Tackling Missing Heritability by Use of an Optimum Curve: A Systematic Review and Meta-Analysis
Abstract
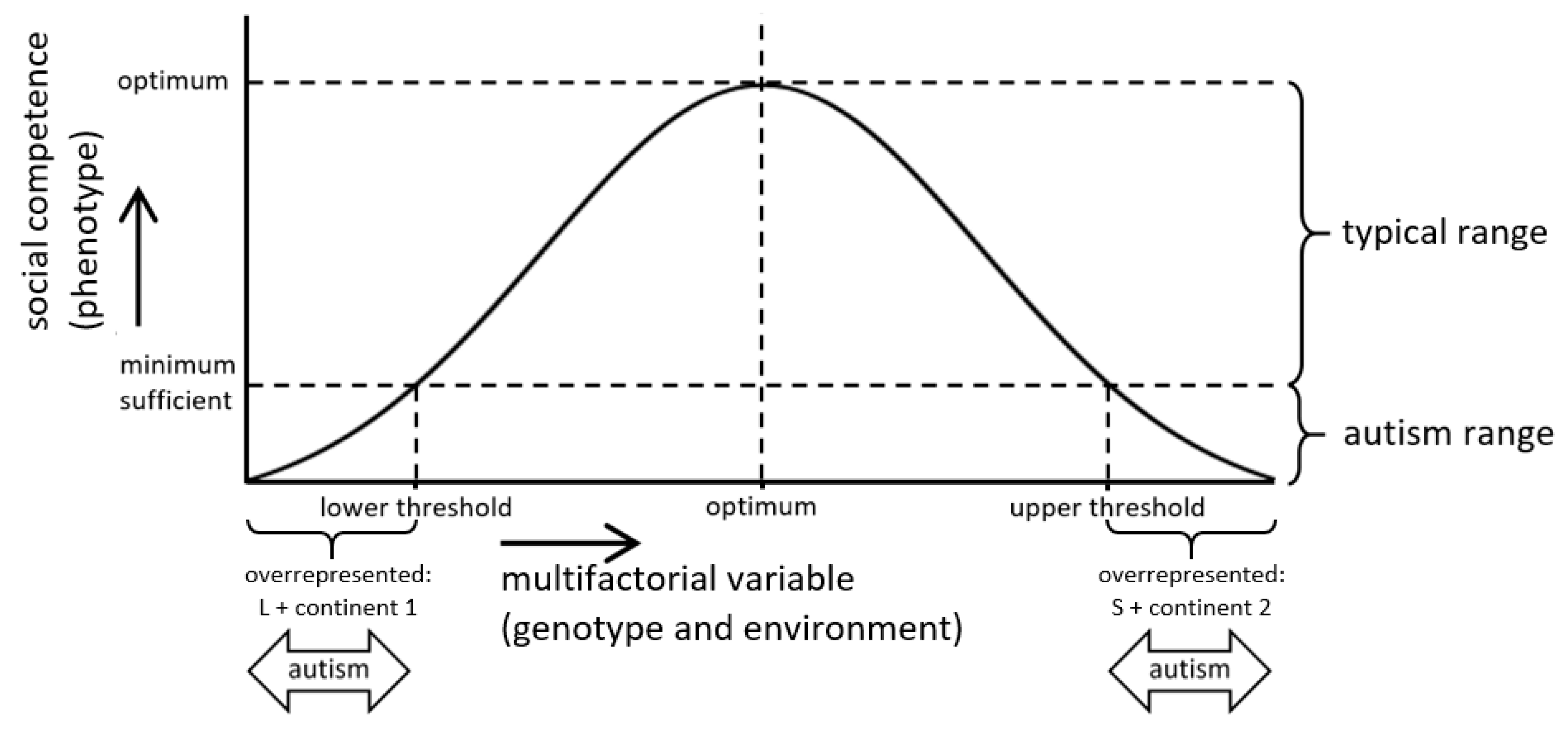
:1. Introduction
2. Results
2.1. Search Results
2.2. Primary Data and Synthesis of the Results
2.3. Across Study Bias
2.4. Sensitivity Analysis
3. Discussion
3.1. Relevance
3.2. Strengths and Limitations
3.3. Suggestions for Future Research
4. Methods
4.1. Search Strategy
4.2. Selection Criteria
4.3. Data Extraction
4.4. Statistical Analysis
4.5. Synthesis of Results
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Mouti, A.; Reddihough, D.; Marraffa, C.; Hazell, P.; Wray, J.; Lee, K.; Kohn, M. Fluoxetine for Autistic Behaviors (FAB trial): Study protocol for a randomized controlled trial in children and adolescents with autism. Trials 2014, 15, 230. [Google Scholar] [CrossRef] [PubMed]
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, 5th ed.; American Psychiatric Publishing: Washington, DC, USA, 2013; p. 50. [Google Scholar]
- Tick, B.; Bolton, P.; Happé, F.; Rutter, M.; Rijsdijk, F. Heritability of autism spectrum disorders: A meta-analysis of twin studies. J. Child Psychol. Psychiatry 2016, 57, 585–595. [Google Scholar] [CrossRef] [PubMed]
- Coleman, M.; Gillberg, C. The Autisms, 4th ed.; Oxford University Press: Oxford, UK, 2012; pp. 151–302. [Google Scholar]
- Geschwind, D.H. Genetics of autism spectrum disorders. Trends Cogn. Sci. 2012, 15, 409–416. [Google Scholar] [CrossRef] [PubMed]
- De la Torre-Ubieta, L.; Won, H.; Stein, J.L.; Geschwind, D.H. Advancing the understanding of autism disease mechanisms through genetics. Nat. Med. 2016, 22, 345–361. [Google Scholar] [CrossRef] [PubMed]
- Maher, B. Personal genomes: The case of the missing heritability. Nature 2008, 456, 18–21. [Google Scholar] [CrossRef]
- Eichler, E.E.; Flint, J.; Gibson, G.; Kong, A.; Leal, S.M.; Moore, J.H.; Nadeau, J.H. Missing heritability and strategies for finding the underlying causes of complex disease. Nat. Rev. Genet. 2010, 11, 446–450. [Google Scholar] [CrossRef] [Green Version]
- Schadt, E.E.; Björkegren, J.L.M. NEW: Network-Enabled Wisdom in biology, medicine, and health care. Sci. Transl. Med. 2012, 4, 115rv1. [Google Scholar] [CrossRef]
- Gabriele, S.; Sacco, R.; Persico, A.M. Blood serotonin levels in autism spectrum disorder: A systematic review and meta-analysis. Eur. Neuropsychopharmacol. 2014, 24, 919–929. [Google Scholar] [CrossRef]
- Veenstra-VanderWeele, J.; Muller, C.L.; Iwamoto, H.; Sauer, J.E.; Owens, W.A.; Shah, C.R.; Cohen, J.; Mannangatti, P.; Jessen, T.; Thompson, B.J.; et al. Autism gene variant causes hyperserotonemia, serotonin receptor hypersensitivity, social impairment and repetitive behavior. Proc. Natl. Acad. Sci. USA 2012, 109, 5469–5474. [Google Scholar] [CrossRef] [Green Version]
- Heils, A.; Teufel, A.; Petri, S.; Stöber, G.; Riederer, P.; Bengel, D.; Lesch, K.P. Allelic variation of human serotonin transporter gene expression. J. Neurochem. 1996, 66, 2621–2624. [Google Scholar] [CrossRef]
- Huang, C.H.; Santangelo, S.L. Autism and serotonin transporter gene polymorphisms: A systematic review and meta-analysis. Am. J. Med. Genet. B 2008, 147B, 903–913. [Google Scholar] [CrossRef] [PubMed]
- Yang, P.Y.; Menga, Y.J.; Li, T.; Huang, Y. Associations of endocrine stress-related gene polymorphisms with risk of autism spectrum disorders: Evidence from an integrated meta-analysis. Autism Res. 2017, 10, 1722–1736. [Google Scholar] [CrossRef] [PubMed]
- Garbarino, V.R.; Gilmana, T.L.; Dawsa, L.C.; Gould, G.G. Extreme enhancement or depletion of serotonin transporter function and serotonin availability in autism spectrum disorder. Pharmacol. Res. 2012, 140, 85–99. [Google Scholar] [CrossRef] [PubMed]
- Cook, E.H.; Courchesne, R.; Lord, C.; Cox, N.J.; Yan, S.; Lincoln, A.; Haas, R.; Courchesne, E.; Leventhal, B.L. Evidence of linkage between the serotonin transporter and autistic disorder. Mol. Psychiatry 1997, 2, 247–250. [Google Scholar] [CrossRef] [Green Version]
- Persico, A.M.; Militerni, R.; Bravaccio, C.; Schneider, C.; Melmed, R.; Conciatori, M.; Damiani, V.; Baldi, A.; Keller, F. Lack of association between serotonin transporter gene promoter variants and autistic disorder in two ethnically distinct samples. Am. J. Med. Genet. 2000, 96, 123–127. [Google Scholar] [CrossRef]
- Kim, S.J.; Cox, N.; Courchesne, R.; Lord, C.; Corsello, C.; Akshoomoff, N.; Guter, S.; Leventhal, B.L.; Courchesne, E.; Cook, E.H., Jr. Transmission disequilibrium mapping at the serotonin transporter gene (SLC6A4) region in autistic disorder. Mol. Psychiatry 2002, 7, 278–288. [Google Scholar] [CrossRef]
- McCauley, J.L.; Olson, L.M.; Dowd, M.; Amin, T.; Steele, A.; Blakely, R.D.; Folstein, S.E.; Haines, J.L.; Sutcliffe, J.S. Linkage and association analysis at the serotonin transporter (SLC6A4) locus in a rigid-compulsive subset of autism. Am. J. Med. Genet. B 2004, 127B, 104–112. [Google Scholar] [CrossRef]
- Devlin, B.; Cook, E.H., Jr.; Coon, H.; Dawson, G.; Grigorenko, E.L.; McMahon, W.; Minshew, N.; Pauls, D.; Smith, M.; Spence, M.A.; et al. Autism and the serotonin transporter: The long and short of it. Mol. Psychiatry 2005, 10, 1110–1116. [Google Scholar] [CrossRef]
- Klauck, S.M.; Poustka, F.; Benner, A.; Lesch, K.P.; Poustka, A. Serotonin transporter (5-HTT) gene variants associated with autism? Hum. Mol. Genet. 1997, 6, 2233–2238. [Google Scholar] [CrossRef] [Green Version]
- Tordjman, S.; Gutknecht, L.; Carlier, M.; Spitz, E.; Antoine, C.; Slama, F.; Carsalade, V.; Cohen, D.J.; Ferrari, P.; Roubertoux, P.L.; et al. Role of the serotonin transporter gene in the behavioral expression of autism. Mol. Psychiatry 2001, 6, 434–439. [Google Scholar] [CrossRef] [Green Version]
- Coutinho, A.M.; Oliveira, G.; Morgadinho, T.; Fesel, C.; Macedo, T.R.; Bento, C.; Marques, C.; Ataíde, A.; Miguel, T.; Borges, L. Variants of the serotonin transporter gene (SLC6A4) significantly contribute to hyperserotonemia in autism. Mol. Psychiatry 2004, 9, 264–271. [Google Scholar] [CrossRef] [PubMed]
- Conroy, J.; Meally, E.; Kearney, G.; Fitzgerald, M.; Gill, M.; Gallagher, L. Serotonin transporter gene and autism: A haplotype analysis in an Irish autistic population. Mol. Psychiatry 2004, 9, 587–593. [Google Scholar] [CrossRef] [PubMed]
- Mulder, E.J.; Anderson, G.M.; Kema, I.P.; Brugman, A.M.; Ketelaars, C.E.; de Bildt, A.; van Lang, N.D.; den Boer, J.A.; Minderaa, R.B. Serotonin transporter intron 2 polymorphism associated with rigid-compulsive behaviors in Dutch individuals with pervasive developmental disorder. Am. J. Med. Genet. B 2005, 133B, 93–96. [Google Scholar] [CrossRef] [PubMed]
- Guerini, F.R.; Manca, S.; Sotgiu, S.; Tremolada, S.; Zanzottera, M.; Agliardi, C.; Zanetta, L.; Saresella, M.; Mancuso, R.; De Silvestri, A.; et al. A family based linkage analysis of HLA and 5-HTTLPR gene polymorphisms in Sardinian children with autism spectrum disorder. Hum. Immunol. 2006, 67, 108–117. [Google Scholar] [CrossRef]
- Yirmiya, N.; Pilowsky, T.; Nemanov, L.; Arbelle, S.; Feinsilver, T.; Fried, I.; Ebstein, R.P. Evidence for an association with the serotonin transporter promoter region polymorphism and autism. Am. J. Med. Genet. 2001, 105, 381–386. [Google Scholar] [CrossRef]
- Guhathakurta, S.; Ghosh, S.; Sinha, S.; Chatterjee, A.; Ahmed, S.; Chowdhury, S.R.; Gangopadhyay, P.K.; Ghosh, S.; Singh, M.; Usha, R. Serotonin transporter promoter variants: Analysis in Indian autistic and control population. Brain Res. 2006, 1092, 28–35. [Google Scholar] [CrossRef]
- Koishi, S.; Yamamoto, K.; Matsumoto, H.; Koishi, S.; Enseki, Y.; Oya, A.; Asakura, A.; Aoki, Y.; Atsumi, M.; Iga, T.; et al. Serotonin transporter gene promoter polymorphism and autism: A family-based genetic association study in Japanese population. Brain Dev. 2006, 28, 257–260. [Google Scholar] [CrossRef]
- Cho, I.H.; Yoo, H.J.; Park, M.; Lee, Y.S.; Kim, S.A. Family-based association study of 5-HTTLPR and the 5-HT2A receptor gene polymorphisms with autism spectrum disorder in Korean trios. Brain Res. 2007, 1139, 34–41. [Google Scholar] [CrossRef]
- Maestrini, E.; Lai, C.; Marlow, A.; Matthews, N.; Wallace, S.; Bailey, A.; Cook, E.H.; Weeks, D.E.; Monaco, A.P. Serotonin transporter (5-HTT) and gamma-aminobutyric acid receptor subunit beta3 (GABRB3) gene polymorphisms are not associated with autism in the IMGSA families. The International Molecular Genetic Study of Autism Consortium. Am. J. Med. Genet. 1999, 88, 492–496. [Google Scholar] [CrossRef]
- Betancur, C.; Corbex, M.; Spielewoy, C.; Philippe, A.; Laplanche, J.L.; Launay, J.M.; Gillberg, C.; Mouren-Siméoni, M.C.; Hamon, M.; Giros, B. Serotonin transporter gene polymorphisms and hyperserotonemia in autistic disorder. Mol. Psychiatry 2002, 7, 67–71. [Google Scholar] [CrossRef]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G. Preferred Reporting Items for Systematic Reviews and Meta-Analyses: The PRISMA statement. PLoS Med. 2009, 6, e1000097. [Google Scholar] [CrossRef] [PubMed]
- Begg, C.B.; Mazumdar, M. Operating characteristics of a rank correlation test for publication bias. Biometrics 1994, 50, 1088–1101. [Google Scholar] [CrossRef] [PubMed]
- Egger, M.; Davey Smith, G.; Schneider, M.; Minder, C. Bias in meta-analysis detected by a simple, graphical test. BMJ 1997, 315, 629–634. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Madden, J.M.; Lakoma, M.D.; Lynch, F.L.; Rusinak, D.; Owen-Smith, A.A.; Coleman, K.J.; Quinn, V.P.; Yau, V.M.; Qian, Y.X.; Croen, L.A. Psychotropic medication use among insured children with autism spectrum disorder. J. Autism Dev. Disord. 2017, 47, 144–154. [Google Scholar] [CrossRef] [PubMed]
- Mann, J.J.; Emslie, G.; Baldessarini, R.J.; Beardslee, W.; Fawcett, J.A.; Goodwin, F.K.; Leon, A.C.; Meltzer, H.Y.; Ryan, N.D.; Shaffer, D.; et al. ACNP Task Force report on SSRIs and suicidal behavior in youth. Neuropsychopharmacology 2006, 31, 473–492. [Google Scholar] [CrossRef] [PubMed]
- Porcelli, S.; Fabbri, C.; Serretti, A. Meta-analysis of serotonin transporter gene promoter polymorphism (5-HTTLPR) association with antidepressant efficacy. Eur. Neuropsychopharmacol. 2012, 22, 239–258. [Google Scholar] [CrossRef] [PubMed]
- Hu, X.Z.; Lipsky, R.H.; Zhu, G.; Akhtar, L.A.; Taubman, J.; Greenberg, B.D.; Xu, K.; Arnold, P.D.; Richter, M.A.; Kennedy, J.L.; et al. Serotonin transporter promoter gain-of-function genotypes are linked to obsessive-compulsive disorder. Am. J. Hum. Genet. 2006, 78, 815–826. [Google Scholar] [CrossRef]
- Spielman, R.S.; McGinnis, R.E.; Ewens, W.J. Transmission test for linkage disequilibrium: The insulin gene region and insulin-dependent diabetes mellitus (IDDM). Am. J. Hum. Genet. 1993, 52, 506–516. [Google Scholar]
- Chang, B.H.; Hoaglin, D.C. Meta-analysis of odds ratios: Current good practices. Med. Care 2017, 55, 328–335. [Google Scholar] [CrossRef]
- Cochran, W.G. The combination of estimates from different experiments. Biometrics 1954, 10, 101–129. [Google Scholar] [CrossRef]
- Higgins, J.P.; Thompson, S.G.; Deeks, J.J.; Altman, D.G. Measuring inconsistency in meta-analyses. BMJ 2003, 327, 557–560. [Google Scholar] [CrossRef] [PubMed] [Green Version]
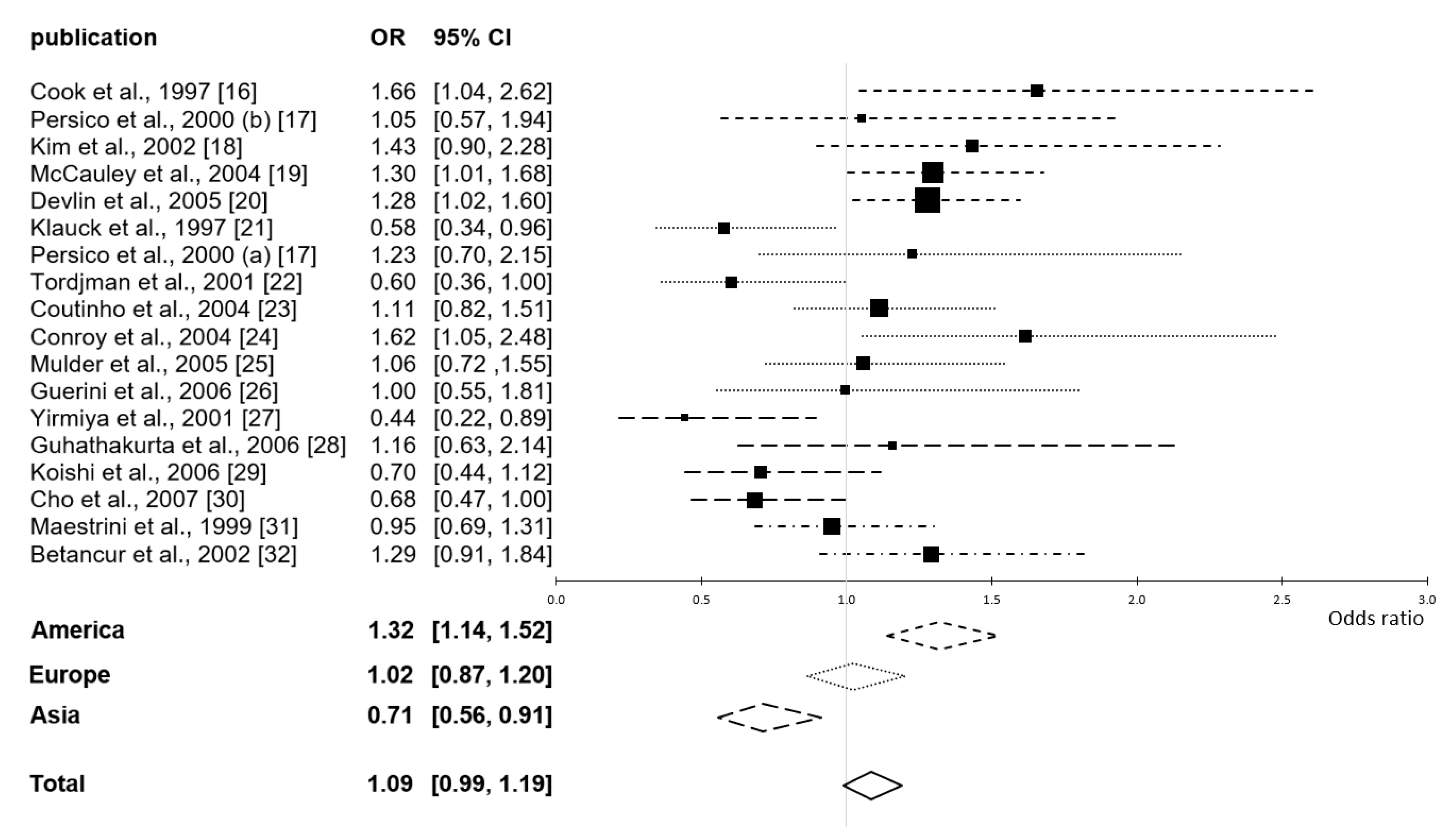
| Publication | Origin of Sample | Samples | T: S NT: L | T: L NT: S | Pref. | p |
|---|---|---|---|---|---|---|
| AMERICA | ||||||
| Cook et al., 1997 [16] | United States | 86 trios | 48 | 29 | S | 0.031 |
| Persico et al., 2000 (sample b) [17] | United States | 32 trios 5 multiplex | 21 | 20 | – | 0.88 |
| Kim et al., 2002 [18] | United States | 115 trios | 43 | 30 | – | 0.13 |
| McCauley et al., 2004 [19] | United States | 123 multiplex | 135 | 104 | S | 0.045 |
| Devlin et al., 2005 [20] | United States | 103 trios 125 multiplex | 175 | 137 | S | 0.031 |
| EUROPE | ||||||
| Klauck et al., 1997 [21] | Germany and Austria | 65 trios | 23 | 40 | L | 0.032 |
| Persico et al., 2000 (sample a) [17] | Italy | 46 trios 8 duos | 27 | 22 | – | 0.48 |
| Tordjman et al., 2001 [22] | France | 42 trios 2 multiplex | 24 | 40 | L | 0.046 |
| Coutinho et al., 2004 [23] | Portugal | 182 families | 88 | 79 | – | 0.49 |
| Conroy et al., 2004 [24] | Ireland | 84 trios | 55 | 34 | S | 0.026 |
| Mulder et al., 2005 [25] | The Netherlands | 111 trios 3 duos 3 multiplex | 54 | 51 | – | 0.77 |
| Guerini et al., 2006 [26] | Italy | 34 singleton 3 multiplex | 22 | 22 | – | 1.0 |
| ASIA | ||||||
| Yirmiya et al., 2001 [27] | Israel | 33 families | 11 | 25 | L | 0.020 |
| Guhathakurta et al., 2006 [28] | India | 61 trios 18 duos | 22 | 19 | – | 0.64 |
| Koishi et al., 2006 [29] | Japan | 104 trios | 31 | 44 | – | 0.13 |
| Cho et al., 2007 [30] | Korea | 126 trios | 45 | 66 | L | 0.046 |
| MIXED | ||||||
| Maestrini et al., 1999 [31] | America and Europe | 8 singleton 82 multiplex | 72 | 76 | – | 0.74 |
| Betancur et al., 2002 [32] | America and Europe | 43 trios 53 multiplex | 71 | 55 | – | 0.15 |
| total American | 589 families | S | <0.001 | |||
| total European | 588 families | – | 0.81 | |||
| total Asian | 342 families | L | 0.007 | |||
| total mixed American/European | 186 families | |||||
| total overall | 1705 families | – | 0.079 | |||
| Samples | Q | p | I2 | Conclusion |
|---|---|---|---|---|
| American | 1.68 | 0.79 | 0.00 | homogeneous |
| European | 14.27 | 0.027 | 0.58 | heterogeneous |
| Asian | 4.23 | 0.24 | 0.29 | homogeneous |
| All | 40.39 | 0.001 | 0.58 | heterogeneous |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wegener Sleeswijk, A.; Heijungs, R.; Durston, S. Tackling Missing Heritability by Use of an Optimum Curve: A Systematic Review and Meta-Analysis. Int. J. Mol. Sci. 2019, 20, 5104. https://doi.org/10.3390/ijms20205104
Wegener Sleeswijk A, Heijungs R, Durston S. Tackling Missing Heritability by Use of an Optimum Curve: A Systematic Review and Meta-Analysis. International Journal of Molecular Sciences. 2019; 20(20):5104. https://doi.org/10.3390/ijms20205104
Chicago/Turabian StyleWegener Sleeswijk, Anneke, Reinout Heijungs, and Sarah Durston. 2019. "Tackling Missing Heritability by Use of an Optimum Curve: A Systematic Review and Meta-Analysis" International Journal of Molecular Sciences 20, no. 20: 5104. https://doi.org/10.3390/ijms20205104
APA StyleWegener Sleeswijk, A., Heijungs, R., & Durston, S. (2019). Tackling Missing Heritability by Use of an Optimum Curve: A Systematic Review and Meta-Analysis. International Journal of Molecular Sciences, 20(20), 5104. https://doi.org/10.3390/ijms20205104





