Biological Properties of Calcium Phosphate Bioactive Glass Composite Bone Substitutes: Current Experimental Evidence
Abstract
1. Introduction
2. Methods
3. Overview
4. The Rationale behind CaP/BG Composite Materials for Application in Bone Tissue Engineering
5. CaP-Types Used as Part of the Composite and Their Influence on Biological Properties
6. BG Compositions Used
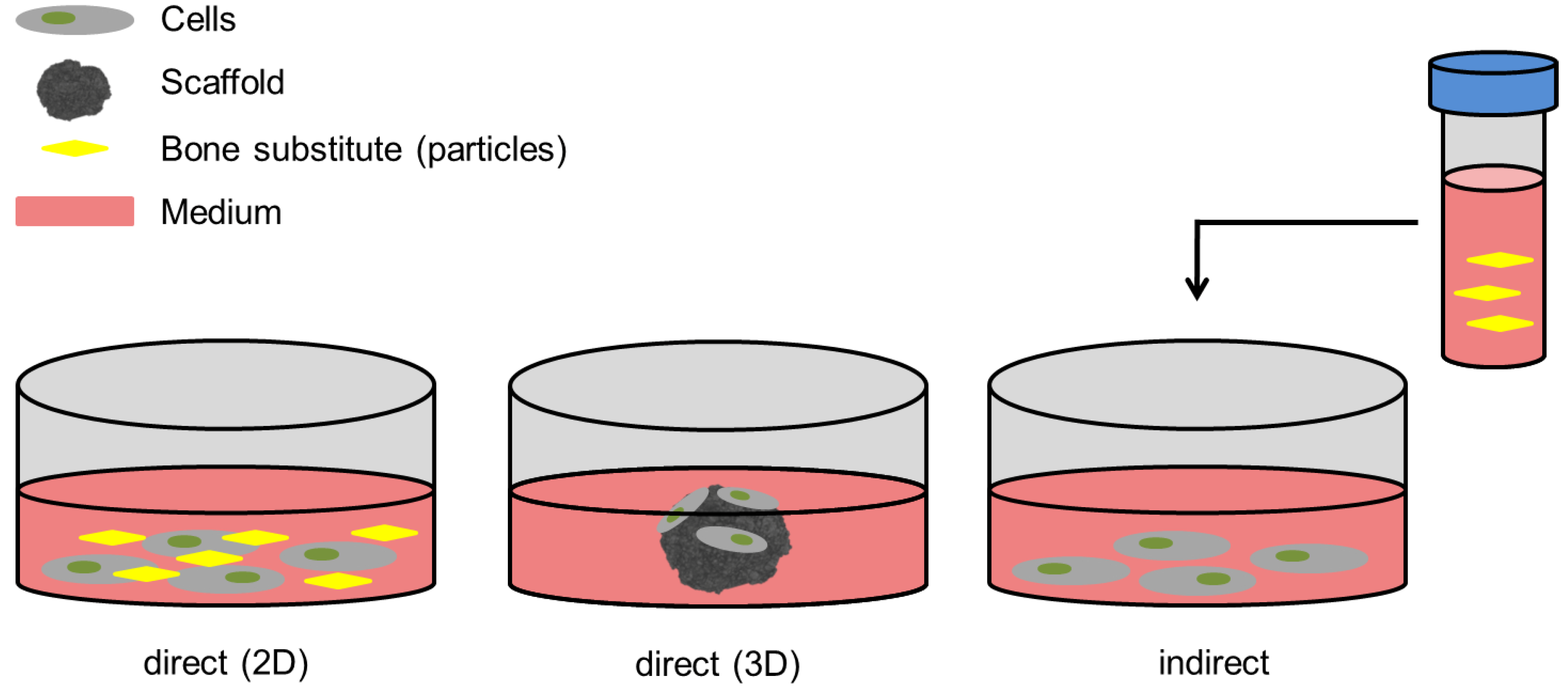
7. In-Vitro Evaluation Models
7.1. Analysis of Cell Morphology, Adhesion and Surface Interaction
7.2. Analysis of Cell Viability, Cytotoxicity, and Proliferation
7.3. Analysis of Osteogenic Differentiation
7.4. Cell Culture Media Used
8. In-Vivo Evaluation Models
9. Biological Properties of Composite Materials
9.1. Cell Vitality (In-Vitro)
9.2. Osteogenic Differentiation (In-Vitro)
9.3. Microscopy (In-Vitro)
9.4. Microscopy (In-Vivo)
10. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| 2D | two-dimensional |
| 3D | three-dimensional |
| AA | ascorbic acid |
| ALP | alkaline phosphatase |
| AOC | alkylene oxide copolymer |
| ASC | adipose tissue-derived mesenchymal stem cells |
| BALB/3T3 | mouse embryonic fibroblast cell line |
| BG | bioactive glass |
| CaP | calcium phosphate |
| Col-I | collagen type I |
| DM | dexamethasone |
| DMEM | Dulbecco’s modified eagle medium |
| EDS | energy dispersion spectroscopy |
| ELISA | Enzyme-linked Immunosorbent Assay |
| G-292 | human osteosarcoma cells |
| GM | growth medium |
| HA | hydroxyapatite |
| hBMSC | human bone marrow stromal cells |
| HCA | hydroxyapatite-like |
| i.m. | intramuscular |
| LDH | lactate dehydrogenase |
| MC3T3-E1 | mouse osteoblast precursor cell line |
| MG 63 | osteoblast-like cells |
| MLO-Y4 | murine long bone osteocyte-like immortalized cell line |
| MM | maintenance medium |
| MSC | mesenchymal stem cells |
| MTT | 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide |
| MTS | 3-(4,5-dimethylthiazol-2-yl)-5-(3-carboxymethoxyphenyl)-2-(4-sulfophenyl)-2H-tetrazolium, inner salt |
| n/s | not specified |
| Na2O | alkali |
| NRU | Neutral Red Uptake Test |
| OC | osteocalcin |
| ODM | osteogenic differentiation medium |
| OP | osteopontin |
| qPCR | quantitative real-time PCR analysis |
| rMSC | rat bone marrow-derived mesenchymal stem cells |
| rOMSC | osteoporotic rat bone marrow-derived mesenchymal stem cells |
| RT-PCR | Reverse transcriptase PCR |
| Runx2 | Runt-related transcription factor 2 |
| SaOS-2 | osteoblast-like cells |
| SEM | scanning electron microscopy |
| TCP | tricalcium phosphate |
| XTT | 2,3-bis(2-methoxy-4-nitro-5-sulfophenyl)-5-[(phenylamino)carbonyl]-2H-tetrazolium hydroxide |
| β-GP | β-glycerophosphate |
References
- Wang, W.; Yeung, K.W.K. Bone grafts and biomaterials substitutes for bone defect repair: A review. Bioact. Mater. 2017, 2, 224–247. [Google Scholar] [CrossRef] [PubMed]
- Giannoudis, P.V.; Einhorn, T.A.; Schmidmaier, G.; Marsh, D. The diamond concept—Open questions. Injury 2008, 39, S5–S8. [Google Scholar] [CrossRef]
- Amini, A.R.; Laurencin, C.T.; Nukavarapu, S.P. Bone tissue engineering: Recent advances and challenges. Crit. Rev. Biomed. Eng. 2012, 40, 363–408. [Google Scholar] [CrossRef] [PubMed]
- Dimitriou, R.; Mataliotakis, G.I.; Angoules, A.G.; Kanakaris, N.K.; Giannoudis, P.V. Complications following autologous bone graft harvesting from the iliac crest and using the RIA: A systematic review. Injury 2011, 42, S3–S15. [Google Scholar] [CrossRef] [PubMed]
- Arrington, E.D.; Smith, W.J.; Chambers, H.G.; Bucknell, A.L.; Davino, N.A. Complications of iliac crest bone graft harvesting. Clin. Orthop. Relat. Res. 1996, 329, 300–309. [Google Scholar] [CrossRef]
- Giannoudis, P.V.; Dinopoulos, H.; Tsiridis, E. Bone substitutes: An update. Injury 2005, 36, S20–S27. [Google Scholar] [CrossRef]
- Westhauser, F.; Senger, A.S.; Reible, B.; Moghaddam, A. In Vivo Models for the Evaluation of the Osteogenic Potency of Bone Substitutes Seeded with Mesenchymal Stem Cells of Human Origin: A Concise Review. Tissue Eng. Part C Methods 2017, 23, 881–888. [Google Scholar] [CrossRef]
- Zimmermann, G.; Moghaddam, A. Allograft bone matrix versus synthetic bone graft substitutes. Injury 2011, 42, S16–S21. [Google Scholar] [CrossRef]
- Cao, W.; Hench, L.L. Bioactive materials. Ceram. Int. 1996, 22, 493–507. [Google Scholar] [CrossRef]
- Hoppe, A.; Guldal, N.S.; Boccaccini, A.R. A review of the biological response to ionic dissolution products from bioactive glasses and glass-ceramics. Biomaterials 2011, 32, 2757–2774. [Google Scholar] [CrossRef]
- Laskus, A.; Kolmas, J. Ionic Substitutions in Non-Apatitic Calcium Phosphates. Int. J. Mol. Sci. 2017, 18, 2542. [Google Scholar] [CrossRef]
- Kolmas, J.; Velard, F.; Jaguszewska, A.; Lemaire, F.; Kerdjoudj, H.; Gangloff, S.C.; Kaflak, A. Substitution of strontium and boron into hydroxyapatite crystals: Effect on physicochemical properties and biocompatibility with human Wharton-Jelly stem cells. Mater. Sci. Eng. C 2017, 79, 638–646. [Google Scholar] [CrossRef] [PubMed]
- Rath, S.N.; Strobel, L.A.; Arkudas, A.; Beier, J.P.; Maier, A.K.; Greil, P.; Horch, R.E.; Kneser, U. Osteoinduction and survival of osteoblasts and bone-marrow stromal cells in 3D biphasic calcium phosphate scaffolds under static and dynamic culture conditions. J. Cell Mol. Med. 2012, 16, 2350–2361. [Google Scholar] [CrossRef] [PubMed]
- El-Rashidy, A.A.; Roether, J.A.; Harhaus, L.; Kneser, U.; Boccaccini, A.R. Regenerating bone with bioactive glass scaffolds: A review of in vivo studies in bone defect models. Acta Biomater. 2017, 62, 1–28. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Chen, S.-K.; Li, L.; Qin, L.; Wang, X.-L.; Lai, Y.-X. Bone defect animal models for testing efficacy of bone substitute biomaterials. J. Orthop. Transl. 2015, 3, 95–104. [Google Scholar] [CrossRef] [PubMed]
- Bellucci, D.; Sola, A.; Cannillo, V. Hydroxyapatite and tricalcium phosphate composites with bioactive glass as second phase: State of the art and current applications. J. Biomed. Mater. Res. A 2016, 104, 1030–1056. [Google Scholar] [CrossRef] [PubMed]
- LeGeros, R.Z. Calcium phosphate-based osteoinductive materials. Chem. Rev. 2008, 108, 4742–4753. [Google Scholar] [CrossRef]
- Bellucci, D.; Anesi, A.; Salvatori, R.; Chiarini, L.; Cannillo, V. A comparative in vivo evaluation of bioactive glasses and bioactive glass-based composites for bone tissue repair. Mater. Sci. Eng. C Mater. Biol. Appl. 2017, 79, 286–295. [Google Scholar] [CrossRef]
- Bellucci, D.; Sola, A.; Anesi, A.; Salvatori, R.; Chiarini, L.; Cannillo, V. Bioactive glass/hydroxyapatite composites: Mechanical properties and biological evaluation. Mater. Sci. Eng. C Mater. Biol. Appl. 2015, 51, 196–205. [Google Scholar] [CrossRef]
- Hannink, G.; Arts, J.J.C. Bioresorbability, porosity and mechanical strength of bone substitutes: What is optimal for bone regeneration? Injury 2011, 42, S22–S25. [Google Scholar] [CrossRef]
- Hench, L.L.; Splinter, R.J.; Allen, W.C.; Greenlee, T.K. Bonding mechanisms at the interface of ceramic prosthetic materials. J. Biomed. Mater. Res. 1971, 5, 117–141. [Google Scholar] [CrossRef]
- Balasubramanian, P.; Hupa, L.; Jokic, B.; Detsch, R.; Grünewald, A.; Boccaccini, A.R. Angiogenic potential of boron-containing bioactive glasses: In vitro study. J. Mater. Sci. 2017, 52, 8785–8792. [Google Scholar] [CrossRef]
- Ojansivu, M.; Vanhatupa, S.; Bjorkvik, L.; Hakkanen, H.; Kellomaki, M.; Autio, R.; Ihalainen, J.A.; Hupa, L.; Miettinen, S. Bioactive glass ions as strong enhancers of osteogenic differentiation in human adipose stem cells. Acta Biomater. 2015, 21, 190–203. [Google Scholar] [CrossRef] [PubMed]
- Gerhardt, L.C.; Boccaccini, A.R. Bioactive Glass and Glass-Ceramic Scaffolds for Bone Tissue Engineering. Materials 2010, 3, 3867–3910. [Google Scholar] [CrossRef]
- Westhauser, F.; Ciraldo, F.; Balasubramanian, P.; Senger, A.S.; Schmidmaier, G.; Moghaddam, A.; Boccaccini, A.R. Micro-Computed-Tomography-Guided Analysis of In Vitro Structural Modifications in Two Types of 45S5 Bioactive Glass Based Scaffolds. Materials 2017, 10, 1341. [Google Scholar] [CrossRef] [PubMed]
- Hench, L.L.; Jones, J.R. Bioactive Glasses: Frontiers and Challenges. Front. Bioeng. Biotechnol. 2015, 3, 194. [Google Scholar] [CrossRef]
- Chen, Q.Z.; Thompson, I.D.; Boccaccini, A.R. 45S5 Bioglass-derived glass-ceramic scaffolds for bone tissue engineering. Biomaterials 2006, 27, 2414–2425. [Google Scholar] [CrossRef] [PubMed]
- Philippart, A.; Boccaccini, A.R.; Fleck, C.; Schubert, D.W.; Roether, J.A. Toughening and functionalization of bioactive ceramic and glass bone scaffolds by biopolymer coatings and infiltration: A review of the last 5 years. Expert Rev. Med. Devices 2015, 12, 93–111. [Google Scholar] [CrossRef]
- Turnbull, G.; Clarke, J.; Picard, F.; Riches, P.; Jia, L.; Han, F.; Li, B.; Shu, W. 3D bioactive composite scaffolds for bone tissue engineering. Bioact. Mater. 2017, 3, 278–314. [Google Scholar] [CrossRef]
- Clupper, D.C.; Hench, L.L. Crystallization kinetics of tape cast bioactive glass 45S5. J. Non-Cryst. Solids 2003, 318, 43–48. [Google Scholar] [CrossRef]
- Boccaccini, A.R.; Chen, Q.; Lefebvre, L.; Gremillard, L.; Chevalier, J. Sintering, crystallisation and biodegradation behaviour of Bioglass®-derived glass-ceramics. Faraday Discuss. 2007, 136, 27. [Google Scholar] [CrossRef] [PubMed]
- Begum, S.; Johnson, W.E.; Worthington, T.; Martin, R.A. The influence of pH and fluid dynamics on the antibacterial efficacy of 45S5 Bioglass. Biomed. Mater. 2016, 11, 015006. [Google Scholar] [CrossRef] [PubMed]
- Rahaman, M.N.; Day, D.E.; Bal, B.S.; Fu, Q.; Jung, S.B.; Bonewald, L.F.; Tomsia, A.P. Bioactive glass in tissue engineering. Acta Biomater. 2011, 7, 2355–2373. [Google Scholar] [CrossRef] [PubMed]
- Kaysinger, K.K.; Ramp, W.K. Extracellular pH modulates the activity of cultured human osteoblasts. J. Cell Biochem. 1998, 68, 83–89. [Google Scholar] [CrossRef]
- Arnett, T.R. Extracellular pH regulates bone cell function. J. Nutr. 2008, 138, 415S–418S. [Google Scholar] [CrossRef] [PubMed]
- Galow, A.M.; Rebl, A.; Koczan, D.; Bonk, S.M.; Baumann, W.; Gimsa, J. Increased osteoblast viability at alkaline pH in vitro provides a new perspective on bone regeneration. Biochem. Biophys. Rep. 2017, 10, 17–25. [Google Scholar] [CrossRef] [PubMed]
- Westhauser, F.; Weis, C.; Prokscha, M.; Bittrich, L.A.; Li, W.; Xiao, K.; Kneser, U.; Kauczor, H.U.; Schmidmaier, G.; Boccaccini, A.R.; et al. Three-dimensional polymer coated 45S5-type bioactive glass scaffolds seeded with human mesenchymal stem cells show bone formation in vivo. J. Mater. Sci. Mater. Med. 2016, 27, 119. [Google Scholar] [CrossRef] [PubMed]
- Deschaseaux, F.; Sensébé, L.; Heymann, D. Mechanisms of bone repair and regeneration. Trends Mol. Med. 2009, 15, 417–429. [Google Scholar] [CrossRef]
- Pryce, R.S.; Hench, L.L. Dissolution Characteristics of Bioactive Glasses. Key Eng. Mater. 2002, 240–242, 201–204. [Google Scholar] [CrossRef]
- Detsch, R.; Alles, S.; Hum, J.; Westenberger, P.; Sieker, F.; Heusinger, D.; Kasper, C.; Boccaccini, A.R. Osteogenic differentiation of umbilical cord and adipose derived stem cells onto highly porous 45S5 Bioglass®-based scaffolds. J. Biomed. Mater. Res. Part A 2015, 103, 1029–1037. [Google Scholar] [CrossRef]
- Ciraldo, F.E.; Boccardi, E.; Melli, V.; Westhauser, F.; Boccaccini, A.R. Tackling bioactive glass excessive in vitro bioreactivity: Preconditioning approaches for cell culture tests. Acta Biomater. 2018, 75, 3–10. [Google Scholar] [CrossRef] [PubMed]
- Liberati, A.; Altman, D.G.; Tetzlaff, J.; Mulrow, C.; Gotzsche, P.C.; Ioannidis, J.P.; Clarke, M.; Devereaux, P.J.; Kleijnen, J.; Moher, D. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: Explanation and elaboration. PLoS Med. 2009, 6, e1000100. [Google Scholar] [CrossRef]
- Barbieri, D.; Yuan, H.; Ismailoglu, A.S.; de Bruijn, J.D. Comparison of Two Moldable Calcium Phosphate-Based Bone Graft Materials in a Noninstrumented Canine Interspinous Implantation Model. Tissue Eng. Part A 2017, 23, 1310–1320. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.Y.; Xu, S.Z.; Wang, X.W.; Yang, X.Y.; Ma, L.; Zhang, L.; Yang, G.J.; Yang, F.; Wang, L.H.; Zhang, X.L.; et al. Systematic comparison of biologically active foreign ions-codoped calcium phosphate microparticles on osteogenic differentiation in rat osteoporotic and normal mesenchymal stem cells. Oncotarget 2017, 8, 36578–36590. [Google Scholar] [CrossRef]
- Lopes, J.H.; Magalhães, J.A.; Gouveia, R.F.; Bertran, C.A.; Motisuke, M.; Camargo, S.E.A.; Trichês, E.D.S. Hierarchical structures of β-TCP/45S5 bioglass hybrid scaffolds prepared by gelcasting. J. Mech. Behav. Biomed. Mater. 2016, 62, 10–23. [Google Scholar] [CrossRef] [PubMed]
- Lu, Z.; Roohani-Esfahani, S.I.; Li, J.; Zreiqat, H. Synergistic effect of nanomaterials and BMP-2 signalling in inducing osteogenic differentiation of adipose tissue-derived mesenchymal stem cells. Nanomedicine 2015, 11, 219–228. [Google Scholar] [CrossRef] [PubMed]
- Bernhardt, A.; Lode, A.; Peters, F.; Gelinsky, M. Comparative evaluation of different calcium phosphate-based bone graft granules—An in vitro study with osteoblast-like cells. Clin. Oral Implants Res. 2013, 24, 441–449. [Google Scholar] [CrossRef]
- Bellucci, D.; Sola, A.; Gazzarri, M.; Chiellini, F.; Cannillo, V. A new hydroxyapatite-based biocomposite for bone replacement. Mater. Sci. Eng. C 2013, 33, 1091–1101. [Google Scholar] [CrossRef]
- Cholewa-Kowalska, K.; Kokoszka, J.; Laczka, M.; Niedzwiedzki, L.; Madej, W.; Osyczka, A.M. Gel-derived bioglass as a compound of hydroxyapatite composites. Biomed. Mater. 2009, 4, 055007. [Google Scholar] [CrossRef]
- Hesaraki, S.; Safari, M.; Shokrgozar, M.A. Composite bone substitute materials based on β-tricalcium phosphate and magnesium-containing sol-gel derived bioactive glass. J. Mater. Sci. Mater. Med. 2009, 20, 2011–2017. [Google Scholar] [CrossRef]
- Yu, X.; Cai, S.; Xu, G.; Zhou, W.; Wang, D. Low temperature fabrication of high strength porous calcium phosphate and the evaluation of the osteoconductivity. J. Mater. Sci. Mater. Med. 2009, 20, 2025–2034. [Google Scholar] [CrossRef] [PubMed]
- Kucukkolbasi, H.; Mutlu, N.; Isik, K.; Celik, I.; Oznurlu, Y. Histological evaluation of the effects of bioglass, hydroxyapatite, or demineralized freeze-dried bone, grafted alone or as composites, on the healing of tibial defects in rabbits. Saudi Med. J. 2009, 30, 329–333. [Google Scholar] [PubMed]
- Haimi, S.; Moimas, L.; Pirhonen, E.; Lindroos, B.; Huhtala, H.; Raty, S.; Kuokkanen, H.; Sandor, G.K.; Miettinen, S.; Suuronen, R. Calcium phosphate surface treatment of bioactive glass causes a delay in early osteogenic differentiation of adipose stem cells. J. Biomed. Mater. Res. A 2009, 91, 540–547. [Google Scholar] [CrossRef] [PubMed]
- Wiltfang, J.; Merten, H.A.; Schlegel, K.A.; Schultze-Mosgau, S.; Kloss, F.R.; Rupprecht, S.; Kessler, P. Degradation characteristics of alpha and beta tri-calcium-phosphate (TCP) in minipigs. J. Biomed. Mater. Res. 2002, 63, 115–121. [Google Scholar] [CrossRef] [PubMed]
- Baino, F.; Hamzehlou, S.; Kargozar, S. Bioactive Glasses: Where Are We and Where Are We Going? J. Funct. Biomater. 2018, 9, 25. [Google Scholar] [CrossRef] [PubMed]
- Xin, R.; Leng, Y.; Chen, J.; Zhang, Q. A comparative study of calcium phosphate formation on bioceramics in vitro and in vivo. Biomaterials 2005, 26, 6477–6486. [Google Scholar] [CrossRef] [PubMed]
- Jones, J.R. Review of bioactive glass: From Hench to hybrids. Acta Biomater. 2013, 9, 4457–4486. [Google Scholar] [CrossRef]
- Qazi, T.H.; Hafeez, S.; Schmidt, J.; Duda, G.N.; Boccaccini, A.R.; Lippens, E. Comparison of the effects of 45S5 and 1393 bioactive glass microparticles on hMSC behavior. J. Biomed. Mater. Res. A 2017, 105, 2772–2782. [Google Scholar] [CrossRef]
- Leibig, N.; Wietbrock, J.O.; Bigdeli, A.K.; Horch, R.E.; Kremer, T.; Kneser, U.; Schmidt, V.J. Flow-Induced Axial Vascularization: The Arteriovenous Loop in Angiogenesis and Tissue Engineering. Plast. Reconstr. Surg. 2016, 138, 825–835. [Google Scholar] [CrossRef]
- Reible, B.; Schmidmaier, G.; Prokscha, M.; Moghaddam, A.; Westhauser, F. Continuous stimulation with differentiation factors is necessary to enhance osteogenic differentiation of human mesenchymal stem cells in-vitro. Growth Factors 2017, 35, 179–188. [Google Scholar] [CrossRef]
- Reible, B.; Schmidmaier, G.; Moghaddam, A.; Westhauser, F. Insulin-Like Growth Factor-1 as a Possible Alternative to Bone Morphogenetic Protein-7 to Induce Osteogenic Differentiation of Human Mesenchymal Stem Cells in Vitro. Int. J. Mol. Sci. 2018, 19, 1674. [Google Scholar] [CrossRef]
- Pettersson, L.F.; Kingham, P.J.; Wiberg, M.; Kelk, P. In Vitro Osteogenic Differentiation of Human Mesenchymal Stem Cells from Jawbone Compared with Dental Tissue. Tissue Eng. Regen. Med. 2017, 14, 763–774. [Google Scholar] [CrossRef] [PubMed]
- Carmichael, J.; DeGraff, W.G.; Gazdar, A.F.; Minna, J.D.; Mitchell, J.B. Evaluation of a tetrazolium-based semiautomated colorimetric assay: Assessment of chemosensitivity testing. Cancer Res. 1987, 47, 936–942. [Google Scholar] [PubMed]
- Ashton, B.A.; Abdullah, F.; Cave, J.; Williamson, M.; Sykes, B.C.; Couch, M.; Poser, J.W. Characterization of cells with high alkaline phosphatase activity derived from human bone and marrow: Preliminary assessment of their osteogenicity. Bone 1985, 6, 313–319. [Google Scholar] [CrossRef]
- Marie, P.J.; Lomri, A.; Sabbagh, A.; Basle, M. Culture and behavior of osteoblastic cells isolated from normal trabecular bone surfaces. In Vitro Cell. Dev. Biol. 1989, 25, 373–380. [Google Scholar] [CrossRef] [PubMed]
- Koshihara, Y.; Kawamura, M.; Oda, H.; Higaki, S. In vitro calcification in human osteoblastic cell line derived from periosteum. Biochem. Biophys. Res. Commun. 1987, 145, 651–657. [Google Scholar] [CrossRef]
- Beresford, J.N.; Gallagher, J.A.; Russell, R.G. 1,25-Dihydroxyvitamin D3 and human bone-derived cells in vitro: Effects on alkaline phosphatase, type I collagen and proliferation. Endocrinology 1986, 119, 1776–1785. [Google Scholar] [CrossRef]
- Tan, I.-K.; Chio, L.-F.; Teow-Suah, L. Heat stability of human serum alkaline phosphatase in bone and liver diseases. Clin. Chim. Acta 1972, 41, 329–334. [Google Scholar] [CrossRef]
- Hinnebusch, B.F.; Siddique, A.; Henderson, J.W.; Malo, M.S.; Zhang, W.; Athaide, C.P.; Abedrapo, M.A.; Chen, X.; Yang, V.W.; Hodin, R.A. Enterocyte differentiation marker intestinal alkaline phosphatase is a target gene of the gut-enriched Kruppel-like factor. Am. J. Physiol. Gastrointest. Liver Physiol. 2004, 286, G23–G30. [Google Scholar] [CrossRef]
- Bonewald, L.F. The amazing osteocyte. J. Bone Miner. Res. 2011, 26, 229–238. [Google Scholar] [CrossRef]
- Ragni, E.; Vigano, M.; Rebulla, P.; Giordano, R.; Lazzari, L. What is beyond a qRT-PCR study on mesenchymal stem cell differentiation properties: How to choose the most reliable housekeeping genes. J. Cell Mol. Med. 2013, 17, 168–180. [Google Scholar] [CrossRef] [PubMed]
- El-Gendy, R.; Yang, X.B.; Newby, P.J.; Boccaccini, A.R.; Kirkham, J. Osteogenic differentiation of human dental pulp stromal cells on 45S5 Bioglass® based scaffolds in vitro and in vivo. Tissue Eng. Part A 2013, 19, 707–715. [Google Scholar] [CrossRef]
- Lievremont, M.; Potus, J.; Guillou, B. Use of alizarin red S for histochemical staining of Ca2+ in the mouse; some parameters of the chemical reaction in vitro. Acta Anatom. 1982, 114, 268–280. [Google Scholar] [CrossRef]
- Stanford, C.M.; Jacobson, P.A.; Eanes, E.D.; Lembke, L.A.; Midura, R.J. Rapidly forming apatitic mineral in an osteoblastic cell line (UMR 106-01 BSP). J. Biol. Chem. 1995, 270, 9420–9428. [Google Scholar] [CrossRef]
- Puchtler, H.; Meloan, S.N.; Terry, M.S. On the history and mechanism of alizarin and alizarin red S stains for calcium. J. Histochem. Cytochem. 1969, 17, 110–124. [Google Scholar] [CrossRef] [PubMed]
- Cai, S.; Xu, G.H.; Yu, X.Z.; Zhang, W.J.; Xiao, Z.Y.; Yao, K.D. Fabrication and biological characteristics of β-tricalcium phosphate porous ceramic scaffolds reinforced with calcium phosphate glass. J. Mater. Sci. Mater. Med. 2009, 20, 351–358. [Google Scholar] [CrossRef] [PubMed]
- Langenbach, F.; Handschel, J. Effects of dexamethasone, ascorbic acid and β-glycerophosphate on the osteogenic differentiation of stem cells in vitro. Stem Cell Res. Ther. 2013, 4, 117. [Google Scholar] [CrossRef]
- Song, I.H.; Caplan, A.I.; Dennis, J.E. In vitro dexamethasone pretreatment enhances bone formation of human mesenchymal stem cells in vivo. J. Orthop. Res. 2009, 27, 916–921. [Google Scholar] [CrossRef]
- Birmingham, E.; Niebur, G.L.; McHugh, P.E.; Shaw, G.; Barry, F.P.; McNamara, L.M. Osteogenic differentiation of mesenchymal stem cells is regulated by osteocyte and osteoblast cells in a simplified bone niche. Eur. Cell Mater. 2012, 23, 13–27. [Google Scholar] [CrossRef]
- Hale, L.V.; Ma, Y.F.; Santerre, R.F. Semi-Quantitative Fluorescence Analysis of Calcein Binding as a Measurement of In Vitro Mineralization. Calcif. Tissue Int. 2000, 67, 80–84. [Google Scholar] [CrossRef]
- Moore, W.R.; Graves, S.E.; Bain, G.I. Synthetic bone graft substitutes. ANZ J. Surg. 2001, 71, 8. [Google Scholar] [CrossRef]
- Sheikh, Z.; Najeeb, S.; Khurshid, Z.; Verma, V.; Rashid, H.; Glogauer, M. Biodegradable Materials for Bone Repair and Tissue Engineering Applications. Materials 2015, 8, 5744–5794. [Google Scholar] [CrossRef] [PubMed]
- Detsch, R.; Boccaccini, A.R. The role of osteoclasts in bone tissue engineering. J. Tissue Eng. Regen. Med. 2015, 9, 1133–1149. [Google Scholar] [CrossRef] [PubMed]
- Brito, A.F.; Antunes, B.; Dos Santos, F.; Fernandes, H.R.; Ferreira, J.M.F. Osteogenic capacity of alkali-free bioactive glasses. In vitro studies. J. Biomed. Mater. Res. B Appl. Biomater. 2017, 105, 2360–2365. [Google Scholar] [CrossRef] [PubMed]
- Detsch, R.; Rubner, M.; Strissel, P.L.; Mohn, D.; Strasser, E.; Stark, W.J.; Strick, R.; Boccaccini, A.R. Nanoscale bioactive glass activates osteoclastic differentiation of RAW 264.7 cells. Nanomedicine 2016, 11, 1093–1105. [Google Scholar] [CrossRef]
- Westhauser, F.; Reible, B.; Hollig, M.; Heller, R.; Schmidmaier, G.; Moghaddam, A. Combining advantages: Direct correlation of two-dimensional microcomputed tomography datasets onto histomorphometric slides to quantify three-dimensional bone volume in scaffolds. J. Biomed. Mater. Res. A 2018, 106, 1812–1821. [Google Scholar] [CrossRef]
- Steffens, L.; Wenger, A.; Stark, G.B.; Finkenzeller, G. In vivo engineering of a human vasculature for bone tissue engineering applications. J. Cell Mol. Med. 2009, 13, 3380–3386. [Google Scholar] [CrossRef]
| First Author | Ref. | Year | Composite | Percentage | Setup | Methods/Endpoints | Biological Outcome | ||
|---|---|---|---|---|---|---|---|---|---|
| 1st Phase | 2nd Phase | 1st Phase | 2nd Phase | ||||||
| Barbieri | [43] | 2017 | HA/β-TCP 4/96 | AOC | 51.7 | 48.3 | in-vivo | Histomorphometry | Compared to CaP/AOC rarely any formation of new bone on CaP/BG composite. |
| β-TCP | 45S5 (45 wt% SiO2, 24.5 wt% CaO, 24.5 wt% Na2O, 6 wt% P2O5)-Collagen mix | 61.8 | 38.2 | ||||||
| Bellucci | [18] | 2017 | HA | BG_Ca/Mix (47.3% SiO2, 45.6% CaO, 2.3% K2O, 2.3% Na2O, 2.6% P2O5) | 70/20/0 | 30/80/100 | in-vivo | X-Ray, histomorphometry | Increased osteoconductivity of pure BG scaffolds compared to composites, with best results for BG_Ca/Mix. Improved osteoconductivity of CaP/BG composites with increasing BG content. |
| 45S5 | / | 100 | 0 | ||||||
| Chen | [44] | 2017 | Si-Sr-Zn-Mg-codoped CaP | 45S5 | 100/6.7/0 | 0/93.3/100 | in-vitro | Cell proliferation, osteogenic differentiation, protein expression | Improved proliferation and differentiation of mesenchymal stem cells in composite materials. |
| Lopes | [45] | 2016 | β-TCP | 45S5 | 100/95/92.5 | 0/5/7.5 | in-vitro | Cell viability, SEM | Increased cell viability and advanced attachment in 7.5%-composites. |
| Bellucci | [19] | 2015 | HA | BG_Ca/Mix | 20/70 | 80/30 | in-vitro | Cell viability, cell proliferation, cytotoxicity | Cell viability in composites equivalent to 45S5-BG. |
| 45S5 | / | 100 | 0 | ||||||
| Lu | [46] | 2015 | HA/β-TCP (40/60) | 58S (60% SiO2, 36% CaO, 4% P2O5) | n/s | n/s | in-vitro | Cell viability, osteogenic differentiation | Synergistic effect of BG and CaP on osteogenic differentiation, further enhanced by BMP-2. |
| HA/β-TCP (40/60) | / | 100 | 0 | ||||||
| Bernhardt | [47] | 2013 | β-TCP | Na-Mg-Si-BG system | 96 | 4 | in-vitro | Cell adhesion, osteogenic differentiation, cell viability | Cell viability and number increased or equal for pure β-TCP vs. β-TCP/BG composites. No differences in osteogenic performance (ALP activity) for pure β-TCP vs. β-TCP/BG after 28 days of incubation. Unless pre-incubated, HA/β-TCP/BG composites and HA/BG composites did reduce cell number compared to pure β-TCP. |
| HA/β-TCP (60/40) | SiO2 matrix | n/s | n/s | ||||||
| HA | SiO2 matrix | 76 | 24 | ||||||
| β-TCP | / | 100 | 0 | ||||||
| Bellucci | [48] | 2013 | HA | BG_Ca (47.3% SiO2, 45.6% CaO, 4.6% Na2O, 2.6% P2O5) | 80/60 | 20/40 | in-vitro | Cell adhesion, cell proliferation, cell viability, osteogenic differentiation | Increased osteogenic differentiation for BG_Ca composites (ALP activity) vs. 45S5-BG composites. Increased osteogenic differentiation with increasing BG content in both composites. No major differences in cell proliferation between the different composites. |
| 45S5 | |||||||||
| Cholewa-Kowalska | [49] | 2009 | HA | S2 (80% SiO2, 16% CaO, 4% P2O5) | 100/90/50/0 | 0/10/50/100 | in-vitro | Cell viability, osteogenic differentiation | Osteogenic differentiation (ALP activity) significantly improved for the 50:50 S2 composite. |
| A2 (40% SiO2, 54% CaO, 6% P2O5) | |||||||||
| Hesaraki | [50] | 2009 | β-TCP | 64% SiO2, 26% CaO, 5% P2O5, 5% MgO | 90/75/60 | 10/25/40 | in-vitro | Cell viability | Improved cell viability in composite materials, regardless of BG content. |
| Yu | [51] | 2009 | β-TCP | 62.04 wt% P2O5, 14.68 wt% CaO, 13 wt% MgO, 10.28 wt% Na2O | 100/80 | 0/20 | in-vivo | SEM, histomorphometry | No differences in in-vivo biocompatibility between pure β-TCP and composite material. |
| Kucukkol-basi | [52] | 2009 | HA | 45S5 | 0/50/100 | 100/50/0 | in-vivo | Histomorphometry | No major differences between the composite and both pure HA and 45S5-BG. |
| Cai | [51] | 2009 | β-TCP | 45 wt% P2O5, 22 wt% CaO, 25 wt% Na2O, 8 wt% MgO | 100/80 | 0/20 | in-vitro | SEM, cell count | BG addition increased cell number as well as attachment and distribution of cells. |
| Haimi | [53] | 2009 | CaP (n/s) | Na2O, K2O, MgO, CaO, B2O3, TiO2, Ca2PO5, SiO2 | n/s | n/s | in-vitro | Cell adhesion, cell viability, cell proliferation, osteogenic differentiation | No remarkable differences between pure BG and CaP-coated BG. |
| First Author | Ref. | Cell Type | Species | Cell Number | Medium | Culture Time | Culture Setting |
|---|---|---|---|---|---|---|---|
| Chen | [44] | rMSC/rOMSC | rat | 100,000/cm2 | MM + Osteogenic factors (β-GP + AA) | 21 d | indirect |
| Lopes | [45] | MG 63 | human | 8000/well (96-well-plate) | MM | 3 d | direct |
| Bellucci | [19] | BALB/3T3 + MLO-Y4 | mouse | n/s | MM | 24 h–48 h | direct and indirect |
| Lu | [46] | ASC | human | 50,000/0.6 cm2/10,000/0.6 cm2 | MM | 14 d | direct |
| Bernhardt | [47] | SaOS-2 | human | 160,000/0.1 2 cm3 | MM + Osteogenic factors (β-GP + AA) | 28 d | direct |
| Bellucci | [48] | MC3T3-E1 | mouse | 50,000/g | GM + ODM (including β-GP + AA) | 21 d | direct |
| Cholewa-Kowalska | [49] | hBMSC | human | 10,000/cm2 | MM + ODM (including DM + AA) | 7 d | direct |
| Hesaraki | [50] | G-292 | human | 30,000/mL | MM | 7 d | direct |
| Cai | [51] | MC3T3-E1 | mouse | 1,000,000/0.375 cm2 | MM | 14 d | direct |
| Haimi | [53] | ASC | human | 500,000/0.98 cm2 | MM | 14 d | direct |
| First Author | Ref. | Species | Location and Defect Size | Implantation Time | Methods | Parameter |
|---|---|---|---|---|---|---|
| Bellucci | [18] | New Zealand Rabbit | Femur (3.5 mm diameter, 7 mm depth) | 2 m | Post-mortem femur X-ray | Graft position |
| Histomorpho-metry | Implant-bone-interface: bone formation, bone healing/contact, cracks of the graft | |||||
| SEM | Bioactive bone bonding mechanism (confirmation of histology) HA-Layer | |||||
| EDS + microradiography | Dissolution kinetics | |||||
| Barbieri | [43] | Mongrel dog | Orthotopic (spine) and ectopic (i.m.) (defect size n/s) | 3 m | Histomorpho-metry | Bone formation, bone-surface-contact, in-vivo resorption rate tissue response (i.m.) |
| Fluorescence microscopy | Bone development (calcein labeling) | |||||
| Yu | [51] | New Zealand Rabbit | Femur (2 mm diameter, depth n/s) | 1/2/3/6 m | Histomorpho-metry | Bone formation, bone-implant-interface, resorption rates |
| SEM | Bone formation, bone-implant-interface, resorption rates | |||||
| Kucukkolbasi | [52] | New Zealand Rabbit | Tibia (3 mm diameter, depth n/s) | 1/3/6 m | Histomorpho-metry | Bone formation, resorption rates, tissue reactions |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Karadjian, M.; Essers, C.; Tsitlakidis, S.; Reible, B.; Moghaddam, A.; Boccaccini, A.R.; Westhauser, F. Biological Properties of Calcium Phosphate Bioactive Glass Composite Bone Substitutes: Current Experimental Evidence. Int. J. Mol. Sci. 2019, 20, 305. https://doi.org/10.3390/ijms20020305
Karadjian M, Essers C, Tsitlakidis S, Reible B, Moghaddam A, Boccaccini AR, Westhauser F. Biological Properties of Calcium Phosphate Bioactive Glass Composite Bone Substitutes: Current Experimental Evidence. International Journal of Molecular Sciences. 2019; 20(2):305. https://doi.org/10.3390/ijms20020305
Chicago/Turabian StyleKaradjian, Maria, Christopher Essers, Stefanos Tsitlakidis, Bruno Reible, Arash Moghaddam, Aldo R. Boccaccini, and Fabian Westhauser. 2019. "Biological Properties of Calcium Phosphate Bioactive Glass Composite Bone Substitutes: Current Experimental Evidence" International Journal of Molecular Sciences 20, no. 2: 305. https://doi.org/10.3390/ijms20020305
APA StyleKaradjian, M., Essers, C., Tsitlakidis, S., Reible, B., Moghaddam, A., Boccaccini, A. R., & Westhauser, F. (2019). Biological Properties of Calcium Phosphate Bioactive Glass Composite Bone Substitutes: Current Experimental Evidence. International Journal of Molecular Sciences, 20(2), 305. https://doi.org/10.3390/ijms20020305







