The Modulating Effect of p-Coumaric Acid on The Surface Charge Density of Human Glioblastoma Cell Membranes
Abstract
1. Introduction
2. Theory
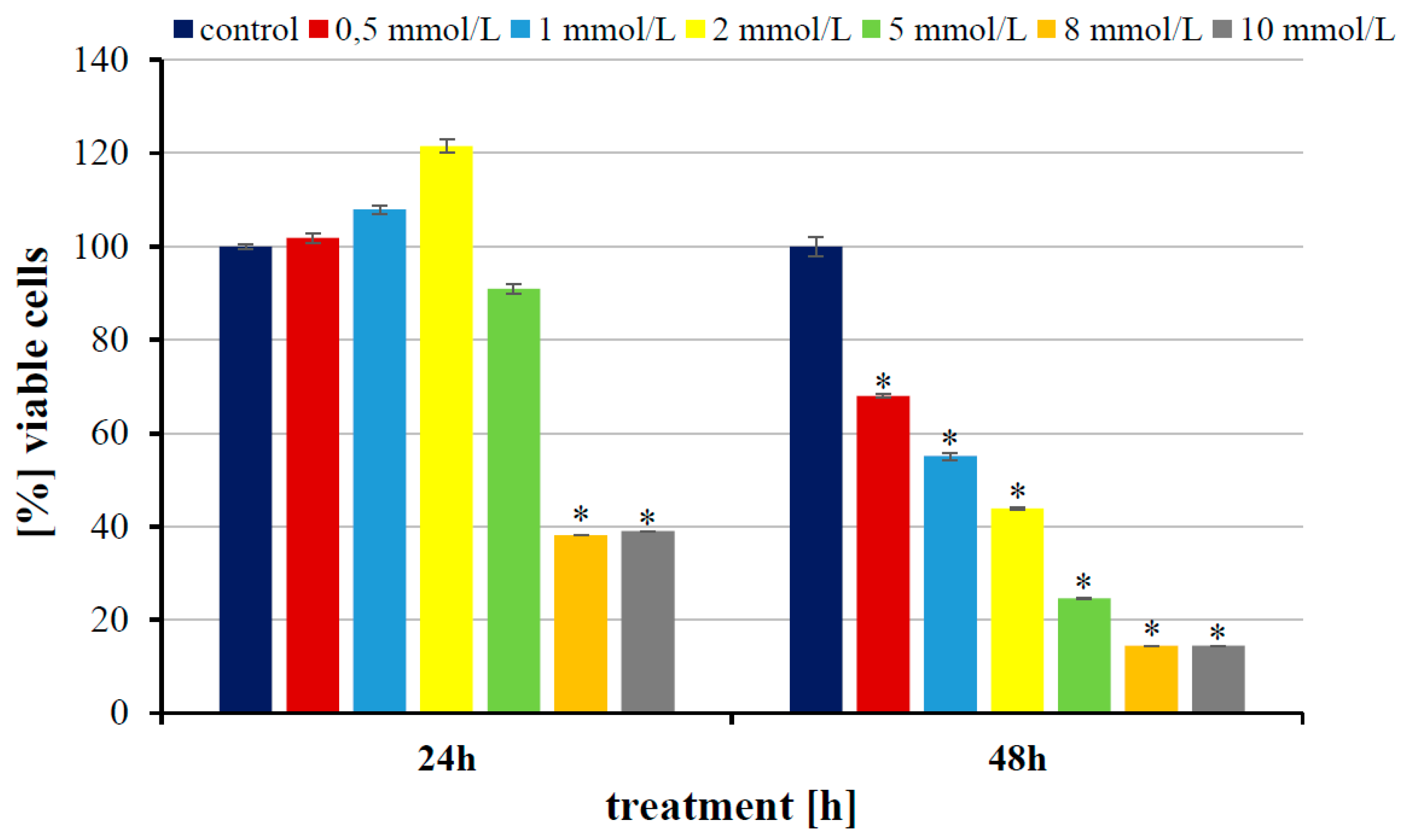
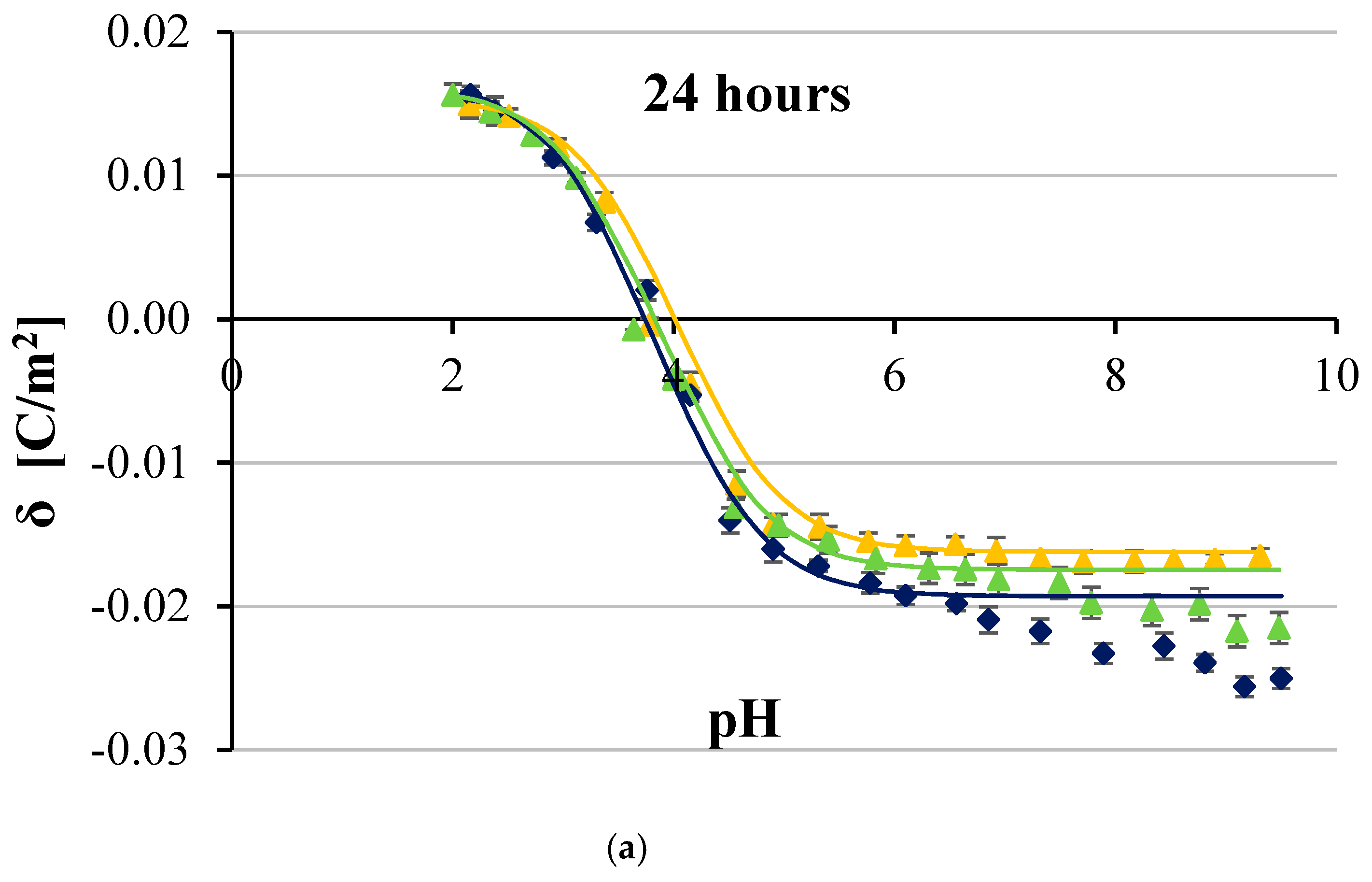
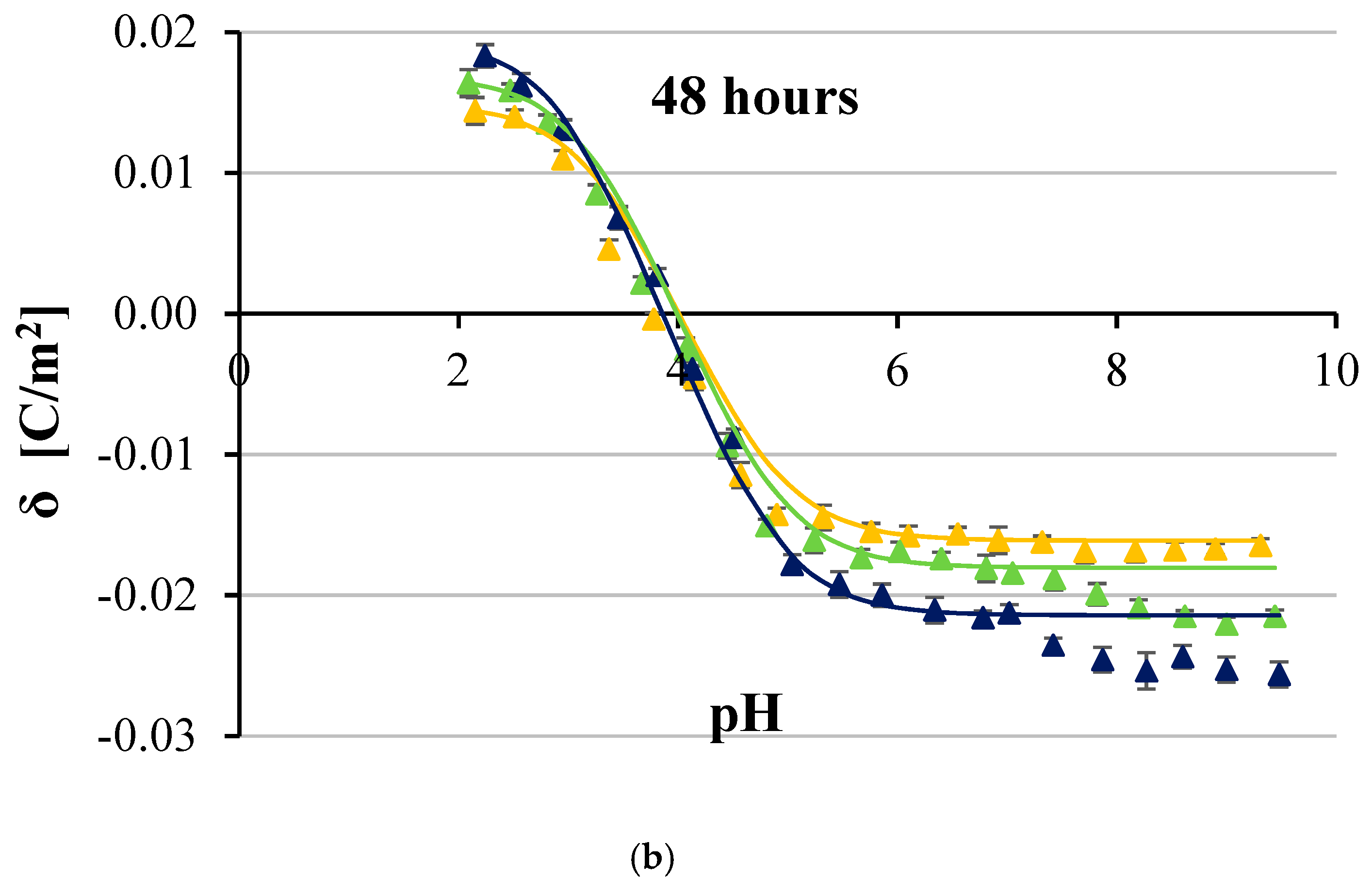
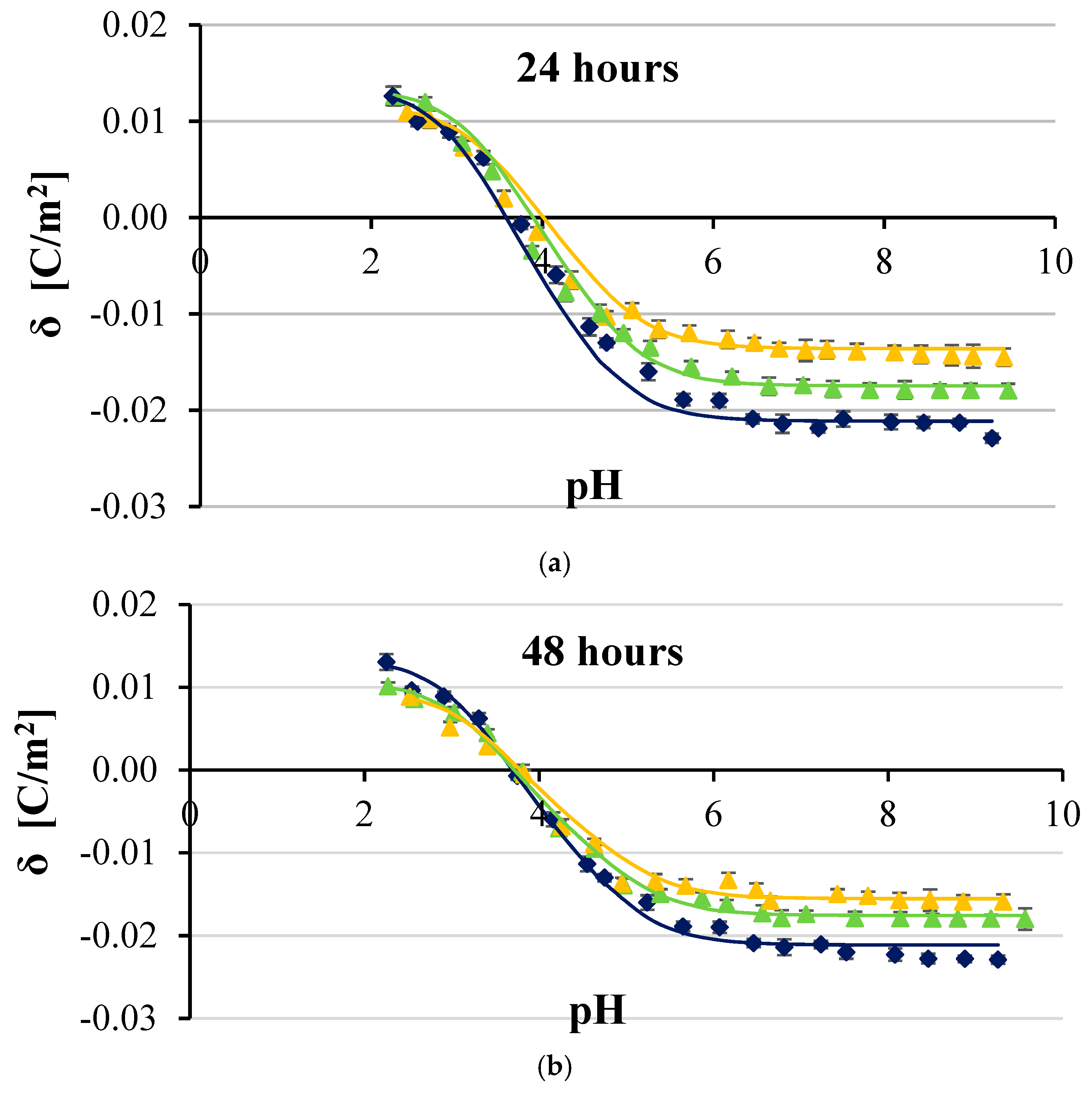
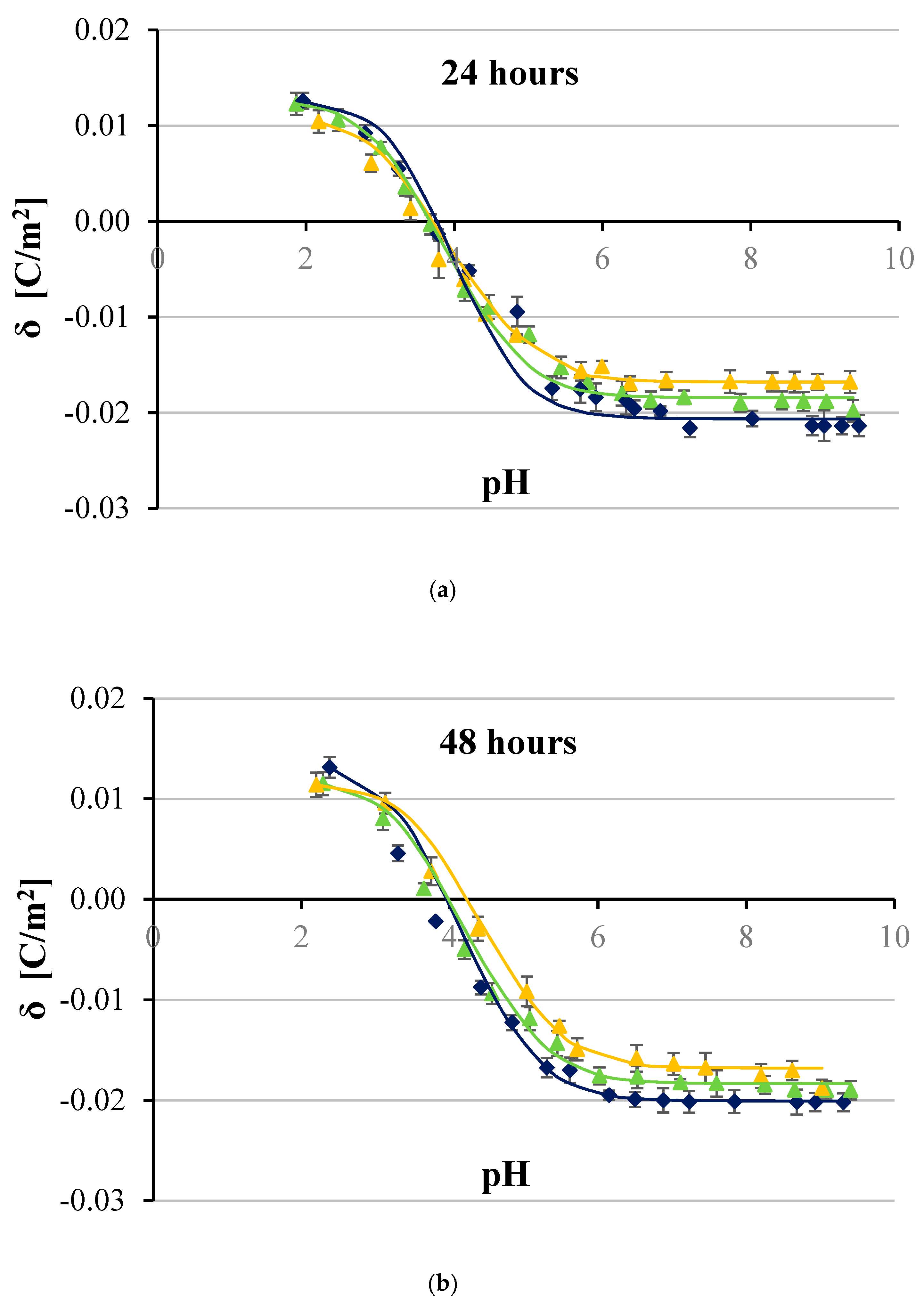
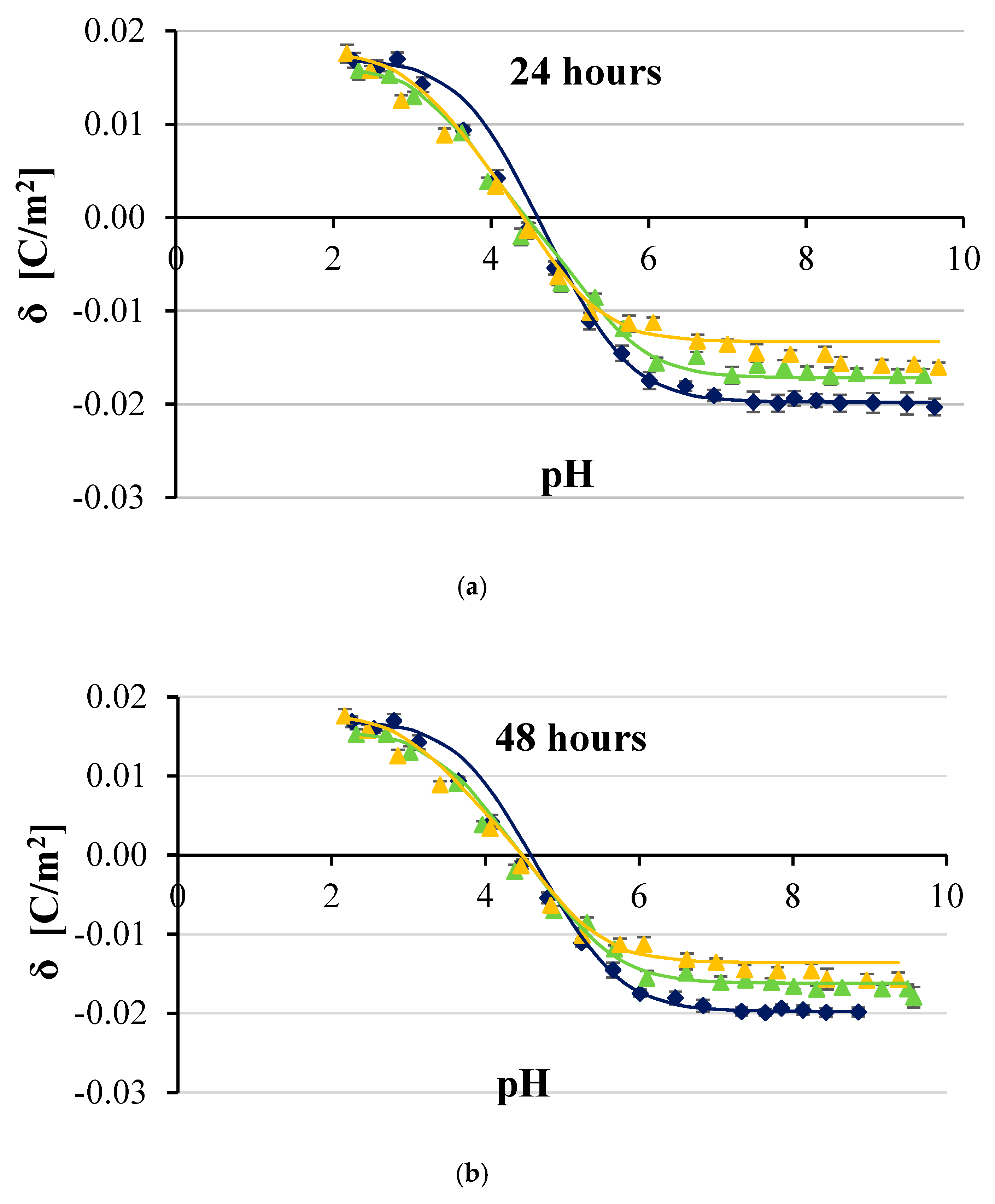
3. Results
4. Discussion
5. Materials and Methods
5.1. Materials and Reagents
5.2. Cell Cultures and Treatment
5.3. Microelectrophoretic Mobility Measurements
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Gordaliza, M. Natural products as leads to anticancer drugs. Clin. Transl. Oncol. 2007, 9, 767–776. [Google Scholar] [CrossRef] [PubMed]
- Carocho, M.; Ferreira, I.C.F.R. A review on antioxidants, prooxidants and related controversy: Natural and synthetic compounds, screening and analysis methodologies and future perspectives. Food Chem. Toxicol. 2013, 51, 15–25. [Google Scholar] [CrossRef] [PubMed]
- Carocho, M.; Ferreira, I.C.F.R. The role of phenolic compounds in the fight against cancer—A review. Anti-Cancer Agents Med. Chem. 2013, 13, 1236–1258. [Google Scholar] [CrossRef]
- Menezes, J.C.J.M.D.S.; Kamat, S.P.; Cavaleiro, J.A.S.; Gaspar, A.; Garrido, J.; Borges, F. Synthesis and antioxidant activity of long chain alkyl hydroxycinnamates. Eur. J. Med. Chem. 2011, 46, 773–777. [Google Scholar] [CrossRef]
- Razzaghi-Asl, N.; Garrido, J.; Khazraei, H.; Borges, F.; Firuzi, O. Antioxidant properties of hydroxycinnamic acids: A review of structure activity relationships. Curr. Med. Chem. 2013, 20, 4436–4450. [Google Scholar] [CrossRef]
- Rocha, L.D.; Monteiro, M.C.; Teodoro, A.J. Anticancer properties of hydroxycinnamic acids—A review. Can. Clin. Oncol. 2012, 1, 109–121. [Google Scholar] [CrossRef]
- Balachandran, C.; Emi, N.; Arun, Y.; Yamamoto, Y.; Ahilan, B.; Sangeetha, B.; Duraipandiyan, V.; Inaguma, Y.; Okamoto, A.; Ignacimuthu, S.; et al. In Vitro anticancer activity of methyl caffeate isolated from Solanum torvum Swartz. fruit. Chem.-Biol. Interact. 2015, 242, 81–90. [Google Scholar] [CrossRef]
- Eroğlu, C.; Seҫme, M.; Bağcı, G.; Dodurga, Y. Assessment of the anticancer mechanism of ferulic acid via cell cycle and apoptotic pathways in human prostate cancer cell lines. Tumor Biol. 2015, 36, 9437–9446. [Google Scholar] [CrossRef]
- Jayaprakasam, B.; Vanisree, M.; Zhang, Y.; Dewitt, D.L.; Nair, M.G. Impact of alkyl esters of caffeic and ferulic acids on tumor cell proliferation, cyclooxygenase enzyme, and lipid peroxidation. J. Agric. Food Chem. 2006, 54, 5375–5381. [Google Scholar] [CrossRef]
- Ferguson, L.R.; Zhu, S.-T.; Harris, P.J. Antioxidant and antigenotoxic effects of plant cell wall hydroxycinnamic acids in cultured HT-29 cells. Mol. Nutr. Food Res. 2005, 49, 585–593. [Google Scholar] [CrossRef]
- Menezes, J.C.J.M.D.S.; Edraki, N.; Kamat, S.P.; Khoshneviszadeh, M.; Kayani, Z.; Mirzaei, H.H.; Miri, R.; Erfani, N.; Nejati, M.; Cavaleiro, J.A.S.; et al. Long chain alkyl esters of hydroxycinnamic acids as promising anticancer agents: Selective induction of apoptosis in cancer cells. J. Agric. Food Chem. 2017, 65, 7228–7239. [Google Scholar] [CrossRef] [PubMed]
- Karthikeyan, R.; Chapala, D.; Babu, P.S. Isolation, characterization, and RP-HPLC estimation of p-coumaric acid from methanolic extract of Durva Grass (Cynodon dactylon Linn.) (Pers.). Int. J. Anal. Chem. 2015, 2015, 201386. [Google Scholar] [CrossRef] [PubMed]
- Galvez, M.C.; Barroso, C.G.; Pѐrez-Bustamante, J.A. Analysis of polyphenolic compounds of different vinegar samples. Z. Lebensm. Unters Forsch. 1994, 199, 29–31. [Google Scholar] [CrossRef]
- Ota, A.; Abramovič, H.; Abram, V.; Poklar Ulrih, N. Interactions of p-coumaric, caffeic and ferulic acids and their styrenes with model lipid membranes. Food Chem. 2011, 125, 1256–1261. [Google Scholar] [CrossRef]
- Fernie, A.R.; Trethewey, R.N.; Krotzky, A.J.; Willmitzer, L. Metabolite profiling: From diagnostics to systems biology. Nat. Rev. Mol. Cell Biol. 2004, 5, 763–769. [Google Scholar] [CrossRef] [PubMed]
- Boros, L.G.; Cascante, M.; Lee, W.N. Metabolic profiling of cell growth and death in cancer: Applications in drug discovery. Drug Discov. Today 2002, 7, 364–372. [Google Scholar] [CrossRef]
- Heileman, K.; Daoud, J.; Tabrizian, M. Dielectric spectroscopy as aviable biosensing tool for cell and tissue characterization and analysis. Biosens. Bioelectron. 2013, 49, 348–359. [Google Scholar] [CrossRef]
- Benga, G.; Holmes, R.P. Interactions between components in biological membranes and their implications for membrane function. Prog. Biophys. Mol. Bio. 1984, 43, 195–257. [Google Scholar] [CrossRef]
- Platre, M.P.; Jaillais, Y. Anionic lipids and the maintenance of membrane electrostatics in eukaryotes. Plant Signal. Behav. 2017, 12, e1282022. [Google Scholar] [CrossRef]
- Klausen, L.K.; Fuhs, T.; Dong, M. Mapping surface charge density of lipid bilayers by quantitative surface conductivity microscopy. Nat. Commun. 2016, 7, 12447. [Google Scholar] [CrossRef]
- Zhou, Y.; Raphael, R.M. Solution pH alters mechanical and electrical properties of phosphatidylcholine membranes: Relation between interfacial electrostatics, intramembrane potential, and bending elasticity. Biophys. J. 2007, 92, 2451–2462. [Google Scholar] [CrossRef] [PubMed]
- Pramanik, S.; Senneca, S.; Ethirajan, A.; Neupane, S.; Renner, F.U.; Losada-Pérez, P. Ionic strength dependent vesicle adsorption and phase behaviour of anionic phospholipids on a gold substrate. Biointerphases 2016, 11, 019006. [Google Scholar] [CrossRef] [PubMed]
- Naumowicz, M.; Kusaczuk, M.; Kruszewski, M.A.; Gál, M.; Krętowski, R.; Cechowska-Pasko, M.; Kotyńska, J. The modulating effect of lipid bilayer/p-coumaric acid interactions on electrical properties of model lipid membranes and human glioblastoma cells. Bioorg. Chem. 2019, in press. [Google Scholar] [CrossRef] [PubMed]
- Dobrzyńska, I.; Skrzydlewska, E.; Figaszewski, Z. Parameters characterizing acid–base equilibria between cell membrane and solution and their application to monitoring the effect of various factors on the membrane. Bioelectrochemistry 2006, 69, 142–147. [Google Scholar] [CrossRef] [PubMed]
- Petelska, A.D.; Kotyńska, J.; Figaszewski, A.Z. The effect of fatal carbon monoxide poisoning on the equilibria between cell membranes and the electrolyte solution. J. Membr. Biol. 2015, 248, 157–161. [Google Scholar] [CrossRef] [PubMed]
- Dobrzyńska, I.; Kotyńska, J.; Figaszewski, Z. Changes in electrical charge of phosphatidylcholine and phosphatidylserine liposomal membranes caused by adsorption of monovalent ions. Chem. Anal. 2007, 52, 931–944. [Google Scholar]
- Brennan, C.; Momota, H.; Hambardzumyan, D.; Ozawa, T.; Tandon, A.; Pedraza, A.; Holland, E. Glioblastoma subclasses can be defined by activity among signal transduction pathways and associated genomic alterations. PLoS ONE 2009, 4, e7752. [Google Scholar] [CrossRef]
- Stupp, R.; Mason, W.P.; van den Bent, M.J.; Weller, M.; Fisher, B.; Taphoorn, M.J.; Belanger, K.; Brandes, A.A.; Marosi, C.; Bogdahn, U.; et al. Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. N. Engl. J. Med. 2005, 352, 987–996. [Google Scholar] [CrossRef]
- Surh, Y.J. Cancer chemoprevention with dietary phytochemicals. Nat. Rev. Cancer 2003, 3, 768–780. [Google Scholar] [CrossRef]
- Surh, Y.J.; Ferguson, L.R. Dietary and medicinal antimutagens and anticarcinogens: Molecular mechanisms and chemopreventive potential-highlights of a symposium. Mutat. Res. 2003, 523–524, 1–8. [Google Scholar] [CrossRef]
- Yang, C.S.; Landau, J.M.; Huang, M.T.; Newmark, H.L. Inhibition of carcinogenesis by dietary polyphenolic compounds. Annu. Rev. Nutr. 2001, 21, 381–406. [Google Scholar] [CrossRef] [PubMed]
- Middleton, E., Jr.; Kandaswami, C.; Theoharides, T.C. The effects of plant flavonoids on mammalian cells: Implications for inflammation, heart disease, and cancer. Pharmacol. Rev. 2000, 52, 673–751. [Google Scholar] [PubMed]
- El-Seedi, H.R.; El-Said, A.M.; Khalifa, S.A.; Göransson, U.; Bohlin, L.; Borg-Karlson, A.K.; Verpoorte, R. Biosynthesis, natural sources, dietary intake, pharmacokinetic properties, and biological activities of hydroxycinnamic acids. J. Agric. Food Chem. 2012, 60, 10877–10895. [Google Scholar] [CrossRef] [PubMed]
- Bouzaiene, N.N.; Kilani-Jaziri, S.; Kovacic, H.; Chekir-Ghedira, L.; Ghedira, K.; Luis, J. The effects of caffeic, coumaric and ferulic acids on proliferation, superoxide production, adhesion and migration of human tumor cells in vitro. Eur. J. Pharmacol. 2015, 766, 99–105. [Google Scholar] [CrossRef] [PubMed]
- Kumar Jaganathan, S.; Supriyanto, E.; Mandal, M. Events associated with apoptotic effect of p-coumaric acid in HCT-15 colon cancer cells. World J. Gastroenterol. 2013, 19, 7726–7734. [Google Scholar] [CrossRef] [PubMed]
- Sitarek, P.; Skała, E.; Toma, M.; Wielanek, M.; Szemraj, J.; Nieborowska-Skorska, M.; Kolasa, M.; Skorski, T.; Wysokińska, H.; Śliwiński, T. A preliminary study of apoptosis induction in glioma cells via alteration of the Bax/Bcl-2-p53 axis by transformed and non-transformed root extracts of Leonurus sibiricus L. Tumor Biol. 2016, 37, 8753–8764. [Google Scholar] [CrossRef]
- Košinová, P.; Berka, K.; Wykes, M.; Otyepka, M.; Trouillas, P. Positioning of antioxidant quercetin and its metabolites in lipid bilayer membranes: Implication for their lipid peroxidation inhibition. J. Phys. Chem. B 2012, 116, 1309–1318. [Google Scholar] [CrossRef]
- Movileanu, L.; Neagoe, I.; Flonta, M.L. Interaction of the antioxidant flavonoid quercetin with planar lipid bilayers. Int. J. Pharm. 2000, 205, 135–146. [Google Scholar] [CrossRef]
- Galiano, V.; Villalaín, J. Oleuropein aglycone in lipid bilayer membranes. A molecular dynamics study. Biochim. Biophys. Acta 2015, 1848, 2849–2858. [Google Scholar] [CrossRef]
- Saija, A.; Scalese, M.; Lanza, M.; Marzullo, D.; Bonina, F.; Castelli, F. Flavonoids as antioxidant agents: Importance of their interaction with biomembranes. Free Radic. Biol. Med. 1995, 19, 481–486. [Google Scholar] [CrossRef]
- Hendrich, A.B.; Malon, R.; Pola, A.; Shirataki, Y.; Motohashi, N.; Michalak, K. Differential interaction of Sophora isoflavonoids with lipid bilayers. Eur. J. Pharm. Sci. 2002, 16, 201–208. [Google Scholar] [CrossRef]
- Pawlikowska-Pawlega, B.; Gruszecki, W.; Misiak, L.; Paduch, R.; Piersiak, T.; Zarzyka, B.; Pawelec, J.; Gawron, A. Modification of membranes by quercetin, a naturally occurring flavonoid, via its incorporation in the polar head group. Biochim. Biophys. Acta-Biomembr. 2007, 1768, 2195–2204. [Google Scholar] [CrossRef] [PubMed]
- Nałęcz, M.J.; Wojtczak, L. Surface charge of biological membranes and its regulatory functions. Postępy Biochem. 1982, 28, 191–225. [Google Scholar] [PubMed]
- Riedl, S.; Rinner, B.; Asslaber, M.; Schaider, H.; Walzer, S.; Novak, A.; Lohner, K.; Zweytick, D. In search of a novel target-phosphatidylserine exposed by non-apoptotic tumor cells and metastases of malignancies with poor treatment efficacy. Biochim. Biophys. Acta-Biomembr. 2011, 1808, 2638–2645. [Google Scholar] [CrossRef] [PubMed]
- Utsugi, T.; Schroit, A.J.; Connor, J.; Bucana, C.D.; Fidler, I.J. Elevated expression of phosphatidylserine in the outermembrane leaflet of human tumor cells and recognition by activated human blood monocytes. Cancer Res. 1991, 51, 3062–3066. [Google Scholar] [PubMed]
- Ran, S.; Downes, A.; Thorpe, P.E. Increased exposure of anionic phospholipids on the surface of tumor blood vessels. Cancer Res. 2002, 62, 6132–6140. [Google Scholar]
- Ventresca, E.M.; Lecht, S.; Jakubowski, P.; Chiaverelli, R.A.; Weaver, M.; Del Valle, L.; Ettinger, K.; Gincberg, G.; Priel, A.; Braiman, A.; et al. Association of p75NTR and α9β1 integrin modulates NGF dependent cellular responses. Cell. Signal. 2015, 27, 1225–1236. [Google Scholar] [CrossRef]
- Kusaczuk, M.; Krętowski, R.; Bartoszewicz, M.; Cechowska-Pasko, M. Phenylbutyrate—A pan-HDAC inhibitor—Suppresses proliferation of glioblastoma LN-229 cell line. Tumor Biol. 2016, 37, 931–942. [Google Scholar] [CrossRef]
- Kusaczuk, M.; Krętowski, R.; Naumowicz, M.; Stypułkowska, A.; Cechowska-Pasko, M. Silica nanoparticle-induced oxidative stress and mitochondrial damage is followed by activation of intrinsic apoptosis pathway in glioblastoma cells. Int. J. Nanomed. 2018, 13, 2279–2294. [Google Scholar] [CrossRef]
- Carmichael, J.; DeGraff, W.G.; Gazdar, A.F.; Minna, J.D.; Mitchell, J.B. Evaluation of a tetrazolium-based semiautomated colorimetric assay: Assessment of chemosensitivity testing. Cancer Res. 1987, 47, 936–942. [Google Scholar]
- Kruszewski, M.; Kusaczuk, M.; Kotyńska, J.; Gál, M.; Krętowski, R.; Cechowska-Pasko, M.; Naumowicz, M. The effect of quercetin on the electrical properties of model lipid membranes and human glioblastoma cells. Bioelectrochemistry 2018, 124, 133–141. [Google Scholar] [CrossRef] [PubMed]
- Alexander, A.E.; Johnson, P. Colloid Science; Clarendon Press: Oxford, UK, 1949; pp. 30–79. [Google Scholar]
- Barrow, G.M. Physical Chemistry; McGraw-Hill: New York, NY, USA, 1996; pp. 42–89. [Google Scholar]
| Cell Line | Groups | Parameters | |||
|---|---|---|---|---|---|
| CTA (10−6 mol/m2) | CTB (10−6 mol/m2) | KAH (10 m3/mol) | KBOH (107 m3/mol) | ||
| LN18 | Control (24 h) | 4.82 ± 0.09 | 1.33 ± 0.02 | 63.30 ± 0.11 | 1.52 ± 0.09 |
| + 5 mM CA (24 h) | 5.24 ± 0.10 | 1.38 ± 0.02 | 56.10 ± 0.12 | 1.97 ± 0.08 | |
| + 8 mM CA (24 h) | 5.78 ± 0.09 | 2.17 ± 0.05 | 53.20 ± 0.09 | 3.39 ± 0.11 | |
| Control (48 h) | 4.68 ± 0.07 | 1.54 ± 0.03 | 134.00 ± 0.11 | 3.55 ± 0.07 | |
| + 5 mM CA (48 h) | 5.24 ± 0.06 | 1.75 ± 0.02 | 123.00 ± 0.07 | 3.67 ± 0.09 | |
| + 8 mM CA (48 h) | 6.23 ± 0.07 | 2.03 ± 0.05 | 81.10 ± 0.10 | 3.96 ± 0.11 | |
| LN229 | Control (24 h) | 3.96 ± 0.08 | 1.21 ± 0.04 | 133.00 ± 0.11 | 2.53 ± 0.02 |
| + 5 mM CA (24 h) | 5.08 ± 0.07 | 1.39 ± 0.07 | 121.00 ± 0.12 | 2.56 ± 0.02 | |
| + 8 mM CA (24 h) | 6.14 ± 0.09 | 1.42 ± 0.03 | 78.40 ± 0.09 | 3.56 ± 0.05 | |
| Control (48 h) | 4.52 ± 0.10 | 1.02 ± 0.04 | 121.00 ± 0.08 | 1.51 ± 0.06 | |
| + 5 mM CA (48 h) | 5.10 ± 0.06 | 1.11 ± 0.03 | 112.00 ± 0.06 | 1.62 ± 0.07 | |
| + 8 mM CA (48 h) | 6.14 ± 0.07 | 1.41 ± 0.03 | 98.50 ± 0.07 | 2.12 ± 0.04 | |
| LBC3 | Control (24 h) | 4.88 ± 0.09 | 1.15 ± 0.05 | 96.40 ± 0.09 | 2.38 ± 0.02 |
| + 5 mM CA (24 h) | 5.31 ± 0.08 | 1.31 ± 0.06 | 90.30 ± 0.07 | 3.51 ± 0.03 | |
| + 8 mM CA (24 h) | 6.00 ± 0.09 | 1.77 ± 0.09 | 88.90 ± 0.06 | 3.83 ± 0.07 | |
| Control (48 h) | 4.88 ± 0.06 | 1.20 ± 0.02 | 328.00 ± 0.09 | 1.00 ± 0.08 | |
| + 5 mM CA (48 h) | 5.33 ± 0.04 | 1.24 ± 0.02 | 196.00 ± 0.08 | 1.48 ± 0.09 | |
| + 8 mM CA (48 h) | 5.83 ± 0.10 | 1.44 ± 0.03 | 175.00 ± 0.08 | 2.06 ± 0.06 | |
| T98G | Control (24 h) | 5.75 ± 0.06 | 1.75 ± 0.03 | 52.90 ± 0.15 | 0.70 ± 0.05 |
| + 5 mM CA (24 h) | 6.29 ± 0.08 | 1.89 ± 0.05 | 47.60 ± 0.11 | 0.97 ± 0.08 | |
| + 8 mM CA (24 h) | 6.68 ± 0.09 | 2.25 ± 0.08 | 35.10 ± 0.09 | 1.87 ± 0.11 | |
| Control (48 h) | 5.75 ± 0.06 | 1.75 ± 0.03 | 52.90 ± 0.15 | 0.70 ± 0.05 | |
| + 5 mM CA (48 h) | 6.71 ± 0.09 | 1.85 ± 0.02 | 24.90 ± 0.12 | 0.85 ± 0.06 | |
| + 8 mM CA (48 h) | 6.95 ± 0.10 | 1.92 ± 0.02 | 12.80 ± 0.11 | 1.54 ± 0.07 | |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kruszewski, M.A.; Kotyńska, J.; Kusaczuk, M.; Gál, M.; Naumowicz, M. The Modulating Effect of p-Coumaric Acid on The Surface Charge Density of Human Glioblastoma Cell Membranes. Int. J. Mol. Sci. 2019, 20, 5286. https://doi.org/10.3390/ijms20215286
Kruszewski MA, Kotyńska J, Kusaczuk M, Gál M, Naumowicz M. The Modulating Effect of p-Coumaric Acid on The Surface Charge Density of Human Glioblastoma Cell Membranes. International Journal of Molecular Sciences. 2019; 20(21):5286. https://doi.org/10.3390/ijms20215286
Chicago/Turabian StyleKruszewski, Marcin Andrzej, Joanna Kotyńska, Magdalena Kusaczuk, Miroslav Gál, and Monika Naumowicz. 2019. "The Modulating Effect of p-Coumaric Acid on The Surface Charge Density of Human Glioblastoma Cell Membranes" International Journal of Molecular Sciences 20, no. 21: 5286. https://doi.org/10.3390/ijms20215286
APA StyleKruszewski, M. A., Kotyńska, J., Kusaczuk, M., Gál, M., & Naumowicz, M. (2019). The Modulating Effect of p-Coumaric Acid on The Surface Charge Density of Human Glioblastoma Cell Membranes. International Journal of Molecular Sciences, 20(21), 5286. https://doi.org/10.3390/ijms20215286






