cGAS/STING/TBK1/IRF3 Signaling Pathway Activates BMDCs Maturation Following Mycobacterium bovis Infection
Abstract
1. Introduction
2. Results
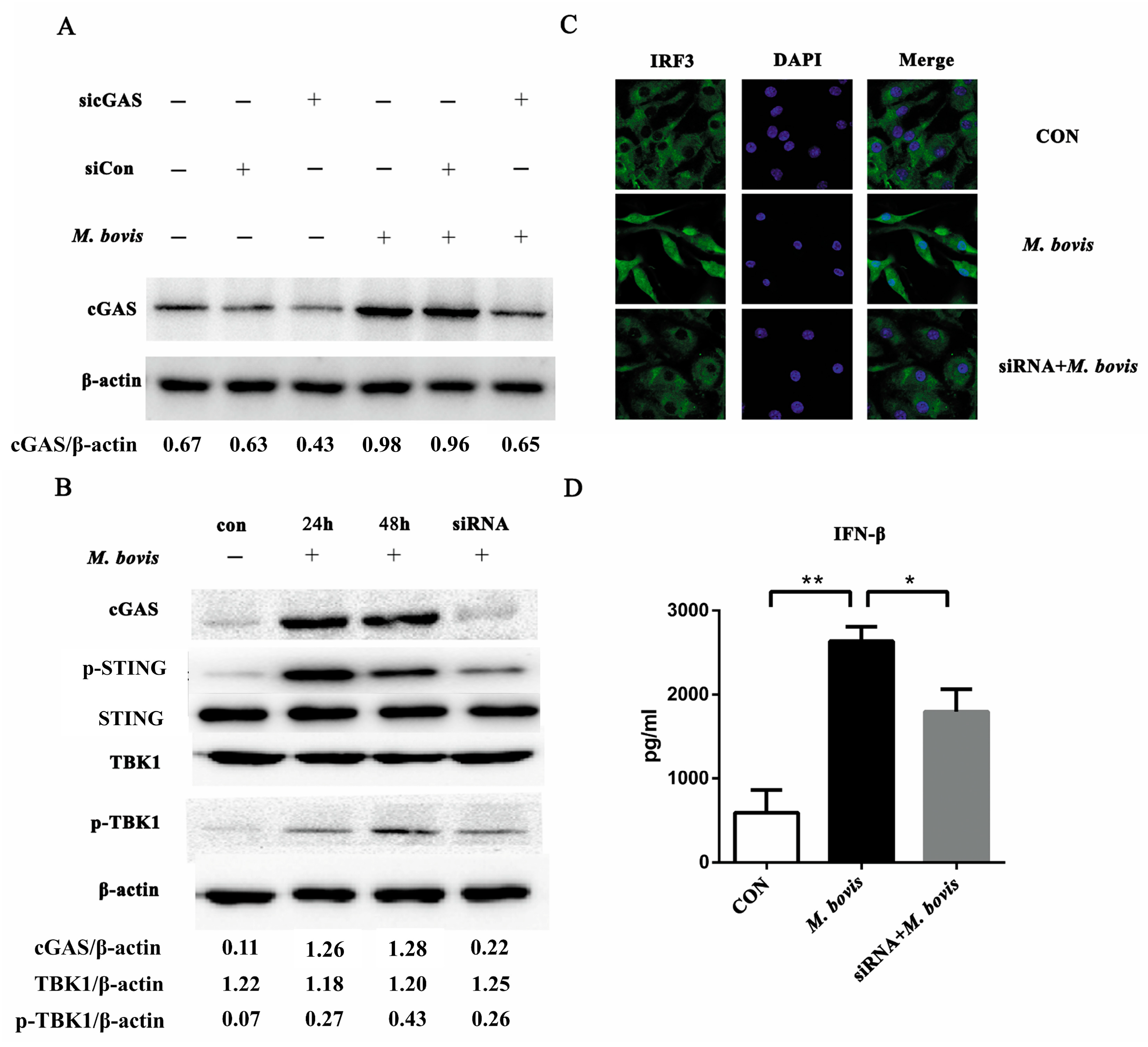
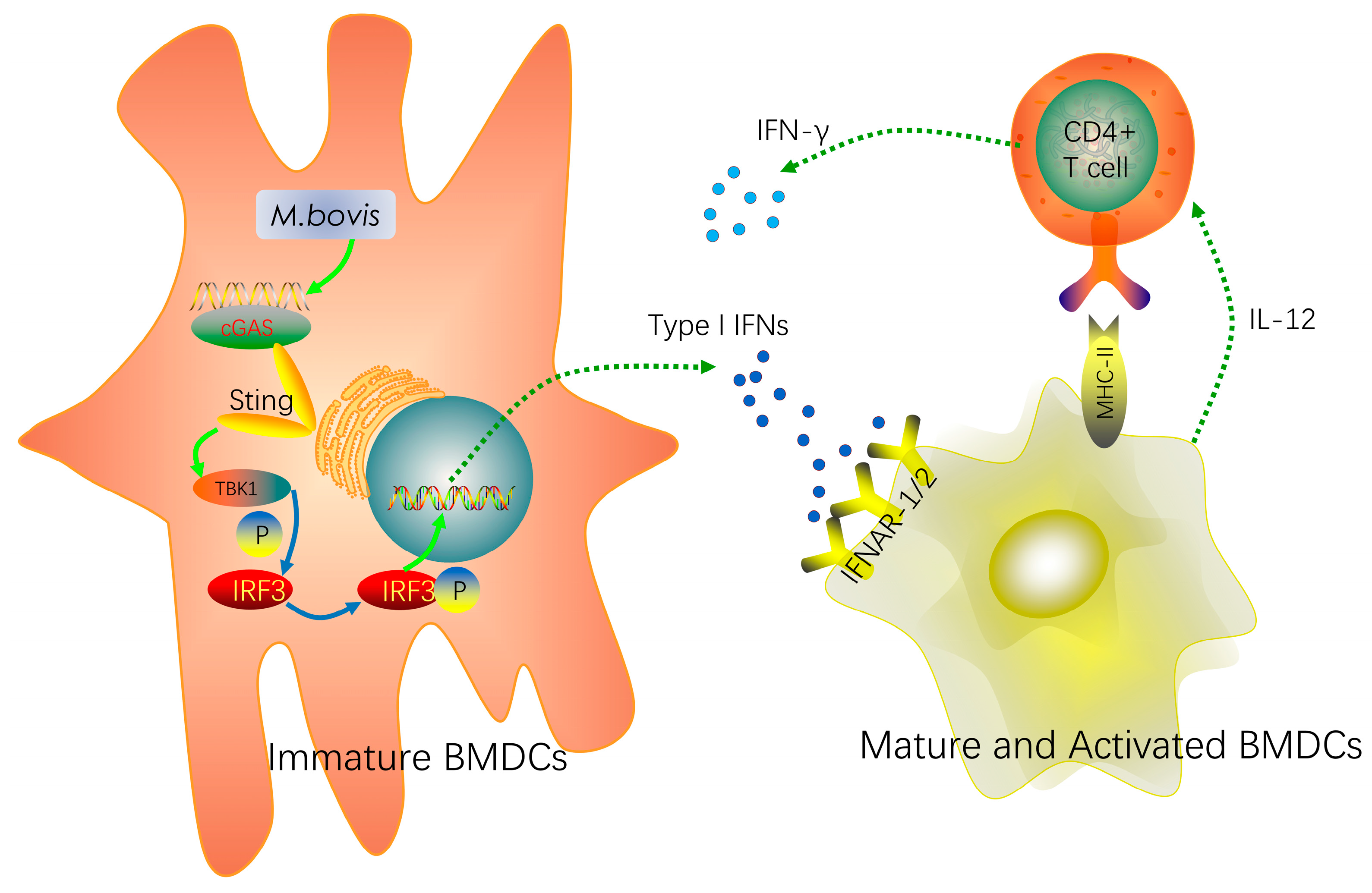
2.1. cGAS Pathway Is Activated in BMDCs During Mycobacterium bovis Infection
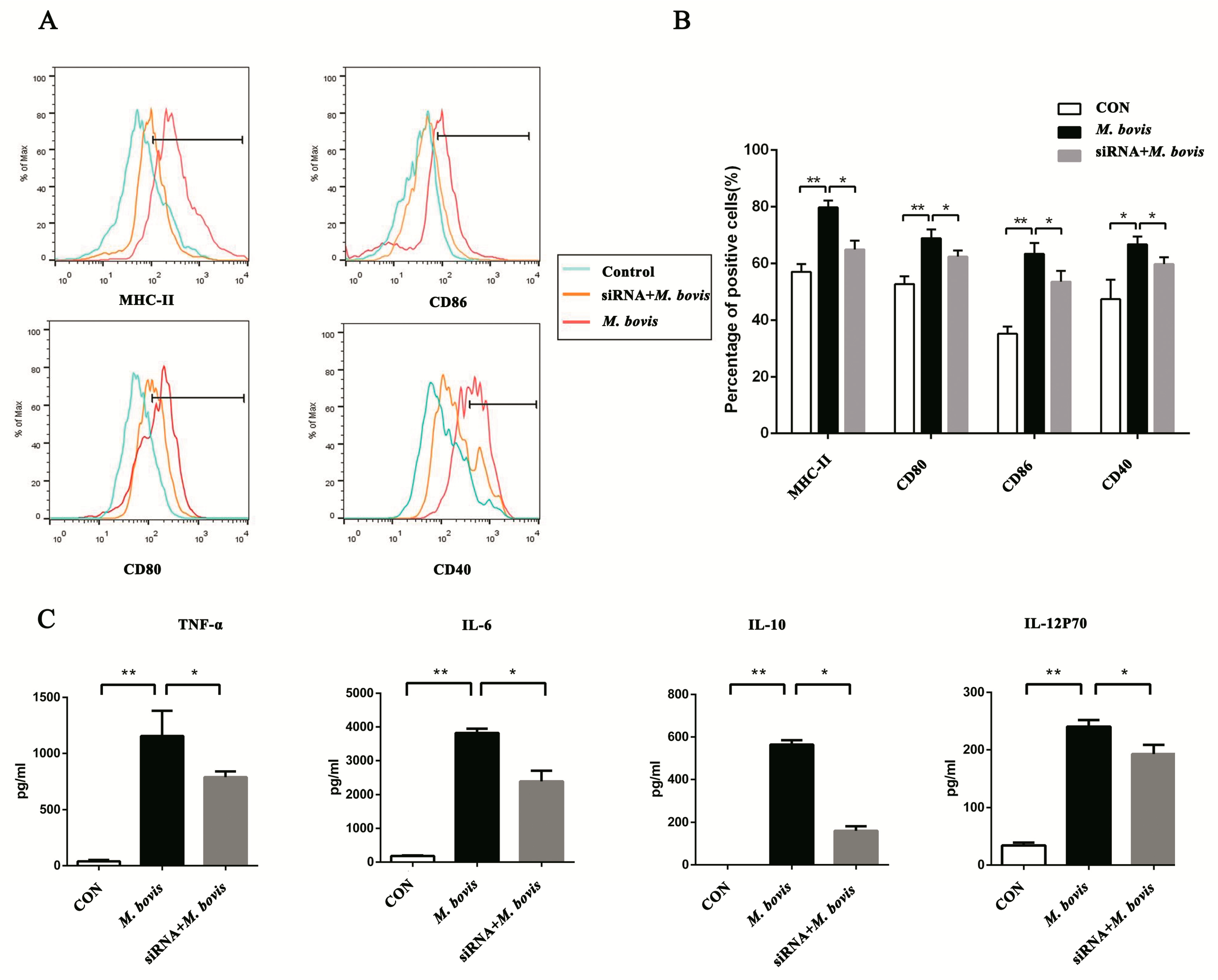
2.2. cGAS Pathway Promotes Maturation and Activation of BMDCs
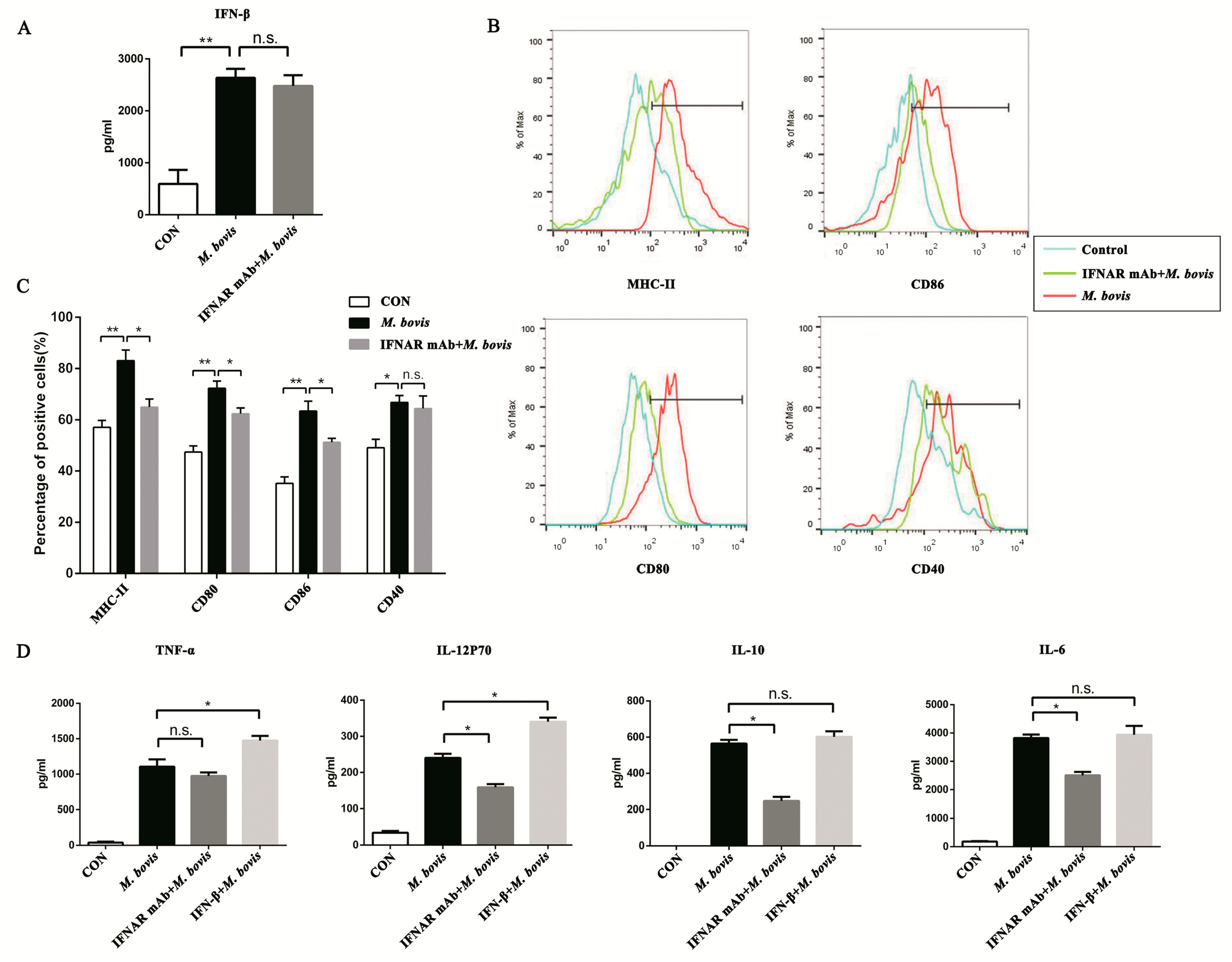
2.3. Type I Interferon and Its Receptor Contribute to the cGAS Pathway in BMDCs
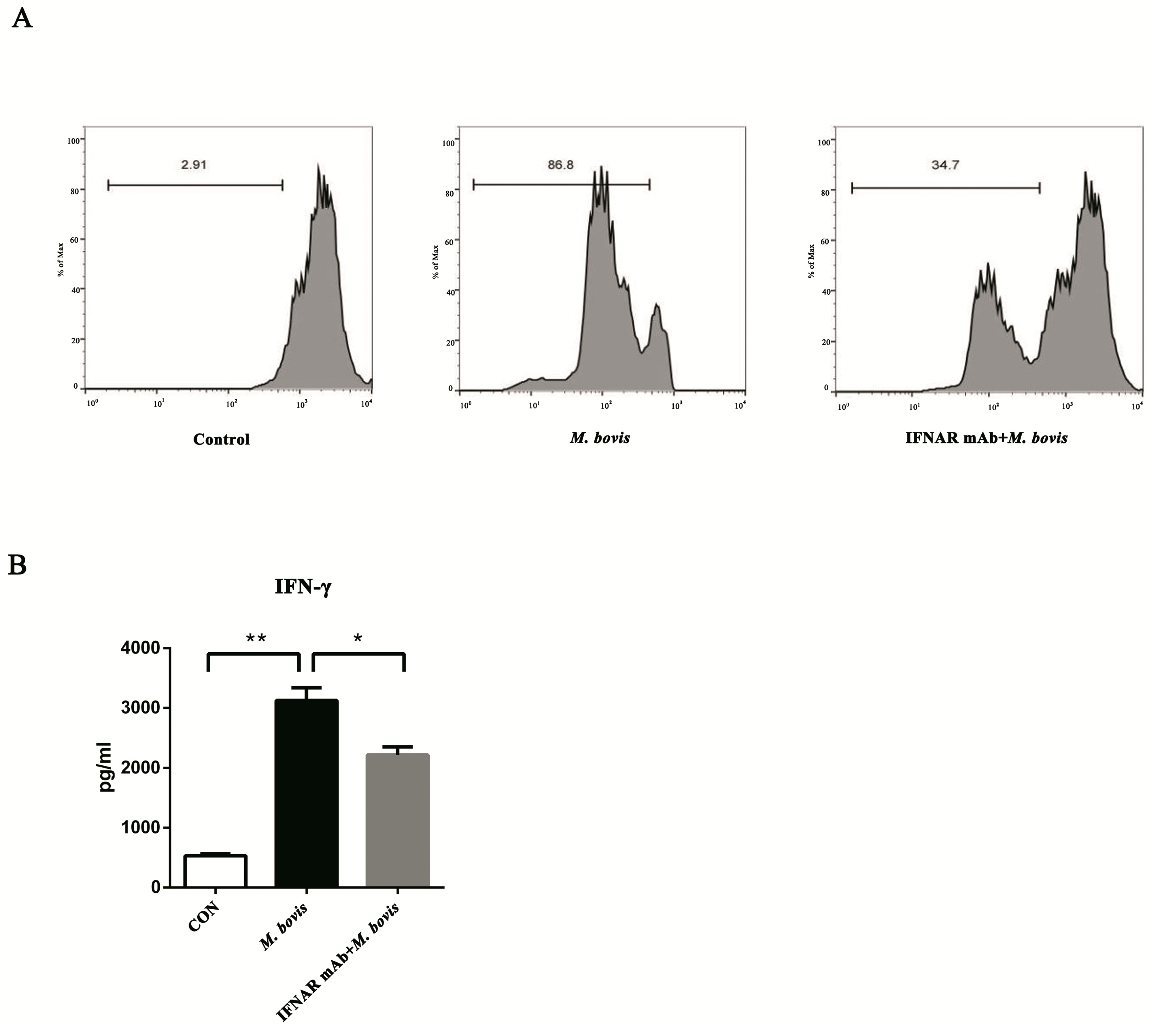
2.4. BMDCs Promote T cell Activity in the Presence of Type I Interferons
3. Discussion
4. Materials and Methods
4.1. Ethics Statement
4.2. Mice
4.3. Cell Preparation
4.4. siRNA Transfection
4.5. M. bovis Infection
4.6. Western Blotting
4.7. Enzyme-Linked Immunosorbent Assay
4.8. Flow Cytometry
4.9. Proliferation of CD4+ T Cells
4.10. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Glaziou, P.; Sismanidis, C.; Floyd, K.; Raviglione, M. Global Epidemiology of Tuberculosis. Cold Spring Harb. Perspect. Med. 2015, 5, a017798. [Google Scholar] [CrossRef] [PubMed]
- Evans, J.T.; Smith, E.G.; Banerjee, A.; Smith, R.M.; Dale, J.; Innes, J.A.; Hunt, D.; Tweddell, A.; Wood, A.; Anderson, C.; Hewinson, R.G.; et al. Cluster of human tuberculosis caused by Mycobacterium bovis: Evidence for person-to-person transmission in the UK. Lancet 2007, 369, 1270–1276. [Google Scholar] [CrossRef]
- Muller, B.; Durr, S.; Alonso, S.; Hattendorf, J.; Laisse, C.J.; Parsons, S.D.; van Helden, P.D.; Zinsstag, J. Zoonotic Mycobacterium bovis-induced tuberculosis in humans. Emerg. Infect. Dis. 2013, 19, 899–908. [Google Scholar]
- Scott, C.; Cavanaugh, J.S.; Pratt, R.; Silk, B.J.; LoBue, P.; Moonan, P.K. Human Tuberculosis Caused by Mycobacterium bovis in the United States, 2006–2013. Clin. Infect. Dis. 2016, 63, 594–601. [Google Scholar] [CrossRef] [PubMed]
- Sun, L.J.; Wu, J.; Du, F.; Chen, X.; Chen, Z.J. Cyclic GMP-AMP Synthase Is a Cytosolic DNA Sensor That Activates the Type I Interferon Pathway. Science 2013, 339, 786–791. [Google Scholar] [CrossRef] [PubMed]
- Lam, E.; Stein, S.; Falck-Pedersen,, E. Adenovirus Detection by the cGAS/STING/TBK1 DNA Sensing Cascade. J. Virol. 2014, 88, 974–981. [Google Scholar] [CrossRef] [PubMed]
- Bridgeman, A.; Maelfait, J.; Davenne, T.; Partridge, T.; Peng, Y.; Mayer, A.; Dong, T.; Kaever, V.; Borrow, P.; Rehwinkel, J. Viruses transfer the antiviral second messenger cGAMP between cells. Science 2015, 349, 1228–1232. [Google Scholar] [CrossRef] [PubMed]
- Carroll, E.C.; Jin, L.; Mori, A.; Munoz-Wolf, N.; Oleszycka, E.; Moran, H.B.T.; Mansouri, S.; McEntee, C.P.; Lambe, E.; Agger, E.M.; et al. The Vaccine Adjuvant Chitosan Promotes Cellular Immunity via DNA Sensor cGAS-STING-Dependent Induction of Type I Interferons. Immunity 2016, 44, 597–608. [Google Scholar] [CrossRef]
- Schoggins, J.W.; MacDuff, D.A.; Imanaka, N.; Gainey, M.D.; Shrestha, B.; Eitson, J.L.; Mar, K.B.; Richardson, R.B.; Ratushny, A.V.; Litvak, V.; et al. Pan-viral specificity of IFN-induced genes reveals new roles for cGAS in innate immunity. Nature 2014, 505, 691. [Google Scholar] [CrossRef]
- Zhang, Z.Q.; Yuan, B.; Bao, M.; Lu, N.; Kim, T.; Liu, Y.J. The helicase DDX41 senses intracellular DNA mediated by the adaptor STING in dendritic cells. Nat. Immunol. 2011, 12, 959. [Google Scholar] [CrossRef]
- Wassermann, R.; Gulen, M.F.; Sala, C.; Perin, S.G.; Lou, Y.; Rybniker, J.; Schmid-Burgk, J.L.; Schmidt, T.; Hornung, V.; Cole, S.T.; et al. Mycobacterium tuberculosis Differentially Activates cGAS- and Inflammasome-Dependent Intracellular Immune Responses through ESX-1. Cell Host Microbe 2015, 17, 799–810. [Google Scholar]
- Arnold-Schrauf, C.; Dudek, M.; Dielmann, A.; Pace, L.; Swallow, M.; Kruse, F.; Kuhl, A.A.; Holzmann, B.; Berod, L.; Sparwasser, T. Dendritic cells coordinate innate immunity via MyD88 signaling to control Listeria monocytogenes infection. Cell Rep. 2014, 6, 698–708. [Google Scholar] [CrossRef] [PubMed]
- Khan, N.; Vidyarthi, A.; Pahari, S.; Agrewala, J.N. Distinct Strategies Employed by Dendritic Cells and Macrophages in Restricting Mycobacterium tuberculosis Infection: Different Philosophies but Same Desire. Int. Rev. Immunol. 2016, 35, 386–398. [Google Scholar] [CrossRef] [PubMed]
- Byun, E.H.; Kim, W.S.; Kim, J.S.; Jung, I.D.; Park, Y.M.; Kim, H.J.; Cho, S.N.; Shin, S.J. Mycobacterium tuberculosis Rv0577, a novel TLR2 agonist, induces maturation of dendritic cells and drives Th1 immune response. FASEB J. 2012, 26, 2695–2711. [Google Scholar] [CrossRef] [PubMed]
- Satchidanandam, V.; Kumar, N.; Jumani, R.S.; Challu, V.; Elangovan, S.; Khan, N.A. The Glycosylated Rv1860 Protein of Mycobacterium tuberculosis Inhibits Dendritic Cell Mediated TH1 and TH17 Polarization of T Cells and Abrogates Protective Immunity Conferred by BCG. PLoS Pathog. 2014, 10, e1004176. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Yue, R.; Yang, Y.; Cui, Y.; Yang, L.; Zhao, D.; Zhou, X. AIM2 inhibits autophagy and IFN-beta production during M. bovis infection. Oncotarget 2016, 7, 46972–46987. [Google Scholar] [PubMed]
- Tailleux, L.; Schwartz, O.; Herrmann, J.-L.; Pivert, E.; Jackson, M.; Amara, A.; Legres, L.; Dreher, D.; Nicod, L.P.; Gluckman, J.C.; et al. DC-SIGN is the major Mycobacterium tuberculosis receptor on human dendritic cells. J. Exp. Med. 2003, 197, 121–127. [Google Scholar] [CrossRef] [PubMed]
- Steinman, R.M. Dendritic cells: Understanding immunogenicity. Eur. J. Immunol. 2007, 37, S53–S60. [Google Scholar] [CrossRef]
- Li, X.; Shu, C.; Yi, G.; Chaton, C.T.; Shelton, C.L.; Diao, J.; Zuo, X.; Kao, C.C.; Herr, A.B.; Li, P. Cyclic GMP-AMP synthase is activated by double-stranded DNA-induced oligomerization. Immunity 2013, 39, 1019–1031. [Google Scholar] [CrossRef]
- Shu, C.; Yi, G.; Watts, T.; Kao, C.C.; Li, P. Structure of STING bound to cyclic di-GMP reveals the mechanism of cyclic dinucleotide recognition by the immune system. Nat. Struct. Mol. Biol. 2012, 19, 722. [Google Scholar] [CrossRef]
- Civril, F.; Deimling, T.; de Oliveira Mann, C.C.; Ablasser, A.; Moldt, M.; Witte, G.; Hornung, V.; Hopfner, K.P. Structural mechanism of cytosolic DNA sensing by cGAS. Nature 2013, 498, 332–337. [Google Scholar] [CrossRef]
- Kranzusch, P.J.; Lee, A.S.; Berger, J.M.; Doudna, J.A. Structure of Human cGAS Reveals a Conserved Family of Second-Messenger Enzymes in Innate Immunity. Cell Rep. 2013, 3, 1362–1368. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Wu, J.; Du, F.; Xu, H.; Sun, L.; Chen, Z.; Brautigam, C.A.; Zhang, X.; Chen, Z.J. The Cytosolic DNA Sensor cGAS Forms an Oligomeric Complex with DNA and Undergoes Switch-like Conformational Changes in the Activation Loop. Cell Rep. 2014, 6, 421–430. [Google Scholar] [CrossRef] [PubMed]
- Gao, P.; Ascano, M.; Zillinger, T.; Wang, W.; Dai, P.; Serganov, A.A.; Gaffney, B.L.; Shuman, S.; Jones, R.A.; Deng, L.; et al. Structure-Function Analysis of STING Activation by c[G(2′,5′) pA(3′,5′)p] and Targeting by Antiviral DMXAA. Cell 2013, 154, 748–762. [Google Scholar] [CrossRef]
- Collins, A.C.; Cai, H.; Li, T.; Franco, L.H.; Li, X.D.; Nair, V.R.; Scharn, C.R.; Stamm, C.E.; Levine, B.; Chen, Z.J.; et al. Cyclic GMP-AMP Synthase Is an Innate Immune DNA Sensor for Mycobacterium tuberculosis. Cell Host Microbe 2015, 17, 820–828. [Google Scholar]
- Teles, R.M.B.; Graeber, T.G.; Krutzik, S.R.; Montoya, D.; Schenk, M.; Lee, D.J.; Komisopoulou, E.; Kelly-Scumpia, K.; Chun, R.; Iyer, S.S.; Sarno, E.N.; et al. Type I Interferon Suppresses Type II Interferon-Triggered Human Anti-Mycobacterial Responses. Science 2013, 339, 1448–1453. [Google Scholar] [CrossRef]
- Radtke, A.J.; Kastenmuller, W.; Espinosa, D.A.; Gerner, M.Y.; Tse, S.W.; Sinnis, P.; Germain, R.N.; Zavala, F.P.; Cockburn, I.A. Lymph-node resident CD8α+ dendritic cells capture antigens from migratory malaria sporozoites and induce CD8+ T cell responses. PLoS Pathog. 2015, 11, e1004637. [Google Scholar] [CrossRef] [PubMed]
- Bode, C.; Fox, M.; Tewary, P.; Steinhagen, A.; Ellerkmann, R.K.; Klinman, D.; Baumgarten, G.; Hornung, V.; Steinhagen, F. Human plasmacytoid dentritic cells elicit a Type I Interferon response by sensing DNA via the cGAS-STING signaling pathway. Eur. J. Immunol. 2016, 46, 1615–1621. [Google Scholar] [CrossRef]
- Manzanillo, P.S.; Shiloh, M.U.; Portnoy, D.A.; Cox, J.S. Mycobacterium tuberculosis Activates the DNA-Dependent Cytosolic Surveillance Pathway within Macrophages. Cell Host & Microbe 2012, 11, 469–480. [Google Scholar]
- Hansen, M.; Met, O.; Svane, I.M.; Andersen, M.H. Cellular Based Cancer Vaccines: Type 1 Polarization of Dendritic Cells. Curr. Med. Chem. 2012, 19, 4239–4246. [Google Scholar] [CrossRef]
- Montoya, M.; Schiavoni, G.; Mattei, F.; Gresser, I.; Belardelli, F.; Borrow, P.; Tough, D.F. Type I interferons produced by dendritic cells promote their phenotypic and functional activation. Blood 2002, 99, 3263–3271. [Google Scholar] [CrossRef]
- Stanley, S.A.; Johndrow, J.E.; Manzanillo, P.; Cox, J.S. The type I IFN response to infection with Mycobacterium tuberculosis requires ESX-1-mediated secretion and contributes to pathogenesis. J. Immunol. 2007, 178, 3143–3152. [Google Scholar] [CrossRef] [PubMed]
- Ma, F.; Li, B.; Liu, S.Y.; Iyer, S.S.; Yu, Y.; Wu, A.; Cheng, G. Positive Feedback Regulation of Type I IFN Production by the IFN-Inducible DNA Sensor cGAS. J. Immunol. 2015, 194, 1545–1554. [Google Scholar] [CrossRef] [PubMed]
- Pelka, K.; Latz, E. IRF5, IRF8, and IRF7 in human pDCs—The good, the bad, and the insignificant? Eur J. Immunol. 2013, 43, 1693–1697. [Google Scholar] [CrossRef] [PubMed]
- Abe, T.; Barber, G.N. Cytosolic-DNA-mediated, STING-dependent proinflammatory gene induction necessitates canonical NF-kappaB activation through TBK1. J. Virol. 2014, 88, 5328–5341. [Google Scholar] [CrossRef] [PubMed]
- Lienard, J.; Movert, E.; Valfridsson, C.; Sturegard, E.; Carlsson, F. ESX-1 exploits type I IFN-signalling to promote a regulatory macrophage phenotype refractory to IFNgamma-mediated autophagy and growth restriction of intracellular mycobacteria. Cell Microbiol. 2016, 18, 1471–1485. [Google Scholar] [CrossRef]
- Ersoy, E.; Kus, C.N.; Sener, U.; Coker, I.; Zorlu, Y. The effects of interferon-beta on interleukin-10 in multiple sclerosis patients. Eur. J. Neurol. 2005, 12, 208–211. [Google Scholar] [CrossRef]
- Zhang, D.; Zhang, W.; Ng, T.W.; Wang, Y.; Liu, Q.; Gorantla, V.; Lakkis, F.; Zheng, X.X. Adoptive cell therapy using antigen-specific CD4−CD8−T regulatory cells to prevent autoimmune diabetes and promote islet allograft survival in NOD mice. Diabetologia 2011, 54, 2082–2092. [Google Scholar] [CrossRef]
- Feng, X.; Petraglia, A.L.; Chen, M.; Byskosh, P.V.; Boos, M.D.; Reder, A.T. Low expression of interferon-stimulated genes in active multiple sclerosis is linked to subnormal phosphorylation of STAT1. J. Neuroimmunol. 2002, 129, 205–215. [Google Scholar] [CrossRef]
- Etna, M.P.; Giacomini, E.; Severa, M.; Coccia, E.M. Pro- and anti-inflammatory cytokines in tuberculosis: A two-edged sword in TB pathogenesis. Semin. Immunol. 2014, 26, 543–551. [Google Scholar] [CrossRef]
- Lutz, M.B.; Kukutsch, N.; Ogilvie, A.L.; Rossner, S.; Koch, F.; Romani, N.; Schuler, G. An advanced culture method for generating large quantities of highly pure dendritic cells from mouse bone marrow. J. Immunol. Methods. 1999, 223, 77–92. [Google Scholar] [CrossRef]
- Zhou, Y.; Zhao, D.; Yue, R.; Khan, S.H.; Shah, S.Z.; Yin, X.; Yang, L.; Zhang, Z.; Zhou, X. Inflammasomes-dependent regulation of IL-1 beta secretion induced by the virulent Mycobacterium bovis Beijing strain in THP-1 macrophages. Antonie Van Leeuwenhoek. 2015, 108, 163–171. [Google Scholar] [CrossRef]
- Kim, J.S.; Kim, W.S.; Choi, H.G.; Jang, B.; Lee, K.; Park, J.H.; Kim, H.J.; Cho, S.N.; Shin, S.J. Mycobacterium tuberculosis RpfB drives Th1-type T cell immunity via a TLR4-dependent activation of dendritic cells. J. Leukoc. Biol. 2013, 94, 733–749. [Google Scholar] [CrossRef] [PubMed]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, Q.; Liu, C.; Yue, R.; El-Ashram, S.; Wang, J.; He, X.; Zhao, D.; Zhou, X.; Xu, L. cGAS/STING/TBK1/IRF3 Signaling Pathway Activates BMDCs Maturation Following Mycobacterium bovis Infection. Int. J. Mol. Sci. 2019, 20, 895. https://doi.org/10.3390/ijms20040895
Li Q, Liu C, Yue R, El-Ashram S, Wang J, He X, Zhao D, Zhou X, Xu L. cGAS/STING/TBK1/IRF3 Signaling Pathway Activates BMDCs Maturation Following Mycobacterium bovis Infection. International Journal of Molecular Sciences. 2019; 20(4):895. https://doi.org/10.3390/ijms20040895
Chicago/Turabian StyleLi, Qiang, Chunfa Liu, Ruichao Yue, Saeed El-Ashram, Jie Wang, Xiaoli He, Deming Zhao, Xiangmei Zhou, and Lihua Xu. 2019. "cGAS/STING/TBK1/IRF3 Signaling Pathway Activates BMDCs Maturation Following Mycobacterium bovis Infection" International Journal of Molecular Sciences 20, no. 4: 895. https://doi.org/10.3390/ijms20040895
APA StyleLi, Q., Liu, C., Yue, R., El-Ashram, S., Wang, J., He, X., Zhao, D., Zhou, X., & Xu, L. (2019). cGAS/STING/TBK1/IRF3 Signaling Pathway Activates BMDCs Maturation Following Mycobacterium bovis Infection. International Journal of Molecular Sciences, 20(4), 895. https://doi.org/10.3390/ijms20040895





