Expression of Histidine Decarboxylase and Its Roles in Inflammation
Abstract
1. Introduction
2. Biological Effects of Histamine
2.1. Signaling and Distribution of Histamine Receptors
2.2. Negative Regulation of Immune and Inflammatory Cell Functions by H2 Receptors In Vitro
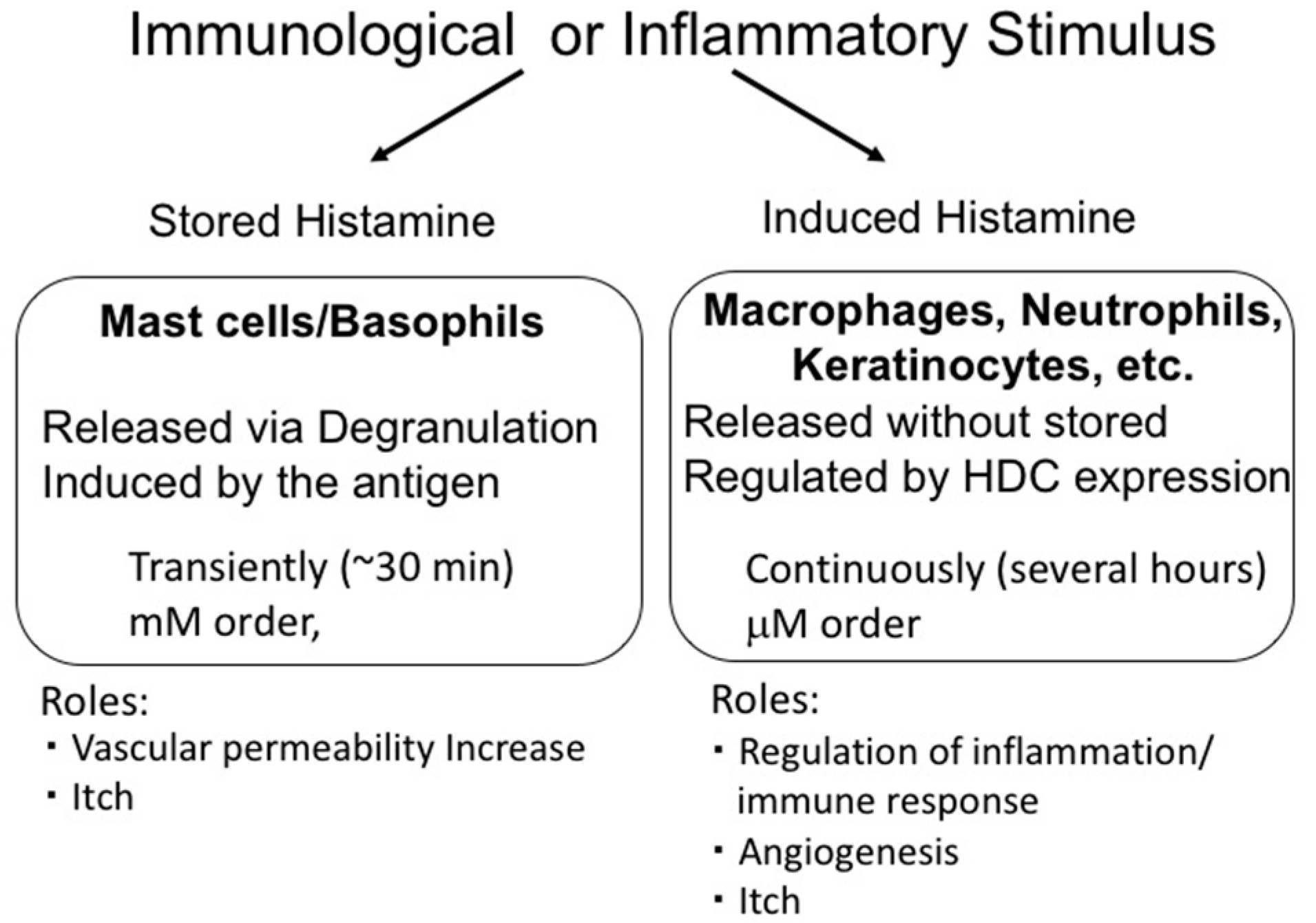
3. Two Modes of Histamine Release
4. Histamine Release from Mast Cells
5. Histamine Production In Vivo and the Functions of Induced Histamine
5.1. Endotoxin-Induced Inflammation
5.2. Dermatitis
5.3. Allergic Inflammation
5.4. Metal-Induced Inflammation
5.5. Granulation Tissues
6. Regulation of HDC Expression
6.1. Biochemical Features of HDC
6.2. HDC Expression in Mast Cells
6.3. HDC Expression in Non-Mast Cells
7. Conclusions
Funding
Conflicts of Interest
Abbreviations
| HDC | histidine decarboxylase |
| IL | interleukin |
| MCP-1 | monocyte chemoattractant protein-1 |
| MIP-2 | macrophage inflammatory protein-2 |
| SDS | sodium dodecyl sulfate |
| TNF | tumor necrosis factor |
| VEGF | vascular endothelial growth factor |
References
- Branco, A.; Yoshikawa, F.S.Y.; Pietrobon, A.J.; Sato, M.N. Role of Histamine in Modulating the Immune Response and Inflammation. Mediat. Inflamm. 2018, 2018, 9524075. [Google Scholar] [CrossRef] [PubMed]
- Deng, X.; Wu, X.; Yu, Z.; Arai, I.; Sasano, T.; Sugawara, S.; Endo, Y. Inductions of histidine decarboxylase in mouse tissues following systemic antigen challenge: Contributions made by mast cells, non-mast cells and IL-1. Int. Arch. Allergy Immunol. 2007, 144, 69–78. [Google Scholar] [CrossRef] [PubMed]
- Pini, A.; Grange, C.; Veglia, E.; Argenziano, M.; Cavalli, R.; Guasti, D.; Calosi, L.; Ghe, C.; Solarino, R.; Thurmond, R.L.; et al. Histamine H4 receptor antagonism prevents the progression of diabetic nephropathy in male DBA2/J mice. Pharmacol. Res. 2018, 128, 18–28. [Google Scholar] [CrossRef]
- Cowden, J.M.; Yu, F.; Banie, H.; Farahani, M.; Ling, P.; Nguyen, S.; Riley, J.P.; Zhang, M.; Zhu, J.; Dunford, P.J.; et al. The histamine H4 receptor mediates inflammation and Th17 responses in preclinical models of arthritis. Ann. Rheum. Dis. 2014, 73, 600–608. [Google Scholar] [CrossRef] [PubMed]
- Varga, C.; Horvath, K.; Berko, A.; Thurmond, R.L.; Dunford, P.J.; Whittle, B.J. Inhibitory effects of histamine H4 receptor antagonists on experimental colitis in the rat. Eur. J. Pharmacol. 2005, 522, 130–138. [Google Scholar] [CrossRef]
- Rivera, J.; Fierro, N.A.; Olivera, A.; Suzuki, R. New insights on mast cell activation via the high affinity receptor for IgE. Adv. Immunol. 2008, 98, 85–120. [Google Scholar] [PubMed]
- Tiligade, E.; Ennis, M. Histamine pharmacology: From Sir Henry Dale to the 21st century. Br. J. Pharmacol. 2018. [Google Scholar] [CrossRef]
- Akdis, C.A.; Simons, F.E. Histamine receptors are hot in immunopharmacology. Eur. J. Pharmacol. 2006, 533, 69–76. [Google Scholar] [CrossRef]
- Lippert, U.; Artuc, M.; Grutzkau, A.; Babina, M.; Guhl, S.; Haase, I.; Blaschke, V.; Zachmann, K.; Knosalla, M.; Middel, P.; et al. Human skin mast cells express H2 and H4, but not H3 receptors. J. Investig. Dermatol. 2004, 123, 116–123. [Google Scholar] [CrossRef]
- Masini, E.; Blandina, P.; Brunelleschi, S.; Mannaioni, P.F. Evidence for H2-receptor-mediated inhibition of histamine release from isolated rat mast cells. Agents Actions 1982, 12, 85–88. [Google Scholar] [CrossRef]
- Lichtenstein, L.M.; Gillespie, E. Inhibition of histamine release by histamine controlled by H2 receptor. Nature 1973, 244, 287–288. [Google Scholar] [CrossRef] [PubMed]
- Bissonnette, E.Y. Histamine inhibits tumor necrosis factor alpha release by mast cells through H2 and H3 receptors. Am. J. Respir. Cell Mol. Biol. 1996, 14, 620–626. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.R.; Zweiman, B. Histamine suppression of human lymphocyte responses to mitogens. Cell. Immunol. 1978, 36, 28–36. [Google Scholar] [CrossRef]
- Anderson, R.; Glover, A.; Rabson, A.R. The in vitro effects of histamine and metiamide on neutrophil motility and their relationship to intracellular cyclic nucleotide levels. J. Immunol. 1977, 118, 1690–1696. [Google Scholar] [PubMed]
- Lett-Brown, M.A.; Leonard, E.J. Histamine-induced inhibition of normal human basophil chemotaxis to C5a. J. Immunol. 1977, 118, 815–818. [Google Scholar] [PubMed]
- Azuma, Y.; Shinohara, M.; Wang, P.L.; Hidaka, A.; Ohura, K. Histamine inhibits chemotaxis, phagocytosis, superoxide anion production, and the production of TNFalpha and IL-12 by macrophages via H2-receptors. Int. Immunopharmacol. 2001, 1, 1867–1875. [Google Scholar] [CrossRef]
- Elenkov, I.J.; Webster, E.; Papanicolaou, D.A.; Fleisher, T.A.; Chrousos, G.P.; Wilder, R.L. Histamine potently suppresses human IL-12 and stimulates IL-10 production via H2 receptors. J. Immunol. 1998, 161, 2586–2593. [Google Scholar]
- Nishibori, M.; Takahashi, H.K.; Mori, S. The regulation of ICAM-1 and LFA-1 interaction by autacoids and statins: A novel strategy for controlling inflammation and immune responses. J. Pharmacol. Sci. 2003, 92, 7–12. [Google Scholar] [CrossRef]
- Caron, G.; Delneste, Y.; Roelandts, E.; Duez, C.; Bonnefoy, J.Y.; Pestel, J.; Jeannin, P. Histamine polarizes human dendritic cells into Th2 cell-promoting effector dendritic cells. J. Immunol. 2001, 167, 3682–3686. [Google Scholar] [CrossRef]
- Mazzoni, A.; Young, H.A.; Spitzer, J.H.; Visintin, A.; Segal, D.M. Histamine regulates cytokine production in maturing dendritic cells, resulting in altered T cell polarization. J. Clin. Investig. 2001, 108, 1865–1873. [Google Scholar] [CrossRef]
- Zhang, J.; Takahashi, H.K.; Liu, K.; Wake, H.; Liu, R.; Sadamori, H.; Matsuda, H.; Yagi, T.; Yoshino, T.; Mori, S.; et al. Histamine inhibits adhesion molecule expression in human monocytes, induced by advanced glycation end products, during the mixed lymphocyte reaction. Br. J. Pharmacol. 2010, 160, 1378–1386. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, H.K.; Yoshida, A.; Iwagaki, H.; Yoshino, T.; Itoh, H.; Morichika, T.; Yokoyama, M.; Akagi, T.; Tanaka, N.; Mori, S.; et al. Histamine regulation of interleukin-18-initiating cytokine cascade is associated with down-regulation of intercellular adhesion molecule-1 expression in human peripheral blood mononuclear cells. J. Pharmacol. Exp. Ther. 2002, 300, 227–235. [Google Scholar] [CrossRef]
- Teuscher, C.; Poynter, M.E.; Offner, H.; Zamora, A.; Watanabe, T.; Fillmore, P.D.; Zachary, J.F.; Blankenhorn, E.P. Attenuation of Th1 effector cell responses and susceptibility to experimental allergic encephalomyelitis in histamine H2 receptor knockout mice is due to dysregulation of cytokine production by antigen-presenting cells. Am. J. Pathol. 2004, 164, 883–892. [Google Scholar] [CrossRef]
- Ohtsu, H.; Tanaka, S.; Terui, T.; Hori, Y.; Makabe-Kobayashi, Y.; Pejler, G.; Tchougounova, E.; Hellman, L.; Gertsenstein, M.; Hirasawa, N.; et al. Mice lacking histidine decarboxylase exhibit abnormal mast cells. FEBS Lett. 2001, 502, 53–56. [Google Scholar] [CrossRef]
- Kobayashi, T.; Tonai, S.; Ishihara, Y.; Koga, R.; Okabe, S.; Watanabe, T. Abnormal functional and morphological regulation of the gastric mucosa in histamine H2 receptor-deficient mice. J. Clin. Investig. 2000, 105, 1741–1749. [Google Scholar] [CrossRef] [PubMed]
- Ma, R.Z.; Gao, J.; Meeker, N.D.; Fillmore, P.D.; Tung, K.S.; Watanabe, T.; Zachary, J.F.; Offner, H.; Blankenhorn, E.P.; Teuscher, C. Identification of Bphs, an autoimmune disease locus, as histamine receptor H1. Science 2002, 297, 620–623. [Google Scholar] [CrossRef] [PubMed]
- Ali, H. Mas-related G protein coupled receptor-X2: A potential new target for modulating mast cell-mediated allergic and inflammatory diseases. J. Immunobiol. 2016. [Google Scholar] [CrossRef]
- Subramanian, H.; Gupta, K.; Ali, H. Roles of Mas-related G protein-coupled receptor X2 on mast cell-mediated host defense, pseudoallergic drug reactions, and chronic inflammatory diseases. J. Allergy Clin. Immunol. 2016, 138, 700–710. [Google Scholar] [CrossRef]
- Fernandes, H.S.; Ramos, M.J.; Cerqueira, N. The Catalytic Mechanism of the Pyridoxal-5′-phosphate-Dependent Enzyme, Histidine Decarboxylase: A Computational Study. Chemistry 2017, 23, 9162–9173. [Google Scholar] [CrossRef]
- Yamaguchi, K.; Motegi, K.; Kurimoto, M.; Endo, Y. Induction of the activity of the histamine-forming enzyme, histidine decarboxylase, in mice by IL-18 and by IL-18 plus IL-12. Inflamm Res. 2000, 49, 513–519. [Google Scholar] [CrossRef]
- Yamaguchi, K.; Motegi, K.; Endo, Y. Induction of histidine decarboxylase, the histamine-forming enzyme, in mice by interleukin-12. Toxicology 2000, 156, 57–65. [Google Scholar] [CrossRef]
- Kawaguchi-Nagata, K.; Okamura, H.; Tamura, T.; Yamatodani, A.; Watanabe, T.; Wada, H.; Taguchi, T.; Kitamura, Y.; Shoji, K. Induction of histidine decarboxylase activity in the spleen of mice treated with staphylococcal enterotoxin A and demonstration of its non-mast cell origin. Biochem. Biophys. Res. Commun. 1985, 129, 187–192. [Google Scholar] [CrossRef]
- Endo, Y.; Nakamura, M.; Nitta, Y.; Kumagai, K. Effects of macrophage depletion on the induction of histidine decarboxylase by lipopolysaccharide, interleukin 1 and tumour necrosis factor. Br. J. Pharmacol. 1995, 114, 187–193. [Google Scholar] [CrossRef] [PubMed]
- Hori, Y.; Nihei, Y.; Kurokawa, Y.; Kuramasu, A.; Makabe-Kobayashi, Y.; Terui, T.; Doi, H.; Satomi, S.; Sakurai, E.; Nagy, A.; et al. Accelerated clearance of Escherichia coli in experimental peritonitis of histamine-deficient mice. J. Immunol. 2002, 169, 1978–1983. [Google Scholar] [CrossRef]
- Hattori, M.; Yamazaki, M.; Ohashi, W.; Tanaka, S.; Hattori, K.; Todoroki, K.; Fujimori, T.; Ohtsu, H.; Matsuda, N.; Hattori, Y. Critical role of endogenous histamine in promoting end-organ tissue injury in sepsis. Intensive Care Med. Exp. 2016, 4, 36. [Google Scholar] [CrossRef] [PubMed]
- Inami, Y.; Sasaki, A.; Andoh, T.; Kuraishi, Y. Surfactant-induced chronic pruritus: Role of L-histidine decarboxylase expression and histamine production in epidermis. Acta Derm. Venereol. 2014, 94, 645–650. [Google Scholar] [CrossRef]
- Inami, Y.; Andoh, T.; Sasaki, A.; Kuraishi, Y. Topical surfactant-induced pruritus: Involvement of histamine released from epidermal keratinocytes. J. Pharmacol. Exp. Ther. 2013, 344, 459–466. [Google Scholar] [CrossRef]
- Gutowska-Owsiak, D.; Greenwald, L.; Watson, C.; Selvakumar, T.A.; Wang, X.; Ogg, G.S. The histamine-synthesizing enzyme histidine decarboxylase is upregulated by keratinocytes in atopic skin. Br. J. Dermatol. 2014, 171, 771–778. [Google Scholar] [CrossRef]
- Hirasawa, N.; Ohuchi, K.; Watanabe, M.; Tsurufuji, S. Role of endogenous histamine in postanaphylactic phase of allergic inflammation in rats. J. Pharmacol. Exp. Ther. 1987, 241, 967–973. [Google Scholar]
- Ohuchi, K.; Hirasawa, N.; Takeda, H.; Asano, K.; Watanabe, M.; Tsurufuji, S. Mechanism of antianaphylactic action of beta-agonists in allergic inflammation of air pouch type in rats. Int. Arch. Allergy Appl. Immunol. 1987, 82, 26–32. [Google Scholar] [CrossRef]
- Shiraishi, M.; Hirasawa, N.; Oikawa, S.; Kobayashi, Y.; Ohuchi, K. Analysis of histamine-producing cells at the late phase of allergic inflammation in rats. Immunology 2000, 99, 600–606. [Google Scholar] [CrossRef] [PubMed]
- Hirasawa, N.; Funaba, Y.; Hirano, Y.; Kawarasaki, K.; Omata, M.; Watanabe, M.; Mue, S.; Tsurufuji, S.; Ohuchi, K. Inhibition by dexamethasone of histamine production in allergic inflammation in rats. J. Immunol. 1990, 145, 3041–3046. [Google Scholar] [PubMed]
- Hirasawa, N.; Ohtsu, H.; Watanabe, T.; Ohuchi, K. Enhancement of neutrophil infiltration in histidine decarboxylase-deficient mice. Immunology 2002, 107, 217–221. [Google Scholar] [CrossRef] [PubMed]
- Ohsawa, Y.; Hirasawa, N. The antagonism of histamine H1 and H4 receptors ameliorates chronic allergic dermatitis via anti-pruritic and anti-inflammatory effects in NC/Nga mice. Allergy 2012, 67, 1014–1022. [Google Scholar] [CrossRef]
- Asakawa, S.; Onodera, R.; Kasai, K.; Kishimoto, Y.; Sato, T.; Segawa, R.; Mizuno, N.; Ogasawara, K.; Moriya, T.; Hiratsuka, M.; et al. Nickel ions bind to HSP90beta and enhance HIF-1alpha-mediated IL-8 expression. Toxicology 2018, 395, 45–53. [Google Scholar] [CrossRef] [PubMed]
- Wong, B.W.; Kuchnio, A.; Bruning, U.; Carmeliet, P. Emerging novel functions of the oxygen-sensing prolyl hydroxylase domain enzymes. Trends Biochem. Sci. 2013, 38, 3–11. [Google Scholar] [CrossRef] [PubMed]
- Hirasawa, N.; Goi, Y.; Tanaka, R.; Ishihara, K.; Ohtsu, H.; Ohuchi, K. Involvement of prostaglandins and histamine in nickel wire-induced acute inflammation in mice. J. Biomed. Mater. Res. A 2010, 93, 1306–1311. [Google Scholar] [CrossRef] [PubMed]
- Kishimoto, Y.; Asakawa, S.; Sato, T.; Takano, T.; Nakajyo, T.; Mizuno, N.; Segawa, R.; Yoshikawa, T.; Hiratsuka, M.; Yanai, K.; et al. Induced histamine regulates Ni elution from an implanted Ni wire in mice by downregulating neutrophil migration. Exp. Dermatol. 2017, 26, 868–874. [Google Scholar] [CrossRef] [PubMed]
- Sato, T.; Kishimoto, Y.; Asakawa, S.; Mizuno, N.; Hiratsuka, M.; Hirasawa, N. Involvement of COX-2 in nickel elution from a wire implanted subcutaneously in mice. Toxicology 2016, 363–364, 37–45. [Google Scholar] [CrossRef]
- Sandberg, N. Enhanced Rate of Healing in Rats with an Increased Rate of Histamine Formation. Acta Chir. Scand. 1964, 127, 9–21. [Google Scholar]
- Hirasawa, N.; Watanabe, M.; Mue, S.; Tsurufuji, S.; Ohuchi, K. Downward regulation of neutrophil infiltration by endogenous histamine without affecting vascular permeability responses in air-pouch-type carrageenin inflammation in rats. Inflammation 1991, 15, 117–126. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, A.K.; Hirasawa, N.; Ohtsu, H.; Watanabe, T.; Ohuchi, K. Defective angiogenesis in the inflammatory granulation tissue in histidine decarboxylase-deficient mice but not in mast cell-deficient mice. J. Exp. Med. 2002, 195, 973–982. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, A.K.; Hirasawa, N.; Ohuchi, K. Enhancement by histamine of vascular endothelial growth factor production in granulation tissue via H(2) receptors. Br. J. Pharmacol. 2001, 134, 1419–1428. [Google Scholar] [CrossRef] [PubMed]
- Ohmori, E.; Fukui, T.; Imanishi, N.; Yatsunami, K.; Ichikawa, A. Purification and characterization of l-histidine decarboxylase from mouse mastocytoma P-815 cells. J. Biochem. 1990, 107, 834–839. [Google Scholar] [CrossRef] [PubMed]
- Taguchi, Y.; Watanabe, T.; Kubota, H.; Hayashi, H.; Wada, H. Purification of histidine decarvoxylase from the liver of fetal rats and its immunochemical and immunohistochemical characterization. J. Bio. Chem. 1984, 259, 5214–5221. [Google Scholar]
- Yamamoto, J.; Yatsunami, K.; Ohmori, E.; Sugimoto, Y.; Fukui, T.; Katayama, T.; Ichikawa, A. cDNA-derived amino acid sequence of L-histidine decarboxylase from mouse mastocytoma P-815 cells. FEBS Lett. 1990, 276, 214–218. [Google Scholar] [CrossRef]
- Joseph, D.R.; Sullivan, P.M.; Wang, Y.M.; Kozak, C.; Fenstermacher, D.A.; Behrendsen, M.E.; Zahnow, C.A. Characterization and expression of the complementary DNA encoding rat histidine decarboxylase. Proc. Natl. Acad. Sci. USA 1990, 87, 733–737. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, S.; Nemoto, K.; Yamamura, E.; Ichikawa, A. Intracellular localization of the 74- and 53-kDa forms of L-histidine decarboxylase in a rat basophilic/mast cell line, RBL-2H3. J. Biol. Chem. 1998, 273, 8177–8182. [Google Scholar] [CrossRef]
- Fennell, L.M.; Fleming, J.V. Differential processing of mammalian L-histidine decarboxylase enzymes. Biochem. Biophys. Res. Commun. 2014, 445, 304–309. [Google Scholar] [CrossRef]
- Fleming, J.V.; Wang, T.C. Amino- and carboxy-terminal PEST domains mediate gastrin stabilization of rat L-histidine decarboxylase isoforms. Mol. Cell. Biol. 2000, 20, 4932–4947. [Google Scholar] [CrossRef]
- Fleming, J.V.; Wang, T.C. The production of 53-55-kDa isoforms is not required for rat L-histidine decarboxylase activity. J. Biol. Chem. 2003, 278, 686–694. [Google Scholar] [CrossRef] [PubMed]
- Yatsunami, K.; Ohtsu, H.; Tsuchikawa, M.; Higuchi, T.; Ishibashi, K.; Shida, A.; Shima, Y.; Nakagawa, S.; Yamauchi, K.; Yamamoto, M.; et al. Structure of the L-histidine decarboxylase gene. J. Biol. Chem. 1994, 269, 1554–1559. [Google Scholar] [PubMed]
- Ohtsu, H.; Kuramasu, A.; Suzuki, S.; Igarashi, K.; Ohuchi, Y.; Sato, M.; Tanaka, S.; Nakagawa, S.; Shirato, K.; Yamamoto, M.; et al. Histidine decarboxylase expression in mouse mast cell line P815 is induced by mouse peritoneal cavity incubation. J. Biol. Chem. 1996, 271, 28439–28444. [Google Scholar] [CrossRef] [PubMed]
- Kuramasu, A.; Saito, H.; Suzuki, S.; Watanabe, T.; Ohtsu, H. Mast cell-/basophil-specific transcriptional regulation of human L-histidine decarboxylase gene by CpG methylation in the promoter region. J. Biol. Chem. 1998, 273, 31607–31614. [Google Scholar] [CrossRef] [PubMed]
- Suzuki-Ishigaki, S.; Numayama-Tsuruta, K.; Kuramasu, A.; Sakurai, E.; Makabe, Y.; Shimura, S.; Shirato, K.; Igarashi, K.; Watanabe, T.; Ohtsu, H. The mouse L-histidine decarboxylase gene: Structure and transcriptional regulation by CpG methylation in the promoter region. Nucleic Acids Res. 2000, 28, 2627–2633. [Google Scholar] [CrossRef]
- Imanishi, N.; Nakayama, T.; Asano, M.; Yatsunami, K.; Tomita, K.; Ichikawa, A. Induction of histidine decarboxylase by dexamethasone in mastocytoma P-815 cells. Biochim. Biophys. Acta 1987, 928, 227–234. [Google Scholar] [CrossRef]
- Li, Y.; Liu, B.; Harmacek, L.; Long, Z.; Liang, J.; Lukin, K.; Leach, S.M.; O’Connor, B.; Gerber, A.N.; Hagman, J.; et al. The transcription factors GATA2 and microphthalmia-associated transcription factor regulate Hdc gene expression in mast cells and are required for IgE/mast cell-mediated anaphylaxis. J. Allergy Clin. Immunol. 2018, 142, 1173–1184. [Google Scholar] [CrossRef]
- Tanaka, S.; Takasu, Y.; Mikura, S.; Satoh, N.; Ichikawa, A. Antigen-independent induction of histamine synthesis by immunoglobulin E in mouse bone marrow-derived mast cells. J. Exp. Med. 2002, 196, 229–235. [Google Scholar] [CrossRef]
- Miyazaki, T.; Ohgoh, M.; Ohmori, E.; Yamamoto, J.; Emoto, S.; Yatsunami, K.; Ichikawa, A. Synergistic effects of cyclic AMP and Ca2+ ionophore A23187 on de novo synthesis of histidine decarboxylase in mastocytoma P-815 cells. Biochim. Biophys. Acta 1992, 1133, 179–186. [Google Scholar] [CrossRef]
- Kawai, H.; Ohgoh, M.; Emoto, S.; Ohmori, E.; Imanishi, N.; Yatsunami, K.; Ichikawa, A. Synergistic effects of 12-O-tetradecanoylphorbol-13-acetate and dexamethasone on de novo synthesis of histidine decarboxylase in mouse mastocytoma P-815 cells. Biochim. Biophys. Acta 1992, 1133, 172–178. [Google Scholar] [CrossRef]
- Mori, Y.; Akedo, H.; Taniguchi, Y.; Tanaka, K.M.; Okada, M.; Nakamuar, N. Effect of sodium butyrate on the production of serotonin, histamine and glycosaminoglycans by cultured murine mastocytoma cells. Exp. Cell Res. 1980, 127, 465–470. [Google Scholar] [CrossRef]
- Hirasawa, N.; Torigoe, M.; Ohgawara, R.; Murakami, A.; Ohuchi, K. Involvement of MAP kinases in lipopolysaccharide-induced histamine production in RAW 264 cells. Life Sci. 2006, 80, 36–42. [Google Scholar] [CrossRef] [PubMed]
- Shiraishi, M.; Hirasawa, N.; Kobayashi, Y.; Oikawa, S.; Murakami, A.; Ohuchi, K. Participation of mitogen-activated protein kinase in thapsigargin- and TPA-induced histamine production in murine macrophage RAW 264.7 cells. Br. J. Pharmacol. 2000, 129, 515–524. [Google Scholar] [CrossRef] [PubMed]
- Hirasawa, N.; Torigoe, M.; Kano, K.; Ohuchi, K. Involvement of Sp1 in lipopolysaccharide-induced expression of HDC mRNA in RAW 264 cells. Biochem. Biophys. Res. Commun. 2006, 349, 833–837. [Google Scholar] [CrossRef]
- Hirasawa, N.; Murakami, A.; Ohuchi, K. Expression of 74-kDa histidine decarboxylase protein in a macrophage-like cell line RAW 264.7 and inhibition by dexamethasone. Eur. J. Pharmacol. 2001, 418, 23–28. [Google Scholar] [CrossRef]
- Nakazawa, S.; Sakanaka, M.; Furuta, K.; Natsuhara, M.; Takano, H.; Tsuchiya, S.; Okuno, Y.; Ohtsu, H.; Nishibori, M.; Thurmond, R.L.; et al. Histamine synthesis is required for granule maturation in murine mast cells. Eur. J. Immunol. 2014, 44, 204–214. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, S.; Mikura, S.; Hashimoto, E.; Sugimoto, Y.; Ichikawa, A. Ca2+ influx-mediated histamine synthesis and IL-6 release in mast cells activated by monomeric IgE. Eur. J. Immunol. 2005, 35, 460–468. [Google Scholar] [CrossRef]
- Kawaguchi-Nagata, K.; Watanabe, T.; Yamatodani, A.; Inoue, M.; Asai, H.; Tamura, T.; Wada, H.; Shoji, K.; Kitamura, Y. In vitro increase of histidine decarboxylase activity and release of histamine by peritoneal resident cells of mast cell-deficient W/Wv mice; possible involvement of macrophages. J. Biochem. 1988, 103, 24–30. [Google Scholar] [CrossRef]
- Takamatsu, S.; Nakano, K. Histamine synthesis by bone marrow-derived macrophages. Biosci. Biotechnol. Biochem. 1994, 58, 1918–1919. [Google Scholar] [CrossRef]
- Kawaguchi-Nagata, K.; Watanabe, T.; Maeyama, K.; Yamatodani, A.; Okamura, H.; Tamura, T.; Shoji, K.; Kitamura, Y. Increase of histidine decarboxylase activity in murine myelomonocytic leukemia cells (WEHI-3B) in parallel to their differentiation into macrophages. Biochim. Biophys. Acta 1988, 972, 249–256. [Google Scholar]
- Alcaniz, L.; Vega, A.; Chacon, P.; El Bekay, R.; Ventura, I.; Aroca, R.; Blanca, M.; Bergstralh, D.T.; Monteseirin, J. Histamine production by human neutrophils. FASEB J. 2013, 27, 2902–2910. [Google Scholar] [CrossRef]
- Aoi, R.; Nakashima, I.; Kitamura, Y.; Asai, H.; Nakano, K. Histamine synthesis by mouse T lymphocytes through induced histidine decarboxylase. Immunology 1989, 66, 219–223. [Google Scholar] [PubMed]
- Kubo, Y.; Nakano, K. Regulation of histamine synthesis in mouse CD4+ and CD8+ T lymphocytes. Inflamm. Res. 1999, 48, 149–153. [Google Scholar] [CrossRef] [PubMed]
- Nagashima, Y.; Kako, K.; Kim, J.D.; Fukamizu, A. Enhanced histamine production through the induction of histidine decarboxylase expression by phorbol ester in Jurkat cells. Mol. Med. Rep. 2012, 6, 944–948. [Google Scholar] [CrossRef] [PubMed]
- Tippens, A.S.; Davis, S.V.; Hayes, J.R.; Bryda, E.C.; Green, T.L.; Gruetter, C.A. Detection of histidine decarboxylase in rat aorta and cultured rat aortic smooth muscle cells. Inflamm. Res. 2004, 53, 390–395. [Google Scholar] [CrossRef]
- Tippens, A.S.; Gruetter, C.A. Detection of histidine decarboxylase mRNA in human vascular smooth muscle and endothelial cells. Inflamm. Res. 2004, 53, 215–216. [Google Scholar] [CrossRef] [PubMed]
| Cells | Stimulants | Ref. |
|---|---|---|
| Mast cells | differentiation | [66] |
| A23187 + cAMP | [69] | |
| Dex + TPA | [70] | |
| granule maturation | [76] | |
| IgE | [68,77] | |
| Non-mast cells | ||
| Macrophages | ||
| peritoneal macrophages | [78] | |
| BMDM | LPS | [79] |
| RAW264 | LPS | [72] |
| TPA | [73] | |
| Thapsigargin | [73] | |
| WEHI-3B | differentiation, LPS | [80] |
| Neutrophils | LPS | [81] |
| Lymphocytes | ||
| T cells | Con A | [82] |
| CD4+, CD8+ | Con A | [83] |
| Jarkat | TPA | [84] |
| Keratinocytes | SDS | [37] |
| LPS, cytokines | [38] | |
| Endothelial cells | [85] | |
| Smooth muscle cells | [86] | |
© 2019 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hirasawa, N. Expression of Histidine Decarboxylase and Its Roles in Inflammation. Int. J. Mol. Sci. 2019, 20, 376. https://doi.org/10.3390/ijms20020376
Hirasawa N. Expression of Histidine Decarboxylase and Its Roles in Inflammation. International Journal of Molecular Sciences. 2019; 20(2):376. https://doi.org/10.3390/ijms20020376
Chicago/Turabian StyleHirasawa, Noriyasu. 2019. "Expression of Histidine Decarboxylase and Its Roles in Inflammation" International Journal of Molecular Sciences 20, no. 2: 376. https://doi.org/10.3390/ijms20020376
APA StyleHirasawa, N. (2019). Expression of Histidine Decarboxylase and Its Roles in Inflammation. International Journal of Molecular Sciences, 20(2), 376. https://doi.org/10.3390/ijms20020376




