Anti-Müllerian Hormone Expression in Endometrial Cancer Tissue
Abstract
:1. Introduction
2. Results
3. Discussion
4. Materials and Methods
4.1. Ethics Statement and Research Material
4.2. Tissue Microarrays (TMAs)
4.3. Immunohistochemistry (IHC)
4.4. Evaluation of IHC Reactions
4.5. Statistical Analysis
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| MDPI | Multidisciplinary Digital Publishing Institute |
| DOAJ | Directory of open access journals |
| TLA | Three letter acronym |
| LD | linear dichroism |
References
- Jost, A. The age factor in the castration of male rabbit fetuses. Exp. Biol. Med. 1947, 66, 302. [Google Scholar] [CrossRef]
- Cate, R.L.; Mattaliano, R.J.; Hession, C.; Tizard, R.; Farber, N.M.; Cheung, A.; Ninfa, E.G.; Frey, A.Z.; Gash, D.J.; Chow, E.P.; et al. Isolation of the bovine and human genes for müllerian inhibiting substance and expression of the human gene in animal cells. Cell 1986, 45, 685–698. [Google Scholar] [CrossRef]
- Cohen-Haguenauer, O.; Picard, J.Y.; Mattei, M.G.; Serero, S.; Nguyen, V.C.; de Tand, M.F.; Guerrier, D.; Hors-Cayla, M.C.; Josso, N.; Frézal, J. Mapping of the gene for anti-Müllerian hormone to the short arm of human chromosome 19. Cytogenet. Cell Genet. 1987, 44, 2–6. [Google Scholar] [CrossRef]
- Sobel, V.; Zhu, Y.S.; Imperato-McGinley, J. Fetal hormones and sexual differentiation. Obstetr. Gynecol. Clin. N. Am. 2004, 31, 837–856. [Google Scholar] [CrossRef] [PubMed]
- La Marca, A.; Broekmans, F.J.; Volpe, A.; Fauser, B.C.; Macklon, N.S. Anti-Mullerian hormone (AMH): What do we still need to know? Hum. Reprod. 2009, 24, 2264–2275. [Google Scholar] [CrossRef] [PubMed]
- Di Clemente, N.; Wilson, C.; Faure, E.; Boussin, L.; Carmillo, P.; Tizard, R.; Picard, J.Y.; Vigier, B.; Josso, N.; Cate, R. Cloning, expression, and alternative splicing of the receptor for anti-Müllerian hormone. Mol. Endocrinol. 1994, 8, 1006–1020. [Google Scholar]
- Massagué, J.; Gomis, R.R. The logic of TGFβ signaling. FEBS Lett. 2006, 580, 2811–2820. [Google Scholar] [CrossRef]
- Kim, J.H.; MacLaughlin, D.T.; Donahoe, P.K. Mullerian inhibiting substance/anti-Mullerian hormone: A novel treatment for gynecologic tumors. Obstetr. Gynecol. Sci. 2014, 57, 343–357. [Google Scholar] [CrossRef]
- Lee, M.M.; Donahoe, P.K.; Silverman, B.L.; Hasegawa, T.; Hasegawa, Y.; Gustafson, M.L.; Chang, Y.C.; MacLaughlin, D.T. Measurements of serum mullerian inhibiting substance in the evaluation of children with nonpalpable gonads. N. Engl. J. Med. 1997, 336, 1480–1486. [Google Scholar] [CrossRef]
- Rajpert-De Meyts, E.; Jørgensen, N.; Graem, N.; Müller, J.; Cate, R.L.; Skakkebaek, N.E. Expression of anti-Müllerian hormone during normal and pathological gonadal development: Association with differentiation of Sertoli and granulosa cells. J. Clin. Endocrinol. Metab. 1999, 84, 3836–3844. [Google Scholar] [CrossRef]
- Josso, N.; Lamarre, I.; Picard, J.Y.; Berta, P.; Davies, N.; Morichon, N.; Peschanski, M.; Jeny, R. Anti-müllerian hormone in early human development. Early Hum. Dev. 1993, 33, 91–99. [Google Scholar] [CrossRef]
- Zhan, Y.; Fujino, A.; MacLaughlin, D.T.; Manganaro, T.F.; Szotek, P.P.; Arango, N.A.; Teixeira, J.; Donahoe, P.K. Mullerian Inhibiting Substance regulates its receptor/SMAD signaling and causes mesenchymal transition of the coelomic epithelial cells early in Mullerian duct regression. Development 2006, 133, 2359–2369. [Google Scholar] [CrossRef]
- Fujino, A.; Arango, N.A.; Zhan, Y.; Manganaro, T.F.; Li, X.; MacLaughlin, D.T.; Donahoe, P.K. Cell migration and activated PI3K/AKT-directed elongation in the developing rat Müllerian duct. Dev. Biol. 2009, 325, 351–362. [Google Scholar] [CrossRef] [PubMed]
- Wilson, J.D.; George, F.W.; Griffin, J.E. The hormonal control of sexual development. Science 1981, 211, 1278–1284. [Google Scholar] [CrossRef] [PubMed]
- Teixeira, J.; Maheswaran, S.; Donahoe, P.K. Mullerian Inhibiting Substance: An instructive developmental hormone with diagnostic and possible therapeutic applications. Endocr. Rev. 2001, 22, 657–674. [Google Scholar]
- MacLaughlin, D.T.; Donahoe, P.K. Müllerian inhibiting substance/anti-Müllerian hormone: A potential therapeutic agent for human ovarian and other cancers. Future Oncol. 2010, 6, 391–405. [Google Scholar] [CrossRef] [PubMed]
- Zec, I.; Tislaric-Medenjak, D.; Bukovec Megla, Z.; Kucak, I. Anti-Mullerian hormone: A unique biochemical marker of gonadal development and fertility in humans. Biochem. Med. 2011, 21, 219–230. [Google Scholar] [CrossRef]
- Kelsey, T.W.; Wright, P.; Nelson, S.M.; Anderson, R.A.; Wallace, W.H.B. A validated model of serum anti-Müllerian hormone from conception to menopause. PLoS ONE 2011, 6, e22024. [Google Scholar] [CrossRef]
- Shahrokhi, S.Z.; Kazerouni, F.; Ghaffari, F. Anti-Müllerian Hormone: Genetic and environmental effects. Clin. Chim. Acta 2018, 476, 123–129. [Google Scholar] [CrossRef]
- De Vet, A.; Laven, J.; de Jong, F.; Themmen, A. Anti-Mullerian hormone serum levels: A putative marker for ovarían aging. Fertil. Steril. 2002, 77, 357–362. [Google Scholar] [CrossRef]
- La Marca, A.; De Leo, V.; Giulini, S.; Orvieto, R.; Malmusi, S.; Giannella, L.; Volpe, A. Anti-Mullerian hormone in premenopausal women and after AMH and ART 127 spontaneous or surgically induced menopause. J. Soc. Gynecol. Investig. 2005, 12, 545–548. [Google Scholar] [CrossRef] [PubMed]
- Ebner, T.; Sommergruber, M.; Moser, M.; Shebl, O.; Schreier-Lechner, E.; Tews, G. Basal level of anti-Mullerian hormone is associated with oocyte quality in stimulated cycles. Hum. Reprod. 2006, 21, 2022–2026. [Google Scholar] [CrossRef] [PubMed]
- Visser, J.A.; de Jong, F.H.; Laven, J.S.E.; Themmen, A.P.N. Anti-Müllerian hormone: A new marker for ovarian function. Reproduction 2006, 131, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Broer, S.L.; Broekmans, F.J.; Laven, J.S.; Fauser, B.C. Anti-Müllerian hormone: Ovarian reserve testing and its potential clinical implications. Hum. Reprod. Update 2014, 20, 688–701. [Google Scholar] [CrossRef] [PubMed]
- Islam, Y.; Aboulghar, M.M.; El-Din AlEbrashy, A.; Abdel-Aziz, O. The value of different ovarian reserve tests in the prediction of ovarian response in patients with unexplained infertility. Middle East Fertil. Soc. J. 2016, 21, 69–74. [Google Scholar] [CrossRef]
- Podfigurna, A.; Lukaszuk, K.; Czyzyk, A.; Kunicki, M.; Maciejewska-Jeske, M.; Jakiel, G.; Meczekalski, B. Testing ovarian reserve in pre-menopausal women: Why, whom and how? Maturitas 2018, 109, 112–117. [Google Scholar] [CrossRef] [PubMed]
- Abdelazimad, I.A.; Belal, M.M.; Makhlouf, H. Anti-Mullerian hormone and antral follicle count as predictors of ovarian reserve and successful IVF. Int. J. Fertil. Steril. 2017, 11, 40–46. [Google Scholar]
- Anderson, R.A.; Anckaert, E.; Bosch, E.; Dewailly, D.; Dunlop, C.E.; Fehr, D.; Nardo, L.; Smitz, J.; Tremellen, K.; Denk, B.; et al. Prospective study into the value of the automated Elecsys antimüllerian hormone assay for the assessment of the ovarian growing follicle pool. Fertil. Steril. 2015, 103, 1074–1080.e4. [Google Scholar] [CrossRef]
- Nyboe Andersen, A.; Nelson, S.M.; Fauser, B.C.; García-Velasco, J.A.; Klein, B.M.; Arce, J.C. ESTHER-1 study group. Individualized versus conventional ovarian stimulation for in vitro fertilization: A multicenter, randomized, controlled, assessor-blinded, phase 3 noninferiority trial. Fertil. Steril. 2017, 107, 387–396.e4. [Google Scholar] [CrossRef]
- Pilsgaard, F.; Grynnerup, A.G.; Løssl, K.; Bungum, L.; Pinborg, A. The use of anti-Müllerian hormone for controlled ovarian stimulation in assisted reproductive technology, fertility assessment and -counseling. Acta Obstet. Gynecol. Scand. 2018, 97, 1105–1113. [Google Scholar] [CrossRef]
- Hudson, P.L.; Dougas, I.; Donahoe, P.K.; Cate, R.L.; Epstein, J.; Pepinsky, R.B.; MacLaughlin, D.T. An immunoassay to detect human mullerian inhibiting substance in males and females during normal development. J. Clin. Endocrinol. Metab. 1990, 70, 16–22. [Google Scholar] [CrossRef] [PubMed]
- Munsterberg, A.; Lovell-Badge, R. Expression of the mouse anti-mullerian hormone gene suggests a role in both male and female sexual differentiation. Development 1991, 113, 613–624. [Google Scholar] [PubMed]
- Kim, J.H.; Seibel, M.M.; MacLaughlin, D.T.; Donahoe, P.K.; Ransil, B.J.; Hametz, P.A. The inhibitory effects of mullerian-inhibiting substance on epidermal growth factor induced proliferation and progesterone production of human granulosa-luteal cells. J. Clin. Endocrinol. Metab. 1992, 75, 911–917. [Google Scholar] [PubMed]
- Durlinger, A.L.; Gruijters, M.J.; Kramer, P.; Karels, B.; Ingraham, H.A.; Nachtigal, M.W. Anti-Müllerian hormone inhibits initiation of primordial follicle growth in the mouse ovary. Endocrinology 2002, 143, 1076–1084. [Google Scholar] [CrossRef]
- Nilsson, E.; Rogers, N.; Skinner, M.K. Actions of anti-Müllerian hormone on the ovarian transcriptome to inhibit primordial to primary follicle transition. Reproduction 2007, 134, 209–221. [Google Scholar] [CrossRef] [PubMed]
- Nilsson, E.; Schindler, R.; Savenkova, M.I.; Skinner, M.K. Inhibitory actions of anti-Müllerian hormone (AMH) on ovarian primordial follicle assembly. PLoS ONE 2011, 6, e20087. [Google Scholar] [CrossRef] [PubMed]
- Baarends, W.M.; Hoogerbrugge, J.W.; Post, M.; Visser, J.A.; De Rooij, D.G.; Parvinen, M. Anti-mullerian hormone and anti-mullerian hormone type II receptor messenger ribonucleic acid expression during postnatal testis development and in the adult testis of the rat. Endocrinology 1995, 136, 5614–5622. [Google Scholar] [CrossRef]
- Laurich, V.M.; Trbovich, A.M.; O’Neill, F.H.; Houk, C.P.; Sluss, P.M.; Payne, A.H. Müllerian inhibiting substance blocks the protein kinase A-induced expression of cytochrome p 450 17alpha-hydroxylase/C(17-20) lyase mRNA in a mouse Leydig cell line independent of cAMP responsive element binding protein phosphorylation. Endocrinology 2002, 143, 3351–3360. [Google Scholar] [CrossRef] [PubMed]
- Rey, R.; Sabourin, J.C.; Venara, M.; Long, W.Q.; Jaubert, F.; Zeller, W.P. Anti-Müllerian hormone is a specific marker of sertoli- and granulosa-cell origin in gonadal tumors. Hum. Pathol. 2000, 31, 1202–1208. [Google Scholar] [CrossRef]
- Edelsztein, N.Y.; Grinspon, R.P.; Schteingart, H.F.; Rey, R.A. Anti-Müllerian hormone as a marker of steroid and gonadotropin action in the testis of children and adolescents with disorders of the gonadal axis. Int. J. Pediatr. Endocrinol. 2016, 2016, 20. [Google Scholar] [CrossRef] [PubMed]
- Seifer, D.B.; Tal, R. Antimullerian Hormone: Biology, Role in Ovarian Function and Clinical Significance, 1st ed.; Nova Science Publishers: New York, NY, USA, 2016. [Google Scholar]
- Kushnir, V.A.; Seifer, D.B.; Barad, D.H.; Sen, A.; Gleicher, N. Potential therapeutic applications of human anti-Müllerian hormone (AMH) analogues in reproductive medicine. J. Assist. Reprod. Genet. 2017, 34, 1105–1113. [Google Scholar] [CrossRef] [PubMed]
- Renaud, E.J.; MacLaughlin, D.T.; Oliva, E.; Rueda, B.R.; Donahoe, P.K. Endometrial cancer is a receptor-mediated target for Mullerian Inhibiting Substance. Proc. Natl. Acad. Sci. USA 2005, 102, 111–116. [Google Scholar] [CrossRef]
- Masiakos, P.T.; MacLaughlin, D.T.; Maheswaran, S.; Teixeira, J.; Fuller, A.F., Jr.; Shah, P.C. Human ovarian cancer, cell lines, and primary ascites cells express the human Mullerian Inhibiting Substance (MIS) type II receptor, bind, and are responsive to MIS. Clin. Cancer Res. 1999, 7, 3488–3499. [Google Scholar]
- Segev, D.L.; Ha, T.U.; Tran, T.T.; Kennealy, M.; Harkin, P.; Jung, M. Mullerian Inhibiting Substance inhibits breast cancer cell growth through an NFkappa B-mediated pathway. J. Biol. Chem. 2000, 275, 28371–28379. [Google Scholar] [CrossRef]
- Pieretti-Vanmarcke, R.; Donahoe, P.K.; Pearsall, L.A.; Dinulescu, D.M.; Connelly, D.C.; Halpern, E.F. Mullerian Inhibiting Substance enhances subclinical doses of chemotherapeutic agents to inhibit human and mouse ovarian cancer. Proc. Natl. Acad. Sci. USA 2006, 103, 17426–17431. [Google Scholar] [CrossRef] [PubMed]
- Fortner, R.T.; Schock, H.; Jung, S.; Allen, N.E.; Arslan, A.A.; Brinton, L.A.; Egleston, B.L.; Falk, R.T.; Gunter, M.J.; Helzlsouer, K.J.; et al. Anti-Mullerian hormone and endometrial cancer: A multi-cohort study. Br. J. Cancer. 2017, 117, 1412–1418. [Google Scholar] [CrossRef] [PubMed]
- Matias-Guiu, X.; Pons, C.; Prat, J. Müllerian inhibiting substance, alpha-inhibin, and CD99 expression in sex cord-stromal tumors and endometrioid ovarian carcinomas resembling sex cordstromal tumors. Hum. Pathol. 1998, 29, 840–845. [Google Scholar] [CrossRef]
- Geerts, I.; Vergote, I.; Neven, P.; Billen, J. The role of inhibins B and antimüllerian hormone for diagnosis and follow-up of granulosa cell tumors. Int. J. Gynecol. Cancer 2009, 19, 847–855. [Google Scholar] [CrossRef] [PubMed]
- Gustafson, M.L.; Lee, M.M.; Scully, R.E.; Moncure, A.C.; Hirakawa, T.; Goodman, A.; Muntz, H.G.; Donahoe, P.K.; MacLaughlin, D.T.; Fuller, A.F., Jr. Mullerian inhibiting substance as a marker for ovarian sex-cord tumor. N. Engl. J. Med. 1992, 326, 466–471. [Google Scholar] [CrossRef] [PubMed]
- Gustafson, M.L.; Lee, M.M.; Asmundson, L.; MacLaughlin, D.T.; Donahoe, P.K. Mullerian inhibiting substance in the diagnosis and management of intersex and gonadal abnormalities. J. Pediatr. Surg. 1993, 28, 439–444. [Google Scholar] [CrossRef]
- Chang, H.L.; Pahlavan, N.; Halpern, E.F.; MacLaughlin, D.T. Serum Mullerian Inhibiting substance/anti-Mullerian hormone levels in patients with adult granulosa cell tumors directly correlate with aggregate tumor mass as determined by pathology or radiology. Gynecol. Oncol. 2009, 114, 57–60. [Google Scholar] [CrossRef]
- Wang, J.; Dicken, C.; Lustbader, J.W.; Tortoriello, D.V. Evidence for a Mullerian-inhibiting substance autocrine/paracrine system in adult human endometrium. Fertil. Steril. 2009, 91, 1195–1203. [Google Scholar] [CrossRef]
- Wang, P.Y.; Koishi, K.; McGeachie, A.B.; Kimber, M.; Maclaughlin, D.T.; Donahoe, P.K.; McLennan, I.S. Mullerian inhibiting substance acts as a motor neuron survival factor in vitro. Proc. Natl. Acad. Sci. USA 2005, 102, 16421–16425. [Google Scholar] [CrossRef]
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer statistics 2017. CA Cancer J. Clin. 2017, 67, 7–30. [Google Scholar] [CrossRef] [PubMed]
- Martinez-Garcia, E.; Lopez-Gil, C.; Campoy, I.; Vallve, J.; Coll, E.; Cabrera, S.; Ramon, Y.; Cajal, S.; Matias-Guiu, X.; Van Oostrum, J.; et al. Advances in endometrial cancer protein biomarkers for use in the clinic. Expert Rev. Proteomics 2018, 15, 81–99. [Google Scholar] [CrossRef] [PubMed]
- Hecht, J.L.; Mutter, G.L. Molecular and pathologic aspects of endometrial carcinogenesis. J. Clin. Oncol. 2006, 24, 4783–4791. [Google Scholar] [CrossRef]
- Arnold, M.; Karim-Kos, H.E.; Coebergh, J.W.; Byrnes, G.; Antilla, A.; Ferlay, J.; Renehan, A.G.; Forman, D.; Soerjomataram, I. Recent trends in incidence of five common cancers in 26 European countries since 1988: Analysis of the European Cancer Observatory. Eur. J. Cancer. 2015, 51, 1164–1187. [Google Scholar] [CrossRef]
- Cook, C.L.; Siow, Y.; Taylor, S.; Fallat, M.E. Serum müllerian-inhibiting substance levels during normal menstrual cycles. Fertil. Steril. 2000, 73, 759–861. [Google Scholar] [CrossRef]
- La Marca, A.; Volpe, A. Anti-Müllerian hormone (AMH) in female reproduction: Is measurement of circulating AMH a useful tool? Clin. Endocrinol. (Oxf.) 2006, 64, 603–610. [Google Scholar] [CrossRef]
- Ravishankar, S.; Mangray, S.; Kurkchubasche, A.; Yakirevich, E.; Young, R.H. Unusual Sertoli Cell Tumor Associated With Sex Cord Tumor With Annular Tubules in Peutz-Jeghers Syndrome: Report of a Case and Review of the Literature on Ovarian Tumors in Peutz-Jeghers Syndrome. Int. J. Surg. Pathol. 2016, 24, 269–273. [Google Scholar] [CrossRef]
- Lane, A.H.; Lee, M.M.; Fuller, A.F.; Kehas, D.J.; Donahoe, P.K.; MacLaughlin, D.T. Diagnostic utility of Müllerian inhibiting substance determination in patients with primary and recurrent granulosa cell tumors. Gynecol. Oncol. 1999, 73, 51–55. [Google Scholar] [CrossRef]
- Wang, F.; Niu, W.B.; Kong, H.J.; Guo, Y.H.; Sun, Y.P. The role of AMH and its receptor SNP in the pathogenesis of PCOS. Mol. Cell Endocrinol. 2017, 439, 363–368. [Google Scholar] [CrossRef]
- Tal, R.; Seifer, D.B.; Khanimov, M.; Malter, H.E.; Grazi, R.V.; Leader, B. Characterization of women with elevated antimüllerian hormone levels (AMH): Correlation of AMH with polycystic ovarian syndrome phenotypes and assisted reproductive technology outcomes. Am. J. Obstet. Gynecol. 2014, 211. [Google Scholar] [CrossRef]
- Reyes-Muñoz, E.; Sathyapalan, T.; Rossetti, P.; Shah, M.; Long, M.; Buscema, M.; Valenti, G.; La Rosa, V.L.; Cianci, S.; Vitale, S.G. Polycystic Ovary Syndrome: Implication for Drug Metabolism on Assisted Reproductive Techniques-A Literature Review. Adv. Ther. 2018, 35, 1805–1815. [Google Scholar] [CrossRef] [PubMed]
- Di Paola, R.; Garzon, S.; Giuliani, S.; Laganà, A.S.; Noventa, M.; Parissone, F.; Zorzi, C.; Raffaelli, R.; Ghezzi, F.; Franchi, M.; Zaffagnini, S. Are we choosing the correct FSH starting dose during controlled ovarian stimulation for intrauterine insemination cycles? Potential application of a nomogram based on woman’s age and markers of ovarian reserve. Arch. Gynecol. Obstet. 2018, 298, 1029–1035. [Google Scholar] [CrossRef]
- Weiderpass, E.; Gridley, G.; Persson, I.; Nyrén, O.; Ekbom, A.; Adami, H.O. Risk of endometrial and breast cancer in patients with diabetes mellitus. Int. J. Cancer. 1997, 71, 360–363. [Google Scholar] [CrossRef]
- Shoff, S.M.; Newcomb, P.A. Diabetes, body size, and risk of endometrial cancer. Am. J. Epidemiol. 1998, 148, 234–240. [Google Scholar] [CrossRef] [PubMed]
- Kawachi, A.; Shimazu, T.; Budhathoki, S.; Sawada, N.; Yamaji, T.; Iwasaki, M.; Inoue, M.; Tsugane, S. JPHC Study Group. Association of BMI and height with the risk of endometrial cancer, overall and by histological subtype: A population-based prospective cohort study in Japan. Eur. J. Cancer Prev. 2018. [Google Scholar] [CrossRef]
- Hardiman, P.; Pillay, O.C.; Atiomo, W. Polycystic ovary syndrome and endometrial carcinoma. Lancet 2003, 361, 1810–1812. [Google Scholar] [CrossRef]
- Dogan, N.U.; Kerimoglu, O.S.; Karabagli, P.; Pekin, A.; Yilmaz, S.A.; Incesu, F.; Celik, C. Anti-Müllerian hormone is associated with extrauterine involvement and stage of disease in patients with endometrial cancer. J. Obstet. Gynaecol. 2015, 35, 178–182. [Google Scholar] [CrossRef] [PubMed]
- MacLaughlin, D.T.; Donahoe, P.K. Mullerian inhibiting substance: A potential nontoxic, naturally occurring therapeutic agent for certain human cancers. In Transforming Growth Factor-β in Cancer Therapy, 1st ed.; Cancer Drug Discovery and Development, Humana Press: New Yrok, NY, USA, 2008; Volume I, pp. 333–354. [Google Scholar]
- Cimino, I.; Casoni, F.; Liu, X.; Messina, A.; Parkash, J.; Jamin, S.P.; Catteau-Jonardm, S.; Collierm, F.; Baroncini, M.; Dewailly, D.; et al. Novel role for anti-Müllerian hormone in the regulation of GnRH neuron excitability and hormone secretion. Nat. Commun. 2016, 7, 10055. [Google Scholar] [CrossRef]
- Bedecarrats, G.Y.; O’Neill, F.H.; Norwitz, E.R.; Kaiser, U.B.; Teixeira, J. Regulation of gonadotropin gene expression by Müllerian Inhibiting Substance. Proc. Natl Acad. Sci. USA 2003, 100, 9348–9353. [Google Scholar] [CrossRef] [PubMed]
- Pieretti-Vanmarcke, R.; Donahoem, P.K.; Szotek, P.; Manganaro, T.; Lorenzen, M.K.; Lorenzen, J.; Connolly, D.C.; Halpern, E.F.; MacLaughlin, D.T. Recombinant human Mullerian inhibiting substance inhibits long-term growth of MIS type II receptor-directed transgenic mouse ovarian cancers in vivo. Clin. Cancer. Res. 2006, 12, 1593–1598. [Google Scholar] [CrossRef]
- Liu, L.; Chen, F.; Xiu, A.; Du, B.; Ai, H.; Xie, W. Identification of Key Candidate Genes and Pathways in Endometrial Cancer by Integrated Bioinformatical Analysis. Asian Pac. J. Cancer Prev. 2018, 19, 969–975. [Google Scholar]
- Chung, Y.J.; Kim, H.J.; Park, S.H.; Yoon, J.H.; Kim, M.R.; Nam, S.W.; MacLaughlin, D.T.; Donahoe, P.K.; Kim, J.H. Transcriptome analysis reveals that Müllerian inhibiting substance regulates signaling pathways that contribute to endometrial carcinogenesis. Int. J. Oncol. 2015, 46, 2039–2046. [Google Scholar] [CrossRef]
- Namkung, J.; Song, J.Y.; Jo, H.H.; Kim, M.R.; Lew, Y.O.; Donahoe, P.K.; MacLaughlin, D.T.; Kim, J.H. Mullerian inhibiting substance induces apoptosis of human endometrial stromal cells in endometriosis. J. Clin. Endocrinol. Metab. 2012, 97, 3224–3230. [Google Scholar] [CrossRef] [PubMed]
- Ha, T.U.; Segev, D.L.; Barbie, D.; Masiakos, P.T.; Tran, T.T.; Dombkowski, D.; Glander, M.; Clarke, T.R.; Lorenzo, H.K.; Donahoe, P.K.; et al. Mullerian inhibiting substance inhibits ovarian cell growth through an Rb-independent mechanism. J. Biol. Chem. 2000, 275, 37101–37109. [Google Scholar] [CrossRef] [PubMed]
- Barbie, T.U.; Barbie, D.A.; MacLaughlin, D.T.; Maheswaran, S.; Donahoe, P.K. Mullerian Inhibiting Substance inhibits cervical cancer cell growth via a pathway involving p130 and p107. Proc. Natl. Acad. Sci. USA 2003, 100, 15601–15606. [Google Scholar] [CrossRef]
- Kandoth, C.; Schultz, N.; Cherniack, A.D.; Akbani, R.; Liu, Y.; Shen, H.; Robertson, A.G.; Pashtan, I.; Shen, R.; Benz, C.C.; et al. Integrated genomic characterization of endometrial carcinoma. Cancer Genome Atlas Research Network. Nature 2013, 497, 67–73. [Google Scholar] [CrossRef]
- Murali, R.; Soslow, R.A.; Weigelt, B. Classification of endometrial carcinoma: More than two types. Lancet Oncol. 2014, 15, e268–e278. [Google Scholar] [CrossRef]
- Segev, D.L.; Hoshiya, Y.; Stephen, A.E.; Hoshiya, M.; Tran, T.T.; MacLaughlin, D.T.; Donahoe, P.K.; Maheswaran, S. Mullerian inhibiting substance regulates NFkappaB signaling and growth of mammary epithelial cells in vivo. J. Biol. Chem. 2001, 276, 26799–26806. [Google Scholar] [CrossRef] [PubMed]
- Segev, D.L.; Hoshiya, Y.; Hoshiya, M.; Tran, T.T.; Carey, J.L.; Stephen, A.E.; MacLaughlin, D.T.; Donahoe, P.K.; Maheswaran, S. Mullerian-inhibiting substance regulates NF-kappa B signaling in the prostate in vitro and in vivo. Proc. Natl. Acad. Sci. USA 2002, 99, 239–244. [Google Scholar] [CrossRef] [PubMed]
- Feldmann, K.A.; Pittelkow, M.R.; Roche, P.C.; Kumar, R.; Grande, J.P. Expression of an immediate early gene, IEX-1, in human tissues. Histochem. Cell Biol. 2001, 115, 489–497. [Google Scholar] [PubMed]
- Wu, M.X.; Ao, Z.; Prasad, K.V.; Wu, R.; Schlossman, S.F. IEX-1L, an apoptosis inhibitor involved in NF-kappaB-mediated cell survival. Science 1998, 281, 998–1001. [Google Scholar] [CrossRef] [PubMed]
- Anttonen, M.; Färkkilä, A.; Tauriala, H.; Kauppinen, M.; Maclaughlin, D.T.; Unkila-Kallio, L.; Bützow, R.; Heikinheimo, M. Anti-Müllerian hormone inhibits growth of AMH type II receptor-positive human ovarian granulosa cell tumor cells by activating apoptosis. Lab Investig. 2011, 91, 1605–1614. [Google Scholar] [CrossRef]
- Renlund, N.; Pieretti-Vanmarcke, R.; O’Neill, F.H.; Zhang, L.; Donahoe, P.K.; Teixeira, J. c-Jun N-terminal kinase inhibitor II (SP600125) activates Mullerian inhibiting substance type II receptor-mediated signal transduction. Endocrinology 2008, 149, 108–115. [Google Scholar] [CrossRef] [PubMed]
- Slamon, D.J.; Leyland-Jones, B.; Shak, S.; Fuchs, H.; Paton, V.; Bajamonde, A.; Fleming, T.; Eiermann, W.; Wolter, J.; Pegram, M. Use of chemotherapy plus a monoclonal antibody against HER2 for metastatic breast cancer that overexpresses HER2. N. Engl. J. Med. 2001, 344, 783–792. [Google Scholar] [CrossRef] [PubMed]
- Geyer, C.E.; Forster, J.; Lindquist, D.; Chan, S.; Romieu, C.G.; Pienkowski, T.; Jagiello-Gruszfeld, A.; Crown, J.; Chan, A.; Kaufman, B. Lapatinib plus capecitabine for HER2-positive advanced breast cancer. N. Engl. J. Med. 2006, 355, 2733–2743. [Google Scholar] [CrossRef]
- Nagai, M.A. Pleckstrin homology-like domain, family A, member 1 (PHLDA1) and cancer. Biomed. Rep. 2016, 4, 275–281. [Google Scholar] [CrossRef]
- Fearon, A.E.; Carter, E.P.; Clayton, N.S.; Wilkes, E.H.; Baker, A.M.; Kapitonova, E.; Bakhouche, B.A.; Tanner, Y.; Wang, J.; Gadaleta, E.; et al. PHLDA1 Mediates Drug Resistance in Receptor Tyrosine Kinase-Driven. Cell Rep. 2018, 22, 2469–2481. [Google Scholar] [CrossRef]
- Neef, R.; Kuske, M.A.; Pröls, E.; Johnson, J.P. Identification of the human PHLDA1/TDAG51 gene: Down-regulation in metastatic melanoma contributes to apoptosis resistance and growth deregulation. Cancer Res. 2002, 62, 5920–5929. [Google Scholar]
- Moad, A.I.; Muhammad, T.S.; Oon, C.E.; Tan, M.L. Rapamycin induces apoptosis when autophagy is inhibited in T-47D mammary cells and both processes are regulated by Phlda1. Cell Biochem. Biophys. 2013, 66, 567–587. [Google Scholar] [CrossRef] [PubMed]
- Meyer, L.A.; Westin, S.N.; Lu, K.H.; Milam, M.R. Genetic polymorphisms and endometrial cancer risk. Expert Rev. Anticancer Ther. 2008, 8, 1159–1167. [Google Scholar] [CrossRef]
- Kang, S.; Kim, J.W.; Park, N.H.; Song, Y.S.; Kang, S.B.; Lee, H.P. Cyclin D1 polymorphism and the risk of endometrial cancer. Gynecol. Oncol. 2005, 97, 431–435. [Google Scholar] [CrossRef]
- Milam, M.R.; Gu, J.; Yang, H.; Celestino, J.; Wu, W.; Horwitz, I.B.; Lacour, R.A.; Westin, S.N.; Gershenson, D.M.; Wu, X.; et al. STK15 F31I polymorphism is associated with increased uterine cancer risk: A pilot study. Gynecol. Oncol. 2007, 107, 71–74. [Google Scholar] [CrossRef]
- Einarsdottir, K.; Humphreys, K.; Bonnard, C.; Li, Y.; Li, Y.; Chia, K.S.; Liu, E.T.; Hall, P.; Liu, J.; Wedrén, S. Effect of ATM, CHEK2 and ERBB2 TAGSNPs and haplotypes on endometrial cancer risk. Hum. Mol. Genet. 2007, 16, 154–164. [Google Scholar] [CrossRef]
- Roh, J.W.; Kim, J.W.; Park, N.H.; Song, Y.S.; Park, I.A.; Park, S.Y.; Kang, S.B.; Lee, H.P. p53 and p21 genetic polymorphisms and susceptibility to endometrial cancer. Gynecol. Oncol. 2004, 93, 499–505. [Google Scholar] [CrossRef] [PubMed]
- Bougeard, G.; Baert-Desurmont, S.; Tournier, I.; Vasseur, S.; Martin, C.; Brugieres, L.; Chompret, A.; Bressac-de Paillerets, B.; Stoppa-Lyonnet, D.; Bonaiti-Pellie, C.; et al. Impact of the MDM2 SNP309 and p53 Arg72Pro polymorphism on age of tumour onsetin Li-Fraumeni syndrome. J. Med. Genet. 2006, 43, 531–533. [Google Scholar] [CrossRef]
- Chen, J.; Wu, X.; Lin, J.; Levine, A.J. MDM-2 inhibits the G1 arrest and apoptosis functions of the p53 tumor suppressor protein. Mol. Cell Biol. 1996, 16, 2445–2452. [Google Scholar] [CrossRef] [PubMed]
- Toledo, F.; Wahl, G.M. Regulating the p53 pathway: In vitro hypotheses, in vivo veritas. Nat. Rev. Cancer 2007, 6, 909–923. [Google Scholar] [CrossRef]
- Zhang, J.; Zhang, Y.; Zhang, Z. Association of rs2279744 and rs117039649 promoter polymorphism with the risk of gynecological cancer: A meta-analysis of case-control studies. Medicine (Baltimore) 2018, 97, e9554. [Google Scholar] [CrossRef]
- Xue, Z.; Zhu, X.; Teng, Y. Relationship Between Murine Double Minute 2 (MDM2) T309G Polymorphism and Endometrial Cancer Risk: A Meta-Analysis. Med. Sci. Monit. 2016, 22, 3186–3190. [Google Scholar] [CrossRef]
- Picard, J.Y.; Benarous, R.; Guerrier, D.; Josso, N.; Kahn, A. Cloning and expression of cDNA for anti-mullerian hormone. Proc. Natl. Acad. Sci. USA 1986, 83, 5464–5468. [Google Scholar] [CrossRef] [PubMed]
- Zakrzewski, P. Zaburzenia kaskady transformujących czynników wzrostu typu β w wybranych patologiach człowieka. Folia Med. Lodz. 2012, 39/2, 265–292. [Google Scholar]
- Glasgow, E.; Mishra, L. Transforming growth factor-beta signaling and ubiquitinators in cancer. Endocr. Relat. Cancer 2008, 15, 59–72. [Google Scholar] [CrossRef] [PubMed]
- Miyazawa, K.; Shinozaki, M.; Hara, T.; Furuya, T.; Miyazono, K. Two major Smad pathways in TGF-beta superfamily signalling. Genes Cells 2002, 7, 1191–1204. [Google Scholar] [CrossRef] [PubMed]
- Liao, J.H.; Chen, J.S.; Chai, M.Q.; Zhao, S.; Song, J.G. The involvement of p38 MAPK in transforming growth factor beta1-induced apoptosis in murine hepatocytes. Cell Res. 2001, 11, 89–94. [Google Scholar] [CrossRef] [PubMed]
- Papageorgis, P.; Stylianopoulos, T. Role of TGFβ in regulation of the tumor microenvironment and drug delivery (review). Int. J. Oncol. 2015, 46, 933–943. [Google Scholar] [CrossRef]
- Blain, S.W.; Massagué, J. Different sensitivity of the transforming growth factor-beta cell cycle arrest pathway to c-Myc and MDM-2. J. Biol. Chem. 2000, 275, 32066–32070. [Google Scholar] [CrossRef]
- Sun, P.; Dong, P.; Dai, K.; Hannon, G.J.; Beach, D. p53-independent role of MDM-2 in TGF-β resistance. Science 1998, 282, 2270–2272. [Google Scholar] [CrossRef] [PubMed]
- Di Clemente, N.; Josso, N.; Gouédard, L.; Belville, C. Components of the anti-Müllerian hormone signaling pathway in gonads. Mol. Cell. Endocrinol. 2003, 211, 9–14. [Google Scholar] [CrossRef] [PubMed]
- Portoso, M.; Cavalli, G. “The Role of RNAi and Noncoding RNAs in Polycomb Mediated Control of Gene Expression and Genomic Programming”. In RNA and the Regulation of Gene Expression: A Hidden Layer of Complexity, 1st ed.; Morris, K.V., Ed.; Caister Academic Press: Norfolk, UK, 2008; pp. 29–44. [Google Scholar]
- Blackledge, N.P.; Rose, N.R.; Klose, R.J. Targeting Polycomb systems to regulate gene expression: Modifications to a complex story. Nat. Rev. Mol. Cell Biol. 2015, 16, 643–649. [Google Scholar] [CrossRef] [PubMed]
- Schuettengruber, B.; Chourrout, D.; Vervoort, M.; Leblanc, B.; Cavalli, G. Genome regulation by polycomb and trithorax proteins. Cell 2007, 128, 735–745. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Ai, Z.; Chen, J.; Teng, Y.; Zhu, J. Enhancer of zeste homolog 2 blockade by RNA interference is implicated with inhibited proliferation, invasion and promoted apoptosis in endometrial carcinoma. Oncol. Lett. 2018, 15, 9429–9435. [Google Scholar] [CrossRef] [PubMed]
- Viré, E.; Brenner, C.; Deplus, R.; Blanchon, L.; Fraga, M.; Didelot, C.; Morey, L.; Van Eynde, A.; Bernard, D.; Vanderwinden, J.M.; et al. The Polycomb group protein EZH2 directly controls DNA methylation. Nature 2006, 439, 871–874. [Google Scholar] [CrossRef] [PubMed]
- Cincin, Z.B.; Kiran, B.; Baran, Y.; Cakmakoglu, B. Hesperidin promotes programmed cell death by downregulation of nongenomic estrogen receptor signalling pathway in endometrial cancer cells. Biomed. Pharmacother. 2018, 103, 336–345. [Google Scholar] [CrossRef] [PubMed]
- Cho, J.H.; Lee, J.G.; Yang, Y.I.; Kim, J.H.; Ahn, J.H.; Baek, N.I.; Lee, K.T.; Choi, J.H. Eupatilin, a dietary flavonoid, induces G2/M cell cycle arrest in human endometrial cancer cells. Food Chem. Toxicol. 2011, 49, 1737–1744. [Google Scholar] [CrossRef] [PubMed]
- Available online: https://www.who.int/gho/ncd/risk_factors/bmi_text/en/ (accessed on 10 March 2018).
- National Collaborating Centre for Women’s and Children’s Health (UK). Diagnosis of perimenopause and menopause. In Menopause: Full Guideline; NICE Guideline No 23; Chard, J., Sarri, G., Eds.; National Institute for Health and Care Excellence: London, UK, 12 November 2015; pp. 49–62. [Google Scholar]
- Lott, R.; Tunnicliffe, J.; Sheppard, E.; Santiago, J.; Hladik, C.; Nasim, M.; Zeitner, K.; Haas, T.; Kohl, S.; Movahedi-Lankarani, S. Practical Guide to Specimen Handling in Surgical Pathology; College of American Pathologists (CAP): Northfield, IL, USA, November 2015; pp. 24–52. [Google Scholar]
- Remmele, W.; Stegner, H.E. Recommendation for uniform definition of an immunoreactive Score (IRS) for immunohistochemical estrogen receptor detection (ER-ICA) in breast cancer tissue. Pathologe 1987, 8, 138–140. [Google Scholar] [PubMed]

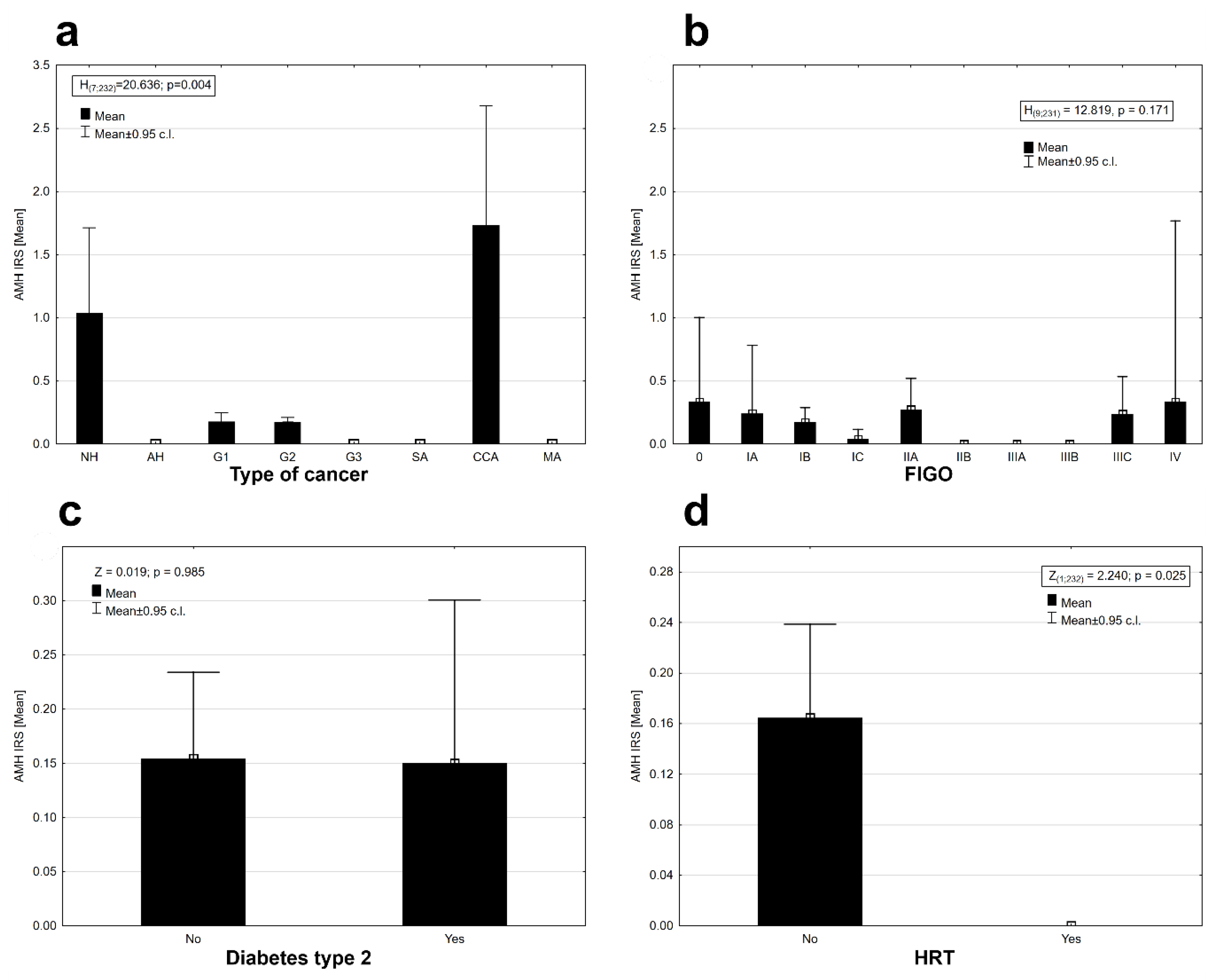
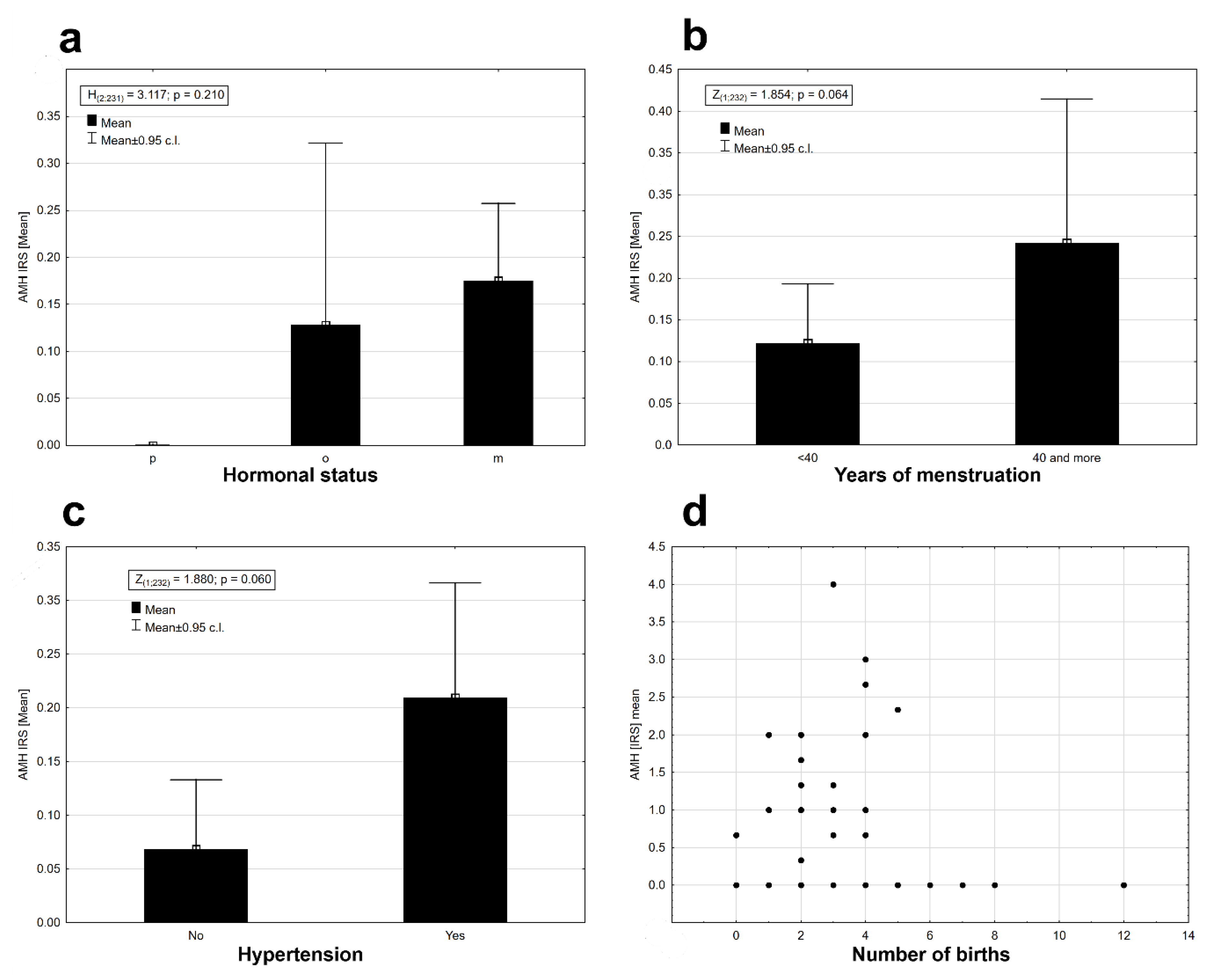
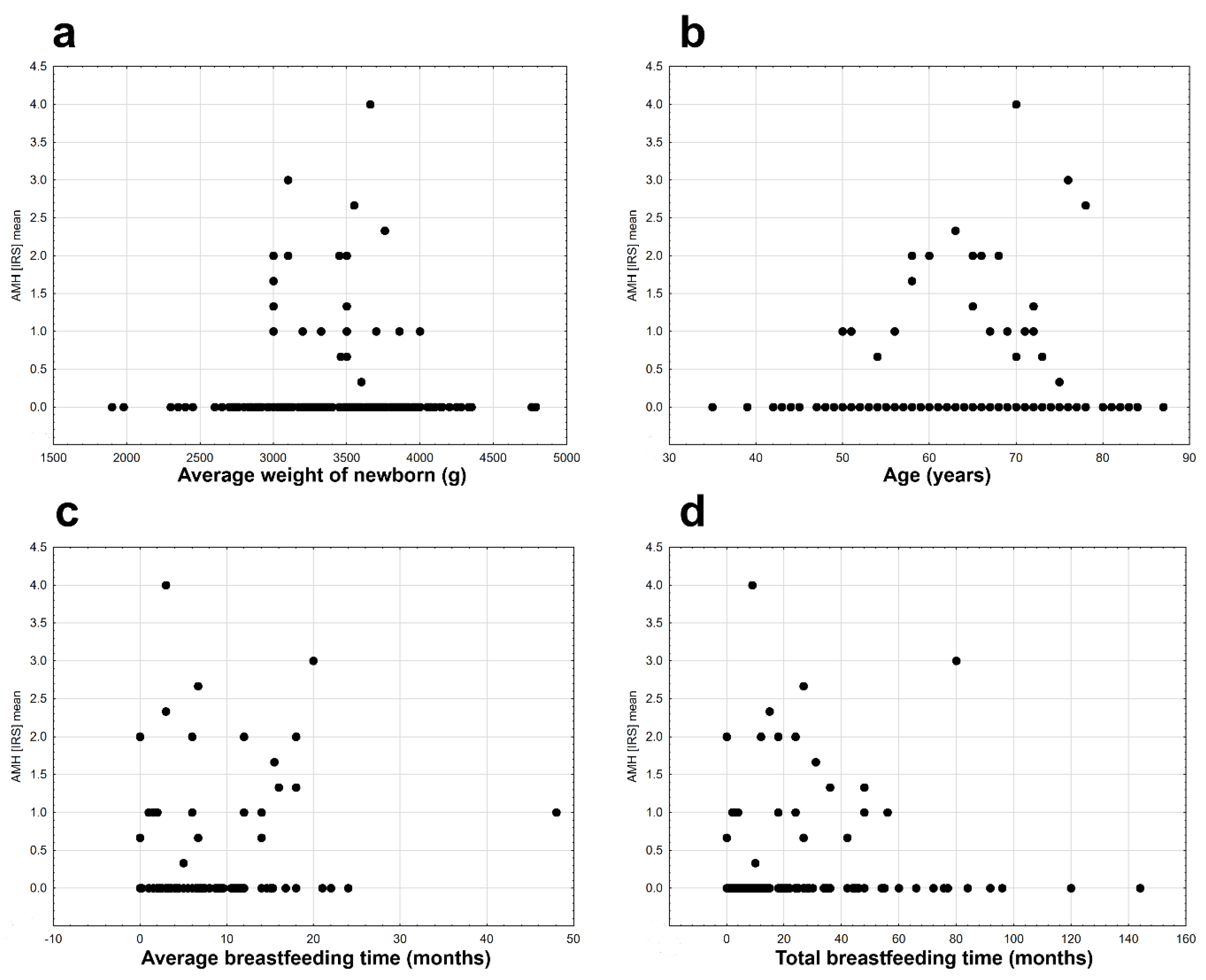
| Histopathological Type of Endometrial Lesion | Number of Patients in Each Group | Number of Patients with Positive AMH Expression | Mean Value of AMH Expression | Minimal AMH Expression | Maximal AMH Expression | SD |
|---|---|---|---|---|---|---|
| Nonatypical endometrial hyperplasia (NH) | 8 | 2 | 2.17 | 0.33 | 4.0 | 2.593 |
| Atypical hyperplasia (AH) | 4 | 0 | - | - | - | - |
| Endometroid adenocarcinoma G1 (G1) | 49 | 3 | 1.78 | 1.0 | 3.0 | 1.072 |
| Endometroid adenocarcinoma G2 (G2) | 147 | 15 | 1.33 | 0.67 | 2.33 | 0.590 |
| Endometroid adenocarcinoma G3 (G3) | 6 | 0 | - | - | - | - |
| Serous adenocarcinoma (SA) | 8 | 0 | - | - | - | - |
| Clear cell adenocarcinoma (CCA) | 5 | 3 | 2.0 | 1.33 | 2.67 | 0.667 |
| Mixed adenocarcinoma (MA) | 5 | 0 | - | - | - | - |
| Variable | df | Max. Likelihood | χ2 | p |
|---|---|---|---|---|
| Average breastfeeding time (continuous variable) | 1 | −58.182 | 8.262 | 0.004 |
| Cancer type (NH, G1, G2, G3, SA, CCA, MA) | 6 | −63.122 | 18.142 | 0.006 |
| Menstrual year (0—below 40; 1—40 and more) | 1 | −56.062 | 4.022 | 0.045 |
| Variable | Level of Variable | Parameter ± SE | Lower 95.0% c.l. | Upper 95.0% c.l. | Wald Function | p |
|---|---|---|---|---|---|---|
| a | −11.274 ± 0.859 | −9.590 | −12.958 | 172.174 | 0.000 | |
| Average breastfeeding time | continuous | 0.091 ± 0.032 | 0.154 | 0.028 | 7.936 | 0.005 |
| Type of cancer | G2 | 8.417 ± 0.925 | 10.230 | 6.604 | 82.794 | 0.000 |
| G1 | 8.125 ± 1.051 | 10.186 | 6.065 | 59.744 | 0.000 | |
| CCA | 12.038 ± 1.445 | 14.870 | 9.205 | 69.362 | 0.000 | |
| Menstrual year | 0—below 40 | −0.555 ± 0.273 | −0.019 | −1.090 | 4.125 | 0.042 |
| Histopathological Type of Endometrial lesion | Number of Patients |
|---|---|
| Nonatypical endometrial hyperplasia | 8 |
| Atypical hyperplasia | 4 |
| Endometrioid adenocarcinoma G1 | 49 |
| Endometrioid adenocarcinoma G2 | 147 |
| Endometrioid adenocarcinoma G3 | 6 |
| Serous adenocarcinoma | 8 |
| Clear cell adenocarcinoma | 5 |
| Mixed adenocarcinoma | 5 |
| A | B |
|---|---|
| 0 pts—no cells with positive reaction | 0 pts—no staining |
| 1 pt—<10% cells with positive reaction | 1 pt—low intensity of staining |
| 2 pts—11%–50% cells with positive reaction | 2 pts—moderate intensity of staining |
| 3 pts—51%–80% cells with positive reaction | 3 pts—intense staining |
| 4 pts—>80% cells with positive reaction |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gowkielewicz, M.; Lipka, A.; Piotrowska, A.; Szadurska-Noga, M.; Nowakowski, J.J.; Dzięgiel, P.; Majewski, M.K.; Jozwik, M.; Majewska, M. Anti-Müllerian Hormone Expression in Endometrial Cancer Tissue. Int. J. Mol. Sci. 2019, 20, 1325. https://doi.org/10.3390/ijms20061325
Gowkielewicz M, Lipka A, Piotrowska A, Szadurska-Noga M, Nowakowski JJ, Dzięgiel P, Majewski MK, Jozwik M, Majewska M. Anti-Müllerian Hormone Expression in Endometrial Cancer Tissue. International Journal of Molecular Sciences. 2019; 20(6):1325. https://doi.org/10.3390/ijms20061325
Chicago/Turabian StyleGowkielewicz, Marek, Aleksandra Lipka, Aleksandra Piotrowska, Marta Szadurska-Noga, Jacek J. Nowakowski, Piotr Dzięgiel, Mariusz Krzysztof Majewski, Marcin Jozwik, and Marta Majewska. 2019. "Anti-Müllerian Hormone Expression in Endometrial Cancer Tissue" International Journal of Molecular Sciences 20, no. 6: 1325. https://doi.org/10.3390/ijms20061325
APA StyleGowkielewicz, M., Lipka, A., Piotrowska, A., Szadurska-Noga, M., Nowakowski, J. J., Dzięgiel, P., Majewski, M. K., Jozwik, M., & Majewska, M. (2019). Anti-Müllerian Hormone Expression in Endometrial Cancer Tissue. International Journal of Molecular Sciences, 20(6), 1325. https://doi.org/10.3390/ijms20061325






