Comparing and Contrasting the Multiple Roles of Butenolide Plant Growth Regulators: Strigolactones and Karrikins in Plant Development and Adaptation to Abiotic Stresses
Abstract
1. Introduction
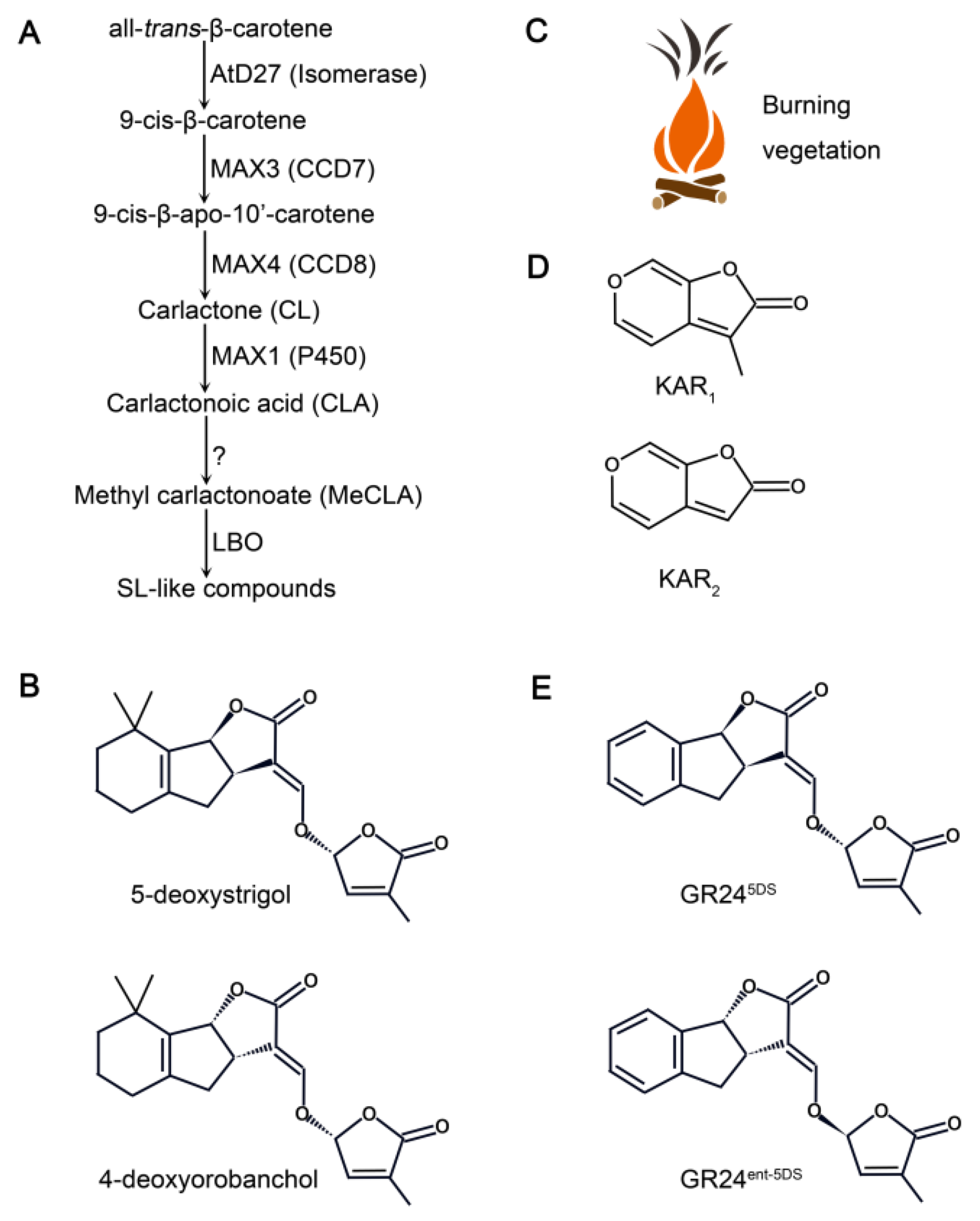
2. Classical Structure and Biosynthesis of SLs and KARs
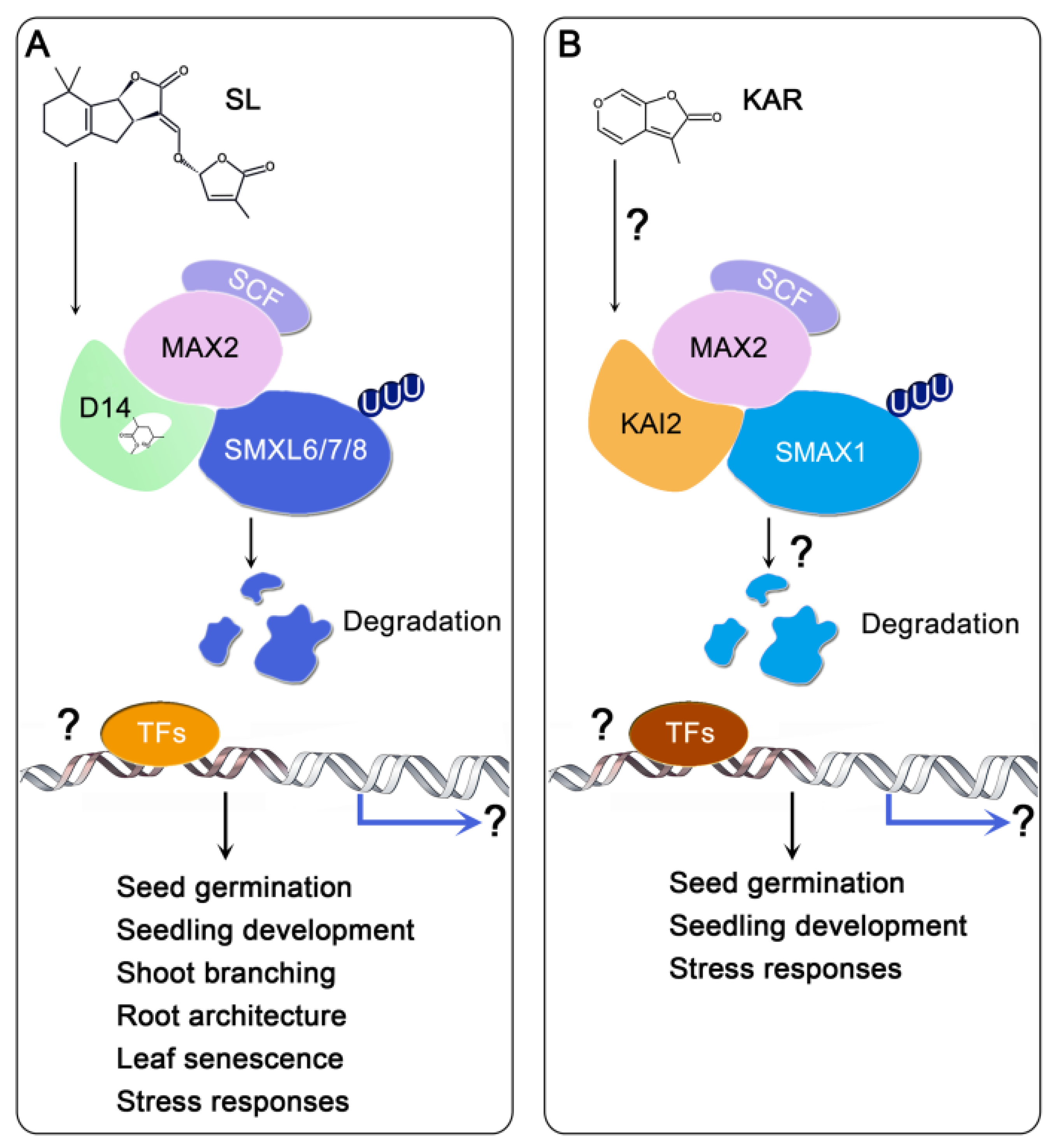
3. Signal Transductions of SL and KAR
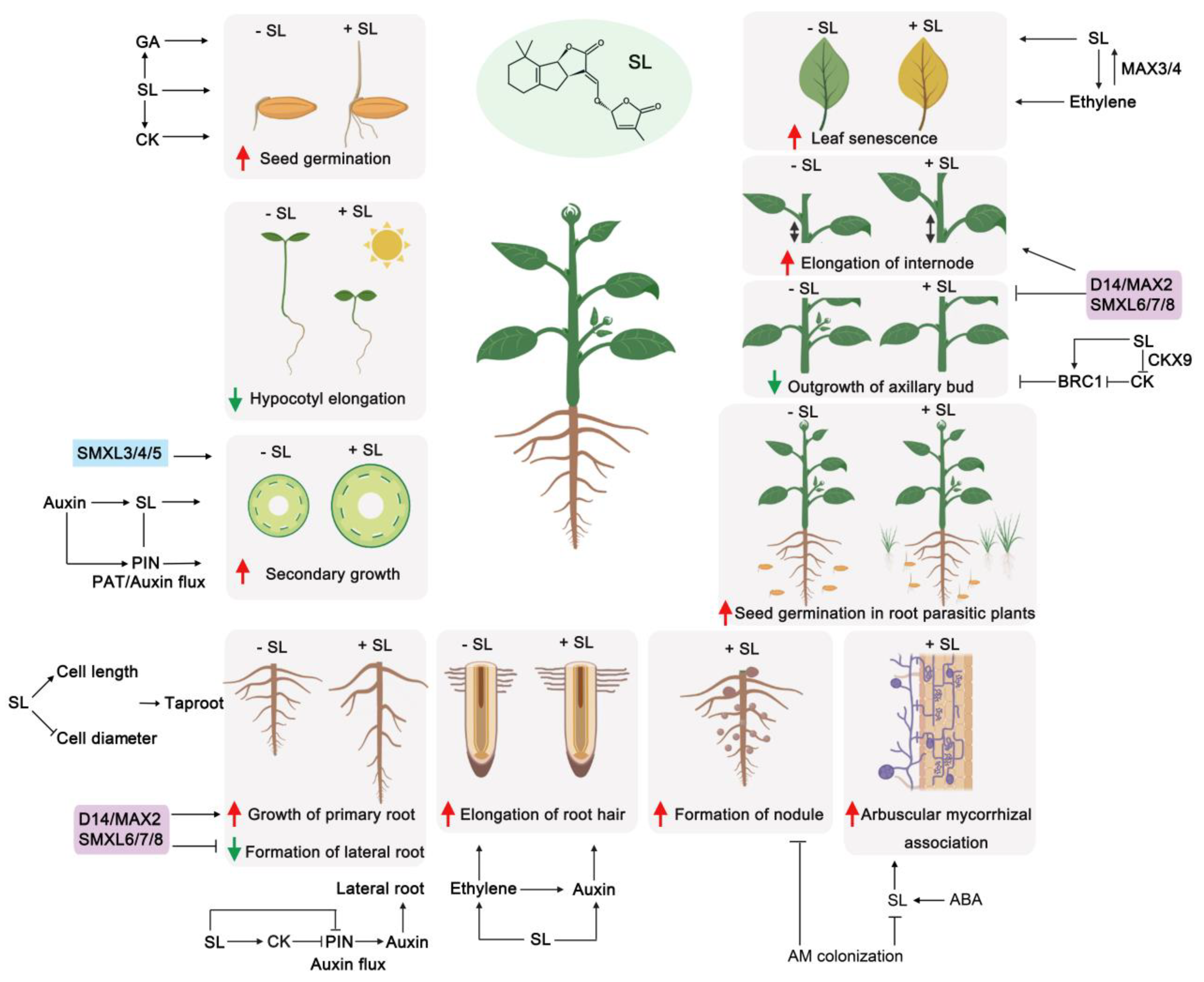
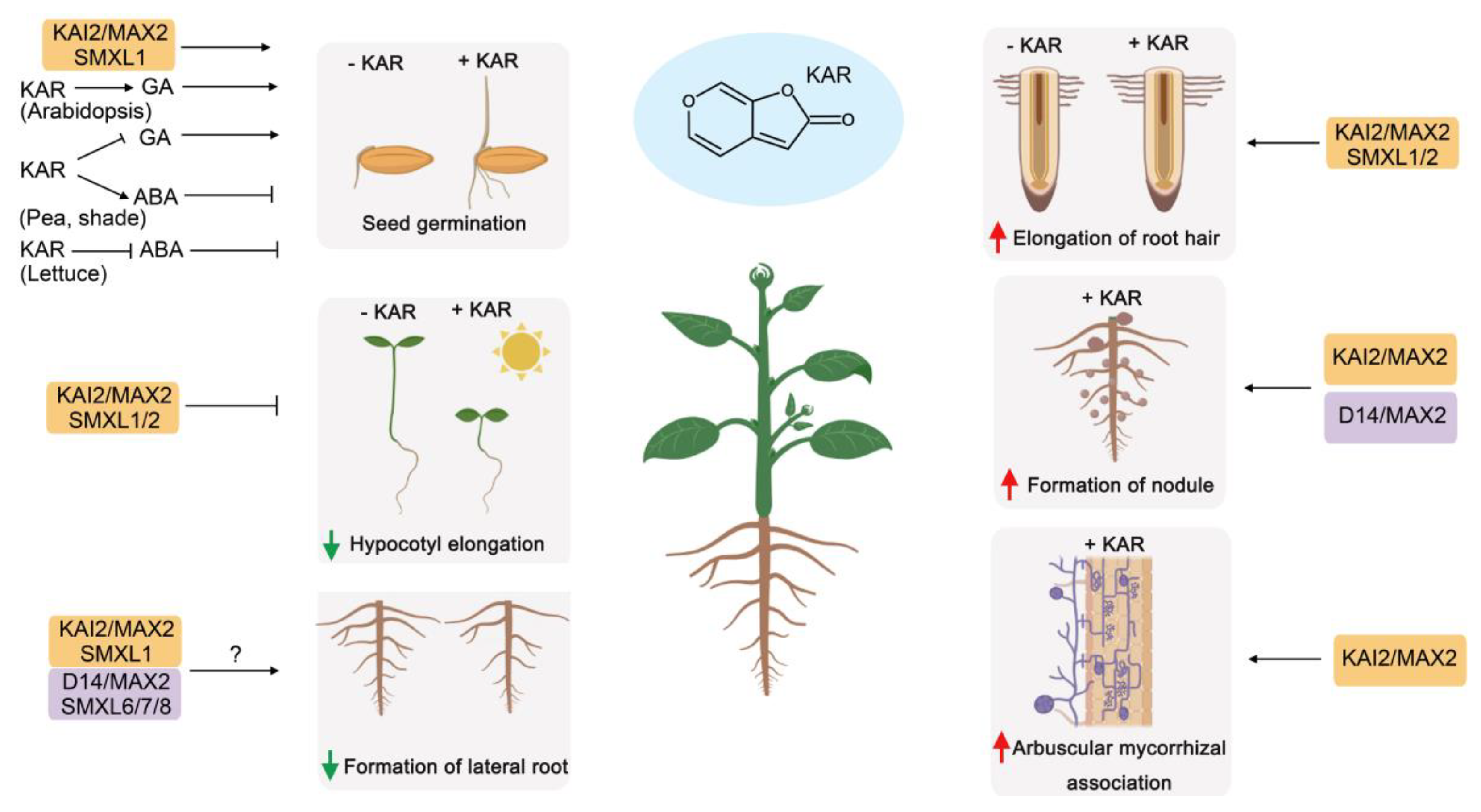
4. Similarities and Differences Between the Functions of SLs and KARs in Plant Development
4.1. Utilization of Storage Reserves in the Early Seed Germination
4.2. Seed Germination
4.3. Leaf Morphogenesis
4.4. Shoot Branching
4.5. Root System Development
4.6. Mycorrhizal Symbiosis
5. Hormone Interactions During SL- or KAR-Mediated Plant Development
5.1. SL or KAR Crosstalk with Auxin
5.2. SL or KAR Crosstalk with Gibberellin
5.3. SL or KAR Crosstalk with Abscisic Acid
5.4. SL or KAR Crosstalk with Ethylene
5.5. SL or KAR Crosstalk with Cytokinin
5.6. SL or KAR Crosstalk with Other Hormones
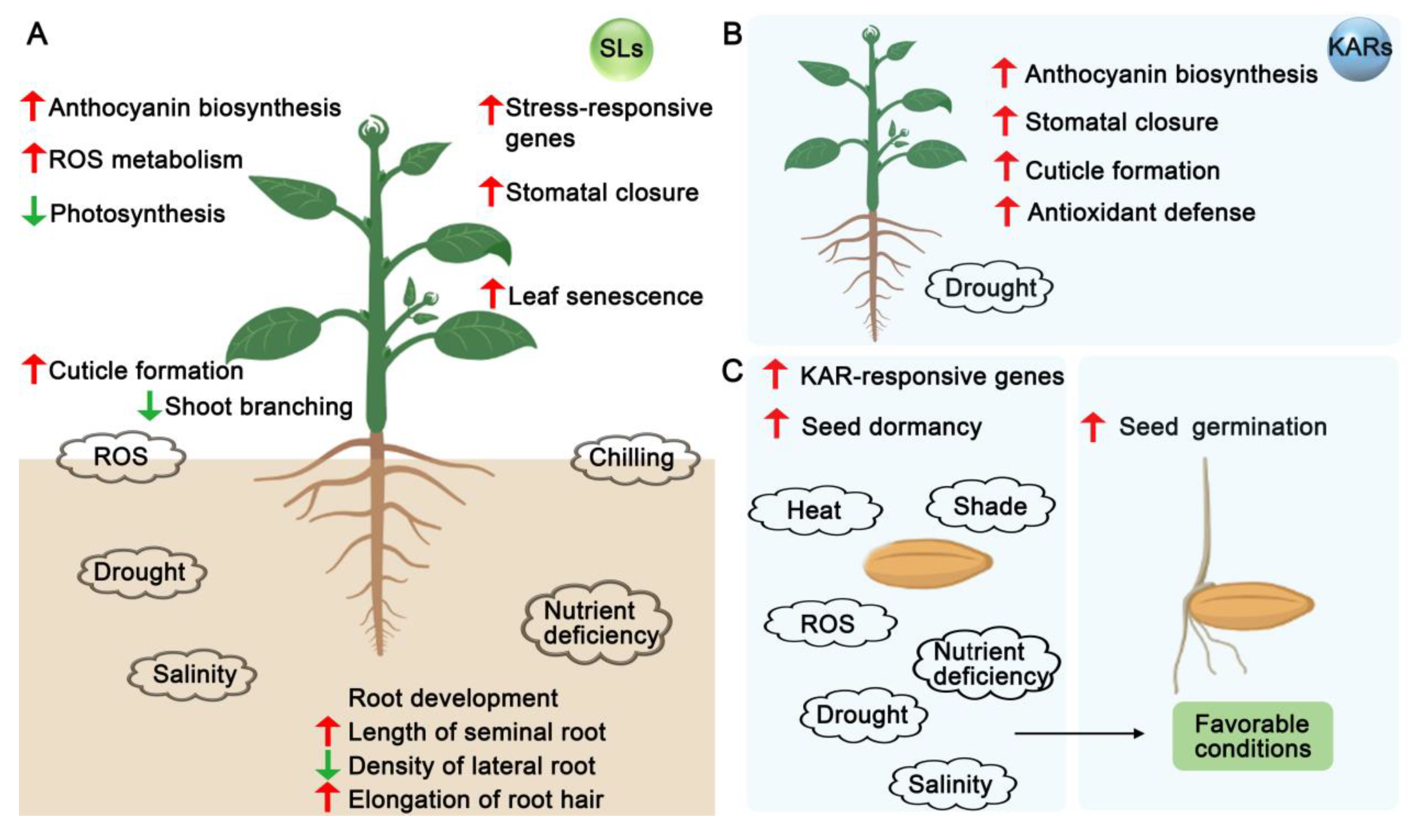
6. SLs and KARs During Abiotic Stress: Responses and Adaptation
6.1. Dynamic Regulation of SLs Under Abiotic Stresses
6.2. SL- and KAR-Mediated Plant Adaptation to Abiotic Stresses
7. SLs and KARs Crosstalk with Other Phytohormones Under Abiotic Stresses
8. Conclusions and Perspectives
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| SL | Strigolactone |
| KAR | Karrikin |
| ABA | Abscisic acid |
| CK | Cytokinin |
| GA | Gibberellic acid |
| ET | Ethylene |
| SA | Salicylic acid |
| JA | Jasmonate |
| BR | Brassinosteroid |
| D10 | DWARF10 |
| D14 | DWARF14 |
| D27 | DWARF27 |
| D53 | DWARF53 |
| KAI2 | KARRIKIN INSENSITIVE 2 |
| MAX2 | MORE AXILLARY GROWTH 2 |
| SMAX1 | SUPPRESSOR OF MAX2 1 |
| SMXL | SMAX1-LIKE |
| LBO | LATERAL BRANCHING OXIDOREDUCTASE |
| CCD | CAROTENOID CLEAVAGE DIOXYGENASE |
| RMS | Ramosus |
| N | Nitrogen |
| P | Phosphate |
| CKX | CYTOKININ OXIDASE |
| CL | Carlactone |
| AMF | Arbuscular mycorrhizal fungus |
| TMB | Trimethylbutenolide |
| SW | Smoke-water |
References
- AbuQamar, S.; Moustafa, K.; Tran, L.S.P. Mechanisms and strategies of plant defense against Botrytis cinerea. Crit. Rev. Biotechnol. 2017, 37, 262–274. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, N.; Rivero, R.M.; Shulaev, V.; Blumwald, E.; Mittler, R. Abiotic and biotic stress combinations. New Phytol. 2014, 203, 32–43. [Google Scholar] [CrossRef] [PubMed]
- Zhu, J.K. Abiotic Stress Signaling and Responses in Plants. Cell 2016, 167, 313–324. [Google Scholar] [CrossRef] [PubMed]
- Lesk, C.; Rowhani, P.; Ramankutty, N. Influence of extreme weather disasters on global crop production. Nature 2016, 529, 84–87. [Google Scholar] [CrossRef]
- Boyer, J.S. Plant Productivity and Environment. Science 1982, 218, 443–448. [Google Scholar] [CrossRef]
- Abdelrahman, M.; Jogaiah, S.; Burritt, D.J.; Tran, L.S.P. Legume genetic resources and transcriptome dynamics under abiotic stress conditions. Plant Cell Environ. 2018, 41, 1972–1983. [Google Scholar] [CrossRef]
- Li, W.Q.; Nguyen, K.H.; Chu, H.D.; Ha, C.V.; Watanabe, Y.; Osakabe, Y.; Leyva-Gonzalez, M.A.; Sato, M.; Toyooka, K.; Voges, L.; et al. The karrikin receptor KAI2 promotes drought resistance in Arabidopsis thaliana. PLoS Genet. 2017, 13. [Google Scholar] [CrossRef]
- Kim, J.M.; To, T.K.; Matsui, A.; Tanoi, K.; Kobayashi, N.I.; Matsuda, F.; Habu, Y.; Ogawa, D.; Sakamoto, T.; Matsunaga, S.; et al. Acetate-mediated novel survival strategy against drought in plants. Nat. Plants 2017, 3. [Google Scholar] [CrossRef]
- Xiong, L.M.; Schumaker, K.S.; Zhu, J.K. Cell signaling during cold, drought, and salt stress. Plant Cell 2002, 14, S165–S183. [Google Scholar] [CrossRef]
- Fabregas, N.; Lozano-Elena, F.; Blasco-Escamez, D.; Tohge, T.; Martinez-Andujar, C.; Albacete, A.; Osorio, S.; Bustamante, M.; Riechmann, J.L.; Nomura, T.; et al. Overexpression of the vascular brassinosteroid receptor BRL3 confers drought resistance without penalizing plant growth. Nat. Commun. 2018, 9. [Google Scholar] [CrossRef]
- Zhang, P.; Fan, Y.; Sun, X.P.; Chen, L.; Terzaghi, W.; Bucher, E.; Li, L.; Dai, M.Q. A large-scale circular RNA profiling reveals universal molecular mechanisms responsive to drought stress in maize and Arabidopsis. Plant J. 2019, 98, 697–713. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.Z.; Hou, L.Y.; Meng, J.J.; You, H.W.; Li, Z.; Gong, Z.Z.; Yang, S.H.; Shi, Y.T. The Antagonistic Action of Abscisic Acid and Cytokinin Signaling Mediates Drought Stress Response in Arabidopsis. Mol. Plant 2018, 11, 970–982. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.; Li, B.S.; Zheng, X.Y.; Li, J.G.; Yang, M.; Dong, X.N.; He, G.M.; An, C.C.; Deng, X.W. Salicylic acid biosynthesis is enhanced and contributes to increased biotrophic pathogen resistance in Arabidopsis hybrids. Nat. Commun. 2015, 6. [Google Scholar] [CrossRef]
- Chen, J.N.; Nolan, T.M.; Ye, H.X.; Zhang, M.C.; Tong, H.N.; Xin, P.Y.; Chu, J.F.; Chu, C.C.; Li, Z.H.; Yin, Y.H. Arabidopsis WRKY46, WRKY54, and WRKY70 Transcription Factors Are Involved in Brassinosteroid-Regulated Plant Growth and Drought Responses. Plant Cell 2017, 29, 1425–1439. [Google Scholar] [CrossRef]
- Ahmad, P.; Rasool, S.; Gul, A.; Sheikh, S.A.; Akram, N.A.; Ashraf, M.; Kazi, A.M.; Gucel, S. Jasmonates: Multifunctional Roles in Stress Tolerance. Front. Plant Sci. 2016, 7. [Google Scholar] [CrossRef]
- Wasternack, C.; Hause, B. Jasmonates: Biosynthesis, perception, signal transduction and action in plant stress response, growth and development. An update to the 2007 review in Annals of Botany. Ann. Bot.-Lond. 2013, 111, 1021–1058. [Google Scholar] [CrossRef]
- Choudhary, S.P.; Yu, J.Q.; Yamaguchi-Shinozaki, K.; Shinozaki, K.; Tran, L.S.P. Benefits of brassinosteroid crosstalk. Trends Plant Sci. 2012, 17, 594–605. [Google Scholar] [CrossRef]
- Wani, S.H.; Kumar, V.; Shriram, V.; Sah, S.K. Phytohormones and their metabolic engineering for abiotic stress tolerance in crop plants. Crop J. 2016, 4, 162–176. [Google Scholar] [CrossRef]
- Ha, S.; Vankova, R.; Yamaguchi-Shinozaki, K.; Shinozaki, K.; Tran, L.S.P. Cytokinins: Metabolism and function in plant adaptation to environmental stresses. Trends Plant Sci. 2012, 17, 172–179. [Google Scholar] [CrossRef]
- Peleg, Z.; Blumwald, E. Hormone balance and abiotic stress tolerance in crop plants. Curr. Opin. Plant Biol. 2011, 14, 290–295. [Google Scholar] [CrossRef]
- Pieterse, C.M.J.; Leon-Reyes, A.; Van der Ent, S.; Van Wees, S.C.M. Networking by small-molecule hormones in plant immunity. Nat. Chem. Biol. 2009, 5, 308–316. [Google Scholar] [CrossRef]
- Robert-Seilaniantz, A.; Grant, M.; Jones, J.D.G. Hormone Crosstalk in Plant Disease and Defense: More Than Just JASMONATE-SALICYLATE Antagonism. Annu. Rev. Phytopathol. 2011, 49, 317–343. [Google Scholar] [CrossRef]
- Santner, A.; Estelle, M. Recent advances and emerging trends in plant hormone signalling. Nature 2009, 459, 1071–1078. [Google Scholar] [CrossRef]
- Netzly, D.H.; Riopel, J.L.; Ejeta, G.; Butler, L.G. Germination Stimulants of Witchweed (Striga-Asiatica) From Hydrophobic Root Exudate of Sorghum (Sorghum-Bicolor). Weed Sci. 1988, 36, 441–446. [Google Scholar] [CrossRef]
- Xie, X.N.; Yoneyama, K.; Yoneyama, K. The Strigolactone Story. Annu. Rev. Phytopathol. 2010, 48, 93–117. [Google Scholar] [CrossRef]
- Spallek, T.; Mutuku, M.; Shirasu, K. The genus Striga: A witch profile. Mol. Plant Pathol. 2013, 14, 861–869. [Google Scholar] [CrossRef]
- Stanga, J.P.; Smith, S.M.; Briggs, W.R.; Nelson, D.C. SUPPRESSOR OF MORE AXILLARY GROWTH2 1 Controls Seed Germination and Seedling Development in Arabidopsis. Plant Physiol. 2013, 163, 318–330. [Google Scholar] [CrossRef]
- Umehara, M.; Hanada, A.; Yoshida, S.; Akiyama, K.; Arite, T.; Takeda-Kamiya, N.; Magome, H.; Kamiya, Y.; Shirasu, K.; Yoneyama, K.; et al. Inhibition of shoot branching by new terpenoid plant hormones. Nature 2008, 455, 195–200. [Google Scholar] [CrossRef]
- Jiang, L.; Liu, X.; Xiong, G.S.; Liu, H.H.; Chen, F.L.; Wang, L.; Meng, X.B.; Liu, G.F.; Yu, H.; Yuan, Y.D.; et al. DWARF 53 acts as a repressor of strigolactone signalling in rice. Nature 2013, 504, 401–405. [Google Scholar] [CrossRef]
- Chevalier, F.; Nieminen, K.; Sanchez-Ferrero, J.C.; Rodriguez, M.L.; Chagoyen, M.; Hardtke, C.S.; Cubas, P. Strigolactone Promotes Degradation of DWARF14, an alpha/beta Hydrolase Essential for Strigolactone Signaling in Arabidopsis. Plant Cell 2014, 26, 1134–1150. [Google Scholar] [CrossRef]
- Wang, L.; Wang, B.; Jiang, L.; Liu, X.; Li, X.L.; Lu, Z.F.; Meng, X.B.; Wang, Y.H.; Smith, S.M.; Li, J.Y. Strigolactone Signaling in Arabidopsis Regulates Shoot Development by Targeting D53-Like SMXL Repressor Proteins for Ubiquitination and Degradation. Plant Cell 2015, 27, 3128–3142. [Google Scholar] [CrossRef] [PubMed]
- Brewer, P.B.; Koltai, H.; Beveridge, C.A. Diverse Roles of Strigolactones in Plant Development. Mol. Plant 2013, 6, 18–28. [Google Scholar] [CrossRef] [PubMed]
- Ueda, H.; Kusaba, M. Strigolactone Regulates Leaf Senescence in Concert with Ethylene in Arabidopsis. Plant Physiol. 2015, 169, 138–147. [Google Scholar] [CrossRef] [PubMed]
- Akiyama, K.; Matsuzaki, K.; Hayashi, H. Plant sesquiterpenes induce hyphal branching in arbuscular mycorrhizal fungi. Nature 2005, 435, 824–827. [Google Scholar] [CrossRef] [PubMed]
- Foo, E.; Yoneyama, K.; Hugill, C.J.; Quittenden, L.J.; Reid, J.B. Strigolactones and the Regulation of Pea Symbioses in Response to Nitrate and Phosphate Deficiency. Mol. Plant 2013, 6, 76–87. [Google Scholar] [CrossRef]
- Lv, S.; Zhang, Y.H.; Li, C.; Liu, Z.J.; Yang, N.; Pan, L.X.; Wu, J.B.; Wang, J.J.; Yang, J.W.; Lv, Y.T.; et al. Strigolactone-triggered stomatal closure requires hydrogen peroxide synthesis and nitric oxide production in an abscisic acid-independent manner. New Phytol. 2018, 217, 290–304. [Google Scholar] [CrossRef]
- Visentin, I.; Vitali, M.; Ferrero, M.; Zhang, Y.X.; Ruyter-Spira, C.; Novak, O.; Strnad, M.; Lovisolo, C.; Schubert, A.; Cardinale, F. Low levels of strigolactones in roots as a component of the systemic signal of drought stress in tomato. New Phytol. 2016, 212, 954–963. [Google Scholar] [CrossRef]
- Ren, C.G.; Kong, C.C.; Xie, Z.H. Role of abscisic acid in strigolactone-induced salt stress tolerance in arbuscular mycorrhizal Sesbania cannabina seedlings. BMC Plant Biol. 2018, 18. [Google Scholar] [CrossRef]
- Haider, I.; Andreo-Jimenez, B.; Bruno, M.; Bimbo, A.; Flokova, K.; Abuauf, H.; Ntui, V.O.; Guo, X.J.; Charnikhova, T.; Al-Babili, S.; et al. The interaction of strigolactones with abscisic acid during the drought response in rice. J. Exp. Bot. 2018, 69, 2403–2414. [Google Scholar] [CrossRef]
- Ha, C.V.; Leyva-Gonzalez, M.A.; Osakabe, Y.; Tran, U.T.; Nishiyama, R.; Watanabe, Y.; Tanaka, M.; Seki, M.; Yamaguchi, S.; Dong, N.V.; et al. Positive regulatory role of strigolactone in plant responses to drought and salt stress. Proc. Natl. Acad. Sci. USA 2014, 111, 851–856. [Google Scholar] [CrossRef]
- Ma, N.; Hu, C.; Wan, L.; Hu, Q.; Xiong, J.L.; Zhang, C.L. Strigolactones Improve Plant Growth, Photosynthesis, and Alleviate Oxidative Stress under Salinity in Rapeseed (Brassica napus L.) by Regulating Gene Expression. Front. Plant Sci. 2017, 8. [Google Scholar] [CrossRef] [PubMed]
- Cooper, J.W.; Hu, Y.; Beyyoudh, L.; Dasgan, H.Y.; Kunert, K.; Beveridge, C.A.; Foyer, C.H. Strigolactones positively regulate chilling tolerance in pea and in Arabidopsis. Plant Cell Environ. 2018, 41, 1298–1310. [Google Scholar] [CrossRef] [PubMed]
- Umehara, M.; Hanada, A.; Magome, H.; Takeda-Kamiya, N.; Yamaguchi, S. Contribution of Strigolactones to the Inhibition of Tiller Bud Outgrowth under Phosphate Deficiency in Rice. Plant Cell Physiol. 2010, 51, 1118–1126. [Google Scholar] [CrossRef] [PubMed]
- Kohlen, W.; Charnikhova, T.; Liu, Q.; Bours, R.; Domagalska, M.A.; Beguerie, S.; Verstappen, F.; Leyser, O.; Bouwmeester, H.; Ruyter-Spira, C. Strigolactones Are Transported through the Xylem and Play a Key Role in Shoot Architectural Response to Phosphate Deficiency in Nonarbuscular Mycorrhizal Host Arabidopsis. Plant Physiol. 2011, 155, 974–987. [Google Scholar] [CrossRef]
- Ruiz-Lozano, J.M.; Aroca, R.; Zamarreno, A.M.; Molina, S.; Andreo-Jimenez, B.; Porcel, R.; Garcia-Mina, J.M.; Ruyter-Spira, C.; Lopez-Raez, J.A. Arbuscular mycorrhizal symbiosis induces strigolactone biosynthesis under drought and improves drought tolerance in lettuce and tomato. Plant Cell Environ. 2016, 39, 441–452. [Google Scholar] [CrossRef]
- Mayzlish-Gati, E.; De-Cuyper, C.; Goormachtig, S.; Beeckman, T.; Vuylsteke, M.; Brewer, P.B.; Beveridge, C.A.; Yermiyahu, U.; Kaplan, Y.; Enzer, Y.; et al. Strigolactones Are Involved in Root Response to Low Phosphate Conditions in Arabidopsis. Plant Physiol. 2012, 160, 1329–1341. [Google Scholar] [CrossRef]
- Bu, Q.Y.; Lv, T.X.; Shen, H.; Luong, P.; Wang, J.; Wang, Z.Y.; Huang, Z.G.; Xiao, L.T.; Engineer, C.; Kim, T.H.; et al. Regulation of Drought Tolerance by the F-Box Protein MAX2 in Arabidopsis(1[C][W][OPEN]). Plant Physiol. 2014, 164, 424–439. [Google Scholar] [CrossRef]
- Andreo-Jimenez, B.; Ruyter-Spira, C.; Bouwmeester, H.J.; Lopez-Raez, J.A. Ecological relevance of strigolactones in nutrient uptake and other abiotic stresses, and in plant-microbe interactions below-ground. Plant Soil 2015, 394, 1–19. [Google Scholar] [CrossRef]
- Min, Z.; Li, R.Y.; Chen, L.; Zhang, Y.; Li, Z.Y.; Liu, M.; Ju, Y.L.; Fang, Y.L. Alleviation of drought stress in grapevine by foliar-applied strigolactones. Plant Physiol. Biochem. 2019, 135, 99–110. [Google Scholar] [CrossRef]
- Cardinale, F.; Krukowski, P.K.; Schubert, A.; Visentin, I. Strigolactones: Mediators of osmotic stress responses with a potential for agrochemical manipulation of crop resilience. J. Exp. Bot. 2018, 69, 2291–2303. [Google Scholar] [CrossRef]
- Lopez-Raez, J.A.; Shirasu, K.; Foo, E. Strigolactones in Plant Interactions with Beneficial and Detrimental Organisms: The Yin and Yang. Trends Plant Sci. 2017, 22, 527–537. [Google Scholar] [CrossRef] [PubMed]
- De Cuyper, C.; Struk, S.; Braem, L.; Gevaert, K.; De Jaeger, G.; Goormachtig, S. Strigolactones, karrikins and beyond. Plant Cell Environ. 2017, 40, 1691–1703. [Google Scholar] [CrossRef] [PubMed]
- Al-Babili, S.; Bouwmeester, H.J. Strigolactones, a Novel Carotenoid-Derived Plant Hormone. Annu. Rev. Plant Biol. 2015, 66, 161–186. [Google Scholar] [CrossRef] [PubMed]
- Flematti, G.R.; Ghisalberti, E.L.; Dixon, K.W.; Trengove, R.D. A compound from smoke that promotes seed germination. Science 2004, 305, 977. [Google Scholar] [CrossRef]
- Nelson, D.C.; Scaffidi, A.; Dun, E.A.; Waters, M.T.; Flematti, G.R.; Dixon, K.W.; Beveridge, C.A.; Ghisalberti, E.L.; Smith, S.M. F-box protein MAX2 has dual roles in karrikin and strigolactone signaling in Arabidopsis thaliana. Proc. Natl. Acad. Sci. USA 2011, 108, 8897–8902. [Google Scholar] [CrossRef]
- Flematti, G.R.; Ghisalberti, E.L.; Dixon, K.W.; Trengove, R.D. Identification of Alkyl Substituted 2H-Furo[2,3-c]pyran-2-ones as Germination Stimulants Present in Smoke. J. Agric. Food Chem. 2009, 57, 9475–9480. [Google Scholar] [CrossRef]
- Nelson, D.C.; Flematti, G.R.; Ghisalberti, E.L.; Dixon, K.; Smith, S.M. Regulation of Seed Germination and Seedling Growth by Chemical Signals from Burning Vegetation. Annu. Rev. Plant Biol. 2012, 63, 107–130. [Google Scholar] [CrossRef]
- Smith, S.M.; Li, J.Y. Signalling and responses to strigolactones and karrikins. Curr. Opin. Plant Biol. 2014, 21, 23–29. [Google Scholar] [CrossRef]
- Ghebrehiwot, H.M.; Kulkarni, M.G.; Kirkman, K.P.; Van Staden, J. Smoke-water and a smoke-isolated butenolide improve germination and seedling vigour of Eragrostis tef (Zucc.) trotter under high temperature and low osmotic potential. J. Agron. Crop Sci. 2008, 194, 270–277. [Google Scholar] [CrossRef]
- Kulkarni, M.G.; Sparg, S.G.; Light, M.E.; van Staden, J. Stimulation of rice (Oryza sativa L.) seedling vigour by smoke-water and butenolide. J. Agron. Crop Sci. 2006, 192, 395–398. [Google Scholar] [CrossRef]
- Morffy, N.; Faure, L.; Nelson, D.C. Smoke and Hormone Mirrors: Action and Evolution of Karrikin and Strigolactone Signaling. Trends Genet. 2016, 32, 176–188. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Waters, M.T.; Smith, S.M. Karrikin-KAI2 signalling provides Arabidopsis seeds with tolerance to abiotic stress and inhibits germination under conditions unfavourable to seedling establishment. New Phytol. 2018, 219, 605–618. [Google Scholar] [CrossRef] [PubMed]
- Banerjee, A.; Tripathi, D.K.; Roychoudhury, A. The karrikin “calisthenics”: Can compounds derived from smoke help in stress tolerance? Physiol. Plant 2019, 165, 290–302. [Google Scholar] [CrossRef] [PubMed]
- Jia, K.P.; Baz, L.; Al-Babili, S. From carotenoids to strigolactones. J. Exp. Bot. 2018, 69, 2189–2204. [Google Scholar] [CrossRef] [PubMed]
- Lumba, S.; Holbrook-Smith, D.; McCourt, P. The perception of strigolactones in vascular plants. Nat. Chem. Biol. 2017, 13, 599–606. [Google Scholar] [CrossRef]
- Alder, A.; Jamil, M.; Marzorati, M.; Bruno, M.; Vermathen, M.; Bigler, P.; Ghisla, S.; Bouwmeester, H.; Beyer, P.; Al-Babili, S. The Path from beta-Carotene to Carlactone, a Strigolactone-Like Plant Hormone. Science 2012, 335, 1348–1351. [Google Scholar] [CrossRef]
- Lin, H.; Wang, R.X.; Qian, Q.; Yan, M.X.; Meng, X.B.; Fu, Z.M.; Yan, C.Y.; Jiang, B.; Su, Z.; Li, J.Y.; et al. DWARF27, an Iron-Containing Protein Required for the Biosynthesis of Strigolactones, Regulates Rice Tiller Bud Outgrowth. Plant Cell 2009, 21, 1512–1525. [Google Scholar] [CrossRef]
- Waters, M.T.; Brewer, P.B.; Bussell, J.D.; Smith, S.M.; Beveridge, C.A. The Arabidopsis Ortholog of Rice DWARF27 Acts Upstream of MAX1 in the Control of Plant Development by Strigolactones. Plant Physiol. 2012, 159, 1073–1085. [Google Scholar] [CrossRef]
- Booker, J.; Auldridge, M.; Wills, S.; McCarty, D.; Klee, H.; Leyser, O. MAX3/CCD7 is a carotenoid cleavage dioxygenase required for the synthesis of a novel plant signaling molecule. Curr. Biol. 2004, 14, 1232–1238. [Google Scholar] [CrossRef]
- Drummond, R.S.M.; Martinez-Sanchez, N.M.; Janssen, B.J.; Templeton, K.R.; Simons, J.L.; Quinn, B.D.; Karunairetnam, S.; Snowden, K.C. Petunia hybrida CAROTENOID CLEAVAGE DIOXYGENASE7 Is Involved in the Production of Negative and Positive Branching Signals in Petunia. Plant Physiol. 2009, 151, 1867–1877. [Google Scholar] [CrossRef]
- Zou, J.H.; Zhang, S.Y.; Zhang, W.P.; Li, G.; Chen, Z.X.; Zhai, W.X.; Zhao, X.F.; Pan, X.B.; Xie, Q.; Zhu, L.H. The rice HIGH-TILLERING DWARF1 encoding an ortholog of Arabidopsis MAX3 is required for negative regulation of the outgrowth of axillary buds. Plant J. 2006, 48, 687–696. [Google Scholar] [CrossRef] [PubMed]
- Arite, T.; Iwata, H.; Ohshima, K.; Maekawa, M.; Nakajima, M.; Kojima, M.; Sakakibara, H.; Kyozuka, J. DWARF10, an RMS1/MAX4/DAD1 ortholog, controls lateral bud outgrowth in rice. Plant J. 2007, 51, 1019–1029. [Google Scholar] [CrossRef] [PubMed]
- Simons, J.L.; Napoli, C.A.; Janssen, B.J.; Plummer, K.M.; Snowden, K.C. Analysis of the DECREASED APICAL DOMINANCE genes of petunia in the control of axillary branching. Plant Physiol. 2007, 143, 697–706. [Google Scholar] [CrossRef] [PubMed]
- Snowden, K.C.; Simkin, A.J.; Janssen, B.J.; Templeton, K.R.; Loucas, H.M.; Simons, J.L.; Karunairetnam, S.; Gleave, A.P.; Clark, D.G.; Klee, H.J. The Decreased apical dominance 1/petunia hybrida carotenoid cleavage dioxygenase8 gene affects branch production and plays a role in leaf senescence, root growth, and flower development. Plant Cell 2005, 17, 746–759. [Google Scholar] [CrossRef] [PubMed]
- Sorefan, K.; Booker, J.; Haurogne, K.; Goussot, M.; Bainbridge, K.; Foo, E.; Chatfield, S.; Ward, S.; Beveridge, C.; Rameau, C.; et al. MAX4 and RMS1 are orthologous dioxygenase-like genes that regulate shoot branching in Arabidopsis and pea. Genes Dev. 2003, 17, 1469–1474. [Google Scholar] [CrossRef]
- Seto, Y.; Sado, A.; Asami, K.; Hanada, A.; Umehara, M.; Akiyama, K.; Yamaguchi, S. Carlactone is an endogenous biosynthetic precursor for strigolactones. Proc. Natl. Acad. Sci. USA 2014, 111, 1640–1645. [Google Scholar] [CrossRef]
- Booker, J.; Sieberer, T.; Wright, W.; Williamson, L.; Willett, B.; Stirnberg, P.; Turnbull, C.; Srinivasan, M.; Goddard, P.; Leyser, O. MAX1 encodes a cytochrome P450 family member that acts downstream of MAX3/4 to produce a carotenoid-derived branch-inhibiting hormone. Dev. Cell 2005, 8, 443–449. [Google Scholar] [CrossRef]
- Drummond, R.S.M.; Sheehan, H.; Simons, J.L.; Martinez-Sanchez, N.M.; Turner, R.M.; Putterill, J.; Snowden, K.C. The expression of petunia strigolactone pathway genes is altered as part of the endogenous developmental program. Front. Plant Sci. 2012, 2. [Google Scholar] [CrossRef]
- Zhang, Y.X.; van Dijk, A.D.J.; Scaffidi, A.; Flematti, G.R.; Hofmann, M.; Charnikhova, T.; Verstappen, F.; Hepworth, J.; van der Krol, S.; Leyser, O.; et al. Rice cytochrome P450 MAX1 homologs catalyze distinct steps in strigolactone biosynthesis. Nat. Chem. Biol. 2014, 10, 1028–1033. [Google Scholar] [CrossRef]
- Brewer, P.B.; Yoneyama, K.; Filardo, F.; Meyers, E.; Scaffidi, A.; Frickey, T.; Akiyama, K.; Seto, Y.; Dun, E.A.; Cremer, J.E.; et al. LATERAL BRANCHING OXIDOREDUCTASE acts in the final stages of strigolactone biosynthesis in Arabidopsis. Proc. Natl. Acad. Sci. USA 2016, 113, 6301–6306. [Google Scholar] [CrossRef]
- Flematti, G.R.; Scaffidi, A.; Dixon, K.W.; Smith, S.M.; Ghisalberti, E.L. Production of the Seed Germination Stimulant Karrikinolide from Combustion of Simple Carbohydrates. J. Agric. Food Chem. 2011, 59, 1195–1198. [Google Scholar] [CrossRef] [PubMed]
- Nelson, D.C.; Riseborough, J.A.; Flematti, G.R.; Stevens, J.; Ghisalberti, E.L.; Dixon, K.W.; Smith, S.M. Karrikins Discovered in Smoke Trigger Arabidopsis Seed Germination by a Mechanism Requiring Gibberellic Acid Synthesis and Light. Plant Physiol. 2009, 149, 863–873. [Google Scholar] [CrossRef] [PubMed]
- Waters, M.T.; Scaffidi, A.; Sun, Y.M.K.; Flematti, G.R.; Smith, S.M. The karrikin response system of Arabidopsis. Plant J. 2014, 79, 623–631. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.H.; Zhou, X.E.; Wu, Z.S.; Yi, W.; Xu, Y.; Li, S.L.; Xu, T.H.; Liu, Y.; Chen, R.Z.; Kovach, A.; et al. Crystal structures of two phytohormone signal-transducing alpha/beta hydrolases: Karrikin-signaling KAI2 and strigolactone-signaling DWARF14. Cell Res. 2013, 23, 436–439. [Google Scholar] [CrossRef] [PubMed]
- Yao, R.F.; Ming, Z.H.; Yan, L.M.; Li, S.H.; Wang, F.; Ma, S.; Yu, C.T.; Yang, M.; Chen, L.; Chen, L.H.; et al. DWARF14 is a non-canonical hormone receptor for strigolactone. Nature 2016, 536, 469–473. [Google Scholar] [CrossRef]
- Waters, M.T.; Gutjahr, C.; Bennett, T.; Nelson, D.C. Strigolactone Signaling and Evolution. Annu. Rev. Plant Biol. 2017, 68, 291–322. [Google Scholar] [CrossRef]
- Nakamura, H.; Xue, Y.L.; Miyakawa, T.; Hou, F.; Qin, H.M.; Fukui, K.; Shi, X.; Ito, E.; Ito, S.; Park, S.H.; et al. Molecular mechanism of strigolactone perception by DWARF14. Nat. Commun. 2013, 4. [Google Scholar] [CrossRef]
- Zhou, F.; Lin, Q.B.; Zhu, L.H.; Ren, Y.L.; Zhou, K.N.; Shabek, N.; Wu, F.Q.; Mao, H.B.; Dong, W.; Gan, L.; et al. D14-SCFD3-dependent degradation of D53 regulates strigolactone signalling. Nature 2013, 504, 406. [Google Scholar] [CrossRef]
- Soundappan, I.; Bennett, T.; Morffy, N.; Liang, Y.Y.; Stang, J.P.; Abbas, A.; Leyser, O.; Nelson, D.C. SMAX1-LIKE/D53 Family Members Enable Distinct MAX2-Dependent Responses to Strigolactones and Karrikins in Arabidopsis. Plant Cell 2015, 27, 3143–3159. [Google Scholar] [CrossRef]
- Guo, Y.X.; Zheng, Z.Y.; La Clair, J.J.; Chory, J.; Noel, J.P. Smoke-derived karrikin perception by the alpha/beta-hydrolase KAI2 from Arabidopsis. Proc. Natl. Acad. Sci. USA 2013, 110, 8284–8289. [Google Scholar] [CrossRef]
- Kagiyama, M.; Hirano, Y.; Mori, T.; Kim, S.Y.; Kyozuka, J.; Seto, Y.; Yamaguchi, S.; Hakoshima, T. Structures of D14 and D14L in the strigolactone and karrikin signaling pathways. Genes Cells 2013, 18, 147–160. [Google Scholar] [CrossRef] [PubMed]
- Waters, M.T.; Scaffidi, A.; Moulin, S.L.Y.; Sun, Y.M.K.; Flematti, G.R.; Smith, S.M. A Selaginella moellendorffii Ortholog of KARRIKIN INSENSITIVE2 Functions in Arabidopsis Development but Cannot Mediate Responses to Karrikins or Strigolactones. Plant Cell 2015, 27, 1925–1944. [Google Scholar] [CrossRef] [PubMed]
- Waters, M.T.; Scaffidi, A.; Flematti, G.; Smith, S.M. Substrate-Induced Degradation of the alpha/beta-Fold Hydrolase KARRIKIN INSENSITIVE2 Requires a Functional Catalytic Triad but Is Independent of MAX2. Mol. Plant 2015, 8, 814–817. [Google Scholar] [CrossRef] [PubMed]
- Machin, D.C.; Hamon-Josse, M.; Bennett, T. Fellowship of the rings: A saga of strigolactones and other small signals. New Phytol. 2019. [Google Scholar] [CrossRef] [PubMed]
- Gupta, S.; Plackova, L.; Kulkarni, M.G.; Dolezal, K.; Van Staden, J. Role of Smoke Stimulatory and Inhibitory Biomolecules in Phytochrome-Regulated Seed Germination of Lactuca sativa. Plant Physiol. 2019, 181, 458–470. [Google Scholar] [CrossRef]
- Toh, S.; Kamiya, Y.; Kawakami, N.; Nambara, E.; McCourt, P.; Tsuchiya, Y. Thermoinhibition Uncovers a Role for Strigolactones in Arabidopsis Seed Germination. Plant Cell Physiol. 2012, 53, 107–117. [Google Scholar] [CrossRef]
- Vesty, E.F.; Saidi, Y.; Moody, L.A.; Holloway, D.; Whitbread, A.; Needs, S.; Choudhary, A.; Burns, B.; McLeod, D.; Bradshaw, S.J.; et al. The decision to germinate is regulated by divergent molecular networks in spores and seeds. New Phytol. 2016, 211, 952–966. [Google Scholar] [CrossRef]
- Nelson, D.C.; Flematti, G.R.; Riseborough, J.A.; Ghisalberti, E.L.; Dixon, K.W.; Smith, S.M. Karrikins enhance light responses during germination and seedling development in Arabidopsis thaliana. Proc. Natl. Acad. Sci. USA 2010, 107, 7095–7100. [Google Scholar] [CrossRef]
- Meng, Y.J.; Chen, F.; Shuai, H.W.; Luo, X.F.; Ding, J.; Tang, S.W.; Xu, S.S.; Liu, J.W.; Liu, W.G.; Du, J.B.; et al. Karrikins delay soybean seed germination by mediating abscisic acid and gibberellin biogenesis under shaded conditions. Sci. Rep. 2016, 6. [Google Scholar] [CrossRef]
- Stirnberg, P.; van de Sande, K.; Leyser, H.M.O. MAX1 and MAX2 control shoot lateral branching in Arabidopsis. Development 2002, 129, 1131–1141. [Google Scholar]
- Gomez-Roldan, V.; Fermas, S.; Brewer, P.B.; Puech-Pages, V.; Dun, E.A.; Pillot, J.P.; Letisse, F.; Matusova, R.; Danoun, S.; Portais, J.C.; et al. Strigolactone inhibition of shoot branching. Nature 2008, 455, 189–194. [Google Scholar] [CrossRef] [PubMed]
- Jamil, M.; Charnikhova, T.; Houshyani, B.; van Ast, A.; Bouwmeester, H.J. Genetic variation in strigolactone production and tillering in rice and its effect on Striga hermonthica infection. Planta 2012, 235, 473–484. [Google Scholar] [CrossRef] [PubMed]
- Tan, M.; Li, G.F.; Chen, X.L.; Xing, L.B.; Ma, J.J.; Zhang, D.; Ge, H.J.; Han, M.Y.; Sha, G.L.; An, N. Role of Cytokinin, Strigolactone, and Auxin Export on Outgrowth of Axillary Buds in Apple. Front. Plant Sci. 2019, 10. [Google Scholar] [CrossRef] [PubMed]
- Mouchel, C.F.; Leyser, O. Novel phytohormones involved in long-range signaling. Curr. Opin. Plant Biol. 2007, 10, 473–476. [Google Scholar] [CrossRef] [PubMed]
- Song, X.G.; Lu, Z.F.; Yu, H.; Shao, G.N.; Xiong, J.S.; Meng, X.B.; Jing, Y.H.; Liu, G.F.; Xiong, G.S.; Duan, J.B.; et al. IPA1 functions as a downstream transcription factor repressed by D53 in strigolactone signaling in rice. Cell Res. 2017, 27, 1128–1141. [Google Scholar] [CrossRef] [PubMed]
- Proust, H.; Hoffmann, B.; Xie, X.N.; Yoneyama, K.; Schaefer, D.G.; Yoneyama, K.; Nogue, F.; Rameau, C. Strigolactones regulate protonema branching and act as a quorum sensing-like signal in the moss Physcomitrella patens. Development 2011, 138, 1531–1539. [Google Scholar] [CrossRef]
- Kretzschmar, T.; Kohlen, W.; Sasse, J.; Borghi, L.; Schlegel, M.; Bachelier, J.B.; Reinhardt, D.; Bours, R.; Bouwmeester, H.J.; Martinoia, E. A petunia ABC protein controls strigolactone-dependent symbiotic signalling and branching. Nature 2012, 483, 341–344. [Google Scholar] [CrossRef]
- Xie, X.D.; Wang, G.H.; Yang, L.; Cheng, T.C.; Gao, J.P.; Wu, Y.Q.; Xia, Q.Y. Cloning and characterization of a novel Nicotiana tabacum ABC transporter involved in shoot branching. Physiol. Plant 2015, 153, 299–306. [Google Scholar] [CrossRef]
- Takeda, T.; Suwa, Y.; Suzuki, M.; Kitano, H.; Ueguchi-Tanaka, M.; Ashikari, M.; Matsuoka, M.; Ueguchi, C. The OsTB1 gene negatively regulates lateral branching in rice. Plant J. 2003, 33, 513–520. [Google Scholar] [CrossRef]
- Aguilar-Martinez, J.A.; Poza-Carrion, C.; Cubas, P. Arabidopsis BRANCHED1 acts as an integrator of branching signals within axillary buds. Plant Cell 2007, 19, 458–472. [Google Scholar] [CrossRef]
- Braun, N.; de Saint Germain, A.; Pillot, J.P.; Boutet-Mercey, S.; Dalmais, M.; Antoniadi, I.; Li, X.; Maia-Grondard, A.; Le Signor, C.; Bouteiller, N.; et al. The Pea TCP Transcription Factor PsBRC1 Acts Downstream of Strigolactones to Control Shoot Branching. Plant Physiol. 2012, 158, 225–238. [Google Scholar] [CrossRef]
- Martin-Trillo, M.; Grandio, E.G.; Serra, F.; Marcel, F.; Rodriguez-Buey, M.L.; Schmitz, G.; Theres, K.; Bendahmane, A.; Dopazo, H.; Cubas, P. Role of tomato BRANCHED1-like genes in the control of shoot branching. Plant J. 2011, 67, 701–714. [Google Scholar] [CrossRef]
- Nicolas, M.; Rodriguez-Buey, M.L.; Franco-Zorrilla, J.M.; Cubas, P. A Recently Evolved Alternative Splice Site in the BRANCHED1a Gene Controls Potato Plant Architecture. Curr. Biol. 2015, 25, 1799–1809. [Google Scholar] [CrossRef]
- Agusti, J.; Herold, S.; Schwarz, M.; Sanchez, P.; Ljung, K.; Dun, E.A.; Brewer, P.B.; Beveridge, C.A.; Sieberer, T.; Sehr, E.M.; et al. Strigolactone signaling is required for auxin-dependent stimulation of secondary growth in plants. Proc. Natl. Acad. Sci. USA 2011, 108, 20242–20247. [Google Scholar] [CrossRef]
- Zou, X.; Wang, Q.; Chen, P.S.; Yin, C.X.; Lin, Y.J. Strigolactones regulate shoot elongation by mediating gibberellin metabolism and signaling in rice (Oryza sativa L.). J. Plant Physiol. 2019, 237, 72–79. [Google Scholar] [CrossRef]
- Kapulnik, Y.; Delaux, P.M.; Resnick, N.; Mayzlish-Gati, E.; Wininger, S.; Bhattacharya, C.; Sejalon-Delmas, N.; Combier, J.P.; Becard, G.; Belausov, E.; et al. Strigolactones affect lateral root formation and root-hair elongation in Arabidopsis. Planta 2011, 233, 209–216. [Google Scholar] [CrossRef]
- Foo, E.; Davies, N.W. Strigolactones promote nodulation in pea. Planta 2011, 234, 1073–1081. [Google Scholar] [CrossRef]
- Arite, T.; Kameoka, H.; Kyozuka, J. Strigolactone Positively Controls Crown Root Elongation in Rice. J. Plant Growth Regul. 2012, 31, 165–172. [Google Scholar] [CrossRef]
- Rasmussen, A.; Mason, M.G.; De Cuyper, C.; Brewer, P.B.; Herold, S.; Agusti, J.; Geelen, D.; Greb, T.; Goormachtig, S.; Beeckman, T.; et al. Strigolactones Suppress Adventitious Rooting in Arabidopsis and Pea. Plant Physiol. 2012, 158, 1976–1987. [Google Scholar] [CrossRef]
- Kohlen, W.; Charnikhova, T.; Lammers, M.; Pollina, T.; Toth, P.; Haider, I.; Pozo, M.J.; de Maagd, R.A.; Ruyter-Spira, C.; Bouwmeester, H.J.; et al. The tomato CAROTENOID CLEAVAGE DIOXYGENASE8 (SlCCD8) regulates rhizosphere signaling, plant architecture and affects reproductive development through strigolactone biosynthesis. New Phytol. 2012, 196, 535–547. [Google Scholar] [CrossRef]
- Urquhart, S.; Foo, E.; Reid, J.B. The role of strigolactones in photomorphogenesis of pea is limited to adventitious rooting. Physiol. Plant 2015, 153, 392–402. [Google Scholar] [CrossRef]
- Liu, G.; Stirnemann, M.; Gubeli, C.; Egloff, S.; Courty, P.E.; Aubry, S.; Vandenbussche, M.; Morel, P.; Reinhardt, D.; Martinoia, E.; et al. Strigolactones Play an Important Role in Shaping Exodermal Morphology via a KAI2-Dependent Pathway. iScience 2019, 17, 144–154. [Google Scholar] [CrossRef]
- Villaecija-Aguilar, J.A.; Hamon-Josse, M.; Carbonnel, S.; Kretschmar, A.; Schmid, C.; Dawid, C.; Bennett, T.; Gutjahr, C. SMAX1/SMXL2 regulate root and root hair development downstream of KAI2-mediated signalling in Arabidopsis. PLoS Genet. 2019, 15. [Google Scholar] [CrossRef]
- Besserer, A.; Puech-Pages, V.; Kiefer, P.; Gomez-Roldan, V.; Jauneau, A.; Roy, S.; Portais, J.C.; Roux, C.; Becard, G.; Sejalon-Delmas, N. Strigolactones stimulate arbuscular mycorrhizal fungi by activating mitochondria. PLoS Biol. 2006, 4, 1239–1247. [Google Scholar] [CrossRef]
- Besserer, A.; Becard, G.; Jauneau, A.; Roux, C.; Sejalon-Delmas, N. GR24, a synthetic analog of strigolactones, stimulates the mitosis and growth of the arbuscular mycorrhizal fungus Gigaspora rosea by boosting its energy metabolism. Plant Physiol. 2008, 148, 402–413. [Google Scholar] [CrossRef]
- Foo, E.; Reid, J.B. Strigolactones: New Physiological Roles for an Ancient Signal. J. Plant Growth Regul. 2013, 32, 429–442. [Google Scholar] [CrossRef]
- Breuillin, F.; Schramm, J.; Hajirezaei, M.; Ahkami, A.; Favre, P.; Druege, U.; Hause, B.; Bucher, M.; Kretzschmar, T.; Bossolini, E.; et al. Phosphate systemically inhibits development of arbuscular mycorrhiza in Petunia hybrida and represses genes involved in mycorrhizal functioning. Plant J. 2010, 64, 1002–1017. [Google Scholar] [CrossRef]
- Lopez-Raez, J.A.; Charnikhova, T.; Fernandez, I.; Bouwmeester, H.; Pozo, M.J. Arbuscular mycorrhizal symbiosis decreases strigolactone production in tomato. J. Plant Physiol. 2011, 168, 294–297. [Google Scholar] [CrossRef]
- Fernandez-Aparicio, M.; Garcia-Garrido, J.M.; Ocampo, J.A.; Rubiales, D. Colonisation of field pea roots by arbuscular mycorrhizal fungi reduces Orobanche and Phelipanche species seed germination. Weed Res. 2010, 50, 262–268. [Google Scholar] [CrossRef]
- Catford, J.G.; Staehelin, C.; Lerat, S.; Piche, Y.; Vierheilig, H. Suppression of arbuscular mycorrhizal colonization and nodulation in split-root systems of alfalfa after pre-inoculation and treatment with Nod factors. J. Exp. Bot. 2003, 54, 1481–1487. [Google Scholar] [CrossRef]
- Ahmad, M.Z.; Rehman, N.U.U.; Yu, S.; Zhou, Y.; Haq, B.U.; Wang, J.; Li, P.; Zeng, Z.; Zhao, J. GmMAX2-D14 and -KAI interactions-mediated SL and KAR signaling play essential roles in soybean root nodulation. Plant J. 2019. [Google Scholar] [CrossRef] [PubMed]
- Waters, M.T.; Nelson, D.C.; Scaffidi, A.; Flematti, G.R.; Sun, Y.K.M.; Dixon, K.W.; Smith, S.M. Specialisation within the DWARF14 protein family confers distinct responses to karrikins and strigolactones in Arabidopsis. Development 2012, 139, 1285–1295. [Google Scholar] [CrossRef] [PubMed]
- Gutjahr, C.; Gobbato, E.; Choi, J.; Riemann, M.; Johnston, M.G.; Summers, W.; Carbonnel, S.; Mansfield, C.; Yang, S.Y.; Nadal, M.; et al. Rice perception of symbiotic arbuscular mycorrhizal fungi requires the karrikin receptor complex. Science 2015, 350, 1521–1524. [Google Scholar] [CrossRef] [PubMed]
- Jia, K.P.; Luo, Q.; He, S.B.; Lu, X.D.; Yang, H.Q. Strigolactone-Regulated Hypocotyl Elongation Is Dependent on Cryptochrome and Phytochrome Signaling Pathways in Arabidopsis. Mol. Plant 2014, 7, 528–540. [Google Scholar] [CrossRef]
- Tsuchiya, Y.; Vidaurre, D.; Toh, S.; Hanada, A.; Nambara, E.; Kamiya, Y.; Yamaguchi, S.; McCourt, P. A small-molecule screen identifies new functions for the plant hormone strigolactone. Nat. Chem. Biol. 2010, 6, 741–749. [Google Scholar] [CrossRef]
- Hamiaux, C.; Drummond, R.S.M.; Janssen, B.J.; Ledger, S.E.; Cooney, J.M.; Newcomb, R.D.; Snowden, K.C. DAD2 Is an alpha/beta Hydrolase Likely to Be Involved in the Perception of the Plant Branching Hormone, Strigolactone. Curr. Biol. 2012, 22, 2032–2036. [Google Scholar] [CrossRef]
- Ruyter-Spira, C.; Kohlen, W.; Charnikhova, T.; van Zeijl, A.; van Bezouwen, L.; de Ruijter, N.; Cardoso, C.; Lopez-Raez, J.A.; Matusova, R.; Bours, R.; et al. Physiological Effects of the Synthetic Strigolactone Analog GR24 on Root System Architecture in Arabidopsis: Another Belowground Role for Strigolactones? Plant Physiol. 2011, 155, 721–734. [Google Scholar] [CrossRef]
- van Rongen, M.; Bennett, T.; Ticchiarelli, F.; Leyser, O. Connective auxin transport contributes to strigolactone-mediated shoot branching control independent of the transcription factor BRC1. PLoS Genet. 2019, 15. [Google Scholar] [CrossRef]
- Ito, S.; Yamagami, D.; Umehara, M.; Hanada, A.; Yoshida, S.; Sasaki, Y.; Yajima, S.; Kyozuka, J.; Ueguchi-Tanaka, M.; Matsuoka, M.; et al. Regulation of Strigolactone Biosynthesis by Gibberellin Signaling. Plant Physiol. 2017, 174, 1250–1259. [Google Scholar] [CrossRef]
- Lopez-Raez, J.A.; Kohlen, W.; Charnikhova, T.; Mulder, P.; Undas, A.K.; Sergeant, M.J.; Verstappen, F.; Bugg, T.D.H.; Thompson, A.J.; Ruyter-Spira, C.; et al. Does abscisic acid affect strigolactone biosynthesis? New Phytol. 2010, 187, 343–354. [Google Scholar] [CrossRef]
- Shen, H.; Zhu, L.; Bu, Q.Y.; Huq, E. MAX2 Affects Multiple Hormones to Promote Photomorphogenesis. Mol. Plant 2012, 5, 750–762. [Google Scholar] [CrossRef]
- Liu, J.W.; He, H.Z.; Vitali, M.; Visentin, I.; Charnikhova, T.; Haider, I.; Schubert, A.; Ruyter-Spira, C.; Bouwmeester, H.J.; Lovisolo, C.; et al. Osmotic stress represses strigolactone biosynthesis in Lotus japonicus roots: Exploring the interaction between strigolactones and ABA under abiotic stress. Planta 2015, 241, 1435–1451. [Google Scholar] [CrossRef]
- Rodriguez, J.A.M.; Morcillo, R.L.; Vierheilig, H.; Ocampo, J.A.; Ludwig-Muller, J.; Garrido, J.M.G. Mycorrhization of the notabilis and sitiens tomato mutants in relation to abscisic acid and ethylene contents. J. Plant Physiol. 2010, 167, 606–613. [Google Scholar] [CrossRef]
- Kapulnik, Y.; Resnick, N.; Mayzlish-Gati, E.; Kaplan, Y.; Wininger, S.; Hershenhorn, J.; Koltai, H. Strigolactones interact with ethylene and auxin in regulating root-hair elongation in Arabidopsis. J. Exp. Bot. 2011, 62, 2915–2924. [Google Scholar] [CrossRef]
- Yamada, Y.; Furusawa, S.; Nagasaka, S.; Shimomura, K.; Yamaguchi, S.; Umehara, M. Strigolactone signaling regulates rice leaf senescence in response to a phosphate deficiency. Planta 2014, 240, 399–408. [Google Scholar] [CrossRef]
- Dun, E.A.; de Saint Germain, A.; Rameau, C.; Beveridge, C.A. Antagonistic Action of Strigolactone and Cytokinin in Bud Outgrowth Control. Plant Physiol. 2012, 158, 487–498. [Google Scholar] [CrossRef]
- Minakuchi, K.; Kameoka, H.; Yasuno, N.; Umehara, M.; Luo, L.; Kobayashi, K.; Hanada, A.; Ueno, K.; Asami, T.; Yamaguchi, S.; et al. FINE CULM1 (FC1) Works Downstream of Strigolactones to Inhibit the Outgrowth of Axillary Buds in Rice. Plant Cell Physiol. 2010, 51, 1127–1135. [Google Scholar] [CrossRef]
- Duan, J.B.; Yu, H.; Yuan, K.; Liao, Z.G.; Meng, X.B.; Jing, Y.H.; Liu, G.F.; Chu, J.F.; Li, J.Y. Strigolactone promotes cytokinin degradation through transcriptional activation of CYTOKININ OXIDASE/DEHYDROGENASE 9 in rice. Proc. Natl. Acad. Sci. USA 2019, 116, 14319–14324. [Google Scholar] [CrossRef]
- Jiang, L.X.; Matthys, C.; Marquez-Garcia, B.; De Cuyper, C.; Smet, L.; De Keyser, A.; Boyer, F.D.; Beeckman, T.; Depuydt, S.; Goormachtig, S. Strigolactones spatially influence lateral root development through the cytokinin signaling network. J. Exp. Bot. 2016, 67, 379–389. [Google Scholar] [CrossRef]
- Wang, Y.; Sun, S.Y.; Zhu, W.J.; Jia, K.P.; Yang, H.Q.; Wang, X.L. Strigolactone/MAX2-Induced Degradation of Brassinosteroid Transcriptional Effector BES1 Regulates Shoot Branching. Dev. Cell 2013, 27, 681–688. [Google Scholar] [CrossRef]
- Reinhardt, D.; Pesce, E.R.; Stieger, P.; Mandel, T.; Baltensperger, K.; Bennett, M.; Traas, J.; Friml, J.; Kuhlemeier, C. Regulation of phyllotaxis by polar auxin transport. Nature 2003, 426, 255–260. [Google Scholar] [CrossRef] [PubMed]
- Wang, R.H.; Estelle, M. Diversity and specificity: Auxin perception and signaling through the TIR1/AFB pathway. Curr. Opin. Plant Biol. 2014, 21, 51–58. [Google Scholar] [CrossRef] [PubMed]
- Szemenyei, H.; Hannon, M.; Long, J.A. TOPLESS mediates auxin-dependent transcriptional repression during Arabidopsis embryogenesis. Science 2008, 319, 1384–1386. [Google Scholar] [CrossRef] [PubMed]
- Brewer, P.B.; Dun, E.A.; Ferguson, B.J.; Rameau, C.; Beveridge, C.A. Strigolactone Acts Downstream of Auxin to Regulate Bud Outgrowth in Pea and Arabidopsis. Plant Physiol. 2009, 150, 482–493. [Google Scholar] [CrossRef] [PubMed]
- Zha, M.; Imran, M.; Wang, Y.; Xu, J.; Ding, Y.; Wang, S.H. Transcriptome analysis revealed the interaction among strigolactones, auxin, and cytokinin in controlling the shoot branching of rice. Plant Cell Rep. 2019, 38, 279–293. [Google Scholar] [CrossRef]
- Sun, H.W.; Tao, J.Y.; Liu, S.J.; Huang, S.J.; Chen, S.; Xie, X.N.; Yoneyama, K.; Zhang, Y.L.; Xu, G.H. Strigolactones are involved in phosphate- and nitrate-deficiency-induced root development and auxin transport in rice. J. Exp. Bot. 2014, 65, 6735–6746. [Google Scholar] [CrossRef]
- Bennett, T.; Sieberer, T.; Willett, B.; Booker, J.; Luschnig, C.; Leyser, O. The Arabidopsis MAX pathway controls shoot branching by regulating auxin transport. Curr. Biol. 2006, 16, 553–563. [Google Scholar] [CrossRef]
- Kumar, M.; Pandya-Kumar, N.; Dam, A.; Haor, H.; Mayzlish-Gati, E.; Belausov, E.; Wininger, S.; Abu-Abied, M.; McErlean, C.S.P.; Bromhead, L.J.; et al. Arabidopsis response to low-phosphate conditions includes active changes in actin filaments and PIN2 polarization and is dependent on strigolactone signalling. J. Exp. Bot. 2015, 66, 1499–1510. [Google Scholar] [CrossRef]
- Crawford, S.; Shinohara, N.; Sieberer, T.; Williamson, L.; George, G.; Hepworth, J.; Muller, D.; Domagalska, M.A.; Leyser, O. Strigolactones enhance competition between shoot branches by dampening auxin transport. Development 2010, 137, 2905–2913. [Google Scholar] [CrossRef]
- Shinohara, N.; Taylor, C.; Leyser, O. Strigolactone Can Promote or Inhibit Shoot Branching by Triggering Rapid Depletion of the Auxin Efflux Protein PIN1 from the Plasma Membrane. PLoS Biol. 2013, 11. [Google Scholar] [CrossRef]
- Koltai, H. Strigolactones are regulators of root development. New Phytol. 2011, 190, 545–549. [Google Scholar] [CrossRef] [PubMed]
- Koltai, H.; Dor, E.; Hershenhorn, J.; Joel, D.M.; Weininger, S.; Lekalla, S.; Shealtiel, H.; Bhattacharya, C.; Eliahu, E.; Resnick, N.; et al. Strigolactones’ Effect on Root Growth and Root-Hair Elongation May Be Mediated by Auxin-Efflux Carriers. J. Plant Growth Regul. 2010, 29, 129–136. [Google Scholar] [CrossRef]
- Bertheloot, J.; Barbier, F.; Boudon, F.; Perez-Garcia, M.D.; Peron, T.; Citerne, S.; Dun, E.; Beveridge, C.; Godin, C.; Sakr, S. Sugar availability suppresses the auxin-induced strigolactone pathway to promote bud outgrowth. New Phytol. 2019. [Google Scholar] [CrossRef] [PubMed]
- Hayward, A.; Stirnberg, P.; Beveridge, C.; Leyser, O. Interactions between Auxin and Strigolactone in Shoot Branching Control. Plant Physiol. 2009, 151, 400–412. [Google Scholar] [CrossRef]
- Xu, J.X.; Zha, M.R.; Li, Y.; Ding, Y.F.; Chen, L.; Ding, C.Q.; Wang, S.H. The interaction between nitrogen availability and auxin, cytokinin, and strigolactone in the control of shoot branching in rice (Oryza sativa L.). Plant Cell Rep. 2015, 34, 1647–1662. [Google Scholar] [CrossRef]
- Cheng, X.; Ruyter-Spira, C.; Bouwmeester, H. The interaction between strigolactones and other plant hormones in the regulation of plant development. Front. Plant Sci. 2013, 4. [Google Scholar] [CrossRef]
- Meng, Y.J.; Shuai, H.W.; Luo, X.F.; Chen, F.; Zhou, W.G.; Yang, W.Y.; Shu, K. Karrikins: Regulators Involved in Phytohormone Signaling Networks during Seed Germination and Seedling Development. Front. Plant Sci. 2017, 7. [Google Scholar] [CrossRef]
- Yamaguchi, S. Gibberellin metabolism and its regulation. Annu. Rev. Plant Biol. 2008, 59, 225–251. [Google Scholar] [CrossRef]
- Lo, S.F.; Yang, S.Y.; Chen, K.T.; Hsing, Y.L.; Zeevaart, J.A.D.; Chen, L.J.; Yu, S.M. A Novel Class of Gibberellin 2-Oxidases Control Semidwarfism, Tillering, and Root Development in Rice. Plant Cell 2008, 20, 2603–2618. [Google Scholar] [CrossRef]
- Silverstone, A.L.; Chang, C.W.; Krol, E.; Sun, T.P. Developmental regulation of the gibberellin biosynthetic gene GA1 in Arabidopsis thaliana. Plant J. 1997, 12, 9–19. [Google Scholar] [CrossRef]
- Ueguchi-Tanaka, M.; Ashikari, M.; Nakajima, M.; Itoh, H.; Katoh, E.; Kobayashi, M.; Chow, T.Y.; Hsing, Y.I.C.; Kitano, H.; Yamaguchi, I.; et al. GIBBERELLIN INSENSITIVE DWARF1 encodes a soluble receptor for gibberellin. Nature 2005, 437, 693–698. [Google Scholar] [CrossRef]
- Sun, T.P. Gibberellin-GID1-DELLA: A Pivotal Regulatory Module for Plant Growth and Development. Plant Physiol. 2010, 154, 567–570. [Google Scholar] [CrossRef] [PubMed]
- Zentella, R.; Zhang, Z.L.; Park, M.; Thomas, S.G.; Endo, A.; Murase, K.; Fleet, C.M.; Jikumaru, Y.; Nambara, E.; Kamiya, Y.; et al. Global analysis of DELLA direct targets in early gibberellin signaling in Arabidopsis. Plant Cell 2007, 19, 3037–3057. [Google Scholar] [CrossRef] [PubMed]
- Kepczynski, J.; Cembrowska-Lech, D.; Van Staden, J. Necessity of gibberellin for stimulatory effect of KAR(1) on germination of dormant Avena fatua L. caryopses. Acta Physiol. Plant. 2013, 35, 379–387. [Google Scholar] [CrossRef]
- Kepczynski, J. Induction of agricultural weed seed germination by smoke and smoke-derived karrikin (KAR(1)), with a particular reference to Avena fatua L. Acta Physiol. Plant. 2018, 40. [Google Scholar] [CrossRef]
- de Saint Germain, A.; Ligerot, Y.; Dun, E.A.; Pillot, J.P.; Ross, J.J.; Beveridge, C.A.; Rameau, C. Strigolactones Stimulate Internode Elongation Independently of Gibberellins. Plant Physiol. 2013, 163, 1012–1025. [Google Scholar] [CrossRef]
- Bassel, G.W.; Mullen, R.T.; Bewley, J.D. Procera is a putative DELLA mutant in tomato (Solanum lycopersicum): Effects on the seed and vegetative plant. J. Exp. Bot. 2008, 59, 585–593. [Google Scholar] [CrossRef]
- Luo, L.; Takahashi, M.; Kameoka, H.; Qin, R.Y.; Shiga, T.; Kanno, Y.; Seo, M.; Ito, M.; Xu, G.H.; Kyozuka, J. Developmental analysis of the early steps in strigolactone-mediated axillary bud dormancy in rice. Plant J. 2019, 97, 1006–1021. [Google Scholar] [CrossRef]
- Brun, G.; Thoiron, S.; Braem, L.; Pouvreau, J.B.; Montiel, G.; Lechat, M.M.; Simier, P.; Gevart, K.; Goormachtig, S.; Delavault, P. CYP707As are effectors of karrikin and strigolactone signalling pathways in Arabidopsis thaliana and parasitic plants. Plant Cell Environ. 2019, 42, 2612–2626. [Google Scholar] [CrossRef]
- Matilla, A.J. Ethylene in seed formation and germination. Seed Sci. Res. 2000, 10, 111–126. [Google Scholar] [CrossRef]
- Sugimoto, Y.; Ali, A.M.; Yabuta, S.; Kinoshita, H.; Inanaga, S.; Itai, A. Germination strategy of Striga hermonthica involves regulation of ethylene biosynthesis. Physiol. Plant 2003, 119, 137–145. [Google Scholar] [CrossRef]
- Jackson, M.B.; Parker, C. Induction of Germination by a Strigol Analog Requires Ethylene Action in Striga-Hermonthica but Not in S-Forbesii. J. Plant Physiol. 1991, 138, 383–386. [Google Scholar] [CrossRef]
- Zehar, N.; Ingouff, M.; Bouya, D.; Fer, A. Possible involvement of gibberellins and ethylene in Orobanche ramosa germination. Weed Res. 2002, 42, 464–469. [Google Scholar] [CrossRef]
- Rasmussen, A.; Hu, Y.M.; Depaepe, T.; Vandenbussche, F.; Boyer, F.D.; Van Der Straeten, D.; Geelen, D. Ethylene Controls Adventitious Root Initiation Sites in Arabidopsis Hypocotyls Independently of Strigolactones. J. Plant Growth Regul. 2017, 36, 897–911. [Google Scholar] [CrossRef]
- Hu, Z.Y.; Yamauchi, T.; Yang, J.H.; Jikumaru, Y.; Tsuchida-Mayama, T.; Ichikawa, H.; Takamure, I.; Nagamura, Y.; Tsutsumi, N.; Yamaguchi, S.; et al. Strigolactone and Cytokinin Act Antagonistically in Regulating Rice Mesocotyl Elongation in Darkness. Plant Cell Physiol. 2014, 55, 30–41. [Google Scholar] [CrossRef] [PubMed]
- Koren, D.; Resnick, N.; Gati, E.M.; Belausov, E.; Weininger, S.; Kapulnik, Y.; Koltai, H. Strigolactone signaling in the endodermis is sufficient to restore root responses and involves SHORT HYPOCOTYL 2 (SHY2) activity. New Phytol. 2013, 198, 866–874. [Google Scholar] [CrossRef]
- Kulkarni, M.G.; Rengasamy, K.R.R.; Pendota, S.C.; Gruz, J.; Plackova, L.; Novak, O.; Dolezal, K.; Van Staden, J. Bioactive molecules derived from smoke and seaweed Ecklonia maxima showing phytohormone-like activity in Spinacia oleracea L. New Biotechnol. 2019, 48, 83–89. [Google Scholar] [CrossRef]
- Aremu, A.O.; Plackova, L.; Novak, O.; Stirk, W.A.; Dolezal, K.; Van Staden, J. Cytokinin profiles in ex vitro acclimatized Eucomis autumnalis plants pre-treated with smoke-derived karrikinolide. Plant Cell Rep. 2016, 35, 227–238. [Google Scholar] [CrossRef]
- Du, H.; Huang, F.; Wu, N.; Li, X.H.; Hu, H.H.; Xiong, L.H. Integrative Regulation of Drought Escape through ABA-Dependent and -Independent Pathways in Rice. Mol. Plant 2018, 11, 584–597. [Google Scholar] [CrossRef]
- Mostofa, M.G.; Li, W.; Nguyen, K.H.; Fujita, M.; Tran, L.P. Strigolactones in plant adaptation to abiotic stresses: An emerging avenue of plant research. Plant Cell Environ. 2018, 41, 2227–2243. [Google Scholar] [CrossRef]
- Lopez-Bucio, J.; Cruz-Ramirez, A.; Herrera-Estrella, L. The role of nutrient availability in regulating root architecture. Curr. Opin. Plant Biol. 2003, 6, 280–287. [Google Scholar] [CrossRef]
- Sun, H.W.; Bi, Y.; Tao, J.Y.; Huang, S.J.; Hou, M.M.; Xue, R.; Liang, Z.H.; Gu, P.Y.; Yoneyama, K.; Xie, X.N.; et al. Strigolactones are required for nitric oxide to induce root elongation in response to nitrogen and phosphate deficiencies in rice. Plant Cell Environ. 2016, 39, 1473–1484. [Google Scholar] [CrossRef] [PubMed]
- Walton, A.; Stes, E.; Goeminne, G.; Braem, L.; Vuylsteke, M.; Matthys, C.; De Cuyper, C.; Staes, A.; Vandenbussche, J.; Boyer, F.D.; et al. The Response of the Root Proteome to the Synthetic Strigolactone GR24 in Arabidopsis. Mol. Cell. Proteom. 2016, 15, 2744–2755. [Google Scholar] [CrossRef] [PubMed]
- Pourcel, L.; Routaboul, J.M.; Cheynier, V.; Lepiniec, L.; Debeaujon, I. Flavonoid oxidation in plants: From biochemical properties to physiological functions. Trends Plant Sci. 2007, 12, 29–36. [Google Scholar] [CrossRef] [PubMed]
- Ferreyra, M.L.F.; Rius, S.P.; Casati, P. Flavonoids: Biosynthesis, biological functions, and biotechnological applications. Front. Plant Sci. 2012, 3. [Google Scholar] [CrossRef]
- Nakabayashi, R.; Yonekura-Sakakibara, K.; Urano, K.; Suzuki, M.; Yamada, Y.; Nishizawa, T.; Matsuda, F.; Kojima, M.; Sakakibara, H.; Shinozaki, K.; et al. Enhancement of oxidative and drought tolerance in Arabidopsis by overaccumulation of antioxidant flavonoids. Plant J. 2014, 77, 367–379. [Google Scholar] [CrossRef]
- Koltai, H. Strigolactones activate different hormonal pathways for regulation of root development in response to phosphate growth conditions. Ann. Bot. 2013, 112, 409–415. [Google Scholar] [CrossRef]
- Koltai, H. Cellular events of strigolactone signalling and their crosstalk with auxin in roots. J. Exp. Bot. 2015, 66, 4855–4861. [Google Scholar] [CrossRef]
- Marzec, M. Strigolactones and Gibberellins: A New Couple in the Phytohormone World? Trends Plant Sci. 2017, 22, 813–815. [Google Scholar] [CrossRef]
- Lantzouni, O.; Klermund, C.; Schwechheimer, C. Largely additive effects of gibberellin and strigolactone on gene expression in Arabidopsis thaliana seedlings. Plant J. 2017, 92, 924–938. [Google Scholar] [CrossRef]
- Matusova, R.; Rani, K.; Verstappen, F.W.A.; Franssen, M.C.R.; Beale, M.H.; Bouwmeester, H.J. The strigolactone germination stimulants of the plant-parasitic Striga and Orobanche spp. are derived from the carotenoid pathway. Plant Physiol. 2005, 139, 920–934. [Google Scholar] [CrossRef] [PubMed]
- Lopez-Raez, J.A.; Charnikhova, T.; Gomez-Roldan, V.; Matusova, R.; Kohlen, W.; De Vos, R.; Verstappen, F.; Puech-Pages, V.; Becard, G.; Mulder, P.; et al. Tomato strigolactones are derived from carotenoids and their biosynthesis is promoted by phosphate starvation. New Phytol. 2008, 178, 863–874. [Google Scholar] [CrossRef] [PubMed]
- Torres-Vera, R.; Garcia, J.M.; Pozo, M.J.; Lopez-Raez, J.A. Do strigolactones contribute to plant defence? Mol. Plant Pathol. 2014, 15, 211–216. [Google Scholar] [CrossRef] [PubMed]
- Marzec, M.; Muszynska, A. In Silico Analysis of the Genes Encoding Proteins that Are Involved in the Biosynthesis of the RMS/MAX/D Pathway Revealed New Roles of Strigolactones in Plants. Int. J. Mol. Sci. 2015, 16, 6757–6782. [Google Scholar] [CrossRef] [PubMed]
- Zhuang, L.; Wang, J.; Huang, B. Drought inhibition of tillering in Festuca arundinacea associated with axillary bud development and strigolactone signaling. Environ. Exp. Bot. 2017, 142, 15–23. [Google Scholar] [CrossRef]
- Kong, C.-C.; Ren, C.-G.; Li, R.-Z.; Xie, Z.-H.; Wang, J.-P. Hydrogen peroxide and strigolactones signaling are involved in alleviation of salt stress induced by arbuscular mycorrhizal fungus in sesbania cannabina seedlings. J. Plant Growth Regul. 2017, 36, 734–742. [Google Scholar] [CrossRef]
- Mackova, H.; Hronkova, M.; Dobra, J.; Tureckova, V.; Novak, O.; Lubovska, Z.; Motyka, V.; Haisel, D.; Hajek, T.; Prasil, I.T.; et al. Enhanced drought and heat stress tolerance of tobacco plants with ectopically enhanced cytokinin oxidase/dehydrogenase gene expression. J. Exp. Bot. 2013, 64, 2805–2815. [Google Scholar] [CrossRef]
- Nishiyama, R.; Watanabe, Y.; Fujita, Y.; Le, D.T.; Kojima, M.; Werner, T.; Vankova, R.; Yamaguchi-Shinozaki, K.; Shinozaki, K.; Kakimoto, T.; et al. Analysis of Cytokinin Mutants and Regulation of Cytokinin Metabolic Genes Reveals Important Regulatory Roles of Cytokinins in Drought, Salt and Abscisic Acid Responses, and Abscisic Acid Biosynthesis. Plant Cell 2011, 23, 2169–2183. [Google Scholar] [CrossRef]
- Li, W.Q.; Herrera-Estrella, L.; Tran, L.S.P. Do Cytokinins and Strigolactones Crosstalk during Drought Adaptation? Trends Plant Sci. 2019, 24, 669–672. [Google Scholar] [CrossRef]
- Lim, P.O.; Kim, H.J.; Nam, H.G. Leaf senescence. Annu. Rev. Plant Biol. 2007, 58, 115–136. [Google Scholar] [CrossRef]
| Species | Mutants | Effects of Mutant | Functions | Interactions with Phytohormones | References |
|---|---|---|---|---|---|
| Rice | d3, d14 and d53 | Increased branching | D53 acts as a repressor of the SL signaling to promoting axillary bud outgrowth | [89] | |
| pea | ccd8 | Endogenous SLs inhibit shoot branching in plants | [102] | ||
| pea | ramosus (rms) | SLs regulate shoot branching | [76] | ||
| Arabidopsis | max4 | ||||
| petunia | dad | [138] | |||
| ipa1 | Regulated tiller number | IPA1 interacts with D53 to mediate tiller regulated by SL | [106] | ||
| Arabidopsis | max1 and max2 | Increased branching, round leaves, elongated hypocotyl | MAX1 and MAX2 control shoot branching by repressing primordia formation of the axillary meristem | [101] | |
| smxl6, smxl7, smxl8 | Reduced shoot branching in smxl6/7/8 | SMXL6, SMXL7, and SMXL8 promote shoot branching by repressing BRC1/TCP18 expression in axillary buds | Auxin | [90] | |
| Reduced auxin transport in smxl6/7/8 | SMXL6, SMXL7, and SMXL8 promote auxin transport in a MAX2-dependent manner | ||||
| Lower lateral root density in smxl6/7/8 Reduced auxin transport in smxl6/7/8 | SMXL6, SMXL7, and SMXL8 promote lateral root density | ||||
| smxl6, smxl7, smxl8, max2, smax1, d14 and kai2 | In short day: elongated petiole in smxl6/7/8, shortened petiole in max2 and d14, increased both blade length and width in kai2 | SMAX1 and SMXL6,7,8 regulate the complementary aspects of leaf morphology in different signaling pathways | |||
| petunia | pdr1 | Increased branching | PaPDR1 acts as a transporter of SL to regulate branching | [108] | |
| dad1/Phccd8 | Increased branching | Mutations of PhCCD8 caused a high branching of dad1 | [75] | ||
| Smaller flowers | Loss of Dad1 reduces the overall height of the plant, root and flowering development | ||||
| Reduced internode length | |||||
| Reduced root growth | |||||
| Arabidopsis | ore9/max2 | Delayed senescence of leaves | Dad1/PhCCD8 and ORE9/MAX2 regulate the leaf senescence by affecting the same signaling pathway | [101] | |
| Rice | d17 and D10 | SLs affect leaf senescence | |||
| Arabidopsis | max1, max2, pin1 and pin3 | Reduced cambium activity | SLs stimulate the secondary growth in auxin-dependent | Auxin | [115] |
| Arabidopsis | max4 | Reduced auxin content in the leaf | SLs reduce the content of auxin | Auxin | [33] |
| Arabidopsis | max2, max4 and pin3pin4pin7 | pin3/4/7 was restored high branching of max2 and max4 | PIN3, 4, and 5 of CAT contribute to branching mediated by SL | Auxin | [139] |
| Arabidopsis | max1-4, ipt1,5,7 and ahk3,4 | Increased adventitious roots | SLs suppress adventitious root formation, SLs could partially restore the stimulating effect of auxin on adventitious root formation | Auxin | [120] |
| Pea | rms1, rms4 and rms5 | SLs also suppress the adventitious root by reducing the size of rooting zone in Pea | |||
| Rice | d3, d10, d14, | Higher epi-5DS levels by feedback relationship of SL pathway | GA3 regulates SL biosynthesis in a D3 and D14 independent manner | GA | [140] |
| slr1-5, gid1-3, and gid2-2 | Reduced the levels of SLs | GAs negatively regulates the level of SLs in a GID1- and GID2-dependent manner | |||
| Species | Mutants | Effects of mutant | Functions | Interactions with phytohormones | References |
| Maize | up14 | Reduced content of SL in root secretion | ABA and SL affect each other’s synthesis | ABA | [141] |
| tomato | notabilis and sitiens | ||||
| Arabidopsis | max | Lower sensitivity to ABA | The synthesis of SL is regulated by ABA | [41] | |
| Arabidopsis | max1 and max2 | Reduced seed germination | Application of GR24 can restore thermoinhibition in max1 and max2 caused by ABA inhibition of GA synthesis and signal and increase GA4 content | ABA and GA | [97] |
| max2 | Lower sensitivity to ABA | MAX2 participates in ABA signaling pathway as an important component of SL signaling pathway | ABA | [142] | |
| Lotus japonicus | ljccd7 | Decreased ABA content | SLs interaction with ABA to regulate the abiotic stress | ABA | [143] |
| tomato | sitiens | Decreased AM colonization | ABA plays a role in arbuscular mycorrhizal fungi symbiosis by regulating the production of SLs | ABA | [144] |
| Physcomitrella | ccd8 | Increased pore germination | SLs inhibits the germination of Physcomitrella pore germination | [98] | |
| Arabidopsis | max2, ein 2-1 and etr1-1 | Reduced root hair | SL’s effect on RH elongation is dependent on both auxin and ethylene signaling | Ethylene and Auxin | [145] |
| tir1, arf7arf19 and aux1 | Increased root hair elongation | ||||
| Arabidopsis | max2 | Delayed senescence | SL interacts with ethylene to regulate leaf senescence | Ethylene | [101] |
| Pea | dad1/Phccd8 | [75] | |||
| Rice | dwarf | [146] | |||
| Pea | rms1 | rms1 are more sensitive to CK | CKs and SLs contribute to bud outgrowth in pea | CK | [147] |
| Rice | fc1, d10, d3 | Increased tillering | The branching related gene FC1 (FINECULM1) is insensitive to SLs, but is inhibited by CK | CK | [148] |
| Rice | d53 | Increased CK content | SLs promote CK degradation | CK | [149] |
| Arabidopsis | ahk3, arr1 and arr12 | Lateral root development insensitive to GR24 and affected polar auxin transport | SLs connects with auxins and CKs to regulate LR development | CK and Auxin | [150] |
| Arabidopsis | max2 and bes1 | Enhanced rosette branching | MAX2 interacts with BES1 to regulate branching | BR | [151] |
| Arabidopsis | hy5 | Shortened hypocotyl | KARs restore the hypocotyl elongation inhibited by red light in hy5 | [99] | |
| ga1 | KAR1 cannot promote the germination of ga1-3 of GA synthesis defect | KAR1 promotes germination is required for GA biosynthesis | GA | [83] | |
| sleepy1 | Delay seed germination | KAR1 promotes germination is partly dependent on DELLA | |||
| Soybean | Wild type | KARs delay seed germination under shaded conditions by inhibiting GA synthesis and promoting ABA synthesis | GA and ABA | [100] | |
| Arabidopsis | kai2, kai2/d14 | Decreased root hair density; Exaggerated skewing and waving | KAI2 signaling pathway regulated root hair and root development | [124] | |
| Lactuca sativa | Wild type | Smoke-water and KAR1 promote seed germination | Application of smoke-water and KAR1 decrease ABA content and enhance hydrolase activity to mobilize stored reserves | ABA | [96] |
| Genotypes | Types of Stresses | Mutants or Transgenes | Effects | Interactions with Phytohormones | References |
|---|---|---|---|---|---|
| Arabidopsis | Drought stress and salinity | max2-3, max2-4, max3-11, max3-12, max4-7, and max4-8 | SLs positively regulate plant responses to drought and salt stress | ABA and CK | [41] |
| Arabidopsis | Drought stress | max2-1, max2-2, max1, max3, and max4 | MAX2 play an important role in plant responses to abiotic stress | ABA | [48] |
| Lotus japonicus | Phosphate starvation and osmotic stress | LjCCD7-silenced line (Ljccd7) | SLs contribute to drought resistance in Lotus japonicus | ABA | [143] |
| Arabidopsis | Wounding, heat, UV-B, salinity | Wild type | Abiotic stresses responses | ABA, CK, IAA, BR, ET, GA, and MeJA | [205] |
| Tomato | Drought | SlCCD7-silenced line (Ljccd7) | Low levels of SLs in roots act as components of the systemic signal of drought stress | ABA | [38] |
| Tomato | Drought and AMF | Wild type | AMF induces SL biosynthesis under drought and improves drought tolerance | ABA | [46] |
| Festuca arundinacea | PEG-induced drought stress | Wild type | Drought-inhibition of tiller development and growth in grass species are associated with SL accumulation and signaling | [206] | |
| Rapeseed | salinity | Wild type | Salinity depresses the shoots and roots growth, whereas GR24 improves the growth under salt stress | [42] | |
| Sesbania cannabina | Salinity and AMF | Wild type | SLs enhance salt stress tolerance, and the H2O2-induced SL accumulation was accompanied by increased salt tolerance | [207] | |
| Rice | Drought stress | d10, d17, d27, and d3, D27-overexpressing plants | SL biosynthesis/perception interferes with ABA formation, and D27 plays a crucial role in determining ABA and SL content | ABA | [40] |
| Sesbania cannabina | Salinity and AMF | Wild type | ABA is regulating the induction of salt tolerance by SL in AM seedlings | ABA | [39] |
| Arabidopsis | Dark | max1-1, max3-9, max4-11, Atd14-1, and max2-4 | ET synthesis and consequent SL synthesis are required for the efficient progression of dark-induced leaf senescence. | ET | [34] |
| Arabidopsis | Phosphate deficiency | max2-1 and max4-1 | SLs regulate the response of plants to low Pi | Auxin | [34] |
| Rice | Phosphate- and nitrate-deficiency | d3, d10, and d27 | SLs affect root growth in rice under phosphate and nitrate limitation by decreasing auxin transport from shoots to roots | Auxin | [157] |
| Rice | Phosphate- and nitrate-deficiency | d3, d10, and d53 | SLs involve in NO-activated elongation of seminal root under nitrogen and phosphate deficiency conditions. | [193] | |
| Arabidopsis and pea | Dark chilling | max2-1, max3-9, and max4-1 in Arabidopsis; rms5-3, rms4-1, and rms3-1 in pea | SLs positively regulate chilling tolerance in pea and in Arabidopsis | [44] | |
| Arabidopsis | Drought stress | kai2-2, kai2-4, and d14-2 | The KAR receptor KAI2 promotes drought resistance | ABA | [8] |
| Arabidopsis | Osmotic stress and salinity | kai2-2, d14-1, max2-1 and max2-7 | Karrikin-KAI2 signaling system can protect against abiotic stress | GA and ABA | [63] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yang, T.; Lian, Y.; Wang, C. Comparing and Contrasting the Multiple Roles of Butenolide Plant Growth Regulators: Strigolactones and Karrikins in Plant Development and Adaptation to Abiotic Stresses. Int. J. Mol. Sci. 2019, 20, 6270. https://doi.org/10.3390/ijms20246270
Yang T, Lian Y, Wang C. Comparing and Contrasting the Multiple Roles of Butenolide Plant Growth Regulators: Strigolactones and Karrikins in Plant Development and Adaptation to Abiotic Stresses. International Journal of Molecular Sciences. 2019; 20(24):6270. https://doi.org/10.3390/ijms20246270
Chicago/Turabian StyleYang, Tao, Yuke Lian, and Chongying Wang. 2019. "Comparing and Contrasting the Multiple Roles of Butenolide Plant Growth Regulators: Strigolactones and Karrikins in Plant Development and Adaptation to Abiotic Stresses" International Journal of Molecular Sciences 20, no. 24: 6270. https://doi.org/10.3390/ijms20246270
APA StyleYang, T., Lian, Y., & Wang, C. (2019). Comparing and Contrasting the Multiple Roles of Butenolide Plant Growth Regulators: Strigolactones and Karrikins in Plant Development and Adaptation to Abiotic Stresses. International Journal of Molecular Sciences, 20(24), 6270. https://doi.org/10.3390/ijms20246270




