Altered Metabolic Profile and Adipocyte Insulin Resistance Mark Severe Liver Fibrosis in Patients with Chronic Liver Disease
Abstract
1. Introduction
2. Results
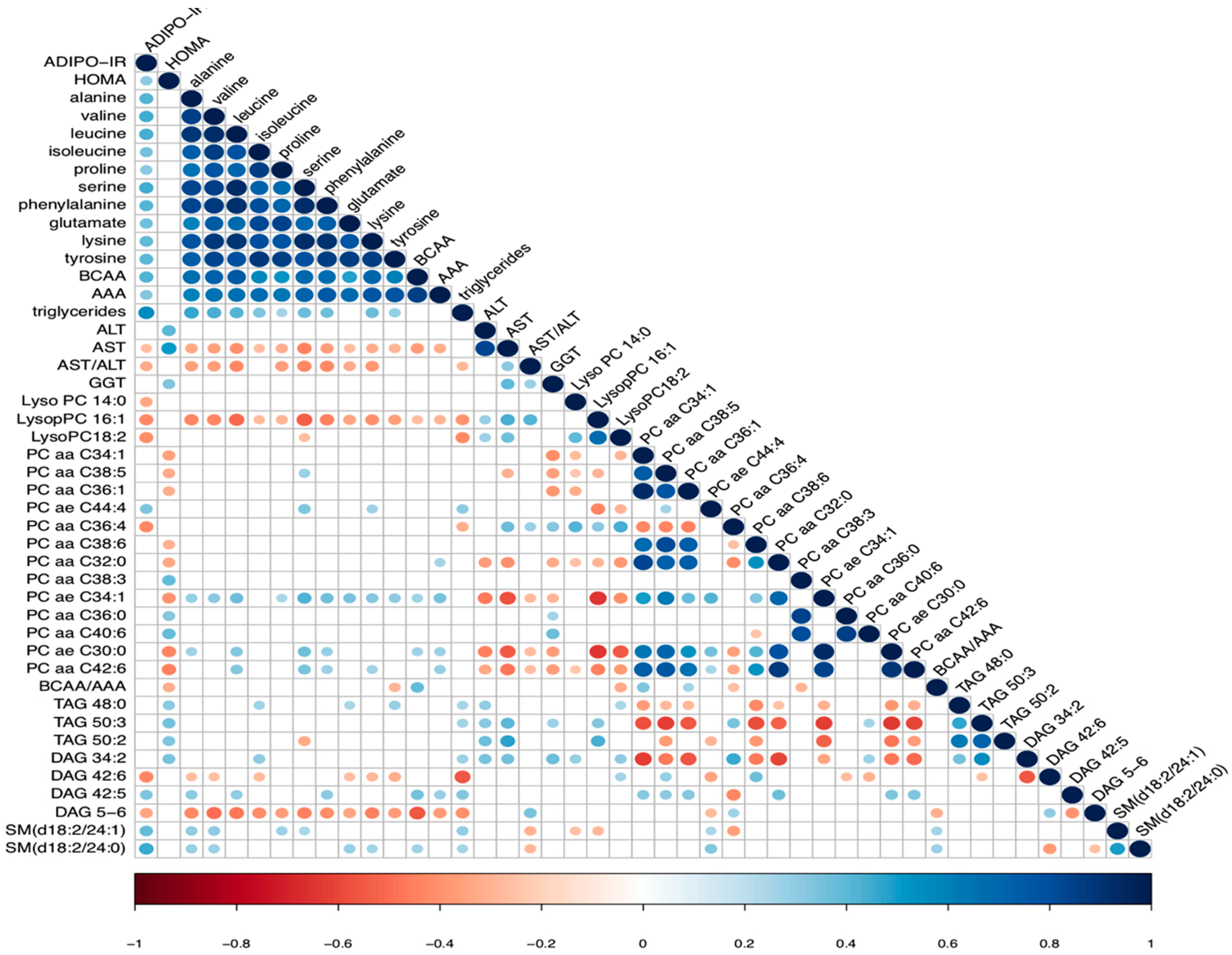
2.1. Markers of Insulin Resistance and Inflammation in CLD
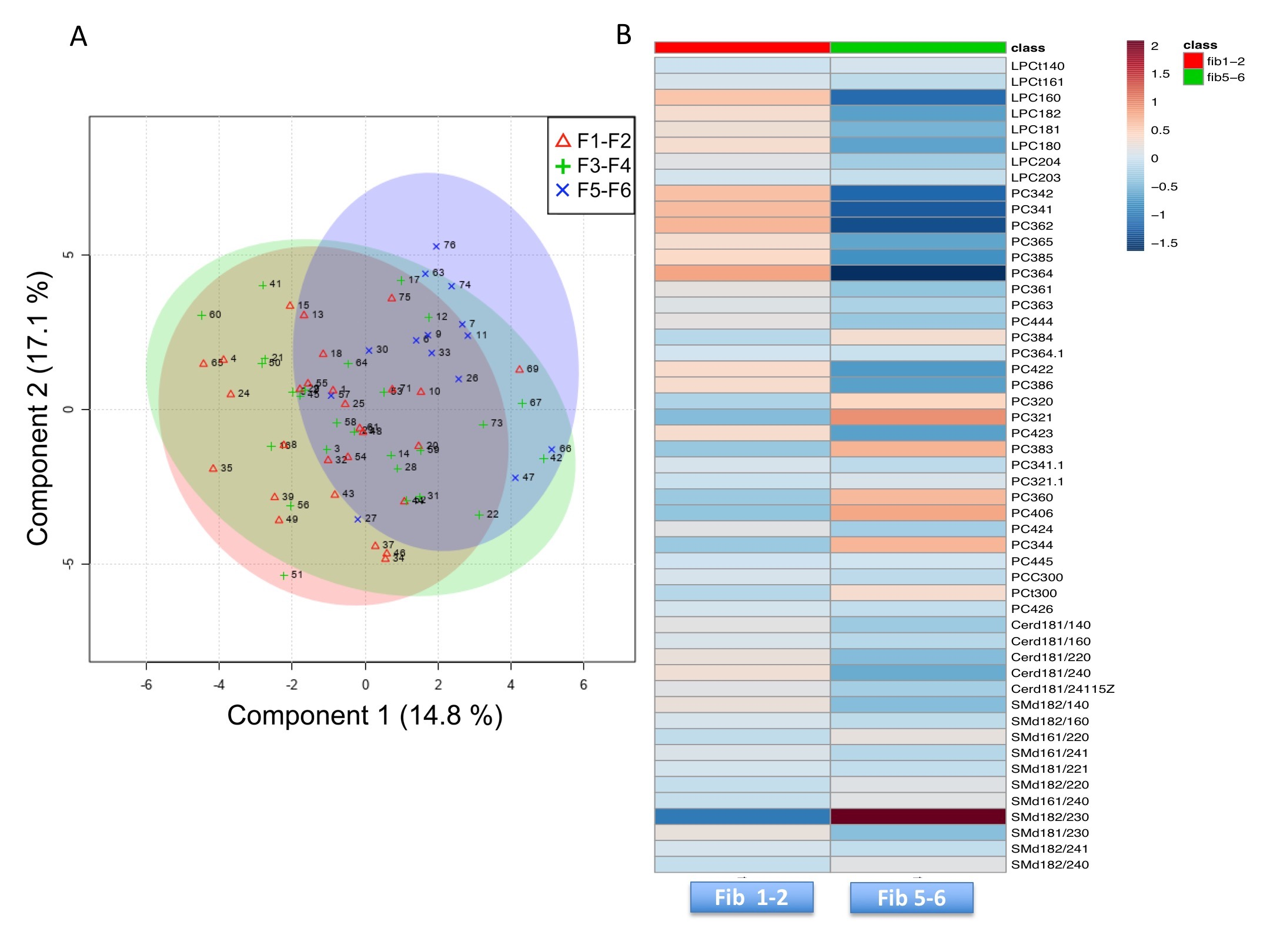
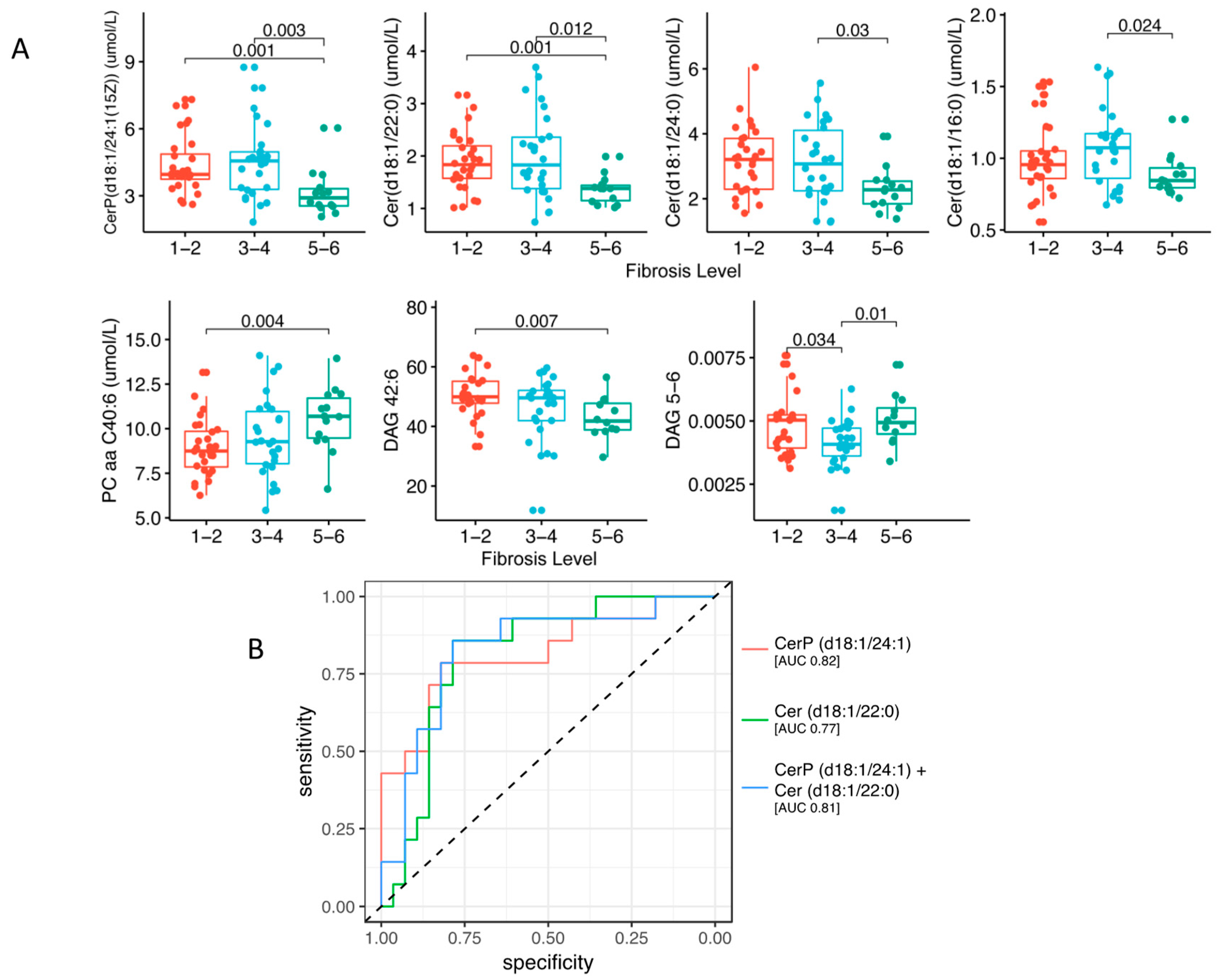
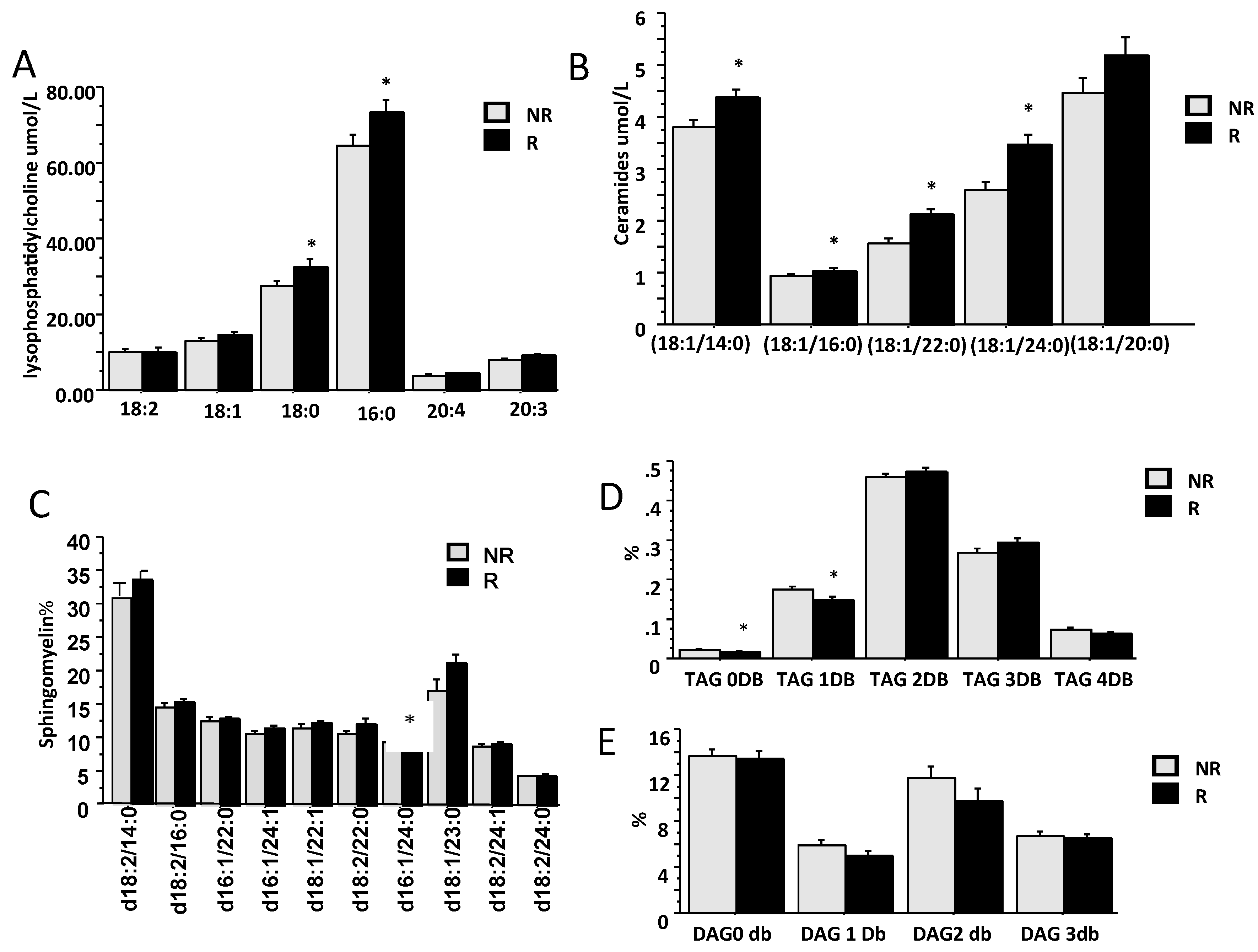
2.2. Lipidomic Profile in Relation to Fibrosis Stage and Insulin Resistance
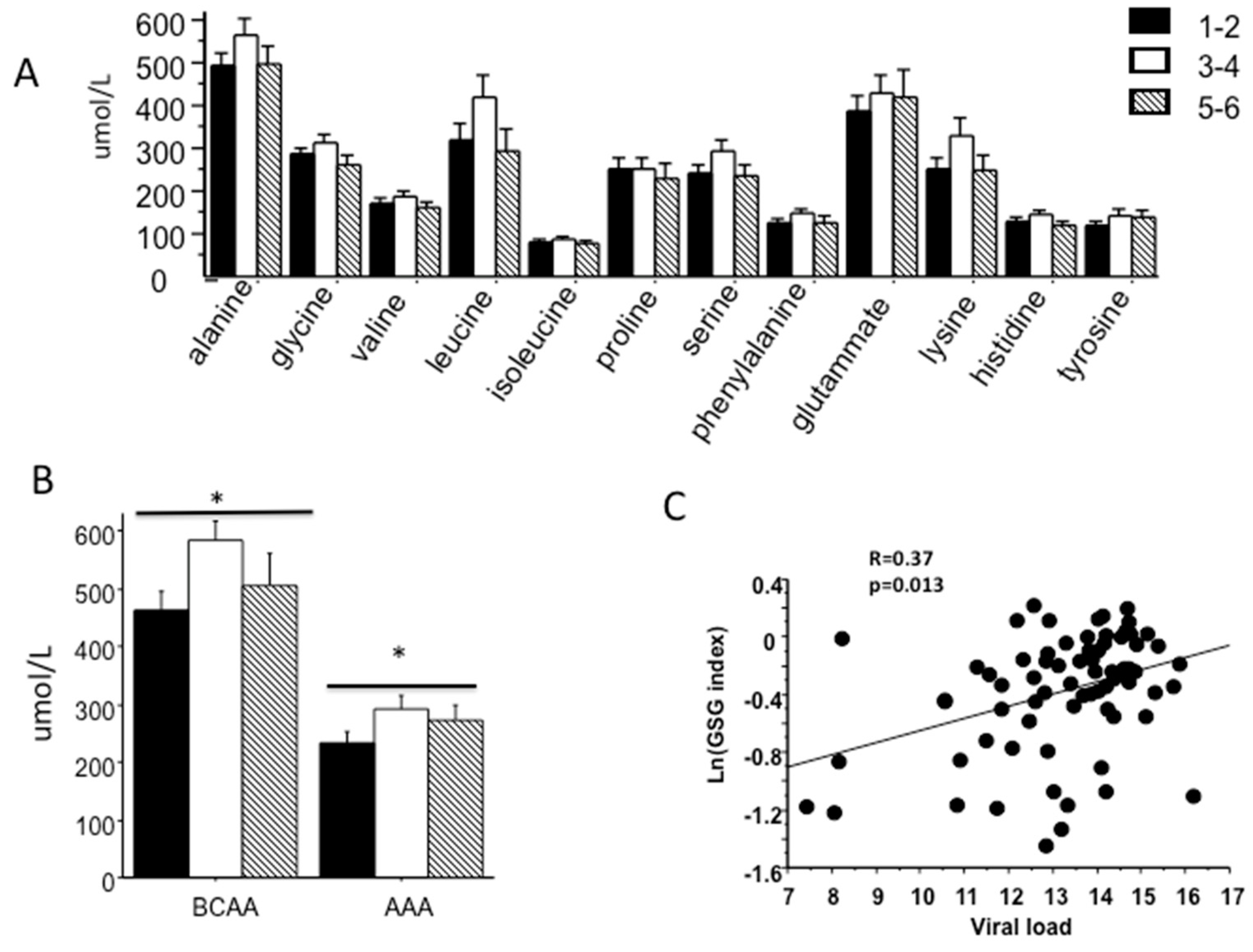
2.3. Association between Amino Acid Profile, Fibrosis Stage and Insulin Resistance
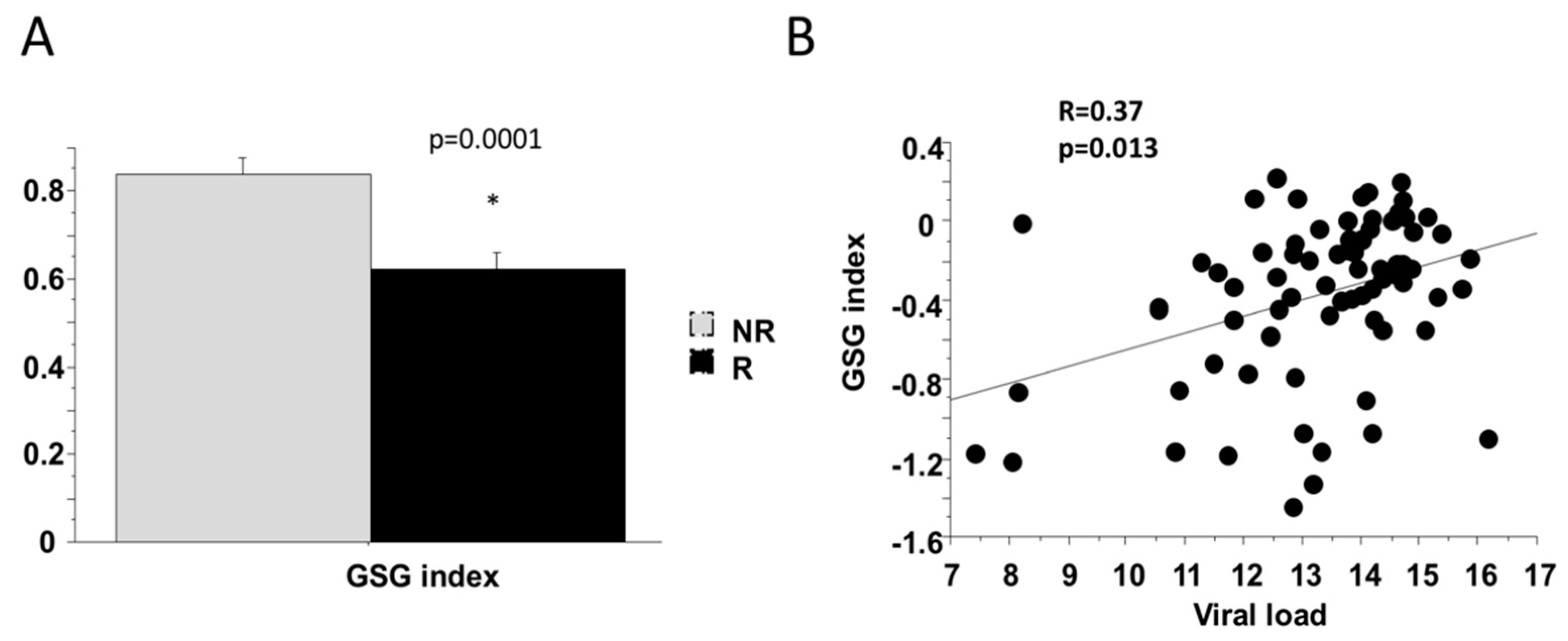
2.4. Baseline Markers of Response to Therapy (SVR)
3. Discussion
4. Materials and Methods
4.1. Lipidomic and Metabolomic Measurements
4.2. Calculation and Statistical Analysis
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| CLD | Chronic Liver Disease |
| IR | Insulin Resistance |
| DAA | Directly Acting Antivirals- |
| SVR | Sustained Virological Response |
| PEG-IFNa | Pegylated-Interferon-A |
| RBV | Ribavirin |
| GC/MS | Gas Chromatography Mass Spectrometry |
| UHPLC/MS-QTOF | Ultra-High Performance Liquid Chromatography-Quadrupole Time-Of-Flight Mass Spectrometry |
| HOMA | Homeostasis Model Assessment |
| BCAA | Branched-Chain Amino Acids |
| AAA | Aromatic Amino Acids |
| SFA | Saturated Fatty Acids |
| Cer | Ceramides |
| PC | Phosphocholines |
| LysoPC | Lysophosphtidilcholines |
| MAG | Monoacylglycerols |
| DAG | Diacylglycerols |
| TAG | Triacylglycerols |
| SM | Sphingomyelin |
References
- Lent-Schochet, D.; McLaughlin, M.; Ramakrishnan, N.; Jialal, I. Exploratory metabolomics of metabolic syndrome: A status report. World J. Diabetes 2019, 10, 23–36. [Google Scholar] [CrossRef] [PubMed]
- Peng, B.; Li, H.; Peng, X.X. Functional metabolomics: From biomarker discovery to metabolome reprogramming. Protein Cell 2015, 6, 628–637. [Google Scholar] [CrossRef] [PubMed]
- Kralj, D.; Virovic Jukic, L.; Stojsavljevic, S.; Duvnjak, M.; Smolic, M.; Curcic, I.B. Hepatitis C Virus, Insulin Resistance, and Steatosis. J. Clin. Transl. Hepatol. 2016, 4, 66–75. [Google Scholar] [CrossRef] [PubMed]
- Vanni, E.; Abate, M.L.; Gentilcore, E.; Hickman, I.; Gambino, R.; Cassader, M.; Smedile, A.; Ferrannini, E.; Rizzetto, M.; Marchesini, G.; et al. Sites and mechanisms of insulin resistance in nonobese, nondiabetic patients with chronic hepatitis C. Hepatology 2009, 50, 697–706. [Google Scholar] [CrossRef]
- Khatun, M.; Ray, R.B. Mechanisms Underlying Hepatitis C Virus-Associated Hepatic Fibrosis. Cells 2019, 8. [Google Scholar] [CrossRef]
- Bose, S.K.; Ray, R. Hepatitis C virus infection and insulin resistance. World J. Diabetes 2014, 5, 52–58. [Google Scholar] [CrossRef]
- Adinolfi, L.E.; Restivo, L.; Zampino, R.; Lonardo, A.; Loria, P. Metabolic alterations and chronic hepatitis C: Treatment strategies. Expert Opin. Pharmacother 2011, 12, 2215–2234. [Google Scholar] [CrossRef]
- Sebastiani, G.; Gkouvatsos, K.; Pantopoulos, K. Chronic hepatitis C and liver fibrosis. World J. Gastroenterol. 2014, 20, 11033–11053. [Google Scholar] [CrossRef]
- Engin, A.B. What Is Lipotoxicity? Adv. Exp. Med. Biol 2017, 960, 197–220. [Google Scholar] [CrossRef]
- Saponaro, C.; Gaggini, M.; Carli, F.; Gastaldelli, A. The Subtle Balance between Lipolysis and Lipogenesis: A Critical Point in Metabolic Homeostasis. Nutrients 2015, 7, 9453–9474. [Google Scholar] [CrossRef]
- Lim, T.R.; Hazlehurst, J.M.; Oprescu, A.I.; Armstrong, M.J.; Abdullah, S.F.; Davies, N.P.; Flintham, R.; Balfe, P.; Mutimer, D.J.; McKeating, J.A.; et al. Hepatitis C virus infection is associated with hepatic and adipose tissue insulin resistance that improves after viral cure. Clin. Endocrinol. 2019, 90, 440–448. [Google Scholar] [CrossRef] [PubMed]
- Lomonaco, R.; Ortiz-Lopez, C.; Orsak, B.; Webb, A.; Hardies, J.; Darland, C.; Finch, J.; Gastaldelli, A.; Harrison, S.; Tio, F.; et al. Effect of adipose tissue insulin resistance on metabolic parameters and liver histology in obese patients with nonalcoholic fatty liver disease. Hepatology 2012, 55, 1389–1397. [Google Scholar] [CrossRef] [PubMed]
- Rosso, C.; Kazankov, K.; Younes, R.; Esmaili, S.; Marietti, M.; Sacco, M.; Carli, F.; Gaggini, M.; Salomone, F.; Moller, H.J.; et al. Crosstalk between adipose tissue insulin resistance and liver macrophages in non-alcoholic fatty liver disease. J. Hepatol. 2019, 71, 1012–1021. [Google Scholar] [CrossRef] [PubMed]
- Bell, L.N.; Wang, J.; Muralidharan, S.; Chalasani, S.; Fullenkamp, A.M.; Wilson, L.A.; Sanyal, A.J.; Kowdley, K.V.; Neuschwander-Tetri, B.A.; Brunt, E.M.; et al. Relationship between adipose tissue insulin resistance and liver histology in nonalcoholic steatohepatitis: A pioglitazone versus vitamin E versus placebo for the treatment of nondiabetic patients with nonalcoholic steatohepatitis trial follow-up study. Hepatology 2012, 56, 1311–1318. [Google Scholar] [CrossRef] [PubMed]
- Dejong, C.H.; van de Poll, M.C.; Soeters, P.B.; Jalan, R.; Olde Damink, S.W. Aromatic amino acid metabolism during liver failure. J. Nutr. 2007, 137, 1579S–1585S. [Google Scholar] [CrossRef] [PubMed]
- Gaggini, M.; Carli, F.; Rosso, C.; Buzzigoli, E.; Marietti, M.; Della Latta, V.; Ciociaro, D.; Abate, M.L.; Gambino, R.; Cassader, M.; et al. Altered amino acid concentrations in NAFLD: Impact of obesity and insulin resistance. Hepatology 2018, 67, 145–158. [Google Scholar] [CrossRef]
- Morgan, M.Y.; Milsom, J.P.; Sherlock, S. Plasma ratio of valine, leucine and isoleucine to phenylalanine and tyrosine in liver disease. Gut 1978, 19, 1068–1073. [Google Scholar] [CrossRef]
- Chang, M.L.; Tsou, Y.K.; Hu, T.H.; Lin, C.H.; Lin, W.R.; Sung, C.M.; Chen, T.H.; Cheng, M.L.; Chang, K.C.; Chiu, C.T.; et al. Distinct patterns of the lipid alterations between genotype 1 and 2 chronic hepatitis C patients after viral clearance. PLoS ONE 2014, 9, e104783. [Google Scholar] [CrossRef]
- Masini, M.; Campani, D.; Boggi, U.; Menicagli, M.; Funel, N.; Pollera, M.; Lupi, R.; Del Guerra, S.; Bugliani, M.; Torri, S.; et al. Hepatitis C virus infection and human pancreatic beta-cell dysfunction. Diabetes Care 2005, 28, 940–941. [Google Scholar] [CrossRef][Green Version]
- Cacoub, P.; Comarmond, C.; Domont, F.; Savey, L.; Desbois, A.C.; Saadoun, D. Extrahepatic manifestations of chronic hepatitis C virus infection. Ther. Adv. Infect. Dis. 2016, 3, 3–14. [Google Scholar] [CrossRef]
- Patel, S.; Jinjuvadia, R.; Patel, R.; Liangpunsakul, S. Insulin Resistance is Associated With Significant Liver Fibrosis in Chronic Hepatitis C Patients: A Systemic Review and Meta-Analysis. J. Clin. Gastroenterol. 2016, 50, 80–84. [Google Scholar] [CrossRef] [PubMed]
- Muzzi, A.; Leandro, G.; Rubbia-Brandt, L.; James, R.; Keiser, O.; Malinverni, R.; Dufour, J.F.; Helbling, B.; Hadengue, A.; Gonvers, J.J.; et al. Insulin resistance is associated with liver fibrosis in non-diabetic chronic hepatitis C patients. J. Hepatol. 2005, 42, 41–46. [Google Scholar] [CrossRef] [PubMed]
- Saxena, N.K.; Ikeda, K.; Rockey, D.C.; Friedman, S.L.; Anania, F.A. Leptin in hepatic fibrosis: Evidence for increased collagen production in stellate cells and lean littermates of ob/ob mice. Hepatology 2002, 35, 762–771. [Google Scholar] [CrossRef] [PubMed]
- Grasso, A.; Malfatti, F.; Andraghetti, G.; Marenco, S.; Mazzucchelli, C.; Labanca, S.; Cordera, R.; Testa, R.; Picciotto, A. HOMA, BMI, and Serum Leptin Levels Variations during Antiviral Treatment Suggest Virus-Related Insulin Resistance in Noncirrhotic, Nonobese, and Nondiabetic Chronic Hepatitis C Genotype 1 Patients. Gastroenterol Res. Pract. 2015, 2015, 975695. [Google Scholar] [CrossRef] [PubMed]
- Gulbins, E.; Li, P.L. Physiological and pathophysiological aspects of ceramide. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2006, 290, R11–R26. [Google Scholar] [CrossRef] [PubMed]
- Aizaki, H.; Morikawa, K.; Fukasawa, M.; Hara, H.; Inoue, Y.; Tani, H.; Saito, K.; Nishijima, M.; Hanada, K.; Matsuura, Y.; et al. Critical role of virion-associated cholesterol and sphingolipid in hepatitis C virus infection. J. Virol. 2008, 82, 5715–5724. [Google Scholar] [CrossRef] [PubMed]
- Osawa, Y.; Uchinami, H.; Bielawski, J.; Schwabe, R.F.; Hannun, Y.A.; Brenner, D.A. Roles for C16-ceramide and sphingosine 1-phosphate in regulating hepatocyte apoptosis in response to tumor necrosis factor-alpha. J. Biol. Chem. 2005, 280, 27879–27887. [Google Scholar] [CrossRef]
- Hartmann, D.; Lucks, J.; Fuchs, S.; Schiffmann, S.; Schreiber, Y.; Ferreiros, N.; Merkens, J.; Marschalek, R.; Geisslinger, G.; Grosch, S. Long chain ceramides and very long chain ceramides have opposite effects on human breast and colon cancer cell growth. Int. J. Biochem. Cell Biol. 2012, 44, 620–628. [Google Scholar] [CrossRef]
- Mesicek, J.; Lee, H.; Feldman, T.; Jiang, X.; Skobeleva, A.; Berdyshev, E.V.; Haimovitz-Friedman, A.; Fuks, Z.; Kolesnick, R. Ceramide synthases 2, 5, and 6 confer distinct roles in radiation-induced apoptosis in HeLa cells. Cell Signal. 2010, 22, 1300–1307. [Google Scholar] [CrossRef]
- Clore, J.N.; Harris, P.A.; Li, J.; Azzam, A.; Gill, R.; Zuelzer, W.; Rizzo, W.B.; Blackard, W.G. Changes in phosphatidylcholine fatty acid composition are associated with altered skeletal muscle insulin responsiveness in normal man. Metabolism 2000, 49, 232–238. [Google Scholar] [CrossRef]
- Hagenfeldt, L.; Eriksson, L.S.; Wahren, J. Amino acids in liver disease. Proc. Nutr Soc. 1983, 42, 497–506. [Google Scholar] [CrossRef] [PubMed]
- Morgan, M.Y.; Marshall, A.W.; Milsom, J.P.; Sherlock, S. Plasma amino-acid patterns in liver disease. Gut 1982, 23, 362–370. [Google Scholar] [CrossRef] [PubMed]
- Petta, S.; Rosso, C.; Leung, R.; Abate, M.L.; Booth, D.; Salomone, F.; Gambino, R.; Rizzetto, M.; Caviglia, P.; Smedile, A.; et al. Effects of IL28B rs12979860 CC genotype on metabolic profile and sustained virologic response in patients with genotype 1 chronic hepatitis C. Clin. Gastroenterol. Hepatol. 2013, 11, 311–317. [Google Scholar] [CrossRef] [PubMed]
- Ishak, K.; Baptista, A.; Bianchi, L.; Callea, F.; De Groote, J.; Gudat, F.; Denk, H.; Desmet, V.; Korb, G.; MacSween, R.N.; et al. Histological grading and staging of chronic hepatitis. J. Hepatol. 1995, 22, 696–699. [Google Scholar] [CrossRef]
- Isokuortti, E.; Zhou, Y.; Peltonen, M.; Bugianesi, E.; Clement, K.; Bonnefont-Rousselot, D.; Lacorte, J.M.; Gastaldelli, A.; Schuppan, D.; Schattenberg, J.M.; et al. Use of HOMA-IR to diagnose non-alcoholic fatty liver disease: A population-based and inter-laboratory study. Diabetologia 2017, 60, 1873–1882. [Google Scholar] [CrossRef]
- Gastaldelli, A.; Gaggini, M.; DeFronzo, R.A. Role of Adipose Tissue Insulin Resistance in the Natural History of Type 2 Diabetes: Results From the San Antonio Metabolism Study. Diabetes 2017, 66, 815–822. [Google Scholar] [CrossRef]
- Chong, J.; Xia, J. MetaboAnalystR: An R package for flexible and reproducible analysis of metabolomics data. Bioinformatics 2018, 34, 4313–4314. [Google Scholar] [CrossRef]
| Variable | F1–F2 | F3–F4 | F5–F6 | p Value |
|---|---|---|---|---|
| Gender (M/F) | 15/14 | 11/15 | 6/6 | ns |
| Age (y) | 45.5 ± 2.14 | 48.3 ± 2.3 | 52.5 ± 3.1 | 0.026 |
| BMI | 23.2 ± 0.6 | 23.8 ± 0.6 | 24.7 ± 1.3 | ns |
| Cholesterol (mg/dL) | 169.2 ± 7 | 169.3 ± 8.4 | 161.1 ± 7.7 | ns |
| LDL (mg/dL) | 102.5 ± 4.9 | 101.4 ± 6.6 | 97.5 ± 6.8 | ns |
| HDL (mg/dL) | 41.9 ± 2.6 | 38.7 ± 3.7 | 39.5 ± 4.3 | ns |
| Triglycerides(mg/dL) | 123.7 ± 9.9 | 146.2 ± 17.5 | 120.6 ± 13 | ns |
| AST (mU/L) | 8.0 ± 0.9 | 6.2 ± 0.7 | 8.9 ± 1.3 | ns |
| ALT (mU/L) | 9.3 ± 1.1 | 7.7 ± 0.9 | 8.2 ± 1.13 | ns |
| GGT (mU/L) | 38 ± 3.6 | 31 ± 4.4 | 58.6 ± 10.5 | 0.021 |
| Glucose (mg/dL) | 67.8 ± 2.3 | 68.2 ± 3.2 | 76.4 ± 5.2 | ns |
| Insulin ulU/mL | 11.9 ± 0.9 | 12.8 ± 1.3 | 16.1 ± 1.8 | ns |
| MCP-1 pg/mL | 223.9 ± 16 | 218.8 ± 26. | 188.4 ± 22.2 | ns |
| Leptin pg/mL | 3000 ± 632 | 3149 ± 547 | 65,340 ± 2345 | ns |
| HOMA-IR | 36.6 ± 1.2 | 38.7 ± 3.8 | 54.6 ± 6.3 | 0.024 |
| Adipo-IR | 10.7 ± 1.2 | 18.1 ± 2.4 | 18.8 ± 3.2 | 0.042 |
| Gender (M/F) | 15/14 | 11/15 | 6/6 | ns |
| Age (y) | 45.5 ± 2.14 | 48.3 ± 2.3 | 52.5 ± 3.1 | 0.026 |
| BMI | 23.2 ± 0.6 | 23.8 ± 0.6 | 24.7 ± 1.3 | ns |
| Cholesterol (mg/dL) | 169.2 ± 7 | 169.3 ± 8.4 | 161.1 ± 7.7 | ns |
| LDL (mg/dL) | 102.5 ± 4.9 | 101.4 ± 6.6 | 97.5 ± 6.8 | ns |
| HDL (mg/dL) | 41.9 ± 2.6 | 38.7 ± 3.7 | 39.5 ± 4.3 | ns |
| Triglycerides(mg/dL) | 123.7 ± 9.9 | 146.2 ± 17.5 | 120.6 ± 13 | ns |
| AST (mU/L) | 8.0 ± 0.9 | 6.2 ± 0.7 | 8.9 ± 1.3 | ns |
| ALT (mU/L) | 9.3 ± 1.1 | 7.7 ± 0.9 | 8.2 ± 1.13 | ns |
| GGT (mU/L) | 38 ± 3.6 | 31 ± 4.4 | 58.6 ± 10.5 | 0.021 |
| Glucose (mg/dL) | 67.8 ± 2.3 | 68.2 ± 3.2 | 76.4 ± 5.2 | ns |
| Insulin ulU/mL | 11.9 ± 0.9 | 12.8 ± 1.3 | 16.1 ± 1.8 | ns |
| MCP-1 pg/mL | 223.9 ± 16 | 218.8 ± 26. | 188.4 ± 22.2 | ns |
| Leptin pg/mL | 3000 ± 632 | 3149 ± 547 | 65,340 ± 2345 | ns |
| HOMA-IR | 36.6 ± 1.2 | 38.7 ± 3.8 | 54.6 ± 6.3 | 0.024 |
| Adipo-IR | 10.7 ± 1.2 | 18.1 ± 2.4 | 18.8 ± 3.2 | 0.042 |
| Amino Acids mmol/L | Non Responders | Responders | NR vs R (p Value) | Viral Load (R) |
|---|---|---|---|---|
| Alanine | 517.38 ± 25.4 | 508.10 ± 29.88 | ns | ns |
| Phenylalanine | 132.73 ± 7.65 | 129.78 ± 9.6 | ns | |
| Glycine | 287.4 ± 12.6 | 289.13 ± 15.62 | ns | |
| Glutamate | 455.5 ± 29.08 | 346.12 ± 33.52 | 0.006 | R = 0.38 |
| Histidine | 131.75 ± 16.57 | 133.92 ± 8.12 | ns | |
| Leucine | 348.1 ± 34.73 | 323.98 ± 40.71 | ns | |
| Isoleucine | 83.68 ± 3.82 | 78.53 ± 5.41 | ns | R = 0.25 |
| Lysine | 282.2 ± 22.08 | 262.76 ± 31.81 | ns | R = 0.23 |
| Proline | 275.13 ± 21.44 | 213.46 ± 20.74 | 0.04 | R = 0.34 |
| Serine | 259.5 ± 15.53 | 247.81 ± 20.47 | ns | |
| Tyrosine | 137 ± 8.58 | 120.27 ± 11.14 | ns | R = 0.27 |
| Valine | 174.81 ± 8.9 | 169. ± 10.7 | ns | |
| BCAAs | 528.92 ± 31.25 | 496.7 ± 31.1 | ns | R = 0.32 |
| AAAs | 258.26 ± 13.92 | 258.33 ± 21.17 | ns | R = 0.32 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gaggini, M.; Carli, F.; Rosso, C.; Younes, R.; D’Aurizio, R.; Bugianesi, E.; Gastaldelli, A. Altered Metabolic Profile and Adipocyte Insulin Resistance Mark Severe Liver Fibrosis in Patients with Chronic Liver Disease. Int. J. Mol. Sci. 2019, 20, 6333. https://doi.org/10.3390/ijms20246333
Gaggini M, Carli F, Rosso C, Younes R, D’Aurizio R, Bugianesi E, Gastaldelli A. Altered Metabolic Profile and Adipocyte Insulin Resistance Mark Severe Liver Fibrosis in Patients with Chronic Liver Disease. International Journal of Molecular Sciences. 2019; 20(24):6333. https://doi.org/10.3390/ijms20246333
Chicago/Turabian StyleGaggini, Melania, Fabrizia Carli, Chiara Rosso, Ramy Younes, Romina D’Aurizio, Elisabetta Bugianesi, and Amalia Gastaldelli. 2019. "Altered Metabolic Profile and Adipocyte Insulin Resistance Mark Severe Liver Fibrosis in Patients with Chronic Liver Disease" International Journal of Molecular Sciences 20, no. 24: 6333. https://doi.org/10.3390/ijms20246333
APA StyleGaggini, M., Carli, F., Rosso, C., Younes, R., D’Aurizio, R., Bugianesi, E., & Gastaldelli, A. (2019). Altered Metabolic Profile and Adipocyte Insulin Resistance Mark Severe Liver Fibrosis in Patients with Chronic Liver Disease. International Journal of Molecular Sciences, 20(24), 6333. https://doi.org/10.3390/ijms20246333







