Comparing the Protection Imparted by Different Fraction Extracts of Garlic (Allium sativum L.) against Der p–Induced Allergic Airway Inflammation in Mice
Abstract
1. Introduction
2. Results
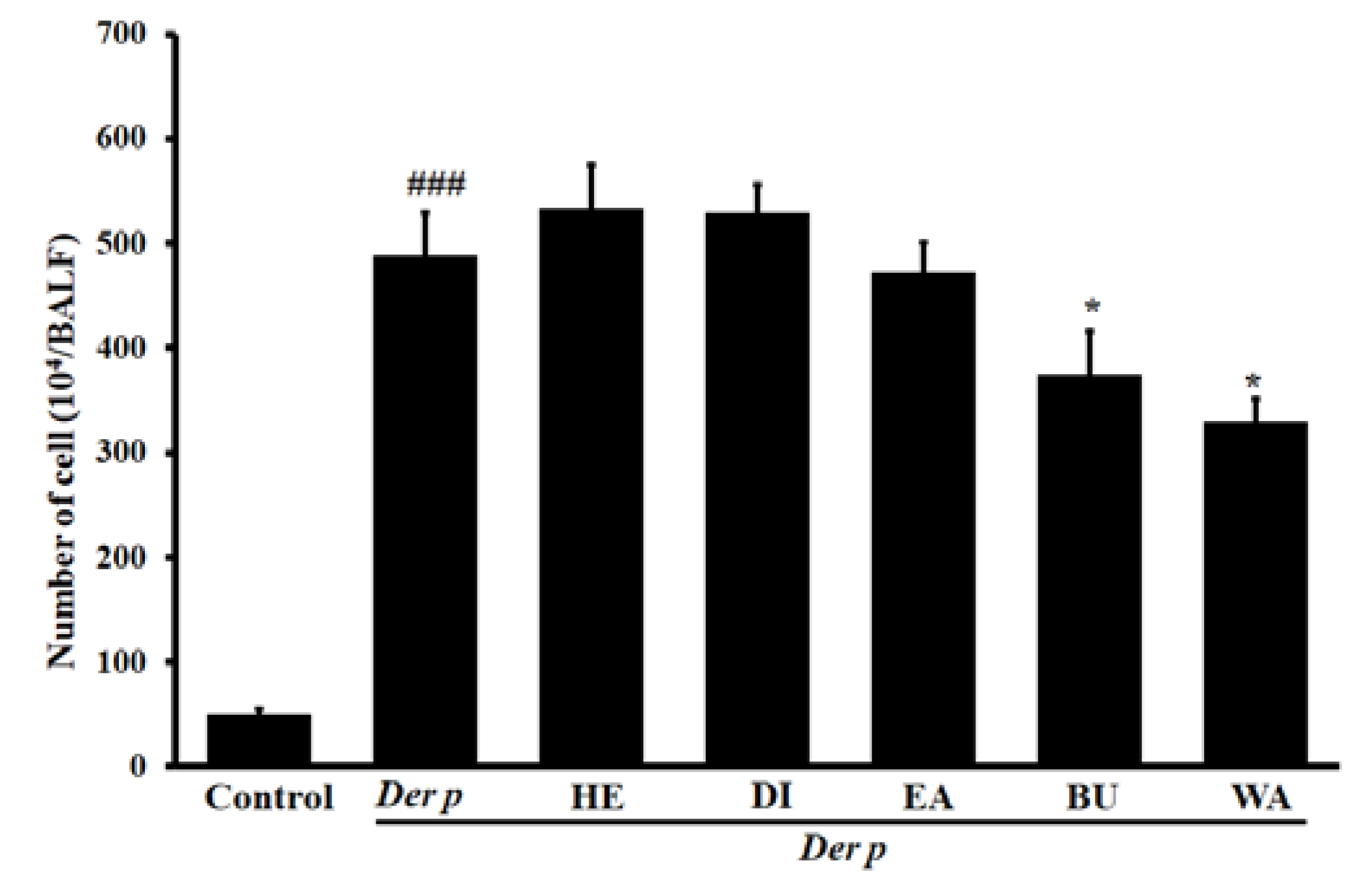
2.1. Effects Exerted on Total Inflammatory Cell Count in Bronchoalveolar Lavage Fluid (BALF)
2.2. Pathological Results on Der p–Induced Lung and Trachea Injuries
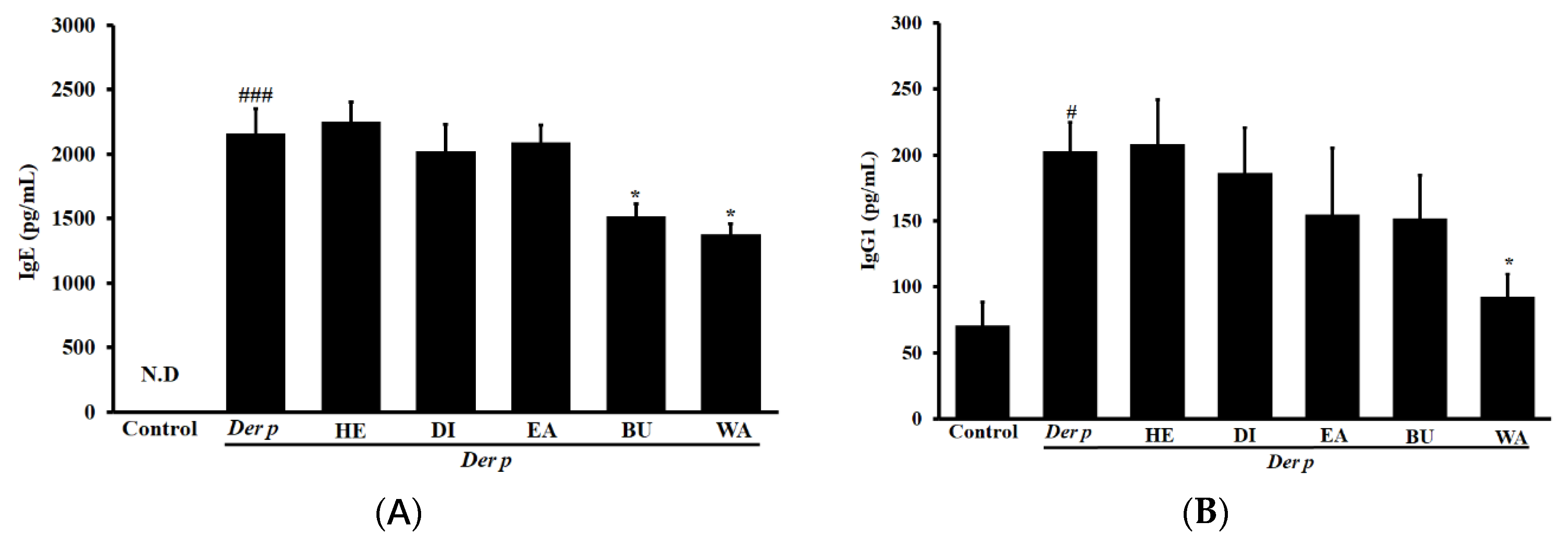
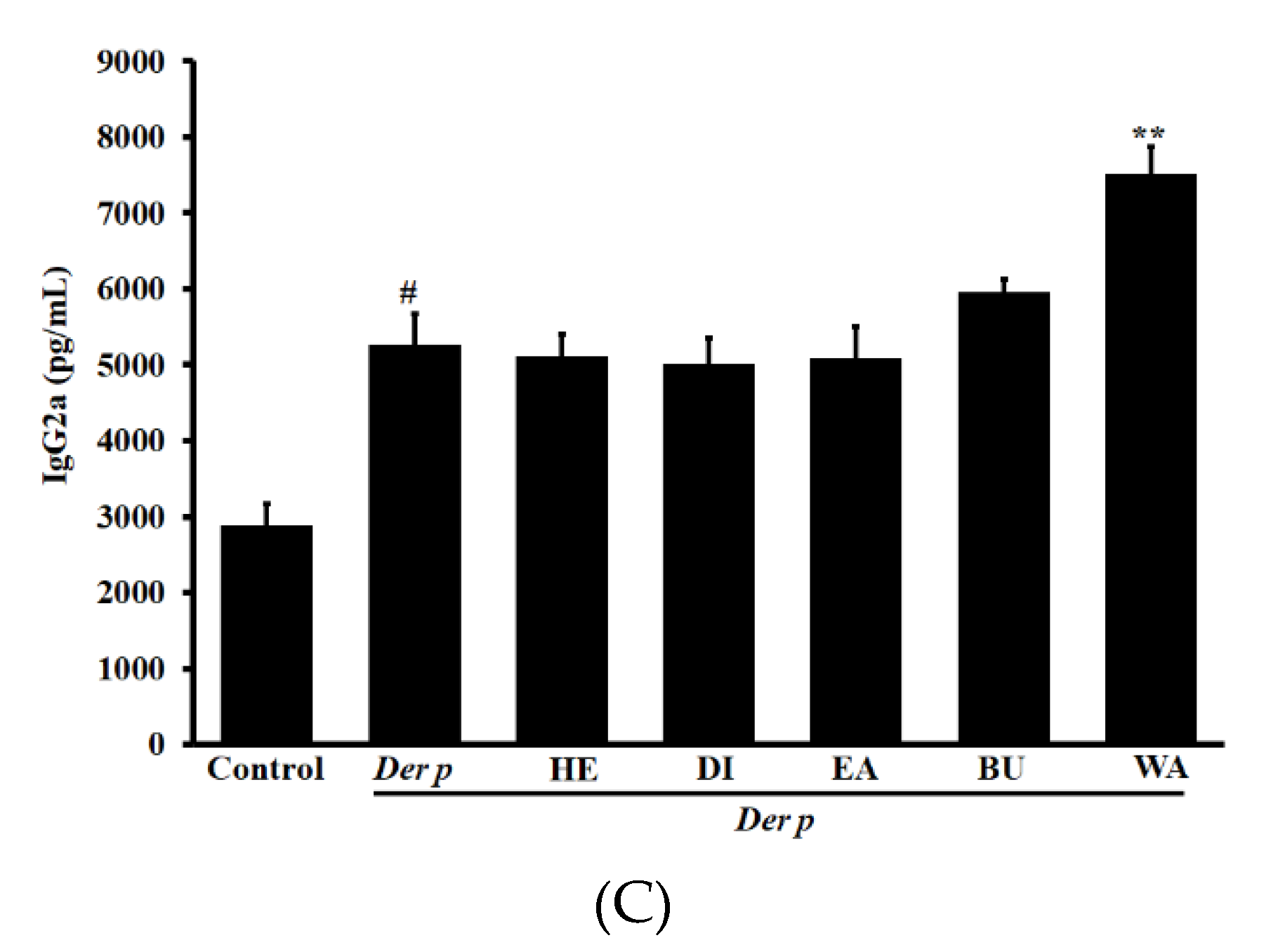
2.3. Effects on Der p–specific serum levels of Immunoglobulin G2a (IgG2a), Immunoglobulin G1 ( IgG1) and IgE
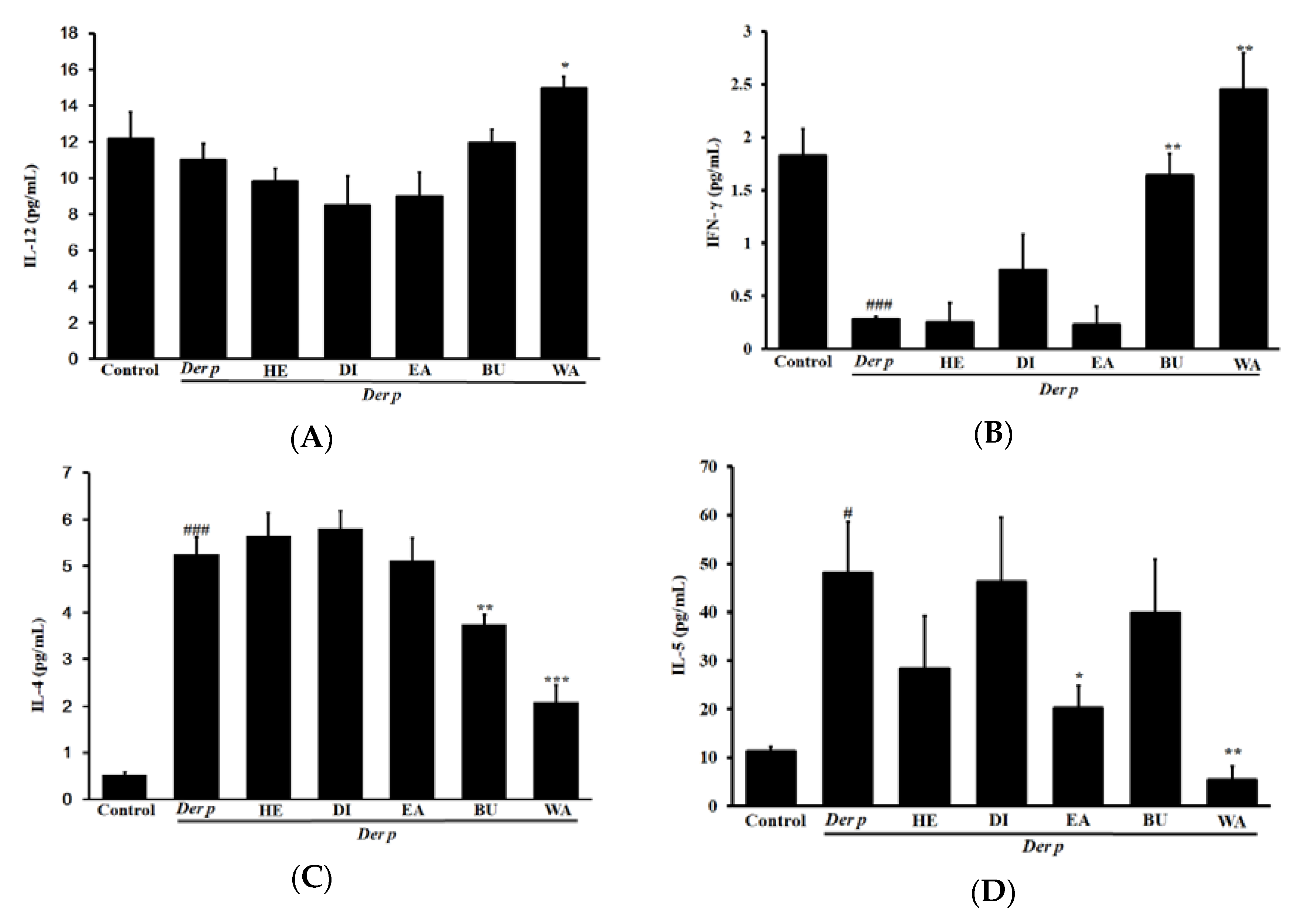
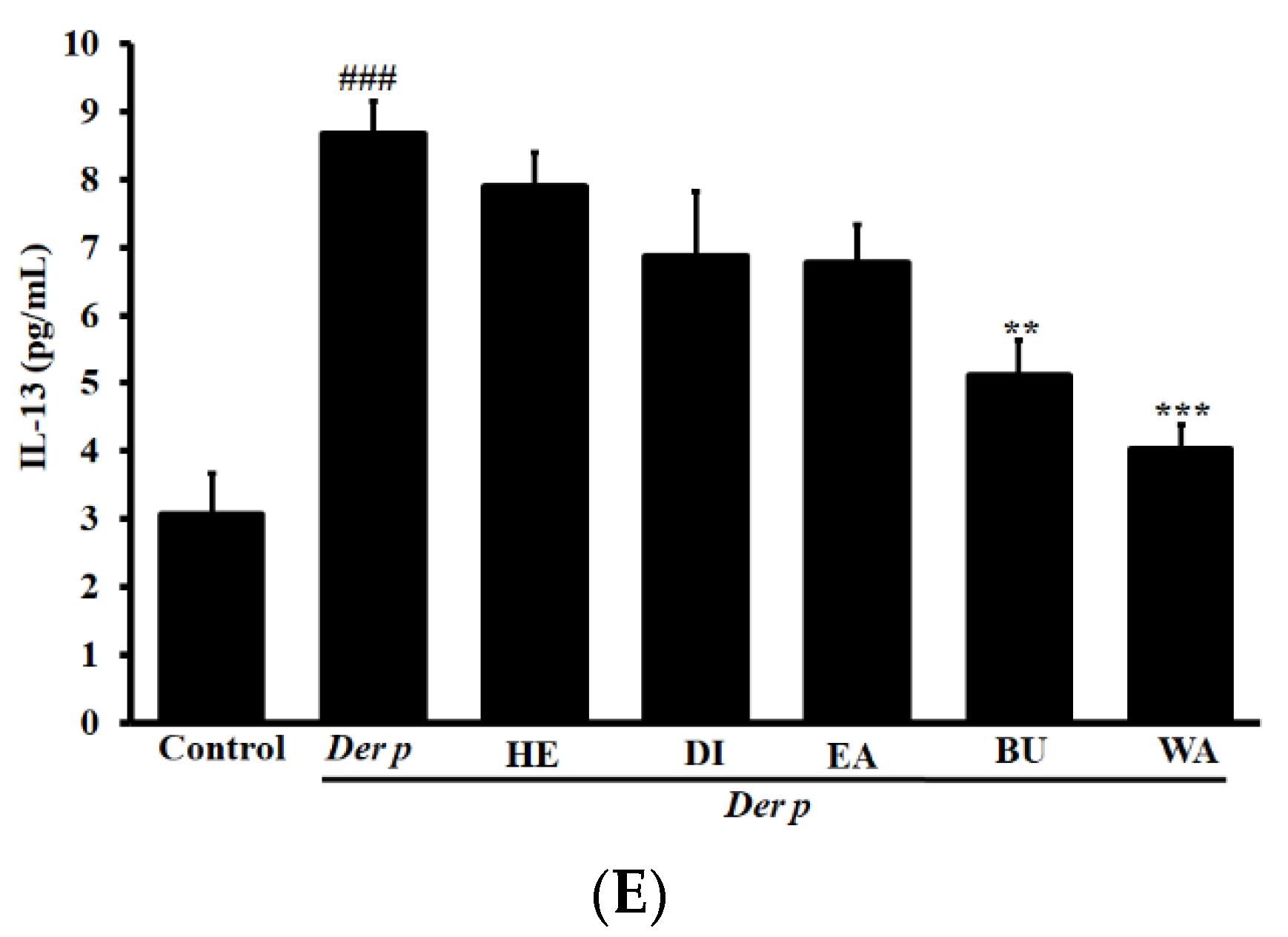
2.4. Effects on Cytokine Levels in the BALF of Mice with Der p–Induced Allergic Asthma
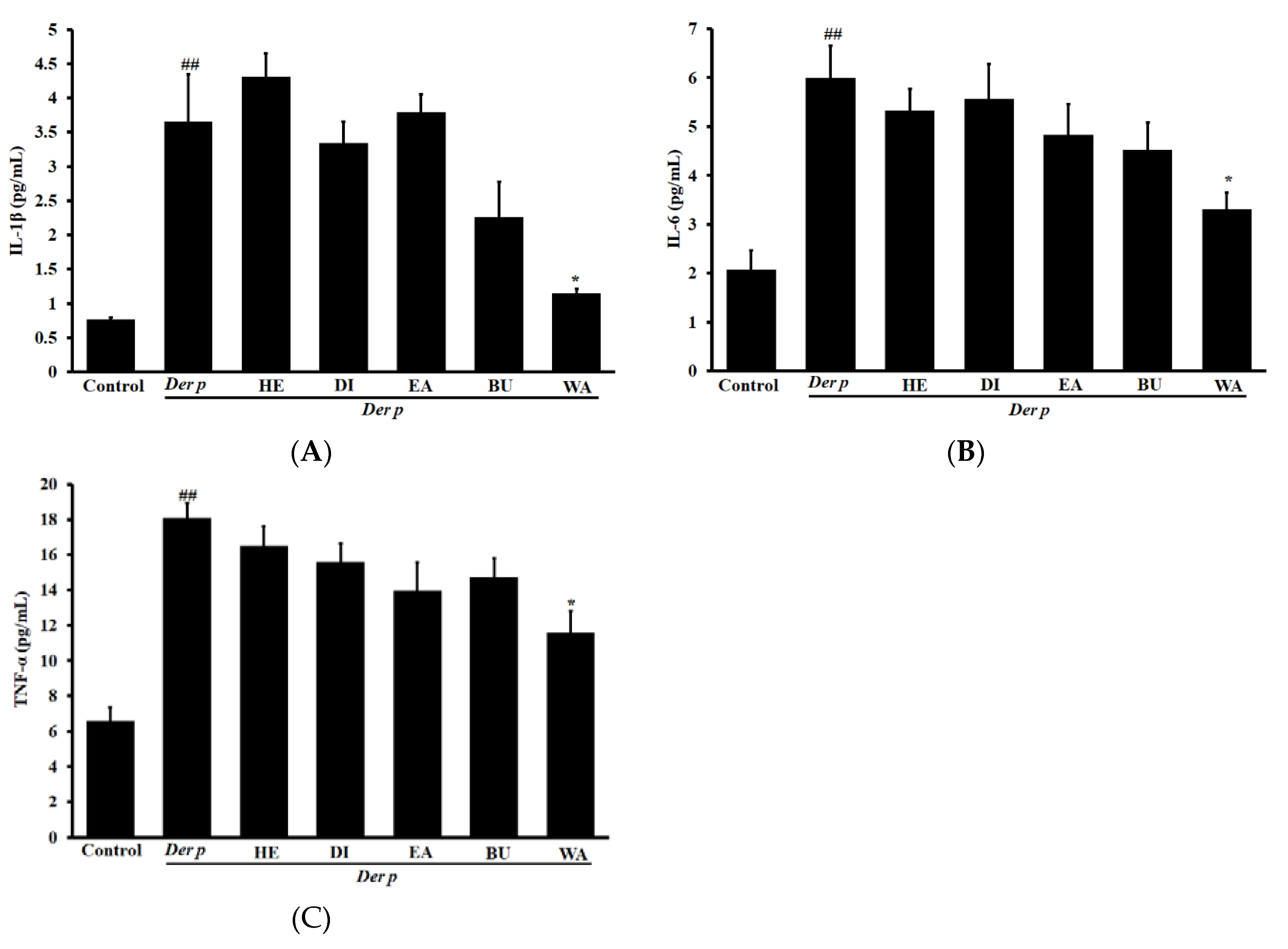
2.5. Effects on Pro-Inflammatory Cytokine Levels in Der p–Induced Lung Tissues of Mice
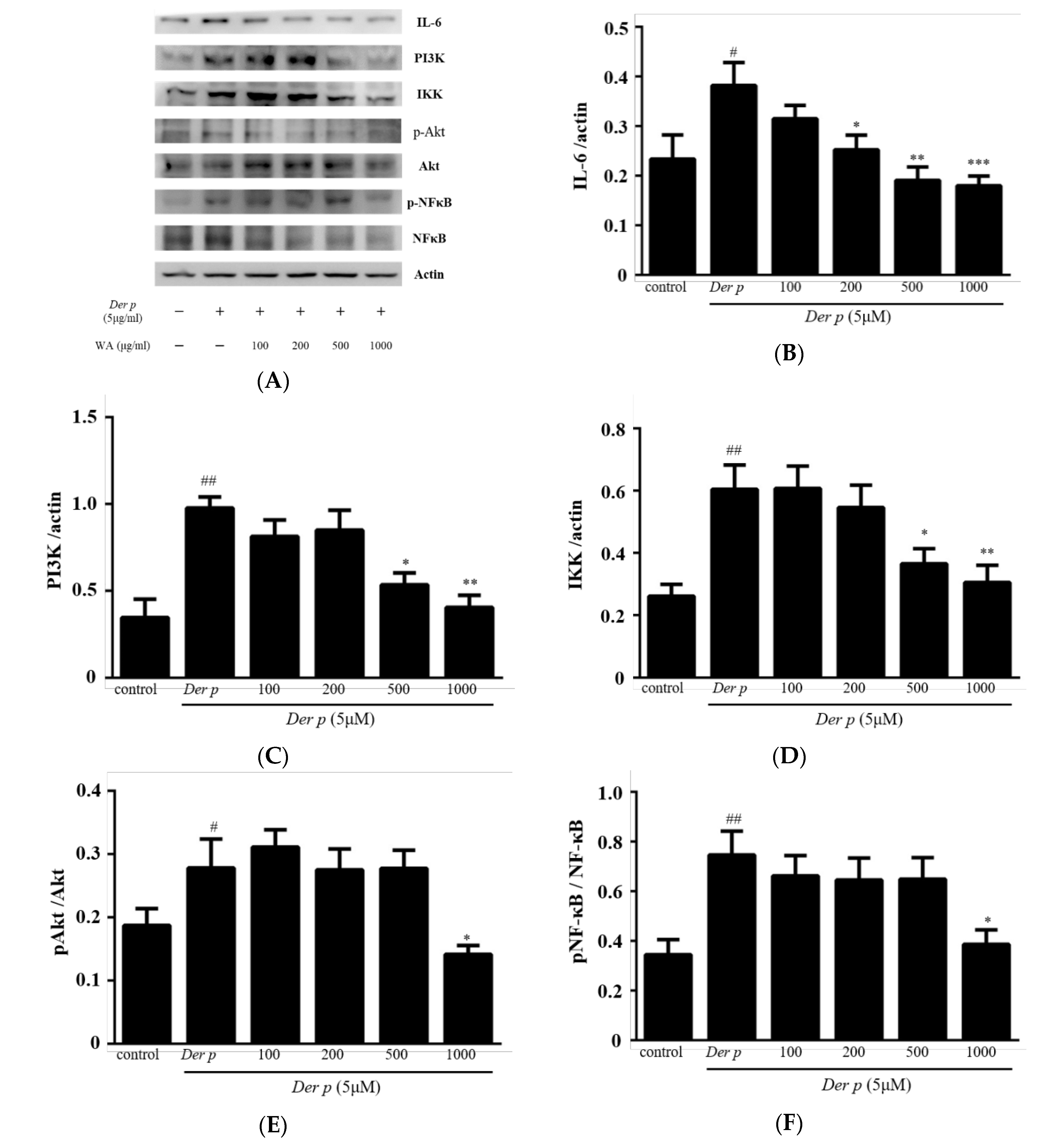
2.6. Effect of Water Fraction Extracts (WA) on IL-6/PI3K/Akt/NF-κB Pathway in Der p-Stimulated A549 Cells
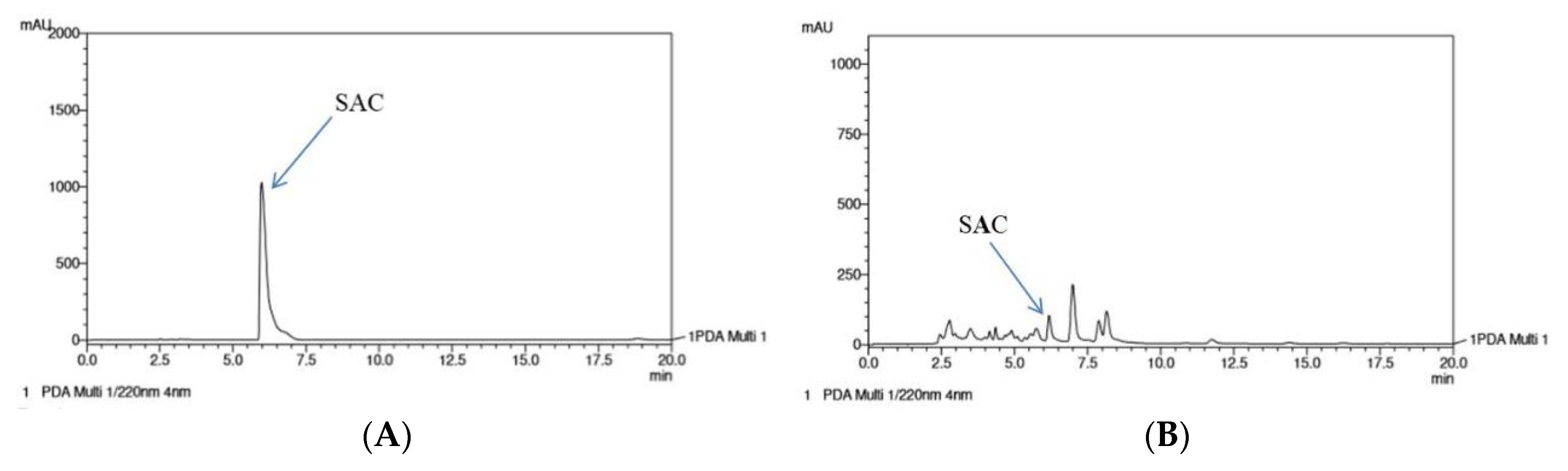
2.7. High-Performance Liquid Chromatography (HPLC) Analysis of the WA Fraction
3. Discussion
4. Materials and Methods
4.1. Chemicals and Drugs
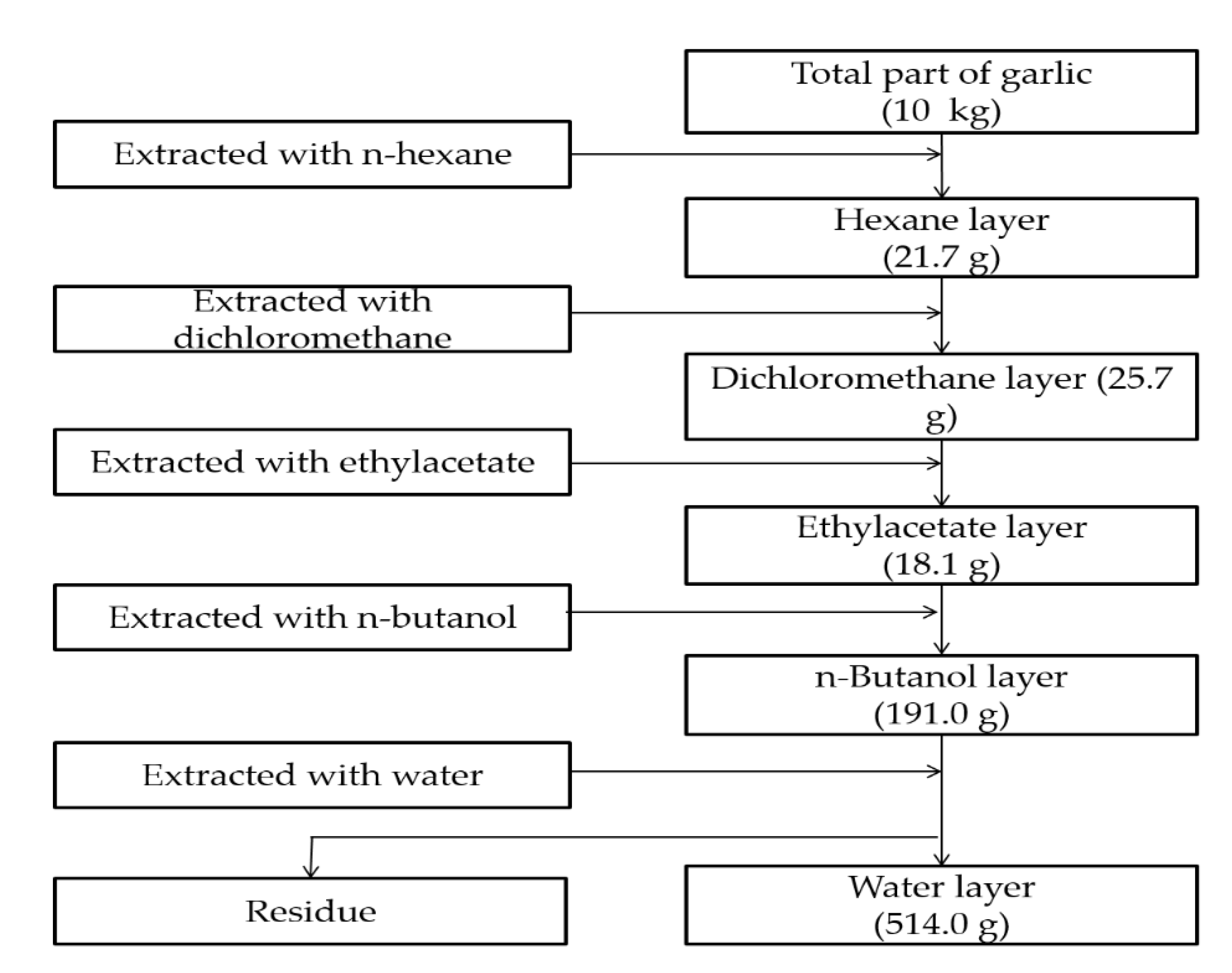
4.2. Preparation of Extracts
4.3. Experimental Animals
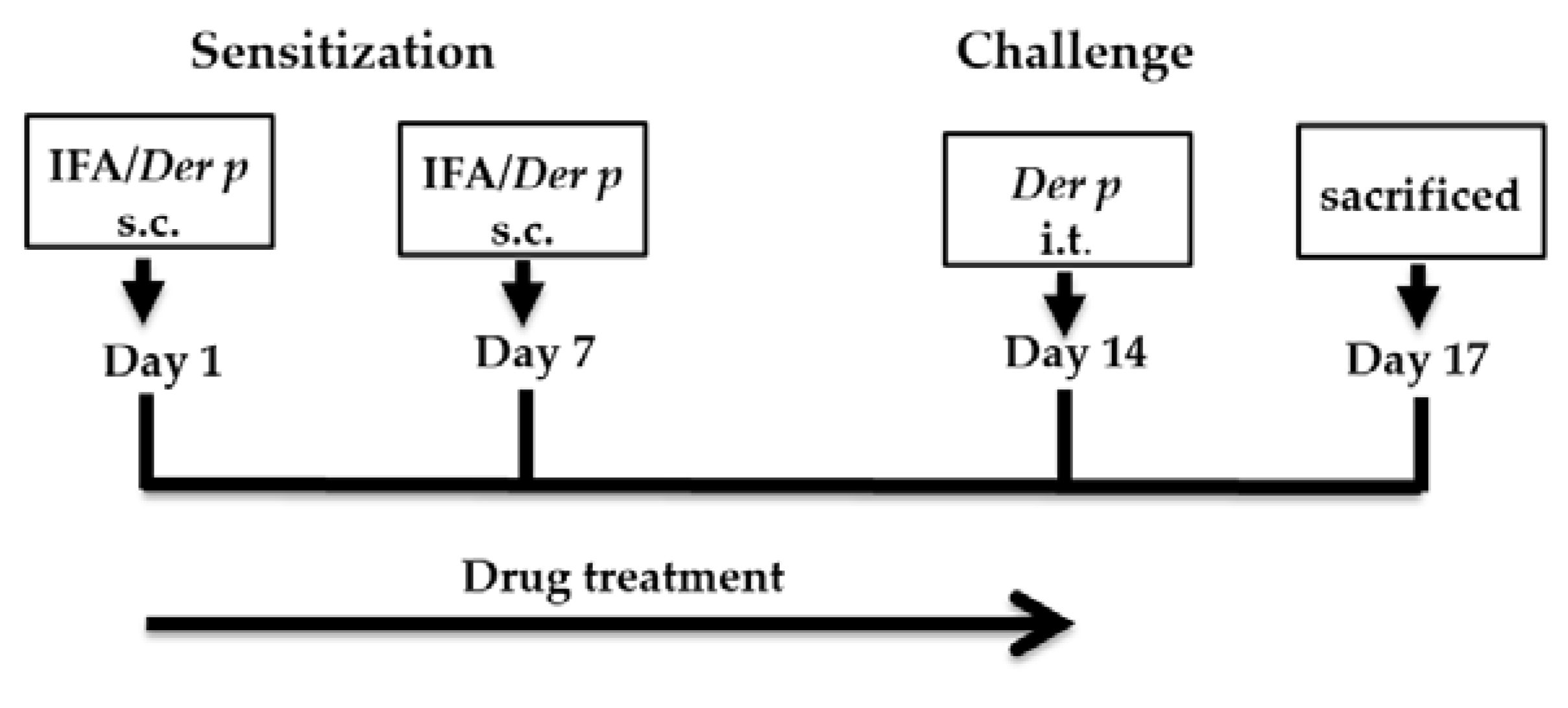
4.4. Establishing an Allergic Asthma Model
4.5. Preparing BALF and Counting Inflammatory Cells
4.6. Histological Lung Assessment
4.7. Measurement of Type 1 T helper (Th1) and Th2 Cytokine Concentrations in BALF
4.8. Der p–Specific Serum IgE/ IgG1/ IgG2a Concentrations
4.9. Measurement of Pro-Inflammatory Cytokine Concentrations in Lung Tissues
4.10. Cell Culture
4.11. Western Blotting Analysis
4.12. HPLC Analysis of the WA
4.13. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| Der p | Dermatophagoides pteronyssinus |
| BALF | bronchoalveolar lavage fluid |
| Th1 | Type 1 T helper |
| Th2 | Type 2 T helper |
| IgE | Immunoglobulin E |
| IgG1 | Immunoglobulin G1 |
| IgG2a | Immunoglobulin G2a |
| IL-12 | Interleukin-12 |
| IL-13 | Interleukin-13 |
| IL-4 | Interleukin-4 |
| IL-5 | Interleukin-5 |
| IL-6 | Interleukin-6 |
| IL-1β | Interleukin-1β |
| IFA | Incomplete Freund’s adjuvant |
| TNF-α | Tumor necrosis factor-α |
| PI3K | Phosphoinositide 3-kinases |
| NF-κB | Nuclear factor-κB |
| IKK | IκB kinase |
| HE | n-Hexane extracts |
| DI | Dichloromethane extracts |
| EA | Ethyl acetate extracts |
| BU | n-Butanol extracts |
| WA | Water extracts |
| SAC | S-allyl cysteine sulfoxide |
References
- Zhang, G.; Wang, P.; Yang, G.; Cao, Q.; Wang, R. The inhibitory role of hydrogen sulfide in airway hyperresponsiveness and inflammation in a mouse model of asthma. Am. J. Pathol. 2013, 182, 1188–1195. [Google Scholar] [CrossRef] [PubMed]
- Gohy, S.T.; Hupin, C.; Pilette, C.; Ladjemi, M.Z. Chronic inflammatory airway diseases: The central role of the epithelium revisited. Clin. Exp. Allergy 2016, 46, 529–542. [Google Scholar] [CrossRef] [PubMed]
- Holgate, S.T. The airway epithelium is central to the pathogenesis of asthma. Allergol. Int. 2008, 57, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Guilbert, T.W.; Bacharier, L.B.; Fitzpatrick, A.M. Severe asthma in children. J. Allergy Clin. Immunol. Pract. 2014, 2, 489–500. [Google Scholar] [CrossRef] [PubMed]
- Potaczek, D.P.; Harb, H.; Michel, S.; Alhamwe, B.A.; Renz, H.; Tost, J. Epigenetics and allergy: From basic mechanisms to clinical applications. Epigenomics. 2017, 9, 539–571. [Google Scholar] [CrossRef]
- Alaskhar Alhamwe, B.; Khalaila, R.; Wolf, J.; von Bulow, V.; Harb, H.; Alhamdan, F.; Hii, C.S.; Prescott, S.L.; Ferrante, A.; Renz, H.; et al. Histone modifications and their role in epigenetics of atopy and allergic diseases. Allergy. Asthma. Clin. Immunol. 2018, 14, 39. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Jin, R.G.; Xiao, L.; Wang, Q.J.; Yan, T.H. Anti-asthma effects of synthetic salidroside through regulation of Th1/Th2 balance. Chin. J. Nat. Med. 2014, 12, 500–504. [Google Scholar] [CrossRef]
- Ashraf, M.I.; Shahzad, M.; Shabbir, A. Oxyresveratrol ameliorates allergic airway inflammation via attenuation of IL-4, IL-5, and IL-13 expression levels. Cytokine 2015, 76, 375–381. [Google Scholar] [CrossRef]
- Maezawa, Y.; Nakajima, H.; Kumano, K.; Kubo, S.; Karasuyama, H.; Iwamoto, I. Role of IgE in Th2 cell-mediated allergic airway inflammation. Int. Arch. Allergy Immunol. 2003, 131 (Suppl. 1), 2–6. [Google Scholar] [CrossRef]
- Thomson, N.C.; Spears, M. Inhaled corticosteroids for asthma: On-demand or continuous use. Expert. Rev. Respir. Med. 2013, 7, 687–699. [Google Scholar] [CrossRef]
- Mazzon, E.; Cuzzocrea, S. Role of TNF-alpha in lung tight junction alteration in mouse model of acute lung inflammation. Respir. Res. 2007, 8, 75. [Google Scholar] [CrossRef] [PubMed]
- Guimaraes, P.M.; Scavuzzi, B.M.; Stadtlober, N.P.; Franchi Santos, L.; Lozovoy, M.A.B.; Iriyoda, T.M.V.; Costa, N.T.; Reiche, E.M.V.; Maes, M.; Dichi, I.; et al. Cytokines in systemic lupus erythematosus: Far beyond Th1/Th2 dualism lupus: Cytokine profiles. Immunol. Cell Biol. 2017, 95, 824–831. [Google Scholar] [CrossRef] [PubMed]
- Hunter, C.A.; Jones, S.A. IL-6 as a keystone cytokine in health and disease. Nat. Immunol. 2015, 16, 448–457. [Google Scholar] [CrossRef] [PubMed]
- Jo, H.A.; Kim, J.Y.; Yang, S.H.; Han, S.S.; Joo, K.W.; Kim, Y.S.; Kim, D.K. The role of local IL6/JAK2/STAT3 signaling in high glucose-induced podocyte hypertrophy. Kidney Res. Clin. Pract. 2016, 35, 212–218. [Google Scholar] [CrossRef] [PubMed]
- Gagliardo, R.; Chanez, P.; Profita, M.; Bonanno, A.; Albano, G.D.; Montalbano, A.M.; Pompeo, F.; Gagliardo, C.; Merendino, A.M.; Gjomarkaj, M. IkappaB kinase-driven nuclear factor-kappaB activation in patients with asthma and chronic obstructive pulmonary disease. J. Allergy Clin. Immunol. 2011, 128, 635-45.e1-2. [Google Scholar] [CrossRef] [PubMed]
- Gu, X.; Zhang, Q.; Du, Q.; Shen, H.; Zhu, Z. Pinocembrin attenuates allergic airway inflammation via inhibition of NF-kappaB pathway in mice. Int. Immunopharmacol. 2017, 53, 90–95. [Google Scholar] [CrossRef] [PubMed]
- Alali, F.Q.; El-Elimat, T.; Khalid, L.; Hudaib, R.; Al-Shehabi, T.S.; Eid, A.H. Garlic for Cardiovascular Disease: Prevention or Treatment? Curr. Pharm. Des. 2017, 23, 1028–1041. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.E.; Wang, W.; Qin, J. Anti-hyperlipidemia of garlic by reducing the level of total cholesterol and low-density lipoprotein: A meta-analysis. Medicine (Baltimore) 2018, 97, e0255. [Google Scholar] [CrossRef]
- Padiya, R.; Banerjee, S.K. Garlic as an anti-diabetic agent: Recent progress and patent reviews. Recent Pat. Food Nutr. Agric. 2013, 5, 105–127. [Google Scholar] [CrossRef]
- Zamani, A.; Vahidinia, A.; Ghannad, M.S. The effect of garlic consumption on Th1/Th2 cytokines in phytohemagglutinin (PHA) activated rat spleen lymphocytes. Phytother. Res. 2009, 23, 579–581. [Google Scholar] [CrossRef]
- Hsieh, C.C.; Peng, W.H.; Tseng, H.H.; Liang, S.Y.; Chen, L.J.; Tsai, J.C. The Protective Role of Garlic on Allergen-Induced Airway Inflammation in Mice. Am. J. Chin. Med. 2019, 47, 1099–1112. [Google Scholar] [CrossRef] [PubMed]
- Shackelford, C.; Long, G.; Wolf, J.; Okerberg, C.; Herbert, R. Qualitative and quantitative analysis of nonneoplastic lesions in toxicology studies. Toxicol. Pathol. 2002, 30, 93–96. [Google Scholar] [CrossRef] [PubMed]
- van Halteren, A.G.; van der Cammen, M.J.; Cooper, D.; Savelkoul, H.F.; Kraal, G.; Holt, P.G. Regulation of antigen-specific IgE, IgG1, and mast cell responses to ingested allergen by mucosal tolerance induction. J. Immunol. 1997, 159, 3009–3015. [Google Scholar] [PubMed]
- Yano, S.; Umeda, D.; Yamashita, T.; Ninomiya, Y.; Sumida, M.; Fujimura, Y.; Yamada, K.; Tachibana, H. Dietary flavones suppresses IgE and Th2 cytokines in OVA-immunized BALB/c mice. Eur. J. Nutr. 2007, 46, 257–263. [Google Scholar] [CrossRef] [PubMed]
- HuangFu, T.; Lim, L.H.; Chua, K.Y. Efficacy evaluation of Der p 1 DNA vaccine for allergic asthma in an experimental mouse model. Vaccine 2006, 24, 4576–4581. [Google Scholar] [CrossRef] [PubMed]
- Roche, N.; Chinet, T.C.; Huchon, G.J. Allergic and nonallergic interactions between house dust mite allergens and airway mucosa. Eur. Respir. J. 1997, 10, 719–726. [Google Scholar] [PubMed]
- Shin, I.S.; Park, J.W.; Shin, N.R.; Jeon, C.M.; Kwon, O.K.; Kim, J.S.; Kim, J.C.; Oh, S.R.; Ahn, K.S. Melatonin reduces airway inflammation in ovalbumin-induced asthma. Immunobiology 2014, 219, 901–908. [Google Scholar] [CrossRef]
- Halwani, R.; Vazquez-Tello, A.; Sumi, Y.; Pureza, M.A.; Bahammam, A.; Al-Jahdali, H.; Soussi-Gounni, A.; Mahboub, B.; Al-Muhsen, S.; Hamid, Q. Eosinophils induce airway smooth muscle cell proliferation. J. Clin. Immunol. 2013, 33, 595–604. [Google Scholar] [CrossRef]
- Debes, G.F.; Dahl, M.E.; Mahiny, A.J.; Bonhagen, K.; Campbell, D.J.; Siegmund, K.; Erb, K.J.; Lewis, D.B.; Kamradt, T.; Hamann, A. Chemotactic responses of IL-4-, IL-10-, and IFN-gamma-producing CD4+ T cells depend on tissue origin and microbial stimulus. J. Immunol. 2006, 176, 557–566. [Google Scholar] [CrossRef]
- Profumo, E.; Buttari, B.; Saso, L.; Capoano, R.; Salvati, B.; Rigano, R. T lymphocyte autoreactivity in inflammatory mechanisms regulating atherosclerosis. Sci. World J. 2012, 2012, 157534. [Google Scholar] [CrossRef]
- Leiria, L.O.; Martins, M.A.; Saad, M.J. Obesity and asthma: Beyond T(H)2 inflammation. Metabolism 2015, 64, 172–181. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.; Lee, S.; Kim, Y.S.; Lawler, S.; Gho, Y.S.; Kim, Y.K.; Hwang, H.J. Regulation of Th1/Th2 cells in asthma development: A mathematical model. Math. Biosci. Eng. 2013, 10, 1095–1133. [Google Scholar] [PubMed]
- Yun, X.; Shang, Y.; Li, M. Effect of Lactobacillus salivarius on Th1/Th2 cytokines and the number of spleen CD4+ CD25+ Foxp3+ Treg in asthma Balb/c mouse. Int. J. Clin. Exp. Pathol. 2015, 8, 7661–7674. [Google Scholar] [PubMed]
- Maeda, S.; Yanagihara, Y. [Inflammatory cytokines (IL-4, IL-5 and IL-13)]. Nihon Rinsho. 2001, 59, 1894–1899. [Google Scholar] [PubMed]
- Wang, J. Casticin alleviates lipopolysaccharide-induced inflammatory responses and expression of mucus and extracellular matrix in human airway epithelial cells through Nrf2/Keap1 and NF-kappaB pathways. Phytother. Res. 2018, 32, 1346–1353. [Google Scholar] [CrossRef] [PubMed]
- Schuliga, M. NF-kappaB Signaling in Chronic Inflammatory Airway Disease. Biomolecules 2015, 5, 1266–1283. [Google Scholar] [CrossRef] [PubMed]
- Schepetkin, I.A.; Kirpotina, L.N.; Khlebnikov, A.I.; Balasubramanian, N.; Quinn, M.T. Neutrophil Immunomodulatory Activity of Natural Organosulfur Compounds. Molecules 2019, 24, 1809. [Google Scholar] [CrossRef] [PubMed]
- Potaczek, D.P. Links between allergy and cardiovascular or hemostatic system. Int. J. Cardiol. 2014, 170, 278–285. [Google Scholar] [CrossRef]
- Miethe, S.; Guarino, M.; Alhamdan, F.; Simon, H.U.; Renz, H.; Dufour, J.F.; Potaczek, D.P.; Garn, H. Effects of obesity on asthma: Immunometabolic links. Pol. Arch. Intern. Med. 2018, 128, 469–477. [Google Scholar] [CrossRef]
- Arreola, R.; Quintero-Fabian, S.; Lopez-Roa, R.I.; Flores-Gutierrez, E.O.; Reyes-Grajeda, J.P.; Carrera-Quintanar, L.; Ortuno-Sahagun, D. Immunomodulation and anti-inflammatory effects of garlic compounds. J. Immunol. Res. 2015, 2015, 401630. [Google Scholar] [CrossRef]
- Johnson, P.; Loganathan, C.; Iruthayaraj, A.; Poomani, K.; Thayumanavan, P. S-allyl cysteine as potent anti-gout drug: Insight into the xanthine oxidase inhibition and anti-inflammatory activity. Biochimie 2018, 154, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Gilhotra, N.; Dhingra, D. GABAergic and nitriergic influence in antianxiety-like Activity of Garlic in Mice. J. Appl. Pharm. Sci. 2016, 6, 77–85. [Google Scholar] [CrossRef]
- Chen, C.L.; Wang, S.D.; Zeng, Z.Y.; Lin, K.J.; Kao, S.T.; Tani, T.; Yu, C.K.; Wang, J.Y. Serine protease inhibitors nafamostat mesilate and gabexate mesilate attenuate allergen-induced airway inflammation and eosinophilia in a murine model of asthma. J. Allergy Clin. Immunol. 2006, 118, 105–112. [Google Scholar] [CrossRef] [PubMed]
- He, J.; Lv, L.; Wang, Z.; Huo, C.; Zheng, Z.; Yin, B.; Jiang, P.; Yang, Y.; Li, J.; Gao, Y.; et al. Pulvis Fellis Suis extract attenuates ovalbumin-induced airway inflammation in murine model of asthma. J. Ethnopharmacol. 2017, 207, 34–41. [Google Scholar] [CrossRef] [PubMed]
- Woolard, M.D.; Hudig, D.; Tabor, L.; Ivey, J.A.; Simecka, J.W. NK cells in gamma-interferon-deficient mice suppress lung innate immunity against Mycoplasma spp. Infect. Immun. 2005, 73, 6742–6751. [Google Scholar] [CrossRef]
- Sabo-Attwood, T.; Ramos-Nino, M.; Bond, J.; Butnor, K.J.; Heintz, N.; Gruber, A.D.; Steele, C.; Taatjes, D.J.; Vacek, P.; Mossman, B.T. Gene expression profiles reveal increased mClca3 (Gob5) expression and mucin production in a murine model of asbestos-induced fibrogenesis. Am. J. Pathol. 2005, 167, 1243–1256. [Google Scholar] [CrossRef]
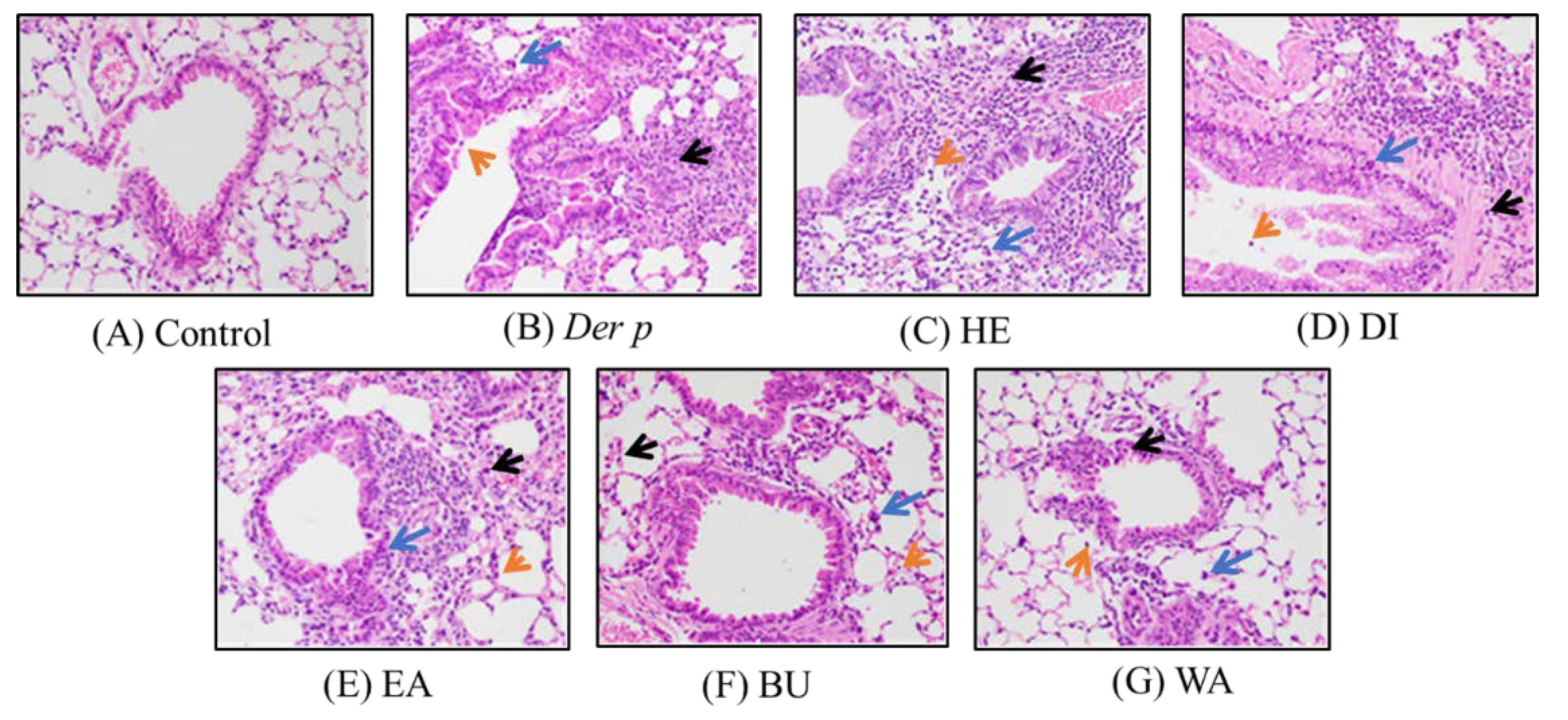
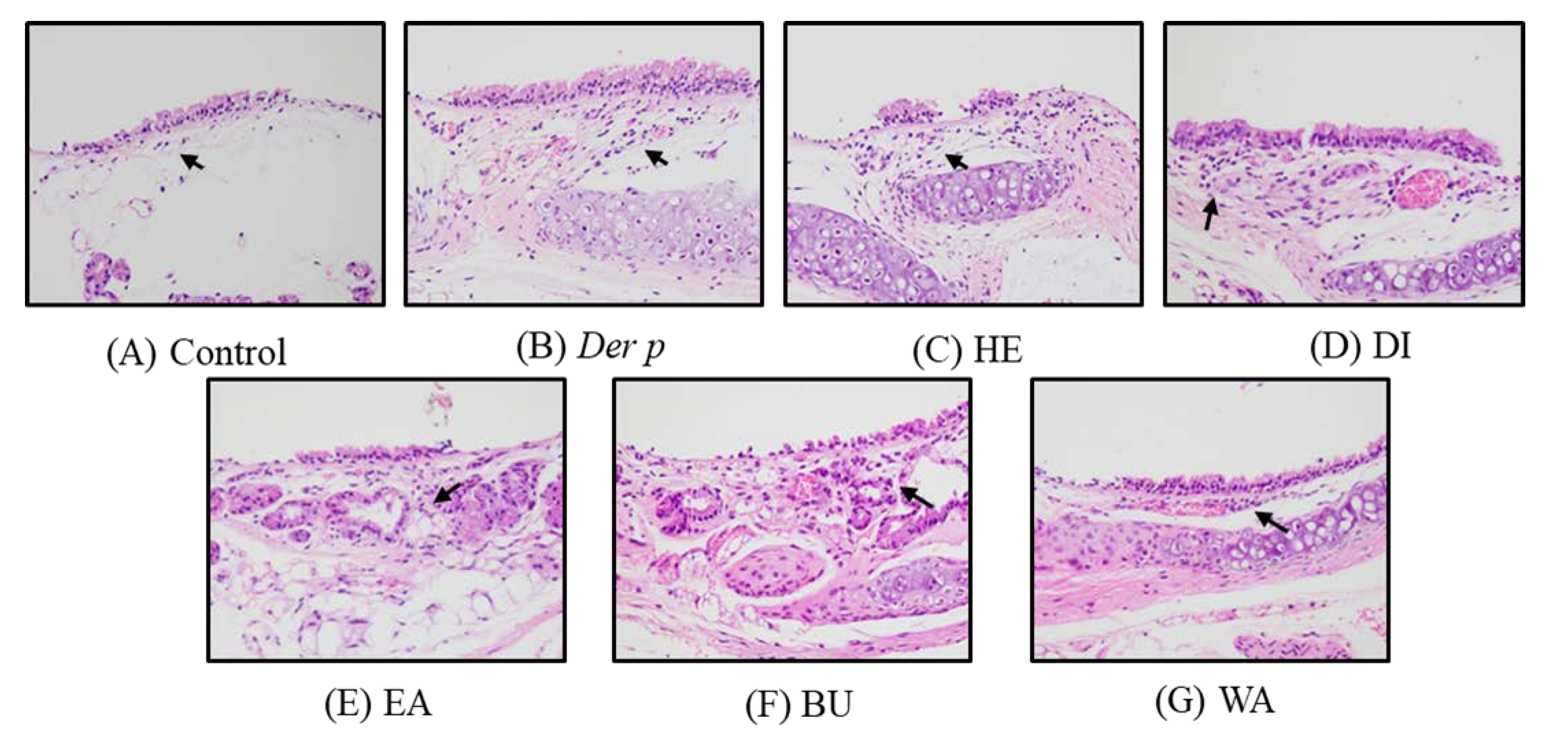
| Organ | Histopathological Findings | Groups 1 | ||||||
|---|---|---|---|---|---|---|---|---|
| Control | Der p | Der p | ||||||
| HE | DI | EA | BU | WH | ||||
| Lung | Inflammation, eosinophilic and lymphocytic cells, perivascular and per bronchial, focal | 0.0 ± 0.00 | 3.3 ± 0.19 # | 3.0 ± 0.00 | 2.5 ± 0.50 | 2.8 ± 0.19 | 2.8 ± 0.19 | 1.8 ± 0.19 * |
| Aggregation, macrophage and giant cells, alveolar, focal | 0.0 ± 0.00 | 2.5 ± 0.22 # | 2.0 ± 0.00 | 1.8 ± 0.38 | 1.8 ± 0.19 | 1.5 ± 0.22 | 0.8 ± 0.19 * | |
| Epithelial hyperplasia, bronchial, focal | 0.0 ± 0.00 | 2.0 ± 0.00 # | 2.0 ± 0.00 | 1.5 ± 0.38 | 1.5 ± 0.22 | 1.0 ± 0.31 * | 1.0 ± 0.31 * | |
| Mucification, goblet, bronchial, focal | 0.0 ± 0.00 | 2.0 ± 0.00 # | 1.8 ± 0.19 | 1.5 ± 0.38 | 1.5 ± 0.22 | 1.0 ± 0.31 * | 1.0 ± 0.31 * | |
| Brachus | Inflammation, submucosal, focal | 1.0 ± 0.00 | 3.3 ± 0.58 # | 2.3 ± 0.58 | 2.0 ± 0.82 | 2.0 ± 0.82 | 1.5 ± 0.58 | 1.3 ± 0.43 * |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hsieh, C.-C.; Liu, K.-F.; Liu, P.-C.; Ho, Y.-T.; Li, W.-S.; Peng, W.-H.; Tsai, J.-C. Comparing the Protection Imparted by Different Fraction Extracts of Garlic (Allium sativum L.) against Der p–Induced Allergic Airway Inflammation in Mice. Int. J. Mol. Sci. 2019, 20, 4879. https://doi.org/10.3390/ijms20194879
Hsieh C-C, Liu K-F, Liu P-C, Ho Y-T, Li W-S, Peng W-H, Tsai J-C. Comparing the Protection Imparted by Different Fraction Extracts of Garlic (Allium sativum L.) against Der p–Induced Allergic Airway Inflammation in Mice. International Journal of Molecular Sciences. 2019; 20(19):4879. https://doi.org/10.3390/ijms20194879
Chicago/Turabian StyleHsieh, Chia-Chen, Keng-Fan Liu, Pei-Chun Liu, Yaw-Tsan Ho, Wei-Sung Li, Wen-Huang Peng, and Jen-Chieh Tsai. 2019. "Comparing the Protection Imparted by Different Fraction Extracts of Garlic (Allium sativum L.) against Der p–Induced Allergic Airway Inflammation in Mice" International Journal of Molecular Sciences 20, no. 19: 4879. https://doi.org/10.3390/ijms20194879
APA StyleHsieh, C.-C., Liu, K.-F., Liu, P.-C., Ho, Y.-T., Li, W.-S., Peng, W.-H., & Tsai, J.-C. (2019). Comparing the Protection Imparted by Different Fraction Extracts of Garlic (Allium sativum L.) against Der p–Induced Allergic Airway Inflammation in Mice. International Journal of Molecular Sciences, 20(19), 4879. https://doi.org/10.3390/ijms20194879






