Comparison the Effect of Ferutinin and 17β-Estradiol on Bone Mineralization of Developing Zebrafish (Danio rerio) Larvae
Abstract
1. Introduction
2. Results
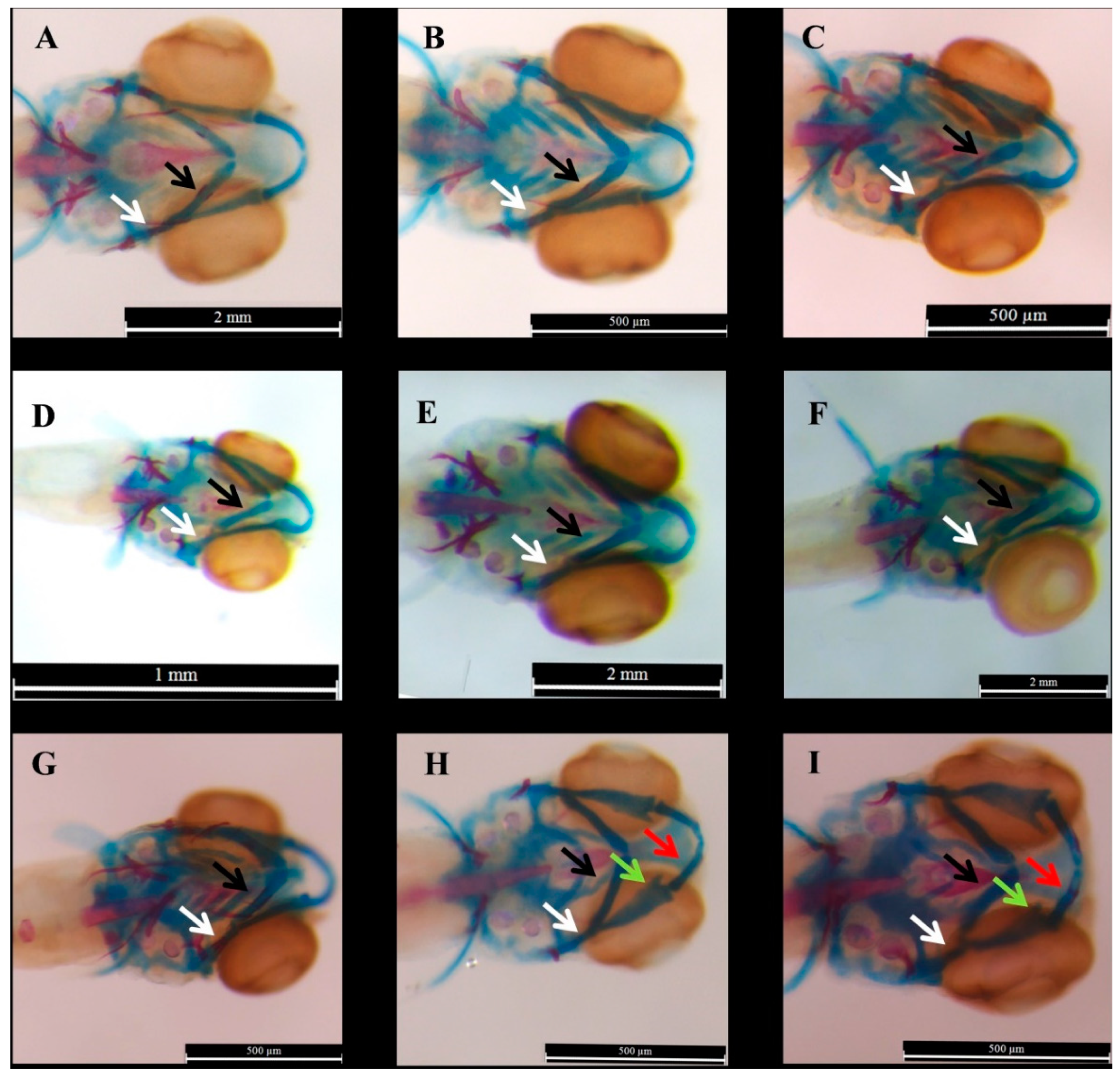
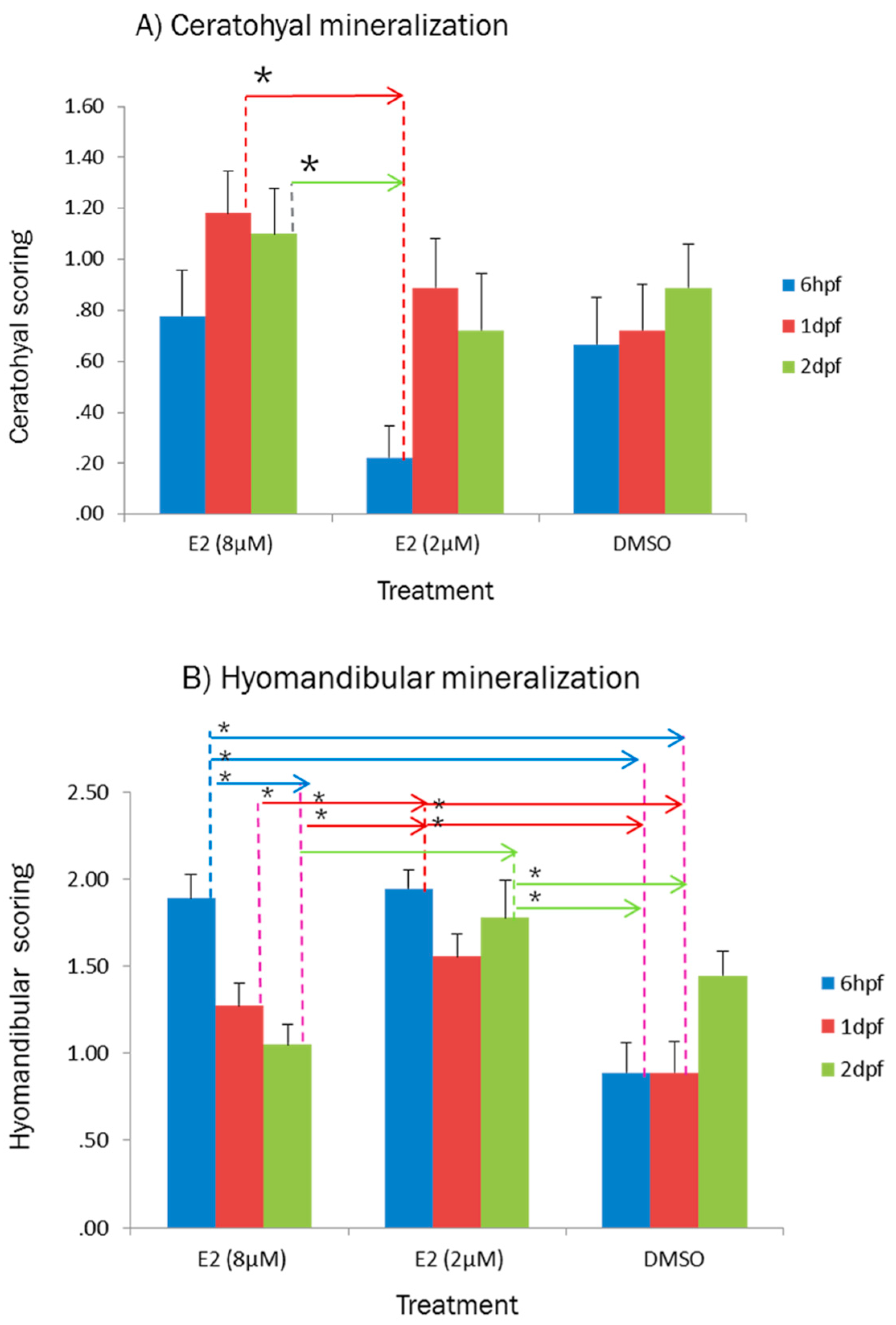
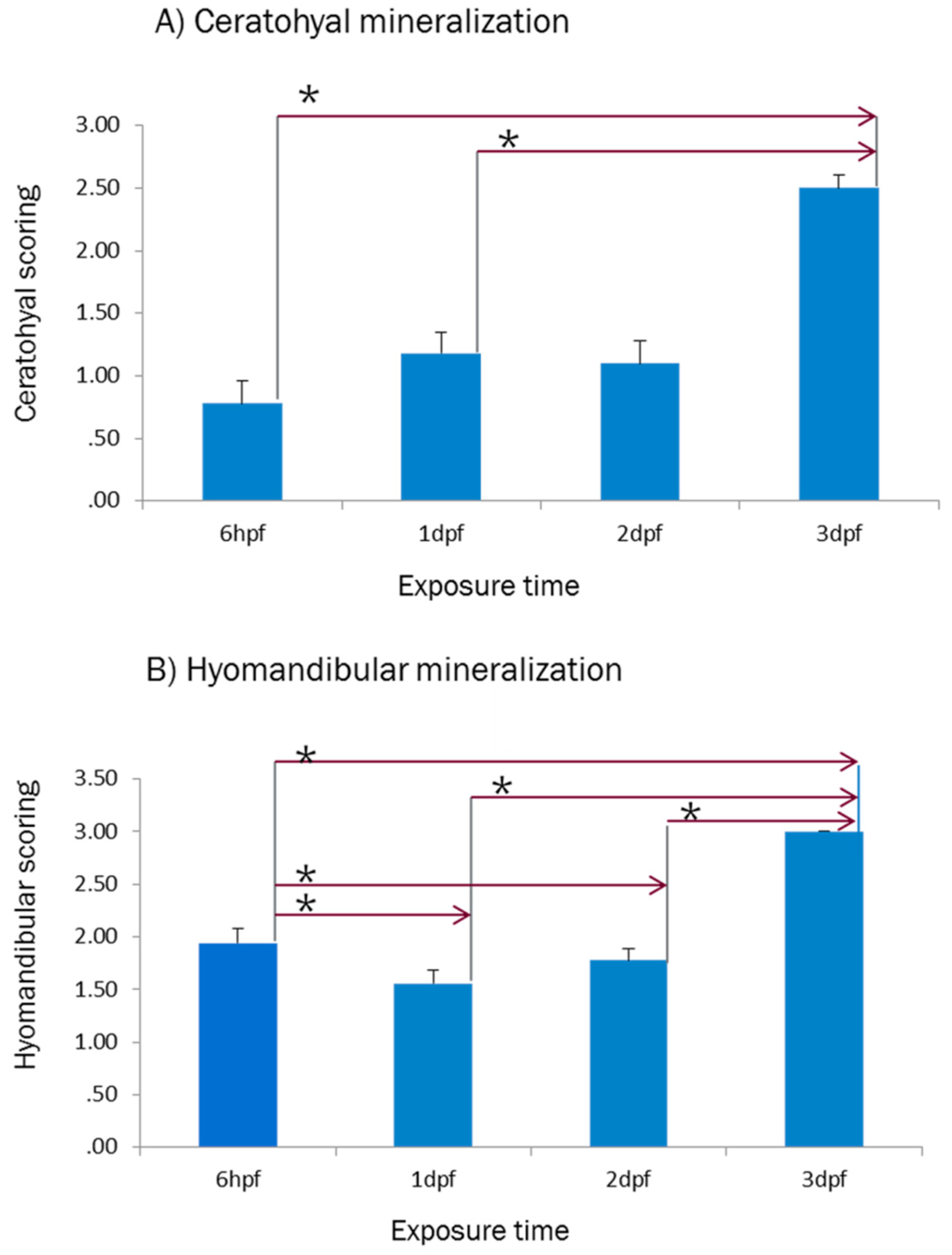
2.1. Effect of E2 on the Phenotype of Bone Mineralization
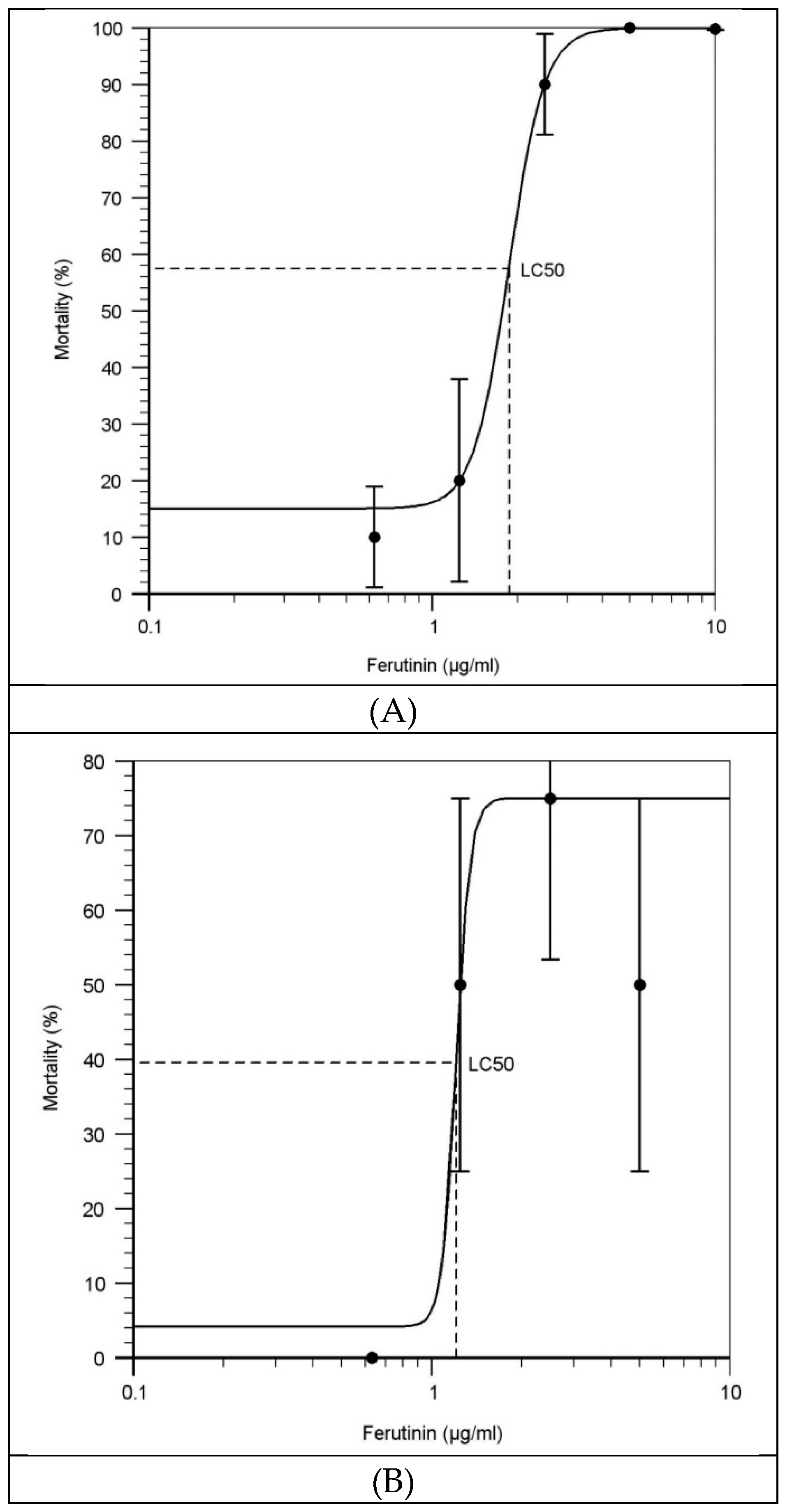
2.2. Effect of Ferutinin on Mortality of Zebrafish Larvae
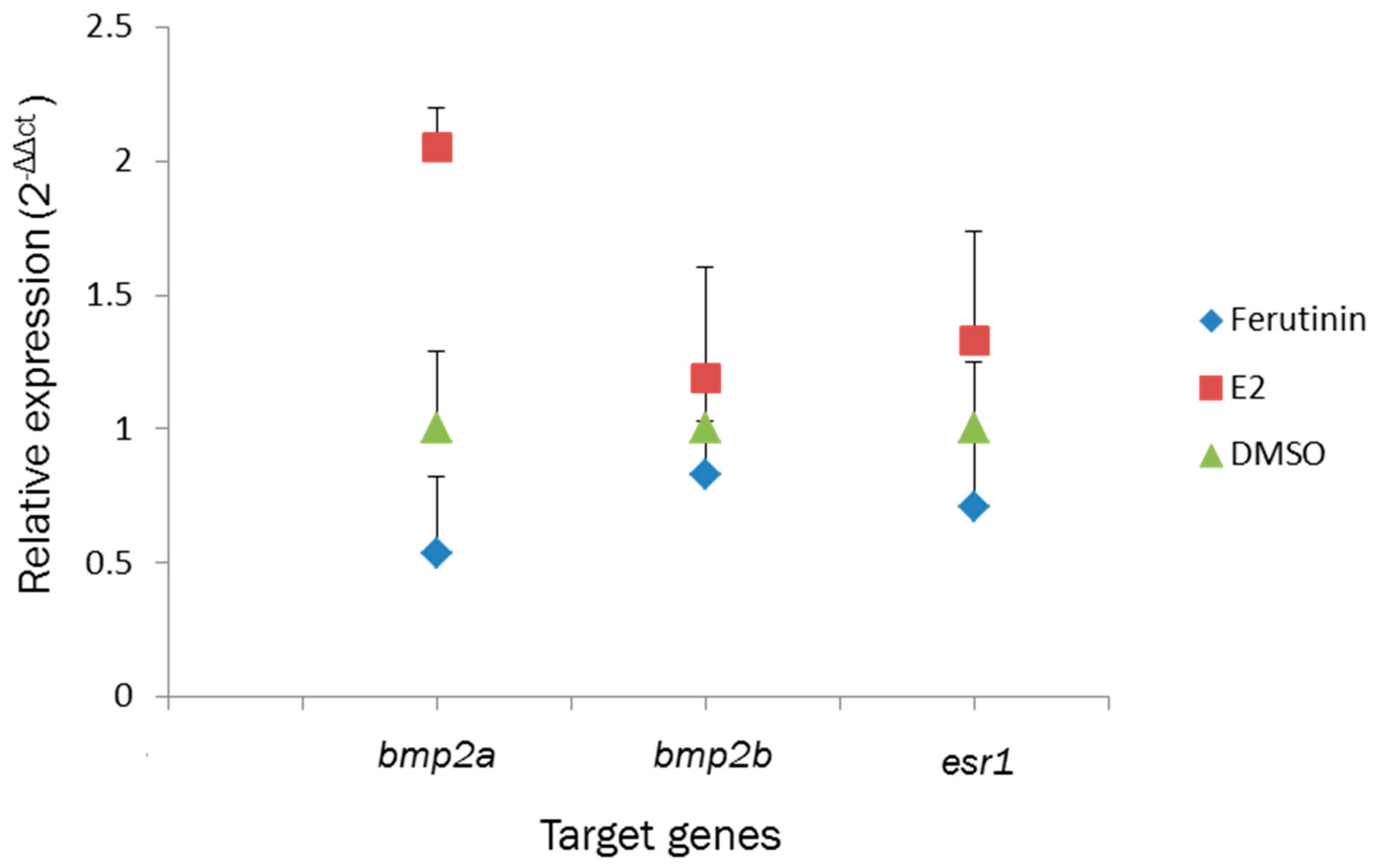
2.3. Effect of Ferutinin on Gene Expression
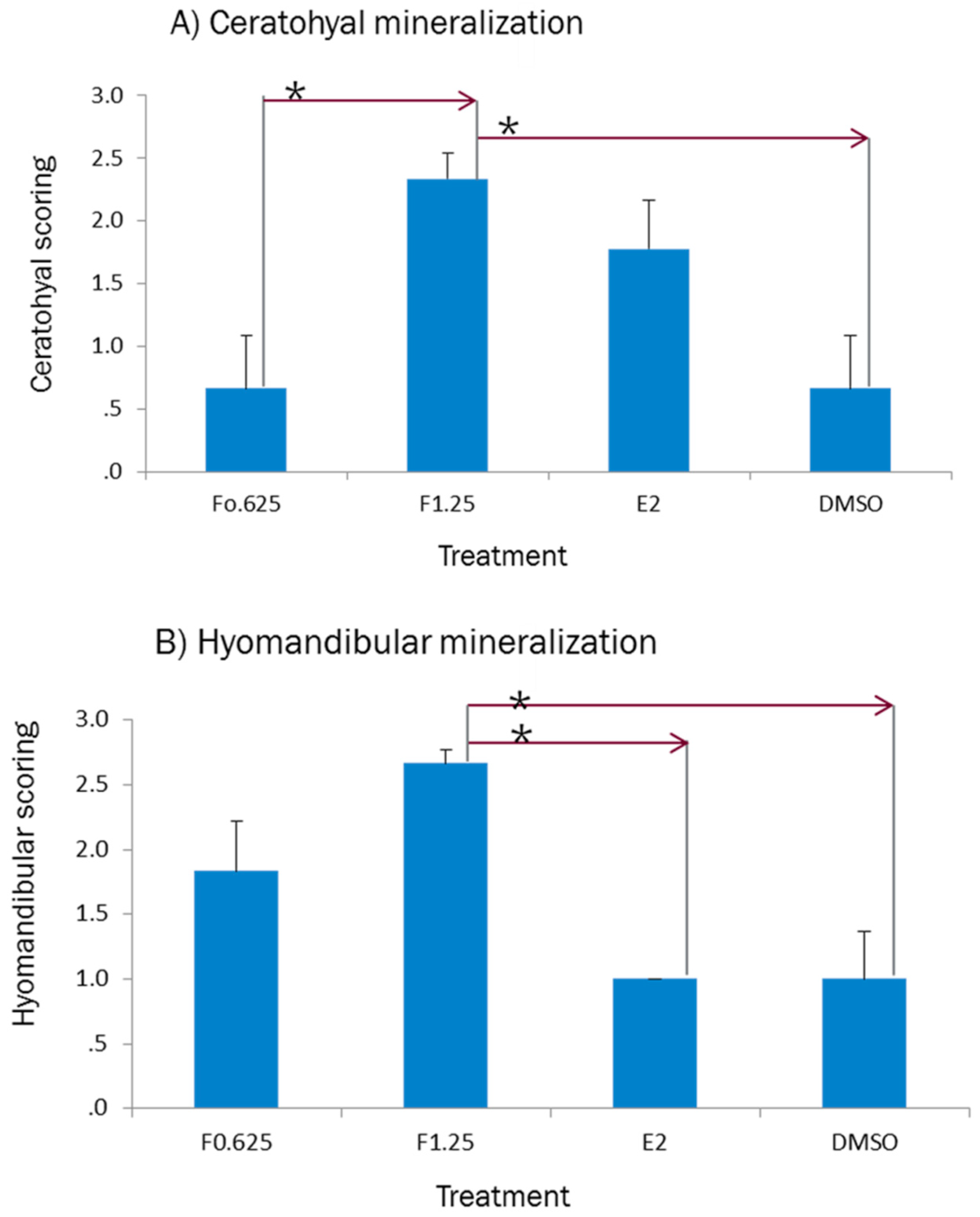
2.4. Phenotypic Readout of Treated Larval Fish
3. Discussion
3.1. Effect of E2 on the Phenotype of Bone Mineralization
3.2. Evaluation of the LC50 of Ferutinin at Each Exposure Time
3.3. Effect of Ferutinin on Target Gene Expression
3.4. Effect of Ferutinin on Bone Mineralization of Zebrafish Larvae
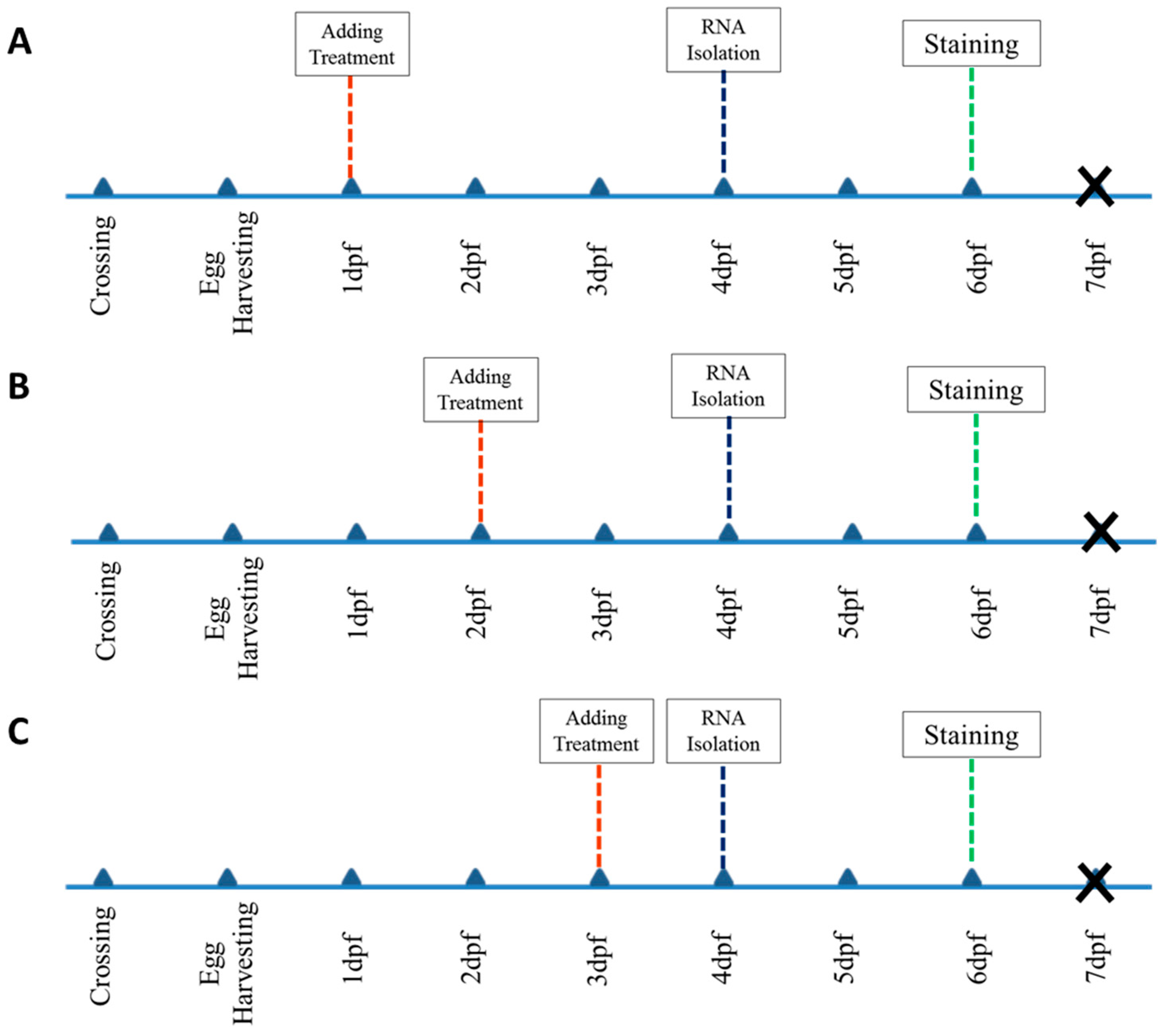
4. Materials and Methods
4.1. Zebrafish Maintain, Husbandry, and Embryo Care
4.2. Chemical Treatments
4.3. Sample Selection and Screening
4.4. Calculation the LC50 of Ferutinin
4.5. RNA Isolation and cDNA Synthesis
4.6. RT-PCR and Real-Time PCR
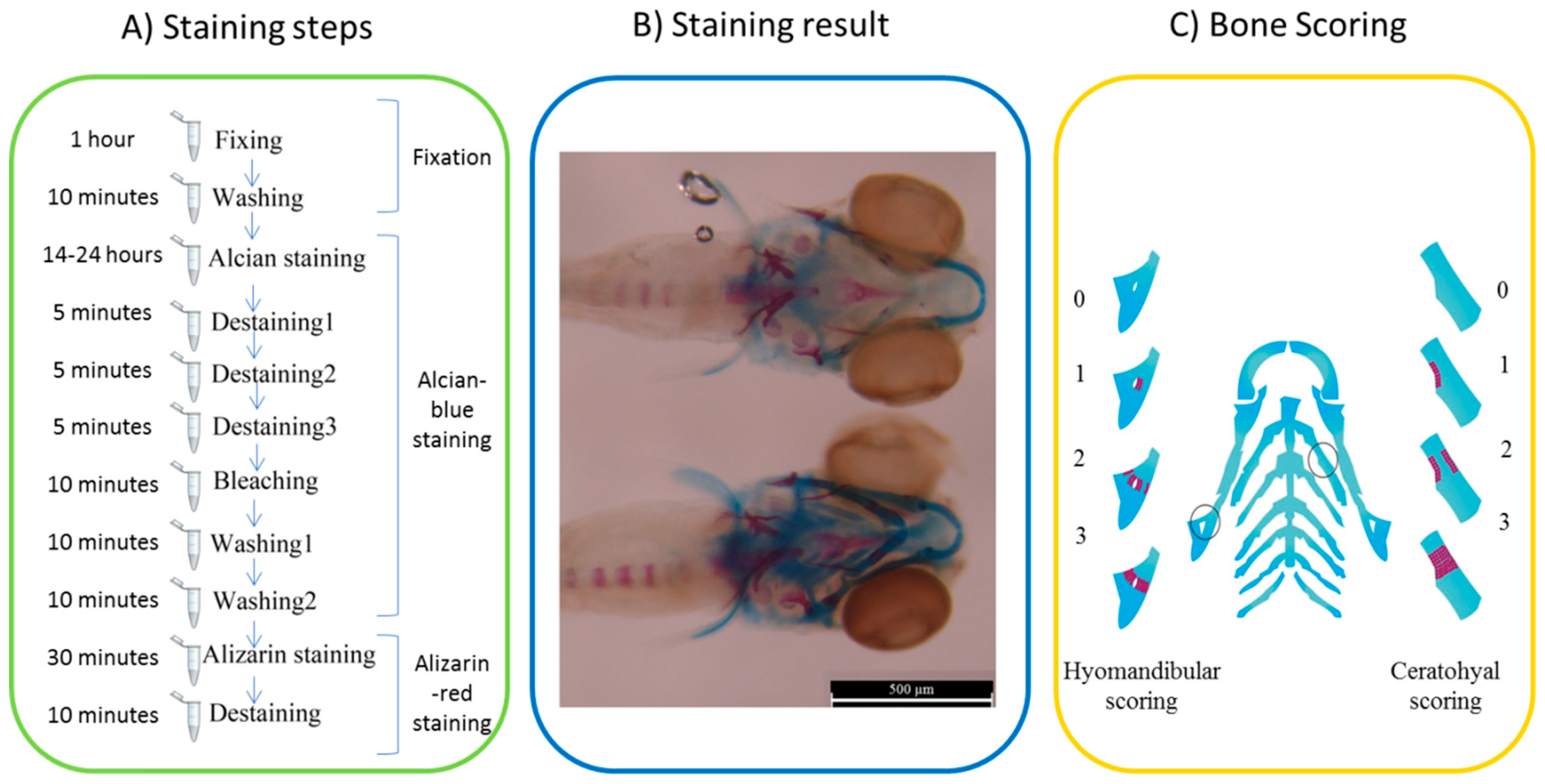
4.7. Staining and Scoring
4.8. Data Analysis and Statistics
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| E2 | 17β-estradiol |
| hpf | Hours post fertilization |
| dpf | Days post fertilization |
| bmp2a | Bone morphogenetic protein 2a |
| bmp2b | Bone morphogenetic protein 2b |
| esr1 | Estrogen receptor 1 |
| esrra | Estrogen-related receptor 1 |
| rpl8 | Ribosomal protein L8 |
| hER | Human estrogen receptor |
| zfERs | Zebrafish estrogen receptor |
| hAFSCs | Human amniotic fluid stem cells |
References
- Khalid, A.B.; Krum, S.A. Estrogen receptors alpha and beta in bone. Bone 2016, 87, 130–135. [Google Scholar] [CrossRef]
- Syed, F.A.; Fraser, D.G.; Monroe, D.G.; Khosla, S. Distinct effects of loss of classical estrogen receptor signaling versus complete deletion of estrogen receptor alpha on bone. Bone 2011, 49, 208–216. [Google Scholar] [CrossRef] [PubMed]
- Pinto, C.; Grimaldi, M.; Boulahtouf, A.; Pakdel, F.; Brion, F.; Aït-Aïssa, S.; Cavaillès, V.; Bourguet, W.; Gustafsson, J.A.; Bondesson, M.; et al. Selectivity of natural, synthetic and environmental estrogens for zebrafish estrogen receptors. Toxicol. Appl. Pharmacol. 2014, 280, 60–69. [Google Scholar] [CrossRef] [PubMed]
- Zavatti, M.; Bertoni, L.; Maraldi, T.; Resca, E.; Beretti, F.; Guida, M.; La Sala, G.B.; De Pol, A. Critical-size bone defect repair using amniotic fluid stem cell/collagen constructs: Effect of oral ferutinin treatment in rats. Life Sci. 2015, 121, 174–183. [Google Scholar] [CrossRef]
- Palumbo, C.; Ferretti, M.; Bertoni, L.; Cavani, F.; Resca, E.; Casolari, B.; Carnevale, G.; Zavatti, M.; Montanari, C.; Benelli, A.; et al. Influence of ferutinin on bone metabolism in ovariectomized rats. I: Role in preventing osteoporosis. J. Bone Miner. Metab. 2009, 27, 538–545. [Google Scholar] [CrossRef] [PubMed]
- Ferretti, M.; Bertoni, L.; Cavani, F.; Zavatti, M.; Resca, E.; Carnevale, G.; Benelli, A.; Zanoli, P.; Palumbo, C. Influence of ferutinin on bone metabolism in ovariectomized rats. II: Role in recovering osteoporosis. J. Anat. 2010, 217, 48–56. [Google Scholar] [CrossRef] [PubMed]
- Ferretti, M.; Cavani, F.; Bertoni, L.; Zavatti, M.; Taronna, A.; Carnevale, G.; Benelli, A.; Zanoli, P.; Marotti, G.; Palumbo, C. New aspects of Ferutinin effect in preventing osteoporosis. Ital. J. Anat. Embryol. 2010, 115, 67. [Google Scholar]
- Ferretti, M.; Bertoni, L.; Cavani, F.; Benincasa, M.; Sena, P.; Carnevale, G.; Zavatti, M.; Viesti, V.D.; Zanoli, P.; Palumbo, C. Structural and histomorphometric evaluations of ferutinin effects on the uterus of ovariectomized rats during osteoporosis treatment. Life Sci. 2012, 90, 161–168. [Google Scholar] [CrossRef]
- Cavani, F.; Ferretti, M.; Carnevale, G.; Bertoni, L.; Zavatti, M.; Palumbo, C. Effects of different doses of ferutinin on bone formation/resorption in ovariectomized rats. J. Bone Miner. Metab. 2012, 30, 619–629. [Google Scholar] [CrossRef]
- Zavatti, M.; Montanari, C.; Zanoli, P. Role of ferutinin in the impairment of female sexual function induced by Ferula hermonis. Physiol. Behav. 2006, 89, 656–661. [Google Scholar] [CrossRef]
- Zavatti, M.; Guida, M.; Maraldi, T.; Beretti, F.; Bertoni, L.; La Sala, G.B.; De Pol, A. Estrogen receptor signaling in the ferutinin-induced osteoblastic differentiation of human amniotic fluid stem cells. Life Sci. 2016, 164, 15–22. [Google Scholar] [CrossRef]
- Zavatti, M.; Benelli, A.; Montanari, C.; Zanoli, P. The phytoestrogen ferutinin improves sexual behavior in ovariectomized rats. Phytomedicine 2009, 16, 547–554. [Google Scholar] [CrossRef]
- Zavatti, M.; Resca, E.; Bertoni, L.; Maraldi, T.; Guida, M.; Carnevale, G.; Ferrari, A.; De Pol, A. Ferutinin promotes proliferation and osteoblastic differentiation in human amniotic fluid and dental pulp stem cells. Life Sci. 2013, 92, 993–1003. [Google Scholar] [CrossRef]
- Crawford, A.; Liekens, S.; Kamuhabwa, A.; Maes, J.; Munck, S.; Busson, R.; Rozenski, J.; Esguerra, C. Zebrafish bioassay-guided natural product discovery: Isolation of angiogenesis inhibitors from East African medicinal plants. PLoS ONE 2011, 6, e14694. [Google Scholar] [CrossRef] [PubMed]
- Crawford, A.D.; Esguerra, C.V.; De Witte, P.A.M. Fishing for drugs from nature: Zebrafish as a technology platform for natural product discovery. Planta Medica 2008, 74, 624–629. [Google Scholar] [CrossRef] [PubMed]
- Howe, K.; Clark, M.D.; Torroja, C.F.; Torrance, J.; Berthelot, C.; Muffato, M.; Collins, J.E.; Humphray, S.; McLaren, K.; Matthews, L.; et al. The zebrafish reference genome sequence and its relationship to the human genome. Nature 2013, 496, 498–503. [Google Scholar] [CrossRef] [PubMed]
- Machado, R.G.; Eames, B.F. Using Zebrafish to test the genetic basis of human craniofacial diseases. J. Dent. Res. 2017, 96, 1192–1199. [Google Scholar] [CrossRef] [PubMed]
- Luo, S.; Yang, Y.; Chen, J.; Zhong, Z.; Huang, H.; Zhang, J.; Cui, L. Tanshinol stimulates bone formation and attenuates dexamethasone-induced inhibition of osteogenesis in larval zebrafish. J. Orthop. Transl. 2016, 4, 35–45. [Google Scholar] [CrossRef] [PubMed]
- Vrieze, E.; Kessel, M.; Peters, H.; Spanings, F.; Flik, G.; Metz, J. Prednisolone induces osteoporosis-like phenotype in regenerating zebrafish scales. 2014. Osteoporos. Int. 2414, 25, 567–578. [Google Scholar] [CrossRef]
- Zhang, N.D.; Han, T.; Huang, B.K.; Rahman, K.H.; Jiang, Y.P.; Xu, H.T.; Qin, L.P.; Xin, H.L.; Zhang, Q.Y.; Li, Y.M. Traditional Chinese medicine formulas for the treatment of osteoporosis: Implication for antiosteoporotic drug discovery. J. Ethnopharmacol. 2016, 189, 61–80. [Google Scholar] [CrossRef]
- Gorelick, D.A.; Halpern, M.E. Visualization of estrogen receptor transcriptional activation in zebrafish. Endocrinology 2011, 52, 2690–2703. [Google Scholar] [CrossRef] [PubMed]
- Hao, R.; Bondesson, M.; Singh, A.V.; Riu, A.; McCollum, C.W.; Knudsen, T.B.; Gorelick, D.A.; Gustafsson, J.A. Identification of estrogen target genes during Zebrafish embryonic development through transcriptomic analysis. PLoS ONE 2013, 8, e79020. [Google Scholar] [CrossRef] [PubMed]
- Souder, J.P.; Gorelick, D.A. Quantification of estradiol uptake in Zebrafish embryos and larvae. Toxicol. Sci. 2017, 158, 465–474. [Google Scholar] [CrossRef] [PubMed]
- Fushimi, S.; Wada, N.; Nohno, T.; Tomita, M.; Saijoh, K.; Sunami, S.; Katsuyama, H. 17β-Estradiol inhibits chondrogenesis in the skull development of zebrafish embryos. Aquat. Toxicol. 2009, 95, 292–298. [Google Scholar] [CrossRef]
- Cohen, S.P.; Lachappelle, A.R.; Walker, B.S.; Lassiter, C.S. Modulation of estrogen causes disruption of craniofacial chondrogenesis in Danio rerio. Aquat. Toxicol. 2014, 152, 113–120. [Google Scholar] [CrossRef] [PubMed]
- Pashai Ahi, E.; Walker, B.S.; Lassiter, C.S.; Jónsson, Z.O.; Heath, J. Investigation of the effects of estrogen on skeletal gene expression during zebrafish larval head development. PeerJ 2016, 4, 1–29. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Yang, F.; Chen, Z.; Shi, L.; Zhang, B.; Pan, J.; Li, X.; Sun, D.; Yang, H. Zebrafish on a Chip: A Novel Platform for Real-Time Monitoring of Drug-Induced Developmental Toxicity. PLoS ONE 2014, 9, e94792. [Google Scholar] [CrossRef]
- Lammer, E.; Kamp, H.G.; Hisgen, V.; Koch, M.; Reinhard, D.; Salinas, E.R.; Wendler, K.; Zok, S.; Braunbeck, T.h. Development of a flow-through system for the fish embryo toxicity test (FET) with the zebrafish (Danio rerio). Toxicol. In Vitro 2009, 23, 1436–1442. [Google Scholar] [CrossRef] [PubMed]
- Khoshmanesh, K.; Akagi, J.; Hall, C.J.; Crosier, K.E.; Crosier, P.S.; Cooper, J.M.; Wlodkowic, D. New rationale for large metazoan embryo manipulations on chip-based devices. Biomicrofluidics 2012, 6, 024102–024114. [Google Scholar] [CrossRef] [PubMed]
- Akagi, J.; Khoshmanesh, K.; Evans, B.; Hall, C.J.; Crosier, K.E.; Cooper, J.M.; Crosier, P.S.; Wlodkowic, D.C. Miniaturized Embryo Array for Automated Trapping, Immobilization and Microperfusion of Zebrafish Embryos. PLoS ONE 2012, 7, e36630. [Google Scholar] [CrossRef]
- AAT Bioquest, Inc. Quest Graph™ LC50 Calculator. Available online: https://www.aatbio.com/tools/lc50-calculator (accessed on 20 February 2019).
- Chen, T.; Wang, Y.; Wu, Y. Developmental exposures to ethanol or dimethylsulfoxide at low concentrations alter locomotor activity in larval zebrafish: Implications for behavioral toxicity bioassays. Aquat. Toxicol. 2011, 102, 162–166. [Google Scholar] [CrossRef] [PubMed]
- Kais, B.; Schneider, K.E.; Keiter, S.; Henn, K.; Ackermann, C.; Braunbeck, T. DMSO modifies the permeability of the zebrafish (Danio rerio) chorion-Implications for the fish embryo test (FET). Aquat. Toxicol. 2013, 140–141, 229–238. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.; Ho, N.Y.; Alshut, R.; Legradi, J.; Weiss, C.; Reischl, M.; Mikut, R.; Liebel, U.; Müller, F.; Strähle, U. Zebrafish embryos as models for embryotoxic and teratological effects of chemicals. Reprod. Toxicol. 2009, 28, 245–253. [Google Scholar] [CrossRef]
- Hogan, N.S.; Lean David, R.S.; Trudeau Vance, L. Exposures to Estradiol, Ethinylestradiol and Octylphenol Affect Survival and Growth of Rana pipiens and Rana sylvatica Tadpoles. J. Toxicol. Environ. Health Part A 2006, 69, 1555–1569. [Google Scholar] [CrossRef] [PubMed]
- Walker, M.B.; Kimmel, C.B. A two-color acid-free cartilage and bone stain for zebrafish larvae. Biotech. Histochem. 2007, 82, 23–28. [Google Scholar] [CrossRef] [PubMed]
- Aceto, J.; Nourizadeh-Lillabadi, R.; Marée, R.; Dardenne, N.; Jeanray, N.; Wehenkel, L.; Aleström, P.; Loon, W.; Muller, M. Zebrafish Bone and General Physiology Are Differently Affected by Hormones or Changes in Gravity. PLoS ONE 2015, 10, e0126928. [Google Scholar] [CrossRef] [PubMed]
- Eames, B.F.; DeLaurier, A.; Ullmann, B.; Huycke, T.R.; Nichols, J.T.; Dowd, J.; McFadden, M.; Sasaki, M.M.; Kimmel, C.B. Fishface: Interactive Atlas of zebrafish craniofacial development at cellular resolution. BMC Dev. Biol. 2013, 13, 23. [Google Scholar] [CrossRef] [PubMed]
- Pashai Ahi, E. Signalling pathways in trophic skeletal development and morphogenesis: Insights from studies on teleost fish. Dev. Biol. 2016, 420, 11–31. [Google Scholar]
- Brenna, T.A.C.; Chang, T.; Eames, B.F. A Novel Role of Extracellular Chondroitin Sulfate Proteoglycan in Cell Signaling and Bone Formation; The University of Saskatchewan, College of Medicine: Saskatoon, SK, Canada, 2016. [Google Scholar]
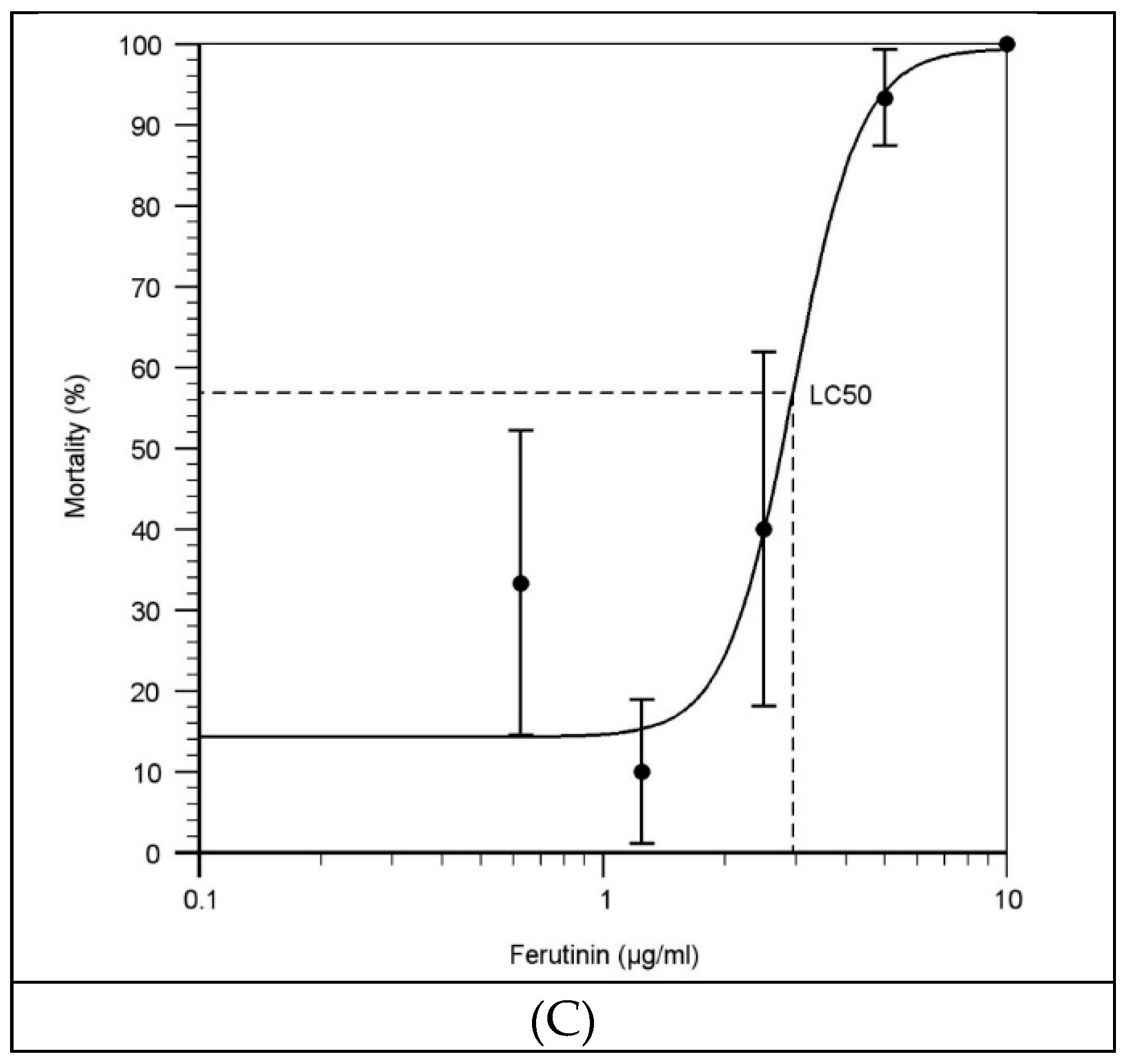
| Conc. of Ferutinin (µg/mL) | Exposure Time | |||||||
|---|---|---|---|---|---|---|---|---|
| 1 dpf | 2 dpf | 3 dpf | ||||||
| Number | Mortality (%) | Number | Mortality (%) | Number | Mortality (%) | |||
| 0 | 16 | 15.02 | 16 | 4.14 | 16 | 14.26 | ||
| 0.625 | 16 | 15.07 | 16 | 4.14 | 16 | 14.29 | ||
| 1.25 | 16 | 19.91 | 16 | 50 | 16 | 15.24 | ||
| 2.5 | 16 | 90.03 | 16 | 75 | 16 | 39.51 | ||
| 5 | 16 | 99.85 | 16 | 75 | 16 | 94.2 | ||
| 10 | 16 | 99.93 | 16 | 75 | 16 | 99.28 | ||
| LC50 | 1.869 | 1.209 | 2.954 | |||||
| Y in toxicity curves (x = conc. of ferutinin) | ||||||||
| Treatment | Genes | ||||
|---|---|---|---|---|---|
| Esr1 | esrra | Bmp2a | Bmp2b | Rpl8 | |
| Ferutinin 0.625 μg/mL | + | + | + | + | + |
| Ferutinin 1.25 μg/mL | − | + | + | + | + |
| Ferutinin 5 μg/mL | + | + | − | + | + |
| Ferutinin at 1 dpf | + | + | − | + | + |
| Ferutinin at 2 dpf | − | + | + | + | + |
| Ferutinin at 3 dpf | + | + | + | + | + |
| DMSO at 1 dpf | + | + | + | − | + |
| DMSO at 2 dpf | + | + | + | + | + |
| DMSO at 3 dpf | + | + | + | + | + |
| 17β-estradiol 10µM at 3 dpf | − | + | + | + | + |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zare Mirakabad, H.; Farsi, M.; Malekzadeh Shafaroudi, S.; Bagheri, A.; Iranshahi, M.; Moshtaghi, N. Comparison the Effect of Ferutinin and 17β-Estradiol on Bone Mineralization of Developing Zebrafish (Danio rerio) Larvae. Int. J. Mol. Sci. 2019, 20, 1507. https://doi.org/10.3390/ijms20061507
Zare Mirakabad H, Farsi M, Malekzadeh Shafaroudi S, Bagheri A, Iranshahi M, Moshtaghi N. Comparison the Effect of Ferutinin and 17β-Estradiol on Bone Mineralization of Developing Zebrafish (Danio rerio) Larvae. International Journal of Molecular Sciences. 2019; 20(6):1507. https://doi.org/10.3390/ijms20061507
Chicago/Turabian StyleZare Mirakabad, Hoda, Mohammad Farsi, Saeed Malekzadeh Shafaroudi, Abdolreza Bagheri, Mehrdad Iranshahi, and Nasrin Moshtaghi. 2019. "Comparison the Effect of Ferutinin and 17β-Estradiol on Bone Mineralization of Developing Zebrafish (Danio rerio) Larvae" International Journal of Molecular Sciences 20, no. 6: 1507. https://doi.org/10.3390/ijms20061507
APA StyleZare Mirakabad, H., Farsi, M., Malekzadeh Shafaroudi, S., Bagheri, A., Iranshahi, M., & Moshtaghi, N. (2019). Comparison the Effect of Ferutinin and 17β-Estradiol on Bone Mineralization of Developing Zebrafish (Danio rerio) Larvae. International Journal of Molecular Sciences, 20(6), 1507. https://doi.org/10.3390/ijms20061507






