Physiological and Transcriptomic Analyses Elucidate That Exogenous Calcium Can Relieve Injuries to Potato Plants (Solanum tuberosum L.) under Weak Light
Abstract
1. Introduction
2. Results
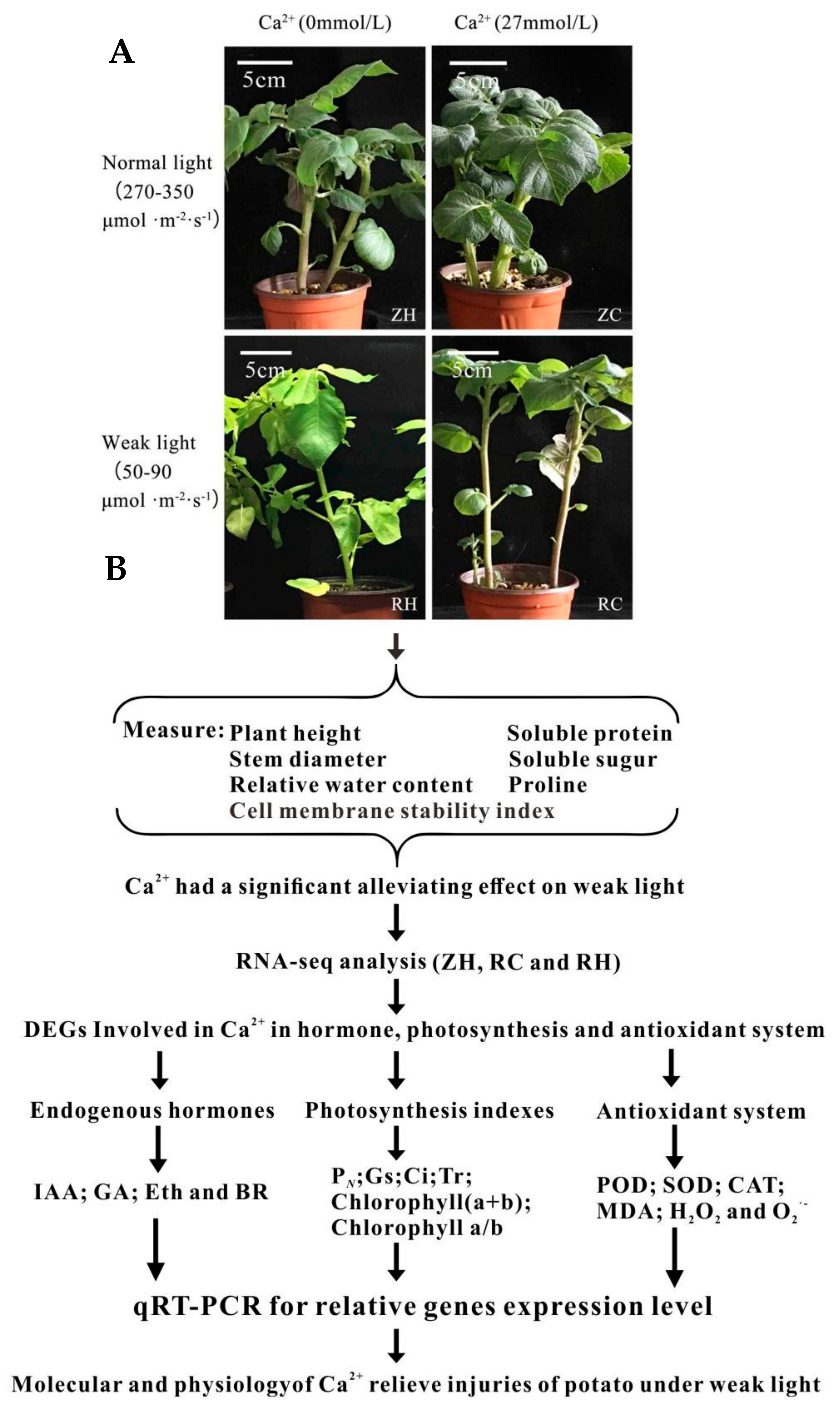
2.1. Effects of Exogenous Ca2+ Treatment Morphological Indexes under Weak Light of Potato
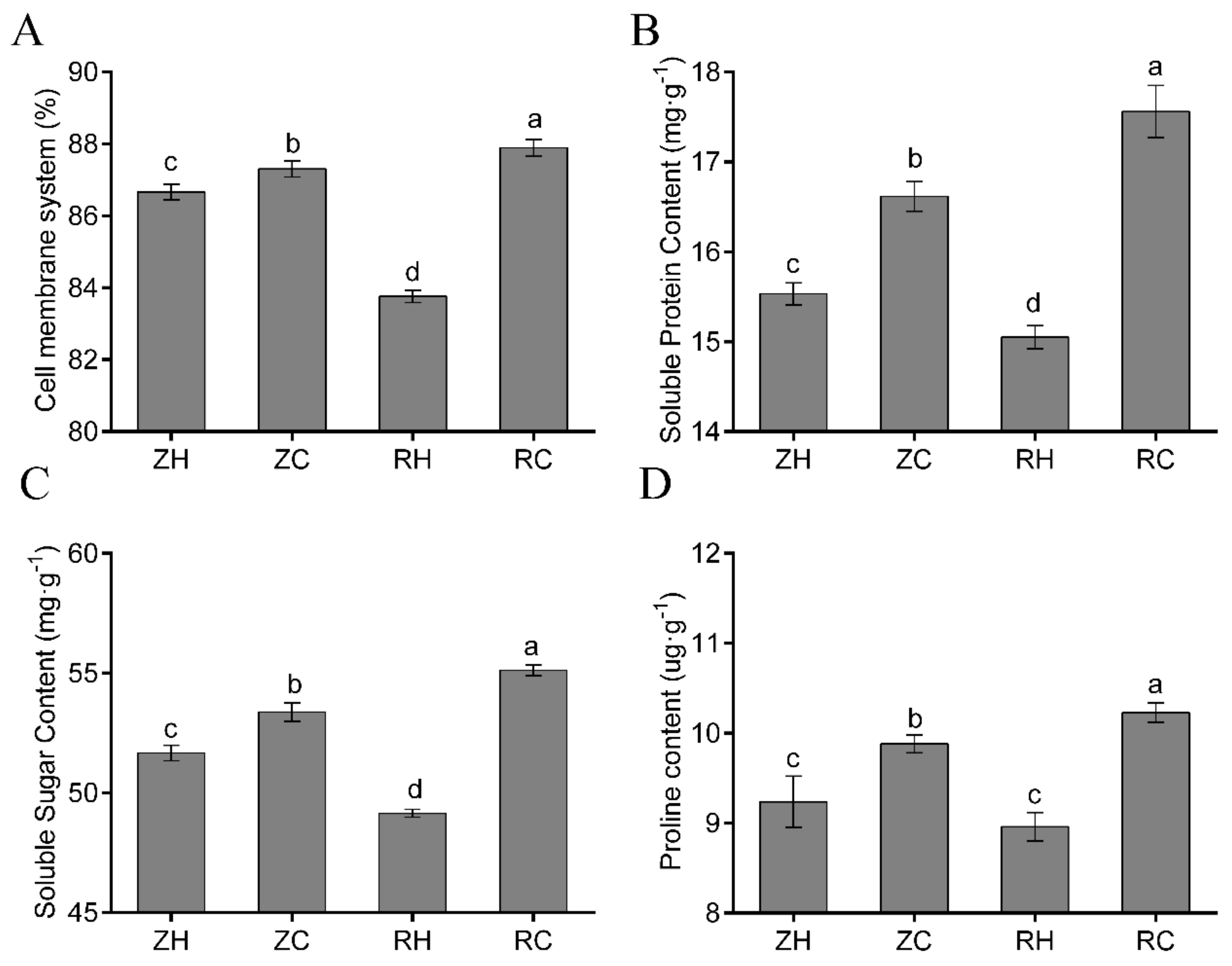
2.2. Physiological Indexes of Exogenous Ca2+ Related to Homeostasis
2.3. RNA-seq Assembly of the Potato Transcriptome Reference
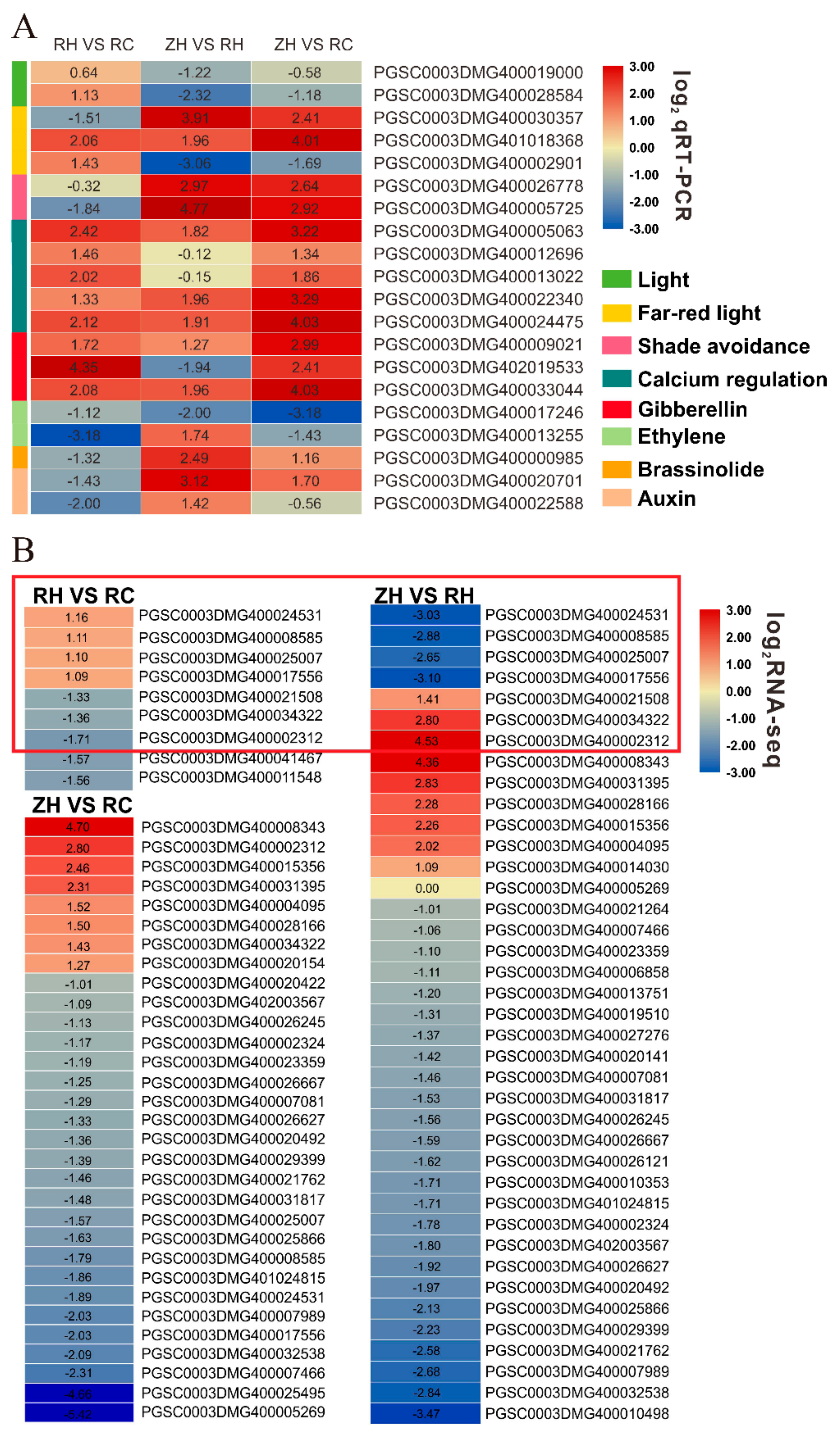
2.4. Comparative Analysis of DEGs
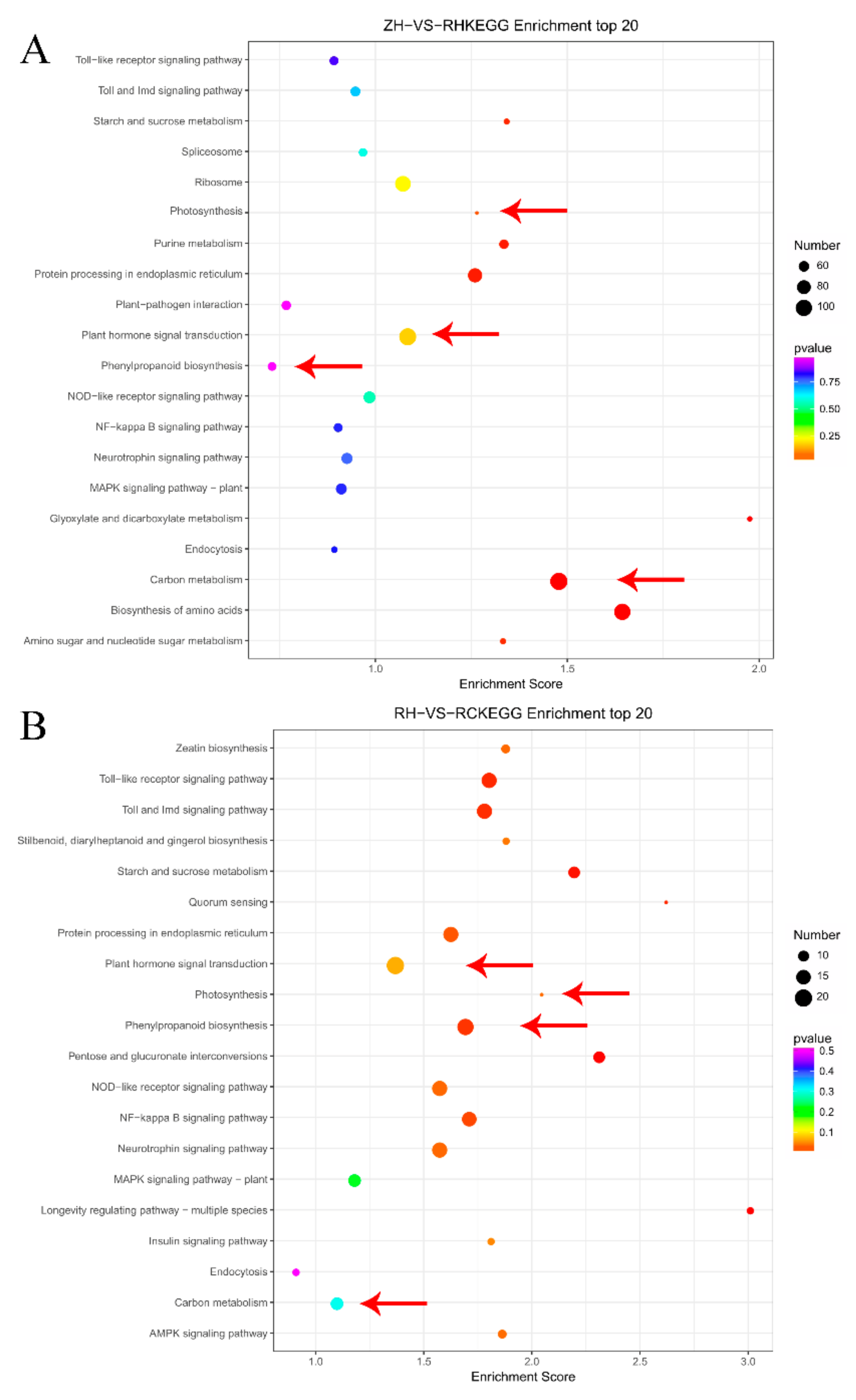
2.5. GO and KEGG Enrichment of Potatoes under Weak Light and Exogenous Ca2+ Treatment
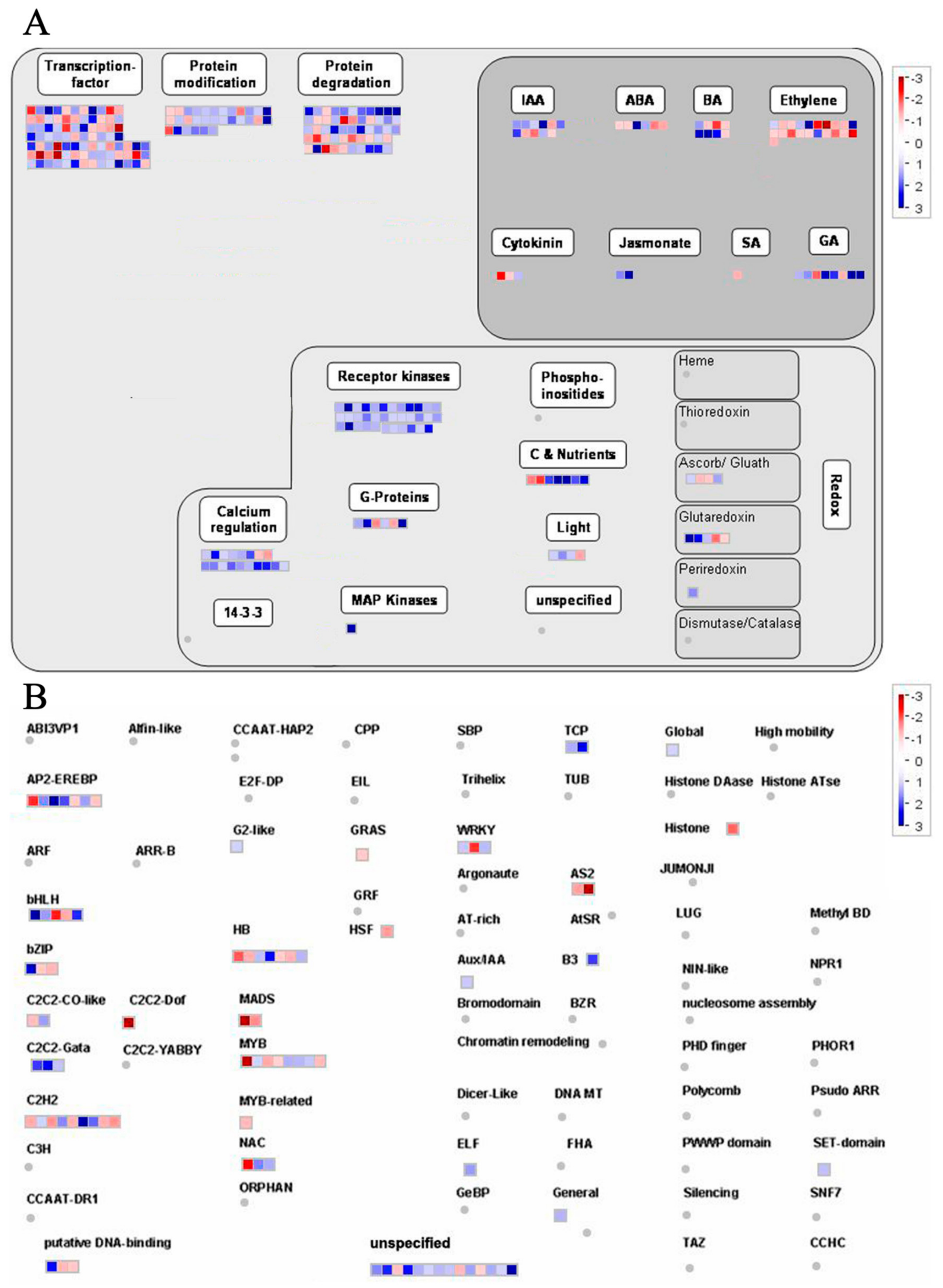
2.6. Overview and Analysis of Regulatory Pathways of DEGs
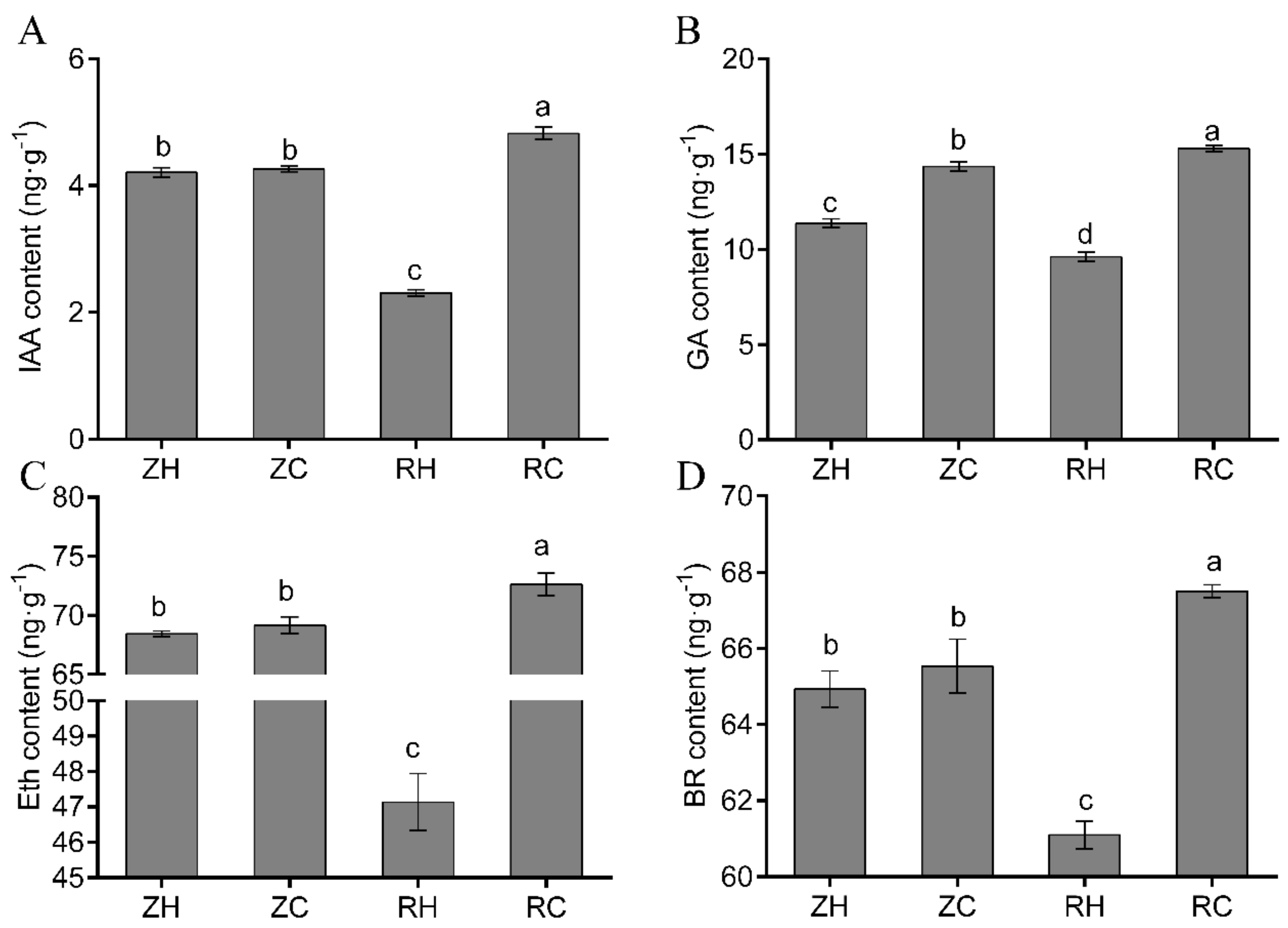
2.7. Changes in Endogenous Hormone Content and Related DEGs in Potatoes
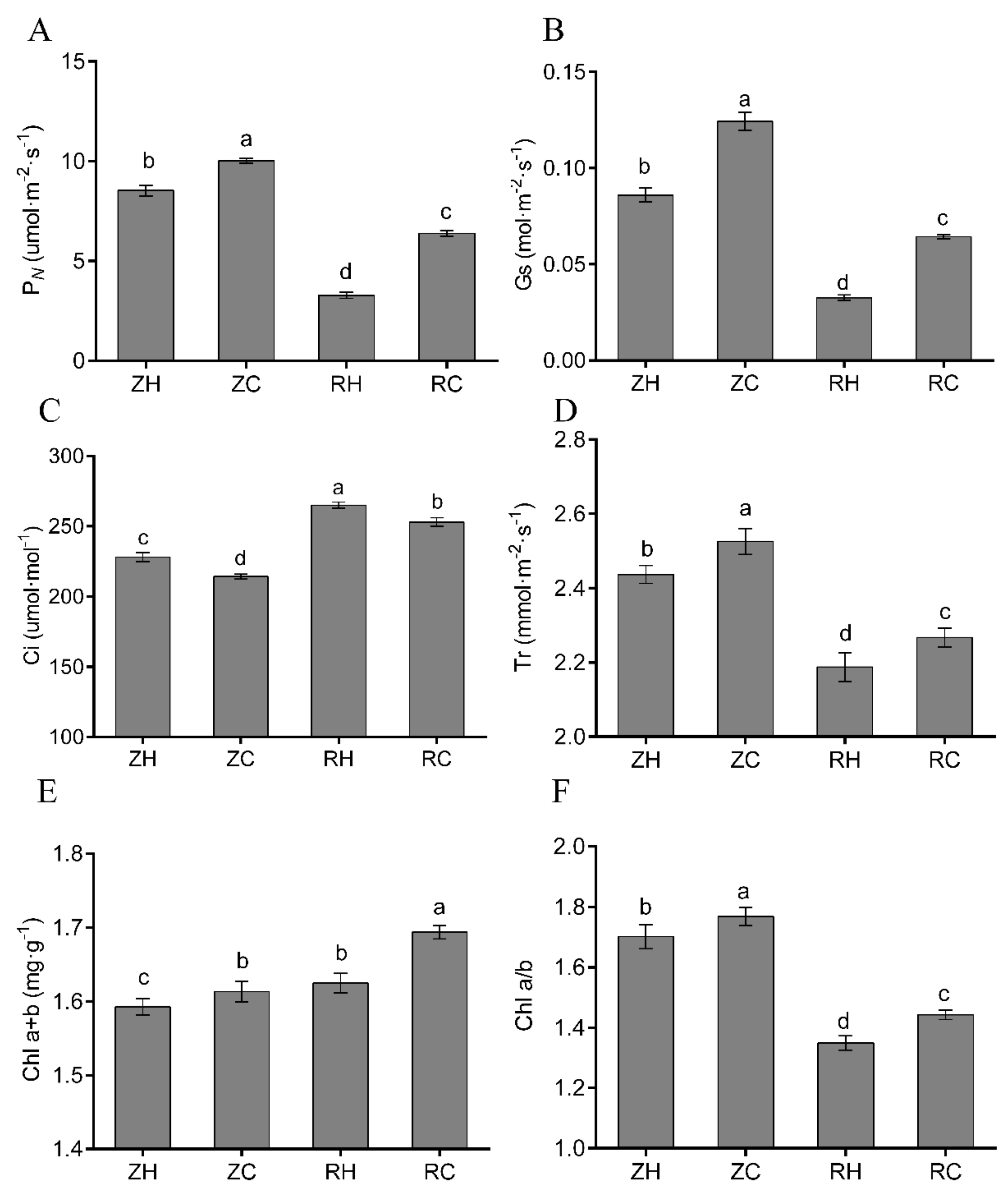
2.8. Effects of Exogenous Ca2+ on Chlorophyll Content and Photosynthesis under Weak Light
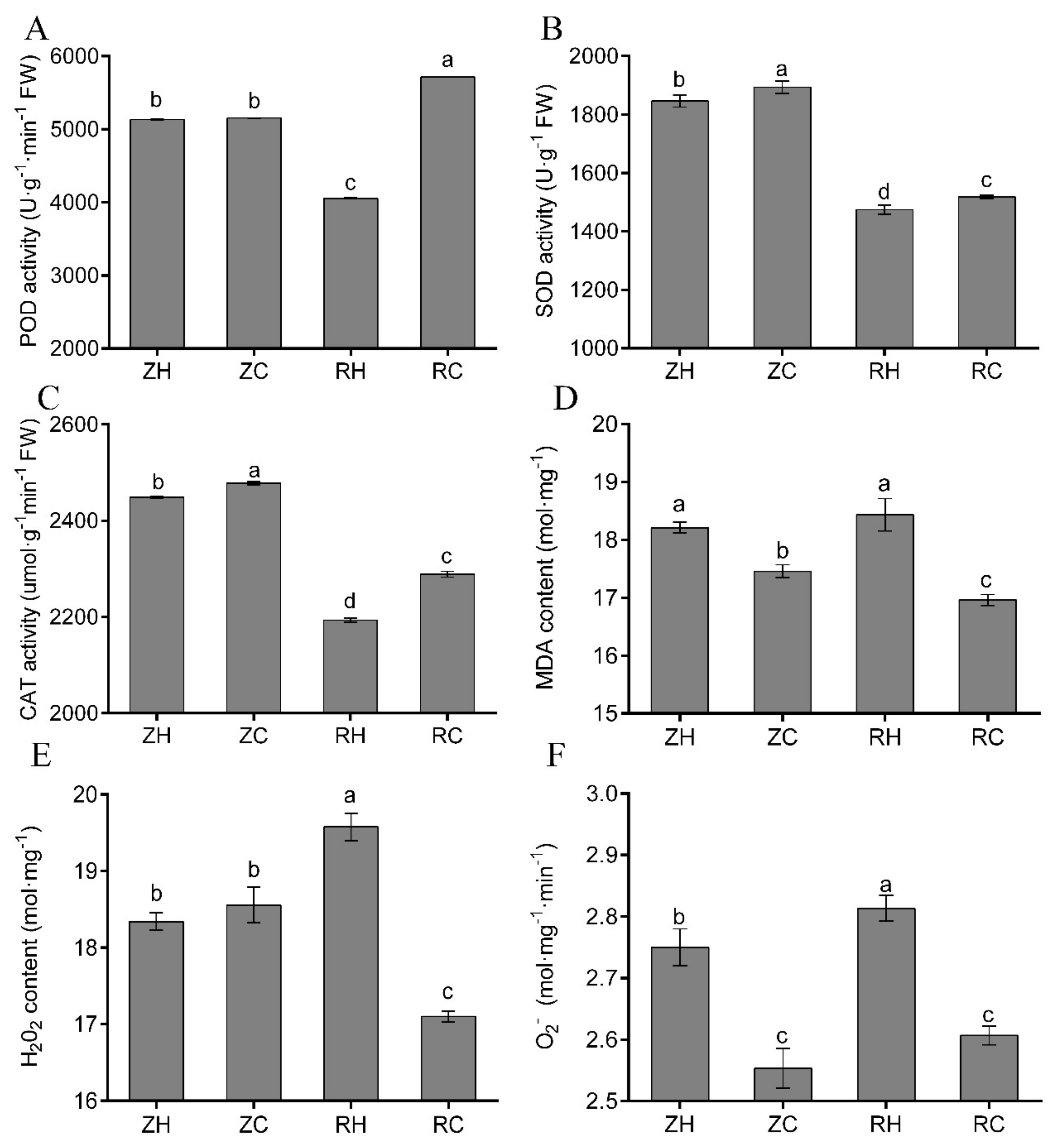
2.9. Effects of Exogenous Ca2+ on the Antioxidant System under Weak Light
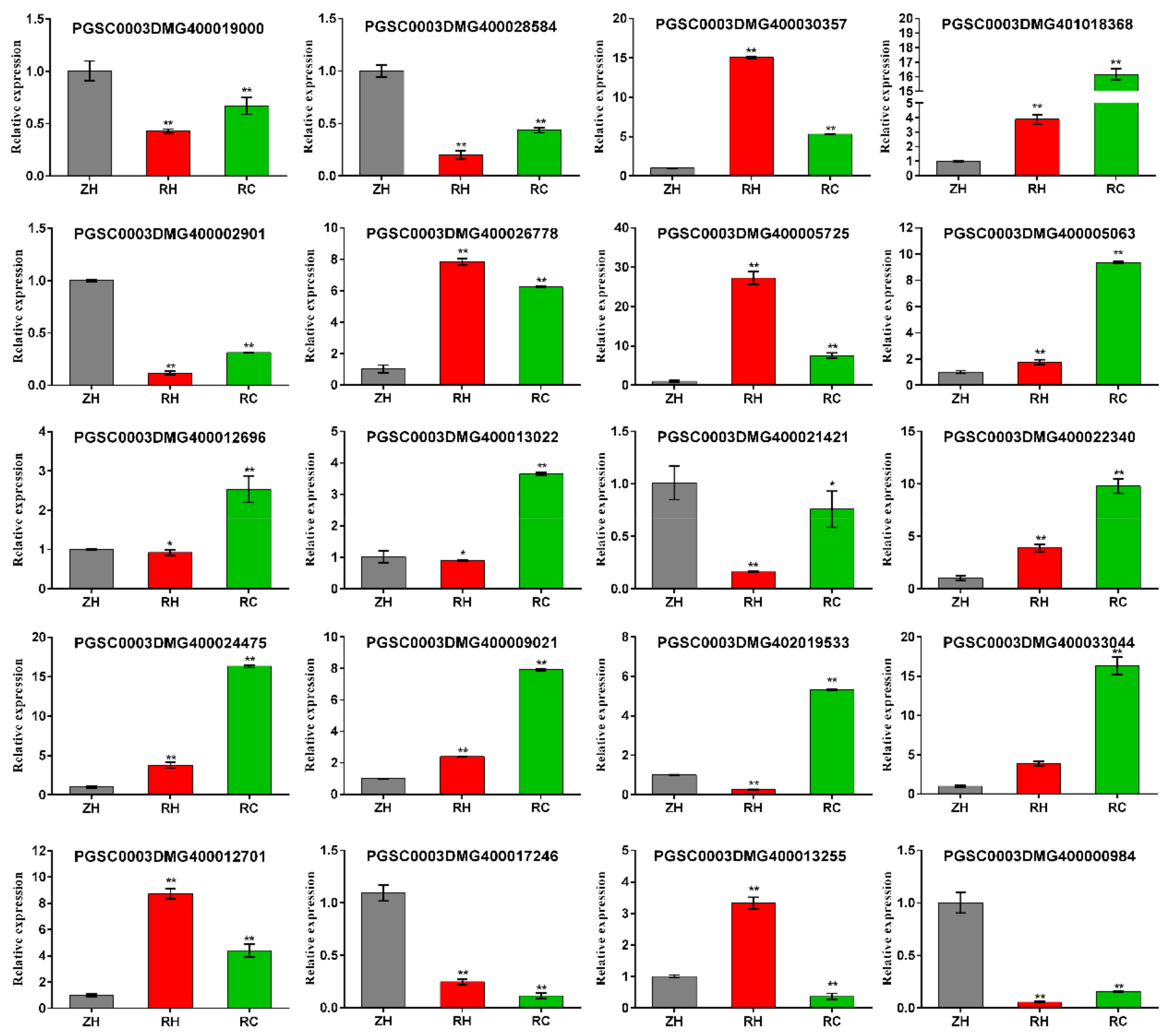
2.10. Relative DEGs Measured by qRT-PCR
3. Discussion
3.1. RNA-seq Analysis Reveal DEGs under Weak Light
3.2. Effects of Exogenous Ca2+ Treatment on Endogenous IAA, GA, BR, and Eth Contents under Weak Light
3.3. Exogenous Ca2+ Relieves The Changes in Physiological Indexes under Weak Light Stress
3.4. Exogenous Ca2+ Relieves The Changes in Photosynthesis and The Antioxidant System under Weak Light Stress
3.5. Exogenous Ca2+ Relieves Injuries to Potatoes under Weak Light Stress
4. Materials and Methods
4.1. Plant Materials
4.2. Determination of Exogenous Ca2+ Treatment Morphology and Osmotic Adjustment Substances under Weak Light
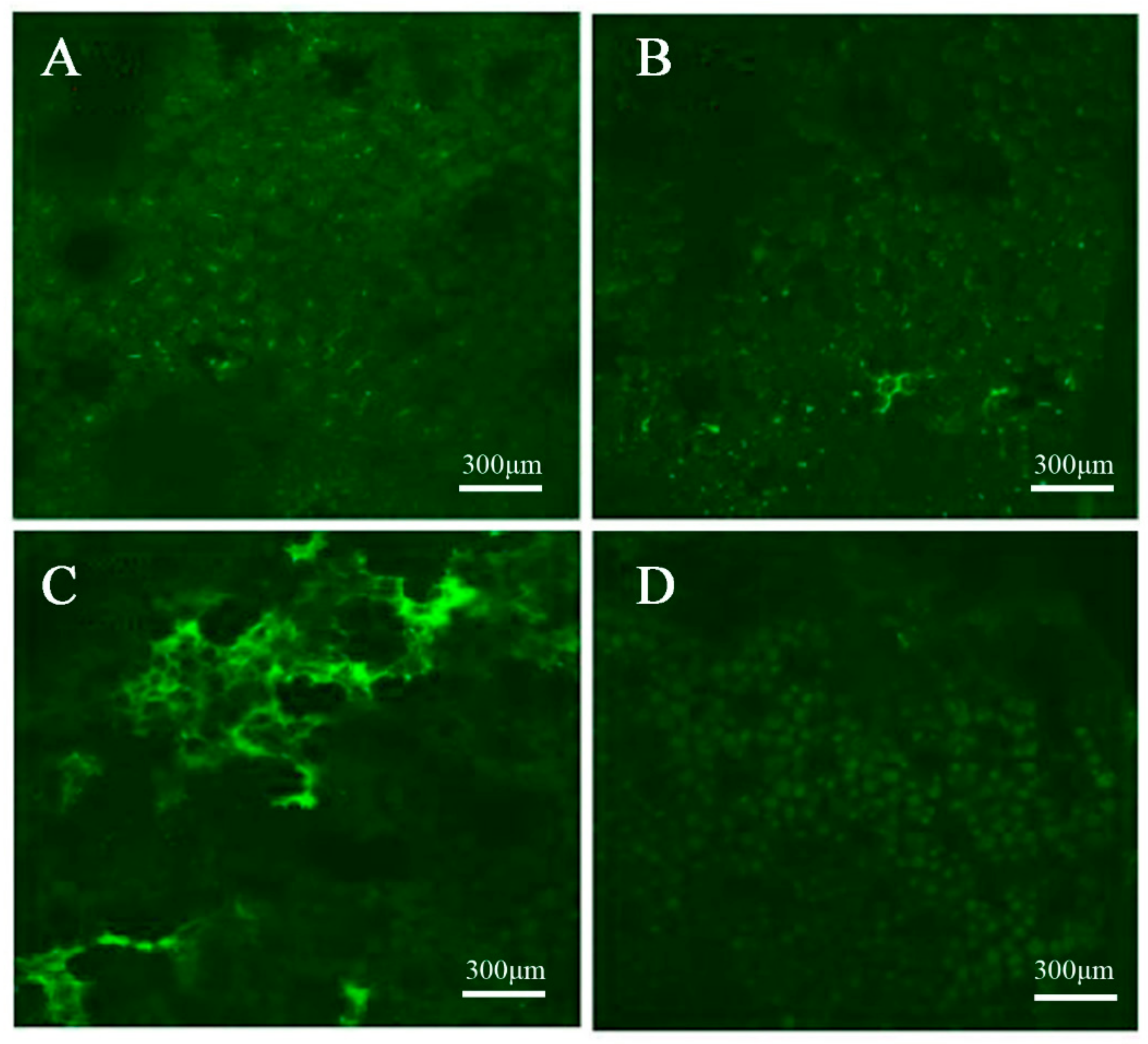
4.3. Ca2+ Fluorescence Signal Localization
4.4. Determination of Endogenous IAA, Eth, GA and BRs Contents
4.5. Determination of Exogenous Ca2+ on The Photosynthetic Capacity and Chlorophyll Content under Weak Light
4.6. Determination of Exogenous Ca2+ on Activities of Antioxidant Enzymes under Weak Light
4.7. cDNA Library Construction and RNA-seq Analysis
4.8. Analysis of DEGs, Cluster Analysis, GO, and KEGG Enrichment
4.9. Functional Annotation and KEGG Enrichment Pathway Analysis
4.10. qRT-PCR Analysis
4.11. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| RWC | Relative water content |
| SD | Stem diameter |
| H | Height |
| CMSI | Cell membrane stability index |
| SS | Soluble sugar |
| SP | Soluble protein |
| PR | Proline |
| O2− | Superoxide anion |
| Tr | Transpiration rate |
| PIF | Phytochrome interaction factor |
| Ci | Intercellular CO2 concentration |
| Gs | Stomatal conductance |
| PN | Net photosynthetic rate |
| Chl | Chlorophyll |
| IAA | Auxin |
| GA | Gibberellin |
| BR | Brassinolide |
| MDA | Malondialdehyde |
| CAT | Catalase |
| POD | Peroxidase |
| SOD | superoxide dismutase |
| CK | Control Check |
| DEGs | Different expressed genes |
| BP | Biological process |
| CC | Cellular component |
| MF | Molecular function |
| TF | Transcription factor |
| GO | Gene ontology |
| KEGG | Kyoto Encyclopedia of Genes and Genomes |
| ROS | Reactive oxygen species |
| FPKM | Fragments per kb per million reads |
References
- Schittenhelm, S.; Mengehartmann, U.; Oldenburg, E. Photosynthesis, carbohydrate metabolism, and yield of phytochrome-B-overexpressing potatoes under different light regimes. Crop. Sci. 2004, 44, 131–143. [Google Scholar] [CrossRef]
- Fankhauser, C.; Christie, J.M. Plant phototropic growth. Curr. Biol. 2015, 25, 384–389. [Google Scholar] [CrossRef] [PubMed]
- Franklin, K.A.; Whitelam, G.C. Phytochromes and shade-avoidance responses in plants. Ann. Bot. 2005, 96, 169–175. [Google Scholar] [CrossRef] [PubMed]
- Fu, W.G.; Li, P.P.; Wu, Y.Y. Effects of different light intensities on chlorophyll fluorescence characteristics and yield in lettuce. Sci. Hortic. 2012, 135, 45–51. [Google Scholar] [CrossRef]
- Han, S.; Tang, R.; Anderson, L.K.; Woerner, T.E.; Pei, Z.M. A cell surface receptor mediates extracellular Ca2+ sensing in guard cells. Nature 2003, 425, 196–200. [Google Scholar] [CrossRef]
- Cardenas, L.; Vidali, L.; Perez, H.; Sanchez, F.; Kunkel, J.; Feijo, J.; Hepler, P.; Quinto, C. Intracellular and Extracellular Calcium Changes in Plant. Root Hair Cells Responding to Nodulation Signals; Springer International Publishing: Cham, Switzerland, 1998; Available online: https://link.springer.com/chapter/10.1007/978-94-011-5159-7_123 (accessed on 28 September 2019).
- Reid, R.J.; Tester, M.A.; Smith, F.A. Calcium/aluminium interactions in the cell wall and plasma membrane of Chara. Planta 1995, 195, 362–368. [Google Scholar] [CrossRef]
- Tan, W.; Meng, Q.; Brestic, M.; Olsovska, K.; Yang, X. Photosynthesis is improved by exogenous calcium in heat-stressed tobacco plants. J. Plant Physiol. 2011, 168, 2063–2071. [Google Scholar] [CrossRef]
- Colomer, J.; Means, A.R. Physiological roles of the Ca2+/CaM-dependent protein kinase cascade in health and disease. Subcell. Biochem. 2007, 45, 169–214. [Google Scholar]
- Clack, T.; Mathews, S.; Sharrock, R.A. The phytochrome apoprotein family in Arabidopsis is encoded by five genes: The sequences and expression of PHYD and PHYE. Plant Mol. Biol. 1994, 25, 413–427. [Google Scholar] [CrossRef]
- Castillon, A.; Shen, H.; Huq, E. Phytochrome Interacting Factors: Central players in phytochrome-mediated light signaling networks. Trends Plant Sci. 2007, 12, 514–521. [Google Scholar] [CrossRef]
- Vandenbussche, F.; Vriezen, W.H.; Smalle, J.; Laarhoven, L.J.; Harren, F.J.; Van Der Straeten, D. Ethylene and auxin control the Arabidopsis response to decreased light intensity. Plant Physiol. 2003, 133, 517–527. [Google Scholar] [CrossRef] [PubMed]
- Leivar, P.; Quail, P.H. PIFs: Pivotal components in a cellular signaling hub. Trends Plant Sci. 2011, 16, 19–28. [Google Scholar] [CrossRef] [PubMed]
- Callis, J.; Vierstra, R.D. Protein degradation in signaling. Curr. Opin. Plant Biol. 2000, 3, 381–386. [Google Scholar] [CrossRef]
- Pierik, R.; Cuppens, M.L.; Voesenek, L.A.; Visser, E.J. Interactions between ethylene and gibberellins in phytochrome-mediated shade avoidance responses in tobacco. Plant Physiol. 2004, 136, 2928–2936. [Google Scholar] [CrossRef]
- Stamm, P.; Kumar, P.P. The phytohormone signal network regulating elongation growth during shade avoidance. J. Exp. Bot. 2010, 61, 2889–2903. [Google Scholar] [CrossRef]
- Browse, J.; Howe, G.A. New weapons and a rapid response against insect attack. Plant Physiol. 2008, 146, 832–838. [Google Scholar] [CrossRef]
- Keuskamp, D.H.; Pollmann, S.; Voesenek, L.A.; Peeters, A.J.; Pierik, R. Auxin transport through PIN-FORMED 3 (PIN3) controls shade avoidance and fitness during competition. Proc. Natl. Acad. Sci. USA 2010, 107, 22740–22744. [Google Scholar] [CrossRef]
- Redman, J.C.; Haas, B.J.; Tanimoto, G.; Town, C.D. Development and evaluation of an Arabidopsis whole genome Affymetrix probe array. Plant J. 2004, 38, 545–561. [Google Scholar] [CrossRef]
- Lorrain, S.; Allen, T.; Duek, P.D.; Whitelam, G.C.; Fankhauser, C. Phytochrome-mediated inhibition of shade avoidance involves degradation of growth-promoting bHLH transcription factors. Plant J. 2008, 53, 312–323. [Google Scholar] [CrossRef]
- Khanna, R.; Shen, Y.; Marion, C.M.; Tsuchisaka, A.; Theologis, A.; Schafer, E.; Quail, P.H. The basic helix-loop-helix transcription factor PIF5 acts on ethylene biosynthesis and phytochrome signaling by distinct mechanisms. Plant Cell 2007, 19, 3915–3929. [Google Scholar] [CrossRef]
- Ren, H.; Gray, W.M. SAUR Proteins as Effectors of Hormonal and Environmental Signals in Plant Growth. Mol. Plant 2015, 8, 1153–1164. [Google Scholar] [CrossRef] [PubMed]
- Zou, M.Q.; Yuan, L.Y.; Zhu, S.D.; Liu, S.; Ge, J.T.; Wang, C.G. Effects of heat stress on photosynthetic characteristics and chloroplast ultrastructure of a heat-sensitive and heat-tolerant cultivar of wucai (Brassica campestris L.). Acta Physiol. Plant 2017, 39, 30. [Google Scholar] [CrossRef]
- Zhao, D.; Oosterhuis, D.M. Influence of shade on mineral nutrient status of field-grown cotton. J. Plant Nutr. 1998, 21, 1681–1695. [Google Scholar] [CrossRef]
- Fay, P.A.; Knapp, A.K. Responses to Short-Term Reductions in Light in Soybean Leaves: Effects of Leaf Position and Drought Stress. Int. J. Plant Sci. 1998, 159, 805–811. [Google Scholar] [CrossRef]
- Deng, Y.M.; Shao, Q.S.; Li, C.C.; Ye, X.Q.; Tang, R.S. Differential responses of double petal and multi petal jasmine to shading: II. Morphology, anatomy and physiology. Sci. Hortic. 2012, 144, 19–28. [Google Scholar] [CrossRef]
- Shin, J.; Kim, K.; Kang, H.; Zulfugarov, I.S.; Bae, G.; Lee, C.H.; Lee, D.; Choi, G. Phytochromes promote seedling light responses by inhibiting four negatively-acting phytochrome-interacting factors. Proc. Natl. Acad. Sci. USA 2009, 106, 7660–7665. [Google Scholar] [CrossRef]
- Yang, X.Y.; Ye, X.F.; Liu, G.S.; Wei, H.Q.; Wang, Y. Effects of light intensity on morphological and physiological characteristics of tobacco seedlings. Ying Yong Sheng Tai Xue Bao 2007, 18, 2642–2645. [Google Scholar]
- Batutis, E.J.; Ewing, E.E. Far-Red Reversal of Red light Effect during Long-Night Induction of Potato (Solanum tuberosum L.) Tuberization. Plant Physiol. 1982, 69, 672–674. [Google Scholar] [CrossRef]
- Wuyts, N.; Dhondt, S.; Inze, D. Measurement of plant growth in view of an integrative analysis of regulatory networks. Curr. Opin. Plant Biol. 2015, 25, 90–97. [Google Scholar] [CrossRef]
- Elliott, D.C. Calcium Involvement in Plant Hormone Action. 1986. Available online: https://link.springer.com/chapter/10.1007/978-1-4613-2177-4_35 (accessed on 28 September 2019).
- Achard, P.; Gusti, A.; Cheminant, S.; Alioua, M.; Dhondt, S.; Coppens, F.; Beemster, G.T.; Genschik, P. Gibberellin signaling controls cell proliferation rate in Arabidopsis. Curr. Biol. 2009, 19, 1188–1193. [Google Scholar] [CrossRef]
- Grove, M.D.; Spencer, G.F.; Rohwedder, W.K.; Mandava, N.; Worley, J.F.; Warthen, J.D.; Steffens, G.L.; Flippen-Anderson, J.L.; Cook, J.C. Brassinolide, a plant growth-promoting steroid isolated from Brassica napus pollen. Nature 1979, 281, 216–217. [Google Scholar] [CrossRef]
- Sami, F.; Yusuf, M.; Faizan, M.; Faraz, A.; Hayat, S. Role of sugars under abiotic stress. Plant Physiol. Biochem. 2016, 109, 54–61. [Google Scholar] [CrossRef] [PubMed]
- Catola, S.; Marino, G.; Emiliani, G.; Huseynova, T.; Musayev, M.; Akparov, Z.; Maserti, B.E. Physiological and metabolomic analysis of Punica granatum (L.) under drought stress. Planta 2016, 243, 441–449. [Google Scholar] [CrossRef] [PubMed]
- Apel, K.; Hirt, H. Reactive oxygen species: Metabolism, oxidative stress, and signal transduction. Annu. Rev. Plant Biol. 2004, 55, 373–399. [Google Scholar] [CrossRef]
- Bhatnagar-Mathur, P.; Vadez, V.; Sharma, K.K. Transgenic approaches for abiotic stress tolerance in plants: Retrospect and prospects. Plant Cell Rep. 2008, 27, 411–424. [Google Scholar] [CrossRef]
- Tripathy, B.C.; Oelmuller, R. Reactive oxygen species generation and signaling in plants. Plant Signal Behav. 2012, 7, 1621–1633. [Google Scholar] [CrossRef]
- Wu, S.W.; Hu, C.X.; Tan, Q.L.; Li, L.; Shi, K.L.; Zheng, Y.; Sun, X.C. Drought stress tolerance mediated by zinc-induced antioxidative defense and osmotic adjustment in cotton (Gossypium Hirsutum). Acta Physiol. Plant 2015, 37, 167. [Google Scholar] [CrossRef]
- Mohammed, A.R.; Tarpley, L. Effects of high night temperature and spikelet position on yield-related parameters of rice (Oryza sativa L.) plants. Eur. J. Agron. 2010, 33, 117–123. [Google Scholar] [CrossRef]
- Greer, D.H.; Weedon, M.M. Modelling photosynthetic responses to temperature of grapevine (Vitis vinifera cv. Semillon) leaves on vines grown in a hot climate. Plant Cell Environ. 2012, 35, 1050–1064. [Google Scholar] [CrossRef]
- Poorter, H.; Niinemets, U.; Ntagkas, N.; Siebenkas, A.; Maenpaa, M.; Matsubara, S.; Pons, T. A meta-analysis of plant responses to light intensity for 70 traits ranging from molecules to whole plant performance. New Phytol. 2019, 223, 1073–1105. [Google Scholar] [CrossRef]
- He, P.; Wright, I.J.; Zhu, S.; Onoda, Y.; Liu, H.; Li, R.; Liu, X.; Hua, L.; Oyanoghafo, O.O.; Ye, Q. Leaf mechanical strength and photosynthetic capacity vary independently across 57 subtropical forest species with contrasting light requirements. New Phytol. 2019, 223, 607–618. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Jiang, W.; Yu, H. Effects of exogenous epibrassinolide on photosynthetic characteristics in tomato (Lycopersicon esculentum Mill) seedlings under weak light stress. J. Agric. Food Chem. 2010, 58, 3642–3645. [Google Scholar] [CrossRef] [PubMed]
- Pospíšil, P.; Šnyrychová, I.; Nauš, J. Dark production of reactive oxygen species in photosystem II membrane particles at elevated temperature: EPR spin-trapping study. Bba-Bioenergetics 2007, 1767, 854–859. [Google Scholar] [CrossRef]
- Wu, H.; Wu, X.; Li, Z.; Duan, L.; Zhang, M. Physiological Evaluation of Drought Stress Tolerance and Recovery in Cauliflower (Brassica oleracea L.) Seedlings Treated with Methyl Jasmonate and Coronatine. J. Plant Growth Regul. 2012, 31, 113–123. [Google Scholar] [CrossRef]
- Li, J.; Guan, Y.; Yuan, L.; Hou, J.; Wang, C.; Liu, F.; Yang, Y.; Lu, Z.; Chen, G.; Zhu, S. Effects of exogenous IAA in regulating photosynthetic capacity, carbohydrate metabolism and yield of Zizania latifolia. Sci. Hortic. 2019, 253, 276–285. [Google Scholar] [CrossRef]
- Zou, M.Q.; Yuan, L.Y.; Zhu, S.D.; Liu, S.; Ge, J.T.; Wang, C.G. Response of osmotic adjustment and ascorbate-glutathione cycle to heat stress in a heat-sensitive and a heat-tolerant genotype of wucai (Brassica campestris L.). Sci. Hortic. 2016, 211, 87–94. [Google Scholar] [CrossRef]
- Walczysko, P.; Wagner, E.; Albrechtova, J.T. Use of coloaded Fluo-3 and Fura Red fluorescent indicators for studying the cytosolic Ca2+ concentrations distribution in living plant tissue. Cell Calcium. 2000, 28, 23–32. [Google Scholar] [CrossRef]
- Sun, Y.D.; Li, Z.X.; Luo, W.R.; Chang, H.C.; Wang, G.Y. Effects of Indole Acetic Acid on Growth and Endogenous Hormone Levels of Cucumber Fruit. Int. J. Agric. Biol. 2018, 20, 197–202. [Google Scholar]
- Strain, H.H.; Svec, W.A. Extraction, Separation, Estimation, and Isolation of the Chlorophylls. Chlorophylls 1966. [Google Scholar] [CrossRef]
- Aebi, H. Catalase in vitro. Methods Enzym. 1984, 105, 121–126. [Google Scholar]
- Heath, R.L.; Packer, L. Photoperoxidation in isolated chloroplasts. I. Kinetics and stoichiometry of fatty acid peroxidation. Arch. Biochem. Biophys. 1968, 125, 189–198. [Google Scholar] [CrossRef]
- Li, J.; Hu, L.; Zhang, L.; Pan, X.; Hu, X. Exogenous spermidine is enhancing tomato tolerance to salinity-alkalinity stress by regulating chloroplast antioxidant system and chlorophyll metabolism. BMC Plant Biol. 2015, 15, 303. [Google Scholar] [CrossRef] [PubMed]
- Bolger, A.M.; Lohse, M.; Usadel, B. Trimmomatic: A flexible trimmer for Illumina sequence data. Bioinformatics 2014, 30, 2114–2120. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.; Langmead, B.; Salzberg, S.L. HISAT: A fast spliced aligner with low memory requirements. Nat. Methods 2015, 12, 357–360. [Google Scholar] [CrossRef] [PubMed]
- Anders, S.; Pyl, P.T.; Huber, W. HTSeq—a Python framework to work with high-throughput sequencing data. Bioinformatics 2015, 31, 166–169. [Google Scholar] [CrossRef] [PubMed]
- Roberts, A.; Pachter, L. Streaming fragment assignment for real-time analysis of sequencing experiments. Nat. Methods 2013, 10, 71–73. [Google Scholar] [CrossRef] [PubMed]
- Pertea, M.; Pertea, G.M.; Antonescu, C.M.; Chang, T.C.; Mendell, J.T.; Salzberg, S.L. StringTie enables improved reconstruction of a transcriptome from RNA-seq reads. Nat. Biotechnol. 2015, 33, 290–295. [Google Scholar] [CrossRef]
- Xu, X.; Pan, S.; Cheng, S.; Zhang, B.; Visser, R.G.F. Genome sequence and analysis of tuber crop potato. Nature 2011, 475, 189–195. [Google Scholar]
- Young, M.D.; Wakefield, M.J.; Smyth, G.K.; Oshlack, A. Gene ontology analysis for RNA-seq: Accounting for selection bias. Genome Biol. 2010, 11. [Google Scholar] [CrossRef]
- Mao, X.; Cai, T.; Olyarchuk, J.G.; Wei, L. Automated genome annotation and pathway identification using the KEGG Orthology (KO) as a controlled vocabulary. Bioinformatics 2005, 21, 3787–3793. [Google Scholar] [CrossRef]
- Chen, G.; Ye, X.; Zhang, S.; Zhu, S.; Yuan, L.; Hou, J.; Wang, C. Comparative Transcriptome Analysis between Fertile and CMS Flower Buds in Wucai (Brassica campestris L.). Bmc Genom. 2018, 19, 908. [Google Scholar] [CrossRef] [PubMed]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef] [PubMed]



| Sample | ZH | ZC | RH | RC |
|---|---|---|---|---|
| H | 18.94 ± 0.62b | 19.17 ± 0.56b | 23.42 ± 0.62a | 18.06 ± 0.31b |
| SD | 10.72 ± 0.51ab | 11.39 ± 0.32a | 10.28 ± 0.25b | 11.31 ± 0.35a |
| RWC | 83.37 ± 0.90ab | 83.59 ± 0.70ab | 82.22 ± 0.78b | 85.11 ± 0.66a |
| Sample | Clean Reads (Million) | Clean Bases (Gigabytes) | Valid Bases (%) | Q30 (%) | GC (%) |
|---|---|---|---|---|---|
| RC1 | 83.61 | 12.53 | 97.93% | 94.30% | 44.00% |
| RC2 | 89.95 | 13.48 | 97.75% | 93.97% | 44.00% |
| RC3 | 67.45 | 10.11 | 96.79% | 93.47% | 44.00% |
| RH1 | 81.78 | 12.26 | 97.63% | 93.77% | 44.00% |
| RH2 | 70.08 | 10.50 | 97.79% | 94.08% | 43.50% |
| RH3 | 92.74 | 13.90 | 97.27% | 93.44% | 44.50% |
| ZH1 | 85.06 | 12.75 | 97.88% | 94.12% | 43.00% |
| ZH2 | 83.99 | 12.59 | 97.81% | 94.06% | 43.00% |
| ZH3 | 84.96 | 12.73 | 97.28% | 93.39% | 43.00% |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hou, J.; Li, J.; Yang, Y.; Wang, Z.; Chang, B.; Yu, X.; Yuan, L.; Wang, C.; Chen, G.; Tang, X.; et al. Physiological and Transcriptomic Analyses Elucidate That Exogenous Calcium Can Relieve Injuries to Potato Plants (Solanum tuberosum L.) under Weak Light. Int. J. Mol. Sci. 2019, 20, 5133. https://doi.org/10.3390/ijms20205133
Hou J, Li J, Yang Y, Wang Z, Chang B, Yu X, Yuan L, Wang C, Chen G, Tang X, et al. Physiological and Transcriptomic Analyses Elucidate That Exogenous Calcium Can Relieve Injuries to Potato Plants (Solanum tuberosum L.) under Weak Light. International Journal of Molecular Sciences. 2019; 20(20):5133. https://doi.org/10.3390/ijms20205133
Chicago/Turabian StyleHou, Jinfeng, Jie Li, Yang Yang, Zixin Wang, Bowen Chang, Xiaowei Yu, Lingyun Yuan, Chenggang Wang, Guohu Chen, Xiaoyan Tang, and et al. 2019. "Physiological and Transcriptomic Analyses Elucidate That Exogenous Calcium Can Relieve Injuries to Potato Plants (Solanum tuberosum L.) under Weak Light" International Journal of Molecular Sciences 20, no. 20: 5133. https://doi.org/10.3390/ijms20205133
APA StyleHou, J., Li, J., Yang, Y., Wang, Z., Chang, B., Yu, X., Yuan, L., Wang, C., Chen, G., Tang, X., & Zhu, S. (2019). Physiological and Transcriptomic Analyses Elucidate That Exogenous Calcium Can Relieve Injuries to Potato Plants (Solanum tuberosum L.) under Weak Light. International Journal of Molecular Sciences, 20(20), 5133. https://doi.org/10.3390/ijms20205133





