Brain-Derived Neurotrophic Factor Is Required for the Neuroprotective Effect of Mifepristone on Immature Purkinje Cells in Cerebellar Slice Culture
Abstract
1. Introduction
2. Results
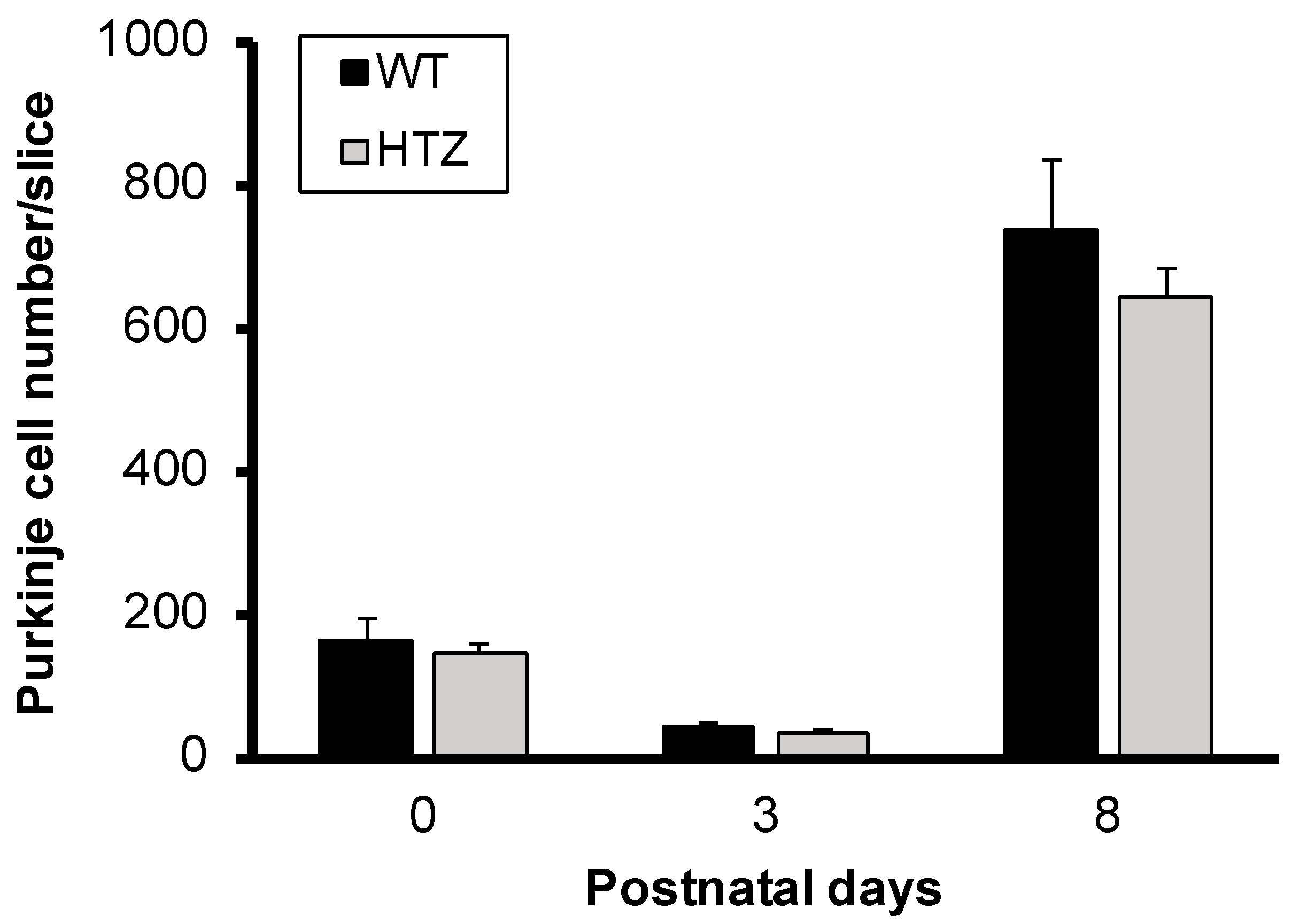
2.1. The Neurotrophin BDNF Is Not Involved in Immature Purkinje Cell Survival in Organotypic Cerebellar Slice Culture
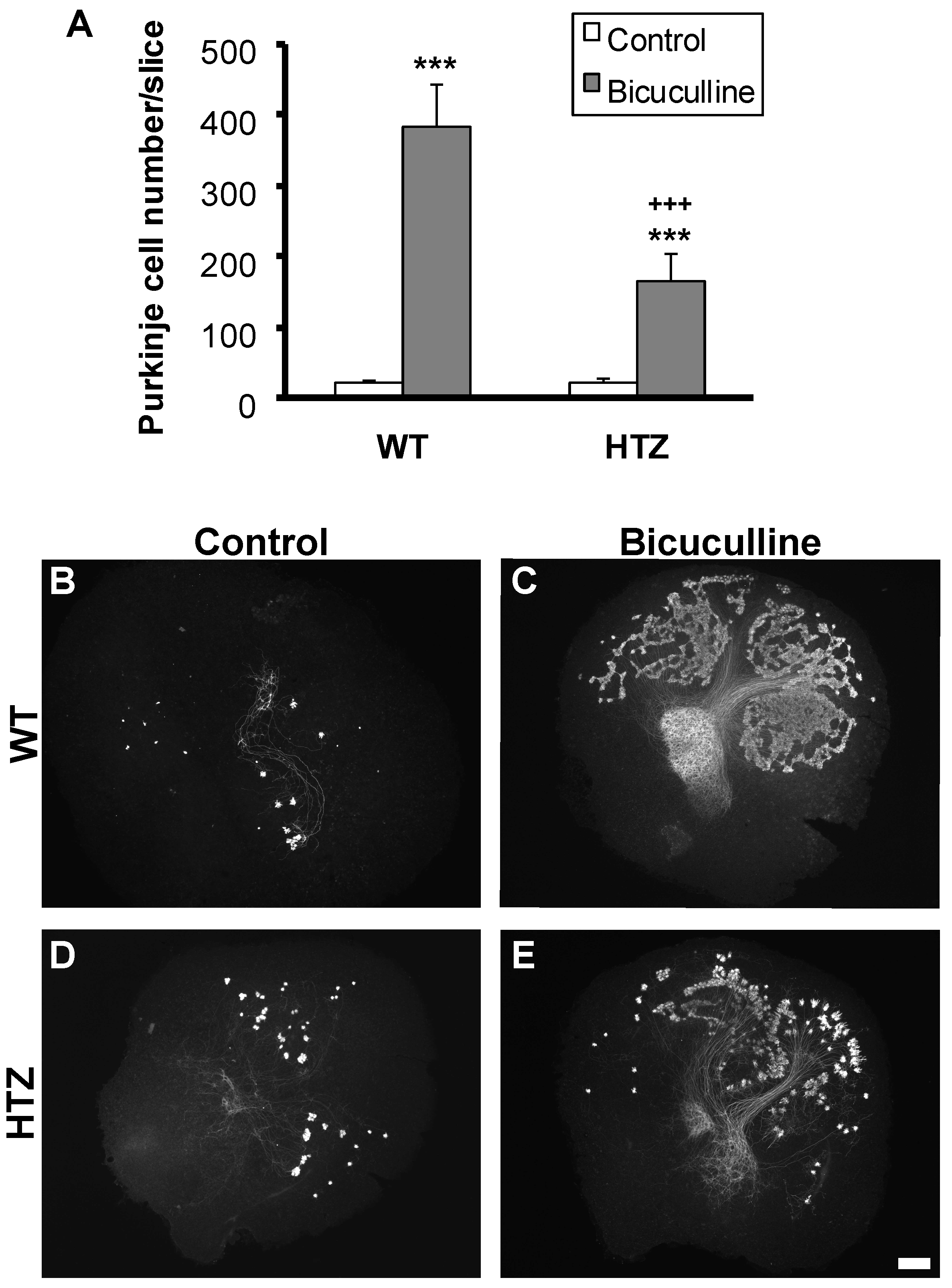
2.2. BDNF Haploinsufficiency Impairs Neuroprotection by Bicuculline in Cerebellar Slice Culture
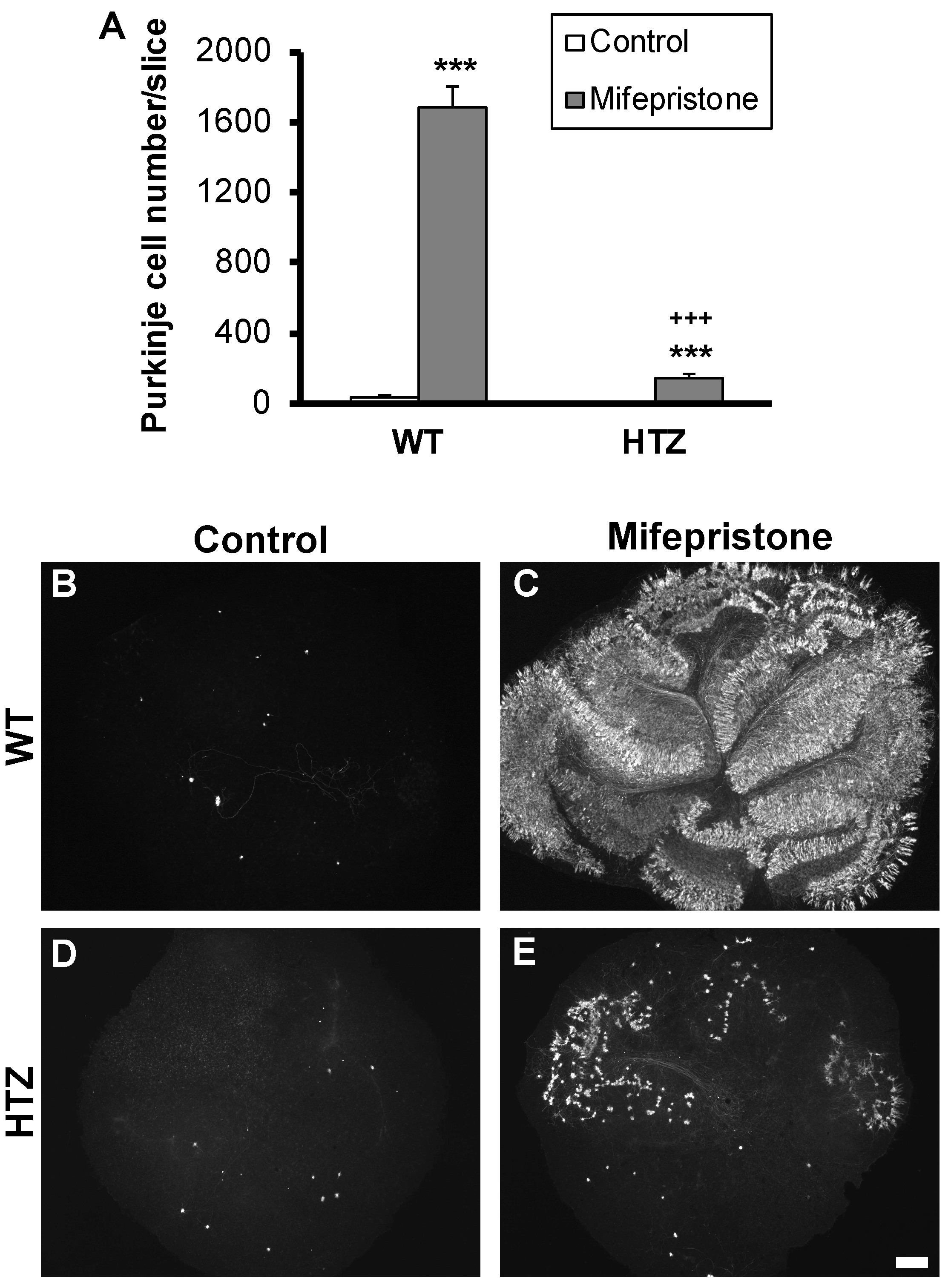
2.3. The Neuroptrophin BDNF Full Expression Is Necessary for Neuroprotection with Mifepristone
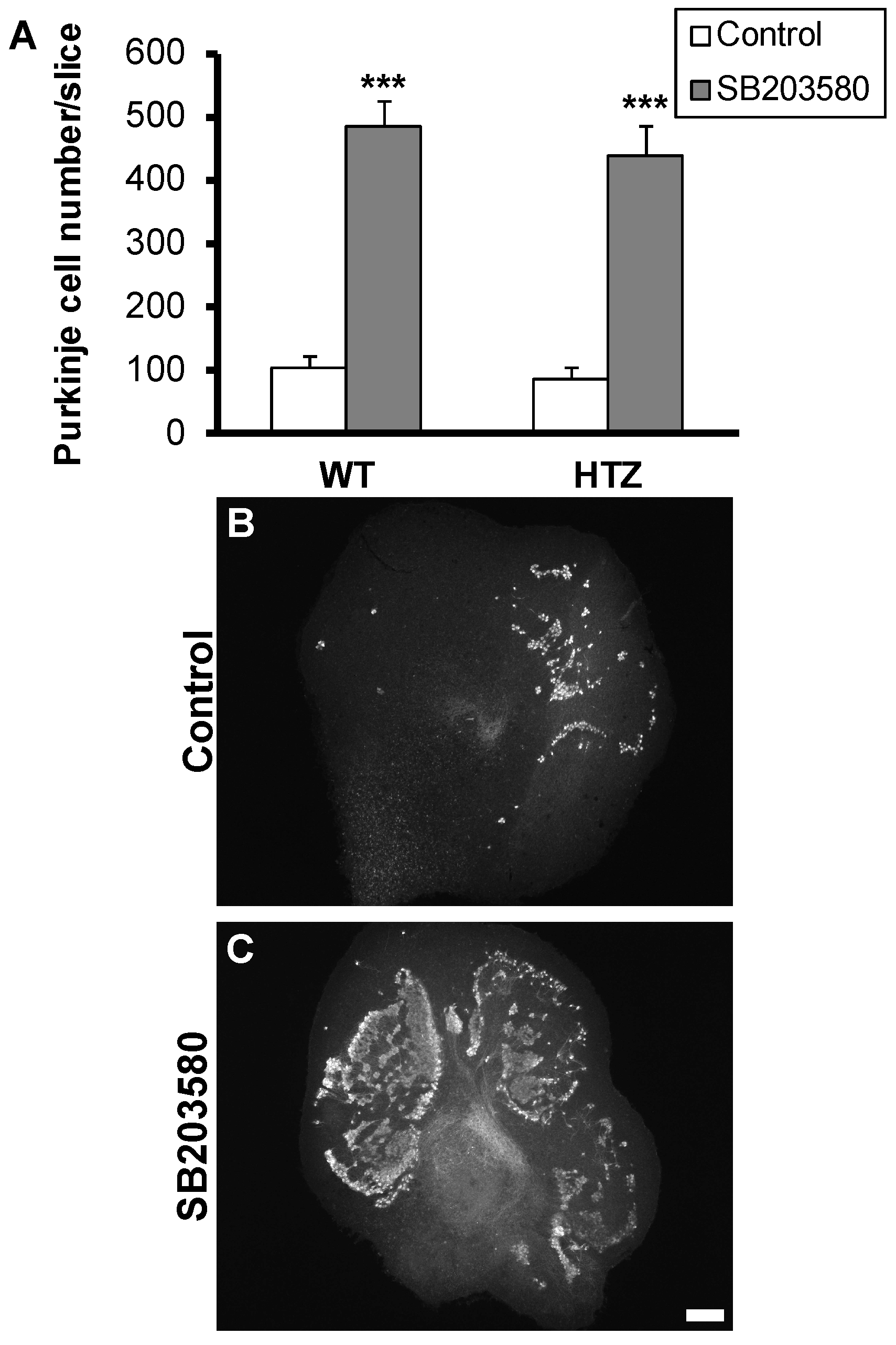
2.4. Neuroprotective Effect of the p38 MAP-Kinase Inhibitor, SB203580, Is Not Affected by BDNF Haploinsufficiency
3. Discussion
4. Materials and Methods
4.1. Animals and Organotypic Slice Culture
4.2. Immunofluorescence Analysis
4.3. Statistical Analysis
Author Contributions
Funding
Conflicts of Interest
References
- Dusart, I.; Airaksinen, M.S.; Sotelo, C. Purkinje cell survival and axonal regeneration are age dependent: An in vitro study. J. Neurosci. 1997, 17, 3710–3726. [Google Scholar] [CrossRef] [PubMed]
- Bosman, L.W.; Konnerth, A. Activity-dependent plasticity of developing climbing fiber-Purkinje cell synapses. Neuroscience 2009, 162, 612–623. [Google Scholar] [CrossRef] [PubMed]
- Sotelo, C. Development of “Pinceaux” formations and dendritic translocation of climbing fibers during the acquisition of the balance between glutamatergic and gamma-aminobutyric acidergic inputs in developing Purkinje cells. J. Comp. Neurol. 2008, 506, 240–262. [Google Scholar] [CrossRef]
- Rakotomamonjy, J.; Levenes, C.; Baulieu, E.E.; Schumacher, M.; Ghoumari, A.M. Novel protective effect of mifepristone on detrimental GABAA receptor activity to immature Purkinje neurons. FASEB J. 2011, 25, 3999–4010. [Google Scholar] [CrossRef] [PubMed]
- Eilers, J.; Plant, T.D.; Marandi, N.; Konnerth, A. GABA-mediated Ca2+ signalling in developing rat cerebellar Purkinje neurones. J. Physiol. 2001, 536, 429–437. [Google Scholar] [CrossRef] [PubMed]
- Watt, A.J.; Cuntz, H.; Mori, M.; Nusser, Z.; Sjostrom, P.J.; Hausser, M. Traveling waves in developing cerebellar cortex mediated by asymmetrical Purkinje cell connectivity. Nat. Neurosci. 2009, 12, 463–473. [Google Scholar] [CrossRef] [PubMed]
- Ghoumari, A.M.; Dusart, I.; El-Etr, M.; Tronche, F.; Sotelo, C.; Schumacher, M.; Baulieu, E.E. Mifepristone (RU486) protects Purkinje cells from cell death in organotypic slice cultures of postnatal rat and mouse cerebellum. Proc. Natl. Acad. Sci. USA 2003, 100, 7953–7958. [Google Scholar] [CrossRef] [PubMed]
- Ghoumari, A.M.; Piochon, C.; Tomkiewicz, C.; Eychenne, B.; Levenes, C.; Dusart, I.; Schumacher, M.; Baulieu, E.E. Neuroprotective effect of mifepristone involves neuron depolarization. FASEB J. 2006, 20, 1377–1386. [Google Scholar] [CrossRef]
- Poo, M.M. Neurotrophins as synaptic modulators. Nat. Rev. Neurosci. 2001, 2, 24–32. [Google Scholar] [CrossRef]
- Klein, R.; Nanduri, V.; Jing, S.A.; Lamballe, F.; Tapley, P.; Bryant, S.; Cordon-Cardo, C.; Jones, K.R.; Reichardt, L.F.; Barbacid, M. The trkB tyrosine protein kinase is a receptor for brain-derived neurotrophic factor and neurotrophin-3. Cell 1991, 66, 395–403. [Google Scholar] [CrossRef]
- Bosman, L.W.; Hartmann, J.; Barski, J.J.; Lepier, A.; Noll-Hussong, M.; Reichardt, L.F.; Konnerth, A. Requirement of TrkB for synapse elimination in developing cerebellar Purkinje cells. Brain Cell Biol. 2006, 35, 87–101. [Google Scholar] [CrossRef] [PubMed]
- Lindholm, D.; Hamner, S.; Zirrgiebel, U. Neurotrophins and cerebellar development. Perspect. Dev. Neurobiol. 1997, 5, 83–94. [Google Scholar] [PubMed]
- Gorski, J.A.; Zeiler, S.R.; Tamowski, S.; Jones, K.R. Brain-derived neurotrophic factor is required for the maintenance of cortical dendrites. J. Neurosci. 2003, 23, 6856–6865. [Google Scholar] [CrossRef]
- Zhou, X.P.; Wu, K.Y.; Liang, B.; Fu, X.Q.; Luo, Z.G. TrkB-mediated activation of geranylgeranyltransferase I promotes dendritic morphogenesis. Proc. Natl. Acad. Sci. USA 2008, 105, 17181–17186. [Google Scholar] [CrossRef]
- Schwartz, P.M.; Borghesani, P.R.; Levy, R.L.; Pomeroy, S.L.; Segal, R.A. Abnormal Cerebellar Development and Foliation in BDNF−/− Mice Reveals a Role for Neurotrophins in CNS Patterning. Neuron 1997, 19, 269–281. [Google Scholar] [CrossRef]
- Cheng, Q.; Yeh, H.H. PLCgamma signaling underlies BDNF potentiation of Purkinje cell responses to GABA. J. Neurosci. Res. 2005, 79, 616–627. [Google Scholar] [CrossRef] [PubMed]
- Kafitz, K.W.; Rose, C.R.; Thoenen, H.; Konnerth, A. Neurotrophin-evoked rapid excitation through TrkB receptors. Nature 1999, 401, 918–921. [Google Scholar] [CrossRef]
- Choo, M.; Miyazaki, T.; Yamazaki, M.; Kawamura, M.; Nakazawa, T.; Zhang, J.; Tanimura, A.; Uesaka, N.; Watanabe, M.; Sakimura, K.; et al. Retrograde BDNF to TrkB signaling promotes synapse elimination in the developing cerebellum. Nat. Commun. 2017, 8, 195. [Google Scholar] [CrossRef]
- Hu, P.; Kalb, R.G. BDNF heightens the sensitivity of motor neurons to excitotoxic insults through activation of TrkB. J. Neurochem. 2003, 84, 1421–1430. [Google Scholar] [CrossRef]
- Ishikawa, Y.; Ikeuchi, T.; Hatanaka, H. Brain-derived neurotrophic factor accelerates nitric oxide donor-induced apoptosis of cultured cortical neurons. J. Neurochem. 2000, 75, 494–502. [Google Scholar] [CrossRef]
- Kim, S.H.; Won, S.J.; Sohn, S.; Kwon, H.J.; Lee, J.Y.; Park, J.H.; Gwag, B.J. Brain-derived neurotrophic factor can act as a pronecrotic factor through transcriptional and translational activation of NADPH oxidase. J. Cell Biol. 2002, 159, 821–831. [Google Scholar] [CrossRef] [PubMed]
- Morrison, M.E.; Mason, C.A. Granule neuron regulation of Purkinje cell development: Striking a balance between neurotrophin and glutamate signaling. J. Neurosci. 1998, 18, 3563–3573. [Google Scholar] [CrossRef] [PubMed]
- Jiang, C.; Zhang, S.; Liu, H.; Guan, Z.; Zeng, Q.; Zhang, C.; Lei, R.; Xia, T.; Wang, Z.; Yang, L.; et al. Low glucose utilization and neurodegenerative changes caused by sodium fluoride exposure in rat’s developmental brain. Neuromol. Med. 2014, 16, 94–105. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Niu, Q.; Xia, T.; Zhou, G.; Li, P.; Zhao, Q.; Xu, C.; Dong, L.; Zhang, S.; Wang, A. ERK1/2-mediated disruption of BDNF—TrkB signaling causes synaptic impairment contributing to fluoride-induced developmental neurotoxicity. Toxicology 2018, 410, 222–230. [Google Scholar] [CrossRef] [PubMed]
- Ghoumari, A.M.; Wehrle, R.; De Zeeuw, C.I.; Sotelo, C.; Dusart, I. Inhibition of protein kinase C prevents Purkinje cell death but does not affect axonal regeneration. J. Neurosci. 2002, 22, 3531–3542. [Google Scholar] [CrossRef]
- Obrietan, K.; Gao, X.B.; Van Den Pol, A.N. Excitatory actions of GABA increase BDNF expression via a MAPK-CREB-dependent mechanism—A Positive Feedback Circuit in Developing Neurons. J. Neurophysiol. 2002, 88, 1005–1015. [Google Scholar] [CrossRef]
- Gubellini, P.; Ben-Ari, Y.; Gaiarsa, J.L. Endogenous neurotrophins are required for the induction of GABAergic long-term potentiation in the neonatal rat hippocampus. J. Neurosci. 2005, 25, 5796–5802. [Google Scholar] [CrossRef]
- Mizoguchi, Y.; Ishibashi, H.; Nabekura, J. The action of BDNF on GABA(A) currents changes from potentiating to suppressing during maturation of rat hippocampal CA1 pyramidal neurons. J. Physiol. 2003, 548, 703–709. [Google Scholar] [CrossRef]
- Goggi, J.; Pullar, I.A.; Carney, S.L.; Bradford, H.F. Modulation of neurotransmitter release induced by brain-derived neurotrophic factor in rat brain striatal slices in vitro. Brain Res. 2002, 941, 34–42. [Google Scholar] [CrossRef]
- Kim, J.; Lee, S.; Kang, S.; Kim, S.H.; Kim, J.C.; Yang, M.; Moon, C. Brain-derived neurotropic factor and GABAergic transmission in neurodegeneration and neuroregeneration. Neural Regen. Res. 2017, 12, 1733–1741. [Google Scholar]
- Han, B.H.; Holtzman, D.M. BDNF protects the neonatal brain from hypoxic-ischemic injury in vivo via the ERK pathway. J. Neurosci. 2000, 20, 5775–5781. [Google Scholar] [CrossRef] [PubMed]
- Kubo, T.; Nishimura, S.; Murasugi, T.; Kaneko, I.; Meguro, M.; Marumoto, S.; Kogen, H.; Koyama, K.; Oda, T.; Nakagami, Y. 6-Ethyl-N,N’-bis(3-hydroxyphenyl)[1,3,5]triazine-2,4-diamine (RS-0466) enhances the protective effect of brain-derived neurotrophic factor on amyloid beta-induced cytotoxicity in cortical neurones. Pharmacol. Toxicol. 2003, 93, 264–268. [Google Scholar] [CrossRef] [PubMed]
- Firozan, B.; Goudarzi, I.; Elahdadi Salmani, M.; Lashkarbolouki, T.; Rezaei, A.; Abrari, K. Estradiol increases expression of the brain-derived neurotrophic factor after acute administration of ethanol in the neonatal rat cerebellum. Eur. J. Pharmacol. 2014, 732, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Zhao, H.; Alam, A.; San, C.Y.; Eguchi, S.; Chen, Q.; Lian, Q.; Ma, D. Molecular mechanisms of brain-derived neurotrophic factor in neuro-protection: Recent developments. Brain Res. 2017, 1665, 1–21. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Ko, H.; Cheung, Z.H.; Yung, K.K.; Yao, T.; Wang, J.J.; Morozov, A.; Ke, Y.; Ip, N.Y.; Yung, W.H. Dual actions of brain-derived neurotrophic factor on GABAergic transmission in cerebellar Purkinje neurons. Exp. Neurol. 2012, 233, 791–798. [Google Scholar] [CrossRef] [PubMed]
- Mizoguchi, Y.; Kanematsu, T.; Hirata, M.; Nabekura, J. A rapid increase in the total number of cell surface functional GABAA receptors induced by brain-derived neurotrophic factor in rat visual cortex. J. Biol. Chem. 2003, 278, 44097–44102. [Google Scholar] [CrossRef] [PubMed]
- Palizvan, M.R.; Sohya, K.; Kohara, K.; Maruyama, A.; Yasuda, H.; Kimura, F.; Tsumoto, T. Brain-derived neurotrophic factor increases inhibitory synapses, revealed in solitary neurons cultured from rat visual cortex. Neuroscience 2004, 126, 955–966. [Google Scholar] [CrossRef]
- Matsumoto, T.; Numakawa, T.; Yokomaku, D.; Adachi, N.; Yamagishi, S.; Numakawa, Y.; Kunugi, H.; Taguchi, T. Brain-derived neurotrophic factor-induced potentiation of glutamate and GABA release: Different dependency on signaling pathways and neuronal activity. Mol. Cell. Neurosci. 2006, 31, 70–84. [Google Scholar] [CrossRef]
- Katoh-Semba, R.; Kaneko, R.; Kitajima, S.; Tsuzuki, M.; Ichisaka, S.; Hata, Y.; Yamada, H.; Miyazaki, N.; Takahashi, Y.; Kato, K. Activation of p38 mitogen-activated protein kinase is required for in vivo brain-derived neurotrophic factor production in the rat hippocampus. Neuroscience 2009, 163, 352–361. [Google Scholar] [CrossRef]
- Trang, T.; Beggs, S.; Wan, X.; Salter, M.W. P2 × 4-Receptor-Mediated Synthesis and Release of Brain-Derived Neurotrophic Factor in Microglia is dependent on Calcium and p38-Mitogen-Activated Protein Kinase Activation. J. Neurosci. 2009, 29, 3518–3528. [Google Scholar] [CrossRef]
- Yamagishi, S.; Matsumoto, T.; Yokomaku, D.; Hatanaka, H.; Shimoke, K.; Yamada, M.; Ikeuchi, T. Comparison of inhibitory effects of brain-derived neurotrophic factor and insulin-like growth factor on low potassium-induced apoptosis and activation of p38 MAPK and c-Jun in cultured cerebellar granule neurons. Brain Res. Mol. Brain Res. 2003, 119, 184–191. [Google Scholar] [CrossRef] [PubMed]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rakotomamonjy, J.; Ghoumari, A.M. Brain-Derived Neurotrophic Factor Is Required for the Neuroprotective Effect of Mifepristone on Immature Purkinje Cells in Cerebellar Slice Culture. Int. J. Mol. Sci. 2019, 20, 285. https://doi.org/10.3390/ijms20020285
Rakotomamonjy J, Ghoumari AM. Brain-Derived Neurotrophic Factor Is Required for the Neuroprotective Effect of Mifepristone on Immature Purkinje Cells in Cerebellar Slice Culture. International Journal of Molecular Sciences. 2019; 20(2):285. https://doi.org/10.3390/ijms20020285
Chicago/Turabian StyleRakotomamonjy, Jennifer, and Abdel Mouman Ghoumari. 2019. "Brain-Derived Neurotrophic Factor Is Required for the Neuroprotective Effect of Mifepristone on Immature Purkinje Cells in Cerebellar Slice Culture" International Journal of Molecular Sciences 20, no. 2: 285. https://doi.org/10.3390/ijms20020285
APA StyleRakotomamonjy, J., & Ghoumari, A. M. (2019). Brain-Derived Neurotrophic Factor Is Required for the Neuroprotective Effect of Mifepristone on Immature Purkinje Cells in Cerebellar Slice Culture. International Journal of Molecular Sciences, 20(2), 285. https://doi.org/10.3390/ijms20020285




