Effect of Resveratrol-Enriched Rice on Skin Inflammation and Pruritus in the NC/Nga Mouse Model of Atopic Dermatitis
Abstract
1. Introduction
2. Results
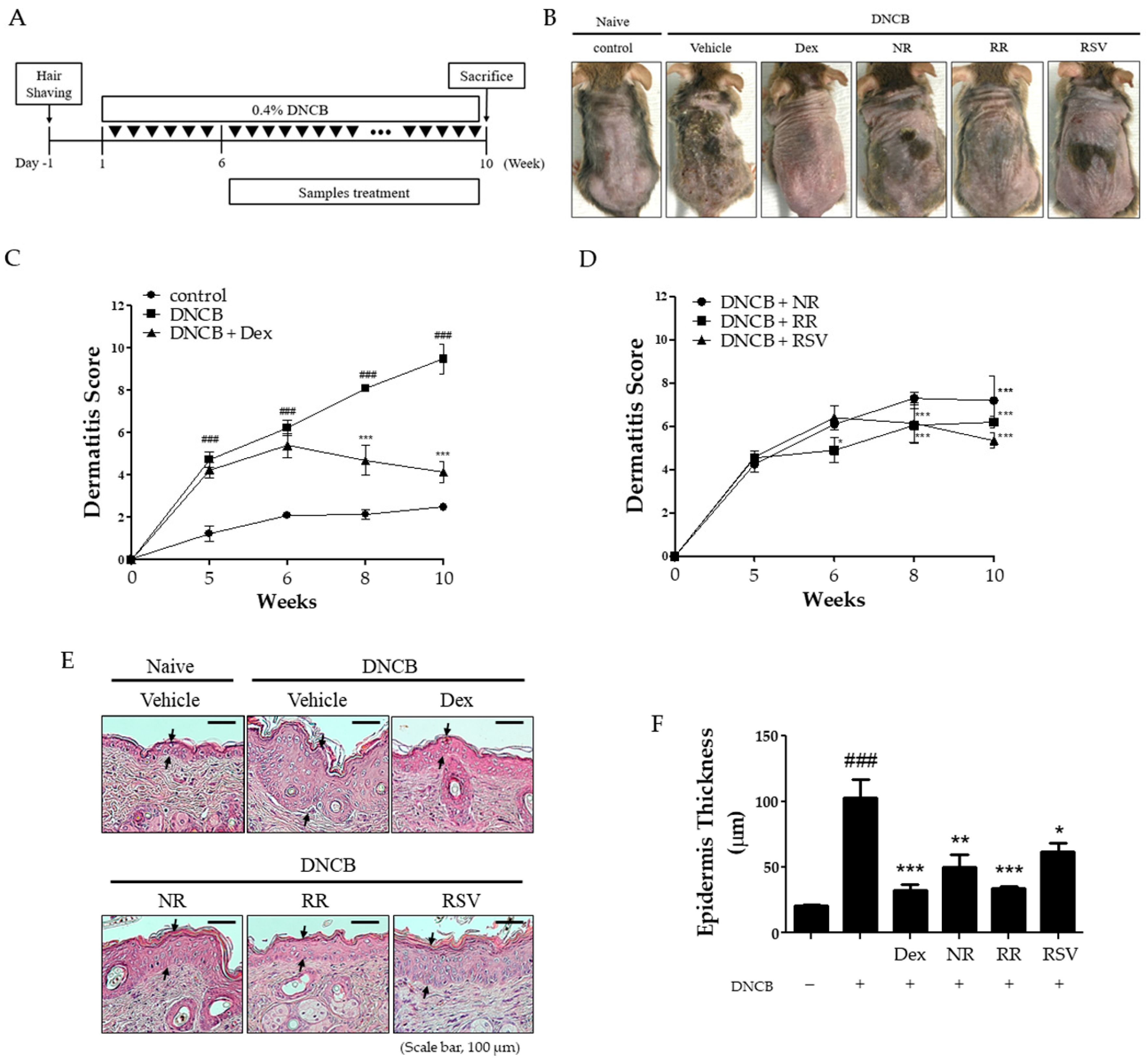
2.1. Resveratrol-Enriched Rice Suppressed AD-like Skin Severity in DNCB-Induced NC/Nga Mice
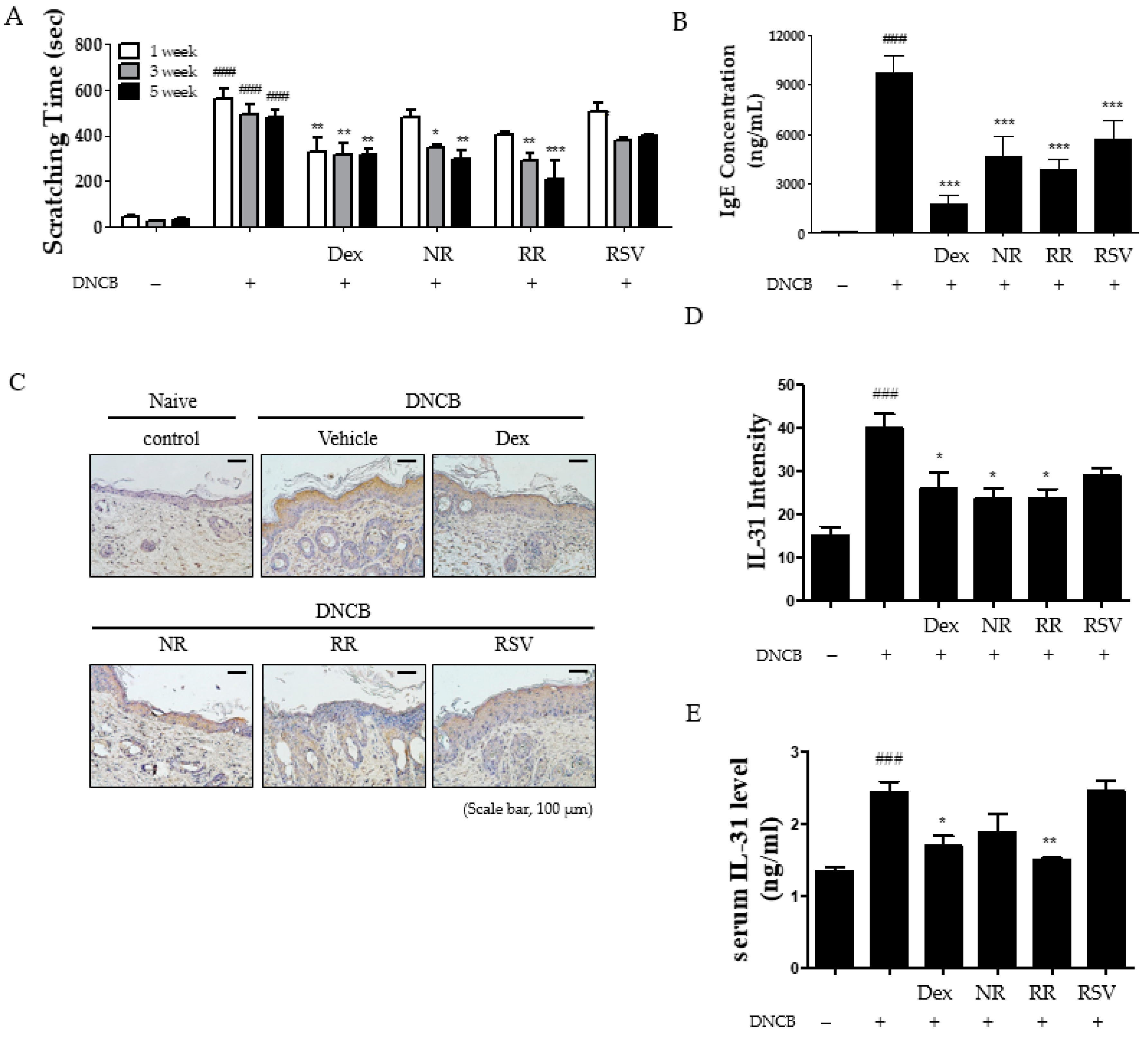
2.2. Resveratrol-Enriched Rice Reduced Scratching Behavior, Serum IgE, and IL-31 Expression in DNCB-Induced NC/Nga Mice
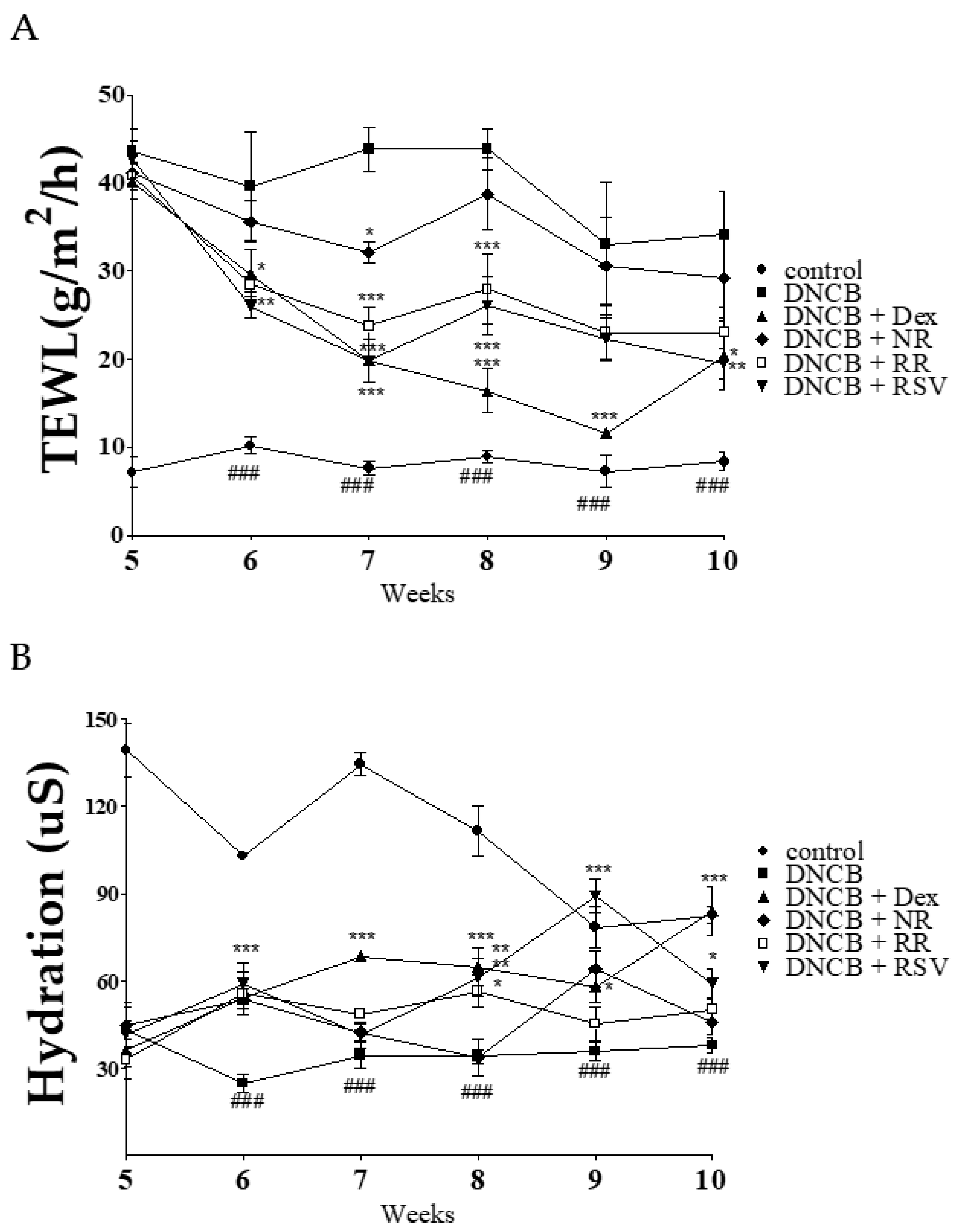
2.3. Resveratrol-Enriched Rice Ameliorates TEWL and Skin Hydration in DNCB-Induced NC/Nga Mice
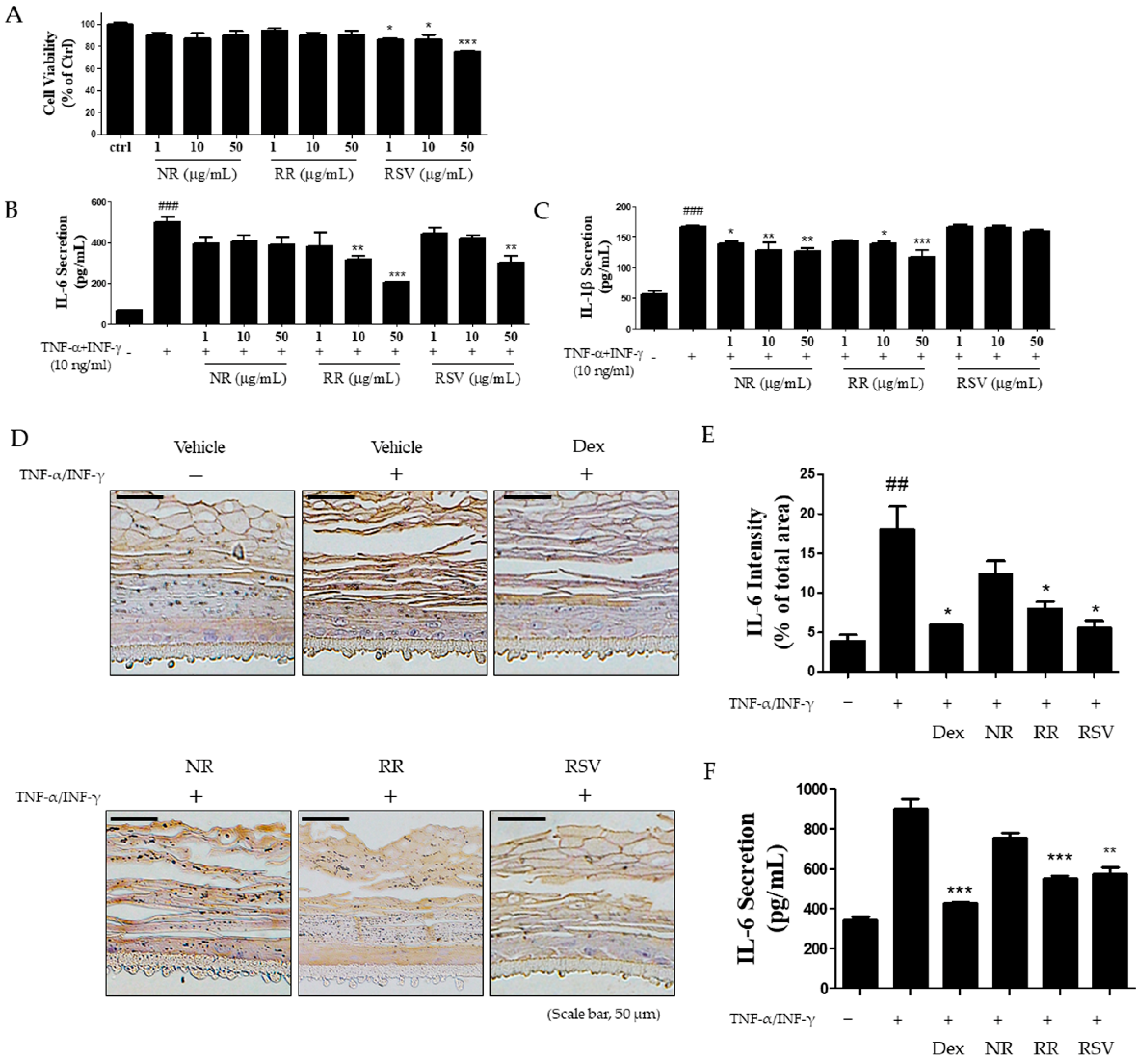
2.4. Resveratrol-Enriched Rice Suppressed TNF-α/INF-γ-Increased Inflammatory Cytokine Secretion in HaCaT Cells and 3D Skin Model
3. Discussion
4. Materials and Methods
4.1. Preparation of Rice Extracts
4.2. Animal Experiments
4.3. Scratching Behavior
4.4. TEWL and Skin Hydration
4.5. Cell Culture and 3D Skin Model
4.6. Cell Viability
4.7. ELISA
4.8. Histology and Immunohistochemistry
4.9. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Pastore, S.; Mascia, F.; Giustizieri, M.L.; Giannetti, A.; Girolomoni, G. Pathogenetic mechanisms of atopic dermatitis. Arch. Immunol. Et Ther. Exp. 2000, 48, 497–504. [Google Scholar]
- Flohr, C.; Mann, J. New insights into the epidemiology of childhood atopic dermatitis. Allergy 2014, 69, 3–16. [Google Scholar] [CrossRef] [PubMed]
- Leung, D.Y. Atopic dermatitis: Immunobiology and treatment with immune modulators. Clin. Exp. Immunol. 1997, 107 (Suppl. 1), 25–30. [Google Scholar]
- David Boothe, W.; Tarbox, J.A.; Tarbox, M.B. Atopic Dermatitis: Pathophysiology. Adv. Exp. Med. Biol. 2017, 1027, 21–37. [Google Scholar] [CrossRef] [PubMed]
- Yosipovitch, G. Dry skin and impairment of barrier function associated with itch–new insights. Int. J. Cosmet. Sci. 2004, 26, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Jin, H.; He, R.; Oyoshi, M.; Geha, R.S. Animal models of atopic dermatitis. J. Investig. Dermatol. 2009, 129, 31–40. [Google Scholar] [CrossRef] [PubMed]
- Matsuda, H.; Watanabe, N.; Geba, G.P.; Sperl, J.; Tsudzuki, M.; Hiroi, J.; Matsumoto, M.; Ushio, H.; Saito, S.; Askenase, P.W.; et al. Development of atopic dermatitis-like skin lesion with IgE hyperproduction in NC/Nga mice. Int. Immunol. 1997, 9, 461–466. [Google Scholar] [CrossRef] [PubMed]
- Fujii, Y.; Takeuchi, H.; Sakuma, S.; Sengoku, T.; Takakura, S. Characterization of a 2,4-dinitrochlorobenzene-induced chronic dermatitis model in rats. Ski. Pharmacol. Physiol. 2009, 22, 240–247. [Google Scholar] [CrossRef]
- Suto, H.; Matsuda, H.; Mitsuishi, K.; Hira, K.; Uchida, T.; Unno, T.; Ogawa, H.; Ra, C. NC/Nga mice: A mouse model for atopic dermatitis. Int. Arch. Allergy Immunol. 1999, 120 (Suppl. 1), 70–75. [Google Scholar] [CrossRef]
- Gutfreund, K.; Bienias, W.; Szewczyk, A.; Kaszuba, A. Topical calcineurin inhibitors in dermatology. Part I: Properties, method and effectiveness of drug use. Postepy Dermatol. I Alergol. 2013, 30, 165–169. [Google Scholar] [CrossRef]
- Hengge, U.R.; Ruzicka, T.; Schwartz, R.A.; Cork, M.J. Adverse effects of topical glucocorticosteroids. J. Am. Acad. Dermatol. 2006, 54, 1–15; quiz 16–18. [Google Scholar] [CrossRef] [PubMed]
- Buys, L.M. Treatment options for atopic dermatitis. Am. Fam. Phys. 2007, 75, 523–528. [Google Scholar]
- Kundu, J.K.; Shin, Y.K.; Surh, Y.J. Resveratrol modulates phorbol ester-induced pro-inflammatory signal transduction pathways in mouse skin in vivo: NF-kappaB and AP-1 as prime targets. Biochem. Pharmacol. 2006, 72, 1506–1515. [Google Scholar] [CrossRef]
- Caglayan Sozmen, S.; Karaman, M.; Cilaker Micili, S.; Isik, S.; Arikan Ayyildiz, Z.; Bagriyanik, A.; Uzuner, N.; Karaman, O. Resveratrol ameliorates 2,4-dinitrofluorobenzene-induced atopic dermatitis-like lesions through effects on the epithelium. PeerJ 2016, 4, e1889. [Google Scholar] [CrossRef] [PubMed]
- Seo, M.Y.; Chung, S.Y.; Choi, W.K.; Seo, Y.K.; Jung, S.H.; Park, J.M.; Seo, M.J.; Park, J.K.; Kim, J.W.; Park, C.S. Anti-aging effect of rice wine in cultured human fibroblasts and keratinocytes. J. Biosci. Bioeng. 2009, 107, 266–271. [Google Scholar] [CrossRef]
- Shimoda, H.; Terazawa, S.; Hitoe, S.; Tanaka, J.; Nakamura, S.; Matsuda, H.; Yoshikawa, M. Changes in ceramides and glucosylceramides in mouse skin and human epidermal equivalents by rice-derived glucosylceramide. J. Med. Food 2012, 15, 1064–1072. [Google Scholar] [CrossRef] [PubMed]
- Baek, S.H.; Chung, H.J.; Lee, H.K.; D’Souza, R.; Jeon, Y.; Kim, H.J.; Kweon, S.J.; Hong, S.T. Treatment of obesity with the resveratrol-enriched rice DJ-526. Sci. Rep. 2014, 4, 3879. [Google Scholar] [CrossRef] [PubMed]
- Subedi, L.; Lee, T.H.; Wahedi, H.M.; Baek, S.H.; Kim, S.Y. Resveratrol-Enriched Rice Attenuates UVB-ROS-Induced Skin Aging via Downregulation of Inflammatory Cascades. Oxidative Med. Cell. Longev. 2017, 2017, 8379539. [Google Scholar] [CrossRef]
- Lee, T.H.; Seo, J.O.; Do, M.H.; Ji, E.; Baek, S.H.; Kim, S.Y. Resveratrol-Enriched Rice Down-Regulates Melanin Synthesis in UVB-Induced Guinea Pigs Epidermal Skin Tissue. Biomol. Ther. 2014, 22, 431–437. [Google Scholar] [CrossRef]
- Potenzieri, C.; Undem, B.J. Basic mechanisms of itch. Clin. Exp. Allergy J. Br. Soc. Allergy Clin. Immunol. 2012, 42, 8–19. [Google Scholar] [CrossRef]
- Lavery, M.J.; Kinney, M.O.; Mochizuki, H.; Craig, J.; Yosipovitch, G. Pruritus: An overview. What drives people to scratch an itch? Ulst. Med. J. 2016, 85, 164–173. [Google Scholar]
- Darsow, U.; Pfab, F.; Valet, M.; Huss-Marp, J.; Behrendt, H.; Ring, J.; Stander, S. Pruritus and atopic dermatitis. Clin. Rev. Allergy Immunol. 2011, 41, 237–244. [Google Scholar] [CrossRef] [PubMed]
- Cho, K.; Parveen, A.; Kang, M.C.; Subedi, L.; Lee, J.H.; Park, S.Y.; Jin, M.R.; Yoon, H.; Son, Y.K.; Kim, S.Y. Pyrus ussuriensis Maxim. leaves extract ameliorates DNCB-induced atopic dermatitis-like symptoms in NC/Nga mice. Phytomed. Int. J. Phytother. Phytopharm. 2018, 48, 76–83. [Google Scholar] [CrossRef] [PubMed]
- Lyons, J.J.; Milner, J.D.; Stone, K.D. Atopic dermatitis in children: Clinical features, pathophysiology, and treatment. Immunol. Allergy Clin. N. Am. 2015, 35, 161–183. [Google Scholar] [CrossRef] [PubMed]
- Rabenhorst, A.; Hartmann, K. Interleukin-31: A novel diagnostic marker of allergic diseases. Curr. Allergy Asthma Rep. 2014, 14, 423. [Google Scholar] [CrossRef] [PubMed]
- Zaniboni, M.C.; Samorano, L.P.; Orfali, R.L.; Aoki, V. Skin barrier in atopic dermatitis: Beyond filaggrin. Anais Bras. Dermatol. 2016, 91, 472–478. [Google Scholar] [CrossRef] [PubMed]
- Ozceker, D.; Bulut, M.; Ozbay, A.C.; Dilek, F.; Koser, M.; Tamay, Z.; Guler, N. Assessment of IL-31 levels and disease severity in children with atopic dermatitis. Allergol. Immunopathol. 2018, 46, 322–325. [Google Scholar] [CrossRef] [PubMed]
- Dillon, S.R.; Sprecher, C.; Hammond, A.; Bilsborough, J.; Rosenfeld-Franklin, M.; Presnell, S.R.; Haugen, H.S.; Maurer, M.; Harder, B.; Johnston, J.; et al. Interleukin 31, a cytokine produced by activated T cells, induces dermatitis in mice. Nat. Immunol. 2004, 5, 752–760. [Google Scholar] [CrossRef] [PubMed]
- Takamori, A.; Nambu, A.; Sato, K.; Yamaguchi, S.; Matsuda, K.; Numata, T.; Sugawara, T.; Yoshizaki, T.; Arae, K.; Morita, H.; et al. IL-31 is crucial for induction of pruritus, but not inflammation, in contact hypersensitivity. Sci. Rep. 2018, 8, 6639. [Google Scholar] [CrossRef] [PubMed]
- Novak, N.; Bieber, T.; Leung, D.Y. Immune mechanisms leading to atopic dermatitis. J. Allergy Clin. Immunol. 2003, 112, S128–S139. [Google Scholar] [CrossRef]
- Kasraie, S.; Niebuhr, M.; Baumert, K.; Werfel, T. Functional effects of interleukin 31 in human primary keratinocytes. Allergy 2011, 66, 845–852. [Google Scholar] [CrossRef] [PubMed]
- Huth, S.; Schmitt, L.; Marquardt, Y.; Heise, R.; Luscher, B.; Amann, P.M.; Baron, J.M. Effects of a ceramide-containing water-in-oil ointment on skin barrier function and allergen penetration in an IL-31 treated 3D model of the disrupted skin barrier. Exp. Dermatol. 2018, 27, 1009–1014. [Google Scholar] [CrossRef] [PubMed]
- Boguniewicz, M.; Leung, D.Y. Atopic dermatitis: A disease of altered skin barrier and immune dysregulation. Immunol. Rev. 2011, 242, 233–246. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.H.; Yu, H.S. Biomarkers for itch and disease severity in atopic dermatitis. Curr. Probl. Dermatol. 2011, 41, 136–148. [Google Scholar] [CrossRef] [PubMed]
- Brandt, E.B.; Sivaprasad, U. Th2 Cytokines and Atopic Dermatitis. J. Clin. Cell. Immunol. 2011, 2. [Google Scholar] [CrossRef] [PubMed]
- Leung, D.Y.; Hirsch, R.L.; Schneider, L.; Moody, C.; Takaoka, R.; Li, S.H.; Meyerson, L.A.; Mariam, S.G.; Goldstein, G.; Hanifin, J.M. Thymopentin therapy reduces the clinical severity of atopic dermatitis. J. Allergy Clin. Immunol. 1990, 85, 927–933. [Google Scholar] [CrossRef]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kang, M.C.; Cho, K.; Lee, J.H.; Subedi, L.; Yumnam, S.; Kim, S.Y. Effect of Resveratrol-Enriched Rice on Skin Inflammation and Pruritus in the NC/Nga Mouse Model of Atopic Dermatitis. Int. J. Mol. Sci. 2019, 20, 1428. https://doi.org/10.3390/ijms20061428
Kang MC, Cho K, Lee JH, Subedi L, Yumnam S, Kim SY. Effect of Resveratrol-Enriched Rice on Skin Inflammation and Pruritus in the NC/Nga Mouse Model of Atopic Dermatitis. International Journal of Molecular Sciences. 2019; 20(6):1428. https://doi.org/10.3390/ijms20061428
Chicago/Turabian StyleKang, Min Cheol, Kyohee Cho, Jae Hyuk Lee, Lalita Subedi, Silvia Yumnam, and Sun Yeou Kim. 2019. "Effect of Resveratrol-Enriched Rice on Skin Inflammation and Pruritus in the NC/Nga Mouse Model of Atopic Dermatitis" International Journal of Molecular Sciences 20, no. 6: 1428. https://doi.org/10.3390/ijms20061428
APA StyleKang, M. C., Cho, K., Lee, J. H., Subedi, L., Yumnam, S., & Kim, S. Y. (2019). Effect of Resveratrol-Enriched Rice on Skin Inflammation and Pruritus in the NC/Nga Mouse Model of Atopic Dermatitis. International Journal of Molecular Sciences, 20(6), 1428. https://doi.org/10.3390/ijms20061428




