 1 g/kg, ig, qd | 5 days | Fine powders of FTB was superior to control in reducing the amount of phlegm secretion. |
| Wang et al., 1993 [86] | In vivo | Alcohol extract | 40 Wistar rats | Control: starch paste, 15 g/kg, ig,qd; FTB: 15 g/kg, ig, qd; CBM: 15 g/kg, ig, qd; WBM: 15 g/kg, ig, qd | 5 h | Alcohol extract of FTB was superior to control in reducing the amount of phlegm secretion. |
| 5. Anti-inflammation | ||||||
| Kim et al., 2016 [33] | In vitro | Peimine; Ebeiedine; Suchengbeisine | Human mucoepidermoid carcinoma cells NCI-H292 | Peimine; Ebeiedine; Suchengbeisine | 24 h | Peimine, ebeiedine, or suchengbeisine was superior to control in decreasing the expression of MUC5AC mucin gene. |
| Zhou et al., 2017 [73] | In vitro | Puqiedine; Zhebeiresinol, 2-monopalmitin, N-demethylpuqietinone; Isoverticine | Human embryonic kidney cells HEK293 | FTB 10, 3, 1 mg/mL respectively; Dexamethasone 10−5 mol/L | 6 h | Puqiedine, zhebeiresinol, 2-monopalmitin, n-demethylpuqietinone or isoverticine was superior to control in reducing the expression of NF-Κb level in cells. |
| Xia et al., 2011 [87] | In vivo | Aqueous extract | 30 mice (CP/CPPS model) | Blank control and blank normal: NS; FTB 0.1 mL/10 g, ig, qd | 7 days | Aqueous extract of FTB was superior to control in relieving the inflammation and over proliferation of fibroblasts of the prostate and reducing the serum level of nitric oxide in mice. |
| Zhang et al., 1998 [93] | In vivo | Alcohol extract | 40 ICR mice (xylene induced ear swelling) | Blank control: Purified water, ig,qd; Ethenzamide 0.3 g/kg, ig,qd; FTB 0.8, 2.4 g/kg respectively, ig, qd | 4 h | Alcohol extract of FTB was superior to control in releving the swelling of ear. |
| In vivo | Alcohol extract | 40 ICR mice (carrageenin induced foot plantar swelling) | Blank control: Purified water, ig,qd; Ethenzamide 0.3 g/kg, ig,qd; FTB 0.8, 2.4 g/kg respectively, ig, qd | 6 h | Alcohol extract of FTB was superior to control in releving the swelling of foot plantar. | |
| Xu et al., 2016 [88] | In vitro | Peimine | Human Embryonic Kidney Cells HEK293 | Peimine concentrations 1, 3 10, 30, 100, 300 μM | 300 s | Peimine could inhibit the Nav 1.3 channel. |
| 6. Pain suppression | ||||||
| Zhang et al., 1998 [92] | In vivo | Alcohol extract | 44 ICR mice (acetic acid induced pain) | Blank control: Purified water, ig,qd; Ethenzamide 0.3 g/kg, ig,qd; FTB 0.8, 2.4 g/kg respectively, ig, qd | 8 h | Alcohol extract of FTB was superior to control in reducing the frequency of mouse writhing. |
| 40 ICR mice (thermal stimulus pain) | Blank control: Purified water, ig,qd; Ethenzamide 0.3 g/kg, ig,qd; FTB 0.8, 2.4 g/kg respectively, ig, qd | 3 h | Alcohol extract of FTB was superior to control in increasing the remission period of tail-flick latency. | |||
| Xu et al., 2016 [88] | In vitro | Peimine | Human Embryonic Kidney Cells HEK293 | Peimine concentrations 1, 3 10, 30, 100, 300 μM | 300 s | Peimine could inhibit the Nav 1.7 channel. |
| 7. Antioxidation | ||||||
| Ruan et al., 2016 [85] | In vitro | Total alkaloids | DPPH radical; ABTS radical; FRAP reagent | DPPH: Total alkaloids 50 μL; ABTS: Total alkaloids 50 μL; FRAP: Total alkaloids 20 μL, control ethanol 20 μL | DPPH: 30 min; ABTS: Immediate; FRAP: Immediate | Total alkaloids in FTB have strong a antioxidative capacity evidence by the results of DPPH, ABTS and FRAP. |
| Ma, 2014 [84] | In vitro | polysaccharide | DPPH radical | polysaccharide 0.1, 0.2, 0.4, 0.6, 0.8, 1 mg/mL | 30 min | Polysaccharide in FTB has strong antioxidative capacity evidence by the results of DPPH and absorption spectroscopy test. |
| 8. Antiulcer | ||||||
| Zhang et al., 2018 [92] | In vivo | Alcohol extract | 90 SD rats (oral ulcer model) | Blank model: NS; FTB 4, 2, 1 g/kg respectively, external, qid; Gui Lin Xi Gua Shuang, 1 g/kg, external, qid | 6 days | Alcohol extract of FTB was superior to control in inhibiting oral ulcer. |
| In vivo | Alcohol extract | 120 ICR mice (gastric ulcer model) | Blank control: Purified water, ig,qd; Mepirizole 0.05 g/kg, ig,qd; FTB 0.8, 2.4 g/kg respectively, ig, qd; | 2 h | Alcohol extract of FTB was superior to control in inhibiting gastric ulcer. | |
| 9. Anti-muscarinic | ||||||
| Zhou et al., 2006 [75] | In vitro | Peimine; Peiminine; Puqietinone | Human Embryonic Kidney Cells HEK293 | Five alkaloids: 10μM (final concentration each); Negative control: carbachol, 0.3 μM (final concentration each); Positive control: Atropine 1 μM (final concentration each) | 48 h | Peimine, peiminine or puqietinone was superior to control in raising the cAMP level in cells transfected with muscarinic M2 receptor. |
| 10. Anti-thyroid | ||||||
| Lin et al., 2010 [82] | In vivo | Extract | 180 SD rats; 60 SPF mice (hyperthyroidism model) | Blank control and blank model: NS, ig, qd; Tapazole 0.02 g/kg, ig, qd; FTB 3, 1.5, 0.75 g/kg respectively, ig, qd | 14 days | FTB was superior to control in reducing the serum level of T3, T4, cAMP, cGMP and raising the abilities of hypoxia tolerance. |
| 11. Regulation of blood rheology | ||||||
| Jiang et al., 2002 [78] | In vivo | Aqueous extract | 50 SD rats | Blank control: Purified water, ig,qd; FTB 1 mL respectively, ig, qd | 1 week | Aqueous extract of FTB was superior to control in reducing the whole blood viscosity, restricting the trythrocyte aggregation and raising the erythrocyte deformability. |
| 12. Anti-diarrhea | ||||||
| Zhang et al., 1998 [93] | In vivo | Alcohol extract | 40 ICR mice (castor oil induced diarrhea); 40 ICR mice (FXY induced diarrhea) | Blank control: Purified water, ig,qd; Ethenzamide 0.3 g/kg, ig,qd; FTB 0.8, 2.4 g/kg respectively, ig, qd | 8 h | Alcohol extract of FTB was superior to control in reducing the frequency of diarrhea. |
| 13. Neuroprotection | ||||||
| Suh et al., 2018 [8] | In vitro | Frithunbol B; Ebeiedinone; 3β-hydroxy-5α-jervanin-12-en-6-one; Suchengbeisine | lipopolysaccharide-activated BV-2 cells; C6 glioma cells | NO study: 100 ng/mL isolates; NGF study: 20 μM isolates | 24 h | Frithunbol B, ebeiedinone, and suchengbeisine significantly reduce the nitric oxide level, compared to control; 3β-hydroxy-5α-jervanin-12-en-6 -one was superior to control in increasing the nerve growth factor level. |
| Included Studies | Study Type | Methods | Animals | Interventions | T1/2 (h) | Tmax (h) | CL/F (L/h/kg) | V/F (L/kg) | Cmax (μg/L) | AUC0-t (μg h/L) | AUC0-∞ (μg h/L) |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Chen et al., 2011 [76] | In vivo | LC-MS-MS; DAS 2.0 package | 12 female SD rats | Peimine, 4.25 g/kg, ig | 4.8 ± 0.8 | 1.5 ± 0.6 | 119.6 ± 40.1 | 854.8 ± 363.9 | 43.2 ± 5.4 | N/A | 260.5 ± 119.8 |
| Peiminine, 4.25 g/kg, ig | 6.6 ± 3.2 | 4.5 ± 1.9 | 34.1 ± 4.8 | 321.1 ± 155.4 | 57.6 ± 23.0 | N/A | 618.3 ± 94.8 | ||||
| Chen et al., 2013 [77] | In vivo | LC-MS-MS; DAS 2.0 package | 6 female SD rats | Peimine, 4.25 g/kg, ig | 4.2 ± 2.0 | 1.5 ± 0.7 | 128.9 ± 32.6 | 781.3 ± 305.6 | 43.7 ± 22.7 | 214.2 ± 84.6 | 214.3 ± 84.5 |
| Peiminine, 4.25 g/kg, ig | 3.4 ± 1.7 | 2.8 ± 0.9 | 36.3 ± 15.8 | 268.8 ± 163.9 | 64.2 ± 40.0 | 571.0 ± 243.4 | 571.1 ± 243.9 | ||||
| 6 SD male rats | Peimine, 4.25 g/kg, ig | 6.2 ± 1.9 | 2.9 ± 1.7 | 41.5 ± 20.1 | 374.1 ± 186.2 | 57.6 ± 21.6 | 662.4 ± 277.9 | 665.3 ± 213.3 | |||
| Peiminine, 4.25 g/kg, ig | 5.1 ± 1.4 | 3.0 ± 1.4 | 10.5 ± 2.6 | 92.2 ± 55.1 | 135.6 ± 40.2 | 1965.5 ± 433.3 | 1969.6 ± 433.5 | ||||
| Tong, 2016 [49] | In vivo | UHPLC-MS/MS; DAS 2.0 package | 6 SD male rats; 6 female SD rats | Peimine, 0.45 g/kg, ig | 2.7 ± 0.5 | 0.5 ± 0.2 | N/A | 40.8 ± 17.6 | 3.7 ± 0.9 | 10.5 ± 1.7 | 10.6 ± 1.7 |
| Peiminine, 0.45 g/kg, ig | 1.9 ± 0.8 | 0.7 ± 0.2 | N/A | 9.6 ± 5.0 | 12.6 ± 2.0 | 37.1 ± 13.7 | 37.1 ± 13.8 | ||||
| Peimisine, 0.45 g/kg, ig | 4.0 ± 1.0 | 0.8 ± 0.3 | N/A | 12.1 ± 4. 9 | 1.2 ± 0.2 | 4.6 ± 2.0 | 4.7 ± 2.1 | ||||
| Xu et al., 2017 [59] | In vivo | UHPLC-MS/MS; DAS 2.0 package | 6 male SD rats | Peimine, 20 g/kg, ig | 2.3 ±0.8 | 2.8 ± 1.4 | N/A | N/A | 74.7 ± 18.2 | 474.5 ± 143.4 | N/A |
| Peimisine, 20 g/kg, ig | 2.68 ± 0.78 | 3.3 ± 1.1 | N/A | N/A | 15.1 ± 2.0 | 120.7 ± 31.3 | N/A |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, H.; Hung, A.; Li, M.; Yang, A.W.H. Fritillariae Thunbergii Bulbus: Traditional Uses, Phytochemistry, Pharmacodynamics, Pharmacokinetics and Toxicity. Int. J. Mol. Sci. 2019, 20, 1667. https://doi.org/10.3390/ijms20071667
Li H, Hung A, Li M, Yang AWH. Fritillariae Thunbergii Bulbus: Traditional Uses, Phytochemistry, Pharmacodynamics, Pharmacokinetics and Toxicity. International Journal of Molecular Sciences. 2019; 20(7):1667. https://doi.org/10.3390/ijms20071667
Chicago/Turabian StyleLi, Hong, Andrew Hung, Mingdi Li, and Angela Wei Hong Yang. 2019. "Fritillariae Thunbergii Bulbus: Traditional Uses, Phytochemistry, Pharmacodynamics, Pharmacokinetics and Toxicity" International Journal of Molecular Sciences 20, no. 7: 1667. https://doi.org/10.3390/ijms20071667
APA StyleLi, H., Hung, A., Li, M., & Yang, A. W. H. (2019). Fritillariae Thunbergii Bulbus: Traditional Uses, Phytochemistry, Pharmacodynamics, Pharmacokinetics and Toxicity. International Journal of Molecular Sciences, 20(7), 1667. https://doi.org/10.3390/ijms20071667






