Interaction of Nerve Growth Factor β with Adiponectin and SPARC Oppositely Modulates its Biological Activity
Abstract
1. Introduction
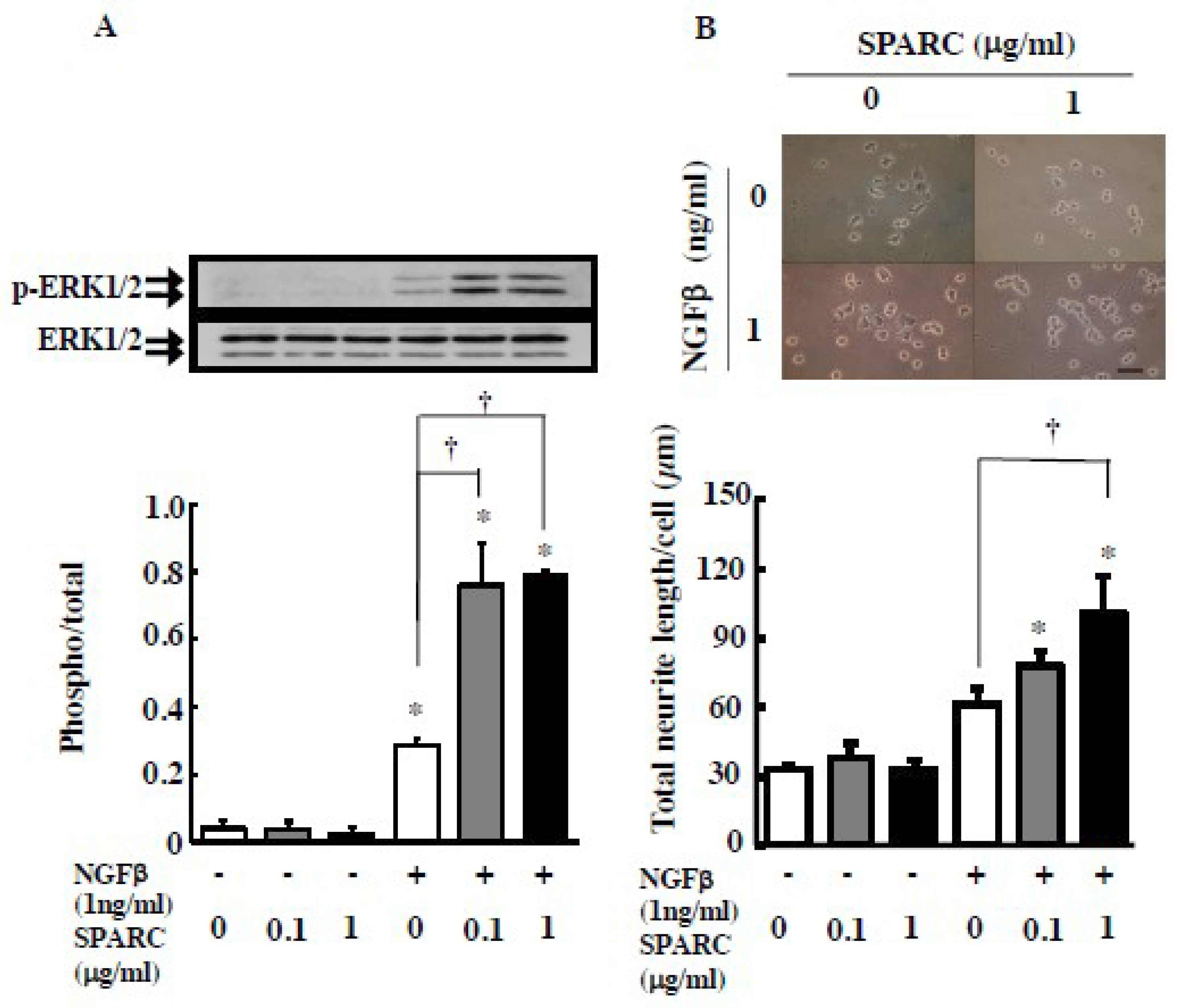
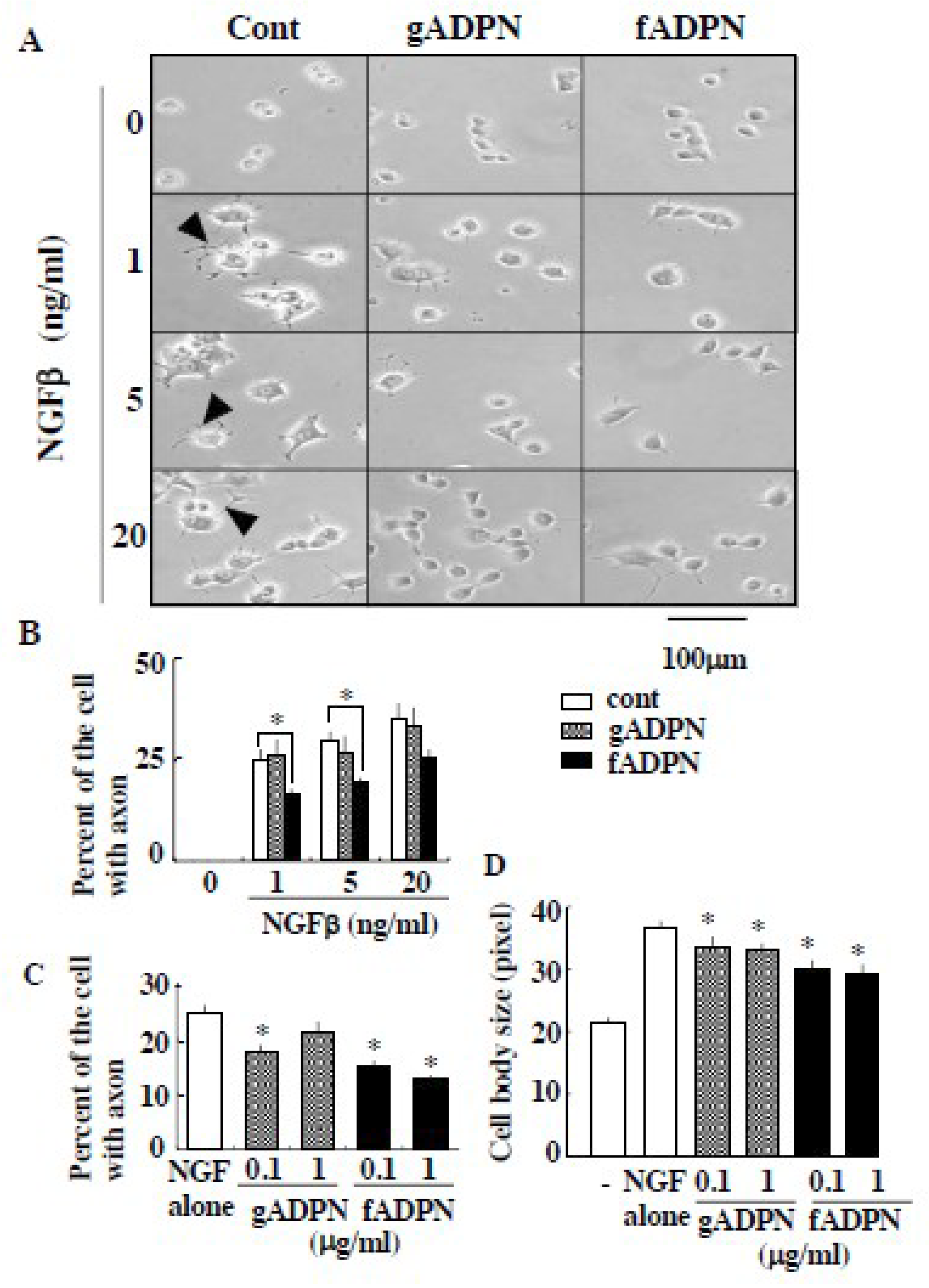
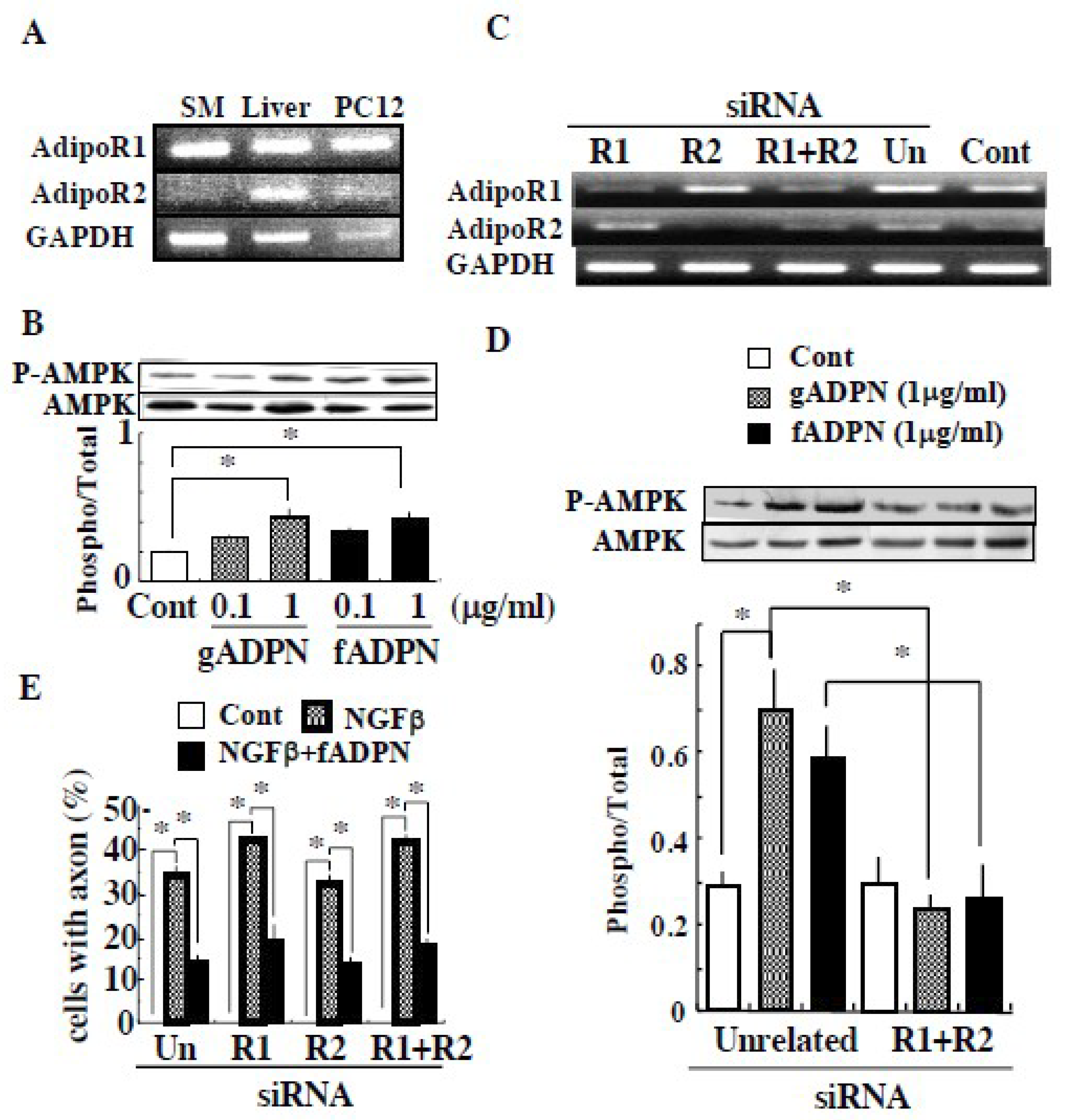
2. Results
3. Discussion
4. Materials and Methods
4.1. Materials
4.2. Analysis of Protein-Protein Interaction with Surface Plasmon Resonance (SPR) Method
4.3. Assay of Biological Activity of NGF
4.4. Expression of AdipoR1 and AdipoR2 in PC12 Cells and Their Silencing
4.5. MAP Kinase and AMP-Activated Protein Kinase Activation
4.6. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| AMPK | AMP-activated protein kinase |
| ECM | extracellular matrix |
| ERK | extracellular signal regulated kinase |
| FCS | fetal calf serum |
| FGF | fibroblast growth factor |
| GAPDH | glyceraldehyde 3-phosphate dehydrogenase |
| MAPK | mitogen-activated protein kinase |
| NGF | nerve growth factor |
| NT | neurotrophin |
| PDGF | platelet-derived growth factor |
| RU | resonance unit |
| SPARC | secreted protein, acidic and rich in cysteine |
| SPR | surface plasmon resonance |
| TGF | transforming growth factor |
| TNF | tumor necrosis factor |
| VEGF | vascular endothelial growth factor |
References
- Matsuzawa, Y.; Funahashi, T.; Kihara, S.; Shimomura, I. Adiponectin and metabolic syndrome. Arterioscler. Thromb. Vasc. Biol. 2004, 24, 29–33. [Google Scholar] [CrossRef]
- Oh, D.K.; Ciaraldi, T.; Henry, R.R. Adiponectin in health and disease. Diabetes Obes. Metab. 2007, 9, 282–289. [Google Scholar] [CrossRef] [PubMed]
- Schäffler, A.; Buechler, C. CTRP family: Linking immunity to metabolism. Trends Endocrinol. Metab. 2012, 23, 194–204. [Google Scholar] [CrossRef]
- Liu, M.; Liu, F. Regulation of adiponectin multimerization, signaling and function. Best Pract. Res. Clin. Endocrinol. Metab. 2014, 28, 25–31. [Google Scholar] [CrossRef]
- Arita, Y.; Kihara, S.; Ouchi, N.; Takahashi, M.; Maeda, K.; Miyagawa, J.; Hotta, K.; Shimomura, I.; Nakamura, T.; Miyaoka, K.; et al. Paradoxical decrease of an adipose- specific protein, adiponectin, in obesity. Biochem. Biophys. Res. Commun. 1999, 257, 79–83. [Google Scholar] [CrossRef]
- Kondo, H.; Shimomura, I.; Matsukawa, Y.; Kumada, M.; Takahashi, M.; Matsuda, M.; Ouchi, N.; Kihara, S.; Kawamoto, T.; Sumitsuji, S.; et al. Association of adiponectin mutation with type 2 diabetes: A candidate gene for the insulin resistance syndrome. Diabetes 2002, 51, 2325–2328. [Google Scholar] [CrossRef] [PubMed]
- Orlandi, A.; Bochaton-Piallat, M.L.; Gabbiani, G.; Spagnoli, L.G. Aging, smooth muscle cells and vascular pathobiology: Implications for atherosclerosis. Atherosclerosis 2006, 188, 221–230. [Google Scholar] [CrossRef] [PubMed]
- Arita, Y.; Kihara, S.; Ouchi, N.; Maeda, K.; Kuriyama, H.; Okamoto, Y.; Kumada, M.; Hotta, K.; Nishida, M.; Takahashi, M.; et al. Adipocyte-derived plasma protein adiponectin acts as a platelet-derived growth factor-BB–binding protein and regulates growth factor–induced common postreceptor signal in vascular smooth muscle cell. Circulation 2002, 105, 2893–2898. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Lam, K.S.L.; Xu, J.U.; Lu, G.; Xu, L.Y.; Cooper, G.J.S.; Xu, A. Adiponectin inhibits cell proliferation by interacting with several growth factors in an oligomerization-dependent manner. J. Biol. Chem. 2005, 280, 18341–18347. [Google Scholar] [CrossRef]
- Rivera, L.B.; Bradshaw, A.D.; Brekken, R.A. The regulatory function of SPARC in vascular biology. Cell Mol. Life Sci. 2011, 68, 3165–3173. [Google Scholar] [CrossRef]
- Bradshaw, A.D. Diverse biological functions of the SPARC family of proteins. Int. J. Biochem. Cell Biol. 2012, 44, 480–488. [Google Scholar] [CrossRef] [PubMed]
- Bradshaw, A.D.; Sage, E.H. SPARC, a matricellular protein that functions in cellular differentiation and tissue response to injury. J. Clin. Invest. 2001, 107, 1049–1054. [Google Scholar] [CrossRef] [PubMed]
- Nagaraju, G.P.; Dontula, R.; El-Rayes, B.F.; Lakka, S.S. Molecular mechanisms underlying the divergent roles of SPARC in human carcinogenesis. Carcinogenesis 2014, 35, 967–973. [Google Scholar] [CrossRef] [PubMed]
- Kos, K.; Wilding, J.P.H. SPRAC: A key player in the pathologies associated with obesity and diabetes. Nat. Rev. Endocrinol. 2010, 6, 225–235. [Google Scholar] [CrossRef] [PubMed]
- Raines, E.W.; Lane, T.F.; Iruela-Arispe, M.L.; Ross, R.; Sage, E.H. The extracellular glycoprotein SPARC interacts with platelet-derived growth factor (PDGF)-AB and -BB and inhibits the binding of PDGF to its receptors. Proc. Natl. Acad. Sci. USA 1992, 89, 1281–1285. [Google Scholar] [CrossRef] [PubMed]
- Motamed, K.; Blake, D.J.; Angello, J.C.; Allen, B.L.; Rapraeger, A.C.; Hauschka, S.D.; Sage, E.H. Fibroblast growth factor receptor-1 mediates the inhibition of endothelial cell proliferation and the promotion of skeletal myoblast differentiation by SPARC: A role for protein kinase A. J. Cell. Biochem. 2003, 90, 408–423. [Google Scholar] [CrossRef] [PubMed]
- Jayakumar, A.R.; Apeksha, A.; Norenberg, M.D. Role of matricellular proteins in disorders of the central nervous system. Neurochem. Res. 2017, 42, 858–875. [Google Scholar] [CrossRef]
- Gilmour, D.T.; Lyon, G.J.; Carlton, M.B.; Sanes, J.R.; Cunningham, J.M.; Anderson, J.R.; Hogan, B.L.; Evans, M.J.; Colledge, W.H. Mice deficient for the secreted glycoprotein SPARC/osteonectin/BM40 develop normally but show severe age-onset cataract formation and disruption of the lens. EMBO J. 1998, 17, 1860–1870. [Google Scholar] [CrossRef]
- Kucukdereli, H.; Allen, N.J.; Lee, A.T.; Feng, A.; Ozlu, M.I.; Conatser, L.M.; Chakraborty, C.; Workman, G.; Weaver, M.; Sage, E.H.; et al. Control of excitatory CNS synaptogenesis by astrocyte-secreted proteins Hevin and SPARC. Proc. Natl. Acad. Sci. USA 2011, 108, E440–E449. [Google Scholar] [CrossRef] [PubMed]
- López-Murcia, F.J.; Terni, B.; Llobet, A. SPARC triggers a cell-autonomous program of synapse elimination. Proc. Natl. Acad. Sci. USA 2015, 112, 13366–13371. [Google Scholar] [CrossRef]
- Au, E.; Richter, M.W.; Vincent, A.J.; Tetzlaff, W.; Aebersold, R.; Sage, E.H.; Roskams, A.J. SPARC from olfactory ensheathing cells stimulates Schwann cells to promote neurite outgrowth and enhances spinal cord repair. J. Neurosci. 2007, 27, 7208–7221. [Google Scholar] [CrossRef]
- Ma, C.H.; Palmer, A.; Taylor, J.S. Synergistic effects of osteonectin and NGF in promoting survival and neurite outgrowth of superior cervical ganglion neurons. Brain Res. 2009, 1289, 1–13. [Google Scholar] [PubMed]
- Lorber, B.; Chew, D.J.; Hauck, S.M.; Chong, R.S.; Fawcett, J.W.; Martin, K.R. Retinal glia promote dorsal root ganglion axon regeneration. PLoS ONE 2015, 10, e0115996. [Google Scholar] [CrossRef]
- Pepeu, G.; Grazia Giovannini, M. The fate of the brain cholinergic neurons in neurodegenerative diseases. Brain Res. 2017, 1670, 173–184. [Google Scholar] [CrossRef] [PubMed]
- Peeraully, M.R.; Jenkins, J.R.; Trayhurn, P. NGF gene expression and secretion in white adipose tissue: Regulation in 3T3-L1 adipocytes by hormones and inflammatory cytokines. Am. J. Physiol. Endocrinol. Metab. 2004, 287, E331–E339. [Google Scholar] [CrossRef]
- Bulló, M.; Peeraully, M.R.; Trayhurn, P.; Folch, J.; Salas-Salvadó, J. Circulating nerve growth factor levels in relation to obesity and the metabolic syndrome in women. Eur. J. Endocrinol. 2007, 157, 303–310. [Google Scholar] [CrossRef] [PubMed]
- Atanassova, P.; Hrischev, P.; Orbetzova, M.; Nikolov, P.; Nikolova, J.; Georgieva, E. Expression of leptin, NGF and adiponectin in metabolic syndrome. Folia Biol. 2014, 62, 301–306. [Google Scholar] [CrossRef]
- Skaper, S.D. Nerve growth factor: A neuroimmune crosstalk mediator for all seasons. Immunology 2017, 151, 1–15. [Google Scholar] [CrossRef]
- Minnone, G.; De Benedetti, F.; Bracci-Laudiero, L. NGF and its receptors in the regulation of inflammatory response. Int. J. Mol. Sci. 2017, 18, 1028. [Google Scholar] [CrossRef]
- Francki, A.; McClure, T.D.; Brekken, R.A.; Motamed, K.; Murri, C.; Wang, T.; Sage, E.H. SPARC regulates TGF-beta1-dependent signaling in primary glomerular mesangial cells. J. Cell. Biochem. 2004, 91, 915–925. [Google Scholar] [CrossRef] [PubMed]
- Kupprion, C.; Motamed, K.; Sage, E.H. SPARC (BM-40, osteonectin) inhibits the mitogenic effect of vascular endothelial growth factor on microvascular endothelial cells. J. Biol. Chem. 1998, 273, 29635–29640. [Google Scholar] [CrossRef] [PubMed]
- Meldolesi, J. Neurotrophin receptors in the pathogenesis, diagnosis and therapy of neurodegenerative diseases. Pharmacol. Res. 2017, 121, 129–137. [Google Scholar] [CrossRef] [PubMed]
- Ravichandran, A.; Low, B.C. SmgGDS antagonizes BPGAP1-induced Ras/ERK activation and neuritogenesis in PC12 cell differentiation. Mol. Biol. Cell. 2013, 24, 145–156. [Google Scholar] [CrossRef] [PubMed]
- Lombardi, L.; Persiconi, I.; Gallo, A.; Hoogenraad, C.C.; De Stefano, M.E. NGF-dependent axon growth and regeneration are altered in sympathetic neurons of dystrophic mdx mice. Mol. Cell. Neurosci. 2017, 80, 1–17. [Google Scholar] [CrossRef]
- Albrecht, D.; López-Murcia, F.J.; Pérez-González, A.P.; Lichtner, G.; Solsona, C.; Llobet, A. SPARC prevents maturation of cholinergic presynaptic terminals. Mol. Cell. Neurosci. 2012, 49, 364–374. [Google Scholar] [CrossRef] [PubMed]
- Waki, H.; Yamauchi, T.; Kamon, J.; Kita, S.; Ito, Y.; Hada, Y.; Uchida, S.; Tsuchida, A.; Takekawa, S.; Kadowaki, T. Generation of globular fragment of adiponectin by leukocyte elastase secreted by monocytic cell line THP-1. Endocrinology 2005, 146, 790–796. [Google Scholar] [CrossRef]
- Kritas, S.K.; Saggini, A.; Cerulli, G.; Caraffa, A.; Antinolfi, P.; Pantalone, A.; Frydas, S.; Rosatt, M.; Tei, M.; Speziali, A.; et al. Neuropeptide NGF mediates neuro-immune response and inflammation through mast cell activation. J. Biol. Regulators Homeostatic agents 2014, 28, 177–181. [Google Scholar]
- Żelechowska, P.; Agier, J.; Kozłowska, E.; Brzezińska-Błaszczyk, E. Mast cells participate in chronic low-grade inflammation within adipose tissue. Obes. Rev. 2018, 19, 686–697. [Google Scholar]
- Demir, I.E.; Tieftrunk, E.; Schrn, S.; FriEss, H.; Ceyhan, G.O. Nerve growth factor & TrkA as novel therapeutic targets in cancer. Biochim. Biophys. Acta 2016, 1866, 37–50. [Google Scholar]
- Aloe, L.; Rocco, M.L.; Balzamino, B.O.; Micera, A. Nerve growth factor: Role in growth, differentiation and controlling cancer cell development. J. Exp. Clin. Cancer Res. 2016, 35, 116. [Google Scholar] [CrossRef]
- Denk, F.; Bennett, D.L.; McMahon, S.B. Nerve growth factor and pain mechanisms. Annu. Rev. Neurosci. 2017, 40, 307–325. [Google Scholar] [CrossRef] [PubMed]
- O’Shannessy, D.J.; Brigham-Burke, M.; Soneson, K.K.; Hensley, P.; Brooks, I. Determination of rate and equilibrium binding constants for macromolecular interactions by surface plasmon resonance. Methods Enzymol. 1994, 240, 323–349. [Google Scholar]
- Schuster, S.C.; Swanson, R.V.; Alex, L.A.; Bourret, R.B.; Simon, M.I. Assembly and function of a quaternary signal transduction complex monitored by surface plasmon resonance. Nature 1993, 365, 343–347. [Google Scholar] [CrossRef]
- Komagome, R.; Shuto, B.; Moriishi, K.; Kimura, K.; Saito, M. Neuronal and glial differentiation of neuroblastoma and glioma cell lines by Rho inhibitory exoenzyme C3. Neuropathology 1999, 19, 288–293. [Google Scholar] [CrossRef]
- Fujioka, D.; Kawabata, K.; Saito, Y.; Kobayashi, T.; Nakamura, T.; Kodama, Y.; Takano, H.; Obata, J.E.; Kitta, Y.; Umetani, K.; et al. Role of adiponectin receptors in endothelin-induced cellular hypertrophy in cultured cardiomyocytes and their expression in infarcted heart. Am. J. Physiol. Heart Circul. Physiol. 2006, 290, H2409–H2416. [Google Scholar] [CrossRef] [PubMed]
| Analytes | Association Constant (ka) | Dissociation Constant (kd) | Binding Constant (KD = kd/ka) |
|---|---|---|---|
| PDGF-BB | 3.71 × 104 | 9.03 × 10−4 | 2.43 × 10−8 |
| VEGF-165 | 7.58 × 104 | 4.13 × 10−3 | 5.44 × 10−8 |
| FGF2 | 1.35 × 102 | 1.95 × 10−3 | 1.44 × 10−5 |
| TGFβ1 | 7.90 × 102 | 1.92 × 10−2 | 2.43 × 10−5 |
| NGFβ | 1.86 × 105 | 1.10 × 10−2 | 5.94 × 10−8 |
| Analytes | Association Constant (ka) | Dissociation Constant (kd) | Binding Constant (KD = kd/ka) |
|---|---|---|---|
| (ligand: full length adiponectin) | |||
| PDGF-BB | 1.15 × 103 | 2.82 × 10−5 | 2.45 × 10−8 |
| FGF2 | 7.16 × 102 | 5.76 × 10−5 | 8.02 × 10−8 |
| NGFβ | 5.76 × 104 | 5.97 × 10−3 | 1.03 × 10−7 |
| (ligand: globular adiponectin) | |||
| PDGF-BB | 1.05 × 105 | 7.38 × 10−3 | 7.04 × 10−8 |
| NGFβ | 1.15 × 104 | 1.45 × 10−2 | 1.26 × 10−6 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Okura, Y.; Imao, T.; Murashima, S.; Shibata, H.; Kamikavwa, A.; Okamatsu-Ogura, Y.; Saito, M.; Kimura, K. Interaction of Nerve Growth Factor β with Adiponectin and SPARC Oppositely Modulates its Biological Activity. Int. J. Mol. Sci. 2019, 20, 1541. https://doi.org/10.3390/ijms20071541
Okura Y, Imao T, Murashima S, Shibata H, Kamikavwa A, Okamatsu-Ogura Y, Saito M, Kimura K. Interaction of Nerve Growth Factor β with Adiponectin and SPARC Oppositely Modulates its Biological Activity. International Journal of Molecular Sciences. 2019; 20(7):1541. https://doi.org/10.3390/ijms20071541
Chicago/Turabian StyleOkura, Yuu, Takeshi Imao, Seisuke Murashima, Haruki Shibata, Akihiro Kamikavwa, Yuko Okamatsu-Ogura, Masayuki Saito, and Kazuhiro Kimura. 2019. "Interaction of Nerve Growth Factor β with Adiponectin and SPARC Oppositely Modulates its Biological Activity" International Journal of Molecular Sciences 20, no. 7: 1541. https://doi.org/10.3390/ijms20071541
APA StyleOkura, Y., Imao, T., Murashima, S., Shibata, H., Kamikavwa, A., Okamatsu-Ogura, Y., Saito, M., & Kimura, K. (2019). Interaction of Nerve Growth Factor β with Adiponectin and SPARC Oppositely Modulates its Biological Activity. International Journal of Molecular Sciences, 20(7), 1541. https://doi.org/10.3390/ijms20071541





