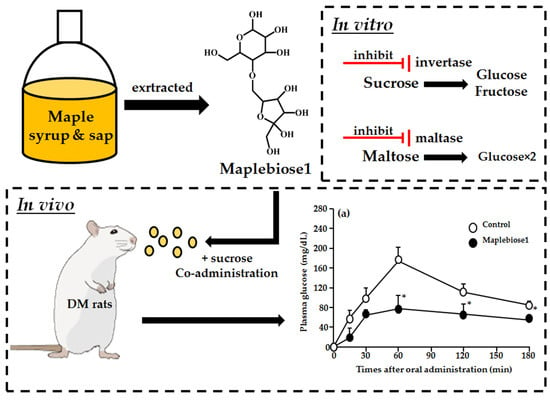
Identification of a Novel Oligosaccharide in Maple Syrup as a Potential Alternative Saccharide for Diabetes Mellitus Patients
Abstract
:1. Introduction
2. Results
2.1. Profile and Characteristics of the Saccharides in Maple Syrup
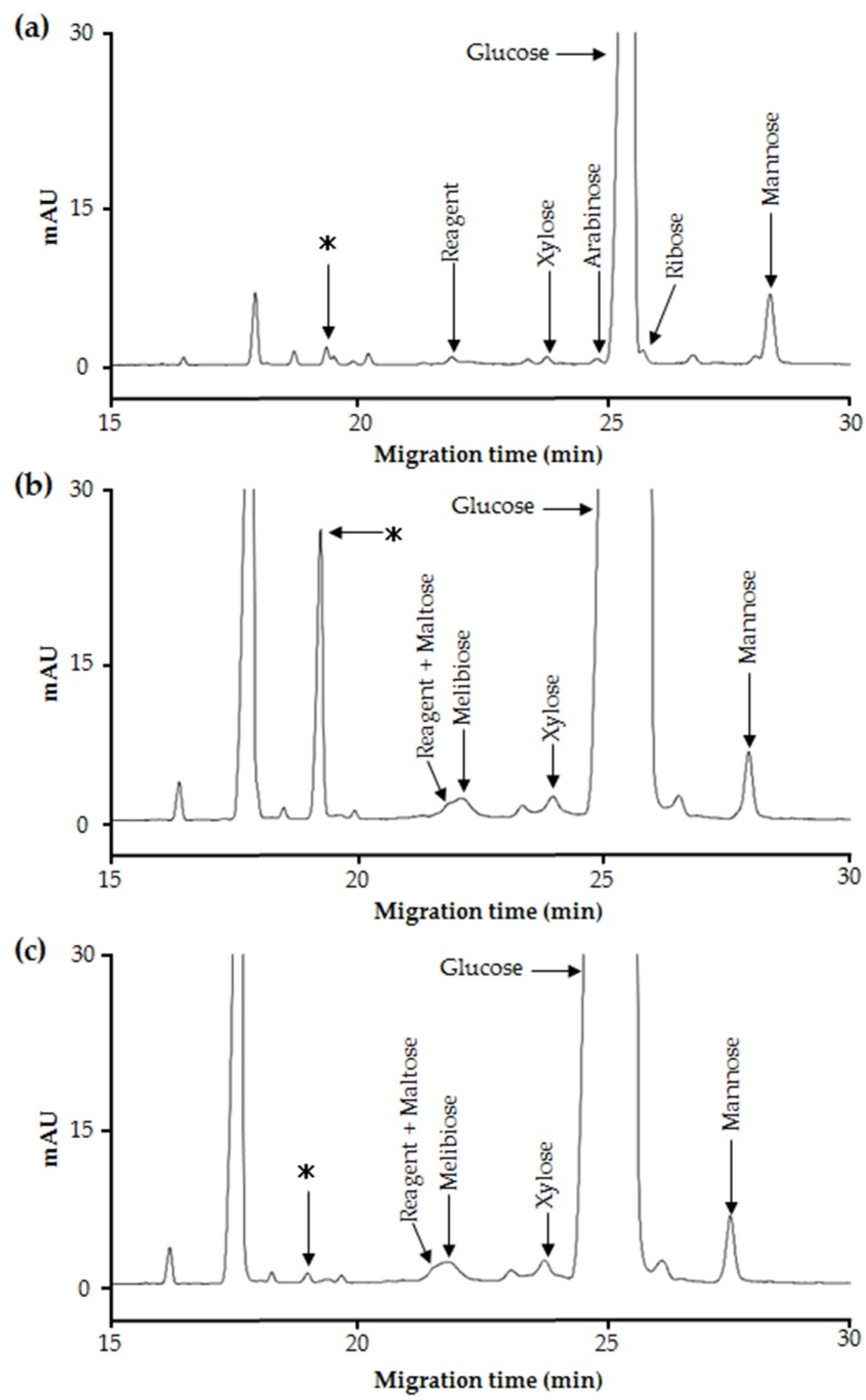
2.2. Purification and Structural Analysis of the Unidentified Oligosaccharide in Maple Sap
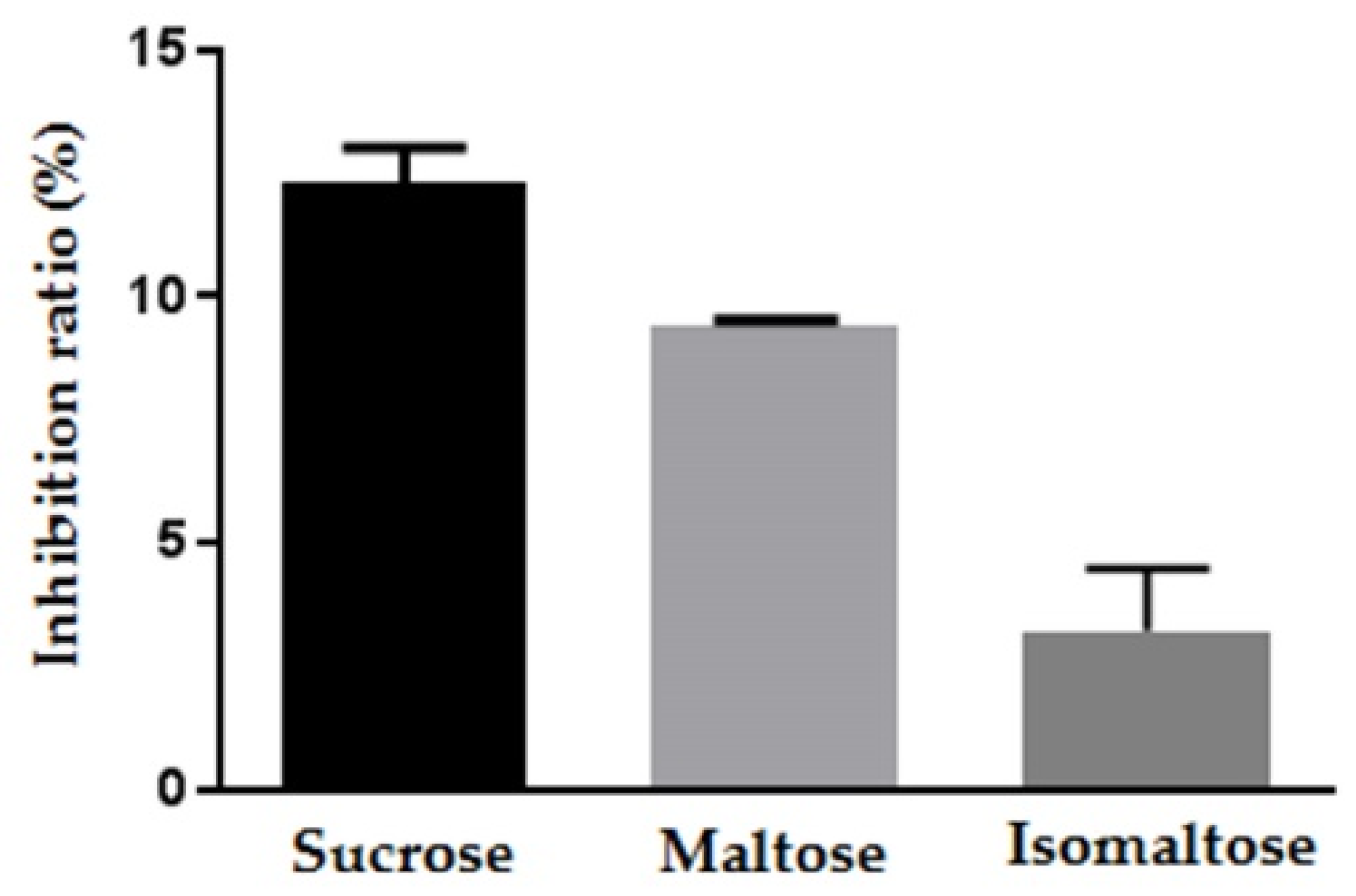
2.3. The Inhibitory Effect of Maplebiose1 on Enzymatic Glycolysis
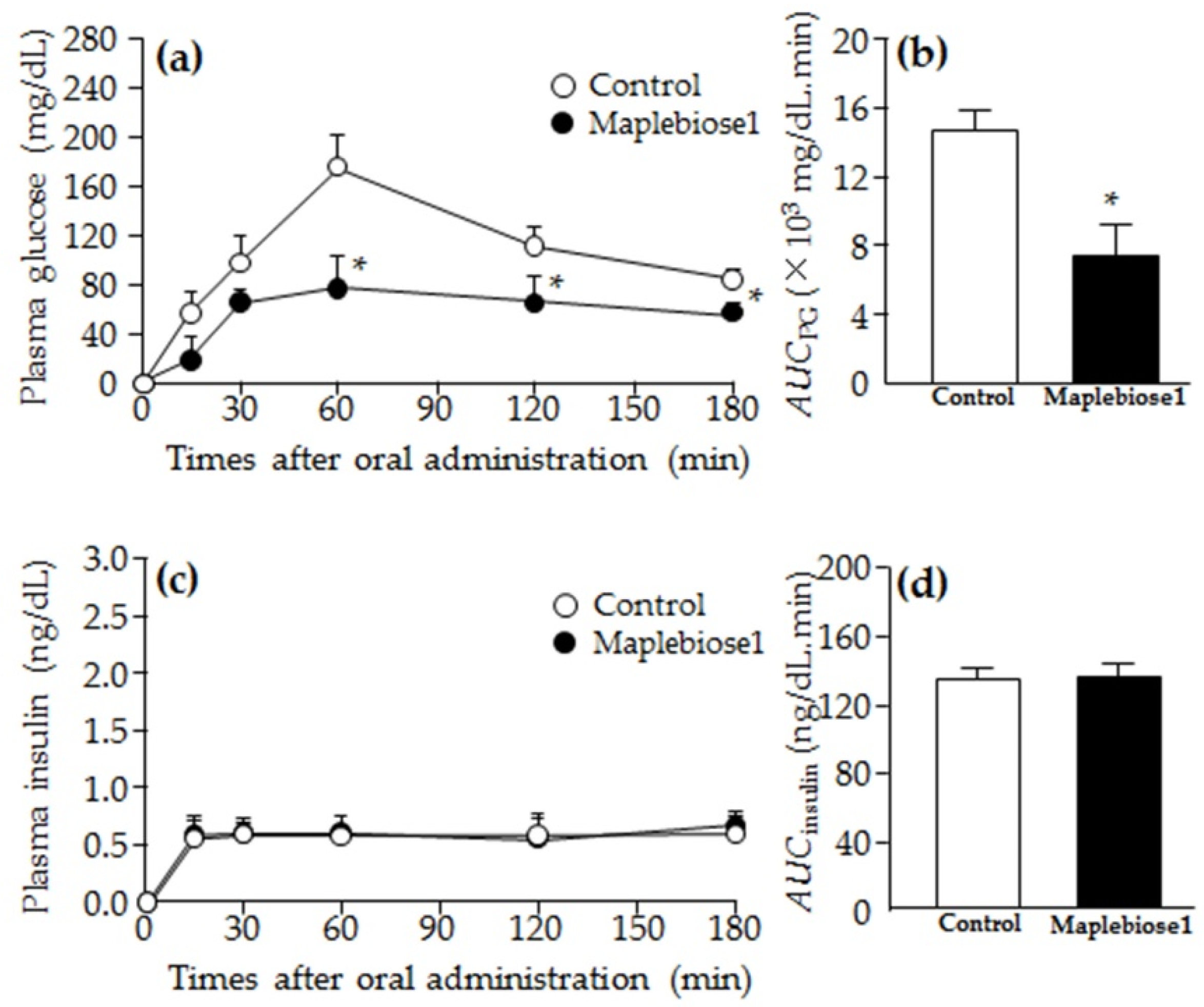
2.4. Prevention of the Hyperglycemia Induced in Rats Following Oral Administration of a Sucrose Load by Maplebiose1
3. Discussion
4. Material and Methods
4.1. Materials and Animals
4.2. PMP Derivatization of Maple Syrup and Sap
4.3. Capillary Electrophoresis (CE)
4.4. Invertase Digestion of Maple Syrup
4.5. Size Exclusion Chromatography
4.6. High-Performance Liquid Chromatography with Charged Aerosol Detection (HPLC-CAD)
4.7. Hydrolysis of the Unidentified Oligosaccharide
4.8. Liquid Chromatography (LC)-Electrospray Ionization (ESI)-Mass Spectrometry (MS)
4.9. Nuclear Magnetic Resonance (NMR)
4.10. Invertase Inhibition Assay
4.11. Glycosidase Inhibition Assay
4.12. Biochemical Assays Conducted on Blood Samples from Rats
4.13. Oral Sucrose Tolerance Testing
4.14. Statistical Analysis
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| BGE | Background electrolyte |
| CAD | Charged aerosol detection |
| CE | Capillary electrophoresis |
| CID | Collision introduced dissolution |
| DM | Diabetes mellitus |
| HMBC | 1H-13C heteronuclear multiple-bond correlation spectroscopy |
| HPLC | High-performance liquid chromatography |
| IC50 | Half-maximal inhibitory concentration |
| LC-ESI-MS | Liquid chromatography-electrospray ionization-mass spectrometry |
| NMR | Nuclear magnetic resonance |
| PMP | 1-phenyl-3-methyl-5-pyrazolone |
| UV | Ultraviolet |
References
- Haffner, S.M.; Valdez, R.A.; Hazuda, H.P.; Mitchell, B.D.; Morales, P.A.; Stern, M.P. Prospective analysis of the insulin-resistance syndrome (syndrome X). Diabetes 1992, 41, 715–722. [Google Scholar] [CrossRef] [PubMed]
- Isomaa, B.; Almgren, P.; Tuomi, T.; Forsén, B.; Lahti, K.; Nissén, M.; Taskinen, M.R.; Groop, L. Cardiovascular morbidity and mortality associated with the metabolic syndrome. Diabetes Care 2001, 24, 683–689. [Google Scholar] [CrossRef] [PubMed]
- Suez, J.; Korem, T.; Zeevi, D.; Zilberman-Schapira, G.; Thaiss, C.A.; Maza, O.; Israeli, D.; Zmora, N.; Gilad, S.; Weinberger, A.; et al. Artificial sweeteners induce glucose intolerance by altering the gut microbiota. Nature 2014, 514, 181–186. [Google Scholar] [CrossRef] [PubMed]
- Ball, D.W. The Chemical Composition of Maple Syrup. J. Chem. Educ. 2009, 84, 1647. [Google Scholar] [CrossRef]
- Stinson, E.E.; Dooley, C.J.; Purcell, J.M.; Ard, J.S. Quebrachitol—A New Component of Maple Sap and Sirup. J. Agric. Food Chem. 1967, 15, 394–397. [Google Scholar] [CrossRef]
- Stuckel, J.G.; Low, N.H. The chemical composition of 80 pure maple syrup samples produced in North America. Food Res. Int. 1996, 29, 373–379. [Google Scholar] [CrossRef]
- Yamamoto, T.; Uemura, K.; Moriyama, K.; Mitamura, K.; Taga, A. Inhibitory effect of maple syrup on the cell growth and invasion of human colorectal cancer cells. Oncol. Rep. 2015, 33, 1579–1584. [Google Scholar] [CrossRef]
- Yamamoto, T.; Sato, K.; Kubota, Y.; Mitamura, K.; Taga, A. Effect of dark-colored maple syrup on cell proliferation of human gastrointestinal cancer cell. Biomed. Rep. 2017, 7, 6–10. [Google Scholar] [CrossRef] [Green Version]
- Taga, A.; Kodama, S. Analysis of reducing carbohydrates and fructosyl saccharides in maple syrup and maple sugar by CE. Chromatographia 2012, 75, 1009–1016. [Google Scholar] [CrossRef]
- Xiao, L.; Van’t Land, B.; Engen, P.A.; Naqib, A.; Green, S.J.; Nato, A.; Leusink-Muis, T.; Garssen, J.; Keshavarzian, A.; Stahl, B.; et al. Human milk oligosaccharides protect against the development of autoimmune diabetes in NOD-mice. Sci. Rep. 2018, 8, 3829. [Google Scholar] [CrossRef]
- Zheng, J.; Yuan, X.; Cheng, G.; Jiao, S.; Feng, C.; Zhao, X.; Yin, H.; Du, Y.; Liu, H. Chitosan oligosaccharides improve the disturbance in glucose metabolism and reverse the dysbiosis of gut microbiota in diabetic mice. Carbohydr. Polym. 2018, 190, 77–86. [Google Scholar] [CrossRef] [PubMed]
- Pitt, J.J.; Gorman, J.J. Oligosaccharide characterization and quantitation using 1-phenyl-3- methyl-5-pyrazolone derivatization and matrix-assisted laser desorption/ionization time-of-flight mass spectrometry. Anal. Biochem. 1997, 248, 63–75. [Google Scholar] [CrossRef] [PubMed]
- Taga, A.; Uegaki, K.; Yabusako, Y.; Kitano, A.; Honda, S. Simultaneous determination of the association constants of oligosaccharides to a lectin by capillary electrophoresis. J. Chromatogr. A. 1999, 837, 221–229. [Google Scholar] [CrossRef]
- Homann, A.; Biedendieck, R.; Götze, S.; Jahn, D.; Seibel, J. Insights into polymer versus oligosaccharide synthesis: Mutagenesis and mechanistic studies of a novel levansucrase from Bacillus megaterium. Biochem. J. 2007, 407, 189–198. [Google Scholar] [CrossRef]
- Kawano, K.; Hirashima, T.; Mori, S.; Saitoh, Y.; Kurosumi, M.; Natori, T. Spontaneous long-term hyperglycemic rat with diabetic complications: Otsuka Long-Evans Tokushima Fatty (OLETF) strain. Diabetes 1992, 41, 1422–1428. [Google Scholar] [CrossRef] [PubMed]
- Meng, G.; Fütterer, K. Structural framework of fructosyl transfer in Bacillus subtilis levansucrase. Nat. Struct. Biol. 2003, 10, 935–941. [Google Scholar] [CrossRef]
- Raga-Carbajal, E.; López-Munguía, A.; Alvarez, L.; Olvera, C. Understanding the transfer reaction network behind the non-processive synthesis of low molecular weight levan catalyzed by Bacillus subtilis levansucrase. Sci. Rep. 2018, 8, 15035. [Google Scholar] [CrossRef]
- Gupta, S.; Mahmood, S.; Khan, R.H.; Mahmood, A. Inhibition of brush border sucrase by polyphenols in mouse intestine. Biosci. Rep. 2009, 30, 111–117. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pyner, A.; Nyambe-Silavwe, H.; Williamson, G. Inhibition of human and rat sucrase and maltase activities to assess antiglycemic potential: Optimization of the assay using acarbose and polyphenols. J. Agric. Food Chem. 2017, 65, 8643–8651. [Google Scholar] [CrossRef]
- Williamson, G. Possible effects of dietary polyphenols on sugar absorption and digestion. Mol. Nutr. Food Res. 2013, 57, 48–57. [Google Scholar] [CrossRef]
- Jaynes, T.A.; Nelson, O.E. An Invertase Inactivator in Maize Endosperm and Factors Affecting Inactivation. Plant Physiol. 2008, 47, 629–634. [Google Scholar] [CrossRef]
- Pressey, R. Separation and properties of potato invertase and invertase inhibitor. Arch. Biochem. Biophys. 1966, 113, 667–674. [Google Scholar] [CrossRef]
- Pressey, R. Invertase Inhibitor from Potatoes: Purification, Characterization, and Reactivity with Plant Invertases. Plant. Physiol. 2008, 42, 1780–1786. [Google Scholar] [CrossRef]
- Hauri, H.-P.; Quaroni, A.; Isselbacher, K.J. Biogenesis of intestinal plasma membrane: Posttranslational route and cleavage of sucrase--isomaltase. Proc. Natl. Acad. Sci. USA 1979, 76, 5183–5186. [Google Scholar] [CrossRef] [PubMed]
- Shapiro, G.L.; Bulow, S.D.; Conklin, K.A.; Scheving, L.A.; Gray, G.M. Postinsertional processing of sucrase-alpha-dextrinase precursor to authentic subunits: Multiple step cleavage by trypsin. Am. J. Physiol. Liver Physiol. 2017, 261, G847–G857. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, M.; Shindo, D.; Suzuki, R.; Shirataki, Y.; Waki, H. Combined long-term caffeine intake and exercise inhibits the development of diabetic nephropathy in OLETF rats. J. Appl. Physiol. 2017, 122, 1321–1328. [Google Scholar] [CrossRef] [PubMed]
- Nagai, N.; Ito, Y.; Taga, A. Comparison of the Enhancement of Plasma Glucose Levels in Type 2 Diabetes Otsuka Long-Evans Tokushima Fatty Rats by Oral Administration of Sucrose or Maple Syrup. J. Oleo Sci. 2013, 62, 737–743. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nagai, N.; Yamamoto, T.; Tanabe, W.; Ito, Y.; Kurabuchi, S.; Mitamura, K.; Taga, A. Changes in Plasma Glucose in Otsuka Long-Evans Tokushima Fatty Rats After Oral Administration of Maple Syrup. J. Oleo Sci. 2015, 64, 331–335. [Google Scholar] [CrossRef] [Green Version]
- Yabuki, A.; Tahara, T.; Taniguchi, K.; Matsumoto, M.; Suzuki, S. Neuronal Nitric Oxide Synthase and Cyclooxygenase-2 in Diabetic Nephropathy of Type 2 Diabetic OLETF Rats. Exp. Anim. 2006, 55, 17–25. [Google Scholar] [CrossRef] [Green Version]
- Honda, S.; Akao, E.; Suzuki, S.; Okuda, M.; Kakehi, K.; Nakamura, J. High-performance liquid chromatography of reducing carbohydrates as strongly ultraviolet-absorbing and electrochemically sensitive 1-phenyl-3-methyl5-pyrazolone derivatives. Anal. Biochem. 1989, 180, 351–357. [Google Scholar] [CrossRef]
- Sharma, M.L.; Newbrun, E. Continuous spectrophotometric assay of glucosyltransferase and β-fructofuranosidase activity. Carbohydr. Res. 1973, 29, 165–172. [Google Scholar] [CrossRef]
- Deguchi, Y.; Osada, K.; Uchida, K.; Kimura, H.; Yoshikawa, M.; Kudo, T.; Yasui, H.; Watanuki, M. Effects of extract of guava leaves on the development of diabetes in the db/db mouse and on the postprandial blood glucose of human subjects. J. Agric. Chem. Soc. Japan 1998, 72, 923–931. [Google Scholar]

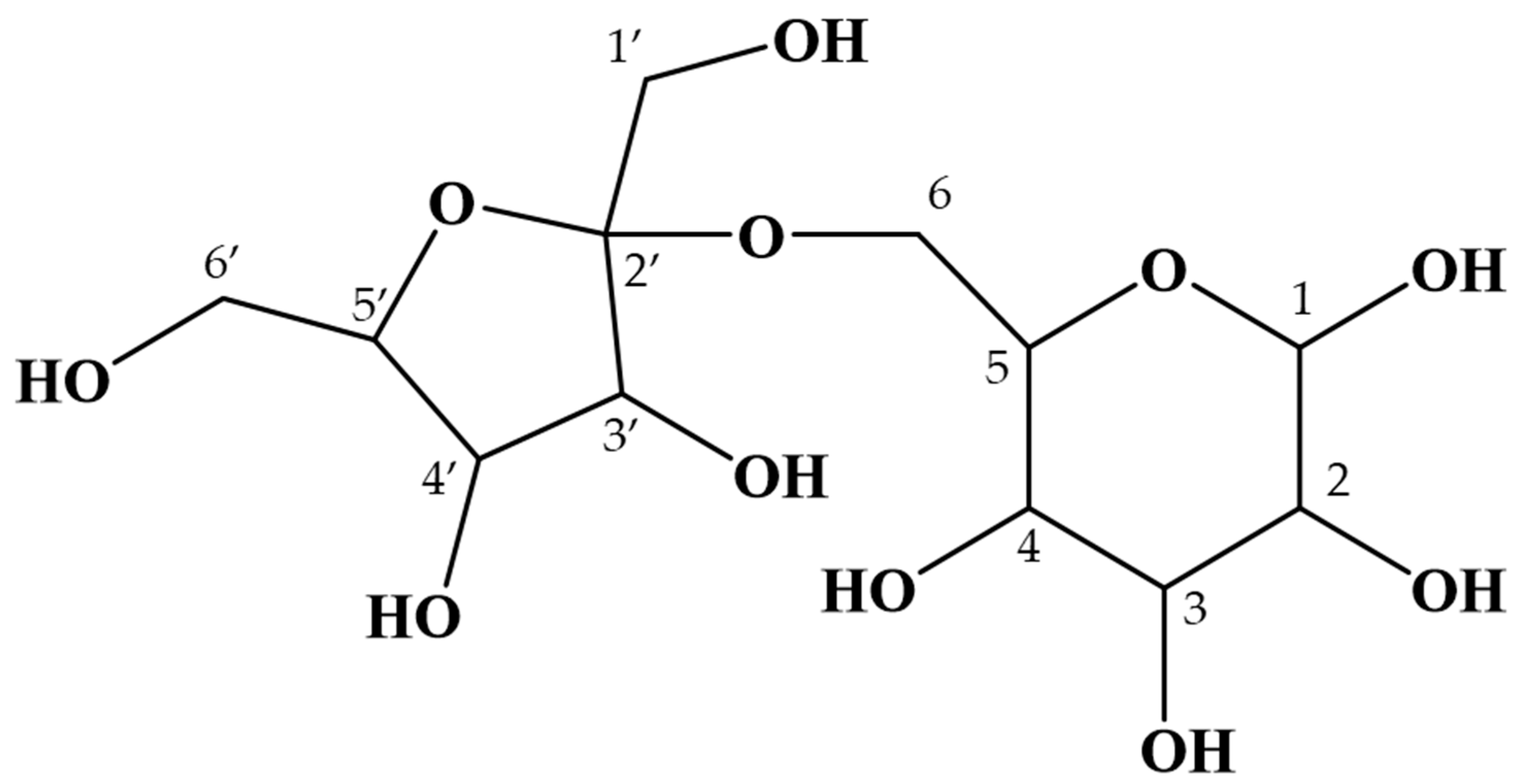
| Chemical Shift | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Residue | Position | δC | δH | JH,H | Type | Residue | Position | δC | δH | JH,H | Type |
| Glc α | 1 | 94.72 | 5.07 | 3.8 | d | Glc β | 1 | 98.57 | 4.49 | 8.0 | d |
| 2 | 74.05 | 3.38 | 3.8, 9.8 | dd | 2 | 76.67 | 3.09 | 8.0, 9.3 | dd | ||
| 3 | 75.25 | 3.54 | - | m | 3 | 78.23 | 3.33 | 9.3, 9.3 | dd | ||
| 4 | 72.32 | 3.29 | 9.0, 9.0 | dd | 4 | 72.25 | 3.29 | 9.0, 9.0 | dd | ||
| 5 | 73.27 | 3.77 | 1.9, 9.2, 9.7 | ddd | 5 | 77.58 | 3.40 | 2.1, 8.7, 9.7 | ddd | ||
| 6 | 63.38 | 3.82 | 2.1, 10.9 | dd | 6 | 63.38 | 3.87 | 2.0, 11.0 | dd | ||
| Fru β | 1’ | 62.75 | 3.60 | - | m | Fru β | 1’ | 62.72 | 3.54 | - | m |
| 2’ | 106.31 | - | - | - | 2’ | 106.34 | - | - | - | ||
| 3’ | 79.42 | 4.03 | 8.5 | d | 3’ | 79.52 | 4.02 | 8.5 | d | ||
| 4’ | 77.06 | 3.97 | 8.1, 8.2 | dd | 4’ | 77.21 | 3.96 | 8.0, 8.3 | dd | ||
| 5’ | 83.75 | 3.72 | 7.4 | t | 5’ | 83.81 | 3.72 | 7.4 | t | ||
| 6’ | 64.98 | 3.54–3.66 | - | m | 6’ | 65.10 | 3.54–3.66 | - | m | ||
| Glycolytic Enzyme | Inhibitor (µg) | Peak Response (D2) | Peak Response (D3) | Inhibition Rate (%) | IC50 (mmol/L) |
|---|---|---|---|---|---|
| Invertase | - | 30.5 | - | - | 1.17 |
| 1 | 18.2 | N.D | 40.3 | ||
| 10 | 17.2 | N.D | 43.6 | ||
| 100 | 23.1 | 12.5 | 65.2 | ||
| Maltase | - | 44.3 | - | - | 1.72 |
| 1 | 27 | N.D | 39.1 | ||
| 10 | 22.4 | N.D | 49.4 | ||
| 100 | 20.3 | N.D | 54.2 | ||
| Isomaltase | - | 31.6 | - | - | - |
| 1 | 21.1 | N.D | 33.2 | ||
| 10 | 31.9 | N.D | - | ||
| 100 | 46.7 | 6.7 | - |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sato, K.; Nagai, N.; Yamamoto, T.; Mitamura, K.; Taga, A. Identification of a Novel Oligosaccharide in Maple Syrup as a Potential Alternative Saccharide for Diabetes Mellitus Patients. Int. J. Mol. Sci. 2019, 20, 5041. https://doi.org/10.3390/ijms20205041
Sato K, Nagai N, Yamamoto T, Mitamura K, Taga A. Identification of a Novel Oligosaccharide in Maple Syrup as a Potential Alternative Saccharide for Diabetes Mellitus Patients. International Journal of Molecular Sciences. 2019; 20(20):5041. https://doi.org/10.3390/ijms20205041
Chicago/Turabian StyleSato, Kanta, Noriaki Nagai, Tetsushi Yamamoto, Kuniko Mitamura, and Atsushi Taga. 2019. "Identification of a Novel Oligosaccharide in Maple Syrup as a Potential Alternative Saccharide for Diabetes Mellitus Patients" International Journal of Molecular Sciences 20, no. 20: 5041. https://doi.org/10.3390/ijms20205041
APA StyleSato, K., Nagai, N., Yamamoto, T., Mitamura, K., & Taga, A. (2019). Identification of a Novel Oligosaccharide in Maple Syrup as a Potential Alternative Saccharide for Diabetes Mellitus Patients. International Journal of Molecular Sciences, 20(20), 5041. https://doi.org/10.3390/ijms20205041