UDP-Glycosyltransferase Genes in the Striped Rice Stem Borer, Chilo suppressalis (Walker), and Their Contribution to Chlorantraniliprole Resistance
Abstract
1. Introduction
2. Results
2.1. Chlorantraniliprole Toxicity and Synergists Assessment
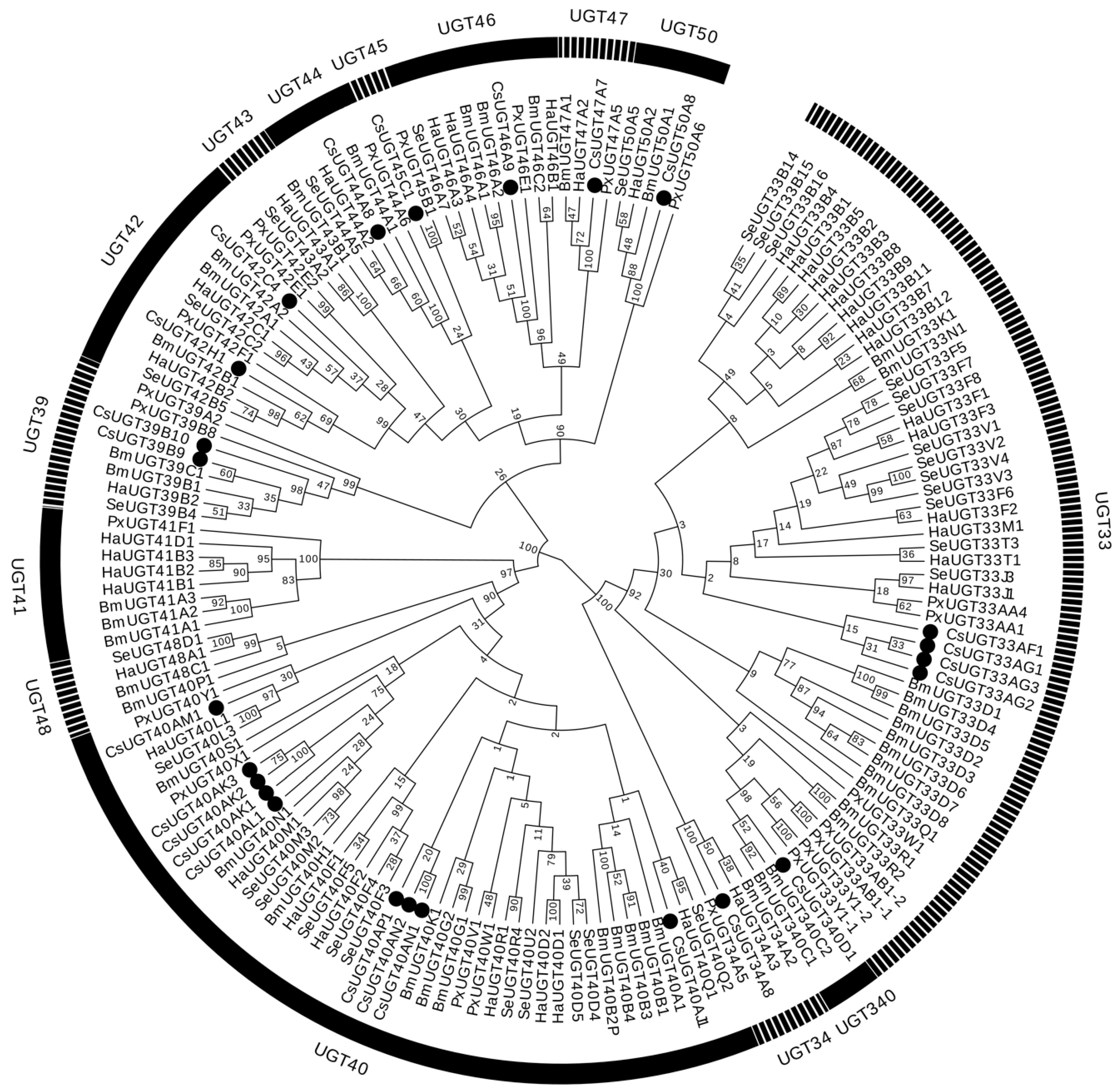
2.2. Identification and Classification of UGT Genes
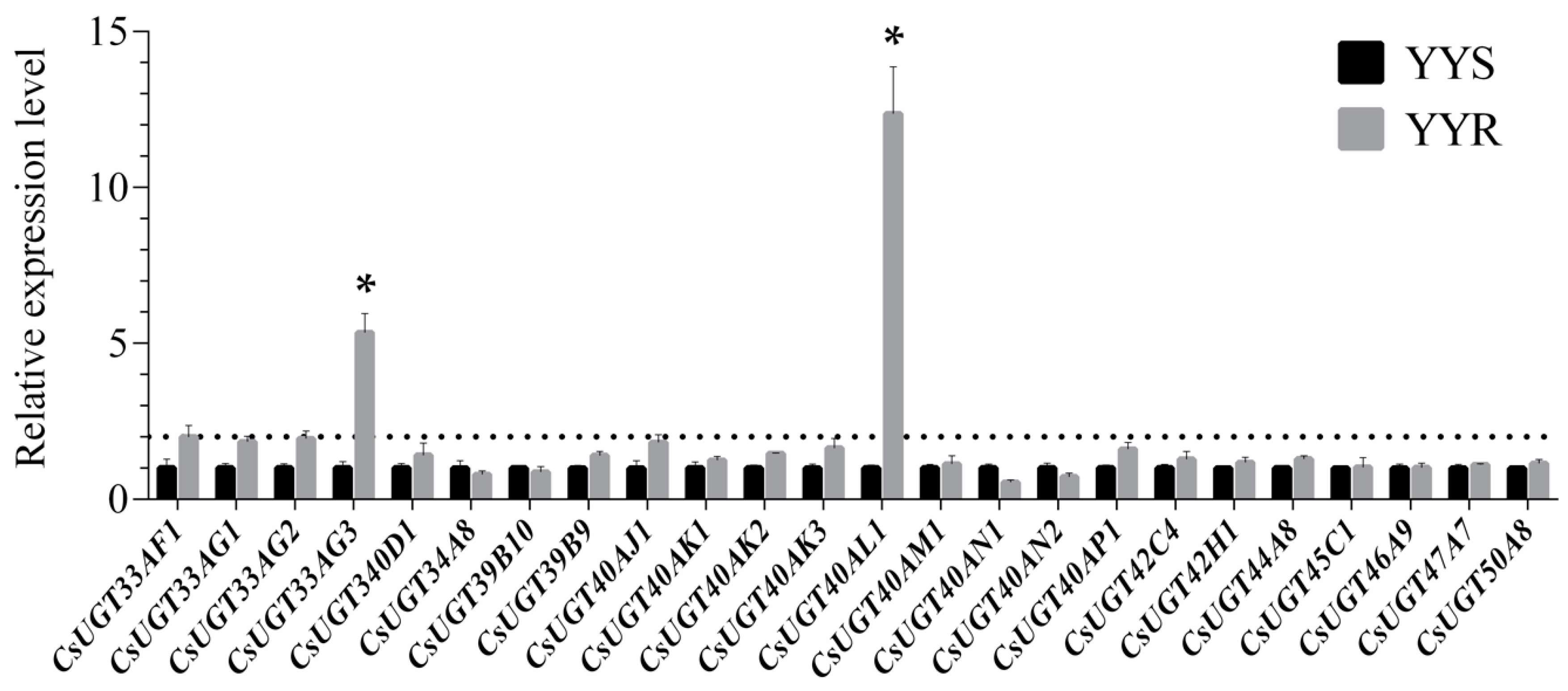
2.3. Screening of Overexpressed UGTs in C. suppressalis with Chlorantraniliprole Resistance
2.4. Expression Patterns of Overexpressed UGTs in Different Tissues
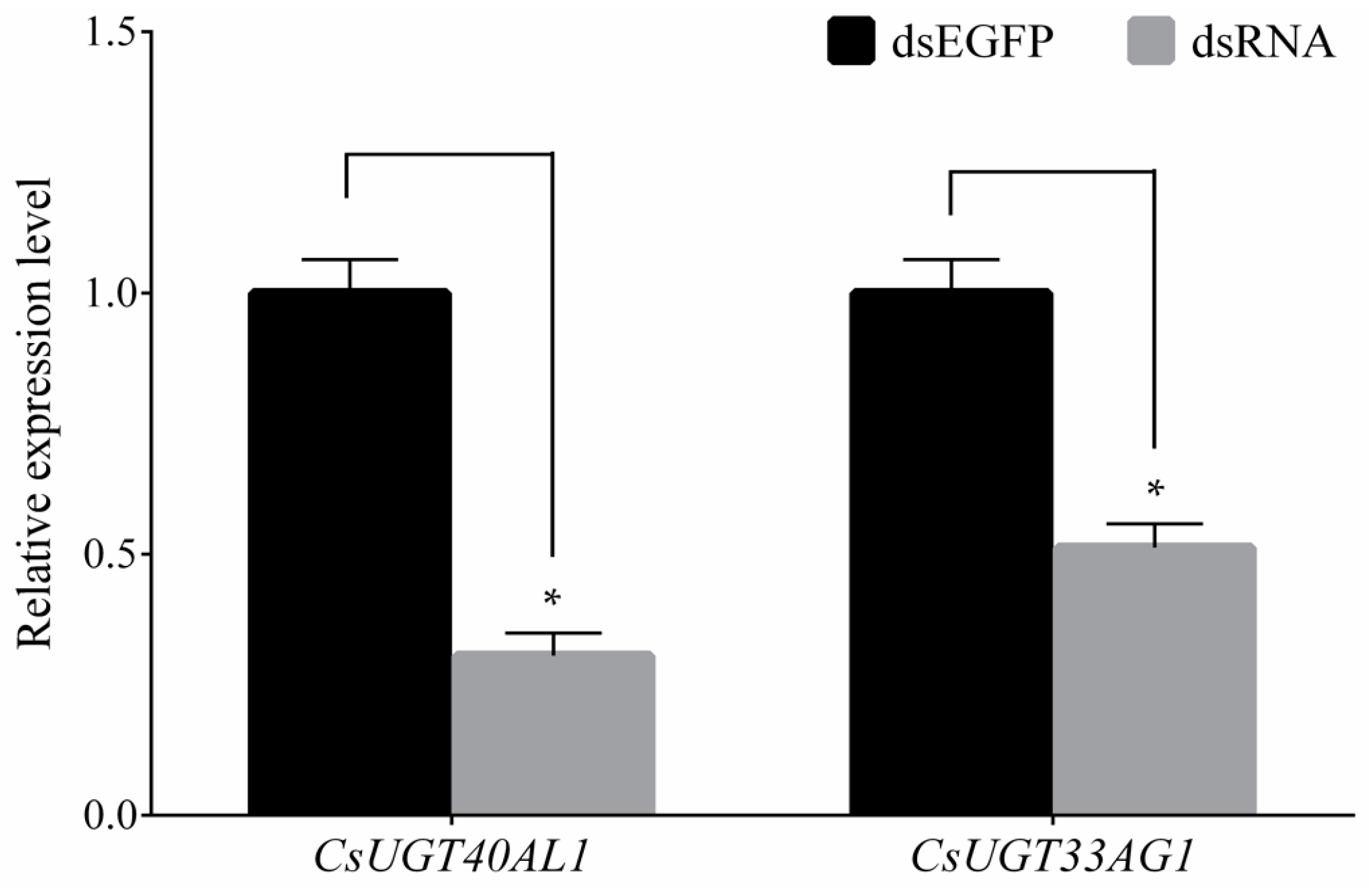
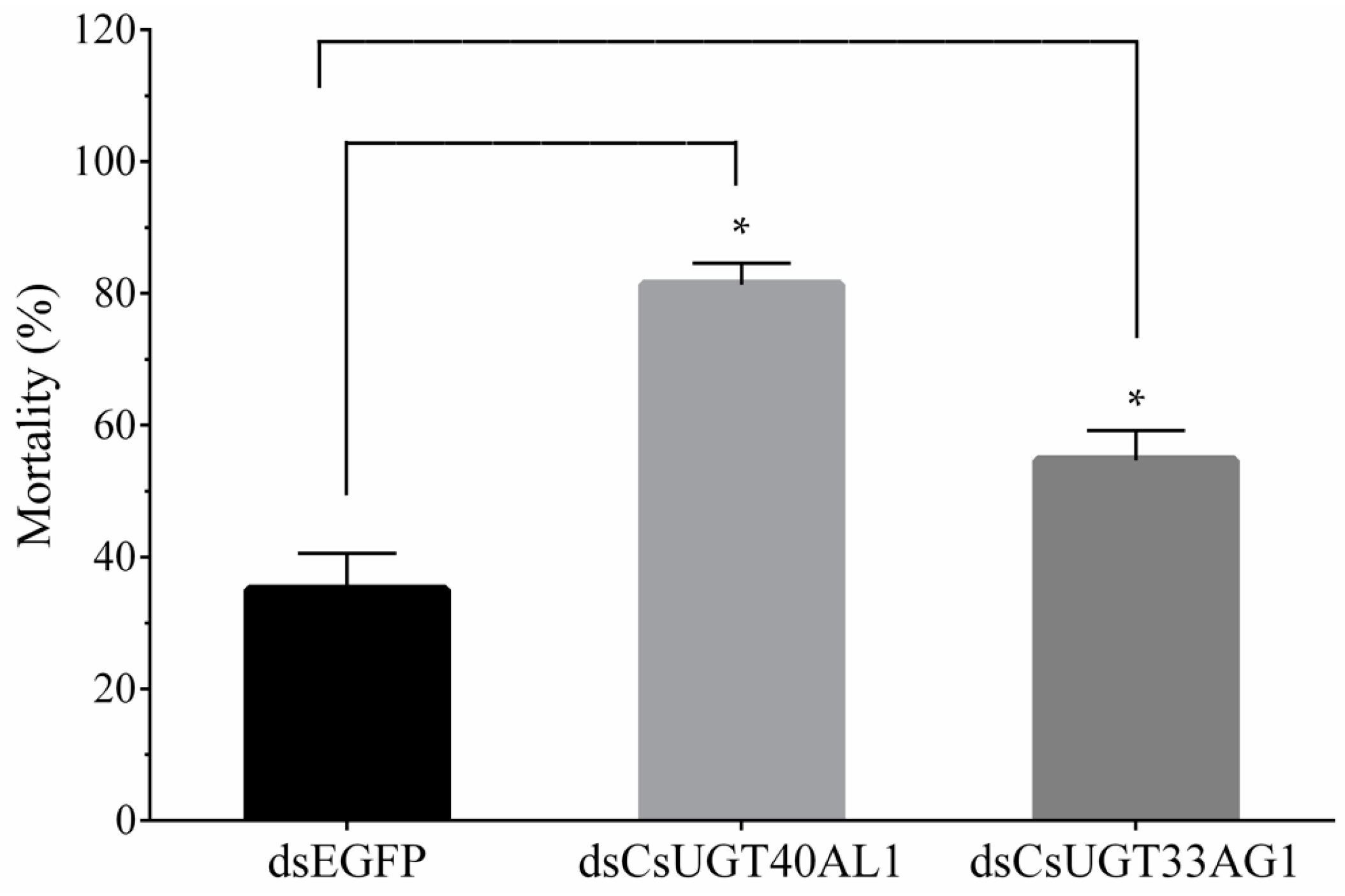
2.5. Confirmation of Overexpressed UGT Genes Function
3. Discussion
4. Materials and Methods
4.1. Insects and Sample Preparation
4.2. Toxicity and Synergism Bioassays
4.3. RNA Isolation and cDNA Synthesis
4.4. Identification of UGT Genes and Phylogenetic Analysis
4.5. Expression Patterns of UGT Genes in Resistant and Susceptible Strains
4.6. Functional Analysis of Overexpressed UGT Genes with RNAi
4.7. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| UGT | Uridine diphosphate glycosyltransferase |
| UDP | Uridine diphosphate |
| RyR | Ryanodine receptor |
| RNAi | RNA interference |
| dsRNA | Double-stranded RNA |
| 5-NU | 5-nitrouracil |
| SUL | Sulfinpyrazone |
| RR | Resistance ratio |
| SR | Synergism ratio |
| CL | Confidence limits |
| ORF | Open reading frame |
| MW | Molecular weight |
| PI | Isoelectric point |
| SP | Signal peptide |
| EGFP | Enhanced green fluorescent protein |
| qRT-PCR | Real-time quantitative reverse transcription polymerase chain reaction |
| RT-PCR | Reverse transcription polymerase chain reaction |
| PBS | Phosphate-buffered saline |
| SD | Standard deviation |
| ANOVA | Analysis of variance |
| Tukey’s HSD | Tukey’s honest significant difference |
References
- Lu, Y.H.; Wang, G.R.; Zhong, L.Q.; Zhang, F.C.; Bai, Q.; Zheng, X.S.; Lu, Z.X. Resistance monitoring of Chilo suppressalis (Walker) (Lepidoptera: Crambidae) to chlorantraniliprole in eight field populations from east and central China. Crop. Prot. 2017, 100, 196–202. [Google Scholar] [CrossRef]
- Huang, S.J.; Chen, Q.; Qin, W.J.; Sun, Y.; Qin, H.G. Resistance monitoring of four insecticides and a description of an artificial diet incorporation method for Chilo suppressalis (Lepidoptera: Crambidae). J. Econ. Entomol. 2017, 110, 2554–2561. [Google Scholar] [CrossRef]
- Su, J.Y.; Zhang, Z.Z.; Wu, M.; Gao, C.F. Geographic susceptibility of Chilo suppressalis Walker (Lepidoptera: Crambidae), to chlorantraniliprole in China. Pest Manag. Sci. 2014, 70, 989–995. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Huang, S.J.; Wang, S.P.; Guo, D.H.; Ge, C.; Xiao, H.M.; Jie, W.C.; Yang, Q.P.; Teng, X.L.; Li, F. Large-scale identification of differentially expressed genes during pupa development reveals solute carrier gene is essential for pupal pigmentation in Chilo suppressalis. J. Insect Physiol. 2017, 98, 117–125. [Google Scholar] [CrossRef] [PubMed]
- Cordova, D.; Benner, E.A.; Sacher, M.D.; Rauh, J.J.; Sopa, J.S.; Lahm, G.P.; Selby, T.P.; Stevenson, T.M.; Flexner, L.; Gutteridge, S.; et al. Anthranilic diamides: A new class of insecticides with a novel mode of action, ryanodine receptor activation. Pestic. Biochem. Physiol. 2006, 84, 196–214. [Google Scholar] [CrossRef]
- He, Y.P.; Zhang, J.F.; Gao, C.F.; Su, J.Y.; Chen, J.M.; Shen, J.L. Regression analysis of dynamics of insecticide resistance in field populations of Chilo suppressalis (Lepidoptera: Crambidae) during 2002–2011 in China. J. Econ. Entomol. 2013, 106, 1832–1837. [Google Scholar] [CrossRef] [PubMed]
- Yao, R.; Zhao, D.D.; Zhang, S.; Zhou, L.Q.; Wang, X.; Gao, C.F.; Wu, S.F. Monitoring and mechanisms of insecticide resistance in Chilo suppressalis (Lepidoptera: Crambidae), with special reference to diamides. Pest Manag. Sci. 2017, 73, 1169–1178. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Xu, L.; Chen, Q.; Qin, W.J.; Huang, S.J.; Jiang, Y.; Qin, H.G. Chlorantraniliprole resistance and its biochemical and new molecular target mechanisms in laboratory and field strains of Chilo suppressalis (Walker). Pest Manag. Sci. 2018, 74, 1416–1423. [Google Scholar] [CrossRef] [PubMed]
- Xu, L.; Zhao, J.; Sun, Y.; Xu, D.; Xu, G.; Xu, X.; Zhang, Y.; Huang, S.; Han, Z.; Gu, Z. Constitutive overexpression of cytochrome P450 monooxygenase genes contributes to chlorantraniliprole resistance in Chilo suppressalis (Walker). Pest Manag. Sci. 2019, 75, 718–725. [Google Scholar] [CrossRef] [PubMed]
- Mackenzie, P.I.; Owens, I.S.; Burchell, B.; Bock, K.W.; Bélanger, A.; Belanger, A.; Fournel-Gigleux, S.; Green, M.; Hum, D.W.; Iyanagi, T.; et al. The UDP glycosyltransferase gene superfamily: Recommended nomenclature update based on evolutionary divergence. Pharmacogenetics 1997, 7, 255–269. [Google Scholar] [CrossRef] [PubMed]
- Bock, K.W. Vertebrate UDP-glucuronosyltransferases: functional and evolutionary aspects. Biochem. Pharmacol. 2003, 66, 691–696. [Google Scholar] [CrossRef]
- Bull, D.L.; Whitten, C.J. Factors influencing organophosphorus insecticide resistance in tobacco budworms. J. Agric. Food. Chem. 1972, 20, 561. [Google Scholar] [CrossRef]
- Lee, S.W.; Ohta, K.; Tashiro, S.; Shono, T. Metabolic resistance mechanisms of the housefly (Musca domestica) resistant to pyraclofos. Pestic. Biochem. Physiol. 2006, 85, 76–83. [Google Scholar] [CrossRef]
- Kaplanoglu, E.; Chapman, P.; Scott, I.M.; Donly, C. Overexpression of a cytochrome P450 and a UDP-glycosyltransferase is associated with imidacloprid resistance in the Colorado potato beetle, Leptinotarsa decemlineata. Sci. Rep. 2017, 7, 1762. [Google Scholar] [CrossRef] [PubMed]
- Pan, Y.O.; Tian, F.Y.; Wei, X.; Wu, Y.Q.; Gao, X.W.; Xi, J.H.; Shang, Q.L. Thiamethoxam resistance in Aphis gossypii glover relies on multiple UDP-glucuronosyltransferases. Front. Physiol. 2018, 9, 322. [Google Scholar] [CrossRef] [PubMed]
- Li, X.X.; Zhu, B.; Gao, X.W.; Liang, P. Over-expression of UDP-glycosyltransferase gene UGT2B17 is involved in chlorantraniliprole resistance in Plutella xylostella (L.). Pest Manag. Sci. 2017, 73, 1402–1409. [Google Scholar] [CrossRef] [PubMed]
- Mackenzie, P.I.; Bock, K.W.; Burchell, B.; Guillemette, C.; Ikushiro, S.; Iyanagi, T.; Miners, J.O.; Owens, I.S.; Nebert, D.W. Nomenclature update for the mammalian UDP glycosyltransferase (UGT) gene superfamily. Pharmacogenet. Genom. 2005, 15, 677–685. [Google Scholar] [CrossRef]
- Wang, X.L.; Wu, Y.D. High levels of resistance to chlorantraniliprole evolved in field populations of Plutella xylostella. J. Econ. Entomol. 2012, 105, 1019–1023. [Google Scholar] [CrossRef] [PubMed]
- Lai, T.C.; Li, J.; Su, J.Y. Monitoring of beet armyworm Spodoptera exigua (Lepidoptera: Noctuidae) resistance to chlorantraniliprole in China. Pestic. Biochem. Physiol. 2011, 101, 198–205. [Google Scholar] [CrossRef]
- Su, J.Y.; Lai, T.C.; Li, J. Susceptibility of field populations of Spodoptera litura (Fabricius) (Lepidoptera: Noctuidae) in China to chlorantraniliprole and the activities of detoxification enzymes. Crop Prot. 2012, 42, 217–222. [Google Scholar] [CrossRef]
- Ahn, S.J.; Vogel, H.; Heckel, D.G. Comparative analysis of the UDP-glycosyltransferase multigene family in insects. Insect Biochem. Mol. Biol. 2012, 42, 133–147. [Google Scholar] [CrossRef] [PubMed]
- Huang, F.F.; Chai, C.L.; Zhang, Z.; Liu, Z.H.; Dai, F.Y.; Lu, C.; Xiang, Z.H. The UDP-glucosyltransferase multigene family in Bombyx mori. BMC Genomics 2008, 9, 563. [Google Scholar] [CrossRef] [PubMed]
- Luque, T.; O’Reilly, D.R. Functional and phylogenetic analyses of a putative Drosophila melanogaster UDP-glycosyltransferase gene. Insect Biochem. Mol. Biol. 2002, 32, 1597–1604. [Google Scholar] [CrossRef]
- Bozzolan, F.; Siaussat, D.; Maria, A.; Durand, N.; Pottier, M.A.; Chertemps, T.; Maïbèche-Coisne, M. Antennal uridine diphosphate (UDP)-glycosyltransferases in a pest insect: diversity and putative function in odorant and xenobiotics clearance. Insect Mol. Biol. 2014, 23, 539–549. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Shi, H.; Gao, X.; Liang, P. Characterization of UDP-glucuronosyltransferase genes and their possible roles in multi-insecticide resistance in Plutella xylostella (L.). Pest Manag. Sci. 2018, 74, 695–704. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.N.; Ma, J.F.; Xu, L.; Dong, Z.P.; Xu, J.W.; Li, M.Y.; Zhu, X.Y. Identification and expression patterns of UDP-glycosyltransferase (UGT) genes from insect pest Athetis lepigone (Lepidoptera: Noctuidae). J. Asia-Pac. Entomol. 2017, 20, 253–259. [Google Scholar] [CrossRef]
- Krempl, C.; Sporer, T.; Reichelt, M.; Ahn, S.; Heidel-Fischer, H.; Vogel, H.; Heckel, D.G.; Joußen, N. Potential detoxification of gossypol by UDP-glycosyltransferases in the two Heliothine moth species Helicoverpa armigera and Heliothis virescens. Insect Biochem. Mol. Biol. 2016, 71, 49–57. [Google Scholar] [CrossRef] [PubMed]
- Xu, L.; Zhao, C.Q.; Xu, D.J.; Xu, G.C.; Xu, X.L.; Han, Z.J.; Zhang, Y.N.; Gu, Z.Y. RNAi suppression of nuclear receptor genes results in increased susceptibility to sulfoxaflor in brown planthopper, Nilaparvata lugens. J. Asia-Pac. Entomol. 2017, 20, 645–653. [Google Scholar] [CrossRef]
- Tian, F.; Wang, Z.; Li, C.; Liu, J.; Zeng, X. UDP-Glycosyltransferases are involved in imidacloprid resistance in the Asian citrus psyllid, Diaphorina citri (Hemiptera: Lividae). Pestic. Biochem. Physiol. 2019, 154, 23–31. [Google Scholar] [CrossRef] [PubMed]
- Yin, C.L.; Shen, G.Y.; Guo, D.H.; Wang, S.P.; Ma, X.Z.; Xiao, H.M.; Liu, J.D.; Zhang, Z.; Liu, Y.; Zhang, Y.Q.; et al. InsectBase: a resource for insect genomes and transcriptomes. Nucleic Acids Res. 2016, 44, D801–D807. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Liu, Y.; Zhou, J.J.; Yi, J.K.; Pan, Y.; Wang, J.; Zhang, X.X.; Wang, J.X.; Yang, S.; Xi, J.H. Identification and tissue expression profiling of candidate UDP-glycosyltransferase genes expressed in Holotrichia parallela motschulsky antennae. Bull. Entomol. Res. 2018, 108, 807–816. [Google Scholar] [CrossRef] [PubMed]
- Larkin, M.A.; Blackshields, G.; Brown, N.P.; Chenna, R.; McGettigan, P.A.; McWilliam, H.; Valentin, F.; Wallace, I.M.; Wilm, A.; Lopez, R.; et al. Clustal W and Clustal X version 2.0. Bioinformatics 2007, 23, 2947–2948. [Google Scholar] [CrossRef] [PubMed]
- Tamura, K.; Peterson, D.; Peterson, N.; Stecher, G.; Nei, M.; Kumar, S. MEGA5: molecular gvolutionary genetics analysis using maximum likelihood, evolutionary distance, and maximum parsimony methods. Mol. Biol. Evol. 2011, 28, 2731–2739. [Google Scholar] [CrossRef] [PubMed]
- Thompson, J.D.; Higgins, D.G.; Gibson, T.J. Clustal W: improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position-specific gap penalties and weight matrix choice. Nucleic Acids Res. 1994, 22, 4673–4680. [Google Scholar] [CrossRef] [PubMed]
- Hellemans, J.; Mortier, G.; Paepe, D.A.; Speleman, F.; Vandesompele, J. qBase relative quantification framework and software for management and automated analysis of real-time quantitative PCR data. Genome Biol. 2007, 8, R19. [Google Scholar] [CrossRef] [PubMed]


| Strain | Treatment | LC50 (95% CL) (mg·L−1) | Slope ± SE | x2 | RR | SR |
|---|---|---|---|---|---|---|
| YYS | Chlorantraniliprole | 12.76 (11.08–14.76) | 2.72 ± 0.31 | 5.96 | 1.00 | 1.00 |
| Chlorantraniliprole + 5-NU | 10.48 (8.95–12.27) | 2.42 ± 0.35 | 5.17 | - | 1.22 | |
| Chlorantraniliprole + SUL | 11.27 (9.76–13.03) | 2.70 ± 0.32 | 7.72 | - | 1.13 | |
| YYR | Chlorantraniliprole | 565.48 (510.42–626.47) | 3.67 ± 0.22 | 7.31 | 44.32 | 1.00 |
| Chlorantraniliprole + 5-NU | 167.47 (146.78–191.08) | 2.97 ± 0.29 | 2.68 | - | 3.38 * | |
| Chlorantraniliprole + SUL | 158.71 (139.32–180.79) | 3.08 ± 0.28 | 7.75 | - | 3.56 * |
| Gene Name | Accession Number | ORF Size (aa) | PI/MW (KDa) | SP | ‘UDPGT’ Domain | Best Blastx Match | ||
|---|---|---|---|---|---|---|---|---|
| Species Name 1 | GenBank Accession Number | Identity | ||||||
| CsUGT33AF1 | MK135471 | 518 | 7.77/59.32 | 1–19 | 22–517 | Ha | XP_021197637.1 | 62% |
| CsUGT33AG1 | MK135472 | 524 | 6.91/60.06 | 1–22 | 24–517 | Pp | XP_013139422.1 | 61% |
| CsUGT33AG2 | MK135473 | 520 | 6.77/59.27 | 1–20 | 24–517 | Ha | AEW43120.1 | 55% |
| CsUGT33AG3 | MK135474 | 520 | 9.14/59.67 | 1–23 | 25–519 | Ha | AEW43118.1 | 57% |
| CsUGT340D1 | MK135475 | 523 | 7.24/59.54 | 1–19 | 21–519 | Bm | NP_001243963.1 | 52% |
| CsUGT34A8 | MK135476 | 523 | 8.30/59.31 | – | 22–510 | At | XP_013184861.1 | 62% |
| CsUGT39B10 | MK135477 | 528 | 9.00/61.43 | – | 110–517 | Bm | NP_001243980.1 | 69% |
| CsUGT39B9 | MK135478 | 528 | 9.01/60.69 | 1–21 | 95–516 | Se | ANI21999.1 | 66% |
| CsUGT40AJ1 | MK135479 | 514 | 8.87/58.20 | – | 19–513 | Ha | XP_021183292.1 | 56% |
| CsUGT40AK1 | MK135480 | 519 | 8.39/59.76 | – | 29–518 | Ha | AEW43128.1 | 52% |
| CsUGT40AK2 | MK135481 | 519 | 8.83/59.17 | – | 37–515 | Ha | AEW43128.1 | 52% |
| CsUGT40AK3 | MK135482 | 520 | 8.76/59.33 | 1–18 | 27–516 | Ha | XP_021191114.1 | 54% |
| CsUGT40AL1 | MK135483 | 531 | 9.32/61.01 | – | 32–531 | Ha | AEW43128.1 | 54% |
| CsUGT40AM1 | MK135484 | 521 | 8.13/59.30 | 1–20 | 22–520 | Dp | OWR51610.1 | 59% |
| CsUGT40AN1 | MK135485 | 515 | 9.00/57.36 | – | 22–514 | At | XP_013186135.1 | 56% |
| CsUGT40AN2 | MK135486 | 514 | 8.94/57.42 | – | 22–513 | At | XP_013189977.1 | 56% |
| CsUGT40AP1 | MK135487 | 519 | 9.22/58.68 | 1–19 | 24–518 | Bm | XP_012550238.1 | 56% |
| CsUGT42C4 | MK135488 | 505 | 7.63/57.49 | 1–21 | 24–500 | Se | ANI22012.1 | 65% |
| CsUGT42H1 | MK135489 | 524 | 8.67/59.39 | 1–22 | 24–523 | Pm | XP_014358643.1 | 61% |
| CsUGT44A8 | MK135490 | 515 | 6.79/59.79 | – | 30–511 | Ha | XP_021186279.1 | 62% |
| CsUGT45C1 | MK135491 | 515 | 8.33/58.49 | 1–19 | 24–502 | Pp | XP_013141171.1 | 54% |
| CsUGT46A9 | MK135492 | 533 | 8.96/60.29 | 1–23 | 27–528 | At | XP_013183121.1 | 66% |
| CsUGT47A7 | MK135493 | 530 | 8.94/60.44 | 1–18 | 19–510 | At | XP_013192144.1 | 78% |
| CsUGT50A8 | MK135494 | 544 | 8.88/62.11 | 1–21 | 79–516 | Ha | XP_021184423.1 | 74% |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhao, J.; Xu, L.; Sun, Y.; Song, P.; Han, Z. UDP-Glycosyltransferase Genes in the Striped Rice Stem Borer, Chilo suppressalis (Walker), and Their Contribution to Chlorantraniliprole Resistance. Int. J. Mol. Sci. 2019, 20, 1064. https://doi.org/10.3390/ijms20051064
Zhao J, Xu L, Sun Y, Song P, Han Z. UDP-Glycosyltransferase Genes in the Striped Rice Stem Borer, Chilo suppressalis (Walker), and Their Contribution to Chlorantraniliprole Resistance. International Journal of Molecular Sciences. 2019; 20(5):1064. https://doi.org/10.3390/ijms20051064
Chicago/Turabian StyleZhao, Jun, Lu Xu, Yang Sun, Pingping Song, and Zhaojun Han. 2019. "UDP-Glycosyltransferase Genes in the Striped Rice Stem Borer, Chilo suppressalis (Walker), and Their Contribution to Chlorantraniliprole Resistance" International Journal of Molecular Sciences 20, no. 5: 1064. https://doi.org/10.3390/ijms20051064
APA StyleZhao, J., Xu, L., Sun, Y., Song, P., & Han, Z. (2019). UDP-Glycosyltransferase Genes in the Striped Rice Stem Borer, Chilo suppressalis (Walker), and Their Contribution to Chlorantraniliprole Resistance. International Journal of Molecular Sciences, 20(5), 1064. https://doi.org/10.3390/ijms20051064





