Polyunsaturated Fatty Acids: What is Their Role in Treatment of Psychiatric Disorders?
Abstract
1. Introduction
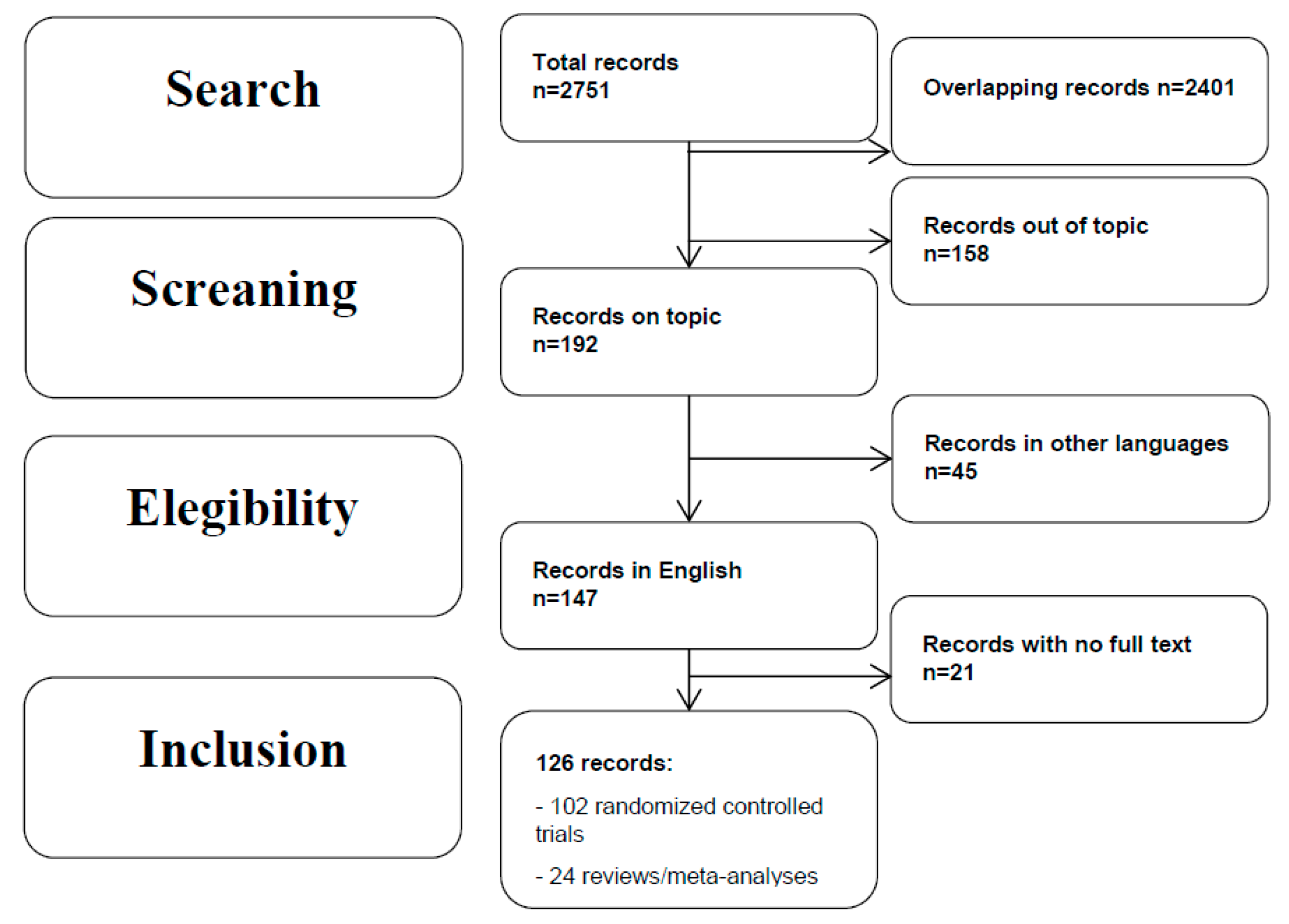
2. Methods
3. Results
4. Discussion
4.1. Omega-3 Fatty Acids in Schizophrenia
4.2. Omega-3 Fatty Acids in Major Depressive Disorder
4.3. Omega-3 Fatty Acids in Bipolar Disorder
4.4. Omega-3 Fatty Acids in Anxiety Disorders
4.5. Omega-3 Fatty Acids in Obsessive-Compulsive Disorder
4.6. Omega-3 Fatty Acids in Post-Traumatic STRESS Disorder (PTSD)
4.7. Omega-3 Fatty Acids in Substance Use Disorder
4.8. Omega-3 Fatty Acids in Attention Deficit Hyperactivity Disorder (ADHD)
4.9. Omega-3 Fatty Acids in Autism Spectrum Disorders
4.10. Omega-3 Fatty Acids in Anorexia Nervosa
4.11. Omega-3 Fatty Acids in Borderline Personality Disorder
5. Adverse Effects
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Lee, S.; Gura, K.M.; Kim, S.; Arsenault, D.A.; Bistrian, B.R.; Puder, M. Current clinical applications of omega-6 and omega-3 fatty acids. Nutr. Clin. Pr. 2006, 21, 323–341. [Google Scholar] [CrossRef]
- Pusceddu, M.M.; Nolan, Y.M.; Green, H.F.; Robertson, R.C.; Stanton, C.; Kelly, P.; Cryan, J.F.; Dinan, T.G. The Omega-3 polyunsaturated fatty acid docosahexaenoic acid (DHA) reverses corticosterone-induced changes in cortical neurons. Int. J. Neuropsychopharmacol. 2016, 6, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Calder, P.C.; Bosco, N.; Bourdet-Sicard, R.; Capuron, L.; Delzenne, N.; Doré, J.; Franceschi, C.; Lehtinen, M.J.; Recker, T.; Salvioli, S.; et al. Health relevance of the modification of low grade inflammation in ageing (inflammageing) and the role of nutrition. Ageing Res. Rev. 2017, 40, 95–119. [Google Scholar] [CrossRef] [PubMed]
- Ruiz-León, A.M.; Lapuente, M.; Estruch, R.; Casas, R. Clinical advances in immunonutrition and atherosclerosis: A review. Front. Immunol. 2019, 10, 837. [Google Scholar] [CrossRef] [PubMed]
- Simopoulos, A.P. Essential fatty acids in health and chronic disease. Am. J. Clin. Nutr. 1999, 70 (Suppl. 3), 560–569. [Google Scholar] [CrossRef] [PubMed]
- Ergas, D.; Eilat, E.; Mendlovic, S.; Sthoeger, Z.M. n-3 fatty acids and the immune system in autoimmunity. Isr. Med. Assoc. J. 2002, 4, 34–38. [Google Scholar]
- Saunders, E.F.; Ramsden, C.E.; Sherazy, M.S.; Gelenberg, A.J.; Davis, J.M.; Rapoport, S.I. Omega-3 and omega-6 polyunsaturated fatty acids in bipolar disorder: A review of biomarker and treatment studies. J. Clin. Psychiatry 2016, 77, 1301–1308. [Google Scholar] [CrossRef]
- De la Pressa, O.S.; Innis, S.M. Docosahexanoic and arachidonic acid prevent a decrease in dopaminergic and serotoninergic neurotrasmitters in frontal cortex caused by a linoleic and alpha-linoleic acid deficient diet in formula-fed piglets. J. Nutr. 1999, 129, 2088–2093. [Google Scholar]
- Sinn, N.; Milte, C.; Howe, P.R. Oiling the brain: A review of randomized controlled trials of omega-3 fatty acids in psychopathology across the lifespan. Nutrients 2010, 2, 128–170. [Google Scholar] [CrossRef]
- Bozzatello, P.; Brignolo, E.; De Grandi, E.; Bellino, S. Supplementation with omega-3 fatty acids in psychiatric disorders: A review of literature data. J. Clin. Med. 2016, 5, 67. [Google Scholar] [CrossRef]
- Mischoulon, D.; Freeman, M.P. Omega-3 fatty acids in psychiatry. Psychiatr. Clin. N. Am. 2013, 36, 15–23. [Google Scholar] [CrossRef] [PubMed]
- Glen, A.I.; Glen, E.M.; Horrobin, D.F.; Vaddadi, K.S.; Spellman, M.; Morse-Fisher, N.; Ellis, K.; Skinner, F.S. A red cell membrane abnormality in a subgroup of schizophrenic patients: Evidence for two diseases. Schizophr. Res. 1994, 12, 53–61. [Google Scholar] [CrossRef]
- Reddy, R.D.; Keshavan, M.S.; Yao, J.K. Reduced red blood cell membrane essential polyunsatu- rated fatty acids in first episode schizophrenia at neuroleptic-naive baseline. Schizophr. Bull. 2004, 30, 901–911. [Google Scholar] [CrossRef] [PubMed]
- Schlögelhofer, M.; Amminger, G.P.; Schaefer, M.R.; Fusar-Poli, P.; Smesny, S.; McGorry, P.; Berger, G.; Mossaheb, N. Polyunsaturated fatty acids in emerging psychosis: A safer alternative? Early Interv. Psychiatry 2014, 8, 199–208. [Google Scholar] [CrossRef] [PubMed]
- Cadenhead, K.S.; Minichino, A.; Kelsven, S.; Addington, J.; Bearden, C.; Cannon, T.D.; Cornblatt, B.A.; Mathalon, D.; McGlashan, T.H.; Perkins, D.O.; et al. Metabolic abnormalities and low dietary Omega 3 are associated with symptom severity and worse functioning prior to the onset of psychosis: Findings from the North American Prodrome Longitudinal Studies Consortium. Schizophr. Res. 2019, 204, 96–103. [Google Scholar] [CrossRef]
- Sethom, M.M.; Fares, S.; Bouaziz, N.; Melki, W.; Jemaa, R.; Feki, M.; Hechmi, Z.; Kaabachi, N. Polyunsaturated fatty acids deficits are associated with psychotic state and negative symptoms in patients with schizophrenia. Prostaglandins Leukot. Essent. Fat. Acids 2010, 83, 131–136. [Google Scholar] [CrossRef]
- Sumiyoshi, T.; Higuchi, Y.; Matsui, M.; Itoh, H.; Uehara, T.; Itoh, T.; Arai, H.; Takamiya, C.; Suzuki, M.; Kurachi, M. Membrane fatty acid levels as a predictor of treatment response in chronic schizophrenia. Psychiatry Res. 2011, 186, 23–27. [Google Scholar] [CrossRef]
- Amminger, G.P.; Schäfer, M.R.; Papageorgiou, K.; Klier, C.M.; Cotton, S.M.; Harrigan, S.M.; Mackinnon, A.; McGorry, P.D.; Berger, G.E. Long-chain omega-3 fatty acids for indicated prevention of psychotic disorders: A randomized, placebo-controlled trial. Arch. Gen. Psychiatry 2010, 67, 146–154. [Google Scholar] [CrossRef]
- Amminger, G.P.; Chanen, A.M.; Ohmann, S.; Klier, C.M.; Mossaheb, N.; Bechdolf, A.; Nelson, B.; Thompson, A.; McGorry, P.D.; Yung, A.R.; et al. Omega-3 fatty acid supplementation in adolescents with borderline personality disorder and ultra-high risk criteria for psychosis: A post hoc subgroup analysis of a double-blind, randomized controlled trial. Can. J. Psychiatry 2013, 58, 402–408. [Google Scholar] [CrossRef]
- Amminger, G.P.; Schäfer, M.R.; Schlögelhofer, M.; Klier, C.; McGorry, P. Longer-term outcome in the prevention of psychotic disorders by the Vienna omega-3 study. Nat. Commun. 2015, 6, 7934. [Google Scholar] [CrossRef]
- Smesny, S.; Milleit, B.; Hipler, U.-C.; Milleit, C.; Schäfer, M.; Klier, C.; Holub, M.; Holzer, I.; Berger, G.; Otto, M.; et al. Omega-3 fatty acid supplementation changes intracellular phospholipase A2 activity and membrane fatty acid profiles in individuals at ultra-high risk for psychosis. Mol. Psychiatry 2014, 19, 317–324. [Google Scholar] [CrossRef] [PubMed]
- McGorry, P.D.; Nelson, B.; Markulev, C.; Yuen, H.; Schäfer, M.; Mossaheb, N.; Schlögelhofer, M.; Smesny, S.; Hickie, I.; Berger, G. Effect of ω-3 Polyunsaturated Fatty Acids in Young People at Ultrahigh Risk for Psychotic Disorders. JAMA Psychiatry 2017, 74, 19. [Google Scholar] [CrossRef] [PubMed]
- Berger, G.E.; Proffitt, T.-M.; McConchie, M.; Yuen, H.; Wood, S.; Amminger, G.; Brewer, W.; McGorry, P. Ethyl-eicosapentaenoic acid in first-episode psychosis: A randomized, placebo-controlled trial. J. Clin. Psychiatry 2007, 68, 1867–1875. [Google Scholar] [CrossRef] [PubMed]
- Berger, G.E.; Wood, S.J.; Wellard, R.M.; Proffitt, T.; McConchie, M.; Amminger, G.; Jackson, G.; Velakoulis, D.; Pantelis, C.; McGorry, P. Ethyl-eicosapentaenoic acid in first-episode psychosis. A 1H-MRS study. Neuropsychopharmacology 2008, 33, 2467–2473. [Google Scholar] [CrossRef] [PubMed]
- Wood, S.J.; Cocchi, L.; Proffitt, T.M.; McConchie, M.; Jackson, G.D.; Takahashi, T.; Berger, G.E. Neuroprotective effects of ethyl-eicosapentaenoic acid in first episode psychosis: A longitudinal T2 relaxometry pilot study. Psychiatry Res. Neuroimaging 2010, 182, 180–182. [Google Scholar] [CrossRef] [PubMed]
- Emsley, R.; Chiliza, B.; Asmal, L.; Emsley, R.; Chiliza, B.; Asmal, L.; du Plessis, S.; Phahladira, L.; van Niekerk, E.; van Rensburg, S.J.; et al. A randomized, controlled trial of omega-3 fatty acids plus an antioxidant for relapse prevention after antipsychotic discontinuation in first-episode schizophrenia. Schizophr. Res. 2014, 158, 230–235. [Google Scholar] [CrossRef] [PubMed]
- Pawełczyk, T.; Grancow-Grabka, M.; Kotlicka-Antczak, M.; Trafalska, E.; Pawełczyk, A. A randomized controlled study of the efficacy of six-month supplementation with concentrated fish oil rich in omega-3 polyunsaturated fatty acids in first episode schizophrenia. J. Psychiatr. Res. 2016, 73, 34–44. [Google Scholar] [CrossRef] [PubMed]
- Pawelczyk, T.; Grancow-Grabka, M.; Trafalska, E.; Szemraj, J.; Pawelczyk, A. Oxidative stress reduction related to the efficacy of n-3 polyunsaturated fatty acids in first episode schizophrenia: Secondary outcome analysis of the OFFER randomized trial. Prostaglandins Leukot. Essent. Fat. Acids 2017, 121, 7–13. [Google Scholar] [CrossRef]
- Pawelczyk, T.; Grancow-Grabka, M.; Trafalska, E.; Szemraj, J.; Zurner, N.; Pawelczyk, A. Telomerase level increase is related to n-3 polyunsaturated fatty acid efficacy in first episode schizophrenia: Secondary outcome analysis of the OFFER randomized clinical trial. Prog. Neuropsychopharm. Biol. Psychiatry 2018, 83, 142–148. [Google Scholar] [CrossRef]
- Peet, M.; Brind, J.; Ramchand, C.N.; Shah, S.; Vankar, G.K. Two double-blind placebo-controlled pilot studies of eicosapentaenoic acid in the treatment of schizophrenia. Schizophr. Res. 2001, 49, 243–251. [Google Scholar] [CrossRef]
- Peet, M.; Horrobin, D.F.; Study Group. A dose-ranging exploratory study of the effects of ethyl-eicosapentaenoate in patients with persistent schizophrenic symptoms. J. Psychiat. Res. 2002, 36, 7–18. [Google Scholar] [CrossRef]
- Emsley, R.; Myburgh, C.; Oosthuizen, P.; van Rensburg, S.J. Randomized, placebo-controlled study of ethyl-eicosapentaenoic acid as supplemental treatment in schizophrenia. Am. J. Psychiatry 2002, 159, 1596–1598. [Google Scholar] [CrossRef] [PubMed]
- Emsley, R.; Niehaus, D.J.; Koen, L.; Oosthuizen, P.P.; Turner, H.J.; Carey, P.; Murck, H. The effects of eicosapentaenoic acid in tardive dyskinesia: A randomized, placebo-controlled trial. Schizophr. Res. 2006, 84, 112–120. [Google Scholar] [CrossRef] [PubMed]
- Jamilian, H.; Solhi, H.; Jamilian, M. Randomized, placebo-controlled clinical trial of omega-3 as supplemental treatment in schizophrenia. Glob. J. Health Sci. 2014, 18, 103–108. [Google Scholar] [CrossRef]
- Fenton, W.S.; Dickerson, F.; Boronow, J.; Hibbeln, J.R.; Knable, M. A placebo-controlled trial of omega-3 fatty acid (ethyl eicosapentaenoic acid) supplementation for residual symptoms and cognitive impairment in schizophrenia. Am. J. Psychiatry 2001, 158, 2071–2074. [Google Scholar] [CrossRef]
- Bentsen, H.; Osnes, K.; Refsum, H. A randomized placebo-controlled trial of an omega-3 fatty acid and vitamins E + C in schizophrenia. Transl. Psychiatry 2013, 3, e335. [Google Scholar] [CrossRef]
- Qiao, Y.; Mei, Y.; Han, H.; Liu, F.; Yang, X.M.; Yang, S.; Xie, B.; Long, B. Effects of Omega-3 in the treatment of violent schizophrenia patients. Schizophr. Res. 2018, 195, 283–285. [Google Scholar] [CrossRef]
- Robinson, D.G.; Schooler, N.R.; Rosenheck, R.A.; Lin, H.; Sint, K.J.; Marcy, P.; Kane, J.M. Predictors of hospitalization of individuals with first-episode psychosis: Data from a 2-year follow-up of the raise-ETP. Psychiatr. Serv. 2019, 7, 569–577. [Google Scholar] [CrossRef]
- Joy, C.B.; Mumby-Croft, R.; Joy, L.A. Polyunsaturated fatty acid supplementation for schizophrenia. Cochrane Database Syst. Rev. 2006, 19, CD001257. [Google Scholar]
- Freeman, M.P.; Hibbeln, J.R.; Wisner, K.L.; Davis, J.M.; Mischoulon, D.; Peet, M.; Keck, P.E., Jr.; Marangell, L.B.; Richardson, A.J.; Lake, J.; et al. Omega-3 fatty acids: Evidence basis for treatment and future research in psychiatry. J. Clin. Psychiatry 2006, 67, 1954–1967. [Google Scholar] [CrossRef]
- Fusar-Poli, P.; Berger, G. Eicosapentaenoic acid interventions in schizophrenia: Meta-analysis of randomized, placebo-controlled studies. J. Clin. Psychopharmacol. 2012, 32, 179–185. [Google Scholar] [CrossRef] [PubMed]
- Akter, K.; Gallo, D.A.; Martin, S.A.; Myronyuk, N.; Roberts, R.T.; Stercula, K.; Raffa, R.B. A review of the possible role of the essential fatty acids and fish oils in the aetiology, prevention or pharmacotherapy of schizophrenia. J. Clin. Pharm. Ther. 2012, 37, 132–139. [Google Scholar] [CrossRef] [PubMed]
- Agostoni, C.; Nobile, M.; Ciappolino, V.; Delvecchio, G.; Tesei, A.; Turolo, S.; Crippa, A.; Mazzocchi, A.; Altamura, C.A.; Brambilla, P. The Role of omega-3 fatty acids in developmental psychopathology: A systematic review on early psychosis, Autism, and ADHD. Int. J. Mol. Sci. 2017, 18, 2608. [Google Scholar] [CrossRef] [PubMed]
- Joseph Firth, J.; Carney, R.; Stubbs, B.; Teasdale, S.B.; Vancampfort, D.; Ward, P.B.; Berk, M.; Sarris, J. Nutritional deficiencies and clinical correlates in first-episode psychosis: A systematic review and meta-analysis. Schizophr. Bull. 2018, 44, 1275–1292. [Google Scholar] [CrossRef]
- Balanzá Martínez, V. Nutritional supplements in psychotic disorders. Actas Esp. Psiquiatr. 2017, 45, 16–25. [Google Scholar]
- Peet, M.; Murphy, B.; Shay, J.; Horrobin, D. Depletion of omega-3 fatty acid levels in red blood cell membranes of depressive patients. Biol. Psychiatry 1998, 43, 315–319. [Google Scholar] [CrossRef]
- Lin, P.Y.; Huang, S.Y.; Su, K.P. A meta-analytic review of polyunsaturated fatty acid compositions in patients with depression. Biol. Psychiatry 2010, 68, 140–147. [Google Scholar] [CrossRef]
- Chrysohoou, C.; Tsitsinakis, G.; Siassos, G.; Psaltopoulou, T.; Galiatsatos, N.; Metaxa, V.; Lazaros, G.; Miliou, A.; Giakoumi, E.; Mylonakis, C.; et al. Fish consumption moderates depressive symptomatology in elderly men and women from the IKARIA study. Cardiol. Res. Pr. 2010, 2011, 219578. [Google Scholar] [CrossRef]
- Hoffmire, C.A.; Block, R.C.; Thevenet-Morrison, K.; Van Wijngaarden, E. Associations between omega-3 poly-unsaturated fatty acids from fish consumption and severity of depressive symptoms: An analysis of the 2005–2008 national health and nutrition examination survey. Prostaglandins Leukot. Essent. Fat. Acids 2012, 86, 155–160. [Google Scholar] [CrossRef]
- Beydoun, M.A.; Kuczmarski, M.T.F.; Beydoun, H.A.; Hibbeln, J.R.; Evans, M.K.; Zonderman, A.B. Ω−3 fatty acid intakes are inversely related to elevated depressive symptoms among United States women. J. Nutr. 2013, 143, 1743–1752. [Google Scholar] [CrossRef]
- Song, C. Essential fatty acids as potential anti-inflammatory agents in the treatment of affective disorders. Mod. Trends Pharmacopsychiatr. 2013, 28, 75–89. [Google Scholar]
- Lotrich, F.E. Inflammatory cytokine-associated depression. Brain Res. 2015, 1617, 113–125. [Google Scholar] [CrossRef] [PubMed]
- Song, C.; Shieh, C.H.; Wu, Y.S.; Kalueff, A.; Gaikwad, S.; Su, K.P. The role of omega-3 polyunsaturated fatty acids eicosapentaenoic and docosahexaenoic acids in the treatment of major depression and Alzheimer’s disease: Acting separately or synergistically? Prog. Lipid Res. 2016, 62, 41–54. [Google Scholar] [CrossRef] [PubMed]
- Chhetry, B.T.; Hezghia, A.; Miller, J.M.; Lee, S.; Rubin-Falcone, H.; Cooper, T.B.; Oquendo, M.A.; Mann, J.J.; Sublette, M.E. Omega-3 polyunsaturated fatty acid supplementation and white matter changes in major depression. J. Psychiatr. Res. 2016, 75, 65–74. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-Villegas, A.; Álvarez-Pérez, J.; Toledo, E.; Salas-Salvadó, J.; Ortega-Azorín, C.; Zomeño, M.D.; Vioque, J.; Martínez, J.A.; Romaguera, D.; Pérez-López, J.; et al. Seafood consumption, omega-3 fatty acids intake, and life-time prevalence of depression in the PREDIMED-plus trial. Nutrients 2018, 10, 2000. [Google Scholar] [CrossRef] [PubMed]
- Nemets, B.; Stahl, Z.; Belmaker, R.H. Addition of omega-3 fatty acid to maintenance medication treatment for recurrent unipolar depressive disorder. Am. J. Psychiatry 2002, 159, 477–479. [Google Scholar] [CrossRef] [PubMed]
- Peet, M.; Horrobin, D.F. A dose-ranging study of the effects of ethyl- eicosapentaenoate in patients with ongoing depression despite apparently adequate treatment with standard drugs. Arch. Gen. Psychiatry 2002, 59, 913–919. [Google Scholar] [CrossRef]
- Llorente, A.M.; Jensen, C.L.; Voigt, R.G.; Fraley, J.K.; Berretta, M.C.; Heird, W.C. Effect of maternal docosahexaenoic acid supplementation on postpartum depression and information processing. Am. J. Obs. Gynecol. 2003, 188, 1348–1353. [Google Scholar] [CrossRef]
- Marangell, L.B.; Martinez, J.M.; Zboyan, H.A.; Kertz, B.; Kim, H.F.; Puryear, L.J. A double-blind, placebo-controlled study of the omega-3 fatty acid docosahexaenoic acid in the treatment of major depression. Am. J. Psychiatry 2003, 160, 996–998. [Google Scholar] [CrossRef]
- Su, K.P.; Huang, S.Y.; Chiu, C.C.; Shen, W.W. Omega-3 fatty acids in major depressive disorder. A preliminary double-blind, placebo-controlled trial. Eur. Neuropsychopharmacol. 2003, 13, 267–271. [Google Scholar] [CrossRef]
- Silvers, K.M.; Woolley, C.C.; Hamilton, F.C.; Watts, P.M.; Watson, R.A. Randomised double-blind placebo-controlled trial of fish oil in the treatment of depression. Prostaglandins Leukot. Ess. Fat. Acids 2005, 72, 211–218. [Google Scholar] [CrossRef] [PubMed]
- Nemets, H.; Nemets, B.; Apter, A.; Bracha, Z.; Belmaker, R.H. Omega-3 treatment of childhood depression: A controlled, double-blind pilot study. Am. J. Psychiatry 2006, 163, 1098–1100. [Google Scholar] [CrossRef] [PubMed]
- Grenyer, B.F.; Crowe, T.; Meyer, B.; Owen, A.J.; Grigonis-Deane, E.M.; Caputi, P.; Howe, P.R.C. Fish oil supplementation in the treatment of major depression: A randomised double- blind placebo-controlled trial. Prog. Neuropsychopharmacol. Biol. Psychiatry 2007, 31, 1393–1396. [Google Scholar] [CrossRef] [PubMed]
- Freeman, M.P.; Davis, M.; Sinha, P.; Wisner, K.L.; Hibbeln, J.R.; Gelenberg, A.J. Omega-3 fatty acids and supportive psychotherapy for perinatal depression: A randomized placebo-controlled study. J. Affect. Disord. 2008, 110, 142–148. [Google Scholar] [CrossRef]
- Jazayeri, S.; Tehrani-Doost, M.; Keshavarz, S.A.; Hosseini, M.; Djazayery, A.; Amini, H.; Jalali, M.; Peet, M. Comparison of therapeutic effects of omega-3 fatty acid eicosapentaenoic acid and fluoxetine, separately and in combination, in major depressive disorder. Aust. N. Z. J. Psychiatry 2008, 42, 192–198. [Google Scholar] [CrossRef]
- Rees, A.M.; Austin, M.P.; Parker, G.B. Omega-3 fatty acids as a treatment for perinatal depression: Randomized double-blind placebo-controlled trial. Aust. N. Z. J. Psychiatry 2008, 42, 199–205. [Google Scholar] [CrossRef]
- Rogers, P.J.; Appleton, K.M.; Kessler, D.; Peters, T.J.; Gunnell, D.; Hayward, R.C.; Heatherley, S.V.; Christian, L.M.; McNaughton, S.A.; Ness, A.R. No effect of n-3 long-chain polyunsaturated fatty acid (EPA and DHA) supplementation on depressed mood and cognitive function: A randomised controlled trial. Br. J. Nutr. 2008, 99, 421–431. [Google Scholar] [CrossRef]
- Doornbos, B.; Van Goor, S.A.; Dijck-Brouwer, D.A.; Schaafsma, A.; Korf, J.; Muskiet, F.A.J. Supplementation of a low dose of DHA or DHA+AA does not prevent peripartum depressive symptoms in a small population based sample. Prog. Neuropsychopharmacol. Biol. Psychiatry 2008, 33, 49–52. [Google Scholar] [CrossRef]
- Lucas, M.; Asselin, G.; Merette, C.; Poulin, M.J.; Dodin, S. Ethyleicosapentaenoic acid for the treatment of psychological distress and depressive symptoms in middle- aged women: A double-blind, placebo-controlled, randomized clinical trial. Am. J. Clin. Nutr. 2009, 89, 641–651. [Google Scholar] [CrossRef]
- Mischoulon, D.; Papakostas, G.I.; Dording, C.M.; Farabaugh, A.H.; Sonawalla, S.B.; Agoston, A.M.; Smith, J.; Beaumont, E.C.; Dahan, L.E.; Alpert, J.E.; et al. A double-blind, randomized controlled trial of ethyl- eicosapentaenoate for major depressive disorder. J. Clin. Psychiatry 2009, 70, 1636–1644. [Google Scholar] [CrossRef]
- Makrides, M.; Gibson, R.A.; McPhee, A.J.; Yelland, L.; Quinlivan, J.; Ryan, P. DOMInO Investigative Team, Effect of DHA supplementation during pregnancy on maternal depression and neurodevelopment of young children: A randomized controlled trial. JAMA 2010, 304, 1675–1683. [Google Scholar] [CrossRef] [PubMed]
- Rondanelli, M.; Giacosa, A.; Opizzi, A.; Pelucchi, C.; La Vecchia, C.; Montorfano, G.; Negroni, M.; Berra, B.; Politi, P.; Rizzo, A.M. Effect of omega-3 fatty acids supplementation on depressive symptoms and on health-related quality of life in the treatment of elderly women with depression: A double-blind, placebo-controlled, randomized clinical trial. J. Am. Coll. Nutr. 2010, 29, 55–64. [Google Scholar] [CrossRef] [PubMed]
- Rondanelli, M.; Giacosa, A.; Opizzi, A.; Pelucchi, C.; La Vecchia, C.; Montorfano, G.; Negroni, M.; Berra, B.; Politi, P.; Rizzo, A.M. Long chain omega 3 polyunsaturated fatty acids supplementation in the treatment of elderly depression: Effects on depressive symptoms, on phospholipids fatty acids profile and on health-related quality of life. J. Nutr. Health Aging 2011, 15, 37–44. [Google Scholar] [CrossRef] [PubMed]
- Lespérance, F.; Frasure-Smith, N.; St-André, E.; Lespérance, F.; Frasure-Smith, N.; St-André, E.; Turecki, G.; Lespérance, P.; Wisniewski, S.R. The efficacy of omega-3 supplementation for major depression: A randomized controlled trial. J. Clin. Psychiatry 2011, 72, 1054–1062. [Google Scholar] [CrossRef]
- Tajalizadekhoob, Y.; Sharifi, F.; Fakhrzadeh, H.; Mirarefin, M.; Ghaderpanahi, M.; Badamchizade, Z.; Azimipour, S. The effect of low-dose omega 3 fatty acids on the treatment of mild to moderate depression in the elderly: A double-blind, randomized, placebocontrolled study. Eur. Arch. Psychiatry Clin. Neurosci. 2011, 261, 539–549. [Google Scholar] [CrossRef]
- Antypa, N.; Smelt, A.H.; Strengholt, A.; Van der Does, A.J. Effects of omega-3 fatty acid supplementation on mood and emotional information processing in recovered depressed individuals. J. Psychopharmacol. 2012, 26, 738–743. [Google Scholar] [CrossRef]
- Gertsik, L.; Poland, R.E.; Bresee, C.; Rapaport, M.H. Omega-3 fatty acid augmentation of citalopram treatment for patients with major depressive disorder. J. Clin. Psychopharmacol. 2012, 32, 61–64. [Google Scholar] [CrossRef]
- Krawczyk, K.; Rybakowski, J. Augmentation of antidepressants with unsaturated fatty acids omega-3 in drug-resistant depression. Psychiatr. Pol. 2012, 46, 585–598. [Google Scholar]
- Rizzo, A.M.; Corsetto, P.A.; Montorfano, G.; Opizzi, A.; Faliva, M.; Giacosa, A.; Ricevuti, G.; Pelucchi, C.; Berra, B.; Rondanelli, M. Comparison between the AA/EPA ratio in depressed and non depressed elderly females: Omega-3 fatty acid supplementation correlates with improved symptoms but does not change immunological parameters. Nutr. J. 2012, 11, 82. [Google Scholar] [CrossRef]
- Mozaffari-Khosravi, H.; Yassini-Ardakani, M.; Karamati, M.; Shariati-Bafghi, S.E. Eicosapentaenoic acid versus docosahexaenoic acid in mild-to-moderate depression: A randomized, doubleblind, placebo-controlled trial. Eur. Neuropsychopharmacol. 2013, 23, 636–644. [Google Scholar] [CrossRef]
- Mozurkewich, E.L.; Clinton, C.M.; Chilimigras, J.L.; Hamilton, S.E.; Allbaugh, L.J.; Berman, D.R.; Marcus, S.M.; Romero, V.C.; Treadwell, M.C.; Keeton, K.L.; et al. The mothers, Omega-3, and mental health study: A double-blind, randomized controlled trial. Am. J. Obs. Gynecol. 2013, 313, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Judge, M.P.; Beck, C.T.; Durham, H.; McKelvey, M.M.; Lammi-Keefe, C.J. Pilot trial evaluating maternal docosahexaenoic acid consumption during pregnancy: Decreased postpartum depressive symptomatology. Int. J. Nursing Sci. 2014, 1, 339–345. [Google Scholar] [CrossRef][Green Version]
- Ginty, A.T.; Conklin, S.M. Short-term supplementation of acute long-chain omega-3 polyunsaturated fatty acids may alter depression status and decrease symptomology among young adults with depression: A preliminary randomized and placebo controlled trial. Psychiatry Res. 2015, 229, 485–489. [Google Scholar] [CrossRef] [PubMed]
- Mischoulon, D.; Nierenberg, A.A.; Schettler, P.J.; Kinkead, B.L.; Fehling, K.; Martinson, M.A.; Hyman Rapaport, M. A double-blind, randomized controlled clinical trial comparing eicosapentaenoic acid versus docosahexaenoic acid for depression. J. Clin. Psychiat. 2015, 76, 54–61. [Google Scholar] [CrossRef] [PubMed]
- Park, Y.; Park, Y.S.; Kim, S.H.; Oh, D.H.; Park, Y.C. Supplementation of n-3 polyunsaturated fatty acids for major depressive disorder: A randomized, double-blind, 12-week, placebo-controlled trial in Korea. Ann. Nutr. Metab. 2015, 66, 141–148. [Google Scholar] [CrossRef]
- Young, A.S.; Arnold, L.E.; Wolfson, H.L.; Fristad, M.A. Psychoeducational psychotherapy and omega-3 supplementation improve co-occurring behavioral problems in youth with depression: Results from a pilot RCT. J. Abnorm. Child. Psychol. 2017, 45, 1025–1037. [Google Scholar] [CrossRef]
- Gabbay, V.; Freed, R.D.; Alonso, C.M.; Senger, S.; Stadterman, J.; Davison, B.A.; Klein, R.G. A Double-blind placebo-controlled trial of omega-3 fatty acids as a monotherapy for adolescent depression. J. Clin. Psychiatry 2018, 26, 79. [Google Scholar] [CrossRef]
- Hsu, M.C.; Tung, C.Y.; Chen, H.E. Omega-3 polyunsaturated fatty acid supplementation in prevention and treatment of maternal depression: Putative mechanism and recommendation. J. Affect. Disord. 2018, 238, 47–61. [Google Scholar] [CrossRef]
- Jahangard, L.; Sadeghi, A.; Ahmadpanah, M.; Holsboer-Trachsler, E.; Sadeghi Bahmani, D.; Haghighi, M.; Brand, S. Influence of adjuvant omega-3-polyunsaturated fatty acids on depression, sleep, and emotion regulation among outpatients with major depressive disorders—Results from a double-blind, randomized and placebo-controlled clinical trial. J. Psychiatr Res. 2018, 107, 48–56. [Google Scholar] [CrossRef]
- Tayama, J.; Ogawa, S.; Nakaya, N.; Sone, T.; Hamaguchi, T.; Takeoka, A.; Hamazaki, K.; Okamura, H.; Yajima, J.; Kobayashi, M.; et al. Omega-3 polyunsaturated fatty acids and psychological intervention for workers with mild to moderate depression: A double-blind randomized controlled trial. J. Affect. Disord. 2019, 245, 364–370. [Google Scholar] [CrossRef]
- Rapaport, M.H.; Nierenberg, A.A.; Schettler, P.J.; Kinkeaad, B.; Cardoos, A.; Walker, R.; Mischoulon, D. Inflammation as a predictive biomarker for response to omega-3 fatty acids in major depressive disorder a proof-of-concept study. Mol. Psychiatry 2016, 21, 71–79. [Google Scholar] [CrossRef] [PubMed]
- Bloch, M.H.; Hannestad, J. Omega-3 fatty acids for the treatment of depression: Systematic review and meta-analysis. Mol. Psychiatry 2012, 17, 1272–1282. [Google Scholar] [CrossRef] [PubMed]
- Grosso, G.; Pajak, A.; Marventano, S.; Castellano, S.; Galvano, F.; Bucolo, C.; Drago, F.; Caraci, F. Role of omega-3 fatty acids in the treatment of depressive disorders: A comprehensive meta-analysis of randomized clinical trials. PLoS ONE 2014, 9, e96905. [Google Scholar] [CrossRef] [PubMed]
- Appleton, K.M.; Sallis, H.; Perry, R.; Ness, A.; Churchill, R. Omega-3 fatty acids for depression in adults. Cochrane Database Syst. Rev. 2015, 11, CD004692. [Google Scholar] [CrossRef]
- Sublette, M.E.; Galfalvy, H.C.; Hibbeln, J.R.; Keilp, J.G.; Malone, K.M.; Oquendo, M.A.; Mann, J.J. Polyunsaturated fatty acid associations with dopaminergic indices in major depressive disorder. Int. J. Neuropsychopharmacol. 2014, 17, 383–391. [Google Scholar] [CrossRef]
- Sarris, J.; Kavanagh, D.J.; Byrne, G. Adjuvant use of nutritional and herbal medicines with antidepressants, mood stabilizers and benzodiazepines. J. Psychiatr. Res. 2010, 44, 32–41. [Google Scholar] [CrossRef]
- Rocha, C.M.; Kac, G. High dietary ratio of omega-6 to omega-3 polyunsaturated acids during pregnancy and prevalence of post-partum depression. Matern. Child. Nutr. 2012, 8, 36–48. [Google Scholar] [CrossRef]
- Ravindran, A.V.; Balneaves, L.G.; Faulkner, G.; Ortiz, A.; McIntosh, D.; Morehouse, R.L.; Ravindran, L.; Yatham, L.N.; Kennedy, S.H.; Lam, R.W.; et al. Canadian network for mood and anxiety treatments (CANMAT) 2016 clinical guidelines for the management of adults with major depressive disorder: Section 5. complementary and alternative medicine treatments. Can. J. Psychiatry 2016, 61, 576–587. [Google Scholar] [CrossRef]
- Appleton, K.M.; Sallis, H.; Perry, R.; Ness, A.; Churchill, R. ω-3 Fatty acids for major depressive disorder in adults: An abridged Cochrane review. BMJ Open 2016, 6, e010172. [Google Scholar] [CrossRef]
- Hallahan, B.; Ryan, T.; Hibbeln, J.R.; Murray, I.T.; Glynn, S.; Ramsden, C.E.; SanGiovanni, J.P.; Davis, J.M. Efficacy of omega-3 highly unsaturated fatty acids in the treatment of depression. Br. J. Psychiatry 2016, 209, 192–201. [Google Scholar] [CrossRef] [PubMed]
- Mocking, R.J.T.; Harmsen, I.; Assies, J.; Koeter, M.W.J.; Ruhé, H.G.; Schene, H.A. Meta-analysis and meta-regression of omega-3 polyunsaturated fatty acid supplementation for major depressive disorder. Transl. Psychiatry 2016, 6, e756. [Google Scholar] [CrossRef] [PubMed]
- Schefft, C.; Kilarski, L.L.; Bschor, T.; Köhler, S. Efficacy of adding nutritional supplements in unipolar depression: A systematic review and meta-analysis. Eur. Neuropsychopharmacol. 2017, 27, 1090–1109. [Google Scholar] [CrossRef] [PubMed]
- Ciappolino, V.; Delvecchio, G.; Agostoni, C.; Mazzocchi, A.; Altamura, A.C.; Brambilla, P. The role of n-3 polyunsaturated fatty acids (n-3PUFAs) in affective disorders. J. Affect. Disord. 2017, 224, 32–47. [Google Scholar] [CrossRef] [PubMed]
- Martins, J.G. EPA but not DHA appears to be responsible for the efficacy of omega-3 long chain polyunsaturated fatty acids supplementation in depression evidence from a meta-analysis of randomized controlled trials. J. Am. Coll. Nutr. 2009, 28, 525–542. [Google Scholar] [CrossRef]
- Sublette, M.E.; Ellis, S.P.; Geant, A.L.; Mann, J.J. Meta-analysis of the effects of eicosapentaenoic acid (EPA) in clinical trials in depression. J. Clin. Psychiatry 2011, 72, 1577–1584. [Google Scholar] [CrossRef]
- Rutkofsky, I.H.; Khan, A.S.; Sahito, S.; Kumar, V. The Psychoneuroimmunological role of omega-3 polyunsaturated fatty acids in major depressive disorder and bipolar disorder. Adv. Mind Body Med. 2017, 31, 8–16. [Google Scholar]
- Chiu, C.C.; Huang, S.Y.; Chen, C.C.; Su, K.P. Omega-3 fatty acids are more beneficial in the depressive phase than in the manic phase in patients with bipolar I disorder. J. Clin. Psychiatry 2005, 66, 1613–1614. [Google Scholar] [CrossRef]
- McNamara, R.K.; Jandacek, R.; Rider, T.; Tso, P.; Stanford, K.E.; Hahn, C.G.; Richtand, N.M. Deficits in docosahexaenoic acid and associated elevations in the metabolism of arachidonic acid and saturated fatty acids in the postmortem orbitofrontal cortex of patients with bipolar disorder. Psychiatry Res. 2008, 160, 285–299. [Google Scholar] [CrossRef]
- Hirashima, F.; Parow, A.M.; Stoll, A.L.; Demopulos, C.M.; Damico, K.E.; Rohan, M.L.; Eskesen, J.G.; Zuo, C.S.; Cohen, B.M.; Renshaw, P.F. Omega-3 fatty acid treatment and T(2) whole brain relaxation times in bipolar disorder. Am. J. Psychiatry 2019, 161, 1922–1924. [Google Scholar] [CrossRef]
- Keck, P.E., Jr.; Mintz, J.; McElroy, S.L.; Freeman, M.P.; Suppes, T.; Frye, M.A.; Altshuler, L.L.; Kupka, R.; Nolen, W.A.; Leverich, G.S.; et al. Double-blind, randomized, placebo-controlled trials of ethyl-eicosapentanoate in the treatment of bipolar depression and rapid cycling bipolar disorder. Biol. Psychiatry 2006, 60, 1020–1022. [Google Scholar] [CrossRef]
- Frangou, S.; Lewis, M.; McCrone, P. Efficacy of ethyl-eicosapentaenoic acid in bipolar depression: Randomised double-blind placebo-controlled study. Br. J. Psychiatry 2006, 188, 46–50. [Google Scholar] [CrossRef] [PubMed]
- Murphy, B.L.; Stoll, A.L.; Harris, P.Q.; Ravichandran, C.; Babb, S.M.; Carlezon, W.A., Jr.; Cohen, B.M. Omega-3 fatty acid treatment, with or without cytidine, fails to show therapeutic properties in bipolar disorder: A double-blind, randomized add-on clinical trial. J. Clin. Psychopharmacol. 2012, 32, 699–703. [Google Scholar] [CrossRef] [PubMed]
- Stoll, A.L.; Severus, W.E.; Freeman, M.P.; Rueter, S.; Zboyan, H.A.; Diamond, E.; Cress, K.K.; Marangell, L.B. Omega 3 fatty acids in bipolar disorder: A preliminary double-blind, placebo-controlled trial. Arch. Gen. Psychiatry 1999, 56, 407–412. [Google Scholar] [CrossRef] [PubMed]
- Gracious, B.L.; Chirieac, M.C.; Costescu, S.; Finucane, T.L.; Youngstrom, E.A.; Hibbeln, J.R. Randomized, placebo-controlled trial of flax oil in pediatric bipolar disorder. Bipolar Disord. 2010, 12, 142–154. [Google Scholar] [CrossRef]
- Turnbull, T.; Cullen-Drill, M.; Smaldone, A. Efficacy of omega-3 fatty acid supplementation on improvement of bipolar symptoms: A systematic review. Arch. Psychiat. Nurs. 2008, 22, 305–311. [Google Scholar] [CrossRef]
- Montgomery, P.; Richardson, A.J. Omega-3 fatty acids for bipolar disorder. Cochrane Database Syst. Rev. 2008, 2, CD005169. [Google Scholar] [CrossRef]
- Kraguljac, N.V.; Montori, V.M.; Pavuluri, M.; Chai, H.S.; Wilson, B.S.; Unal, S.S. Efficacy of omega-3 fatty acids in mood disorders—A systematic review and metaanalysis. Psychopharmacol. Bull. 2009, 42, 39–54. [Google Scholar]
- Sarris, J.; Mischoulon, D.; Schweitzer, I. Omega-3 for bipolar disorder: Meta-analyses of use in mania and bipolar depression. J. Clin. Psychiatry 2012, 73, 81–86. [Google Scholar] [CrossRef]
- Khandaker, G.M.; Zammit, S.; Lewis, G.; Jones, P.B. Association between serum C-reactive protein and DSM-IV generalized anxiety disorder in adolescence: Findings from the ALSPAC cohort. Neurobiol. Stress. 2016, 4, 55–61. [Google Scholar] [CrossRef]
- Polokowski, A.R.; Shakil, H.; Carmichael, C.L.; Reigada, L.C. Omega-3 fatty acids and anxiety: A systematic review of the possible mechanisms at play. Nutr. Neurosci. 2018, 28, 1–11. [Google Scholar] [CrossRef]
- Thesing, C.S.; Bot, M.; Milaneschi, Y.; Giltay, E.J.; Penninx, B.W.J.H. Omega-3 polyunsaturated fatty acid levels and dysregulations in biological stress systems. Psychoneuroendocrinology 2018, 97, 206–215. [Google Scholar] [CrossRef] [PubMed]
- Buydens-Branchey, L.; Branchey, M. n-3 polyunsaturated fatty acids decrease anxiety feelings in a population of substance abusers. J. Clin. Psychopharmacol. 2006, 26, 661–665. [Google Scholar] [CrossRef] [PubMed]
- Su, K.P.; Tseng, P.T.; Lin, P.Y.; Okubo, R.; Chen, T.Y.; Chen, Y.W.; Matsuoka, Y.J. Association of Use of Omega-3 Polyunsaturated Fatty Acids With Changes in Severity of Anxiety Symptoms: A Systematic Review and Meta-analysis. JAMA Netw. Open. 2018, 1, e182327. [Google Scholar] [CrossRef] [PubMed]
- Fux, M.; Benjamin, J.; Nemets, B. A placebo-controlled cross-over trial of adjunctive EPA in OCD. J. Psychiatr. Res. 2004, 38, 323–335. [Google Scholar] [CrossRef]
- Matsuoka, Y.; Nishi, D.; Hamazaki, K.; Yonemoto, N.; Matsumura, K.; Noguchi, H.; Hashimoto, K.; Hamazaki, T. Docosahexaenoic acid for selective prevention of posttraumatic stress disorder among severely injured patients: A randomized, placebo-controlled trial. J. Clin. Psychiatry 2015, 76, e1015-22. [Google Scholar] [CrossRef]
- Matsuoka, Y.J.; Hamazaki, K.; Nishi, D.; Hamazaki, T. Change in blood levels of eicosapentaenoic acid and posttraumatic stress symptom: A secondary analysis of data from a placebo-controlled trial of omega3 supplements. J. Affect. Disord. 2016, 205, 289–291. [Google Scholar] [CrossRef] [PubMed]
- Matsumura, K.; Noguchi, H.; Nishi, D.; Hamazaki, K.; Hamazaki, T.; Matsuoka, Y.J. Effects of omega-3 polyunsaturated fatty acids on psychophysiological symptoms of post-traumatic stress disorder in accident survivors: A randomized, double-blind, placebo-controlledtrial. J. Affect. Disord. 2017, 224, 27–31. [Google Scholar] [CrossRef]
- Buydens-Branchey, L.; Branchey, M.; Hibbeln, J.R. Associations between increases in plasma n-3 polyunsaturated fatty acids following supplementation and decreases in anger and anxiety in substance abusers. Prog. Neuropsychopharmacol. Biol. Psychiatry 2008, 32, 568–575. [Google Scholar] [CrossRef]
- De Vries, G.J.; Mocking, R.; Lok, A.; Assies, J.; Schene, A.; Olff, M. Fatty acid concentrations in patients with posttraumatic stress disorder compared to healthy controls. J. Affect. Disord. 2016, 205, 351–359. [Google Scholar] [CrossRef]
- Alquraan, L.; Alzoubi, K.H.; Hammad, H.; Rababa’h, S.Y.; Mayyas, F. Omega-3 fatty acids prevent post-traumatic stress disorder-induced memory impairment. Biomolecules 2019, 9, 100. [Google Scholar] [CrossRef]
- Beltz, B.S.; Tlusty, M.F.; Benton, J.L.; Sandeman, D.C. Omega-3 fatty acids upregulate adult neurogenesis. Neurosci. Lett. 2007, 415, 154–158. [Google Scholar] [CrossRef] [PubMed]
- Calderon, F.; Kim, H.Y. Docosahexaenoic acid promotes neurite growth in hippocampal neurons. J. Neurochem. 2004, 90, 979–988. [Google Scholar] [CrossRef] [PubMed]
- Kitamura, T.; Saitoh, Y.; Takashima, N.; Murayama, A.; Niibori, Y.; Ageta, H.; Sekiguchi, M.; Sugiyama, H.; Inokuchi, K. Adult neurogenesis modulates the hippocampus-dependent period of associative fear memory. Cell 2009, 139, 814–827. [Google Scholar] [CrossRef] [PubMed]
- Matsuoka, Y.; Nishi, D.; Nakaya, N.; Sone, T.; Hamazaki, K.; Hamazaki, T.; Koido, Y. Attenuating posttraumatic distress with omega-3 polyunsaturated fatty acids among disaster medical assistance team members after the Great East Japan Earthquake: The APOP randomized controlled trial. BMC Psychiatry 2011, 11, 132. [Google Scholar] [CrossRef] [PubMed]
- Ginty, A.T.; Conklin, S.M. Preliminary evidence that acute long-chain omega-3 supplementation reduces cardiovascular reactivity to mental stress: A randomized and placebo controlled trial. Biol. Psychol. 2012, 89, 269–272. [Google Scholar] [CrossRef] [PubMed]
- Hamazaki, K.; Itomura, M.; Huan, M.; Nishizawa, H.; Sawazaki, S.; Tanouchi, M.; Watanabe, S.; Hamazaki, T.; Terasawa, K.; Yazawa, K. Effect of omega-3 fatty acid-containing phospholipids on blood catecholamine concentrations in healthy volunteers: A randomized, placebo-controlled, double-blind trial. Nutrition 2005, 21, 705–710. [Google Scholar] [CrossRef] [PubMed]
- Matsumura, K.; Noguchi, H.; Nishi, D.; Matsuoka, Y. The effect of omega-3 fatty acids on psychophysiological assessment for the secondary prevention of posttraumatic stress disorder: An open-label pilot study. Glob. J. Health Sci. 2012, 4, 3–9. [Google Scholar] [CrossRef]
- Spence, J.D.; Thornton, T.; Muir, A.D.; Westcott, N.D. The effect of flax seed cultivars with differing content of alpha-linolenic acid and lignans on responses to mental stress. J. Am. Coll. Nutr. 2003, 22, 494–501. [Google Scholar] [CrossRef]
- Song, C.; Li, X.; Leonard, B.E.; Horrobin, D.F. Effects of dietary n-3 or n6 fatty acids on interleukin-1beta-induced anxiety, stress, and inflammatory responses in rats. J. Lipid Res. 2003, 44, 1984–1991. [Google Scholar] [CrossRef]
- Stevens, L.J.; Zentall, S.S.; Deck, J.L.; Abate, M.; Watkins, B.; Lipp, S.; Burgess, J. Essential fatty acid metabolism in boys with attention-deficit hyperactivity disorder. Am. J. Clin. Nutr. 1995, 62, 761–768. [Google Scholar] [CrossRef]
- Bonvicini, C.; Faraone, S.V.; Scassellati, C. Attention-deficit hyperactivity disorder in adults: A systematic review and meta-analysis of genetic, pharmacogenetic and biochemical studies. Mol. Psychiatry 2016, 21, 872–884. [Google Scholar] [CrossRef] [PubMed]
- Martins, B.P.; Bandarra, N.M.; Figueiredo-Braga, M. The role of marine omega-3 in human neurodevelopment, including autism spectrum disorders and attention-deficit/hyperactivity disorder—a review. Crit. Rev. Food Sci. Nutr. 2019, 18, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Voigt, R.G.; Llorente, A.M.; Jensen, C.L.; Fraley, J.; Berretta, M.; Heird, W. A randomized, double-blind, placebo-controlled trial of docosahexaenoic acid supplementation in children with attention-deficit/hyperactivity disorder. J. Pediatr. 2001, 139, 189–196. [Google Scholar] [CrossRef] [PubMed]
- Richardson, A.J.; Puri, B.K. A randomized double-blind, placebo-controlled study of the effects of supplementation with highly unsaturated fatty acids on ADHD-related symptoms in children with specific learning difficulties. Prog. Neuropsychopharmacol. Biol. Psychiatry 2002, 26, 233–239. [Google Scholar] [CrossRef]
- Stevens, L.; Zhang, W.; Peck, L.; Kuczek, T.; Grevstad, N.; Mahon, A.; Zentall, S.S.; Eugene Arnold, L.; Burgess, J.R. EFA supplementation in children with inattention, hyperactivity, and other disruptive behaviors. Lipids 2003, 38, 1007–1021. [Google Scholar] [CrossRef]
- Hirayama, S.; Hamazaki, T.; Terasawa, K. Effect of docosahexaenoic acid-containing food administration on symptoms of attention-deficit/hyperactivity disorder—A placebo-controlled double-blind study. Eur. J. Clin. Nutr. 2004, 58, 467–473. [Google Scholar] [CrossRef]
- Sinn, N.; Bryan, J.; Wilson, C. Cognitive effects of polyunsaturated fatty acids in children with attention deficit hyperactivity disorder symptoms: A randomised controlled trial. Prostaglandins Leukort. Essent. Fat. Acids 2008, 78, 311–326. [Google Scholar] [CrossRef]
- Vaisman, N.; Kaysar, N.; Zaruk-Adasha, Y.; Pelled, D.; Brichon, G.; Zwingelstein, G.; Bodennec, J. Correlation between changes in blood fatty acid composition and visual sustained attention performance in children with inattention: Effect of dietary n-3 fatty acids containing phospholipids. Am. J. Clin. Nutr. 2008, 87, 1170–1180. [Google Scholar] [CrossRef]
- Johnson, M.; Ostlund, S.; Fransson, G.; Kadesjö, B.; Gillberg, C. Omega-3/omega-6 fatty acids for attention deficit hyperactivity disorder: A randomized placebo-controlled trial in children and adolescents. J. Atten. Disord. 2009, 12, 394–401. [Google Scholar] [CrossRef]
- Bélanger, S.A.; Vanasse, M.; Spahis, S.; Sylvestre, M.P.; Lippé, S.; L’heureux, F.; Ghadirian, P.; Vanasse, C.M.; Levy, E. Omega-3 fatty acid treatment of children with attention-deficit hyperactivity disorder: A randomized, double-blind, placebo-controlled study. Paediatr. Child. Health 2009, 14, 89–98. [Google Scholar] [CrossRef]
- Gustafsson, P.A.; Birberg-Thornberg, U.; Duchén, K.; Landgren, M.; Malmberg, K.; Pelling, H.; Strandvik, B.; Karlsson, T. EPA supplementation improves teacher-rated behaviour and oppositional symptoms in children with ADHD. Acta Paediatr. 2010, 99, 1540–1549. [Google Scholar] [CrossRef] [PubMed]
- Perera, H.; Jeewandara, K.C.; Seneviratne, S.; Guruge, C. Combined !3 and !6 supplementation in children with attention-deficit hyperactivity disorder (ADHD) refractory to methylphenidate treatment. J. Child. Neurol. 2012, 27, 747–753. [Google Scholar] [CrossRef] [PubMed]
- Manor, I.; Magen, A.; Keidar, D.; Rosen, S.; Tasker, H.; Cohen, T.; Richter, Y.; Zaaroor-Regev, D.; Manor, Y.; Weizman, A. The effect of phosphatidylserine containing Omega3 fatty-acids on attention-deficit hyperactivity disorder symptoms in children: A double-blind placebo-controlled trial, followed by an open-label extension. Eur. Psychiatry 2012, 27, 335–342. [Google Scholar] [CrossRef] [PubMed]
- Milte, C.M.; Sinn, N.; Buckley, J.D.; Coates, A.M.; Young, R.M.; Howe, P.R. Polyunsaturated fatty acids, cognition and literacy in children with ADHD with and without learning difficulties. J. Child. Health Care 2011, 15, 299–311. [Google Scholar] [CrossRef] [PubMed]
- Widenhorn-Müller, K.; Schwanda, S.; Scholz, E.; Spitzer, M.; Bode, H. Effect of supplementation with long-chain ω-3 polyunsaturated fatty acids on behavior and cognition in children with attention deficit/hyperactivity disorder (ADHD): A randomized placebo-controlled intervention trial. Prostaglandins Leukort. Essent. Fat. Acids 2014, 91, 49–60. [Google Scholar] [CrossRef] [PubMed]
- Bos, D.J.; Oranje, B.; Veerhoek, E.S.; van Diepen, R.; Weusten, J.; Demmelmair, H.; Koletzko, B.; De Sain-Van Der Velden, M.G.; Eilander, A.; Hoeksma, M.; et al. Reduced symptoms of inattention after dietary omega-3 fatty acid supplementation in boys with and without attention deficit/hyperactivity disorder. Neuropsychopharmacology 2015, 40, 2298–2306. [Google Scholar] [CrossRef]
- Bloch, M.H.; Qawasmi, A. Omega-3 fatty acid supplementation for the treatment of children with attention-deficit/hyperactivity disorder symptomatology: Systematic review and meta-analysis. J. Am. Acad. Child. Adolesc. Psychiatry 2011, 50, 991–1000. [Google Scholar] [CrossRef]
- Sonuga-Barke, E.J.S.; Brandeis, D.; Cortese, S.; Daley, D.; Ferrin, M.; Holtmann, M.; Stevenson, J.; Danckaerts, M.; van der Oord, S.; Döpfner, M.; et al. Nonpharmacological interventions for ADHD: Systematic review and metaanalyses of randomized controlled trials of dietary and psychological treatments. Am. J. Psychiatry 2013, 170, 275–289. [Google Scholar] [CrossRef]
- Gillies, D.; Sinn, J.K.h.; Lad, S.S.; Leach, M.J.; Ross, M.J. Polyunsaturated fatty acids (PUFA) for attention deficit hyperactivity disorder (ADHD) in children and adolescents. Cochrane Database Syst. Rev. 2012, 7, CD007986. [Google Scholar] [CrossRef]
- Gow, R.V.; Hibbeln, J.R.; Parletta, N. Current evidence and future directions for research with omega-3 fatty acids and attention deficit hyperactivity disorder. Curr. Opin. Clin. Nutr. Metab. Care 2015, 18, 133–138. [Google Scholar] [CrossRef]
- Chang, J.P.; Su, K.P.; Mondelli, V.; Pariante, C.M. Omega-3 polyunsaturated fatty acids in youths with Attention deficit hyperactivity disorder: A systematic review and meta-analysis of clinical trials and biological studies. Neuropsychopharmacology 2018, 43, 534–545. [Google Scholar] [CrossRef] [PubMed]
- Bell, J.G.; Sargent, J.R.; Tocher, D.R.; Dick, J.R. Red blood cell fatty acid compositions in a patient with autistic spectrum disorder: A characteristic abnormality in neurodevelopmental disorders? Prostaglandins Leukort. Essent. Fat. Acids 2000, 63, 21–25. [Google Scholar] [CrossRef]
- Bell, J.G.; MacKinlay, E.E.; Dick, J.R.; MacDonald, D.J.; Boyle, R.M.; Glen, A.C.A. Essential fatty acids and phospholipase A2 in autistic spectrum disorders. Prostaglandins Leukort. Essent. Fat. Acids 2004, 71, 201–204. [Google Scholar] [CrossRef] [PubMed]
- Vancassel, S.; Durand, G.; Barthelemy, C. Plasma fatty acid levels in autistic children. Prostaglandins Leukort. Essent. Fat. Acids 2001, 65, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Amminger, G.P.; Berger, G.E.; Schäfer, M.R.; Klier, C.; Friedrich, M.H.; Feucht, M. Omega-3 fatty acids supplementation in children with autism: A double-blind randomized, placebo controlled pilot study. Biol. Psychiatry 2007, 61, 551–553. [Google Scholar] [CrossRef]
- Bent, S.; Bertoglio, K.; Ashwood, P.; Bostrom, A.; Hendren, R.L. A pilot randomized controlled trial of omega-3 fatty acids for autism spectrum disorder. J. Autism Dev. Disord. 2011, 41, 545–554. [Google Scholar] [CrossRef]
- Bent, S.; Hendren, R.L.; Zandi, T.; Law, K.; Choi, J.; Widjaja, F.; Kalb, L.; Nestle, J.; Law, P. Internet-based, randomized, controlled trial of omega-3 fatty acids for hyperactivity in Autism. J. Am. Acad. Child. Adolesc. Psychiatry 2014, 53, 658–666. [Google Scholar] [CrossRef]
- Yui, K.; Koshiba, M.; Nakamura, S.; Kobayashi, Y. Effects of large doses of arachidonic acid added to docosahexaenoic acid on social impairment in individuals with Autism spectrum disorders. J. Clin. Psychopharmacol. 2012, 32, 200–206. [Google Scholar] [CrossRef]
- Voigt, R.G.; Mellon, M.W.; Katusic, S.K.; Weaver, A.L.; Matern, D.; Mellon, B.; Jensen, C.L.; Barbaresi, W.J. Dietary docosahexaenoic acid supplementation in children with autism. J. Pediatr. Gastroenterol. Nutr. 2014, 58, 715–722. [Google Scholar]
- Mankad, D.; Dupuis, A.; Smile, S.; Roberts, W.; Brian, J.; Lui, T.; Genore, L.; Zaghloul, D.; Iaboni, A.; Marcon, P.M.; et al. A randomized, placebo controlled trial of omega-3 fatty acids in the treatment of young children with autism. Mol. Autism 2015, 6, 18. [Google Scholar] [CrossRef]
- Sheppard, K.W.; Boone, K.M.; Gracious, B.; Klebanoff, M.A.; Rogers, L.K.; Rausch, J.; Bartlett, C.; Coury, D.L.; Keim, S.A. Effect of Omega-3 and -6 supplementation on language in preterm toddlers exhibiting Autism spectrum disorder symptoms. J. Autism Dev. Disord. 2017, 47, 3358–3369. [Google Scholar] [CrossRef] [PubMed]
- Mazahery, H.; Conlon, C.A.; Beck, K.L.; Mugridge, O.; Kruger, M.C.; Stonehouse, W.; Camargo, C.A.; Meyer, B.J.; Jones, B.; von Hurst, P.R. A randomised controlled trial of vitamin D and omega-3 long chain polyunsaturated fatty acids in the treatment of irritability and hyperactivity among children with autism spectrum disorder. J. Steroid Biochem. Mol. Biol. 2019, 187, 9–16. [Google Scholar] [CrossRef] [PubMed]
- Horvath, A.; Łukasik, J.; Szajewska, H. ω-3 fatty acid supplementation does not affect Autism spectrum disorder in children: A systematic review and meta-analysis. J. Nutr. 2017, 147, 367–376. [Google Scholar] [CrossRef] [PubMed]
- Cheng, J.; Eskenazi, B.; Widjaja, F.; Cordero, J.F.; Hendren, R.L. Improving autism perinatal risk factors: A systematic review. Med. Hypotheses 2019, 127, 26–33. [Google Scholar] [CrossRef]
- Swenne, I.; Rosling, A. Omega-3 essential fatty acid status is improved during nutritional rehabilitation of adolescent girls with eating disorders and weight loss. Acta Paediatr. 2012, 101, 858–861. [Google Scholar] [CrossRef]
- Caspar-Bauguil, S.; Montastier, E.; Galinon, F.; Frisch-Benarous, D.; Salvayre, R.; Ritz, P. Anorexia nervosa patients display a deficit in membrane long chain poly-unsaturated fatty acids. Clin. Nutr. 2012, 31, 386–390. [Google Scholar] [CrossRef]
- Langan, S.M.; Farrell, P.M. Vitamin E, vitamin A and essential fatty acid status of patients hospitalized for anorexia nervosa. Am. J. Clin. Nutr. 1985, 41, 1054–1056. [Google Scholar] [CrossRef]
- Sirinathsinghji, D.J.; Mills, I.H. Concentration patterns of plasma dehydroepian- drosterone, delta 5-androstenediol and their sulphates, testosterone and cortisol in normal healthy women and in women with anorexia nervosa. Acta Endocrinol. (Copenh.) 1985, 108, 255–260. [Google Scholar] [CrossRef]
- Swenne, I.; Rosling, A.; Tengblad, S.; Vessby, B. Omega-3 polyunsaturated essential fatty acids are associated with depression in adolescents with eating disorders and weight loss. Acta Paediatr. 2011, 100, 1610–1615. [Google Scholar] [CrossRef]
- Shih, P.A.; Woodside, D.B. Contemporary views on the genetics of anorexia nervosa. Eur. Neuropsychopharmacol. 2016, 26, 663–673. [Google Scholar] [CrossRef]
- Ayton, A.K.; Azaz, A.; Horrobin, D.F. A pilot open case series of ethyl EPA supplementation in thetreatment of anorexia nervosa. Prostaglandins Leukort. Essent. Fat. Acids 2004, 71, 205–209. [Google Scholar] [CrossRef] [PubMed]
- Barbarich, N.C.; McConaha, C.W.; Halmi, K.A.; Gendall, K.; Sunday, S.R.; Gaskill, J.; laVia, M.; Frank, G.K.; Brooks, S.; Plotnicov, K.H.; et al. Use of nutritional supplements to increase the efficacy of fluoxetine in the treatment of anorexia nervosa. Int. J. Eat. Disord. 2004, 35, 10–15. [Google Scholar] [CrossRef]
- Balcerzak, P.; Bazynska, A.; Bragoszewska, J.; Niwinski, P.; Popek, L.; Remberk, B.; Rybakowski, F. Polyunsaturated omega-3 fatty acids are not effective for depressive and compulsive symptoms in adolescent girls with anorexia nervosa. Eur. Neuropsychopharmacol. 2016, 26, 729–730. [Google Scholar] [CrossRef]
- Woo, J.; Couturier, J.; Pindiprolu, B.; Picard, L.; Maertens, C.; Leclerc, A.; Findlay, S.; Johnson, N.; Grant, C.; Kimber, M. Acceptability and tolerability of omega-3 fatty acids as adjunctive treatment for children and adolescents with eating disorders. Eat. Disord. 2017, 25, 114–121. [Google Scholar] [CrossRef] [PubMed]
- Manos, B.E.; Bravender, T.D.; Harrison, T.M.; Lange, H.L.H.; Cottrill, C.B.; Abdel-Rasoul, M.; Bonny, A.E. A pilot randomized controlled trial of omega-3 fatty acid supplementation for the treatment of anxiety in adolescents with anorexia nervosa. Int. J. Eat Disord. 2018, 51, 1367–1372. [Google Scholar] [CrossRef]
- Satogami, K.; Tseng, P.T.; Su, K.P.; Takahashi, S.; Ukai, S.; Li, D.J.; Chen, T.Y.; Lin, P.Y.; Chen, Y.W.; Matsuoka, Y.J. Relationship between polyunsaturated fatty acid and eating disorders: Systematic review and meta-analysis. Prostaglandins Leukot. Essent. Fat. Acids 2019, 142, 11–19. [Google Scholar] [CrossRef]
- Di Giacomo, E.; Aspesi, F.; Fotiadou, M.; Arntz, A.; Aguglia, E.; Barone, L.; Bellino, S.; Carpiniello, B.; Colmegna, F.; Lazzari, M.; et al. Unblending borderline personality and bipolar disorders. J. Psychiatr. Res. 2017, 91, 90–97. [Google Scholar] [CrossRef]
- Gajos, J.M.; Beaver, K.M. The effect of omega-3 fatty acids on aggression: A meta-analysis. Neurosci. Biobehav. Rev. 2016, 69, 147–158. [Google Scholar] [CrossRef]
- Bègue, L.; Zaalberg, A.; Shankland, R.; Duke, A.; Jacquet, J.; Kaliman, P.; Pennel, L.; Chanove, M.; Arvers, P.; Bushman, B.J. Omega-3 supplements reduce self-reported physical aggression in healthy adults. Psychiatry Res. 2018, 261, 307–311. [Google Scholar] [CrossRef]
- Choy, O.; Raine, A. Omega-3 supplementation as a dietary intervention to reduce aggressive and antisocial behavior. Curr. Psychiatry Rep. 2018, 20, 32. [Google Scholar] [CrossRef]
- Zanarini, M.C.; Frankenburg, F.R. Omega-3 fatty acid treatment of women with borderline personality disorder: A double blind, placebo-controlled pilot study. Am. J. Psychiatry 2003, 160, 167–169. [Google Scholar] [CrossRef] [PubMed]
- Hallahan, B.; Hibblen, J.R.; Davis, J.M.; Garland, M.R. Omega-3 fatty acids supplementation in patients with recurrent self-harm: Single center double bind randomized controlled trial. Br. J. Psychiatry 2007, 190, 118–122. [Google Scholar] [CrossRef] [PubMed]
- Bellino, S.; Bozzatello, P.; Rocca, G.; Bogetto, F. Efficacy of omega-3fatty acids in the treatment of borderline personality disorder: A study of the association with valproic acid. J. Psychopharmacol. 2014, 28, 125–132. [Google Scholar] [CrossRef] [PubMed]
- Gallagher, A.T.; Byrne, F.; Murray, I.T.; Lally, J.; Davis, J.M.; Garland, M.; Hibbeln, J.R.; Hallahan, B. Lipids and polyunsaturated fatty acid levels in deliberate self-harm: A 10-year follow-up study. Ir. J. Psychol. Med. 2017, 34, 117–126. [Google Scholar] [CrossRef] [PubMed]
- Bozzatello, P.; Rocca, P.; Bellino, S. Combination of omega-3 fatty acids and valproic acid in treatment of borderline personality disorder: A follow-up study. Clin. Drug Investig. 2018, 38, 367–372. [Google Scholar] [CrossRef]
- Stoffers, J.; Völlm, B.A.; Rücker, G.; Timmer, A.; Huband, N.; Lieb, K. Pharmacological interventions for borderline personality disorder. Cochrane Database Syst Rev. 2010, 16, CD005653. [Google Scholar] [CrossRef]
- Lieb, K.; Völlm, B.; Rücker, G.; Timmer, A.; Stoffers, J.M. Pharmacotherapy for borderline personality disorder: Cochrane systematic review of randomised trials. Br. J. Psychiatry 2010, 196, 4–12. [Google Scholar] [CrossRef]
- Boutron, I.; Tubach, F.; Giraudeau, B.; Ravaud, P. Blinding was judged more difficult to achieve and maintain in nonpharmacologic than pharmacologic trials. J. Clin. Epidemiol. 2004, 57, 543–550. [Google Scholar] [CrossRef]
- Freeman, M.P.; Fava, M.; Lake, J.; Trivedi, M.H.; Wisner, K.L.; Mischoulon, D. Complementary and alternative medicine in major depressive disorder: The American psychiatric association task force report. J. Clin. Psychiatry. 2010, 71, 669–681. [Google Scholar] [CrossRef]
- European Food Safety Authority (EFSA) Panelon Dietetic Products, Nutrition and Allergies (NDA). Scientific Opinion on the Tolerable Upper Intake Level of eicosapentaenoic acid (EPA), docosahexaenoic acid (DHA) and docosapentaenoic acid (DPA). EFSA J. 2012, 10, 2815. [Google Scholar]
- Yokoyama, M.; Origasa, H.; Matsuzaki, M.; Matsuzawa, Y.; Saito, Y.; Ishikawa, Y.; Oikawa, S.; Sasaki, J.; Hishida, H.; Itakura, H.; et al. Effects of eicosapentaenoic acid on major coronary events in hypercholesterolaemic patients (JELIS): A randomised open-label, blinded endpoint analysis. Lancet 2007, 369, 1090–1098. [Google Scholar] [CrossRef]
- Tanaka, K.; Ishikawa, Y.; Yokoyama, M.; Origasa, H.; Matsuzaki, M.; Saito, Y.; Matsuzawa, Y.; Sasaki, J.; Oikawa, S.; Hishida, H.; et al. Reduction in the recurrence of stroke by eicosapentaenoic acid for hypercholesterolemic patients: Subanalysis of the JELIS trial. Stroke 2008, 39, 2052–2058. [Google Scholar] [CrossRef] [PubMed]
- MacLean, C.H.; Mojica, W.A.; Morton, S.C.; Pencharz, J.; Hasenfeld Garland, R. Effects of omega-3 fatty acids on lipids and glycemic control in type II diabetes and the metabolic syndrome and on inflammatory bowel disease, rheumatoid arthritis, renal disease, systemic lupus erythematosus, and osteoporosis. Evid. Rep. Technol. Assess. (Summ.) 2004, 89, 1–4. [Google Scholar]
- Hartweg, J.; Perera, R.; Montori, V.; Dinneen, S.; Neil, H.A.; Farmer, A. Omega-3 polyunsaturated fatty acids (HUFA) for type 2 diabetes mellitus. Cochrane Database Syst. Rev. 2008, 1, CD003205. [Google Scholar]
- Hartweg, J.; Farmer, A.J.; Holman, R.R.; Neil, A. Potential impact of omega-3 treatment on cardiovascular disease in type 2 diabetes. Curr. Opin. Lipidol. 2009, 20, 30–38. [Google Scholar] [CrossRef]
- Bloomer, R.J.; Larson, D.E.; Fisher Wellman, K.H.; Galpin, A.J.; Schilling, B.K. Effectt of eicosapentaenoic and docosahexaenoic acid on resting and exercise-induced inflammatory and oxidative stress biomarkers: A randomized, placebo controlled, cross-over study. Lipids Health Dis. 2009, 8, 36. [Google Scholar] [CrossRef]
- VKM (Norwegian Scientific Committee for Food Safety). Opinion of the steering committee of the norwegian scientific committee for food safety: Evaluation of negative and positive health effects of n-3 fatty acids as constituents of food supplements and fortified foods. Available online: http://english.vkm.no/dav/031c000d1a.pdf (accessed on 20 June 2016).
- Freeman, L.M. Beneficial effects of omega-3 fatty acids in cardiovascular disease. J. Small Anim. Pract. 2010, 51, 462–470. [Google Scholar] [CrossRef]
| High-Risk Psychosis | ||||
|---|---|---|---|---|
| Study | Drugs and Dose | Sample | Treatment Duration | Results |
| Amminger et al., 2010 [18] | EPA 700 mg/day + DHA 480 mg/day | 81 individuals UHR | 12 weeks | ↓ progression in psychosis in young UHR patients |
| Amminger et al., 2013 [19] | EPA 700 mg/day + DHA 480 mg/day + 7.6 mg vit. E | 81 young individuals at UHR | 12 weeks | ↓ positive symptoms, negative symptoms and general symptoms, ↑ level of functioning |
| Amminger et al., 2015 [20] | EPA 700 mg/day + DHA 480 mg/day | 81 young individuals at UHR | 12 weeks | ↓ both risk of progression to psychotic disorder and psychiatric morbidity |
| Smesny et al., 2014 [21] | EPA 700 mg/day + DHA 480 mg/day | 81 young individuals at UHR | 12 weeks | normalizing PLA2 activity and d-6-desaturase-mediated metabolism of o-3 and o-6 PUFAs |
| McGorry et al., 2017 [22] | EPA 840 mg/day + DHA 560 mg/day + CBCM | 304 individuals UHR | 24 weeks | not effective under conditions where evidence-based psychosocial treatment is available |
| First-episode Psychosis | ||||
| Study | Drugs and Dose | Sample | Treatment Duration | Results |
| Berger et al., 2007 [23] | Ethyl-EPA 3 g/day | 69 patients | 12 weeks | accelerated treatment response |
| Berger et al., 2008 [24] | EPA 2 g/day | 24 patients | 12 weeks | ↓ of negative symptoms |
| Wood et al., 2010 [25] | EPA 2 g/day | 17 patients | 12 weeks | increased water in hippocampal tissues and positive effect on negative symptoms |
| Emsley et al., 2014 [26] | EPA 2 g/day + DHA 1 g/day + α-LA 300 mg/day | 33 patients | 2 years | relapse prevention of psychotic symptoms |
| Pawelzcyk et al., 2016 [27] | EPA + DHA 2.2 g/day | 71 patients | 26 weeks | ↓ psychotic symptoms measured with PANSS ↓ depressive symptoms ↑ level of functioning |
| Pawelzcyk et al., 2017 [28] | EPA + DHA 2.2 g/day | 71 patients | 26 weeks | improved PANSS negative and general symptoms, along with global functioning |
| Pawelzcyk et al., 2018 [29] | EPA + DHA 2.2 g/day | 71 patients | 26 weeks | ↑ level of telomerase in peripheral blood cells with ↓ depressive symptoms |
| Stable Schizophrenia | ||||
| Study | Drugs and Dose | Sample | Treatment Duration | Results |
| Peet et al., 2001 [30] | EPA or DHA 2 g/day | 45 patients | 12 weeks | ↓ psychotic symptoms measured with PANSS in the group treated with EPA |
| Peet and Horrobin, 2002 [31] | E-EPA 1–4 g/day | 115 patients | 12 weeks | ↓ positive symptoms measured with PANSS, ↓ depressive symptoms |
| Emsley et al., 2002 [32] | ethyl-EPA 3 g/day | 40 patients | 12 weeks | ↓ positive symptoms and negative symptoms measured with PANSS |
| Emsley et al., 2006 [33] | ethyl-EPA 2 g/day | 77 patients | 12 weeks | no efficacy on specific psychotic symptoms |
| Jamilian et al., 2014 [34] | 1 g/day | 60 patients | 8 weeks | ↓ psychotic symptoms measured with PANSS |
| Fenton et al., 2001 [35] | ethyl-EPA 3 g/day | 87 patients | 16 weeks | no significant differences in positive, negative symptoms, mood or cognition |
| Bentsen et al., 2013 [36] | ethyl-EPA 2 g/day | 99 patients | 16 weeks | ↓ impairment of the course of psychosis |
| Qiao et al., 2018 [37] | DHA 360 mg/day + EPA 540 mg/day | 50 patients | 12 weeks | ↓ violence, but no improvement in positive and negative symptoms |
| Robinson et al., 2019 [38] | EPA 740 mg and DHA 400 mg/day | 50 patients | 16 weeks | ↓ confusion, anxiety, depression, irritability and tiredness/fatigue |
| Study | Drugs and Dose | Sample | Treatment Duration | Results |
|---|---|---|---|---|
| Nemets et al., 2002 [56] | ethyl-EPA 2 g/day | 20 patients | 4 weeks | ↓ depressive symptoms measured with HDRS from the second week of treatment |
| Peet and Horrobin, 2002 [57] | ethyl-EPA 1, 2 or 4 g/day add-on standard antidepressant treatment | 70 patients resistant to antidepressant treatment | 12 weeks | ↓ depressive symptoms measured with HDRS, MADRS and BDI in the group treated with 1 g/day of PUFAs |
| LIorente et al., 2003 [58] | DHA 0.2 g/day monotherapy | 99 healthy pregnant women | 16 weeks | no effect on depression |
| Marangell et al., 2003 [59] | add on to standard therapy DHA 2 g/day monotherapy | 36 depressed patients | 12 weeks | no significant differences |
| Su et al., 2003 [60] | ethyl-EPA 4.4 g/day + DHA 2.2 g/day add-on existing antidepressant treatment | 22 patients | 8 weeks | ↓ depressive symptoms measured with HDRS |
| Silvers et al., 2005 [61] | EPA 0.6 g/day +DHA 2.4 g/day added to standard therapy | 77 MDD patients | 12 weeks | no evidence that n-3PUFAs improved mood compared to placebo. Mood improved in both groups within the first 2 weeks of the study |
| Nemets et al., 2006 [62] | ethyl-EPA 0.4 g/day + DHA 0.2 g/day | 20 patients 6–12 years-old | 16 weeks | ↓ depressive symptoms measured with CDRS, CDI and CGI |
| Greyner et al., 2007 [63] | EPA 0.6 g/day + DHA 2.2 g/day add to standard therapy | 83 MDD patients | 16 weeks | no significant differences |
| Freeman et al., 2008 [64] | EPA 1.1 g/day + DHA 0.8 g/day | 59 women | 8 weeks | no benefit on perinatal depressive symptoms |
| Jazayeri et al., 2008 [65] | EPA 1 g/day versus fluoxetine 20 mg/day | 60 patients | 8 weeks | ↓ depressive symptoms in both groups |
| Rees et al., 2008 [66] | ethyl-EPA 0.4 g/day + DHA 1.6 g/day | 26 pregnant patients | 6 weeks | no benefits on depressive symptoms |
| Rogers et al., 2008 [67] | EPA 0.63 g/day + DHA 0.85 g/day monotherapy | 218 mild to moderate depressed patients untreated | 12 weeks | n-3PUFAs not have beneficial or harmful effects on mood in mild to moderate depression. |
| Doornbos et al., 2009 [68] | DHA 0.22 g/day or DHA 0.22 g/day + AA (0.22 g/day arachidonic acid) monotherapy | 119 healthy pregnant women | 28 weeks | red blood cell DHA, AA and DHA/AA ratio did not correlate with EPDS or blues scores |
| Lucas et al., 2009 [69] | EPA 1.05 g/day + DHA 0.25 g/day mono-therapy | 120 patients with psychological distress with or without MDD in comorbidity | 8 weeks | no significant differences |
| Mischoulon et al., 2009 [70] | EPA 1 g/day + (+0.2% dl alphatocopherol) monotherapy | 57 MDD patients | 8 weeks | ↓ depressive symptoms assessed with HDRS, but no statistical significance |
| Makrides et al., 2010 [71] | DHA-rich tuna oil capsules 0.5 g/day mono therapy | 2399 healthy pregnant women at 21 weeks’ gestation | women received assigned capsules daily, from study entry until birth of their child | DHA during pregnancy did not lower levels of postpartum depression |
| Rondanelli et al., 2010, 2011 [72,73] | EPA 1.67 g/day + DHA 0.83 g/day added to existing antidepressant treatment | 46 elderly female residents in a nursing home | 8 weeks | ↓ depressive symptoms assessed with GDS, improvement of phospholipids fatty acids profile |
| Lespérance et al., 2011 [74] | EPA 1.05 g/day + DHA 0.15 g/day | 432 patients with a major depressive episode | 8 weeks | ↓ depressive symptoms only for patients without comorbid anxiety disorders |
| Tajalizadekhoob et al., 2011 [75] | EPA 0.18 g/day + DHA 0.12 g/day add to standard therapy in 55 patients while in 11 monotherapy | 66 patients with mild-to moderate depression aged > 66 years | 24 weeks | low-dose n-3PUFAs have some efficacy in mild to moderate depression |
| Antypa et al., 2012 [76] | EPA 1.74 g/day+ DHA 0.25 g/day added to standard therapy | 71 patients with history of at least one MDD | 4 weeks | no significant effects on memory, attention, cognitive reactivity and depressive symptoms |
| Gertsik et al., 2012 [77] | EPA 0.9 g/day + DHA 0.2 g/day + other n-3 PUFAs (0.1 g/day) added to citalopram | 42 MDD patients taking citalopram | 9 weeks | significantly greater improvement in HDRS scores |
| Krawczyk et al., 2012 [78] | EPA 2.2 g/day + DHA 0.7 g/day + GLA (0.24 g/day) + vit. E added to standard therapy | 21 patients with severe episode of treatment resistant recurring depression | 8 weeks | n-3PUFAs significantly improved HDRS scores |
| Rizzo et al., 2012 [79] | EPA/DHA 2.1/2.5 g of n3-PUFA monotherapy | 46 MMD patients (only women > 66 years old) | 8 weeks | mean GDS score and AA/EPA ratio, in whole blood and RBC membrane phospholipids, were significantly lower |
| Mozzafarri Khoshari et al., 2013 [80] | EPA 1 g/day or DHA 1 g/day added to standard therapy | 81 mild to moderate depressed patients | 12 weeks | ↓ HDRS score compared with those in the DHA or placebo groups |
| Mozurkewich et al., 2013 [81] | EPA 1.06 g/day+ DHA 0.27 g/day or EPA 0.18 g/day + DHA 0.9 g/day mono-therapy | 126 healthy pregnant women | 6-8 weeks | no differences between groups in BDI scores or other depression endpoints |
| Judge et al., 2014 [82] | DHA 0.3 g/day | 42 healthy pregnant women | 8 weeks | ↓ depressive symptoms assessed with PDSS |
| Ginty et al., 2015 [83] | EPA + DHA 1.4 g/day monotherapy | 23 depressed patients | 3 weeks | n-3PUFAs group had a significant reduction in BDI scores over time |
| Mischoulon et al., 2015 [84] | EPA 1 g/day or DHA 1 g/day | 196 patients | 8 weeks | EPA and DHA were not superior to placebo |
| Park et al., 2015 [85] | EPA 1140 g/day + DHA 0.6 g/day add to standard therapy | 35 MDD patients | 12 weeks | no significant differences |
| Young et al., 2017 [86] | PEP + EPA 1.4g/day + DHA 0.2 g/day + 0.4 g/day other | 72 patients 7–14 years old | 12 weeks | ↓ co-occurring behaviour symptoms in youth with depression. |
| Gabbay et al., 2018 [87] | 2:1 ratio of EPA to DHA: Initial dose of 1.2 g/day. Doses were raised in increments of 0.6 g/day every 2 weeks (maximum possible dose of 3.6 g/day, combined EPA 2.4 g + DHA 1.2 g) | 51 psychotropic medication-free adolescents with MDD aged 12–19 years | 10 weeks | n-3PUFAs do not appear to be superior to placebo. |
| Jahangard et al., 2018 [89] | n-3 PUFAs (1000 mg/day) + sertraline (50–200 mg/day) | 50 MDD patients | 12 weeks | ↓ depression, anxiety, sleep and patients’ competencies to regulate their emotions. |
| Tayama et al., 2019 [90] | DHA 500 mg/day + EPA 1000 mg/day | 20 patients with mild to moderate depression | 12 weeks | no significant differences |
| Study | Drugs and Dose | Sample | Treatment Duration | Results |
|---|---|---|---|---|
| Hirashima et al., 2004 [109] | High dose: EPA, 5.0–5.2 g/day; DHA, 3.0–3.4 g/day; other, 0.3–1.7 g/day | 21 patients | 4 weeks | no significant differences |
| Chiu et al., 2005 [107] | valproate 2 g/day and 4.4g/day EPA + 2.4 g/day DHA | 16 newly hospitalised patients in the acute manic phase of bipolar disorder | 4 weeks | no significant differences |
| Keck et al., 2006 [110] | EPA 6 g/day in addition to at least one mood stabilizer | 121 patients with bipolar depression or rapid cycling bipolar disorder | 4 months | no significant differences |
| Frangou et al., 2006 [111] | ethyl-EPA 1 or 2 g/day added to stable psychotropic medications | 75 patients | 12 weeks | ↓ depressive symptoms measured with HDRS |
| Murphy et al., 2012 [112] | omega-3 fatty acids plus cytidine, omega-3 fatty acid plus placebo or only placebo in addition to a mood stabilizer | 45 patients with type I bipolar disorder | 4 months | no benefits of omega-3 fatty acids on affective symptoms |
| Stoll et al., 1999 [113] | EPA 6.2 g/day + DHA 3.4 g/day | 30 patients | 16 weeks | ↓ depressive symptoms measured with HDRS |
| Gracious et al., 2010 [114] | ALA in addition to psychotropic medication | children and adolescent with bipolar I or II disorder | 16 weeks | significant improvement of overall symptom severity compared with placebo |
| PTSD | ||||
|---|---|---|---|---|
| Study | Drugs and Dose | Sample | Treatment Duration | Results |
| Matsuoka et al., 2015 [125] | DHA 1.47 g/day + EPA 0.147 g/day | 110 patients | 12 weeks | not superior to placebo for the secondary prevention of PTSD symptoms |
| Matsuoka et al., 2016 [126] | DHA 1.47 g/day + EPA 0.147 g/day | 110 patients | 12 weeks | ↑ erythrocyte level of EPA associated with ↓ PTSD symptoms |
| Matsumura et al., 2017 [127] | DHA 1.47 g/day and EPA 0.147 g/day | 83 patients | 12 weeks | effective for the secondary prevention of psychophysiological symptoms of PTSD |
| OCD | ||||
| Study | Drugs and Dose | Sample | Treatment Duration | Results |
| Fux et al., 2004 [124] | EPA 2 g/day + stable dose of SSRIs | 11 patients with OCD | 6 weeks | no effect on anxious, obsessive-compulsive and depressive symptoms |
| Substance use disorder | ||||
| Study | Drugs and Dose | Sample | Treatment Duration | Results |
| Buydens-Branchey & Barnchey, 2006 [122] | EPA + DHA at high dose (3 g/day) | 13 patients with substance abuse | 12 weeks | ↓ anxious symptoms |
| Buydens-Branchey et al., 2008 [128] | EPA + DHA at high dose (3 g/day) | 22 patients with substance abuse | 12 weeks | ↓ anger and anxiety levels |
| Study | Drugs and Dose | Sample | Treatment Duration | Results |
|---|---|---|---|---|
| Voigt et al., 2001 [143] | DHA 0.345 g/day versus placebo. With ADHD medication | 63 children (6–12 years old) with ADHD | 4 months | no statistically significant improvement in any ADHD symptoms compared to placebo |
| Richardson et al., 2002 [144] | EPA 0.186 g/day + DHA 0.480 g/day + linolenic acid 0.864 g/day + arachidonic acid 0.042 g/day versus placebo | 41 children with ADHD-like symptoms | 12 weeks | improvement of cognitive problems and general behaviour in the group treated with PUFAs than placebo |
| Stevens et al., 2003 [145] | DHA 0.48 g/day + EPA 0.08 g/day + arachidonic acid 0.04 g/day + gamma-linolenic acid 0.096 g/day versus placebo. No ADHD medications | 50 children with ADHD-like symptoms | 4 months | no significant differences |
| Hirayama et al., 2004 [146] | EPA 0.1 g/day + DHA 0.5 g/day versus placebo Mostly without ADHD medications (only six subjects had been under medications) | 40 children with ADHD | 2 months | no evidence of efficacy of omega-3 fatty acids compared to placebo |
| Sinn and Bryan., 2008 [147] | EPA 93 mg/day + DHA 29 mg/day + gamma-linolenic acid 10 mg/day versus placebo. No ADHD medications | 132 children (7 to 12 years) with ADHD | improved in inattention, hyperactivity and impulsivity in most ADHD scales in parents reports; no improvement in teachers reports Limits: No ADHD diagnosis (reported ADHD symptoms) | |
| Vaisman et al., 2008 [148] | EPA 0.156 g/day + DHA 0.095 g/day or EPA 0.153 g/day + DHA 0.096 g/day or placebo | 83 children with ADHD | 12 weeks | significantly improved executive functioning |
| Johnson et al., 2009 [149] | EPA 0.558 g/day + DHA 0.174 g/day + gamma linoleic acid 0.06 g/day versus placebo. Only one patient with ADHD medication | 75 children and adolescents 8–18 years old with ADHD | 3 months | no evidence of efficacy of omega-3 fatty acids compared to placebo |
| Bélanger et al. 2009 [150] | EPA 0.02–0.025 g/kg/day + DHA 0.85–0.105 g/kg/day versus placebo. No ADHD medications | 26 children | 16 weeks | improvement in inattention and global ADHD symptoms only in the first phase of the study (weeks 0 to 15) |
| Gustafsson et al., 2010 [151] | EPA 0.5 g/day | 92 children (7 to 12 years) with ADHD | 15 weeks | two ADHD subgroups (oppositional and less hyperactive/impulsive children) improved symptoms |
| Perera et al., 2012 [152] | omega-3 + omega-6 versus placebo. With ADHD medications | 98 children (6 to 12 years) with ADHD diagnosis | 6 months | improved behavior and learning in restlessness, aggressiveness, completing work and academic performance, but not in inattention, impulsiveness and cooperation with parents and teachers |
| Manor et al., 2012 [153] | 0.3 g of PS and 0.120 g of EPA + DHA (EPA/DHA ratio of 2:1) | 200 children with ADHD | 15 weeks | improved ADHD symptoms |
| Milte et al., 2012 [154] | EPA-rich oil (providing EPA 1.109 g/day and DHA 0.108 g/day, DHA-rich oil (providing EPA 0.264 g/day and DHA 1.032 g/day) versus an omega-6 PUFAs oil. No ADHD medications | 90 children (7 to 12 years old) with ADHD | 4 months | no statistically significant differences between the two groups |
| Widenhorn-Müller et al., 2014 [155] | EPA 0.6 g/day + DHA 0.120 g/day. No ADHD medications | 95 children (6 to 12 years) with ADHD | 16 weeks | improved working memory function, but no effects on other cognitive measures or behavioural symptoms in the study population |
| Bos et al., 2015 [156] | EPA 0.65 g/day + DHA 0.65 g/day | 40 young boys (8 to14 years ) with ADHD | 16 weeks | ↓ symptoms of ADHD, both for individuals with ADHD and typically developing children. |
| Study | Drugs and Dose | Sample | Treatment Duration | Results |
|---|---|---|---|---|
| Amminger et al., 2007 [165] | EPA 0.84 /day + DHA 0.7 g/day | 13 children (aged 5 to 17 years) with autistic disorders accompanied by severe tantrums, aggression or self-injurious behavior | 6 weeks | improvement of hyperactivity and stereotypy |
| Bent et al., 2011 [166] | 1.3 g/day of omega-3 fatty acids (and 1.1 g of DHA + EPA) | 27 children (aged 3 to 8 years) with autism spectrum disorder | 12 weeks | improvement of hyperactivity |
| Bent et al., 2014 [167] | 1.3 g/day of omega-3 fatty acids (and 1.1 g of DHA + EPA) | 57 children with autism spectrum disorder | 6 weeks | improvement of hyperactivity |
| Yui et al., 2012 [168] | large doses of ARA (40 mg/day) added to DHA (40 mg/day) | 13 patients with autism spectrum disorder | 16 weeks | improved impaired social interaction by up-regulating signal transduction. |
| Voigt et al., 2014 [169] | DHA 0.2 g/day or placebo | 48 children (3 to 10 years) with autism | 6 months | no improvement core symptoms of autism |
| Mankad et al., 2015 [170] | from 0.75 to 1.5 g/day of EPA + DHA | 38 children (2 to 5 years) with autism spectrum disorder | 6 months | no significant differences |
| Sheppard et al., 2017 [171] | Daily doses of Omega-3-6-9 Junior treatment (including 338 mg EPA, 225 mg DHA, 83 mg GLA and 306 mg total omega-9 fatty acids) | 31 children 18–38 months of age born at ≤29 weeks of gestation | 3 months | evidence of efficacy of omega-3 and -6 fatty acid supplementation in improving aspects of early language development in children at risk for ASD |
| Mazaheri et al., 2019 [172] | vitamin D (2000 IU/day, VID), omega-3 LCPUFA (722 mg/day DHA, OM) or both (2000 IU/day vitamin D + 722 mg/day DHA, VIDOM) | 117 children (2,5 to 8 years) with autism spectrum disorder | 12 months | vitamin D and omega-3 LCPUFA reduced irritability symptoms; vitamin D also reduced hyperactivity symptoms |
| Study | Drugs and Dose | Sample | Treatment Duration | Results |
|---|---|---|---|---|
| Ayton et al., 2004 [181] | 1 g/day E-EPA in addition to standard treatment | 7 young patients with anorexia nervosa | 3 months | significant growth during E-EPA supplementation |
| Barbarich et al., 2004 [182] | tryptophan, vitamins, minerals and essential fatty acids (DHA 0.6 g/day and arachadonic acid 0.18 g/day) + fluoxetine | 26 patients with anorexia nervosa | 6 months | no significant difference in weight gain, anxiety or obsessive-compulsive symptoms |
| Pirog-Balcerzak et al., 2016 [183] | EPA 0.558 g/day DHA 0.174 g/day + gamma linolenic acid 0.06 g/day, vs. placebo | 61 patients with anorexia nervosa | 10 weeks | not effective for depressive and compulsive symptoms |
| Woo et al., 2017 [184] | EPA 300 mg/day + DHA 200 mg/day + standard treatment | 21 patients with eating disorders | 8 weeks | significant increase in mean percent ideal body weight, but no significant differences in eating disorder, anxiety and depression symptoms |
| Manos et al., 2018 [185] | EPA 2.12 g/day + DHA 0.6 g/day | 24 adolescent females with anorexia nervosa | 12 weeks | no significant differences |
| Study | Drugs and Dose | Sample | Treatment Duration | Results |
|---|---|---|---|---|
| Zanarini and Frankenburg, 2003 [191] | EPA 1 g/day (with no standard psychiatric therapies) | 30 BPD females | 8 weeks | ↓ aggression, ↓ depression |
| Hallahan et al., 2007 [192] | EPA 1.2 g/day + DHA 0.9 g/day (added to the standard psychiatric therapies) | 49 patients with self-defeating behaviors (39 BPD patients) | 12 weeks | ↓ depression, ↓ parasuicidal behaviours, ↓ stress reactivity |
| Bellino et al., 2014 [193] | EPA (1.2 g/day) + DHA (0.6 g/day) in combination with valproic acid (800–1300 mg/day) versus valproic acid (800–1300 mg/day) (plasma range: 50–100 μg/mL) | 43 BPD patients | 12 weeks | ↓ severity of BPDSI, ↓ impulsive behavioural dyscontrol, ↓ anger, ↓ self-mutilating conduct |
| Gallagher et al., 2017 [194] | n-3 PUFAs including EPA or DHA | 40 individuals who self-harmed and 40 controls | 10 years | ↓ self-harm, ↓depressive symptoms and ↓ impulsivity |
| Bozzatello et al., 2018 [195] | EPA + DHA + valproic acid | 34 patients with borderline personality disorder | 24 weeks | ↓ outbursts of anger |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bozzatello, P.; Rocca, P.; Mantelli, E.; Bellino, S. Polyunsaturated Fatty Acids: What is Their Role in Treatment of Psychiatric Disorders? Int. J. Mol. Sci. 2019, 20, 5257. https://doi.org/10.3390/ijms20215257
Bozzatello P, Rocca P, Mantelli E, Bellino S. Polyunsaturated Fatty Acids: What is Their Role in Treatment of Psychiatric Disorders? International Journal of Molecular Sciences. 2019; 20(21):5257. https://doi.org/10.3390/ijms20215257
Chicago/Turabian StyleBozzatello, Paola, Paola Rocca, Emanuela Mantelli, and Silvio Bellino. 2019. "Polyunsaturated Fatty Acids: What is Their Role in Treatment of Psychiatric Disorders?" International Journal of Molecular Sciences 20, no. 21: 5257. https://doi.org/10.3390/ijms20215257
APA StyleBozzatello, P., Rocca, P., Mantelli, E., & Bellino, S. (2019). Polyunsaturated Fatty Acids: What is Their Role in Treatment of Psychiatric Disorders? International Journal of Molecular Sciences, 20(21), 5257. https://doi.org/10.3390/ijms20215257






