Ibuprofen-Loaded Hyaluronic Acid Nanofibrous Membranes for Prevention of Postoperative Tendon Adhesion through Reduction of Inflammation
Abstract
1. Introduction
2. Results and Discussion
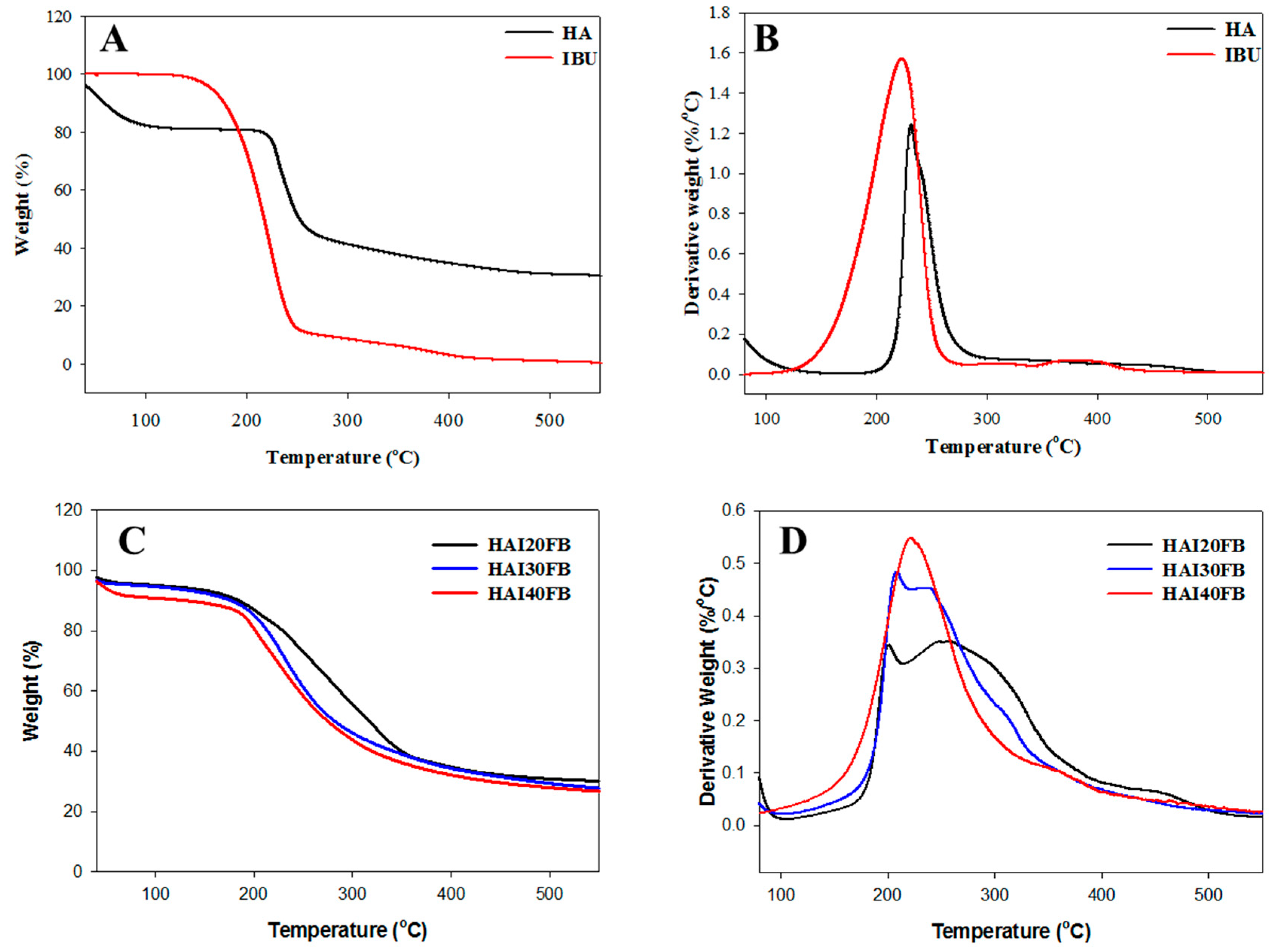
2.1. Characteristics of the NFMs
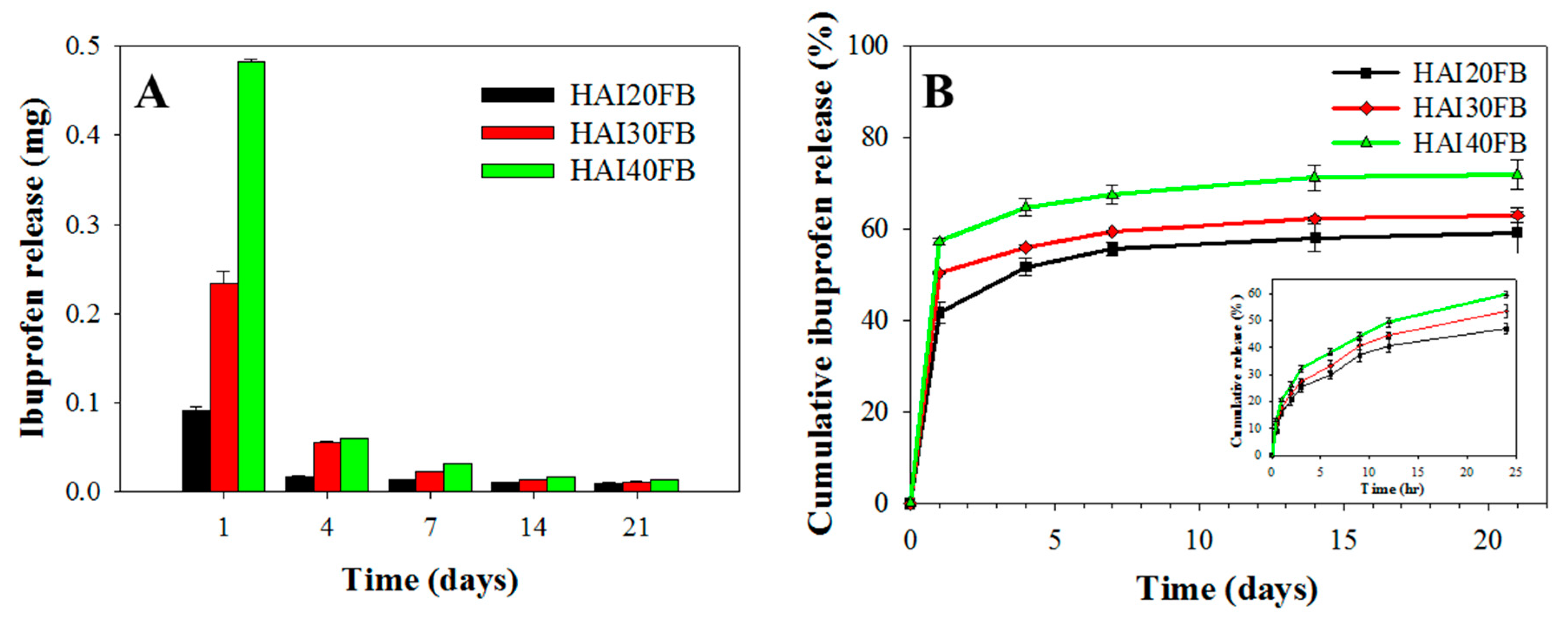
2.2. Release of IBU from NFMs
2.3. In Vitro Cell Culture
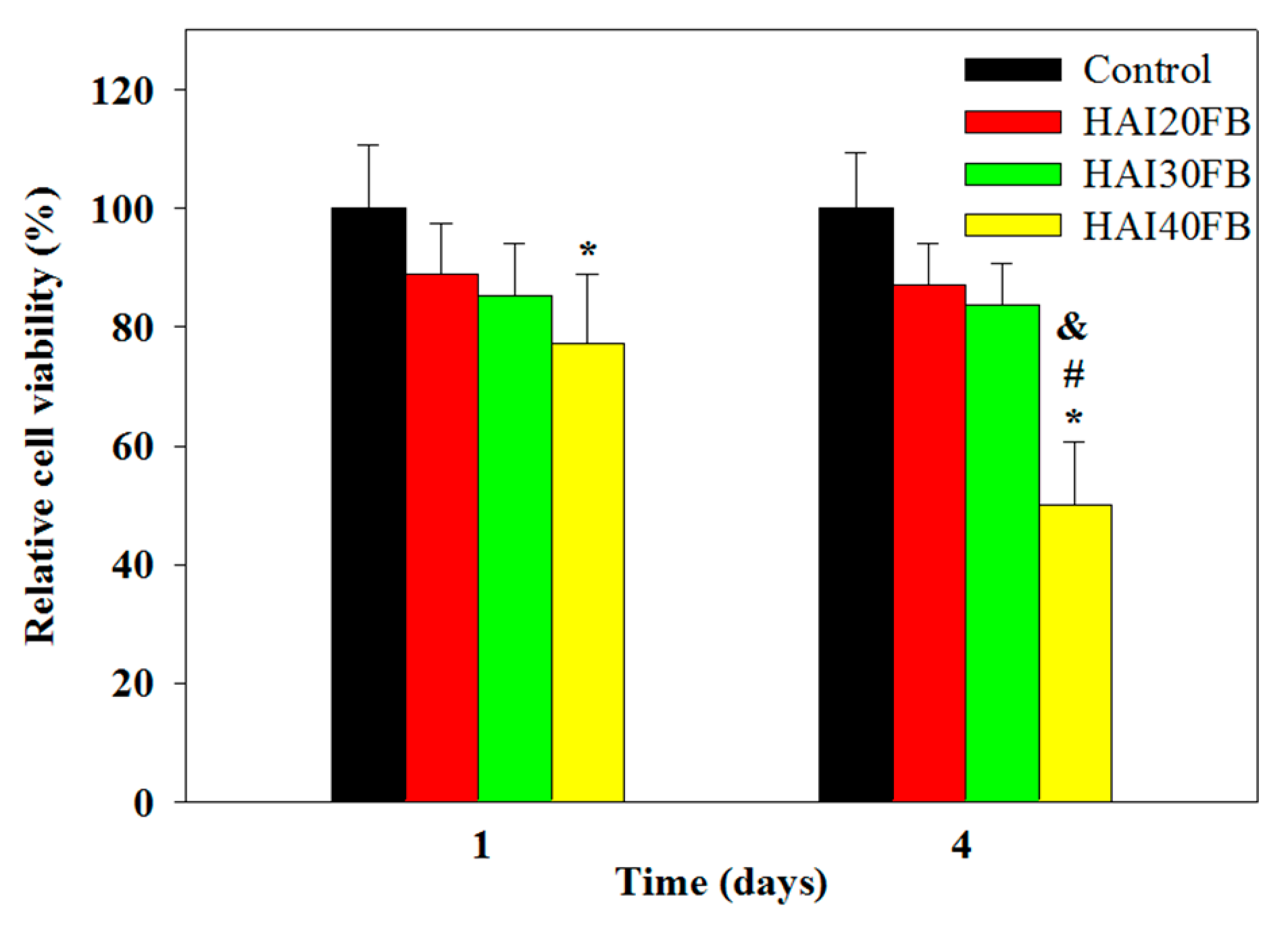
2.3.1. Cytotoxicity of NFMs
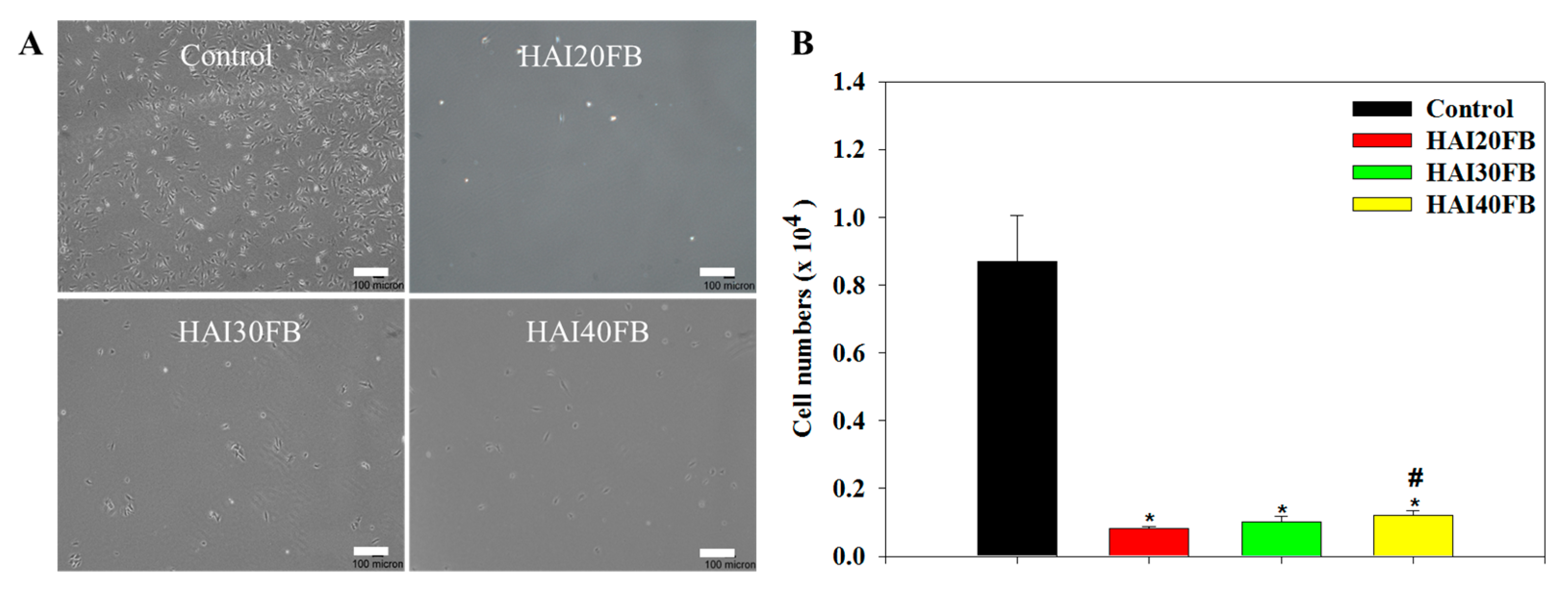
2.3.2. Penetration of Cells through NFMs
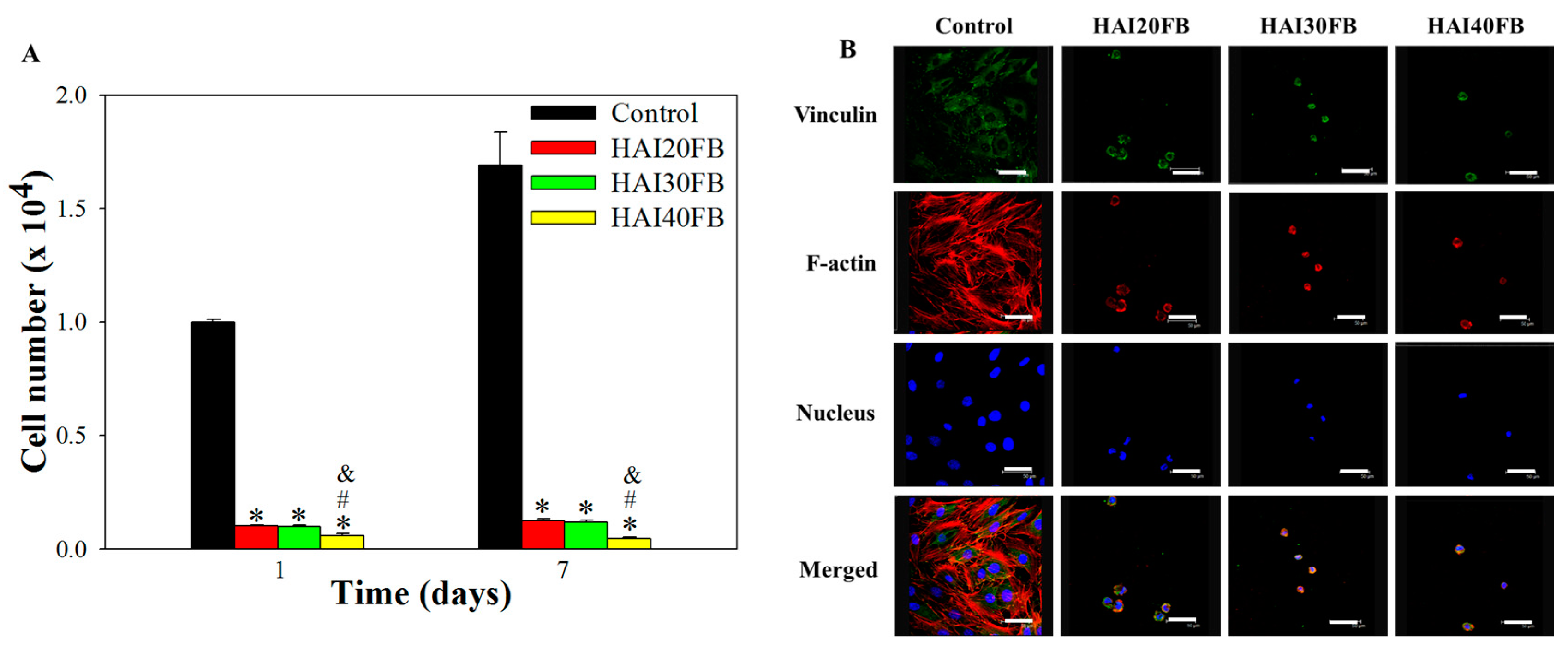
2.3.3. Cell Attachment to NFMs
2.4. In Vivo Studies
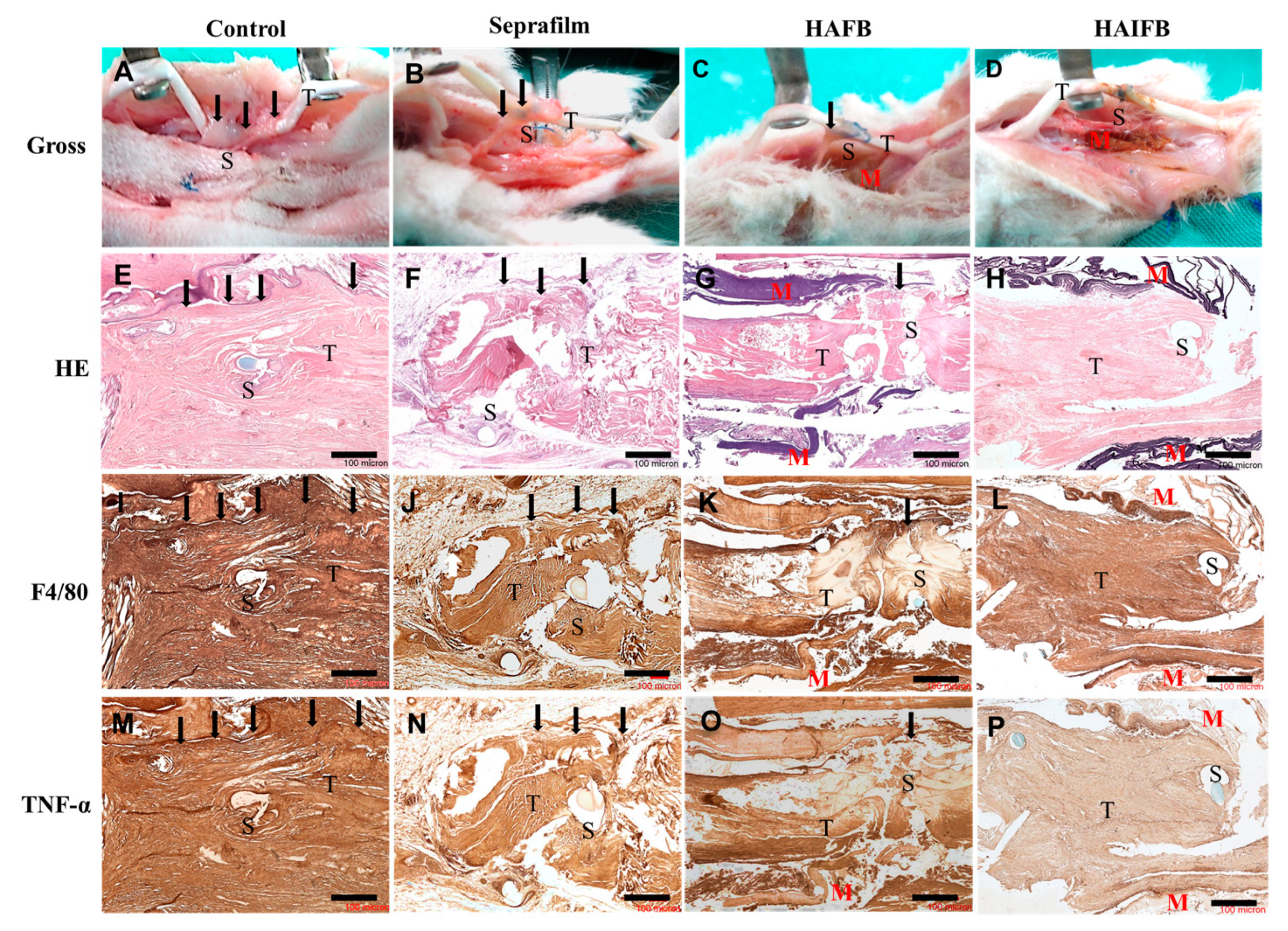
2.4.1. Gross Observation
2.4.2. Histological Staining
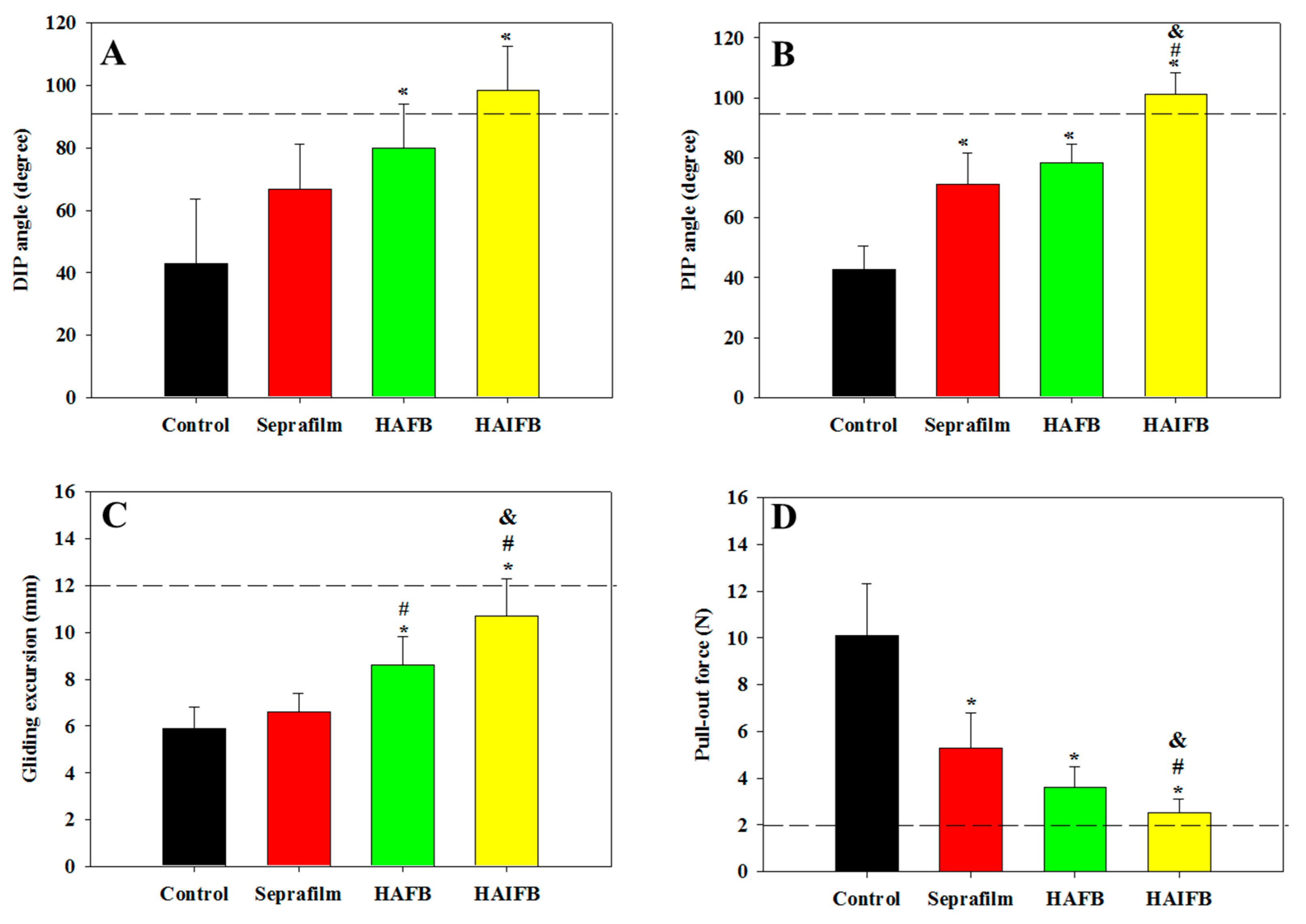
2.4.3. Evaluation of Tendon Adhesion by Biomechanical Evaluation
3. Materials and Methods
3.1. Materials
3.2. Fabrication of Nanofibrous Membranes (NFMs)
3.3. Characteristics of Nanofibrous Membranes (NFMs)
3.4. In Vitro Cell Culture Studies
3.4.1. Cell Penetration through NFMs
3.4.2. Cell Attachment and Proliferation
3.4.3. Cytoskeleton and Focal Adhesion Analyses by Confocal Microscopy
3.5. Animal Studies
3.5.1. Flexor Tendon Adhesion Animal Model
3.5.2. Gross Observation
3.5.3. Histological Evaluation
3.5.4. Biomechanical Evaluation
3.6. Statistical Analysis
4. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
Abbreviations
| ATR-FTIR | Attenuated total reflection Fourier-transform infrared |
| BDDE | 1,4-butanediol diglycidyl |
| DMEM | Dulbecco’s modified Eagle medium |
| DTG | Derivative thermogravimetry |
| DAPI | 4′,6-diamidino-2-phenylindole |
| DIP | Distal interphalangeal |
| ELISA | Enzyme-linked immunosorbent assay |
| FDP | Flexor digitorum profundus |
| FDS | Flexor digitorum superficialis |
| FBS | Fetal bovine serum |
| FITC | Fluorescein isothiocyanate |
| GMP | Good manufacturing practice |
| HA | Hyaluronic acid |
| H&E | Hematoxylin and eosin |
| IBU | Ibuprofen |
| IHC | Immunohistochemical |
| NFM | Nanofibrous membrane |
| NSAIDs | Nonsteroidal anti-inflammatory drugs |
| OD | Optical density |
| PBS | Phosphate buffered saline |
| PCL | Poly(caprolactone) |
| PEG | Poly(ethylene glycol) |
| PEO | Poly(ethylene oxide) |
| PLA | Poly(lactic acid) |
| PIP | Proximal interphalangeal |
| PIP | Proximal interphalangeal |
| SEM | Scanning electron microscopy |
| TGA | Thermogravimetric analysis |
| TCPS | Tissue culture polystyrene |
| TNF-α | Tumor necrosis factor alpha |
References
- Bolgen, N.; Vargel, I.; Korkusuz, P.; Menceloglu, Y.Z.; Piskin, E. In vivo performance of antibiotic embedded electrospun PCL membranes for prevention of abdominal adhesions. J. Biomed. Mater. Res. B Appl. Biomater. 2007, 81, 530–543. [Google Scholar] [CrossRef] [PubMed]
- Diamond, M.P.; Burns, E.L.; Accomando, B.; Mian, S.; Holmdahl, L. Seprafilm® adhesion barrier: (1) a review of preclinical, animal, and human investigational studies. Gynecol. Surg. 2012, 9, 237–245. [Google Scholar] [CrossRef] [PubMed]
- Hwang, H.; An, M.; Ha, T.; Kim, K.; Kim, T.; Choi, C.; Hong, K.; Jung, S.; Kim, S.-H.; Rho, K.; et al. All the commercially available adhesion barriers have the same effect on adhesion prophylaxis?; A comparison of barrier agents using a newly developed, severe intra-abdominal adhesion model. Int. J. Colorectal Dis. 2013, 28, 1117–1125. [Google Scholar] [CrossRef] [PubMed]
- Gruber-Blum, S.; Petter-Puchner, A.H.; Brand, J.; Fortelny, R.H.; Walder, N.; Oehlinger, W.; Koenig, F.; Redl, H. Comparison of three separate antiadhesive barriers for intraperitoneal onlay mesh hernia repair in an experimental model. Br. J. Surg. 2011, 98, 442–449. [Google Scholar] [CrossRef]
- Shalumon, K.T.; Sheu, C.; Chen, C.-H.; Chen, S.-H.; Jose, G.; Kuo, C.-Y.; Chen, J.-P. Multi-functional electrospun antibacterial core-shell nanofibrous membranes for prolonged prevention of post-surgical tendon adhesion and inflammation. Acta Biomater. 2018, 72, 121–136. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.-H.; Chen, S.-H.; Shalumon, K.T.; Chen, J.-P. Prevention of peritendinous adhesions with electrospun polyethylene glycol/polycaprolactone nanofibrous membranes. Colloids Surf. B Biointerfaces 2015, 133, 221–230. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.-H.; Chen, C.-H.; Shalumon, K.T.; Chen, J.-P. Preparation and characterization of antiadhesion barrier film from hyaluronic acid-grafted electrospun poly(caprolactone) nanofibrous membranes for prevention of flexor tendon postoperative peritendinous adhesion. Int. J. Nanomed. 2014, 9, 4079–4092. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Zhao, J.; Ruan, H.; Tang, T.; Liu, G.; Yu, D.; Cui, W.; Fan, C. Biomimetic sheath membrane via electrospinning for antiadhesion of repaired tendon. Biomacromolecules 2012, 13, 3611–3619. [Google Scholar] [CrossRef] [PubMed]
- Hu, C.; Liu, S.; Zhang, Y.; Li, B.; Yang, H.; Fan, C.; Cui, W. Long-term drug release from electrospun fibers for in vivo inflammation prevention in the prevention of peritendinous adhesions. Acta Biomater. 2013, 9, 7381–7388. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Hu, C.; Li, F.; Li, X.-j.; Cui, W.; Fan, C. Prevention of peritendinous adhesions with electrospun ibuprofen-loaded poly (l-lactic acid)-polyethylene glycol fibrous membranes. Tissue Eng. A 2012, 19, 529–537. [Google Scholar] [CrossRef] [PubMed]
- James, R.; Kesturu, G.; Balian, G.; Chhabra, A.B. Tendon: Biology, biomechanics, repair, growth factors, and evolving treatment options. J. Hand Surg. 2008, 33, 102–112. [Google Scholar] [CrossRef] [PubMed]
- Kulick, M.I.; Smith, S.; Hadler, K. Oral ibuprofen: Evaluation of its effect on peritendinous adhesions and the breaking strength of a tenorrhaphy. J. Hand Surg. 1986, 11, 110–120. [Google Scholar] [CrossRef]
- Rouhani, A.; Tabrizi, A.; Ghavidel, E. Effects of non-steroidal anti-inflammatory drugs on flexor tendon rehabilitation after repair. Arch. Bone Jt. Surg. 2013, 1, 28–30. [Google Scholar] [PubMed]
- Chen, S.; Wang, G.; Wu, T.; Zhao, X.; Liu, S.; Li, G.; Cui, W.; Fan, C. Silver nanoparticles/ibuprofen-loaded poly(l-lactide) fibrous membrane: Anti-infection and anti-adhesion effects. Int. J. Mol. Sci. 2014, 15, 14014–14025. [Google Scholar] [CrossRef]
- Zhang, W.; Li, X.; Franchini, M.C.; Xu, K.; Locatelli, E.; Martin, R.C.; Monaco, I.; Li, Y.; Cui, S. Controlled release of curcumin from curcumin-loaded nanomicelles to prevent peritendinous adhesion during Achilles tendon healing in rats. Int. J. Nanomed. 2016, 11, 2873–2881. [Google Scholar]
- Lee, S.; Jin, G.; Jang, J.-H. Electrospun nanofibers as versatile interfaces for efficient gene delivery. J. Biol. Eng. 2014, 8, 30. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, L.H.; Gao, M.; Lin, J.; Wu, W.; Wang, J.; Chew, S.Y. Three-dimensional aligned nanofibers-hydrogel scaffold for controlled non-viral drug/gene delivery to direct axon regeneration in spinal cord injury treatment. Sci. Rep. 2017, 7, 42212. [Google Scholar] [CrossRef]
- Nagarajan, S.; Bechelany, M.; Kalkura, N.S.; Miele, P.; Bohatier, C.P.; Balme, S. Electrospun nanofibers for drug delivery in regenerative medicine. In Applications of Targeted Nano Drugs and Delivery Systems; Elsevier: Amsterdam, The Netherlands, 2019; pp. 595–625. [Google Scholar]
- Figueroa-Lopez, K.; Castro-Mayorga, J.; Andrade-Mahecha, M.; Cabedo, L.; Lagaron, J. Antibacterial and barrier properties of gelatin coated by electrospun polycaprolactone ultrathin fibers containing black pepper oleoresin of interest in active food biopackaging applications. Nanomaterials 2018, 8, 199. [Google Scholar] [CrossRef]
- Swann, D.A.; Radin, E.L.; Nazimiec, M.; Weisser, P.A.; Curran, N.; Lewinnek, G. Role of hyaluronic acid in joint lubrication. Ann. Rheum. Dis. 1974, 33, 318–326. [Google Scholar] [CrossRef]
- Galandáková, A.; Ulrichová, J.; Langová, K.; Hanáková, A.; Vrbka, M.; Hartl, M.; Gallo, J. Characteristics of synovial fluid required for optimization of lubrication fluid for biotribological experiments. J. Biomed. Mater. Res. B Appl. Biomater. 2016, 105, 1422–1431. [Google Scholar] [CrossRef]
- Diamond, M.P.; Burns, E.L.; Accomando, B.; Mian, S.; Holmdahl, L. Seprafilm adhesion barrier: (2) a review of the clinical literature on intraabdominal use. Gynecol. Surg. 2012, 9, 247–257. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.H.; Go, A.K.; Oh, S.H.; Lee, K.E.; Yuk, S.H. Tissue anti-adhesion potential of ibuprofen-loaded PLLA-PEG diblock copolymer films. Biomaterials 2005, 26, 671–678. [Google Scholar] [CrossRef] [PubMed]
- Yeo, Y.; Kohane, D.S. Polymers in the prevention of peritoneal adhesions. Eur. J. Pharm. Biopharm. 2008, 68, 57–66. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Ni, J.; Chen, L.; Yu, L.; Xu, J.; Ding, J. Biodegradable and thermoreversible PCLA–PEG–PCLA hydrogel as a barrier for prevention of post-operative adhesion. Biomaterials 2011, 32, 4725–4736. [Google Scholar] [CrossRef] [PubMed]
- Yamaoka, T.; Njatawidjaja, E.; Kasai, A.; Agudelo, C.A.; Ehashi, T.; Kakinoki, S.; Kato, S.; Mahara, A. Elastic/adhesive double-layered PLA-PEG multiblock copolymer membranes for postoperative adhesion prevention. Polym. Degrad. Stab. 2013, 98, 2168–2176. [Google Scholar] [CrossRef]
- Angajala, K.K.; Vianala, S.; Macha, R.; Raghavender, M.; Thupurani, M.K.; Pathi, P.J. Synthesis, anti-inflammatory, bactericidal activities and docking studies of novel 1,2,3-triazoles derived from ibuprofen using click chemistry. SpringerPlus 2016, 5, 423. [Google Scholar] [CrossRef] [PubMed]
- Tan, S.C.; Patel, B.K.; Jackson, S.H.D.; Swift, C.G.; Hutt, A.J. Influence of age on the enantiomeric disposition of ibuprofen in healthy volunteers. Br. J. Clin. Pharmacol. 2003, 55, 579–587. [Google Scholar] [CrossRef]
- Tran, T.; Hernandez, M.; Patel, D.; Burns, E.; Peterman, V.; Wu, J. Controllable and switchable drug delivery of ibuprofen from temperature responsive composite nanofibers. Nano Converg. 2015, 2, 15. [Google Scholar] [CrossRef]
- Lewandowska, K.; Sionkowska, A.; Grabska, S.; Kaczmarek, B. Surface and thermal properties of collagen/hyaluronic acid blends containing chitosan. Int. J. Biol. Macromol. 2016, 92, 371–376. [Google Scholar] [CrossRef]
- Sheu, C.; Shalumon, K.T.; Chen, C.-H.; Kuo, C.-Y.; Fong, Y.T.; Chen, J.-P. Dual crosslinked hyaluronic acid nanofibrous membranes for prolonged prevention of post-surgical peritoneal adhesion. J. Mater. Chem. B 2016, 4, 6680–6693. [Google Scholar] [CrossRef]
- Ofokansi, K.C.; Kenechukwu, F.C.; Ezugwu, R.O.; Attama, A.A. Improved dissolution and anti-inflammatory activity of ibuprofen-polyethylene glycol 8000 solid dispersion systems. Int. J. Pharm. Investig. 2016, 6, 139–147. [Google Scholar] [PubMed]
- Chevalier, E.; Viana, M.; Cazalbou, S.; Makein, L.; Dubois, J.; Chulia, D. Ibuprofen-loaded calcium phosphate granules: Combination of innovative characterization methods to relate mechanical strength to drug location. Acta Biomater. 2010, 6, 266–274. [Google Scholar] [CrossRef] [PubMed]
- Docheva, D.; Müller, S.A.; Majewski, M.; Evans, C.H. Biologics for tendon repair. Adv. Drug Deliv. Rev. 2015, 84, 222–239. [Google Scholar] [CrossRef] [PubMed]
- Brain, P.; Leyva, R.; Doyle, G.; Kellstein, D. Onset of analgesia and efficacy of ibuprofen sodium in postsurgical dental pain: A randomized, placebo-controlled study versus standard ibuprofen. Clin. J. Pain 2015, 31, 444–450. [Google Scholar] [CrossRef] [PubMed]
- Manrique-Moreno, M.; Villena, F.; Sotomayor, C.P.; Edwards, A.M.; Muñoz, M.A.; Garidel, P.; Suwalsky, M. Human cells and cell membrane molecular models are affected in vitro by the nonsteroidal anti-inflammatory drug ibuprofen. Biochim. Biophys. Acta Biomembr. 2011, 1808, 2656–2664. [Google Scholar] [CrossRef] [PubMed]
- Koga, T.; Fujiwara, R.; Nakajima, M.; Yokoi, T. Toxicological evaluation of acyl glucuronides of nonsteroidal anti-inflammatory drugs using human embryonic kidney 293 cells stably expressing human UDP-glucuronosyltransferase and human hepatocytes. Drug Metab. Dispos 2011, 39, 54–60. [Google Scholar] [CrossRef] [PubMed]
- Aghaei, H.; Nourbakhsh, A.A.; Karbasi, S.; JavadKalbasi, R.; Rafienia, M.; Nourbakhsh, N.; Bonakdar, S.; Mackenzie, K.J.D. Investigation on bioactivity and cytotoxicity of mesoporous nano-composite MCM-48/hydroxyapatite for ibuprofen drug delivery. Ceramics Int. 2014, 40, 7355–7362. [Google Scholar] [CrossRef]
- Pozzi, A.; Gallelli, L. Pain management for dentists: The role of ibuprofen. Ann. Stomatol. 2011, 2, 3–24. [Google Scholar]
- Chen, S.H.; Chou, P.Y.; Chen, Z.Y.; Lin, F.H. Electrospun water-borne polyurethane nanofibrous membrane as a barrier for preventing postoperative peritendinous adhesion. Int. J. Mol. Sci. 2019, 20, 1625. [Google Scholar] [CrossRef]
- Ishiyama, N.; Moro, T.; Ohe, T.; Miura, T.; Ishihara, K.; Konno, T.; Ohyama, T.; Kimura, M.; Kyomoto, M.; Saito, T.; et al. Reduction of peritendinous adhesions by hydrogel containing biocompatible phospholipid polymer mpc for tendon repair. J. Bone Joint Surg. 2011, 93, 142–149. [Google Scholar] [CrossRef]
- diZerega, G.S. Contemporary adhesion prevention. Fertil. Steril. 1994, 61, 219–235. [Google Scholar] [CrossRef]
- Hellebrekers, B.W.; Kooistra, T. Pathogenesis of postoperative adhesion formation. Br. J. Surg. 2011, 98, 1503–1516. [Google Scholar] [CrossRef] [PubMed]
- Köwitsch, A.; Yang, Y.; Ma, N.; Kuntsche, J.; Mäder, K.; Groth, T. Bioactivity of immobilized hyaluronic acid derivatives regarding protein adsorption and cell adhesion. Biotechnol. Appl. Biochem. 2011, 58, 376–389. [Google Scholar] [CrossRef]
- Tsai, W.-C.; Tang, F.-T.; Hsu, C.-C.; Hsu, Y.-H.; Pang, J.-H.S.; Shiue, C.-C. Ibuprofen inhibition of tendon cell proliferation and upregulation of the cyclin kinase inhibitor p21cip1. J. Orthop. Res. 2004, 22, 586–591. [Google Scholar] [CrossRef] [PubMed]
- Yagi, M.; Sato, N.; Mitsui, Y.; Gotoh, M.; Hamada, T.; Nagata, K. Hyaluronan modulates proliferation and migration of rabbit fibroblasts derived from flexor tendon epitenon and endotenon. J. Hand Surg. 2010, 35, 791–796. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.-H.; Chen, S.-H.; Shalumon, K.T.; Chen, J.-P. Dual functional core–sheath electrospun hyaluronic acid/polycaprolactone nanofibrous membranes embedded with silver nanoparticles for prevention of peritendinous adhesion. Acta Biomater. 2015, 26, 225–235. [Google Scholar] [CrossRef] [PubMed]
- Meier Bürgisser, G.; Calcagni, M.; Bachmann, E.; Fessel, G.; Snedeker, J.G.; Giovanoli, P.; Buschmann, J. Rabbit achilles tendon full transection model–wound healing, adhesion formation and biomechanics at 3, 6 and 12 weeks post-surgery. Biol. Open 2016, 5, 1324–1333. [Google Scholar] [CrossRef] [PubMed]
- Kwan, K.H.L.; Yeung, K.W.K.; Liu, X.; Wong, K.K.Y.; Shum, H.C.; Lam, Y.W.; Cheng, S.H.; Cheung, K.M.C.; To, M.K.T. Silver nanoparticles alter proteoglycan expression in the promotion of tendon repair. Nanomed. Nanotechnol. 2014, 10, 1375–1383. [Google Scholar] [CrossRef]
- Tan, V.; Nourbakhsh, A.; Capo, J.; Cottrell, J.A.; Meyenhofer, M.; O′Connor, J.P. Effects of nonsteroidal anti-inflammatory drugs on flexor tendon adhesion. J. Hand Surg. 2010, 35, 941–947. [Google Scholar] [CrossRef]
- Parameswaran, N.; Patial, S. Tumor necrosis factor-alpha signaling in macrophages. Crit. Rev. Eukaryot. Gene Expr. 2010, 20, 87–103. [Google Scholar] [CrossRef]
- Dinarvand, P.; Hassanian, S.M.; Weiler, H.; Rezaie, A.R. Intraperitoneal administration of activated protein c prevents postsurgical adhesion band formation. Blood 2015, 125, 1339–1348. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Donlin, L.T.; Jayatilleke, A.; Giannopoulou, E.G.; Kalliolias, G.D.; Ivashkiv, L.B. Modulation of TNF-induced macrophage polarization by synovial fibroblasts. J. Immunol. 2014, 193, 2373–2383. [Google Scholar] [CrossRef] [PubMed]
- Chang, J.; Thunder, R.; Most, D.; Longaker, M.T.; Lineaweaver, W.C. Studies in flexor tendon wound healing: Neutralizing antibody to TGF-[beta]1 increases postoperative range of motion. Plast. Reconstr. Surg. 2000, 105, 148–155. [Google Scholar] [CrossRef] [PubMed]
- Namba, J.; Shimada, K.; Saito, M.; Murase, T.; Yamada, H.; Yoshikawa, H. Modulation of peritendinous adhesion formation by alginate solution in a rabbit flexor tendon model. J. Biomed. Mater. Res. B Appl. Biomater. 2007, 80, 273–279. [Google Scholar] [CrossRef]
- Vorvolakos, K.; Isayeva, I.S.; Do Luu, H.-M.; Patwardhan, D.V.; Pollack, S.K. Ionically cross-linked hyaluronic acid: Wetting, lubrication, and viscoelasticity of a modified adhesion barrier gel. Med. Devices (Auckl.) 2011, 4, 1. [Google Scholar] [CrossRef] [PubMed]

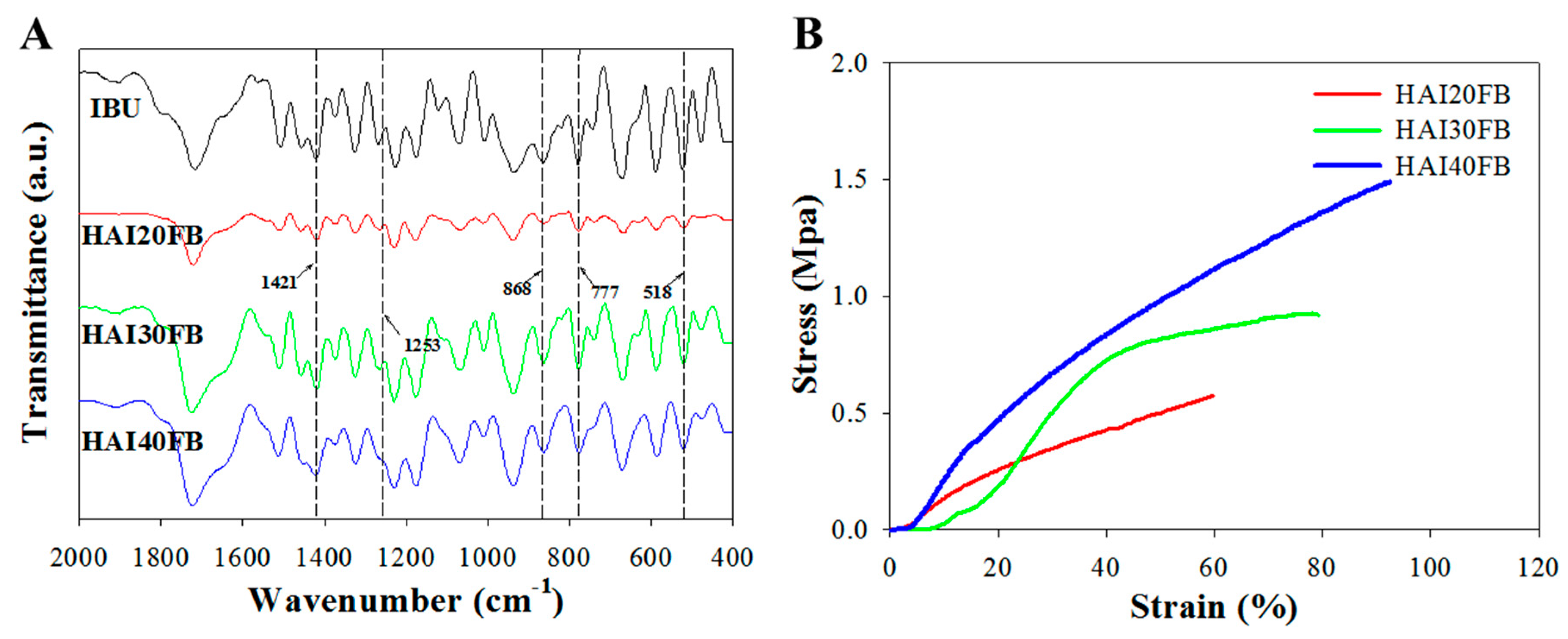
| Membrane | Ultimate Stress (MPa) | Ultimate Strain (%) | Young’s Modulus |
|---|---|---|---|
| HAI20FB | 0.63 ± 0.53 | 61.46 ± 11.42 | 9.42 ± 0.83 |
| HAI30FB | 0.94 ± 0.89 * | 81.22 ± 8.23 * | 10.57 ± 0.84 |
| HAI40FB | 1.43 ± 0.13 *, # | 90.11 ± 8.75 *, # | 14.16 ± 1.25 *, # |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, C.-T.; Chen, C.-H.; Sheu, C.; Chen, J.-P. Ibuprofen-Loaded Hyaluronic Acid Nanofibrous Membranes for Prevention of Postoperative Tendon Adhesion through Reduction of Inflammation. Int. J. Mol. Sci. 2019, 20, 5038. https://doi.org/10.3390/ijms20205038
Chen C-T, Chen C-H, Sheu C, Chen J-P. Ibuprofen-Loaded Hyaluronic Acid Nanofibrous Membranes for Prevention of Postoperative Tendon Adhesion through Reduction of Inflammation. International Journal of Molecular Sciences. 2019; 20(20):5038. https://doi.org/10.3390/ijms20205038
Chicago/Turabian StyleChen, Chien-Tzung, Chih-Hao Chen, Chialin Sheu, and Jyh-Ping Chen. 2019. "Ibuprofen-Loaded Hyaluronic Acid Nanofibrous Membranes for Prevention of Postoperative Tendon Adhesion through Reduction of Inflammation" International Journal of Molecular Sciences 20, no. 20: 5038. https://doi.org/10.3390/ijms20205038
APA StyleChen, C.-T., Chen, C.-H., Sheu, C., & Chen, J.-P. (2019). Ibuprofen-Loaded Hyaluronic Acid Nanofibrous Membranes for Prevention of Postoperative Tendon Adhesion through Reduction of Inflammation. International Journal of Molecular Sciences, 20(20), 5038. https://doi.org/10.3390/ijms20205038






