The Antithrombotic Function of Sphingosine-1-Phosphate on Human Adipose-Stem-Cell-Recellularized Tissue Engineered Vascular Graft In Vitro
Abstract
1. Introduction
2. Materials and Methods
2.1. Ethics Assurance
2.2. Preparation of the DHUA (Decellularized Human Umbilical Arteries)
2.3. Isolation, Culture, and Characterization of hASC and Human Umbilical Vein Endothelial Cells (HUVEC)
2.3.1. The hASC (Human Adipose Stem Cells)
2.3.2. HUVEC
2.4. Adhesion and Proliferation of hASCs on DHUA with S1P
2.5. Antithrombotic Assay for TEVGs (Tissue Engineerined Vascular Grafts)
2.5.1. Reconstitution of TEVGs In Vitro
2.5.2. Coagulation and Kinetic Clotting Tests
2.5.3. Platelet Adhesion Test
2.6. The Effect of S1P on the Expression of Syndecan-1 and CD 31 of hASCs
2.7. Statistical Analysis
3. Results
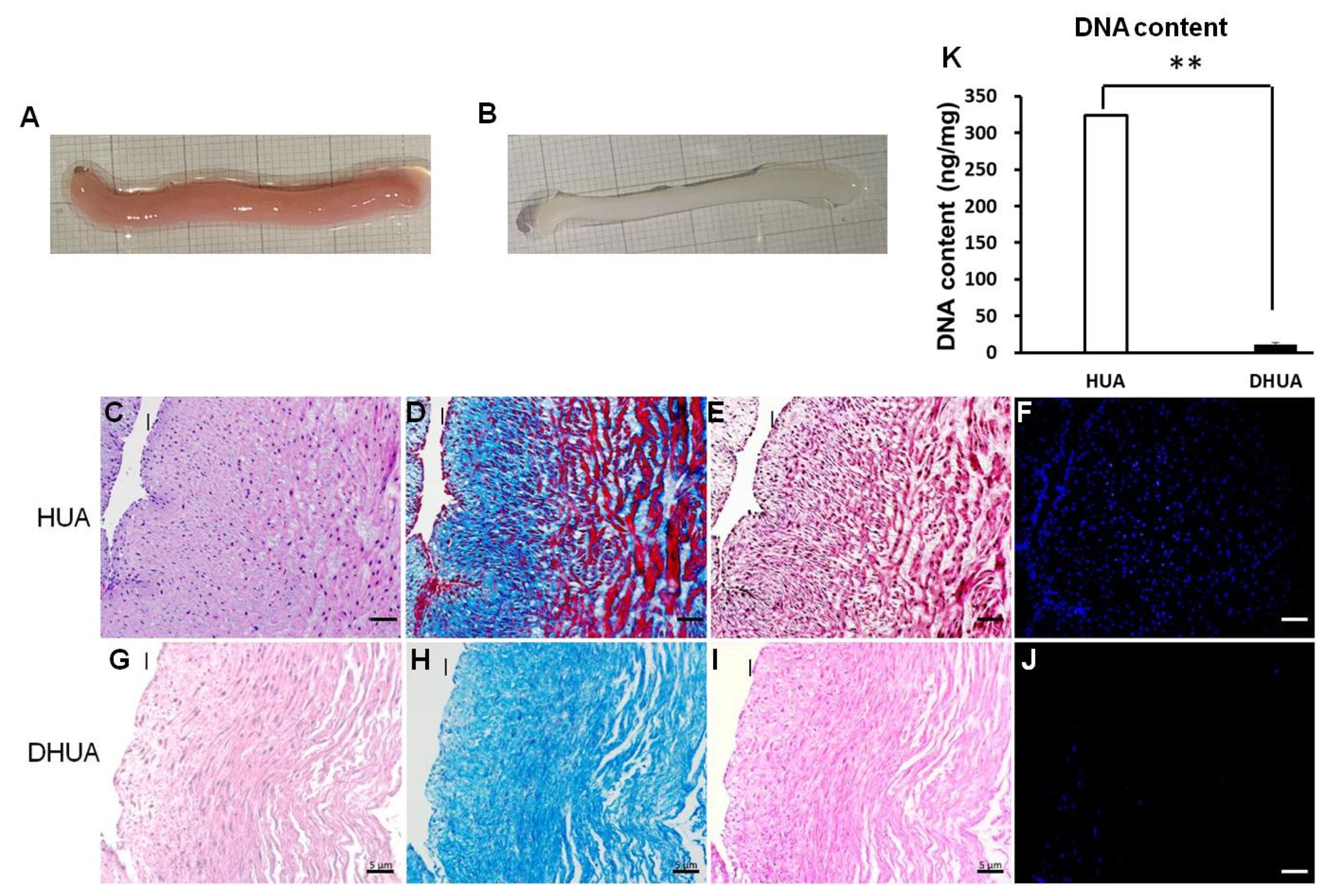
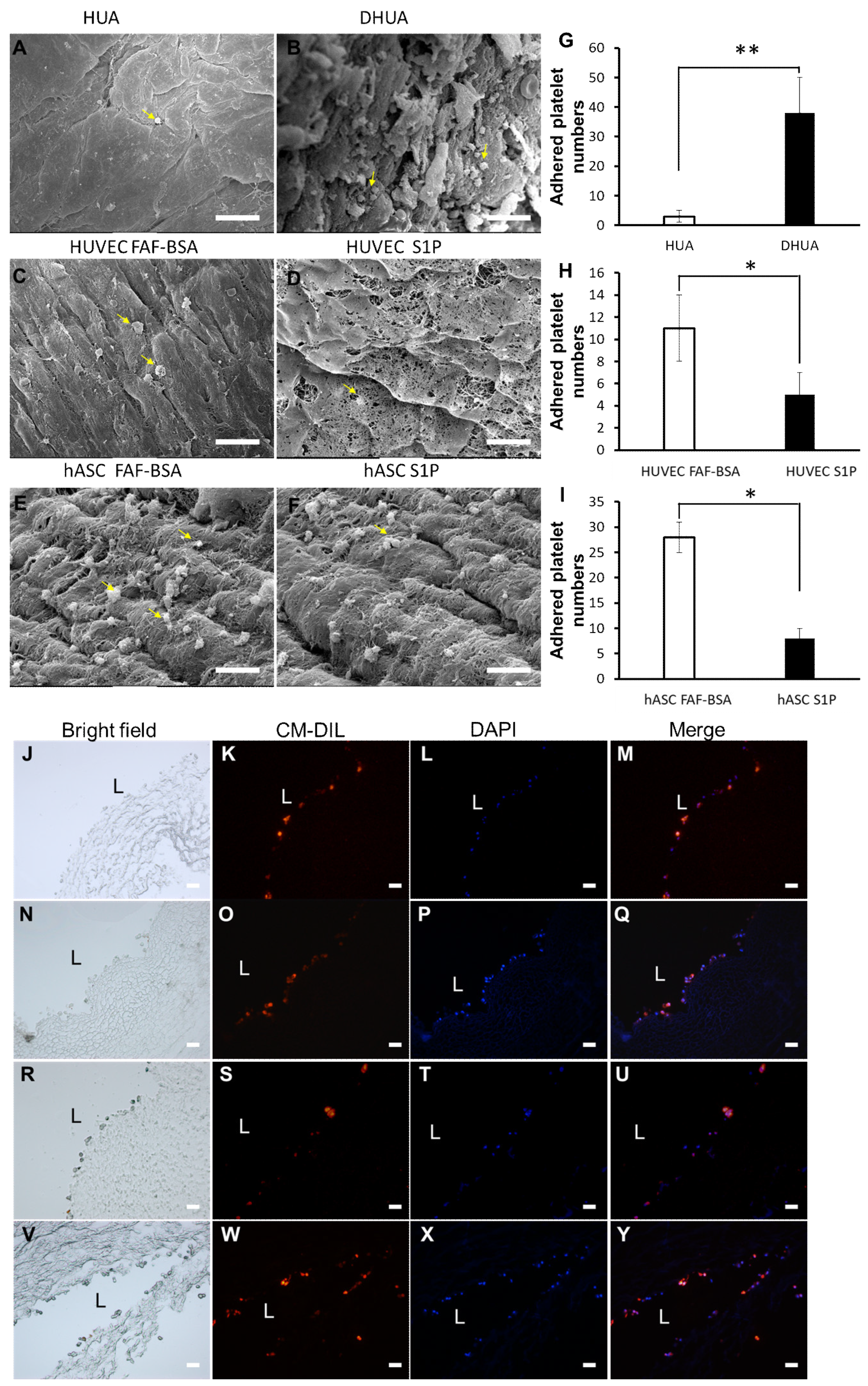
3.1. Characterization of DHUA
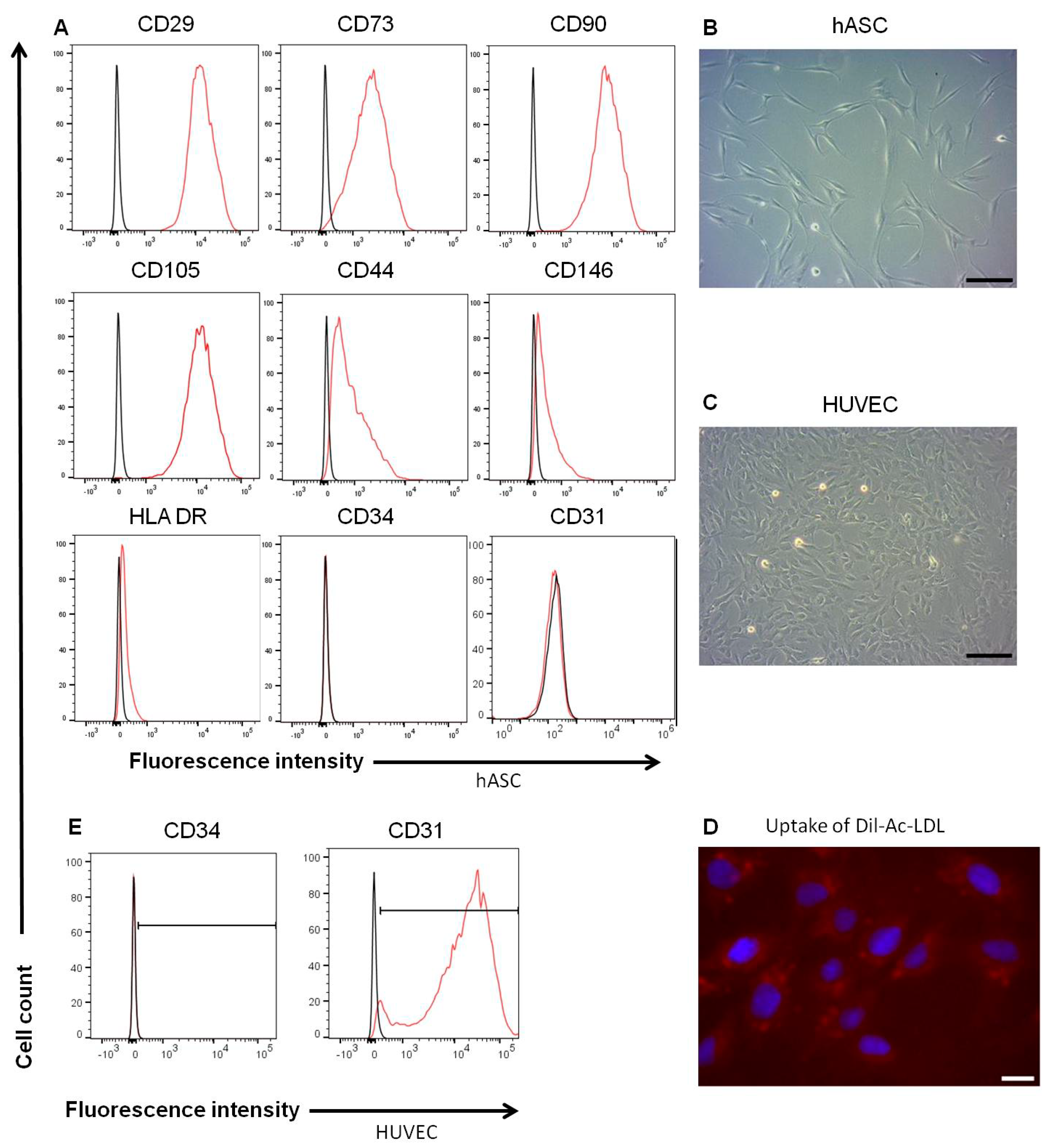
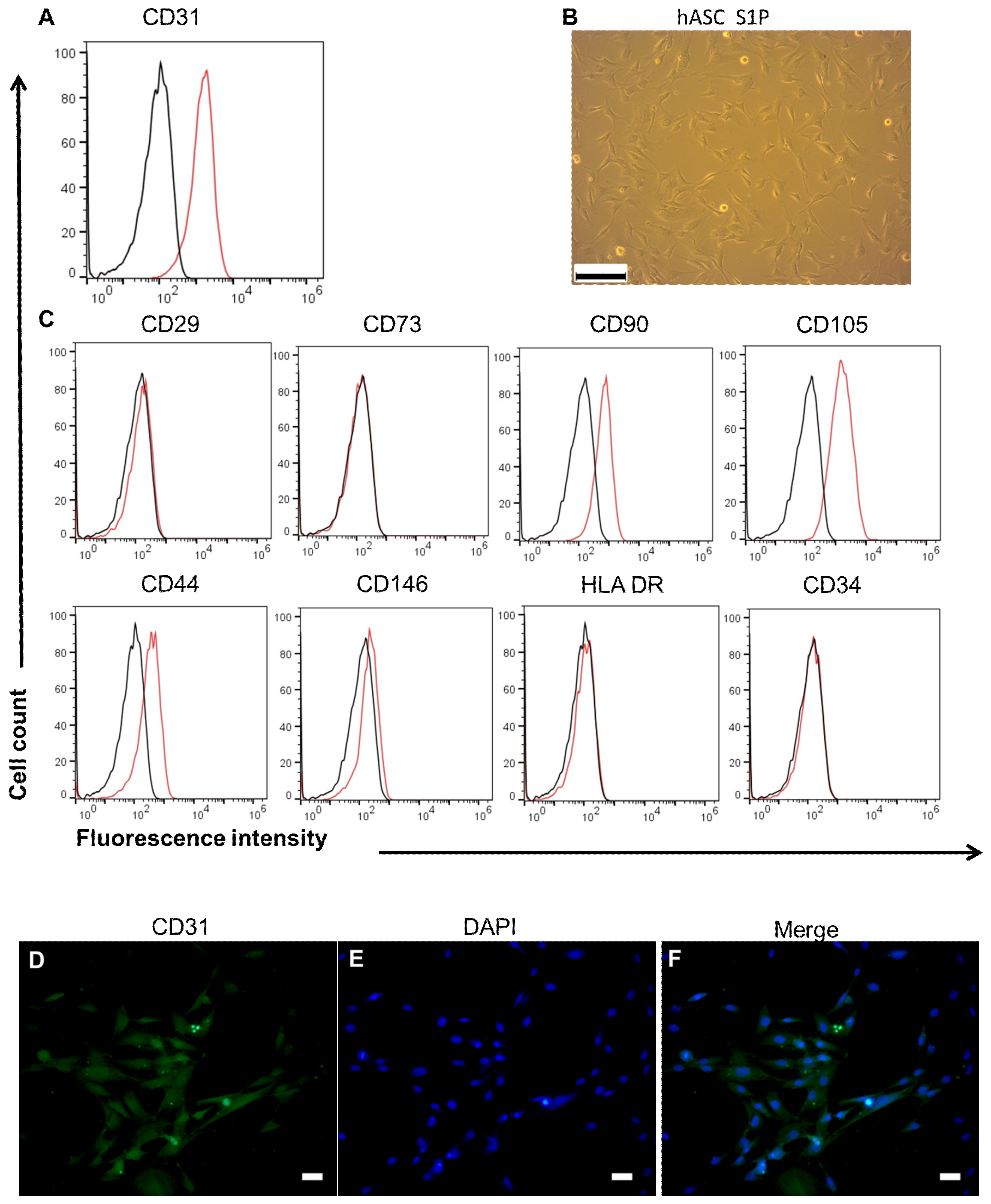
3.2. Characterics of hASC and HUVEC
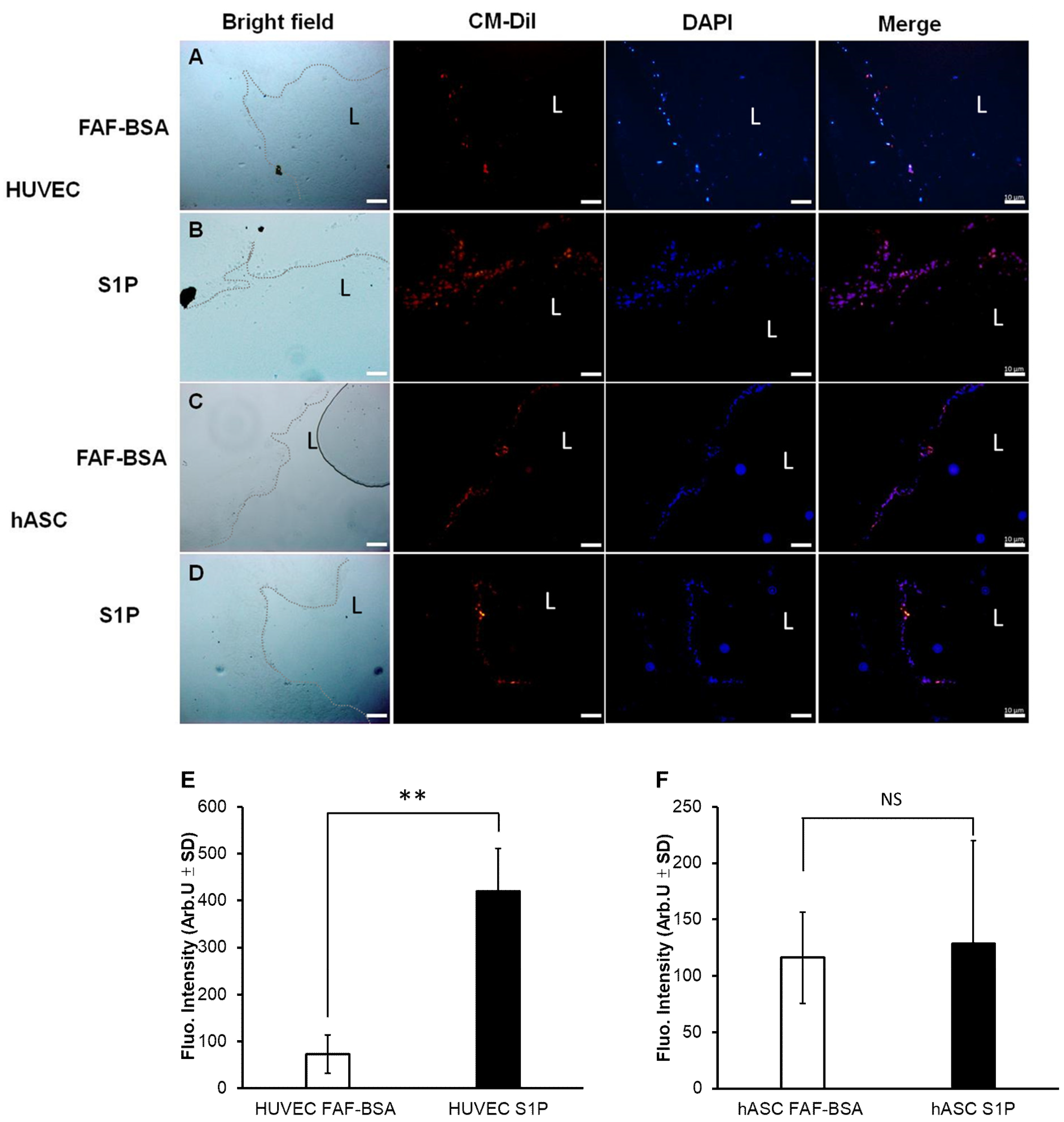
3.3. The Effect of S1P on Adhesion and Proliferation of hASCs on DHUA
3.4. The Effect of S1P on Coagulation and Kinetic Clotting Time of hASC-Recellularized DHUA
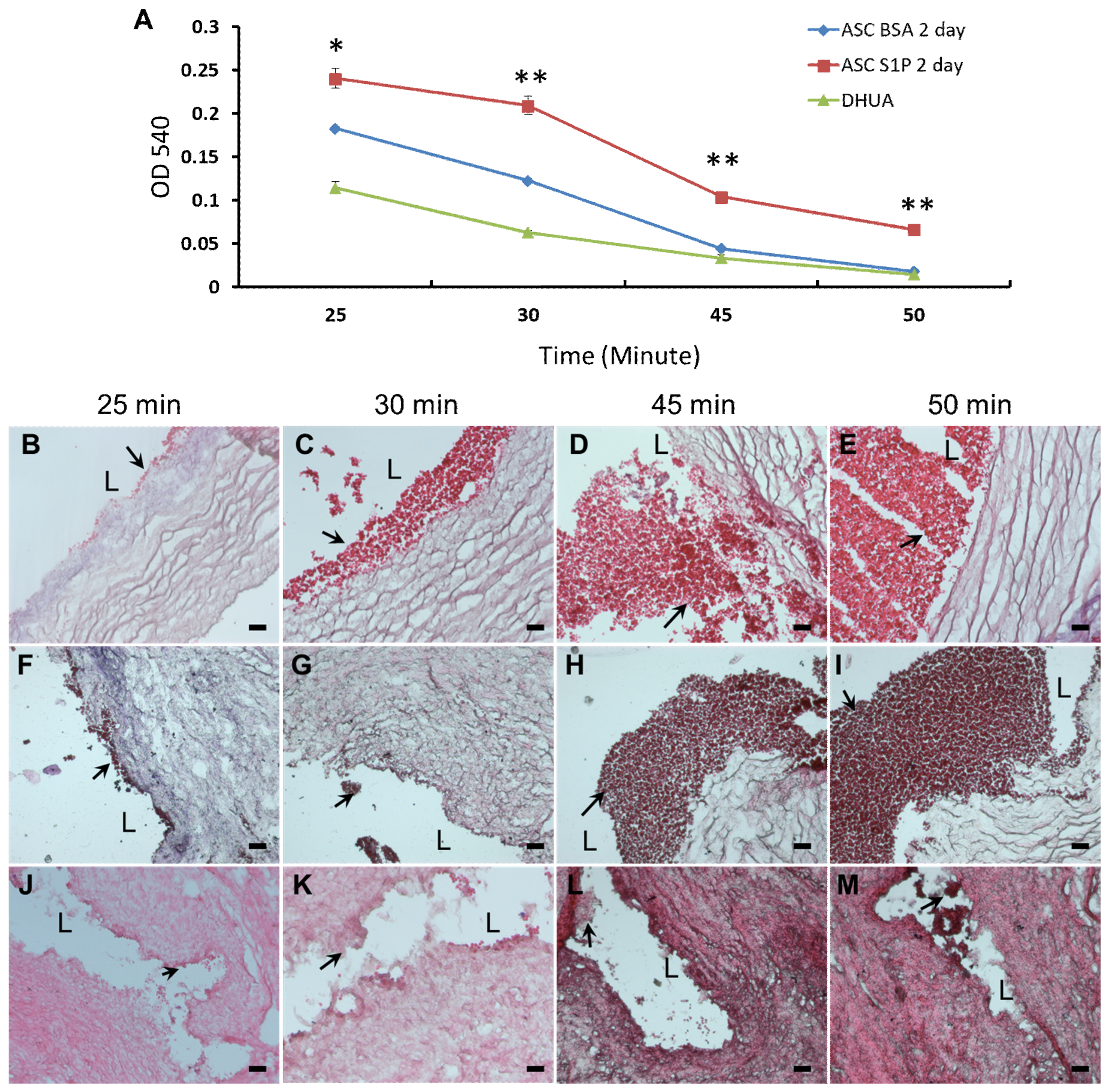
3.5. The Effect of S1P on the Platelet Adhesion of hASC-Recellularized DHUA
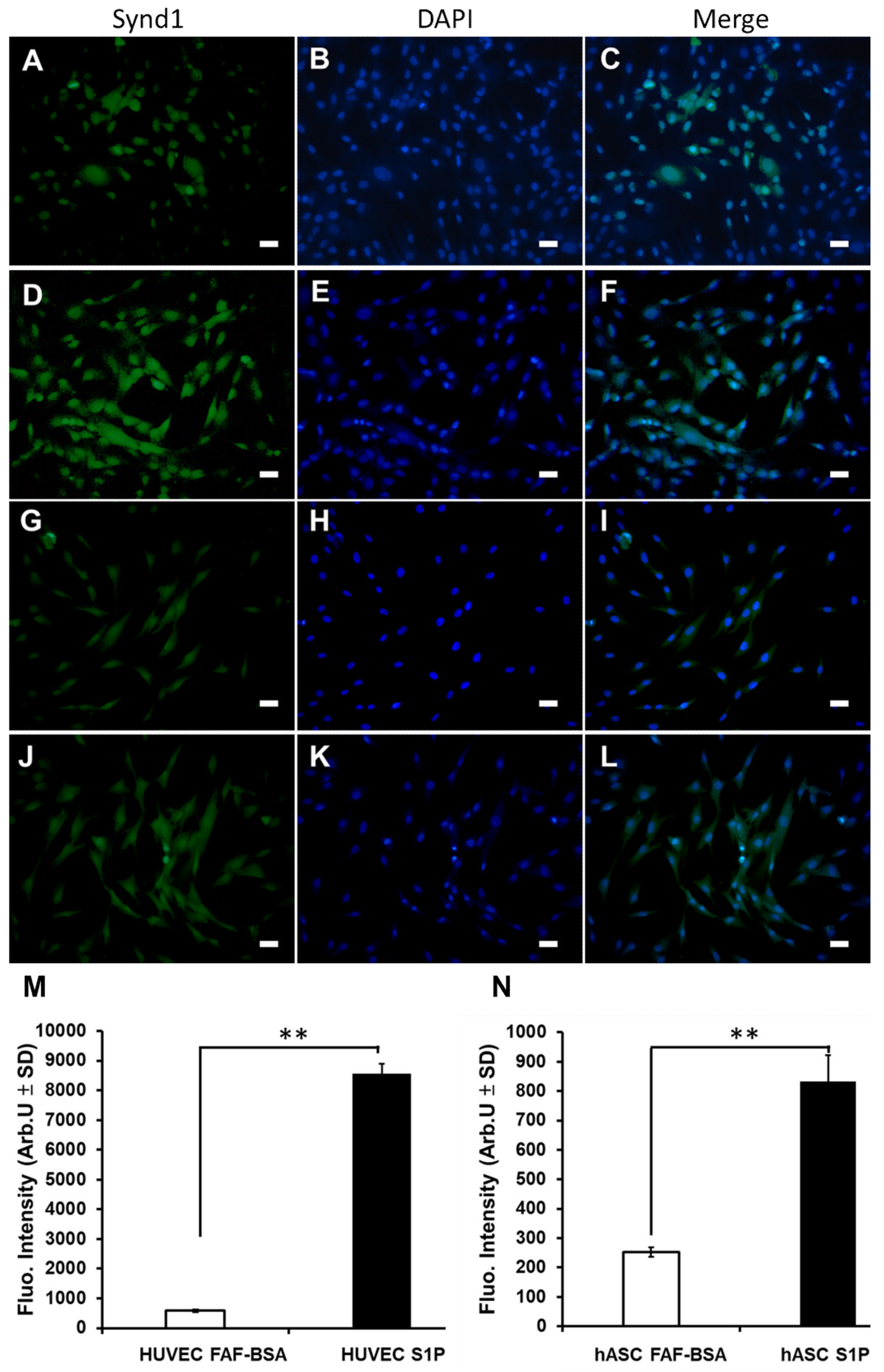
3.6. The Effect of S1P on EC Differentiation and SDC-1 Expression of hASCs In Vitro
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Mizuno, H.; Tobita, M.; Uysal, A.C. Concise review: Adipose-derived stem cells as a novel tool for future regenerative medicine. Stem Cells 2012, 30, 804–810. [Google Scholar] [CrossRef] [PubMed]
- Argentati, C.; Morena, F.; Bazzucchi, M.; Armentano, I.; Emiliani, C.; Martino, S. Adipose stem cell translational applications: From Bench-to-Bedside. Int. J. Mol. Sci. 2018, 19, 3475. [Google Scholar] [CrossRef]
- Dragoo, J.; Samimi, B.; Zhu, M.; Hame, S.; Thomas, B.; Lieberman, J.; Hedrick, M.; Benhaim, P. Tissue-engineered cartilage and bone using stem cells from human infrapatellar fat pads. J. Bone Joint Surg. Br. 2003, 85, 740–747. [Google Scholar] [CrossRef] [PubMed]
- Mauney, J.R.; Nguyen, T.; Gillen, K.; Kirker-Head, C.; Gimble, J.M.; Kaplan, D.L. Engineering adipose-like tissue in vitro and in vivo utilizing human bone marrow and adipose-derived mesenchymal stem cells with silk fibroin 3D scaffolds. Biomaterials 2007, 28, 5280–5290. [Google Scholar] [CrossRef] [PubMed]
- James, R.; Kumbar, S.; Laurencin, C.; Balian, G.; Chhabra, A. Tendon tissue engineering: Adipose-derived stem cell and GDF-5 mediated regeneration using electrospun matrix systems. Biomed. Mater. 2011, 6, 025011. [Google Scholar] [CrossRef]
- Klar, A.S.; Güven, S.; Biedermann, T.; Luginbühl, J.; Böttcher-Haberzeth, S.; Meuli-Simmen, C.; Meuli, M.; Martin, I.; Scherberich, A.; Reichmann, E. Tissue-engineered dermo-epidermal skin grafts prevascularized with adipose-derived cells. Biomaterials 2014, 35, 5065–5078. [Google Scholar] [CrossRef]
- DiMuzio, P.; Fischer, L.; McIlhenny, S.; DiMatteo, C.; Golesorhki, N.; Grabo, D.; Tarola, N.; Mericli, A.; Shapiro, I.; Tulenko, T. Development of a tissue-engineered bypass graft seeded with stem cells. Vascular 2006, 14, 338–342. [Google Scholar] [CrossRef]
- Borschel, G.H.; Huang, Y.-C.; Calve, S.; Arruda, E.M.; Lynch, J.B.; Dow, D.E.; Kuzon, W.M.; Dennis, R.G.; Brown, D.L. Tissue engineering of recellularized small-diameter vascular grafts. Tissue Eng. 2005, 11, 778–786. [Google Scholar] [CrossRef]
- Skovrind, I.; Harvald, E.B.; Belling, H.J.; Jørgensen, C.D.; Lindholt, J.S.; Andersen, D.C. Concise review: Patency of small-diameter tissue-engineered vascular grafts: A meta-analysis of preclinical trials. Stem. Cells. Transl. Med. 2019, 8, 671–680. [Google Scholar] [CrossRef]
- Krawiec, J.T.; Weinbaum, J.S.; Liao, H.T.; Ramaswamy, A.K.; Pezzone, D.J.; Josowitz, A.D.; D’Amore, A.; Rubin, J.P.; Wagner, W.R.; Vorp, D.A. In vivo functional evaluation of tissue-engineered vascular grafts fabricated using human adipose-derived stem cells from high cardiovascular risk populations. Tissue Eng. Part A 2016, 22, 765–775. [Google Scholar] [CrossRef]
- Arya, D.; Chang, S.; DiMuzio, P.; Carpenter, J.; Tulenko, T.N. Sphingosine-1-phosphate promotes the differentiation of adipose-derived stem cells into endothelial nitric oxide synthase (eNOS) expressing endothelial-like cells. J. Biomed. Sci. 2014, 21, 55. [Google Scholar] [CrossRef] [PubMed]
- Cao, Y.; Sun, Z.; Liao, L.; Meng, Y.; Han, Q.; Zhao, R.C. Human adipose tissue-derived stem cells differentiate into endothelial cells in vitro and improve postnatal neovascularization in vivo. Biochem. Biophys. Res. Commun. 2005, 332, 370–379. [Google Scholar] [CrossRef] [PubMed]
- Fischer, L.J.; McIlhenny, S.; Tulenko, T.; Golesorkhi, N.; Zhang, P.; Larson, R.; Lombardi, J.; Shapiro, I.; DiMuzio, P.J. Endothelial differentiation of adipose-derived stem cells: Effects of endothelial cell growth supplement and shear force. J. Surg. Res. 2009, 152, 157–166. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez, L.V.; Alfonso, Z.; Zhang, R.; Leung, J.; Wu, B.; Ignarro, L.J. Clonogenic multipotent stem cells in human adipose tissue differentiate into functional smooth muscle cells. Proc. Natl. Acad. Sci. USA 2006, 103, 12167–12172. [Google Scholar] [CrossRef] [PubMed]
- DiMuzio, P.; Tulenko, T. Tissue engineering applications to vascular bypass graft development: The use of adipose-derived stem cells. J. Vasc. Surg. 2007, 45, A99–A103. [Google Scholar] [CrossRef]
- Heydarkhan-Hagvall, S.; Schenke-Layland, K.; Yang, J.Q.; Heydarkhan, S.; Xu, Y.; Zuk, P.A.; MacLellan, W.R.; Beygui, R.E. Human adipose stem cells: A potential cell source for cardiovascular tissue engineering. Cells Tissues Organs 2008, 187, 263–274. [Google Scholar] [CrossRef]
- Krawiec, J.T.; Liao, H.T.; Kwan, L.L.; D’Amore, A.; Weinbaum, J.S.; Rubin, J.P.; Wagner, W.R.; Vorp, D.A. Evaluation of the stromal vascular fraction of adipose tissue as the basis for a stem cell-based tissue-engineered vascular graft. J. Vasc. Surg. 2017, 66, 883–890. [Google Scholar] [CrossRef]
- Lin, C.-H.; Hsia, K.; Tsai, C.-H.; Ma, H.; Lu, J.-H.; Tsay, R.-Y. Decellularized porcine coronary artery with adipose stem cells for vascular tissue engineering. Biomed. Mater. 2019, 14, 045014. [Google Scholar] [CrossRef]
- Bassaneze, V.; Barauna, V.G.; Lavini-Ramos, C.; Kalil, J.; Schettert, I.T.; Miyakawa, A.A.; Krieger, J.E. Shear stress induces nitric oxide-mediated vascular endothelial growth factor production in human adipose tissue mesenchymal stem cells. Stem Cells Dev. 2010, 19, 371–378. [Google Scholar] [CrossRef]
- Chang, C.H.; Huang, Y.L.; Shyu, M.K.; Chen, S.U.; Lin, C.H.; Ju, T.K.; Lu, J.; Lee, H. Sphingosine-1-phosphate induces VEGF-C expression through a MMP-2/FGF-1/FGFR-1-dependent pathway in endothelial cells in vitro. Acta Pharmacol. Sin. 2013, 34, 360–366. [Google Scholar] [CrossRef]
- Lee, H.; Goetzl, E.J.; An, S. Lysophosphatidic acid and sphingosine 1-phosphate stimulate endothelial cell wound healing. Am. J. Physiol. Cell. Physiol. 2000, 278, C612–C618. [Google Scholar] [CrossRef] [PubMed]
- An, S.; Goetzl, E.J.; Lee, H. Signaling mechanisms and molecular characteristics of G protein-coupled receptors for lysophosphatidic acid and sphingosine 1-phosphate. J. Cell. Biochem. Suppl. 1998, 30–31, 147–157. [Google Scholar] [CrossRef]
- Lin, C.I.; Chen, C.N.; Lin, P.W.; Lee, H. Sphingosine 1-phosphate regulates inflammation-related genes in human endothelial cells through S1P1 and S1P3. Biochem. Biophys. Res. Commun. 2007, 355, 895–901. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Zeng, M.; Fan, J.; Tarbell, J.M.; Curry, F.R.; Fu, B.M. Sphingosine-1-phosphate maintains normal vascular permeability by preserving endothelial surface glycocalyx in intact microvessels. Microcirculation 2016, 23, 301–310. [Google Scholar] [CrossRef]
- Hsia, K.; Yang, M.J.; Chen, W.M.; Yao, C.L.; Lin, C.H.; Loong, C.C.; Huang, Y.L.; Lin, Y.T.; Lander, A.D.; Lee, H.; et al. Sphingosine-1-phosphate improves endothelialization with reduction of thrombosis in recellularized human umbilical vein graft by inhibiting syndecan-1 shedding in vitro. Acta Biomater. 2017, 51, 341–350. [Google Scholar] [CrossRef]
- Hsia, K.; Lin, C.-H.; Lee, H.-Y.; Chen, W.-M.; Yao, C.-L.; Chen, C.-C.; Ma, H.; Wang, S.-J.; Lu, J.-H. Sphingosine-1-phosphate in endothelial cell recellularization improves patency and endothelialization of decellularized vascular grafts in vivo. Int. J. Mol. Sci. 2019, 20, 1641. [Google Scholar] [CrossRef]
- Gui, L.; Chan, S.A.; Breuer, C.K.; Niklason, L.E. Novel utilization of serum in tissue decellularization. Tissue Eng. Part C Methods 2010, 16, 173–184. [Google Scholar] [CrossRef]
- Gui, L.; Muto, A.; Chan, S.A.; Breuer, C.K.; Niklason, L.E. Development of decellularized human umbilical arteries as small-diameter vascular grafts. Tissue Eng. Part A 2009, 15, 2665–2676. [Google Scholar] [CrossRef]
- Chang, Y.J.; Shih, D.T.; Tseng, C.P.; Hsieh, T.B.; Lee, D.C.; Hwang, S.M. Disparate mesenchyme-lineage tendencies in mesenchymal stem cells from human bone marrow and umbilical cord blood. Stem Cells 2006, 24, 679–685. [Google Scholar] [CrossRef]
- Kao, I.T.; Yao, C.L.; Chang, Y.J.; Hsieh, T.B.; Hwang, S.M. Chondrogenic differentiation of human mesenchymal stem cells from umbilical cord blood in chemically synthesized thermoreversible polymer. Chin. J. Physiol. 2008, 51, 252–258. [Google Scholar]
- Huang, N.; Chen, Y.R.; Luo, J.M.; Yi, J.; Lu, R.; Xiao, J.; Xue, Z.N.; Liu, X.H. In vitro investigation of blood compatibility of Ti with oxide layers of rutile structure. J. Biomater. Appl. 1994, 8, 404–412. [Google Scholar] [CrossRef] [PubMed]
- Metcalfe, P.; Williamson, L.M.; Reutelingsperger, C.P.; Swann, I.; Ouwehand, W.H.; Goodall, A.H. Activation during preparation of therapeutic platelets affects deterioration during storage: A comparative flow cytometric study of different production methods. Br. J. Haematol. 1997, 98, 86–95. [Google Scholar] [CrossRef]
- Ratcliffe, A. Tissue engineering of vascular grafts. Matrix Biol. 2000, 19, 353–357. [Google Scholar] [CrossRef]
- Thomas, L.V.; Lekshmi, V.; Nair, P.D. Tissue engineered vascular grafts—Preclinical aspects. Int. J. Cardiol. 2013, 167, 1091–1100. [Google Scholar] [CrossRef] [PubMed]
- De Valence, S.; Tille, J.-C.; Mugnai, D.; Mrowczynski, W.; Gurny, R.; Möller, M.; Walpoth, B.H. Long term performance of polycaprolactone vascular grafts in a rat abdominal aorta replacement model. Biomaterials 2012, 33, 38–47. [Google Scholar] [CrossRef] [PubMed]
- Ramaswamy, A.K.; Vorp, D.A.; Weinbaum, J.S. Functional vascular tissue engineering inspired by matricellular proteins. Front. Cardiovasc. Med. 2019, 6, 74. [Google Scholar] [CrossRef]
- Cunnane, E.M.; Weinbaum, J.S.; O’Brien, F.J.; Vorp, D.A. Future perspectives on the role of stem cells and extracellular vesicles in vascular tissue regeneration. Front. Cardiovasc. Med. 2018, 5, 86. [Google Scholar] [CrossRef]
- Roh, J.D.; Sawh-Martinez, R.; Brennan, M.P.; Jay, S.M.; Devine, L.; Rao, D.A.; Yi, T.; Mirensky, T.L.; Nalbandian, A.; Udelsman, B. Tissue-engineered vascular grafts transform into mature blood vessels via an inflammation-mediated process of vascular remodeling. Proc. Natl. Acad. Sci. USA 2010, 107, 4669–4674. [Google Scholar] [CrossRef]
- Naito, Y.; Shinoka, T.; Duncan, D.; Hibino, N.; Solomon, D.; Cleary, M.; Rathore, A.; Fein, C.; Church, S.; Breuer, C. Vascular tissue engineering: Towards the next generation vascular grafts. Adv. Drug Deliv. Rev. 2011, 63, 312–323. [Google Scholar] [CrossRef]
- Kurobe, H.; Maxfield, M.W.; Breuer, C.K.; Shinoka, T. Concise review: Tissue-engineered vascular grafts for cardiac surgery: Past, present, and future. Stem Cells Transl. Med. 2012, 1, 566–571. [Google Scholar] [CrossRef]
- Row, S.; Peng, H.; Schlaich, E.M.; Koenigsknecht, C.; Andreadis, S.T.; Swartz, D.D. Arterial grafts exhibiting unprecedented cellular infiltration and remodeling in vivo: The role of cells in the vascular wall. Biomaterials 2015, 50, 115–126. [Google Scholar] [CrossRef] [PubMed]
- Chen, R.; Cai, X.; Liu, J.; Bai, B.; Li, X. Sphingosine 1-phosphate promotes mesenchymal stem cell-mediated cardioprotection against myocardial infarction via ERK1/2-MMP-9 and Akt signaling axis. Life Sci. 2018, 215, 31–42. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Lv, Y.; Chen, F.; Wang, X.; Zhu, J.; Li, H.; Xiao, J. Co-stimulation of LPAR1 and S1PR1/3 increases the transplantation efficacy of human mesenchymal stem cells in drug-induced and alcoholic liver diseases. Stem Cell Res. Ther. 2018, 9, 161. [Google Scholar] [CrossRef] [PubMed]
- Marycz, K.; Krzak, J.; Maredziak, M.; Tomaszewsk, K.A.; Szczurek, A.; Moszak, K. The influence of metal-based biomaterials functionalized with sphingosine-1-phosphate on the cellular response and osteogenic differentaion potenial of human adipose derived mesenchymal stem cells in vitro. J. Biomater. Appl. 2016, 30, 1517–1533. [Google Scholar] [CrossRef] [PubMed]
- Ishii, I.; Ye, X.; Friedman, B.; Kawamura, S.; Contos, J.J.; Kingsbury, M.A.; Yang, A.H.; Zhang, G.; Brown, J.H.; Chun, J. Marked perinatal lethality and cellular signaling deficits in mice null for the two sphingosine 1-phosphate (S1P) receptors, S1P(2)/LP(B2)/EDG-5 and S1P(3)/LP(B3)/EDG-3. J. Biol. Chem. 2002, 277, 25152–25159. [Google Scholar] [CrossRef]
- Shen, H.; Zhou, E.; Wei, X.; Fu, Z.; Niu, C.; Li, Y.; Pan, B.; Mathew, A.V.; Wang, X.; Pennathur, S. High density lipoprotein promotes proliferation of adipose-derived stem cells via S1P1 receptor and Akt, ERK1/2 signal pathways. Stem Cell Res. Ther. 2015, 6, 95. [Google Scholar] [CrossRef]
- Marycz, K.; Śmieszek, A.; Jeleń, M.; Chrząstek, K.; Grzesiak, J.; Meissner, J. The effect of the bioactive sphingolipids S1P and C1P on multipotent stromal cells—New opportunities in regenerative medicine. Cell. Mol. Biol. Lett. 2015, 20, 510–533. [Google Scholar] [CrossRef]
- Cooper, G.M.; Hausman, R.E.; Hausman, R.E. The Cell: A Molecular Approach; DC2000; ASM Press Washington: Washington, DC, USA, 1996. [Google Scholar]
- Nincheri, P.; Luciani, P.; Squecco, R.; Donati, C.; Bernacchioni, C.; Borgognoni, L.; Luciani, G.; Benvenuti, S.; Francini, F.; Bruni, P. Sphingosine 1-phosphate induces differentiation of adipose tissue-derived mesenchymal stem cells towards smooth muscle cells. Cell. Mol. Life Sci. 2009, 66, 1741–1754. [Google Scholar] [CrossRef]
- Zeng, Y.; Adamson, R.H.; Curry, F.R.; Tarbell, J.M. Sphingosine-1-phosphate protects endothelial glycocalyx by inhibiting syndecan-1 shedding. Am. J. Physiol. Heart. Circ. Physiol. 2014, 306, H363–H372. [Google Scholar] [CrossRef]
- Yoshimura, K.; Suga, H.; Eto, H. Adipose-derived stem/progenitor cells: Roles in adipose tissue remodeling and potential use for soft tissue augmentation. Regen. Med. 2009, 4, 265–273. [Google Scholar] [CrossRef]
- Melchiorri, A.J.; Hibino, N.; Fisher, J.P. Strategies and techniques to enhance the in situ endothelialization of small-diameter biodegradable polymeric vascular grafts. Tissue Eng. Part B Rev. 2013, 19, 292–307. [Google Scholar] [CrossRef] [PubMed]
- Hanson, S.; D’Souza, R.N.; Hematti, P. Biomaterial–mesenchymal stem cell constructs for immunomodulation in composite tissue engineering. Tissue Eng. Part A 2014, 20, 2162–2168. [Google Scholar] [CrossRef] [PubMed]
- Yatomi, Y.; Igarashi, Y.; Yang, L.; Hisano, N.; Qi, R.; Asazuma, N.; Satoh, K.; Ozaki, Y.; Kume, S. Sphingosine 1-phosphate, a bioactive sphingolipid abundantly stored in platelets, is a normal constituent of human plasma and serum. J. Biochem. 1997, 121, 969–973. [Google Scholar] [CrossRef] [PubMed]
- Panetti, T.S. Differential effects of sphingosine 1-phosphate and lysophosphatidic acid on endothelial cells. Biochim. Biophys. Acta 2002, 1582, 190–196. [Google Scholar] [CrossRef]
- McVerry, B.J.; Garcia, J.G. Endothelial cell barrier regulation by sphingosine 1-phosphate. J. Cell. Biochem. 2004, 92, 1075–1085. [Google Scholar] [CrossRef]
- Siess, W. Athero- and thrombogenic actions of lysophosphatidic acid and sphingosine-1-phosphate. Biochim. Biophys. Acta 2002, 1582, 204–215. [Google Scholar] [CrossRef]
- Ushiyama, A.; Kataoka, H.; Iijima, T. Glycocalyx and its involvement in clinical pathophysiologies. J. Intensive Care 2016, 4, 59. [Google Scholar] [CrossRef]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lin, C.-H.; Lu, J.-H.; Hsia, K.; Lee, H.; Yao, C.-L.; Ma, H. The Antithrombotic Function of Sphingosine-1-Phosphate on Human Adipose-Stem-Cell-Recellularized Tissue Engineered Vascular Graft In Vitro. Int. J. Mol. Sci. 2019, 20, 5218. https://doi.org/10.3390/ijms20205218
Lin C-H, Lu J-H, Hsia K, Lee H, Yao C-L, Ma H. The Antithrombotic Function of Sphingosine-1-Phosphate on Human Adipose-Stem-Cell-Recellularized Tissue Engineered Vascular Graft In Vitro. International Journal of Molecular Sciences. 2019; 20(20):5218. https://doi.org/10.3390/ijms20205218
Chicago/Turabian StyleLin, Chih-Hsun, Jen-Her Lu, Kai Hsia, Hsinyu Lee, Chao-Ling Yao, and Hsu Ma. 2019. "The Antithrombotic Function of Sphingosine-1-Phosphate on Human Adipose-Stem-Cell-Recellularized Tissue Engineered Vascular Graft In Vitro" International Journal of Molecular Sciences 20, no. 20: 5218. https://doi.org/10.3390/ijms20205218
APA StyleLin, C.-H., Lu, J.-H., Hsia, K., Lee, H., Yao, C.-L., & Ma, H. (2019). The Antithrombotic Function of Sphingosine-1-Phosphate on Human Adipose-Stem-Cell-Recellularized Tissue Engineered Vascular Graft In Vitro. International Journal of Molecular Sciences, 20(20), 5218. https://doi.org/10.3390/ijms20205218






