Caprate Modulates Intestinal Barrier Function in Porcine Peyer’s Patch Follicle-Associated Epithelium
Abstract
1. Introduction
2. Results
2.1. Cell Culture Study
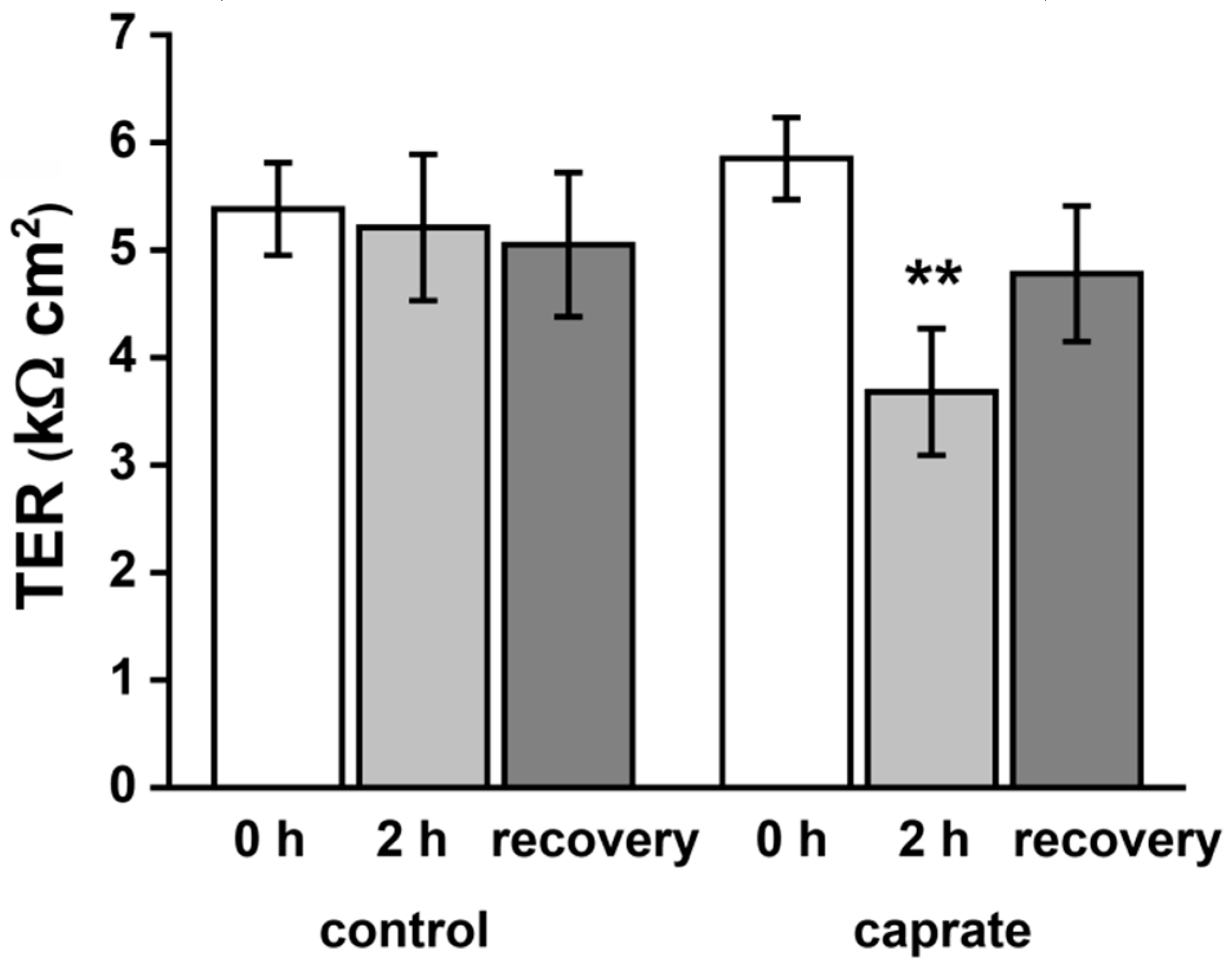
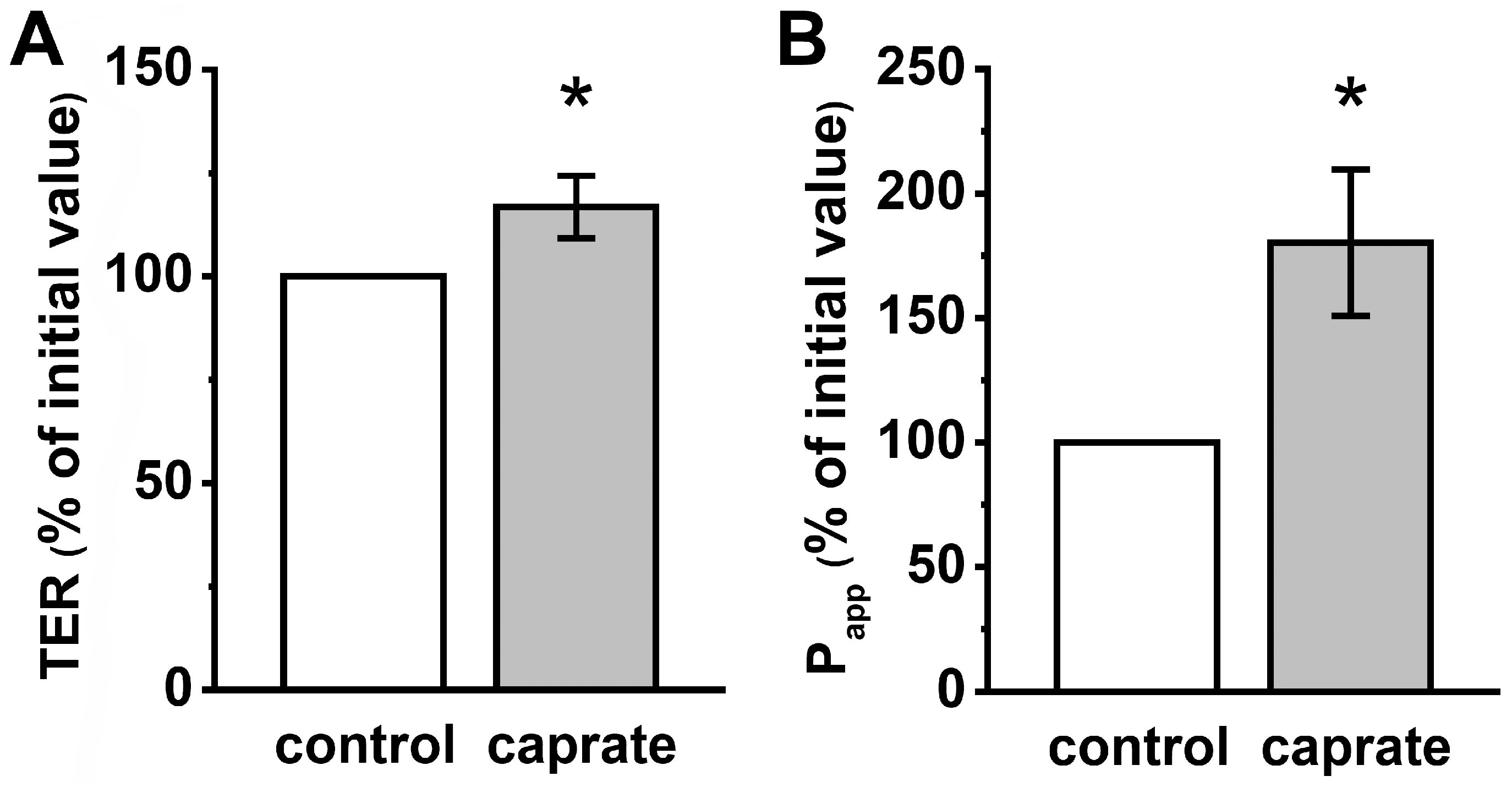
2.2. Ussing Chamber Experiments
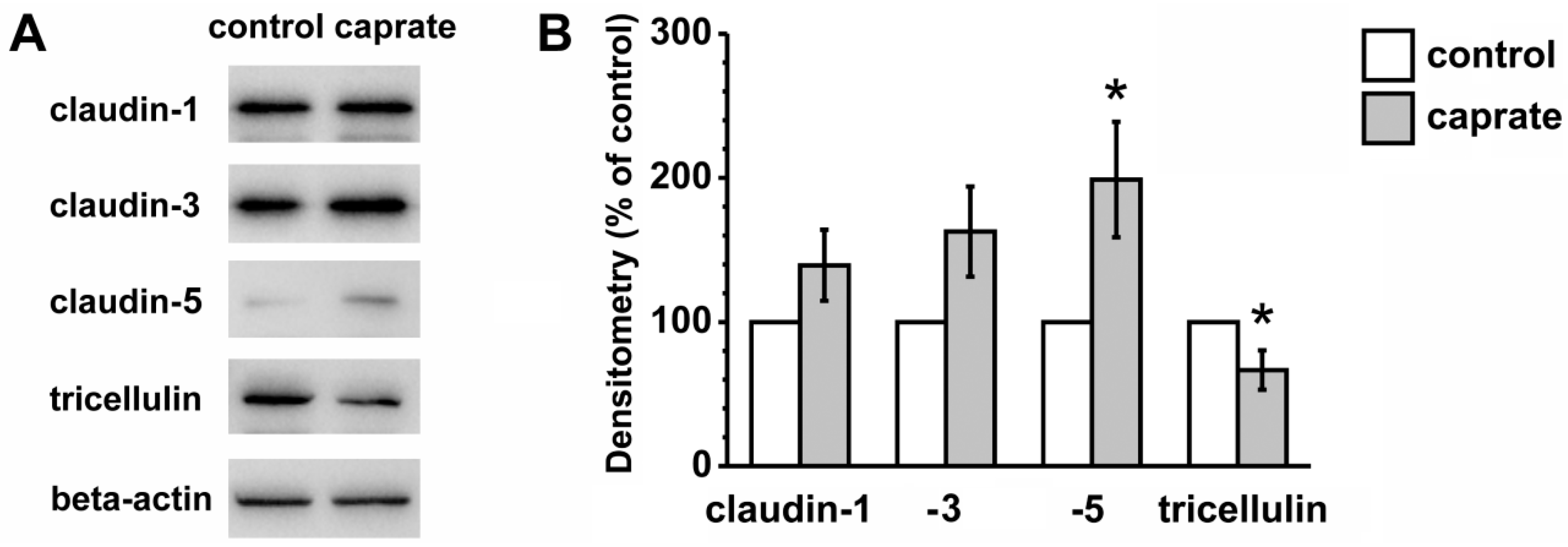
2.3. Immunoblotting
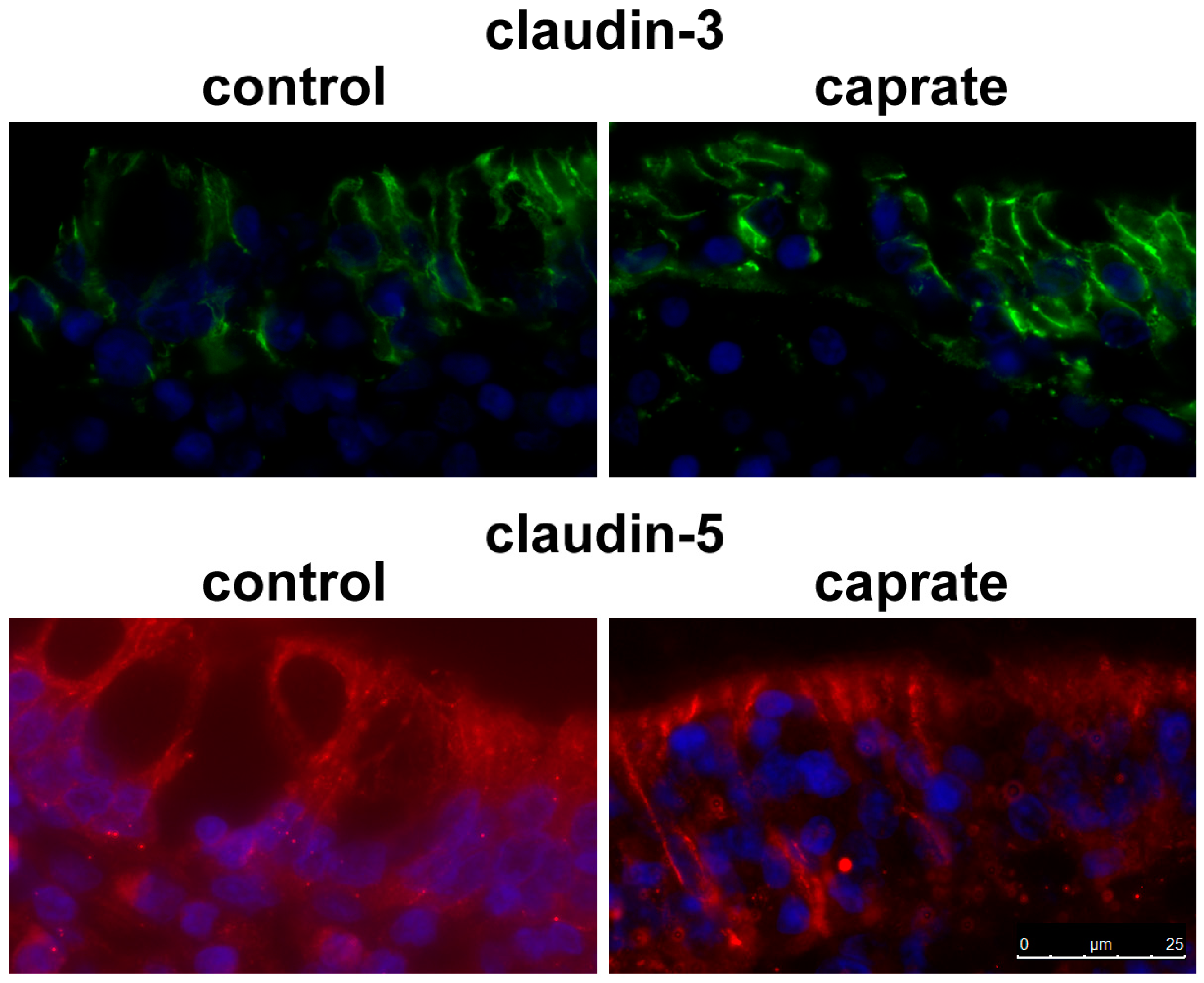
2.4. Immunohistochemistry
3. Discussion
3.1. Suitability of a Porcine Model for Human Gastrointestinal Pathophysiology
3.2. Caprate in Modern Foods
3.3. Effects of Caprate
3.4. Caprate Effects on Tight Junction Protein Expression
4. Materials and Methods
4.1. Cell Culturing and Experiments
4.2. Tissue Samples
4.3. Ussing Chamber Experiments
4.4. Permeability Measurements
4.5. Protein Extraction and Quantification
4.6. Immunoblotting and Densitometry
4.7. Immunohistochemistry
4.8. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| Cldn | Claudin |
| DC | Dendritic cells |
| FAE | Follicle-associated epithelium |
| GALT | Gut-associated lymphocytic tissue |
| IgE | Immunoglobulin E |
| M-cell | Microfold cell |
| PP | Peyer’s patch |
| TER | Transepithelial electrical resistance |
| VE | Villous epithelium |
References
- Neutra, M.R.; Mantis, N.J.; Kraehenbuhl, J.P. Collaboration of epithelial cells with organized mucosal lymphoid tissues. Nat. Immunol. 2001, 2, 1004–1009. [Google Scholar] [CrossRef] [PubMed]
- Markov, A.G.; Falchuk, E.F.; Kruglova, N.M.; Radloff, J.; Amasheh, S. Claudin expression in follicle-associated epithelium of rat Peyer’s patches defines a major restriction of the paracellular pathway. Acta Physiol. 2016, 216, 112–119. [Google Scholar] [CrossRef] [PubMed]
- Radloff, J.; Falchuk, E.L.; Markov, A.G.; Amasheh, S. Molecular Characterization of Barrier Properties in Follicle-Associated Epithelium of Porcine Peyer’s Patches Reveals Major Sealing Function of Claudin-4. Front. Physiol. 2017, 8, 579. [Google Scholar] [CrossRef] [PubMed]
- Owen, R.L.; Jones, A.L. Epithelial cell specialization within human Peyer’s patches: An ultrastructural study of intestinal lymphoid follicles. Gastroenterology 1974, 66, 189–203. [Google Scholar] [CrossRef]
- Takeuchi, T.; Gonda, T. Distribution of the pores of epithelial basement membrane in the rat small intestine. J. Vet. Med. Sci. 2004, 66, 695–700. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Rescigno, M.; Urbano, M.; Valzasina, B.; Francolini, M.; Rotta, G.; Bonasio, R.; Granucci, F.; Kraehenbuhl, J.P.; Ricciardi-Castagnoli, P. Dendritic cells express tight junction proteins and penetrate gut epithelial monolayers to sample bacteria. Nat. Immunol. 2001, 2, 361–367. [Google Scholar] [CrossRef]
- Tamagawa, H.; Takahashi, I.; Furuse, M.; Yoshitake-Kitano, Y.; Tsukita, S.; Ito, T.; Matsuda, H.; Kiyono, H. Characteristics of claudin expression in follicle-associated epithelium of Peyer’s patches: Preferential localization of claudin-4 at the apex of the dome region. Lab. Invest. 2003, 83, 1045–1053. [Google Scholar] [CrossRef]
- Krug, S.M.; Amasheh, M.; Dittmann, I.; Christoffel, I.; Fromm, M.; Amasheh, S. Sodium caprate as an enhancer of macromolecule permeation across tricellular tight junctions of intestinal cells. Biomaterials 2013, 34, 275–282. [Google Scholar] [CrossRef]
- Dittmann, I.; Amasheh, M.; Krug, S.M.; Markov, A.G.; Fromm, M.; Amasheh, S. Laurate permeates the paracellular pathway for small molecules in the intestinal epithelial cell model HT-29/B6 via opening the tight junctions by reversible relocation of claudin-5. Pharm. Res. 2014, 31, 2539–2548. [Google Scholar] [CrossRef]
- Radloff, J.; Zakrzewski, S.S.; Pieper, R.; Markov, A.G.; Amasheh, S. Porcine milk induces a strengthening of barrier function in porcine jejunal epithelium in vitro. Ann. N. Y. Acad. Sci. 2017, 1397, 110–118. [Google Scholar] [CrossRef]
- Li, W.; Sun, K.; Ji, Y.; Wu, Z.; Wang, W.; Dai, Z.; Wu, G. Glycine regulates expression and distribution of claudin-7 and ZO-3 proteins in intestinal porcine epithelial cells. J. Nutr. 2016, 146, 964–969. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.; Wu, Z.; Ji, Y.; Sun, K.; Dai, Z.; Wu, G. L-Glutamine enhances tight junction integrity by activating CaMK kinase 2-AMP-activated protein kinase signaling in intestinal porcine epithelial cells. J. Nutr. 2016, 146, 501–508. [Google Scholar] [CrossRef]
- Amasheh, M.; Schlichter, S.; Amasheh, S.; Mankertz, J.; Zeitz, M.; Fromm, M.; Schulzke, J.D. Quercetin enhances epithelial barrier function and increases claudin-4 expression in Caco-2 cells. J. Nutr. 2008, 138, 1067–1073. [Google Scholar] [CrossRef]
- Amasheh, M.; Fromm, A.; Krug, S.M.; Amasheh, S.; Andres, S.; Zeitz, M.; Fromm, M.; Schulzke, J.D. TNFalpha-induced and berberine-antagonized tight junction barrier impairment via tyrosine kinase, Akt and NFkappaB signaling. J. Cell Sci. 2010, 123, 4145–4155. [Google Scholar] [CrossRef] [PubMed]
- Berin, M.C. Pathogenesis of IgE-mediated food allergy. Clin. Exp. Allergy 2015, 45, 1483–1496. [Google Scholar] [CrossRef] [PubMed]
- Boyce, J.A.; Assa’ad, A.; Burks, A.W.; Jones, S.M.; Sampson, H.A.; Wood, R.A.; Plaut, M.; Cooper, S.F.; Fenton, M.J.; Arshad, S.H.; et al. Guidelines for the Diagnosis and Management of Food Allergy in the United States: Summary of the NIAID-Sponsored Expert Panel Report. J. Allergy Clin. Immunol. 2010, 126, 1105–1118. [Google Scholar] [CrossRef] [PubMed]
- Tang, M.L.; Mullins, R.J. Food allergy: Is prevalence increasing? Intern. Med. J. 2017, 47, 256–261. [Google Scholar] [CrossRef]
- Lindmark, T.; Söderholm, J.D.; Olaison, G.; Alvan, G.; Ocklind, G.; Artursson, P. Mechanism of Absorption Enhancement in Humans After Rectal Administration of Ampicillin in Suppositories Containing Sodium Caprate. Pharm. Res. 1997, 14, 930–935. [Google Scholar] [CrossRef] [PubMed]
- Beare-Rogers, J.L.; Dieffenbacher, A.; Holm, J.V. Lexicon of lipid nutrition (IUPAC Technical Report). Pure. Appl. Chem. 2001, 73, 685–744. [Google Scholar] [CrossRef]
- Tomita, M.; Hayashi, M.; Awazu, S. Absorption-enhancing mechanism of sodium caprate and decanoylcarnitine in Caco-2 cells. J. Pharmacol. Exp. Ther. 1995, 272, 739–743. [Google Scholar] [CrossRef] [PubMed]
- Lv, X.Y.; Li, J.; Zhang, M.; Wang, C.M.; Fan, Z.; Wang, C.Y.; Chen, L. Enhancement of Sodium Caprate on Intestine Absorption and Antidiabetic Action of Berberine. AAPS PharmSciTech. 2010, 11, 372–382. [Google Scholar] [CrossRef]
- Humphray, S.J.; Scott, C.E.; Clark, R.; Marron, B.; Bender, C.; Camm, N.; Davis, J.; Jenks, A.; Noon, A.; Patel, M.; et al. A high utility integrated map of the pig genome. Genome Biol. 2007, 8, R139. [Google Scholar] [CrossRef] [PubMed]
- Kararli, T.T. Comparison of the gastrointestinal anatomy, physiology, and biochemistry of humans and commonly used laboratory animals. Biopharm. Drug. Dispos. 1995, 16, 351–380. [Google Scholar] [CrossRef] [PubMed]
- Rowan, A.M.; Moughan, P.J.; Wilson, M.N.; Maher, K.; Tasman-Jones, C. Comparison of the ileal and faecal digestibility of dietary amino acids in adult humans and evaluation of the pig as a model animal for digestion studies in man. Br. J. Nutr. 1994, 71, 29–42. [Google Scholar] [CrossRef] [PubMed]
- Patterson, J.K.; Lei, X.G.; Miller, D.D. The Pig as an Experimental Model for Elucidating the Mechanisms Governing Dietary Influence on Mineral Absorption. Exp. Biol. Med. 2008, 233, 651–664. [Google Scholar] [CrossRef] [PubMed]
- Miller, E.R.; Ullrey, D.E. The pig as a model for human nutrition. Annu. Rev. Nutr. 1987, 7, 361–382. [Google Scholar] [CrossRef] [PubMed]
- Schierack, P.; Nordhoff, M.; Pollmann, M.; Weyrauch, K.D.; Amasheh, S.; Lodemann, U.; Jores, J.; Tachu, B.; Kleta, S.; Blikslager, A.; et al. Characterization of a porcine intestinal epithelial cell line for in vitro studies of microbial pathogenesis in swine. Histochem. Cell. Biol. 2006, 125, 293–305. [Google Scholar] [CrossRef] [PubMed]
- Vergauwen, H. The IPEC-J2 Cell Line. In The Impact of Food Bioactives on Health: In Vitro and Ex Vivo Models; Springer: Cham, Switzerland, 2015. [Google Scholar] [CrossRef]
- Sankararaman, S.; Sferra, T.J. Are We Going Nuts on Coconut Oil? Curr. Nutr. Rep. 2018, 7, 107–115. [Google Scholar] [CrossRef] [PubMed]
- Davies, D.T.; Holt, C.; Christie, W.W. The composition of milk. In Biochemistry of Lactation; Mepham, T.B., Ed.; Elsevier Science Publishers: New York, NY, USA, 1983; pp. 71–117. [Google Scholar]
- Prentice, A.M. Dairy products in global public health. Am. J. Clin. Nutr. 2014, 99, 1212–1216. [Google Scholar] [CrossRef]
- Guillot, E.; Lemarchal, P.; Dhorne, T.; Rerat, A. Intestinal absorption of medium chain fatty acids: In vivo studies in pigs devoid of exocrine pancreatic secretion. Br. J. Nutr. 1994, 72, 545–553. [Google Scholar] [CrossRef]
- You, Y.Q.; Ling, P.R.; Qu, J.Z.; Bistrian, B.R. Effects of medium-chain triglycerides, long-chain triglycerides, or 2-monododecanoin on fatty acid composition in the portal vein, intestinal lymph, and systemic circulation in rats. JPEN J. Parenter. Enteral. Nutr. 2008, 32, 169–175. [Google Scholar] [CrossRef]
- Quann, E.E.; Fulgoni, V.L., III; Auestad, N. Consuming the daily recommended amounts of dairy products would reduce the prevalence of inadequate micronutrient intakes in the United States: Diet modeling study based on NHANES 2007–2010. Nutr. J. 2015, 14, 90. [Google Scholar] [CrossRef]
- Martínez-Vallespín, B.; Vahjen, W.; Zentek, J. Effects of medium chain fatty acids on the structure and immune response of IPEC-J2 cells. Cytotechnology 2016, 68, 1925–1936. [Google Scholar] [CrossRef]
- Rego Costa, A.; Rosado, E.L.; Soares-Mota, M. Influence of the dietary intake of medium chain triglycerides on body composition, energy expenditure and satiety: A systematic review. Nutr. Hosp. 2012, 27, 103–108. [Google Scholar] [CrossRef]
- Husain, Z.; Schwartz, R.A. Food allergy update: More than a peanut of a problem. Int. J. Dermatol. 2013, 52, 286–294. [Google Scholar] [CrossRef]
- Fujita, K.; Katahira, J.; Horiguchi, Y.; Sonoda, N.; Furuse, M.; Tskuita, S. Clostridium perfringens enterotoxin binds to the second extracellular loop of claudin-3, a tight junction membrane protein. FEBS Lett. 2000, 476, 258–261. [Google Scholar] [CrossRef]
- Farquhar, M.G.; Palade, G.E. Junctional complexes in various epithelia. J. Cell Biol. 1963, 17, 375–412. [Google Scholar] [CrossRef]
- Martinez-Palomo, A.; Erlij, D.; Bracho, H. Localization of permeability barriers in the frog skin epithelium. J. Cell. Biol. 1971, 50, 277–287. [Google Scholar] [CrossRef]
- Günzel, D.; Fromm, M. Claudins and other tight junction proteins. Compr. Physiol. 2012, 2, 1819–1852. [Google Scholar] [CrossRef]
- Markov, A.G.; Veshnyakova, A.; Fromm, M.; Amasheh, M.; Amasheh, S. Segmental expression of claudin proteins correlates with tight junction barrier properties in rat intestine. J. Comp. Physiol. B 2010, 180, 591–598. [Google Scholar] [CrossRef]
- Wen, H.; Watry, D.D.; Marcondes, M.C.; Fox, H.S. Selective decrease in paracellular conductance of tight junctions: Role of the first extracellular domain of claudin-5. Mol. Cell. Biol. 2004, 24, 8408–8417. [Google Scholar] [CrossRef] [PubMed]
- Amasheh, S.; Schmidt, T.; Mahn, M.; Florian, P.; Mankertz, J.; Tavalali, S.; Gitter, A.H.; Schulzke, J.D.; Fromm, M. Contribution of claudin-5 to barrier properties in tight junctions of epithelial cells. Cell Tissue Res. 2005, 321, 89–96. [Google Scholar] [CrossRef] [PubMed]
- Ikenouchi, J.; Furuse, M.; Furuse, K.; Sasaki, K.; Tsukita, S.; Tsukita, S. Tricellulin constitutes a novel barrier at tricellular contacts of epithelial cells. J. Cell Biol. 2005, 171, 939–945. [Google Scholar] [CrossRef] [PubMed]
- Krug, S.M.; Amasheh, S.; Richter, J.F.; Milatz, S.; Günzel, D.; Westphal, J.K.; Huber, O.; Schulzke, J.D.; Fromm, M. Tricellulin forms a barrier to macromolecules in tricellular tight junctions without affecting ion permeability. Mol. Biol. Cell. 2009, 20, 3713–3724. [Google Scholar] [CrossRef] [PubMed]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Radloff, J.; Cornelius, V.; Markov, A.G.; Amasheh, S. Caprate Modulates Intestinal Barrier Function in Porcine Peyer’s Patch Follicle-Associated Epithelium. Int. J. Mol. Sci. 2019, 20, 1418. https://doi.org/10.3390/ijms20061418
Radloff J, Cornelius V, Markov AG, Amasheh S. Caprate Modulates Intestinal Barrier Function in Porcine Peyer’s Patch Follicle-Associated Epithelium. International Journal of Molecular Sciences. 2019; 20(6):1418. https://doi.org/10.3390/ijms20061418
Chicago/Turabian StyleRadloff, Judith, Valeria Cornelius, Alexander G. Markov, and Salah Amasheh. 2019. "Caprate Modulates Intestinal Barrier Function in Porcine Peyer’s Patch Follicle-Associated Epithelium" International Journal of Molecular Sciences 20, no. 6: 1418. https://doi.org/10.3390/ijms20061418
APA StyleRadloff, J., Cornelius, V., Markov, A. G., & Amasheh, S. (2019). Caprate Modulates Intestinal Barrier Function in Porcine Peyer’s Patch Follicle-Associated Epithelium. International Journal of Molecular Sciences, 20(6), 1418. https://doi.org/10.3390/ijms20061418




