Increased HLA-G Expression in Term Placenta of Women with a History of Recurrent Miscarriage Despite Their Genetic Predisposition to Decreased HLA-G Levels
Abstract
1. Introduction
2. Results
2.1. Patient Characteristics
2.2. HLA-G Polymorphisms and Haplotypes
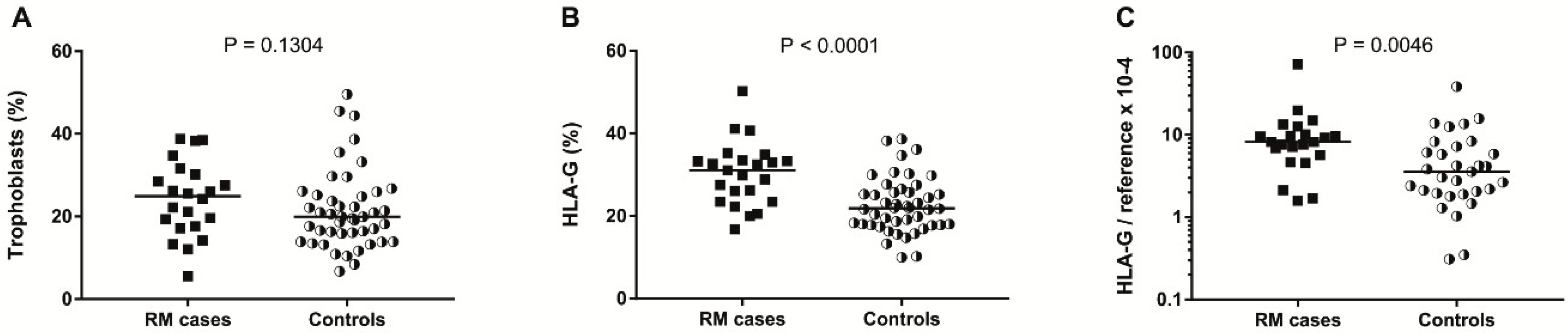
2.3. Placental HLA-G Expression Is Elevated in Women with History of RM
2.4. HLA-G Expression in First Trimester Miscarriage Material and Elective Abortions Is Similar
3. Discussion
4. Materials and Methods
4.1. Subjects and Materials
4.2. HLA-G Polymorphisms and Haplotypes
4.3. Immunohistochemistry
4.4. Quantification of Immunohistochemical Stainings
4.5. RNA Isolation and qPCR
4.6. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| 3′UTR | Three Prime Untranslated Region |
| DNA | Deoxyribonucleic Acid |
| EVT | Extravillous Trophoblast |
| GA | Gestational Age |
| HLA | Human Leukocyte Antigen |
| MMP | Matrix Metalloproteinase |
| NK | Natural Killer |
| OR | Odds Ratio |
| PCR | Polymerase Chain Reaction |
| RM | Recurrent Miscarriage |
| sHLA | Soluble HLA |
| SNP | Single Nucleotide Polymorphism |
References
- Regan, L.; Backos, M.; Rai, R. Recurrent Miscarriage, Investigation and Treatment of Couples; Greentop Guideline 17; Royal College of Obstetricians and Gynaecologists: London, UK, 2011. [Google Scholar]
- Practice Committee of the American Society for Reproductive Medicine. Evaluation and treatment of recurrent pregnancy loss: A committee opinion. Fertil. Steril. 2012, 98, 1103–1111. [Google Scholar] [CrossRef] [PubMed]
- Kovats, S.; Main, E.K.; Librach, C.; Stubblebine, M.; Fisher, S.J.; DeMars, R. A class I antigen, HLA-G, expressed in human trophoblasts. Science 1990, 248, 220–223. [Google Scholar] [CrossRef] [PubMed]
- Ishitani, A.; Sageshima, N.; Lee, N.; Dorofeeva, N.; Hatake, K.; Marquardt, H.; Geraghty, D.E. Protein expression and peptide binding suggest unique and interacting functional roles for HLA-E, F, and G in maternal-placental immune recognition. J. Immunol. 2003, 171, 1376–1384. [Google Scholar] [CrossRef] [PubMed]
- Kofod, L.; Lindhard, A.; Bzorek, M.; Eriksen, J.O.; Larsen, L.G.; Hviid, T.V.F. Endometrial immune markers are potential predictors of normal fertility and pregnancy after in vitro fertilization. Am. J. Reprod. Immunol. 2017, 78. [Google Scholar] [CrossRef] [PubMed]
- Pfeiffer, K.A.; Rebmann, V.; Passler, M.; van der Ven, K.; van der Ven, H.; Krebs, D.; Grosse-Wilde, H. Soluble HLA levels in early pregnancy after in vitro fertilization. Hum. Immunol. 2000, 61, 559–564. [Google Scholar] [CrossRef]
- Lee, N.; Malacko, A.R.; Ishitani, A.; Chen, M.C.; Bajorath, J.; Marquardt, H.; Geraghty, D.E. The membrane-bound and soluble forms of HLA-G bind identical sets of endogenous peptides but differ with respect to TAP association. Immunity 1995, 3, 591–600. [Google Scholar] [CrossRef]
- Rebmann, V.; Pfeiffer, K.; Passler, M.; Ferrone, S.; Maier, S.; Weiss, E.; Grosse-Wilde, H. Detection of soluble HLA-G molecules in plasma and amniotic fluid. Tissue Antigens 1999, 53, 14–22. [Google Scholar] [CrossRef] [PubMed]
- Shaikly, V.R.; Morrison, I.E.; Taranissi, M.; Noble, C.V.; Withey, A.D.; Cherry, R.J.; Blois, S.M.; Fernandez, N. Analysis of HLA-G in maternal plasma, follicular fluid, and preimplantation embryos reveal an asymmetric pattern of expression. J. Immunol. 2008, 180, 4330–4337. [Google Scholar] [CrossRef] [PubMed]
- Larsen, M.H.; Bzorek, M.; Pass, M.B.; Larsen, L.G.; Nielsen, M.W.; Svendsen, S.G.; Lindhard, A.; Hviid, T.V. Human leukocyte antigen-G in the male reproductive system and in seminal plasma. Mol. Hum. Reprod. 2011, 17, 727–738. [Google Scholar] [CrossRef] [PubMed]
- Rizzo, R.; Trentini, A.; Bortolotti, D.; Manfrinato, M.C.; Rotola, A.; Castellazzi, M.; Melchiorri, L.; Di Luca, D.; Dallocchio, F.; Fainardi, E.; et al. Matrix metalloproteinase-2 (MMP-2) generates soluble HLA-G1 by cell surface proteolytic shedding. Mol. Cell. Biochem. 2013, 381, 243–255. [Google Scholar] [CrossRef] [PubMed]
- Rousseau, P.; Le Discorde, M.; Mouillot, G.; Marcou, C.; Carosella, E.D.; Moreau, P. The 14 bp deletion-insertion polymorphism in the 3′UT region of the HLA-G gene influences HLA-G mRNA stability. Hum. Immunol. 2003, 64, 1005–1010. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.Y.; Yan, W.H.; Lin, A.; Xu, H.H.; Zhang, J.G.; Wang, X.X. The 14 bp deletion polymorphisms in HLA-G gene play an important role in the expression of soluble HLA-G in plasma. Tissue Antigens 2008, 72, 335–341. [Google Scholar] [CrossRef] [PubMed]
- Yie, S.M.; Li, L.H.; Xiao, R.; Librach, C.L. A single base-pair mutation in the 3′-untranslated region of HLA-G mRNA is associated with pre-eclampsia. Mol. Hum. Reprod. 2008, 14, 649–653. [Google Scholar] [CrossRef] [PubMed]
- Tan, Z.; Randall, G.; Fan, J.; Camoretti-Mercado, B.; Brockman-Schneider, R.; Pan, L.; Solway, J.; Gern, J.E.; Lemanske, R.F.; Nicolae, D.; et al. Allele-specific targeting of microRNAs to HLA-G and risk of asthma. Am. J. Hum. Genet. 2007, 81, 829–834. [Google Scholar] [CrossRef] [PubMed]
- Hviid, T.V.; Hylenius, S.; Rorbye, C.; Nielsen, L.G. HLA-G allelic variants are associated with differences in the HLA-G mRNA isoform profile and HLA-G mRNA levels. Immunogenetics 2003, 55, 63–79. [Google Scholar] [PubMed]
- Bland, J.M.; Altman, D.G. Statistical methods for assessing agreement between two methods of clinical measurement. Lancet 1986, 1, 307–310. [Google Scholar] [CrossRef]
- Mori, A.; Nishi, H.; Sasaki, T.; Nagamitsu, Y.; Kawaguchi, R.; Okamoto, A.; Kuroda, M.; Isaka, K. HLA-G expression is regulated by miR-365 in trophoblasts under hypoxic conditions. Placenta 2016, 45, 37–41. [Google Scholar] [CrossRef] [PubMed]
- Manaster, I.; Goldman-Wohl, D.; Greenfield, C.; Nachmani, D.; Tsukerman, P.; Hamani, Y.; Yagel, S.; Mandelboim, O. MiRNA-mediated control of HLA-G expression and function. PLoS ONE 2012, 7, e33395. [Google Scholar] [CrossRef] [PubMed]
- Djurisic, S.; Teiblum, S.; Tolstrup, C.K.; Christiansen, O.B.; Hviid, T.V. Allelic imbalance modulates surface expression of the tolerance-inducing HLA-G molecule on primary trophoblast cells. Mol. Hum. Reprod. 2015, 21, 281–295. [Google Scholar] [CrossRef] [PubMed]
- Kalotra, V.; Lall, M.; Verma, I.C.; Kaur, A.; Kaur, A. The HLA-G 14 bp insertion/deletion polymorphism and its association with soluble HLA-G levels in women with recurrent miscarriages. HLA 2018, 91, 167–174. [Google Scholar] [CrossRef] [PubMed]
- Xue, S.; Yang, J.; Yao, F.; Xu, L.; Fan, L. Recurrent spontaneous abortions patients have more −14 bp/+14 bp heterozygotes in the 3′UT region of the HLA-G gene in a Chinese Han population. Tissue Antigens 2007, 69 (Suppl. S1), 153–155. [Google Scholar] [CrossRef] [PubMed]
- Tripathi, P.; Abbas, A.; Naik, S.; Agrawal, S. Role of 14-bp deletion in the HLA-G gene in the maintenance of pregnancy. Tissue Antigens 2004, 64, 706–710. [Google Scholar] [CrossRef] [PubMed]
- Vargas, R.G.; Sarturi, P.R.; Mattar, S.B.; Bompeixe, E.P.; Silva Jdos, S.; Pirri, A.; Bicalho Mda, G. Association of HLA-G alleles and 3′-UTR 14 bp haplotypes with recurrent miscarriage in Brazilian couples. Hum. Immunol. 2011, 72, 479–485. [Google Scholar] [CrossRef] [PubMed]
- Hviid, T.V.; Hylenius, S.; Hoegh, A.M.; Kruse, C.; Christiansen, O.B. HLA-G polymorphisms in couples with recurrent spontaneous abortions. Tissue Antigens 2002, 60, 122–132. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Jiang, W.; Zhang, D. Association of 14-bp insertion/deletion polymorphism of HLA-G gene with unexplained recurrent spontaneous abortion: A meta-analysis. Tissue Antigens 2013, 81, 108–115. [Google Scholar] [CrossRef] [PubMed]
- Fan, W.; Li, S.; Huang, Z.; Chen, Q. Relationship between HLA-G polymorphism and susceptibility to recurrent miscarriage: A meta-analysis of non-family-based studies. J. Assist. Reprod. Genet. 2014, 31, 173–184. [Google Scholar] [CrossRef] [PubMed]
- Yazdani, N.; Shekari Khaniani, M.; Bastami, M.; Ghasemnejad, T.; Afkhami, F.; Mansoori Derakhshan, S. HLA-G regulatory variants and haplotypes with susceptibility to recurrent pregnancy loss. Int. J. Immunogenet. 2018, 45, 181–189. [Google Scholar] [CrossRef] [PubMed]
- Amodio, G.; Canti, V.; Maggio, L.; Rosa, S.; Castiglioni, M.T.; Rovere-Querini, P.; Gregori, S. Association of genetic variants in the 3′UTR of HLA-G with Recurrent Pregnancy Loss. Hum. Immunol. 2016, 77, 886–891. [Google Scholar] [CrossRef] [PubMed]
- Meuleman, T.; Drabbels, J.; van Lith, J.M.M.; Dekkers, O.M.; Rozemuller, E.; Cretu-Stancu, M.; Claas, F.H.J.; Bloemenkamp, K.W.M.; Eikmans, M. Lower frequency of the HLA-G UTR-4 haplotype in women with unexplained recurrent miscarriage. J. Reprod. Immunol. 2018, 126, 46–52. [Google Scholar] [CrossRef]
- Emmer, P.M.; Steegers, E.A.; Kerstens, H.M.; Bulten, J.; Nelen, W.L.; Boer, K.; Joosten, I. Altered phenotype of HLA-G expressing trophoblast and decidual natural killer cells in pathological pregnancies. Hum. Reprod. 2002, 17, 1072–1080. [Google Scholar] [CrossRef]
- Bhalla, A.; Stone, P.R.; Liddell, H.S.; Zanderigo, A.; Chamley, L.W. Comparison of the expression of human leukocyte antigen (HLA)-G and HLA-E in women with normal pregnancy and those with recurrent miscarriage. Reproduction 2006, 131, 583–589. [Google Scholar] [CrossRef]
- Dahl, M.; Klitkou, L.; Christiansen, O.B.; Djurisic, S.; Piosik, Z.M.; Skovbo, P.; Moller, A.M.; Steffensen, R.; Hviid, T.V. Human leukocyte antigen (HLA)-G during pregnancy part II: Associations between maternal and fetal HLA-G genotypes and soluble HLA-G. Hum. Immunol. 2015, 76, 260–271. [Google Scholar] [CrossRef] [PubMed]
- Goldman-Wohl, D.S.; Ariel, I.; Greenfield, C.; Hanoch, J.; Yagel, S. HLA-G expression in extravillous trophoblasts is an intrinsic property of cell differentiation: A lesson learned from ectopic pregnancies. Mol. Hum. Reprod. 2000, 6, 535–540. [Google Scholar] [CrossRef] [PubMed]
- Okimaa, V.; Oksjoki, S.; Kujari, H.; Vuorio, E.; Anttila, L. Altered expression of genes involved in the production and degradation of endometrial extracellular matrix in patients with unexplained infertility and recurrent miscarriages. Mol. Hum. Reprod. 2002, 8, 1111–1116. [Google Scholar]
- Meuleman, T.; Cohen, D.; Swings, G.M.; Veraar, K.; Claas, F.H.; Bloemenkamp, K.W. Increased complement C4d deposition at the maternal-fetal interface in unexplained recurrent miscarriage. J. Reprod. Immunol. 2016, 113, 54–60. [Google Scholar] [CrossRef] [PubMed]
- Hua, Y.J.; Tu, K.; Tang, Z.Y.; Li, Y.X.; Xiao, H.S. Comparison of normalization methods with microRNA microarray. Genomics 2008, 92, 122–128. [Google Scholar] [CrossRef]
- Wotschofsky, Z.; Meyer, H.A.; Jung, M.; Fendler, A.; Wagner, I.; Stephan, C.; Busch, J.; Erbersdobler, A.; Disch, A.C.; Mollenkopf, H.J.; et al. Reference genes for the relative quantification of microRNAs in renal cell carcinomas and their metastases. Anal. Biochem. 2011, 417, 233–241. [Google Scholar] [CrossRef] [PubMed]
- Lancaster, A.K.; Single, R.M.; Solberg, O.D.; Nelson, M.P.; Thomson, G. PyPop update—A software pipeline for large-scale multilocus population genomics. Tissue Antigens 2007, 69, 192–197. [Google Scholar] [CrossRef]
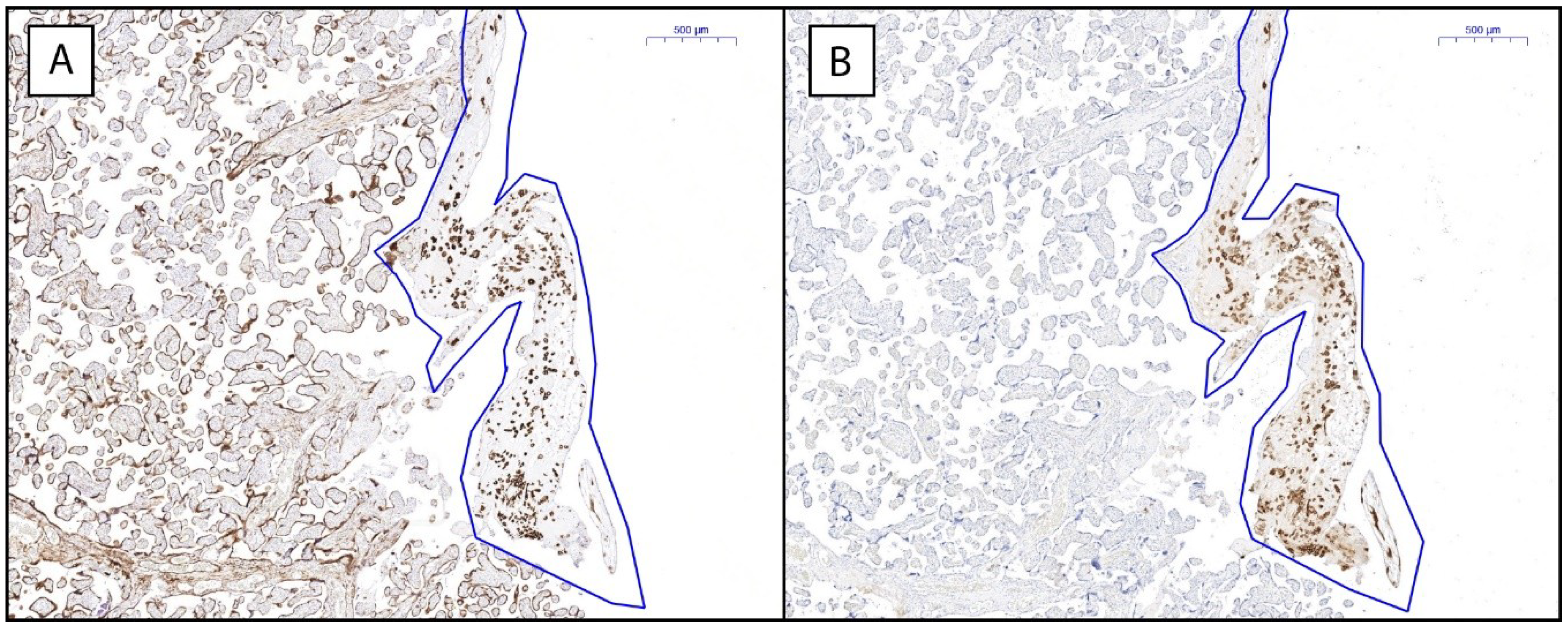

| Parameters | Pregnancy after RM (n = 23) | Uneventful Pregnancy (n = 46) | p-Value * |
|---|---|---|---|
| Maternal age at time of index pregnancy in years | 34 (22–39) # | 33 (20–41) # | 0.548 |
| Gestational age at time of birth in weeks | 39 (37–41) # | 39 (37–42) # | 0.109 |
| Gravidity at time of index pregnancy | 5 (4–9) # | 3 (1–7) # | <0.001 |
| Parity at time of index pregnancy | 0 (0–2) # | 1 (0–5) # | <0.001 |
| Number of previous miscarriages | 4 (3–7) # | 0 (0–1) # | <0.001 |
| RM Women (n = 23) | Control Women (n = 44) * | OR | 95% CI | p-Value $ | |||
|---|---|---|---|---|---|---|---|
| Genotype Frequency | |||||||
| Del/Del | 4 | 17.4% | 19 | 43.2% | 0.28 | 0.08–0.95 | 0.039 |
| Ins/Del | 15 | 65.2% | 16 | 36.4% | 3.28 | 1.14–9.43 | 0.026 |
| Ins/Ins | 4 | 17.4% | 9 | 20.5% | 0.82 | 0.22–3.01 | 0.810 |
| Phenotype Frequency | |||||||
| Ins phenotype # | 19 | 82.6% | 25 | 56.8% | 3.61 | 1.05–12.38 | 0.039 |
| Del phenotype & | 19 | 82.6% | 35 | 79.6% | 1.22 | 0.33–4.50 | 0.810 |
| Allele Frequency | |||||||
| Insertion | 23 | 50.0% | 34 | 38.6% | 1.59 | 0.77–3.26 | 0.205 |
| Deletion | 23 | 50.0% | 54 | 61.4% | 0.63 | 0.31–1.29 | 0.205 |
| RM Offspring (n = 23) | Control Offspring (n = 45) * | OR | 95% CI | p-Value $ | |||
|---|---|---|---|---|---|---|---|
| Genotype Frequency | |||||||
| Del/Del | 8 | 34.8% | 16 | 34.0% | 0.97 | 1.33–2.77 | 0.969 |
| Ins/Del | 11 | 47.8% | 22 | 46.8% | 0.96 | 0.35–2.62 | 0.936 |
| Ins/Ins | 4 | 17.4% | 7 | 14.9% | 1.14 | 0.30–4.39 | 0.789 |
| Phenotype Frequency | |||||||
| Ins phenotype # | 15 | 65.2% | 29 | 61.7% | 1.03 | 0.36–2.97 | 0.969 |
| Del phenotype & | 19 | 82.6% | 38 | 84.4% | 0.88 | 0.23–3.36 | 0.789 |
| Allele Frequency | |||||||
| Insertion | 19 | 43.3% | 36 | 40.0% | 1.06 | 0.51–2.17 | 0.875 |
| Deletion | 27 | 58.7% | 54 | 60.0% | 0.95 | 0.46–1.95 | 0.875 |
| RM Women (n = 23) | Control Women (n = 44) * | OR | 95% CI | p-Value | |||
|---|---|---|---|---|---|---|---|
| Low HLA-G protein expression | 3 | 13.0% | 23 | 50.0% | 6.67 | 1.74–25.57 | 0.006 $ |
| High HLA-G protein expression | 20 | 87.0% | 23 | 50.0% | |||
| RM Women (n = 22) * | Control Women (n = 32) * | OR | 95% CI | p-Value | |||
|---|---|---|---|---|---|---|---|
| Low HLA-G mRNA expression | 3 | 13.6% | 16 | 50.0% | 6.33 | 1.56–25.71 | 0.010 $ |
| High HLA-G mRNA expression | 19 | 86.4% | 16 | 50.0% | |||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Craenmehr, M.H.C.; Nederlof, I.; Cao, M.; Drabbels, J.J.M.; Spruyt-Gerritse, M.J.; Anholts, J.D.H.; Kapsenberg, H.M.; Stegehuis, J.A.; van der Keur, C.; Fasse, E.; et al. Increased HLA-G Expression in Term Placenta of Women with a History of Recurrent Miscarriage Despite Their Genetic Predisposition to Decreased HLA-G Levels. Int. J. Mol. Sci. 2019, 20, 625. https://doi.org/10.3390/ijms20030625
Craenmehr MHC, Nederlof I, Cao M, Drabbels JJM, Spruyt-Gerritse MJ, Anholts JDH, Kapsenberg HM, Stegehuis JA, van der Keur C, Fasse E, et al. Increased HLA-G Expression in Term Placenta of Women with a History of Recurrent Miscarriage Despite Their Genetic Predisposition to Decreased HLA-G Levels. International Journal of Molecular Sciences. 2019; 20(3):625. https://doi.org/10.3390/ijms20030625
Chicago/Turabian StyleCraenmehr, Moniek H. C., Iris Nederlof, Milo Cao, Jos J. M. Drabbels, Marijke J. Spruyt-Gerritse, Jacqueline D. H. Anholts, Hanneke M. Kapsenberg, Janine A. Stegehuis, Carin van der Keur, Esther Fasse, and et al. 2019. "Increased HLA-G Expression in Term Placenta of Women with a History of Recurrent Miscarriage Despite Their Genetic Predisposition to Decreased HLA-G Levels" International Journal of Molecular Sciences 20, no. 3: 625. https://doi.org/10.3390/ijms20030625
APA StyleCraenmehr, M. H. C., Nederlof, I., Cao, M., Drabbels, J. J. M., Spruyt-Gerritse, M. J., Anholts, J. D. H., Kapsenberg, H. M., Stegehuis, J. A., van der Keur, C., Fasse, E., Haasnoot, G. W., van der Hoorn, M.-L. P., Claas, F. H. J., Heidt, S., & Eikmans, M. (2019). Increased HLA-G Expression in Term Placenta of Women with a History of Recurrent Miscarriage Despite Their Genetic Predisposition to Decreased HLA-G Levels. International Journal of Molecular Sciences, 20(3), 625. https://doi.org/10.3390/ijms20030625





