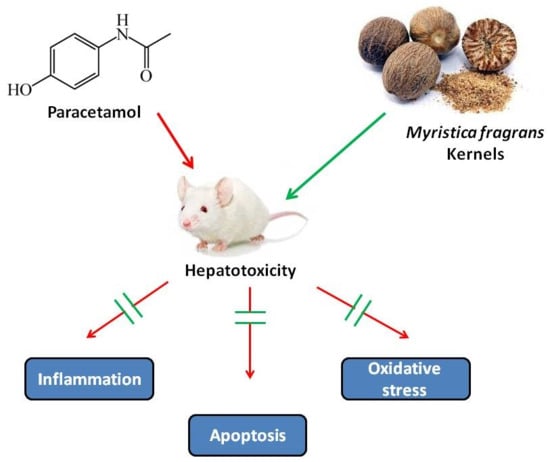
Myristica fragrans Kernels Prevent Paracetamol-Induced Hepatotoxicity by Inducing Anti-Apoptotic Genes and Nrf2/HO-1 Pathway
Abstract
1. Introduction
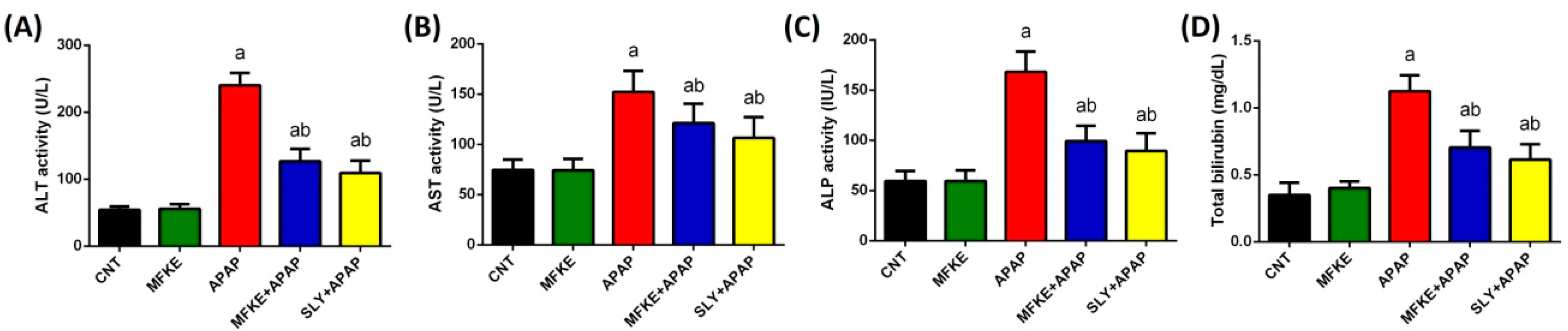
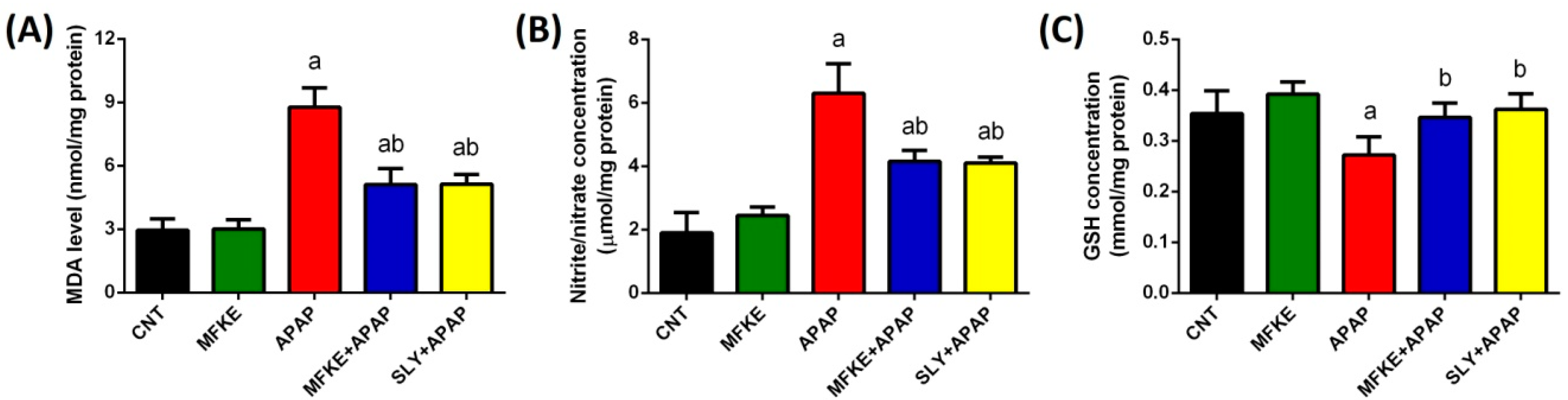
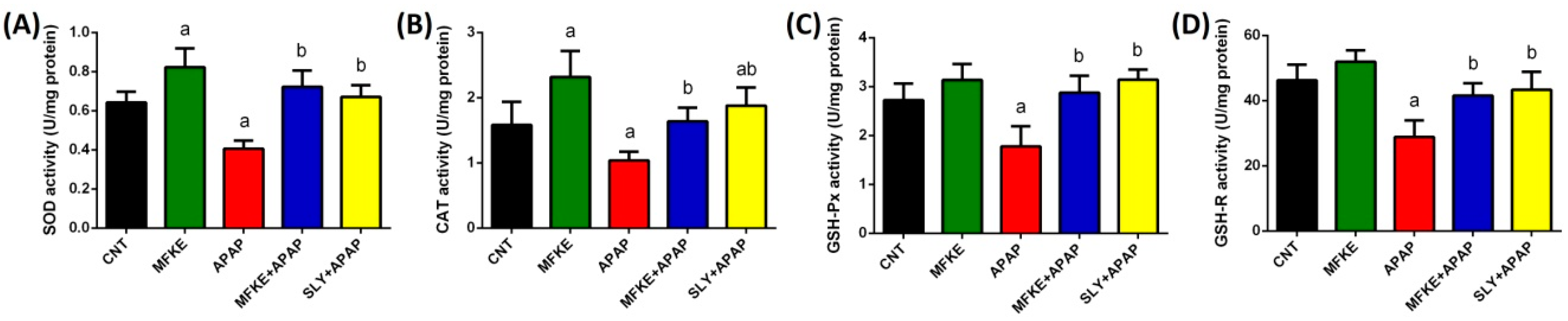
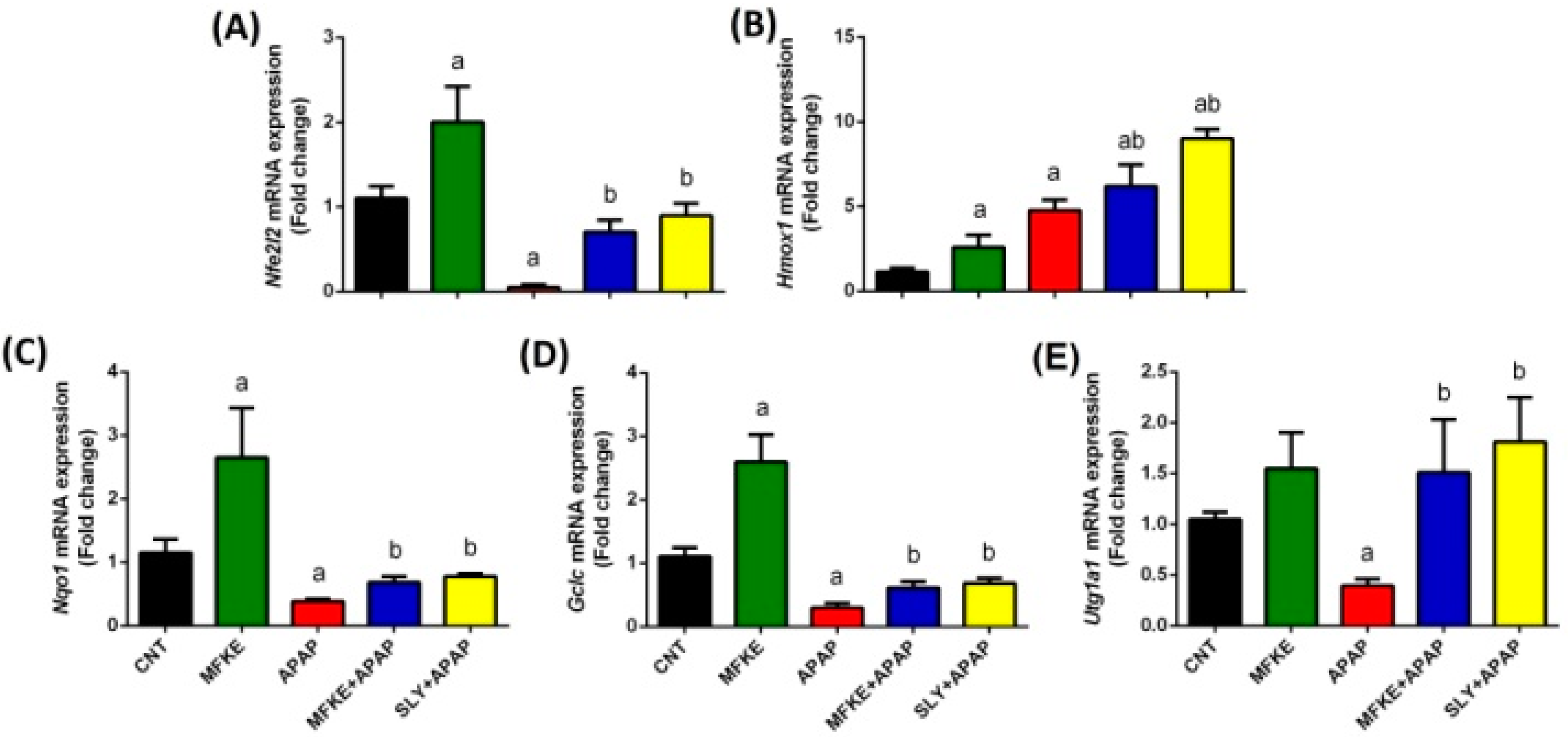
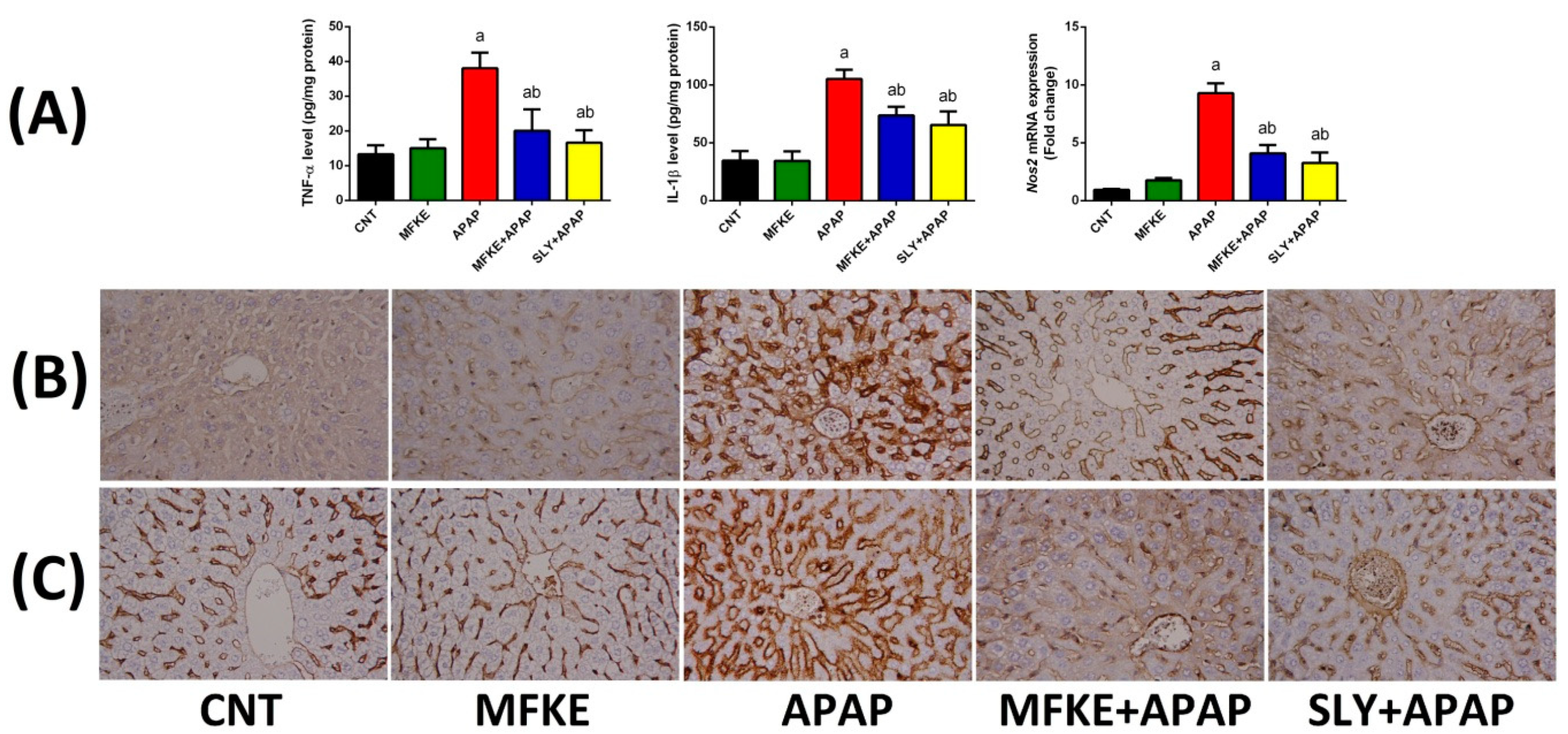
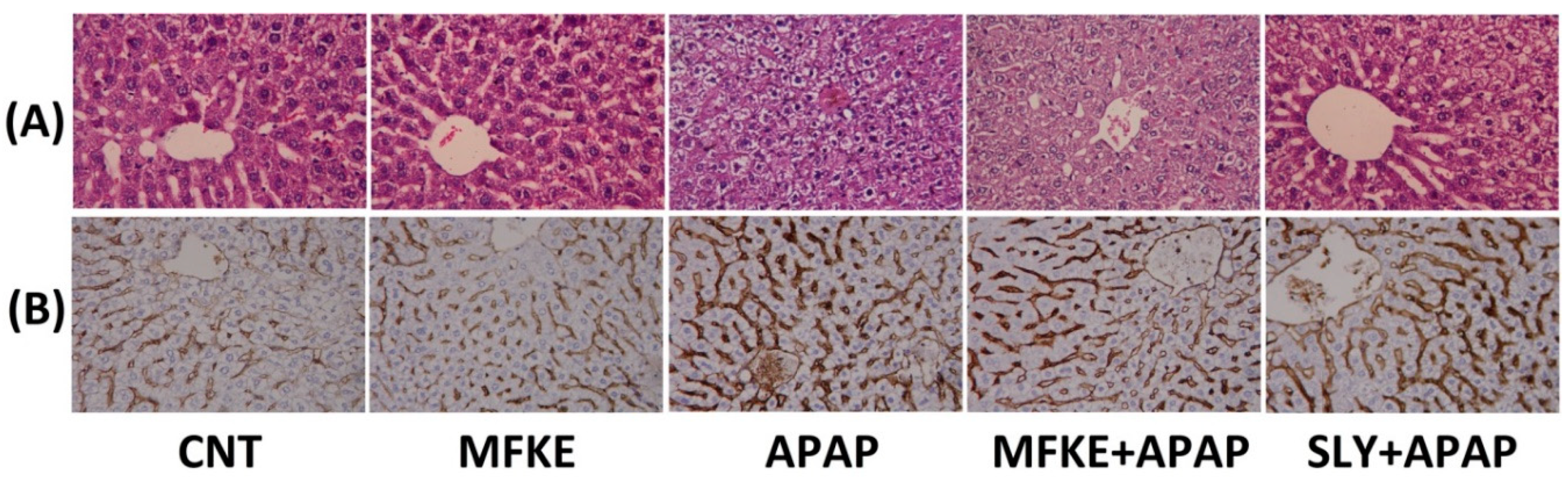
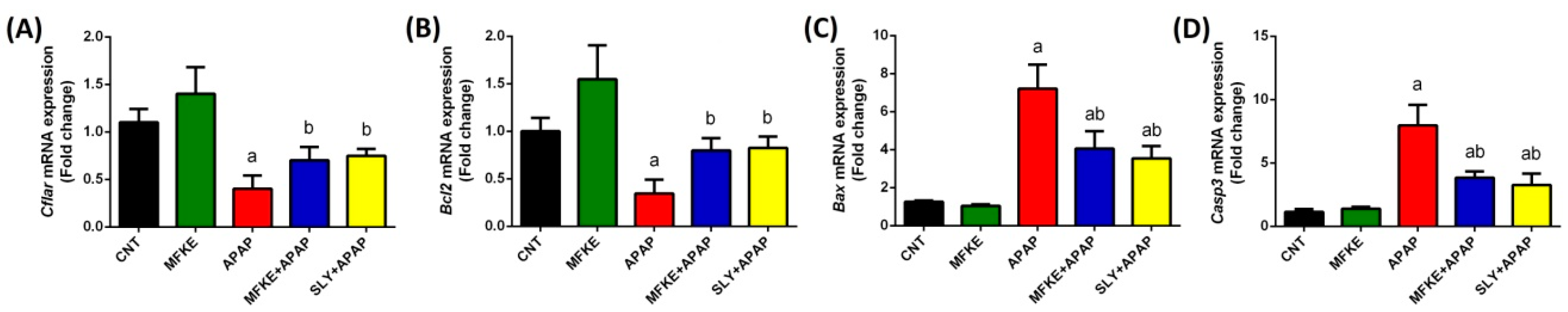
2. Results
3. Discussion
4. Materials and Methods
4.1. Chemicals
4.2. Plant Materials and Extraction Procedure
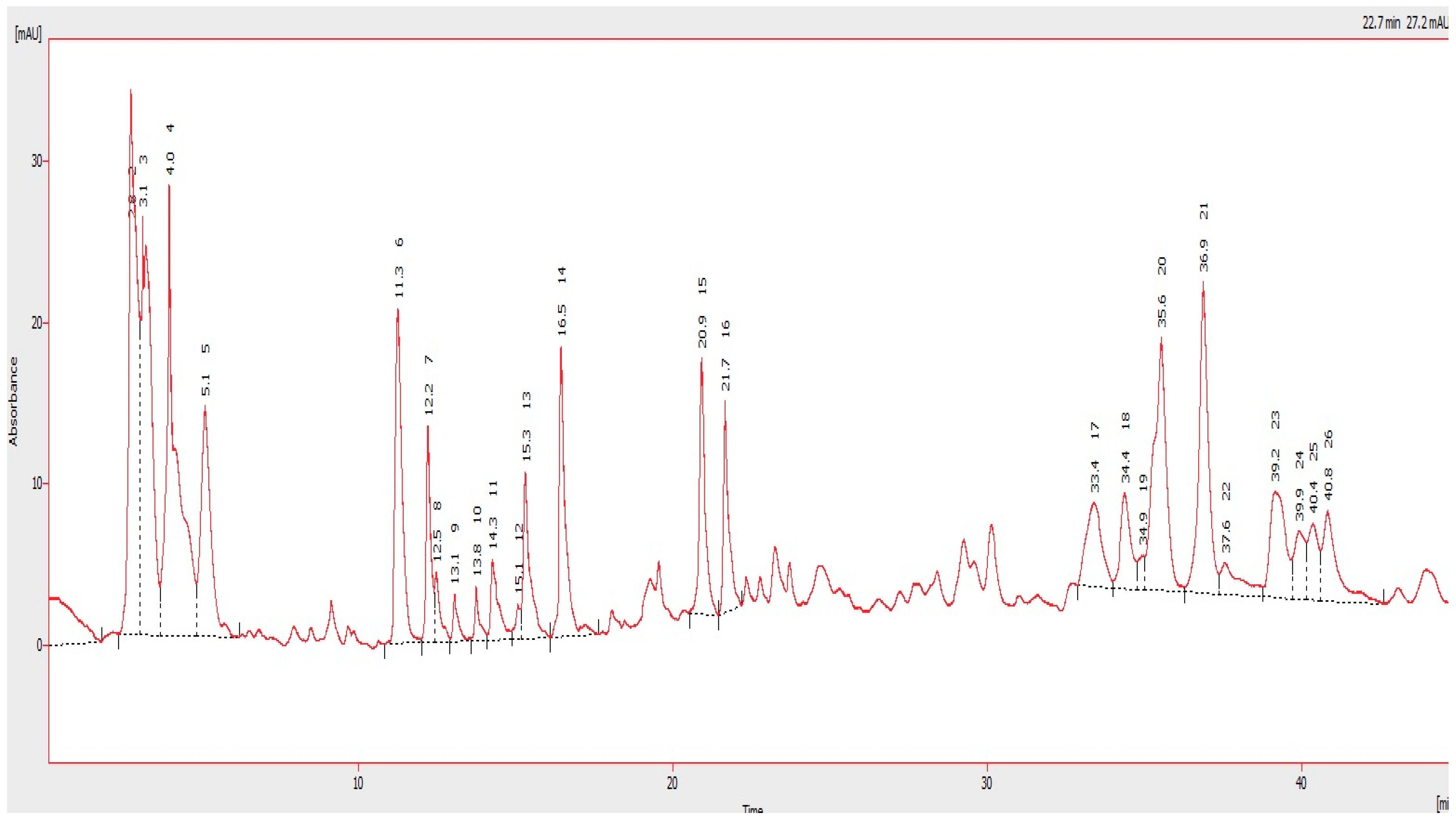
4.3. HPLC Analysis
4.4. Animals
4.5. Experimental Design
4.6. Biochemical Parameters
4.6.1. Liver Functions Tests
4.6.2. Determination of Malondialdehyde and Nitric Oxide
4.6.3. Nonenzymatic and Enzymatic Antioxidant Molecules
4.6.4. Determination of Proinflammation Markers
4.6.5. Quantitative Reverse Transcription-Polymerase Chain Reaction (qRT-PCR) Analysis
4.7. Histopathological Examination
4.8. Immunohistochemistry Analysis
4.9. Statistical Analysis
Supplementary Materials
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Almeer, R.S.; Alarifi, S.; Alkahtani, S.; Ibrahim, S.R.; Ali, D.; Moneim, A. The potential hepatoprotective effect of royal jelly against cadmium chloride-induced hepatotoxicity in mice is mediated by suppression of oxidative stress and upregulation of nrf2 expression. Biomed. Pharmacother. 2018, 106, 1490–1498. [Google Scholar] [CrossRef] [PubMed]
- Bunchorntavakul, C.; Reddy, K.R. Acetaminophen-related hepatotoxicity. Clin. Liver Dis. 2013, 17, 587–607. [Google Scholar] [CrossRef] [PubMed]
- Herndon, C.M.; Dankenbring, D.M. Patient perception and knowledge of acetaminophen in a large family medicine service. J. Pain Palliat. Care Pharmacother. 2014, 28, 109–116. [Google Scholar] [CrossRef] [PubMed]
- Blieden, M.; Paramore, L.C.; Shah, D.; Ben-Joseph, R. A perspective on the epidemiology of acetaminophen exposure and toxicity in the united states. Expert Rev. Clin. Pharmacol. 2014, 7, 341–348. [Google Scholar] [CrossRef] [PubMed]
- Yoon, E.; Babar, A.; Choudhary, M.; Kutner, M.; Pyrsopoulos, N. Acetaminophen-induced hepatotoxicity: A comprehensive update. J. Clin. Transl. Hepatol. 2016, 4, 131–142. [Google Scholar] [PubMed]
- Mahmood, N.D.; Mamat, S.S.; Kamisan, F.H.; Yahya, F.; Kamarolzaman, M.F.F.; Nasir, N.; Mohtarrudin, N.; Tohid, S.F.M.; Zakaria, Z.A. Amelioration of paracetamol-induced hepatotoxicity in rat by the administration of methanol extract of muntingia calabura l. Leaves. BioMed Res. Int. 2014, 2014, 10. [Google Scholar] [CrossRef] [PubMed]
- El-Kott, A.F.; Bin-Meferij, M.M. Use of arctium lappa extract against acetaminophen-induced hepatotoxicity in rats. Curr. Ther. Res. 2015, 77, 73–78. [Google Scholar] [CrossRef] [PubMed]
- Abirami, A.; Nagarani, G.; Siddhuraju, P. Hepatoprotective effect of leaf extracts from citrus hystrix and c. Maxima against paracetamol induced liver injury in rats. Food Sci. Hum. Wellness 2015, 4, 35–41. [Google Scholar] [CrossRef]
- Kareem, M.A.; Gadhamsetty, S.K.; Shaik, A.H.; Prasad, E.M.; Kodidhela, L.D. Protective effect of nutmeg aqueous extract against experimentally-induced hepatotoxicity and oxidative stress in rats. J. Ayurveda Integr. Med. 2013, 4, 216–223. [Google Scholar] [CrossRef] [PubMed]
- Francis, S.K.; James, B.; Varughese, S.; Nair, M.S. Phytochemical investigation on myristica fragrans stem bark. Nat. Prod. Res. 2018. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.K.; Tao, S.S.; Li, T.T.; Li, Y.S.; Li, X.J.; Tang, H.B.; Cong, R.H.; Ma, F.L.; Wan, C.J. Nutmeg oil alleviates chronic inflammatory pain through inhibition of cox-2 expression and substance p release in vivo. Food Nutr. Res. 2016, 60, 30849. [Google Scholar] [CrossRef] [PubMed]
- Broadhurst, C.L.; Polansky, M.M.; Anderson, R.A. Insulin-like biological activity of culinary and medicinal plant aqueous extracts in vitro. J. Agric. Food Chem. 2000, 48, 849–852. [Google Scholar] [CrossRef] [PubMed]
- Kimura, Y.; Ito, H.; Hatano, T. Effects of mace and nutmeg on human cytochrome p450 3a4 and 2c9 activity. Biol. Pharm. Bull. 2010, 33, 1977–1982. [Google Scholar] [CrossRef] [PubMed]
- Stec, D.E.; John, K.; Trabbic, C.J.; Luniwal, A.; Hankins, M.W.; Baum, J.; Hinds, T.D., Jr. Bilirubin binding to pparalpha inhibits lipid accumulation. PLoS ONE 2016, 11, e0153427. [Google Scholar] [CrossRef] [PubMed]
- Hinds, T.D., Jr.; Sodhi, K.; Meadows, C.; Fedorova, L.; Puri, N.; Kim, D.H.; Peterson, S.J.; Shapiro, J.; Abraham, N.G.; Kappas, A. Increased ho-1 levels ameliorate fatty liver development through a reduction of heme and recruitment of fgf21. Obesity 2014, 22, 705–712. [Google Scholar] [CrossRef] [PubMed]
- Morita, T.; Jinno, K.; Kawagishi, H.; Arimoto, Y.; Suganuma, H.; Inakuma, T.; Sugiyama, K. Hepatoprotective effect of myristicin from nutmeg (myristica fragrans) on lipopolysaccharide/d-galactosamine-induced liver injury. J. Agric. Food Chem. 2003, 51, 1560–1565. [Google Scholar] [CrossRef] [PubMed]
- Sharma, M.; Kumar, M. Radioprotection of swiss albino mice by myristica fragrans houtt. J. Radiat. Res. 2007, 48, 135–141. [Google Scholar] [CrossRef] [PubMed]
- Du, K.; Ramachandran, A.; Jaeschke, H. Oxidative stress during acetaminophen hepatotoxicity: Sources, pathophysiological role and therapeutic potential. Redox Biol. 2016, 10, 148–156. [Google Scholar] [CrossRef] [PubMed]
- Du, K.; McGill, M.R.; Xie, Y.; Bajt, M.L.; Jaeschke, H. Resveratrol prevents protein nitration and release of endonucleases from mitochondria during acetaminophen hepatotoxicity. Food Chem. Toxicol. 2015, 81, 62–70. [Google Scholar] [CrossRef] [PubMed]
- Papackova, Z.; Heczkova, M.; Dankova, H.; Sticova, E.; Lodererova, A.; Bartonova, L.; Poruba, M.; Cahova, M. Silymarin prevents acetaminophen-induced hepatotoxicity in mice. PLoS ONE 2018, 13, e0191353. [Google Scholar] [CrossRef] [PubMed]
- Shanmugam, S.; Thangaraj, P.; Lima, B.D.S.; Chandran, R.; de Souza Araujo, A.A.; Narain, N.; Serafini, M.R.; Junior, L.J.Q. Effects of luteolin and quercetin 3-beta-d-glucoside identified from passiflora subpeltata leaves against acetaminophen induced hepatotoxicity in rats. Biomed. Pharmacother. 2016, 83, 1278–1285. [Google Scholar] [CrossRef] [PubMed]
- Biswas, S.K. Does the interdependence between oxidative stress and inflammation explain the antioxidant paradox? Oxid. Med. Cell. Longev. 2016, 2016, 9. [Google Scholar] [CrossRef] [PubMed]
- Posadas, I.; Santos, P.; Cena, V. Acetaminophen induces human neuroblastoma cell death through nfkb activation. PLoS ONE 2012, 7, e50160. [Google Scholar] [CrossRef] [PubMed]
- Yoshioka, H.; Usuda, H.; Fujii, H.; Nonogaki, T. Sasa veitchii extracts suppress acetaminophen-induced hepatotoxicity in mice. Environ. Health Prev. Med. 2017, 22, 54. [Google Scholar] [CrossRef] [PubMed]
- Akinboro, A.; Mohamed, K.B.; Asmawi, M.Z.; Sulaiman, S.F.; Sofiman, O.A. Antioxidants in aqueous extract of myristica fragrans (houtt.) suppress mitosis and cyclophosphamide-induced chromosomal aberrations in allium cepa l. Cells. J. Zhejiang Univ. Sci. B 2011, 12, 915–922. [Google Scholar] [CrossRef] [PubMed]
- Calliste, C.A.; Kozlowski, D.; Duroux, J.L.; Champavier, Y.; Chulia, A.J.; Trouillas, P. A new antioxidant from wild nutmeg. Food Chem. 2010, 118, 489–496. [Google Scholar] [CrossRef]
- Kapoor, I.P.S.; Singh, B.; Singh, G.; De Heluani, C.S.; De Lampasona, M.P.; Catalan, C.A.N. Chemical composition and antioxidant activity of essential oil and oleoresins of nutmeg (myristica fragrans houtt.) fruits. Int. J. Food Prop. 2013, 16, 1059–1070. [Google Scholar] [CrossRef]
- Erukainure, O.L.; Ajiboye, J.A.; Abbah, U.A.; Asieba, G.O.; Mamuru, S.; Zaruwa, M.Z.; Manhas, N.; Singh, P.; Islam, M.S. Monodora myristica (African nutmeg) modulates redox homeostasis and alters functional chemistry in sickled erythrocytes. Hum. Exp. Toxicol. 2018, 37, 458–467. [Google Scholar] [CrossRef] [PubMed]
- Tisserand, R.; Young, R. 14—Constituent profiles. In Essential Oil Safety, 2nd ed.; Tisserand, R., Young, R., Eds.; Churchill Livingstone: St. Louis, MO, USA, 2014; pp. 483–647. [Google Scholar]
- Jeong, H.G.; Yun, C.H. Induction of rat hepatic cytochrome p450 enzymes by myristicin. Biochem. Biophys. Res. Commun. 1995, 217, 966–971. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.Y.; Park, W. Anti-inflammatory effect of myristicin on raw 264.7 macrophages stimulated with polyinosinic-polycytidylic acid. Molecules 2011, 16, 7132–7142. [Google Scholar] [CrossRef] [PubMed]
- Serhan, C.N.; Chiang, N.; Van Dyke, T.E. Resolving inflammation: Dual anti-inflammatory and pro-resolution lipid mediators. Nat. Rev. Immunol. 2008, 8, 349–361. [Google Scholar] [CrossRef] [PubMed]
- Acuña, U.M.; Blanco Carcache, P.J.; Matthew, S.; Carcache de Blanco, E.J. New acyclic bis phenylpropanoid and neolignans, from myristica fragrans houtt., exhibiting parp-1 and nf-κb inhibitory effects. Food Chem. 2016, 202, 269–275. [Google Scholar] [CrossRef] [PubMed]
- Watelet, J.; Laurent, V.; Bressenot, A.; Bronowicki, J.P.; Larrey, D.; Peyrin-Biroulet, L. Toxicity of chronic paracetamol ingestion. Aliment. Pharmacol. Ther. 2007, 26, 1543–1544. [Google Scholar] [CrossRef] [PubMed]
- Bai, Q.; Yan, H.; Sheng, Y.; Jin, Y.; Shi, L.; Ji, L.; Wang, Z. Long-term acetaminophen treatment induced liver fibrosis in mice and the involvement of egr-1. Toxicology 2017, 382, 47–58. [Google Scholar] [CrossRef] [PubMed]
- Almeer, R.S.; El-Khadragy, M.F.; Abdelhabib, S.; Abdel Moneim, A.E. Ziziphus spina-christi leaf extract ameliorates schistosomiasis liver granuloma, fibrosis, and oxidative stress through downregulation of fibrinogenic signaling in mice. PLoS ONE 2018, 13, e0204923. [Google Scholar] [CrossRef] [PubMed]
- Shen, K.; Chang, W.; Gao, X.; Wang, H.; Niu, W.; Song, L.; Qin, X. Depletion of activated hepatic stellate cell correlates with severe liver damage and abnormal liver regeneration in acetaminophen-induced liver injury. Acta Biochim. Biophys. Sin. 2011, 43, 307–315. [Google Scholar] [CrossRef] [PubMed]
- Park, E.Y.; Shin, S.M.; Ma, C.J.; Kim, Y.C.; Kim, S.G. Meso-dihydroguaiaretic acid from machilus thunbergii down-regulates tgf-beta1 gene expression in activated hepatic stellate cells via inhibition of ap-1 activity. Planta Med. 2005, 71, 393–398. [Google Scholar] [CrossRef] [PubMed]
- Chan, K.; Han, X.D.; Kan, Y.W. An important function of nrf2 in combating oxidative stress: Detoxification of acetaminophen. Proc. Natl. Acad. Sci. USA 2001, 98, 4611–4616. [Google Scholar] [CrossRef] [PubMed]
- Song, J.S.; Kim, E.K.; Choi, Y.W.; Oh, W.K.; Kim, Y.M. Hepatocyte-protective effect of nectandrin b, a nutmeg lignan, against oxidative stress: Role of nrf2 activation through erk phosphorylation and ampk-dependent inhibition of gsk-3beta. Toxicol. Appl. Pharmacol. 2016, 307, 138–149. [Google Scholar] [CrossRef] [PubMed]
- Kim, E.K.; Song, J.S.; Choi, D.G.; Choi, Y.W.; Oh, W.K.; Kim, Y.-M. Therapeutic potential of nectandrin b, a nutmeg lignan, in nonalcoholic fatty liver disease: Anti-lipogenic and hepatocyte-protective effects through amp-activated protein kinase and nrf2 activation. FASEB J. 2017, 31. [Google Scholar] [CrossRef]
- Roubalová, L.; Dinkova-Kostova, A.T.; Biedermann, D.; Křen, V.; Ulrichová, J.; Vrba, J. Flavonolignan 2,3-dehydrosilydianin activates nrf2 and upregulates nad(p)h:Quinone oxidoreductase 1 in hepa1c1c7 cells. Fitoterapia 2017, 119, 115–120. [Google Scholar] [CrossRef] [PubMed]
- Johnson, J.A.; Johnson, D.A.; Kraft, A.D.; Calkins, M.J.; Jakel, R.J.; Vargas, M.R.; Chen, P.C. The nrf2-are pathway: An indicator and modulator of oxidative stress in neurodegeneration. Ann. N. Y. Acad. Sci. 2008, 1147, 61–69. [Google Scholar] [CrossRef] [PubMed]
- Lv, X.; Xia, Y.; Finel, M.; Wu, J.; Ge, G.; Yang, L. Recent progress and challenges in screening and characterization of ugt1a1 inhibitors. Acta Pharm. Sin. B 2018. [Google Scholar] [CrossRef]
- Kostrubsky, S.E.; Sinclair, J.F.; Strom, S.C.; Wood, S.; Urda, E.; Stolz, D.B.; Wen, Y.H.; Kulkarni, S.; Mutlib, A. Phenobarbital and phenytoin increased acetaminophen hepatotoxicity due to inhibition of udp-glucuronosyltransferases in cultured human hepatocytes. Toxicol. Sci. 2005, 87, 146–155. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Fujino, M.; Takahara, T.; Li, X.K. Protective role of heme oxygenase-1 in fatty liver ischemia-reperfusion injury. Med. Mol. Morphol. 2018. [Google Scholar] [CrossRef] [PubMed]
- Bauer, M.; Bauer, I. Heme oxygenase-1: Redox regulation and role in the hepatic response to oxidative stress. Antioxid. Redox Signal. 2002, 4, 749–758. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.H.; Chau, L.Y.; Chen, J.W.; Lin, S.J. Serum bilirubin and ferritin levels link heme oxygenase-1 gene promoter polymorphism and susceptibility to coronary artery disease in diabetic patients. Diabetes Care 2008, 31, 1615–1620. [Google Scholar] [CrossRef] [PubMed]
- Bauer, I.; Vollmar, B.; Jaeschke, H.; Rensing, H.; Kraemer, T.; Larsen, R.; Bauer, M. Transcriptional activation of heme oxygenase-1 and its functional significance in acetaminophen-induced hepatitis and hepatocellular injury in the rat. J. Hepatol. 2000, 33, 395–406. [Google Scholar] [CrossRef]
- Gao, Y.; Cao, Z.; Yang, X.; Abdelmegeed, M.A.; Sun, J.; Chen, S.; Beger, R.D.; Davis, K.; Salminen, W.F.; Song, B.J.; et al. Proteomic analysis of acetaminophen-induced hepatotoxicity and identification of heme oxygenase 1 as a potential plasma biomarker of liver injury. Proteom. Clin. Appl. 2017, 11, 1600123. [Google Scholar] [CrossRef] [PubMed]
- Sharma, S.; Singh, R.L.; Kakkar, P. Modulation of bax/bcl-2 and caspases by probiotics during acetaminophen induced apoptosis in primary hepatocytes. Food Chem. Toxicol. 2011, 49, 770–779. [Google Scholar] [CrossRef] [PubMed]
- Hu, J.; Yan, D.; Gao, J.; Xu, C.; Yuan, Y.; Zhu, R.; Xiang, D.; Weng, S.; Han, W.; Zang, G.; et al. Rhil-1ra reduces hepatocellular apoptosis in mice with acetaminophen-induced acute liver failure. Lab. Investig. 2010, 90, 1737–1746. [Google Scholar] [CrossRef] [PubMed]
- Li, G.; Chen, J.B.; Wang, C.; Xu, Z.; Nie, H.; Qin, X.Y.; Chen, X.M.; Gong, Q. Curcumin protects against acetaminophen-induced apoptosis in hepatic injury. World J. Gastroenterol. 2013, 19, 7440–7446. [Google Scholar] [CrossRef] [PubMed]
- Ramachandran, A.; Jaeschke, H. Mechanisms of acetaminophen hepatotoxicity and their translation to the human pathophysiology. J. Clin. Transl. Res. 2017, 3, 157–169. [Google Scholar] [CrossRef] [PubMed]
- Vaux, D.L. Apoptogenic factors released from mitochondria. Biochim. Biophys. Acta-Mol. Cell Res. 2011, 1813, 546–550. [Google Scholar] [CrossRef] [PubMed]
- Safa, A.R. C-flip, a master anti-apoptotic regulator. Exp. Oncol. 2012, 34, 176–184. [Google Scholar] [PubMed]
- Zhao, Q.; Liu, C.; Shen, X.; Xiao, L.; Wang, H.; Liu, P.; Wang, L.; Xu, H. Cytoprotective effects of myristicin against hypoxiainduced apoptosis and endoplasmic reticulum stress in rat dorsal root ganglion neurons. Mol. Med. Rep. 2017, 15, 2280–2288. [Google Scholar] [CrossRef] [PubMed]
- Abdel Moneim, A.E. The neuroprotective effects of purslane (portulaca oleracea) on rotenone-induced biochemical changes and apoptosis in brain of rat. CNS Neurol. Disord. Drug Targets 2013, 12, 830–841. [Google Scholar] [CrossRef] [PubMed]
- Abdel Moneim, A.E. Indigofera oblongifolia prevents lead acetate-induced hepatotoxicity, oxidative stress, fibrosis and apoptosis in rats. PLoS ONE 2016, 11, e0158965. [Google Scholar] [CrossRef] [PubMed]
- Sehitoglu, M.H.; Yayla, M.; Kiraz, A.; Oztopuz, R.O.; Bayir, Y.; Karaca, T.; Khalid, S.; Akpinar, E. The effects of apomorphine on paracetamol-induced hepatotoxicity in rats. Cell. Mol. Biol. 2017, 63, 40–44. [Google Scholar] [CrossRef] [PubMed]
- El-Maddawy, Z.K.; El-Sayed, Y.S. Comparative analysis of the protective effects of curcumin and n-acetyl cysteine against paracetamol-induced hepatic, renal, and testicular toxicity in wistar rats. Environ. Sci. Pollut. Res. 2018, 25, 3468–3479. [Google Scholar] [CrossRef] [PubMed]
- Reitman, S.; Frankel, S. A colorimetric method for the determination of serum glutamic oxalacetic and glutamic pyruvic transaminases. Am. J. Clin. Pathol. 1957, 28, 56–63. [Google Scholar] [CrossRef] [PubMed]
- Shephard, M.D.; Peake, M.J. Quantitative method for determining serum alkaline phosphatase isoenzyme activity i. Guanidine hydrochloride: New reagent for selectively inhibiting major serum isoenzymes of alkaline phosphatase. J. Clin. Pathol. 1986, 39, 1025–1030. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, M.; Eisenburg, J. Serum bilirubin determination in newborn infants. A new micromethod for the determination of serum of plasma bilirubin in newborn infants. Fortschr. Med. 1975, 93, 1461–1466. [Google Scholar] [PubMed]
- Ohkawa, H.; Ohishi, N.; Yagi, K. Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal. Biochem. 1979, 95, 351–358. [Google Scholar] [CrossRef]
- Green, L.C.; Wagner, D.A.; Glogowski, J.; Skipper, P.L.; Wishnok, J.S.; Tannenbaum, S.R. Analysis of nitrate, nitrite, and [15n]nitrate in biological fluids. Anal. Biochem. 1982, 126, 131–138. [Google Scholar] [CrossRef]
- Ellman, G.L. Tissue sulfhydryl groups. Arch. Biochem. Biophys. 1959, 82, 70–77. [Google Scholar] [CrossRef]
- Sun, Y.; Oberley, L.W.; Li, Y. A simple method for clinical assay of superoxide dismutase. Clin. Chem. 1988, 34, 497–500. [Google Scholar] [PubMed]
- Luck, H. Catalase. In Methods of Enzymatic Analysis; Bergmeyer, H.U., Ed.; Academic Press: New York, NY, USA, 1965; pp. 855–888. [Google Scholar]
- Paglia, D.E.; Valentine, W.N. Studies on the quantitative and qualitative characterization of erythrocyte glutathione peroxidase. J. Lab. Clin. Med. 1967, 70, 158–169. [Google Scholar] [PubMed]
- Factor, V.M.; Kiss, A.; Woitach, J.T.; Wirth, P.J.; Thorgeirsson, S.S. Disruption of redox homeostasis in the transforming growth factor-alpha/c-myc transgenic mouse model of accelerated hepatocarcinogenesis. J. Biol. Chem. 1998, 273, 15846–15853. [Google Scholar] [CrossRef] [PubMed]
- Almeer, R.S.; Abdel Moneim, A.E. Evaluation of the protective effect of olive leaf extract on cisplatin-induced testicular damage in rats. Oxid. Med. Cell. Longev. 2018, 2018, 11. [Google Scholar] [CrossRef] [PubMed]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dkhil, M.A.; Abdel Moneim, A.E.; Hafez, T.A.; Mubaraki, M.A.; Mohamed, W.F.; Thagfan, F.A.; Al-Quraishy, S. Myristica fragrans Kernels Prevent Paracetamol-Induced Hepatotoxicity by Inducing Anti-Apoptotic Genes and Nrf2/HO-1 Pathway. Int. J. Mol. Sci. 2019, 20, 993. https://doi.org/10.3390/ijms20040993
Dkhil MA, Abdel Moneim AE, Hafez TA, Mubaraki MA, Mohamed WF, Thagfan FA, Al-Quraishy S. Myristica fragrans Kernels Prevent Paracetamol-Induced Hepatotoxicity by Inducing Anti-Apoptotic Genes and Nrf2/HO-1 Pathway. International Journal of Molecular Sciences. 2019; 20(4):993. https://doi.org/10.3390/ijms20040993
Chicago/Turabian StyleDkhil, Mohamed A., Ahmed E. Abdel Moneim, Taghreed A. Hafez, Murad A. Mubaraki, Walid F. Mohamed, Felwa A. Thagfan, and Saleh Al-Quraishy. 2019. "Myristica fragrans Kernels Prevent Paracetamol-Induced Hepatotoxicity by Inducing Anti-Apoptotic Genes and Nrf2/HO-1 Pathway" International Journal of Molecular Sciences 20, no. 4: 993. https://doi.org/10.3390/ijms20040993
APA StyleDkhil, M. A., Abdel Moneim, A. E., Hafez, T. A., Mubaraki, M. A., Mohamed, W. F., Thagfan, F. A., & Al-Quraishy, S. (2019). Myristica fragrans Kernels Prevent Paracetamol-Induced Hepatotoxicity by Inducing Anti-Apoptotic Genes and Nrf2/HO-1 Pathway. International Journal of Molecular Sciences, 20(4), 993. https://doi.org/10.3390/ijms20040993