Highly Selective Cleavage of TH2-Promoting Cytokines by the Human and the Mouse Mast Cell Tryptases, Indicating a Potent Negative Feedback Loop on TH2 Immunity
Abstract
:1. Introduction
2. Results
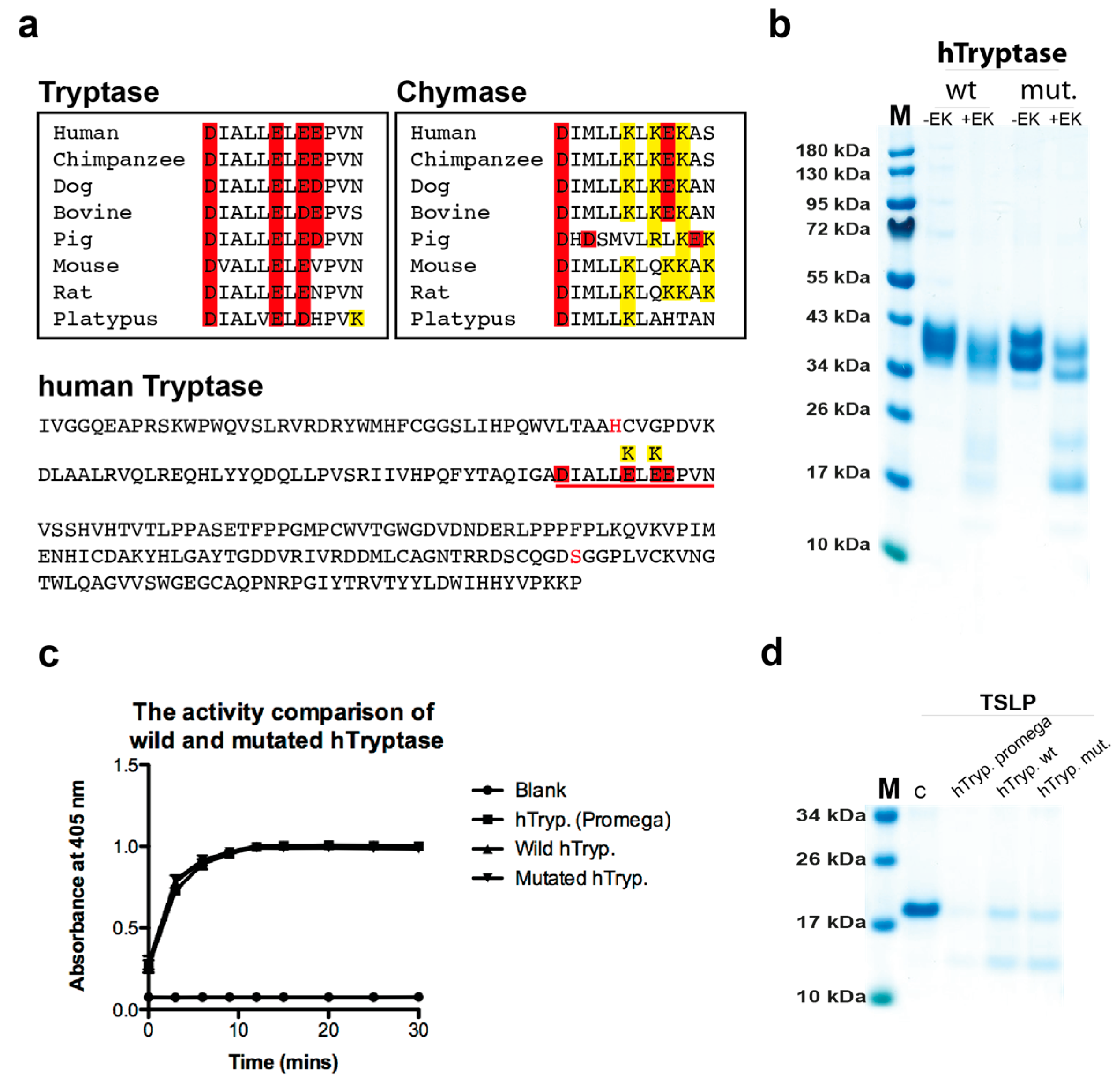
2.1. Analysis of the Purity and Activity of the Recombinant Human and Mouse Mast Cell Tryptases
2.2. Analysis of Cleavage Sensitivity against a Panel of 69 Cytokines and Chemokines by the Recombinant Human Tryptase
2.3. Analysis of Cleavage Sensitivity against a Panel of 56 Cytokines and Chemokines by the Recombinant Mouse Tryptase, mMCP-6
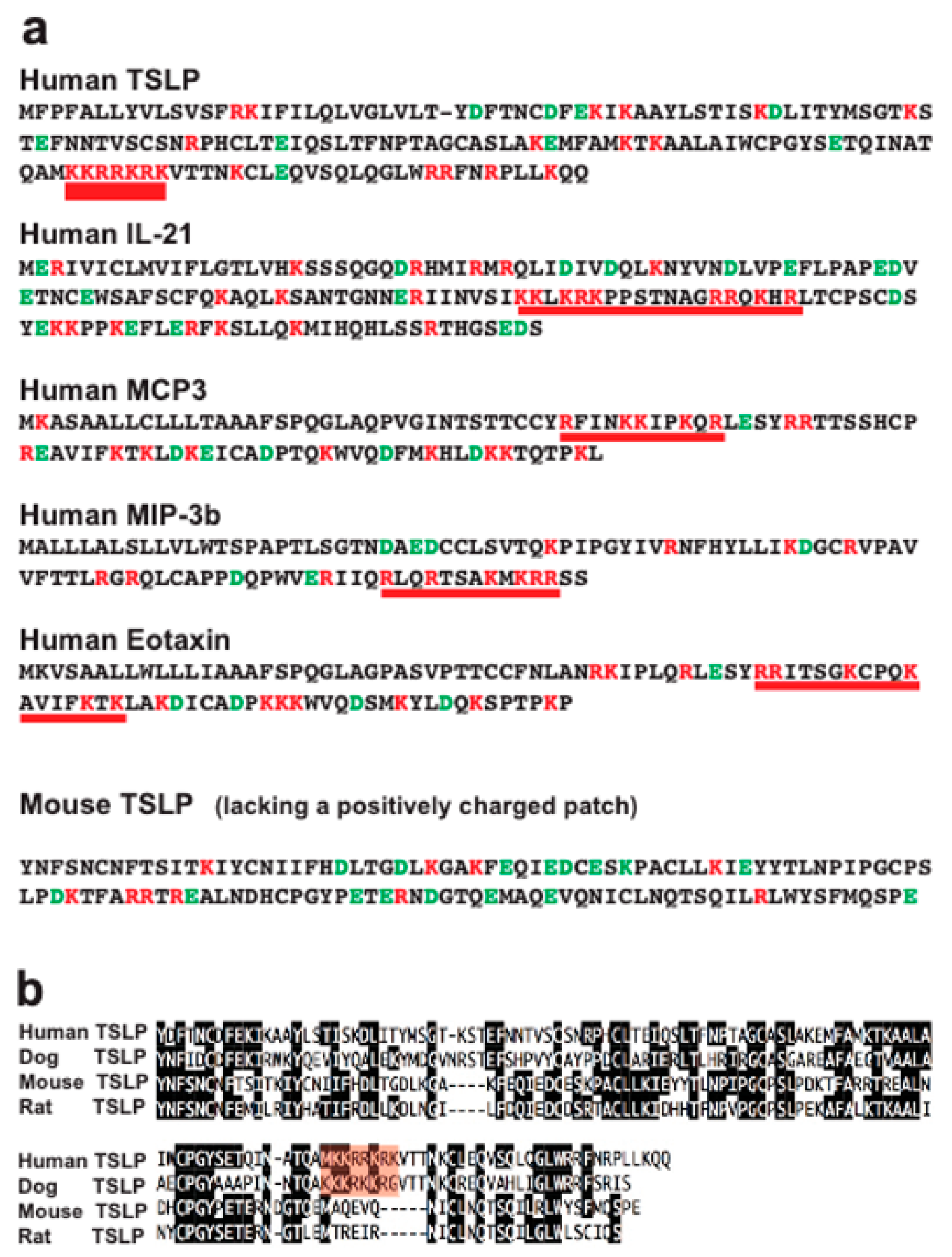
2.4. Analysis of Structural Similarities between the Three Efficiently Cleaved Cytokines and Chemokines
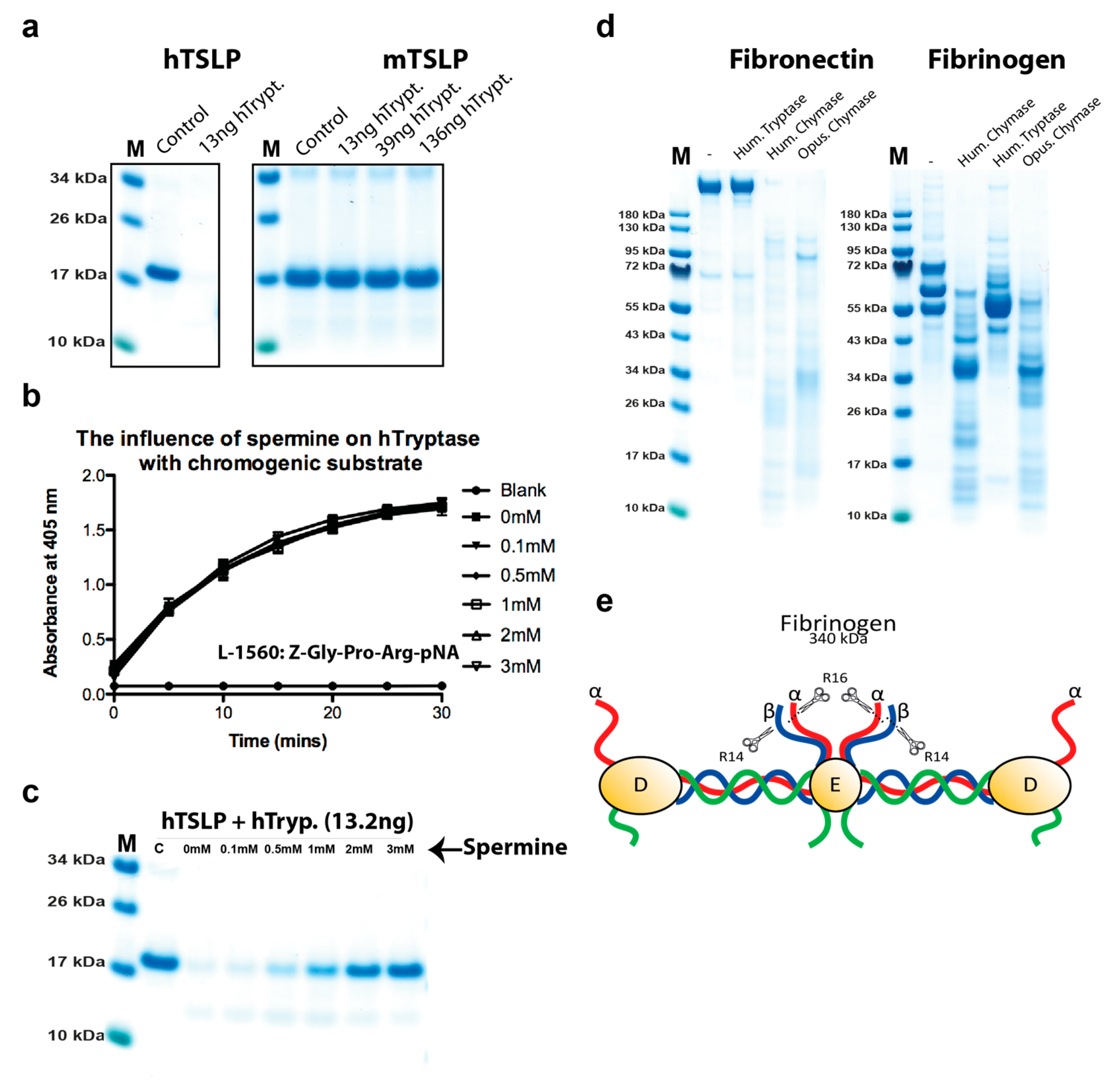
2.5. Analysis of the Effect of Spermine on Tryptase Activity
2.6. Cleavage of other Substrates by Human Tryptase
2.7. Cleavage Selectivity of Wild Type (WT) and Mutant Human Tryptase
3. Discussion
4. Materials and Methods
4.1. Enzymes and other Reagents
Recombinant Human and Mouse Cytokines and Chemokines
4.2. Analysis of the Sensitivity to Cleavage by the Recombinant Human and Mouse Tryptase
4.3. Analysis of the Cleavage of Human Fibrinogen and Fibronectin
Author Contributions
Funding
Conflicts of Interest
References
- Hallgren, J.; Pejler, G. Biology of mast cell tryptase. An inflammatory mediator. Febs. J. 2006, 273, 1871–1895. [Google Scholar] [CrossRef]
- Korkmaz, B.; Moreau, T.; Gauthier, F. Neutrophil elastase proteinase 3, cathepsin G: Physicochemical properties activity physiopathological functions. Biochimie 2008, 90, 227–242. [Google Scholar] [CrossRef]
- Pejler, G.; Rönnberg, E.; Waern, I.; Wernersson, S. Mast cell proteases: Multifaceted regulators of inflammatory disease. Blood 2010, 115, 4981–4990. [Google Scholar] [CrossRef]
- Caughey, G.H. Mast cell proteases as protective and inflammatory mediators. Single Mol. Single Cell Seq. 2011, 716, 212–234. [Google Scholar]
- Hellman, L.; Thorpe, M. Granule proteases of hematopoietic cells, a family of versatile inflammatory mediators – an update on their cleavage specificity, in vivo substrates, and evolution. Boil. Chem. 2014, 395, 15–49. [Google Scholar] [CrossRef]
- Schwartz, L.B.; Irani, A.M.; Roller, K.; Castells, M.C.; Schechter, N.M. Quantitation of histamine, tryptase, and chymase in dispersed human T and TC mast cells. J. Immunol. 1987, 138, 2611–2615. [Google Scholar]
- Galli, S.J.; Starkl, P.; Marichal, T.; Tsai, M. Mast cells and IgE in defense against venoms: Possible “good side” of allergy? Allergol. Int. 2016, 65, 3–15. [Google Scholar] [CrossRef]
- Piliponsky, A.M.; Chen, C.C.; Rios, E.J.; Treuting, P.M.; Lahiri, A.; Abrink, M.; Pejler, G.; Tsai, M.; Galli, S.J. The chymase mouse mast cell protease 4 degrades TNF, limits inflammation, and promotes survival in a model of sepsis. Am. J. Pathol. 2012, 181, 875–886. [Google Scholar] [CrossRef]
- Mizutani, H.; Schechter, N.; Lazarus, G.; Black, R.A.; Kupper, T.S. Rapid and specific conversion of precursor interleukin 1 beta (IL-1 beta) to an active IL-1 species by human mast cell chymase. J. Exp. Med. 1991, 174, 821–825. [Google Scholar] [CrossRef]
- Omoto, Y.; Tokime, K.; Yamanaka, K.; Habe, K.; Morioka, T.; Kurokawa, I.; Tsutsui, H.; Yamanishi, K.; Nakanishi, K.; Mizutani, H. Human mast cell chymase cleaves pro-IL-18 and generates a novel and biologically active IL-18 fragment. J. Immunol. 2006, 177, 8315–8319. [Google Scholar] [CrossRef]
- Longley, B.J.; Tyrrell, L.; Ma, Y.; Williams, D.A.; Halaban, R.; Langley, K.; Lu, H.S.; Schechter, N.M. Chymase cleavage of stem cell factor yields a bioactive, soluble product. Proc. Natl. Acad. Sci. USA 1997, 94, 9017–9021. [Google Scholar] [CrossRef] [Green Version]
- Guillabert, A.; Wittamer, V.; Bondue, B.; Godot, V.; Imbault, V.; Parmentier, M.; Communi, D. Role of neutrophil proteinase 3 and mast cell chymase in chemerin proteolytic regulation. J. Leukoc. Boil. 2008, 84, 1530–1538. [Google Scholar] [CrossRef] [Green Version]
- Gela, A.; Kasetty, G.; Jovic, S.; Ekoff, M.; Nilsson, G.; Mörgelin, M.; Kjellstrom, S.; Pease, J.E.; Schmidtchen, A.; Egesten, A. Eotaxin-3 (CCL26) exerts innate host defense activities that are modulated by mast cell proteases. Allergy 2015, 70, 161–170. [Google Scholar] [CrossRef]
- Waern, I.; Lundequist, A.; Pejler, G.; Wernersson, S. Mast cell chymase modulates IL-33 levels and controls allergic sensitization in dust-mite induced airway inflammation. Mucosal Immunol. 2013, 6, 911–920. [Google Scholar] [CrossRef]
- Roy, A.; Ganesh, G.; Sippola, H.; Bolin, S.; Sawesi, O.; Dagälv, A.; Schlenner, M.S.; Feyerabend, T.; Rodewald, R.H.; Kjellén, L.; et al. Mast cell chymase degrades the alarmins heat shock protein 70, biglycan, HMGB1, and interleukin-33 (IL-33) and limits danger-induced inflammation. J. Biol. Chem. 2014, 289, 237–250. [Google Scholar] [CrossRef]
- Pang, L.; Nie, M.; Corbett, L.; Sutcliffe, A.; Knox, A.J. Mast cell beta-tryptase selectively cleaves eotaxin and RANTES and abrogates their eosinophil chemotactic activities. F1000 Post Publ. Peer Rev. Biomed. Lit. 2006, 176, 3788–3795. [Google Scholar]
- Fu, Z.; Thorpe, M.; Alemayehu, R.; Roy, A.; Kervinen, J.; De Garavilla, L.; Abrink, M.; Hellman, L. Highly selective cleavage of cytokines and chemokines by the human mast cell chymase and neutrophil cathepsin G. J. Immunol. 2017, 198, 1474–1483. [Google Scholar] [CrossRef]
- Le, Q.T.; Lyons, J.J.; Naranjo, A.N.; Olivera, A.; Lazarus, A.R.; Metcalfe, D.D.; Milner, J.D.; Schwartz, L.B. Impact of naturally forming human alpha/beta-tryptase heterotetramers in the pathogenesis of hereditary alpha-tryptasemia. J. Exp. Med. 2019, 216, 2348–2361. [Google Scholar] [CrossRef]
- Hallgren, J.; Karlson, U.; Poorafshar, M.; Hellman, L.; Pejler, G.; Martinsson, J.H. Mechanism for activation of mouse mast cell tryptase: Dependence on heparin and acidic pH for formation of active tetramers of mouse mast cell protease 6†. Biochemistry 2000, 39, 13068–13077. [Google Scholar] [CrossRef]
- Pereira, P.J.; Bergner, A.; Macedo-Ribeiro, S.; Huber, R.; Matschiner, G.; Fritz, H.; Sommerhoff, C.P.; Bode, W. Human beta-tryptase is a ring-like tetramer with active sites facing a central pore. Nature 1998, 392, 306–311. [Google Scholar] [CrossRef]
- Rückert, R.; Herz, U.; Paus, R.; Ungureanu, D.; Pohl, T.; Renz, H.; Bulfone-Paus, S. IL-15-IgG2b fusion protein accelerates and enhances a Th2 but not a Th1 immune response in vivo, while IL-2-IgG2b fusion protein inhibits both. Eur. J. Immunol. 1998, 28, 3312–3320. [Google Scholar] [CrossRef]
- Rückert, R.; Brandt, K.; Braun, A.; Hoymann, H.-G.; Herz, U.; Budagian, V.; Dürkop, H.; Renz, H.; Bulfone-Paus, S. Blocking IL-15 prevents the induction of allergen-specific T cells and allergic inflammation in vivo. J. Immunol. 2005, 174, 5507–5515. [Google Scholar] [CrossRef] [PubMed]
- Wurster, A.L.; Rodgers, V.L.; Satoskar, A.R.; Whitters, M.J.; Young, D.A.; Collins, M.; Grusby, M.J. Interleukin 21 is a T helper (Th) cell 2 cytokine that specifically inhibits the differentiation of naive Th cells into interferon gamma-producing Th1 cells. J. Exp. Med. 2002, 196, 969–977. [Google Scholar] [CrossRef] [PubMed]
- Niles, A.L.; Maffitt, M.; Haak-Frendscho, M.; Wheeless, C.J.; Johnson, D.A. Recombinant human mast cell tryptase beta: Stable expression in Pichia pastoris and purification of fully active enzyme. Biotechnol. Appl. Biochem. 1998, 28, 125–131. [Google Scholar] [PubMed]
- Doolittle, R.F. Structural basis of the fibrinogen-fibrin transformation: Contributions from X-ray crystallography. Blood Rev. 2003, 17, 33–41. [Google Scholar] [CrossRef]
- Schwartz, L.B.; Bradford, T.R.; Littman, B.H.; Wintroub, B.U. The fibrinogenolytic activity of purified tryptase from human lung mast cells. J. Immunol. 1985, 135, 2762–2767. [Google Scholar]
- Kaminska, R.; Helisalmi, P.; Harvima, R.J.; Horsmanheimo, M.; Harvima, I.T.; Naukkarinen, A. Focal Dermal–epidermal separation and fibronectin cleavage in basement membrane by human mast cell tryptase. J. Investig. Dermatol. 1999, 113, 567–573. [Google Scholar] [CrossRef]
- Im, E.; Chung, K.C. Precise assembly and regulation of 26S proteasome and correlation between proteasome dysfunction and neurodegenerative diseases. BMB Rep. 2016, 49, 459–473. [Google Scholar] [CrossRef]
- Vanderlinden, R.T.; Hemmis, C.W.; Yao, T.; Robinson, H.; Hill, C.P. Structure and energetics of pairwise interactions between proteasome subunits RPN2, RPN13, and ubiquitin clarify a substrate recruitment mechanism. J. Boil. Chem. 2017, 292, 9493–9504. [Google Scholar] [CrossRef] [Green Version]
- Chahal, G.; Thorpe, M.; Hellman, L. The importance of exosite interactions for substrate cleavage by human thrombin. PLoS ONE 2015, 10, e0129511. [Google Scholar] [CrossRef]
- Forsberg, E.; Pejler, G.; Ringvall, M.; Lunderius, C.; Tomasini-Johansson, B.; Kusche-Gullberg, M.; Eriksson, I.; Ledin, J.; Hellman, L.; Kjellén, L. Abnormal mast cells in mice deficient in a heparin-synthesizing enzyme. Nature 1999, 400, 773–776. [Google Scholar] [CrossRef]
- Pejler, G.; Sadler, J.E. Mechanism by which heparin proteoglycan modulates mast cell chymase activity†. Biochemistry 1999, 38, 12187–12195. [Google Scholar] [CrossRef]
- Tchougounova, E.; Forsberg, E.; Angelborg, G.; Kjellén, L.; Pejler, G. Altered processing of fibronectin in mice lacking heparin. a role for heparin-dependent mast cell chymase in fibronectin degradation. J. Biol. Chem. 2001, 276, 3772–3777. [Google Scholar] [CrossRef]
- Tchougounova, E. Regulation of extravascular coagulation and fibrinolysis by heparin-dependent mast cell chymase. FASEB J. 2001, 15, 2763–2765. [Google Scholar] [CrossRef]
- Caughey, G.H.; Leidig, F.; Viro, N.F.; A Nadel, J. Substance P and vasoactive intestinal peptide degradation by mast cell tryptase and chymase. J. Pharmacol. Exp. Ther. 1988, 244, 133–137. [Google Scholar]
- Tam, E.K.; Caughey, G.H. Degradation of airway neuropeptides by human lung tryptase. Am. J. Respir. Cell Mol. Boil. 1990, 3, 27–32. [Google Scholar] [CrossRef]
- Hellman, L. Regulation of IgE homeostasis, and the identification of potential targets for therapeutic intervention. Biomed. Pharmacother. 2007, 61, 34–49. [Google Scholar] [CrossRef]
- Hellman, L. Therapeutic vaccines against IgE-mediated allergies. Expert Rev. Vaccines 2008, 7, 193–208. [Google Scholar] [CrossRef]
- Lei, Y.; Boinapally, V.; Zoltowska, A.; Adner, M.; Hellman, L.; Nilsson, G. Vaccination against IL-33 inhibits airway hyperresponsiveness and inflammation in a house dust mite model of asthma. PLoS ONE 2015, 10, e0133774. [Google Scholar] [CrossRef]
- Nakanishi, K. Unique action of interleukin-18 on T cells and other immune cells. Front. Immunol. 2018, 9, 763. [Google Scholar] [CrossRef]
- Punnonen, J.; Aversa, G.; Cocks, B.G.; McKenzie, A.N.; Menon, S.; Zurawski, G.; Malefyt, R.D.W.; De Vries, J.E. Interleukin 13 induces interleukin 4-independent IgG4 and IgE synthesis and CD23 expression by human B cells. Proc. Natl. Acad. Sci. USA 1993, 90, 3730–3734. [Google Scholar] [CrossRef]
- Miller, H.R.; Wright, S.H.; Knight, P.A.; Thornton, E.M. A novel function for transforming growth factor-beta1: Upregulation of the expression and the IgE-independent extracellular release of a mucosal mast cell granule-specific beta-chymase, mouse mast cell protease-1. Blood 1999, 93, 3473–3486. [Google Scholar] [CrossRef]
- Waldmann, T.A.; Tagaya, Y. The multifaceted regulation of interleukin-15 expression and the role of this cytokine in NK cell differentiation and host response to intracellular pathogens 1. Annu. Rev. Immunol. 1999, 17, 19–49. [Google Scholar] [CrossRef]
- Vernersson, M. Generation of therapeutic antibody responses against IgE through vaccination. FASEB J. 2002, 16, 875–877. [Google Scholar] [CrossRef]
- Reimer, J.M.; Enoksson, M.; Samollow, P.B.; Hellman, L. Extended substrate specificity of opossum chymase—Implications for the origin of mast cell chymases. Mol. Immunol. 2008, 45, 2116–2125. [Google Scholar] [CrossRef]
- Andersson, M.K.; Enoksson, M.; Gallwitz, M.; Hellman, L. The extended substrate specificity of the human mast cell chymase reveals a serine protease with well-defined substrate recognition profile. Int. Immunol. 2009, 21, 95–104. [Google Scholar] [CrossRef]
- Neuhoff, V.; Arold, N.; Taube, D.; Ehrhardt, W. Improved staining of proteins in polyacrylamide gels including isoelectric focusing gels with clear background at nanogram sensitivity using Coomassie Brilliant Blue G-250 and R-250. Electrophoresis 1988, 9, 255–262. [Google Scholar] [CrossRef]


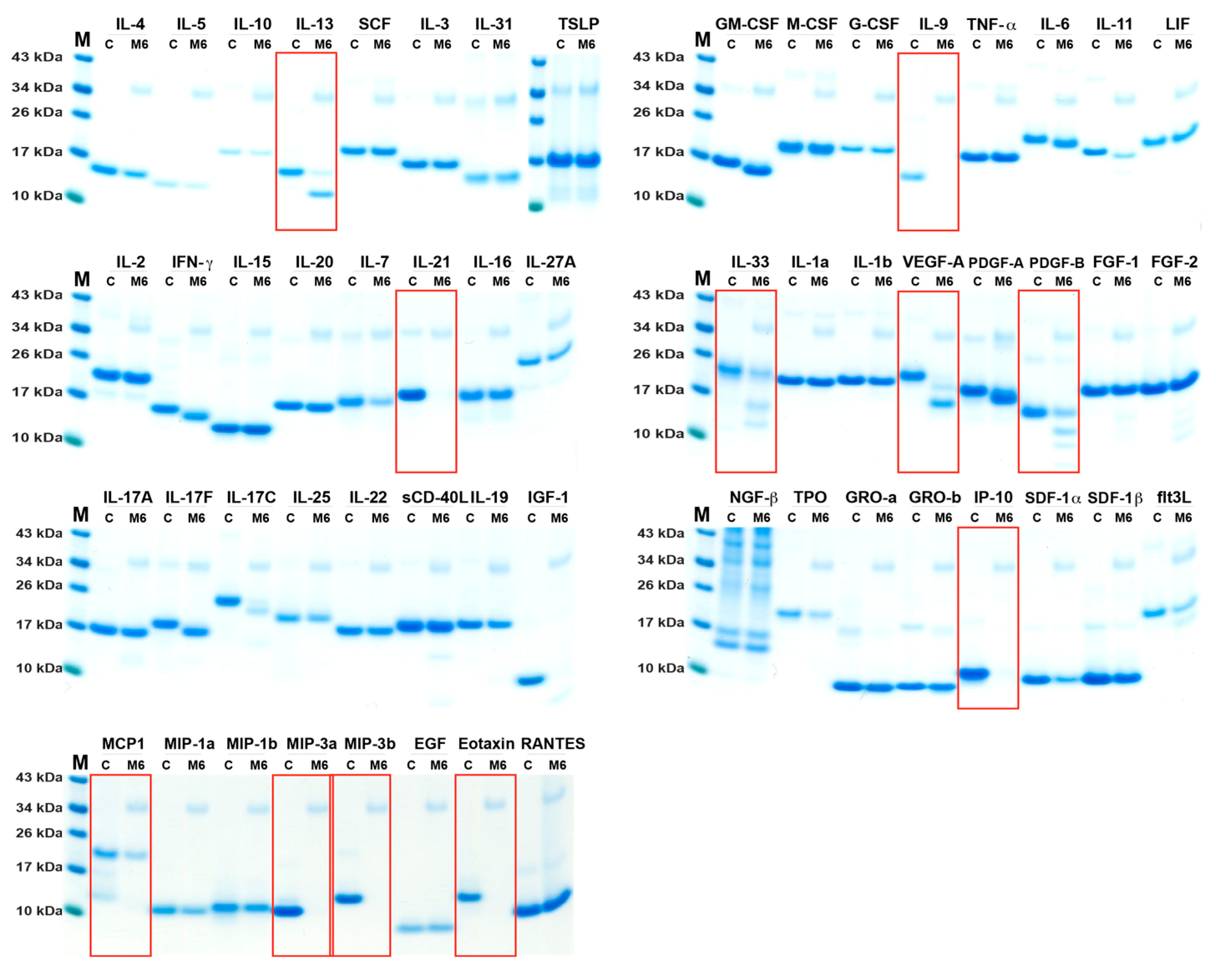

| Cytokine | hTryp. | M6 | HC | Cytokine | hTryp. | M6 | HC |
|---|---|---|---|---|---|---|---|
| IL-4 | − | − | − | IL-1α | − | − | |
| IL-5 | − | − | − | IL-1β | − | − | − |
| IL-10 | − | − | − | IL-1RA | + | ||
| TSLP | ++++ | − | + | IL-33 | − | + | ++++ |
| IL-13 | − | +++ | + | IL-18 | − | +++ | |
| SCF | − | − | − | IGF-1 | − | ++++ | |
| IL-3 | − | − | + | IGF-2 | − | ||
| IL-31 | − | − | − | HGF | − | ||
| GM-CSF | − | + | − | IL-8 | ++ | − | |
| M-CSF | − | − | IP-10 | − | ++++ | − | |
| G-CSF | − | − | − | MCP-1 | − | + | − |
| TGF- β3 | − | MCP-2 | − | − | |||
| TNF-α | − | − | − | MCP-3 | ++++ | − | |
| IL-6 | − | + | ++ | RANTES | − | − | − |
| IL-11 | − | + | + | SDF-1α | − | ++ | − |
| LIF | − | − | + | SDF-1β | + | − | − |
| IL-2 | − | − | − | EGF | − | − | − |
| IFN-γ | +++ | + | + | MIP-1α | − | − | - |
| IL-15 | − | − | ++++ | flt3L | − | + | ++ |
| IL-12 | − | − | FGF-9 | − | |||
| IL-20 | + | − | − | FGF-19 | − | ||
| IL-7 | + | + | − | BMP-14 | − | ||
| IL-21 | ++++ | ++++ | − | GAL-7H | − | ||
| IL-16 | − | − | pF4V1 | − | |||
| VEGF-A | + | ++++ | − | IL-25 | − | − | − |
| VEGF-121 | − | IL-9 | − | ++++ | |||
| PDGF-A | − | + | − | MIP-3a | − | ++++ | |
| PDGF-B | − | +++ | − | MIP-3b | ++++ | ++++ | |
| BMP-2 | − | Eotaxin | +++ | ++++ | |||
| CTGF | ++ | + | IL-27A | - | |||
| FGF-1 | + | − | + | IL17C | ++++ | ||
| FGF-2 | − | + | + | NGF-β | − | ||
| IL-17A | + | − | − | TPO | + | ||
| IL-17F | + | + | − | GRO-a | − | ||
| IL-22 | − | − | − | GRO-b | − | ||
| CD40L | − | + | − | MIP-1b | − | ||
| BAFF | − | + | |||||
| IL-19 | − | − | − | ||||
| sIL-6R | − | ||||||
| CNTF | − |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fu, Z.; Akula, S.; Thorpe, M.; Hellman, L. Highly Selective Cleavage of TH2-Promoting Cytokines by the Human and the Mouse Mast Cell Tryptases, Indicating a Potent Negative Feedback Loop on TH2 Immunity. Int. J. Mol. Sci. 2019, 20, 5147. https://doi.org/10.3390/ijms20205147
Fu Z, Akula S, Thorpe M, Hellman L. Highly Selective Cleavage of TH2-Promoting Cytokines by the Human and the Mouse Mast Cell Tryptases, Indicating a Potent Negative Feedback Loop on TH2 Immunity. International Journal of Molecular Sciences. 2019; 20(20):5147. https://doi.org/10.3390/ijms20205147
Chicago/Turabian StyleFu, Zhirong, Srinivas Akula, Michael Thorpe, and Lars Hellman. 2019. "Highly Selective Cleavage of TH2-Promoting Cytokines by the Human and the Mouse Mast Cell Tryptases, Indicating a Potent Negative Feedback Loop on TH2 Immunity" International Journal of Molecular Sciences 20, no. 20: 5147. https://doi.org/10.3390/ijms20205147






