Enterovirus-Associated Hand-Foot and Mouth Disease and Neurological Complications in Japan and the Rest of the World
Abstract
:1. Introduction
2. Enterovirus A
3. Enterovirus Infection
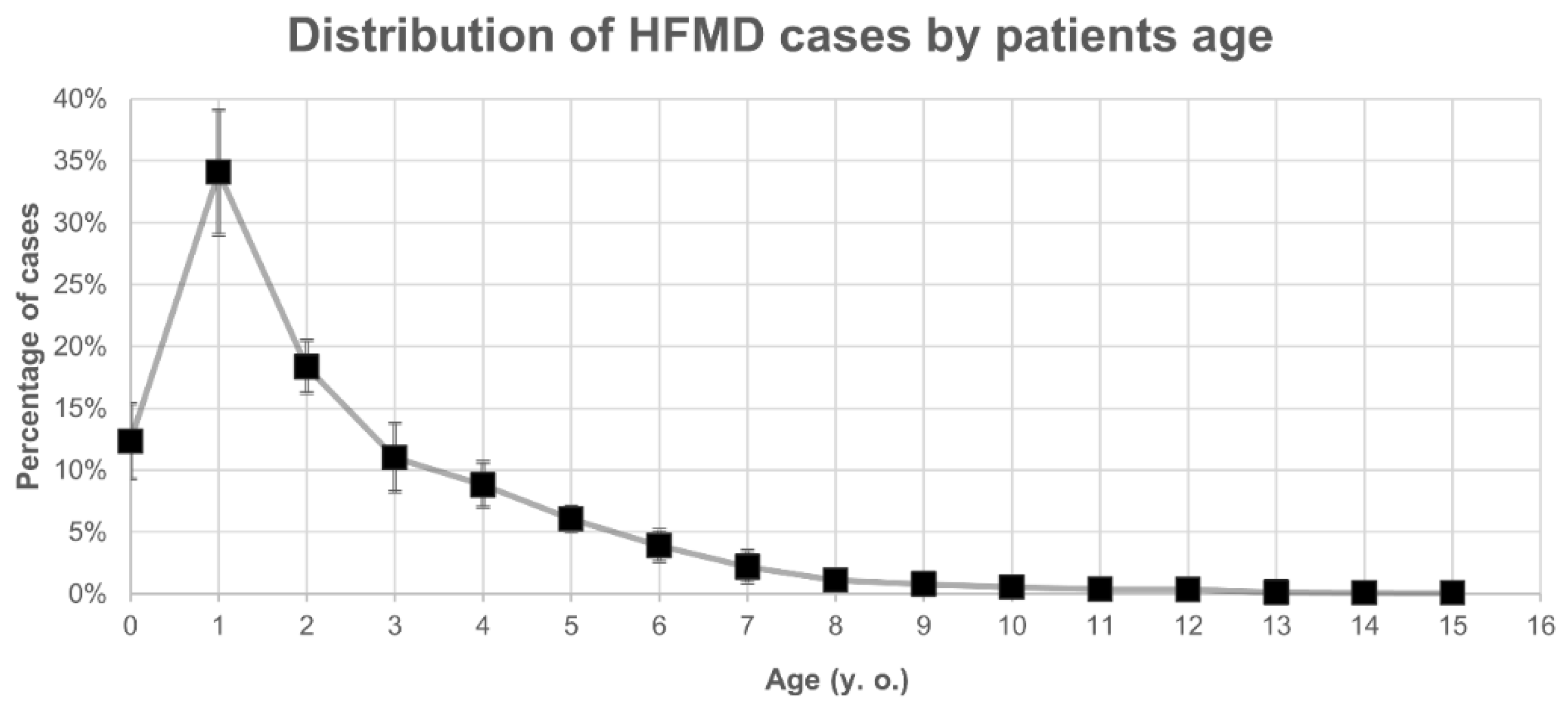
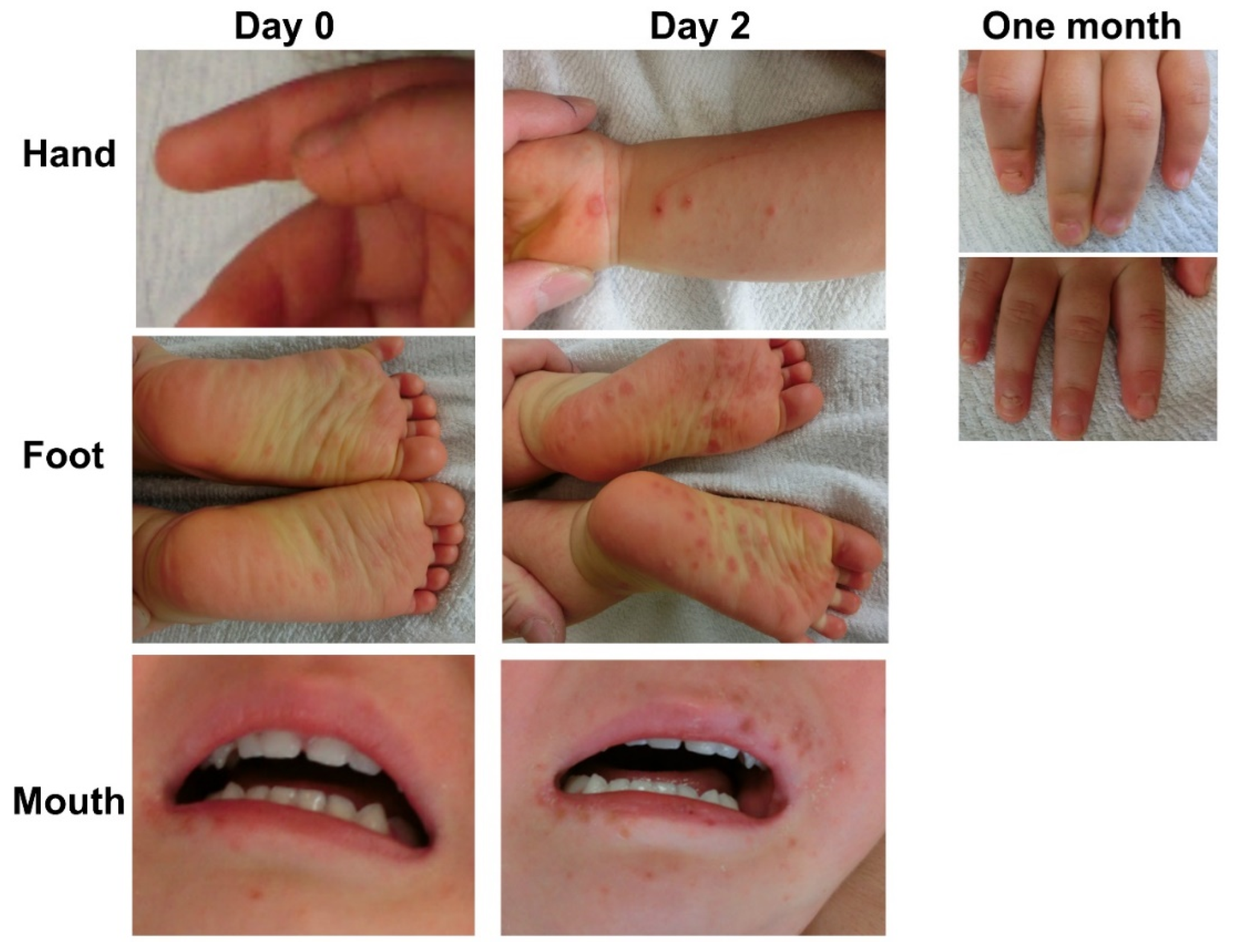
4. Hand, Foot, and Mouth Disease (HFMD)
5. EV Infections in the Central Nervous System
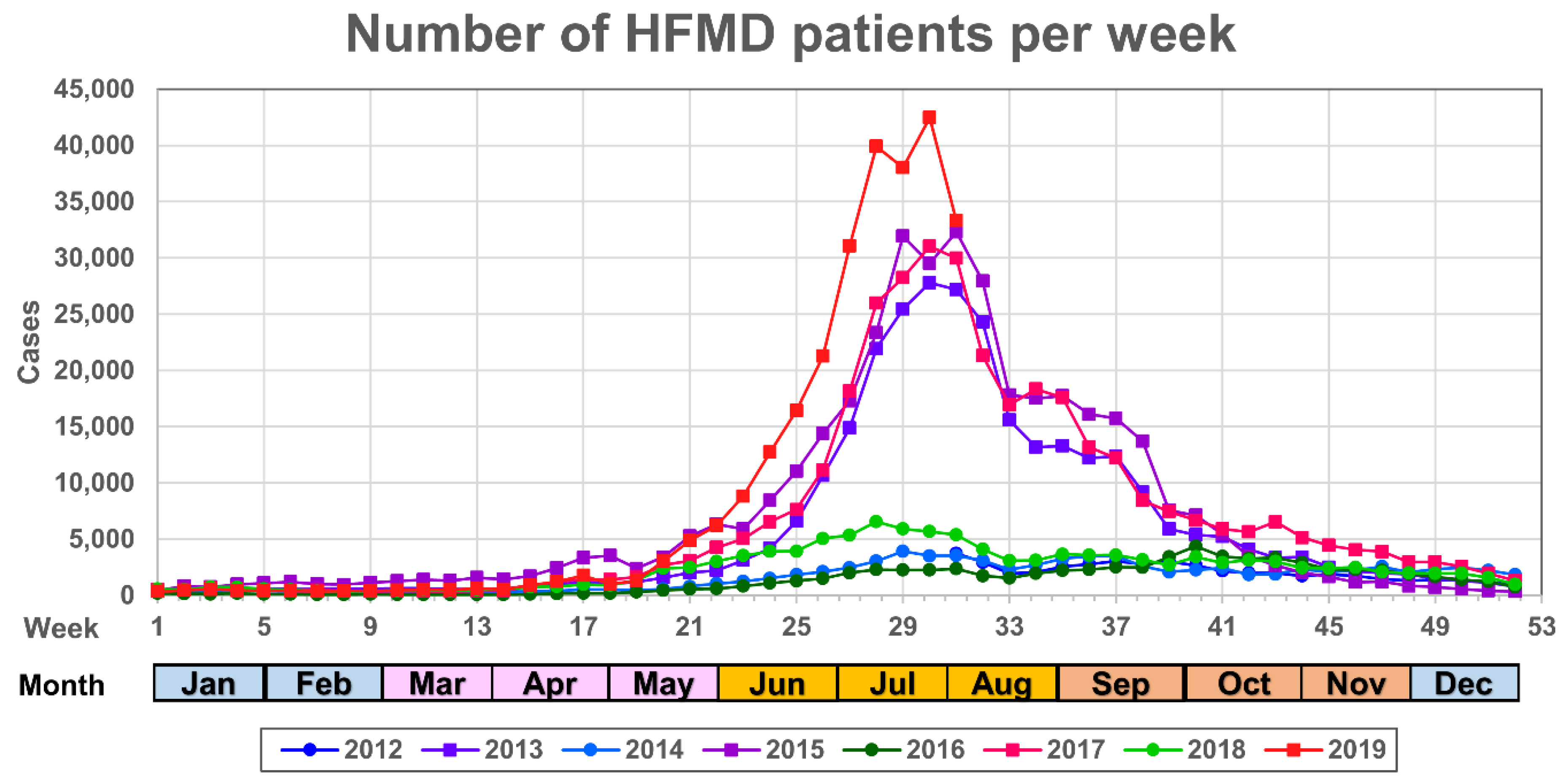
6. Surveillance of HFMD and AM in Japan
7. Enterovirus Detection and Isolation
8. Molecular Characterization of EV-A71
9. Treatment and Vaccines against HFMD
10. Risk Factors for Severity in HFMD Cases
11. Summary
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| EV | Enterovirus |
| CV | coxsackievirus |
| HFMD | hand-foot and mouth disease |
| AM | aseptic meningitis |
| CNS | Central Nervous System |
| NESID | National Epidemiological Surveillance of Infectious Diseases |
| HEV-A | Human enterovirus A |
| AFP | acute flaccid paralysis |
| CSF | cerebrospinal fluid |
| RdRp | RNA-dependent RNA polymerase |
References
- Pallansch, M.A.; Oberste, M.S.; Whitton, J.L. Enteroviruses: Polioviruses, coxsackieviruses, echoviruses, and newer enteroviruses. In Fields Virology, 6th ed.; Knipe, D.M., Howley, P.M., Eds.; Lippincott Williams & Wilkins: Philadelphia, PA, USA, 2007; Volume 1, pp. 490–530. [Google Scholar]
- WHO. W.H.O. a Guide to Clinical Management and Public Health Response for Hand, Foot and Mouth Disease (HFMD); WHO Regional Office for the Western Pacific: Manila, Philippines, 2011. [Google Scholar]
- Broccolo, F.; Drago, F.; Ciccarese, G.; Genoni, A.; Puggioni, A.; Rosa, G.M.; Parodi, A.; Manukyan, H.; Laassri, M.; Chumakov, K.; et al. Severe atypical hand-foot-and-mouth disease in adults due to coxsackievirus A6: Clinical presentation and phylogenesis of CV-A6 strains. J. Clin. Virol. 2019, 110, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Huang, J.; Liao, Q.; Ooi, M.H.; Cowling, B.J.; Chang, Z.; Wu, P.; Liu, F.; Li, Y.; Luo, L.; Yu, S.; et al. Epidemiology of Recurrent Hand, Foot and Mouth Disease, China, 2008–2015. Emerg. Infect. Dis. 2018, 24. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Hu, T.; Sun, D.; Ding, S.; Carr, M.J.; Xing, W.; Li, S.; Wang, X.; Shi, W. Epidemiological characteristics of hand, foot, and mouth disease in Shandong, China, 2009–2016. Sci. Rep. 2017, 7, 8900. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.Y.; Lee, Y.-J.; Kim, T.H.; Cheon, D.-S.; Nam, S.-O. Clinico-radiological spectrum in enterovirus 71 infection involving the central nervous system in children. J. Clin. Neurosci. 2014, 21, 416–420. [Google Scholar] [CrossRef] [PubMed]
- Infectious Disease Surveillance Center. IASR Summer Diseases (Herpangina/Hand-Foot-and-Mouth Disease). Available online: http://www.niid.go.jp/niid/ja/iasr/510-surveillance/iasr/graphs/4892-iasrgnatus.html (accessed on 19 October 2019).
- Fujimoto, T.; Chikahira, M.; Yoshida, S.; Ebira, H.; Hasegawa, A.; Totsuka, A.; Nishio, O. Outbreak of central nervous system disease associated with hand, foot, and mouth disease in Japan during the summer of 2000: Detection and molecular epidemiology of enterovirus 71. Microbiol. Immunol. 2002, 46, 621–627. [Google Scholar] [CrossRef]
- Sun, B.J.; Chen, H.J.; Chen, Y.; An, X.D.; Zhou, B.S. The Risk Factors of Acquiring Severe Hand, Foot, and Mouth Disease: A Meta-Analysis. Can. J. Infect. Dis. Med Microbiol. 2018, 2018, 1–12. [Google Scholar] [CrossRef] [Green Version]
- Zell, R.; Delwart, E.; Gorbalenya, A.E.; Hovi, T.; King, A.M.Q.; Knowles, N.J.; Lindberg, A.M.; Pallansch, M.A.; Palmenberg, A.C.; Reuter, G.; et al. ICTV Virus Taxonomy Profile: Picornaviridae. J. Gen. Virol. 2017, 98, 2421–2422. [Google Scholar] [CrossRef]
- Solomon, T.; Lewthwaite, P.; Perera, D.; Cardosa, M.J.; McMINN, P.; Ooi, M.H. Virology, epidemiology, pathogenesis, and control of enterovirus 71. Lancet Infect. Dis. 2010, 10, 778–790. [Google Scholar] [CrossRef]
- Wang, H.; Li, Y. Recent Progress on Functional Genomics Research of Enterovirus 71. Virol. Sin. 2019, 34, 9–21. [Google Scholar] [CrossRef]
- Young, N.A. Polioviruses, Coxsackie-Viruses, and Echoviruses-Comparison of Genomes by Rna Hybridization. J. Virol. 1973, 11, 832–839. [Google Scholar]
- Oberste, M.S.; Peñaranda, S.; Maher, K.; Pallansch, M.A. Complete genome sequences of all members of the species Human enterovirus A. J. Gen. Virol. 2004, 85, 1597–1607. [Google Scholar] [CrossRef] [PubMed]
- Guerra, J.A.; Waters, A.; Kelly, A.; Morley, U.; O’Reilly, P.; O’Kelly, E.; Dean, J.; Cunney, R.; O’Lorcain, P.; Cotter, S.; et al. Seroepidemiological and phylogenetic characterization of neurotropic enteroviruses in Ireland, 2005–2014. J. Med Virol. 2017, 89, 1550–1558. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Ren, Q.; Zhang, Z.; Zhang, L.; Carr, M.J.; Li, J.; Zhou, H.; Shi, W. Rapid detection of hand, foot and mouth disease enterovirus genotypes by multiplex PCR. J. Virol. Methods 2018, 258, 7–12. [Google Scholar] [CrossRef] [PubMed]
- Oberste, M.S.; Maher, K.; Kilpatrick, D.R.; Flemister, M.R.; Brown, B.A.; Pallansch, M.A. Typing of Human Enteroviruses by Partial Sequencing of VP1. J. Clin. Microbiol. 1999, 37, 1288–1293. [Google Scholar] [Green Version]
- International Committee on Taxonomy of Viruses. Genus: Enterovirus. Available online: https://talk.ictvonline.org/ictv-reports/ictv_online_report/positive-sense-rna-viruses/picornavirales/w/picornaviridae/681/genus-enterovirus (accessed on 19 October 2019).
- Ooi, M.H.; Wong, S.C.; Lewthwaite, P.; Cardosa, M.J.; Solomon, T. Clinical features, diagnosis, and management of enterovirus 71. Lancet Neurol. 2010, 9, 1097–1105. [Google Scholar] [CrossRef]
- Hatayama, K.; Goto, S.; Yashiro, M.; Mori, H.; Fujimoto, T.; Hanaoka, N.; Tanaka-Taya, K.; Zuzan, T.; Inoue, M. Acute flaccid myelitis associated with enterovirus D68 in a non-epidemic setting. IDCases 2019, 17, e00549. [Google Scholar] [CrossRef]
- Funakoshi, Y.; Ito, K.; Morino, S.; Kinoshita, K.; Morikawa, Y.; Kono, T.; Doan, Y.H.; Shimizu, H.; Hanaoka, N.; Konagaya, M.; et al. Enterovirus D68 respiratory infection in a children’s hospital in Japan in 2015. Pediatr. Int. 2019. [Google Scholar] [CrossRef]
- Poelman, R.; Schuffenecker, I.; Van Leer-Buter, C.; Josset, L.; Niesters, H.G.; Lina, B. European surveillance for enterovirus D68 during the emerging North-American outbreak in 2014. J. Clin. Virol. 2015, 71, 1–9. [Google Scholar] [CrossRef] [Green Version]
- Tan, Y.; Hassan, F.; Schuster, J.E.; Simenauer, A.; Selvarangan, R.; Halpin, R.A.; Lin, X.; Fedorova, N.; Stockwell, T.B.; Lam, T.T.-Y.; et al. Molecular Evolution and Intraclade Recombination of Enterovirus D68 during the 2014 Outbreak in the United States. J. Virol. 2016, 90, 1997–2007. [Google Scholar] [CrossRef] [Green Version]
- Schmidt, N.J.; Lennette, E.H.; Ho, H.H. An Apparently New Enterovirus Isolated from Patients with Disease of the Central Nervous System. J. Infect. Dis. 1974, 129, 304–309. [Google Scholar] [CrossRef]
- Wang, Y.-F.; Chou, C.-T.; Lei, H.-Y.; Liu, C.-C.; Wang, S.-M.; Yan, J.-J.; Su, I.-J.; Wang, J.-R.; Yeh, T.-M.; Chen, S.-H.; et al. A Mouse-Adapted Enterovirus 71 Strain Causes Neurological Disease in Mice after Oral Infection. J. Virol. 2004, 78, 7916–7924. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chang, L.-Y.; Tsao, K.-C.; Hsia, S.-H.; Shih, S.-R.; Huang, C.-G.; Chan, W.-K.; Hsu, K.-H.; Fang, T.-Y.; Huang, Y.-C.; Lin, T.-Y. Transmission and Clinical Features of Enterovirus 71 Infections in Household Contacts in Taiwan. JAMA 2004, 291, 222–227. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Muehlenbachs, A.; Bhatnagar, J.; Zaki, S.R. Tissue tropism, pathology and pathogenesis of enterovirus infection. J. Pathol. 2015, 235, 217–228. [Google Scholar] [CrossRef] [PubMed]
- Tang, J.W.; Nicolle, A.D.; Klettner, C.A.; Pantelic, J.; Wang, L.; Bin Suhaimi, A.; Tan, A.Y.L.; Ong, G.W.X.; Su, R.; Sekhar, C.; et al. Airflow Dynamics of Human Jets: Sneezing and Breathing-Potential Sources of Infectious Aerosols. PLoS ONE 2013, 8, e59970. [Google Scholar] [CrossRef] [PubMed]
- Yin-Murphy, M.; Almond, J.W. Chapter 53 Picornaviruses. Galveston (TX): Univ. of Texas Medical Branch at Galveston; 1996; pp. 1–18. Available online: https://www.ncbi.nlm.nih.gov/books/NBK7687/ (accessed on 19 October 2019).
- Li, X.W.; Ni, X.; Qian, S.Y.; Wang, Q.; Jiang, R.M.; Xu, W.B.; Zhang, Y.C.; Yu, G.J.; Chen, Q.; Shang, Y.X.; et al. Chinese guidelines for the diagnosis and treatment of hand, foot and mouth disease (2018 edition). World J. Pediatr. 2018, 14, 437–447. [Google Scholar] [CrossRef]
- Cox, J.A.; Hiscox, J.A.; Solomon, T.; Ooi, M.-H.; Ng, L.F.P. Immunopathogenesis and Virus–Host Interactions of Enterovirus 71 in Patients with Hand, Foot and Mouth Disease. Front. Microbiol. 2017, 8, 2249. [Google Scholar] [CrossRef]
- Fujimoto, T.; Yoshida, S.; Munemura, T.; Taniguchi, K.; Shinohara, M.; Nishio, O.; Chikahira, M.; Okabe, N. Detection and quantification of enterovirus 71 genome from cerebrospinal fluid of an encephalitis patient by PCR applications. Jpn. J. Infect. Dis. 2008, 61, 497–499. [Google Scholar]
- Chong, P.F.; Kira, R.; Mori, H.; Okumura, A.; Torisu, H.; Yasumoto, S.; Shimizu, H.; Fujimoto, T.; Hanaoka, N.; Kusunoki, S.; et al. Clinical Features of Acute Flaccid Myelitis Temporally Associated With an Enterovirus D68 Outbreak: Results of a Nationwide Survey of Acute Flaccid Paralysis in Japan, August-December 2015. Clin. Infect. Dis. 2018, 66, 653–664. [Google Scholar] [CrossRef]
- Shah, V.A.; Chong, C.Y.; Chan, K.P.; Ng, W.; Ling, A.E. Clinical characteristics of an outbreak of hand, foot and mouth disease in Singapore. Ann. Acad. Med. Singap. 2003, 32, 381–387. [Google Scholar]
- Pons-Salort, M.; Grassly, N.C. Serotype-specific immunity explains the incidence of diseases caused by human enteroviruses. Science 2018, 361, 800–803. [Google Scholar] [CrossRef] [Green Version]
- Kobayashi, M.; Makino, T.; Hanaoka, N.; Shimizu, H.; Enomoto, M.; Okabe, N.; Kanou, K.; Konagaya, M.; Oishi, K.; Fujimoto, T. Clinical manifestations of coxsackievirus A6 infection associated with a major outbreak of hand, foot, and mouth disease in Japan. Jpn. J. Infect. Dis. 2013, 66, 260–261. [Google Scholar] [CrossRef]
- Martinez, M.; Shukla, H.; Nikulin, J.; Wadood, M.Z.; Hadler, S.; Mbaeyi, C.; Tangermann, R.; Jorba, J.; Ehrhardt, D. Progress Toward Poliomyelitis Eradication-Afghanistan, January 2016–June 2017. MMWR. Morb. Mortal. Wkly. Rep. 2017, 66, 854–858. [Google Scholar] [CrossRef]
- Nathanson, N.; Kew, O.M. From emergence to eradication: The epidemiology of poliomyelitis deconstructed. Am. J. Epidemiol. 2010, 172, 1213–1229. [Google Scholar] [CrossRef]
- Xing, W.; Liao, Q.; Viboud, C.; Zhang, J.; Sun, J.; Wu, J.T.; Chang, Z.; Liu, F.; Fang, V.J.; Zheng, Y.; et al. Hand, foot, and mouth disease in China, 2008–2012: An epidemiological study. Lancet Infect. Dis. 2014, 14, 308–318. [Google Scholar] [CrossRef]
- Casas-Alba, D.; de Sevilla, M.F.; Valero-Rello, A.; Fortuny, C.; Garcia-Garcia, J.J.; Ortez, C.; Muchart, J.; Armangue, T.; Jordan, I.; Luaces, C.; et al. Outbreak of brainstem encephalitis associated with enterovirus-A71 in Catalonia, Spain (2016): A clinical observational study in a children’s reference centre in Catalonia. Clin. Microbiol. Infect. 2017, 23, 874–881. [Google Scholar] [CrossRef]
- Chen, C.-S.; Yao, Y.-C.; Lin, S.-C.; Lee, Y.-P.; Wang, Y.-F.; Wang, J.-R.; Liu, C.-C.; Lei, H.-Y.; Yu, C.-K. Retrograde Axonal Transport: A Major Transmission Route of Enterovirus 71 in Mice. J. Virol. 2007, 81, 8996–9003. [Google Scholar] [CrossRef]
- Knoester, M.; Helfferich, J.; Poelman, R.; Van Leer-Buter, C.; Brouwer, O.F.; Niesters, H.G. Twenty-nine Cases of Enterovirus-D68-associated Acute Flaccid Myelitis in Europe 2016: A Case Series and Epidemiologic Overview. Pediatr. Infect. Dis. J. 2019, 38, 16–21. [Google Scholar] [CrossRef]
- Bitnun, A.; Yeh, E.A. Acute Flaccid Paralysis and Enteroviral Infections. Curr. Infect. Dis. Rep. 2018, 20, 34. [Google Scholar] [CrossRef]
- Takahashi, S.; Metcalf, C.J.E.; Arima, Y.; Fujimoto, T.; Shimizu, H.; Van Doorn, H.R.; Le Van, T.; Chan, Y.-F.; Farrar, J.J.; Oishi, K.; et al. Epidemic dynamics, interactions and predictability of enteroviruses associated with hand, foot and mouth disease in Japan. J. R. Soc. Interface 2018, 15, 20180507. [Google Scholar] [CrossRef] [Green Version]
- Rao, D.C.; Naidu, J.R.; Maiya, P.P.; Babu, A.; Bailly, J.-L. Large-scale HFMD epidemics caused by Coxsackievirus A16 in Bangalore, India during 2013 and 2015. Infect. Genet. Evol. 2017, 55, 228–235. [Google Scholar] [CrossRef]
- Dong, W.; Li, X.; Yang, P.; Liao, H.; Wang, X.; Wang, Q. The Effects of Weather Factors on Hand, Foot and Mouth Disease in Beijing. Sci. Rep. 2016, 6, 19247. [Google Scholar] [CrossRef]
- Second, J.; Velter, C.; Calès, S.; Truchetet, F.; Lipsker, D.; Cribier, B. Clinicopathologic analysis of atypical hand, foot, and mouth disease in adult patients. J. Am. Acad. Dermatol. 2017, 76, 722–729. [Google Scholar] [CrossRef]
- Ramirez-Fort, M.K.; Downing, C.; Doan, H.Q.; Benoist, F.; Oberste, M.S.; Khan, F.; Tyring, S.K. Coxsackievirus A6 associated hand, foot and mouth disease in adults: Clinical presentation and review of the literature. J. Clin. Virol. 2014, 60, 381–386. [Google Scholar] [CrossRef]
- Robinson, C.R.; Doane, F.W.; Rhodes, A.J. Report of an outbreak of febrile illness with pharyngeal lesions and exanthem: Toronto, summer 1957—Isolation of group A Coxsackie virus. Can. Med. Assoc. J. 1958, 79, 615–621. [Google Scholar]
- Van der Sanden, S.; Koopmans, M.; Uslu, G.; van der Avoort, H.; Dutch Working Group for Clinical, V. Epidemiology of enterovirus 71 in the Netherlands, 1963 to 2008. J. Clin. Microbiol. 2009, 47, 2826–2833. [Google Scholar] [CrossRef]
- Nakamura, K.; Watanabe, K.; Kitayama, T.; Drano, J.; Fukumi, H.; Nishikawa, F. Febrile disease with stomatic enanthem caused by Coxsackievirus group A. Shonika-Shinryo 1965, 28, 1004–1012. [Google Scholar]
- Tagaya, I.; Moritsugu, Y. EPIDEMIC OF HAND, FOOT AND MOUTH DISEASE IN JAPAN. Jpn. J. Med. Sci. Boil. 1973, 26, 143–147. [Google Scholar] [CrossRef] [Green Version]
- Pallansch, M.A.; Oberste, M.S. Enterovirus 71 encephalitis: A new vaccine on the horizon? Lancet 2013, 381, 976–977. [Google Scholar] [CrossRef]
- Ishimaru, Y.; Nakano, S.; Yamaoka, K.; Takami, S. Outbreaks of hand, foot, and mouth disease by enterovirus 71. High incidence of complication disorders of central nervous system. Arch. Dis. Child. 1980, 55, 583–588. [Google Scholar] [CrossRef]
- Nagy, G.; Takátsy, S.; Kukán, E.; Mihály, I.; Dömök, I. Virological diagnosis of enterovirus type 71 infections: Experiences gained during an epidemic of acute CNS diseases in Hungary in 1978. Arch. Virol. 1982, 71, 217–227. [Google Scholar] [CrossRef]
- Chu, P.-Y.; Lin, K.-H.; Hwang, K.-P.; Chou, L.-C.; Wang, C.-F.; Shih, S.-R.; Wang, J.-R.; Shimada, Y.; Ishiko, H. Molecular epidemiology of enterovirus 71 in Taiwan. Arch. Virol. 2001, 146, 589–600. [Google Scholar] [CrossRef]
- Sabanathan, S.; Thwaites, L.; Wills, B.; Qui, P.T.; van Doorn, H.R. Enterovirus 71 related severe hand, foot and mouth disease outbreaks in South-East Asia: Current situation and ongoing challenges. J. Epidemiol. Community Health 2014, 68, 500–502. [Google Scholar] [CrossRef]
- Chakraborty, R.; Iturriza-Gómara, M.; Musoke, R.; Palakudy, T.; D’Agostino, A.; Gray, J. An epidemic of enterovirus 71 infection among HIV-1-infected orphans in Nairobi. AIDS 2004, 18, 1968–1970. [Google Scholar] [CrossRef]
- Österback, R.; Vuorinen, T.; Linna, M.; Susi, P.; Hyypiä, T.; Waris, M. Coxsackievirus A6 and Hand, Foot, and Mouth Disease, Finland. Emerg. Infect. Dis. 2009, 15, 1485–1488. [Google Scholar] [CrossRef]
- Fujimoto, T.; Iizuka, S.; Enomoto, M.; Abe, K.; Yamashita, K.; Hanaoka, N.; Okabe, N.; Yoshida, H.; Yasui, Y.; Kobayashi, M.; et al. Hand, foot, and mouth disease caused by coxsackievirus A6, Japan, 2011. Emerg. Infect. Dis. 2012, 18, 337–339. [Google Scholar] [CrossRef]
- Bian, L.; Wang, Y.; Yao, X.; Mao, Q.; Xu, M.; Liang, Z. Coxsackievirus A6: A new emerging pathogen causing hand, foot and mouth disease outbreaks worldwide. Expert Rev. Anti-Infect. Ther. 2015, 13, 1061–1071. [Google Scholar] [CrossRef]
- Yang, B.; Liu, F.; Liao, Q.; Wu, P.; Chang, Z.; Huang, J.; Long, L.; Luo, L.; Li, Y.; Leung, G.M.; et al. Epidemiology of hand, foot and mouth disease in China, 2008 to 2015 prior to the introduction of EV-A71 vaccine. Eurosurveillance 2017, 22, 50. [Google Scholar] [CrossRef]
- Mao, Q.-Y.; Wang, Y.; Bian, L.; Xu, M.; Liang, Z. EV71 vaccine, a new tool to control outbreaks of hand, foot and mouth disease (HFMD). Expert Rev. Vaccines 2016, 15, 599–606. [Google Scholar] [CrossRef]
- Antona, D.; Kossorotoff, M.; Schuffenecker, I.; Mirand, A.; Leruez-Ville, M.; Bassi, C.; Aubart, M.; Moulin, F.; Levy-Bruhl, D.; Henquell, C.; et al. Severe paediatric conditions linked with EV-A71 and EV-D68, France, May to October 2016. Eurosurveillance 2016, 21, 30402. [Google Scholar] [CrossRef]
- Nhan, L.N.T.; Hong, N.T.T.; Nhu, L.N.T.; Nguyet, L.A.; Ny, N.T.H.; Thanh, T.T.; Han, D.D.K.; Van, H.M.T.; Thwaites, C.L.; Hien, T.T.; et al. Severe enterovirus A71 associated hand, foot and mouth disease, Vietnam, 2018: Preliminary report of an impending outbreak. Eurosurveillance 2018, 23, 1800590. [Google Scholar] [CrossRef]
- Wang, Y.-F.; Yu, C.-K. Animal models of enterovirus 71 infection: Applications and limitations. J. Biomed. Sci. 2014, 21, 31. [Google Scholar] [CrossRef]
- Chia, M.-Y.; Chung, W.-Y.; Chiang, P.-S.; Chien, Y.-S.; Ho, M.-S.; Lee, M.-S. Monitoring Antigenic Variations of Enterovirus 71: Implications for Virus Surveillance and Vaccine Development. PLoS Negl. Trop. Dis. 2014, 8, e3044. [Google Scholar] [CrossRef]
- Bessaud, M.; Joffret, M.-L.; Blondel, B.; Delpeyroux, F. Exchanges of genomic domains between poliovirus and other cocirculating species C enteroviruses reveal a high degree of plasticity. Sci. Rep. 2016, 6, 38831. [Google Scholar] [CrossRef]
- Gonzalez, G.; Yawata, N.; Aoki, K.; Kitaichi, N. Challenges in management of epidemic keratoconjunctivitis with emerging recombinant human adenoviruses. J. Clin. Virol. 2019, 112, 1–9. [Google Scholar] [CrossRef]
- Andrés, C.; Piñana, M.; Vila, J.; Esperalba, J.; Trejo-Zahínos, J.; Codina, M.G.; Martín, M.C.; Fuentes, F.; Rubio, S.; Pumarola, T.; et al. The high genetic similarity between rhinoviruses and enteroviruses remains as a pitfall for molecular diagnostic tools: A three-year overview. Infect. Genet. Evolut. 2019, 75, 103996. [Google Scholar] [CrossRef]
- Leber, A.L.; Everhart, K.; Balada-Llasat, J.M.; Cullison, J.; Daly, J.; Holt, S.; Lephart, P.; Salimnia, H.; Schreckenberger, P.C.; DesJarlais, S.; et al. Multicenter Evaluation of BioFire FilmArray Meningitis/Encephalitis Panel for Detection of Bacteria, Viruses, and Yeast in Cerebrospinal Fluid Specimens. J. Clin. Microbiol. 2016, 54, 2251–2261. [Google Scholar] [CrossRef] [Green Version]
- Huang, S.-W.; Tai, C.-H.; Fonville, J.M.; Lin, C.-H.; Wang, S.-M.; Liu, C.-C.; Su, I.-J.; Smith, D.J.; Wang, J.-R. Mapping Enterovirus A71 Antigenic Determinants from Viral Evolution. J. Virol. 2015, 89, 11500–11506. [Google Scholar] [CrossRef] [Green Version]
- Bessaud, M.; Razafindratsimandresy, R.; Nougairède, A.; Joffret, M.-L.; Deshpande, J.M.; Dubot-Pérès, A.; Héraud, J.-M.; De Lamballerie, X.; Delpeyroux, F.; Bailly, J.-L. Molecular Comparison and Evolutionary Analyses of VP1 Nucleotide Sequences of New African Human Enterovirus 71 Isolates Reveal a Wide Genetic Diversity. PLoS ONE 2014, 9, e90624. [Google Scholar] [CrossRef]
- Tee, K.K.; Lam, T.T.-Y.; Chan, Y.F.; Bible, J.M.; Kamarulzaman, A.; Tong, C.Y.W.; Takebe, Y.; Pybus, O.G. Evolutionary Genetics of Human Enterovirus 71: Origin, Population Dynamics, Natural Selection, and Seasonal Periodicity of the VP1 Gene. J. Virol. 2010, 84, 3339–3350. [Google Scholar] [CrossRef] [Green Version]
- Yee, P.T.I.; Poh, C.L. Impact of genetic changes, pathogenicity and antigenicity on Enterovirus- A71 vaccine development. Virology 2017, 506, 121–129. [Google Scholar] [CrossRef] [Green Version]
- Mandary, M.B.; Poh, C.L. Changes in the EV-A71 Genome through Recombination and Spontaneous Mutations: Impact on Virulence. Viruses 2018, 10, 320. [Google Scholar] [CrossRef] [PubMed]
- Nikolaidis, M.; Mimouli, K.; Kyriakopoulou, Z.; Tsimpidis, M.; Tsakogiannis, D.; Markoulatos, P.; Amoutzias, G.D. Large-scale genomic analysis reveals recurrent patterns of intertypic recombination in human enteroviruses. Virology 2019, 526, 72–80. [Google Scholar] [CrossRef] [PubMed]
- Ronquist, F.; Teslenko, M.; Van Der Mark, P.; Ayres, D.L.; Darling, A.; Höhna, S.; Larget, B.; Liu, L.; Suchard, M.A.; Huelsenbeck, J.P. MrBayes 3.2: Efficient Bayesian phylogenetic inference and model choice across a large model space. Syst. Boil. 2012, 61, 539–542. [Google Scholar] [CrossRef]
- Zhu, F.; Xu, W.; Xia, J.; Liang, Z.; Liu, Y.; Zhang, X.; Tan, X.; Wang, L.; Mao, Q.; Wu, J.; et al. Efficacy, safety, and immunogenicity of an enterovirus 71 vaccine in China. N. Engl. J. Med. 2014, 370, 818–828. [Google Scholar] [CrossRef]
- Pourianfar, H.R.; Grollo, L. Development of antiviral agents toward enterovirus 71 infection. J. Microbiol. Immunol. Infect. 2015, 48, 1–8. [Google Scholar] [CrossRef] [Green Version]
- Zhang, D.; Li, R.; Zhang, W.; Li, G.; Ma, Z.; Chen, X.; Du, Z.; Li, Z.; Guo, P.; Lin, Z.; et al. A Case-control Study on Risk Factors for Severe Hand, Foot and Mouth Disease. Sci. Rep. 2017, 7, 40282. [Google Scholar] [CrossRef]
- Esposito, S.; Picciolli, I.; Semino, M.; Principi, N. Steroids and Childhood Encephalitis. Pediatr. Infect. Dis. J. 2012, 31, 759–760. [Google Scholar] [CrossRef] [Green Version]
- Li, Z.; Li, C.; Ling, P.; Shen, F.; Chen, S.; Liu, C.; Yu, C.; Chen, S. Ribavirin Reduces Mortality in Enterovirus 71–Infected Mice by Decreasing Viral Replication. J. Infect. Dis. 2008, 197, 854–857. [Google Scholar] [CrossRef]
- Zhang, H.-P.; Wang, L.; Qian, J.-H.; Cai, K.; Chen, Y.-H.; Zhang, Q.-L.; Yu, H.-J. Efficacy and safety of ribavirin aerosol in children with hand-foot-mouth disease. Zhongguo Dang Dai Er Ke Za Zhi 2014, 16, 272–276. [Google Scholar]
- Ooi, M.H.; Wong, S.C.; Mohan, A.; Podin, Y.; Perera, D.; Clear, D.; del Sel, S.; Chieng, C.H.; Tio, P.H.; Cardosa, M.J.; et al. Identification and validation of clinical predictors for the risk of neurological involvement in children with hand, foot, and mouth disease in Sarawak. BMC Infect. Dis. 2009, 9, 3. [Google Scholar] [CrossRef]
- Aswathyraj, S.; Arunkumar, G.; Alidjinou, E.K.; Hober, D. Hand, foot and mouth disease (HFMD): Emerging epidemiology and the need for a vaccine strategy. Med Microbiol. Immunol. 2016, 205, 397–407. [Google Scholar] [CrossRef] [PubMed]
- Chong, C.Y.; Chan, K.P.; Shah, V.A.; Ng, W.Y.M.; Lau, G.; Teo, T.E.S.; Lai, S.H.; Ling, A.E. Hand, foot and mouth disease in Singapore: A comparison of fatal and non-fatal cases. Acta Paediatr. 2003, 92, 1163–1169. [Google Scholar] [CrossRef] [PubMed]
- Lu, H.-K.; Lin, T.-Y.; Hsia, S.-H.; Chiu, C.-H.; Huang, Y.-C.; Tsao, K.-C.; Chang, L.-Y. Prognostic implications of myoclonic jerk in children with enterovirus infection. J. Microbiol. Immunol. Infect. 2004, 37, 82–87. [Google Scholar] [PubMed]
- Fang, Y.; Wang, S.; Zhang, L.; Guo, Z.; Huang, Z.; Tu, C.; Zhu, B.-P. Risk factors of severe hand, foot and mouth disease: A meta-analysis. Scand. J. Infect. Dis. 2014, 46, 515–522. [Google Scholar] [CrossRef] [PubMed]
- Ku, Z.; Liu, Q.; Ye, X.; Cai, Y.; Wang, X.; Shi, J.; Li, D.; Jin, X.; An, W.; Huang, Z. A virus-like particle based bivalent vaccine confers dual protection against enterovirus 71 and coxsackievirus A16 infections in mice. Vaccine 2014, 32, 4296–4303. [Google Scholar] [CrossRef]
- Zhang, Z.; Dong, Z.; Wang, Q.; Carr, M.J.; Li, J.; Liu, T.; Li, D.; Shi, W. Characterization of an inactivated whole-virus bivalent vaccine that induces balanced protective immunity against coxsackievirus A6 and A10 in mice. Vaccine 2018, 36, 7095–7104. [Google Scholar] [CrossRef]
- Zhang, Z.; Dong, Z.; Li, J.; Carr, M.J.; Zhuang, D.; Wang, J.; Zhang, Y.; Ding, S.; Tong, Y.; Li, D.; et al. Protective Efficacies of Formaldehyde-Inactivated Whole-Virus Vaccine and Antivirals in a Murine Model of Coxsackievirus A10 Infection. J. Virol. 2017, 91, e00333-17. [Google Scholar] [CrossRef] [Green Version]
- Zhang, Z.; Dong, Z.; Wei, Q.; Carr, M.J.; Li, J.; Ding, S.; Tong, Y.; Li, D.; Shi, W. A Neonatal Murine Model of Coxsackievirus A6 Infection for Evaluation of Antiviral and Vaccine Efficacy. J. Virol. 2017, 91, e02450-16. [Google Scholar] [CrossRef] [Green Version]

| Disease | Virus * | 2012 | 2013 | 2014 | 2015 | 2016 | 2017 | 2018 |
|---|---|---|---|---|---|---|---|---|
| HFMD | 46530 | 301071 | 83219 | 379368 | 68834 | 355686 | 122041 | |
| EV-A71 | 46% | 82% | 43% | 0% | 22% | 64% | 65% | |
| CV-A16 | 54% | 18% | 57% | 100% | 78% | 36% | 35% | |
| AM | 453 | 1058 | 854 | 1035 | 1332 | 928 | 760 | |
| Enterovirus A | 15% | 11% | 0% | 17% | 0% | 19% | 12% | |
| CV-4 | 1% | - | - | - | - | - | - | |
| CV-6 | - | 2% | - | - | - | 5% | - | |
| CV-9 | 12% | - | - | 17% | - | 4% | 4% | |
| EV-A71 | 2% | 9% | - | - | - | 10% | 7% | |
| Enterovirus B | 66% | 81% | 92% | 71% | 91% | 52% | 65% | |
| CV-B1 | - | 3% | - | - | 3% | - | - | |
| CV-B2 | - | - | 7% | 3% | 2% | 10% | 3% | |
| CV-B3 | - | 13% | 5% | 4% | 8% | - | - | |
| CV-B4 | 2% | 2% | 4% | - | - | 2% | 12% | |
| CV-B5 | 13% | 9% | 12% | 9% | 41% | - | 9% | |
| Echovirus 11 | - | - | 29% | - | - | - | 25% | |
| Echovirus 16 | - | - | - | 4% | - | - | - | |
| Echovirus 18 | 1% | 6% | 9% | 33% | 4% | - | 8% | |
| Echovirus 3 | - | - | 7% | 3% | 2% | 6% | - | |
| Echovirus 30 | - | 22% | 15% | 5% | 6% | - | - | |
| Echovirus 6 | 25% | 26% | 3% | - | 20% | 20% | 4% | |
| Echovirus 7 | 14% | - | - | - | - | 3% | 3% | |
| Echovirus 9 | 11% | - | - | 10% | 4% | 10% | - | |
| Mumps virus + | 19% | 7% | 8% | 12% | 9% | 30% | 23% |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gonzalez, G.; Carr, M.J.; Kobayashi, M.; Hanaoka, N.; Fujimoto, T. Enterovirus-Associated Hand-Foot and Mouth Disease and Neurological Complications in Japan and the Rest of the World. Int. J. Mol. Sci. 2019, 20, 5201. https://doi.org/10.3390/ijms20205201
Gonzalez G, Carr MJ, Kobayashi M, Hanaoka N, Fujimoto T. Enterovirus-Associated Hand-Foot and Mouth Disease and Neurological Complications in Japan and the Rest of the World. International Journal of Molecular Sciences. 2019; 20(20):5201. https://doi.org/10.3390/ijms20205201
Chicago/Turabian StyleGonzalez, Gabriel, Michael J. Carr, Masaaki Kobayashi, Nozomu Hanaoka, and Tsuguto Fujimoto. 2019. "Enterovirus-Associated Hand-Foot and Mouth Disease and Neurological Complications in Japan and the Rest of the World" International Journal of Molecular Sciences 20, no. 20: 5201. https://doi.org/10.3390/ijms20205201
APA StyleGonzalez, G., Carr, M. J., Kobayashi, M., Hanaoka, N., & Fujimoto, T. (2019). Enterovirus-Associated Hand-Foot and Mouth Disease and Neurological Complications in Japan and the Rest of the World. International Journal of Molecular Sciences, 20(20), 5201. https://doi.org/10.3390/ijms20205201







