Sevoflurane, Propofol and Carvedilol Block Myocardial Protection by Limb Remote Ischemic Preconditioning
Abstract
1. Introduction
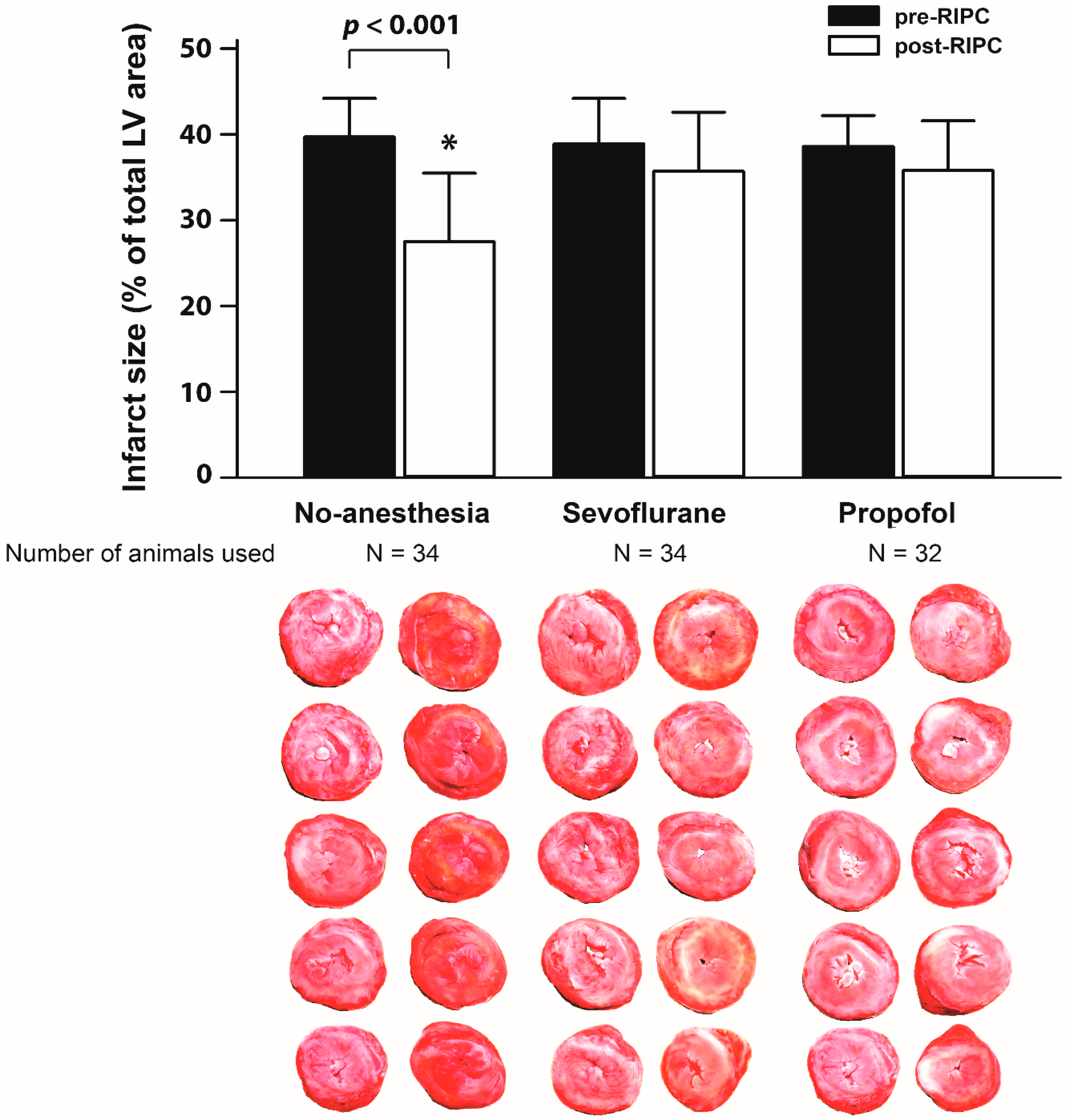
2. Results
3. Discussion
4. Materials and Methods
4.1. Ethical Approval
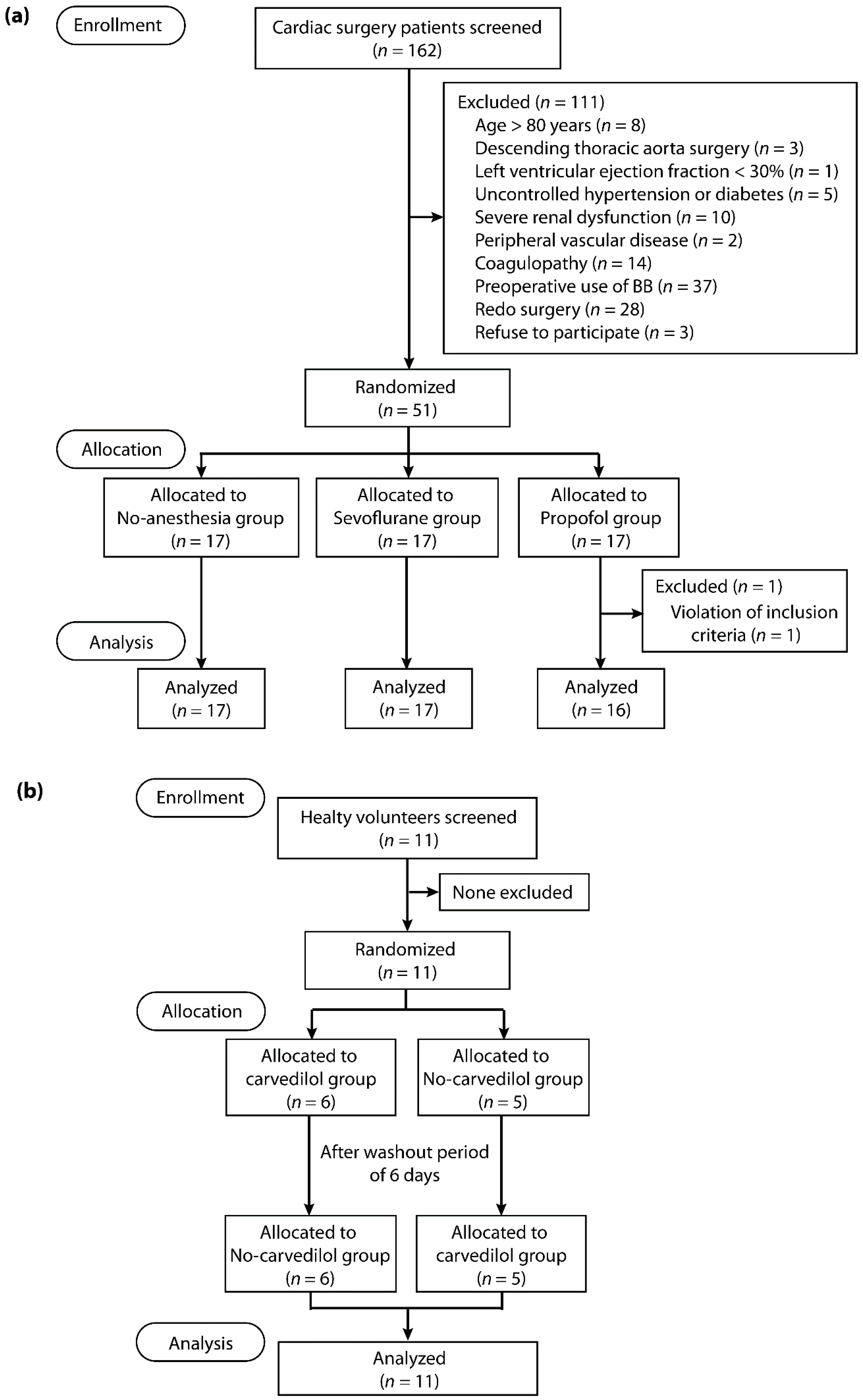
4.2. Study Population
4.3. Randomization and Masking
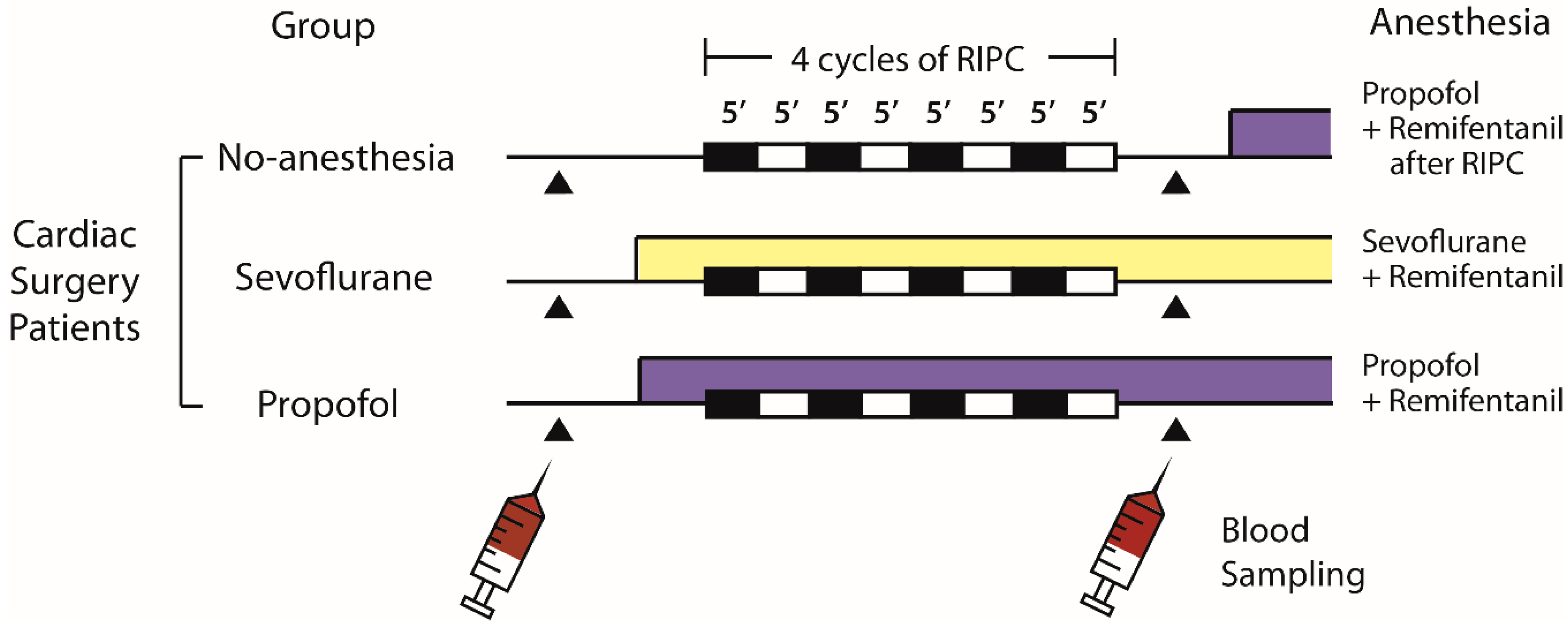
4.4. Cardiac Surgery Study Design
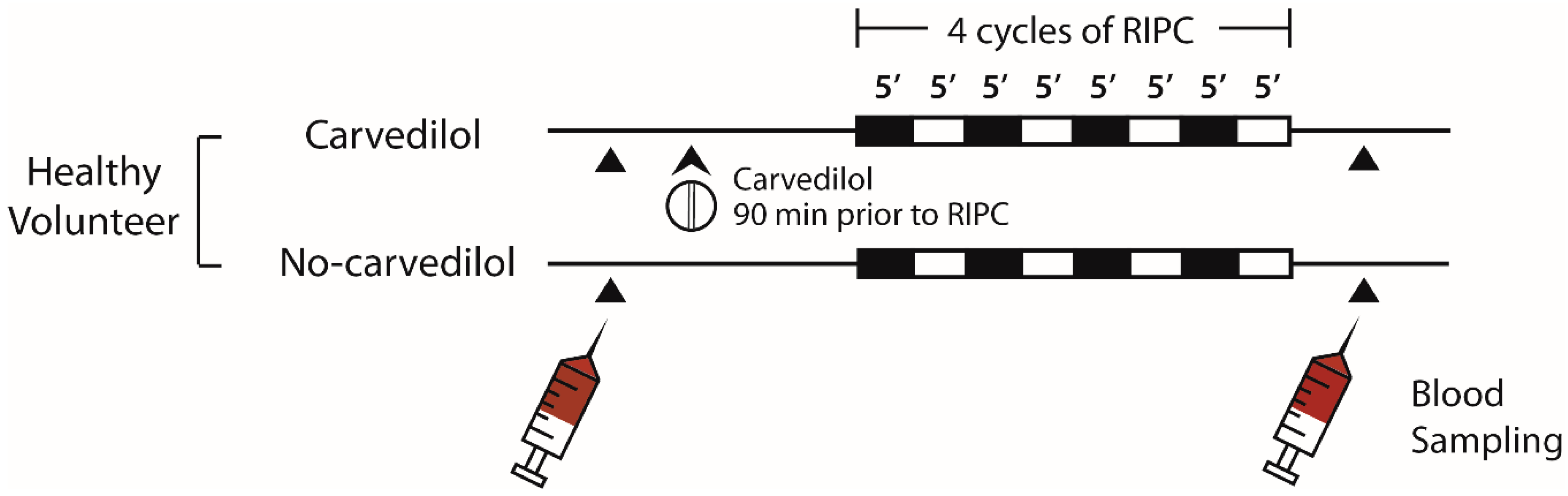
4.5. Healthy Volunteer Study Design
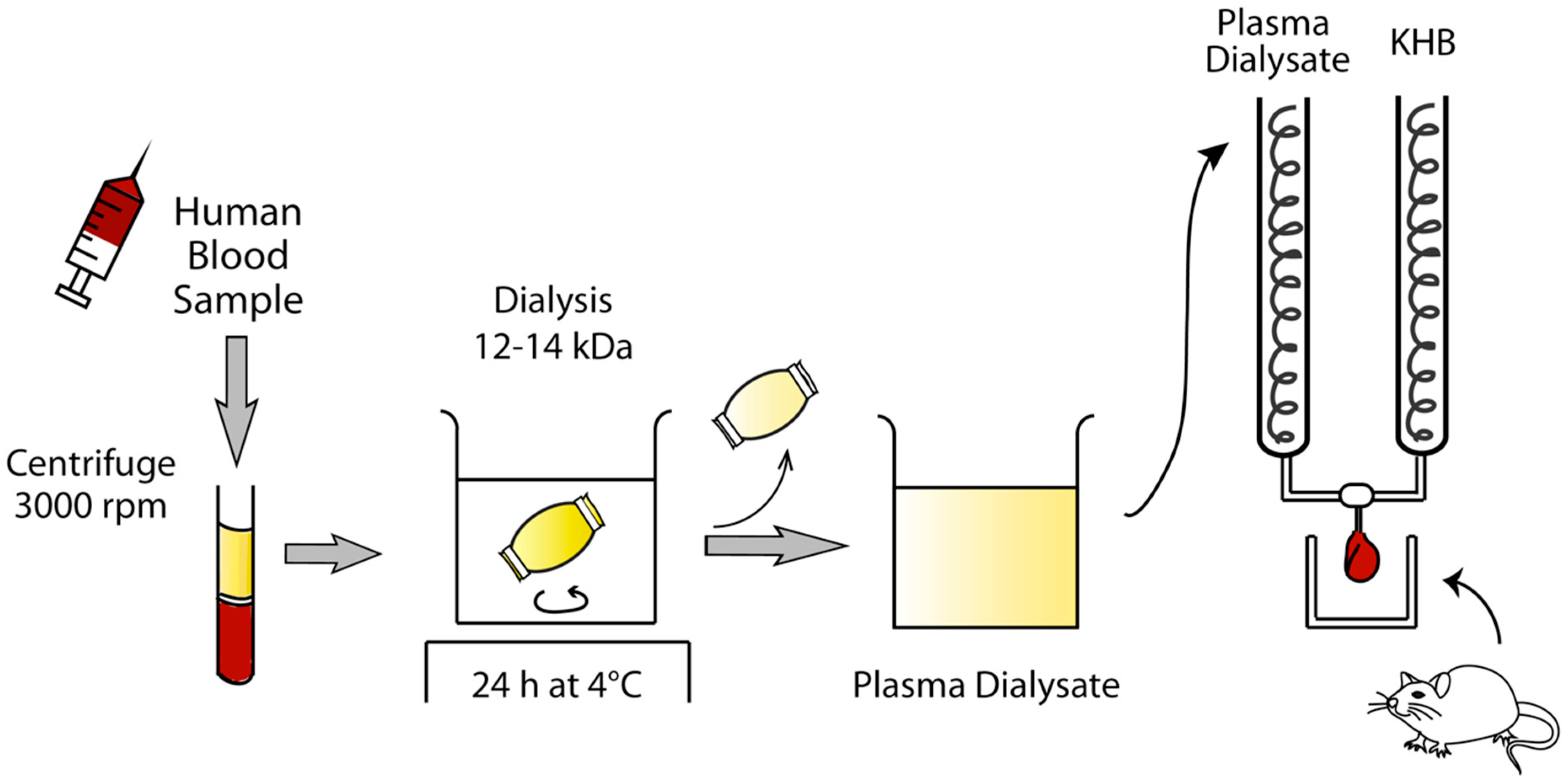
4.6. Preparation of Human Plasma Dialysate
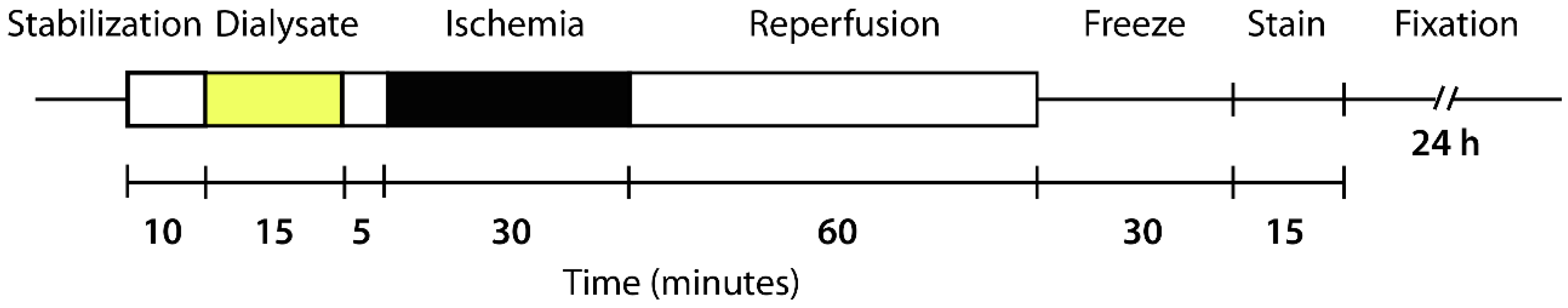
4.7. Langendorff-Perfused Rat Heart Model
4.8. Study Endpoints and Sample Size Calculation
4.9. Statistical Analyses
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| RIPC | remote ischemic preconditioning |
| BB | beta-blocker |
| MI | myocardial infarct |
| IR | ischemia/reperfusion |
| CABG | coronary artery bypass graft |
| cTnI | cardiac troponin I |
| IACUC | Institutional Animal Care and Use Committee |
| LV | left ventricular |
| BP | blood pressure |
| CI | confidence interval |
References
- Kharbanda, R.K.; Mortensen, U.M.; White, P.A.; Kristiansen, S.B.; Schmidt, M.R.; Hoschtitzky, J.A.; Vogel, M.; Sorensen, K.; Redington, A.N.; MacAllister, R. Transient limb ischemia induces remote ischemic preconditioning in vivo. Circulation 2002, 106, 2881–2883. [Google Scholar] [CrossRef] [PubMed]
- Hoole, S.P.; Heck, P.M.; Sharples, L.; Khan, S.N.; Duehmke, R.; Densem, C.G.; Clarke, S.C.; Shapiro, L.M.; Schofield, P.M.; O’Sullivan, M.; et al. Cardiac Remote Ischemic Preconditioning in Coronary Stenting (CRISP Stent) Study: A prospective, randomized control trial. Circulation 2009, 119, 820–827. [Google Scholar] [CrossRef] [PubMed]
- Botker, H.E.; Kharbanda, R.; Schmidt, M.R.; Bottcher, M.; Kaltoft, A.K.; Terkelsen, C.J.; Munk, K.; Andersen, N.H.; Hansen, T.M.; Trautner, S.; et al. Remote ischaemic conditioning before hospital admission, as a complement to angioplasty, and effect on myocardial salvage in patients with acute myocardial infarction: A randomised trial. Lancet 2010, 375, 727–734. [Google Scholar] [CrossRef]
- White, S.K.; Frohlich, G.M.; Sado, D.M.; Maestrini, V.; Fontana, M.; Treibel, T.A.; Tehrani, S.; Flett, A.S.; Meier, P.; Ariti, C.; et al. Remote ischemic conditioning reduces myocardial infarct size and edema in patients with ST-segment elevation myocardial infarction. JACC Cardiovasc. Interv. 2015, 8, 178–188. [Google Scholar] [CrossRef] [PubMed]
- Gaspar, A.; Lourenco, A.P.; Pereira, M.A.; Azevedo, P.; Roncon-Albuquerque, R., Jr.; Marques, J.; Leite-Moreira, A.F. Randomized controlled trial of remote ischaemic conditioning in ST-elevation myocardial infarction as adjuvant to primary angioplasty (RIC-STEMI). Basic Res. Cardiol. 2018, 113, 14. [Google Scholar] [CrossRef] [PubMed]
- Hausenloy, D.J.; Mwamure, P.K.; Venugopal, V.; Harris, J.; Barnard, M.; Grundy, E.; Ashley, E.; Vichare, S.; Di Salvo, C.; Kolvekar, S.; et al. Effect of remote ischaemic preconditioning on myocardial injury in patients undergoing coronary artery bypass graft surgery: A randomised controlled trial. Lancet 2007, 370, 575–579. [Google Scholar] [CrossRef]
- Thielmann, M.; Kottenberg, E.; Kleinbongard, P.; Wendt, D.; Gedik, N.; Pasa, S.; Price, V.; Tsagakis, K.; Neuhauser, M.; Peters, J.; et al. Cardioprotective and prognostic effects of remote ischaemic preconditioning in patients undergoing coronary artery bypass surgery: A single-centre randomised, double-blind, controlled trial. Lancet 2013, 382, 597–604. [Google Scholar] [CrossRef]
- Hong, D.M.; Lee, E.H.; Kim, H.J.; Min, J.J.; Chin, J.H.; Choi, D.K.; Bahk, J.H.; Sim, J.Y.; Choi, I.C.; Jeon, Y. Does remote ischaemic preconditioning with postconditioning improve clinical outcomes of patients undergoing cardiac surgery? Remote Ischaemic Preconditioning with Postconditioning Outcome Trial. Eur. Heart J. 2014, 35, 176–183. [Google Scholar] [CrossRef]
- Hausenloy, D.J.; Candilio, L.; Evans, R.; Ariti, C.; Jenkins, D.P.; Kolvekar, S.; Knight, R.; Kunst, G.; Laing, C.; Nicholas, J.; et al. Remote Ischemic Preconditioning and Outcomes of Cardiac Surgery. N. Engl. J. Med. 2015, 373, 1408–1417. [Google Scholar] [CrossRef]
- Meybohm, P.; Bein, B.; Brosteanu, O.; Cremer, J.; Gruenewald, M.; Stoppe, C.; Coburn, M.; Schaelte, G.; Boning, A.; Niemann, B.; et al. A Multicenter Trial of Remote Ischemic Preconditioning for Heart Surgery. N. Engl. J. Med. 2015, 373, 1397–1407. [Google Scholar] [CrossRef]
- Cheung, C.X.; Healy, D.A.; Walsh, S.R. Remote preconditioning and cardiac surgery: Regrouping after Remote Ischemic Preconditioning for Heart Surgery (RIPHeart) and Effect of Remote Ischemic Preconditioning on Clinical Outcomes in Patients Undergoing Coronary Artery Bypass Surgery (ERICCA). J. Thorac. Dis. 2016, 8, E197–E199. [Google Scholar] [CrossRef] [PubMed]
- Heusch, G.; Gersh, B.J. ERICCA and RIPHeart: Two nails in the coffin for cardioprotection by remote ischemic conditioning? Probably not! Eur. Heart J. 2016, 37, 200–202. [Google Scholar] [CrossRef] [PubMed]
- Benstoem, C.; Goetzenich, A.; Stoppe, C. The role of propofol for remote ischaemic preconditioning in the setting of cardiac surgery—A Cochrane systematic review. Br. J. Anaesth. 2017, 119, 1234–1235. [Google Scholar] [CrossRef] [PubMed]
- Lange, M.; Smul, T.M.; Blomeyer, C.A.; Redel, A.; Klotz, K.N.; Roewer, N.; Kehl, F. Role of the beta1-adrenergic pathway in anesthetic and ischemic preconditioning against myocardial infarction in the rabbit heart in vivo. Anesthesiology 2006, 105, 503–510. [Google Scholar] [CrossRef] [PubMed]
- Pickard, J.M.; Davidson, S.M.; Hausenloy, D.J.; Yellon, D.M. Co-dependence of the neural and humoral pathways in the mechanism of remote ischemic conditioning. Basic Res. Cardiol. 2016, 111, 50. [Google Scholar] [CrossRef] [PubMed]
- Mastitskaya, S.; Basalay, M.; Hosford, P.S.; Ramage, A.G.; Gourine, A.; Gourine, A.V. Identifying the Source of a Humoral Factor of Remote (Pre)Conditioning Cardioprotection. PLoS ONE 2016, 11, e0150108. [Google Scholar] [CrossRef] [PubMed]
- Shimizu, M.; Tropak, M.; Diaz, R.J.; Suto, F.; Surendra, H.; Kuzmin, E.; Li, J.; Gross, G.; Wilson, G.J.; Callahan, J.; et al. Transient limb ischaemia remotely preconditions through a humoral mechanism acting directly on the myocardium: Evidence suggesting cross-species protection. Clin. Sci. 2009, 117, 191–200. [Google Scholar] [CrossRef]
- Jensen, R.V.; Stottrup, N.B.; Kristiansen, S.B.; Botker, H.E. Release of a humoral circulating cardioprotective factor by remote ischemic preconditioning is dependent on preserved neural pathways in diabetic patients. Basic Res. Cardiol. 2012, 107, 285. [Google Scholar] [CrossRef]
- Heusch, G.; Botker, H.E.; Przyklenk, K.; Redington, A.; Yellon, D. Remote ischemic conditioning. J. Am. Coll. Cardiol. 2015, 65, 177–195. [Google Scholar] [CrossRef]
- McDonald, M.A.; Braga, J.R.; Li, J.; Manlhiot, C.; Ross, H.J.; Redington, A.N. A randomized pilot trial of remote ischemic preconditioning in heart failure with reduced ejection fraction. PLoS ONE 2014, 9, e105361. [Google Scholar] [CrossRef]
- Przyklenk, K.; Bauer, B.; Ovize, M.; Kloner, R.A.; Whittaker, P. Regional ischemic ‘preconditioning’ protects remote virgin myocardium from subsequent sustained coronary occlusion. Circulation 1993, 87, 893–899. [Google Scholar] [CrossRef] [PubMed]
- Davies, W.R.; Brown, A.J.; Watson, W.; McCormick, L.M.; West, N.E.; Dutka, D.P.; Hoole, S.P. Remote ischemic preconditioning improves outcome at 6 years after elective percutaneous coronary intervention: The CRISP stent trial long-term follow-up. Circ. Cardiovasc. Interv. 2013, 6, 246–251. [Google Scholar] [CrossRef]
- Sloth, A.D.; Schmidt, M.R.; Munk, K.; Kharbanda, R.K.; Redington, A.N.; Schmidt, M.; Pedersen, L.; Sorensen, H.T.; Botker, H.E.; CONDI Investigators. Improved long-term clinical outcomes in patients with ST-elevation myocardial infarction undergoing remote ischaemic conditioning as an adjunct to primary percutaneous coronary intervention. Eur. Heart J. 2014, 35, 168–175. [Google Scholar] [CrossRef] [PubMed]
- Candilio, L.; Malik, A.; Ariti, C.; Barnard, M.; Di Salvo, C.; Lawrence, D.; Hayward, M.; Yap, J.; Roberts, N.; Sheikh, A.; et al. Effect of remote ischaemic preconditioning on clinical outcomes in patients undergoing cardiac bypass surgery: A randomised controlled clinical trial. Heart 2015, 101, 185–192. [Google Scholar] [CrossRef] [PubMed]
- Candilio, L.; Hausenloy, D. Is there a role for ischaemic conditioning in cardiac surgery? F1000Research 2017, 6, 563. [Google Scholar] [CrossRef] [PubMed]
- Vasileiou, I.; Xanthos, T.; Koudouna, E.; Perrea, D.; Klonaris, C.; Katsargyris, A.; Papadimitriou, L. Propofol: A review of its non-anaesthetic effects. Eur. J. Pharmacol. 2009, 605, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Gan, X.; Xing, D.; Su, G.; Li, S.; Luo, C.; Irwin, M.G.; Xia, Z.; Li, H.; Hei, Z. Propofol Attenuates Small Intestinal Ischemia Reperfusion Injury through Inhibiting NADPH Oxidase Mediated Mast Cell Activation. Oxid. Med. Cell. Longev. 2015, 2015, 167014. [Google Scholar] [CrossRef]
- Mullenheim, J.; Ebel, D.; Frassdorf, J.; Preckel, B.; Thamer, V.; Schlack, W. Isoflurane preconditions myocardium against infarction via release of free radicals. Anesthesiology 2002, 96, 934–940. [Google Scholar] [CrossRef]
- Tanaka, K.; Weihrauch, D.; Kehl, F.; Ludwig, L.M.; LaDisa, J.F., Jr.; Kersten, J.R.; Pagel, P.S.; Warltier, D.C. Mechanism of preconditioning by isoflurane in rabbits: A direct role for reactive oxygen species. Anesthesiology 2002, 97, 1485–1490. [Google Scholar] [CrossRef]
- Kottenberg, E.; Musiolik, J.; Thielmann, M.; Jakob, H.; Peters, J.; Heusch, G. Interference of propofol with signal transducer and activator of transcription 5 activation and cardioprotection by remote ischemic preconditioning during coronary artery bypass grafting. J. Thorac. Cardiovasc. Surg. 2014, 147, 376–382. [Google Scholar] [CrossRef]
- Kottenberg, E.; Thielmann, M.; Bergmann, L.; Heine, T.; Jakob, H.; Heusch, G.; Peters, J. Protection by remote ischemic preconditioning during coronary artery bypass graft surgery with isoflurane but not propofol—A clinical trial. Acta. Anaesthesiol. Scand. 2012, 56, 30–38. [Google Scholar] [CrossRef] [PubMed]
- Zarbock, A.; Schmidt, C.; Van Aken, H.; Wempe, C.; Martens, S.; Zahn, P.K.; Wolf, B.; Goebel, U.; Schwer, C.I.; Rosenberger, P.; et al. Effect of remote ischemic preconditioning on kidney injury among high-risk patients undergoing cardiac surgery: A randomized clinical trial. JAMA 2015, 313, 2133–2141. [Google Scholar] [CrossRef] [PubMed]
- Green, T.R.; Bennett, S.R.; Nelson, V.M. Specificity and properties of propofol as an antioxidant free radical scavenger. Toxicol. Appl. Pharmacol. 1994, 129, 163–169. [Google Scholar] [CrossRef] [PubMed]
- Xu, Z.; Yu, J.; Wu, J.; Qi, F.; Wang, H.; Wang, Z.; Wang, Z. The Effects of Two Anesthetics, Propofol and Sevoflurane, on Liver Ischemia/Reperfusion Injury. Cell. Physiol. Biochem. 2016, 38, 1631–1642. [Google Scholar] [CrossRef] [PubMed]
- Woodbridge, P.D. The Components of General Anesthesia. A Plea for the Blocking of Sensory Pathways. JAMA 1963, 186, 641–645. [Google Scholar] [CrossRef] [PubMed]
- Benstoem, C.; Stoppe, C.; Liakopoulos, O.J.; Ney, J.; Hasenclever, D.; Meybohm, P.; Goetzenich, A. Remote ischaemic preconditioning for coronary artery bypass grafting (with or without valve surgery). Cochrane Database Syst. Rev. 2017, 5, CD011719. [Google Scholar] [CrossRef] [PubMed]
- Behmenburg, F.; van Caster, P.; Bunte, S.; Brandenburger, T.; Heinen, A.; Hollmann, M.W.; Huhn, R. Impact of Anesthetic Regimen on Remote Ischemic Preconditioning in the Rat Heart In Vivo. Anesth. Analg. 2018, 126, 1377–1380. [Google Scholar] [CrossRef]
- Kleinbongard, P.; Heusch, G. Extracellular signalling molecules in the ischaemic/reperfused heart—Druggable and translatable for cardioprotection? Br. J. Pharmacol. 2015, 172, 2010–2025. [Google Scholar] [CrossRef]
- Wu, Q.; Wang, T.; Chen, S.; Zhou, Q.; Li, H.; Hu, N.; Feng, Y.; Dong, N.; Yao, S.; Xia, Z. Cardiac protective effects of remote ischaemic preconditioning in children undergoing tetralogy of fallot repair surgery: A randomized controlled trial. Eur. Heart J. 2018, 39, 1028–1037. [Google Scholar] [CrossRef]
- Song, Y.; Song, J.W.; Lee, S.; Jun, J.H.; Kwak, Y.L.; Shim, J.K. Effects of remote ischemic preconditioning in patients with concentric myocardial hypertrophy: A randomized, controlled trial with molecular insights. Int. J. Cardiol. 2017, 249, 36–41. [Google Scholar] [CrossRef]
- Feuerstein, G.; Liu, G.L.; Yue, T.L.; Cheng, H.Y.; Hieble, J.P.; Arch, J.R.; Ruffolo, R.R., Jr.; Ma, X.L. Comparison of metoprolol and carvedilol pharmacology and cardioprotection in rabbit ischemia and reperfusion model. Eur. J. Pharmacol. 1998, 351, 341–350. [Google Scholar] [CrossRef]
- Gao, F.; Chen, J.; Lopez, B.L.; Christopher, T.A.; Gu, J.; Lysko, P.; Ruffolo, R.R., Jr.; Ohlstein, E.H.; Ma, X.L.; Yue, T.L. Comparison of bisoprolol and carvedilol cardioprotection in a rabbit ischemia and reperfusion model. Eur. J. Pharmacol. 2000, 406, 109–116. [Google Scholar] [CrossRef]
- Suematsu, Y.; Anttila, V.; Takamoto, S.; del Nido, P. Cardioprotection afforded by ischemic preconditioning interferes with chronic beta-blocker treatment. Scand. Cardiovasc. J. 2004, 38, 293–299. [Google Scholar] [CrossRef] [PubMed]
- See Hoe, L.E.; Schilling, J.M.; Busija, A.R.; Haushalter, K.J.; Ozberk, V.; Keshwani, M.M.; Roth, D.M.; Toit, E.D.; Headrick, J.P.; Patel, H.H.; et al. Chronic beta1-adrenoceptor blockade impairs ischaemic tolerance and preconditioning in murine myocardium. Eur. J. Pharmacol. 2016, 789, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Kleinbongard, P.; Neuhauser, M.; Thielmann, M.; Kottenberg, E.; Peters, J.; Jakob, H.; Heusch, G. Confounders of Cardioprotection by Remote Ischemic Preconditioning in Patients Undergoing Coronary Artery Bypass Grafting. Cardiology 2016, 133, 128–133. [Google Scholar] [CrossRef] [PubMed]
- Headrick, J.P.; See Hoe, L.E.; Du Toit, E.F.; Peart, J.N. Opioid receptors and cardioprotection—’Opioidergic conditioning’ of the heart. Br. J. Pharmacol. 2015, 172, 2026–2050. [Google Scholar] [CrossRef] [PubMed]
- Lemoine, S.; Zhu, L.; Massetti, M.; Gerard, J.L.; Hanouz, J.L. Continuous administration of remifentanil and sufentanil induces cardioprotection in human myocardium, in vitro. Acta Anaesthesiol. Scand. 2011, 55, 758–764. [Google Scholar] [CrossRef]
- Kuzume, K.; Kuzume, K.; Wolff, R.A.; Chien, G.L.; Van Winkle, D.M. Remifentanil limits infarct size but attenuates preconditioning-induced infarct limitation. Coron. Artery Dis. 2004, 15, 449–455. [Google Scholar] [CrossRef]
- Ferrera, R.; Benhabbouche, S.; Bopassa, J.C.; Li, B.; Ovize, M. One hour reperfusion is enough to assess function and infarct size with TTC staining in Langendorff rat model. Cardiovasc. Drugs Ther. 2009, 23, 327–331. [Google Scholar] [CrossRef]
- Bell, R.M.; Mocanu, M.M.; Yellon, D.M. Retrograde heart perfusion: The Langendorff technique of isolated heart perfusion. J. Mol. Cell. Cardiol. 2011, 50, 940–950. [Google Scholar] [CrossRef]
- Heinen, A.; Behmenburg, F.; Aytulun, A.; Dierkes, M.; Zerbin, L.; Kaisers, W.; Schaefer, M.; Meyer-Treschan, T.; Feit, S.; Bauer, I.; et al. The release of cardioprotective humoral factors after remote ischemic preconditioning in humans is age- and sex-dependent. J. Transl. Med. 2018, 16, 112. [Google Scholar] [CrossRef] [PubMed]
- Lateef, R.; Al-Masri, A.; Alyahya, A. Langendorff’s isolated perfused rat heart technique: A review. Int. J. Basic Clin. Pharmacol. 2015, 4, 1314–1322. [Google Scholar] [CrossRef]
- Redington, K.L.; Disenhouse, T.; Li, J.; Wei, C.; Dai, X.; Gladstone, R.; Manlhiot, C.; Redington, A.N. Electroacupuncture reduces myocardial infarct size and improves post-ischemic recovery by invoking release of humoral, dialyzable, cardioprotective factors. J. Physiol. Sci. 2013, 63, 219–223. [Google Scholar] [CrossRef] [PubMed]
- Jean-St-Michel, E.; Manlhiot, C.; Li, J.; Tropak, M.; Michelsen, M.M.; Schmidt, M.R.; McCrindle, B.W.; Wells, G.D.; Redington, A.N. Remote preconditioning improves maximal performance in highly trained athletes. Med. Sci. Sports Exerc. 2011, 43, 1280–1286. [Google Scholar] [CrossRef] [PubMed]


| Characteristics | Cardiac Surgical Patients | p Value | ||
|---|---|---|---|---|
| No-Anesthesia (n = 17) | Sevoflurane (n = 17) | Propofol (n = 16) | ||
| Age (yr) | 66 ± 14 | 59 ± 15 | 63 ± 15 | 0.453 |
| Male sex | 6 (35%) | 13 (77%) | 7 (41%) | 0.040 |
| Height (cm) | 158.1 ± 11.9 | 168.7 ± 10.4 | 160.4 ± 8.3 | 0.011 |
| Weight (kg) | 62.5 ± 10.5 | 66.4 ± 10.3 | 62.2 ± 8.1 | 0.395 |
| BMI (kg/m2) | 25.0 ± 3.5 | 23.2 ± 2.4 | 24.2 ± 3.0 | 0.242 |
| BSA (m2) | 1.6 ± 0.2 | 1.8 ± 0.2 | 1.7 ± 0.1 | 0.082 |
| Smoking status (never/current/ex-smoker) | 16 (94%)/1 (6%)/0 | 11 (64%)/3 (18%)/3 (18%) | 12 (75%) /2 (12%) /2 (12%) | 0.314 |
| Baseline LV EF (%) | 62 ± 6 | 60 ± 7 | 59 ± 8 | 0.480 |
| Baseline troponin I (ng/mL) | 0.007 ± 0.014 | 0.018 ± 0.038 | 0.026 ± 0.049 | 0.332 |
| Baseline hematocrit (%) | 40 ± 3 | 41 ± 6 | 39 ± 4 | 0.357 |
| Baseline eGFR (mL/min/1.73m2) | 78.1 ± 19.3 | 86.9 ± 20.6 | 88.5 ± 27.7 | 0.371 |
| Comorbidities | No-Anesthesia | Sevoflurane | Propofol | |
| Hypertension | 8 (47%) | 2 (12%) | 6 (37%) | 0.075 |
| Diabetes mellitus | 2 (12%) | 1 (6%) | 3 (18%) | 0.524 |
| Ischemic heart disease | 3 (19%) | 1 (6%) | 1 (6%) | 0.456 |
| Previous PCI | 2 (12%) | 2 (12%) | 0 | 0.360 |
| Previous stroke | 1 (6%) | 0 | 0 | 0.371 |
| Preoperative medication | No-Anesthesia | Sevoflurane | Propofol | |
| Aspirin | 7 (41%) | 4 (24%) | 2 (13%) | 0.165 |
| Clopidogrel | 1 (6%) | 1 (6%) | 0 | 0.613 |
| ACE inhibitor | 0 | 0 | 1 (6%) | 0.338 |
| ARB | 2 (12%) | 2 (12%) | 3 (18%) | 0.802 |
| CCB | 9 (53%) | 4 (24%) | 3 (18%) | 0.071 |
| Diuretics | 7 (41%) | 4 (24%) | 6 (37%) | 0.520 |
| Nitroglycerin | 0 | 0 | 1 (6%) | 0.338 |
| Digoxin | 0 | 1 (6%) | 1 (6%) | 0.584 |
| OHA | 0 | 1 (6%) | 3 (18%) | 0.129 |
| Insulin | 0 | 1 (6%) | 0 | 0.371 |
| Statin | 8 (47%) | 4 (24%) | 5 (31%) | 0.337 |
| Type of surgery | No-Anesthesia | Sevoflurane | Propofol | 0.381 |
| Valve | 8 (47%) | 10 (59%) | 13 (82%) | |
| Valve + Aorta | 7 (41%) | 5 (29%) | 1 (6%) | |
| Valve + CABG | 1 (6%) | 0 | 1 (6%) | |
| Aorta | 1 (6%) | 1 (6%) | 1 (6%) | |
| Other* | 0 | 1 (6%) | 0 | |
| Characteristics | Healthy Volunteers (n = 11) |
|---|---|
| Age (yr) | 27 ± 6 |
| Height (cm) | 174.1 ± 4.8 |
| Weight (kg) | 71.3 ± 8.2 |
| BMI (kg/m2) | 23.5 ± 2.2 |
| BSA (m2) | 1.9 ± 0.1 |
| Variables | No-Anesthesia (n = 17) | Sevoflurane (n = 17) | Propofol (n = 16) | p Value |
|---|---|---|---|---|
| Peak troponin I in 72 h (ng/mL) | 13.07 ± 8.02 | 11.92 ± 7.33 * | 17.44 ± 20.01 | 0.456 |
| Chest tube drain in 24 h (mL) | 697 ± 433 | 859 ± 538 | 739 ± 612 | 0.656 |
| New onset atrial fibrillation | 5 (31%) | 7 (41%) | 7 (47%) | 0.671 |
| Intensive care unit stay (h) | 42 ± 28 | 35 ± 27 | 42 ± 36 | 0.743 |
| Postoperative hospital stay (days) | 12 ± 6 | 10 ± 4 | 11 ± 8 | 0.618 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cho, Y.J.; Nam, K.; Kim, T.K.; Choi, S.W.; Kim, S.J.; Hausenloy, D.J.; Jeon, Y. Sevoflurane, Propofol and Carvedilol Block Myocardial Protection by Limb Remote Ischemic Preconditioning. Int. J. Mol. Sci. 2019, 20, 269. https://doi.org/10.3390/ijms20020269
Cho YJ, Nam K, Kim TK, Choi SW, Kim SJ, Hausenloy DJ, Jeon Y. Sevoflurane, Propofol and Carvedilol Block Myocardial Protection by Limb Remote Ischemic Preconditioning. International Journal of Molecular Sciences. 2019; 20(2):269. https://doi.org/10.3390/ijms20020269
Chicago/Turabian StyleCho, Youn Joung, Karam Nam, Tae Kyong Kim, Seong Woo Choi, Sung Joon Kim, Derek J Hausenloy, and Yunseok Jeon. 2019. "Sevoflurane, Propofol and Carvedilol Block Myocardial Protection by Limb Remote Ischemic Preconditioning" International Journal of Molecular Sciences 20, no. 2: 269. https://doi.org/10.3390/ijms20020269
APA StyleCho, Y. J., Nam, K., Kim, T. K., Choi, S. W., Kim, S. J., Hausenloy, D. J., & Jeon, Y. (2019). Sevoflurane, Propofol and Carvedilol Block Myocardial Protection by Limb Remote Ischemic Preconditioning. International Journal of Molecular Sciences, 20(2), 269. https://doi.org/10.3390/ijms20020269





