Identification of c.1531C>T Pathogenic Variant in the CDH1 Gene as a Novel Germline Mutation of Hereditary Diffuse Gastric Cancer
Abstract
1. Introduction
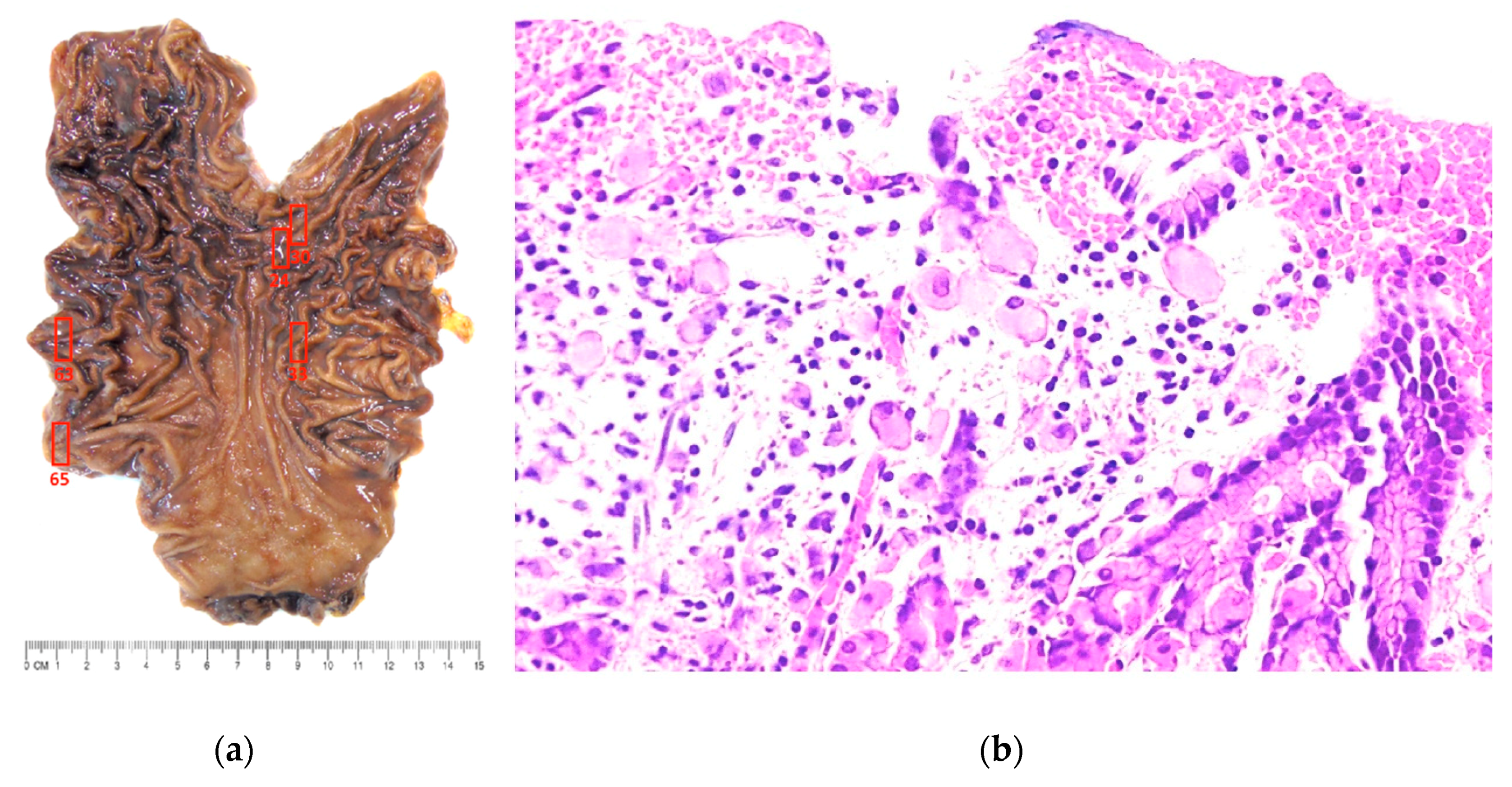
2. Results
3. Discussion
4. Materials and Methods
4.1. DNA Extraction
4.2. Sequence Analysis
4.3. Statistical Analyses
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ferlay, J.; Colombet, M.; Soerjomataram, I.; Mathers, C.; Parkin, D.M.; Pineros, M.; Znaor, A.; Bray, F. Estimating the global cancer incidence and mortality in 2018: GLOBOCAN sources and methods. Int. J. Cancer 2019, 144, 1941. [Google Scholar] [CrossRef] [PubMed]
- Zanghieri, G.; Di Gregorio, C.; Sacchetti, C.; Fante, R.; Sassatelli, R.; Cannizzo, G.; Carriero, A.; Ponz de Leon, M. Familial occurrence of Gastric Cancer in the 2-year experience of a population-based registry. Cancer 1990, 66, 2047–2051. [Google Scholar] [CrossRef]
- Jones, E.G. Familial Gastric Cancer. N. Z. Med. J. 1964, 63, 287–296. [Google Scholar] [PubMed]
- Guilford, P.; Hopkins, J.; Harraway, J.; McLeod, M.; McLeod, N.; Harawira, P.; Taite, H.; Scoular, R.; Miller, A.; Reeve, A.E. E-cadherin germline mutations in familial gastric cancer. Nature 1998, 392, 402–405. [Google Scholar] [CrossRef] [PubMed]
- Guilford, P.; Humar, B.; Blair, V. Hereditary diffuse gastric cancer: Translation of CDH1 germline mutations into clinical practice. Gastric Cancer 2010, 13, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Park, J.G.; Yang, H.K.; Kim, W.H.; Caldas, C.; Yokota, J.; Guilford, P.J. Report on the first meeting of the International Collaborative Group on Hereditary Gastric Cancer. J. Natl. Cancer Inst. 2000, 92, 1781–1782. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Humar, B.; Guilford, P. Hereditary diffuse gastric cancer: A manifestation of lost cell polarity. Cancer Sci. 2009, 100, 1151–1157. [Google Scholar] [CrossRef]
- Van der Post, R.S.; Vogelaar, I.P.; Carneiro, F.; Guilford, P.; Huntsman, D.; Hoogerbrugge, N.; Caldas, C.; Schreiber, K.E.; Hardwick, R.H.; Ausems, M.G.; et al. Hereditary diffuse gastric cancer: Updated clinical guidelines with an emphasis on germline CDH1 mutation carriers. J. Med. Genet. 2015, 52, 361–374. [Google Scholar] [CrossRef]
- Fitzgerald, R.C.; Hardwick, R.; Huntsman, D.; Carneiro, F.; Guilford, P.; Blair, V.; Chung, D.C.; Norton, J.; Ragunath, K.; Van Krieken, J.H.; et al. Hereditary diffuse gastric cancer: Updated consensus guidelines for clinical management and directions for future research. J. Med. Genet. 2010, 47, 436–444. [Google Scholar] [CrossRef]
- Hansford, S.; Kaurah, P.; Li-Chang, H.; Woo, M.; Senz, J.; Pinheiro, H.; Schrader, K.A.; Schaeffer, D.F.; Shumansky, K.; Zogopoulos, G.; et al. Hereditary Diffuse Gastric Cancer Syndrome: CDH1 Mutations and Beyond. JAMA Oncol. 2015, 1, 23–32. [Google Scholar] [CrossRef]
- Pharoah, P.D.; Guilford, P.; Caldas, C. Incidence of Gastric Cancer and breast cancer in CDH1 (E-cadherin) mutation carriers from hereditary diffuse Gastric Cancer families. Gastroenterology 2001, 121, 1348–1353. [Google Scholar] [CrossRef] [PubMed]
- Seevaratnam, R.; Coburn, N.; Cardoso, R.; Dixon, M.; Bocicariu, A.; Helyer, L. A systematic review of the indications for genetic testing and prophylactic gastrectomy among patients with hereditary diffuse gastric cancer. Gastric Cancer 2012, 15, 153–163. [Google Scholar] [CrossRef] [PubMed]
- Corso, G.; Marrelli, D.; Pascale, V.; Vindigni, C.; Roviello, F. Frequency of CDH1 germline mutations in gastric carcinoma coming from high- and low-risk areas: Metanalysis and systematic review of the literature. BMC Cancer 2012, 12, 8. [Google Scholar] [CrossRef] [PubMed]
- Melo, S.; Figueiredo, J.; Fernandes, M.S.; Goncalves, M.; Morais-de-Sa, E.; Sanches, J.M.; Seruca, R. Predicting the Functional Impact of CDH1 Missense Mutations in Hereditary Diffuse Gastric Cancer. Int. J. Mol. Sci. 2017, 18, 2687. [Google Scholar] [CrossRef] [PubMed]
- Cancer Genome Atlas Research, N. Comprehensive molecular characterization of gastric adenocarcinoma. Nature 2014, 513, 202–209. [Google Scholar] [CrossRef] [PubMed]
- Van der Post, R.S.; Vogelaar, I.P.; Manders, P.; van der Kolk, L.E.; Cats, A.; van Hest, L.P.; Sijmons, R.; Aalfs, C.M.; Ausems, M.G.; Gomez Garcia, E.B.; et al. Accuracy of Hereditary Diffuse Gastric Cancer Testing Criteria and Outcomes in Patients with a Germline Mutation in CDH1. Gastroenterology 2015, 149, 897–906. [Google Scholar] [CrossRef] [PubMed]
- Kaurah, P.; MacMillan, A.; Boyd, N.; Senz, J.; De Luca, A.; Chun, N.; Suriano, G.; Zaor, S.; Van Manen, L.; Gilpin, C.; et al. Founder and recurrent CDH1 mutations in families with hereditary diffuse gastric cancer. Jama 2007, 297, 2360–2372. [Google Scholar] [CrossRef]
- De Campos, E.C.; Ribeiro, S.; Higashi, R.; Manfredini, R.; Kfouri, D.; Cavalcanti, T.C. Hereditary diffuse gastric cancer: Laparoscopic surgical approach associated to rare mutattion of CDH1 gene. Arq. Bras. Cir. 2015, 28, 149–151. [Google Scholar] [CrossRef][Green Version]
- Moreira-Nunes, C.A.; Barros, M.B.L.; do Nascimento Borges, B.; Montenegro, R.C.; Lamarao, L.M.; Ribeiro, H.F.; Bona, A.B.; Assumpcao, P.P.; Rey, J.A.; Pinto, G.R.; et al. Genetic screening analysis of patients with hereditary diffuse Gastric Cancer from northern and northeastern Brazil. Hered. Cancer Clin. Pract. 2014, 12, 18. [Google Scholar] [CrossRef]
- Landrum, M.J.; Lee, J.M.; Benson, M.; Brown, G.; Chao, C.; Chitipiralla, S.; Gu, B.; Hart, J.; Hoffman, D.; Hoover, J.; et al. ClinVar: Public archive of interpretations of clinically relevant variants. Nucleic Acids Res. 2016, 44, 862–868. [Google Scholar] [CrossRef]
- Li, M.M.; Datto, M.; Duncavage, E.J.; Kulkarni, S.; Lindeman, N.I.; Roy, S.; Tsimberidou, A.M.; Vnencak-Jones, C.L.; Wolff, D.J.; Younes, A.; et al. Standards and Guidelines for the Interpretation and Reporting of Sequence Variants in Cancer: A Joint Consensus Recommendation of the Association for Molecular Pathology, American Society of Clinical Oncology, and College of American Pathologists. J. Mol. Diagn. 2017, 19, 4–23. [Google Scholar] [CrossRef] [PubMed]
- Richards, S.; Aziz, N.; Bale, S.; Bick, D.; Das, S.; Gastier-Foster, J.; Grody, W.W.; Hegde, M.; Lyon, E.; Spector, E.; et al. Standards and guidelines for the interpretation of sequence variants: A joint consensus recommendation of the American College of Medical Genetics and Genomics and the Association for Molecular Pathology. Genet. Med. 2015, 17, 405–424. [Google Scholar] [CrossRef] [PubMed]
- Norero, E.; Vega, E.A.; Diaz, C.; Cavada, G.; Ceroni, M.; Martinez, C.; Briceno, E.; Araos, F.; Gonzalez, P.; Baez, S.; et al. Improvement in postoperative mortality in elective gastrectomy for gastric cancer: Analysis of predictive factors in 1066 patients from a single centre. Eur. J. Surg. Oncol. 2017, 43, 1330–1336. [Google Scholar] [CrossRef] [PubMed]
- Lynch, H.T.; Lynch, J.F. Hereditary diffuse gastric cancer: Lifesaving total gastrectomy for CDH1 mutation carriers. J. Med. Genet. 2010, 47, 433–435. [Google Scholar] [CrossRef] [PubMed]
- Kunisaki, C.; Makino, H.; Takagawa, R.; Kimura, J.; Ota, M.; Ichikawa, Y.; Kosaka, T.; Akiyama, H.; Endo, I. A systematic review of laparoscopic total gastrectomy for gastric cancer. Gastric Cancer 2015, 18, 218–226. [Google Scholar] [CrossRef]
- Moisan, F.; Norero, E.; Slako, M.; Varas, J.; Palominos, G.; Crovari, F.; Ibanez, L.; Perez, G.; Pimentel, F.; Guzman, S.; et al. Completely laparoscopic versus open gastrectomy for early and advanced gastric cancer: A matched cohort study. Surg. Endosc. 2012, 26, 661–672. [Google Scholar] [CrossRef] [PubMed]
- Norero, E.; Baez, S.; Briceno, E.; Martinez, C.; Ceroni, M.; Escalona, A.; Aguayo, G.; Gonzalez, P.; Araos, F.; Calvo, A.; et al. Totally laparoscopic gastrectomy for the treatment of gastric tumors. Rev. Med. Chil. 2015, 143, 281–288. [Google Scholar] [CrossRef]
- Haverkamp, L.; Weijs, T.J.; van der Sluis, P.C.; van der Tweel, I.; Ruurda, J.P.; van Hillegersberg, R. Laparoscopic total gastrectomy versus open total gastrectomy for cancer: A systematic review and meta-analysis. Surg. Endosc. 2013, 27, 1509–1520. [Google Scholar] [CrossRef]
- Norero, E.; Munoz, R.; Ceroni, M.; Manzor, M.; Crovari, F.; Gabrielli, M. Two-Layer Hand-Sewn Esophagojejunostomy in Totally Laparoscopic Total Gastrectomy for Gastric Cancer. J. Gastric Cancer 2017, 17, 267–276. [Google Scholar] [CrossRef]
- Strong, V.E.; Gholami, S.; Shah, M.A.; Tang, L.H.; Janjigian, Y.Y.; Schattner, M.; Selby, L.V.; Yoon, S.S.; Salo-Mullen, E.; Stadler, Z.K.; et al. Total Gastrectomy for Hereditary Diffuse Gastric Cancer at a Single Center: Postsurgical Outcomes in 41 Patients. Ann. Surg. 2017, 266, 1006–1012. [Google Scholar] [CrossRef]
- Hakkaart, C.; Ellison-Loschmann, L.; Day, R.; Sporle, A.; Koea, J.; Harawira, P.; Cheng, S.; Gray, M.; Whaanga, T.; Pearce, N.; et al. Germline CDH1 mutations are a significant contributor to the high frequency of early-onset diffuse Gastric Cancer cases in New Zealand Maori. Fam. Cancer 2018, 18, 83–90. [Google Scholar] [CrossRef] [PubMed]
- Schrader, K.A.; Masciari, S.; Boyd, N.; Salamanca, C.; Senz, J.; Saunders, D.N.; Yorida, E.; Maines-Bandiera, S.; Kaurah, P.; Tung, N.; et al. Germline mutations in CDH1 are infrequent in women with early-onset or familial lobular breast cancers. J. Med. Genet. 2011, 48, 64–68. [Google Scholar] [CrossRef] [PubMed]
- Vogelaar, I.P.; Figueiredo, J.; van Rooij, I.A.; Simoes-Correia, J.; van der Post, R.S.; Melo, S.; Seruca, R.; Carels, C.E.; Ligtenberg, M.J.; Hoogerbrugge, N. Identification of germline mutations in the cancer predisposing gene CDH1 in patients with orofacial clefts. Hum. Mol. Genet. 2013, 22, 919–926. [Google Scholar] [CrossRef] [PubMed]
- Molinaro, V.; Pensotti, V.; Marabelli, M.; Feroce, I.; Barile, M.; Pozzi, S.; Laghi, L.; Serrano, D.; Bernard, L.; Bonanni, B.; et al. Complementary molecular approaches reveal heterogeneous CDH1 germline defects in Italian patients with hereditary diffuse Gastric Cancer (HDGC) syndrome. Genes Chromosomes Cancer 2014, 53, 432–445. [Google Scholar] [CrossRef] [PubMed]
- Brooks-Wilson, A.R.; Kaurah, P.; Suriano, G.; Leach, S.; Senz, J.; Grehan, N.; Butterfield, Y.S.; Jeyes, J.; Schinas, J.; Bacani, J.; et al. Germline E-cadherin mutations in hereditary diffuse gastric cancer: Assessment of 42 new families and review of genetic screening criteria. J. Med. Genet. 2004, 41, 508–517. [Google Scholar] [CrossRef] [PubMed]
- Van Domselaar, F.; Correa, D.; Vaccaro, C.; Redal, M.; Van Domselaar, R.; Huntsman, D.; Kaurah, P.; Senz, J.; Lynch, H. Hereditary diffuse Gastric Cancer (HDGC): Presentation of a family with a new mutation of the CDH1 gene. Acta Gastroenterol. Latinoam. 2007, 37, 158–163. [Google Scholar]
- Bray, F.; Ferlay, J.; Soerjomataram, I.; Siegel, R.L.; Torre, L.A.; Jemal, A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2018, 68, 394–424. [Google Scholar] [CrossRef]
- Sierra, M.S.; Cueva, P.; Bravo, L.E.; Forman, D. Stomach cancer burden in Central and South America. Cancer Epidemiol. 2016, 44, s62–s73. [Google Scholar] [CrossRef]
- Sahasrabudhe, R.; Lott, P.; Bohorquez, M.; Toal, T.; Estrada, A.P.; Suarez, J.J.; Brea-Fernandez, A.; Cameselle-Teijeiro, J.; Pinto, C.; Ramos, I.; et al. Germline Mutations in PALB2, BRCA1, and RAD51C, Which Regulate DNA Recombination Repair, in Patients with Gastric Cancer. Gastroenterology 2017, 152, 983–986. [Google Scholar] [CrossRef]
- Slavin, T.; Neuhausen, S.L.; Rybak, C.; Solomon, I.; Nehoray, B.; Blazer, K.; Niell-Swiller, M.; Adamson, A.W.; Yuan, Y.C.; Yang, K.; et al. Genetic Gastric Cancer Susceptibility in the International Clinical Cancer Genomics Community Research Network. Cancer Genet. 2017, 216–217, 111–119. [Google Scholar] [CrossRef]
- Jung, I.; Gurzu, S.; Turdean, G.S. Current status of familial gastrointestinal polyposis syndromes. World J. Gastrointest. Oncol. 2015, 7, 347–355. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.; Chung, J.W.; Jeong, T.D.; Park, Y.S.; Lee, J.H.; Ahn, J.Y.; Kim, D.H.; Choi, K.D.; Lee, W.; Song, H.J.; et al. Searching for E-cadherin gene mutations in early onset diffuse Gastric Cancer and hereditary diffuse Gastric Cancer in Korean patients. Fam. Cancer 2013, 12, 503–507. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, C.; Senz, J.; Kaurah, P.; Pinheiro, H.; Sanges, R.; Haegert, A.; Corso, G.; Schouten, J.; Fitzgerald, R.; Vogelsang, H.; et al. Germline CDH1 deletions in hereditary diffuse Gastric Cancer families. Hum. Mol. Genet. 2009, 18, 1545–1555. [Google Scholar] [CrossRef] [PubMed]
- Bennett, R.L.; French, K.S.; Resta, R.G.; Doyle, D.L. Standardized human pedigree nomenclature: Update and assessment of the recommendations of the National Society of Genetic Counselors. J. Genet. Couns. 2008, 17, 424–433. [Google Scholar] [CrossRef] [PubMed]
- Marrelli, D.; Morgagni, P.; de Manzoni, G.; Coniglio, A.; Marchet, A.; Saragoni, L.; Tiberio, G.; Roviello, F. Prognostic value of the 7th AJCC/UICC TNM classification of noncardia gastric cancer: Analysis of a large series from specialized Western centers. Ann. Surg. 2012, 255, 486–491. [Google Scholar] [CrossRef] [PubMed]
- Lauren, P. The Two Histological Main Types of Gastric Carcinoma: Diffuse and So-Called Intestinal-Type Carcinoma. An Attempt at a Histo-Clinical Classification. Acta Pathol. Microbiol. Scand. 1965, 64, 31–49. [Google Scholar] [CrossRef] [PubMed]
- Den Dunnen, J.T.; Dalgleish, R.; Maglott, D.R.; Hart, R.K.; Greenblatt, M.S.; McGowan-Jordan, J.; Roux, A.F.; Smith, T.; Antonarakis, S.E.; Taschner, P.E. HGVS Recommendations for the Description of Sequence Variants: 2016 Update. Hum. Mutat. 2016, 37, 564–569. [Google Scholar] [CrossRef] [PubMed]
 : Proband.
: Proband.  : Died from gastric cancer.
: Died from gastric cancer.  : Male.
: Male.  : Female. E1: endoscopic evaluation. E2: molecular evaluation. In red, the two individuals in which DGC was confirmed.
: Female. E1: endoscopic evaluation. E2: molecular evaluation. In red, the two individuals in which DGC was confirmed.
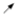 : Proband.
: Proband.  : Died from gastric cancer.
: Died from gastric cancer.  : Male.
: Male.  : Female. E1: endoscopic evaluation. E2: molecular evaluation. In red, the two individuals in which DGC was confirmed.
: Female. E1: endoscopic evaluation. E2: molecular evaluation. In red, the two individuals in which DGC was confirmed.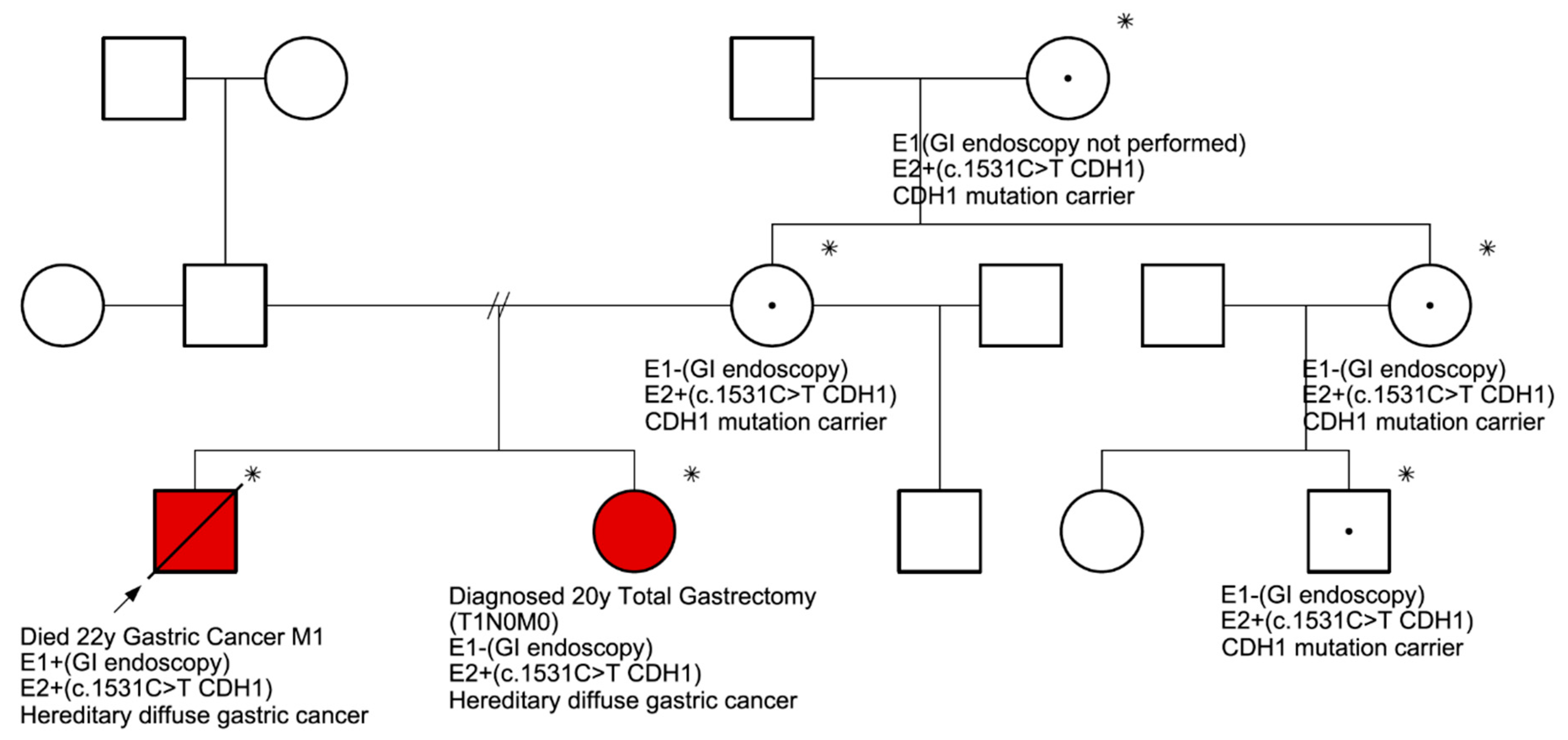
| Characteristics | N (%) |
|---|---|
| Total | 36 |
| Gender | |
| Male | 18 (50) |
| Female | 18 (50) |
| Age (years) | 44 (range: 22–68) |
| ≤ 29 | 7 (19) |
| 30–39 | 7 (19) |
| 40–49 | 12 (33) |
| 50–59 | 4 (11) |
| 60 or older | 6 (16) |
| Smoking status | |
| Current smoker | 16 (44) |
| Previously smoker | 3 (8) |
| Non smoker | 17 (47) |
| Stage | |
| I | 5 (14) |
| II | 6 (17) |
| III | 12 (33) |
| IV | 13 (36) |
| Family history of gastric cancer * | |
| 1 family member (only proband) | 17 (47) |
| 2 family members | 6 (17) |
| 3 family members | 9 (25) |
| 4 or more family members | 4 (11) |
| Family history of breast cancer | 6 (16) |
| Family history of colorectal cancer | 2 (6) |
| Family history of prostate cancer | 3 (8) |
| Family history of uterine cancer | 2 (6) |
| Family history of skin cancer | 1 (3) |
| Family history of esophageal cancer | 1 (3) |
| Family history of gallbladder cancer | 1 (3) |
| Family history of lung cancer | 1 (3) |
| Family history of thyroid cancer | 1 (3) |
| Family history of ovarian cancer | 1 (3) |
| Family history of other cancer unknown | 6 (16) |
| Helicobacter pylori infection | |
| Infected | 15 (42) |
| Not infected | 10 (28) |
| Unknown | 11 (30) |
| SNP ID | Sequence Variant | Protein Change | Probands (n) | Location | Class | Significance α |
|---|---|---|---|---|---|---|
| rs16260 | c.-285C>A | 19 | Promoter | Promoter | Non-coding | |
| rs28372783 | c.-197A>C | 11 | Promoter | Promoter | Non-coding | |
| rs3743674 | c.48+6C>T | 36 | Intron 1 | Splice site | Benign | |
| rs139866691 | c.88C>A | p.Pro30Thr | 1 | Exon 2 | Missense | Benign |
| rs33963999 | c.531+10G>C | 1 | Intron 4 | Splice site | Benign | |
| rs61756284 | c.1272A>T | p.Thr424Thr | 1 | Exon 9 | Synonymous | Benign |
| rs1131690810 | c.1531C>T | p.Gln511 * | 1 | Exon 10 | Nonsense | Pathogenic |
| rs786201452 | c.1893A>T | p.Thr631Thr | 1 | Exon 12 | Synonymous | Likely benign |
| rs764379691 | c.2052C>T | p.Ser684Ser | 1 | Exon 13 | Synonymous | Likely benign |
| rs1801552 | c.2076T>C | p.Ala692Ala | 30 | Exon 13 | Synonymous | Benign |
| rs33964119 | c.2253C>T | p.Asn751Asn | 4 | Exon 14 | Synonymous | Benign |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Norero, E.; Alarcon, M.A.; Hakkaart, C.; de Mayo, T.; Mellado, C.; Garrido, M.; Aguayo, G.; Lagos, M.; Torres, J.; Calvo, A.; et al. Identification of c.1531C>T Pathogenic Variant in the CDH1 Gene as a Novel Germline Mutation of Hereditary Diffuse Gastric Cancer. Int. J. Mol. Sci. 2019, 20, 4980. https://doi.org/10.3390/ijms20204980
Norero E, Alarcon MA, Hakkaart C, de Mayo T, Mellado C, Garrido M, Aguayo G, Lagos M, Torres J, Calvo A, et al. Identification of c.1531C>T Pathogenic Variant in the CDH1 Gene as a Novel Germline Mutation of Hereditary Diffuse Gastric Cancer. International Journal of Molecular Sciences. 2019; 20(20):4980. https://doi.org/10.3390/ijms20204980
Chicago/Turabian StyleNorero, Enrique, M. Alejandra Alarcon, Christopher Hakkaart, Tomas de Mayo, Cecilia Mellado, Marcelo Garrido, Gloria Aguayo, Marcela Lagos, Javiera Torres, Alfonso Calvo, and et al. 2019. "Identification of c.1531C>T Pathogenic Variant in the CDH1 Gene as a Novel Germline Mutation of Hereditary Diffuse Gastric Cancer" International Journal of Molecular Sciences 20, no. 20: 4980. https://doi.org/10.3390/ijms20204980
APA StyleNorero, E., Alarcon, M. A., Hakkaart, C., de Mayo, T., Mellado, C., Garrido, M., Aguayo, G., Lagos, M., Torres, J., Calvo, A., Guilford, P., & Corvalan, A. H. (2019). Identification of c.1531C>T Pathogenic Variant in the CDH1 Gene as a Novel Germline Mutation of Hereditary Diffuse Gastric Cancer. International Journal of Molecular Sciences, 20(20), 4980. https://doi.org/10.3390/ijms20204980





