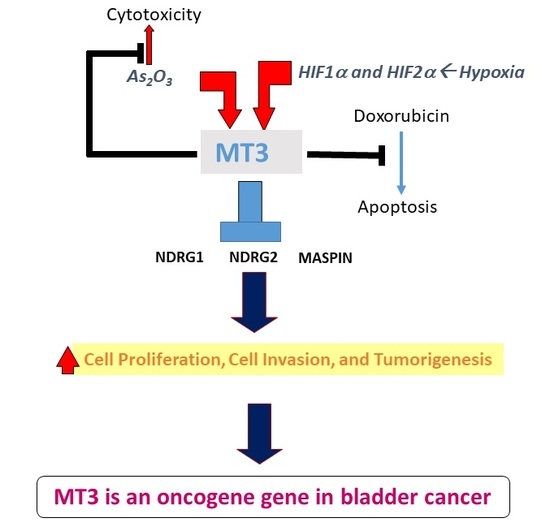
Metallothionein 3 Is a Hypoxia-Upregulated Oncogene Enhancing Cell Invasion and Tumorigenesis in Human Bladder Carcinoma Cells
Abstract
1. Introduction
2. Results
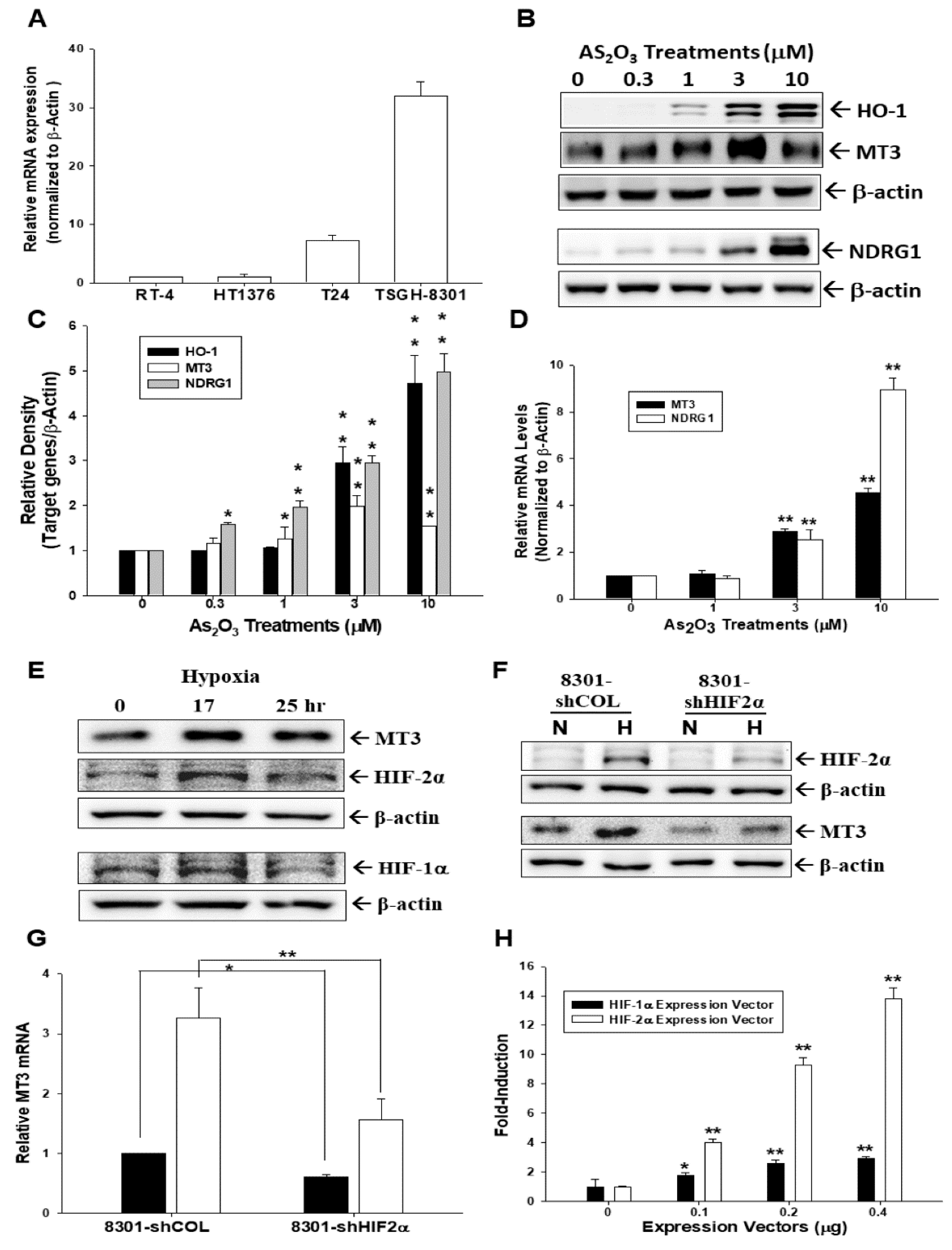
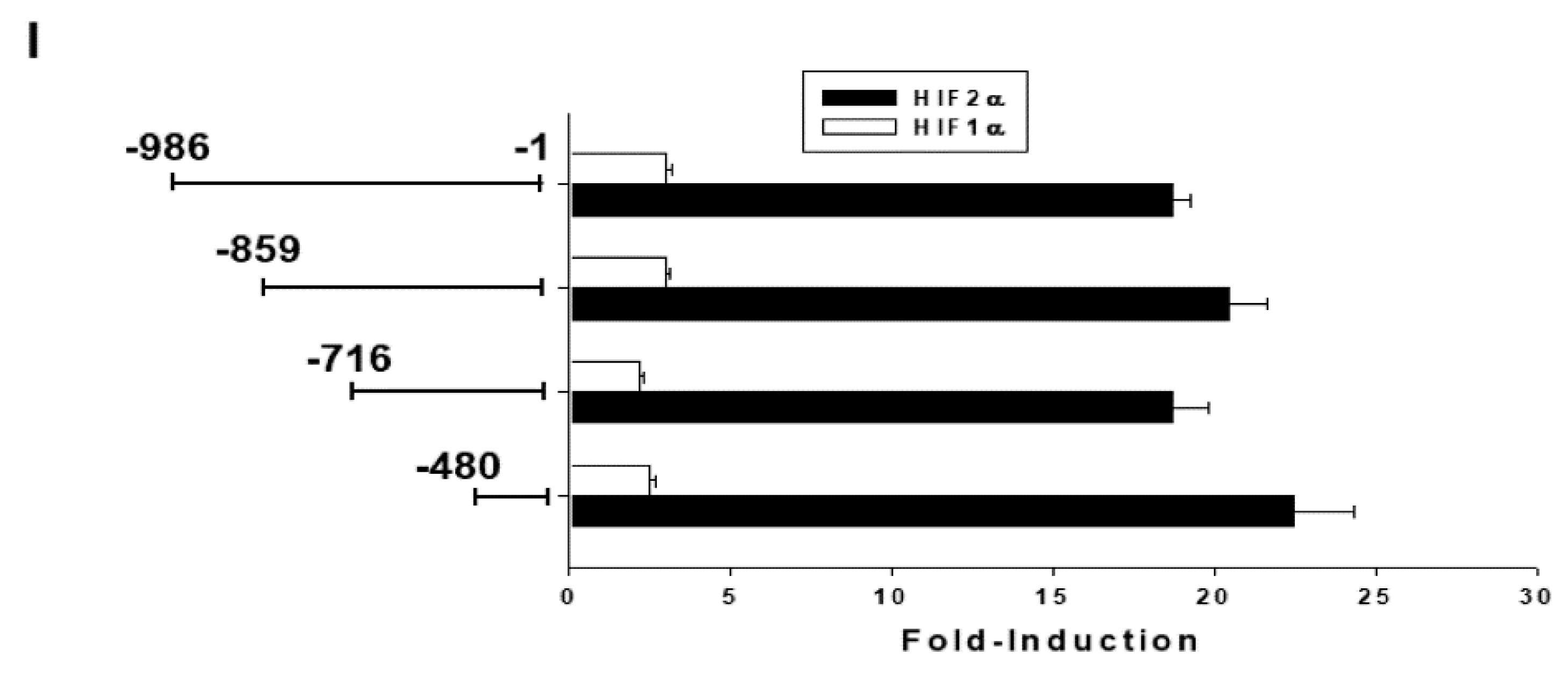
2.1. Arsenic and Hypoxia Upregulate Metallothionein 3 (MT3) Expression in Bladder Carcinoma Cells
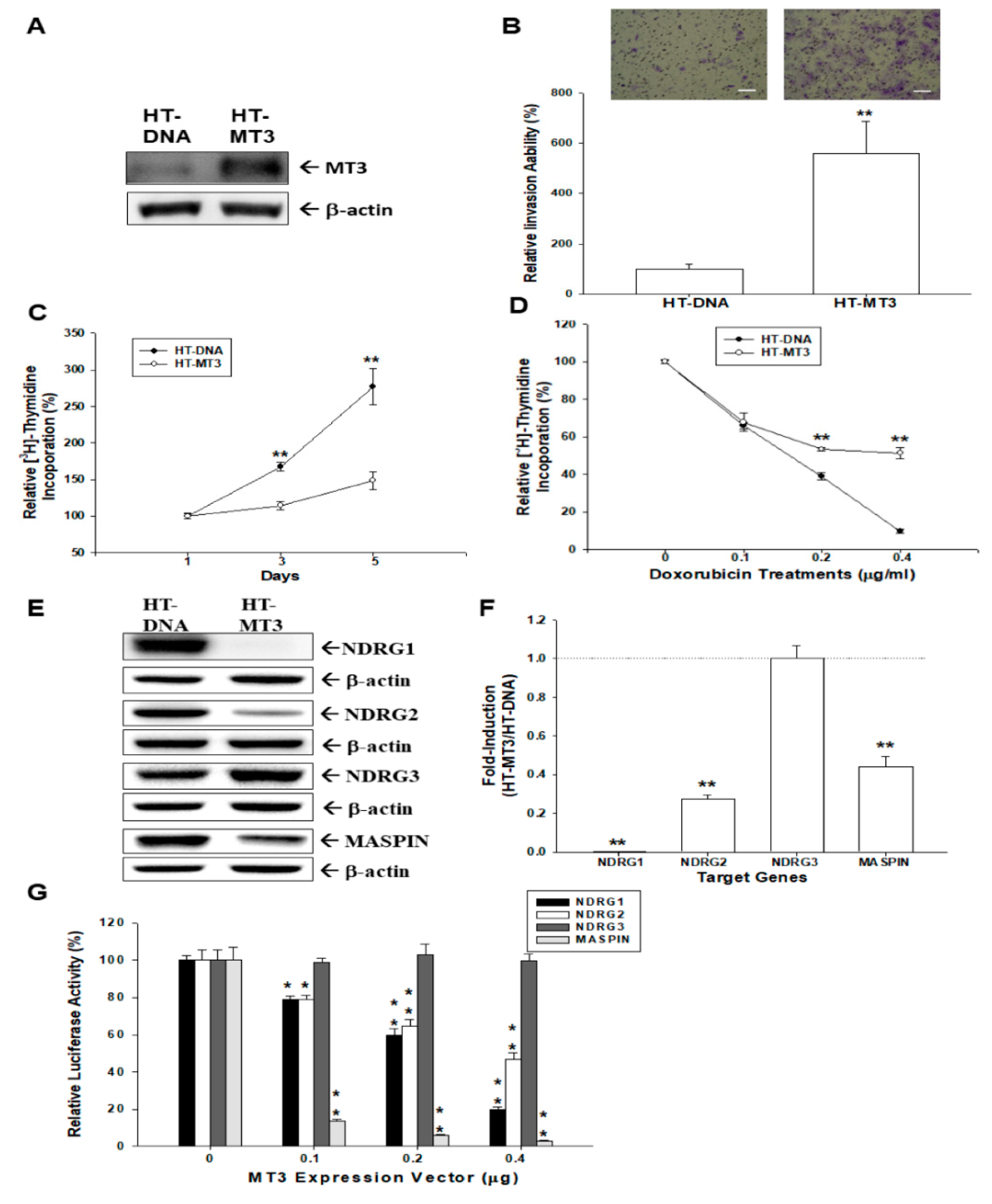
2.2. Effects of Ectopic Overexpression of MT3 on Proliferation and Invasion of Bladder Carcinoma HT1376 Cells
2.3. Ectopic Overexpression MT3 Modulates N-myc Downstream Regulated Gene 1 (NDRG1), NDRG2, NDRG3, and MASPIN Gene Expressions in Bladder Carcinoma HT1376 Cells
2.4. Effect of MT3-Knockdown on Proliferation and Invasion of Bladder Carcinoma TSGH-8301 Cells
2.5. Effect of Overexpression of MT3 on Proliferation and Invasion of Bladder Carcinoma T24 Cells
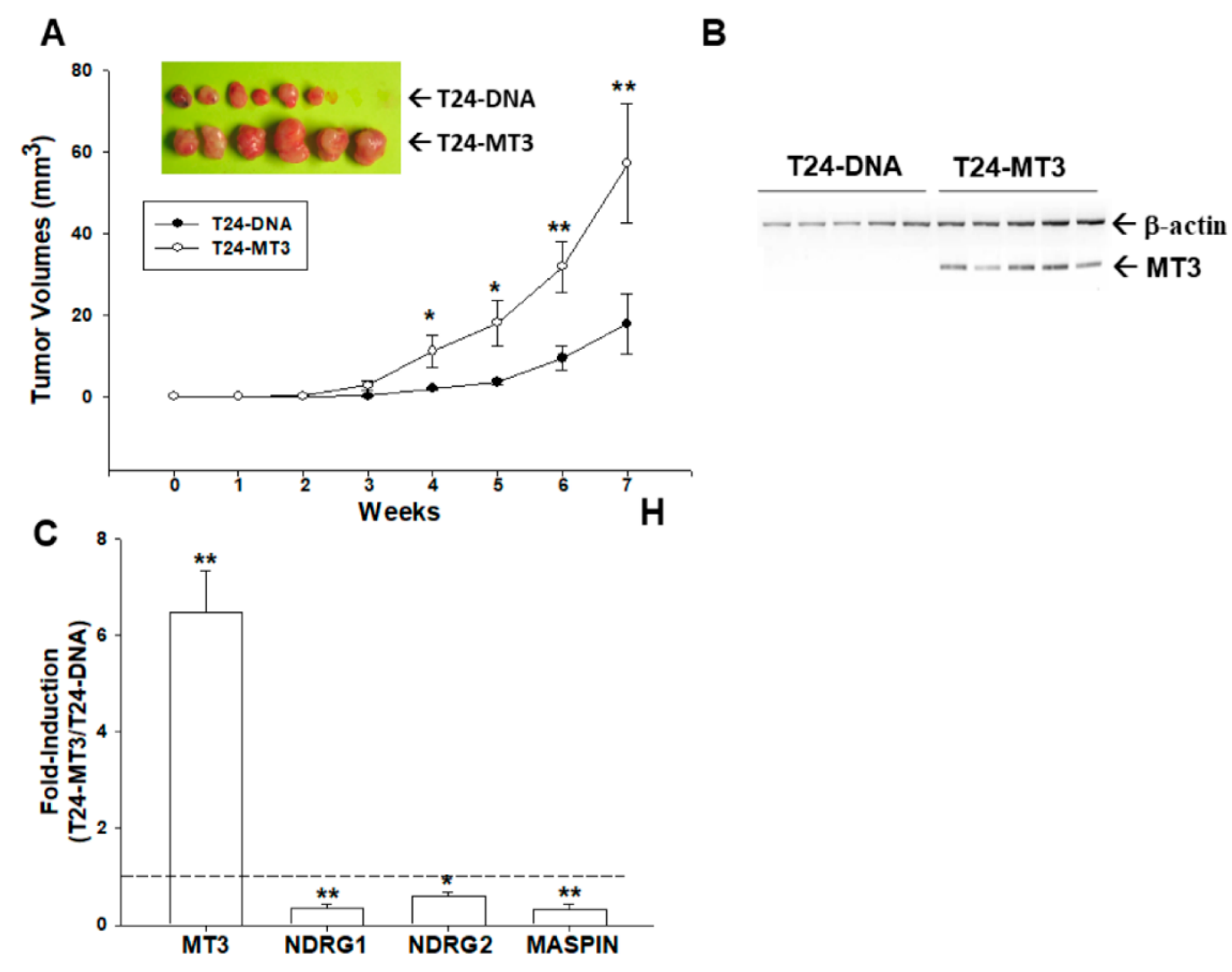
2.6. Overexpression of MT3 Downregulates NDRG1, NDRG2, and MASPIN Gene Expressions in Bladder Carcinoma T24 Cells
2.7. Effects of Ectopic Overexpression of MT3 on Tumorigenesis of Bladder Carcinoma T24 cells
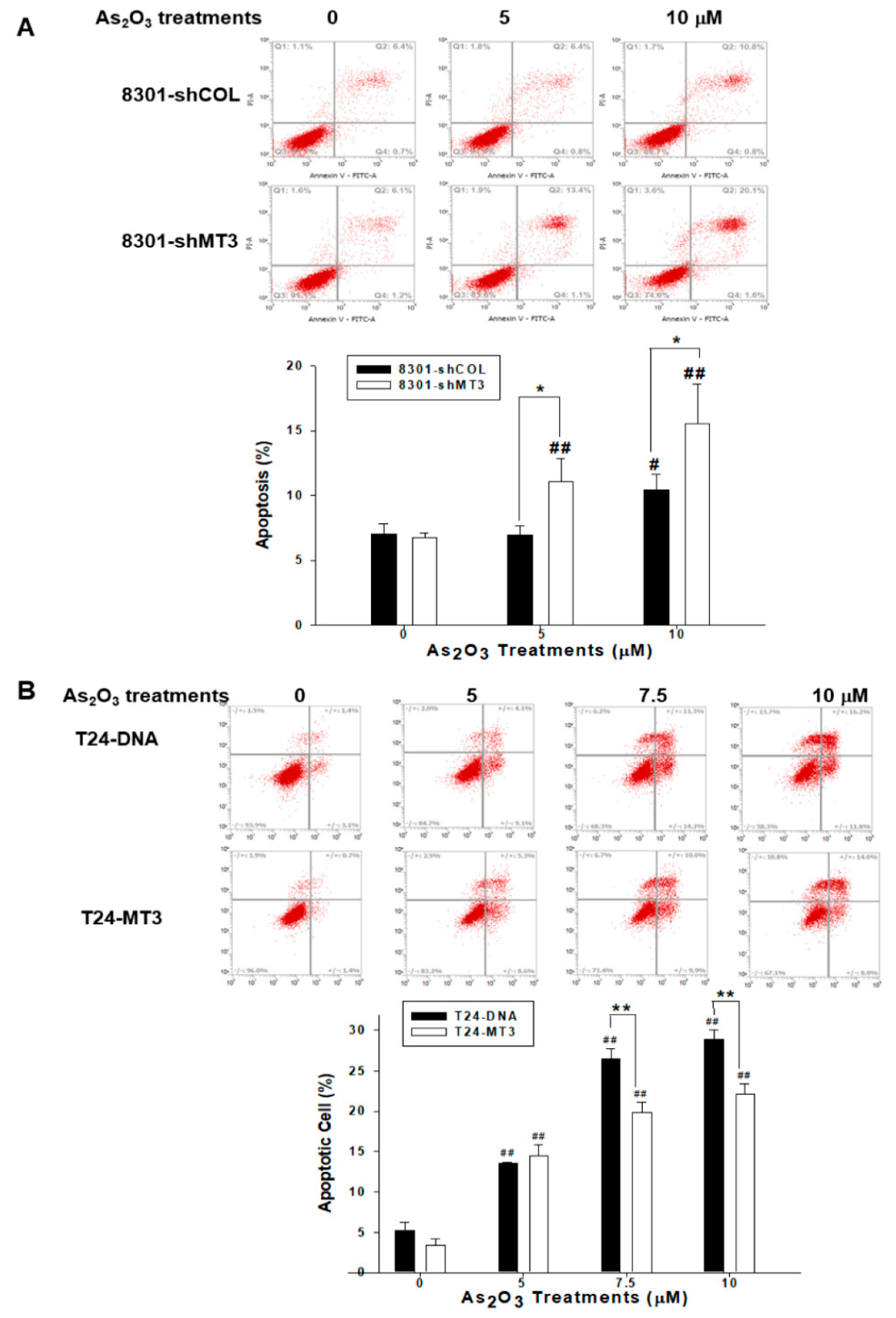
2.8. MT3 Modulates Cell Apoptosis Induced by As2O3 in Bladder Carcinoma Cells
3. Discussion
4. Materials and Methods
4.1. Cell Cultures and Chemicals
4.2. Knock-down MT3 and HIF-2α
4.3. Expression Vectors and Stable Transfection
4.4. Immunoblot Assays
4.5. Real-Time Reverse Transcriptase-Polymerase Chain Reaction (RT-qPCR)
4.6. [3H]thymidine Incorporation Assay
4.7. Matrigel Invasion Assay
4.8. Annexin V-FITC Apoptosis Detection
4.9. Reporter Vector Constructs and Reporter Assay
4.10. Tumor Xenograft Study
4.11. Statistical Analysis
5. Conclusion
Supplementary Materials
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Antoni, S.; Ferlay, J.; Soerjomataram, I.; Znaor, A.; Jemal, A.; Bray, F. Bladder cancer incidence and mortality: A global overview and recent trends. Eur. Urol. 2017, 71, 96–108. [Google Scholar] [CrossRef] [PubMed]
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer statistics, 2017. CA Cancer J. Clin. 2017, 67, 7–30. [Google Scholar] [CrossRef] [PubMed]
- Goodison, S.; Rosser, C.J.; Urquidi, V. Bladder cancer detection and monitoring: Assessment of urine- and blood based marker tests. Mol. Diag. Ther. 2013, 17, 71–84. [Google Scholar] [CrossRef] [PubMed]
- Narayan, V.M.; Adejoro, O.; Schwartz, I.; Ziegelmann, M.; Elliott, S.; Konety, B.R. The prevalence and impact of urinary marker testing in patients with bladder cancer. J. Urol. 2018, 199, 74–80. [Google Scholar] [CrossRef] [PubMed]
- Si, M.; Lang, J. The role of metallothioneins in carcinogenesis. J. Hematol. Oncol. 2018, 11, 107. [Google Scholar] [CrossRef] [PubMed]
- Cherian, M.G.; Jayasurya, A.; Bay, B.H. Metallothioneins in human tumors and potential roles in carcinogenesis. Mutat. Res. 2003, 533, 201–209. [Google Scholar] [CrossRef] [PubMed]
- Gumulec, J.; Raudenska, M.; Adam, V.; Kizek, R.; Masarik, M. Metallothionein—Immunohistochemical cancer biomarker: A meta-analysis. PLoS ONE 2014, 9, e85346. [Google Scholar] [CrossRef] [PubMed]
- Moleirinho, A.; Carneiro, J.; Matthiesen, R.; Silva, R.M.; Amorim, A.; Azevedo, L. Grains, losses and changes of function after gene duplication: Study of the metallothionein family. PLoS ONE 2011, 6, e18487. [Google Scholar] [CrossRef] [PubMed]
- Nordberg, M. Trace elements and metallothionein related to geo-environment. J. Trace Elem. Exp. Med. 2000, 13, 97–104. [Google Scholar] [CrossRef]
- Wang, G.; Fowler, B.A. Roles of biomarkers in evaluating interactions among mixtures of lead, cadmium and arsenic. Toxicol. Appl. Pharmacol. 2008, 233, 92–99. [Google Scholar] [CrossRef] [PubMed]
- Kojima, I.; Tanaka, T.; Inagi, R.; Nishi, H.; Aburatani, H.; Kato, H.; Miyata, T.; Fujita, T.; Nangaku, M. Metallothionein is upregulated by hypoxia and stabilizes hypoxia-inducible factor in the kidney. Kidney Int. 2009, 75, 268–277. [Google Scholar] [CrossRef] [PubMed]
- Hozumi, I.; Suzuki, J.S.; Kanazawa, H.; Hara, A.; Saio, M.; Inuzuka, T.; Miyairi, S.; Naganuma, A.; Tohyama, C. Metallothionein-3 is expressed in the brain and various peripheral organs of the rat. Neurosci. Lett. 2008, 438, 54–58. [Google Scholar] [CrossRef] [PubMed]
- Vašák, M.; Meloni, G. Mammalian metallothionein-3: New functional and structural insights. Int. J. Mol. Sci. 2017, 18, 1117. [Google Scholar] [CrossRef] [PubMed]
- Sens, M.A.; Somji, S.; Lamm, D.L.; Garrett, S.H.; Slovinsky, F.; Todd, J.H.; Sens, D.A. Metallothionein isoform 3 as a potential biomarker for human bladder cancer. Environ. Health Persp. 2000, 108, 413. [Google Scholar] [CrossRef] [PubMed]
- Juang, H.H.; Chung, L.C.; Feng, T.H.; Lee, Y.H.; Chang, P.L.; Tsui, K.H. Metallothionein 3: An androgen-upregulated gene enhances cell invasion and tumorigenesis of prostate carcinoma cells. Prostate 2013, 73, 1495–1506. [Google Scholar] [CrossRef] [PubMed]
- Wei, H.; Desouki, M.M.; Lin, S.; Xiao, D.; Franklin, R.B.; Feng, P. Differential expression of metallothioneins (MTs) 1, 2, and 3 in response to zinc treatment in human prostate normal and malignant cells and tissues. Mol. Cancer 2008, 7, 7. [Google Scholar] [CrossRef] [PubMed]
- Davis, A.P.; Murphyu, C.G.; Rosenstein, M.C.; Wiegers, T.C.; Mattingly, C.J. The comparative toxicogenomics database facilitates identification and understanding of chemical-gene-disease associations: Arsenic as a case study. BMC Med. Genomics 2008, 1, 48. [Google Scholar] [CrossRef] [PubMed]
- Somji, S.; Garrett, S.H.; Toni, C.; Zhou, X.D.; Zeng, Y.; Ajjimaporn, A.; Sens, M.A.; Sens, D.A. Differences in epigenetic regulation of MT-3 gene expression between parental and Cd+2 or As+3 transformed human urothelial cells. Cancer Cell Int. 2011, 11, 2. [Google Scholar] [CrossRef] [PubMed]
- Geleta, B.; Makonnen, E.; Abay, S.M. N-myc downstream regulated gene (NDRG): Role in cancer metastasis suppression and as drug target in cancer therapeutics. J. Cancer Sci. Ther. 2016, 8, 6. [Google Scholar] [CrossRef]
- Fang, B.A.; Kovačević, Ž.; Park, K.C.; Kalinowski, D.S.; Jansson, P.J.; Lane, D.J.; Sahni, S.; Richardson, D.R. Molecular functions of the iron-regulated metastasis suppressor, NDRG1, and its potential as a molecular target for cancer therapy. Biochim. Biophys. Acta 2014, 1845, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Kimura, T.; Kambe, T. The functions of metallothionein and ZIP and ZnT transporters: An overview and perspective. Int. J. Mol. Sci. 2016, 17, 336. [Google Scholar] [CrossRef] [PubMed]
- Krizkova, S.; Fabrik, I.; Adam, V.; Hrabeta, J.; Eckschlager, T.; Kizek, R. Metallothionein—A promising tool for cancer diagnostics. Bratisl. Lek. Listy. 2009, 110, 93–97. [Google Scholar] [PubMed]
- Saga, Y.; Hashimoto, H.; Yachiku, S.; Tokumitsu, M.; Kaneko, S. Immunohistochemical expression of metallothionein in human bladder cancer: Correlation with histopathological parameters and patient survival. J. Urol. 2002, 168, 2227–2231. [Google Scholar] [CrossRef]
- Yamasaki, Y.; Smith, C.; Weisz, D.; van Huizen, I.; Xuan, J.; Moussa, M.; Stitt, L.; Hideki, S.; Cherian, M.G.; Izawa, J.I. Metallothionein expression as prognostic factor for transitional cell carcinoma of bladder. Urology 2006, 67, 530–535. [Google Scholar] [CrossRef] [PubMed]
- Palmiter, R.D.; Findley, S.D.; Whitmore, T.E.; Durnam, D.M. MT-III, a brain-specific member of the metallothionein gene family. Proc. Nat. Acad. Sci. USA 1992, 89, 6333–6337. [Google Scholar] [CrossRef] [PubMed]
- Amoureux, M.C.; Wurch, T.; Pauwels, P.J. Modulation of metallothionein-III mRNA content and growth rate of rat C6-glial cells by transfection with human 5-HT1D receptor genes. Biochem. Biophys. Res. Commun. 1995, 214, 639–645. [Google Scholar] [CrossRef] [PubMed]
- Garrett, S.H.; Sens, M.A.; Shukla, D.; Nestor, S.; Somji, S.; Todd, J.H.; Sens, D.A. Metallothionein isoform 3 expression in the human prostate and cancer-derived cell lines. Prostate 1999, 41, 196–202. [Google Scholar] [CrossRef]
- Garrett, S.H.; Sens, M.A.; Todd, J.H.; Somji, S.; Sens, D.A. Expression of MT-3 protein in the human kidney. Toxicol. Lett. 1999, 105, 207–214. [Google Scholar] [CrossRef]
- Yeh, T.C.; Tai, Y.S.; Pu, Y.S.; Chen, C.H. Characteristics of arsenic-related bladder cancer: A study from Nationwide Cancer Registry Database in Taiwan. Urol. Sci. 2015, 26, 103–108. [Google Scholar] [CrossRef]
- Zhou, X.D.; Sens, M.A.; Garrett, S.H.; Somji, S.; Park, S.; Gurel, V.; Sens, D.A. Enhanced expression of metallothionein isoform 3 protein in tumor heterotransplants derived from As+3- and Cd+2-transformed human urothelial cells. Toxicol. Sci. 2006, 93, 322–330. [Google Scholar] [CrossRef] [PubMed]
- Gunther, V.; Lindert, U.; Shaffner, W. The taste of heavy metals: Gene regulation by MTF-1. Biochim. Biophys. Acta 2012, 1823, 1416–1425. [Google Scholar] [CrossRef] [PubMed]
- Ruiz-Ramos, R.; Lopez-Carrillo, L.; Rios-Perez, A.D.; De Vizcaya-Ruíz, A.; Cebrian, M.E. Sodium arsenite induces ROS generation, DNA oxidative damage, HO-1 and c-Myc proteins, NF-kappaB activation and cell proliferation in human breast cancer MCF-7 cells. Mutat. Res. 2009, 674, 109–115. [Google Scholar] [CrossRef] [PubMed]
- Murphy, B.J.; Kimura, T.; Sato, B.G.; Shi, Y.; Andrews, G.K. Metallothionein induction by hypoxia involves cooperative interactions between metal-responsive transcription factor-1 and hypoxia-inducible transcription factor-1alpha. Mol. Cancer Res. 2008, 6, 483–490. [Google Scholar] [CrossRef] [PubMed]
- Won, Y.; Shin, Y.; Chun, C.H.; Cho, Y.; Ha, C.W.; Kim, J.H.; Chun, J.S. Pleiotropic roles of metallothioneins as regulators of chondrocyte apoptosis and catabolic and anabolic pathways during osteoarthritis pathogenesis. Ann. Rheum. Dis. 2016, 75, 2045–2052. [Google Scholar] [CrossRef] [PubMed]
- Yamasaki, M.; Nomura, T.; Sato, F.; Mimata, H. Methallothionein is up-regulated under hypoxia and promotes the survival of human prostate cancer cells. Oncol. Rep. 2007, 18, 1145–1154. [Google Scholar] [PubMed]
- Ioachim, E.; Michael, M.; Salmas, M.; Michael, M.M.; Stavropoulos, N.E.; Malamou-Mitsi, V. Hypoxia-inducible factors HIF-1α and HIF-2α expression in bladder cancer and their associations with other angiogenesis-related proteins. Urol. Int. 2006, 77, 255–263. [Google Scholar] [CrossRef] [PubMed]
- Dutta, R.; Sens, D.A.; Somji, S.; Sens, M.A.; Garrett, S.H. Metallothionein isoform 3 inhibits cell growth and increases drug resistance of PC-3 prostate cancer cells. Prostate 2002, 52, 89–97. [Google Scholar] [CrossRef] [PubMed]
- Rahman, M.T.; De Ley, M. Arsenic induction of metallothionen and metallothionein induction against arsenic cytotoxicity. Rev. Environ. Contam. Toxicol. 2017, 240, 151–168. [Google Scholar] [PubMed]
- Kurdistani, S.K.; Arizti, P.; Reimer, C.L.; Sugrue, M.M.; Aaronson, S.A.; Lee, S.W. Inhibition of tumor cell growth by RTP/rit42 and its responsiveness to p53 and DNA damage. Cancer Res. 1998, 58, 4439–4444. [Google Scholar] [PubMed]
- Li, R.; Yu, C.; Jiang, F.; Gao, L.; Li, J.; Wang, Y.; Beckwith, N.; Yao, L.; Zhang, J.; Wu, G. Overexpression of N-Myc downstream-regulated gene 2 (NDRG2) regulates the proliferation and invasion of bladder cancer cells in vitro and in vivo. PLoS ONE 2013, 8, e76689. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Qi, F.; Cao, Y.; Zu, X.; Chen, M.; Li, Z.; Qi, L. 5-Aza-2’-deoxycytidine enhances maspin expression and inhibits proliferation, migration, and invasion of the bladder cancer T24 cell line. Cancer Biother. Radiopharm. 2013, 28, 343–350. [Google Scholar] [CrossRef] [PubMed]
- Tao, Y.F.; Xu, L.X.; Lu, J.; Cao, L.; Li, Z.H.; Hu, S.Y.; Wang, N.N.; Du, X.J.; Sun, L.C.; Zao, W.L.; et al. Metallothionein III (MT3) is putative tumor suppressor gene that is frequently inactivated in pediatric acute myeloid leukemia by promoter hypermethylation. J. Transl. Med. 2014, 12, 182. [Google Scholar] [CrossRef] [PubMed]
- Tsui, K.H.; Lin, Y.H.; Chung, L.C.; Chuang, S.T.; Feng, T.H.; Chiang, K.C.; Chang, P.L.; Yen, C.L.; Juang, H.H. Prostate-derived ets factor represses tumorigenesis and modulates epithelial-to-mesenchymal transition in bladder carcinoma cells. Cancer Lett. 2016, 375, 142–151. [Google Scholar] [CrossRef] [PubMed]
- Rigby, C.C.; Franks, L. A human tissue culture cell line from a transitional cell tumour of the urinary bladder: Growth, chromosome pattern and ultrastructure. Br. J. Cancer 1970, 24, 746–754. [Google Scholar] [CrossRef] [PubMed]
- Rasheed, S.; Gardner, M.B.; Rongey, R.W.; Nelson-Rees, W.A.; Arnstein, P. Human bladder carcinoma: Characterization of two new tumor cell lines and search for tumor viruses. J. Natl. Cancer Inst. 1977, 58, 881–890. [Google Scholar] [CrossRef] [PubMed]
- Yeh, M.Y.; Yu, D.S.; Chen, S.C.; Lin, M.S.; Chang, S.Y.; Ma, C.P.; Han, S.H. Establishment and characterization of a human urinary bladder carcinoma cell line (TSGH-8301). J. Surg. Oncol. 1988, 37, 177–184. [Google Scholar] [CrossRef] [PubMed]
- Bubenik, J.; Barešová, M.; Viklický, V.; Jakoubkova, J.; Sainerova, H.; Donner, J. Established cell line of urinary bladder carcinoma (T24) containing tumour-specific antigen. Int. J. Cancer. 1973, 11, 765–773. [Google Scholar] [CrossRef] [PubMed]
- Tsui, K.H.; Chang, P.L.; Juang, H.H. Zinc blocks gene expression of mitochondrial aconitase in human prostatic carcinoma cells. Int. J. Cancer 2006, 118, 609–615. [Google Scholar] [CrossRef] [PubMed]
- Tsui, K.H.; Hsu, S.Y.; Chung, L.C.; Lin, Y.H.; Feng, T.H.; Lee, T.Y.; Chang, P.L.; Juang, H.H. Growth differentiation factor-15: A p53- and demethylation-upregulating gene represses cell proliferation, invasion, and tumorigenesis in bladder carcinoma cells. Sci. Rep. 2015, 5, 12870. [Google Scholar] [CrossRef] [PubMed]
- Tsui, K.H.; Chung, L.C.; Wang, S.W.; Feng, T.H.; Chang, P.L.; Juang, H.H. Hypoxia upregulates the gene expression of mitochondrial aconitase in prostate carcinoma cells. J. Mol. Endocrinol. 2013, 51, 131–141. [Google Scholar] [CrossRef] [PubMed]
- Tsui, K.H.; Chang, Y.L.; Feng, T.H.; Hou, C.P.; Lin, Y.H.; Yang, P.S.; Lee, B.W.; Juang, H.H. Capillarisin blocks prostate-specific antigen expression on activation of androgen receptor in prostate carcinoma cells. Prostate 2018, 78, 242–249. [Google Scholar] [CrossRef] [PubMed]
- Chung, L.C.; Chiang, K.C.; Feng, T.H.; Chang, K.S.; Chuang, S.T.; Chen, Y.J.; Tsui, K.H.; Lee, J.C.; Juang, H.H. Caffeic acid phenethyl ester upregulates N-myc downstream regulated gene 1 via ERK pathway to inhibit human oral cancer cell growth in vitro and in vivo. Mol. Nutr. Food Res. 2017, 61, 1600842. [Google Scholar] [CrossRef] [PubMed]

© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tsui, K.-H.; Hou, C.-P.; Chang, K.-S.; Lin, Y.-H.; Feng, T.-H.; Chen, C.-C.; Shin, Y.-S.; Juang, H.-H. Metallothionein 3 Is a Hypoxia-Upregulated Oncogene Enhancing Cell Invasion and Tumorigenesis in Human Bladder Carcinoma Cells. Int. J. Mol. Sci. 2019, 20, 980. https://doi.org/10.3390/ijms20040980
Tsui K-H, Hou C-P, Chang K-S, Lin Y-H, Feng T-H, Chen C-C, Shin Y-S, Juang H-H. Metallothionein 3 Is a Hypoxia-Upregulated Oncogene Enhancing Cell Invasion and Tumorigenesis in Human Bladder Carcinoma Cells. International Journal of Molecular Sciences. 2019; 20(4):980. https://doi.org/10.3390/ijms20040980
Chicago/Turabian StyleTsui, Ke-Hung, Chen-Pang Hou, Kang-Shuo Chang, Yu-Hsiang Lin, Tsui-Hsia Feng, Chiu-Chun Chen, Yi-Syuan Shin, and Horng-Heng Juang. 2019. "Metallothionein 3 Is a Hypoxia-Upregulated Oncogene Enhancing Cell Invasion and Tumorigenesis in Human Bladder Carcinoma Cells" International Journal of Molecular Sciences 20, no. 4: 980. https://doi.org/10.3390/ijms20040980
APA StyleTsui, K.-H., Hou, C.-P., Chang, K.-S., Lin, Y.-H., Feng, T.-H., Chen, C.-C., Shin, Y.-S., & Juang, H.-H. (2019). Metallothionein 3 Is a Hypoxia-Upregulated Oncogene Enhancing Cell Invasion and Tumorigenesis in Human Bladder Carcinoma Cells. International Journal of Molecular Sciences, 20(4), 980. https://doi.org/10.3390/ijms20040980