Electron Transfer Induced Decomposition in Potassium–Nitroimidazoles Collisions: An Experimental and Theoretical Work
Abstract
1. Introduction
2. Results and Discussion
2.1. Nitroimidazoles and Methylated Compounds
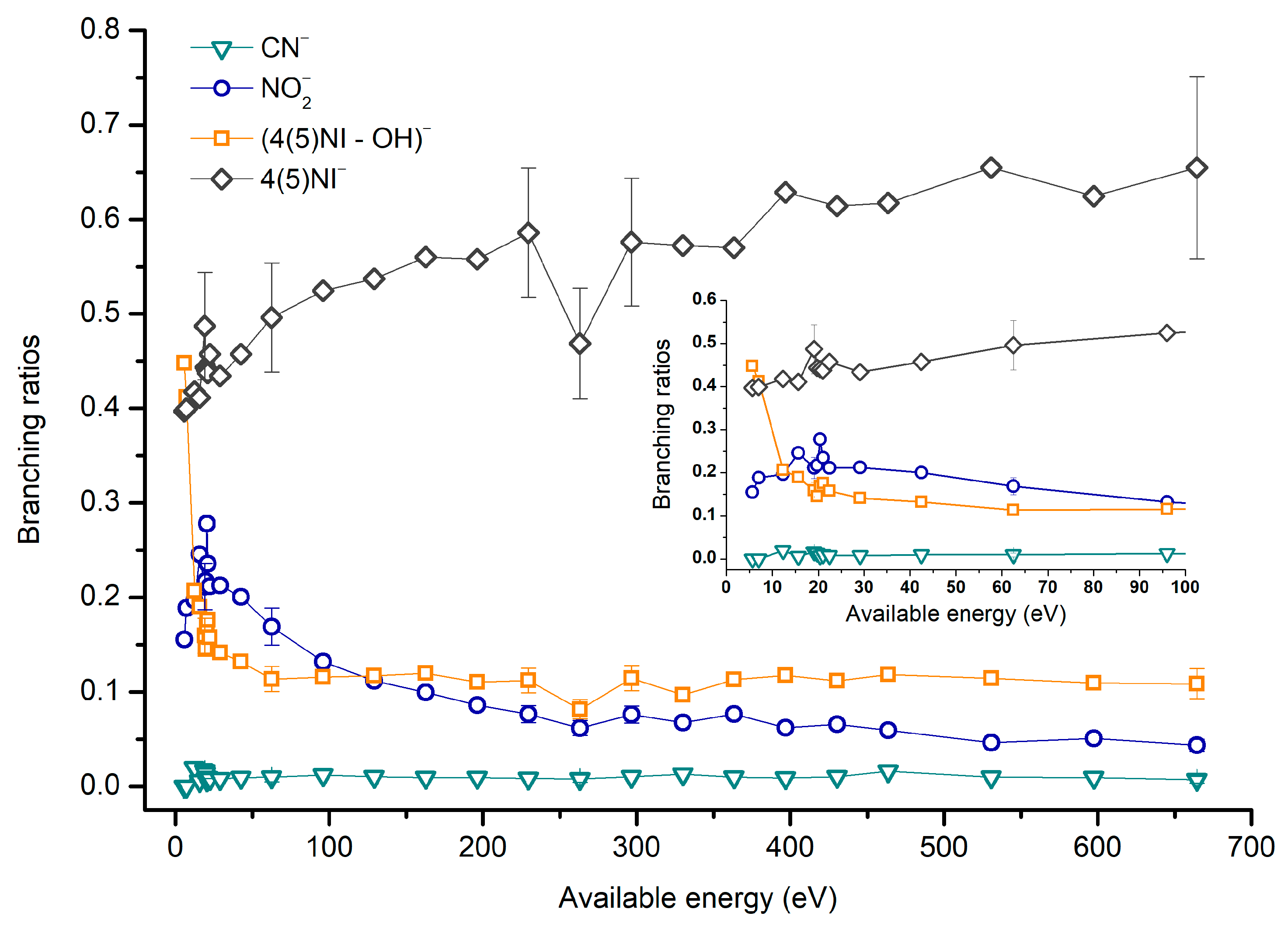
2.1.1. Parent Anion Formation
2.1.2. Formation of NO2−, Loss of Neutral NO2, and Related Multiple Dehydrogenation (HNO2−, H2NO2−, and H3NO2−)
2.1.3. (M–NO)‒ and (M–OH)‒ Formation
2.1.4. CN− Formation
2.2. Imidazole Molecule
2.2.1. (IMI−H)− Formation
2.2.2. C2H2N−, CH− and CN− Formation
3. Experimental Methods
4. Theoretical Method
5. Conclusions
Supplementary Materials
Supplementary File 1Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| DEA | dissociative electron attachment |
| SMC | Schwinger multichannel |
| IMI | imidazole |
| 4(5)NI | 4(5)-nitroimidazole |
| 2NI | 2-nitroimidazole |
| Me4NI | 1-methyl-4-nitroimidazole |
| Me5NI | 1-methyl-5-nitroimidazole |
| VAE | vertical attachment energy |
| TOF | time-of-flight |
| BR | branching ratio |
| TNI | temporary negative ion |
| MO | molecular orbital |
| DBS | dipole bound state |
References
- Wardman, P. Chemical Radiosensitizers for Use in Radiotherapy. Clin. Oncol. 2007, 19, 397–417. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Mu, X.; He, H.; Zhang, X.D. Cancer Radiosensitizers. Trends Pharm. Sci. 2018, 39, 24–48. [Google Scholar] [CrossRef] [PubMed]
- Overgaard, J.; Hansen, H.S.; Overgaard, M.; Bastholt, L.; Berthelsen, A.; Specht, L.; Lindeløv, B.; Jørgensen, K. A randomized double-blind phase III study of nimorazole as a hypoxic radiosensitizer of primary radiotherapy in supraglottic larynx and pharynx carcinoma. Results of the Danish Head and Neck Cancer Study (DAHANCA) Protocol 5-85. Radiother. Oncol. 1998, 46, 135–146. [Google Scholar] [CrossRef]
- Overgaard, J. Hypoxic modification of radiotherapy in squamous cell carcinoma of the head and neck—A systematic review and meta-analysis. Radiother. Oncol. 2011, 100, 22–32. [Google Scholar] [CrossRef]
- Feketeová, L.; Albright, A.L.; Sørensen, B.S.; Horsman, M.R.; White, J.; O’Hair, R.A.J.; Bassler, N. Formation of radical anions of radiosensitizers and related model compounds via electrospray ionization. Int. J. Mass Spectrom. 2014, 365, 56–63. [Google Scholar] [CrossRef]
- Pandeti, S.; Feketeová, L.; Reddy, T.J.; Abdoul-Carime, H.; Farizon, B.; Farizon, M.; Märk, T.D. Binding preference of nitroimidazolic radiosensitizers to nucleobases and nucleosides probed by electrospray ionization mass spectrometry and density functional theory. J. Chem. Phys. 2019, 150, 014302. [Google Scholar] [CrossRef]
- Feketeová, L.; Plekan, O.; Goonewardane, M.; Ahmed, M.; Albright, A.L.; White, J.; O’Hair, R.A.J.; Horsman, M.R.; Wang, F.; Prince, K.C. Photoelectron Spectra and Electronic Structures of the Radiosensitizer Nimorazole and Related Compounds. J. Phys. Chem. A 2015, 119, 9986–9995. [Google Scholar] [CrossRef]
- Meißner, R.; Kočišek, J.; Feketeová, L.; Fedor, J.; Fárník, M.; Limão-Vieira, P.; Illenberger, E.; Denifl, S. Low-energy electrons transform the nimorazole molecule into a radiosensitiser. Nat. Commun. 2019, 10, 2388. [Google Scholar] [CrossRef]
- Meißner, R.; Feketeová, L.; Illenberger, E.; Denifl, S. Reactions in the Radiosensitizer Misonidazole Induced by Low-Energy (0–10 eV) Electrons. Int. J. Mol. Sci. 2019, 20, 3496. [Google Scholar] [CrossRef]
- Tanzer, K.; Feketeová, L.; Puschnigg, B.; Scheier, P.; Illenberger, E.; Denifl, S. Reactions in Nitroimidazole Triggered by Low-Energy (0–2 eV) Electrons: Methylation at N1-H Completely Blocks Reactivity. Angew. Chem. Int. Ed. Engl. 2014, 53, 12240–12243. [Google Scholar] [CrossRef]
- Tanzer, K.; Feketeová, L.; Puschnigg, B.; Scheier, P.; Illenberger, E.; Denifl, S. Reactions in Nitroimidazole and Methylnitroimidazole Triggered by Low-Energy (0–8 eV) Electrons. J. Phys. Chem. A 2015, 119, 6668–6675. [Google Scholar] [CrossRef] [PubMed]
- Yu, Z.; Bernstein, E.R. Experimental and theoretical studies of the decomposition of new imidazole based energetic materials: Model systems. J. Chem. Phys. 2012, 137, 114303. [Google Scholar] [CrossRef] [PubMed]
- Cartoni, A.; Casavola, A.R.; Bolognesi, P.; Castrovilli, M.C.; Catone, D.; Chiarinelli, J.; Richter, R.; Avaldi, L. Insights into 2- and 4(5)-Nitroimidazole Decomposition into Relevant Ions and Molecules Induced by VUV Ionization. J. Phys. Chem. A 2018, 122, 4031–4041. [Google Scholar] [CrossRef] [PubMed]
- Kossoski, F.; Varella, M.T.D.N. How does methylation suppress the electron-induced decomposition of 1-methyl-nitroimidazoles? J. Chem. Phys. 2017, 147, 164310. [Google Scholar] [CrossRef]
- Brown, E.G. Imidazoles and Benzimidazoles. In Ring Nitrogen and Key Biomolecules; Springer: Dordrecht, The Netherlands, 1998; pp. 40–59. ISBN 978-94-010-6058-5. [Google Scholar]
- Kumar, M.; Kumar, D.; Raj, V. Studies on Imidazole and Its Derivatives with Particular Emphasis on Their Chemical/biological Applications as Bioactive Molecules/Intermediated to Bioactive Molecule. Curr. Synth. Syst. Biol. 2017, 5, 1–10. [Google Scholar] [CrossRef]
- Ribar, A.; Fink, K.; Li, Z.; Ptasinska, S.; Carmichael, I.; Feketeova, L.; Denifl, S. Stripping off Hydrogens in Imidazole Triggered by the Attachment of a Single Electron. Phys. Chem. Chem. Phys. 2017, 19, 6406–6415. [Google Scholar] [CrossRef]
- Modelli, A.; Burrow, P.D. Electron attachment to the aza-derivatives of furan, pyrrole, and thiophene. J. Phys. Chem. A 2004, 108, 5721–5726. [Google Scholar] [CrossRef]
- Carles, S.; Lecomte, F.; Schermann, J.P.; Desfrançois, C. Gas-phase experimental and theoretical studies of adenine, imidazole, pyrrole, and water non-covalent complexes. J. Phys. Chem. A 2000, 104, 10662–10668. [Google Scholar] [CrossRef]
- Gianola, A.J.; Ichino, T.; Hoenigman, R.L.; Kato, S.; Bierbaum, V.M.; Lineberger, W.C. Photoelectron spectra and ion chemistry of imidazolide. J. Phys. Chem. A 2005, 109, 11504–11514. [Google Scholar] [CrossRef]
- Bolognesi, P.; Casavola, A.R.; Cartoni, A.; Richter, R.; Markus, P.; Borocci, S.; Chiarinelli, J.; Tošić, S.; Sa’adeh, H.; Masič, M.; et al. Communication: “Position” does matter: The photofragmentation of the nitroimidazole isomers. J. Chem. Phys. 2016, 145, 191102. [Google Scholar] [CrossRef]
- Jimenez, P.; Laynes, J.; Claramunt, R.M.; Sanz, D.; Fayet, J.P.; Vertut, M.C.; Catalán, J.; Paz, J.L.G.; Pfister-Guillouzo, G.; Guimon, C.; et al. The Problem of the Tautomerism of 4(5)-Nitroimidazole—A Theoretical and Experimental-Study. New J. Chem. 1989, 13, 151–156. [Google Scholar]
- Mendes, M.; Probst, M.; García, G.; Limão-Vieira, P. Selective Bond Excision of Nitroimidazoles in Electron Transfer Experiments. J Phys Chem A 2019, 123, 4068–4073. [Google Scholar] [CrossRef]
- Cunha, T.; Mendes, M.; Ferreira Da Silva, F.; Eden, S.; García, G.; Bacchus-Montabonel, M.C.; Limão-Vieira, P. Electron transfer driven decomposition of adenine and selected analogs as probed by experimental and theoretical methods. J. Chem. Phys. 2018, 148, 134301. [Google Scholar] [CrossRef]
- Mendes, M.; Pamplona, B.; Kumar, S.; da Silva, F.F.; Aguilar, A.; García, G.; Bacchus-Montabonel, M.C.; Limão-Vieira, P. Ion-pair formation in neutral potassium-neutral pyrimidine collisions: Electron transfer experiments. Front. Chem. 2019, 7, 264. [Google Scholar] [CrossRef]
- Kleyn, A.; Los, J.; Gislason, E.A. Vibronic Coupling At Intersections of Covalent and Ionic States. Phys. Rep. 1982, 90, 1–71. [Google Scholar] [CrossRef]
- Lacmann, K. Collisional Ionization. In Advances in Chemical Physics (Potential Energy Surfaces); Lawley, K.P., Ed.; John Wiley & Sons Ltd.: London, UK, 1980; pp. 513–583. [Google Scholar]
- NIST Chemistry WebBook. Available online: http://webbook.nist.gov/chemistry/ (accessed on 1 September 2019).
- Illenberger, E.; Momingy, J. Gaseous Molecular Ions: An Introduction to Elementary Processes Induced by Ionization, 1st ed.; Baumgartel, H., Frank, E.U., Grunbein, W., Eds.; Steinkopff-Verlag: Heidelberg, Germany; New York, NY, USA, 1992; ISBN 3-7985-0870-4. [Google Scholar]
- Kleyn, A.W.; Moutinho, A.M.C. Negative ion formation in alkali-atom-molecule collisions. J. Phys. B 2001, 34, R1. [Google Scholar] [CrossRef]
- Limão-Vieira, P.; Moutinho, A.M.C.; Los, J. Dissociative ion-pair formation in collisions of fast potassium atoms with benzene and fluorobenzene. J. Chem. Phys. 2006, 124, 054306. [Google Scholar] [CrossRef]
- Ribar, A.; Fink, K.; Probst, M.; Huber, S.E.; Feketeová, L.; Denifl, S. Isomer Selectivity in Low-Energy Electron Attachment to Nitroimidazoles. Chem. Eur. J. 2017, 23, 12892–12899. [Google Scholar] [CrossRef]
- Cadet, J.; Douki, T.; Ravanat, J.L. Oxidatively generated base damage to cellular DNA. Free Radic. Biol. Med. 2010, 49, 9–21. [Google Scholar] [CrossRef]
- Cadet, J.; Douki, T.; Ravanat, J.L. Measurement of oxidatively generated base damage in cellular DNA. Mutat. Res. Fundam. Mol. Mech. Mutagen. 2011, 711, 3–12. [Google Scholar] [CrossRef]
- Ravanat, J.-L.; Cadet, J.; Douki, T. Oxidatively Generated DNA Lesions as Potential Biomarkers of In Vivo Oxidative Stress. Curr. Mol. Med. 2012, 12, 655–671. [Google Scholar] [CrossRef]
- Dumont, E.; Monari, A. Understanding DNA under oxidative stress and sensitization: The role of molecular modeling. Front. Chem. 2015, 3, 43. [Google Scholar] [CrossRef]
- Almeida, D.; Antunes, R.; Martins, G.; Eden, S.; Ferreira da Silva, F.; Nunes, Y.; Garcia, G.; Limão-Vieira, P. Electron transfer-induced fragmentation of thymine and uracil in atom-molecule collisions. Phys. Chem. Chem. Phys. 2011, 13, 15657–15665. [Google Scholar] [CrossRef]
- Cunha, T.; Mendes, M.; Ferreira Da Silva, F.; Eden, S.; García, G.; Limão-Vieira, P. Communication: Site-selective bond excision of adenine upon electron transfer. J. Chem. Phys. 2018, 148, 021101. [Google Scholar] [CrossRef]
- Antunes, R.; Almeida, D.; Martins, G.; Mason, N.J.; Garcia, G.; Maneira, M.J.P.; Nunes, Y.; Limão-Vieira, P. Negative ion formation in potassium–nitromethane collisions. Phys. Chem. Chem. Phys. 2010, 12, 12513–12519. [Google Scholar] [CrossRef]
- Almeida, D.; Bacchus-Montabonel, M.-C.; Ferreira da Silva, F.; García, G.; Limão-Vieira, P. Potassium-uracil/thymine ring cleavage enhancement as studied in electron transfer experiments and theoretical calculations. J. Phys. Chem. A 2014, 118, 6547–6552. [Google Scholar] [CrossRef]
- Bacchus-Montabonel, M.C.; Tergiman, Y.S. Radiation damage on biomolecular systems: Dynamics of ion induced collision processes. Comput. Theor. Chem. 2012, 990, 177–184. [Google Scholar] [CrossRef]
- Bene, E.; Vibók, Á.; Halász, G.J.; Bacchus-Montabonel, M.C. Ab initio molecular treatment of charge transfer processes induced by collision of C2+ ions with the OH radical: A linear approach. Chem. Phys. Lett. 2008, 455, 159–163. [Google Scholar] [CrossRef]
- Bacchus-Montabonel, M.C.; Tergiman, Y.S. Charge transfer dynamics of carbon ions with uracil and halouracil targets at low collision energies. Chem. Phys. Lett. 2011, 503, 45–48. [Google Scholar] [CrossRef]
- Salem, L. Electrons in Chemical Reactions: First Principles; Wiley Interscience: New York, NY, USA, 1982. [Google Scholar]
- Bacchus-Montabonel, M.C.; Tergiman, Y.S. Anisotropic effect in the charge transfer of Cq+ ions with uracil. Phys. Rev. A At. Mol. Opt. Phys. 2006, 74, 8–11. [Google Scholar] [CrossRef]
- Calvo, F.; Bacchus-Montabonel, M.C.; Clavaguéra, C. Stepwise Hydration of 2-Aminooxazole: Theoretical Insight into the Structure, Finite Temperature Behavior and Proton-Induced Charge Transfer. J. Phys. Chem. A 2016, 120, 2380–2389. [Google Scholar] [CrossRef]
- Bacchus-Montabonel, M.C.; Tergiman, Y.S. An ab initio study of ion induced charge transfer dynamics in collision of carbon ions with thymine. Phys. Chem. Chem. Phys. 2011, 13, 9761–9767. [Google Scholar] [CrossRef]
- Werner, H.-J.; Knowles, P.J.; Knizia, G.; Manby, F.R.; Schütz, M.; Celani, P.; Györffy, W.; Kats, D.; Korona, T.; Lindh, R.; et al. MOLPRO, Version 2015.1 2015, Orsay, France.
- Nicklass, A.; Dolg, M.; Stoll, H.; Preuss, H. Ab initio energy-adjusted pseudopotentials for the noble gases Ne through Xe: Calculation of atomic dipole and quadrupole polarizabilities. J. Chem. Phys. 1995, 102, 8942–8952. [Google Scholar] [CrossRef]

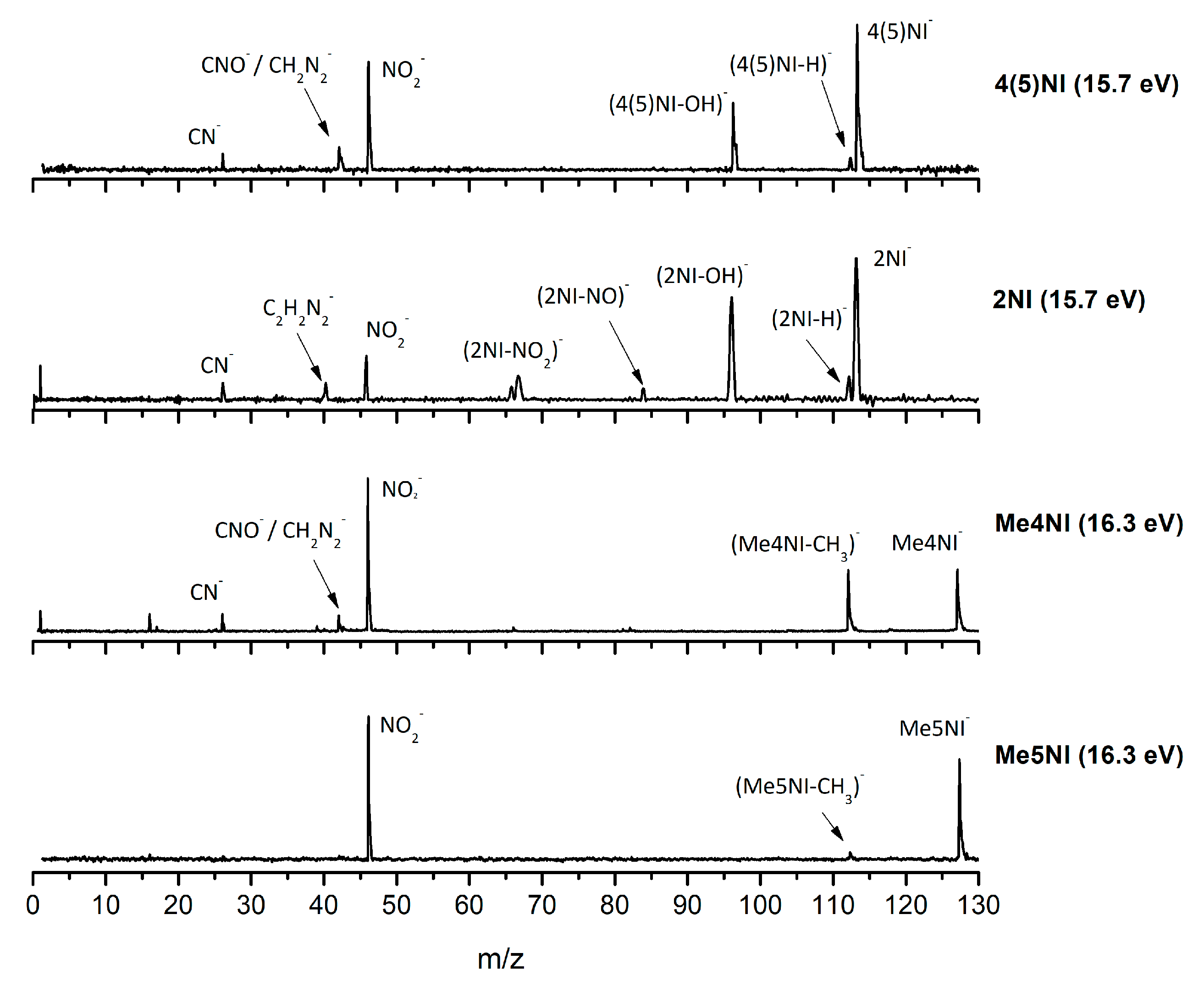
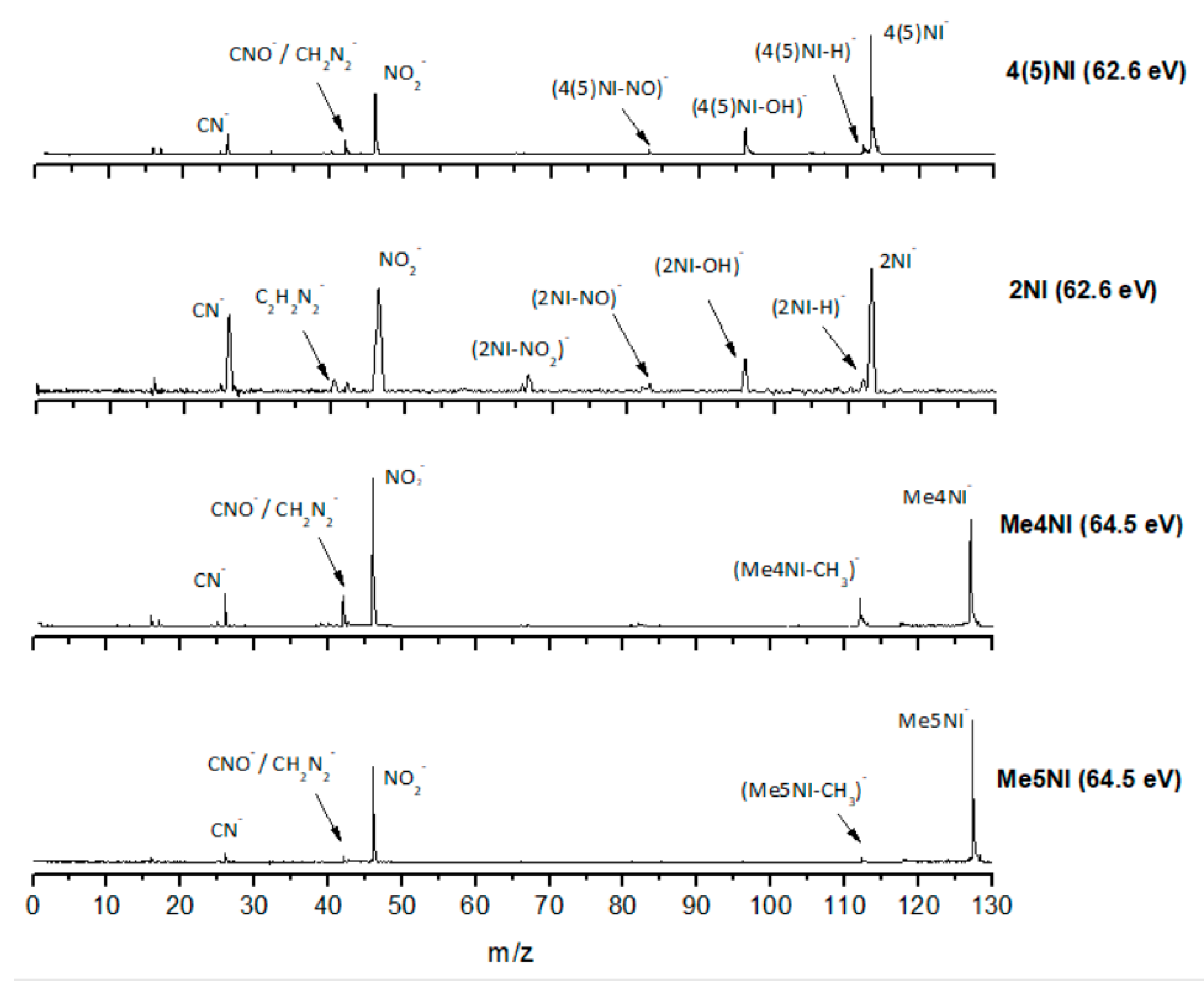
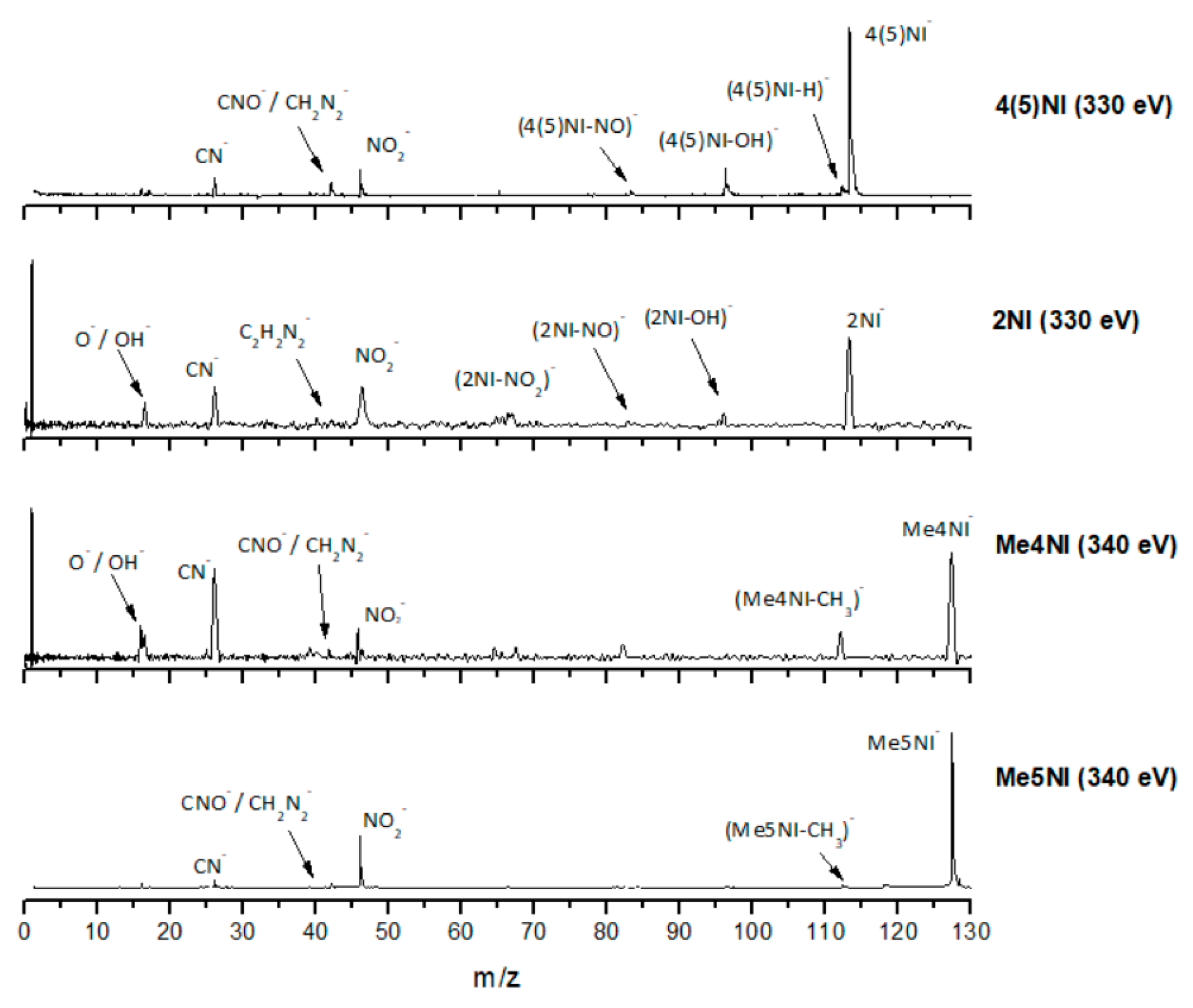
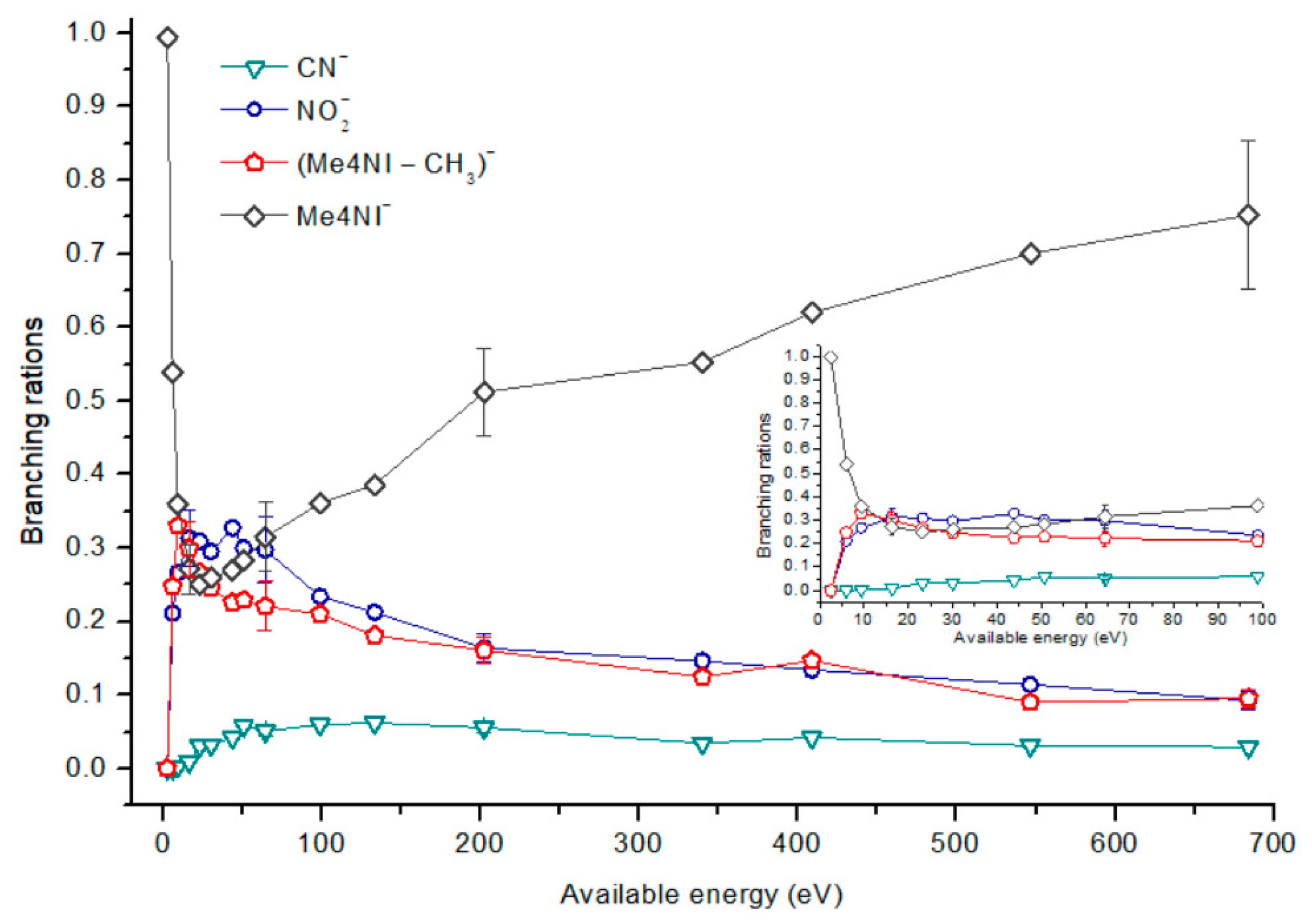
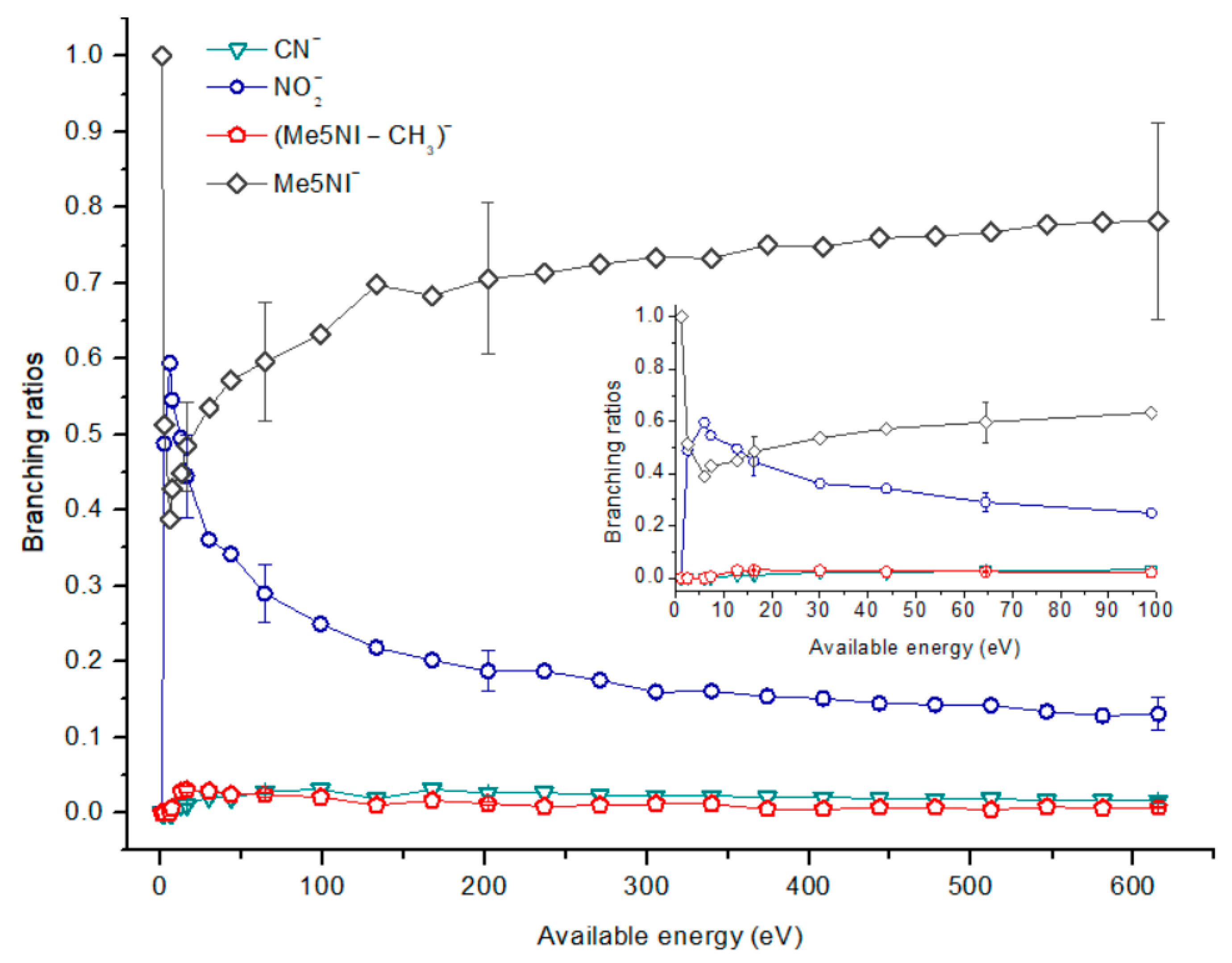


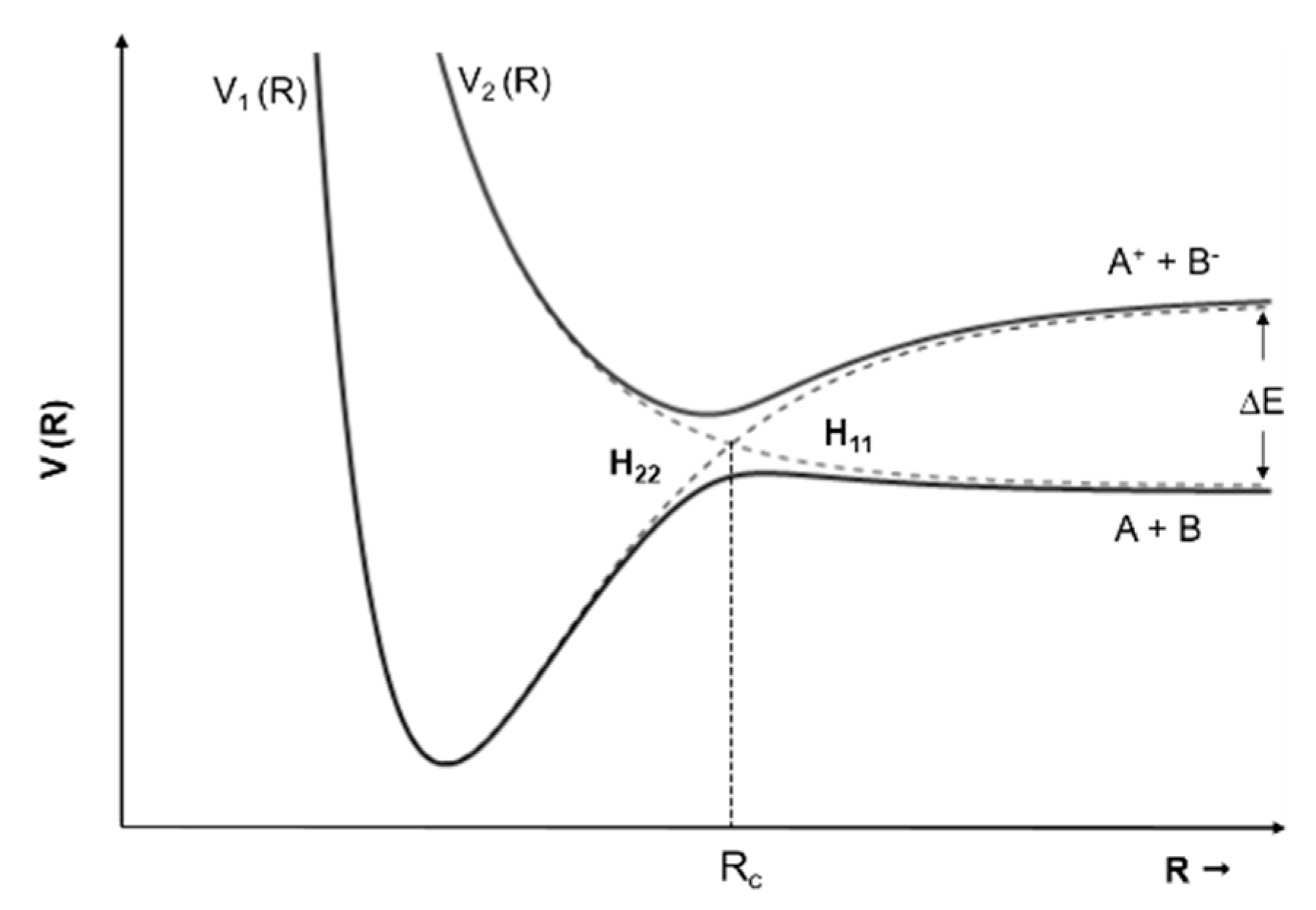
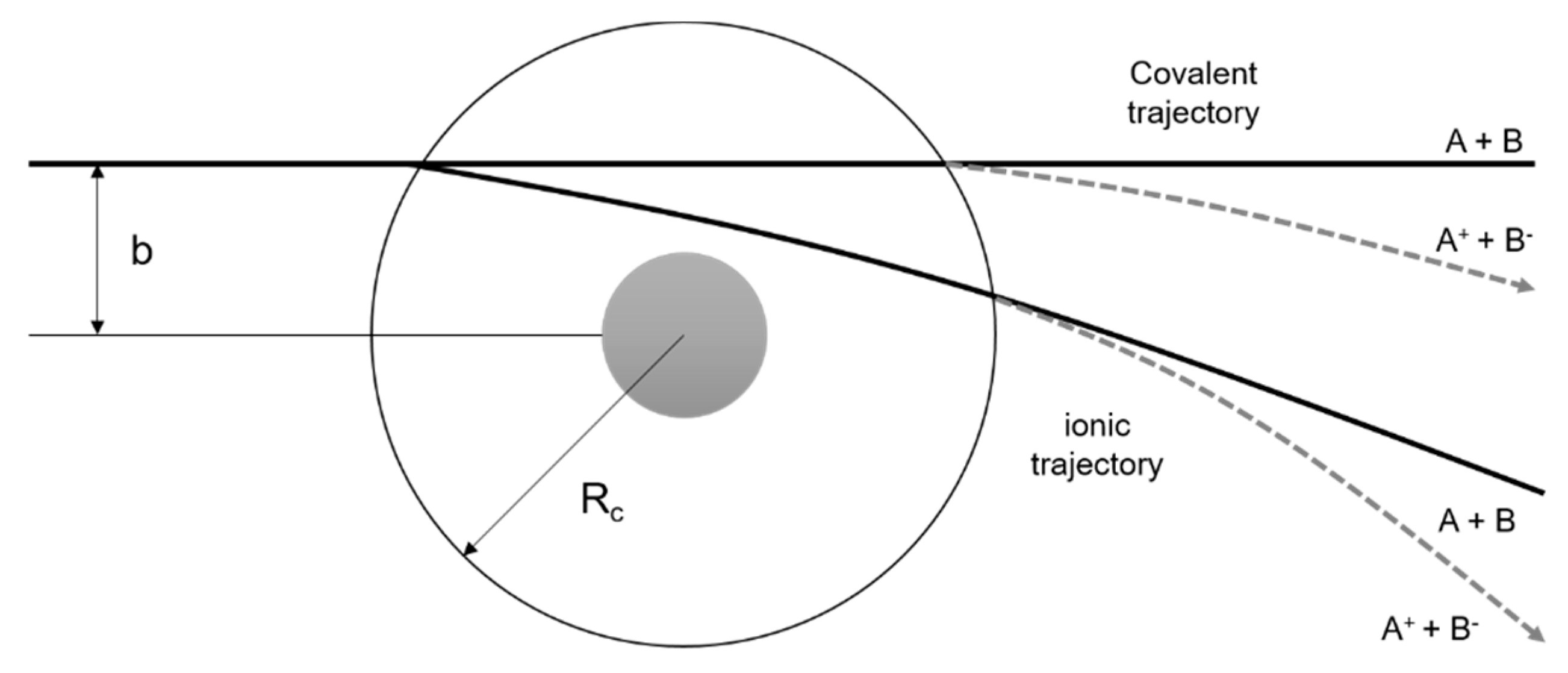
| m/z | 4(5)NI | 2NI | Me4NI | Me5NI | IMI |
|---|---|---|---|---|---|
| 1 | H− | H− | H− | H− | H− |
| 12 | C− | ||||
| 13 | CH− | CH− | |||
| 14 | CH2− | ||||
| 15 | CH3−/NH− | CH3−/NH− | |||
| 16 | O−/NH2− | O−/NH2− | O−/NH2− | O−/NH2− | O−/NH2− |
| 17 | OH− | OH− | OH− | OH− | |
| 24 | C2− | C2− | C2− | ||
| 25 | C2H− | C2H− | C2H− | C2H− | |
| 26 | CN− | CN− | CN− | CN− | CN− |
| 39 | C2HN−/C3H3− | C2HN−/C3H3− | C2HN−/C3H3− | C2HN−/C3H3− | |
| 40 | C2H2N− | C2H2N− | C2H2N− | C2H2N− | C2H2N− |
| 41 | C2H3N−/CHN− | ||||
| 42 | CNO−/CH2N− | CNO−/CH2N− | CNO−/CH2N− | CNO−/CH2N− | |
| 46 | NO2− | NO2− | NO2− | NO2− | |
| 50 | C3N− | ||||
| 64 | (4(5)NI–H3NO2)− | ||||
| 65 | (4(5)NI–H2NO2)− | ||||
| 66 | (4(5)NI–HNO2)− | (2NI–HNO2)− | (Me4NI–CH3NO2)− | (Me5NI–CH3NO2)− | |
| 67 | (4(5)NI–NO2)− | (2NI–NO2)− | (IMI–H)− | ||
| 68 | (IMI–H)− * | ||||
| 81 | (Me4NI–NO2)− | (Me5NI–NO2)− | |||
| 82 | (4(5)NI–HNO)− | (Me4NI–CH3NO)− | (Me5NI–CH3NO)− | ||
| 83 | (4(5)NI–NO)− | (2NI–NO)− | |||
| 96 | (4(5)NI–OH)− | (2NI–OH)− | (Me4NI–HNO)− | (Me5NI–HNO)− | |
| 112 | (4(5)NI–H)− | (2NI–H)− | (Me4NI–CH3)− | (Me5NI–CH3)− | |
| 113 | 4(5)NI− | 2NI− | |||
| 114 | 4(5)NI− * | ||||
| 127 | Me4NI− | Me5NI− | |||
| 128 | Me4NI− * | Me5NI− * |
| IMI | 2NI | 4NI | Me4NI | Me5NI |
|---|---|---|---|---|
| HOMO πC=C −9.3 eV 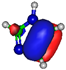 | HOMO πC=C −10.4 eV 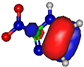 | HOMO πC=N −11.8 eV 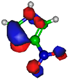 | HOMO πC=N −11.4 eV 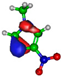 | HOMO πC=N/C=C −11.4 eV 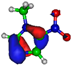 |
| LUMO π*ring 5.2 eV 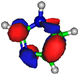 | LUMO π*ring 4.9 eV 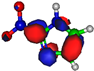 | LUMO σ*C=C 6.5 eV  | LUMO σ*C=C 7.3 eV  | LUMO σ*C=C 8.9 eV 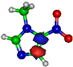 |
| π*C=C 7.2 eV  | π*C=C 6.1 eV 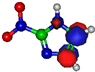 | π*C=C 10.9 eV  | π*C=C 11.3 eV 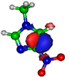 | π*C=C 11.7 eV 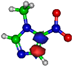 |
| π*CH 8.3 eV  | π*CH 7.4 eV  | π*C=C 13.2 eV 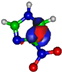 | π*C=C 14.9 eV 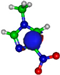 | π*C=C 15.4 eV 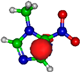 |
| σ*CH 10.1 eV 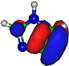 | σ*CH 9.0 eV  |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mendes, M.; García, G.; Bacchus-Montabonel, M.-C.; Limão-Vieira, P. Electron Transfer Induced Decomposition in Potassium–Nitroimidazoles Collisions: An Experimental and Theoretical Work. Int. J. Mol. Sci. 2019, 20, 6170. https://doi.org/10.3390/ijms20246170
Mendes M, García G, Bacchus-Montabonel M-C, Limão-Vieira P. Electron Transfer Induced Decomposition in Potassium–Nitroimidazoles Collisions: An Experimental and Theoretical Work. International Journal of Molecular Sciences. 2019; 20(24):6170. https://doi.org/10.3390/ijms20246170
Chicago/Turabian StyleMendes, Mónica, Gustavo García, Marie-Christine Bacchus-Montabonel, and Paulo Limão-Vieira. 2019. "Electron Transfer Induced Decomposition in Potassium–Nitroimidazoles Collisions: An Experimental and Theoretical Work" International Journal of Molecular Sciences 20, no. 24: 6170. https://doi.org/10.3390/ijms20246170
APA StyleMendes, M., García, G., Bacchus-Montabonel, M.-C., & Limão-Vieira, P. (2019). Electron Transfer Induced Decomposition in Potassium–Nitroimidazoles Collisions: An Experimental and Theoretical Work. International Journal of Molecular Sciences, 20(24), 6170. https://doi.org/10.3390/ijms20246170





