Omega-3 Polyunsaturated Fatty Acids and Stroke Burden
Abstract
1. Introduction
2. The Impact of Dietary Intake of n-3 PUFAs on Stroke
2.1. United States of America
2.2. Europe
2.3. Eastern Asia
2.4. Review of the Epidemiological Studies Worldwide and Their Limitations
3. Therapeutic Use of n-PUFAs and Stroke Burden
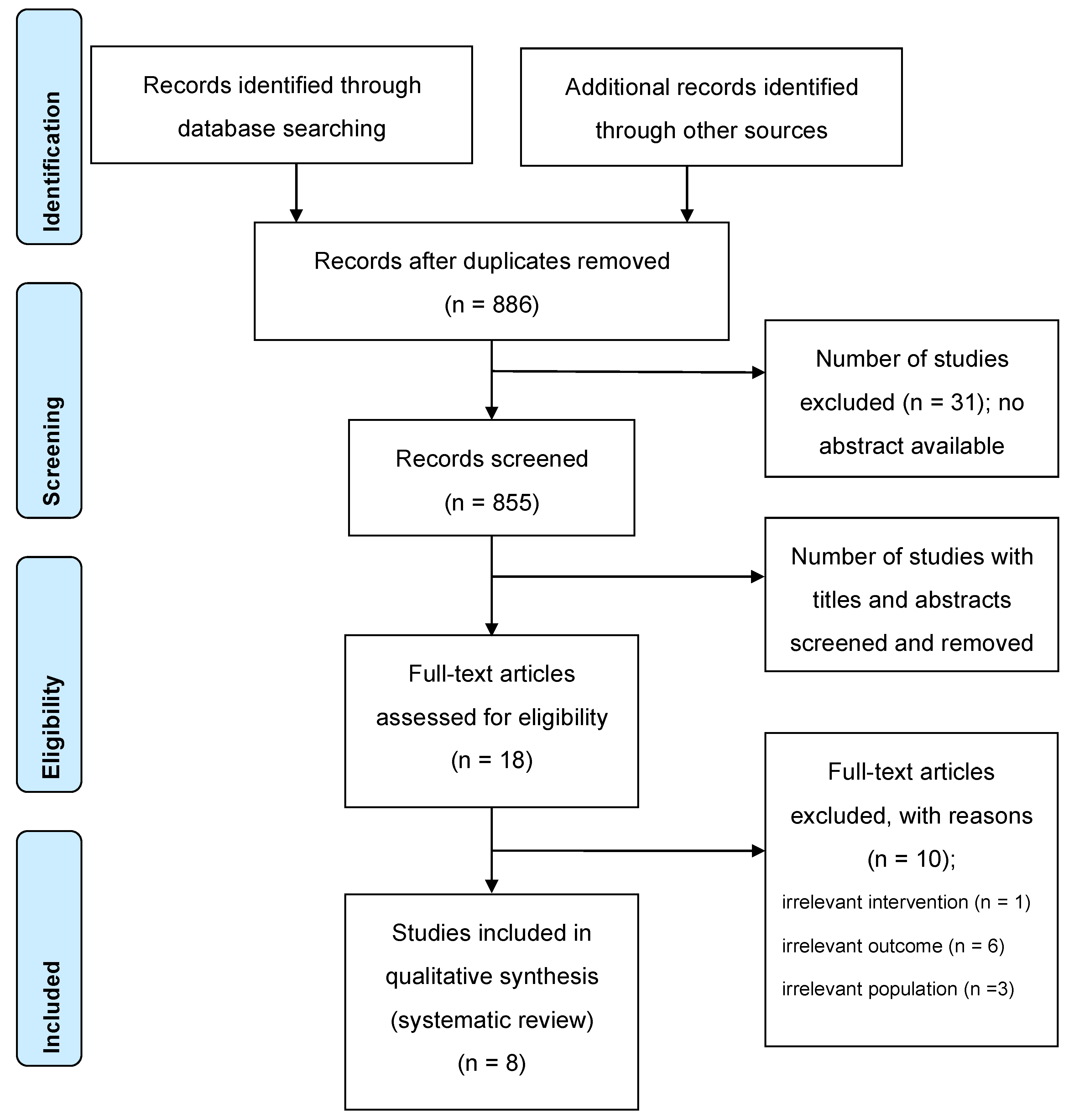
3.1. Systematic Literature Review
3.2. EPA/DHA
3.3. EPA
3.4. Insights into Controversial Results from Clinical Trials
4. Levels of n-3 PUFAs as Surrogate Markers of Stroke
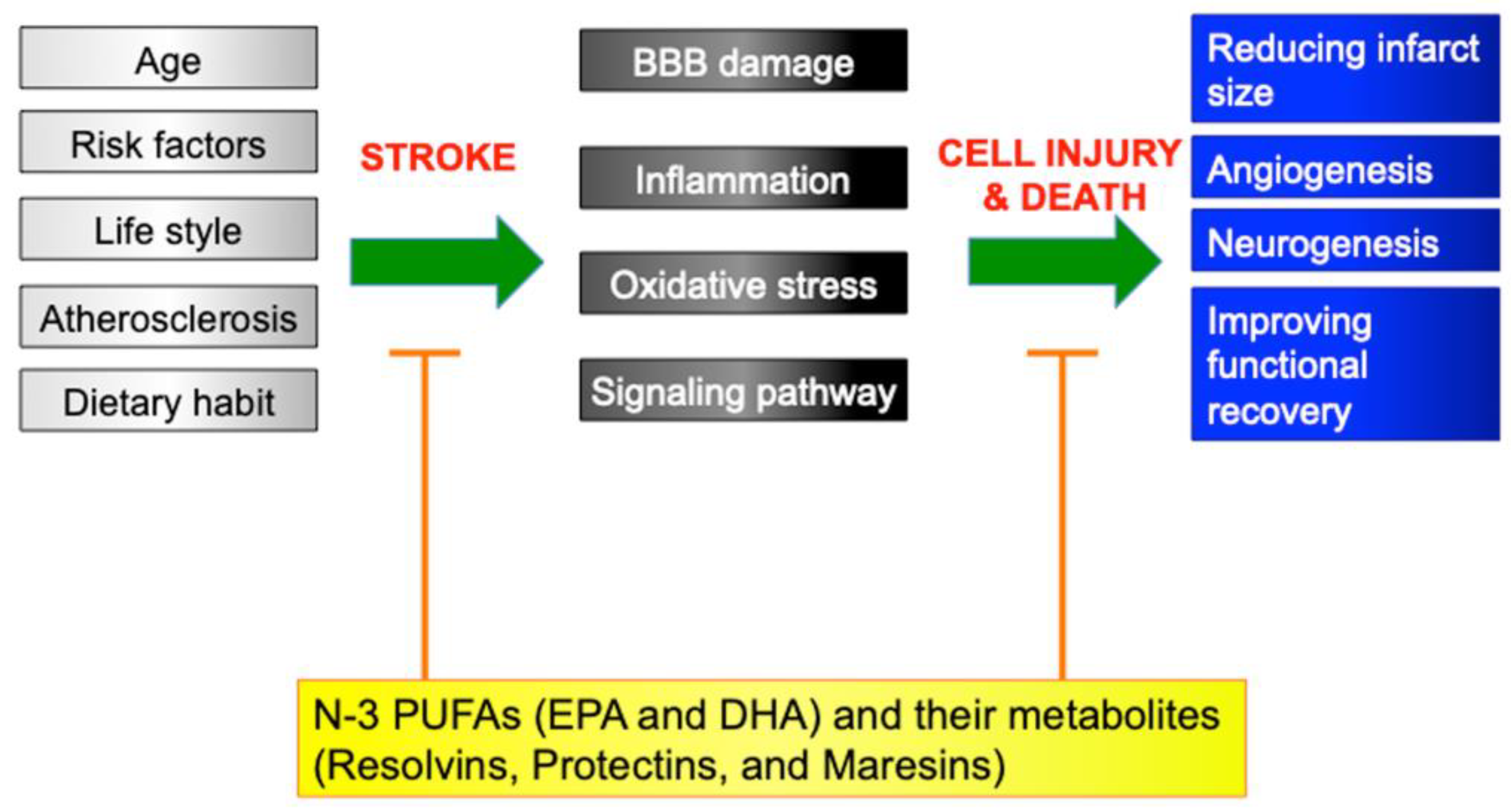
5. Biological Effects of Omega-3 PUFAs for Stroke
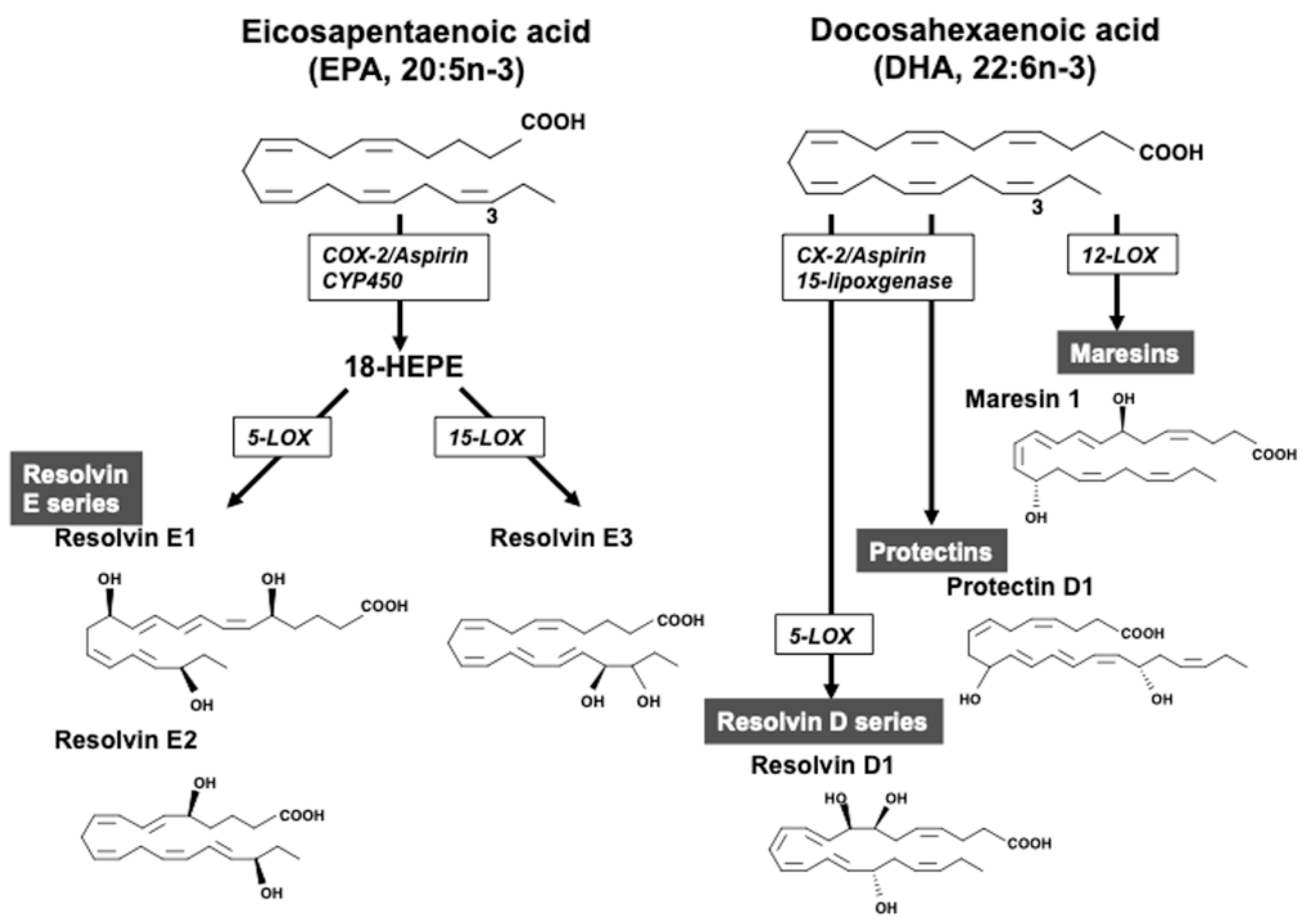
5.1. Omega-3 PUFAs and Their Metabolites
5.2. Treatment with Omega-3 PUFAs in Experimental Stroke
5.3. Role of Omega-3 PUFAs and Their Metabolites before and after Stroke
6. Conclusions
Funding
Conflicts of Interest
References
- Roth, G.A.; Johnson, C.; Abajobir, A.; Abd-Allah, F.; Abera, S.F.; Abyu, G.; Ahmed, M.; Aksut, B.; Alam, T.; Alam, K.; et al. Global, regional, and national burden of cardiovascular diseases for 10 causes, 1990 to 2015. J. Am. Coll. Cardiol. 2017, 70, 1–25. [Google Scholar] [CrossRef] [PubMed]
- Feigin, V.L.; Norrving, B.; Mensah, G.A. Global burden of stroke. Circ. Res. 2017, 120, 439–448. [Google Scholar] [CrossRef] [PubMed]
- GBD 2015 Neurological Disorders Collaborator Group. Global, regional, and national burden of neurological disorders during 1990-2015: A systematic analysis for the Global Burden of Disease Study 2015. Lancet Neurol. 2017, 16, 877–897. [Google Scholar] [CrossRef]
- Boehme, A.K.; Esenwa, C.; Elkind, M.S. Stroke risk factors, genetics, and prevention. Circ. Res. 2017, 120, 472–495. [Google Scholar] [CrossRef] [PubMed]
- Hata, J.; Kiyohara, Y. Epidemiology of stroke and coronary artery disease in Asia. Circ. J. 2013, 77, 1923–1932. [Google Scholar] [CrossRef] [PubMed]
- Radu, R.A.; Terecoasa, E.O.; Bajenaru, O.A.; Tiu, C. Etiologic classification of ischemic stroke: Where do we stand? Clin. Neurol. Neurosurg. 2017, 159, 93–106. [Google Scholar] [CrossRef]
- Chiuve, S.E.; Rexrode, K.M.; Spiegelman, D.; Logroscino, G.; Manson, J.E.; Rimm, E.B. Primary prevention of stroke by healthy lifestyle. Circulation 2008, 118, 947–954. [Google Scholar] [CrossRef]
- Larsson, S.C.; Akesson, A.; Wolk, A. Healthy diet and lifestyle and risk of stroke in a prospective cohort of women. Neurology 2014, 83, 1699–1704. [Google Scholar] [CrossRef]
- Zhang, Y.; Tuomilehto, J.; Jousilahti, P.; Wang, Y.; Antikainen, R.; Hu, G. Lifestyle factors on the risks of ischemic and hemorrhagic stroke. Arch. Intern. Med. 2011, 171, 1811–1818. [Google Scholar] [CrossRef]
- Lee, C.D.; Folsom, A.R.; Blair, S.N. Physical activity and stroke risk: A meta-analysis. Stroke 2003, 34, 2475–2481. [Google Scholar] [CrossRef]
- Kaplan, R.C.; Tirschwell, D.L.; Longstreth, W.T., Jr.; Manolio, T.A.; Heckbert, S.R.; Lefkowitz, D.; El-Saed, A.; Psaty, B.M. Vascular events, mortality, and preventive therapy following ischemic stroke in the elderly. Neurology 2005, 65, 835–842. [Google Scholar] [CrossRef] [PubMed]
- Niewada, M.; Michel, P. Lifestyle modification for stroke prevention: Facts and fiction. Curr. Opin. Neurol. 2016, 29, 9–13. [Google Scholar] [CrossRef] [PubMed]
- Mozaffarian, D.; Benjamin, E.J.; Go, A.S.; Arnett, D.K.; Blaha, M.J.; Cushman, M.; de Ferranti, S.; Despres, J.P.; Fullerton, H.J.; Howard, V.J.; et al. Heart disease and stroke statistics—2015 update: A report from the American Heart Association. Circulation 2015, 131, e29–e322. [Google Scholar] [CrossRef] [PubMed]
- Zeng, R.; Xu, C.H.; Xu, Y.N.; Wang, Y.L.; Wang, M. The effect of folate fortification on folic acid-based homocysteine-lowering intervention and stroke risk: A meta-analysis. Public Health Nutr. 2015, 18, 1514–1521. [Google Scholar] [CrossRef]
- Adebamowo, S.N.; Spiegelman, D.; Flint, A.J.; Willett, W.C.; Rexrode, K.M. Intakes of magnesium, potassium, and calcium and the risk of stroke among men. Int. J. Stroke 2015, 10, 1093–1100. [Google Scholar] [CrossRef]
- Huo, Y.; Li, J.; Qin, X.; Huang, Y.; Wang, X.; Gottesman, R.F.; Tang, G.; Wang, B.; Chen, D.; He, M.; et al. Efficacy of folic acid therapy in primary prevention of stroke among adults with hypertension in China: The CSPPT randomized clinical trial. JAMA 2015, 313, 1325–1335. [Google Scholar] [CrossRef]
- Hu, D.; Huang, J.; Wang, Y.; Zhang, D.; Qu, Y. Fruits and vegetables consumption and risk of stroke: A meta-analysis of prospective cohort studies. Stroke 2014, 45, 1613–1619. [Google Scholar] [CrossRef]
- Lagiou, P.; Sandin, S.; Lof, M.; Trichopoulos, D.; Adami, H.O.; Weiderpass, E. Low carbohydrate-high protein diet and incidence of cardiovascular diseases in Swedish women: Prospective cohort study. BMJ 2012, 344, e4026. [Google Scholar] [CrossRef]
- Thies, F.; Garry, J.M.; Yaqoob, P.; Rerkasem, K.; Williams, J.; Shearman, C.P.; Gallagher, P.J.; Calder, P.C.; Grimble, R.F. Association of n-3 polyunsaturated fatty acids with stability of atherosclerotic plaques: A randomised controlled trial. Lancet 2003, 361, 477–485. [Google Scholar] [CrossRef]
- Satoh, N.; Shimatsu, A.; Kotani, K.; Sakane, N.; Yamada, K.; Suganami, T.; Kuzuya, H.; Ogawa, Y. Purified eicosapentaenoic acid reduces small dense LDL, remnant lipoprotein particles, and C-reactive protein in metabolic syndrome. Diabetes Care 2007, 30, 144–146. [Google Scholar] [CrossRef]
- Kramer, H.J.; Stevens, J.; Grimminger, F.; Seeger, W. Fish oil fatty acids and human platelets: Dose-dependent decrease in dienoic and increase in trienoic thromboxane generation. Biochem. Pharmacol. 1996, 52, 1211–1217. [Google Scholar] [CrossRef]
- Watanabe, Y.; Tatsuno, I. Omega-3 polyunsaturated fatty acids for cardiovascular diseases: Present, past and future. Expert Rev. Clin. Pharmacol. 2017, 10, 865–873. [Google Scholar] [CrossRef] [PubMed]
- Calder, P.C. The role of marine omega-3 (n-3) fatty acids in inflammatory processes, atherosclerosis and plaque stability. Mol. Nutr. Food Res. 2012, 56, 1073–1080. [Google Scholar] [CrossRef] [PubMed]
- Konishi, T.; Sunaga, D.; Funayama, N.; Yamamoto, T.; Murakami, H.; Hotta, D.; Nojima, M.; Tanaka, S. Eicosapentaenoic acid therapy is associated with decreased coronary plaque instability assessed using optical frequency domain imaging. Clin. Cardiol. 2019, 42, 618–628. [Google Scholar] [CrossRef] [PubMed]
- Morris, M.C.; Manson, J.E.; Rosner, B.; Buring, J.E.; Willett, W.C.; Hennekens, C.H. Fish consumption and cardiovascular disease in the physicians’ health study: A prospective study. Am. J. Epidemiol. 1995, 142, 166–175. [Google Scholar] [CrossRef]
- Iso, H.; Rexrode, K.M.; Stampfer, M.J.; Manson, J.E.; Colditz, G.A.; Speizer, F.E.; Hennekens, C.H.; Willett, W.C. Intake of fish and omega-3 fatty acids and risk of stroke in women. JAMA 2001, 285, 304–312. [Google Scholar] [CrossRef]
- He, K.; Rimm, E.B.; Merchant, A.; Rosner, B.A.; Stampfer, M.J.; Willett, W.C.; Ascherio, A. Fish consumption and risk of stroke in men. JAMA 2002, 288, 3130–3136. [Google Scholar] [CrossRef]
- de Goede, J.; Verschuren, W.M.; Boer, J.M.; Kromhout, D.; Geleijnse, J.M. Gender-specific associations of marine n-3 fatty acids and fish consumption with 10-year incidence of stroke. PLoS ONE 2012, 7, e33866. [Google Scholar] [CrossRef]
- Larsson, S.C.; Virtamo, J.; Wolk, A. Dietary fats and dietary cholesterol and risk of stroke in women. Atherosclerosis 2012, 221, 282–286. [Google Scholar] [CrossRef]
- Wallstrom, P.; Sonestedt, E.; Hlebowicz, J.; Ericson, U.; Drake, I.; Persson, M.; Gullberg, B.; Hedblad, B.; Wirfalt, E. Dietary fiber and saturated fat intake associations with cardiovascular disease differ by sex in the Malmo Diet and Cancer Cohort: A prospective study. PLoS ONE 2012, 7, e31637. [Google Scholar] [CrossRef]
- Veno, S.K.; Bork, C.S.; Jakobsen, M.U.; Lundbye-Christensen, S.; McLennan, P.L.; Bach, F.W.; Overvad, K.; Schmidt, E.B. Marine n-3 polyunsaturated fatty acids and the risk of ischemic stroke. Stroke 2019, 50, 274–282. [Google Scholar] [CrossRef] [PubMed]
- Sauvaget, C.; Nagano, J.; Allen, N.; Grant, E.J.; Beral, V. Intake of animal products and stroke mortality in the Hiroshima/Nagasaki Life Span Study. Int. J. Epidemiol. 2003, 32, 536–543. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, Y.; Ueshima, H.; Okamura, T.; Kadowaki, T.; Hayakawa, T.; Kita, Y.; Tamaki, S.; Okayama, A.; Group, N.D.R. Association between fish consumption and all-cause and cause-specific mortality in Japan: NIPPON DATA80, 1980–1999. Am. J. Med. 2005, 118, 239–245. [Google Scholar] [CrossRef] [PubMed]
- Yamagishi, K.; Iso, H.; Date, C.; Fukui, M.; Wakai, K.; Kikuchi, S.; Inaba, Y.; Tanabe, N.; Tamakoshi, A.; Japan Collaborative Cohort Study for Evaluation of Cancer Risk Study Group. Fish, omega-3 polyunsaturated fatty acids, and mortality from cardiovascular diseases in a nationwide community-based cohort of Japanese men and women the JACC (Japan Collaborative Cohort Study for Evaluation of Cancer Risk) Study. J. Am. Coll. Cardiol. 2008, 52, 988–996. [Google Scholar] [PubMed]
- Yuan, J.M.; Ross, R.K.; Gao, Y.T.; Yu, M.C. Fish and shellfish consumption in relation to death from myocardial infarction among men in Shanghai, China. Am. J. Epidemiol. 2001, 154, 809–816. [Google Scholar] [CrossRef] [PubMed]
- Takata, Y.; Zhang, X.; Li, H.; Gao, Y.T.; Yang, G.; Gao, J.; Cai, H.; Xiang, Y.B.; Zheng, W.; Shu, X.O. Fish intake and risks of total and cause-specific mortality in 2 population-based cohort studies of 134,296 men and women. Am. J. Epidemiol. 2013, 178, 46–57. [Google Scholar] [CrossRef]
- Koh, A.S.; Pan, A.; Wang, R.; Odegaard, A.O.; Pereira, M.A.; Yuan, J.M.; Koh, W.P. The association between dietary omega-3 fatty acids and cardiovascular death: The Singapore Chinese Health Study. Eur. J. Prev. Cardiol. 2015, 22, 364–372. [Google Scholar] [CrossRef]
- Larsson, S.C.; Orsini, N.; Wolk, A. Long-chain omega-3 polyunsaturated fatty acids and risk of stroke: A meta-analysis. Eur. J. Epidemiol. 2012, 27, 895–901. [Google Scholar] [CrossRef]
- Qin, Z.Z.; Xu, J.Y.; Chen, G.C.; Ma, Y.X.; Qin, L.Q. Effects of fatty and lean fish intake on stroke risk: A meta-analysis of prospective cohort studies. Lipids Health Dis. 2018, 17, 264. [Google Scholar] [CrossRef]
- Iso, H. Changes in coronary heart disease risk among Japanese. Circulation 2008, 118, 2725–2729. [Google Scholar] [CrossRef]
- Stark, K.D.; Van Elswyk, M.E.; Higgins, M.R.; Weatherford, C.A.; Salem, N., Jr. Global survey of the omega-3 fatty acids, docosahexaenoic acid and eicosapentaenoic acid in the blood stream of healthy adults. Prog. Lipid Res. 2016, 63, 132–152. [Google Scholar] [CrossRef] [PubMed]
- Luscher, T.F. In search of the right word: A statement of the HEART Group on scientific language. Eur. Heart J. 2013, 34, 7–9. [Google Scholar] [CrossRef] [PubMed]
- Fukushima, H.; Takemura, K.; Suzuki, H.; Koga, F. Impact of sarcopenia as a prognostic biomarker of bladder cancer. Int. J. Mol. Sci. 2018, 19, 2999. [Google Scholar] [CrossRef] [PubMed]
- GISSI-Prevenzione Investigators. Dietary supplementation with n-3 polyunsaturated fatty acids and vitamin E after myocardial infarction: Results of the GISSI-Prevenzione trial. Gruppo Italiano per lo Studio della Sopravvivenza nell’Infarto miocardico. Lancet 1999, 354, 447–455. [Google Scholar]
- Tavazzi, L.; Maggioni, A.P.; Marchioli, R.; Barlera, S.; Franzosi, M.G.; Latini, R.; Lucci, D.; Nicolosi, G.L.; Porcu, M.; Tognoni, G.; et al. Effect of n-3 polyunsaturated fatty acids in patients with chronic heart failure (the GISSI-HF trial): A randomised, double-blind, placebo-controlled trial. Lancet 2008, 372, 1223–1230. [Google Scholar]
- Tavazzi, L.; Maggioni, A.P.; Marchioli, R.; Barlera, S.; Franzosi, M.G.; Latini, R.; Lucci, D.; Nicolosi, G.L.; Porcu, M.; Tognoni, G.; et al. Effect of rosuvastatin in patients with chronic heart failure (the GISSI-HF trial): A randomised, double-blind, placebo-controlled trial. Lancet 2008, 372, 1231–1239. [Google Scholar]
- Galan, P.; Kesse-Guyot, E.; Czernichow, S.; Briancon, S.; Blacher, J.; Hercberg, S.; SU.FOL.OM3 Collaborative Group. Effects of B vitamins and omega 3 fatty acids on cardiovascular diseases: A randomised placebo controlled trial. BMJ 2010, 341, c6273. [Google Scholar] [CrossRef]
- Investigators, O.T.; Bosch, J.; Gerstein, H.C.; Dagenais, G.R.; Diaz, R.; Dyal, L.; Jung, H.; Maggiono, A.P.; Probstfield, J.; Ramachandran, A.; et al. n-3 fatty acids and cardiovascular outcomes in patients with dysglycemia. N. Engl. J. Med. 2012, 367, 309–318. [Google Scholar] [CrossRef]
- Manson, J.E.; Cook, N.R.; Lee, I.M.; Christen, W.; Bassuk, S.S.; Mora, S.; Gibson, H.; Albert, C.M.; Gordon, D.; Copeland, T.; et al. Marine n-3 fatty acids and prevention of cardiovascular disease and cancer. N. Engl. J. Med. 2019, 380, 23–32. [Google Scholar] [CrossRef]
- ASCEND Study Collaborative Group; Bowman, L.; Mafham, M.; Wallendszus, K.; Stevens, W.; Buck, G.; Barton, J.; Murphy, K.; Aung, T.; Haynes, R.; et al. Effects of n-3 fatty acid supplements in diabetes mellitus. N. Engl. J. Med. 2018, 379, 1540–1550. [Google Scholar]
- Yokoyama, M.; Origasa, H.; Matsuzaki, M.; Matsuzawa, Y.; Saito, Y.; Ishikawa, Y.; Oikawa, S.; Sasaki, J.; Hishida, H.; Itakura, H.; et al. Effects of eicosapentaenoic acid on major coronary events in hypercholesterolaemic patients (JELIS): A randomised open-label, blinded endpoint analysis. Lancet 2007, 369, 1090–1098. [Google Scholar] [CrossRef]
- Tanaka, K.; Ishikawa, Y.; Yokoyama, M.; Origasa, H.; Matsuzaki, M.; Saito, Y.; Matsuzawa, Y.; Sasaki, J.; Oikawa, S.; Hishida, H.; et al. Reduction in the recurrence of stroke by eicosapentaenoic acid for hypercholesterolemic patients: Subanalysis of the JELIS trial. Stroke 2008, 39, 2052–2058. [Google Scholar] [CrossRef] [PubMed]
- Bhatt, D.L.; Steg, P.G.; Brinton, E.A.; Jacobson, T.A.; Miller, M.; Tardif, J.C.; Ketchum, S.B.; Doyle, R.T., Jr.; Murphy, S.A.; Soni, P.N.; et al. Rationale and design of REDUCE-IT: Reduction of cardiovascular events with Icosapent Ethyl-intervention trial. Clin. Cardiol. 2017, 40, 138–148. [Google Scholar] [CrossRef] [PubMed]
- Bhatt, D.L.; Steg, P.G.; Miller, M.; Brinton, E.A.; Jacobson, T.A.; Ketchum, S.B.; Doyle, R.T., Jr.; Juliano, R.A.; Jiao, L.; Granowitz, C.; et al. Cardiovascular risk reduction with Icosapent Ethyl for Hypertriglyceridemia. N. Engl. J. Med. 2019, 380, 11–22. [Google Scholar] [CrossRef] [PubMed]
- Hosogoe, N.; Ishikawa, S.; Yokoyama, N.; Kozuma, K.; Isshiki, T. Add-on antiplatelet effects of Eicosapentaenoic acid with tailored dose setting in patients on dual antiplatelet therapy. Int. Heart J. 2017, 58, 481–485. [Google Scholar] [CrossRef]
- Nicholls, S.J.; Lincoff, A.M.; Bash, D.; Ballantyne, C.M.; Barter, P.J.; Davidson, M.H.; Kastelein, J.J.P.; Koenig, W.; McGuire, D.K.; Mozaffarian, D.; et al. Assessment of omega-3 carboxylic acids in statin-treated patients with high levels of triglycerides and low levels of high-density lipoprotein cholesterol: Rationale and design of the STRENGTH trial. Clin. Cardiol. 2018, 41, 1281–1288. [Google Scholar] [CrossRef]
- Sabatine, M.S.; Giugliano, R.P.; Keech, A.C.; Honarpour, N.; Wiviott, S.D.; Murphy, S.A.; Kuder, J.F.; Wang, H.; Liu, T.; Wasserman, S.M.; et al. Investigators, Evolocumab and clinical outcomes in patients with cardiovascular disease. N. Engl. J. Med. 2017, 376, 1713–1722. [Google Scholar] [CrossRef]
- Ridker, P.M.; Everett, B.M.; Thuren, T.; MacFadyen, J.G.; Chang, W.H.; Ballantyne, C.; Fonseca, F.; Nicolau, J.; Koenig, W.; Anker, S.D.; et al. Antiinflammatory therapy with Canakinumab for atherosclerotic disease. N. Engl. J. Med. 2017, 377, 1119–1131. [Google Scholar] [CrossRef]
- Yang, B.; Ren, X.L.; Huang, H.; Guo, X.J.; Ma, A.G.; Li, D. Circulating long-chain n-3 polyunsaturated fatty acid and incidence of stroke: A meta-analysis of prospective cohort studies. Oncotarget 2017, 8, 83781–83791. [Google Scholar] [CrossRef]
- Park, Y.; Park, S.; Yi, H.; Kim, H.Y.; Kang, S.J.; Kim, J.; Ahn, H. Low level of n-3 polyunsaturated fatty acids in erythrocytes is a risk factor for both acute ischemic and hemorrhagic stroke in Koreans. Nutr. Res. 2009, 29, 825–830. [Google Scholar] [CrossRef]
- Suda, S.; Katsumata, T.; Okubo, S.; Kanamaru, T.; Suzuki, K.; Watanabe, Y.; Katsura, K.; Katayama, Y. Low serum n-3 polyunsaturated fatty acid/n-6 polyunsaturated fatty acid ratio predicts neurological deterioration in Japanese patients with acute ischemic stroke. Cerebrovasc. Dis. 2013, 36, 388–393. [Google Scholar] [CrossRef]
- Ueno, Y.; Tanaka, R.; Yamashiro, K.; Miyamoto, N.; Hira, K.; Kurita, N.; Sakurai, M.; Urabe, T.; Shimada, K.; Miyazaki, T.; et al. Age stratification and impact of Eicosapentaenoic acid and Docosahexaenoic acid to Arachidonic acid ratios in ischemic stroke patients. J. Atheroscler. Thromb. 2018, 25, 593–605. [Google Scholar] [CrossRef] [PubMed]
- Harris, W.S.; Tintle, N.L.; Etherton, M.R.; Vasan, R.S. Erythrocyte long-chain omega-3 fatty acid levels are inversely associated with mortality and with incident cardiovascular disease: The Framingham Heart Study. J. Clin. Lipidol. 2018, 12, 718–727. e6. [Google Scholar] [CrossRef] [PubMed]
- Saber, H.; Yakoob, M.Y.; Shi, P.; Longstreth, W.T., Jr.; Lemaitre, R.N.; Siscovick, D.; Rexrode, K.M.; Willett, W.C.; Mozaffarian, D. Omega-3 fatty acids and incident ischemic stroke and its Atherothrombotic and Cardioembolic subtypes in 3 US cohorts. Stroke 2017, 48, 2678–2685. [Google Scholar] [CrossRef] [PubMed]
- Song, T.J.; Cho, H.J.; Chang, Y.; Choi, K.; Jung, A.R.; Youn, M.; Shin, M.J.; Kim, Y.J. Low plasma proportion of omega 3-polyunsaturated fatty acids predicts poor outcome in acute non-cardiogenic ischemic stroke patients. J. Stroke 2015, 17, 168–176. [Google Scholar] [CrossRef]
- Hodson, L.; Skeaff, C.M.; Fielding, B.A. Fatty acid composition of adipose tissue and blood in humans and its use as a biomarker of dietary intake. Prog. Lipid Res. 2008, 47, 348–380. [Google Scholar] [CrossRef]
- Araujo, P.; Belghit, I.; Aarsaether, N.; Espe, M.; Lucena, E.; Holen, E. The effect of omega-3 and omega-6 polyunsaturated fatty acids on the production of Cyclooxygenase and Lipoxygenase metabolites by human umbilical vein endothelial cells. Nutrients 2019, 11, 966. [Google Scholar] [CrossRef]
- Calder, P.C. Intravenous lipid emulsions to deliver bioactive omega-3 fatty acids for improved patient outcomes. Mar. Drugs 2019, 17, 274. [Google Scholar] [CrossRef]
- Allaire, J.; Couture, P.; Leclerc, M.; Charest, A.; Marin, J.; Lepine, M.C.; Talbot, D.; Tchernof, A.; Lamarche, B. A randomized, crossover, head-to-head comparison of eicosapentaenoic acid and docosahexaenoic acid supplementation to reduce inflammation markers in men and women: The Comparing EPA to DHA (ComparED) Study. Am. J. Clin. Nutr. 2016, 104, 280–287. [Google Scholar] [CrossRef]
- Halade, G.V.; Rahman, M.M.; Bhattacharya, A.; Barnes, J.L.; Chandrasekar, B.; Fernandes, G. Docosahexaenoic acid-enriched fish oil attenuates kidney disease and prolongs median and maximal life span of autoimmune lupus-prone mice. J. Immunol. 2010, 184, 5280–5286. [Google Scholar] [CrossRef]
- Miyata, J.; Arita, M. Role of omega-3 fatty acids and their metabolites in asthma and allergic diseases. Allergol. Int. 2015, 64, 27–34. [Google Scholar] [CrossRef]
- Duvall, M.G.; Levy, B.D. DHA- and EPA-derived resolvins, protectins, and maresins in airway inflammation. Eur. J. Pharmacol. 2016, 785, 144–155. [Google Scholar] [CrossRef] [PubMed]
- Belayev, L.; Hong, S.H.; Menghani, H.; Marcell, S.J.; Obenaus, A.; Freitas, R.S.; Khoutorova, L.; Balaszczuk, V.; Jun, B.; Oria, R.B.; et al. Docosanoids promote neurogenesis and angiogenesis, blood-brain barrier integrity, penumbra protection, and neurobehavioral recovery after experimental ischemic stroke. Mol. Neurobiol. 2018, 55, 7090–7106. [Google Scholar] [CrossRef] [PubMed]
- Calandria, J.M.; Asatryan, A.; Balaszczuk, V.; Knott, E.J.; Jun, B.K.; Mukherjee, P.K.; Belayev, L.; Bazan, N.G. NPD1-mediated stereoselective regulation of BIRC3 expression through cREL is decisive for neural cell survival. Cell Death Differ. 2015, 22, 1363–1377. [Google Scholar] [CrossRef] [PubMed]
- Nielsen, M.M.; Lambertsen, K.L.; Clausen, B.H.; Meyer, M.; Bhandari, D.R.; Larsen, S.T.; Poulsen, S.S.; Spengler, B.; Janfelt, C.; Hansen, H.S. Mass spectrometry imaging of biomarker lipids for phagocytosis and signalling during focal cerebral ischaemia. Sci. Rep. 2016, 6, 39571. [Google Scholar] [CrossRef] [PubMed]
- Serhan, C.N.; Levy, B.D. Resolvins in inflammation: Emergence of the pro-resolving superfamily of mediators. J. Clin. Investig. 2018, 128, 2657–2669. [Google Scholar] [CrossRef] [PubMed]
- Gonzalo-Gobernado, R.; Ayuso, M.I.; Sansone, L.; Bernal-Jimenez, J.J.; Ramos-Herrero, V.D.; Sanchez-Garcia, E.; Ramos, T.L.; Abia, R.; Muriana, F.J.G.; Bermudez, B.; et al. Neuroprotective effects of diets containing olive oil and DHA/EPA in a mouse model of cerebral ischemia. Nutrients 2019, 11, 1109. [Google Scholar] [CrossRef] [PubMed]
- Luo, C.; Ren, H.; Yao, X.; Shi, Z.; Liang, F.; Kang, J.X.; Wan, J.B.; Pei, Z.; Su, K.P.; Su, H. Enriched brain omega-3 polyunsaturated fatty acids confer neuroprotection against microinfarction. EBioMedicine 2018, 32, 50–61. [Google Scholar] [CrossRef]
- Ueda, M.; Inaba, T.; Nito, C.; Kamiya, N.; Katayama, Y. Therapeutic impact of eicosapentaenoic acid on ischemic brain damage following transient focal cerebral ischemia in rats. Brain Res. 2013, 1519, 95–104. [Google Scholar] [CrossRef]
- Sumiyoshi, M.; Satomi, J.; Kitazato, K.T.; Yagi, K.; Shimada, K.; Kurashiki, Y.; Korai, M.; Miyamoto, T.; Suzue, R.; Kuwayama, K.; et al. PPARgamma-dependent and -independent inhibition of the HMGB1/TLR9 pathway by Eicosapentaenoic acid attenuates ischemic brain damage in ovariectomized rats. J. Stroke Cerebrovasc. Dis. 2015, 24, 1187–1195. [Google Scholar] [CrossRef]
- Berressem, D.; Koch, K.; Franke, N.; Klein, J.; Eckert, G.P. Intravenous treatment with a long-chain omega-3 lipid emulsion provides neuroprotection in a Murine model of ischemic stroke—A pilot study. PLoS ONE 2016, 11, e0167329. [Google Scholar] [CrossRef]
- Jiang, X.; Pu, H.; Hu, X.; Wei, Z.; Hong, D.; Zhang, W.; Gao, Y.; Chen, J.; Shi, Y. A post-stroke therapeutic regimen with omega-3 polyunsaturated fatty acids that promotes white matter integrity and beneficial microglial responses after cerebral ischemia. Transl. Stroke Res. 2016, 7, 548–561. [Google Scholar] [CrossRef] [PubMed]
- Cai, M.; Zhang, W.; Weng, Z.; Stetler, R.A.; Jiang, X.; Shi, Y.; Gao, Y.; Chen, J. Promoting neurovascular recovery in aged mice after ischemic stroke—Prophylactic effect of omega-3 polyunsaturated fatty acids. Aging Dis. 2017, 8, 531–545. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Zhang, H.; Pu, H.; Wang, G.; Li, W.; Leak, R.K.; Chen, J.; Liou, A.K.; Hu, X. n-3 PUFA supplementation benefits microglial responses to myelin pathology. Sci. Rep. 2014, 4, 7458. [Google Scholar] [CrossRef]
- Zhang, M.; Wang, S.; Mao, L.; Leak, R.K.; Shi, Y.; Zhang, W.; Hu, X.; Sun, B.; Cao, G.; Gao, Y.; et al. Omega-3 fatty acids protect the brain against ischemic injury by activating Nrf2 and upregulating heme oxygenase 1. J. Neurosci. 2014, 34, 1903–1915. [Google Scholar] [CrossRef] [PubMed]
- Belayev, L.; Khoutorova, L.; Atkins, K.D.; Eady, T.N.; Hong, S.; Lu, Y.; Obenaus, A.; Bazan, N.G. Docosahexaenoic acid therapy of experimental ischemic stroke. Transl. Stroke Res. 2011, 2, 33–41. [Google Scholar] [CrossRef]
- Eady, T.N.; Belayev, L.; Khoutorova, L.; Atkins, K.D.; Zhang, C.; Bazan, N.G. Docosahexaenoic acid signaling modulates cell survival in experimental ischemic stroke penumbra and initiates long-term repair in young and aged rats. PLoS ONE 2012, 7, e46151. [Google Scholar] [CrossRef]
- Hong, S.H.; Khoutorova, L.; Bazan, N.G.; Belayev, L. Docosahexaenoic acid improves behavior and attenuates blood-brain barrier injury induced by focal cerebral ischemia in rats. Exp. Transl. Stroke Med. 2015, 7, 3. [Google Scholar] [CrossRef]
| Study | GISSI-P | JELIS | GISSI-HF | ORIGIN | SU.FOL.OM3 | ASCEND | VITAL | REDUCE-IT |
|---|---|---|---|---|---|---|---|---|
| Subjects | Post MI (< 3 mo) | T-C ≥6.5 mmol/L1, or LDL-C ≥4.4 mmol/L | Chronic heart failure (NYHA class II–IV) | ≥50 y, diabetes or prediabetes, and high risk of CVD | MI, unstable angina, and ischemic stroke | ≥40 y, with diabetes mellitus, and without CVD | Men ≥50 y, women ≥55 y | ≥45 y with established CVD or ≥50 y with diabetes and ≥1 additional RF, and with fasting TG level of 1.69 to 5.63 mmol/L, LDL-C, 1.06 to 2.59 mmol/L, and had been treated with statins for ≥4 weeks |
| Components of n-3 agents | EPA/DHA | EPA | EPA/DHA | EPA/DHA | EPA/DHA | EPA/DHA | EPA/DHA | EPA |
| Dosage (g/day) | 1 | 1.8 | 1 | 1 | 0.6 | 1 | 1 | 4 |
| No. of subjects | 11,324 | 18,645 | 6975 | 12,536 | 2501 | 15,480 | 25,871 | 8179 |
| Enrollment period | 1993–1995 | 1996–1999 | 2002–2005 | 2003–2005 | 2003–2007 | 2005–2011 | 2011–2014 | 2011–2016 |
| Follow-up (median, y) | 3.5 | 4.6 | 3.9 | 6.2 | 4.7 | 7.4 | 5.3 | 4.9 |
| Primary endpoints | All-cause death, non-fatal MI, and non-fatal stroke | Major coronary event(b) | All-cause death | Cardiovascular death | Non-fatal MI, ischemic stroke, and | Non-fatal MI, stroke, TIA, and vascular death excluding ICH | Major cardiovascular events(e) | Cardiovascular death, non-fatal MI, nonfatal stroke, coronary revascularization, and unstable angina |
| n-3 PUFA treatment results for PE, HR or RR (95% CI) | 0.85 (0.74–0.98) (a) | 0.81 (0.69–0.95) | 0.91 (0.833–0.998) | 0.98 (0.87–1.10) | 1.08 (0.79–1.47) | 0.97 (0.87–1.08) | 0.92 (0.80–1.06) | 0.75 (0.68–0.83) |
| Stroke outcome | Fatal and non-fatal stroke | Fatal and non-fatal stroke | Fatal and non-fatal stroke | Fatal and non-fatal stroke | Fatal and non-fatal stroke, and death from cardiovascular diseases | Non-fatal ischemic stroke | Fatal and non-fatal stroke | Fatal and non-fatal stroke |
| n–3 PUFA treatment results for SO, HR or RR (95% CI) | 0.95 (0.61–1.47) (a) | 1.08 (0.95–1.72) (c) | 1.16 (0.89–1.51) | 0.92 (0.79–1.08) | 1.04 (0.62–1.75) | 1.01 (0.84–1.22) | 1.04 (0.83–1.31) | 0.72 (0.55–0.93) |
| 0.8 (0.64–0.997) (d) | ||||||||
| Reference number | [44] | [51,52] | [45,46] | [48] | [47] | [50] | [49] | [53,54] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ueno, Y.; Miyamoto, N.; Yamashiro, K.; Tanaka, R.; Hattori, N. Omega-3 Polyunsaturated Fatty Acids and Stroke Burden. Int. J. Mol. Sci. 2019, 20, 5549. https://doi.org/10.3390/ijms20225549
Ueno Y, Miyamoto N, Yamashiro K, Tanaka R, Hattori N. Omega-3 Polyunsaturated Fatty Acids and Stroke Burden. International Journal of Molecular Sciences. 2019; 20(22):5549. https://doi.org/10.3390/ijms20225549
Chicago/Turabian StyleUeno, Yuji, Nobukazu Miyamoto, Kazuo Yamashiro, Ryota Tanaka, and Nobutaka Hattori. 2019. "Omega-3 Polyunsaturated Fatty Acids and Stroke Burden" International Journal of Molecular Sciences 20, no. 22: 5549. https://doi.org/10.3390/ijms20225549
APA StyleUeno, Y., Miyamoto, N., Yamashiro, K., Tanaka, R., & Hattori, N. (2019). Omega-3 Polyunsaturated Fatty Acids and Stroke Burden. International Journal of Molecular Sciences, 20(22), 5549. https://doi.org/10.3390/ijms20225549






